Abstract
Primary tumors actively and specifically prime pre-metastatic niches (PMNs), the future sites of organotropic metastasis, preparing these distant microenvironments for disseminated tumor cell arrival. While initial studies of the PMN focused on extracellular matrix alterations and stromal reprogramming, it is increasingly clear that the far-reaching effects of tumors are in great part achieved through systemic and local PMN immunosuppression. Here, we discuss recent advances in our understanding of the tumor immune microenvironment and provide a comprehensive overview of the immune determinants of the PMN’s spatiotemporal evolution. Moreover, we depict the PMN immune landscape, based on functional pre-clinical studies as well as mounting clinical evidence, and the dynamic, reciprocal crosstalk with systemic changes imposed by cancer progression. Finally, we outline emerging therapeutic approaches that alter the dynamics of the interactions driving PMN formation and reverse immunosuppression programs in the PMN ensuring early anti-tumor immune responses.
Introduction
The systemic effects of cancer are crucial determinants of patient outcome. Tumor-secreted factors, including tumor-derived extracellular vesicles and particles (EVPs), circulate and alter distant organs, governing metastatic progression, which is responsible for the majority of cancer-related deaths. The concept that tumors have a predilection to metastasize to specific organs was first introduced in Stephen Paget’s ‘seed and soil’ theory of cancer metastasis in 1889. However, only in the 1970s did Isaiah Fidler provide experimental evidence of metastatic organotropism, demonstrating that cancer cell-intrinsic properties dictate preferential homing to specific target sites, independently of their nonspecific distribution following injection1.
Decades later, our lab and others demonstrated that primary tumors induced early changes in the microenvironment of secondary distant organs devoid of cancer cells, thereby providing a milieu favorable for cancer cell settlement and metastasis initiation, creating niches termed ‘pre-metastatic niches’ (PMNs)2,3. The PMN is shaped by interactions between cancer-secreted factors and resident stromal cells at these distant sites as well as bone marrow-derived cells (BMDCs)2. The priming of a distant organ consists of stepwise processes that skew tissue homeostasis towards a dysfunctional environment receptive to circulating tumor cell (CTC) colonization: vascular leakiness4,5, lymphangiogenesis3, extracellular matrix (ECM) remodeling, and the generation of an immunosuppressive microenvironment (Fig. 1).
Figure 1. Pre-metastatic niche formation.
Primary tumors secrete soluble factors, such as cytokines, chemokines, hormones, and metabolites as well as extracellular vesicles and particles (EVPs) which disseminate systemically, reprogramming distant organ microenvironments. Primary tumor-derived factors exert both direct effects, via tumor-derived secreted factors (TDSFs) and EVPs circulating through the hematogenous and lymphatic systems to distant future sites of metastasis and indirect effects, mediated through their impact on non-metastatic organs that then influence host physiology. The indirect factors promote the mobilization of bone-marrow-derived cells (BMDCs), secretion of metabolites by the gut microbiome, and of stress hormones following hypothalamic-pituitary-adrenal (HPA) axis activation. The direct factors alter distant microenvironments by affecting the tissue resident cells, such as endothelial cells and fibroblasts, leading to vascular leakiness and extracellular matrix (ECM) remodeling while the indirect effects induce the recruitment of non-resident immune cells. Together, these changes generate an immunosuppressive and inflammatory environment which includes myeloid-derived suppressor cells (MDSC) BMDCs, such as VEGFR1+ HPCs and CD11b+ myeloid cells, tumor-associated macrophages (TAMs), tumor-associated neutrophils (TANs), T regulatory (Treg) and B regulatory (Breg) cells, γδT cells, dysfunctional dendritic cells (DCs) and natural killer (NK) cells, suppressed CD4+/CD8+ T cells, and cancer-associated fibroblasts (CAFs). The reprogrammed microenvironment then attracts disseminated tumor cells to home and colonize this permissive soil. The most common PMN sites studied to date are the LN, lung, liver, omentum, bone, and brain.
This review offers a holistic overview of the immune determinants driving the PMN’s spatiotemporal evolution (Fig.2), clinical evidence of PMN and methods for clinical detection of PMN in patients, exploring the impact of current therapies on PMN formation, and potential therapeutic strategies to promote anti-tumor immunity throughout PMN evolution (Fig. 3).
Figure 2. Pre-metastatic niche evolution.
Tumor-derived factors such as TDSF, pro-angiogenic, pro-inflammatory factors, and EVPs trigger the formation of an early PMN characterized by ECM remodeling and inflammatory changes that reprogram the tissue-resident immune and stromal cells in distant organs that orchestrate the PMN initiation. The mid PMN, is characterized by increased fibronectin deposition and upregulation of tenascin C, enhance ECM deposition and remodeling and vascular permeability that facilitates BMDCs recruitment. In the late PMN, local secretion of inflammatory and chemotactic molecules reinforce the continued recruitment of hematopoietic progenitor cells and immature myeloid cells, neutrophils, macrophages, Tregs, and dysfunctional DCs and NK cells that crosstalk with the PMN resident cells to maintain an inflammatory and immunosuppressive circuitry. The resulting ECM modifications accompanied by enhanced immune cell recruitment, persistent inflammation, and immunosuppression support CTC adhesion and extravasation, and CTC outgrowth in the Metastatic niche.
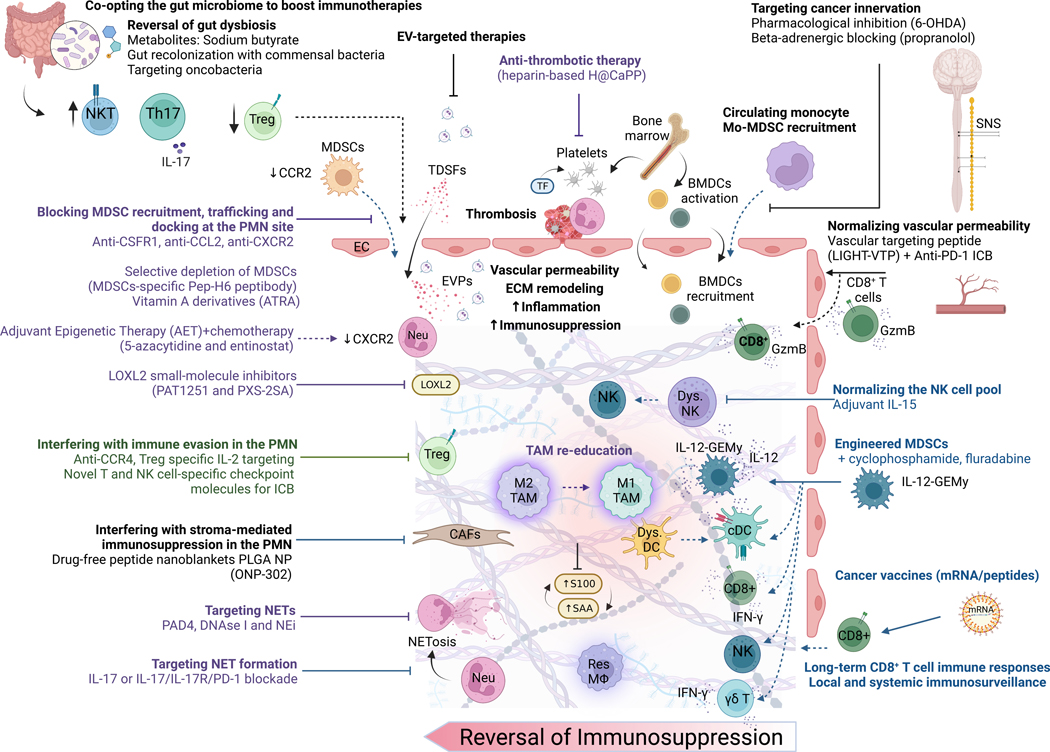
Figure 3. Therapeutic strategies to target the PMN and associated immunosuppression.
Direct interference with innate immunity-mediated PMN formation (purple) includes platelet targeting, blocking of myeloid cell recruitment, trafficking or docking at the PMN site, TAM re-education, MDSC depletion, and NET formation. Direct interference with adaptive immunity-mediated PMN formation (green) employs immune checkpoint blockade molecules on T and NK cells, as well as specific blockade of Tregs. Reversal of PMN immunosuppression by co-opting the anti-tumor immunity (blue) include the use of engineered MDSCs, normalization of the NK cell pool, and the use of cancer vaccines to enhance long-term CD8+ T cell immune responses. Therapies indirectly targeting immunity (black) that result in reduced immune evasion and boost immunotherapies include the normalization of vascular permeability, targeting pro-metastatic effects of CAFs in the PMN, targeting cancer innervation, co-opting the gut microbiome to exert anti-metastatic immunomodulation, and EV-based therapies.
Pre-metastatic niche development and evolution
Organotropism: Paget’s hypothesis solved
Organotropism represents the propensity of cancer cells to metastasize to PMNs in specific organs (Fig.1D)1,2 and is driven by selective interactions between cancer cells, their secretome, and the cellular and extracellular microenvironmental components of the distant future metastatic organ. While some organs, such as the lymph nodes (LNs), bones, lungs, omentum, brain, and liver, are more susceptible to metastasis, other organs, such as the muscle6 and kidney7 are rarely targeted by metastasis (Box 1). Furthermore, both cancer type (e.g., pancreatic vs breast) and subtype (e.g. luminal vs. basal breast cancer) dictate the particular organ tumor cells invade8,9. Metastatic site specificity was evident in the earliest PMN studies, as tumor-secreted VEGF selectively induced MMP-9 expression in lung endothelial cells and macrophages10. Tumor-secreted factors also simultaneously mobilized VEGFR1+ very late antigen–4+ (VLA-4/integrin α4β1) bone marrow-derived hematopoietic progenitor cells (HPCs) and upregulated fibronectin, an integrin α4β1 ligand, in selective PMNs, such as the lung, which cooperated to recruit BMDCs to the altered distant sites2. Thus, cancer cell integrin expression is a critical mediator of organotropism: β1 and β3 integrins mediate bone metastasis11,12 while integrin α3β1 correlates with lung colonization13. Integrin α5 overexpression in either early-stage primary breast cancer (BC) or bone metastases, but not lung, liver, or brain metastases, correlated with osteoclast-mediated bone resorption and colonization by CTCs14. The key role of integrins in mediating lymphocyte homing highlights PMN dependence on these adhesion molecules. Lymphocyte function-associated antigen 1 (LFA-1/integrin αLβ2) and integrin α4β1 are crucial for leukocyte rolling and arrest in blood vessels15,16. Myeloid cells use integrins LFA-1, α4, and β2 to interact with the vasculature while neutrophil extravasation requires LFA-1 for attachment and integrin β2 for crawling to an endothelial junction17,18.
Box 1: Anti-metastatic niches
Certain tissue microenvironments, collectively referred to as “anti-metastatic niches,” are inhospitable to DTCs279. Clinical meta-analysis of autopsies revealed that, paradoxically, arterial output of the heart in tissues does not correlate with their colonization by DTCs280–282, thereby suggesting, yet again, that the “soil” of the target organ and not hematogenous access, determines metastatic outcome. However, few studies have explored the specific features of these “soils” that reject metastatic colonization. Proposed mechanisms for DTC outgrowth suppression in the four anti-metastatic niches (i.e., skeletal, spleen, thyroid, and yellow bone marrow) are reviewed elsewhere279.
Despite seeding by DTCs, tumors do not grow in skeletal muscle. Mechanistically, the intrinsic sustained oxidative stress in muscle imposes a profound “metabolic bottleneck” on DTCs, which are not molecularly equipped to overcome such environmental challenges. Functionally, disruption of redox homeostatic balance toward ROS production can impair DTC outgrowth in the lung, whereas the enhancement of anti-oxidant enzymatic tumor cell defense mechanisms (e.g., catalase ectopic expression) allowed DTCs to colonize the skeletal muscle6.
Similar mechanisms driving DTC death via metabolic restraints may also exist in the spleen and thyroid: blood filtration in the spleen is a source of redox-active iron that could promote ROS-mediated lipid peroxidation and DNA damage or ferroptosis283,284, while the thyroid gland stores the majority of body iodine, a highly oxidizing element leading to ROS production285. Moreover, the spleen and thyroid are also immune-rich niches populated by mature immune cells, which may allow early anti-tumor surveillance and hinder DTC outgrowth. Thus, the outstanding question is: given our knowledge of immunometabolism and the crosstalk between exercise and cancer prevention, do specific immunosurveillance mechanisms contribute to the skeletal muscle and other, yet to be characterized, anti-metastatic niches? Understanding the underlying mechanisms responsible for supporting inhospitable, anti-metastatic niches could lead to new therapeutic strategies to prevent PMN formation and metastasis.
Box 2: Anti-PMN innate immunity
While most studies report PMN-promoting functions of innate immunity, some evidence of PMN-suppressing effects have been suggested. For example, EVPs from non-metastatic cell lines induce the development of anti-PMN patrolling monocytes. These “non-metastatic” exosomes stimulated the expansion of Ly6Clo patrolling monocytes in the bone marrow, which upon PMN arrival caused cancer cell clearance via the recruitment of NK cells and TRAIL-dependent killing of melanoma cells by macrophages286. These findings unveil a novel view of metastatic organotropism, in which the non-metastatic organs may in fact have an active anti-PMN function. In another study, B220+CD11b+NK1.1+ liver-entrained leukocytes travelled to the pre-metastatic site and efficiently blocked deposition of FN, an essential ECM component of the PMN, through coagulation factor X and thrombospondin expression287. Lastly, tumor-entrained neutrophils could also inhibit metastatic seeding in the lungs by generating H2O243. These findings highlight the dynamic nature of the PMN and the potential for anti-PMN treatments by tipping the balance in favor of anti-tumor reponses.
Box 3: PMN modulation by Diet, Exercise, and Aging
An intriguing but understudied area is how systemic changes in homeostasis influence the formation of PMN-like tissue microenvironments. Aging is the most significant risk factor for cancer overall, as a result of various molecular and cellular processes that progress over time. Interestingly, “aged” tissues resemble PMNs, and may serve as pre-existing “fertile soils”, explaining the higher cancer incidence in the elderly population. inflammaging, hampered DNA damage repair mechanisms, and accumulation of epigenetic modifications and somatic mutations. “Aged” tissues are characterized by an inflammatory49 and immunosuppressive288,289 state which may be a critical contributor to PMN evolution. These similarities imply that lessons from studying the elderly population may shed light on parallel PMN mechanisms which are often less accessible and less prevalent.
Exercise, diet, and other lifestyle factors, such as smoking and alcohol consumption, may also modulate PMN progression. PMN formation have been associated with high cholesterol, obesity, and high fat diet179,188,193,201,290. Several metabolic changes associated with obesity correlate with cancer 290,193). Conversely, dietary restriction enhances T cell fitness and protects against tumors 291,292. Smoking and alcohol consumption induce chronic inflammation and can exert detrimental effects on the immune system and recent studies showed that chronic nicotine exposure supports lung PMNs225. Extended lifespans and low cancer incidence in Blue Zones support the idea that healthy diets and lifestyles can reduce cancer incidence.
Exercise boosts the immune system and prevents cancer recurrence and metastasis293,294. Exercise-induced metabolic changes, as well as muscle cell-derived EVPs are likely to play a role in reprogramming distal PMNs295. Exercise facilitates immune cell infiltration/homing at the primary tumor or PMN, thereby augmenting the efficacy of immunotherapies293,296,297. Given that in terms of cancer progression, metastasis prevention has the highest potential to impact outcome, nutrition and exercise as tools to minimize inflammation associated with aging are promising interventions to affect PMN development that should be further studied294.
Integrins on EVPs also direct organotropism by their specific interactions with cells and ECM molecules in PMNs19,20. For example, laminin-binding integrins α6β4 and α6β1 are enriched in lung-tropic EVPs, RGD-motif binding integrin αvβ5 in liver-tropic EVPs, and integrin β3 in brain-tropic EVPs21, and α5 integrin, partnering with cadherin 11, in bone-tropic EVPs22. Importantly, the integrin profiles of plasma-derived EVPs from cancer patients were consistent with pre-clinical model findings and, indeed, predicted future metastatic patterns11,19. As tumor-derived EVPs release precedes CTC arrival during PMN formation, differential EVP integrin expression essentially dictates the site prepared for metastasis, and could be targeted to control metastasis. PMN-promoting alterations supporting metastatic organotropism include cellular, extracellular, and structural features of the affected sites20,23 and these functions are organ-specific. At the cellular level, platelet depletion reduced lung metastasis, but enhanced LN metastases24. ECM components are critical for organotropism to PMN20. Proteomic analysis of ECM composition of brain, lung, liver, and bone marrow BC metastatic niches, as well as loss of function studies of ECM modulators, revealed that distinct molecules comprise and regulate spread to each metastatic site25. Tissue architecture may also contribute to PMN organotropism: lungs, for example, have dense capillary beds and serve as a lymphatic drainage reservoir, properties which enhance the probability of CTC arrest and extravasation23.
To enable the systematic investigation of key biological features of PMN and organotropism, 3D models that mimic tissue complexity were bioengineered26–28. Hydrogel scaffolds seeded with human peripheral blood mononuclear cells and subdermally implanted in mice induced vascularized and pro-inflammatory microenvironments that recruited disseminated tumor cells (DTCs) from an orthotopic prostate tumor xenograft28. These bioengineered PMNs advance our understanding of the biological processes underlying PMN formation and organotropism and allow high-throughput testing of anti-tumor therapies.
PMN induction by tumor-secreted factors
Tumor-derived soluble factors (TDSFs)
PMN establishment requires the combined systemic effects of a wide variety of soluble factors: cytokines, growth factors, enzymes, and EVPs secreted by the primary tumor and its milieu. Primary tumors secrete key pro-angiogenic factors VEGF-A and placental growth factor 2 that mobilize and recruit VEGFR1+VLA-4+ BM-derived HPCs to fibronectin-rich pre-metastatic organs. VEGFR1+ HPC clusters are a common denominator for several pre-metastatic organs in numerous murine and human cancers, highlighting their clinical relevance2. Several TDSFs, including VEGF-A, TNF-α, and TGF-β, induce the expression of inflammatory chemoattractant proteins S100A8 and S100A9, which promote Mac1+ myeloid cell lung PMN infiltration29. Functionally, VEGF secretion by primary BC promotes endothelial cell-focal adhesion kinase (FAK) activation, inducing E-selectin-mediated vascular hyperpermeability foci in the pre-metastatic lung30. Moreover, VEGF-induced PGE2 production in the pre-metastatic lung potentiates tumor cell adhesion and ensures late PMN to metastatic niche transition31.
Other pro-inflammatory growth factors, angiopoietin-like protein 2 (ANGPTL2), G-CSF, IL-6, and CCL2/MCP-1, are critical for PMN formation through their effects on myelopoiesis. ANGPTL2 binds integrin α5β1 and mediates osteosarcoma cell intravasation32, whereas CXCL chemokine production by lung epithelial cells and recruitment of pro-metastatic neutrophils to the pre-metastatic lung support metastasis33. Increased circulating levels of tumor-derived G-CSF by early-stage BC, reprogram BM hematopoiesis toward a myeloid lineage expansion and aberrant differentiation towards immunosuppressive, ROS-producing neutrophils, which then egress into the pre-metastatic lung34.
Secreted by both primary tumor cells and associated stroma, IL-6 promotes PMN formation via STAT3 activation in both tumor cells and tumor-infiltrating myeloid cells, facilitating intravasation and trafficking to the lung PMN35. Tumor-secreted IL-6 induces expression of HIF-1α, VEGF, and CCL5 in lymphatic endothelial cells, thereby promoting angiogenesis and CTC lung and LN colonization36. IL-6 secreted by pancreatic cancer α-SMA+ cancer-associated fibroblasts (CAFs) also induced liver PMN via STAT3 signaling in hepatocytes through production of serum amyloid A (SAA) 1, SAA2, FN, and collagen I leading to Ly6G+ myeloid cell recruitment37.
Tumor-derived CCL2 is a potent chemoattractant for monocytes, macrophages, memory T lymphocytes, and natural killer (NK) cells38. Elevated systemic CCL2 levels were associated with poor prognosis in BC patients39, potentially due to preferential recruitment of CCR2+ inflammatory monocytes to pulmonary metastases40. Moreover, CCL2 recruited mature macrophages that favored homing and outgrowth of tumor cells in the pre-metastatic lung and enhanced osteoclast differentiation in the pre-metastatic bone41. CCL2 derived from hypoxic primary tumors also promoted lung PMN infiltration by dysfunctional myeloid and NK cells with decreased capacity to eliminate incoming CTCs42. Interestingly, CCL2 can also promote anti-tumor responses, by recruiting to the PMN tumor-entrained neutrophils that exert cytotoxic effects and interfere with tumor cell seeding43.
Tumor-derived EV-driven immunosuppression in the PMN
EVPs mediate intercellular communication by transporting proteins, lipids, nucleic acids, and metabolites, orchestrating a complex crosstalk between innate and adaptive immune cell types in the PMN and systemically, ultimately supporting the formation of an inflammatory and immunosuppressive microenvironment conducive to tumor growth44–49. Of note, EVPs are extremely heterogeneous in biogenesis, cargo, size, and function, including newly discovered amembraneous extracellular particles, such as exomeres50–52 and supermeres53, both of which have yet unexplored, distinct contributions to PMN formation and immunosuppression.
Seminal studies revealed that melanoma-derived EVPs reprogram BM progenitor cells towards pro-tumorigenic phenotypes that migrate to the PMN47. In melanoma patients, primary tumor-derived EVPs package multiple immune-modulatory proteins, including S100A9, which travel via lymphatics to sub-capsular macrophages in the pre-metastatic sentinel LNs where they inhibit DC dendritic cell (DC) maturation46,54. Melanoma EVPs transfer MHC class I molecules to antigen-presenting cells (APCs), downregulating CD86 and CD40 co-stimulatory molecules and suppressing DC function55. Smilarly, EVP uptake by monocytic cells and DCs in ovarian ascites induced inflammatory mediators, i.e., IL-6, IL-1β, and TNF-α, thereby generating an immunosuppressive environment56.
Tumor-derived EVPs also induce suppression of T cell function in the PMN. Ex vivo, they reduced proliferation and enhanced apoptosis of CD8+ T cells by downregulating CD69, suppressing CD4+ T cell activation, and inducing regulatory T cell (Treg) expansion57–59. Ovarian cancer-derived EVPs suppressed T cell function both directly by GD3 ganglioside production48,60 and indirectly through production of immune-suppressive adenosine by Treg cells57. Disease activity directly correlated with tumor-derived EVP-dependent CD8+ T cell apoptosis, decreased CD4+ T cell proliferation, elevated Treg function, and NK suppression61. Importantly, immune checkpoint molecules packaged in EVPs contribute to PMN immune suppression, a finding with critical implications for immunotherapy outcomes. EVPs derived from plasma of head and neck cancer patients carried active PD-L1, which inhibited activated T cells and correlated with LN metastasis62. Consistent with these findings, administration of PD-L1+FasL+ tumor-derived EVPs at a pre-malignant stage promoted disease progression and reduced immune infiltration in oral squamous cell carcinoma63. The role of PD-L1-expressing EVPs in PMN formation remains to be evaluated. Taken together these data reveal that tumor-derived EVPs induce PMN immunosuppression through multiple mechanisms impacting both innate and adaptive immunity, as well as their crosstalk, and can serve as biomarkers of disease activity and adaptive immune dysfunction.
PMN hallmarks: through the lens of immunity
Vascular permeability and lymphangiogenesis
Vascular permeability is a hallmark of PMN formation64, where prostaglandins, nitric oxide (NO), matrix metalloproteinases (MMPs), VEGF, and cytokines disrupt endothelial cell junctions, leading to vascular permeabilization5,65, enabling extravasation of immune and cancer cells into the tissue. In turn, tumor-secreted VEGF further supports vascular permeability by recruiting tumor-entrained VEGFR1+ BMDCs. Several MMPs, such as MMP−1, −2, −3, and −10, upregulated in the PMN, interact with angiopoietin-2 and cyclooxygenase-2 (COX-2) to promote vascular permeability5,66. Immune cells also contribute to vascular permeability, through activation of innate immune cell CCL2 signaling that upregulates secretion of SAA3 and S100A8 permeability factors67. Perivascular macrophages induce permeabilization and metastasis in the brain68 and the lung via tenascin C, NO, and TNF secretion69. Neutrophils also contribute to vascular instability through the production of neutrophil extracellular traps (NETs), networks of extracellular DNA containing histones and enzymes designed to entrap and destroy pathogens. NETs anchor to the vessel wall and release neutrophil elastase (NE) that proteolytically disrupts vascular integrity70,71. Moreover, activation of platelets via tumor-derived CD97 leads to granule secretion and ATP release, disrupting endothelial junctions72. Lastly, CD4+ T cell inhibition prevents vessel normalization73 while secretion of IL-17A by γδ T cells promotes tumor-endothelial transmigration74, supporting a role for circulating T cells in shaping vascular leakiness.
EVPs are also potent promoters of vascular leakiness in the lung and brain19,47,75. Brain metastatic cell-derived EVPs carrying cell migration-inducing and hyaluronan-binding protein (CEMIP) induced endothelial cell branching and inflammation in the perivascular niche by upregulating the prostaglandin-endoperoxide synthase 2 (Ptgs2), Tnf, and Cxcl genes encoding pro-inflammatory, pro-metastatic cytokines75. Breast cancer EVPs also promoted brain metastases by transferring the microRNA miR-181c to endothelial cells, altering actin localization via PDPK1 downregulation, leading to blood-brain barrier disruption76.
Lymphatic alterations have also been implicated in PMN formation. In triple-negative breast cancer (TNBC), tumor-secreted IL-6-induced CCL5 expression on lymphatic endothelial cells, promoting cancer cell migration to the lung and LN PMN36. Furthermore, lymphatic ablation increased the frequency of immunosuppressive cells in the PMN, such as regulatory T cells (Tregs) and myeloid-derived suppressor cells (MDSCs)77,78. Thus, both blood and lymphatic vasculature normalization would be required to inhibit PMN formation.
PMN-associated thrombosis
Thrombosis is an early PMN feature orchestrated by platelets that promote ECM remodeling and vascular dysfunction, leading to inflammation and immunosuppression. The coagulation cascade, its interactions with tumor cells and TDSFs are critical for PMN formation and seeding by CTCs. Tumor cells directly promote coagulation via secretion of tissue-factor (TF) and EVPs79 that induce platelet activation and aggregation through coagulation factor VII, triggering a proteolytic cascade that generates thrombin80. Platelet-derived thrombin catalyzes fibrinogen to fibrin conversion and crosslinking to fibronectin, resulting in platelet aggregation and thrombus formation. This initiates a feed-forward loop, where PMN fibronectin deposition acts as a scaffold for microthrombi that then recruit macrophages, creating a platform for CTC arrest and survival in the lung PMN81, protecting CTCs from hemodynamic shear stress and patrolling NK cells. Importantly, platelet activation in the PMN is indirectly regulated by the activation of the adenosine diphosphate (ADP) receptor, as P2Y12 loss attenuates fibronectin deposition, VEGFR1+ BMDC recruitment, and lung metastasis82. Conversely, platelet-derived cytokines, such as thrombospondin-1 and TGF-β1, mediate osteoclast differentiation and BMDC mobilization to the PMN in bone-tropic cancers83.
ECM remodeling in the PMN
ECM remodeling is the defining feature of PMN development. ECM components, such as fibronectin, versican, collagens, and periostin, are modified by resident fibroblast and endothelial cells, as well as by resident and recruited immune cells84. Hypoxia-induced matrix-degrading enzymes of the lysyl oxidase (LOX) family play pivotal roles in PMN collagen remodeling and ECM preconditioning to allow immune cell recruitment42,85. Importantly, systemic release of LOX, LOX-like (LOXL), and FN1 into the circulation by primary BC favors CD11b+ myeloid cell accumulation in the pre-metastatic lung86 and bone, ensuring adhesion and formation of osteolytic lesions, respectively87. Alternatively, HIF-1-mediated upregulation of carbonic anhydrase IX (CAIX) induced G-CSF production downstream of NF-kB activation and subsequent mobilization of granulocytic MDSCs to the lung PMN88. Another ECM-remodeling molecule, TIMP-1, induces specific proteolytic cleavage of MMPs and controls their local activities89. High systemic levels of TIMP-1 mediated SDF-1/CXCR3-dependent recruitment of neutrophils to the pre-metastatic liver and TIMP-1 upregulation was sufficient to promote liver metastasis in various cancers90.
Among stromal cells, fibroblasts are central to ECM deposition and remodeling in the PMN. In the primary tumor microenvironment91,92 fibroblasts are pivotal mediators of immunosuppression, and accumulating evidence suggests that similar interactions occur in the PMN. Following activation by TDSFs, PMN fibroblasts deposit fibronectin, to which incoming BMDCs adhere via integrins2,47,93,94 (Fig. 1, 2). Upon arrival, CD11b+ BMDCs produce MMP-2, which cleaves collagen fibers, thus promoting BMDC recruitment and the colonization and proliferation of CTCs 95. Lung PMN fibroblasts secrete periostin, which also supports MDSC accumulation96 and CTC colonization97. CAFs are also the main source of CXCL12, a CXCR4 ligand, an immunosuppressive signaling axis98 that recruits Tregs to the bone marrow, supporting bone metastasis in prostate cancer patients99. High CXCL12 levels were evident in pre-metastatic lungs of patients with colorectal cancer compared to controls with benign lung disease100. These pleiotropic functions of fibroblasts in supporting an immunosuppressive PMN environment highlight their potential as targets for therapies to hinder PMN formation.
The PMN Immune Landscape and Inflammation
Modulation of immune cell function at pre-metastatic organ sites, (e.g. lung, liver, bone, and LNs) is a critical step in PMN formation. These cells interact with resident stromal cells, such as fibroblasts and endothelial cells, as well as each other, to reprogram the distal microenvironment. A variety of recruited and resident innate and adaptive immune cells shape PMN evolution, immunosuppression, and inflammation (Fig.1). Early changes in the PMN initiate an inflammatory niche that facilitates recruitment of innate immune cells (Fig.1). Subsequent cooperation between recruited cells and factors secreted by remote primary tumors induces changes in resident immune and stromal cells, enhances BMDC chemotaxis to the pre-metastatic organ and establishes a persistent inflammatory yet immune-suppressive PMN circuitry (Fig.2).
Innate cell recruitment to the PMN
Among innate immune populations, recruited myeloid cells are critical drivers of PMN formation2,101 and inflammation. PMN inflammation is shaped by production of damage-associated molecular pattern (DAMP) molecules (e.g., S100 proteins, SAA 3, high-mobility group box1 [HMGB1]102) and expression of their pattern recognition receptors (PRRs) (e.g., receptor for advanced glycation end-products (RAGE) and TLR4103,104) that induce potent STAT335 and NF-kB-mediated103,105 inflammatory signaling, regulating PMN myeloid cell composition and function.
Myeloid cells recruited to the PMN by S100 chemotactic signals subsequently produce S100 proteins in a pro-inflammatory autocrine loop, thereby reinforcing S100 signaling105,106. Through their diverse autocrine and paracrine functions107,108, S100 proteins, small molecular Ca2+ sensors, collectively orchestrate PMN inflammation, mediating the crosstalk between resident cells of the distant organ, recruited immune cells, and CTCs29,103,107,109. S100 proteins are also involved in EV-mediated PMN reprogramming. For example, S100A4 is a tumor EVP-specific DAMP that induces PMN inflammation19,110, while S100A8 and S100A9 are upregulated in the pre-metastatic lung by tumor EVPs19,47 and tumor-secreted VEGF-A, TNF-α, and TGF-β29. Indeed, calprotectin, the S100A8/A9 heterodimer, a ligand/agonist for PRRs, amplifies the production of pro-inflammatory mediators (e.g., SAA3) in the lung PMN to further attract CD11b+ MDSCs103,104. TNF-α accumulated in the PMN then hinders MDSC maturation by regulating S100A8/A9, their receptor RAGE and thus NF-kB signaling105,111. Recently, additional S100A8/A9 receptors were identified, such as basigin (BSG/CD147)112,113, although the role of these interactions in PMN evolution remains unexplored.
Once in the PMN, CD11b+Gr1+ immature myeloid cells downregulate IFN-γ and upregulate pro-inflammatory cytokine and VEGF-A production, facilitating tumor cell extravasation and cancer cell-macrophage interactions114,115. Gr1+CCR2+ inflammatory monocytes recruited to the PMN lung via tumor/stroma-derived CCL2 differentiate into tumor-associated macrophages (TAMs) and further recruit metastasis-promoting macrophages (MAMs) that secrete CSF-1 in a VEGF-A-dependent manner (Table 1)40,116. Meanwhile, pro-tumorigenic M2 TAMs with poor antigen presenting capacity that secrete low IL-12 and high IL-10, IL-4, and IL-13117 differentiated from recruited monocytes reinforce PMN immunosuppression through PD-L1 expression118,119. Moreover, monocytic MDSCs (mo-MDSCs) accumulate in the lung PMN in response to microenvironmental CCL12 chemotactic signals, stimulating endothelial activation, E-selectin expression, CTC arrest, and metastasis via IL-1β secretion120.
Table 1.
The immune landscape of the PMN
| Cell type | PMN site | Function | References |
|---|---|---|---|
| INNATE IMMUNE CELLS | |||
| BMDCs and BM HPCs | LU, LI, K, SPL, LN, BO | Recruitment of BMDCs to FN-rich areas, enhancing further BMDC recruitmment | 2,298 |
| Myeloid cells | |||
| Recruited mo-MDSC | LU | Inflammation, immunosuppression, endothelial activation (IL-1β/E-selectin) and CTC arrest. Recruitment of MAM. | 40,120 |
| Patrolling mo | LU | Immune surveillance | 286 |
| Recruited/res mo, Φ, TAM, Perivascular Φ | LU, BO | Pro-metastatic vascular niche induction, CTC arrest and survival, extravasation, and outgrowth. Secrete MMP-9, S100A8, and FN. Polarized towards an M2 phenotype. | 41,69,81,141,143,148 |
| MAM | LU | VEGFA-dependent CTC extravasation, seeding, proliferation., and regulation of inflammation via secreting CSF-1 | 40,116 |
| Recruited g-MDSC | LU, BR | PMN inflammation and vascular remodeling. Reduced IFN-γ and production of S100A8, S100A9, SAA3, CCL2, MMP-2. | 42,103,114,299 |
| Neutrophils | LU, LI, Peripheral blood | Dysregulate the cytokine mileu via a pro-inflammatory signature: Bv8, MMP-9, S100A8, S100A9, IL-1β, TNF-α, IL-6, and COX-2. Upregulate PD-L1 and impair NK and T cell function. TAN release NETs that promote PMN formation. | 33,90,125,127,136,300 |
| TENs | LU | Inhibit metastatic seeding by exerting cytotoxic effects | 43 |
| Platelets | LU, BO | Promote ECM remodeling, thrombosis and vascular dysfunction, osteoclast differentiation and BMDC mobilization via TGF-β | 72,82, 83 |
| DCs | LU, LN | Inhibit NK cell cytotoxicity, DC dysfunction via COX-2 | 150,151 |
| Tissue-resident macrophages | |||
| Lung-res (AM, IM) | LU | Promote inflammation (TNF-α), ECM degradation (MMP-9), and immunosuppression (PD-L1) | 10,119,137 |
| Kupffer cells | LI | ECM remodeling (FN) mediating BMDC recruitment Promote immunosuppression via reduction of serum IL-2, TNF-α levels and CD4+ T cell levels in the PMN | 44,145,147 |
| Microglia | BR | Polarized towards M2 pro-tumorigenic phenotype Initiate neuroinflammation and regulate cell adhesion | 75,140,141,148 |
| Osteoclasts | BO | Formation of osteolytic lesions and bone resorption, critical for tumor cell seeding and colonization | 14,83,217 |
| Omental-res | PER | Omental CD163+Tim4+ macrophages are critical for PER PMN | 142 |
| ILCs : NK cells | LU, LI | Reduced infiltration and anti-tumor immunity | 42,178 |
| ADAPTIVE IMMUNE CELLS | |||
| CD4+ T cells | LI, BO | Marked reduction of CD4+ T cell numbers, dysfunctional CD4+ T cells and conversion to Tregs with immunosuppressive role | 147,168 |
| T helper 17 (Th17) | LI | Reduced frequency. Production of IL-17. Produce RANKL and induce osteolytic disease before CTC homing | 170,178 |
| Regulatory T cells (Tregs) | LU, BO, LN, LI | Inhibit proliferation and function of T and NK cells Enriched and maintained via CXCL12 and CXCR4 signaling Protect infiltrating CTCs from immune responses | 99,163–165,178 |
| CD8+ T cells | LI, LN | Constrain myeloid cell infiltration via immunosurveillance. Reduced frequency and cytotoxic function. Increased exhaustion phenotype. | 168,178,227 |
| γδ T cells (res) | - | Prevalent at common future metastatic sites | 172 |
| NKT-like | Peripheral blood | Associated with shorter disease-free survival | 301 |
| NKT type I | LI | Immune surveillance by CXCR6+ NKT cell. Reduced frequency. | 176,178,188 |
| Regulatory B cells (Bregs) | LU | Promote CD4+ T cells conversion to Tregs and mediate TGF-βdependent crosstalk with MDSCs and stroma | 163 |
| Liver-res leukocytes | LU | Anti-metastatic effects via IFN-γ induction and elimination of fibrinogen depositions | 287 |
PMN sites were abbreviated as follows: BO = bone, BR = brain, LU = lung, K = kidney, LI = liver, LN = lymph node, PER = peritoneum, SPL = spleen; mo = monocyte; Φ = macrophage; res = resident; TAN = tumor-associated neutrophils; TEN = tumor-entrained neutrophils; AM = alveolar macrophages; IM = interstitial macrophages; ILC = innate lymphoid cells.
Neutrophils have recently garnered attention for their roles in cancer metastasis121. Chemotactic signals for neutrophil recruitment to the lung PMN include CXCL1/2/5/12, S100A8/A9, and Lin28B33,122,123,124,125, while neutrophil recruitment to the pre-metastatic liver is stromal cell-derived factor-1 (SDF-1)-dependent90.
Upon PMN arrival, neutrophils initiate inflammatory responses via leukotriene signaling and ROS generation, the latter strongly impairing NK and T cell function. Neutrophil leukotriene signaling supports metastasis-initiating cell proliferation in BC pre-metastatic lungs126. In melanoma, UV-damaged epidermal keratinocytes release HMGB1 to activate TLR4-MYD88 inflammatory signaling in neutrophils, stimulating angiogenesis as well as lung and LN metastasis102, while Bv8, MMP-9, and S100A8/A9 secreted by lung PMN-infiltrating neutrophils promote cancer cell extravasation and proliferation127. In the liver, neutrophils induce PMN formation downstream of SDF-1/CXCR4 signaling90. Notably, systemic expansion and polarization of PMN neutrophils in response to IL-17 production by IL-1β-activated γδT cells underscores the cooperation between innate and adaptive immune cells during PMN induction128.
Neutrophils support each stage of the PNM evolution via multiple mechanisms129. First, upon exposure to tumor-derived EVPs130, PMN neutrophils form NETs that sequester CTCs via integrin β1131, thus promoting colonization. NETs also induce resistance to chemo-, immuno-, and radiation therapies through IL-17 mediated NETosis, driving T cell exhaustion via CEACAM1/TIM-3 interactions132 and the PD-1/PD-L1 axis133, as well as by mediating drug detoxification134 and NET-dependent epithelial to mesenchymal transition (EMT)135. GM-CSF secreted by the primary tumor activates a positive feedback loop, where neutrophil JAK-STAT5β signaling induces synthesis of transferrin, an iron-transporting protein and mitogenic factor for incoming CTCs. Lastly, PMN neutrophils release elastase and cathepsin G that degrade thrombospondin-1, enhancing metastasis136. Thus, monocytic and granulocytic cells recruited to the PMN cooperate to generate an inflammatory and immunosuppressive PMN through a feed-forward loop that sustains chemoattractive recruitment.
PMN-promoting Innate Resident Cells
Resident cells in future metastatic organs are subject to active reprogramming that facilitates the acquisition of a pro-metastatic phenotype by recruited immune cells and can be diverse and organ-specific, of epithelial, fibroblast, endothelial, or hematopoietic origin. For example, lung resident club cells are non-ciliated epithelial cells that maintain a chronic homeostatic inflammatory state in the lung via S100A8–SAA3–TLR4 signaling. In a metastatic lung model, TLR4+ club cells auto-amplified SAA3, inducing TNF-α production by lung alveolar type II cells and macrophages. Meanwhile, club cell ablation halted the recruitment of CD11b+TLR4+ cells and spontaneous lung metastasis137. Thus, club cell-mediated homeostatic inflammatory imbalance due to constant exposure to airborne microbial endotoxins may contribute to lung metastatic susceptibility.
Critically, tissue resident macrophages, such as lung alveolar and interstitial macrophages138, liver Kupffer cells139 or brain microglia140, are not only rheostats regulating inflammation in their home organs, but also central effectors of PMN formation44,75,119,141. In the omentum PMN, CD163+Tim4+ resident macrophages were pivotal for ovarian cancer metastatic spread142. Both lung resident macrophages and endothelial cells upregulate MMP-9 secretion in response to primary tumor-secreted VEGF-A, thereby promoting lung metastasis by ECM remodeling10. CYP4A+ lung macrophages also regulate PMN formation as depletion of this population significantly decreased the number of circulating VEGFR1+ myeloid cells and the levels of MMP-9, S100A8, and FN in pre-metastatic lungs143. In addition to soluble factors, tumor-derived EVPs also reprogram PMN tissue resident interstitial macrophages towards an immunosuppressive function via de novo PD-L1 expression119.
Liver-resident Kupffer cells are first-line defense macrophages/phagocytes surveilling the early PMN, but can be corrupted to exert pro-metastatic functions144,145. Human liver-tropic pancreatic cancer EVPs containing integrin αvβ5 localize to fibronectin-rich areas and upregulate S100P and S100A8 in Kupffer cells via Src activation, thereby promoting S100-dependent inflammation19. Pancreatic cancer-derived EVPs preferentially uptaken by Kupffer cells trigger TGF-β secretion and subsequent fibronectin upregulation by hepatic stellate cells, generating a fibrotic liver microenvironment supportive of BMDC recruitment and PMN establishment44. EVP-packaged ANGPTL1 in turn reprogrammed the Kupffer cell secretome by suppressing MMP-9 production via JAK2-STAT3 signaling in colorectal cancer (CRC) models146. Phagocytosis of hypoxia-induced EVP miR-135a-5p in the liver PMN promoted MMP-7 production and CD30-TNF receptor–associated factor 2-p65 signaling in Kupffer cells, exerting immunosuppression by reducing TNF-α and IL-2 serum levels and intrahepatic CD4+ T cell numbers147. These studies suggest that Kupffer cells, due to their intrinsic function, are highly susceptible to reprogramming towards tumor-supporting phenotypes.
Microglia are brain-resident innate immune cells that respond to TDSF and EVPs75,141,148 by adopting an M2-like phenotype and releasing pro-tumor cytokines (e.g., IL-4, IL-10, IL-13, TGF-β) typically involved in neurovascular network repair149. M2-like microglia impair T cell proliferation in the brain PMN by upregulating PD-L1 expression. Loss of X-inactive–specific transcript, which correlates clinically with brain metastasis in BC patients, led to the production of EVP miRNA-503 responsible for M2 microglia polarization in brain metastatic BC models141. In lung cancer models, EVP uptake by brain microvascular endothelial cells induced endogenous DKK-1 secretion, which activated and skewed M1-like microglia dominant in the PMN towards a pro-tumorigenic M2-like one, concurrently upregulating both M1 (IL-1β, iNOS, TNFα, and TLR4) and M2 (ARG1 and CD206)148 markers. Importantly, the uptake of brain tropic human BC-derived EVPs containing CEMIP by brain endothelial cells and microglia induced perivascular inflammation and vascular remodeling19,75. Thus, PMN-resident macrophages establish a sustained pro-tumor inflammatory milieu at a secondary site and reprogram recruited immune cells, sealing the fate of tumor cells seeding the PMN.
Dendritic cell antigen-presenting functions are also altered in the PMN. Lung DC function is suppressed by PGE2 secreted by resident fibroblasts at steady state but is reinforced by tumor-secreted IL-1β, supporting PMN formation. Fibroblast-specific deletion of COX-2, the enzyme that produces PGE2, reverses DC suppression, leading to enhanced immune activation and reduced lung metastasis in multiple BC models150. Consistent with these findings, COX-2 expression in DCs promoted LN metastasis, which was markedly suppressed by COX-2 inhibition151.
These diverse myeloid cell functions highlight their key roles in PMN formation. Data on involvement of other innate immune populations, such as basophils, eosinophils, and mast cells, in the PMN are limited, but warrant further exploration, especially in barrier tissues, such as the lung, in the context of chronic inflammation induced by environmental exposures, infections, and aging.
The PMN complement system
The complement system functionally bridges innate and adaptive immunity, clearing pathogens and modulating host immune responses through antibody-mediated activation or anaphylatoxins152,153. Anaphylatoxins (C3a, C4a, C5a) recruit myeloid cells that express their cognate 7-transmembrane G-protein-coupled receptors, thereby regulating myeloid cell chemotaxis, migration, and phagocytosis152. Recent studies highlighted the role of C3a-C3aR crosstalk between activated CAF and macrophage/neutrophil chemotaxis during metastatic progression154–156. In the pre-metastatic lung of BC models, pro-tumor inflammatory Th2 cytokines induced sustained C3 expression in mesenchymal stromal cells in an IL-4/13-STAT6-dependent manner, enhancing metastasis through recruitment of C3aR+ neutrophils and induction of NETosis157. Importantly, the complement system may also account for acquired resistance to chemotherapy early during metastasis through C3a and C5a crosstalk between resident fibroblasts and recruited myeloid cells that restricts T cell function158. These studies highlight the involvement of the complement system in PMN formation and stress the need to investigate the therapeutic potential of targeting specific components of the complement cascade to hinder PMN establishment.
Adaptive immune landscape
The systemic immune surveillance exerted by adaptive immune cells, such as cytotoxic T lymphocytes (CTLs; i.e., CD8+ T cells) and NK T (NKT) cells restricts the survival of DTCs and regulates metastatic colonization and outgrowth159–161. By recruiting and promoting the differentiation and expansion of tumor-infiltrating lymphocytes (TILs) into Treg cells, T helper 17 (TH17) cells, γδT cells, and regulatory B (Breg) cells in the primary tumor and potentially at future metastatic sites, cancer cells avert anti-tumor immunity, thus initiating the metastatic cascade162 (Table 1).
The most prevalent adaptive immune cells in the PMN are Tregs, which have regulatory and suppressive functions. Treg recruitment to the PMN relies on a complex interplay between primary tumor-derived signals, systemic Breg-mediated secretion of TGF-β, crosstalk with immunosuppressive myeloid populations, and PMN stroma-derived signals162–164. In turn, Treg enrichment maintains an immunosuppressive environment, supporting PMN progression. Although the complex Treg-mediated PMN interactions remain to be explored, they shield DTCs from immune attack by inhibiting T cell proliferation, cytokine production, and cytotoxic function in several cancers162,165,166. Moreover, local and systemic TGF-β and MDSCs cooperate to expand Tregs in distant organs, such as draining LNs, liver, lung, and bone marrow167. In the bone marrow PMN, Treg enrichment and maintenance depends on CXCL12 and CXCR4 signaling99. A recent study exploring immune cell dynamics in the rhabdomyosarcoma lung PMN168 reported a significant increase in exhausted, dysfunctional PD1hiCD44int T cells at the expense of CD4+ helper T cells169. Interestingly, the PMN-promoting role of dysfunctional Th17 cells was described in BC murine models, where production of RANK ligand, a major regulator of osteoclastogenesis, and IL-17F by these cells induced osteolysis in a manner proportional with the metastatic potential of the tumor model 170.
Immune surveillance is achieved by the three main lymphocyte lineages: αβT cells (CD4+ and CD8+), γδT cells, and B cells171. While αβT cells and B cells exert both systemic and local effects, γδT cells (mostly CD4−CD8−) are present at lower frequencies in the circulation but are highly abundant in tissues, especially at sites prone to metastases172. Since these cells are not MHC-restricted, they contribute to an unconventional form of immunosurveillance and have different roles depending on their molecular make-up and residence. As suggested by pre-clinical and clinical data, γδT cells may hold the key to understanding mechanisms of immunosenescence, anti-tumor immunity, and tumor regression. Future discoveries will unveil the role of these cells in PMN initiation and will assist the development of therapeutic strategies for preventing PMN evolution (e.g.,through γδT cell-mediated tissue protection from fibrosis).
Although humoral immunity is critical in modulating tumor progression, few studies have investigated the role of B cells in the PMN. A novel subset of tumor-evoked Bregs, characterized by constitutively active STAT3, induced TGF-β-dependent conversion of resting CD4+ T cells into Tregs, which protected metastatic cells from host immune responses163 (Table 1). Moreover, the primary tumor educated pre-metastatic lungs to express TARC/CCL17 and MDC/CCL22 chemokines, which attracted C-C motif chemokine receptor 4 (CCR4)-positive tumor and immune cells 164. PMN infiltration by CCR4+ Tregs induced NK cell apoptosis via secretion of β-galactoside-binding protein, supporting metastatic cell seeding164. The role of B cells and immunoglobulins (Ig) in LN metastasis was recently investigated in spontaneous BC models. Primary tumors induced B cell accumulation in pre-metastatic tumor-draining LNs and production of pathogenic IgG that targeted HSPA4, a tumor cell glycosylated membrane protein, which led to integrin β5 activation. This in turn triggered Src-NF-κB activation in cancer cells, which further promoted CXCR4/SDF1α-mediated metastasis173.
Local and systemic factors contributing to PMN formation
Gut and tumor microbiome contributions to PMN formation
Groundbreaking discoveries in the past decade highlighted that the microbiome shapes host immune responses, modulates inflammation, and orchestrates anti-tumor immunity174. During CRC metastasis, the combined effects of gut dysbiosis and intestinal inflammation extend to the liver, generating a pro-inflammatory and immunosuppressive hepatic microenvironment175–178. Environmental factors, such as a high-fat diet, also promoted lung PMN formation in murine models of BC and melanoma, an effect mediated by gut dysbiosis via M1 polarization, TNF-α and CCL2 secretion by gut macrophages, and subsequent recruitment of S100A8/A9-producing MDSCs to future metastatic sites 179. Similar to PMN vascular leakiness, gut vascular barrier disruption is an essential step for CRC resident Escherichia coli bacteria translocation to the liver, PMN formation, and metastasis 180. These observations spurred further studies that showed that each tumor type harbors its own microbiome, dwelling intracellularly in both tumor and immune cells, although systematic comparisons with pre-metastatic and metastatic niches have yet to be made 181.
Among commensals turned onco-pathobionts, Fusobacterium nucleatum, a biomarker of inflammatory bowel disease, associated with poor prognosis in CRC patients182,183. F. nucleatum, found to reside not only in primary patient-derived xenografts (PDXs) but also within liver metastatic cells 184, could evade the immune system during CRC progression by shedding its wall and invading cancer cells, thus modulating host E-cadherin-β-catenin signaling185. Studies in immunocompetent mice suggest that F. nucleatum may drive liver PMN formation by promoting MDSC and Treg infiltration, decreasing NK and CD8+ and Th17 T cell frequencies and elevating CXCL1 levels, all defining features of CRC liver PMN186,187,178.
Importantly, gut dysbiosis and inflammation alter microbial metabolites, resulting in local and systemic metabolic changes. Liver metastasis was inhibited by secondary bile acid conversion by gut commensal bacteria, through upregulation of CXCL16 in liver sinusoidal ECs, and immunosurveillance by recruited CXCR6+ NKT177. However, capsaicin-induced disruption of gut vascular barrier and altered bile acid metabolism reduced NKT cell infiltration in the liver and increased inflammation (IL-12, TNF-α, IFN-γ) that facilitated PMN formation188. Moreover, sodium butyrate rescued gut dysbiosis, significantly reducing liver metastasis in pre-clinical models of CRC176,177. These findings suggest that remodeling of the liver immune microenvironment by gut-derived metabolites may occur prior to tumor dissemination and shape the liver PMN in gastrointestinal cancers.
The role of local and systemic metabolism in PMN induction
The role of glucose metabolism in PMN formation
During metastatic progression in cancer patients, the differential availability of circulating nutrients and metabolites is marked by accumulation of glucose metabolism byproducts, such as lipid, lactate, pyruvate, and glutamate, with a concomitant decrease in glutamine, a precursor of gluconeogenesis189, which together contribute to systemic immune suppression. For instance, lactate accumulation impairs NK cell function, leading to MDSC and Treg expansion, as well as M2 macrophage polarization190. Proteomic characterization of early lung PMN changes in BC identified metabolic changes in glucose metabolism, while late PMN changes revealed an increase in Ca2+ signaling/sensing, likely due to extensive lung tissue remodeling, ECM reorganization, and inflammatory signaling191. Despite increasing interest in metabolic rewiring during cancer progression and metastasis, few studies have investigated PMN immunometabolic rewiring, with those that did identifying EVPs as the main drivers of systemic and local metabolic reprogramming.
EV-mediated immunometabolic reprogramming of resident macrophages in the lung PMN may promote immunosuppression via several mechanisms119. On one hand, tumor-derived EVPs triggered HMGB-1-TLR2-NF-kB signaling in lung PMN interstitial macrophages leading to immunometabolic reprogramming toward an immunosuppressive phenotype, while NOS2 induction shifted metabolism from mitochondrial oxidative phosphorylation towards glycolysis. On the other hand, increasing lactate levels further fueled HIF-1α activation downstream of NF-kB, increasing PD-L1 expression on the surface of macrophages119.
Notably, BC can alter systemic glucose metabolism and modulate the metabolic environment of distant organs favoring PMN formation. BC-derived EVPs limit glucose availability and utilization by PMN resident cells (e.g., lung fibroblasts, brain astrocytes, and neurons) via miR-122-mediated downregulation of pyruvate kinase and glucose transporters (i.e., GLUT-1) to instead provide this nutrient for the outgrowth of infiltrating tumor cells192. Enrichment in EVPs, and particularly exomeres in metabolic enzymes and metabolites50 indicates they may play a role in local and systemic metabolic reprogramming of target cells in distant organs.
Lipid metabolism and metabolites involved in PMN formation
Recent studies suggest that abnormal cancer cell and cancer-associated stroma lipid metabolism fuel tumor progression193–195. Increased fatty acid accumulation and subsequent oxidation represent major ATP sources and one of the key metabolic adaptations associated with metastasis. For example, acetyl-coenzyme A carboxylase alpha stabilizes fatty acid synthesis in BC lung PMN196, while fatty acid oxidation (FAO) is involved in sequential steps of the pre-metastatic cascade and LN metastasis197. A recent study documented palmitate enrichment in pre-metastatic lungs and lung interstitial fluid of murine models and patients with BC without lung metastasis, and that this fatty acid was used for acetyl-CoA synthesis198. Thus, FAO and related lipid metabolic processes might be involved in reprogramming PMN-associated immune cells, contributing to immunosuppression.
Lipid metabolism also favors eicosanoid synthesis, converting arachidonic acid (AA) to PGE2, which is central to PMN formation. PGE2 metabolism promoted immunosuppression and immune escape in a murine bladder carcinoma model, upregulating PD-L1 on BMDCs199. Notably, lung PMN pro-inflammatory S100A8/A9 chemokines complex with AA to facilitate its uptake by endothelial cells via CD36200. Thus, PGE2 and S100A8/A9 cooperate, linking inflammation and metabolism in the pre-metastatic lung, suggesting that inhibiting BMDCs PGE2 metabolism may effectively target their immunosuppressive function. Expression of CYP4A, another enzyme involved in AA metabolism, in macrophages was also associated with M2 polarization, pro-tumor cytokine production, and recruitment of VEGFR1+ myeloid cells to the lung PMN143. Given that high cholesterol, obesity, and high fat diets are major risk factors for BC recurrence and metastasis201, it is not surprising that cholesterol and sterol hydroxylation in BC models resulting in high levels of pro-metastatic 27-hydroxycholesterol directly altered myeloid cell function, increased γδT cell frequency, and neutrophil infiltration, and suppressed cytotoxic CD8+ T cells in the lung niche201.
Nutrient, oxygen, and mineral sensing in immune cell function
Cancer cells are often exposed to metabolically challenging environments and their metabolic plasticity enabled by nutrient-, oxygen-, and cation-sensing molecules (e.g., Ca2+, Fe2+) ensures their adaptation. The lack of studies on the metabolic crosstalk in the PMN raises the question regarding the roles of Fe2+ and Ca2+ sensing in PMN immunometabolism, and whether they may drive inflammatory and immunosuppressive processes, likely through the S100 Ca2+-sensing proteins and ferroptotic cell death. Recent studies suggest a link between ferroptosis and PMN formation, as CD36-mediated AA uptake by CD8+ T cells resulted in lipid peroxidation and ferroptosis, followed by impairment of the anti-tumor immunity202. Among immune cells, T cells are exquisitely dependent on Ca2+ signaling downstream of TCR activation for effector functions, as well as self-tolerance programs and homeostasis and Ca2+ sensing dysregulation results in autoimmunity, inflammation, and immunodeficiency203,204. Intriguingly, the late lung PMN is dominated by upregulation of Ca2+ signaling via autocrine and paracrine chemotactic loops orchestrated by the S100 Ca2+ sensor proteins191. Thus, whether competition for Ca2+ in the PMN and Ca2+ sequestering by PMN stromal cells limit Ca2+ availability to effector PMN immune cells remains an important question.
Innervation in cancer and its implications for PMN formation
The role of cancer innervation and stress in PMN evolution
The sympathetic nervous system (SNS) mediates acute stress responses following hypothalamic-pituitary-adrenal (HPA) axis activation205 by modulating adrenergic and glucocorticoid signaling206 and supports cancer progression in response to chronic stress207. The immunosuppressive effects of stress205,208,209, including suppression of NK210 and CD8+ T cells211, and MDSC accumulation212 extend to the lymphatic system, as chronic stress induces remodeling of tumor and peritumor lymphatic networks via tumor-derived VEGF-C and TAM-mediated COX-2-PGE2 inflammatory signaling to provide lymphatic dissemination routes for tumor cells213.
Recent studies identified stress-related catecholamine as a primary factor in regulating pre-metastatic to metastatic evolution in the lung niche214 and favoring colonization by BC CTC. Pharmacological inhibition of sympathetic nerve function or blocking the β-adrenergic signaling markedly suppressed stress-induced lung metastasis along with the reduction of circulating monocytes and lung resident macrophages214. Critically, a clinical trial in patients with early BC assessing the efficacy of combined blockade of β-adrenergic receptors and PGE2-reducing COX-2 inhibitors during the perioperative period, when stress flares due to both the surgical procedure and psychological stressors, revealed reduced primary tumor STAT3 levels, lower circulating IL6, C-reactive protein (CRP) and monocyte levels and increased NK cell activation215. Given these data, an outstanding question is what are the potential contributions of stress and cancer innervation to PMN generation, and more specifically, to modulating immune function in the PMN?
Neuro-immune interactions in the PMN
The effects of neuro-immune interactions on the PMN were first explored in the bone marrow (BM), a highly innervated tissue where the SNS regulates the physiological HSC niche216. Indeed, stress-induced or pharmacological sympathetic activation in bone metastatic BC increased RANKL levels in the bone microenvironment, induced bone lesions, altered the cytokine profile of the bone PMN and rendered the BM microenvironment more receptive for CTC colonization217. Direct neuro-immune interaction of SNS fibers with BMDCs also favor CTC colonization by disrupting Sema3A-mediated neuron-macrophage interactions in healthy lungs. Chemical sympathectomy inhibited tumor angiogenesis, prevented bone loss, and decreased lung mo-MDSC recruitment and metastatic burden, whereas specific adrenoreceptor beta 2 (ADRB2) inhibition reduced S100A8 levels and CTC recruitment to the lung218. These findings ascribe cancer innervation and sympathetic activation an active role in PMN development, thereby providing a rationale for blocking SNS signaling in cancer patients by repurposing FDA-approved drugs (e.g., βAR or β-blockers) as anti-PMN therapies.
Clinical implications of the PMN for cancer detection and management
Systemic detection of PMN formation in cancer patients
While pre-clinical cancer models furthered our understanding of PMN biology, overcoming challenges associated with PMN detection, characterization, and therapeutic targeting in patients is essential to translating these advances into improved patient outcomes. Several mediators of PMN formation, such as ECM-remodeling molecules, detected in patient liquid biopsies, correlated with metastatic outcome. High MMP serum levels correlated with metastasis and poor survival in gastric219, breast220 and ovarian cancer221, while elevated systemic levels of TIMP-1 associated with liver metastasis and poor prognosis in colon and pancreatic cancers90,222. Similarly, ANGPTL2 was elevated in osteosarcoma patients and promoted lung PMN formation33.
Blood-based liquid biopsies could reveal distant PMN formation and inform on metastatic progression through EVP cargo. In head and neck cancer patients, PD-L1 on EVPs, but not free-circulating PD-L1, associated with disease progression62. In pancreatic cancer, levels of MIF, a cytokine associated with liver PMN formation, were markedly higher in EVPs isolated from the plasma of stage I PDAC patients who later developed liver metastasis compared to patients without metastasis44. Comparison of EVP-packaged pyruvate kinase M2 in localized versus metastatic prostate cancer patients identified it as a potential marker of bone pre-metastatic niche formation 223. Unbiased proteomic profiling of EVPs from tumor explants and plasma of patients identified pan-cancer EVp markers (e.g., VCAN, tenascin C, and THBS2), as well as tumor-type-specific EV proteins that, in stage I patients, may allow PMN biomarker identification110 to improve early detection.
Urine EVP biomarkers may also indicate PMN status not only in the urinary system but also in other cancers such as BC224,225. Given the accessibility of urine samples, this biofluid is a convenient liquid biopsy source. Human lymphatic fluid may offer unique insight into systemic immune responses: in early-stage melanoma, immune-modulating S100A9 EVP cargo correlated with IHC staining of non-metastatic LNs46. Large studies are however required to evaluate correlations between lymphatic fluid biomarkers and metastasis.
Profiling PMN-associated immune cells
Profiling the frequency, phenotype, and signaling pathways activated in BMDCs, MDSCs, neutrophils, and T cells, can offer additional insight into PMN evolution in cancer patients. MDSC frequencies correlated with cancer stage and metastatic burden in BC patients226. In melanoma patients, STAT3 activation in myeloid cells inversely correlated with the number of CD8+ T cells in pre-metastatic LNs227. CD68+ myeloid cells exhibiting elevated S1PR1 and p-STAT3 levels accumulated in tumor-free LNs of high-risk prostate cancer and melanoma patients35. Similarly, CYP4A+ TAM accumulation in primary tumors correlated with reduced overall and recurrence-free survival in BC patients143. Lastly, circulating neutrophils in HCC patients bearing lung or liver metastasis showed enhanced NET formation compared to non-metastatic patients or controls228. Interestingly, while NET deposition occurred in non-metastatic and metastatic LNs, high LN NETs correlated with reduced survival, even in the absence of apparent metastasis229, suggesting NETosis is a LN PMN biomarker predictive of patient outcome. Thus, patient validation of PMN immune hallmarks identified in pre-clinical studies highlights their conserved role and warrants clinical trials to establish their prognostic power.
PMN tissue biopsies during the clinical workup
Most clinical evidence of PMN reprogramming is based on sentinel LN (SLN) samples from cancer patients as these tissues are routinely resected for staging. LNs are a common initial site of metastasis in most solid tumors and reliable indicators of patient outcomes. A PMN hallmark, VEGFR1+ myeloid clusters were detected in cancer-free pre-metastatic LNs of patients2,230 and their abundance predicted prostate cancer recurrence, marked by the rise of prostate-specific antigen (PSA) blood levels, and metastatic progression230. ECM-remodeling enzyme MT1-MMP and LOX expression in LN macrophages correlated with lymphatic metastasis in oral cancer231. Comparison of non-metastatic, metastatic SLNs, and non-metastatic distal LNs in early-stage cervical and oral squamous cell carcinoma revealed that non-metastatic and metastatic SLNs shared similar features absent from distal LNs. Specifically, high lymphatic vessel density was observed in both SLN groups, suggesting this characteristic as a biomarker of LN metastasis232,233. Similarly, high endothelial vessel remodeling in BC pre-metastatic LNs associated with accumulation of CCL21+ lymphocytes234. Moreover, primary BC tumor CYP4A+ TAM levels correlated with several PMN markers, including VEGFR1+ cell infiltration, S100A8 expression, and fibronectin deposition in uninvolved LNs143. Nevertheless, in patients, LN PMN-associated immunosuppression is mainly uncharted235. Thus, despite routine collection and analysis of LN, it is still unknown which PMN LN signatures can risk-stratify patients or predict their potential involvement in LN and distant metastases, highlighting the need of extensive profiling of these patient tissues.
Clinical evidence of bone and lung PMN formation also exists. High primary tumor LOX expression correlated with metastatic disease and poor survival236,as well as with bone tropism and osteolytic lesions in ER-negative BC patients87. A comprehensive analysis of lung autopsy samples suggested that MMP-9 initiates lung PMN formation in patients, as high MMP-9 levels were detected in lung endothelial cells of uninvolved lungs from patients with nine distinct cancers 10.
Another PMN site that has garnered attention is the omentum, a common site of ovarian cancer metastasis. Adhesion molecules, such as integrin α2, mediate cancer cell adherence to collagen-rich omentum, initiating ovarian cancer metastasis237. A correlation between integrin α2 expression and altered collagen expression in pre-metastatic omentum predicts poor outcome in patients237. Moreover, neutrophil recruitment promotes omental PMN formation in early-stage, high-grade serous carcinoma patients. Neutrophils accumulated around blood vessels and high levels of neutrophil elastase were detected in omental tissues from cancer patients but not healthy subjects238. EVP proteomic analysis revealed an omental PMN signature characterized by IL-6 and IL-8 expression239.
While liver, bone, and especially brain PMN patient tissues are challenging to obtain, their prognostic potential is immense. Our multi-omics characterization of pre-metastatic livers collected during primary tumor resection from pancreatic cancer patients with localized disease revealed the presence of various PMN hallmarks, such as inflammation, metabolic dysfunction, ECM changes, and immunosuppression. Importantly, liver neutrophil infiltration at the pre-metastatic stage correlated with metastatic progression in these patients at 8-year follow-up240, a finding that could inform treatment course. Such biomarkers may predict not only the presence of a PMN, but also the specific site where these processes occur, thus allowing tailored interventions. The development of novel technologies that require less material may enable clinicians to perform analyze fine needle biopsies, which will facilitate future studies.
Imaging tools for clinical PMN detection
Monitoring PMN progression non-invasively can also be achieved via imaging, either by repurposing current methods or by developing novel, more sensitive imaging techniques to detect PMN-related features. A few pre-clinical and clinical studies reported such techniques; however, they have yet to be adopted for routine patient screening.
Imaging the PMN in the clinic
Several pre-clinical imaging techniques were developed to detect PMN-associated changes (Table 2(. These studies underscore the potential such applications have in monitoring PMN formation while highlighting the need for additional imaging techniques, focusing on key high-translational determinants, such as integrins. Multiple imaging methods, including fluorescent antibodies241 and positron-emission-tomography (PET) scans242,243, enable visualization of integrin localization, activation, and function. In patients, noninvasive imaging of αvβ3244 and αvβ6245 integrins was well tolerated and revealed significant uptake in primary tumors and multiple metastatic foci. Such methodologies, with improved sensitivity, may hold great promise in PMN detection.
Table 2.
Pre-clinical imaging of the PMN
In the clinical setting, several studies employed radiomic methods to identify unexplored PMN-related features in lungs of BC patients246,247 or in liver of pancreatic248 and rectal249 cancer patients. By comparing computed tomography (CT) scans of metastatic and non-metastatic regions at two time points, prior to and after detection of evident metastasis, five features that distinguish the regions prior to metastasis formation were identified, suggesting this imaging method could clinically detect lung PMN formation246. Another study analyzed lung scans of BC patients with or without lung metastasis and healthy controls. Increased lung density, indicative of inflammation, was evident in metastatic and non-metastatic, but not control lungs. Notably, enhanced lung density was detected prior to the clinical diagnosis of pulmonary metastasis and correlated with disease score, demonstrating the potential of CT scans in clinically diagnosing the lung PMN247.
Similarly, a retrospective study in pancreatic cancer patients aimed to predict early liver metastasis development (within 6 months from primary tumor excision) based on contrast-enhanced CT before primary tumor resection. Analysis of pre-operative enhanced CT scans of livers from a total of 688 resected PDAC patients found a predictive ability of a multifactored model using radiomic data combined with clinicopathological variables. Six radiomic texture features that reflect hepatic parenchymal heterogeneity and fibrosis250 were significantly elevated in patients that developed early liver metastasis. Combined with tumor size and differentiation, this analysis identified patients at high risk for early liver metastases248. Retrospective analysis of contrast-enhanced CT radiomics predicted liver metastasis within two years from primary tumor resection in rectal cancer patients with no pre-operative evidence of liver metastases, based on a mixed model including both radiomic data and clinical parameters249. Another imaging method assessed in an ongoing clinical trial is the FibroScan, an ultrasound-based transient elastography. This study aims to identify pre-metastatic liver biomarkers in stage I-IV colon cancer patients, correlating the EV cargo of liver biopsies with FibroScan imaging (NCT03432806), as measures of liver fibrosis (tissue stiffness) and steatosis (fatty liver), indicative of pre-metastatic modifications.
Therapeutic strategies targeting the PMN
Risk of promoting PMN with current therapeutic approaches
Epidemiological studies have shown that current cancer therapies, including surgery, radiotherapy, and chemotherapy may promote distant metastases by triggering BMDC-mediated repair resembling wound healing and systemic inflammation251. Several chemotherapies support PMN formation systemically by eliciting release of tumor-derived EVPs and locally by activating CAFs and endothelial cells as well as by recruiting MDSCs, ultimately impairing T cell function158,251–253. Similarly, radiotherapy promotes the recruitment of pro-tumor TAMs and MDSCs in various cancers254, while NETosis is a common consequence of both chemotherapy and radiotherapy134,255. Notably, primary tumor resection releases tumor cells in the circulation, increasing the frequency of CTCs256–259 thus promoting metastasis, should these cells encounter a favorable niche generated by adjuvant chemotherapy, radiation, and pre-existing inflammation.
Changing the status quo: targeting PMN formation
To date, tailored therapies targeting PMN formation are nonexistent and metastasis-directed therapies are scarce (Fig. 3). Among existing therapies, cytoreductive surgery may also hinder PMN formation and enhance patients survival by blocking the systemic effects of the primary tumor260,261, including the recruitment of tumor-supporting innate immune subsets.
The most promising therapy for metastatic cancers is immune checkpoint blockade (ICB) targeting T cell checkpoints (e.g., PD-1, PD-L1, CTLA-4); however, therapeutic success has been limited to cancers with high mutational burden and hindered by development of therapy resistance. Recent therapeutic advances aim to overcome ICB failure by targeting new inhibitory checkpoint molecules on T cells or NK cells as well as co-stimulatory molecules to enhance endogenous anti-tumor immunity262. Moreover, potential targets for interfering with pro-metastatic immune cell function and PMN formation have emerged. For instance, blocking immune cell recruitment to the PMN (e.g., antibodies targeting CCL2 for BM monocytes, CCR4 for Tregs, CXCR2 for neutrophils, CSF-R1 for overall myeloid cell infiltration) or reverting macrophages to M1, anti-tumor phenotypes, could impair PMN formation. Selective targeting of IL-2 receptors on Tregs could overcome immune escape by compromising their survival while antibody blocking of CD47 “don’t eat me” signals upregulated by metastatic cancer cells may enhance phagocytosis262. Using these drugs as adjuvants or as monotherapy could provide new therapeutic approaches for treating PMN and metastases.
Reversing PMN immunosuppression
Therapeutic strategies targeting PMN formation would represent a paradigm shift in treating cancer, by preventing or eradicating metastases before they manifest clinically. This could be achieved by interfering with PMN immunosuppression by targeting cells with crucial roles in PMN formation or by activating anti-tumor immunity (Box 2), thus preventing seeding and outgrowth of CTCs at these distal sites (Fig.3).
Co-opting anti-tumor immunity
Targeting S100A4 that perpetuates MDSC-driven PMN inflammation with the 6B12 antibody reduced metastatic burden by restoring Th1 anti-tumorigenic phenotype in the lung PMN263. Future studies should investigate the translational immunomodulatory potential of the 6B12-based therapy in cancer patients. Kaplan and colleagues pioneered a promising proof-of-principle pre-clinical study that could rebalance the core immunosuppressive PMN environment and reduce metastatic burden. Genetically engineered myeloid cells (GEMys) were used for local delivery of immunomodulatory cargo (IL-12) to the lung PMN in a rhabdomyosarcoma model. This approach activated APCs, and T and NK cells168, extending survival of 90% of mice for over one year. This study provides a rationale for IL12-GEMy as adjuvant therapy after surgery and in combination with chemotherapy for unresectable tumors, highlighting the potential of engineered myeloid cells as promising reconfigurable cell-based therapies that boost anti-tumor immune responses and reverse immunosuppression in the PMN.
Normalizing NK cell function
NK cells are promising cellular targets for immunotherapies given their critical role in controlling metastatic dissemination. Dormant DTCs that remain undetectable after primary tumor resection offer a therapeutic window to prevent their reactivation. In BC models, NK cells enforce DTC dormancy in the liver, whereas hepatic stellate cells suppress NK cell proliferation allowing cancer cell outgrowth. Adjuvant IL-15-based immunotherapy normalized the NK cell pool sustaining DTC dormancy via IFN-γ signaling, preventing hepatic metastases and significantly prolonging survival without affecting dormant cells264. Thus, IL-15 or IFN-γ therapy, or CXCR4 inhibition in early-stage cancer could prevent PMN formation by normalizing the NK cell pool and rescuing their anti-tumor function.
Normalizing vascular permeability and anti-tumor immune cell trafficking in the PMN
Recent attempts to reverse PMN-promoting vascular permeability employing a protein fusion between LIGHT (a TNF family cytokine) and a vascular targeting peptide (LIGHT-VTP) that homes to angiogenic blood vessels in primary tumors normalized intra-metastatic blood vessels and increased infiltration by GZMB+ effector T cells that further sensitized lung metastases to antiPD-1 checkpoint inhibitors265.
Targeting innate cell-mediated immunosuppression in the PMN
Epigenetic therapy targeting MDSC trafficking
Following surgical resection of primary lung, breast, and esophageal cancers, low-dose adjuvant epigenetic therapy (LD-AET) selectively disrupted MDSC PMN recruitment, reprogramming them towards an interstitial macrophage-like phenotype266, thus reversing immunosuppression and improving disease-free and overall survival267. A recent randomized, phase-II adjuvant LD-AET clinical trial in patients with stage-I NSCLC undergoing tumor resection, reduced tumor recurrence twofold267.
Obstructing myeloid cell recruitment by the PMN ECM
Disrupting MDSC recruitment to the PMN may represent another viable strategy to reverse the effects of PMN immunosuppression. As ECM regulators and docking sites for BMDCs, PMN-targeting LOX proteins hold promise, especially in preventing osteolytic bone PMN lesions86 and could benefit BC and renal cell carcinoma patients who exhibit LOXL2 overexpression268. In clinical trials, achieving high serum concentrations of vitamin derivatives, such as all-trans retinoic acid, decreased peripheral MDSCs and improved T cell immune responses in cancer patients and thus could block PMN formation269.
Targeting NET formation
Neutrophils, the most abundant cell type in the human blood, have remarkably plasticity and represent our first defense against blood pathogens through NETosis270. Although cancers employ NETs to protect themselves by inducing cytotoxic T cell exhaustion in an IL-17-dependent manner132,133,271, indirect NET targeting via IL-17 blockade potentiated responses to ICB271. Moreover, pharmacological inhibition of IL-17/IL-17RA signaling using monoclonal antibodies in combination with ICB, proved highly effective in enhancing CD8+ T cell recruitment and cytotoxicity in pancreatic cancer, normally refractory to ICB271. A humanized mAb against IL17A is already FDA-approved, suggesting that anti–PD-1+anti-IL-17 combination therapy could be easily translated to the clinic271. Additional strategies blocking NET formation include targeting PAD4, the enzyme responsible for NET formation, inducing NET degradation via DNAse I treatment and neutrophil elastase inhibition (NEi) could also be combined with ICB in patients otherwise resistant to immunotherapy238,134.
Anti-thrombotic therapies that prevent PMN formation
Anti-thrombotic agents could also prevent PMN formation and metastatic progression. A calcium phosphate nanoparticle nanotherapeutic containing an anti-inflammatory agent and decorated with an anti-thrombotic agent, low molecular weight heparin could target systemic metastases272. In a BC model, administration of the nanotherapeutic as an adjuvant to paclitaxel chemotherapy or surgery reduced tumor foci by 10–15-fold and increased survival. Moreover, the nanotherapeutic agent targeted both the primary tumor and the lung pre-metastatic site where it markedly reduced expression of PMN proteins272.
Vaccine-based anti-tumor immunotherapies
The implementation of therapeutic strategies based on cancer vaccines in patients who are at risk for developing metastasis, could boost systemic immunosurveillance programs and prevent PMN progression. Liposomal RNA-based cancer vaccines targeting tumor antigens to APC273 could overcome systemic as well as local PMN immunosuppression and T cell tolerance and improve therapeutic outcome. The ongoing, first phase I trial in patients with advanced melanoma (NCT02410733) has yielded encouraging responses so far, with shrinkage or complete regression of multiple LN and lung metastases, while also hindering the development of novel metastases in responsive patients with lower mutational burden that previously received ICB therapy. These effects were achieved through expansion and endurance of tumor-specific memory CD8+ T cells and enhanced cytotoxic responses.
Co-opting gut microbiota to boost immunotherapies
Strategies that reshape microbiota to improve anti-cancer treatment are being evaluated. High-fat diet (HFD) are linked to increased risk of metastatic cancer274,275 (Box 3), an effect attributed to the induction of microbial dysbiosis and PMN formation179,275. Glycyrrhizic acid (GA) prevented HFD-induced PMN formation and metastasis by reversing microbiota dysbiosis179. Treatment with GA obstructed myeloid cell migration and S100A8/A9 expression in the lung PMN. Antibiotic treatment or recolonization of the gut with beneficial bacteria exerted effects similar to GA, highlighting the gut microbiome role in systemic crosstalk with PMNs in distal organs179. Similarly, sodium butyrate, another major gut microbiome metabolite, rescued gut dysbiosis and significantly reduced liver metastasis in CRC pre-clinical models by increasing intrahepatic NKT and Th17 cell infiltration, along with decreasing Treg and IL-10 accumulation176,177.
EVP-based therapies
EVPs are promising reconfigurable therapeutic tools. Chimeric EVPs produced by macrophage-tumor hybrid cells accumulated in the LN where they activated T cells, inducing tumor regression and reducing metastases in lymphoma, breast, and melanoma models276. In the metastatic lung of breast cancer models, these EVPs increased CD8+ T cell/Treg ratio and in combination with PD-1 ICB, lung metastasis was significantly suppressed276. This study warrants further exploration of using personalized EVPs for cancer therapy. Meanwhile, EVP-targeted therapeutic approaches focused on disrupting circulating EVPs could interfere with the capacity of primary tumor-derived EVPs to prepare a metastasis-supportive microenvironment at early stages, thereby preventing PMN formation. For example, targeting key EVP proteins in systemic circulation using monoclonal antibodies against S100A4 or integrins could interfere with EVP homing hindering PMN development 110,277,278.
Discussion
Indisputable evidence underscores PMN’s prominent role in determining cancer progression and outcome. However, translation of this knowledge to the clinic has been slow. To better characterize pre-metastatic alterations in patients, we propose that biopsies from metastasis-prone organs, such as LN, bone marrow, liver, and when possible, lung, and omentum, should be regularly collected and studied in parallel to liquid biopsies. Systematically correlating the signatures of these samples collected at diagnosis/during primary tumor resection with the subsequent metastatic site and patient survival data will accelerate biomarker discovery and early cancer detection, prior to the metastatic colonization. Such findings will enable a paradigm shift in cancer treatment, where personalized prediction of metastatic sites will allow early intervention and tailoring treatments to specific PMN sites/characteristics. Such approaches may reduce or even prevent metastasis by reverting the “soil” of these distant sites to one unfavorable for CTC growth. Moreover, as current therapies may promote PMN formation, sensitive biomarkers or imaging methods could improve patient stratification for current standard of care therapies, sparing patients that show no evidence of systemic disease or metastases aggressive treatments. As metastasis remains the most critical factor determining cancer patient survival, such approaches bear unprecedented potential in changing patient care and outcomes.
Acknowledgements
The authors gratefully acknowledge support from the National Cancer Institute (CA232093, CA163117, CA210240 and CA207983, and CA218513 to D.L. and CA270998 to I.M), the Thompson Family Foundation, the Tortolani Foundation, the Pediatric Oncology Experimental Therapeutics Investigator’s Consortium, the Malcolm Hewitt Weiner Foundation, the Manning Foundation, the Sohn Foundation, the Theodore A. Rapp Foundation, the Hartwell Foundation, the Children’s Cancer and Blood Foundation, the AHEPA Vth District Cancer Research Foundation (all to D.L.) and the STARR Cancer Consortium SCC I16-0057 to I.M. L.S. is supported by the Thompson Family Foundation and by Zuckerman STEM Leadership Program. We are also grateful to Doru M. Paul, M.D., Ph.D. and the Association of Transdisciplinary Society of Personalized Oncology for Combating Cancer, for financial support through the postdoctoral research fellowship STOP Cancer (to L.P.). Last but not least, the authors apologize to those colleagues whose work could not be cited due to length and reference number restrictions. All illustrations were created with BioRender.com.
Footnotes
Patras et. al review the spatiotemporal evolution of the perimetastatic niche (PMN) with a special focus on the immune determinants of this. They also review the clinical evidence of PMN formation and discuss potential therapeutic approaches to reduce immunosuppression in the PMN to prevent and treat metastases.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References:
- 1.Fidler IJ & Nicolson GL Organ selectivity for implantation survival and growth of B16 melanoma variant tumor lines. J Natl Cancer Inst 57, 1199–1202, doi: 10.1093/jnci/57.5.1199 (1976). [DOI] [PubMed] [Google Scholar]
- 2.Kaplan RN et al. VEGFR1-positive haematopoietic bone marrow progenitors initiate the pre-metastatic niche. Nature 438, 820–827, doi: 10.1038/nature04186 (2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hirakawa S. et al. VEGF-C-induced lymphangiogenesis in sentinel lymph nodes promotes tumor metastasis to distant sites. Blood 109, 1010–1017, doi: 10.1182/blood-2006-05-021758 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Psaila B. & Lyden D. The metastatic niche: adapting the foreign soil. Nat Rev Cancer 9, 285–293, doi: 10.1038/nrc2621 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Huang Y. et al. Pulmonary vascular destabilization in the premetastatic phase facilitates lung metastasis. Cancer Res 69, 7529–7537, doi: 10.1158/0008-5472.CAN-08-4382 (2009). [DOI] [PubMed] [Google Scholar]
- 6.Crist SB et al. Unchecked oxidative stress in skeletal muscle prevents outgrowth of disseminated tumour cells. Nat Cell Biol 24, 538–553, doi: 10.1038/s41556-022-00881-4 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Riihimaki M, Thomsen H, Sundquist K, Sundquist J. & Hemminki K. Clinical landscape of cancer metastases. Cancer Med 7, 5534–5542, doi: 10.1002/cam4.1697 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Arpino G, Bardou VJ, Clark GM & Elledge RM Infiltrating lobular carcinoma of the breast: tumor characteristics and clinical outcome. Breast Cancer Res 6, R149–156, doi: 10.1186/bcr767 (2004). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kennecke H. et al. Metastatic behavior of breast cancer subtypes. J Clin Oncol 28, 3271–3277, doi: 10.1200/JCO.2009.25.9820 (2010). [DOI] [PubMed] [Google Scholar]
- 10.Hiratsuka S. et al. MMP9 induction by vascular endothelial growth factor receptor-1 is involved in lung-specific metastasis. Cancer Cell 2, 289–300, doi: 10.1016/s1535-6108(02)00153-8 (2002). [DOI] [PubMed] [Google Scholar]
- 11.Schneider JG, Amend SR & Weilbaecher KN Integrins and bone metastasis: integrating tumor cell and stromal cell interactions. Bone 48, 54–65, doi: 10.1016/j.bone.2010.09.016 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hall CL et al. Type I collagen receptor (alpha2beta1) signaling promotes prostate cancer invasion through RhoC GTPase. Neoplasia 10, 797–803, doi: 10.1593/neo.08380 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Zhou B. et al. Integrin alpha3beta1 can function to promote spontaneous metastasis and lung colonization of invasive breast carcinoma. Mol Cancer Res 12, 143–154, doi: 10.1158/1541-7786.MCR-13-0184 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Pantano F. et al. Integrin alpha5 in human breast cancer is a mediator of bone metastasis and a therapeutic target for the treatment of osteolytic lesions. Oncogene 40, 1284–1299, doi: 10.1038/s41388-020-01603-6 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Tsuzuki Y. et al. alpha 4 integrin plays a critical role in early stages of T lymphocyte migration in Peyer’s patches of rats. Int Immunol 8, 287–295, doi: 10.1093/intimm/8.3.287 (1996). [DOI] [PubMed] [Google Scholar]
- 16.Berlin-Rufenach C. et al. Lymphocyte migration in lymphocyte function-associated antigen (LFA)-1-deficient mice. J Exp Med 189, 1467–1478, doi: 10.1084/jem.189.9.1467 (1999). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Henderson RB et al. The use of lymphocyte function-associated antigen (LFA)-1-deficient mice to determine the role of LFA-1, Mac-1, and alpha4 integrin in the inflammatory response of neutrophils. J Exp Med 194, 219–226, doi: 10.1084/jem.194.2.219 (2001). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Phillipson M. et al. Intraluminal crawling of neutrophils to emigration sites: a molecularly distinct process from adhesion in the recruitment cascade. J Exp Med 203, 2569–2575, doi: 10.1084/jem.20060925 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hoshino A. et al. Tumour exosome integrins determine organotropic metastasis. Nature 527, 329–335, doi: 10.1038/nature15756 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Deasy SK & Erez N. A glitch in the matrix: organ-specific matrisomes in metastatic niches. Trends Cell Biol 32, 110–123, doi: 10.1016/j.tcb.2021.08.001 (2022). [DOI] [PubMed] [Google Scholar]
- 21.Ruoslahti E. Brain extracellular matrix. Glycobiology 6, 489–492, doi: 10.1093/glycob/6.5.489 (1996). [DOI] [PubMed] [Google Scholar]
- 22.Li XQ, Zhang R, Lu H, Yue XM & Huang YF Extracellular Vesicle-Packaged CDH11 and ITGA5 Induce the Premetastatic Niche for Bone Colonization of Breast Cancer Cells. Cancer Res 82, 1560–1574, doi: 10.1158/0008-5472.CAN-21-1331 (2022). [DOI] [PubMed] [Google Scholar]
- 23.Cucanic O, Farnsworth RH & Stacker SA The cellular and molecular mediators of metastasis to the lung. Growth Factors 40, 119–152, doi: 10.1080/08977194.2022.2087520 (2022). [DOI] [PubMed] [Google Scholar]
- 24.Thiele W. et al. Platelet deficiency in Tpo(−/−) mice can both promote and suppress the metastasis of experimental breast tumors in an organ-specific manner. Clin Exp Metastasis 35, 679–689, doi: 10.1007/s10585-018-9924-8 (2018). [DOI] [PubMed] [Google Scholar]
- 25.Hebert JD et al. Proteomic Profiling of the ECM of Xenograft Breast Cancer Metastases in Different Organs Reveals Distinct Metastatic Niches. Cancer Res 80, 1475–1485, doi: 10.1158/0008-5472.CAN-19-2961 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Colombo MV et al. Engineering the early bone metastatic niche through human vascularized immuno bone minitissues. Biofabrication 13, doi: 10.1088/1758-5090/abefea (2021). [DOI] [PubMed] [Google Scholar]
- 27.Kwak TJ & Lee E. Rapid multilayer microfabrication for modeling organotropic metastasis in breast cancer. Biofabrication 13, doi: 10.1088/1758-5090/abbd28 (2020). [DOI] [PubMed] [Google Scholar]
- 28.Carpenter RA, Kwak JG, Peyton SR & Lee J. Implantable pre-metastatic niches for the study of the microenvironmental regulation of disseminated human tumour cells. Nat Biomed Eng 2, 915–929, doi: 10.1038/s41551-018-0307-x (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hiratsuka S, Watanabe A, Aburatani H. & Maru Y. Tumour-mediated upregulation of chemoattractants and recruitment of myeloid cells predetermines lung metastasis. Nat Cell Biol 8, 1369–1375, doi: 10.1038/ncb1507 (2006). [DOI] [PubMed] [Google Scholar]
- 30.Hiratsuka S. et al. Endothelial focal adhesion kinase mediates cancer cell homing to discrete regions of the lungs via E-selectin up-regulation. Proc Natl Acad Sci U S A 108, 3725–3730, doi: 10.1073/pnas.1100446108 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Liu S. et al. Vascular endothelial growth factor plays a critical role in the formation of the pre-metastatic niche via prostaglandin E2. Oncol Rep 32, 2477–2484, doi: 10.3892/or.2014.3516 (2014). [DOI] [PubMed] [Google Scholar]
- 32.Odagiri H. et al. The secreted protein ANGPTL2 promotes metastasis of osteosarcoma cells through integrin alpha5beta1, p38 MAPK, and matrix metalloproteinases. Sci Signal 7, ra7, doi: 10.1126/scisignal.2004612 (2014). [DOI] [PubMed] [Google Scholar]
- 33.Charan M. et al. Tumor secreted ANGPTL2 facilitates recruitment of neutrophils to the lung to promote lung pre-metastatic niche formation and targeting ANGPTL2 signaling affects metastatic disease. Oncotarget 11, 510–522, doi: 10.18632/oncotarget.27433 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Casbon AJ. et al. Invasive breast cancer reprograms early myeloid differentiation in the bone marrow to generate immunosuppressive neutrophils. Proc Natl Acad Sci U S A 112, E566–575, doi: 10.1073/pnas.1424927112 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Deng J. et al. S1PR1-STAT3 signaling is crucial for myeloid cell colonization at future metastatic sites. Cancer Cell 21, 642–654, doi: 10.1016/j.ccr.2012.03.039 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Lee E. et al. Breast cancer cells condition lymphatic endothelial cells within pre-metastatic niches to promote metastasis. Nat Commun 5, 4715, doi: 10.1038/ncomms5715 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Lee JW et al. Hepatocytes direct the formation of a pro-metastatic niche in the liver. Nature 567, 249–252, doi: 10.1038/s41586-019-1004-y (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Melgarejo E, Medina MA, Sanchez-Jimenez F. & Urdiales JL Monocyte chemoattractant protein-1: a key mediator in inflammatory processes. Int J Biochem Cell Biol 41, 998–1001, doi: 10.1016/j.biocel.2008.07.018 (2009). [DOI] [PubMed] [Google Scholar]
- 39.Lebrecht A. et al. Monocyte chemoattractant protein-1 serum levels in patients with breast cancer. Tumour Biol 25, 14–17, doi: 10.1159/000077718 (2004). [DOI] [PubMed] [Google Scholar]
- 40.Qian BZ et al. CCL2 recruits inflammatory monocytes to facilitate breast-tumour metastasis. Nature 475, 222–225, doi: 10.1038/nature10138 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Lu X. & Kang Y. Chemokine (C-C motif) ligand 2 engages CCR2+ stromal cells of monocytic origin to promote breast cancer metastasis to lung and bone. J Biol Chem 284, 29087–29096, doi: 10.1074/jbc.M109.035899 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Sceneay J. et al. Primary tumor hypoxia recruits CD11b+/Ly6Cmed/Ly6G+ immune suppressor cells and compromises NK cell cytotoxicity in the premetastatic niche. Cancer Res 72, 3906–3911, doi: 10.1158/0008-5472.CAN-11-3873 (2012). [DOI] [PubMed] [Google Scholar]
- 43.Granot Z. et al. Tumor entrained neutrophils inhibit seeding in the premetastatic lung. Cancer Cell 20, 300–314, doi: 10.1016/j.ccr.2011.08.012 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Costa-Silva B. et al. Pancreatic cancer exosomes initiate pre-metastatic niche formation in the liver. Nat Cell Biol 17, 816–826, doi: 10.1038/ncb3169 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Deep G. et al. Exosomes secreted by prostate cancer cells under hypoxia promote matrix metalloproteinases activity at pre-metastatic niches. Mol Carcinog 59, 323–332, doi: 10.1002/mc.23157 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Maus RLG et al. Identification of novel, immune-mediating extracellular vesicles in human lymphatic effluent draining primary cutaneous melanoma. Oncoimmunology 8, e1667742, doi: 10.1080/2162402X.2019.1667742 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Peinado H. et al. Melanoma exosomes educate bone marrow progenitor cells toward a pro-metastatic phenotype through MET. Nat Med 18, 883–891, doi: 10.1038/nm.2753 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Shenoy GN et al. Exosomes Associated with Human Ovarian Tumors Harbor a Reversible Checkpoint of T-cell Responses. Cancer Immunol Res 6, 236–247, doi: 10.1158/2326-6066.CIR-17-0113 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Zhang X. & Xiang J. Remodeling the Microenvironment before Occurrence and Metastasis of Cancer. Int J Biol Sci 15, 105–113, doi: 10.7150/ijbs.28669 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Zhang H. et al. Identification of distinct nanoparticles and subsets of extracellular vesicles by asymmetric flow field-flow fractionation. Nat Cell Biol 20, 332–343, doi: 10.1038/s41556-018-0040-4 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Zhang Q. et al. Transfer of Functional Cargo in Exomeres. Cell Rep 27, 940–954 e946, doi: 10.1016/j.celrep.2019.01.009 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Jeppesen DK et al. Reassessment of Exosome Composition. Cell 177, 428–445 e418, doi: 10.1016/j.cell.2019.02.029 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Zhang Q. et al. Supermeres are functional extracellular nanoparticles replete with disease biomarkers and therapeutic targets. Nat Cell Biol 23, 1240–1254, doi: 10.1038/s41556-021-00805-8 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Dieterich LC Mechanisms of extracellular vesicle-mediated immune evasion in melanoma. Front Immunol 13, 1002551, doi: 10.3389/fimmu.2022.1002551 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Duchler M. et al. Melanoma-Derived Extracellular Vesicles Bear the Potential for the Induction of Antigen-Specific Tolerance. Cells 8, doi: 10.3390/cells8070665 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Bretz NP et al. Body fluid exosomes promote secretion of inflammatory cytokines in monocytic cells via Toll-like receptor signaling. J Biol Chem 288, 36691–36702, doi: 10.1074/jbc.M113.512806 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Szajnik M, Czystowska M, Szczepanski MJ, Mandapathil M. & Whiteside TL Tumor-derived microvesicles induce, expand and up-regulate biological activities of human regulatory T cells (Treg). PLoS One 5, e11469, doi: 10.1371/journal.pone.0011469 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Wieckowski EU et al. Tumor-derived microvesicles promote regulatory T cell expansion and induce apoptosis in tumor-reactive activated CD8+ T lymphocytes. J Immunol 183, 3720–3730, doi: 10.4049/jimmunol.0900970 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Muller L, Mitsuhashi M, Simms P, Gooding WE & Whiteside TL Tumor-derived exosomes regulate expression of immune function-related genes in human T cell subsets. Sci Rep 6, 20254, doi: 10.1038/srep20254 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Shenoy GN et al. Sialic Acid-Dependent Inhibition of T Cells by Exosomal Ganglioside GD3 in Ovarian Tumor Microenvironments. J Immunol 201, 3750–3758, doi: 10.4049/jimmunol.1801041 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Ludwig S. et al. Suppression of Lymphocyte Functions by Plasma Exosomes Correlates with Disease Activity in Patients with Head and Neck Cancer. Clin Cancer Res 23, 4843–4854, doi: 10.1158/1078-0432.CCR-16-2819 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Theodoraki MN, Yerneni SS, Hoffmann TK, Gooding WE & Whiteside TL Clinical Significance of PD-L1(+) Exosomes in Plasma of Head and Neck Cancer Patients. Clin Cancer Res 24, 896–905, doi: 10.1158/1078-0432.CCR-17-2664 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Razzo BM et al. Tumor-derived exosomes promote carcinogenesis of murine oral squamous cell carcinoma. Carcinogenesis 41, 625–633, doi: 10.1093/carcin/bgz124 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Peinado H. et al. Pre-metastatic niches: organ-specific homes for metastases. Nat Rev Cancer 17, 302–317, doi: 10.1038/nrc.2017.6 (2017). [DOI] [PubMed] [Google Scholar]
- 65.Wautier JL & Wautier MP Vascular Permeability in Diseases. Int J Mol Sci 23, doi: 10.3390/ijms23073645 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Gupta GP et al. Mediators of vascular remodelling co-opted for sequential steps in lung metastasis. Nature 446, 765–770, doi: 10.1038/nature05760 (2007). [DOI] [PubMed] [Google Scholar]
- 67.Hiratsuka S. et al. Primary tumours modulate innate immune signalling to create pre-metastatic vascular hyperpermeability foci. Nat Commun 4, 1853, doi: 10.1038/ncomms2856 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.You H, Baluszek S. & Kaminska B. Supportive roles of brain macrophages in CNS metastases and assessment of new approaches targeting their functions. Theranostics 10, 2949–2964, doi: 10.7150/thno.40783 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Hongu T. et al. Perivascular tenascin C triggers sequential activation of macrophages and endothelial cells to generate a pro-metastatic vascular niche in the lungs. Nat Cancer 3, 486–504, doi: 10.1038/s43018-022-00353-6 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Kolaczkowska E. et al. Molecular mechanisms of NET formation and degradation revealed by intravital imaging in the liver vasculature. Nat Commun 6, 6673, doi: 10.1038/ncomms7673 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.De Meo ML & Spicer JD The role of neutrophil extracellular traps in cancer progression and metastasis. Semin Immunol 57, 101595, doi: 10.1016/j.smim.2022.101595 (2021). [DOI] [PubMed] [Google Scholar]
- 72.Ward Y. et al. Platelets Promote Metastasis via Binding Tumor CD97 Leading to Bidirectional Signaling that Coordinates Transendothelial Migration. Cell Rep 23, 808–822, doi: 10.1016/j.celrep.2018.03.092 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Tian L. et al. Mutual regulation of tumour vessel normalization and immunostimulatory reprogramming. Nature 544, 250–254, doi: 10.1038/nature21724 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Kulig P. et al. IL17A-Mediated Endothelial Breach Promotes Metastasis Formation. Cancer Immunol Res 4, 26–32, doi: 10.1158/2326-6066.CIR-15-0154 (2016). [DOI] [PubMed] [Google Scholar]
- 75.Rodrigues G. et al. Tumour exosomal CEMIP protein promotes cancer cell colonization in brain metastasis. Nat Cell Biol 21, 1403–1412, doi: 10.1038/s41556-019-0404-4 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Tominaga N. et al. Brain metastatic cancer cells release microRNA-181c-containing extracellular vesicles capable of destructing blood-brain barrier. Nat Commun 6, 6716, doi: 10.1038/ncomms7716 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Kataru RP et al. Tumor Lymphatic Function Regulates Tumor Inflammatory and Immunosuppressive Microenvironments. Cancer Immunol Res 7, 1345–1358, doi: 10.1158/2326-6066.CIR-18-0337 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Li CY, Brown S, Mehrara BJ & Kataru RP Lymphatics in Tumor Progression and Immunomodulation. Int J Mol Sci 23, doi: 10.3390/ijms23042127 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Yanez-Mo M. et al. Biological properties of extracellular vesicles and their physiological functions. J Extracell Vesicles 4, 27066, doi: 10.3402/jev.v4.27066 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Versteeg HH & Ruf W. Emerging insights in tissue factor-dependent signaling events. Semin Thromb Hemost 32, 24–32, doi: 10.1055/s-2006-933337 (2006). [DOI] [PubMed] [Google Scholar]
- 81.Gil-Bernabe AM et al. Recruitment of monocytes/macrophages by tissue factor-mediated coagulation is essential for metastatic cell survival and premetastatic niche establishment in mice. Blood 119, 3164–3175, doi: 10.1182/blood-2011-08-376426 (2012). [DOI] [PubMed] [Google Scholar]
- 82.Wang Y. et al. Platelet P2Y12 is involved in murine pulmonary metastasis. PLoS One 8, e80780, doi: 10.1371/journal.pone.0080780 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Kerr BA et al. Platelet TSP-1 controls prostate cancer-induced osteoclast differentiation and bone marrow-derived cell mobilization through TGFbeta-1. Am J Clin Exp Urol 9, 18–31 (2021). [PMC free article] [PubMed] [Google Scholar]
- 84.Zhuyan J. et al. Critical steps to tumor metastasis: alterations of tumor microenvironment and extracellular matrix in the formation of pre-metastatic and metastatic niche. Cell Biosci 10, 89, doi: 10.1186/s13578-020-00453-9 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Wong CC et al. Hypoxia-inducible factor 1 is a master regulator of breast cancer metastatic niche formation. Proc Natl Acad Sci U S A 108, 16369–16374, doi: 10.1073/pnas.1113483108 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Erler JT et al. Hypoxia-induced lysyl oxidase is a critical mediator of bone marrow cell recruitment to form the premetastatic niche. Cancer Cell 15, 35–44, doi: 10.1016/j.ccr.2008.11.012 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Cox TR et al. The hypoxic cancer secretome induces pre-metastatic bone lesions through lysyl oxidase. Nature 522, 106–110, doi: 10.1038/nature14492 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 88.Chafe SC. et al. Carbonic anhydrase IX promotes myeloid-derived suppressor cell mobilization and establishment of a metastatic niche by stimulating G-CSF production. Cancer Res 75, 996–1008, doi: 10.1158/0008-5472.CAN-14-3000 (2015). [DOI] [PubMed] [Google Scholar]
- 89.Visse R. & Nagase H. Matrix metalloproteinases and tissue inhibitors of metalloproteinases: structure, function, and biochemistry. Circ Res 92, 827–839, doi: 10.1161/01.RES.0000070112.80711.3D (2003). [DOI] [PubMed] [Google Scholar]
- 90.Seubert B. et al. Tissue inhibitor of metalloproteinases (TIMP)-1 creates a premetastatic niche in the liver through SDF-1/CXCR4-dependent neutrophil recruitment in mice. Hepatology 61, 238–248, doi: 10.1002/hep.27378 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Monteran L. & Erez N. The Dark Side of Fibroblasts: Cancer-Associated Fibroblasts as Mediators of Immunosuppression in the Tumor Microenvironment. Front Immunol 10, 1835, doi: 10.3389/fimmu.2019.01835 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Desbois M. & Wang Y. Cancer-associated fibroblasts: Key players in shaping the tumor immune microenvironment. Immunol Rev 302, 241–258, doi: 10.1111/imr.12982 (2021). [DOI] [PubMed] [Google Scholar]
- 93.Lu X. & Kang Y. Organotropism of breast cancer metastasis. J Mammary Gland Biol Neoplasia 12, 153–162, doi: 10.1007/s10911-007-9047-3 (2007). [DOI] [PubMed] [Google Scholar]
- 94.Hynes RO Integrins: a family of cell surface receptors. Cell 48, 549–554, doi: 10.1016/0092-8674(87)90233-9 (1987). [DOI] [PubMed] [Google Scholar]
- 95.Cox TR et al. LOX-mediated collagen crosslinking is responsible for fibrosis-enhanced metastasis. Cancer Res 73, 1721–1732, doi: 10.1158/0008-5472.CAN-12-2233 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Wang Z. et al. Periostin promotes immunosuppressive premetastatic niche formation to facilitate breast tumour metastasis. J Pathol 239, 484–495, doi: 10.1002/path.4747 (2016). [DOI] [PubMed] [Google Scholar]
- 97.Malanchi I. et al. Interactions between cancer stem cells and their niche govern metastatic colonization. Nature 481, 85–89, doi: 10.1038/nature10694 (2011). [DOI] [PubMed] [Google Scholar]
- 98.Mortezaee K. CXCL12/CXCR4 axis in the microenvironment of solid tumors: A critical mediator of metastasis. Life Sci 249, 117534, doi: 10.1016/j.lfs.2020.117534 (2020). [DOI] [PubMed] [Google Scholar]
- 99.Zhao E. et al. Regulatory T cells in the bone marrow microenvironment in patients with prostate cancer. Oncoimmunology 1, 152–161, doi: 10.4161/onci.1.2.18480 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Wang M, Yang X, Wei M. & Wang Z. The Role of CXCL12 Axis in Lung Metastasis of Colorectal Cancer. J Cancer 9, 3898–3903, doi: 10.7150/jca.26383 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.van Deventer HW, Palmieri DA, Wu QP, McCook EC & Serody JS Circulating fibrocytes prepare the lung for cancer metastasis by recruiting Ly-6C+ monocytes via CCL2. J Immunol 190, 4861–4867, doi: 10.4049/jimmunol.1202857 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Bald T. et al. Ultraviolet-radiation-induced inflammation promotes angiotropism and metastasis in melanoma. Nature 507, 109–113, doi: 10.1038/nature13111 (2014). [DOI] [PubMed] [Google Scholar]
- 103.Hiratsuka S. et al. The S100A8-serum amyloid A3-TLR4 paracrine cascade establishes a pre-metastatic phase. Nat Cell Biol 10, 1349–1355, doi: 10.1038/ncb1794 (2008). [DOI] [PubMed] [Google Scholar]
- 104.Ehrchen JM, Sunderkotter C, Foell D, Vogl T. & Roth J. The endogenous Toll-like receptor 4 agonist S100A8/S100A9 (calprotectin) as innate amplifier of infection, autoimmunity, and cancer. J Leukoc Biol 86, 557–566, doi: 10.1189/jlb.1008647 (2009). [DOI] [PubMed] [Google Scholar]
- 105.Sinha P. et al. Proinflammatory S100 proteins regulate the accumulation of myeloid-derived suppressor cells. J Immunol 181, 4666–4675, doi: 10.4049/jimmunol.181.7.4666 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Urban CF et al. Neutrophil extracellular traps contain calprotectin, a cytosolic protein complex involved in host defense against Candida albicans. PLoS Pathog 5, e1000639, doi: 10.1371/journal.ppat.1000639 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Lukanidin E. & Sleeman JP Building the niche: the role of the S100 proteins in metastatic growth. Semin Cancer Biol 22, 216–225, doi: 10.1016/j.semcancer.2012.02.006 (2012). [DOI] [PubMed] [Google Scholar]
- 108.Donato R. et al. Functions of S100 proteins. Curr Mol Med 13, 24–57 (2013). [PMC free article] [PubMed] [Google Scholar]
- 109.Peinado H, Rafii S. & Lyden D. Inflammation joins the “niche”. Cancer Cell 14, 347–349, doi: 10.1016/j.ccr.2008.10.012 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Hoshino A. et al. Extracellular Vesicle and Particle Biomarkers Define Multiple Human Cancers. Cell 182, 1044–1061 e1018, doi: 10.1016/j.cell.2020.07.009 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Cheng P. et al. Inhibition of dendritic cell differentiation and accumulation of myeloid-derived suppressor cells in cancer is regulated by S100A9 protein. J Exp Med 205, 2235–2249, doi: 10.1084/jem.20080132 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Hibino T. et al. S100A9 is a novel ligand of EMMPRIN that promotes melanoma metastasis. Cancer Res 73, 172–183, doi: 10.1158/0008-5472.CAN-11-3843 (2013). [DOI] [PubMed] [Google Scholar]
- 113.Sakaguchi M. et al. Identification of an S100A8 Receptor Neuroplastin-beta and its Heterodimer Formation with EMMPRIN. J Invest Dermatol 136, 2240–2250, doi: 10.1016/j.jid.2016.06.617 (2016). [DOI] [PubMed] [Google Scholar]
- 114.Yan HH et al. Gr-1+CD11b+ myeloid cells tip the balance of immune protection to tumor promotion in the premetastatic lung. Cancer Res 70, 6139–6149, doi: 10.1158/0008-5472.CAN-10-0706 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Chen Q, Zhang XH & Massague J. Macrophage binding to receptor VCAM-1 transmits survival signals in breast cancer cells that invade the lungs. Cancer Cell 20, 538–549, doi: 10.1016/j.ccr.2011.08.025 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Qian BZ et al. FLT1 signaling in metastasis-associated macrophages activates an inflammatory signature that promotes breast cancer metastasis. J Exp Med 212, 1433–1448, doi: 10.1084/jem.20141555 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Mantovani A. et al. The chemokine system in diverse forms of macrophage activation and polarization. Trends Immunol 25, 677–686, doi: 10.1016/j.it.2004.09.015 (2004). [DOI] [PubMed] [Google Scholar]
- 118.Joyce JA & Pollard JW Microenvironmental regulation of metastasis. Nat Rev Cancer 9, 239–252, doi: 10.1038/nrc2618 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Morrissey SM et al. Tumor-derived exosomes drive immunosuppressive macrophages in a pre-metastatic niche through glycolytic dominant metabolic reprogramming. Cell Metab 33, 2040–2058 e2010, doi: 10.1016/j.cmet.2021.09.002 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Shi H. et al. Recruited monocytic myeloid-derived suppressor cells promote the arrest of tumor cells in the premetastatic niche through an IL-1beta-mediated increase in E-selectin expression. Int J Cancer 140, 1370–1383, doi: 10.1002/ijc.30538 (2017). [DOI] [PubMed] [Google Scholar]
- 121.Quail DF et al. Neutrophil phenotypes and functions in cancer: A consensus statement. J Exp Med 219, doi: 10.1084/jem.20220011 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Wang Z. et al. CD62L(dim) Neutrophils Specifically Migrate to the Lung and Participate in the Formation of the Pre-Metastatic Niche of Breast Cancer. Front Oncol 10, 540484, doi: 10.3389/fonc.2020.540484 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Ryckman C, Vandal K, Rouleau P, Talbot M. & Tessier PA Proinflammatory activities of S100: proteins S100A8, S100A9, and S100A8/A9 induce neutrophil chemotaxis and adhesion. J Immunol 170, 3233–3242, doi: 10.4049/jimmunol.170.6.3233 (2003). [DOI] [PubMed] [Google Scholar]
- 124.Goyette J. & Geczy CL Inflammation-associated S100 proteins: new mechanisms that regulate function. Amino Acids 41, 821–842, doi: 10.1007/s00726-010-0528-0 (2011). [DOI] [PubMed] [Google Scholar]
- 125.Qi M. et al. Lin28B-high breast cancer cells promote immune suppression in the lung pre-metastatic niche via exosomes and support cancer progression. Nat Commun 13, 897, doi: 10.1038/s41467-022-28438-x (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Wculek SK & Malanchi I. Neutrophils support lung colonization of metastasis-initiating breast cancer cells. Nature 528, 413–417, doi: 10.1038/nature16140 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Wu CF et al. The lack of type I interferon induces neutrophil-mediated pre-metastatic niche formation in the mouse lung. Int J Cancer 137, 837–847, doi: 10.1002/ijc.29444 (2015). [DOI] [PubMed] [Google Scholar]
- 128.Coffelt SB et al. IL-17-producing gammadelta T cells and neutrophils conspire to promote breast cancer metastasis. Nature 522, 345–348, doi: 10.1038/nature14282 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Wu M, Ma M, Tan Z, Zheng H. & Liu X. Neutrophil: A New Player in Metastatic Cancers. Front Immunol 11, 565165, doi: 10.3389/fimmu.2020.565165 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Leal AC et al. Tumor-Derived Exosomes Induce the Formation of Neutrophil Extracellular Traps: Implications For The Establishment of Cancer-Associated Thrombosis. Sci Rep 7, 6438, doi: 10.1038/s41598-017-06893-7 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Cools-Lartigue J. et al. Neutrophil extracellular traps sequester circulating tumor cells and promote metastasis. J Clin Invest, doi: 10.1172/JCI67484 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Huang YH et al. CEACAM1 regulates TIM-3-mediated tolerance and exhaustion. Nature 517, 386–390, doi: 10.1038/nature13848 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Kaltenmeier C. et al. Neutrophil Extracellular Traps Promote T Cell Exhaustion in the Tumor Microenvironment. Front Immunol 12, 785222, doi: 10.3389/fimmu.2021.785222 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Shahzad MH et al. Neutrophil Extracellular Traps in Cancer Therapy Resistance. Cancers (Basel) 14, doi: 10.3390/cancers14051359 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Xia X. et al. Neutrophil extracellular traps promote metastasis in gastric cancer patients with postoperative abdominal infectious complications. Nat Commun 13, 1017, doi: 10.1038/s41467-022-28492-5 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.El Rayes T. et al. Lung inflammation promotes metastasis through neutrophil protease-mediated degradation of Tsp-1. Proc Natl Acad Sci U S A 112, 16000–16005, doi: 10.1073/pnas.1507294112 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Tomita T, Sakurai Y, Ishibashi S. & Maru Y. Imbalance of Clara cell-mediated homeostatic inflammation is involved in lung metastasis. Oncogene 30, 3429–3439, doi: 10.1038/onc.2011.53 (2011). [DOI] [PubMed] [Google Scholar]
- 138.Aegerter H, Lambrecht BN & Jakubzick CV Biology of lung macrophages in health and disease. Immunity 55, 1564–1580, doi: 10.1016/j.immuni.2022.08.010 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Ju C. & Tacke F. Hepatic macrophages in homeostasis and liver diseases: from pathogenesis to novel therapeutic strategies. Cell Mol Immunol 13, 316–327, doi: 10.1038/cmi.2015.104 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Sevenich L. Brain-Resident Microglia and Blood-Borne Macrophages Orchestrate Central Nervous System Inflammation in Neurodegenerative Disorders and Brain Cancer. Front Immunol 9, 697, doi: 10.3389/fimmu.2018.00697 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Xing F. et al. Loss of XIST in Breast Cancer Activates MSN-c-Met and Reprograms Microglia via Exosomal miRNA to Promote Brain Metastasis. Cancer Res 78, 4316–4330, doi: 10.1158/0008-5472.CAN-18-1102 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Etzerodt A. et al. Tissue-resident macrophages in omentum promote metastatic spread of ovarian cancer. J Exp Med 217, doi: 10.1084/jem.20191869 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Chen XW et al. CYP4A in tumor-associated macrophages promotes pre-metastatic niche formation and metastasis. Oncogene 36, 5045–5057, doi: 10.1038/onc.2017.118 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Mielgo A. & Schmid MC Liver Tropism in Cancer: The Hepatic Metastatic Niche. Cold Spring Harb Perspect Med 10, doi: 10.1101/cshperspect.a037259 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Matsumura H. et al. Kupffer cells decrease metastasis of colon cancer cells to the liver in the early stage. Int J Oncol 45, 2303–2310, doi: 10.3892/ijo.2014.2662 (2014). [DOI] [PubMed] [Google Scholar]
- 146.Jiang K. et al. Exosomal ANGPTL1 attenuates colorectal cancer liver metastasis by regulating Kupffer cell secretion pattern and impeding MMP9 induced vascular leakiness. J Exp Clin Cancer Res 40, 21, doi: 10.1186/s13046-020-01816-3 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Sun H. et al. Hypoxia-Inducible Exosomes Facilitate Liver-Tropic Premetastatic Niche in Colorectal Cancer. Hepatology 74, 2633–2651, doi: 10.1002/hep.32009 (2021). [DOI] [PubMed] [Google Scholar]
- 148.Gan DX et al. Lung Cancer Cells-Controlled Dkk-1 Production in Brain Metastatic Cascade Drive Microglia to Acquire a Pro-tumorigenic Phenotype. Front Cell Dev Biol 8, 591405, doi: 10.3389/fcell.2020.591405 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Hu X. et al. Microglial and macrophage polarization-new prospects for brain repair. Nat Rev Neurol 11, 56–64, doi: 10.1038/nrneurol.2014.207 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Gong Z. et al. Lung fibroblasts facilitate pre-metastatic niche formation by remodeling the local immune microenvironment. Immunity 55, 1483–1500 e1489, doi: 10.1016/j.immuni.2022.07.001 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Ogawa F. et al. Prostanoid induces premetastatic niche in regional lymph nodes. J Clin Invest 124, 4882–4894, doi: 10.1172/JCI73530 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Dunkelberger JR & Song WC Complement and its role in innate and adaptive immune responses. Cell Res 20, 34–50, doi: 10.1038/cr.2009.139 (2010). [DOI] [PubMed] [Google Scholar]
- 153.Reis ES, Mastellos DC, Hajishengallis G. & Lambris JD New insights into the immune functions of complement. Nat Rev Immunol 19, 503–516, doi: 10.1038/s41577-019-0168-x (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Shu C. et al. C3a-C3aR signaling promotes breast cancer lung metastasis via modulating carcinoma associated fibroblasts. J Exp Clin Cancer Res 39, 11, doi: 10.1186/s13046-019-1515-2 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Davidson S. et al. Single-Cell RNA Sequencing Reveals a Dynamic Stromal Niche That Supports Tumor Growth. Cell Rep 31, 107628, doi: 10.1016/j.celrep.2020.107628 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Hsu BE et al. C3a elicits unique migratory responses in immature low-density neutrophils. Oncogene 39, 2612–2623, doi: 10.1038/s41388-020-1169-8 (2020). [DOI] [PubMed] [Google Scholar]
- 157.Zheng Z. et al. Lung mesenchymal stromal cells influenced by Th2 cytokines mobilize neutrophils and facilitate metastasis by producing complement C3. Nat Commun 12, 6202, doi: 10.1038/s41467-021-26460-z (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Monteran L. et al. Chemotherapy-induced complement signaling modulates immunosuppression and metastatic relapse in breast cancer. Nat Commun 13, 5797, doi: 10.1038/s41467-022-33598-x (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Bidwell BN et al. Silencing of Irf7 pathways in breast cancer cells promotes bone metastasis through immune escape. Nat Med 18, 1224–1231, doi: 10.1038/nm.2830 (2012). [DOI] [PubMed] [Google Scholar]
- 160.Hanna RN et al. Patrolling monocytes control tumor metastasis to the lung. Science 350, 985–990, doi: 10.1126/science.aac9407 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Molling JW et al. Low levels of circulating invariant natural killer T cells predict poor clinical outcome in patients with head and neck squamous cell carcinoma. J Clin Oncol 25, 862–868, doi: 10.1200/JCO.2006.08.5787 (2007). [DOI] [PubMed] [Google Scholar]
- 162.Kitamura T, Qian BZ & Pollard JW Immune cell promotion of metastasis. Nat Rev Immunol 15, 73–86, doi: 10.1038/nri3789 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Olkhanud PB et al. Tumor-evoked regulatory B cells promote breast cancer metastasis by converting resting CD4(+) T cells to T-regulatory cells. Cancer Res 71, 3505–3515, doi: 10.1158/0008-5472.CAN-10-4316 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Olkhanud PB et al. Breast cancer lung metastasis requires expression of chemokine receptor CCR4 and regulatory T cells. Cancer Res 69, 5996–6004, doi: 10.1158/0008-5472.CAN-08-4619 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Viguier M. et al. Foxp3 expressing CD4+CD25(high) regulatory T cells are overrepresented in human metastatic melanoma lymph nodes and inhibit the function of infiltrating T cells. J Immunol 173, 1444–1453, doi: 10.4049/jimmunol.173.2.1444 (2004). [DOI] [PubMed] [Google Scholar]
- 166.Curiel TJ et al. Specific recruitment of regulatory T cells in ovarian carcinoma fosters immune privilege and predicts reduced survival. Nat Med 10, 942–949, doi: 10.1038/nm1093 (2004). [DOI] [PubMed] [Google Scholar]
- 167.Lai C. et al. Characteristics of immunosuppressive regulatory T cells in cutaneous squamous cell carcinomas and role in metastasis. Lancet 385 Suppl 1, S59, doi: 10.1016/S0140-6736(15)60374-9 (2015). [DOI] [PubMed] [Google Scholar]
- 168.Kaczanowska S. et al. Genetically engineered myeloid cells rebalance the core immune suppression program in metastasis. Cell 184, 2033–2052 e2021, doi: 10.1016/j.cell.2021.02.048 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Xia A, Zhang Y, Xu J, Yin T. & Lu XJ T Cell Dysfunction in Cancer Immunity and Immunotherapy. Front Immunol 10, 1719, doi: 10.3389/fimmu.2019.01719 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Monteiro AC et al. T cells induce pre-metastatic osteolytic disease and help bone metastases establishment in a mouse model of metastatic breast cancer. PLoS One 8, e68171, doi: 10.1371/journal.pone.0068171 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Hayday AC [gamma][delta] cells: a right time and a right place for a conserved third way of protection. Annu Rev Immunol 18, 975–1026, doi: 10.1146/annurev.immunol.18.1.975 (2000). [DOI] [PubMed] [Google Scholar]
- 172.Xu W, Lau ZWX, Fulop T. & Larbi A. The Aging of gammadelta T Cells. Cells 9, doi: 10.3390/cells9051181 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Gu Y. et al. Tumor-educated B cells selectively promote breast cancer lymph node metastasis by HSPA4-targeting IgG. Nat Med 25, 312–322, doi: 10.1038/s41591-018-0309-y (2019). [DOI] [PubMed] [Google Scholar]
- 174.Park EM et al. Targeting the gut and tumor microbiota in cancer. Nat Med 28, 690–703, doi: 10.1038/s41591-022-01779-2 (2022). [DOI] [PubMed] [Google Scholar]
- 175.Arthur JC et al. Intestinal inflammation targets cancer-inducing activity of the microbiota. Science 338, 120–123, doi: 10.1126/science.1224820 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Ma X. et al. Sodium butyrate modulates gut microbiota and immune response in colorectal cancer liver metastatic mice. Cell Biol Toxicol 36, 509–515, doi: 10.1007/s10565-020-09518-4 (2020). [DOI] [PubMed] [Google Scholar]
- 177.Ma C. et al. Gut microbiome-mediated bile acid metabolism regulates liver cancer via NKT cells. Science 360, doi: 10.1126/science.aan5931 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Yin H. et al. Fusobacterium nucleatum promotes liver metastasis in colorectal cancer by regulating the hepatic immune niche and altering gut microbiota. Aging (Albany NY) 14, 1941–1958, doi: 10.18632/aging.203914 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Qiu M. et al. Modulation of intestinal microbiota by glycyrrhizic acid prevents high-fat diet-enhanced pre-metastatic niche formation and metastasis. Mucosal Immunol 12, 945957, doi: 10.1038/s41385-019-0144-6 (2019). [DOI] [PubMed] [Google Scholar]
- 180.Bertocchi A. et al. Gut vascular barrier impairment leads to intestinal bacteria dissemination and colorectal cancer metastasis to liver. Cancer Cell 39, 708–724 e711, doi: 10.1016/j.ccell.2021.03.004 (2021). [DOI] [PubMed] [Google Scholar]
- 181.Nejman D. et al. The human tumor microbiome is composed of tumor type-specific intracellular bacteria. Science 368, 973–980, doi: 10.1126/science.aay9189 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Strauss J. et al. Invasive potential of gut mucosa-derived Fusobacterium nucleatum positively correlates with IBD status of the host. Inflamm Bowel Dis 17, 1971–1978, doi: 10.1002/ibd.21606 (2011). [DOI] [PubMed] [Google Scholar]
- 183.Gevers D. et al. The treatment-naive microbiome in new-onset Crohn’s disease. Cell Host Microbe 15, 382–392, doi: 10.1016/j.chom.2014.02.005 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Bullman S. et al. Analysis of Fusobacterium persistence and antibiotic response in colorectal cancer. Science 358, 1443–1448, doi: 10.1126/science.aal5240 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Rubinstein MR et al. Fusobacterium nucleatum promotes colorectal carcinogenesis by modulating E-cadherin/beta-catenin signaling via its FadA adhesin. Cell Host Microbe 14, 195–206, doi: 10.1016/j.chom.2013.07.012 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Wang D, Sun H, Wei J, Cen B. & DuBois RN CXCL1 Is Critical for Premetastatic Niche Formation and Metastasis in Colorectal Cancer. Cancer Res 77, 3655–3665, doi: 10.1158/0008-5472.CAN-16-3199 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Casasanta MA et al. Fusobacterium nucleatum host-cell binding and invasion induces IL-8 and CXCL1 secretion that drives colorectal cancer cell migration. Sci Signal 13, doi: 10.1126/scisignal.aba9157 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Cheng P. et al. Capsaicin shapes gut microbiota and pre-metastatic niche to facilitate cancer metastasis to liver. Pharmacol Res 188, 106643, doi: 10.1016/j.phrs.2022.106643 (2023). [DOI] [PubMed] [Google Scholar]
- 189.Bergers G. & Fendt SM The metabolism of cancer cells during metastasis. Nat Rev Cancer 21, 162–180, doi: 10.1038/s41568-020-00320-2 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Morrot A. et al. Metabolic Symbiosis and Immunomodulation: How Tumor Cell-Derived Lactate May Disturb Innate and Adaptive Immune Responses. Front Oncol 8, 81, doi: 10.3389/fonc.2018.00081 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Kurpinska A. et al. Proteomic characterization of early lung response to breast cancer metastasis in mice. Exp Mol Pathol 107, 129–140, doi: 10.1016/j.yexmp.2019.02.001 (2019). [DOI] [PubMed] [Google Scholar]
- 192.Fong MY et al. Breast-cancer-secreted miR-122 reprograms glucose metabolism in premetastatic niche to promote metastasis. Nat Cell Biol 17, 183–194, doi: 10.1038/ncb3094 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Ladanyi A. et al. Adipocyte-induced CD36 expression drives ovarian cancer progression and metastasis. Oncogene 37, 2285–2301, doi: 10.1038/s41388-017-0093-z (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Natsume M. et al. Omental adipocytes promote peritoneal metastasis of gastric cancer through the CXCL2-VEGFA axis. Br J Cancer 123, 459–470, doi: 10.1038/s41416-020-0898-3 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Wilde L. et al. Metabolic coupling and the Reverse Warburg Effect in cancer: Implications for novel biomarker and anticancer agent development. Semin Oncol 44, 198–203, doi: 10.1053/j.seminoncol.2017.10.004 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Huang YC et al. Involvement of ACACA (acetyl-CoA carboxylase alpha) in the lung pre-metastatic niche formation in breast cancer by senescence phenotypic conversion in fibroblasts. Cell Oncol (Dordr), doi: 10.1007/s13402-022-00767-5 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Li M, Xian HC, Tang YJ, Liang XH & Tang YL Fatty acid oxidation: driver of lymph node metastasis. Cancer Cell Int 21, 339, doi: 10.1186/s12935-021-02057-w (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Altea-Manzano P. et al. A palmitate-rich metastatic niche enables metastasis growth via p65 acetylation resulting in pro-metastatic NF-kappaB signaling. Nat Cancer, doi: 10.1038/s43018-023-00513-2 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Prima V, Kaliberova LN, Kaliberov S, Curiel DT & Kusmartsev S. COX2/mPGES1/PGE2 pathway regulates PD-L1 expression in tumor-associated macrophages and myeloid-derived suppressor cells. Proc Natl Acad Sci U S A 114, 1117–1122, doi: 10.1073/pnas.1612920114 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Kerkhoff C, Sorg C, Tandon NN & Nacken W. Interaction of S100A8/S100A9-arachidonic acid complexes with the scavenger receptor CD36 may facilitate fatty acid uptake by endothelial cells. Biochemistry 40, 241–248, doi: 10.1021/bi001791k (2001). [DOI] [PubMed] [Google Scholar]
- 201.Baek AE et al. The cholesterol metabolite 27 hydroxycholesterol facilitates breast cancer metastasis through its actions on immune cells. Nat Commun 8, 864, doi: 10.1038/s41467-017-00910-z (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Ma X. et al. CD36-mediated ferroptosis dampens intratumoral CD8(+) T cell effector function and impairs their antitumor ability. Cell Metab 33, 1001–1012 e1005, doi: 10.1016/j.cmet.2021.02.015 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Fracchia KM, Pai CY & Walsh CM Modulation of T Cell Metabolism and Function through Calcium Signaling. Front Immunol 4, 324, doi: 10.3389/fimmu.2013.00324 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Trebak M. & Kinet JP Calcium signalling in T cells. Nat Rev Immunol 19, 154–169, doi: 10.1038/s41577-018-0110-7 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Colon-Echevarria CB, Lamboy-Caraballo R, Aquino-Acevedo AN & Armaiz-Pena GN Neuroendocrine Regulation of Tumor-Associated Immune Cells. Front Oncol 9, 1077, doi: 10.3389/fonc.2019.01077 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Chrousos GP & Kino T. Glucocorticoid signaling in the cell. Expanding clinical implications to complex human behavioral and somatic disorders. Ann N Y Acad Sci 1179, 153–166, doi: 10.1111/j.1749-6632.2009.04988.x (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Chida Y, Hamer M, Wardle J. & Steptoe A. Do stress-related psychosocial factors contribute to cancer incidence and survival? Nat Clin Pract Oncol 5, 466–475, doi: 10.1038/ncponc1134 (2008). [DOI] [PubMed] [Google Scholar]
- 208.Antoni MH & Dhabhar FS The impact of psychosocial stress and stress management on immune responses in patients with cancer. Cancer 125, 1417–1431, doi: 10.1002/cncr.31943 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Qiao G, Chen M, Bucsek MJ, Repasky EA & Hylander BL Adrenergic Signaling: A Targetable Checkpoint Limiting Development of the Antitumor Immune Response. Front Immunol 9, 164, doi: 10.3389/fimmu.2018.00164 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Rosenne E. et al. In vivo suppression of NK cell cytotoxicity by stress and surgery: glucocorticoids have a minor role compared to catecholamines and prostaglandins. Brain Behav Immun 37, 207–219, doi: 10.1016/j.bbi.2013.12.007 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Bucsek MJ et al. beta-Adrenergic Signaling in Mice Housed at Standard Temperatures Suppresses an Effector Phenotype in CD8(+) T Cells and Undermines Checkpoint Inhibitor Therapy. Cancer Res 77, 5639–5651, doi: 10.1158/0008-5472.CAN-17-0546 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Mohammadpour H. et al. beta2 adrenergic receptor-mediated signaling regulates the immunosuppressive potential of myeloid-derived suppressor cells. J Clin Invest 129, 5537–5552, doi: 10.1172/JCI129502 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Le CP et al. Chronic stress in mice remodels lymph vasculature to promote tumour cell dissemination. Nat Commun 7, 10634, doi: 10.1038/ncomms10634 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Chen H. et al. Chronic psychological stress promotes lung metastatic colonization of circulating breast cancer cells by decorating a pre-metastatic niche through activating beta-adrenergic signaling. J Pathol 244, 49–60, doi: 10.1002/path.4988 (2018). [DOI] [PubMed] [Google Scholar]
- 215.Shaashua L. et al. Perioperative COX-2 and beta-Adrenergic Blockade Improves Metastatic Biomarkers in Breast Cancer Patients in a Phase-II Randomized Trial. Clin Cancer Res 23, 4651–4661, doi: 10.1158/1078-0432.CCR-17-0152 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Katayama Y. et al. Signals from the sympathetic nervous system regulate hematopoietic stem cell egress from bone marrow. Cell 124, 407–421, doi: 10.1016/j.cell.2005.10.041 (2006). [DOI] [PubMed] [Google Scholar]
- 217.Campbell JP et al. Stimulation of host bone marrow stromal cells by sympathetic nerves promotes breast cancer bone metastasis in mice. PLoS Biol 10, e1001363, doi: 10.1371/journal.pbio.1001363 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Ieguchi K. et al. The Sympathetic Nervous System Contributes to the Establishment of Pre-Metastatic Pulmonary Microenvironments. Int J Mol Sci 23, doi: 10.3390/ijms231810652 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Kasurinen A. et al. High serum MMP-14 predicts worse survival in gastric cancer. PLoS One 13, e0208800, doi: 10.1371/journal.pone.0208800 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220.Ren F. et al. Overexpression of MMP Family Members Functions as Prognostic Biomarker for Breast Cancer Patients: A Systematic Review and Meta-Analysis. PLoS One 10, e0135544, doi: 10.1371/journal.pone.0135544 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Davidson B. et al. High levels of MMP-2, MMP-9, MT1-MMP and TIMP-2 mRNA correlate with poor survival in ovarian carcinoma. Clin Exp Metastasis 17, 799–808, doi: 10.1023/a:1006723011835 (1999). [DOI] [PubMed] [Google Scholar]
- 222.Grunwald B. et al. Pancreatic Premalignant Lesions Secrete Tissue Inhibitor of Metalloproteinases-1, Which Activates Hepatic Stellate Cells Via CD63 Signaling to Create a Premetastatic Niche in the Liver. Gastroenterology 151, 1011–1024 e1017, doi: 10.1053/j.gastro.2016.07.043 (2016). [DOI] [PubMed] [Google Scholar]
- 223.Dai J. et al. Primary prostate cancer educates bone stroma through exosomal pyruvate kinase M2 to promote bone metastasis. J Exp Med 216, 2883–2899, doi: 10.1084/jem.20190158 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Silvers CR, Messing EM, Miyamoto H. & Lee YF Tenascin-C expression in the lymph node pre-metastatic niche in muscle-invasive bladder cancer. Br J Cancer 125, 1399–1407, doi: 10.1038/s41416-021-01554-z (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Tyagi A. et al. Nicotine promotes breast cancer metastasis by stimulating N2 neutrophils and generating pre-metastatic niche in lung. Nat Commun 12, 474, doi: 10.1038/s41467-020-20733-9 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 226.Diaz-Montero CM et al. Increased circulating myeloid-derived suppressor cells correlate with clinical cancer stage, metastatic tumor burden, and doxorubicin-cyclophosphamide chemotherapy. Cancer Immunol Immunother 58, 49–59, doi: 10.1007/s00262-008-0523-4 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227.Zhang W. et al. CD8+ T-cell immunosurveillance constrains lymphoid premetastatic myeloid cell accumulation. Eur J Immunol 45, 71–81, doi: 10.1002/eji.201444467 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228.Yang LY et al. Increased neutrophil extracellular traps promote metastasis potential of hepatocellular carcinoma via provoking tumorous inflammatory response. J Hematol Oncol 13, 3, doi: 10.1186/s13045-019-0836-0 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 229.SU X. et al. Effect of neutrophil extracellular traps on tumor lymph nodes. Journal of Clinical Oncology 40, 4033–4033, doi: 10.1200/JCO.2022.40.16_suppl.4033 (2022).36054870 [DOI] [Google Scholar]
- 230.Pal SK et al. Clinical and Translational Assessment of VEGFR1 as a Mediator of the Premetastatic Niche in High-Risk Localized Prostate Cancer. Mol Cancer Ther 14, 2896–2900, doi: 10.1158/1535-7163.MCT-15-0367 (2015). [DOI] [PubMed] [Google Scholar]
- 231.Fujita S, Sumi M, Tatsukawa E, Nagano K. & Katase N. Expressions of extracellular matrix-remodeling factors in lymph nodes from oral cancer patients. Oral Dis 26, 1424–1431, doi: 10.1111/odi.13419 (2020). [DOI] [PubMed] [Google Scholar]
- 232.Balsat C. et al. A specific immune and lymphatic profile characterizes the pre-metastatic state of the sentinel lymph node in patients with early cervical cancer. Oncoimmunology 6, e1265718, doi: 10.1080/2162402X.2016.1265718 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Chung MK et al. Lymphatic vessels and high endothelial venules are increased in the sentinel lymph nodes of patients with oral squamous cell carcinoma before the arrival of tumor cells. Ann Surg Oncol 19, 1595–1601, doi: 10.1245/s10434-011-2154-9 (2012). [DOI] [PubMed] [Google Scholar]
- 234.Bekkhus T. et al. Remodeling of the Lymph Node High Endothelial Venules Reflects Tumor Invasiveness in Breast Cancer and is Associated with Dysregulation of Perivascular Stromal Cells. Cancers (Basel) 13, doi: 10.3390/cancers13020211 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 235.du Bois H, Heim TA & Lund AW Tumor-draining lymph nodes: At the crossroads of metastasis and immunity. Sci Immunol 6, eabg3551, doi: 10.1126/sciimmunol.abg3551 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 236.Hou X. et al. Silibinin Inhibits NSCLC Metastasis by Targeting the EGFR/LOX Pathway. Front Pharmacol 9, 21, doi: 10.3389/fphar.2018.00021 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Huang YL et al. Collagen-rich omentum is a premetastatic niche for integrin alpha2-mediated peritoneal metastasis. Elife 9, doi: 10.7554/eLife.59442 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 238.Lee W. et al. Neutrophils facilitate ovarian cancer premetastatic niche formation in the omentum. J Exp Med 216, 176–194, doi: 10.1084/jem.20181170 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239.Feygenzon V. et al. Unique cellular interactions between pancreatic cancer cells and the omentum. PLoS One 12, e0179862, doi: 10.1371/journal.pone.0179862 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 240.Bojmar L. et al. Abstract PR012: Perioperative liver biopsy captures features of the liver pre-metastatic niche and predicts metastatic outcome after pancreatic cancer resection. Cancer Research 82, PR012-PR012, doi: 10.1158/1538-7445.Panca22-pr012 (2022). [DOI] [Google Scholar]
- 241.Cai C, Sun H, Hu L. & Fan Z. Visualization of integrin molecules by fluorescence imaging and techniques. Biocell 45, 229–257, doi: 10.32604/biocell.2021.014338 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 242.Haubner R. et al. Noninvasive imaging of alpha(v)beta3 integrin expression using 18F-labeled RGD-containing glycopeptide and positron emission tomography. Cancer Res 61, 1781–1785 (2001). [PubMed] [Google Scholar]
- 243.Ye Y. & Chen X. Integrin targeting for tumor optical imaging. Theranostics 1, 102–126, doi: 10.7150/thno/v01p0102 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244.Haubner R. et al. Noninvasive visualization of the activated alphavbeta3 integrin in cancer patients by positron emission tomography and [18F]Galacto-RGD. PLoS Med 2, e70, doi: 10.1371/journal.pmed.0020070 (2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 245.Hausner SH et al. Preclinical Development and First-in-Human Imaging of the Integrin alphavbeta6 with [(18)F]alphavbeta6-Binding Peptide in Metastatic Carcinoma. Clin Cancer Res 25, 1206–1215, doi: 10.1158/1078-0432.CCR-18-2665 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246.Qi Y. et al. Radiomics analysis of lung CT image for the early detection of metastases in patients with breast cancer: preliminary findings from a retrospective cohort study. Eur Radiol 30, 4545–4556, doi: 10.1007/s00330-020-06745-5 (2020). [DOI] [PubMed] [Google Scholar]
- 247.Li R. et al. Computed tomography reveals microenvironment changes in premetastatic lung. Eur Radiol 31, 4340–4349, doi: 10.1007/s00330-020-07500-6 (2021). [DOI] [PubMed] [Google Scholar]
- 248.Zambirinis CP et al. Recurrence After Resection of Pancreatic Cancer: Can Radiomics Predict Patients at Greatest Risk of Liver Metastasis? Ann Surg Oncol 29, 4962–4974, doi: 10.1245/s10434-022-11579-0 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 249.Liang M. et al. Whole-liver enhanced CT radiomics analysis to predict metachronous liver metastases after rectal cancer surgery. Cancer Imaging 22, 50, doi: 10.1186/s40644-022-00485-z (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 250.Lubner MG, Jones D, Kloke J, Said A. & Pickhardt PJ CT texture analysis of the liver for assessing hepatic fibrosis in patients with hepatitis C virus. Br J Radiol 92, 20180153, doi: 10.1259/bjr.20180153 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 251.Daenen LG et al. Chemotherapy enhances metastasis formation via VEGFR-1-expressing endothelial cells. Cancer Res 71, 6976–6985, doi: 10.1158/0008-5472.CAN-11-0627 (2011). [DOI] [PubMed] [Google Scholar]
- 252.Keklikoglou I. et al. Chemotherapy elicits pro-metastatic extracellular vesicles in breast cancer models. Nat Cell Biol 21, 190–202, doi: 10.1038/s41556-018-0256-3 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 253.Karagiannis GS, Condeelis JS & Oktay MH Chemotherapy-induced metastasis: mechanisms and translational opportunities. Clin Exp Metastasis 35, 269–284, doi: 10.1007/s10585-017-9870-x (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 254.Vatner RE & Formenti SC Myeloid-derived cells in tumors: effects of radiation. Semin Radiat Oncol 25, 18–27, doi: 10.1016/j.semradonc.2014.07.008 (2015). [DOI] [PubMed] [Google Scholar]
- 255.Shinde-Jadhav S. et al. Role of neutrophil extracellular traps in radiation resistance of invasive bladder cancer. Nat Commun 12, 2776, doi: 10.1038/s41467-021-23086-z (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 256.Hendricks A. et al. Longitudinal Analysis of Circulating Tumor Cells in Colorectal Cancer Patients by a Cytological and Molecular Approach: Feasibility and Clinical Application. Front Oncol 11, 646885, doi: 10.3389/fonc.2021.646885 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 257.Martin OA, Anderson RL, Narayan K. & MacManus MP Does the mobilization of circulating tumour cells during cancer therapy cause metastasis? Nat Rev Clin Oncol 14, 32–44, doi: 10.1038/nrclinonc.2016.128 (2017). [DOI] [PubMed] [Google Scholar]
- 258.Ju M. et al. Application of a telomerase-based circulating tumor cell (CTC) assay in bladder cancer patients receiving postoperative radiation therapy: a case study. Cancer Biol Ther 15, 683–687, doi: 10.4161/cbt.28412 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 259.Dorsey JF et al. Tracking viable circulating tumor cells (CTCs) in the peripheral blood of non-small cell lung cancer (NSCLC) patients undergoing definitive radiation therapy: pilot study results. Cancer 121, 139–149, doi: 10.1002/cncr.28975 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 260.Cifuentes FF, Valenzuela RH, Contreras HR & Castellon EA Surgical cytoreduction of the primary tumor reduces metastatic progression in a mouse model of prostate cancer. Oncol Rep 34, 2837–2844, doi: 10.3892/or.2015.4319 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 261.Mahjoub S. & Heidenreich A. Oligometastatic prostate cancer: definition and the role of local and systemic therapy: a narrative review. Transl Androl Urol 10, 3167–3175, doi: 10.21037/tau-20-1033 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 262.Edwards SC, Hoevenaar WHM & Coffelt SB Emerging immunotherapies for metastasis. Br J Cancer 124, 37–48, doi: 10.1038/s41416-020-01160-5 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 263.Grum-Schwensen B. et al. S100A4-neutralizing antibody suppresses spontaneous tumor progression, pre-metastatic niche formation and alters T-cell polarization balance. BMC Cancer 15, 44, doi: 10.1186/s12885-015-1034-2 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 264.Correia AL et al. Hepatic stellate cells suppress NK cell-sustained breast cancer dormancy. Nature 594, 566–571, doi: 10.1038/s41586-021-03614-z (2021). [DOI] [PubMed] [Google Scholar]
- 265.He B. et al. Remodeling of Metastatic Vasculature Reduces Lung Colonization and Sensitizes Overt Metastases to Immunotherapy. Cell Rep 30, 714–724 e715, doi: 10.1016/j.celrep.2019.12.013 (2020). [DOI] [PubMed] [Google Scholar]
- 266.Low V, Blenis J. & Gomes AP Targeting the premetastatic niche: epigenetic therapies in the spotlight. Signal Transduct Target Ther 5, 68, doi: 10.1038/s41392-020-0165-3 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 267.Lu Z. et al. Epigenetic therapy inhibits metastases by disrupting premetastatic niches. Nature 579, 284–290, doi: 10.1038/s41586-020-2054-x (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 268.Chopra V, Sangarappillai RM, Romero-Canelón I. & Jones AM Lysyl Oxidase Like-2 (LOXL2): An Emerging Oncology Target. Advanced Therapeutics 3, 1900119, doi: 10.1002/adtp.201900119 (2020). [DOI] [Google Scholar]
- 269.Kusmartsev S. et al. Reversal of myeloid cell-mediated immunosuppression in patients with metastatic renal cell carcinoma. Clin Cancer Res 14, 8270–8278, doi: 10.1158/1078-0432.CCR-08-0165 (2008). [DOI] [PubMed] [Google Scholar]
- 270.Brinkmann V. et al. Neutrophil extracellular traps kill bacteria. Science 303, 1532–1535, doi: 10.1126/science.1092385 (2004). [DOI] [PubMed] [Google Scholar]
- 271.Zhang Y. et al. Interleukin-17-induced neutrophil extracellular traps mediate resistance to checkpoint blockade in pancreatic cancer. J Exp Med 217, doi: 10.1084/jem.20190354 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 272.Xu M. et al. Systemic metastasis-targeted nanotherapeutic reinforces tumor surgical resection and chemotherapy. Nat Commun 12, 3187, doi: 10.1038/s41467-021-23466-5 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 273.Sahin U. et al. An RNA vaccine drives immunity in checkpoint-inhibitor-treated melanoma. Nature 585, 107–112, doi: 10.1038/s41586-020-2537-9 (2020). [DOI] [PubMed] [Google Scholar]
- 274.Chen H, Huang P, Chen J. & Song E. Abstract 6123: High-fat diets promote lung metastasis of breast cancer by activating lung fibroblasts. Cancer Research 82, 6123–6123, doi: 10.1158/1538-7445.Am2022-6123 (2022). [DOI] [Google Scholar]
- 275.Yu Y. et al. High-Fat Diet Enhances the Liver Metastasis Potential of Colorectal Cancer through Microbiota Dysbiosis. Cancers (Basel) 14, doi: 10.3390/cancers14112573 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 276.Wang S. et al. Macrophage-tumor chimeric exosomes accumulate in lymph node and tumor to activate the immune response and the tumor microenvironment. Sci Transl Med 13, eabb6981, doi: 10.1126/scitranslmed.abb6981 (2021). [DOI] [PubMed] [Google Scholar]
- 277.Klingelhofer J. et al. Anti-S100A4 antibody suppresses metastasis formation by blocking stroma cell invasion. Neoplasia 14, 1260–1268, doi: 10.1593/neo.121554 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 278.Ji Q. et al. Primary tumors release ITGBL1-rich extracellular vesicles to promote distal metastatic tumor growth through fibroblast-niche formation. Nat Commun 11, 1211, doi: 10.1038/s41467-020-14869-x (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 279.Crist SB & Ghajar CM When a House Is Not a Home: A Survey of Antimetastatic Niches and Potential Mechanisms of Disseminated Tumor Cell Suppression. Annu Rev Pathol 16, 409–432, doi: 10.1146/annurev-pathmechdis-012419-032647 (2021). [DOI] [PubMed] [Google Scholar]
- 280.Paget S. The distribution of secondary growths in cancer of the breast. 1889. Cancer Metastasis Rev 8, 98–101 (1889). [PubMed] [Google Scholar]
- 281.Disibio G. & French SW Metastatic patterns of cancers: results from a large autopsy study. Arch Pathol Lab Med 132, 931–939, doi: 10.1043/1543-2165(2008)132[931:MPOCRF]2.0.CO;2 (2008). [DOI] [PubMed] [Google Scholar]
- 282.Willis RA in Postgrad Med J Vol. 29 160 (1953). [Google Scholar]
- 283.Chiabrando D, Vinchi F, Fiorito V, Mercurio S. & Tolosano E. Heme in pathophysiology: a matter of scavenging, metabolism and trafficking across cell membranes. Front Pharmacol 5, 61, doi: 10.3389/fphar.2014.00061 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 284.Everse J. & Hsia N. The toxicities of native and modified hemoglobins. Free Radic Biol Med 22, 1075–1099, doi: 10.1016/s0891-5849(96)00499-6 (1997). [DOI] [PubMed] [Google Scholar]
- 285.Poncin S. et al. Oxidative stress in the thyroid gland: from harmlessness to hazard depending on the iodine content. Endocrinology 149, 424–433, doi: 10.1210/en.2007-0951 (2008). [DOI] [PubMed] [Google Scholar]
- 286.Plebanek MP et al. Pre-metastatic cancer exosomes induce immune surveillance by patrolling monocytes at the metastatic niche. Nat Commun 8, 1319, doi: 10.1038/s41467-017-01433-3 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 287.Hiratsuka S. et al. Hepato-entrained B220(+)CD11c(+)NK1.1(+) cells regulate pre-metastatic niche formation in the lung. EMBO Mol Med 10, doi: 10.15252/emmm.201708643 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 288.Angelidis I. et al. An atlas of the aging lung mapped by single cell transcriptomics and deep tissue proteomics. Nat Commun 10, 963, doi: 10.1038/s41467-019-08831-9 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 289.Martinez-Jimenez CP et al. Aging increases cell-to-cell transcriptional variability upon immune stimulation. Science 355, 1433–1436, doi: 10.1126/science.aah4115 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 290.Garcia DI et al. High-Fat Diet Drives an Aggressive Pancreatic Cancer Phenotype. J Surg Res 264, 163–172, doi: 10.1016/j.jss.2020.10.007 (2021). [DOI] [PubMed] [Google Scholar]
- 291.Collins N. et al. The Bone Marrow Protects and Optimizes Immunological Memory during Dietary Restriction. Cell 178, 1088–1101 e1015, doi: 10.1016/j.cell.2019.07.049 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 292.Collins N. & Belkaid Y. Control of immunity via nutritional interventions. Immunity 55, 210–223, doi: 10.1016/j.immuni.2022.01.004 (2022). [DOI] [PubMed] [Google Scholar]
- 293.Rundqvist H. et al. Cytotoxic T-cells mediate exercise-induced reductions in tumor growth. Elife 9, doi: 10.7554/eLife.59996 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 294.Sheinboim D. et al. An exercise-induced metabolic shield in distant organs blocks cancer progression and metastatic dissemination. Cancer Res, doi: 10.1158/0008-5472.CAN-22-0237 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 295.Darkwah S. et al. Potential Roles of Muscle-Derived Extracellular Vesicles in Remodeling Cellular Microenvironment: Proposed Implications of the Exercise-Induced Myokine, Irisin. Front Cell Dev Biol 9, 634853, doi: 10.3389/fcell.2021.634853 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 296.Idorn M. & Thor Straten P. Chemokine Receptors and Exercise to Tackle the Inadequacy of T Cell Homing to the Tumor Site. Cells 7, doi: 10.3390/cells7080108 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 297.Gomes-Santos IL et al. Exercise Training Improves Tumor Control by Increasing CD8(+) T-cell Infiltration via CXCR3 Signaling and Sensitizes Breast Cancer to Immune Checkpoint Blockade. Cancer Immunol Res 9, 765–778, doi: 10.1158/2326-6066.CIR-20-0499 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 298.Shokeen M. et al. Molecular imaging of very late antigen-4 (alpha4beta1 integrin) in the premetastatic niche. J Nucl Med 53, 779–786, doi: 10.2967/jnumed.111.100073 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 299.Liu Y. et al. Premetastatic soil and prevention of breast cancer brain metastasis. Neuro Oncol 15, 891–903, doi: 10.1093/neuonc/not031 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 300.Zeng Z. et al. HAO1-mediated oxalate metabolism promotes lung pre-metastatic niche formation by inducing neutrophil extracellular traps. Oncogene 41, 3719–3731, doi: 10.1038/s41388-022-02248-3 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 301.Krijgsman D. et al. Characterization of circulating T-, NK-, and NKT cell subsets in patients with colorectal cancer: the peripheral blood immune cell profile. Cancer Immunol Immunother 68, 1011–1024, doi: 10.1007/s00262-019-02343-7 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 302.Eisenblaetter M. et al. Visualization of Tumor-Immune Interaction - Target-Specific Imaging of S100A8/A9 Reveals Pre-Metastatic Niche Establishment. Theranostics 7, 2392–2401, doi: 10.7150/thno.17138 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 303.Olmeda D. et al. Whole-body imaging of lymphovascular niches identifies pre-metastatic roles of midkine. Nature 546, 676–680, doi: 10.1038/nature22977 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 304.Paidi SK et al. Label-Free Raman Spectroscopy Detects Stromal Adaptations in Premetastatic Lungs Primed by Breast Cancer. Cancer Res 77, 247–256, doi: 10.1158/0008-5472.CAN-16-1862 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]