Abstract
Enterococcus faecalis strain WCH9 displays a moderate level of resistance to vancomycin (MIC = 16 μg/ml) and full susceptibility to teicoplanin but is negative by PCR analysis using primers specific for all known enterococcal vancomycin resistance genotypes (vanA, vanB, vanC, vanD, and vanE). We have isolated and sequenced a novel putative vancomycin resistance locus (designated vanG), which contains seven open reading frames, from this strain. These are organized differently from those of all the other enterococcal van loci, and, furthermore, the individual vanG gene products exhibit less than 50% amino acid sequence identity to other van gene products.
Glycopeptides inhibit cell wall synthesis through the interaction of their N termini with the d-alanyl-d-alanine termini of peptidoglycan precursors (25). The resulting complexes prevent the transglycosylases from incorporating disaccharide pentapeptide subunits into the growing peptidoglycan chain, thereby inhibiting cell wall synthesis (4). In enterococci, both intrinsic and acquired resistance to glycopeptides occurs via a common mechanism involving modification of the peptidoglycan biosynthetic pathway (7). Six enterococcal vancomycin resistance genotypes have been described and characterized. Four of these (vanA, vanB, vanD, and vanE) are acquired mechanisms and the other two (vanC1 and vanC2/C3 specific to Enterococcus gallinarum and Enterococcus casseliflavus/Enterococcus flavescens, respectively [the vanC2 and vanC3 genotypes are closely related, with the loci containing minor sequence differences, and are therefore jointly referred to herein as vanC2/C3; likewise, there is debate over whether E. casseliflavus and E. flavescens are actually separate species, and they are jointly referred to herein as E. casseliflavus/E. flavescens]) are intrinsic properties (15, 18, 21).
The genes for the VanA vancomycin resistance phenotype are located on the 10.8-kb transposable element Tn1546, which encodes 7 polypeptides involved in resistance. These polypeptides function together to confer high-level resistance to vancomycin and teicoplanin (6, 7), as well as variable levels of resistance to all other glycopeptides (18). The VanB resistance phenotype confers variable levels of resistance to vancomycin but leaves the organism susceptible to teicoplanin (8, 13). VanD is characterized by resistance to moderate levels of vancomycin and to low levels of teicoplanin (21), while the VanE resistance phenotype exhibits low-level resistance to vancomycin but full susceptibility to teicoplanin (15).
Enterococci of the VanA, VanB, and VanD phenotypes produce a suite of functionally homologous proteins, including ligases (VanA, VanB, and VanD) resulting in the formation of peptidoglycan precursors terminating in d-Ala-d-Lac (18, 21), dehydrogenases (VanH, VanHB, and VanHD) which reduce pyruvate to d-lactate (7, 10, 11), dipeptidases (VanX, VanXB, and VanXD) which hydrolyze the glycopeptide-susceptible cell wall precursors ending in d-Ala-d-Ala (7, 11, 24), and carboxypeptidases (VanY, VanYB, and VanYD) (5, 11, 14) which cleave the C-terminal d-Ala residue of late peptidoglycan precursors (22). VanE ligase synthesizes the formation of peptides terminating in d-Ala-d-Ser, and serine racemase activity has also been detected in VanE strains (15).
VanC1 and VanC2/C3 are intrinsic enterococcal resistance phenotypes characterized by low-level resistance to vancomycin but susceptibility to teicoplanin (7). Three protein products of the vanC1 operon have been characterized: VanC1, VanXYc, and VanT. VanC1 is a ligase that catalyzes the synthesis of cell wall precursors terminating in d-Ala-d-Ser (19). VanXYc has both d,d-dipeptidase and d,d-carboxypeptidase activities (23), while VanT is a membrane-associated serine racemase which converts l-Ser to d-Ser (3). Genes encoding a putative two-component regulator (VanRc-VanSc) are located downstream of the vanC1 locus (2).
Recently, we have investigated the genotype of Australian isolates of vancomycin-resistant enterococci (VRE) (9). Among the strains examined were four Enterococcus faecalis isolates which exhibited moderate resistance to vancomycin (MIC range, 12 to 16 μg/ml), but full susceptibility to teicoplanin (MIC = 0.5 μg/ml), a phenotype similar to that of VanB and VanE strains. The four isolates were indistinguishable by pulsed-field gel electrophoresis and were isolated from inpatients at the same hospital in Brisbane, Queensland, Australia, in 1997. However, these strains were negative by PCR using primers specific for all enterococcal vancomycin resistance genotypes known at the time (vanA, vanB, vanC1, vanC2/C3, and vanD) (9) and have since tested negative by PCR using primers specific for the recently described vanE genotype (15). In the present study, we isolated and characterized the putative vancomycin resistance locus from one of these isolates (WCH9).
Isolation of putative vancomycin resistance locus from E. faecalis WCH9.
In order to isolate a fragment of the vancomycin resistance locus, E. faecalis WCH9 DNA was extracted using the Wizard genomic DNA purification kit (Promega, Madison, Wis.), and subjected to PCR amplification using the degenerate primers V1 and V2 previously described by Perichon et al. (21). These primers were designed to direct amplification of an internal fragment from genes encoding glycopeptide resistance-associated ligases from a number of gram-positive species. The resultant 1.63-kb PCR product was much larger than expected and was purified using the BRESAspin PCR purification kit (Bresatec Ltd., Adelaide, Australia) and cloned into pGEMT-easy (Promega), generating plasmid pJCP1101. The enterococcal DNA insert of this plasmid was then sequenced using dye terminator chemistry on an ABI model 373A automated DNA sequencer. Interestingly, analysis of the sequence revealed that amplification of the 1.63-kb fragment was a consequence of priming with only one of the degenerate primers (V1), presumably due to the presence of an inverted repeat of the target sequence. Nevertheless, BLASTX analysis (1) indicated that the fragment encoded a putative protein with similarities to the enterococcal VanB and VanA ligases (approximately 50 and 48% amino acid sequence identity, respectively), and another with approximately 50% identity to the VanY d,d-carboxypeptidase of the vanA locus.
In order to confirm that the isolated sequences were specific for non-vanABCDE strains such as WCH9, a variety of VRE and sensitive enterococci were subjected to PCR using primers VANG1 (5′-CGGTTGTGCCGTACTTGGC-3′) and VANG2 (5′-GGGTAAAGCCATAGTCTGGGGC-3′). These primers direct amplification of an 810-bp fragment from the portion of the pJCP1101 insert encoding the ligase homologue. The PCR amplification conditions used were the same as those previously described by Bell et al. (9) for detection of the vanA, vanB, vanC1, and vanC2/C3 genotypes. The only strains that yielded 810-bp PCR products were WCH9 and the three other related non-vanABCDE VRE isolates. No PCR products were obtained when DNA extracts from vancomycin-sensitive Enterococcus faecium, E. faecalis vanA, E. faecalis vanB, E. gallinarum vanC1, or vanC2/C3 E. casseliflavus/E. flavescens strains were tested (result not shown).
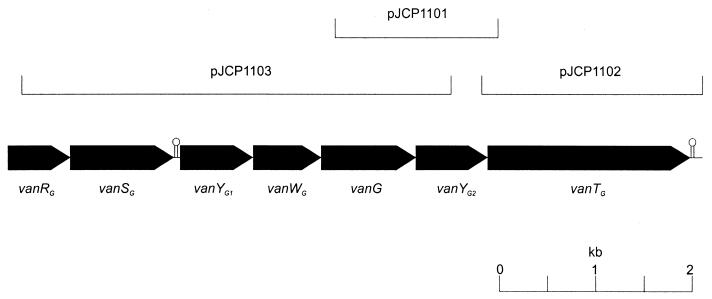
In order to isolate the remainder of the apparently novel van locus, DNA from WCH9 was subjected to PCR using primers on the basis of preliminary sequence analysis of inverse PCR fragments. Primers SM5 (5′-GTTGACGAATTCATATTCCCAGATAAA-3′) and SM6 (5′-CGAAAGGAATTCTGGCATTGATTATTTTAG-3′) directed amplification of a 1,999-bp PCR product, which was cloned into the EcoRI site of pBluescript KS (Stratagene, La Jolla, Calif.) to generate plasmid pJCP1102. Plasmid pJCP1103 was constructed by cloning the 5,004-bp PCR product obtained using primers VANG2 and SM7 (5′-CGGGACAAATAATTGGATGTC-3′) into pGEMT-easy (see Fig. 1).
FIG. 1.
Map of region of E. faecalis WCH9 chromosome containing the vanG locus, showing the location of the various subclones used to determine the DNA sequence. The locations and direction of transcription of the various ORFs are shown as solid pointed boxes. The locations of two putative transcription terminators are also indicated.
DNA sequence analysis.
The sequence of the complete putative vancomycin resistance locus was then determined by analysis of the recombinant plasmids, and nested deletion derivatives thereof, constructed according to the method of Henikoff (16). Sequence data in the 5′ direction of the region cloned in pJCP1103 were also obtained by direct sequence analysis of an additional inverse PCR product using primers SM30 (5′-CGATTGGAAAATCTGATCAATG-3′) and SM31 (5′-CGTAACCTCCCGATATGAGCC-3′) on NdeI-digested, recircularized DNA. Although existing enterococcal vancomycin resistance loci have been named vanA through vanE, we designated this novel locus vanG, because the name vanF was recently used for a vanA-like locus in Paenibacillus popillae (20). Examination of the compiled sequence indicated that the vanG locus was 7,652 nucleotides long and contained seven complete open reading frames (ORFs), arranged as shown in Fig. 1. BLASTX analyses were performed to identify the putative functions of the proteins, and the results are summarized in Table 1. The 5′ end of the locus contains genes designated vanRG and vanSG, encoding a putative two-component regulatory system. The vanSG gene was immediately followed by a stem-loop structure (ΔG = −28.5 kcal/mol), suggesting that these two genes may be transcribed separately from the remainder of the locus, which encodes two putative d,d-peptidases (VanYG1 and VanYG2), the ligase (VanG), a racemase (VanTG), and a protein with unknown function (VanWG). Interestingly, no obvious promoter region could be found upstream of vanYG1, and the nearest site with similarity to consensus Escherichia coli promoter sequences is located near the 3′ end of vanWG. Thus, the ORFs designated vanYG1 and vanWG may or may not be transcribed. Each of the genes in the vanG cluster is preceded by a potential ribosome binding site, although that for vanYG2 is very close to the initiation codon, which may result in a low translation efficiency. An additional stem-loop structure (ΔG = −23.0 kcal/mol) is located approximately 20 nucleotides downstream of the vanTG termination codon. BLASTX analysis of sequences downstream of this indicated a low degree of similarity to a protein of unknown function (ORF9) from the 3′ end of the E. faecalis conjugative transposon Tn916 (GenBank accession number U09422).
TABLE 1.
Summary of ORFs in the vanG locus
| Gene | Putative function | Location in sequencea | Predicted Mr | No. of amino acids | % G+C content | Similar protein(s) (GenBank accession no.; gene) | % Identity, % similarity (no. of aa) |
|---|---|---|---|---|---|---|---|
| vanRG | Response regulator | 1 to 706 | 26,858 | 235 | 42.3 | VanRD of E. faecium (AF130997; vanD) | 54, 71 (238) |
| VanR of Bacillus circulans (Y15706) | 46, 63 (239) | ||||||
| VanR, E. faecium (M68910; vanA) | 46, 61 (238) | ||||||
| vanSG | Sensor protein kinase | 722 to 1825 | 42,027 | 367 | 27.7 | VanSD of E. faecium (AF130997; vanD) | 57, 69 (330) |
| VanS of B. circulans (Y15707) | 39, 56 (364) | ||||||
| VanS of E. faecium (M68910; vanA) | 38, 57 (365) | ||||||
| vanYG1 | d,d-Peptidase | 1962 to 2798 | 31,926 | 278 | 42.9 | VanYB of E. faecalis (U35369; vanB) | 48, 63 (211) |
| Carboxypeptidase of Bacillus subtilis (AF015775) | 31, 47 (246) | ||||||
| YokZ of B. subtilis (AF006665) | 31, 47 (246) | ||||||
| vanWG | Unknown | 2979 to 3713 | 28,301 | 244 | 35.8 | VanW of E. faecalis (U35369; vanB) | 45, 60 (244) |
| vanG | Ligase | 3715 to 4764 | 38,727 | 349 | 39.0 | VanB of E. faecalis (U00456) | 46, 64 (215) |
| VanA of B. circulans (Y15704) | 49, 66 (202) | ||||||
| VanB2 of E. faecium (U81452) | 47, 64 (212) | ||||||
| vanYG2 | d,d-Peptidase | 4761 to 5525 | 29,362 | 253 | 37.3 | VayY of B. circulans (Y17303) | 35, 54 (262) |
| VanY of E. faecium (M90647; vanA) | 34, 53 (256) | ||||||
| YokZ of B. subtilis (AF006665) | 34, 51 (191) | ||||||
| vanTG | Racemase | 5518 to 7653 | 80,159 | 711 | 36.1 | VanT of E. gallinarum (AF077816; vanC1) | 35, 55 (698) |
| Alanine racemase of Bacillus psychrosaccharolyticus (AB021683) | 36, 53 (383) | ||||||
| Alanine racemase of Synechocystis sp. (D64003) | 35, 53 (362) |
Nucleotide positions are given.
Similarities to other enterococcal van loci and gene products.
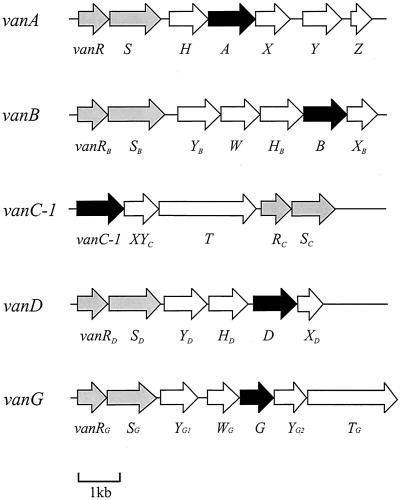
The comparative genetic organization for the five VRE van loci that have been fully sequenced to date is shown in Fig. 2. Although except in the case of vanC1 response regulator (R) and sensor protein kinase (S) genes are located at the 5′ end of the loci, the organization of subsequent genes differs widely between van types, suggesting diverse origins. Marked differences also occur at the deduced amino acid sequence level. The alignment of the putative ligase VanG with the respective ligases VanA, VanB, VanC1, and VanD is shown in Fig. 3, and the percent amino acid identity between each ligase is shown in Table 2. This shows that the degree of similarity between VanG and the other ligases (39 to 47%) is no greater than between any of the other proteins (values observed range from 38% identity between VanC1 and VanD to 76% identity between VanA and VanB). Thus, VanG is clearly not a minor variant of one of the other enterococcal proteins and represents a distinct class of ligases.
FIG. 2.
Genetic organization of the vanG locus, compared with that of other enterococcal vancomycin resistance loci, based on published sequence data for the vanA, vanB, vanC1, and vanD loci (2, 6, 7, 11). ORFs encoding putative ligases and two-component regulatory systems are shown in black and grey, respectively.
FIG. 3.
Comparison of VanG with other enterococcal ligases. The deduced amino acid sequence of VanG was aligned with those for VanA (6), VanB (14), VanC1 (12), and VanD (11) using CLUSTAL (17). Amino acid position numbers are shown at the left of the sequences. Identical residues are shaded black; residues with similar properties are shaded grey. Gaps (dashes) have been introduced by the program to optimize the alignment.
TABLE 2.
Sequence identity between the deduced amino acid sequence of VanG and those of other enterococcal ligases
| Sequence compareda | % Sequence identity (no. of overlapping amino acids)
|
||||
|---|---|---|---|---|---|
| VanG | VanA | VanB | VanC | VanD | |
| VanA | 44 (351) | ||||
| VanB | 47 (354) | 76 (341) | |||
| VanC | 42 (347) | 38 (349) | 39 (350) | ||
| VanD | 46 (347) | 69 (342) | 67 (343) | 38 (350) | |
| VanE | 39 (201) | 44 (206) | 42 (206) | 54 (202) | 44 (206) |
The putative regulatory proteins VanRG and VanSG had greatest identity with VanRD and VanSD (54 and 57%, respectively). In addition, VanRG also has 46% identity with VanR (over 236 amino acids [aa]) and 28% identity with VanRB (over 169 aa), while VanSG has 42% identity with VanS (over 365 aa) and 20% identity with VanSB (over 274 aa). VanRG has similar degrees of amino acid identity between both the N- and C-terminal portions, which are the phosphorylation site and DNA-binding regions, respectively. VanSG has a percent amino acid identity in the C-terminal two-thirds, which include the histidine kinase activity motif, greater than that in the N-terminal third, which is involved in ligand binding.
The putative peptidase VanYG1, which may or may not be transcribed, has a deduced amino acid sequence with 46% identity with that of VanYB (over 214 aa) and 27% identity with the putative protein VanYG2 (over 177 aa). In addition, the putative peptidase VanYG2 has 32% identity (over 247 aa) and 33% identity (over 184 aa) with the VanY and VanYB proteins, respectively. VanYG2 is approximately 49 and 14 amino acids shorter than its VanY and VanYB counterparts, respectively. The vanG locus lacks a putative VanX dipeptidase, indicating that the putative VanYG proteins may be similar to VanXYc of the vanC1 locus and display both carboxypeptidase and dipeptidase activities.
Attempted transfer of vancomycin resistance.
Transfer of vancomycin resistance from E. faecalis WCH9 to E. faecium ATCC 19434 was attempted by filter mating (26), with selection on phenol red agar containing phenol red broth base (1.6% [wt/vol]), Bacto agar (1% [wt/vol]), l-arabinose (1% [wt/vol]), and vancomycin (5 μg/ml). However, although transmission of vancomycin resistance occurred when vanB E. faecalis ATCC 51299 was mated with the sensitive Enterococcus faecium recipient, transmission was not demonstrated when the vanG strain was used as donor. Fines et al. (15) were similarly unable to demonstrate transmission of the VanE phenotype. Transfer of vancomycin resistance from E. faecalis WCH9 to another E. faecalis strain was not attempted due to the lack of a selective marker capable of distinguishing donor and recipient strains.
Conclusions.
In this study, we have isolated and characterized a novel vancomycin resistance locus, designated vanG, from E. faecalis strain WCH9, which locus displays a moderate level of resistance to vancomycin and full susceptibility to teicoplanin. The vanG locus contains seven ORFs, but these are organized differently from all the other enterococcal van loci characterized to date. Moreover, the individual vanG gene products exhibit less than 50% amino acid sequence identity to their respective homologues from the other enterococcal van loci, suggesting diverse evolutionary origins of vancomycin resistance in this genus. VanG VRE appear to be rare, at least in Australia. The four original strains were isolated from patients in a single institution over a relatively short period of time, consistent with nosocomial spread. With the exception of reisolation from one of the four original VanG VRE-carrying patients 12 months later, no further strains belonging to this genotype have been detected. However, its prevalence in other parts of the world remains to be investigated.
Nucleotide sequence accession number.
The DNA sequence of the vanG locus described here has been deposited with GenBank under accession number AF253562.
Acknowledgments
We are grateful to Graeme Nimmo for referring VRE isolates for characterization.
REFERENCES
- 1.Altschul S F, Madden T L, Schäffer A A, Zhang J, Zhang Z, Miller W, Lipman D J. Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res. 1997;25:3389–3402. doi: 10.1093/nar/25.17.3389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Arias C A, Courvalin P, Reynolds P E. vanC cluster of vancomycin-resistant Enterococcus gallinarum BM4174. Antimicrob Agents Chemother. 2000;44:1660–1666. doi: 10.1128/aac.44.6.1660-1666.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Arias C, Martin-Martinez M, Blundell T, Arthur M, Courvalin P, Reynolds P. Characterization and modelling of VanT: a novel, membrane-bound, serine racemase from vancomycin-resistant Enterococcus gallinarum BM4174. Mol Microbiol. 1999;31:1653–1664. doi: 10.1046/j.1365-2958.1999.01294.x. [DOI] [PubMed] [Google Scholar]
- 4.Arthur M, Courvalin P. Genetics and mechanisms of glycopeptide resistance in enterococci. Antimicrob Agents Chemother. 1993;37:1563–1571. doi: 10.1128/aac.37.8.1563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Arthur M, Depardieu F, Snaith H A, Reynolds P E, Courvalin P. Contribution of VanY d,d-carboxypeptidase to glycopeptide resistance in Enterococcus faecalis by hydrolysis of peptidoglycan precursors. Antimicrob Agents Chemother. 1994;38:1899–1903. doi: 10.1128/aac.38.9.1899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Arthur M, Molinas C, Depardieu F, Courvalin P. Characterization of Tn1546, a Tn3-related transposon conferring glycopeptide resistance by synthesis of depsipeptide peptidoglycan precursors in Enterococcus faecium BM4147. J Bacteriol. 1993;175:117–127. doi: 10.1128/jb.175.1.117-127.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Arthur M, Reynolds P E, Courvalin P. Glycopeptide resistance in enterococci. Trends Microbiol. 1996;4:401–407. doi: 10.1016/0966-842X(96)10063-9. [DOI] [PubMed] [Google Scholar]
- 8.Baptista M, Depardieu F, Courvalin P, Arthur M. Specificity of induction of glycopeptide resistance genes in Enterococcus faecalis. Antimicrob Agents Chemother. 1996;40:2291–2295. doi: 10.1128/aac.40.10.2291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bell J M, Paton J C, Turnidge J. Emergence of vancomycin-resistant enterococci in Australia: phenotypic and genotypic characteristics. J Clin Microbiol. 1998;36:2187–2190. doi: 10.1128/jcm.36.8.2187-2190.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Bugg T D, Wright G T, Dutka-Malen S, Arthur M, Courvalin P, Walsh C T. Molecular basis for vancomycin resistance in Enterococcus faecium BM4147: biosynthesis of a depsipeptide peptidoglycan precursor by vancomycin resistance proteins VanH and VanA. Biochemistry. 1991;30:10408–10415. doi: 10.1021/bi00107a007. [DOI] [PubMed] [Google Scholar]
- 11.Casadewall B, Courvalin P. Characterization of the vanD glycopeptide resistance gene cluster from Enterococcus faecium BM4339. J Bacteriol. 1999;181:3644–3648. doi: 10.1128/jb.181.12.3644-3648.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Dutka-Malen S, Molinas C, Arthur M, Courvalin P. Sequence of the vanC gene of Enterococcus gallinarum BM4174 encoding a d-alanine-d-alanine ligase-related protein necessary for vancomycin resistance. Gene. 1992;112:53–58. doi: 10.1016/0378-1119(92)90302-6. [DOI] [PubMed] [Google Scholar]
- 13.Evers S, Courvalin P. Regulation of VanB-Type vancomycin resistance gene expression by the VanSB-VanRB two-component regulatory system in Enterococcus faecalis V583. J Bacteriol. 1996;178:1302–1309. doi: 10.1128/jb.178.5.1302-1309.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Evers S, Reynolds P E, Courvalin P. Sequence of the vanB and ddl genes encoding D-alanine:D-lactate and D-alanine:D-alanine ligases in vancomycin-resistant Enterococcus faecalis V583. Gene. 1994;140:97–102. doi: 10.1016/0378-1119(94)90737-4. [DOI] [PubMed] [Google Scholar]
- 15.Fines M, Perichon B, Reynolds P, Sahm D F, Courvalin P. VanE, a new type of acquired glycopeptide resistance in Enterococcus faecalis BM4405. Antimicrob Agents Chemother. 1999;43:2161–2164. doi: 10.1128/aac.43.9.2161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Henikoff S. Unidirectional digestion with exonuclease III creates targeted breakpoints for DNA sequencing. Gene. 1984;28:351–359. doi: 10.1016/0378-1119(84)90153-7. [DOI] [PubMed] [Google Scholar]
- 17.Higgins D G, Sharp P M. CLUSTAL: a package for performing multiple sequence alignments on a microcomputer. Gene. 1988;73:237–244. doi: 10.1016/0378-1119(88)90330-7. [DOI] [PubMed] [Google Scholar]
- 18.Leclercq R, Courvalin P. Resistance to glycopeptides in enterococci. Clin Infect Dis. 1997;24:545–556. doi: 10.1093/clind/24.4.545. [DOI] [PubMed] [Google Scholar]
- 19.Navarro F, Courvalin P. Analysis of genes encoding d-alanine-d-alanine ligase-related enzymes in Enterococcus casseliflavus and Enterococcus flavescens. Antimicrob Agents Chemother. 1994;38:1788–1793. doi: 10.1128/aac.38.8.1788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Patel R, Piper K, Cockerill III F R, Steckelberg J M, Yousten A A. The biopesticide Paenibacillus popillae has a vancomycin resistance gene cluster homologous to the enterococcal VanA vancomycin resistance gene cluster. Antomicrob Agents Chemother. 2000;44:715–709. doi: 10.1128/aac.44.3.705-709.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Perichon B, Reynolds P, Courvalin P. VanD-type glycopeptide-resistant Enterococcus faecium BM4339. Antimicrob Agents Chemother. 1997;41:2016–2018. doi: 10.1128/aac.41.9.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Reynolds P E. Control of peptidoglycan synthesis in vancomycin-resistant enterococci: D,D-peptidases and D,D-carboxypeptidases. Cell Mol Life Sci. 1998;54:325–331. doi: 10.1007/s000180050159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Reynolds P, Arias C, Courvalin P. Gene vanXYc encodes D,D-dipeptidase (VanX) and D,D-carboxypeptidase (VanY) activities in vancomycin-resistant Enterococcus gallinarum BM4174. Mol Microbiol. 1999;34:341–349. doi: 10.1046/j.1365-2958.1999.01604.x. [DOI] [PubMed] [Google Scholar]
- 24.Reynolds P E, Depardieu F, Dutka-Malen S, Arthur M, Courvalin P. Glycopeptide resistance mediated by enterococcal transposon Tn1546 requires production of VanX for hydrolysis of D-alanyl-D-alanine. Mol Microbiol. 1994;13:1065–1070. doi: 10.1111/j.1365-2958.1994.tb00497.x. [DOI] [PubMed] [Google Scholar]
- 25.Sheldrick G M, Jones P G, Kennard O, Williams D H, Smith G A. Structure of vancomycin and its complex with acetyl-D-alanyl-D-alanine. Nature. 1978;271:223–225. doi: 10.1038/271223a0. [DOI] [PubMed] [Google Scholar]
- 26.Trieu-Cuot P, Carlier C, Courvalin P. Conjugative plasmid transfer from Enterococcus faecalis to Escherichia coli. J Bacteriol. 1988;170:4388–4391. doi: 10.1128/jb.170.9.4388-4391.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]