Abstract
Background
Estrogen-based hormone therapy (HT) may have beneficial cardiovascular effects when initiated in early menopause. This has not been examined in women with human immunodeficiency virus (HIV), who have heightened immune activation and cardiovascular risks.
Methods
Among 609 postmenopausal women (1234 person-visits) in the Women’s Interagency HIV Study, we examined the relationship of ever HT use (oral, patch, or vaginal) with subclinical atherosclerosis: carotid artery intima-media thickness (CIMT), distensibility, and plaque assessed via repeated B-mode ultrasound imaging (2004–2013). We also examined associations of HT with cross-sectional biomarkers of immune activation and D-dimer. Statistical models were adjusted for sociodemographic, behavioral, and cardiometabolic factors.
Results
Women (mean age, 51 years; 80% HIV positive) who ever used HT at baseline were older, and more likely to be non-Hispanic White and report higher income, than never-users. Women who ever used HT had 43% lower prevalence of plaque (prevalence ratio, 0.57 [95% confidence interval {CI}, .40–.80]; P < .01), 2.51 µm less progression of CIMT per year (95% CI, –4.60, to –.41; P = .02), and marginally lower incidence of plaque over approximately 7 years (risk ratio, 0.38 [95% CI, .14–1.03; P = .06), compared with never-users, adjusting for covariates; ever HT use was not associated with distensibility. These findings were similar for women with and without HIV. Ever HT use was associated with lower serum D-dimer, but not with biomarkers of immune activation after covariate adjustment.
Conclusions
HT may confer a subclinical cardiovascular benefit in women with HIV. These results begin to fill a knowledge gap in menopausal care for women with HIV, in whom uptake of HT is very low.
Keywords: menopause, hormone therapy, cardiovascular disease, atherosclerosis, HIV
In this cohort study of women with and without HIV, ever use of estrogen-based hormone therapy was associated with less progression of subclinical atherosclerosis over time. Hormone therapy may confer a similar cardiovascular benefit for women with and without HIV.
Estrogen-based menopausal hormone therapy (HT) has been the subject of controversy regarding its effects on cardiovascular disease (CVD). While many observational studies of recently postmenopausal women have shown reduced rates of coronary heart disease (CHD) in HT users [1], the Women’s Health Initiative (WHI) randomized trials of the 1990s demonstrated increased CHD and stroke with HT use, leading to concerns of harm and widespread declines in HT prescriptions [2]. The discrepancy between the WHI trials, conducted among older women, and observational studies of women in early menopause, led to the HT timing hypothesis—that effects of HT depend on timing of initiation in relation to menopause [3]. Substantial evidence has accumulated supporting the protective effect of HT on cardiometabolic endpoints among women <60 years old when initiated within 10 years of menopause [2–5].
CVD is a particular concern in women with HIV, as HIV infection is associated with increased CVD, more so in women than men [6]. Persistent immune activation in people with HIV may contribute to elevated risk of non-AIDS diseases, including CVD [7], despite antiretroviral therapy. Perturbations in sex hormones in women with HIV may also contribute to CVD risk [8]. HT uptake for menopausal symptoms in women with HIV is very low [9], with a key barrier being lack of knowledge of the effect of HT on CVD in women with HIV [10]. Here, we examine the relationship of menopausal HT with subclinical atherosclerosis and blood immune activation biomarkers in the Women’s Interagency HIV Study (WIHS). Additionally, given concerns regarding increased risk of venous thromboembolism (VTE) with HT, we also assessed the relationship of HT to serum D-dimer, a biomarker of thrombosis associated with VTE risk in women taking HT [11].
METHODS
Study Population
WIHS was a US multicenter cohort of women with and without HIV that conducted semiannual interviews, physical examinations, and laboratory tests [12]. Institutional review boards at all sites approved the study, and participants provided written informed consent.
Women in the current analysis participated in a vascular disease substudy at the original WIHS sites (Bronx and Brooklyn, New York; Chicago; San Francisco; Los Angeles; Washington, D.C.) [13], which featured high-resolution B-mode carotid artery ultrasound at a baseline visit (2004–2006; wave 1) and follow-up ultrasounds every 2–3 years through 2013 (waves 2–4).
From 4325 person-visits in the vascular substudy, we included 1234 postmenopausal person-visits from 609 participants, after exclusions including age <40 years and report of hormonal contraceptive or cancer HT use. Prospective analyses had further exclusions. Person-visits were classified as postmenopausal if they were after a certainly or likely observed natural final menstrual period, after a bilateral ovariectomy, or uncertain status but age ≥45 years. Additional details are shown in the Supplementary Methods, Supplementary Figure 1, and Supplementary Table 1.
Hormone Therapy Use Definition
HT use was determined semiannually based on participant self-report starting in 2001. Ever HT use was defined for each person-visit as any report of HT use with estrogen (oral, patch, or vaginal) at or prior to the person-visit. Past HT use was defined as HT use at a prior but not the current person-visit, while current HT use was defined as reporting HT use at the current person-visit. The Supplementary Methods and Supplementary Figure 2 have additional details.
Subclinical Cardiovascular Disease
High-resolution B-mode ultrasound with automated computerized edge detection software assessed carotid artery arteriosclerosis in the right carotid artery [13]. Outcomes were intima-media thickness of the right common carotid artery (CIMT; µm), presence/absence of carotid artery lesions (plaque) defined as focal CIMT >1.5 mm in any imaged segment (common and internal carotid arteries and bifurcation), and carotid artery distensibility (10−6 × N−1 × m2) measured at the common carotid artery. CIMT was assessed at all 4 waves of the vascular substudy, distensibility at waves 1–3, and plaque at waves 1 and 4 (sample sizes in Supplementary Table 1 and Supplementary Figure 1).
Biomarkers
Biomarkers were measured on subsets of participants at the vascular substudy baseline visit.
T-Cell Activation
Multiparametric flow cytometry of peripheral blood mononuclear cells [14] measured %HLA-DR+CD38+ of CD4+ and CD8+ T cells (n = 168).
Monocyte/Macrophage Activation and D-Dimer
Enzyme-linked immunosorbent assays measured serum soluble CD14 and CD163 (R&D Systems, Minneapolis) (n = 147) and D-dimer (Abcam, Boston, Massachusetts) (n = 142) [15].
Statistical Analysis
In all analyses, we considered nested models to serially adjust for potential confounders/mediators (covariates shown in Table 1 and described in the Supplementary Methods), and explored separate models by HIV serostatus where sample size allowed. P < .05 was considered statistically significant. Analyses were conducted in R version 4.1.2 software.
Table 1.
Characteristics of Postmenopausal Women in the Women’s Interagency HIV Study at the Baseline Vascular Substudy Visit, by Ever Use of Hormone Therapya
| HT Use | ||||
|---|---|---|---|---|
| Characteristic | Overall (N = 609) |
Never (n = 466) |
Ever (n = 143) |
P Valueb |
| No. of prior visits with HT data, mean (SD) | 9.55 (4.98) | 10.06 (5.17) | 7.90 (3.88) | <.001 |
| Age, y, mean (SD) | 50.93 (5.44) | 50.58 (5.26) | 52.08 (5.89) | .001 |
| HIV positive | 487 (80.0) | 369 (79.2) | 118 (82.5) | .45 |
| Race/ethnicity | <.001 | |||
| ȃBlack non-Hispanic | 378 (62.1) | 299 (64.2) | 79 (55.2) | |
| ȃWhite non-Hispanic | 71 (11.7) | 40 (8.6) | 31 (21.7) | |
| ȃHispanic | 144 (23.6) | 114 (24.5) | 30 (21.0) | |
| ȃOther/missing | 16 (2.6) | 13 (2.8) | 3 (2.1) | |
| Annual income | .06 | |||
| ȃ$12 000 or less | 362 (59.4) | 290 (62.2) | 72 (50.3) | |
| ȃ$12 001–$24 000 | 122 (20.0) | 87 (18.7) | 35 (24.5) | |
| ȃ$24 001–$36 000 | 61 (10.0) | 46 (9.9) | 15 (10.5) | |
| ȃ$36 001 or more | 64 (10.5) | 43 (9.2) | 21 (14.7) | |
| Educational attainment | .04 | |||
| ȃLess than high school | 229 (37.6) | 188 (40.3) | 41 (28.7) | |
| ȃCompleted high school | 199 (32.7) | 151 (32.4) | 48 (33.6) | |
| ȃSome college | 144 (23.6) | 102 (21.9) | 42 (29.4) | |
| ȃCompleted college | 37 (6.1) | 25 (5.4) | 12 (8.4) | |
| Employed | 180 (29.6) | 129 (27.7) | 51 (35.7) | .09 |
| Smoking status | .03 | |||
| ȃNever smoker | 111 (18.2) | 80 (17.2) | 31 (21.7) | |
| ȃCurrent smoker | 311 (51.1) | 252 (54.1) | 59 (41.3) | |
| ȃFormer smoker | 187 (30.7) | 134 (28.8) | 53 (37.1) | |
| Current drinking status | .70 | |||
| ȃAbstainer | 364 (59.8) | 282 (60.5) | 82 (57.3) | |
| ȃ>0–7 drinks/wk | 187 (30.7) | 139 (29.8) | 48 (33.6) | |
| ȃ>7 drinks/wk | 58 (9.5) | 45 (9.7) | 13 (9.1) | |
| Current drug use | 138 (22.7) | 109 (23.4) | 29 (20.3) | .51 |
| HCV positive | 307 (50.4) | 244 (52.4) | 63 (44.1) | .10 |
| BMI, kg/m2, mean (SD) | 28.80 (7.34) | 28.75 (7.43) | 28.94 (7.06) | .64 |
| History of hypertension | 290 (47.6) | 220 (47.2) | 70 (49.0) | .79 |
| History of diabetes | 111 (18.2) | 78 (16.7) | 33 (23.1) | .11 |
| History of CVD | 67 (11.0) | 50 (10.7) | 17 (11.9) | .82 |
| Current lipid-lowering medication | 32 (5.3) | 26 (5.6) | 6 (4.2) | .66 |
| Current HIV viral loadc, copies/mL, geometric mean (SD) | 368.7 (24.3) | 350.7 (27.4) | 437.0 (16.3) | .72 |
| Current detectable HIV viral loadc | 253 (52.0) | 198 (53.7) | 55 (46.6) | .22 |
| Current CD4 countc, cells/µL, mean (SD) | 474.57 (305.98) | 461.25 (309.00) | 516.25 (293.73) | .04 |
| Current ART usec | 368 (75.6) | 276 (74.8) | 92 (78.0) | .57 |
| CIMT, µm, mean (SD) | 791.42 (132.15) | 794.39 (132.79) | 781.74 (130.01) | .23 |
| Carotid artery distensibilityd (10−6 × N–1 × m2), mean (SD) | 15.38 (7.07) | 15.34 (7.08) | 15.50 (7.08) | .85 |
| Presence of carotid artery plaquee | 116 (24.9) | 94 (27.8) | 22 (17.2) | .03 |
Data are presented as No. (%) unless otherwise indicated.
Abbreviations: ART, antiretroviral therapy; BMI, body mass index; CIMT, carotid artery intima-media thickness; CVD, cardiovascular disease; HCV, hepatitis C virus; HIV, human immunodeficiency virus; HT, hormone therapy; SD, standard deviation.
Each participant’s first visit in the vascular substudy was used for this summary table.
P values from Wilcoxon rank-sum test or χ2 test for continuous and categorical variables, respectively.
Only in women with HIV (n = 487).
Reduced sample size (n = 531).
Reduced sample size (n = 466).
HT and Subclinical Atherosclerosis
Generalized estimating equations (GEEs) using independence working correlation estimated the population-average effect of HT use on continuous outcomes of CIMT and distensibility (linear model) or the binary outcome of plaque (Poisson model). Covariates were time-varying, excepting time-fixed variables (eg, race/ethnicity).
HT and Subclinical Atherosclerosis Progression
For CIMT and distensibility outcomes, linear mixed-effects regression models with a random intercept were used to examine whether change over time differed by HT use. The effect of interest was the interaction of time and HT use at baseline. Baseline covariate values were used.
For the plaque outcome, only available at waves 1 and 4, we used Poisson regression with robust standard errors to examine whether ever HT use at wave 1 was associated with (1) development of new plaque or (2) incidence of plaque at wave 4. Women were defined as having developed new plaque if the number of lesions increased from wave 1 to 4 (eg, from none to 1 lesion, 1 to 2 lesions). Plaque incidence was considered development of plaque among women with none at wave 1. Models were restricted to women with plaque measured at both waves 1 and 4 (Supplementary Figure 1). Due to small sample size, only the binary ever-HT variable was used as a predictor, and models in women without HIV were not explored separately.
HT Use and Biomarkers
We used linear regression to examine cross-sectional associations of HT use with T-cell activation (%HLADR+CD38+ of CD4+ and CD8+ T cells), serum soluble CD14 and CD163, and D-dimer. Due to small sample size, models in women without HIV were not explored separately.
RESULTS
Participant Characteristics
Among 609 postmenopausal women at baseline during 2004–2006, 143 (23.5%) had ever used HT, and 46 (8%) were currently using HT. Among ever-users, 76% had used HT for menopause-related symptoms (hot flashes, vaginal dryness, sweating). On average, women were 51 years old, and most were people with HIV (80%), non-Hispanic Black (62%), and low-income (≤$12 000; 59%) (Table 1). Ever HT users were older than never-users, more likely to be non-Hispanic White and have more educational attainment, and less likely to be current smokers (Table 1). Differences in characteristics by ever HT use were similar for women with and without HIV; among women with HIV, CD4 cell count was higher in ever-users vs never-users (Supplementary Table 2). Past and current HT users at baseline differed similarly from never-users (Supplementary Table 3).
HT Use and Subclinical Atherosclerosis
In GEE models adjusting for age, HIV status, and sociodemographic, behavioral, and cardiometabolic factors, women who ever used HT had 43% lower prevalence of carotid artery plaque on average than women who never used HT (prevalence ratio [PR], 0.57 [95% confidence interval {CI}, .40–.80], P < .01), with similar associations observed for women with and without HIV (P for interaction ≥ .93) (Table 2). When separating ever HT users into past and current users, only past users had lower prevalence of plaque compared to never-users (PR, 0.47 [95% CI, .31–.72]; P < .01) while current users did not (PR, 0.87 [95% CI, .55–1.38]; P = .55); this pattern was similar for women with and without HIV though nonsignificant in women without HIV (Supplementary Table 4).
Table 2.
Ever Hormone Therapy Use and Subclinical Cardiovascular Disease in the Women’s Interagency HIV Studya
| Model | Allb | Women With HIVc | Women Without HIVd | P interaction e | |||
|---|---|---|---|---|---|---|---|
| β (95% CI) | P Value | β (95% CI) | P Value | β (95% CI) | P Value | ||
| CIMT, µm | |||||||
| ȃUnadjusted | –18.95 (–46.12 to 8.23) | .17 | –21.80 (–48.15 to 4.54) | .10 | –4.61 (–88.59 to 79.37) | .91 | .70 |
| ȃModel 1 | –26.36 (–53.30 to .58) | .06 | –29.27 (–55.29 to –3.26) | .03 | –15.35 (–98.74 to 68.04) | .72 | .71 |
| ȃModel 2 | –21.31 (–47.89 to 5.27) | .12 | –25.22 (–51.50 to 1.05) | .06 | NA | NA | NA |
| ȃModel 3 | –22.41 (–47.84 to 3.03) | .08 | –25.28 (–51.41 to .85) | .06 | NA | NA | NA |
| Carotid artery distensibility (10−6 × N–1 × m2) | |||||||
| ȃUnadjusted | .25 (–.98 to 1.47) | .69 | .28 (–1.08 to 1.64) | .69 | –.02 (–2.70 to 2.67) | .99 | .85 |
| ȃModel 1 | .68 (–.49 to 1.85) | .26 | .72 (–.61 to 2.04) | .29 | .31 (–2.09 to 2.72) | .80 | .72 |
| ȃModel 2 | .32 (–.80 to 1.43) | .57 | .25 (–1.00 to 1.49) | .70 | NA | NA | NA |
| ȃModel 3 | .39 (–.69 to 1.48) | .48 | .26 (–.95 to 1.47) | .67 | NA | NA | NA |
| Carotid artery plaque | |||||||
| ȃUnadjusted | .60f (.41–.86) | .01 | .59f (.40–.87) | .01 | .57f (.17–1.84) | .34 | .94 |
| ȃModel 1 | .55f (.38–.78) | <.01 | .56f (.38–.81) | <.01 | .52f (.19–1.47) | .22 | .93 |
| ȃModel 2 | .59f (.41–.84) | <.01 | .62f (.42–.90) | .01 | NA | NA | NA |
| ȃModel 3 | .57f (.40–.80) | <.01 | .59f (.41–.85) | <.01 | NA | NA | NA |
Abbreviations: CI, confidence interval; CIMT, carotid artery intima-media thickness; HIV, human immunodeficiency virus; HT, hormone therapy; NA, not applicable.
Generalized estimating equations with the working independence correlation structure estimated the population-average effect of ever hormone therapy (HT) use (vs never-use) on the continuous outcomes of CIMT and distensibility (linear model) or the binary outcome of plaque (Poisson model). Model 1 was adjusted for concurrent age and HIV status (or antiretroviral therapy, viral load, and CD4 cell count in women with HIV); model 2 was additionally adjusted for race/ethnicity, income, educational attainment, employment status, smoking status, alcohol drinking status, drug use, and hepatitis C virus serostatus; model 3 was additionally adjusted for body mass index, hypertension, diabetes, history of self-reported cardiovascular disease, and use of lipid-lowering medications. We show models for the overall study population and stratified by HIV status, but modeling in women without HIV was restricted to age adjustment only, due to low sample size.
Number of person-visits (unique persons) total: 1234 (609) for CIMT, 922 (531) for distensibility, and 700 (565) for plaque; number of person-visits (unique persons) ever HT use: 345 (156) for CIMT, 271 (143) for distensibility, and 193 (151) for plaque.
Number of person-visits (unique persons): 974 (487) for CIMT, 731 (428) for distensibility, and 557 (448) for plaque; number of person-visits (unique persons) ever HT use: 277 (128) for CIMT, 219 (117) for distensibility, and 157 (124) for plaque.
Number of person-visits (unique persons): 260 (122) for CIMT, 191 (103) for distensibility, and 143 (117) for plaque; number of person-visits (unique persons) ever HT use: 68 (28) for CIMT, 52 (26) for distensibility, and 36 (27) for plaque.
P value of HIV status × ever HT term.
Prevalence ratio.
Ever HT use was associated with nonsignificantly lower CIMT on average compared to never-use in GEE models (β = −22.4 [95% CI, −47.8 to 3.0] µm; P = .08), with no heterogeneity by HIV status (P for interaction ≥ .70) (Table 2). Current HT use was associated with significantly lower CIMT compared to never-use (β = −34.2 [95% CI, −62.1 to –6.3] µm; P = .02), whereas past HT use was not (β = −18.7 [95% CI, −47.5 to 10.2] µm; P = .20). These patterns were similar for women with and without HIV, although nonsignificant in women without HIV (Supplementary Table 4).
Ever, past, or current HT use were not associated with carotid artery distensibility in GEE models (Table 2; Supplementary Table 4), consistent in women with and without HIV (P for interaction ≥ .72).
HT Use and Subclinical Atherosclerosis Progression
Ever use of HT at baseline compared to never-use was associated with a significantly lower increase in CIMT over time (median follow-up, 4.0 [range, 1.4–7.8] years), in linear mixed-effects models adjusting for baseline age, HIV status, and sociodemographic, behavioral, and cardiometabolic factors (β = −2.51 [95% CI, −4.60 to –.41] µm/year; P = .02) (Table 3, Figure 1). This association was consistent in women with and without HIV, though not reaching significance in either stratum (Table 3). While both past and current HT use at baseline were related to lower increases in CIMT over time compared to never-use, this effect was significant in past HT users (β = −3.70 [95% CI, −6.31 to –1.10] µm/year; P = .01) but not current HT users (β = −2.46 [95% CI, −5.69 to .78] µm/year; P = .14) (Supplementary Table 5).
Table 3.
Ever Hormone Therapy Use and Progression of Carotid Artery Intima-Media Thickness and Distensibility in the Women’s Interagency HIV Studya
| Allb | Women With HIVc | Women Without HIVd | ||||
|---|---|---|---|---|---|---|
| Model | β (95% CI) | P Value | β (95% CI) | P Value | β (95% CI) | P Value |
| CIMT progression, µm/year | ||||||
| ȃUnadjusted | –2.50 (–4.60 to –.41) | .02 | –2.35 (–4.72 to .02) | .05 | –3.29 (–7.73 to 1.16) | .15 |
| ȃModel 1 | –2.50 (–4.59 to –.40) | .02 | –2.33 (–4.70 to .04) | .05 | –3.28 (–7.72 to 1.17) | .15 |
| ȃModel 2 | –2.48 (–4.58 to –.39) | .02 | –2.32 (–4.69 to .06) | .06 | NA | NA |
| ȃModel 3 | –2.51 (–4.60 to –.41) | .02 | –2.33 (–4.71 to .04) | .05 | NA | NA |
| Carotid artery distensibility progression, 10−6 × N–1 × m2/year | ||||||
| ȃUnadjusted | –.33 (–.80 to .13) | .16 | –.45 (–1.00 to .11) | .12 | .10 (–.66 to .87) | .79 |
| ȃModel 1 | –.36 (–.83 to .10) | .13 | –.49 (–1.04 to .07) | .09 | .07 (–.69 to .84) | .86 |
| ȃModel 2 | –.32 (–.79 to .15) | .18 | –.42 (–.97 to .14) | .14 | NA | NA |
| ȃModel 3 | –.30 (–.76 to .17) | .21 | –.41 (–.96 to 0.14) | .15 | NA | NA |
Abbreviations: CI, confidence interval; CIMT, carotid artery intima-media thickness; HIV, human immunodeficiency virus; NA, not applicable.
Linear mixed-effects models estimated the difference in CIMT or distensibility progression over time by ever hormone therapy (HT) use at baseline (ie, interaction of time × ever HT use). Unadjusted model includes terms for time, ever HT use at baseline, and the interaction. Model 1 was adjusted for age at baseline and HIV status (or antiretroviral therapy at baseline, viral load at baseline, and CD4 cell count at baseline in women with HIV); model 2 was additionally adjusted for race/ethnicity, income at baseline, educational attainment at baseline, employment status at baseline, smoking status at baseline, alcohol drinking status at baseline, drug use at baseline, and hepatitis C virus serostatus at baseline; Model 3 was additionally adjusted for body mass index at baseline, hypertension at baseline, diabetes at baseline, and lipid-lowering medication at baseline. We show models for the overall study population and stratified by HIV status, but modeling in women without HIV was restricted to age at baseline adjustment only, due to low sample size.
Number of person-visits (unique persons) total: 911 (303) for CIMT and 627 (246) for distensibility; number of person-visits (unique persons) ever HT use: 260 (80) for CIMT and 198 (74) for distensibility.
Number of person-visits (unique persons): 712 (238) for CIMT and 488 (192) for distensibility; number of person-visits (unique persons) ever HT use: 209 (65) for CIMT and 160 (60) for distensibility.
Number of person-visits (unique persons): 199 (65) for CIMT and 139 (54) for distensibility; number of person-visits (unique persons) ever HT use: 51 (15) for CIMT and 38 (14) for distensibility.
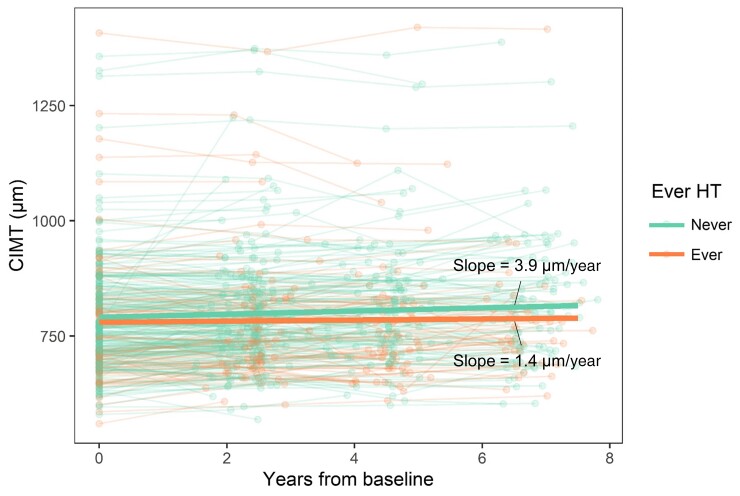
Figure 1.
Progression of carotid artery intima-media thickness (CIMT) over time in ever and never hormone therapy (HT) users. Thin lines represent trajectory of CIMT within each participant from their first visit in the vascular substudy (n = 303 women; n = 911 person-visits). Thick lines represent the estimate of change in CIMT over time for ever and never HT users, from linear mixed-effects models with terms for time, ever HT use at baseline, and the interaction of time × ever HT use, as well as a random intercept. The slope for women who never used HT at baseline (n = 223 women) was significantly different from zero (P < .0001), whereas the slope in women who ever used HT (n = 80 women) was not different from zero (P = .11).
Ever-use of HT at baseline was not associated with change in carotid artery distensibility over time compared to never-use (median follow-up, 2.6 [range, 1.4–5.2] years) (Table 3). However, past use of HT at baseline was associated with a greater decline in distensibility compared to never-use (β = −.57 [95% CI, −1.14 to .00] 10−6 × N–1 × m2/year; P = .05), while current use of HT at baseline was not (β = −.02 [range, −.73 to .69] 10−6 × N–1 × m2/year; P = .95). This pattern was consistent in women with HIV, but not in women without HIV (Supplementary Table 5).
Finally, during a median follow-up of 6.7 (range, 5.4–7.8) years, women who ever used HT at baseline compared to never-users had nonsignificantly lower risk of carotid artery plaque incidence in a covariate-adjusted Poisson regression model (incidence in ever- vs never-users, 10% vs 24%; adjusted risk ratio, 0.38 [95% CI, .14–1.03]; P = .06), with a similar finding for the plaque progression outcome (Table 4). The association of ever HT use with lower risk of plaque was also observed in women with HIV, though somewhat attenuated (incidence in ever- vs never-users, 13% vs 25%) (Table 4).
Table 4.
Ever Hormone Therapy Use and Progression or Incidence of Carotid Artery Plaque in the Women’s Interagency HIV Studya
| Model | All | Women With HIV | ||
|---|---|---|---|---|
| Risk Ratio (95% CI) | P Value | Risk Ratio (95% CI) | P Value | |
| Plaque progressionb | ||||
| ȃUnadjusted | .40 (.15–1.10) | .08 | .48 (.18–1.32) | .16 |
| ȃModel 1 | .41 (.15–1.10) | .08 | .49 (.18–1.39) | .18 |
| ȃModel 2 | .47 (.17–1.29) | .14 | .55 (.20–1.50) | .24 |
| ȃModel 3 | .44 (.17–1.17) | .10 | .52 (.19–1.40) | .19 |
| Incidence of plaquec | ||||
| ȃUnadjusted | .42 (.15–1.16) | .09 | .49 (.18–1.36) | .17 |
| ȃModel 1 | .41 (.15–1.10) | .08 | .53 (.18–1.55) | .25 |
| ȃModel 2 | .45 (.15–1.37) | .16 | .50 (.14–1.74) | .28 |
| ȃModel 3 | .38 (.14–1.03) | .06 | .42 (.14–1.27) | .12 |
Abbreviations: CI, confidence interval; HIV, human immunodeficiency virus.
Poisson regression estimated the relative risk of plaque progression or incidence. Plaque progression was defined as development of any new plaque from waves 1 to 4 of the vascular substudy. Incidence of plaque was defined as development of plaque at wave 4, among women with no plaque at wave 1. Model 1 was adjusted for age at baseline and HIV status (or antiretroviral therapy at baseline, viral load at baseline, and CD4 cell count at baseline in women with HIV); model 2 was additionally adjusted for race/ethnicity, income at baseline, educational attainment at baseline, employment status at baseline, smoking status at baseline, alcohol drinking status at baseline, drug use at baseline, and hepatitis C virus serostatus at baseline; model 3 was additionally adjusted for body mass index at baseline, hypertension at baseline, diabetes at baseline, and lipid-lowering medication at baseline.
n = 131 women (42 ever hormone therapy [HT] users), 105 women with HIV (33 ever HT users).
n = 113 women (39 ever HT users), 91 women with HIV (32 ever HT users).
HT Use and Biomarkers of Immune Activation and D-dimer
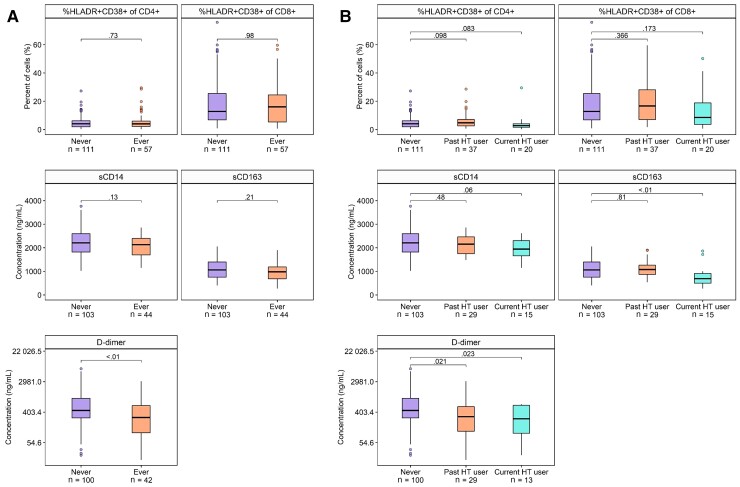
In cross-sectional analysis at baseline, ever HT use was not significantly associated with %HLADR+CD38+ of CD4+ and CD8+ T-cells, or serum soluble CD14 and CD163 (Figure 2, Supplementary Table 6). Current HT users had lower serum soluble CD163 compared to never-users (Figure 2, Supplementary Table 7), but the observed association was somewhat attenuated by adjustment for sociodemographic and behavioral factors (Supplementary Table 7). Findings were similar in women with HIV (Supplementary Tables 6 and 7).
Figure 2.
Hormone therapy (HT) use and biomarkers of immune activation and D-dimer. Boxplots show distribution of biomarkers according to ever or never HT use (A) or past, current, or never HT use (B). P values are from Wilcoxon rank-sum test. Abbreviations: HT, hormone therapy; sCD14, serum soluble CD14; sCD163, serum soluble CD163.
Ever HT use was associated with significantly lower serum levels of D-dimer (Figure 2, Supplementary Table 6), with a stronger association for current than past HT users (Supplementary Table 7). The association of ever HT use with lower D-dimer persisted upon covariate adjustment, and was similar in women with HIV (Supplementary Table 6).
DISCUSSION
In this longitudinal study of women with and without HIV, ever-use of menopausal HT was associated with lower prevalence of carotid artery plaque, reduced progression of CIMT over time, and possibly with lower incidence of carotid artery plaque. Though CIMT is a surrogate endpoint, to our knowledge this is the first study of HT and any cardiovascular outcomes in women with HIV, who have high CVD risk compared to women in the general population [6]. Since our study comprised mostly younger postmenopausal women (93% were <60 years old at baseline), our findings are consistent with the HT timing hypothesis, in which HT is thought to be protective against CVD in younger, recently postmenopausal women [3]. Importantly, associations of HT with subclinical atherosclerosis were largely similar in women with and without HIV, suggesting for the first time that women with HIV may benefit from early menopausal use of HT, similar to women without HIV [4, 5, 16–18]. CIMT and presence of carotid artery plaque measured by ultrasound predict future clinical cardiovascular events [19]. In addition, HIV infection is associated with higher prevalence [20–22] and future development of carotid artery plaque [13], and plaque is associated with higher risk of CVD events and mortality in people with HIV [23, 24]. Taken together, the association of HT use with reduced subclinical atherosclerosis in women with HIV may have clinical implications extending to protection from CVD events and death. While HT is not currently indicated for CVD prevention in any population, our research highlights the unmet need for high-quality clinical studies of the CVD benefits of HT in younger postmenopausal women with HIV.
We found distinct associations of past HT use with lower prevalence of carotid artery plaque, and current HT use with lower CIMT. These findings may be related to the time-frame in which HT influences CIMT, an early anatomic feature in atherosclerosis development. In both the 2-year Estrogen in the Prevention of Atherosclerosis Trial [18] and 5-year Early vs. Late Intervention Trial with Estrogen [4] trials, the difference in CIMT progression between the HT and placebo groups became apparent within 1 year of the trial start, indicating a relatively rapid effect of HT on CIMT. In contrast, carotid artery plaque development may take more years to occur, which could explain why only past HT use was related to lower plaque prevalence. Semiannual data collection on HT use in the WIHS began shortly before the vascular substudy; we were therefore unable to capture length of HT use, which would further clarify the relationship of HT timing and duration with CIMT and plaque.
Estrogens have pleiotropic cardiovascular influences [25]; thus, it is unclear why HT use was not associated with higher carotid artery distensibility (ie, lower stiffness) in this study, and moreover, why past HT use at baseline was associated with greater decline in distensibility over time in women with HIV. Observational studies indicate that distensibility decreases with age and during the menopausal transition [26] and that HT may improve distensibility during the menopausal transition [27]. Acute intravenous injection of estradiol rapidly increases distensibility in postmenopausal women [28], but some clinical trials have not demonstrated an effect of HT on distensibility [29, 30]. Our prospective analysis of distensibility was limited to fewer women and shorter follow-up than for CIMT, warranting a larger replication study to confirm this unexplained finding.
Estrogens are also important immune system modulators [31]. We observed that current HT use was associated with lower serum soluble CD163, a marker of macrophage activation and predictor of mortality in HIV [32]. However, sample size for this analysis was small and the association became attenuated with covariate adjustment. Prior studies in non-HIV women have reported reduced natural killer cell cytotoxicity and lower proinflammatory cytokines with HT use [33–35]. In the context of HIV, estrogens inhibit HIV transcription and maintain HIV latency [36], and downregulate the macrophage response to HIV infection [37]. In vitro effects of estrogen on HIV replication and response are consistent with our finding, albeit nonsignificant, of reduced immune activation with HT.
Our observation that ever HT use was associated with lower serum D-dimer was unexpected, given the well-known increased risk of VTE in postmenopausal women taking HT [38]. D-dimer is a fibrin degradation product and direct biomarker of thrombosis; it is used in VTE diagnosis and also predicts future VTE events [39]. D-dimer has been associated with HIV viremia and is a predictor of mortality in people with HIV [40], but has not been consistently associated with CIMT [41, 42]. It is possible that inhibitory effects of estrogen on HIV replication lead to lower D-dimer and outweigh thrombotic effects of estrogen in women with HIV; or there may be residual confounding. While it may be encouraging that HT use was not associated with higher D-dimer in women with HIV, the conflict of our result with studies from the general population necessitate confirmation in other HIV populations, especially since our sample size for this analysis was small.
Our study was limited by lack of data on duration of HT use, and the crude assessment of HT, which did not differentiate estrogen formulation, dose, or route; the inability to distinguish systemic vs topical estrogens limits causal inference, as systemic absorption of vaginal estrogens is variable and can be very low [43]. Additionally, as most women in this study were <60 years of age, we were unable to examine heterogeneity by age (ie, the HT timing hypothesis). Last, we cannot rule out the influence of residual confounding by the “healthy woman effect” (ie, that health conscious women are more likely to use HT and engage in health-promoting behaviors such as healthful diet and exercise, leading to better cardiovascular health). Alternatively, given that some indications for HT (eg, vasomotor symptoms, premature menopause) are associated with higher CVD risk [44, 45], any confounding by such indications would bias our findings toward a harmful rather than protective association.
Currently, use of menopausal HT is indicated for vasomotor symptoms, genitourinary syndrome of menopause, premature hormonal decline, and prevention of bone loss, for women <60 years old or within 10 years of menopause [46]. However, in peri- and postmenopausal women with HIV, uptake of HT is extremely low despite high prevalence of indications for use, since providers often do not address HT use in women with HIV [10]. As such, better menopause care including HT use has recently been encouraged for women with HIV, with providers urged to review indications, contraindications, and risk of adverse events with patients [10]. Our study begins to fill a critical knowledge gap in menopause care for women with HIV, namely, whether menopausal HT impacts atherosclerotic heart disease in women with HIV. Importantly, our results that suggest HT may confer cardiovascular benefit in women with HIV by reducing subclinical atherosclerosis, a predictor of future clinical cardiovascular events. Additional observational studies and randomized trials are sorely needed to further fill gaps in knowledge regarding effects of menopausal HT in women with HIV, especially concerning risk of VTE, which is elevated in people with HIV [47].
Supplementary Data
Supplementary materials are available at Clinical Infectious Diseases online. Consisting of data provided by the authors to benefit the reader, the posted materials are not copyedited and are the sole responsibility of the authors, so questions or comments should be addressed to the corresponding author.
Supplementary Material
Contributor Information
Brandilyn A Peters, Department of Epidemiology and Population Health, Albert Einstein College of Medicine, Bronx, New York, USA.
David B Hanna, Department of Epidemiology and Population Health, Albert Einstein College of Medicine, Bronx, New York, USA.
Anjali Sharma, Department of Medicine, Albert Einstein College of Medicine, Bronx, New York, USA.
Kathryn Anastos, Department of Medicine, Albert Einstein College of Medicine, Bronx, New York, USA.
Donald R Hoover, Department of Statistics and Institute for Health, Health Care Policy and Aging Research, Rutgers University, New Brunswick, New Jersey, USA.
Qiuhu Shi, School of Health Sciences and Practice, New York Medical College, Valhalla, New York, USA.
Caitlin A Moran, Department of Medicine, Emory University School of Medicine, Atlanta, Georgia, USA.
Elizabeth A Jackson, Department of Medicine, University of Alabama at Birmingham, Birmingham, Alabama, USA.
Maria L Alcaide, Department of Medicine, University of Miami Miller School of Medicine, Miami, Florida, USA.
Igho Ofotokun, Department of Medicine, Emory University School of Medicine, Atlanta, Georgia, USA.
Adaora A Adimora, Department of Medicine, UNC School of Medicine, University of North Carolina at Chapel Hill, Chapel Hill, North Carolina, USA.
Sabina A Haberlen, Department of Epidemiology, Johns Hopkins Bloomberg School of Public Health, Baltimore, Maryland, USA.
Mardge Cohen, Department of Medicine, Stroger Hospital, Cook County Health and Hospital System, Chicago, Illinois, USA.
Phyllis C Tien, Department of Medicine, University of California, San Francisco, San Francisco, California, USA; Department of Veterans Affairs Medical Center, San Francisco, California, USA.
Katherine G Michel, Department of Medicine, Georgetown University, Washington, District of Columbia, USA.
Steven R Levine, Departments of Neurology and Emergency Medicine, State University of New York Downstate Health Sciences University, Brooklyn, New York, USA.
Howard N Hodis, Departments of Medicine and Population and Public Health Sciences, Atherosclerosis Research Unit, Keck School of Medicine, University of Southern California, Los Angeles, California, USA.
Robert C Kaplan, Department of Epidemiology and Population Health, Albert Einstein College of Medicine, Bronx, New York, USA; Fred Hutchinson Cancer Research Center, Division of Public Health Sciences, Seattle, Washington, USA.
Michael T Yin, Department of Medicine, College of Physicians and Surgeons, Columbia University Medical Center, New York, New York, USA.
Notes
Disclaimer. The contents of this publication are solely the responsibility of the authors and do not represent the official views of the National Institutes of Health (NIH).
Financial support. B. A. P., D. B. H., C. A. M., and R. C. K. were supported by the National Heart, Lung, and Blood Institute of the NIH (K01HL160146 to B. A. P., K01HL137557 to D. B. H., K23HL152903 to C. A. M., and R01HL148094 to R. C. K.). K. G. M. was supported by the National Center for Advancing Translational Sciences (KL2TR001432). Data in this manuscript were collected by the Women’s Interagency HIV Study (WIHS), now the Multicenter AIDS Cohort Study/WIHS Combined Cohort Study (MWCCS). MWCCS (Principal Investigators): Atlanta Clinical Research Site (CRS) (Ighovwerha Ofotokun, Anandi Sheth, and Gina Wingood), U01-HL146241; Baltimore CRS (Todd Brown and Joseph Margolick), U01-HL146201; Bronx CRS (Kathryn Anastos and Anjali Sharma), U01-HL146204; Brooklyn CRS (Deborah Gustafson and Tracey Wilson), U01-HL146202; Data Analysis and Coordination Center (Gypsyamber D’Souza, Stephen Gange, and Elizabeth Golub), U01-HL146193; Chicago-Cook County CRS (Mardge Cohen and Audrey French), U01-HL146245; Chicago-Northwestern CRS (Steven Wolinsky), U01-HL146240; Northern California CRS (Bradley Aouizerat, Jennifer Price, and Phyllis Tien), U01-HL146242; Los Angeles CRS (Roger Detels and Matthew Mimiaga), U01-HL146333; Metropolitan Washington CRS (Seble Kassaye and Daniel Merenstein), U01-HL146205; Miami CRS (Maria Alcaide, Margaret Fischl, and Deborah Jones), U01-HL146203; Pittsburgh CRS (Jeremy Martinson and Charles Rinaldo), U01-HL146208; University of Alabama at Birmingham (UAB)–Mississippi CRS (Mirjam-Colette Kempf, Jodie Dionne-Odom, and Deborah Konkle-Parker), U01-HL146192; and University of North Carolina (UNC) CRS (Adaora Adimora and Michelle Floris-Moore), U01-HL146194. The MWCCS is funded primarily by the National Heart, Lung, and Blood Institute, with additional co-funding from the Eunice Kennedy Shriver National Institute of Child Health and Human Development, National Institute on Aging (NIA), National Institute of Dental and Craniofacial Research, National Institute of Allergy and Infectious Diseases, National Institute of Neurological Disorders and Stroke, National Institute of Mental Health, National Institute on Drug Abuse, National Institute of Nursing Research, National Cancer Institute, National Institute on Alcohol Abuse and Alcoholism, National Institute on Deafness and Other Communication Disorders, National Institute of Diabetes and Digestive and Kidney Diseases, and National Institute on Minority Health and Health Disparities, and in coordination and alignment with the research priorities of the NIH, Office of AIDS Research. MWCCS data collection is also supported by UL1-TR000004 (University of California, San Francisco Clinical and Translational Science Award), UL1-TR003098 (Johns Hopkins University Institute for Clinical and Translational Research), UL1-TR001881 (University of California, Los Angeles Clinical & Translational Science Institute), P30-AI-050409 (Atlanta Center for AIDS Research [CFAR]), P30-AI-073961 (Miami CFAR), P30-AI-050410 (UNC CFAR), P30-AI-027767 (UAB CFAR), and P30-MH-116867 (Miami Center for HIV and Research in Mental Health). The authors gratefully acknowledge the contributions of the study participants and dedication of the staff at the MWCCS sites.
References
- 1. Stampfer MJ, Colditz GA. Estrogen replacement therapy and coronary heart disease: a quantitative assessment of the epidemiologic evidence. Prev Med 1991; 20:47–63. [DOI] [PubMed] [Google Scholar]
- 2. El Khoudary SR, Aggarwal B, Beckie TM, et al. Menopause transition and cardiovascular disease risk: implications for timing of early prevention: a scientific statement from the American Heart Association. Circulation 2020; 142:e506–32. [DOI] [PubMed] [Google Scholar]
- 3. Hodis HN, Mack WJ, Shoupe D, et al. Methods and baseline cardiovascular data from the early versus late intervention trial with estradiol testing the menopausal hormone timing hypothesis. Menopause 2015; 22:391–401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Hodis HN, Mack WJ, Henderson VW, et al. Vascular effects of early versus late postmenopausal treatment with estradiol. N Engl J Med 2016; 374:1221–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Boardman HM, Hartley L, Eisinga A, et al. Hormone therapy for preventing cardiovascular disease in post-menopausal women. Cochrane Database Syst Rev 2015; 3:CD002229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Stone L, Looby SE, Zanni MV. Cardiovascular disease risk among women living with HIV in North America and Europe. Curr Opin HIV AIDS 2017; 12:585–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Hunt PW, Lee SA, Siedner MJ. Immunologic biomarkers, morbidity, and mortality in treated HIV infection. J Infect Dis 2016; 214(Suppl 2):S44–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Karim R, Mack WJ, Kono N, et al. Gonadotropin and sex steroid levels in HIV-infected premenopausal women and their association with subclinical atherosclerosis in HIV-infected and -uninfected women in the Women’s Interagency HIV Study (WIHS). J Clin Endocrinol Metab 2013; 98:E610–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. King EM, Kaida A, Mayer U, et al. Undertreated midlife symptoms for women living with HIV linked to lack of menopause discussions with providers. J Acquir Immune Defic Syndr 2022; 89:505–10. [DOI] [PubMed] [Google Scholar]
- 10. King EM, Prior JC, Pick N, et al. Menopausal hormone therapy for women living with HIV. Lancet HIV 2021; 8:e591–8. [DOI] [PubMed] [Google Scholar]
- 11. Cushman M, Larson JC, Rosendaal FR, et al. Biomarkers, menopausal hormone therapy and risk of venous thrombosis: the Women’s Health Initiative. Res Pract Thromb Haemost 2018; 2:310–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Adimora AA, Ramirez C, Benning L, et al. Cohort profile: the Women’s Interagency HIV Study (WIHS). Int J Epidemiol 2018; 47:393–4i. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Hanna DB, Post WS, Deal JA, et al. HIV Infection is associated with progression of subclinical carotid atherosclerosis. Clin Infect Dis 2015; 61:640–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Peters BA, Moon JY, Hanna DB, et al. T-cell immune dysregulation and mortality in women with human immunodeficiency virus. J Infect Dis 2022; 225:675–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Hanna DB, Lin J, Post WS, et al. Association of macrophage inflammation biomarkers with progression of subclinical carotid artery atherosclerosis in HIV-infected women and men. J Infect Dis 2017; 215:1352–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Takahashi K, Tanaka E, Murakami M, et al. Long-term hormone replacement therapy delays the age related progression of carotid intima-media thickness in healthy postmenopausal women. Maturitas 2004; 49:170–7. [DOI] [PubMed] [Google Scholar]
- 17. Tremollieres FA, Cigagna F, Alquier C, Cauneille C, Pouilles J-M, Ribot C. Effect of hormone replacement therapy on age-related increase in carotid artery intima-media thickness in postmenopausal women. Atherosclerosis 2000; 153:81–8. [DOI] [PubMed] [Google Scholar]
- 18. Hodis HN, Mack WJ, Lobo RA, et al. Estrogen in the prevention of atherosclerosis. Ann Intern Med 2001; 135:939–53. [DOI] [PubMed] [Google Scholar]
- 19. Naqvi TZ, Lee MS. Carotid intima-media thickness and plaque in cardiovascular risk assessment. JACC Cardiovasc Imaging 2014; 7:1025–38. [DOI] [PubMed] [Google Scholar]
- 20. Soares C, Samara A, Yuyun MF, et al. Coronary artery calcification and plaque characteristics in people living with HIV: a systematic review and meta-analysis. J Am Heart Assoc 2021; 10:e019291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Mosepele M, Hemphill LC, Moloi W, et al. Pre-clinical carotid atherosclerosis and sCD163 among virally suppressed HIV patients in Botswana compared with uninfected controls. PLoS One 2017; 12:e0179994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Lucas GM, Atta MG, Fine DM, et al. HIV, cocaine use, and hepatitis C virus: a triad of nontraditional risk factors for subclinical cardiovascular disease. Arterioscler Thromb Vasc Biol 2016; 36:2100–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Hanna DB, Moon JY, Haberlen SA, et al. Carotid artery atherosclerosis is associated with mortality in HIV-positive women and men. AIDS 2018; 32:2393–403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Janjua SA, Staziaki PV, Szilveszter B, et al. Presence, characteristics, and prognostic associations of carotid plaque among people living with HIV. Circ Cardiovasc Imaging 2017; 10:e005777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Menazza S, Murphy E. The expanding complexity of estrogen receptor signaling in the cardiovascular system. Circ Res 2016; 118:994–1007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Samargandy S, Matthews KA, Brooks MM, et al. Arterial stiffness accelerates within 1 year of the final menstrual period. Arterioscler Thromb Vasc Biol 2020; 40:1001–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Shufelt C, Elboudwarej O, Johnson BD, et al. Carotid artery distensibility and hormone therapy and menopause: the Los Angeles Atherosclerosis Study. Menopause 2016; 23:150–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Kallikazaros I, Tsioufis C, Zambaras P, Stefanadis C, Toutouzas P. Conjugated estrogen administration improves common carotid artery elastic properties in normotensive postmenopausal women. Clin Cardiol 2002; 25:167–72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Angerer P, Kothny W, Störk S, von Schacky C. Hormone replacement therapy and distensibility of carotid arteries in postmenopausal women: a randomized, controlled trial. J Am Coll Cardiol 2000; 36:1789–96. [DOI] [PubMed] [Google Scholar]
- 30. Westendorp IC, de Kleijn MJ, Bots ML, et al. The effect of hormone replacement therapy on arterial distensibility and compliance in perimenopausal women: a 2-year randomised trial. Atherosclerosis 2000; 152:149–57. [DOI] [PubMed] [Google Scholar]
- 31. Markle JG, Fish EN. SeXX matters in immunity. Trends Immunol 2014; 35:97–104. [DOI] [PubMed] [Google Scholar]
- 32. Knudsen TB, Ertner G, Petersen J, et al. Plasma soluble CD163 level independently predicts all-cause mortality in HIV-1–infected individuals. J Infect Dis 2016; 214:1198–204. [DOI] [PubMed] [Google Scholar]
- 33. Deguchi K, Kamada M, Irahara M, et al. Postmenopausal changes in production of type 1 and type 2 cytokines and the effects of hormone replacement therapy. Menopause 2001; 8:266–73. [DOI] [PubMed] [Google Scholar]
- 34. Stopińska-Głuszak U, Waligóra J, Grzela T, et al. Effect of estrogen/progesterone hormone replacement therapy on natural killer cell cytotoxicity and immunoregulatory cytokine release by peripheral blood mononuclear cells of postmenopausal women. J Reprod Immunol 2006; 69:65–75. [DOI] [PubMed] [Google Scholar]
- 35. Saucedo R, Rico G, Basurto L, Ochoa R, Zárate A. Transdermal estradiol in menopausal women depresses interleukin-6 without affecting other markers of immune response. Gynecol Obstet Invest 2002; 53:114–7. [DOI] [PubMed] [Google Scholar]
- 36. Das B, Dobrowolski C, Luttge B, et al. Estrogen receptor-1 is a key regulator of HIV-1 latency that imparts gender-specific restrictions on the latent reservoir. Proc Natl Acad Sci U S A 2018; 115:E7795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Devadas K, Biswas S, Ragupathy V, Lee S, Dayton A, Hewlett I. Modulation of HIV replication in monocyte derived macrophages (MDM) by steroid hormones. PLoS One 2018; 13:e0191916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Canonico M. Hormone therapy and risk of venous thromboembolism among postmenopausal women. Maturitas 2015; 82:304–7. [DOI] [PubMed] [Google Scholar]
- 39. Hansen ES, Rinde FB, Edvardsen MS, et al. Elevated plasma D-dimer levels are associated with risk of future incident venous thromboembolism. Thromb Res 2021; 208:121–6. [DOI] [PubMed] [Google Scholar]
- 40. Borges AH, O’Connor JL, Phillips AN, et al. Factors associated with D-dimer levels in HIV-infected individuals. PLoS One 2014; 9:e90978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Kaplan RC, Landay AL, Hodis HN, et al. Potential cardiovascular disease risk markers among HIV-infected women initiating antiretroviral treatment. J Acquir Immune Defic Syndr 2012; 60:359–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Hanna DB, Guo M, Bůžková P, et al. HIV Infection and carotid artery intima-media thickness: pooled analyses across 5 cohorts of the NHLBI HIV-CVD Collaborative. Clin Infect Dis 2016; 63:249–56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Santen RJ, Mirkin S, Bernick B, Constantine GD. Systemic estradiol levels with low-dose vaginal estrogens. Menopause 2020; 27:361–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Thurston RC, El Khoudary SR, Tepper PG, et al. Trajectories of vasomotor symptoms and carotid intima media thickness in the study of women’s health across the nation. Stroke 2016; 47:12–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Muka T, Oliver-Williams C, Kunutsor S, et al. Association of age at onset of menopause and time since onset of menopause with cardiovascular outcomes. Intermediate vascular traits, and all-cause mortality: a systematic review and meta-analysis. JAMA Cardiol 2016; 1:767–76. [DOI] [PubMed] [Google Scholar]
- 46. The NAMS 2017 Hormone Therapy Position Statement Advisory Panel . The 2017 hormone therapy position statement of the North American menopause society. Menopause 2017; 24:728–53. [DOI] [PubMed] [Google Scholar]
- 47. Bibas M, Biava G, Antinori A. HIV-associated venous thromboembolism. Mediterr J Hematol Infect Dis 2011; 3:e2011030. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.