Abstract
This study aimed to compare the efficacy and safety of Vernonia cinerea (VC) and nortriptyline for smoking cessation. A randomized, active-comparator, open-label trial was conducted in 2019. A total of 84 patients participated in the study, and equally randomized with 42 participants in each group. Overall, there was no statistically significant difference of continuous abstinence rate (CAR) between VC and nortriptyline group (Odd ratio 0.68, 95%CI 0.25-1.85, P=0.451). After week 12, the end of treatment, CAR between both groups was not different (44.44% vs 45.95%, Odd ratio 0.77, 95%CI 0.23–2.54, P>0.999). After follow up by the end of research at week 24, the CAR in both groups was not different (41.67% vs 43.24%, Odd ratio 0.76, 95%CI 0.23–2.55, P>0.999). After week 24, relapse rate between VC and nortriptyline group was not different (13.89% vs 10.81%, P=0.923). In addition, both groups were effective in reducing the number of cigarettes per day compared to baseline. However, there was no difference between the groups. Overall, the VC group had an 8% smoking rate less than nortriptyline group, but not statistically significant (IRR 0.92, 95%CI 0.59-1.43, P=0.702). They also resulted in reducing the exhaled CO level at treatment period and wash out period (at week 12; 7(-17-20) vs 7(-12-16), mean difference 0.78, 95%CI -3.07-4.63, P>0.999, at week 24; 8(-5-22) vs 8.5(-5-17), mean difference 0.39, 95%CI -3.46-4.24, P>0.999). Overall, there was no difference between either group (mean difference -0.31, 95%CI -3.10-2.47). For safety data, adverse events including tongue bitter taste or numbness were found in VC group to be greater than in nortriptyline group (61.9% vs 30.95%, P=0.004), whereas dry mouth and drowsiness were greater found in nortriptyline group (35.71% vs 90.48%, P<0.001 and 16.67% vs 90.48%, P<0.001, respectively). Serious adverse events were not found. In smoking cessation, efficacy and safety of either VC or nortriptyline showed no difference.
Key Words: Vernonia cinerea, nortriptyline, tobacco dependence, smoking cessation, safety
Introduction
Cigarette is easily accessed and traded legally in Thailand (Campaign for Tobacco Free Kids, 2021). There is a tendency of increasing number of people who are addicted (Baumeister, 2017). It affects every system of the body (WHO, 2008), causing severe and long-term disability and premature death (Raw et al.,1998; Parrott et al.,1998; Clinical Practice Guideline Treating Tobacco Use and Dependence 2008 Update Panel, Liaisons, and Staff 2008; Steinberg et al., 2008). Smoking addiction is associated with the functioning of neurotransmitters in the brain, especially dopamine, with addiction defined by familiarity and social conditions. Therefore, it is so difficult to quit smoking (Haxby, 1995; Piasecki and Newhouse, 2000; Berrettini, 2004; Fahey et al., 2022), even with those who with urgent needs, such as bladder cancer patients (Novesar et al., 2022).
Effective smoking cessation includes adequate advice and pharmacological methods (The Tobacco Use and Dependence Clinical Practice Guideline Panel, Staff, and Consortium Representatives, 2000; Fiore, 2000; Yunipan et al., 2012). Giving advice requires intensive training and may cope with many barriers (Medawela et al., 2021). There are many studies on smoking cessation by pharmacological methods, including nicotine replacement therapy (NRT) and non-NRT. NRT is rather expensive. Consequently, nortriptyline, one of the non-NRT, is often used in current practice. It is inexpensive in comparison, effective, but may induces many adverse events (Hall et al., 2002; Wagena et al., 2005; Haggsträm et al., 2006).
Vernonia cinerea (VC) has the ability to inhibit the degradation of dopamine, which prolong the craving of cigarettes for the next time of smoking (Prasopthum et al., 2015). It has shown the reduction of tobacco dependence effect among tobacco users. VC has eight active compounds (apigenin, chrysoeriol, luteolin, quercetin, and four hirsutinolide-type sesquiterpene lactones) that could inhibit CYP2A6 and MAO-A and MAO-B enzymes. These are two therapeutic targets of nicotine dependence. Therefore, VC could have important roles in smoking cessation (Prasopthum et al., 2015). It also contains nitrate that can make the tongue feeling bitter or cause numbness during smoking, i.e. tongue taste would would significantly change (Wongwiwatthananukit et al., 2018). This might help quit smoking and could have less adverse events. The exact mechanism of nortriptyline is unclear, but it may relate to dopaminergic or adrenergic activity. This could mean that nortriptyline reduces the strength of smoking rewarding properties (Hughes et al., 2005). However, there is still no clinical-based evidence comparing the efficacy and safety between VC and nortriptyline.
Materials and Methods
Study participants
Adults (age 20-60 years) with smoking dependence were screened for participation in the study. Inclusion criteria included moderate to severe nicotine dependence (Heaviness of smoking index, HSI of less than or equal to three for at least 1 month) before randomization. Patients with a history of either VC or nortriptyline allergy, comorbidities (heart disease, cancer, urinary retention, benign prostatic hyperplasia, glaucoma, abnormal vision, dry mouth, constipation, history of gut obstruction, abnormal function of either liver or kidney, abnormal serum potassium level, neurological disorder, depression), using other types of tobacco, taking other drugs, pregnancy, on breastfeeding, or used MAOIs drug at least 14 days before randomization, and may have used drugs that may cause drug interactions with nortriptyline and used other medication for smoking cessation, were excluded. Written informed consent was obtained from all patients.
Study design and oversight
This randomized, active-comparator, open-label trial was conducted at Namphong Hospital, Thailand between December 2018 to August 2019. All the authors were involved in the design and performance of the study, which was conducted according to the Declaration of Helsinki. The Khon Kaen Hospital institute review board in human research approved the study protocol, KE61095.
Study treatments and procedures
Patients were randomly assigned in advance by computer generation sequence, using 1:1 ratio. The sequence of this assignment was concealed by a research assistant nurse and can only be known when new patient come in and ask for it specifically. All patients received the same advice regarding smoking cessation. The intervention group with 2 grams of VC takes pre-meals three times a day. For the control group, and to reduce adverse events, nortriptyline was started at 25 milligrams once daily (before bed) for day 1 through 3, followed by 50 milligrams once daily for day 3 through 7, and then 75 milligrams once daily for day 8 through the end of treatment at week 12. After 10 days of treatment, when therapeutic level of nortriptyline was reached, it is anticipated that patients can quit smoking. Patients received a logbook (personal diary) to record any adverse events or withdrawal symptoms.
Follow-up
The appointment was made at week 1, 2, 4, 8, 12 and 24. Each time, continuous abstinence rates (CAR) were assessed by direct observation, patients’ self-reports, number of cigarettes per day, and exhaled carbon monoxide (CO) level. If the CO level was less than or equal to 6 part per million (ppm), the patient would be considered as a non-smoker. The relapse rate (return to smoking after cessation) was assessed at week 24, which was the washout period. If an individual kept smoking throughout, it meant that no cessation had occurred (current smokers).
Endpoints
We focused on the continuous abstinence rate as the primary outcome (no smoking at all). The secondary outcomes included number of cigarettes per day, exhaled carbon monoxide level, relapse rate, adverse events, and withdrawal symptoms.
Statistical analysis
We calculated that a sample of 76 patients would provide adequate power for the proposed tests in this two-group study. Dropout rate were 10%, so the sample size were 84 patients and equally randomized with 42 participants in each group. After follow up at week 24, intention to treat (ITT) analysis was used.
Baseline characteristics were analyzed with mean±SD, median (min-max), the overall outcomes were analyzed with generalized estimating equations (GEE) model. As GEE were used for multiple-point measurements, OR (Odd Ratios) were used for each point percentages comparison and IRR (Incidence Rate Ratios) were used for each point changes (delta) comparison. Other outcomes were compared among the groups or compared before and after with Bonferroni test. Significant differences were considered at p < 0.05. All data were analyzed by Stata software version 10.1.
Results
Study participants
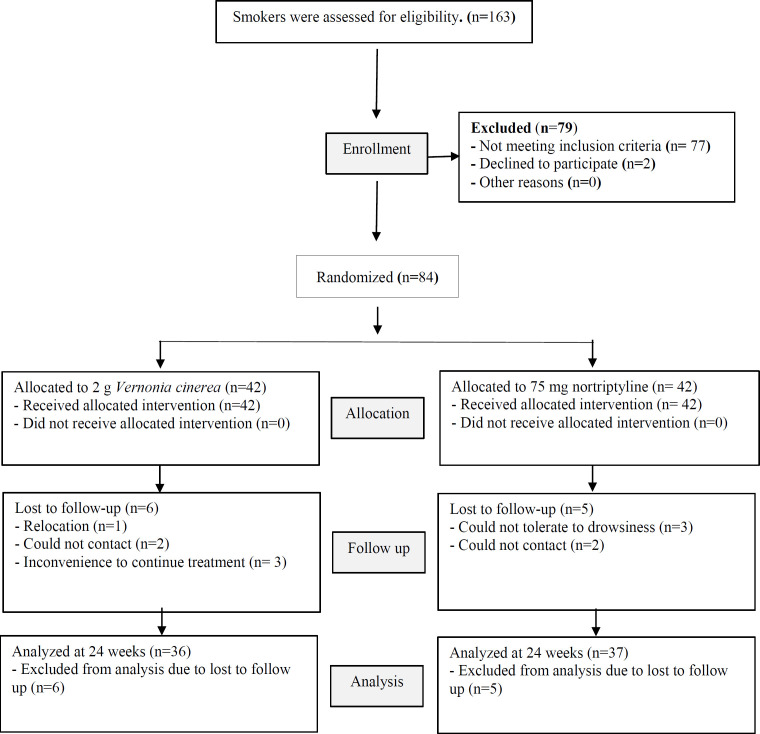
One hundred and sixty-three smokers were assessed for eligibility. Seventy-nine were excluded due to not meeting inclusion criteria, two declined to participate. A total of 84 patients participated in the study, and equally randomized with 42 participants in each group (simple randomization by computer generator). During the research process, six patients were dropped out during follow-up in VC group due to relocation, or could not be contacted, or it was too inconvenient to continue the treatment, because of work related reasons or poor compliance, or both. In nortriptyline group, five patients were eliminated because they cannot tolerate the drowsiness or could not be contacted anymore. Thirty-six patients in VC group and 37 patients in nortriptyline group had received follow-ups until the very end of the treatment and were consequently analyzed (Figure 1). The characteristics of the patients at baseline were similar among the study groups (Table 1).
Figure 1.
CONSORT Diagram
Table 1.
Baseline of Characteristics of the Patients for Both Groups of VC and Nortriptyline
| Characteristics | Vernonia cinerea (N= 42) |
Nortriptyline (N= 42) |
p-value |
|---|---|---|---|
| Male sex – no(%) | 41 (97.62) | 42 (100.00) | >0.999 |
| Age – yr | 52 (33 - 59) | 52 (28 - 60) | 0.597 |
| Marital status – no(%) | 0.542 | ||
| Single | 6 (14.29) | 5 (11.91) | |
| Married | 35 (83.33) | 33 (78.57) | |
| Divorce | 1 (2.38) | 3 (7.14) | |
| Widow | 0 (0.00) | 1 (2.38) | |
| Education – no(%) | 0.897 | ||
| Primary school or lower | 25 (59.52) | 23 (54.76) | |
| High school | 14 (33.33) | 16 (38.1) | |
| College | 3 (7.14) | 3 (7.14) | |
| Occupation – no(%) | 0.553 | ||
| Unemployment | 0 (0.00) | 1 (2.38) | |
| Farmer | 31 (73.81) | 35 (83.33) | |
| Merchant | 6 (14.29) | 4 (9.52) | |
| Employee | 2 (4.76) | 0 (0.00) | |
| State enterprise employee | 0 (0.00) | 0 (0.00) | |
| Civil servants | 2 (4.76) | 1 (2.38) | |
| Others | 1 (2.38) | 1 (2.38) | |
| Comorbidities – no. (%) | 11 (26.19) | 6 (14.29) | 0.175 |
| Stroke | 1 (2.38) | 1 (2.38) | >0.999 |
| Chronic obstructive pulmonary disease | 2 (4.76) | 0 (0.00) | 0.494 |
| Asthma | 1 (2.38) | 0 (0.00) | >0.999 |
| Diabetes mellitus | 5 (11.90) | 3 (7.14) | 0.713 |
| Hypertension | 2 (4.76) | 4 (9.52) | 0.676 |
| Thalassemia | 1 (2.38) | 1 (2.38) | >0.999 |
| Alcohol – no(%) | 0.653 | ||
| Never | 0 (0.00) | 0 (0.00) | |
| Former | 15 (35.71) | 17 (40.48) | |
| Current | 27 (64.29) | 25 (59.52) | |
| Heaviness of smoking index – no(%) | 0.776 | ||
| Moderate | 34 (80.95) | 35 (83.33) | |
| Severe | 8 (19.05) | 7 (16.67) | |
| Duration of smoking – year (mean (SD)) | 28.14 (9.43) | 30.43 (9.33) | 0.268 |
| Number of cigarettes per day - piece (median (min-max)) | 15 (10 - 80) | 17.5 (10 - 60) | 0.416 |
| Other people who smoked in the house – no. (%) | 18 (42.86) | 13 (30.95) | 0.258 |
| Trying to quit smoking – times (median (min-max)) | 1 (0 - 8) | 1 (0 - 10) | 0.876 |
| Duration of smoking cessation – day | 10 (0 - 730) | 30 (0 – 1,095) | 0.09 |
| (median (min-max)) | |||
| Exhaled carbon monoxide level – ppm | 14 (9 - 28) | 14 (5 - 26) | 0.753 |
| (median (min-max)) | |||
| Laboratory | |||
| Hemoglobin – g/dL - (median (min-max)) | 13.8 (7 - 18.1) | 13.75 (7.9 - 15.9) | 0.552 |
| Hematocrit – vol% - (median (min-max)) | 41.65 (21.5 - 51) | 40.7 (23.9 - 46.9) | 0.348 |
| Characteristics | Vernonia cinerea (N= 42) |
Nortriptyline (N= 42) |
p-value |
| Laboratory | |||
| White blood cell – cells/uL - (mean (SD)) | 8.56 (2.00) | 8.69 (1.95) | 0.775 |
| Platelet – cells/uL - (median (min-max)) | 301.5 (171 - 491) | 277.5 (144 - 540) | 0.144 |
| Serum Creatinine – ng/mL - (mean (SD)) | 0.87 (0.15) | 0.92 (0.13) | 0.11 |
| Estimated glomerular filtration rate – ml/min/1.73m2 - (mean (SD)) | 98.82 (13.43) | 94.88 (12.30) | 0.165 |
| Serum potassium – mmol/L - (median (min-max)) | 3.7 (3.5 - 5) | 3.8 (3.5 - 5) | 0.305 |
| Aspartate aminotransferase – U/L - (median (min-max)) | 29.5 (9 - 72) | 25 (12 - 64) | 0.629 |
| Alanine aminotransferase – U/L - (median (min-max)) | 29.5 (10 - 69) | 25 (9 - 78) | 0.558 |
Primary outcome
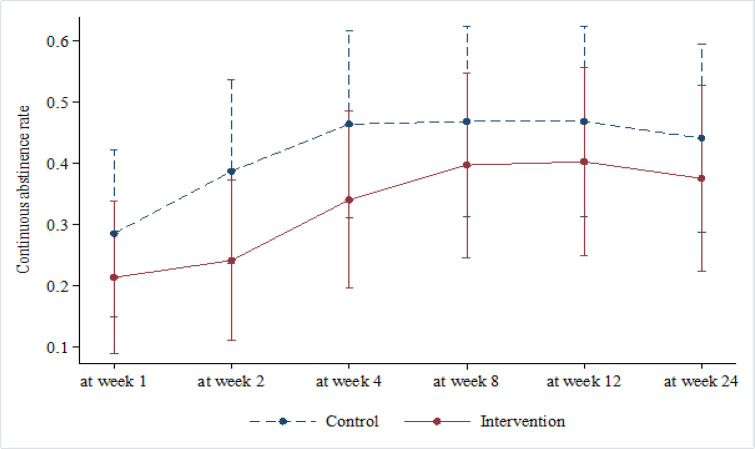
At the end of the research, there was no statistically significant difference of the CAR between VC and nortriptyline group with Odd ratio 0.68, 95%CI 0.25-1.85, P=0.451, but slightly higher in nortriptyline group in the first period (at week 1, 2, 4; 21.43%, 25.00%, 35.00% vs 28.57%, 38.46%, 46.15%, respectively). After week 8 until the end of treatment at week 12, the CAR of both groups were stable. There was also a constant rate of CAR at week 24, which was the wash out period (at week 8, 12, 24; 42.11%, 44.44%, 41.67% vs 45.95% , 45.95%, 43.24%, respectively) (Figure 2 and Table 2).
Figure 2.
Continuous Abstinence Rate and 95%CI at Week 1, 2, 4, 8, 12 and 24
Table 2.
Continuous Abstinence Rate (CAR) for Both Groups of VC and Nortriptyline
| Continuous abstinence rate | Vernonia cinerea | Nortriptyline | OR (95%CI) | p-value |
|---|---|---|---|---|
| Overall | 0.68 (0.25 – 1.85) | 0.451a | ||
| CAR at week 1 – no(%) | (n=42) | (n=42) | 0.68 (0.25 – 1.85) | >0.999b |
| 9 (21.43) | 12 (28.57) | |||
| CAR at week 2 – no(%) | (n=40) | (n=39) | 0.50 (0.16 – 1.60) | 0.465b |
| 10 (25.00)* | 15 (38.46)* | |||
| CAR at week 4 – no(%) | (n=40) | (n=39) | 0.59 (0.20 – 1.76) | 0.752b |
| 14 (35.00)* | 18 (46.15)* | |||
| CAR at week 8 – no(%) | (n = 38) | (n = 37) | 0.75 (0.23 – 2.47) | >0.999b |
| 16 (42.11)* | 17 (45.95)* | |||
| CAR at week 12 – no(%) | (n = 36) | (n = 37) | 0.77 (0.23 – 2.54) | >0.999b |
| 16 (44.44)* | 17 (45.95)* | |||
| CAR at week 24 – no(%) | (n = 36) | (n = 37) | 0.76 (0.23 – 2.55) | >0.999b |
| 15 (41.67)* | 16 (43.24)* |
a By generalized estimating equations (GEE) model; b Comparison of CAR between VC and nortriptyline at week 1, 2, 4, 8, 12 and 24 by Bonferroni test; * Comparison of CAR at week 1, 2, 4, 8, 12 and 24 vs CAR at week 1 by Bonferroni test and significance at p < 0.05
Secondary outcomes
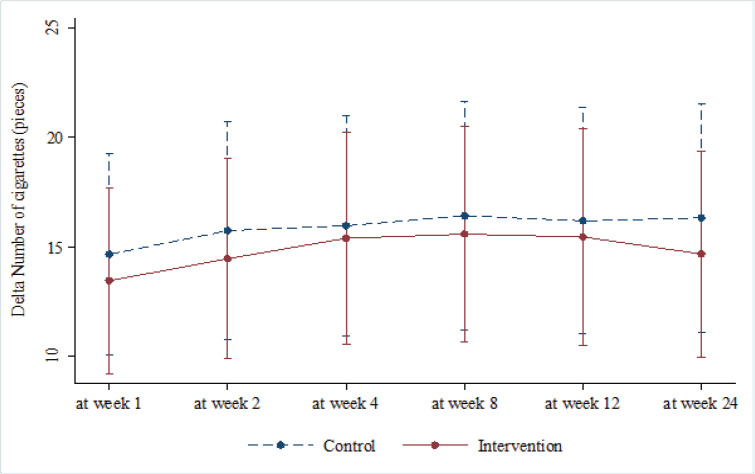
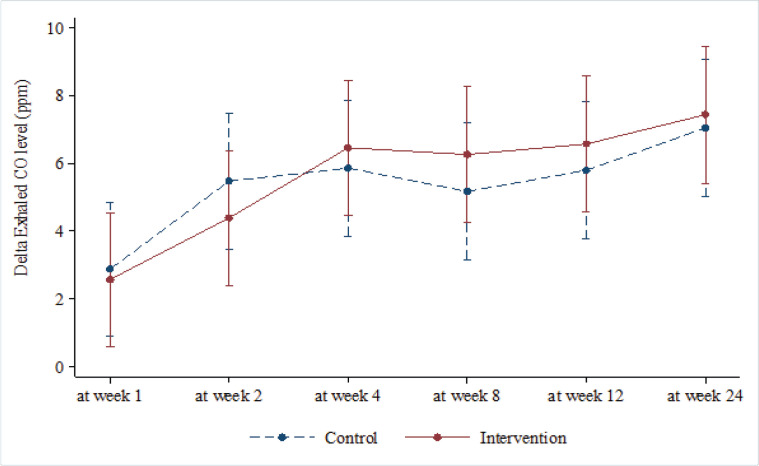
We found that by reducing the number of cigarettes per day by more than 10 in both groups, there was no difference between both groups throughout treatment period and wash out period. At the end of treatment, week 12, the change of number of cigarettes per day was 11.5 (4-30) vs 15 (5-55), IRR 0.95, 95%CI 0.52-1.75, P>0.999, and after week 24, without receiving any medication, both groups had reduced their number of cigarettes per day (12 (0-30) vs 15 (5-57), IRR 0.90, 95%CI 0.49-1.65, P>0.999). Overall, VC group had 8% smoking rate less than nortriptyline group, but with no statistically significant difference (IRR 0.92, 95%CI 0.59-1.43, P=0.702). Moreover, both groups could reduce the exhale CO level in treatment period and wash out period (week 12; 7 (-17-20) vs 7 (-12-16), mean difference 0.78, 95%CI -3.07-4.63, P>0.999, and week 24; 8 (-5-22) vs 8.5 (-5-17), mean difference 0.39, 95%CI -3.46-4.24, P>0.999). Overall, there was no statistically significant difference of the exhale CO level between both groups (mean difference -0.31, 95%CI -3.10-2.47, P=0.829). After 24 weeks of follow up, it can be concluded that the relapse rate showed no difference (13.89% vs 10.81%). The numbers of patients who quit smoking continuously at week 24 did not differ (41.67% vs 43.24%) as well as those that could not quit smoking (44.44% vs 45.95%, P=0.923) (Table 3, 4, and 5, Figure 3 and 4).
Table 3.
Number of Cigarettes Per Day for Both Groups of VC and Nortriptyline
| Number of cigarettes | Vernonia cinerea | Nortriptyline | IRR (95%CI) | p-value |
|---|---|---|---|---|
| Overall | 0.92 (0.59 – 1.43) | 0.702a | ||
| Δ Number of cigarettes | (n=42) | (n=42) | 0.92 (0.53 – 1.58) | >0.999b |
| – pieces at week 1 | 10 (3 - 77) | 12 (4 - 50) | ||
| Δ Number of cigarettes | (n=42) | (n=42) | 0.92 (0.53 – 1.58) | >0.999b |
| – pieces at week 2 | 12 (5 - 76)* | 15 (6 - 50)* | ||
| Δ Number of cigarettes | (n=42) | (n=42) | 0.97 (0.56 – 1.66) | >0.999b |
| – pieces at week 4 | 13 (5 - 77)* | 15 (5 - 51)* | ||
| Δ Number of cigarettes | (n = 38) | (n = 37) | 0.95 (0.52 – 1.74) | >0.999b |
| – pieces at week 8 | 12 (5 - 78)* | 15 (5 - 55)* | ||
| Δ Number of cigarettes | (n = 36) | (n = 36) | 0.95 (0.52 – 1.75) | >0.999b |
| – pieces at week 12 | 11.5 (4 - 30)* | 15 (5 - 55)* | ||
| Δ Number of cigarettes | (n = 36) | (n = 36) | 0.90 (0.49 – 1.65) | >0.999b |
| – pieces at week 24 | 12 (0 - 30)* | 15 (5 - 57)* |
a By generalized estimating equations (GEE) model; b Comparison of number of cigarettes between VC and nortriptyline at week 1, 2, 4, 8, 12 and 24 by Bonferroni test; * Comparison of number of cigarettes at week 1, 2, 4, 8, 12 and 24 vs number of cigarettes at week 1 by Bonferroni test and significance at p < 0.05; Δ Calculation by number of cigarettes – pieces at time point minus number of cigarettes – pieces at baseline
Table 4.
Exhaled carbon Monoxide Level for Both Groups of VC and Nortriptyline
| Exhaled CO level | Vernonia cinerea | Nortriptyline | mean difference (95%CI) | p-value |
|---|---|---|---|---|
| Overall | -0.31 | 0.829a | ||
| (-3.10 - 2.47) | ||||
| Δ Exhaled CO level | (n=42) | (n=42) | -0.31 | >0.999b |
| – ppm at week 1 | 3.5 (-12 - 19) | 3 (-19 - 15) | (-4.06 - 3.44) | |
| Δ Exhaled CO level | (n=40) | (n=39) | -1.08 | >0.999b |
| – ppm at week 2 | 3.5 (-6 - 20)* | 6 (-7 - 17)* | (-4.87 - 2.71) | |
| Δ Exhaled CO level | (n=40) | (n=39) | 0.61 | >0.999b |
| – ppm at week 4 | 6 (-8 - 23)* | 6 (-10 - 21)* | (-3.19 - 4.40) | |
| Δ Exhaled CO level | (n = 38) | (n = 37) | 1.09 | >0.999b |
| – ppm at week 8 | 6 (-9 - 21)* | 6 (-11 - 16)* | (-2.73 - 4.92) | |
| Δ Exhaled CO level | (n = 36) | (n = 36) | 0.78 | >0.999b |
| – ppm at week 12 | 7 (-17 - 20)* | 7 (-12 - 16)* | (-3.07 - 4.63) | |
| Δ Exhaled CO level | (n = 36) | (n = 36) | 0.39 | >0.999b |
| – ppm at week 24 | 8 (-5 - 22)* | 8.5 (-5 - 17)* | (-3.46 - 4.24) |
a By generalized estimating equations (GEE) model; b Comparison of exhaled CO level between VC and nortriptyline at week 1, 2, 4, 8, 12 and 24 by Bonferroni test; * Comparison of exhaled CO level at week 1, 2, 4, 8, 12 and 24 vs exhaled CO level at week 1 by Bonferroni test and significance at p < 0.05; Δ Calculation by exhaled CO level – ppm at time point minus exhaled CO level – ppm at baseline
Table 5.
Relapse of Smoking Rate – no. (%) for Both Groups of VC and Nortriptyline
| Relapse of smoking rate | Vernonia cinerea (N=36) | Nortriptyline (N=37) | p-value |
|---|---|---|---|
| Relapse of smoking rate | 0.923 | ||
| Smoking cessation continuously | 15 (41.67) | 16 (43.24) | |
| Relapse | 5 (13.89) | 4 (10.81) | |
| Current | 16 (44.44) | 17 (45.95) |
Figure 3.
Number of Cigarettes Per Day and 95%CI at week 1, 2, 4, 8, 12 and 24
Figure 4.
Exhaled CO Level and 95%CI at Week 1, 2, 4, 8, 12 and 24
Adverse events and withdrawal symptoms
We found that adverse events, including tongue bitter taste or numbness, were significantly greater in VC group (61.9% vs 30.95%, P=0.004), whereas dry mouth and drowsiness were significantly greater in nortriptyline group (35.71% vs 90.48%, P<0.001 and 16.67% vs 90.48%, P<0.001, respectively). Other adverse events and withdrawal symptoms were not different. Furthermore, serious adverse events were not found (Table 6 and 7).
Table 6.
Adverse Events - no. (%) for Both Groups of VC and Nortriptyline
| Adverse events | Vernonia cinerea (N= 42) | Nortriptyline (N= 42) | p-value |
|---|---|---|---|
| Tongue numbness | 26 (61.9) | 13 (30.95) | 0.004 |
| Dry mouth | 15 (35.71) | 38 (90.48) | <0.001 |
| Dizziness | 9 (21.43) | 8 (19.05) | 0.786 |
| Blurred vision | 1 (2.38) | 1 (2.38) | >0.999 |
| Abdominal pain/Nausea/Vomiting | 3 (7.14) | 2 (4.76) | >0.999 |
| Constipation | 0 (0.00) | 1 (2.38) | >0.999 |
| Urinary incontinence | 0 (0.00) | 0 (0.00) | NA |
| Drowsiness | 7 (16.67) | 38 (90.48) | <0.001 |
| Arrhythmia | 0 (0.00) | 0 (0.00) | NA |
| Palpitation | 2 (4.76) | 0 (0.00) | 0.494 |
| Others | 0 (0.00) | 0 (0.00) | NA |
Table 7.
Withdrawal Symptoms - no. (%) for Both Groups of VC and Nortriptyline
| Withdrawal symptoms | Vernonia cinerea (N= 42) |
Nortriptyline (N= 42) |
p-value |
|---|---|---|---|
| Craving | 31 (73.81) | 37 (88.10) | 0.095 |
| Moody | 19 (45.24) | 16 (38.10) | 0.507 |
| Anxiety | 1 (2.38) | 0 (0.00) | >0.999 |
| Insomnia | 0 (0.00) | 0 (0.00) | NA |
| Others | 0 (0.00) | 0 (0.00) | NA |
Discussion
The efficacy of VC showed non-inferiority to nortriptyline according to the composite outcomes of continuous abstinence rate, number of cigarettes per day, exhaled carbon monoxide level and relapse rate among the two treatment groups. It also showed no significant difference at all time during the treatment and the follow-ups. Patients who received VC and nortriptyline for the first four weeks experienced no difference in smoking cessation throughout, and there was upward trend of the number of patients who can quit smoking in both groups.
These findings were consistent with previous studies of Wongwiwatthananukit et al., (2018), Thripopsakul and Sittipun (2010), and Kitpaiboontawee et al., (2012) which provided shorter courses of treatment (2-4 weeks). In this study, long-term medication was given to patients in 12 weeks and followed up to 24 weeks. There was no statistically significant difference of CAR between VC and nortriptyline group. However, not similar to the previous findings of Chaikoolvatana et al., (2017) with a long-term treatment of 24 weeks, and a statistically significant difference of CAR. This might be from the difference in medication provided to their patients. The intervention group took VC, whereas the control group took a placebo. Therefore, the CAR reflected a statistically significant difference between the two groups. However, in this study, the active-comparator were given to both groups of patients. In a previous study by Chaikoolvatana et al., (2015), they had administered and monitored volunteers also for long-term studies (12 weeks), with the objective to assess the quality of life of patients. VC group gave significantly better results than placebo, but without continuously collecting information about CAR.
In addition, both groups could reduce the number of cigarettes per day compared to baseline. Overall, smoking rate of VC were 8% less than nortriptyline group, but not statistically significant. The data were based on self-report and direct observation (subjective data). Concrete measurements were made to confirm smoking cessation (objective data) by the exhaled carbon monoxide level assessment, which were lower than baseline and there was also no difference between groups. However, there was a limitation in this assessment because of only the 6 hour half-life of carbon monoxide, on average (Olson, 1984). Any low level of carbon monoxide or no measurable detection at all might only reflect that the patient did not smoke for more than 6 hours prior to the examination. It cannot confirm that the patient indeed stopped smoking at all. Moreover, higher carbon monoxide level cannot distinguish whether derived from smoking by themselves or, possibly from secondhand smoking, or by receiving carbon monoxide from other sources.
After followed up to week 24, which was the wash out period and according to the guidelines for patients who wanted to change their behavior (transtheoretical model, TTM) or Stage of Change model (Prochaska et al., 1992), there were patients who quit smoking finally in both groups, but no difference was found. The relapse of smoking rate was not different, and proportions of those who cannot stop smoking at all were also found with no difference between the two groups.
The adverse events in VC group were tongue bitter taste or numbness, which were statistically significantly greater than in the nortriptyline group. As part of the medical properties, VC has important substances; nitrite and nitrate containing substances that are the cause for the taste change. In nortriptyline group, the main adverse events were dry mouth and drowsiness, which were found significantly greater than VC group. Drowsiness is a result from the central nervous system effect of this drug. Dry mouth is a result of anticholinergic effect which can cause fainting, dizziness, blurred vision, constipation, urinary incontinence, gastrointestinal tract irritation which can cause abdominal pain, nausea and vomiting as well. However, the symptoms mentioned in both groups were consider: insignificantly different and no severe or life-threatening adverse events were found.
The strengths of this study was that it was a randomized controlled trial that had a high quality of evidence, with only few allocation biases. We studied about a wash out period, and we are the first who compared the efficacy and safety of VC and nortriptyline in long-term treatment. In addition, the characteristics of our patients were healthy and only some comorbidities. Our findings concluded no serious adverse events. The patients could take their own medication with VC. It can be safely used for long-term smoking cessation (12 weeks) for the chronic illnesses patients who desire to stop smoking.
The main limitation of this study is that it is an open-label trial, in which blinding was not performed. Although this is an open-label trial, we performed the allocation concealment and a good randomization to achieve results of similar characteristics between two treatment groups. Due to validity of the outcomes measured with precise 95% CIs in Table 2, 3 and 4, our findings are summarizable and generalizable to all similar settings and populations. Secondly, receiving lower sample size than previously anticipated due to patients unable to tolerate their drowsiness. Thirdly, in VC group patients had to take the drug 3 times a day, resulting in inconvenience during the day when working. This might cause poor compliance and the outcomes could be variable.
If offered in practice, VC may be appropriate for smokers with nortriptyline allergy, and for patients who currently use CNS depressant drugs. It would assist those who have used nortriptyline to help quit smoking but can’t tolerate its adverse events. It may also be an option (instead of the case with nortriptyline) for patients who have to drive a vehicle or use a machine during their smoking cessation.
In conclusion, the efficacy of Vernonia cinerea (VC) showed non-inferiority to nortriptyline for smoking cessation, reducing number of cigarettes per day, reducing exhaled carbon monoxide level and no difference in relapse rate between two treatment groups. For safety data, the adverse events including tongue bitter taste or numbness were found greater in VC group than in nortriptyline group, whereas dry mouth and drowsiness were found greater in nortriptyline group. Serious adverse events did not arise in either groups.
Author Contribution Statement
KT contributed to the conception, design, data analysis, and manuscript writing. JB contributed to the conception and design, data acquisition, analysis and interpretation of the data, and drafting of the article. NS contributed to the conception, design, and data acquisition. BC contributed to methodological supervision and final approval of the manuscript. Underlying data of this study are available upon reasonable request to the corresponding author. This study was registered in TCTR database (www.thaiclinicaltrials.org), No. TCTR20190506002 on 6 May 2019.
Acknowledgements
Authors appreciate all supports from Namphong Hospital, Khon Kaen, patients, volunteers, and all the staffs of all clinics involving in this study. Authors are grateful to Dr Jitjira Chaiyarit, biostatistician/epidemiologist, in helpful analysis and interpreting of the results. The authors also thank Mr Dirk Krapf for the English editing of the manuscript. Authors received a non-specific funding for this study from Department of Social Medicine, Khon Kaen Hospital (Reference Number: KEXP62005). This study was scientifically approved by the Postgraduate Committee of Medication Education Center, Khon Kaen Hospital, as the Family Medicine Board thesis of Dr Tuenthosarn. The authors have no financial or other conflict of interests to declare. Ethical justification of this study was also approved by Office of the Khon Kaen Hospital Institute Review Board in Human Research on 19 December 2018 (KE61095).
References
- Baumeister RF. Addiction, cigarette smoking, and voluntary control of action: Do cigarette smokers lose their free will? Addict Behav Rep. 2017;5:67–84. doi: 10.1016/j.abrep.2017.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berrettini WH. Nicotine dependence. Psychiatry (Edgmont) 2004;1:12–7. [PMC free article] [PubMed] [Google Scholar]
- Chaikoolvatana A, Ayuthaya ND, Suthipinittharm P, et al. Development and evaluation of the effectiveness of Vernonia cinerea (VC) cookies for smoking cessation. Tob Prev Cessation. 2017;3:45. [Google Scholar]
- Chaikoolvatana A, Sithibutra C, Chandee S, Chaikoolvatana C. A comparison of Smoking Cessation Drug Therapies Related Quality of Life between Vernonia cinerea Tea and 0. 5% Sodium Nitrate Mouthwash. Nurs J. 2015;42:178–92. [Google Scholar]
- Clinical Practice Guideline Treating Tobacco Use, Dependence 2008 Update Panel, Liaisons , Staff A clinical practice guideline for treating tobacco use and dependence: 2008 update. A U.S. Public Health Service report. Am J Prev Med. 2008;35:158–76. doi: 10.1016/j.amepre.2008.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fahey MC, Talcott WG, Robinson LA, et al. Predictors of Cessation Outcomes Among Older Adult Smokers Enrolled in a Proactive Tobacco Quitline Intervention. J Aging Health. 2022;2022:8982643221097679. doi: 10.1177/08982643221097679. [DOI] [PubMed] [Google Scholar]
- Fiore MC. Treating tobacco use and dependence: an introduction to the US Public Health Service Clinical Practice Guideline. Respir Care. 2000;45:1196–9. [PubMed] [Google Scholar]
- Haggsträm FM, Chatkin JM, Sussenbach-Vaz E, et al. A controlled trial of nortriptyline, sustained-release bupropion and placebo for smoking cessation: preliminary results. Pulm Pharmacol Ther. 2006;19:205–9. doi: 10.1016/j.pupt.2005.05.003. [DOI] [PubMed] [Google Scholar]
- Hall SM, Humfleet GL, Reus VI, Muñoz RF, Hartz DT, Maude-Griffin R. Psychological intervention and antidepressant treatment in smoking cessation. Arch Gen Psychiatry. 2002;59:930–6. doi: 10.1001/archpsyc.59.10.930. [DOI] [PubMed] [Google Scholar]
- Haxby DG. Treatment of nicotine dependence. Am J Health Syst Pharmacy. 1995;52:265–315. doi: 10.1093/ajhp/52.3.265. [DOI] [PubMed] [Google Scholar]
- Hughes JR, Stead LF, Lancaster T. Nortriptyline for smoking cessation: a review. Nicotine Tob Res. 2005;7:491–9. doi: 10.1080/14622200500185298. [DOI] [PubMed] [Google Scholar]
- Medawela RMSH, Ratnayake DRDL, Premathilake LKN, Jayasinghe R. Attitudes, Confidence in Practices and Perceived Barriers Towards the Promotion of Tobacco Cessation among Clinical Dental Undergraduates in Sri Lanka. Asian Pac J Cancer Care. 2021;6:175–9. [Google Scholar]
- Novesar A, Pramod S, Safriadi F. Cigarettes Smoking Pattern in Patient with Urothelial Carcinoma of the Bladder after Smoking Cessation in Tertiary Hospital. Asian Pac J Cancer Care. 2022;7:101–10. [Google Scholar]
- Olson KR. Carbon monoxide poisoning: mechanisms, presentation, and controversies in management. J Emerg Med. 1984;1:233–43. doi: 10.1016/0736-4679(84)90078-7. [DOI] [PubMed] [Google Scholar]
- Parrott S, Godfrey C, Raw M, West R, McNeill A. Guidance for commissioners on the cost effectiveness of smoking cessation interventions. Health Educational Authority Thorax. 1998;53:1–38. [PMC free article] [PubMed] [Google Scholar]
- Prasopthum A, Pouyfung P, Sarapusit S, Srisook E, Rongnoparut P. Inhibition effects of Vernonia cinerea active compounds against cytochrome P450 2A6 and human monoamine oxidases, possible targets for reduction of tobacco dependence. Drug Metab Pharmacokinet. 2015;30:174–81. doi: 10.1016/j.dmpk.2014.12.005. [DOI] [PubMed] [Google Scholar]
- Prochaska JO, DiClemente CC, Norcross JC. In search of how people change: Applications to addictive behaviors. Am Psychol. 1992;47:1102–14. doi: 10.1037//0003-066x.47.9.1102. [DOI] [PubMed] [Google Scholar]
- Raw M, McNeill A, West R. Smoking cessation guidelines for health professionals A guide to effective smoking cessation interventions for the health care system. Health Education Authority. Thorax. 1998;53:1–19. doi: 10.1136/thx.53.2008.s1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steinberg MB, Schmelzer AC, Richardson DL, Foulds J. The case for treating tobacco dependence as a chronic disease. Ann Intern Med. 2008;148:554–6. doi: 10.7326/0003-4819-148-7-200804010-00012. [DOI] [PubMed] [Google Scholar]
- Wagena EJ, Knipschild PG, Huibers MJ, Wouters EF, van Schayck CP. Efficacy of bupropion and nortriptyline for smoking cessation among people at risk for or with chronic obstructive pulmonary disease. Arch Intern Med. 2005;165:2286–92. doi: 10.1001/archinte.165.19.2286. [DOI] [PubMed] [Google Scholar]
- Wongwiwatthananukit S, Benjanakaskul P, Songsak T, Suwanamajo S, Verachai V. Efficacy of Vernonia cinerea for Smoking Cessation. J Health Res. 2018;23:31–6. [Google Scholar]
- World Health Organization (WHO) Report on the Global Tobacco Epidemic, 2008: the MPOWER package. Geneva: World Health Organization; 2008. [Google Scholar]
- Yunipan J, Suansane T, Chaosuancharoen T, Namchanthra P, et al. Clinical practice guidelines for the treatment of tobacco addiction in Thailand. Bangkok. Thailand Nursing and Midwifery Council; 2012. [Google Scholar]