Abstract
Background
This study was conducted to assess the efficacy of point-of-care (POC) procalcitonin (PCT) serial measurement in determining the antibiotic treatment duration in patients with ventilator-associated pneumonia (VAP).
Methods
One hundred patients were randomly recruited and then further randomly divided into two groups of 50 patients each. The first group used the POC PCT test along with the standard sepsis parameter monitoring, while the second group had the standard monitoring only (C-reactive protein [CRP] level, total white count, temperature and tracheal aspirate culture). Serial PCT test results and CRP levels were monitored on days 1, 3, 7 and 9. The patients were followed up for 28-day mortality.
Results
Eighty-five patients completed the trial, of whom 43 were in the PCT group and 42 were in the control group. The PCT group had a significantly lower mean (SD) antibiotic treatment duration (10.28 [2.68] days) than the control group (11.52 [3.06]). The mean (SD) difference was −1.25 (95% confidence interval [CI], −2.48 to 0.01; t-statistic [df] = −1.997 [83]; P = 0.049). The PCT group also had a higher number of antibiotic-free days alive during the 28 days after VAP onset than the control group (mean [SD], 10.79 [7.61] vs. 8.72 [6.41]). The Sequential Organ Failure Assessment score was the sole factor for the decrease in duration after VAP onset (regression coefficient b [95% CI], −0.70 [−1.19 to −0.20]; P = 0.006).
Conclusions
The POC procalcitonin test can reduce the antibiotic treatment duration in patients with VAP.
Keywords: procalcitonin, point-of-care, clinical pulmonary infection score, C-reactive protein, ventilator-associated pneumonia
Ventilator-associated pneumonia (VAP) remains a leading cause of nosocomial infections affecting most patients in intensive care units (ICU) worldwide [1]. According to a systematic review from 2019 [1], the incidence of VAP is higher in developing countries than in developed countries (18.5 and 9.0 per 1000 ventilator-days, respectively). Pseudomonas aeruginosa (n = 3176 [22%]), Acinetobacter baumannii (n = 3687 [26%]) and Staphylococcus aureus (n = 1999 [14%]) were the leading causes of VAP with incidence of carbapenem resistance as high as 57.1% [1]. Until now, no single criterion has been established for the diagnosis of VAP. Therefore, VAP should be suspected in the event of deterioration of respiratory function and evidence of infection such as worsening of oxygenation and hypotension with or without new or progressive pulmonary infiltrates [2].
The risk of VAP increases by 1–3% every day the patient is ventilated. The risk may even go higher in patients with a high initial Sequential Organ Failure Assessment (SOFA) score [3]. Rello et al. [4] reported that the incidence of VAP in the United States ranged from 24% to 50% of all nosocomial infections. In Malaysia, the incidence is approximately 1.7 per 1000 ventilated patients based on the Malaysia Regi-stry of Intensive Care 2017 [5]. The most important issues in the management of VAP include: the question of whether to start antibiotic administration, the antibiotic treatment duration, and which parameters should be assessed in managing patients with VAP. Overly long antibiotic treatment is undesirable because of side effects, increased antibiotic resistance and financial burden to the patient and national health care. Nevertheless, many authors have agreed that antibiotic therapy must be started early for VAP, as the mortality rate increases if the initiation of anti-biotic administration is delayed [4].
PCT is released during the inflammatory response, probably due to its anti-inflammatory properties, such as the ability to inhibit the overproduction of tumour necrosis factor α and interleukin-1β. Additionally, it has been postulated that PCT can also reduce the reactivity of lipopolysaccharide in Gram-negative bacteria [6]. Point-of-care (POC) PCT testing provides a great advantage in deciding to discontinue the antibiotics along with other available parameters such as C-reactive protein (CRP) level, total white blood cell count (TWC), temperature and cultures from tracheal aspirate or bronchoalveolar lavage. A systematic review and meta-analysis estimated the accuracy of the PCT test to be 71% for both sensitivity and specificity when used alone in a critically ill patient [7]. Nevertheless, the sensitivity and specificity of this test increase when combined with other septic parame-ters [8].
A European Society of Intensive Care Medicine (ESICM) systematic review and meta-analysis revealed that PCT-guided antibiotic therapy increased the antibiotic-free days within the first 28 days of hospitalisation in comparison with antibiotic therapy without PCT guidance; however, no significant difference in mortality was found [9]. Studies have also shown that PCT guidance can reduce the duration of antibiotic treatment for patients with bacterial infections in the ICU, without compromising safety outcomes [9–11].
The emergence of the multidrug resistant orga-nism (MDRO) and carbapenem-resistant Entero-bacteriaceae has become a challenge to the physician. These could result from an inappropriate antibiotic treatment duration, wrong choice of antibiotics, or the use of antibiotics not indicated for a particular patient [12]. These situations waste resources and can even create new resistance to the last line of antibiotics. However, the use of PCT must be guided by other clinical parameters, as the PCT test can yield false-negative results [13].
Therefore, the aim of this study was to analyse the role of POC PCT in reducing the antibiotic treatment duration as a primary outcome and to observe antibiotic-free days in a 28-day observation period. This is also the first study on the POC PCT test that was conducted in Malaysia for patients with VAP that aimed to promote antibiotic stewardship by reducing the overuse and overprescription of antibiotics in ICUs.
METHODS
Study design and respondents
This was a prospective interventional single-blinded study conducted in the critical care unit of Hospital Universiti Sains Malaysia (HUSM), which included the ICU, surgical ICU, neuro-ICU and trauma ICU, between June 2019 and May 2020. It was approved by the research ethics committee (human; JEPeM of Universiti Sains Malaysia, JEPeM code: USM/JEPeM/17100452). The clinical trial registration number is NCT03982667.
The patients were recruited from medical, surgical, trauma and neurosurgical ICUs of HUSM from June 2019 to May 2020 after written consent was obtained from the patients. All the patients who were admitted in 4 ICUs in the HUSM in a 1-year period were screened. The ventilated patients were identified. The patients who fulfilled the study inclusion criteria were enrolled and analysed.
The Random Version 1.0 allocation software (Isfa-han University of Medical Sciences, Isfahan, Iran) was applied for sample randomisation. By using the randomisation software, the PCT group was labelled as group 1 and the control group as group 2.
The inclusion and exclusion criteria were as follows:
- inclusion criteria:
- age of ≥ 18 years,
- ICU admission,
- ventilation for > 48 hours,
- received the first antibiotic dose at ≤ 24 h before inclusion in the trial for an assumed or proven VAP;
- exclusion criteria:
- systemic antibiotics as prophylaxis or treatment and not for VAP,
- antibiotics solely as part of selective decontamination of the digestive tract,
- prolonged therapy (e.g. endocarditis),
- expected ICU stay of < 24 h,
- severe immunosuppression,
- severe infections due to non-bacterial causes,
- previously enrolled in the study.
No specific antibiotics were allocated to both groups. The antibiotic selection was entirely at the attending physician’s discretion based on either the sensitivity of the organism or as empirical treatment. No intervention for starting the antibiotic treatment was initiated.
Measurements
After patient enrolment based on the inclusion criteria, the patients’ demographic data such as age, height, weight and comorbidities were recorded. The clinical pulmonary infection score (CPIS) was used as a diagnostic tool for VAP. Patients ventilated for > 48 hours who had CPISs > 5 were enrolled in the study. The patients were randomised to PCT and control groups. For the PCT group, 2 mL blood samples were withdrawn from each patient and kept in an ethylene diamine tetraacetic acid bottle. The sample was immediately analysed by the investigator using the Finecare Plus analyser (Wondfo, Guangzhou, China), and the results were available after 15 minutes. The serial PCT levels were monitored on days 1, 3, 7 and 9. In the control group, serial CRP levels were monitored in accordance with the physician’s decision.
In a Pro VAP trial, Stolz et al. [14] made the following recommendations according to PCT level:
if the PCT level is < 0.25 ng mL-1, stopping the antibiotic therapy is strongly encouraged;
if the PCT level decreased by > 80% from the peak PCT level or is > 0.25 but < 0.5 ng mL-1, stopping the antibiotic therapy is encouraged;
if the PCT level decreased by < 80% from the peak or is > 0.5 ng mL-1, continuing the antibiotic therapy is encouraged;
if the PCT level has increased and is > 0.5 ng mL-1, continuing the antibiotic therapy is strongly encouraged.
Nevertheless, the final decision regarding the continuation or termination of antibiotic treatment depends on the treating physician, as other parameters can add extra value in managing the patient (i.e. TWC, temperature, differential count and haemodynamic status).
Statistical analyses
All the statistical analyses were performed using the SPSS software version 26.0 (IBM Corp., New York, United States of America). Discrete variables are expressed as count or percentage. Continuous variables are expressed as mean (SD). The control and PCT groups were compared using the c2 or Fisher exact test for categorical data and the independent t test for continuous variables. Multivariable analysis by linear regression was performed to analyse the influence of the two treatments on the total antibiotic and antibiotic-free days after treatment over 28 days of observation. Statistical significance was defined as P < 0.05.
RESULTS
Of 100 patients, only 85 were included in the analyses. The remaining 15 patients were excluded based on the exclusion criteria. In total, seven patients were excluded from the follow-up because of the identification of MDRO that required longer antibiotic treatment. Five patients died and the other three patients were lost to follow-up as shown in Figure 1.
FIGURE 1.
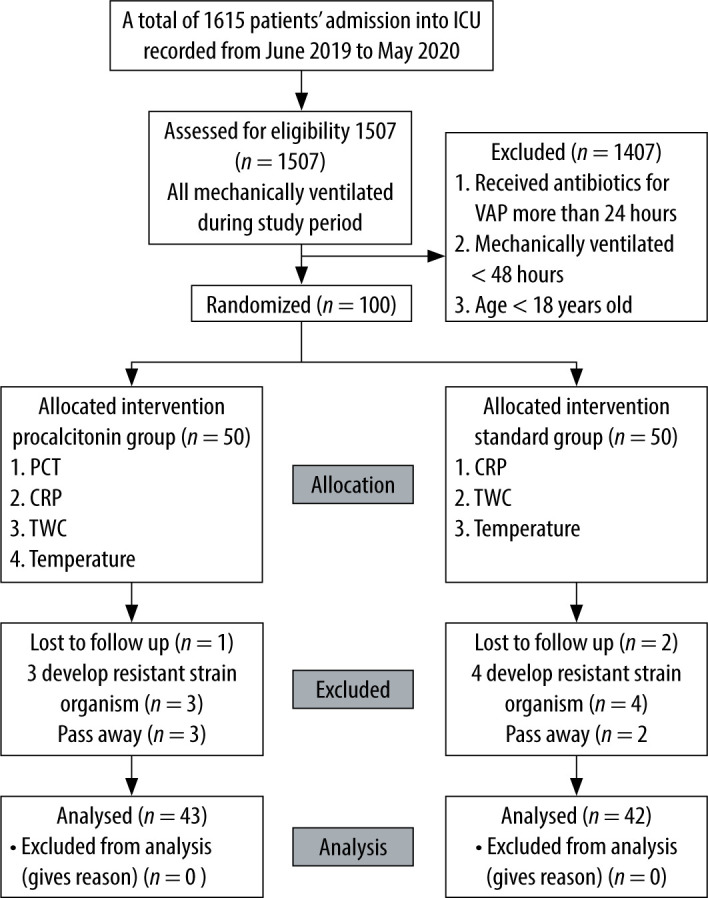
Flowchart of the study
The PCT group was composed of 43 patients, the control group 42 patients. The results showed that the mean (SD) age of the participants was 51.07 (16.81) years. About half of the participants in the total study sample were male (n = 46, 54.1%). No significant differences in SOFA score, Acute Physiologic Assessment and Chronic Health Evaluation II (APACHE II) score or demographic characteristics were found, as shown in Table 1. More than half of the patients in both groups had diabetes, hypertension and acute kidney injury. The most common reason for intubation in both groups was hypoxaemic respiratory failure.
TABLE 1.
Baseline characteristics of the 85 patients randomised to the control or PCT group
| Variable | Total n (%) | Groups | P-value | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PCT n (%) | Control n (%) | |||||||||||
| Age (years)* | 51.07 (16.81) | 49.07 (17.08) | 53.12 (16.49) | 0.270a | ||||||||
| Sex, n (%) | ||||||||||||
| Male | 46 (54.1) | 27 (31.8) | 19 (22.4) | 0.104b | ||||||||
| Female | 39 (45.9) | 16 (18.8) | 23 (27.1) | |||||||||
| APACHE II score* | 18.64 (5.46) | 18.77 (6.23) | 18.50 (4.61) | 0.823a | ||||||||
| SOFA score* | 8.04 (2.99) | 8.05 (3.14) | 8.02 (2.86) | > 0.950a | ||||||||
| Duration (days)* | 10.89 (2.92) | 10.28 (2.68) | 11.52 (3.06) | 0.049a | ||||||||
| Antibiotic-free days* | 9.77 (7.08) | 10.79 (7.61) | 8.72 (6.41) | 0.179a | ||||||||
| Antibiotic used, n (%) | ||||||||||||
| Piperacillin-tazobactam | 39 (45.9) | 20 (46.5) | 19 (45.2) | 0.852c | ||||||||
| Meropenem | 29 (34.1) | 13 (30.2) | 16 (38.1) | |||||||||
| Cefepime | 6 (7.1) | 4 (9.3) | 2 (4.8) | |||||||||
| Polymycin | 1 (1.2) | 1 (2.3) | 0 (0.0) | |||||||||
| Ampicillin-sulbactam | 7 (8.2) | 3 (7.0) | 4 (9.5) | |||||||||
| Ceftriaxone | 3 (3.5) | 2 (4.7) | 1 (2.4) | |||||||||
| Retreatment, n (%) | ||||||||||||
| Yes | 31 (36.5) | 14 (32.6) | 17 (40.5) | 0.448b | ||||||||
| No | 54 (63.5) | 29 (67.4) | 25 (59.5) | |||||||||
| Mortality, n (%) | ||||||||||||
| Alive within 28 days | 60 (70.6) | 31 (72.1) | 29 (69.0) | 0.792b | ||||||||
| Death in the ICU | 13 (15.3) | 7 (16.3) | 6 (14.3) | |||||||||
| Death within 28 days | 12 (14.1) | 5 (11.6) | 7 (16.7) | |||||||||
| Organism, n (%) | ||||||||||||
| Klebsiella pneumoniae | ||||||||||||
| Yes | 38 (44.7) | 18 (41.9) | 20 (47.6) | 0.593b | ||||||||
| No | 48 (55.3) | 25 (58.1) | 22 (52.4) | |||||||||
| Pseudomonas aeruginosa | ||||||||||||
| Yes | 42 (49.4) | 21 (48.8) | 21 (50.0) | 0.915b | ||||||||
| No | 43 (50.6) | 22 (51.2) | 21 (50.0) | |||||||||
| Staphylococcus aureus | ||||||||||||
| Yes | 23 (24.2) | 7 (16.3) | 5 (11.9) | 0.563b | ||||||||
| No | 73 (85.9) | 36 (83.7) | 37 (88.1) | |||||||||
| Acinetobacter spp. | ||||||||||||
| Yes | 9 (10.6) | 4 (9.3) | 5 (11.9) | 0.738c | ||||||||
| No | 76 (89.4) | 39 (90.7) | 37 (88.1) | |||||||||
| Citrobacter koseri | ||||||||||||
| Yes | 7 (8.2) | 5 (11.6) | 2 (4.8) | 0.433c | ||||||||
| No | 78 (91.8) | 38 (88.4) | 40 (95.2) | |||||||||
| Morganella morganii | ||||||||||||
| Yes | 3 (3.5) | 3 (7.0) | 0 (0.0) | 0.241c | ||||||||
| No | 82 (96.5) | 40 (93.0) | 42 (100.0) | |||||||||
| Streptococcus pneumoniae | ||||||||||||
| Yes | 5 (5.9) | 1 (2.3) | 4 (9.5) | 0.202c | ||||||||
| No | 80 (94.1) | 42 (97.7) | 38 (90.5) | |||||||||
| Enterobacter spp. | ||||||||||||
| Yes | 6 (7.1) | 3 (7.0) | 3 (7.1) | > 0.950c | ||||||||
| No | 79 (92.9) | 40 (93.0) | 39 (92.9) | |||||||||
| No growth | ||||||||||||
| Yes | 13 (15.3) | 8 (18.6) | 5 (11.9) | 0.391b | ||||||||
| No | 72 (84.7) | 35 (81.4) | 37 (88.1) | |||||||||
Mean (SD). APACHE – Acute Physiological and Chronic Health Evaluation, SOFA – Sequential Organ Failure Assessment. aIndependent t test was applied. bPearson’s c2 test was applied. cFisher’s exact test was applied.
The mean (SD) antibiotic treatment duration was significantly lower by 1.25 days in the PCT group than in the control group (10.28 [2.68] days vs. 11.52 [3.06]), with a 95% CI of −2.48 to −0.01, t-statistic (df) of −1.997 (83) and P = 0.049). The PCT group also had a higher number of antibiotic-free days alive during the 28 days after VAP onset than the control group (10.79 [7.61] days vs. 8.72 [6.41] days), although the difference was not statistically significant.
On the basis of the analysed data, no significant associations between organism type, antibiotic use, retreatment and mortality were found in the two-group comparison. The distribution was quite similar for all the parameters compared. Multivariable analysis using simple and multiple linear regression revealed that the PCT and control groups were independent predictors of antibiotic treatment duration (b [95% CI], 1.25 [0.01–2.48]; P = 0.049), as shown in Table 2. The control group had a 1.25 times higher chance of experiencing longer antibiotic treatment duration than the procalcitonin group (b [95% CI], 1.25 [0.01–2.48]) when adjustments for other variables were made.
TABLE 2.
Association of variables with higher antibiotic treatment duration
| Variable | Simple linear regression | Multiple linear regression | |||
|---|---|---|---|---|---|
| Crude b (95% CI) | P-value | Adjusted b (95% CI) | P-value | ||
| GroupPCT | 0 | 0.049 | 0 | 0.049 | |
| Control | 1.25(0.01, 2.48) | 1.25(0.01, 2.48) | |||
| SOFA score | 0.09(−0.12, 0.31) | 0.383 | |||
| APACHE IIscore | 0.02(−0.10, 0.13) | 0.803 | |||
APACHE – Acute Physiological and Chronic Health Evaluation, SOFA – Sequential Organ Failure Assessment, b – regression coefficient.
A simple linear regression analysis was applied to examine the association of the groups, SOFA score, and APACHE II score with the antibiotic-free days, as summarised in Table 3. As shown, the variables were significantly related to the SOFA (P = 0.006) and APACHE II score (P = 0.016). We can conclude that the higher the SOFA and APACHE II score, the longer the patient is exposed to antibiotics. The subsequent multiple linear regression model clarifies that the only variable that can be considered an independent risk factor of an increased number of antibiotic-free days from VAP onset was SOFA score (b [95% CI], −0.70 [−1.19 to −0.20]; P = 0.006) when adjustments for other variables were made.
TABLE 3.
Association between the variables for higher of number of antibiotic-free days
| Variable | Simple linear regression | Multiple linear regression | ||
|---|---|---|---|---|
| Crude b (95% CI) | P-value | Adjusted b (95% CI) | P-value | |
| Group | 0.179 | |||
| PCT | 0 | |||
| Control | −2.07(−5.11, 0.97) | |||
| SOFA score | −0.70(−1.19, −0.20) | 0.006 | −0.70(−1.19, −0.20) | 0.006 |
| APACHE IIscore | −0.34(−0.61, −0.07) | 0.016 | ||
APACHE – Acute Physiological and Chronic Health Evaluation, SOFA – Sequential Organ Failure Assessment, b – regression coefficient.
DISCUSSION
A significant association was found between the groups in terms of antibiotic treatment duration (P = 0.049) when adjustments for other variables were made. The antibiotic treatment duration had a 1.25 higher chance of increasing in the PCT group than in the control group (b [95% CI], 1.25 [0.01–2.48]) when adjustments for other variables were made. In this study, the use of POC PCT as an additional biomarker reduced the antibiotic treatment duration by 1.25 days with a significance of P < 0.049. This value is much lower when compared with those reported in previous studies, where the reduction in the number of antibiotic days was up to 3 to 4 days in the patients with VAP [14]. In the ESICM systematic review and meta-analysis on PCT-guided antibiotic therapy (2012), the antibiotic treatment duration was reduced by −3.15 days (95% CI: −4.36 to −1.95; P < 0.001) [15]. Corti et al. [16] reported a median antibiotic exposure duration of 3.5 days (interquartile range [IQR], 0–10 days) in the POC PCT test for patients with COPD. In the control group, the antibiotic duration was 8.5 days (IQR, 1–11 days); P = 0.0169 (Wilcoxon test) [16]. However, these positive results may be a stepping stone to further improve the management of VAP and a cornerstone for using the PCT test as a vital tool in deciding the antibiotic treatment duration.
We also found that although the antibiotic treatment duration was shorter in the PCT group by 1.25 days, the recurrent infection rate, number of antibiotic-free days and mortality rate did not significantly differ from those in the control group. The number of retreatments (further bouts of infection) was 14 (32.6%) in the PCT group and 17 (40.5%) in the control group (P = 0.448). On the other hand, the mean number of antibiotic-free days was 10.79 days in the PCT group and 8.72 days in the control group (P = 0.179). One possible explanation for this situation is that most of the patients admitted to the ICU had similar higher SOFA and APACHE scores. As mentioned in the analysis of the results, the SOFA score directly correlated with the infection recurrence rate. In the PneumA Trial (2004), no significant differences in mortality and recurrent infection rate were found when antibiotic treatment was prolonged for > 8 days as compared with 15 days in the patients with bronchoscopically confirmed infection [9]. The study also showed no significant differences in length of ICU stay, the number of mechanical ventilation-free days and mortality rate at day 60 [9].
Our study shows that the recurrent infection rate was 32.6% (14 cases) in the PCT group and 40.5% (17 cases) in the control group (P = 0.448). Our findings indicate that discontinuation of antibiotic treatment based on PCT levels was associated with high recurrent infection rates. This finding is similar to that of a recent trial conducted in 2019 that revealed a recurrent infection rate of 32.9% after discontinuation of antibiotic within 7 days [17]. Bouadma et al. revealed in the PRORATA trial 2010 that the incidence of superinfection in the PCT group was 34.9% of 307 patients in the PCT arm and 30.9% of the 314 patients in the non-PCT arm [18].
One meta-analysis proved a positive relationship between antibiotic treatment duration and number of antibiotic-free days in a septic patient with Gram-negative bacilli (GNB) growth [9]. This meta-analysis revealed that increased antibiotic treatment duration would only increase the chances of culturing resistant strains of Enterobacteriaceae and other pathogens [9, 10]. De Waele et al. [10] highlighted that prolonged antibiotic exposure increases the development of antimicrobial resistance and that long courses of antibiotic therapy must be avoided in GNB infections. Moreover, some GNB can mutate into a resistant strain, which makes the patient more vulnerable to an infection recurrence [10].
Although many studies have proved the shorter antibiotic course in VAP, adherence to the proposed guidelines is still difficult, as the physician must consider the overall condition of the patient. We found that most of the adherence cases were from among patients with traumatic brain injury, with seven cases from among 43 patients recruited under the PCT group (16.3%). Most of the patients in the neurosurgical ICU had lower SOFA and APACHE scores. The weaning process is slow owing to the poor consciousness level, but the overall response to a single course of antibiotics was good. Most of the neurosurgical patients presented with a non-infectious pathology, so the diagnosis of VAP was easier for us and starting the antibiotic therapy early in this population was easier for us. In the PCT group, in which the protocol was not adhered to, all 12 patients presented more medical and surgical causes, such as septic shock of various aetiologies. How-ever, we found no significant difference between the groups in the recurrent infection rate or number of antibiotic-free days over the 28 days of observation.
In the case of 12 (27.9%) patients from the PCT group, the protocol was not followed. The reasons for this non-adherence included: slow progression in the patient’s general condition despite the improvement in the septic parameters; inability to wean from the ventilator; new onset of sepsis; development of a resistant strain; other septic parameters not consistent with the PCT level (e.g. a significant reduction in PCT level but with persistently high or elevated TWC); physician experience; a need for another surgical intervention for VAP recurrence and isolation of the same organism on repeated cultures of either resistant or sensitive strains.
Diagnosing and managing VAP are challenging and remain controversial in certain areas. Various diagnostic criteria have been suggested for VAP, but one of the commonly used ones is the CPIS, which was applied in this study. The other criteria used for the diagnosis of VAP are Johanson’s criteria for VAP, the criteria of the American Thoracic Society for VAP and the criteria of the European Centre for Disease Prevention and Control VAP. The CPIS has 42% and 90% sensitivity and specificity, respectively, in diagnosing VAP [19, 20]. Nevertheless, on the basis of the CHEST 2014 studies, a CPIS > 6 has a sensitivity of 39.8% and a specificity of 83.3%, with an odds ratio (OR) of 3.309. The introduction of the Chest Echography and Procalcitonin Pulmonary Infection Score from a pilot study in 2014 improved the CPIS accuracy, with a sensitivity of 80.5%, a specificity of 85.2% and an OR of 23.78 [21]. In our study, CPIS was applied for diagnosing VAP, with a sensitivity and specificity of 72% and 85%, respectively [19]. The POC PCT test may improve the CPIS sensitivity and specificity for diagnosing VAP.
The greatest advantage of this study is the POC approach. POC, also known as near-patient testing, is defined as an investigation at the time of consultation with instant availability of results to make an immediate decision on a particular management, especially with regard to antibiotics. Series PCT values provide a general view of whether the patient is receiving appropriate treatment or if the treatment can be optimised further. This series of values also provides clues about patients who could have a recurrent or superimposed infection despite still being in treatment. Hence, this study proved the safety and efficacy of the POC PCT test in patients with VAP.
Non-fermenting GNB were the leading culprit of VAP in our population. The most common organisms were Pseudomonas sp. and Klebsiella sp. These findings are almost similar to those in other established studies where GNB were the main culprit [9]. We also found that some of these positive cultures later developed a higher resistance strength, which required either prolonged antibiotic treatment duration or an escalation of antibiotic dose. Although a 7-day course of antibiotic therapy rather than a longer duration was recommended for patients with VAP, short antibiotic treatment courses were associated with recurrent infection in the subgroup of patients with VAP due to non-fermenting GNB [9]. Meanwhile, a recent systematic review of studies on community-acquired pneumonia (CAP) found that the PCT test was ineffective for the guidance of antibiotic therapy. The PCT test was found to be unable to differentiate viral or bacterial CAP [22]. The current Surviving Sepsis Campaign guidelines suggest that the PCT test is useful for shortening the antibiotic treatment duration but with low-quality evidence. The use of the PCT test was not recommended by the Infectious Diseases Society of America for initiating antibiotic treatment for VAP [23]. Therefore, the use of PCT must be on a case-to-case basis combined with clinical parameters to assist clinicians in promoting antibiotic stewardship [24]. For patients with suspected recurrence of superinfection, an antibiotic treatment that covers Gram-negative organisms, including multiresistant strains, is required [25]. Therefore, in the state of septic shock, successful treatment requires a “hit hard and hit fast” approach with broad-spectrum antibiotics such the carbapenem groups. The value of the PCT test in the management of acute oliguric acute kidney injury must be carefully interpreted in view of the potentially false-positive result due to the slow clearance of inflammatory mediators [26]. The current recommendation of the Food and Drug Admini-stration for the use of the PCT test in lower respiratory tract infections is strong discouragement of antibiotic use for PCT levels < 0.1 ng mL-1 and those ranging from 0.1 to 0.25 ng mL-1, encouragement of antibiotic use for PCT levels ranging from 0.25 to 0.5 ng mL-1 and strong encouragement of antibiotic use for PCT levels > 0.5 ng mL-1 [27].
Another limitation of this study is the relatively small population, although it was large enough to demonstrate the difference in or significance of antibiotic treatment duration. In future studies, we should aim for a larger homogeneous population with a longer study period to support the addition of PCT level as an excellent biomarker that influences the antibiotic treatment duration in patients with VAP.
CONCLUSIONS
POC PCT provides added value to shorten the antibiotic treatment duration in patients with VAP. The PCT level also demonstrated no significant difference in 28-day mortality when the antibiotic treatment duration was shortened as compared with the conventional 14-day antibiotic therapy.
ACKNOWLEDGEMENTS
We would like to express our sincere gratitude to the School of Medical Sciences, Universiti Sains Malaysia, for providing the Bridging grants 304.PPSP.6316494, used to support the study.
Financial support and sponsorship
none.
Conflict of interest
none.
References
- 1.Bonell A, Azarrafiy R, Huong VT, et al. A systematic review and meta-analysis of ventilator-associated pneumonia in adults in Asia: an analysis of national income level on incidence and etiology. Clin Infect Dis 2019; 68: 511-518. doi: 10.1093/cid/ciy543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Papazian L, Klompas M, Luyt CE. Ventilator-associated pneumonia in adults: a narrative review. Intensive Care Med 2020; 46: 888-906. doi: 10.1007/s00134-020-05980-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Čiginskienė A, Dambrauskienė A, Rello J, Adukauskienė D. Ventilator-associated pneumonia due to drug-resistant Acinetobacter baumannii: risk factors and mortality relation with resistance profiles, and independent predictors of in-hospital mortality. Medicina (Kaunas) 2019; 55: 49. doi: 10.3390/medicina55020049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Rello J, Ollendorf DA, Oster G, et al. Epidemiology and outcomes of ventilator-associated pneumonia in a large US database. Chest 2002; 122: 2115-2121. doi: 10.1378/chest.122.6.2115. [DOI] [PubMed] [Google Scholar]
- 5.Tai LL, Lim CH, Mohd Nor MR, Ismail NI, Wan Ismail WN. Malaysian Registry of Intensive Care 2016 report. Available at: https://www.crc.gov.my/wp-content/uploads/documents/report/mric_report_2016.pdf (Accessed: 26.12.2020).
- 6.Link T, Jacobson R, Escorcia I, Fisher J, Nguyen L, Reddy M. Procalcitonin and bacterial infections in the human body. FASEB J 2020; 34: 1.doi: 10.1096/fasebj.2020.34.s1.05445. [DOI] [Google Scholar]
- 7.Tang BM, Eslick GD, Craig JC, McLean AS. Accuracy of procalcitonin for sepsis diagnosis in critically ill patients: systematic review and meta-analysis. Lancet Infect Dis 2007; 7: 210-217. doi: 10.1016/S1473-3099(07)70052-X. [DOI] [PubMed] [Google Scholar]
- 8.Kollef MH, Sherman G, Ward S, Fraser VJ. Inadequate antimicrobial treatment of infections: a risk factor for hospital mortality among critically ill patients. Chest 1999; 115: 462-474. doi: 10.1378/chest.115.2.462. [DOI] [PubMed] [Google Scholar]
- 9.Chastre J, Wolff M, Fagon JY, et al. Comparison of 8 vs 15 days of antibiotic therapy for ventilator-associated pneumonia in adults: a randomised trial. JAMA 2003; 290: 2588-2598. doi: 10.1001/jama.290.19.2588. [DOI] [PubMed] [Google Scholar]
- 10.De Waele JJ, Martin-Loeches I. Optimal duration of antibiotic treatment in Gram-negative infections. Curr Opin Infect Dis 2018; 31: 606-611. doi: 10.1097/QCO.0000000000000491. [DOI] [PubMed] [Google Scholar]
- 11.Wongsurakiat P, Tulatamakit S. Clinical pulmonary infection score and a spot serum procalcitonin level to guide discontinuation of antibiotics in ventilator-associated pneumonia: a study in a single institution with high prevalence of nonfermentative gram-negative bacilli infection. Ther Adv Respir Dis 2018; 12: 1753466618760134. doi: 10.1177/1753466618760134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Porzecanski I, Bowton DL. Diagnosis and treatment of ventilator-associated pneumonia. Chest 2006; 130: 597-604. doi: 10.1378/chest.130.2.597. [DOI] [PubMed] [Google Scholar]
- 13.Caldararo MD. A Gram-negative false-negative: normal procalcitonin level in a patient with multiple Klebsiella pneumoniae intra-abdominal abscesses: a case report and literature review. Med Forum 2019; 20: 36-39. [Google Scholar]
- 14.Stolz D, Smyrnios N, Eggimann P, et al. Procalcitonin for reduced antibiotic exposure in ventilator-associated pneumonia: a randomised study. Eur Respir J 2009; 34: 1364-1375. doi: 10.1183/09031936.00053209. [DOI] [PubMed] [Google Scholar]
- 15.Matthaiou DK, Ntani G, Kontogiorgi M, Poulakou G, Armaganidis A,Dimopoulos G. An ESICM systematic review and meta-analysis of procalcitonin-guided antibiotic therapy algorithms in adult critically ill patients. Intensive Care Med 2012; 38: 940-949. doi: 10.1007/s00134-012-2563-7. [DOI] [PubMed] [Google Scholar]
- 16.Corti C, Fally M, Fabricius-Bjerre A, et al. Point-of-care procalcitonin test to reduce antibiotic exposure in patients hospitalised with acute exacerbation of COPD. Int J Chron Obstruct Pulmon Dis 2016; 11: 1381-1389. doi: 10.2147/COPD.S104051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Wang Q, Hou D, Wang J, An K, Han C, Wang C. Procalcitonin-guided antibiotic discontinuation in ventilator-associated pneumonia: a prospective observational study. Infect Drug Resist 2019; 12: 815-824. doi: 10.2147/IDR.S190859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bouadma L, Luyt CE, Tubach F, et al. Use of procalcitonin to reduce patients’ exposure to antibiotics in intensive care units (PRORATA trial): a multicentre randomised controlled trial. Lancet 2010; 375: 463-474. doi: 10.1016/S0140-6736(09)61879-1. [DOI] [PubMed] [Google Scholar]
- 19.Rea-Neto A, Youssef NC, Tuche F, et al. Diagnosis of ventilator-associated pneumonia: a systematic review of the literature. Crit Care 2008; 12: R56. doi: 10.1186/cc6877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Michaud S, Suzuki S, Harbarth S. Effect of design-related bias in studies of diagnostic tests for ventilator-associated pneumonia. Am J Respir Crit Care Med 2002; 166: 1320-1325. doi: 10.1164/rccm.200202-130CP. [DOI] [PubMed] [Google Scholar]
- 21.Zagli G, Cozzolino M, Terreni A, Biagioli T, Caldini AL, Peris A. Diagnosis of ventilator-associated pneumonia: a pilot, exploratory analysis of a new score based on procalcitonin and chest echography. Chest 2014; 146: 1578-1585. doi: 10.1378/chest.13-2922. [DOI] [PubMed] [Google Scholar]
- 22.Kamat IS, Ramachandran V, Eswaran H, Guffey D, Musher DM. Procalcitonin to distinguish viral from bacterial pneumonia: a systematic review and meta-analysis. Clin Infect Dis 2020; 70: 538-542. doi: 10.1093/cid/ciz545. [DOI] [PubMed] [Google Scholar]
- 23.Kalil AC, Metersky ML, Klompas M, et al. Management of adults with hospital-acquired and ventilator-associated pneumonia: 2016 clinical practice guidelines by the Infectious Diseases Society of America and the American Thoracic Society. Clin Infect Dis 2016; 63: e61-111. doi: 10.1093/cid/ciw353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Hamade B, Huang DT. Procalcitonin: where are we Now? Crit Care Clin 2020; 36: 23-40. doi: 10.1016/j.ccc.2019.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Slama TG. Gram-negative antibiotic resistance: there is a price to pay. Crit Care 2008; 12 Suppl 4: S4. doi: 10.1186/cc6820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Grace E, Turner RM. Use of procalcitonin in patients with various degrees of chronic kidney disease including renal replacement therapy. Clin Infect Dis 2014; 59: 1761-1767. doi: 10.1093/cid/ciu732. [DOI] [PubMed] [Google Scholar]
- 27.Food and Drug Administration . Discussion and recommendations for the application of procalcitonin to the evaluation and management of suspected lower respiratory tract infections and sepsis. FDA Executive Summary; 2016. (Accessed: 25.07.2018). [Google Scholar]


