Introduction
Familial hypercholesterolemia (FH) is an autosomal hereditary disorder characterized by hyper-LDL cholesterolemia (LDL-C), premature coronary artery disease (CAD), and tendon and skin xanthomas. FH is at extremely high-risk of developing CAD and thus requires early diagnosis, rigorous treatment, and family screening (cascade screening). The purpose of these guidelines is to prevent possible future atherosclerosis by providing diagnosis and treatment of FH in real clinical practice.
The criteria for Achilles tendon thickening have changed. The conventional radiographic cutoff value of 9 mm was published in 1977, and while it had high specificity, it was feared to have low sensitivity. In this present study, based on the analysis of 986 cases, including 485 cases of genetically diagnosed FH in Japan, the cutoff values were reviewed according to the report 1) in 2021, which set the cutoff value at 7.6 mm for men and 7.0 mm for females and were changed to 8.0 mm or more for males and 7.5 mm or more for females to increase sensitivity while maintaining specificity. The evaluation by ultrasound has also been standardized and adopted in this guideline. However, further considerations of the validity of the changed reference values and the evaluation of sensitivity and specificity will be necessary in the future.
We omit the treatment of pediatric FH in this adult version and please refer to the newly developed “Guidelines for the Diagnosis and Treatment of Pediatric Familial Hypercholesterolemia 2022” 2) as well.
1. Background Question (BQ)/ Foreground Question (FQ)
BQ1. What is the Prevalence of FH in Japan?
In general, it is found in approximately 1 in 300 people in the general population, 1 in 30 people with CAD, and 1 in 15 people with premature CAD or severe hyperlipidemia. (Level of evidence: E-2)
Traditionally, the prevalence of FH was 1 in 500 people, but in recent years, a series of cross-sectional and cohort studies in the United States and Europe have shown a clearly higher prevalence than that. In Japan, Mabuchi et al. examined the frequency of molecular epidemiology in the Hokuriku region and reported a prevalence rate of 1 in 208 people 3) .
A systematic review/meta-analysis published in 2017 (not including Japanese) reported a frequency of 1 in 250 people in the general population 4) . In addition, a subsequent systematic review/meta-analysis (including Japanese) reported a frequency of 1 in 313 5) and 311 6) people, respectively, in the general population. The meta-analysis that reported a prevalence of 1 in 250 people included literature reporting extremely high frequencies due to the so-called founder effect, whereas the latter two reports of 1 in 313 and 311 people excluded such studies. As increased frequency due to founder effect is presumed to exist in some regions, it is reasonable to assume that the number of cases is about 1 in 300 people, despite the report by Mabuchi et al. in Japan. The number of Japanese in the above meta-analysis is small, and it is assumed that differences in frequency may be due to factors such as consanguineous marriages in some regions and demographic bottleneck effects, but there is no evidence of significant differences among races; thus, the results of the above meta-analysis may be applied to Japanese as well.
BQ2. What are the Prognosis and Main Complications of Patients with FH?
• CAD: odds ratio 10–20 times higher than non-FH (Level of evidence: E-1a*)
• Peripheral arterial disease: odds ratio 5–10 times higher than non-FH (Level of evidence: E-1a)
• Stroke: no clear impact (Level of evidence: E-1a)
• Aortic valve stenosis: no epidemiological association was shown, but there have been case reports of FH complicating the disease. (Level of evidence: E-3)
• Abdominal aortic aneurysm: no epidemiological association was shown, but there have been case reports of FH complicating the disease. (Level of evidence: E-3)
*Although there was no meta-analysis of cohort studies, we chose E-1a because of the existence of multiple cohort studies and identical results.
The most important major complication of systemic atherosclerosis in patients with FH is CAD 7) . In addition, a significantly higher prevalence of peripheral arterial disease and carotid atherosclerosis was reported in FH as compared to non-FH (systematic review/meta-analysis) 8 , 9) . On the other hand, with regard to stroke, many reports state that its effects remain unclear. With regard to aortic disease and valvular disease (aortic aneurysm, aortic stenosis, supravalvular stenosis, etc.), there are case reports showing their association with the disease, although no epidemiological reports have demonstrated such an association.
Although there are no randomized controlled trials (RCTs) or systematic reviews on the prognosis of FH, Mabuchi et al. presented a study comparing the prognosis of FH heterozygotes (HeFH) and FH homozygotes (HoFH) in the pre- and post-statin era in Japan 10) . This report shows that before the advent of statins, 73% of men and 64% of women with HeFH died of cardiac death, the age of death for HeFH increased from an average of 63 years before statins to 76 years after statins, and for HoFH, the average age of death increased from 28 years before statins to 59 years after statins.
FQ1. Can Statins be Recommended as the First Choice in Drug Therapy for HeFH?
Strict lipid management with statins as first-line drugs is recommended for the treatment of HeFH. (Level of evidence: 3; Recommendation level: A)
There are 13 randomized double-blind trials examining the LDL-C lowering effect of statins in HeFH 11 - 23) , including 9 placebo-controlled trials (2 in adult HeFH 11 , 12) , 6 in children to adolescents HeFH 14 - 19) , and 1 in adult FH subjects without mention of whether they are HeFH or HoFH 13) ), and its efficacy and safety have been established in both adult and pediatric cases. Furthermore, a randomized, double-blind, crossover study in HoFH has shown the efficacy 20) of statin. In Japan, reports have been published showing that statin therapy reduces LDL-C in both adult and pediatric patients 22 , 23) , which is consistent with the results of clinical trials in other countries.
A randomized, double-blind, comparative study has been published in adult 24) and pediatric patients 19) , respectively, to investigate the efficacy of high-intensity statin therapy (80 mg of atorvastatin, twice the approved dose in Japan) compared to standard-intensity statin therapy (40 mg simvastatin, twice the approved dose in Japan) in adults and standard-intensity statin therapy (20–40 mg pravastatin, no pediatric indication in Japan at the approved dose of 20 mg) compared to placebo in children. Both studies showed inhibition of the development of Intima–media thickness (IMT). A subanalysis of pediatric cases showed that statin initiation at an earlier age was associated with less IMT thickening 25) . In addition, an observational study examining 2447 cases from the Netherlands has shown that statin use is associated with a lower incidence of CAD and lower all-cause mortality 26) . Although scientific evidence for the prevention of atherosclerotic cardiovascular disease by direct comparison is not sufficient, statins appear to be the most recommended drug therapy at this time, given the abundant evidence in non-FH.
Four clinical studies have been published comparing the LDL-C lowering effects of statins and other lipid-lowering drugs in FH (all in adults, all with pravastatin, two in HeFH, and two in FH subjects). In a report comparing pravastatin 40 mg with cholestyramine 4 g (or colestipol 5 g), both groups had significantly lower LDL-C from baseline, but the group of pravastatin had significantly lower LDL-C than the cholestyramine group 12) . On the other hand, reports comparing 40 mg of pravastatin with 16–24 g of cholestyramine (1.5–2 times the Japanese approved dose) did not show any significant difference between the two groups, although both groups had significantly lower LDL-C from baseline 11 , 13 , 27) . These studies were conducted with pravastatin in the early 1990s, and since the newer generation of statins shows stronger lipid-lowering effects, it is expected to be more effective than cholestyramine. Although there are no direct comparative studies between statins and probucol, small intestinal cholesterol transporter inhibitors (ezetimibe), or proprotein convertase subtilisin/kexin type 9 inhibitors (PCSK9), there are reports of trials of ezetimibe and PCSK9 inhibitors on top of statins 28 - 31) .
FQ2. Is Lipoprotein Apheresis Therapy Recommended for HoFH and Severe HeFH with Drug Resistance?
For HoFH and severe HeFH with drug resistance, strict control of LDL-C with lipoprotein apheresis therapy is recommended. (Level of evidence: 3; Recommendation level: A)
In the 2019 ESC/EAS Guidelines for the Management of Dyslipidaemias: Lipid Modification to Reduce Cardiovascular Risk 32) , although there is no mention of evidence levels, etc., the lipoprotein apheresis has been recommended as a treatment for HoFH.
A systematic review reported in 2016 analyzed a total of 38 articles (8 open-label clinical trials, 11 observational studies, 17 reviews/guidelines, and 2 medical technology evaluations) 33) . Although RCTs were not included, they were noted to have clinical benefits in terms of lowering LDL-C, lowering lipoprotein (a) [Lp (a)], and preventing cardiovascular events. As each country has different rates of diagnosis of FH, availability and access to apheresis treatment, indications, methods, and costs, it will be necessary to evaluate the situation in Japan in the future. However, although this puts physical and social burden on the patient, we believe that lipoprotein apheresis is recommended for patients with FH who do not respond adequately to drug therapy in Japan, where access is relatively easy in terms of transportation and cost.
In 2019, a systematic review examining 76 case reports (209 patients) on HoFH in children was reported 34) . Although it has not been shown whether lipoprotein apheresis is more or less beneficial than drug therapy alone for cardiovascular outcomes, it has been reported to lower LDL-C and reduce xanthomas with fewer adverse events, making it generally safe.
2. Overall Outline
FH is an autosomal hereditary disease characterized by (1) hyper-LDL cholesterolemia (LDL-C), (2) premature CAD, and (3) tendon and skin xanthomas. Except for the rare autosomal recessive hypercholesterolemia (autosomal recessive hypercholesterolemia: ARH), all other cases have a dominant mode of inheritance.
Patients with FH have persistent hyper-LDL-C from the time of birth, and coronary atherosclerosis is known to develop at a young age. Therefore, the condition of FH alone is identified as an extremely high-risk disease for CAD, and CAD such as myocardial infarction and angina pectoris often develop between the ages of 30–50 in untreated men and 50–70 in women 35) . Untreated HeFH have been reported to have an approximately 13-fold higher risk of developing CAD 36) , but hyper-LDL-C itself is asymptomatic. Therefore, when examining a patient with hyper-LDL-C, FH should always be kept in mind, early diagnosis and appropriate treatment should be made, and family screening (cascade screening) should be performed to prevent death at a young age.
In Japan, as in other countries, HeFH exists with a frequency of 1 in 300 people, and it is estimated that the country has more than 300,000 patients 10) . Although there are no accurate reports on the diagnosis rate of FH in Japan, it is expected to be low; thus, improving the diagnosis rate of FH is an urgent issue.
Additionally, by diagnosing a single FH patient, it is possible to detect FH patients in their families and treat them at an early stage. Healthcare providers treating patients with FH should be well aware that the disease is primarily an autosomal dominant genetic disorder and should be included in the diagnosis and treatment of their families.
2.1 Causative Gene for FH
Although genetic testing is not required for diagnosis, the presence of pathogenic mutations in genes involved in LDL metabolism, such as the LDL receptor (LDLR), in addition to hyper-LDL-C, is considered a definite diagnosis. When the genetic test for the proband is performed, the chance of diagnosis for the relatives will then be increased.
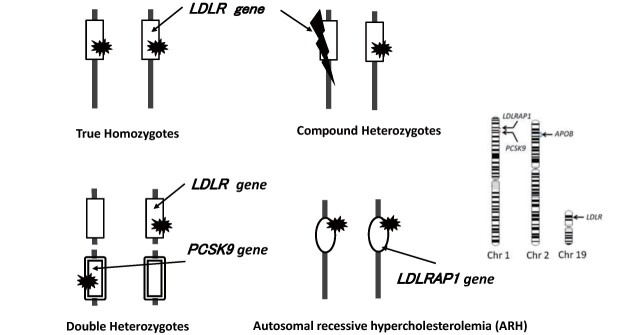
In addition to pathogenic genetic mutations in the LDLR, HeFH is known to be caused by pathogenic genetic mutations in apolipoprotein B-100 (APOB) and pathogenic gain-of-function mutations in the proprotein convertase subtilisin/kexin type 9 (PCSK9) (gain-of-function mutation), both of which play an important role in LDL metabolism. Mutations in the causative gene are identified in 60%–80% of clinically diagnosed HeFH 3 , 37) . HoFH are defined as having abnormalities of the LDLR, APOB, or gain-of-function PCSK9 in both alleles ( Fig.1 ) . ARH is an autosomal recessive inherited disorder caused by biallelic pathogenic mutations in the low-density lipoprotein receptor adaptor protein 1 (LDLRAP1). This situation is considered as HoFH.
Fig.1.
Combination of genetic mutation showing HoFH clinically
In recent years, in addition to FH associated with rare pathogenic genetic mutations of the LDLR and related proteins, as described above, polygenic FH associated with the accumulation of high-frequency genetic polymorphisms at loci involved in LDL metabolism has been suggested to exist 38) . However, there is no evidence that the superposition of high-frequency gene polymorphisms alone, in any known combination, causes the so-called FH. These polymorphisms can affect the phenotype of FH (and other hyper-LDL-C) rather than being involved in the development of FH.
2.1.1 LDL Receptor (LDLR)
The majority of FH (more than 60%) have been attributed to genetic mutations of the LDLR. Many pathogenic gene mutations, numbered at more than 1,000 worldwide, have been reported as causes of FH (http://www.hgmd.cf.ac.uk/ac/gene.php?gene=LDLR). More than 100 mutations have been reported in our country alone 39) .
2.1.2 Apolipoprotein B-100 (APOB)
Genetic mutations in APOB, the ligand for the LDLR, can also cause manifestations of FH and are called familial defective apolipoprotein B-100 (FDB). It is noted to be more frequent in Caucasians in Europe and the United States, but less frequent in other ethnic groups. Serum LDL-C levels are generally lower than those of LDLR mutations. It has also been reported in Japanese 40) . ApoB is a large protein with a molecular weight of 500 kDa that consists of 4,563 amino acids. Therefore, although many gene mutations are usually identified, especially in comprehensive gene analysis, pathogenic gene mutations that cause FH are almost limited to Arg3527Gln mutation (Arg3527Trp is also considered a pathogenic mutation) 41) .
2.1.3 Proprotein Convertase Subtilisin/Kexin Type 9 (PCSK9)
PCSK9 is known to be involved in the degradation of the LDLR, and gain-of-function mutations are known to deplete the LDLR, resulting in hyper-LDL-C. Severe cases of patients homozygous for PCSK9 gain-of-function mutations are clinically indistinguishable from those homozygous for LDLR mutations 42 , 43) . In Japan, the E32K mutation, which is a mild gain-of-function mutation and causes relatively mild elevations in LDL-C, is frequently found in 1%–2% of the general population and 6% of the clinically diagnosed FH patients. When the E32K mutation coexists with another mutation in LDLR, the phenotype resembles that of HoFH, but the response to drug therapy is better than in HoFH for the LDLR 44) . Similarly, V4I mutations are present in 6% of FH patients, and when V4I mutations are associated with LDLR mutations, the phenotype is similar to that of HoFH 37) . The presence of LDLR and PCSK9 mutations in their respective alleles is not genetically homozygous but double heterozygous. Whether PCSK9 mutations cause FH should be determined in a specialized facility.
2.1.4 LDL Receptor Adapter Protein 1 (LDLRAP1)
It is known to be involved in LDLR endocytosis and develops into ARH when the genetic mutation is inherited from both parents. ARH is considered a very rare disease that should be suspected when there is no hyper-LDL-C in the parents, although HoFH is suspected based on massive xanthoma formation and hyper-LDL-C 45 , 46) . This disease should be treated as HoFH.
2.2 Epidemiology of FH
For a long time, the frequency of HeFH was considered to be about 1 in 500 people. However, Mabuchi et al. reported that the frequency of HeFH, calculated by the frequency of HoFH, is estimated to be 1 in 208 people in the general population, which is a higher frequency than before, based on the analysis of LDLR mutations and PCSK9 gain-of-function mutations in the Hokuriku region 3) . Recent meta-analyses of molecular epidemiological studies 5 , 6) suggest that the frequency of FH in the general population worldwide is approximately 1 in 300 people. These meta-analyses of molecular epidemiological studies include data from Japan, with a total sample size of more than several million people and thus are extremely important as data on the frequency of this disease. Furthermore, the frequency of FH in patients with CAD is estimated to be approximately 1 in 30 people, and in patients with premature CAD, it is approximately 1 in 15 people. Therefore, FH should not be considered a rare disease, as it has been reported to account for approximately 8.5% of patients with hyper-LDL-C who undergo treatment 47) .
2.3 Clinical Features of FH
FH is mainly an autosomal dominant genetic disorder characterized by three main features, namely, hyper-LDL-C, premature CAD, and tendon and skin xanthomas.
2.3.1 Hyper-LDL Cholesterolemia
In a survey report conducted in 1996–1997 by the “Research on Primary Hyperlipidemia” group of the Ministry of Health, Labor and Welfare Research on Intractable Diseases, Bujo et al. found that the mean untreated LDL-C of 641 Japanese HeFH (296 males and 345 females, mean age 51 years) was 248 mg/dL, with no difference between men and women 48) . Serum total cholesterol levels in patients with FH and their families are found to be trimodal, with 179±26 in normal subjects, 338±63 in HeFH, and 713±122 in HoFH (mean±SD (mg/dL) for each), almost twice as high as normal subjects in HeFH and four times as high in HoFH 49) . However, there seems to be an overlap between normal subjects and HeFH and between HeFH and HoFH, which may be difficult to distinguish based solely on serum lipid levels ( Fig.2 ) .
Fig.2.
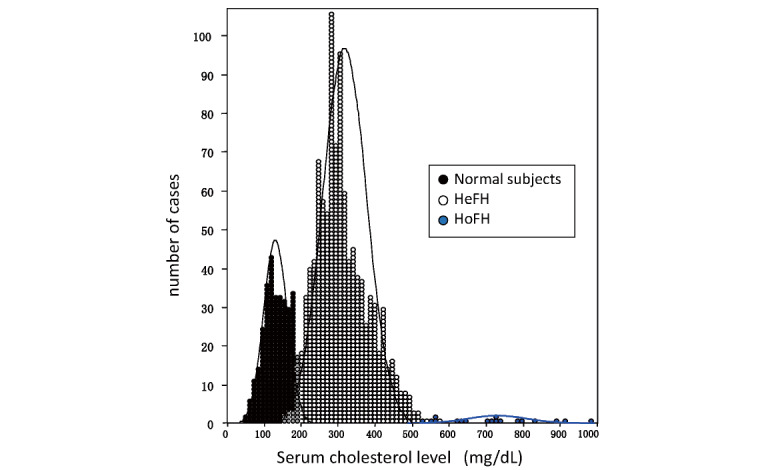
Total cholesterol levels in normal, heterozygous, and homozygous individuals in a family with familial hypercholesterolemia (Modified from Ref. 47 by adding cases)
FH should also be actively suspected when high LDL-C is observed from a young age or when the LDL-C lowering response to drug therapy, such as statins, is noted poor 50) .
2.3.2 Premature CAD
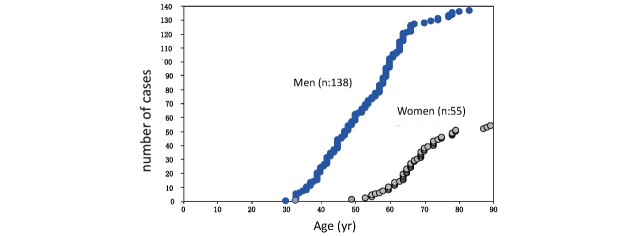
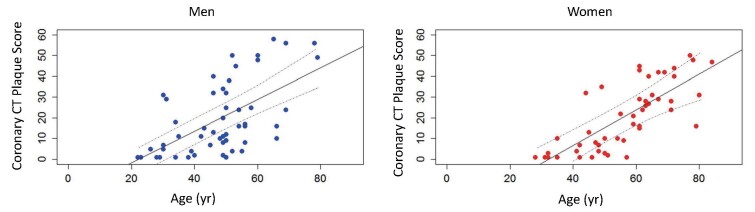
FH should be suspected in patients with premature CAD and high LDL-C levels. Premature CAD is defined as CAD that develops at an age of <55 years in men and <65 years in women. Hyper-LDL-C is the most common cause of atherosclerosis, especially progressive coronary atherosclerosis, and untreated HeFH has a high cardiac death rate of around 60% 10) . Hyper-LDL-C and cerebrovascular disease are associated, but not as common as in HeFH (5%–10%) 49 , 51) . In men, myocardial infarction increases at an almost constant rate after age of 30, while in women, it is rare before the age of 50, indicating a clear gender difference ( Fig.3 ) . Furthermore, the formation of coronary plaque on coronary CT scans has been confirmed from the age of 20 years in men and from the age of 30 years in women 52) ( Fig.4 ) . However, early diagnosis and aggressive LDL-C lowering therapy with statins and other drugs can delay the onset of CAD in HeFH and improve their life expectancy 36 , 53) .
Fig.3.
Cumulative number of myocardial infarction cases in HeFH (Cases were added to Ref. 35 and the display format was modified.)
Fig.4.
Coronary CT plaque score in HeFH (modified from Ref. 52)
2.3.3 Tendon and Skin Xanthomas
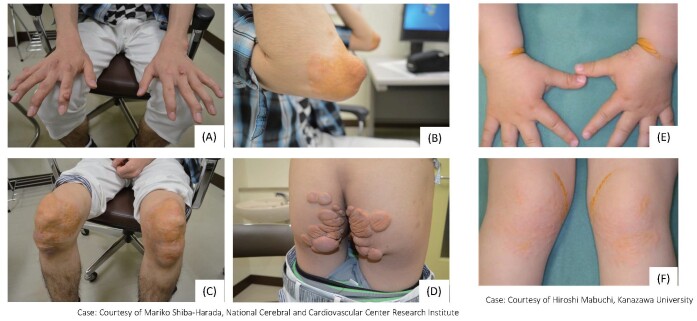
Important physical findings in the clinical diagnosis of FH are the presence of tendon and skin xanthomas. Compared to HeFH, HoFH have more pronounced xanthomas at younger ages ( Fig.5 ) . Xanthomas of the skin occur most frequently in areas subjected to mechanical stimulation, such as the extensor surfaces of the elbow and knee joints, the wrists, and the buttocks. Eyelid xanthomas (xanthelasmas) are less specific and have less diagnostic value because they are often seen outside of FH, but they are a suspicious finding of FH. Tendon xanthomas often present as thickening of the Achilles tendon, and palpation is important for diagnosis. However, FH should not be ruled out even in the absence of xanthomas. In fact, 20%–30% of patients with a confirmed diagnosis of FH by genetic testing do not have xanthomas 54) . In general, xanthomas are present but mild in pre-adulthood, and these often become more evident with age; conversely, there are cases in which tendon xanthomas are absent, even in older patients. It is also difficult to evaluate after Achilles tendon rupture. Therefore, family investigations and genetic testing are important in the absence of xanthoma. Patients with FH may also complain of pain in the Achilles tendon area due to Achilles tendonitis. FH is strongly suspected when calcification and thickening of the Achilles tendon area is observed.
Fig.5. Xanthoma in HoFH.
Skin and tuberous xanthomas in a 22‐year‐old man (A) Finger tuberous xanthomas, (B) Xanthoma planum on the elbows, (C) Xanthoma planum on the knees, (D) Xanthoma planum on the buttocks. (E, F) Skin xanthomas in a 3‐year‐old boy
2.3.4 Corneal Ring
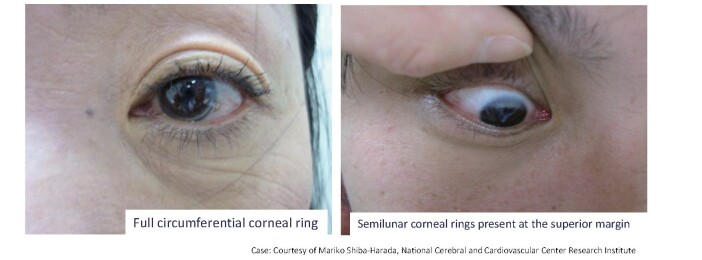
The corneal ring, as shown in Fig.6 , is also a characteristic finding of FH, but its prevalence is approximately 30% in FH patients under the age of 50 48 , 55) . In the elderly population, it can be difficult to distinguish from arcus senilis, as it is common in this age group. However, when observed in patients under 50 years of age, corneal arcus has high diagnostic value.
Fig.6.
Corneal ring in patients with FH
2.3.5 Other Risk Factors of FH
The prevalence of CAD in HeFH patients is known to vary widely in terms of age of onset and rate of progression, but risk factors have been reported in the development of CAD in Japan, as described below. In a study examining 117 HeFH in the Hokuriku region, Yagi et al. determined that diabetes and low HDL-C levels are significant risk factors 56) ; meanwhile, in a study by Harada-Shiba et al., analyzing HeFH in the Kinki region, it was determined that smoking, family history of CAD, Achilles tendon thickening, high LDL-C levels, low HDL-C levels, high TG levels, diabetes, and hypertension are risk factors 57) . Hirobe et al. also noted the involvement of low HDL-C levels in the onset of CAD in FH 57) ; Yanagi et al. noted the involvement of diabetes and impaired glucose tolerance 58) ; Nakamura et al. noted the importance of visceral fat 59) ; and Ogura et al. noted that hypertension and low HDL-C levels (as well as impaired cholesterol efflux capacity) were residual risks in HeFH patients treated with statins 55) .
Furthermore, the Research Team Investigating Primary Hyperlipidemia reported that in patients with FH with CAD, high TG levels and low HDL-C levels were common complications 60) . A subanalysis of FAME also reported that hypo-HDL-C and hypertension are associated with increased carotid IMT, thus indicating the need for comprehensive management of risks rather than reduction of LDL-C levels alone 61) . In a study by Tada et al., an association between arterial wall stiffness and CAD in patients with FH 62) was reported. There are also several reports overseas that hyper-Lp(a) is a risk factor for CAD even in patients with FH 63 - 66) . Among clinically diagnosed FH, there are reports of an increased risk of CAD in patients with genetic mutations associated with FH 67) . Furthermore, Tada and Doi et al. reported that more severe cases were reported in FH with concurrent rare pathogenic mutations in genes related to LDL-C such as ATP-binding cassette transporter G5 (ABCG5), ATP-binding cassette transporter G8 (ABCG8) apolipoprotein E (APOE), LDLRAP1, and PCSK9 68 , 69) .
2.4 Diagnosis of FH
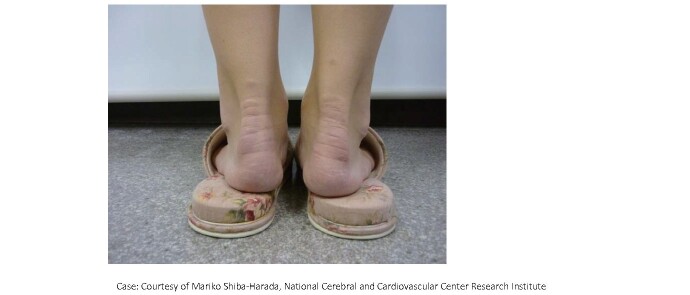
FH is commonly diagnosed with elevated levels of LDL-C, Achilles tendon thickening, skin xanthomas, and familial history. Achilles tendon thickening is diagnosed by visual inspection and palpation ( Fig.7 ) , but if there is any doubt on the diagnosis, radiography or ultrasound is performed to measure the maximum diameter of the Achilles tendon. In radiography, the diagnosis of thickening is made at 8.0 mm or more in men and 7.5 mm or more in women 1) ( Fig.8 ) , whereas in ultrasonography, the diagnosis of thickening is made at 6.0 mm or more in men and 5.5 mm or more in women 70) ( Fig.9 ) . Tendon and skin xanthomas frequently occur on the extensor sides of the hands, elbows, or knees. Due to its inherited form, the development of premature CAD is often found in family members. Therefore, extensive research on the family history is very important to make a correct diagnosis. When FH is diagnosed, screening of blood relatives (cascade screening) should always be considered.
Fig.7.
Achilles tendon thickening in a patient with FH
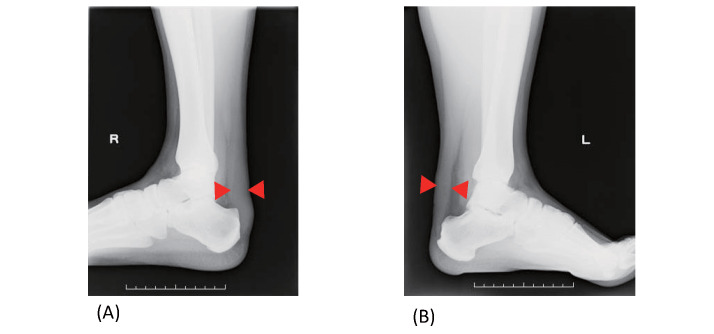
Fig.8. Achilles tendon radiography in patients with FH.
(A) Achilles tendon in a 40‐year‐old man (maximum thickness: 22 mm),
(B) Achilles tendon in a 29‐year‐old man (maximum thickness: 19 mm).
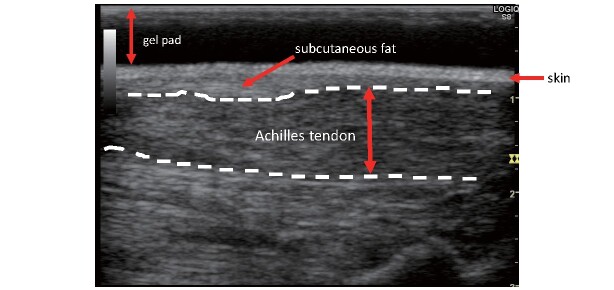
Fig.9. Achilles tendon echo in FH (long-axis cross-sectional image).
Reprinted from Standardized Assessment of Achilles Tendon Thickness Measurement by Ultrasound for Screening Adult Familial Hypercholesterolemia 2018, Japan Atherosclerosis Society and the Japan Society of Ultrasonics in Medicine
The features of HoFH are serum TC ≥ 600 mg/dL, as well as childhood xanthomas and atherosclerotic diseases, and the parents are HeFH. Childhood xanthomas are characteristic, and the initial consultation regarding them may be in a dermatology department; thus, it is extremely important that the dermatologist does not miss the HoFH at this point. It may be difficult to distinguish the serious cases of HeFH, so in order to make a definitive diagnosis of HoFH, genetic analysis is required. In recent years, diagnosis by decreased LDLR activity (less than 20% of normal) 71 in fibroblasts and lymphocytes has rarely been performed 71) .
2.4.1 FH Diagnostic Criteria
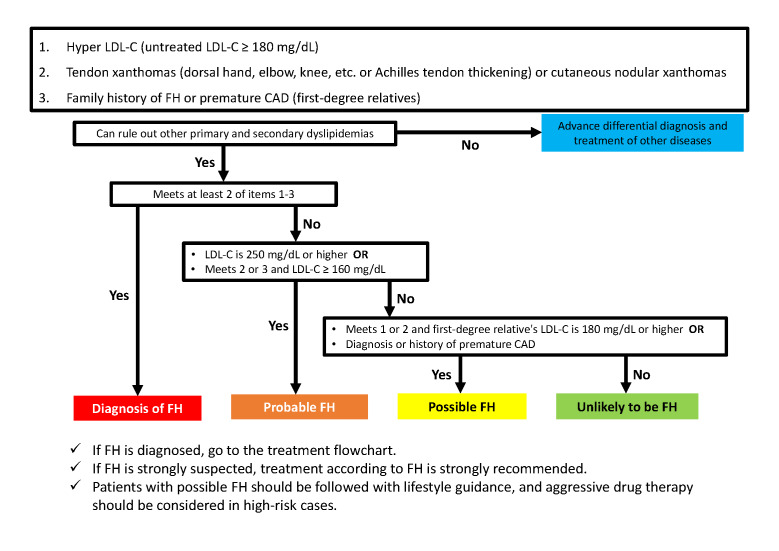
Diagnostic criteria for FH are shown in Table 1 , and the flowchart for diagnosis is presented in Fig.10 . The diagnosis should be made after excluding other primary and secondary dyslipidemias, and if the patient is already on drug therapy, the lipid levels that triggered the therapy should be used as a reference (see 2.4.5 for an evaluation of Achilles tendon thickening). Cutaneous nodular xanthomas do not include eyelid xanthomas. FH is diagnosed if at least two of the three items in Table 1 are met. FH is strongly suspected if LDL-C is ≥ 250 mg/dL or if two or three items are met and LDL-C is ≥ 160 mg/dL, even if at least two items are not met. Even if none of the above applies, if the patient meets one or two items and has a first-degree relative with LDL-C ≥ 180 mg/dL or a diagnosis or history of premature CAD, the patient is considered to have FH and should be followed up with lifestyle guidance. In high-risk cases, aggressive drug therapy should be considered. Genetic testing is useful in difficult to diagnose cases, and referral to a specialist should be considered. FH is diagnosed when pathogenic gene mutations are present; genetic testing is preferred when HoFH is suspected; genetic testing is also found useful for HeFH, which is difficult to diagnose. The same diagnostic criteria apply to HoFH. If a diagnosis of FH is made, it is strongly recommended that family members are also examined. It is important to note that young patients with FH may meet only one of the criteria and may have LDL-C less than 180 mg/dL.
Table 1. Diagnostic criteria for FH in adults (15 years and older).
| 1. Hyper LDL-C (untreated LDL-C ≥180 mg/dL) |
|---|
| 2. Tendon xanthomas (dorsal hand, elbow, knee, etc. or Achilles tendon thickening) or cutaneous nodular xanthomas |
| 3. Family history of FH or premature CAD (first-degree relatives) |
|
|
|
|
|
|
|
|
|
|
Fig.10.
Flowchart of FH diagnosis in adults (15 years and older)
Further, it should be noted that LDL-C levels temporarily decrease when complicated by serious diseases such as acute myocardial infarction, and it is estimated that LDL-C levels decrease by approximately 30% 72) . Therefore, when examining a patient with acute myocardial infarction, the Achilles tendon should be palpated, and a family survey should be conducted. This requires particular attention if the patient is young. The sensitivity and specificity of this diagnostic criterion should be further verified in various cohorts.
2.4.2 Genetic Testing for FH
Ethical considerations must be taken into account for genetic testing (germline human genetic testing) for FH, whether in adults or children. The Japanese Medical Association’s “Guidelines for Genetic Testing and Diagnosis in Medical Care 73) ” is a guideline to conduct the genetic testing. In the case of genetic testing to diagnose patients who have already developed the disease (e.g., high LDL-C), in addition to explaining the significance and purpose of the test at an appropriate time before the test, the situation after the results is obtained, and the possibility that the test results may affect blood relatives should also be explained to the test subject. The test subject should be supported so that he or she can make a voluntary decision to undergo the test or not, after fully understanding these factors. After adequate explanation and support, written consent is then recommended. As a rule, the attending physician should explain the genetic test and confirm consent and understanding (informed consent for adults, informed assent for minors, etc.) prior to the test. Furthermore, ensure that professional genetic counseling and decision-making support are available as needed. The diagnosis of an asymptomatic carrier (with normal LDL-C and no other symptoms) is a test for a person who does not develop the disease and therefore does not require treatment. In principle, the test should not be performed unless there is a special reason in situations where the consent of the patient cannot be obtained.
When testing a minor, it is necessary to obtain the consent of a person who is in a position to consent to the testing on behalf of the minor, and the best interests of the person to be tested should be fully considered. It is also advised to provide explanations that are appropriate to the level of understanding of the examinee to obtain his/her consent (informed consent). Provide genetic counseling as needed. Even if the disease has already developed (e.g., high cholesterol), testing may be considered for minors as needed, as the disease can be treated with a definitive diagnosis. On the other hand, for the diagnosis of an asymptomatic carrier (no symptoms at all, including cholesterol), testing should be postponed until the individual becomes an adult and can make decisions autonomously.
Each principal investigator will establish a policy for handling incidental/secondary findings in genetic testing as a framework for the study, in order to explain it well to participants and make them understand when obtaining informed consent 74) .
Since genetic testing for HoFH has been covered by insurance since April 2022 in Japan, it is expected to be used for the definitive diagnosis and selection of treatment options.
2.4.3 Genetic Counseling in FH
Genetic counseling should be provided at the appropriate time for genetic testing and diagnosis, if necessary. Genetic counseling should not only provide information, but also psychological and social support to enable the patient/subject to make a voluntary choice. Therefore, it is desirable that physicians with extensive experience in the treatment of the disease and people skilled in genetic counseling cooperate to provide genetic counseling as team medical care.
Since the basic form of FH is an autosomal dominant inherited disease, here are its described three etiologic gene abnormalities, that is, LDLR, APOB, and PCSK9. If the child is HeFH, either parent is HeFH. If one parent is HeFH, there is a 50% chance that the child will also be HeFH, and if both parents are HeFH, the child has a 75% chance of developing the disease (25%: HoFH, 50%: HeFH). When both parents of an HeFH child are clinically unremarkable, it is necessary to consider the possibility of a de novo mutation in the affected child, or both parents have mild symptoms, or the absence of a true blood relationship, such as adoption, etc. Prenatal diagnosis and preimplantation diagnosis are attempted in some cases; however, these procedures should be considered very carefully, given that there is no intellectual abnormality and treatments exist even if the child is homozygous for FH.
2.4.4 Notes on Genetic Test Results for FH
Like other genetic diseases, current genetic testing for FH also has its limitations. The detection rate of pathogenic mutations in clinically diagnosed FH cases is reported to be about 60%–80% 39) . Furthermore, approximately 10% of the causative genetic mutations in FH are structural mutations in the LDLR 39) . Therefore, care should be taken because some analytical methods do not identify such structural variations.
2.4.5 Evaluation of Achilles Tendon Thickening
2.4.5.1 Achilles Tendon Radiography
The position is made so that the lower leg bone and plantar surface are on a 90-degree angle, and the incidence is made from the lateral side to the center of the external fibula. The imaging distance is 120 cm, and the imaging conditions are as follows: 50 kV, 5.0 mA (e.g., 100 mA×0.05 s, 50 mA×0.1 s). Achilles tendon is presumed to thicken when a maximum diameter of 8.0 mm or greater for men and 7.5 mm or greater for women is diagnosed. Using these cutoff values, the sensitivity and specificity to predict patients positive for pathogenic mutations are reported to be 82% and 89%, respectively 1) . The sensitivity and specificity for predicting patients positive for FH pathogenic gene mutations in the case of strong suspicion of FH are reported to be 96% and 83%, respectively 1) .
2.4.5.2 Achilles Tendon Ultrasound
As regards the evaluation of Achilles tendon thickness (ATT) via radiography, some general practitioners have commented that radiography is difficult to perform and that the boundary line between the skin and the Achilles tendon is unclear, making measurement difficult. Michikura et al. showed that ultrasonography correlates with radiography in the evaluation of Achilles tendon thickening in FH and were the first in Japan to establish and report a cutoff value for ATT by ultrasonography for the diagnosis of FH 70) . In response, in 2018, the Japan Society of Ultrasonics in Medicine and the Japan Atherosclerosis Society jointly published the “Standard Evaluation Method for Measuring Achilles Tendon Thickness by Ultrasound for Screening Adult Familial Hypercholesterolemia,” setting the diagnostic threshold for thickening of ATT at ≥ 6.0 mm for men and ≥ 5.5 mm for women.
The measurement method is outlined below. The subject is placed in either (1) kneeling or (2) supine position on the bed. The ATT is measured at its thickest point, but since the Achilles tendon attaches to the calcaneus in a twisting motion, a simple long-axis image from the body surface will not provide a correct ATT measurement. Therefore, after observation in the short-axis image, the maximum thickened area is identified, and ATT is measured in the image in which the maximum thickened area of ATT is delineated in the long-axis image considering the torsional direction ( Fig.9 ) . Also, sufficient echo jelly should be used during the examination (gel pads can be used), and caution should be observed to not apply too much pressure to the skin with the probe.
2.4.6 Differential Diagnosis from Other Primary and Secondary Dyslipidemias
The main diseases requiring differentiation from FH are secondary hyperlipidemia characterized by hyper-LDL-C (due to diabetes, hypothyroidism, nephrotic syndrome, cholestatic liver disease, drug-induced hyperlipidemia (by steroids, etc.)) and the familial combined hyperlipidemia (FCHL). FCHL can be distinguished from FH based on the absence of tendon xanthomas, the presence of small dense LDL, and the presence of other types of dyslipidemia within the family (type IIa, type IIb, type IV). Table 2 summarizes the differentiating features of FCHL. Primary dyslipidemias with tendon xanthomas include sitosterolemia and cerebral tendon xanthomatosis.
Table 2. Differentiation between FH (familial hypercholesterolemia) and FCHL (familial combined hyperlipidemia).
| HeFH | FCHL | |
|---|---|---|
| causative gene |
Monogenic causes LDLR, PCSK9, APOB, etc. |
Complex genetic USF‐1 and LPL have been considered as candidates, but the causative gene has not yet been determined. |
| frequency | 1 in 200‐500 | 1 in approximately 100 |
| Lipid profiles | Mostly IIa, sometimes IIb | Both the family and the patient can have three phenotypes during the course of the disease: IIa ⇔ IIb ⇔ IV. |
| Achilles tendon thickening, xanthoma of the skin | Often observed | Not observed |
| Premature corneal rings | Often observed | Not observed |
| Small dense LDL | Rare | Often observed |
| Insulin resistance | Rare | Often observed |
Sitosterolemia is an autosomal recessive hereditary disease with prominent skin and tendon xanthomas from a young age; it is diagnosed based on elevated blood levels of plant sterols (sitosterol, campesterol, etc.). In many cases, serum LDL-C is normal, but when it is elevated to moderate or higher levels, a differential diagnosis from FH or ARH is necessary. The causal gene is ABCG5 or ABCG8 75 - 77) . In the past, the concept of pseudohomozygous type 2 hypercholesterolemia was proposed, in which parents usually do not show overt hypercholesterolemia and respond well to diet and anion exchange resins. However, based on pathogenesis and other factors, it is now considered sitosterolemia 78 , 79) .
Cerebrotendinous xanthomatosis (CTX) is an autosomal recessive hereditary disease diagnosed from symptoms such as tendon xanthomas, typically Achilles tendon xanthomas, reduced intelligence, ataxia, speech impediments, cataract and cerebellar symptoms, as well as high blood cholestanol levels. The essential feature of the disease is impaired bile acid synthesis due to 27α-hydroxylase deficiency 80 , 81) .
2.5 Screening and Follow-Up for atherosclerotic cardiovascular disease (ASCVD) in FH
FH are susceptible to systemic ASCVD, including CAD. Therefore, early detection and treatment for atherosclerotic diseases are required. It is endeavored to conduct patient interviews, auscultation (heart sounds and bruit in carotid artery, subclavian artery, renal artery and femoral artery areas), and palpation on a regular basis. If there are symptoms of suspected CAD, electrocardiography, exercise electrocardiography (treadmill, ergometer), echocardiography, and drug loading or exercise stress myocardial scintigraphy are to be performed. When CAD is suspected on these examinations, coronary CT or coronary angiography is performed to determine the location of coronary artery stenosis. However, FH patients frequently show calcification, which sometimes makes it difficult to make a diagnosis. Characteristic findings of coronary angiography in HeFH include marked stenotic lesions at the origin and dilative lesions downstream (coronary aneurysms).
In evaluating carotid atherosclerosis in HeFH, in addition to listening to noise on physical examination, carry out carotid artery echocardiography, and, if stenosis is suspected, MR, CT, or vascular angiography should be performed. To evaluate the presence of cerebral infarction, MRI and CT should be performed if necessary.
Aortic aneurysms and other complications are common in elderly HeFH patients. Thus, chest X-ray and CT exams should be performed to evaluate thoracic aortic aneurysms and abdominal ultrasonography and CT to evaluate abdominal aortic aneurysms. Treatment needs cooperation with cardiovascular surgeons.
In some HeFH patients, peripheral artery disease (PAD) develops concomitantly; therefore, during the interview, patients should be asked as regards the presence or absence of cold feet and intermittent claudication, and the dorsal and posterior tibial arteries should be palpated. To evaluate atherosclerosis of the femoral artery, the ankle-brachial blood pressure index (ABI) should be measured. When stenosis is suspected, femoral artery ultrasound (Doppler method), CT angiography, and MR angiography should be performed.
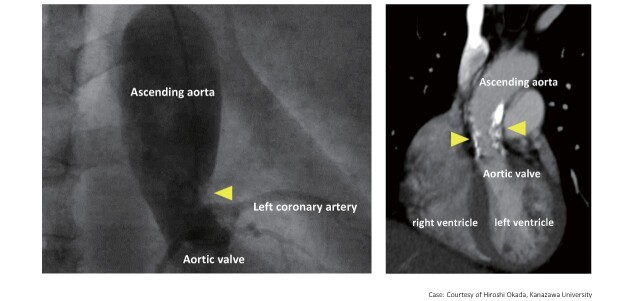
To assess valvular disorders, such as aortic valve stenosis (AS), an echocardiography and, if required, cardiac catheterization should be performed. Surgical Aortic valve replacement or transcatheter aortic valve implantation (TAVI)should be performed in severe cases with narrowed aortic valve orifice area and large aortic valve pressure gradient. In the case of HoFH, supravalvular aortic stenosis should also be evaluated ( Fig.11 ) .
Fig.11.
Supravalvular aortic stenosis in HoFH
3. Treatment of HeFH ( Fig.12 )
Fig.12.
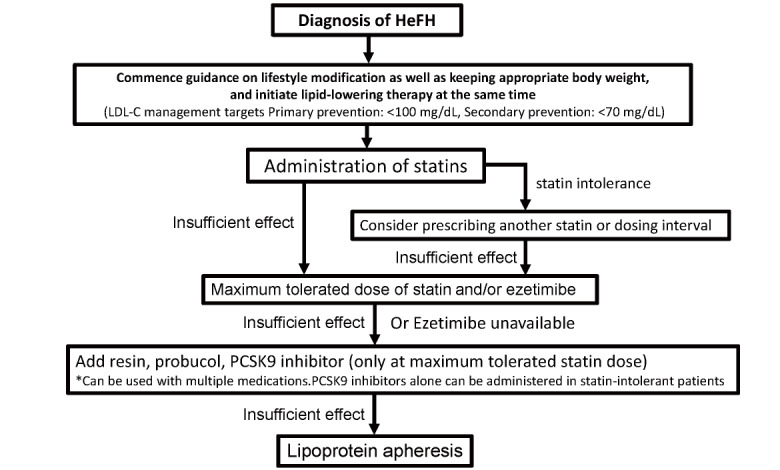
Flowchart of HeFH treatment in adults (15 years and older)
3.1 Target LDL-C Levels for the Management of HeFH
As HeFH are at extremely high-risk of developing ASCVD, especially CAD, the risk of developing ASCVD in primary prevention is at least equivalent to that of usual secondary prevention. Therefore, the target control value of LDL-C for patients with HeFH for primary prevention should be less than 100 mg/dL. It should also be noted that the risk assessment based on the risk chart issued by the Japan Atherosclerosis Society is not applicable in the treatment of FH for primary prevention.
In HeFH patients for secondary prevention, the LDL-C management target level should be less than 70 mg/dL because it can be considered to be at even higher risk.
As it is ethically unacceptable to conduct clinical trials without lipid-lowering therapy in FH, there is no clear evidence for the validity of these numerical targets. The achievement of the management target does not always ensure the absence of future cardiovascular events.
3.2 Dietary Therapy in HeFH
Dietary therapy should also be practiced for FH, and its methods are similar to those for other dyslipidemias. In other words, the Japan Atherosclerosis Society has identified the following points to which patients with hyper-LDL-C should pay attention to in their dietary therapy: (1) maintain appropriate total energy intake, (2) reduce the saturated fatty acid intake from 4.5% or more and less than 7%, (3) reduce trans-fatty acids, (4) keep cholesterol intake below 200 mg/day, and (5) actively consume soy protein, dietary fiber, etc.
3.3 Exercise Therapy in HeFH
Although there are virtually no studies on exercise therapy for FH, exercise therapy should also be practiced in HeFH. However, due to the high-risk of ASCVD, screening for ASCVD before administering exercise therapy is essential. ASCVD should be evaluated by interview, ECG, exercise stress ECG, echocardiography, ABI, etc. If complications of CAD, AS, or PAD are suspected, exercise therapy should be started after appropriate and adequate treatment.
3.4 Drug Therapy in HeFH
Lifestyle interventions such as diet, exercise, smoking cessation, and obesity control alone are often insufficient to achieve adequate lipid control in patients with HeFH; thus, concomitant pharmacotherapy with statins as first-line agents is recommended. A retrospective analysis of 329 HeFH patients in Japan also showed that LDL-C lowering therapy with oral statins significantly delayed the onset of CAD 53) . Based on the efficiency of LDL-C lowering, the so-called strong statins are the main therapeutic agents.
Even in HeFH, the “rate of accumulation of LDL-C” can vary depending on the presence or absence of genetic abnormalities or other concomitant risks (see 2.3.5), which, in turn, affect the timing of onset and risk of developing ASCVD; therefore, the timing of treatment initiation (the earlier, the better) and management goals should be determined based on individual risk considerations 52 , 54 , 82) . In FAME, a FH registry study in Japan, risk factors for developing CAD are as follows: male, age >40 years, heterozygous FH score >20 points, hypertension, low HDL-C, and having a sibling with CAD 83) .
Statins should be started at the usual dose and should be increased while observing their efficacy and side effects. The LDL-C lowering effect of statins is enhanced in a dose-dependent manner, but the frequency and severity of adverse effects may also increase, so care should be exercised. For patients with statin intolerance, try a different statin or a different dose interval (every other day, twice weekly, etc.) with the aim of increasing the maximum tolerated dose 84) .
When sufficient efficacy is not obtained with statin alone, other lipid-lowering agents can be used in combination with statins to achieve even greater LDL-C lowering effects. Concomitant medications include small intestinal cholesterol transporter inhibitors (ezetimibe), anion exchange resins, probucol, and PCSK9 inhibitors 85) . The ENHANCE study reported that the addition of ezetimibe to statin in patients with FH achieved a further LDL-C lowering effect 31) . In the FAME study, LDL-C <100 mg/dL was achieved in 12.3% of patients in primary prevention and only 1.8% in secondary prevention (LDL-C <70 mg/dL) 83) . In daily clinical practice in Japan, it was found that although strong statins are primarily administered, the dose is increased to the maximum tolerated dose in a small number of cases and that the combination of statin and ezetimibe is used in only 26.3% of cases. With regard to PCSK9 inhibitors, the combination of evolocumab or alirocumab in HeFH already treated with statins (and ezetimibe) has been shown to be relatively safe, with additional reduction in LDL-C (approximately 60%) and Lp(a) 28 , 29 , 86) , and long-term safety has been confirmed up to around 3 years of treatment 87 - 89) . Additionally, an average reduction of 35% LDL-C in statin-intolerant FH patients has been noted 90) . Therefore, in HeFH drug treatment, the first choice is statins, and if the effect is deemed inadequate, the dose should be increased to the maximum tolerated dose, or other combination therapies such as ezetimibe combination therapy should be implemented. If the response remains inadequate, additional PCSK9 inhibitors should be administered after careful consideration of individual risk ( Fig.12 ) . In particular, it is desirable to promptly lower and control LDL-C levels in high-risk cases for secondary prevention. HeFH, excluding statin intolerance, who do not achieve the expected LDL-C lowering effect with PCSK9 inhibitor combination therapy in addition to usual oral therapy, should be referred to a specialist, including genetic testing, as they are most likely to be HoFH. However, it is not yet clear whether these combination therapies are more effective in reducing cardiovascular events in patients with FH than statin therapy alone; thus, further evidence needs to be accumulated.
In patients who cannot use statins at all due to side effects such as myalgia (including increased CK) and liver dysfunction, the lipid-lowering drugs mentioned above are administered alone or in combination, but their effectiveness in preventing atherosclerosis has not been fully established. In Japan, a retrospective study suggests that the use of probucol delays the recurrence of CAD in HeFH 91) , but its side effects such as QT prolongation should also be kept in mind.
3.5 Lipoprotein Apheresis for HeFH
For HeFH, the use of statins in combination with ezetimibe and PCSK9 inhibitors has made it possible to achieve LDL-C control targets in many cases in recent years, but the number of cases in which lipoprotein apheresis is indicated has decreased; however, this should not be a reason to hesitate in implementing lipoprotein apheresis if the patient is drug resistant and has advanced CAD. For HeFH, insurance coverage is allowed in cases where serum LDL-C level exceeds 370 mg/dL in steady state (body weight and serum albumin can be maintained) under diet therapy and does not fall below 170 mg/dL, and if coronary atherosclerosis is evident with xanthoma. Additionally, lipoprotein apheresis can be combined with PCSK9 inhibitors, but since anti-PCSK9 antibodies are removed during LDL apheresis, subcutaneous injections should be made after lipoprotein apheresis treatment if the combination is used. On the other hand, it has been reported that the combination of lipoprotein apheresis and treatment with a PCSK9 inhibitor allowed approximately 63% of patients to wean off lipoprotein apheresis and safely achieve good LDL-C control 92) .
4. Treatment of HoFH ( Fig.13 )
Fig.13.
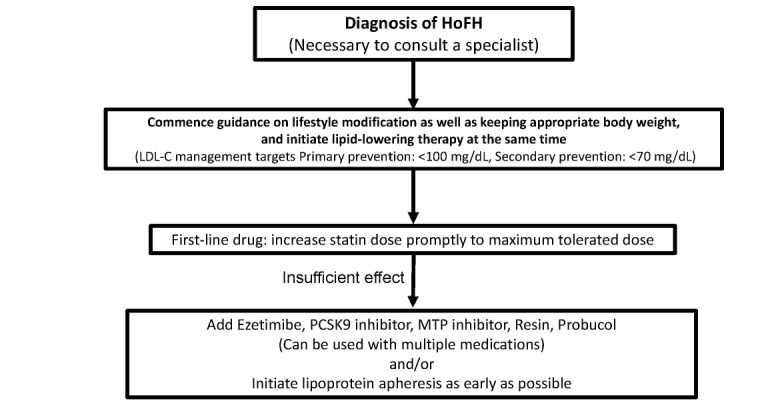
Flowchart of HoFH treatment in adults (15 years and older)
In HoFH, it is essential to lower LDL-C as quickly as possible, and aggressive treatment should be implemented. Ideal LDL-C control targets for HoFH are <100 mg/dL for primary prevention and <70 mg/dL for secondary prevention, although this is often difficult to achieve 93) .
4.2 Lifestyle Interventions in HoFH
As in HeFH patients, lifestyle interventions such as diet, exercise, smoking cessation, and obesity control are fundamental to the treatment of HoFH patients. Since HoFH develops atherosclerosis significantly more rapidly than HeFH, before giving guidance in exercise therapy and initiating it, patients should be carefully evaluated for CAD, as well as for valvular disease (particularly AS, supravalvular aortic stenosis) and aortic aneurysms.
4.3 Drug Therapy in HoFH
In HoFH, the above lifestyle interventions alone are not sufficient to control the disease; thus, strong LDL-C lowering therapy is required from a young age to prevent the development and progression of CAD. However, statins, anion exchange resins, and PCSK9 inhibitors all have the primary mechanism of action for increasing the expression (activity) of the LDLR. For the defective type, in which only a small amount of LDLR activity remains, slight efficacy is observed; however, in the negative type, in which LDLR activity is completely absent, no LDL-C lowering effect is observed 94 - 96) . In a study examining the LDL-C lowering effect of PCSK9 inhibitors in adult HoFH patients, although the LDL-C lowering effect of PCSK9 inhibitors (approximately 30%) was confirmed 30) , PCSK9 inhibitor treatment should be discontinued if LDL-C is not reduced at all. However, a retrospective study found that the administration of statin and other drugs was effective in reducing the mortality rates in HoFH 97) . Furthermore, a microsomal triglyceride transfer protein (MTP) inhibitor, which was developed for patients with HoFH, has been reported to reduce LDL-C by approximately 50% 98) . A study was conducted in HoFH patients in Japan, and it is now on the market 99) . However, since the frequencies of adverse events such as fatty liver and diarrhea are high with MTP inhibitor, it is essential to strictly control fat and alcohol intake 100) . Probucol reportedly exerts a certain LDL-C lowering effect in HoFH and can cause the regression or disappearance of xanthoma on the skin and Achilles tendon 101) . However, for LDL-C control, lipoprotein apheresis therapy is still required once every 1–2 weeks in many cases. Liver transplantation is an option for patients who are resistant or intolerant to all of the above treatments, but there are currently very few cases of liver transplantation in Japan 102 , 103) .
4.4 Lipoprotein Apheresis
4.4.1 Indications for Lipoprotein Apheresis
In patients with HoFH, as it is difficult to sufficiently reduce the LDL-C level using existing drug therapies, continued therapy for lipoprotein apheresis is required from childhood in many cases. Lipoprotein apheresis, which uses an extracorporeal circulatory system to separate and remove LDL from the blood, was developed to remove LDL from the blood of HoFH.
4.4.2 Effects of Lipoprotein Apheresis
Lipoprotein apheresis therapy can be administered safely in children, with no effect on development or growth, and there are reports of not only a reduction or disappearance of cutaneous xanthomas, but also inhibition of progression or improvement of atherosclerotic lesions such as aortic stenosis/ supravalvular stenosis and coronary arteries 104 - 106) . It has also been reported that, in addition to lowering LDL-C, lipoprotein apheresis inhibits the expression of cell adhesion molecules (ICAM-1, ELAM-1, etc.) and suppresses thrombus formation by decreasing fibrinogen and coagulation factors that LDL is not readily oxidized after lipoprotein apheresis and that it has an anti-atherosclerotic action via improvement of LDL subtypes 107) . With the introduction of PCSK9 and MTP inhibitors, there are now a significant number of cases in which LDL-C can be managed after withdrawal from lipoprotein apheresis. However, since lipoprotein apheresis therapy is expected to have not only LDL-C lowering effects but also the above-mentioned effects, careful consideration should be given when considering withdrawal from lipoprotein apheresis 108) . Future prospective studies targeting the new onset of ASCVD after lipoprotein apheresis withdrawal are desirable.
4.4.3 When to Start Lipoprotein Apheresis Therapy
The earlier the age at which lipoprotein apheresis therapy is started, the better to control the progression of ASCVD, but it is difficult to implement until the affected child is able to rest during lipoprotein apheresis. The realistic starting time for treatment is around 4–6 years of age, when the child is bedridden and extracorporeal circulation can be performed, although there have been reports of treatment starting at 3.5 years of age 109) . In some cases, coronary artery stenosis and complete occlusion, coronary valve stenosis, and supravalvular aortic stenosis are already present in infancy, and if lipoprotein apheresis is initiated from the age 10 years onward, the prognosis is poor 110) , and it has been recommended to start treatment as early as possible 104) . In early childhood, simple plasma exchange with low extracorporeal circulatory volume should be performed, or existing LDL adsorption methods should be used with a device to reduce extracorporeal circulatory volume.
4.4.4 Plasma Volume for Lipoprotein Apheresis and Blood Access
The amount of plasma treated for lipoprotein apheresis is determined by the pretreatment LDL-C and the patient’s plasma volume (proportional to body weight). Normally, treatment with 3 to 6 L of plasma can reduce LDL-C by 60 to 80%. However, LDL-C starts to rise soon after treatment and continues to rise until the next treatment. The therapeutic effect of lipoprotein apheresis is considered to be proportional to the time-integrated average value (CAVG), which can be estimated by CAVG=Cmin+0.73 (Cmax-Cmin). In treatment, the volume of plasma processed, the frequency of treatment, and concomitant medications should be set to reduce this integral mean of LDL-C.
Blood access in lipoprotein apheresis is common via the cubital or brachial vein. Unlike hemodialysis in renal failure, blood flow rates range from 50 to 150 mL/min, limiting the need for shunting. FH patients are prone to shunt occlusion and thus require caution because, unlike renal failure patients, they have normal hematocrit.
4.4.5 Methods of LDL Apheresis
4.4.5.1 Simple Plasma Exchange
This was the first apheresis method and was initially conducted by Thompson et al. in 1975. Marked improvement in clinical symptoms such as lower cholesterol, less anginal pain with improvement in coronary artery stenosis, and the disappearance of xanthomas was reported in patients with FH 111) . This method reduces cholesterol by removing the patient’s plasma and replacing it with a human albumin preparation. Therefore, substances necessary for the body, such as immunoglobulins, are also removed in a non-selective manner. For this reason, currently, it is only used in HoFH children under 10 years old to reduce extracorporeal circulating blood volume.
4.4.5.2 Double Membrane Filtration Method
This method has already been developed in Japan. Drs. Yoshikawa and Kishino used two filtration membranes with different diameters to separate blood cells and plasma via the primary membrane and remove LDL and VLDL, which are known to be large particles among plasma components, via the secondary membrane. It selectively removes VLDL and LDL, and the HDL removal rate is lower than that by simple plasma exchange method. However, it has drawbacks, such as the fact that albumin (10%) and globulin (30%–60%) are also removed via the secondary membrane, and the amount of plasma processed is limited due to clogging of the secondary membrane that resulted in in increased membrane pressure.
4.4.5.3 LDL Adsorption Method (Liposorber System)
The LDL adsorption method was developed based on the fact that LDL selectively binds to dextran sulfate. After separating the blood into blood cell components and plasma components, plasma is passed through columns containing dextran sulfate, the negatively charged ligand, in porous beads, and the positively charged apo B containing lipoproteins (VLDL, LDL, lipoprotein (a)) are selectively removed 112) . Two small-volume columns (150 mL) (LA15) are used alternately, with the adsorbed LDL eluted by a high concentration of NaCl (5%).
4.4.6 Adverse Events and Cautions of Lipoprotein Apheresis
Due to a decrease in effective circulating blood volume during apheresis treatment, hypotension can frequently occur; thus, great care is needed, particularly in the case of patients with aortic valve disease and CAD. Iron deficiency anemia is a common adverse event, but patients can easily recover by taking oral iron supplements. Selective LDL adsorption therapy, with its negative charge, binds to apoB-100 in LDL and removes LDL by adsorption, while activating the blood coagulation system and stimulating bradykinin production. Concomitant use of ACE inhibitors, which inhibit bradykinin metabolism and cause severe shock symptoms, is contraindicated.
4.5 Procedure for the Designation of HoFH as Intractable Disease
HoFH is one of the designated intractable diseases in Japan. The certification criteria are as follows: in addition to the definite cases in which the diagnosis is confirmed by genetic testing of genes involved in the LDL metabolic pathway or by measurement of LDLR activity, patients with marked hypercholesterolemia or cutaneous xanthomas since childhood as almost definite cases, and patients who are refractory to drug therapy. Designated intractable diseases are listed on the website of the Ministry of Health, Labor and Welfare’s Intractable Disease Information Center (https://www.mhlw.go.jp/stf/seisakunitsuite/bunya/ 0000084783.html).
Procedures and inquiries are handled at the nearest public health center.
5. Female FH Patients of Childbearing Potential
Lifestyle modification is fundamental, but in many cases, drug therapy must be used to lower LDL-C 113) . In this case, the patient should consult a specialist in the treatment of dyslipidemia after puberty, and the specialist should consider the degree of risk in each individual case, including the initiation of drug treatment and the type of drug. Drug therapy other than anion exchange resins during pregnancy should be used with caution due to concerns about the risk of fetal malformations and other problems. According to the National Institute for Health and Clinical Excellence, if pregnancy is detected during drug treatment, the medication should be stopped immediately. If a woman wishes to have a baby while on medication, she should stop taking the medication for three months before attempting to conceive.
For patients with FH who wish to become pregnant, check the atherosclerotic status beforehand, e.g., by carotid artery echocardiography, in preparation for a safe continuation of pregnancy and delivery. In high-risk cases, screen for complications of CAD and aortic or supravalvular stenosis, and take appropriate measures as needed 114) . As statins have been shown to be teratogenic in animal studies and the safety of lipid-lowering drugs other than anion exchange resins has not been established for administration during human pregnancy, preconception counseling is necessary for HeFH and HoFH women, including explaining the need to stop taking lipid-lowering medications other than anion exchange resins at least 3 months before planning a pregnancy and during the lactation period after delivery. It is important to plan for pregnancy in HoFH patients, and screening for atherosclerosis should be performed prior to pregnancy by echocardiography, electrocardiography, exercise stress echocardiography, and carotid echocardiography. Discontinue lipid-lowering medications other than anion exchange resin (resin) 3 months prior to expected pregnancy. LDL-C and TG are known to increase further during pregnancy in patients with FH, especially after 24 weeks of gestation, when LDL-C and TG increase by approximately 30% and 100%, respectively 115) . It is known that patients with HoFH have increased coagulability, platelet function, and blood viscosity during pregnancy 116) that uteroplacental blood flow in HoFH during pregnancy is decreased compared to normal pregnancy, and that lipoprotein apheresis treatment improves blood flow 117) .ipoprotein apheresis should be performed during pregnancy due to the high stress on the cardiovascular system in the late pregnancy, especially during childbirth. Lipoprotein apheresis therapy can be administered safely during pregnancy, and there have already been reports of successful delivery 106 , 118 - 120) . Lipid-lowering drugs other than anion exchange resins should be discontinued during lactation, and regular lipoprotein apheresis therapy should be continued to ensure adequate LDL-C control.
Systematic Review Committee Member
Hirofumi Okada (Department of Cardiovascular Medicine, Kanazawa University Hospital), Masatsune Ogura (Department of Metabolism and Endocrinology, Eastern Chiba Medical Center/Department of General Medical Science, Chiba University Graduate School of Medicine), Yu Kataoka (Department of Cardiovascular Medicine, National Cerebral and Cardiovascular Center), Yoshihiro Tanaka (Department of Preventive Medicine, Northwestern University), Takahito Doi (Department of Clinical Biochemistry, Copenhagen University Hospital, Herlev and Gentofte/Department of Cardiovascular Medicine, National Cerebral and Cardiovascular Center), Tetsuo Nishikawa (Department of Transplant Medicine, National Cerebral and Cardiovascular Center), Akihiro Nomura (College of Transdisciplinary Sciences for Innovation, Department of Cardiovascular Medicine, Kanazawa University), and Masashi Yamamoto (Clinical Cell Biology and Medicine, Chiba University).
Classification of Level of Evidence for Treatment and Diagnosis
| 1+ | High-quality RCTs* and their MA/SR |
| 1 | Other RCTs and their MA/SR |
| 2 | Prospective cohort studies, their MA/SR, (predefined) RCT subanalysis |
| 3 | Non-randomized controlled trials, before-and-after studies, retrospective cohort studies, case-control studies, and their MA/SR and RCT post hoc subanalysis |
| 4 | Cross-sectional studies, case series |
| Consensus | By consensus of the supervisory committee members and the members who prepared the report |
RCT: randomized controlled trial, MA: meta-analysis, SR: systematic review
*A high-quality RCT is defined as (1) large number of subjects, (2) double-blind, independent assessment, (3) high follow-up rate (low dropout rate), (3) low protocol deviation, (4) clear random allocation method, etc.
Classification of Levels of Evidence for Epidemiological Studies
| E-1a | Meta-analysis of cohort studies |
| E-1b | Cohort studies |
| E-2 | Case-control studies, cross-sectional studies |
| E-3 | Descriptive studies (case series) |
Recommended Level
| A | Strongly Recommended |
| B | Weakly Recommended |
Conflict of Interest
The following discloses the conflicts of interest of the members of the Working Group of the Japan Atherosclerosis Society for the Development of Guidelines for the Treatment of Adult Familial Hypercholesterolemia for the years 2017 - 2021.
| Names | Disclosure item 1 | Disclosure item 2 | Disclosure item 3 | Disclosure item 4 | Disclosure item 5 | Disclosure item 6 |
|---|---|---|---|---|---|---|
| Disclosure item 7 | Disclosure item 8 | Disclosure item 9 | Disclosure item10 | Disclosure item 11 | Disclosure item 12 | |
| Arai H. | None | None | None | Amgen Astellas BioPharma K.K., Astellas Pharma Inc., Abbott Japan LLC, MSD K.K., Otsuka Pharmaceutical Co., Ltd.,Kowa Pharmaceutical Co. Ltd., Sanofi K.K., Daiichi Sankyo Company, Limited, Takeda Pharmaceutical Company Limited, Pfizer Inc. | None | None |
| Daiichi Sankyo Company, Limited | None | None | None | None | None | |
| Ohmura H. | None | None | None | None | None | None |
| None | None | None | None | None | None | |
| Okazaki H. | None | None | None | None | None | Kowa Company, Ltd., Tosoh Corporation, Minophagen Pharmaceutical Co., LTD |
| None | None | None | None | None | None | |
| Okada H. | None | None | None | None | None | None |
| None | None | None | None | None | None | |
| Ogura M. | None | None | None | Astellas Pharma Inc., Amgen K.K., Amgen Astellas BioPharma K.K., Sanofi K.K., Kowa Company, Ltd. | None | None |
| None | None | None | None | None | None | |
| Kataoka Y. | None | None | None | None | None | None |
| None | None | None | None | None | None | |
| Harada-Shiba M. | None | Liid Pharmaceuticals, Inc. | None | Astellas Pharma Inc., Amgen K.K., Amgen Astellas BioPharma K.K., Kaneka Medix Corporation, Sanofi K.K. | None | Aegerion Pharmaceuticals, Inc. , Parexel International Inc. |
| Aegerion Pharmaceuticals, Inc. , MSD K.K., Kaneka Medix Corporation, Kowa Pharmaceutical Co. Ltd., Sanofi K.K., Takeda Pharmaceutical Company Limited, Recordati Rare Diseases | None | None | None | None | None | |
| Sugiyama D. | Ajinomoto Frozen Foods Co., Inc. | None | None | None | None | None |
| None | None | None | None | None | None | |
| Tada H. | None | None | None | None | None | None |
| None | None | None | None | None | None | |
| Tanaka Y. | None | None | None | None | None | None |
|
American Heart Association (AHA), JAPAN LIFELINE Co., Ltd. |
None | None | None | None | None | |
| Doi T. | None | None | None | None | None | None |
| None | None | None | None | None | None | |
| Dobashi K. | None | None | None | None | None | None |
| None | None | None | None | None | None | |
| Nishikawa T. | None | None | None | None | None | None |
| None | None | None | None | None | None | |
| Nomura A. | CureApp, Inc. | None | None | None | None | None |
| None | None | None | None | None | None | |
| Matsuki K. | None | None | None | None | None | None |
| None | None | None | None | None | None | |
| Minamino T. | None | None | None | Daiichi Sankyo Company, Limited, Nippon Boehringer Ingelheim Co., Ltd. | None | A&D Company, Limited, OMRON Corporation, Matsutani Chemical Industry Co.,Ltd., Melody International Ltd. |
| Astellas Pharma Inc., Otsuka Pharmaceutical Co., Ltd.,Kyowa Hakko Kirin Co., Ltd., Kyowa Kirin Co., Ltd., Sanofi K.K., Takeda Pharmaceutical Company Limited, Mitsubishi Tanabe Pharma Corporation | None | None | None | None | None | |
| Yamashita S. | None | None | None |
Amgen K.K., MSD K.K., Otsuka Pharmaceutical Co., Ltd., Kowa Company, Ltd., Skylight Biotech,Inc., Novartis Pharma K.K., Bayer Yakuhin,Ltd,, Hayashibara Co., Ltd., East Japan Institute of Technology Co., Ltd. |
None | Medical Photonics Co.,Ltd. |
|
ONO PHARMACEUTICAL CO., LTD., Nippon Boehringer Ingelheim Co., Ltd., MSD K.K. |
None | None | None | None | None | |
| Yamamoto M. | None | None | None | None | None | None |
| None | None | None | None | None | None | |
| Yokote K. | None | None | None |
Astellas Pharma Inc., Amgen Astellas BioPharma K.K., AstraZeneca K.K., MSD K.K., ONO PHARMACEUTICAL CO., LTD., Kyowa Hakko Kirin Co., Ltd., Kowa Company, Ltd., Kowa Pharmaceutical Co. Ltd., Sanofi K.K., Daiichi Sankyo Company, Limited, Taisho Pharmaceutical Co., Ltd., Taisho Toyama Pharmaceutical Co., Ltd., Sumitomo Dainippon Pharma Co., Ltd., Takeda Pharmaceutical Company Limited, Mitsubishi Tanabe Pharma Corporation, Eli Lilly Japan K.K., Nippon Boehringer Ingelheim Co., Ltd., Novartis Pharma K.K., Novo Nordisk Pharma Ltd., Pfizer Inc., Janssen Pharmaceutical K.K. |
None | Astellas Pharma Inc., Taisho Pharmaceutical Co., Ltd., Taisho Toyama Pharmaceutical Co., Ltd. |
|
Astellas Pharma Inc., Abbott Japan LLC, MSD K.K., ONO PHARMACEUTICAL CO., LTD., Kao Corporation, Kowa Company, Ltd., Kowa Pharmaceutical Co. Ltd., SHIONOGI & Co., Ltd., Daiichi Sankyo Company, Limited, Taisho Pharmaceutical Co., Ltd., Taisho Toyama Pharmaceutical Co., Ltd., Sumitomo Dainippon Pharma Co., Ltd., Takeda Pharmaceutical Company Limited, Mitsubishi Tanabe Pharma Corporation, TEIJIN PHARMA LIMITED, Eli Lilly Japan K.K., Nippon Boehringer Ingelheim Co., Ltd., Novo Nordisk Pharma Ltd., Bayer Yakuhin,Ltd,, Pfizer Inc. |
None | None | None | None | None |
【Disclosure Item】
1. Assuming a position of a board member or advisor in a profit-making business,/ Advisory role (1,000,000 yen or more annual compensation from a single business entity, or group)
2. Stock holdings or options (Annual profit of 1,000,000 yen or more/ownership of 5% or more of total shares)
3. Patent royalties/licensing fees (1,000,000 yen or more annual income per patent)
4. Honoraria (e.g. lecture fees) (500,000 yen or more total annual income from a single company or organization)
5. Fees for promotional materials (e.g. manuscript fee) (500,000 yen or more total annual income from a single company or organization)
6. Total clinical research funding (1,000,000 yen or more total annual research grants paid from a single company or organization to you or your department)
7. Total scholarship grants (1,000,000 yen or more total annual scholarship contributed by a single company or organization to you or your department)
8. Courses endowed by companies (Fill in if you belong to any course endowed by a company, etc.)
9. Others (e.g. trips, travel, or gifts, which are not related to research) (50,000 yen or more annually from a single company or organization)
Matters declared by the author’s spouse, relatives within the first degree of consanguinity, or persons who share income or property
10. Assuming a position of a board member or advisor in a profit-making business,/ Advisory role (1,000,000 yen or more annual compensation from a single business entity, or group)
11. Stock holdings or options (Annual profit of 1,000,000 yen or more/ownership of 5% or more of total shares)
12. Patent royalties/licensing fees (1,000,000 yen or more annual income per patent)
*The company name is the name of the company as it was at the time of filing.
References
- 1).Tada H, Hori M, Matsuki K, Ogura M, Nohara A, Kawashiri MA, Harada-Shiba M: Achilles Tendon Thickness Assessed by X-ray Predicting a Pathogenic Mutation in Familial Hypercholesterolemia Gene. J Atheroscler Thromb, 2022; 29: 816-824 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2).Harada-Shiba M, Ohtake A, Sugiyama D, Tada H, Dobashi K, Matsuki K, Minamino T, Yamashita S, Yamamoto Y: Guidelines for the Diagnosis and Treatment of Pediatric Familial Hypercholesterolemia 2022. J Atheroscler Thromb, 2023; 30: 531-557 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3).Mabuchi H, Nohara A, Noguchi T, Kobayashi J, Kawashiri MA, Tada H, Nakanishi C, Mori M, Yamagishi M, Inazu A, Koizumi J: Molecular genetic epidemiology of homozygous familial hypercholesterolemia in the Hokuriku district of Japan. Atherosclerosis, 2011; 214: 404-407 [DOI] [PubMed] [Google Scholar]
- 4).Akioyamen LE, Genest J, Shan SD, Reel RL, Albaum JM, Chu A, Tu JV: Estimating the prevalence of heterozygous familial hypercholesterolaemia: a systematic review and meta-analysis. BMJ Open, 2017; 7: e016461 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5).Beheshti SO, Madsen CM, Varbo A, Nordestgaard BG: Worldwide Prevalence of Familial Hypercholesterolemia: Meta-Analyses of 11 Million Subjects. J Am Coll Cardiol, 2020; 75: 2553-2566 [DOI] [PubMed] [Google Scholar]
- 6).Hu P, Dharmayat KI, Stevens CAT, Sharabiani MTA, Jones RS, Watts GF, Genest J, Ray KK, Vallejo-Vaz AJ: Prevalence of Familial Hypercholesterolemia Among the General Population and Patients With Atherosclerotic Cardiovascular Disease: A Systematic Review and Meta-Analysis. Circulation, 2020; 141: 1742-1759 [DOI] [PubMed] [Google Scholar]
- 7).Austin MA, Hutter CM, Zimmern RL, Humphries SE: Familial hypercholesterolemia and coronary heart disease: a HuGE association review. Am J Epidemiol, 2004; 160: 421-429 [DOI] [PubMed] [Google Scholar]
- 8).Hutter CM, Austin MA, Humphries SE: Familial hypercholesterolemia, peripheral arterial disease, and stroke: a HuGE minireview. Am J Epidemiol, 2004; 160: 430-435 [DOI] [PubMed] [Google Scholar]
- 9).Akioyamen LE, Tu JV, Genest J, Ko DT, Coutin AJS, Shan SD, Chu A: Risk of Ischemic Stroke and Peripheral Arterial Disease in Heterozygous Familial Hypercholesterolemia: A Meta-Analysis. Angiology, 2019; 70: 726-736 [DOI] [PubMed] [Google Scholar]
- 10).Mabuchi H: Half a Century Tales of Familial Hypercholesterolemia (FH) in Japan. J Atheroscler Thromb, 2017; 24: 189-207 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11).Betteridge DJ, Bhatnager D, Bing RF, Durrington PN, Evans GR, Flax H, Jay RH, Lewis-Barned N, Mann J, Matthews DR.: Treatment of familial hypercholesterolaemia. United Kingdom lipid clinics study of pravastatin and cholestyramine. BMJ, 1992; 304: 1335-1338 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12).Hoogerbrugge N, Mol MJ, Van Dormaal JJ, Rustemeijer C, Muls E, Stalenhoef AF, Birkenhäger JC: The efficacy and safety of pravastatin, compared to and in combination with bile acid binding resins, in familial hypercholesterolaemia. J Intern Med, 1990; 228: 261-266 [DOI] [PubMed] [Google Scholar]
- 13).Wiklund O, Angelin B, Fager G, Eriksson M, Olofsson SO, Berglund L, Lindén T, Sjöberg A, Bondjers G: Treatment of familial hypercholesterolaemia: a controlled trial of the effects of pravastatin or cholestyramine therapy on lipoprotein and apolipoprotein levels. J Intern Med, 1990; 228: 241-247 [DOI] [PubMed] [Google Scholar]
- 14).Avis HJ, Hutten BA, Gagné C, Langslet G, McCrindle BW, Wiegman A, Hsia J, Kastelein JJ, Stein EA: Efficacy and safety of rosuvastatin therapy for children with familial hypercholesterolemia. J Am Coll Cardiol, 2010; 55: 1121-1126 [DOI] [PubMed] [Google Scholar]
- 15).Clauss SB, Holmes KW, Hopkins P, Stein E, Cho M, Tate A, Johnson-Levonas AO, Kwiterovich PO: Efficacy and safety of lovastatin therapy in adolescent girls with heterozygous familial hypercholesterolemia. Pediatrics, 2005; 116: 682-688 [DOI] [PubMed] [Google Scholar]
- 16).de Jongh S, Ose L, Szamosi T, Gagné C, Lambert M, Scott R, Perron P, Dobbelaere D, Saborio M, Tuohy MB, Stepanavage M, Sapre A, Gumbiner B, Mercuri M, van Trotsenburg AS, Bakker HD, Kastelein JJ: Efficacy and safety of statin therapy in children with familial hypercholesterolemia: a randomized, double-blind, placebo-controlled trial with simvastatin. Circulation, 2002; 106: 2231-2237 [DOI] [PubMed] [Google Scholar]
- 17).Knipscheer HC, Boelen CC, Kastelein JJ, van Diermen DE, Groenemeijer BE, van den Ende A, Büller HR, Bakker HD: Short-term efficacy and safety of pravastatin in 72 children with familial hypercholesterolemia. Pediatr Res, 1996; 39: 867-871 [DOI] [PubMed] [Google Scholar]
- 18).Stein EA, Illingworth DR, Kwiterovich PO, Jr., Liacouras CA, Siimes MA, Jacobson MS, Brewster TG, Hopkins P, Davidson M, Graham K, Arensman F, Knopp RH, DuJovne C, Williams CL, Isaacsohn JL, Jacobsen CA, Laskarzewski PM, Ames S, Gormley GJ: Efficacy and safety of lovastatin in adolescent males with heterozygous familial hypercholesterolemia: a randomized controlled trial. JAMA, 1999; 281: 137-144 [DOI] [PubMed] [Google Scholar]
- 19).Wiegman A, Hutten BA, de Groot E, Rodenburg J, Bakker HD, Büller HR, Sijbrands EJ, Kastelein JJ: Efficacy and safety of statin therapy in children with familial hypercholesterolemia: a randomized controlled trial. JAMA, 2004; 292: 331-337 [DOI] [PubMed] [Google Scholar]
- 20).Stein EA, Dann EJ, Wiegman A, Skovby F, Gaudet D, Sokal E, Charng MJ, Mohamed M, Luirink I, Raichlen JS, Sundén M, Carlsson SC, Raal FJ, Kastelein JJP: Efficacy of Rosuvastatin in Children With Homozygous Familial Hypercholesterolemia and Association With Underlying Genetic Mutations. J Am Coll Cardiol, 2017; 70: 1162-1170 [DOI] [PubMed] [Google Scholar]
- 21).Lambert M, Lupien PJ, Gagné C, Lévy E, Blaichman S, Langlois S, Hayden M, Rose V, Clarke JT, Wolfe BM, Clarson C, Parsons H, Stephure DK, Potvin D, Lambert J: Treatment of familial hypercholesterolemia in children and adolescents: effect of lovastatin. Canadian Lovastatin in Children Study Group. Pediatrics, 1996; 97: 619-628 [PubMed] [Google Scholar]
- 22).Harada-Shiba M, Arisaka O, Ohtake A, Okada T, Suganami H: Efficacy and Safety of Pitavastatin in Japanese Male Children with Familial Hypercholesterolemia. J Atheroscler Thromb, 2016; 23: 48-55 [DOI] [PubMed] [Google Scholar]
- 23).Nozue T, Michishita I, Ito Y, Hirano T: Effects of statin on small dense low-density lipoprotein cholesterol and remnant-like particle cholesterol in heterozygous familial hypercholesterolemia. J Atheroscler Thromb, 2008; 15: 146-153 [DOI] [PubMed] [Google Scholar]
- 24).Smilde TJ, van Wissen S, Wollersheim H, Trip MD, Kastelein JJ, Stalenhoef AF: Effect of aggressive versus conventional lipid lowering on atherosclerosis progression in familial hypercholesterolaemia (ASAP): a prospective, randomised, double-blind trial. Lancet, 2001; 357: 577-581 [DOI] [PubMed] [Google Scholar]
- 25).Rodenburg J, Vissers MN, Wiegman A, van Trotsenburg AS, van der Graaf A, de Groot E, Wijburg FA, Kastelein JJ, Hutten BA: Statin treatment in children with familial hypercholesterolemia: the younger, the better. Circulation, 2007; 116: 664-668 [DOI] [PubMed] [Google Scholar]
- 26).Besseling J, Hovingh GK, Huijgen R, Kastelein JJP, Hutten BA: Statins in Familial Hypercholesterolemia: Consequences for Coronary Artery Disease and All-Cause Mortality. J Am Coll Cardiol, 2016; 68: 252-260 [DOI] [PubMed] [Google Scholar]
- 27).Jay RH, Sturley RH, Stirling C, McGarrigle HH, Katz M, Reckless JP, Betteridge DJ: Effects of pravastatin and cholestyramine on gonadal and adrenal steroid production in familial hypercholesterolaemia. Br J Clin Pharmacol, 1991; 32: 417-422 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28).Raal FJ, Stein EA, Dufour R, Turner T, Civeira F, Burgess L, Langslet G, Scott R, Olsson AG, Sullivan D, Hovingh GK, Cariou B, Gouni-Berthold I, Somaratne R, Bridges I, Scott R, Wasserman SM, Gaudet D: PCSK9 inhibition with evolocumab (AMG 145) in heterozygous familial hypercholesterolaemia (RUTHERFORD-2): a randomised, double-blind, placebo-controlled trial. Lancet, 2015; 385: 331-340 [DOI] [PubMed] [Google Scholar]
- 29).Kastelein JJ, Ginsberg HN, Langslet G, Hovingh GK, Ceska R, Dufour R, Blom D, Civeira F, Krempf M, Lorenzato C, Zhao J, Pordy R, Baccara-Dinet MT, Gipe DA, Geiger MJ, Farnier M: ODYSSEY FH I and FH II: 78 week results with alirocumab treatment in 735 patients with heterozygous familial hypercholesterolaemia. Eur Heart J, 2015; 36: 2996-3003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30).Raal FJ, Honarpour N, Blom DJ, Hovingh GK, Xu F, Scott R, Wasserman SM, Stein EA: Inhibition of PCSK9 with evolocumab in homozygous familial hypercholesterolaemia (TESLA Part B): a randomised, double-blind, placebo-controlled trial. Lancet, 2015; 385: 341-350 [DOI] [PubMed] [Google Scholar]
- 31).Kastelein JJ, Akdim F, Stroes ES, Zwinderman AH, Bots ML, Stalenhoef AF, Visseren FL, Sijbrands EJ, Trip MD, Stein EA, Gaudet D, Duivenvoorden R, Veltri EP, Marais AD, de Groot E: Simvastatin with or without ezetimibe in familial hypercholesterolemia. N Engl J Med, 2008; 358: 1431-1443 [DOI] [PubMed] [Google Scholar]
- 32).Authors/Task Force Members; ESC Committee forPractice Guidelines (CPG); ESC National Cardiac Societies: 2019 ESC/EAS guidelines for the management of dyslipidaemias: Lipid modification to reduce cardiovascular risk. Atherosclerosis, 2019; 290: 140-205 [DOI] [PubMed] [Google Scholar]
- 33).Wang A, Richhariya A, Gandra SR, Calimlim B, Kim L, Quek RG, Nordyke RJ, Toth PP: Systematic Review of Low-Density Lipoprotein Cholesterol Apheresis for the Treatment of Familial Hypercholesterolemia. J Am Heart Assoc, 2016; 5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34).Luirink IK, Determeijer J, Hutten BA, Wiegman A, Bruckert E, Schmitt CP, Groothoff JW: Efficacy and safety of lipoprotein apheresis in children with homozygous familial hypercholesterolemia: A systematic review. J Clin Lipidol, 2019; 13: 31-39 [DOI] [PubMed] [Google Scholar]
- 35).Mabuchi H, Koizumi J, Shimizu M, Takeda R: Development of coronary heart disease in familial hypercholesterolemia. Circulation, 1989; 79: 225-232 [DOI] [PubMed] [Google Scholar]
- 36).Nordestgaard BG, Chapman MJ, Humphries SE, Ginsberg HN, Masana L, Descamps OS, Wiklund O, Hegele RA, Raal FJ, Defesche JC, Wiegman A, Santos RD, Watts GF, Parhofer KG, Hovingh GK, Kovanen PT, Boileau C, Averna M, Borén J, Bruckert E, Catapano AL, Kuivenhoven JA, Pajukanta P, Ray K, Stalenhoef AF, Stroes E, Taskinen MR, Tybjærg-Hansen A: Familial hypercholesterolaemia is underdiagnosed and undertreated in the general population: guidance for clinicians to prevent coronary heart disease: consensus statement of the European Atherosclerosis Society. Eur Heart J, 2013; 34: 3478-3490a [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37).Ohta N, Hori M, Takahashi A, Ogura M, Makino H, Tamanaha T, Fujiyama H, Miyamoto Y, Harada-Shiba M: Proprotein convertase subtilisin/kexin 9 V4I variant with LDLR mutations modifies the phenotype of familial hypercholesterolemia. J Clin Lipidol, 2016; 10: 547-555.e545 [DOI] [PubMed] [Google Scholar]
- 38).Talmud PJ, Shah S, Whittall R, Futema M, Howard P, Cooper JA, Harrison SC, Li K, Drenos F, Karpe F, Neil HA, Descamps OS, Langenberg C, Lench N, Kivimaki M, Whittaker J, Hingorani AD, Kumari M, Humphries SE: Use of low-density lipoprotein cholesterol gene score to distinguish patients with polygenic and monogenic familial hypercholesterolaemia: a case-control study. Lancet, 2013; 381: 1293-1301 [DOI] [PubMed] [Google Scholar]
- 39).Tada H, Hori M, Nomura A, Hosomichi K, Nohara A, Kawashiri MA, Harada-Shiba M: A catalog of the pathogenic mutations of LDL receptor gene in Japanese familial hypercholesterolemia. J Clin Lipidol, 2020; 14: 346-351.e349 [DOI] [PubMed] [Google Scholar]
- 40).Hori M, Takahashi A, Son C, Ogura M, Harada-Shiba M: The first Japanese cases of familial hypercholesterolemia due to a known pathogenic APOB gene variant, c.10580 G>A: p.(Arg3527Gln). J Clin Lipidol, 2020; 14: 482-486 [DOI] [PubMed] [Google Scholar]
- 41).Andersen LH, Miserez AR, Ahmad Z, Andersen RL: Familial defective apolipoprotein B-100: A review. J Clin Lipidol, 2016; 10: 1297-1302 [DOI] [PubMed] [Google Scholar]
- 42).Abifadel M, Varret M, Rabès JP, Allard D, Ouguerram K, Devillers M, Cruaud C, Benjannet S, Wickham L, Erlich D, Derré A, Villéger L, Farnier M, Beucler I, Bruckert E, Chambaz J, Chanu B, Lecerf JM, Luc G, Moulin P, Weissenbach J, Prat A, Krempf M, Junien C, Seidah NG, Boileau C: Mutations in PCSK9 cause autosomal dominant hypercholesterolemia. Nat Genet, 2003; 34: 154-156 [DOI] [PubMed] [Google Scholar]
- 43).Cohen J, Pertsemlidis A, Kotowski IK, Graham R, Garcia CK, Hobbs HH: Low LDL cholesterol in individuals of African descent resulting from frequent nonsense mutations in PCSK9. Nat Genet, 2005; 37: 161-165 [DOI] [PubMed] [Google Scholar]
- 44).Noguchi T, Katsuda S, Kawashiri MA, Tada H, Nohara A, Inazu A, Yamagishi M, Kobayashi J, Mabuchi H: The E32K variant of PCSK9 exacerbates the phenotype of familial hypercholesterolaemia by increasing PCSK9 function and concentration in the circulation. Atherosclerosis, 2010; 210: 166-172 [DOI] [PubMed] [Google Scholar]
- 45).Harada-Shiba M, Takagi A, Miyamoto Y, Tsushima M, Ikeda Y, Yokoyama S, Yamamoto A: Clinical features and genetic analysis of autosomal recessive hypercholesterolemia. J Clin Endocrinol Metab, 2003; 88: 2541-2547 [DOI] [PubMed] [Google Scholar]
- 46).D’Erasmo L, Minicocci I, Nicolucci A, Pintus P, Roeters Van Lennep JE, Masana L, Mata P, Sánchez-Hernández RM, Prieto-Matos P, Real JT, Ascaso JF, Lafuente EE, Pocovi M, Fuentes FJ, Muntoni S, Bertolini S, Sirtori C, Calabresi L, Pavanello C, Averna M, Cefalu AB, Noto D, Pacifico AA, Pes GM, Harada-Shiba M, Manzato E, Zambon S, Zambon A, Vogt A, Scardapane M, Sjouke B, Fellin R, Arca M: Autosomal Recessive Hypercholesterolemia: Long-Term Cardiovascular Outcomes. J Am Coll Cardiol, 2018; 71: 279-288 [DOI] [PubMed] [Google Scholar]
- 47). Tada N, Yoshida H, Teramoto T,Oikawa S,Saitoh Y, Yamada N, Shirai K,Ishibashi S, Ishikawa T, Yoshino G,Hirano T: Survey of hyperlipidemia treatment in Japan (Survey 1: Complications and drug of choice in hyper LDL-C patients and familial hypercholesterolemia) - LiMAP1. 37th Annual Meeting of the Japan Atherosclerosis Society, 2005 in Japanese [Google Scholar]
- 48).Bujo H, Takahashi K, Saito Y, Maruyama T, Yamashita S, Matsuzawa Y, Ishibashi S, Shionoiri F, Yamada N, Kita T: Clinical features of familial hypercholesterolemia in Japan in a database from 1996-1998 by the research committee of the ministry of health, labour and welfare of Japan. J Atheroscler Thromb, 2004; 11: 146-151 [DOI] [PubMed] [Google Scholar]
- 49).Mabuchi H: Introduction to Hyperlipidemia, Bunkodo, 2005 [Google Scholar]
- 50).Chaves FJ, Real JT, García-García AB, Civera M, Armengod ME, Ascaso JF, Carmena R: Genetic diagnosis of familial hypercholesterolemia in a South European outbreed population: influence of low-density lipoprotein (LDL) receptor gene mutations on treatment response to simvastatin in total, LDL, and high-density lipoprotein cholesterol. J Clin Endocrinol Metab, 2001; 86: 4926-4932 [DOI] [PubMed] [Google Scholar]
- 51).Neil HA, Hawkins MM, Durrington PN, Betteridge DJ, Capps NE, Humphries SE: Non-coronary heart disease mortality and risk of fatal cancer in patients with treated heterozygous familial hypercholesterolaemia: a prospective registry study. Atherosclerosis, 2005; 179: 293-297 [DOI] [PubMed] [Google Scholar]
- 52).Tada H, Kawashiri MA, Okada H, Teramoto R, Konno T, Yoshimuta T, Sakata K, Nohara A, Inazu A, Kobayashi J, Mabuchi H, Yamagishi M, Hayashi K: Assessment of coronary atherosclerosis in patients with familial hypercholesterolemia by coronary computed tomography angiography. Am J Cardiol, 2015; 115: 724-729 [DOI] [PubMed] [Google Scholar]
- 53).Harada-Shiba M, Sugisawa T, Makino H, Abe M, Tsushima M, Yoshimasa Y, Yamashita T, Miyamoto Y, Yamamoto A, Tomoike H, Yokoyama S: Impact of statin treatment on the clinical fate of heterozygous familial hypercholesterolemia. J Atheroscler Thromb, 2010; 17: 667-674 [DOI] [PubMed] [Google Scholar]
- 54).Tada H, Kawashiri MA, Nohara A, Inazu A, Mabuchi H, Yamagishi M: Impact of clinical signs and genetic diagnosis of familial hypercholesterolaemia on the prevalence of coronary artery disease in patients with severe hypercholesterolaemia. Eur Heart J, 2017; 38: 1573-1579 [DOI] [PubMed] [Google Scholar]
- 55).Ogura M, Hori M, Harada-Shiba M: Association Between Cholesterol Efflux Capacity and Atherosclerotic Cardiovascular Disease in Patients With Familial Hypercholesterolemia. Arterioscler Thromb Vasc Biol, 2016; 36: 181-188 [DOI] [PubMed] [Google Scholar]
- 56).Yagi K, Hifumi S, Nohara A, Higashikata T, Inazu A, Mizuno KO, Namura M, Ueda K, Kobayashi J, Shimizu M, Mabuchi H: Difference in the risk factors for coronary, renal and other peripheral arteriosclerosis in heterozygous familial hypercholesterolemia. Circ J, 2004; 68: 623-627 [DOI] [PubMed] [Google Scholar]
- 57).Hirobe K, Matsuzawa Y, Ishikawa K, Tarui S, Yamamoto A, Nambu S, Fujimoto K: Coronary artery disease in heterozygous familial hypercholesterolemia. Atherosclerosis, 1982; 44: 201-210 [DOI] [PubMed] [Google Scholar]
- 58).Yanagi K, Yamashita S, Kihara S, Nakamura T, Nozaki S, Nagai Y, Funahashi T, Kameda-Takemura K, Ueyama Y, Jiao S, Kubo M, Tokunaga K, Matsuzawa Y: Characteristics of coronary artery disease and lipoprotein abnormalities in patients with heterozygous familial hypercholesterolemia associated with diabetes mellitus or impaired glucose tolerance. Atherosclerosis, 1997; 132: 43-51 [DOI] [PubMed] [Google Scholar]
- 59).Nakamura T, Kobayashi H, Yanagi K, Nakagawa T, Nishida M, Kihara S, Hiraoka H, Nozaki S, Funahashi T, Yamashita S, Kameda-Takemura K, Matsuzawa Y: Importance of intra-abdominal visceral fat accumulation to coronary atherosclerosis in heterozygous familial hypercholesterolaemia. Int J Obes Relat Metab Disord, 1997; 21: 580-586 [DOI] [PubMed] [Google Scholar]
- 60).Tarui S: Primary Hyperlipidemia Research Group, FY 1986 Research Report (Ministry of Health and Welfare specific diseases in 1986), 1987 in Japanese [Google Scholar]
- 61).Ogura M, Harada-Shiba M, Masuda D, Arai H, Bujo H, Ishibashi S, Daida H, Koga N, Oikawa S, Yamashita S: Factors Associated with Carotid Atherosclerosis and Achilles Tendon Thickness in Japanese Patients with Familial Hypercholesterolemia: A Subanalysis of the Familial Hypercholesterolemia Expert Forum (FAME) Study. J Atheroscler Thromb, 2022; 29: 906-922 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62).Tada H, Kawashiri MA, Nohara A, Inazu A, Mabuchi H, Yamagishi M: Assessment of arterial stiffness in patients with familial hypercholesterolemia. J Clin Lipidol, 2018; 12: 397-402.e392 [DOI] [PubMed] [Google Scholar]
- 63).Jensen HK, Hansen PS, Jensen LG, Kristensen MJ, Klausen IC, Kjeldsen M, Lemming L, Bolund L, Gregersen N, Faergeman O: Complexity of molecular genetics of dyslipidemia in a family highly susceptible to ischemic heart disease. Clin Genet, 1995; 48: 23-28 [DOI] [PubMed] [Google Scholar]
- 64).Holmes DT, Schick BA, Humphries KH, Frohlich J: Lipoprotein(a) is an independent risk factor for cardiovascular disease in heterozygous familial hypercholesterolemia. Clin Chem, 2005; 51: 2067-2073 [DOI] [PubMed] [Google Scholar]
- 65).Nenseter MS, Lindvig HW, Ueland T, Langslet G, Ose L, Holven KB, Retterstøl K: Lipoprotein(a) levels in coronary heart disease-susceptible and -resistant patients with familial hypercholesterolemia. Atherosclerosis, 2011; 216: 426-432 [DOI] [PubMed] [Google Scholar]
- 66).Chan DC, Pang J, Hooper AJ, Burnett JR, Bell DA, Bates TR, van Bockxmeer FM, Watts GF: Elevated lipoprotein(a), hypertension and renal insufficiency as predictors of coronary artery disease in patients with genetically confirmed heterozygous familial hypercholesterolemia. Int J Cardiol, 2015; 201: 633-638 [DOI] [PubMed] [Google Scholar]
- 67).Khera AV, Won HH, Peloso GM, Lawson KS, Bartz TM, Deng X, van Leeuwen EM, Natarajan P, Emdin CA, Bick AG, Morrison AC, Brody JA, Gupta N, Nomura A, Kessler T, Duga S, Bis JC, van Duijn CM, Cupples LA, Psaty B, Rader DJ, Danesh J, Schunkert H, McPherson R, Farrall M, Watkins H, Lander E, Wilson JG, Correa A, Boerwinkle E, Merlini PA, Ardissino D, Saleheen D, Gabriel S, Kathiresan S: Diagnostic Yield and Clinical Utility of Sequencing Familial Hypercholesterolemia Genes in Patients With Severe Hypercholesterolemia. J Am Coll Cardiol, 2016; 67: 2578-2589 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68).Tada H, Kawashiri MA, Nomura A, Teramoto R, Hosomichi K, Nohara A, Inazu A, Mabuchi H, Tajima A, Yamagishi M: Oligogenic familial hypercholesterolemia, LDL cholesterol, and coronary artery disease. J Clin Lipidol, 2018; 12: 1436-1444 [DOI] [PubMed] [Google Scholar]
- 69).Doi T, Hori M, Harada-Shiba M, Kataoka Y, Onozuka D, Nishimura K, Nishikawa R, Tsuda K, Ogura M, Son C, Miyamoto Y, Noguchi T, Shimokawa H, Yasuda S: Patients With LDLR and PCSK9 Gene Variants Experienced Higher Incidence of Cardiovascular Outcomes in Heterozygous Familial Hypercholesterolemia. J Am Heart Assoc, 2021; 10: e018263 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70).Michikura M, Ogura M, Yamamoto M, Sekimoto M, Fuke C, Hori M, Arai K, Kihara S, Hosoda K, Yanagi K, Harada-Shiba M: Achilles Tendon Ultrasonography for Diagnosis of Familial Hypercholesterolemia Among Japanese Subjects. Circ J, 2017; 81: 1879-1885 [DOI] [PubMed] [Google Scholar]
- 71).Bilheimer DW, Ho YK, Brown MS, Anderson RG, Goldstein JL: Genetics of the low density lipoprotein receptor. Diminished receptor activity in lymphocytes from heterozygotes with familial hypercholesterolemia. J Clin Invest, 1978; 61: 678-696 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72).Rosenson RS: Myocardial injury: the acute phase response and lipoprotein metabolism. J Am Coll Cardiol, 1993; 22: 933-940 [DOI] [PubMed] [Google Scholar]
- 73).The Japanese Association of Medical Sciences: Guidelines for Genetic Testing and Diagnosis in Health Care, https://jams.med.or.jp/guideline/genetics-diagnosis.pdf, 2011 in Japanese [Google Scholar]
- 74).Ministry of Education, Culture, Sports, Science and Technology, Ministry of Health, Labor and Welfare, Ministry of Economy, Trade and Industry: Ethical guidelines for life science and medical research involving human subjects, https://www.meti.go.jp/press/2020/03/20210323004/20210323004-1.pdf, 2021 in Japanese [Google Scholar]
- 75).Hubacek JA, Berge KE, Cohen JC, Hobbs HH: Mutations in ATP-cassette binding proteins G5 (ABCG5) and G8 (ABCG8) causing sitosterolemia. Hum Mutat, 2001; 18: 359-360 [DOI] [PubMed] [Google Scholar]
- 76).Tada H, Nohara A, Inazu A, Sakuma N, Mabuchi H, Kawashiri MA: Sitosterolemia, Hypercholesterolemia, and Coronary Artery Disease. J Atheroscler Thromb, 2018; 25: 783-789 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77).Tada H, Nomura A, Ogura M, Ikewaki K, Ishigaki Y, Inagaki K, Tsukamoto K, Dobashi K, Nakamura K, Hori M, Matsuki K, Yamashita S, Yokoyama S, Kawashiri MA, Harada-Shiba M: Diagnosis and Management of Sitosterolemia 2021. J Atheroscler Thromb, 2021; 28: 791-801 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78).Morganroth J, Levy RI, McMahon AE, Gotto AM, Jr.: Pseudohomozygous type II hyperlipoproteinemia. J Pediatr, 1974; 85: 639-643 [DOI] [PubMed] [Google Scholar]
- 79).Fujita M, Okamoto S, Shirai K, Saito Y, Yoshida S: Pseudohomozygous type II hyperlipoproteinemia. Dermatologica, 1991; 182: 94-97 [DOI] [PubMed] [Google Scholar]
- 80).Cali JJ, Hsieh CL, Francke U, Russell DW: Mutations in the bile acid biosynthetic enzyme sterol 27-hydroxylase underlie cerebrotendinous xanthomatosis. J Biol Chem, 1991; 266: 7779-7783 [PMC free article] [PubMed] [Google Scholar]
- 81).Koyama S, Sekijima Y, Ogura M, Hori M, Matsuki K, Miida T, Harada-Shiba M: Cerebrotendinous Xanthomatosis: Molecular Pathogenesis, Clinical Spectrum, Diagnosis, and Disease-Modifying Treatments. J Atheroscler Thromb, 2021; 28: 905-925 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82).Luirink IK, Wiegman A, Kusters DM, Hof MH, Groothoff JW, de Groot E, Kastelein JJP, Hutten BA: 20-Year Follow-up of Statins in Children with Familial Hypercholesterolemia. N Engl J Med, 2019; 381: 1547-1556 [DOI] [PubMed] [Google Scholar]
- 83).Yamashita S, Masuda D, Harada-Shiba M, Arai H, Bujo H, Ishibashi S, Daida H, Koga N, Oikawa S: Effectiveness and Safety of Lipid-Lowering Drug Treatments in Japanese Patients with Familial Hypercholesterolemia: Familial Hypercholesterolemia Expert Forum (FAME) Study. J Atheroscler Thromb, 2022; 29: 608-638 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84).Stroes ES, Thompson PD, Corsini A, Vladutiu GD, Raal FJ, Ray KK, Roden M, Stein E, Tokgözoğlu L, Nordestgaard BG, Bruckert E, De Backer G, Krauss RM, Laufs U, Santos RD, Hegele RA, Hovingh GK, Leiter LA, Mach F, März W, Newman CB, Wiklund O, Jacobson TA, Catapano AL, Chapman MJ, Ginsberg HN: Statin-associated muscle symptoms: impact on statin therapy-European Atherosclerosis Society Consensus Panel Statement on Assessment, Aetiology and Management. Eur Heart J, 2015; 36: 1012-1022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85).Kawashiri MA, Nohara A, Noguchi T, Tada H, Nakanishi C, Mori M, Konno T, Hayashi K, Fujino N, Inazu A, Kobayashi J, Mabuchi H, Yamagishi M: Efficacy and safety of coadministration of rosuvastatin, ezetimibe, and colestimide in heterozygous familial hypercholesterolemia. Am J Cardiol, 2012; 109: 364-369 [DOI] [PubMed] [Google Scholar]
- 86).Lipinski MJ, Benedetto U, Escarcega RO, Biondi-Zoccai G, Lhermusier T, Baker NC, Torguson R, Brewer HB, Jr., Waksman R: The impact of proprotein convertase subtilisin-kexin type 9 serine protease inhibitors on lipid levels and outcomes in patients with primary hypercholesterolaemia: a network meta-analysis. Eur Heart J, 2016; 37: 536-545 [DOI] [PubMed] [Google Scholar]
- 87).Dufour R, Bergeron J, Gaudet D, Weiss R, Hovingh GK, Qing Z, Yang F, Andisik M, Torri A, Pordy R, Gipe DA: Open-label therapy with alirocumab in patients with heterozygous familial hypercholesterolemia: Results from three years of treatment. Int J Cardiol, 2017; 228: 754-760 [DOI] [PubMed] [Google Scholar]
- 88).Farnier M, Hovingh GK, Langslet G, Dufour R, Baccara-Dinet MT, Din-Bell C, Manvelian G, Guyton JR: Long-term safety and efficacy of alirocumab in patients with heterozygous familial hypercholesterolemia: An open-label extension of the ODYSSEY program. Atherosclerosis, 2018; 278: 307-314 [DOI] [PubMed] [Google Scholar]
- 89).Santos RD, Stein EA, Hovingh GK, Blom DJ, Soran H, Watts GF, López JAG, Bray S, Kurtz CE, Hamer AW, Raal FJ: Long-Term Evolocumab in Patients With Familial Hypercholesterolemia. J Am Coll Cardiol, 2020; 75: 565-574 [DOI] [PubMed] [Google Scholar]
- 90).Qian LJ, Gao Y, Zhang YM, Chu M, Yao J, Xu D: Therapeutic efficacy and safety of PCSK9-monoclonal antibodies on familial hypercholesterolemia and statin-intolerant patients: A meta-analysis of 15 randomized controlled trials. Sci Rep, 2017; 7: 238 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91).Yamashita S, Hbujo H, Arai H, Harada-Shiba M, Matsui S, Fukushima M, Saito Y, Kita T, Matsuzawa Y: Long-term probucol treatment prevents secondary cardiovascular events: a cohort study of patients with heterozygous familial hypercholesterolemia in Japan. J Atheroscler Thromb, 2008; 15: 292-303 [DOI] [PubMed] [Google Scholar]
- 92).Moriarty PM, Parhofer KG, Babirak SP, Cornier MA, Duell PB, Hohenstein B, Leebmann J, Ramlow W, Schettler V, Simha V, Steinhagen-Thiessen E, Thompson PD, Vogt A, von Stritzky B, Du Y, Manvelian G: Alirocumab in patients with heterozygous familial hypercholesterolaemia undergoing lipoprotein apheresis: the ODYSSEY ESCAPE trial. Eur Heart J, 2016; 37: 3588-3595 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93).Nohara A, Tada H, Ogura M, Okazaki S, Ono K, Shimano H, Daida H, Dobashi K, Hayashi T, Hori M, Matsuki K, Minamino T, Yokoyama S, Harada-Shiba M: Homozygous Familial Hypercholesterolemia. J Atheroscler Thromb, 2021; 28: 665-678 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94).Uauy R, Vega GL, Grundy SM, Bilheimer DM: Lovastatin therapy in receptor-negative homozygous familial hypercholesterolemia: lack of effect on low-density lipoprotein concentrations or turnover. J Pediatr, 1988; 113: 387-392 [DOI] [PubMed] [Google Scholar]
- 95).Stein EA, Honarpour N, Wasserman SM, Xu F, Scott R, Raal FJ: Effect of the proprotein convertase subtilisin/kexin 9 monoclonal antibody, AMG 145, in homozygous familial hypercholesterolemia. Circulation, 2013; 128: 2113-2120 [DOI] [PubMed] [Google Scholar]
- 96).Yamamoto A, Harada-Shiba M, Kawaguchi A, Oi K, Kubo H, Sakai S, Mikami Y, Imai T, Ito T, Kato H, Endo M, Sato I, Suzuki Y, Hori H: The effect of atorvastatin on serum lipids and lipoproteins in patients with homozyous familial hypercholesterolemia undergoing LDL-apheresis therapy. Atherosclerosis, 2000; 153: 89-98 [DOI] [PubMed] [Google Scholar]
- 97).Raal FJ, Pilcher GJ, Panz VR, van Deventer HE, Brice BC, Blom DJ, Marais AD: Reduction in mortality in subjects with homozygous familial hypercholesterolemia associated with advances in lipid-lowering therapy. Circulation, 2011; 124: 2202-2207 [DOI] [PubMed] [Google Scholar]
- 98).Cuchel M, Bloedon LT, Szapary PO, Kolansky DM, Wolfe ML, Sarkis A, Millar JS, Ikewaki K, Siegelman ES, Gregg RE, Rader DJ: Inhibition of microsomal triglyceride transfer protein in familial hypercholesterolemia. N Engl J Med, 2007; 356: 148-156 [DOI] [PubMed] [Google Scholar]
- 99).Harada-Shiba M, Ikewaki K, Nohara A, Otsubo Y, Yanagi K, Yoshida M, Chang Q, Foulds P: Efficacy and Safety of Lomitapide in Japanese Patients with Homozygous Familial Hypercholesterolemia. J Atheroscler Thromb, 2017; 24: 402-411 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100).Kameyama N, Maruyama C, Kitagawa F, Nishii K, Uenomachi K, Katayama Y, Koga H, Chikamoto N, Kuwata Y, Torigoe J, Arimoto M, Tokumaru T, Ikewaki K, Nohara A, Otsubo Y, Yanagi K, Yoshida M, Harada-Shiba M: Dietary Intake during 56 Weeks of a Low-Fat Diet for Lomitapide Treatment in Japanese Patients with Homozygous Familial Hypercholesterolemia. J Atheroscler Thromb, 2019; 26: 72-83 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101).Yamamoto A, Matsuzawa Y, Yokoyama S, Funahashi T, Yamamura T, Kishino B: Effects of probucol on xanthomata regression in familial hypercholesterolemia. Am J Cardiol, 1986; 57: 29h-35h [DOI] [PubMed] [Google Scholar]
- 102).Shirahata Y, Ohkohchi N, Kawagishi N, Syouji M, Tsukamoto S, Sekiguchi S, Koyamada N, Oikawa S, Satomi S: Living-donor liver transplantation for homozygous familial hypercholesterolemia from a donor with heterozygous hypercholesterolemia. Transpl Int, 2003; 16: 276-279 [DOI] [PubMed] [Google Scholar]
- 103).Kawagishi N, Satoh K, Akamatsu Y, Sekiguchi S, Ishigaki Y, Oikawa S, Satomi S: Long-term outcome after living donor liver transplantation for two cases of homozygous familial hypercholesterolemia from a heterozygous donor. J Atheroscler Thromb, 2007; 14: 94-98 [DOI] [PubMed] [Google Scholar]
- 104).Yamamoto A, Harada-Shiba M, Kawaguchi A, Tsushima M: Apheresis technology for prevention and regression of atherosclerosis. Ther Apher, 2001; 5: 221-225 [DOI] [PubMed] [Google Scholar]
- 105).Mabuchi H, Michishita I, Sakai T, Sakai Y, Watanabe A, Wakasugi T, Takeda R: Treatment of homozygous patients with familial hypercholesterolemia by double-filtration plasmapheresis. Atherosclerosis, 1986; 61: 135-140 [DOI] [PubMed] [Google Scholar]
- 106).Makino H, Harada-Shiba M: Long-term effect of low-density lipoprotein apheresis in patients with homozygous familial hypercholesterolemia. Ther Apher Dial, 2003; 7: 397-401 [DOI] [PubMed] [Google Scholar]
- 107).Sampietro T, Tuoni M, Ferdeghini M, Ciardi A, Marraccini P, Prontera C, Sassi G, Taddei M, Bionda A: Plasma cholesterol regulates soluble cell adhesion molecule expression in familial hypercholesterolemia. Circulation, 1997; 96: 1381-1385 [DOI] [PubMed] [Google Scholar]
- 108).Makino H, Koezuka R, Tamanaha T, Ogura M, Matsuki K, Hosoda K, Harada-Shiba M: Familial Hypercholesterolemia and Lipoprotein Apheresis. J Atheroscler Thromb, 2019; 26: 679-687 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109).Stefanutti C, Di Giacomo S, Vivenzio A, Colloridi V, Bosco G, Berni A, Rabbone I, Cerutti F, Bertolini S: Low-density lipoprotein apheresis in a patient aged 3.5 years. Acta Paediatr, 2001; 90: 694-701 [DOI] [PubMed] [Google Scholar]
- 110).Thompson GR: Recommendations for the use of LDL apheresis. Atherosclerosis, 2008; 198: 247-255 [DOI] [PubMed] [Google Scholar]
- 111).Thompson GR, Lowenthal R, Myant NB: Plasma exchange in the management of homozygous familial hypercholesterolaemia. Lancet, 1975; 1: 1208-1211 [DOI] [PubMed] [Google Scholar]
- 112).Yokoyama S, Hayashi R, Satani M, Yamamoto A: Selective removal of low density lipoprotein by plasmapheresis in familial hypercholesterolemia. Arteriosclerosis, 1985; 5: 613-622 [DOI] [PubMed] [Google Scholar]
- 113).Civeira F: Guidelines for the diagnosis and management of heterozygous familial hypercholesterolemia. Atherosclerosis, 2004; 173: 55-68 [DOI] [PubMed] [Google Scholar]
- 114).Thorogood M, Seed M, De Mott K: Management of fertility in women with familial hypercholesterolaemia: summary of NICE guidance. BJOG, 2009; 116: 478-479 [DOI] [PubMed] [Google Scholar]
- 115).Amundsen AL, Khoury J, Iversen PO, Bergei C, Ose L, Tonstad S, Retterstøl K: Marked changes in plasma lipids and lipoproteins during pregnancy in women with familial hypercholesterolemia. Atherosclerosis, 2006; 189: 451-457 [DOI] [PubMed] [Google Scholar]
- 116).Amundsen AL, Khoury J, Sandset PM, Seljeflot I, Ose L, Tonstad S, Henriksen T, Retterstøl K, Iversen PO: Altered hemostatic balance and endothelial activation in pregnant women with familial hypercholesterolemia. Thromb Res, 2007; 120: 21-27 [DOI] [PubMed] [Google Scholar]
- 117).Beigel Y, Bar J, Cohen M, Hod M: Pregnancy outcome in familial homozygous hypercholesterolemic females treated with long-term plasma exchange. Acta Obstet Gynecol Scand, 1998; 77: 603-608 [DOI] [PubMed] [Google Scholar]
- 118).Naoumova RP, Thompson GR, Soutar AK: Current management of severe homozygous hypercholesterolaemias. Curr Opin Lipidol, 2004; 15: 413-422 [DOI] [PubMed] [Google Scholar]
- 119).Klingel R, Göhlen B, Schwarting A, Himmelsbach F, Straube R: Differential indication of lipoprotein apheresis during pregnancy. Ther Apher Dial, 2003; 7: 359-364 [DOI] [PubMed] [Google Scholar]
- 120).Ogura M, Makino H, Kamiya C, Yoshimatsu J, Soran H, Eatough R, Perrone G, Harada-Shiba M, Stefanutti C: Lipoprotein apheresis is essential for managing pregnancies in patients with homozygous familial hypercholesterolemia: Seven case series and discussion. Atherosclerosis, 2016; 254: 179-183 [DOI] [PubMed] [Google Scholar]