Abstract
We investigated whether sleep health (each individual dimension and a composite measure) was associated with better glycemia among a cohort of young adults with type 1 diabetes (T1D) (mean age 21.5 years, mean BMI 24.55 kg/m2). Multiple validated self-report questionnaires were completed, and raw continuous glucose monitor data were shared. One self-reported sleep characteristic for each of the five sleep health dimensions was selected. A composite score was calculated by summing the number of “good” sleep health dimensions. We evaluated the associations between sleep health and glycemia and whether covariates, including age, T1D duration, and sleep apnea risk, influenced the relationships among the study variables using multivariable linear regression. Individual dimensions of sleep satisfaction (β = 0.380, p = .019; β = −0.414, p = .010), timing (β = 0.392, p = .015; β = −0.393, p = .015), and sleep efficiency (β = 0.428, p = .007) were associated with higher achievement of glycemic targets (J-index and time in range) however, these associations did not persist after considering covariates. A better sleep health composite score was associated with higher achievement of glycemic targets even after considering covariates. Using a multidimensional framework can guide future research on causal pathways between sleep and diabetes health, interventions to target sleep health profiles, and may improve sleep screening in routine diabetes care.
Keywords: sleep disturbance, glycemia, glucose regulation, glucoregulation, continuous glucose monitor
Introduction
Sleep health is a broader multidimensional concept characterized across several dimensions (satisfaction, alertness, timing, efficiency, duration; SATED) (Buysse, 2014). Multiple dimensions of sleep health, including satisfaction, alertness, timing, efficiency, and duration, directly or indirectly influence glucose metabolism (i.e., glucoregulation) (Leproult et al., 2014; Spiegel et al., 1999; Van Cauter et al., 2007). Laboratory studies of partial or total sleep deprivation (i.e., duration) and forced desynchrony (i.e., timing) perturb metabolic pathways with an increase in insulin resistance and decrease in glucose tolerance in adults without chronic conditions (eg, healthy) (Leproult et al., 2014; Leproult & Van Cauter, 2010; Spiegel et al., 1999; Van Cauter et al., 2007) and insulin sensitivity adults with T1D (Donga et al., 2010). Specifically alterations to these two prominent sleep health dimensions, duration and timing, lead to an imbalance between glucose production by the liver and glucose utilization by the insulin-dependent tissues (eg muscle and adipose) and non-insulin-dependent tissues (eg brain) (Spiegel et al., 1999; Spiegel et al., 2004; Van Cauter et al., 2007). Impairments in both sleep health and glucoregulation are independent predictors of all-cause mortality (Buysse, 2014; Sondrup et al., 2022).
Type 1 diabetes (T1D) is an autoimmune T cell-mediated condition characterized by destruction of beta cells, absolute insulin deficiency, and a lifelong dependency on exogenous sources (e.g., insulin and glucose) that requires regular self-monitoring to achieve glycemic targets (Burrack et al., 2017). Glycated hemoglobin (HbA1C) is the gold standard long term (chronic) glycemic target measurement for individuals with T1D. Suboptimal achievement (HbA1C <7%) is a major predictor of long-term vascular complications (Nathan & DCCT EDIC Research Group, 2014). Short term glycemic target achievement is often monitored day-to-day during wake and sleep through continuous glucose monitors (i.e., target time-in-range 70 mg −180 mg/dL >80% and glucose management indicator, <7%). Self-monitoring of glucose is a unique challenge for young adults with T1D as they transition away from their childhood homes and providers to adult care and responsibilities (Morrissey et al., 2021).
Promoting modifiable dimensions of sleep health including regularity, timing, efficiency, and duration improves health outcomes among adolescents and young adults without chronic conditions. However less is known about the impact of modifying sleep health dimensions on diabetes health outcomes in individuals with T1D (Beebe et al., 2008; Beebe et al., 2014; Beebe et al., 2015; Van Dyk et al., 2017). In prior research of adolescents or young adults, only one sleep health dimension at a time, predominantly sleep duration or sleep efficiency has been examined with little attention given to other domains (Farabi et al., 2017; Martyn-Nemeth et al., 2018; Patel et al., 2018; Perfect et al., 2012). A combination of sleep health dimensions are likely associated with better diabetes health outcomes. Therefore, there is a need to examine the unique and cumulative contributions of sleep health dimensions on glycemic target achievement in this at-risk population (e.g., multidimensional sleep health studies). The aim of this study was to examine the association between sleep health and glycemia. Each sleep health dimension and a composite measure were the independent variables of interest. We hypothesized that better sleep health (individual dimensions and composite) would be associated with better achievement of glycemic targets (less glucose variability and more time in range).
MATERIALS AND METHODS
Design
We followed the World Medical Association Declaration of Helsinki for research involving human subjects (World Medical Association, 2014) and received approval from the Case Western Reserve University Institutional Review Board (STUDY20201829). Participants were recruited over a 12-month period (February 2021 to April 2022) through social media, ResearchMatch, and newsletters (College Diabetes Network and a University in Cleveland Ohio). A national convenience sample of English-speaking young adults ages 18–25 years, with type 1 diabetes for at least 6-months, who did not work night shift, were not currently pregnant, or did not have a previous history of obstructive sleep apnea, severe mental illness (e.g., bipolar disorder, schizophrenia), or another major complex chronic condition were included.
Outcome
Glycemic target achievement (glucose management indicator, J index, coefficient of variation [CV], and time-in-range) was determined from the objective raw CGM data that were shared from each participant’s existing CGM to capture glucose patterns. J index was calculated as 0.001 × (mean + SD)2) and time in range was calculated as % time spent in target range 70–180 mg/dL. CGM systems provide real-time, dynamic glucose information every five minutes — up to 288 readings in a 24-hour period (Danne et al., 2017). CGMs are accurate across a wide range of test-retest reliability levels ranging from 0.77 – 0.95 (Danne et al., 2017). Glucose variability indices were calculated across the days of monitoring (Danne et al., 2017).
Sleep Health Characteristics and Dimensions
Obstructive sleep apnea risk was assessed with the validated Berlin questionnaire and categorized as low versus high risk (Netzer et al., 1999).
Sleep satisfaction
Sleep satisfaction was measured with the 8-item PROMIS Sleep Disturbance scale (Cronbach’s alpha = 0.95) (Yu et al., 2012). Each item is ranked using a 5-point Likert scale (not at all to very much) (Yu et al., 2012). Scores range from 8–40, with higher scores indicating greater severity of sleep disturbance (Yu et al., 2012). The raw scores on the 8 items are summed to obtain a total raw score. The raw scores are then transformed into a T-score. The Cronbach’s alpha for the PROMIS Sleep Disturbance in the current study was 0.875.
Alertness
Alertness was measured with the 8-item Epworth Sleepiness Scale (ESS) (Cronbach’s α = 0.88) (Johns, 1992). Scores range from 0–24, with higher scores indicating higher sleepiness (Johns, 1992). The scores on the 8 items are summed to obtain a total score. The Cronbach’s alpha for the Epworth Sleepiness Scale in the current study was 0.771.
Timing
Timing was measured with the 19-item Morningness Eveningness Questionnaire (MEQ) (Cronbach’s α = 0.88) (Horne & Ostberg, 1976). The scores on the 19-items are summed to obtain a total score. Scores range from 16–86; scores < 41 indicate evening or later types; scores > 59 indicate morning types, and scores 42–58 indicate intermediate types. The Cronbach’s alpha for the MEQ in the current study was 0.809.
Sleep efficiency
Sleep efficiency was derived from 3-items on the Pittsburgh Sleep Quality Index (PSQI) (sleep duration, getting up time, and bedtime) (Buysse et al., 1989). We calculated sleep efficiency as (sleep duration / time in bed) × 100 (Buysse et al., 1989).
Sleep duration
Sleep duration was measured with one item on the PSQI (“during the past month, how many hours of actual sleep did you get at night? This may be different than the number of hours you spend in bed”) (Buysse et al., 1989).
Derivation of the Sleep Health Composite:
Derivation of the Sleep Health Composite was based on several considerations. We decided to dichotomize each dimension as “good” or “poor.” This approach is more feasible and deployable in clinical practice and has been documented in prior studies (Dong et al., 2019). The Sleep Health Composite was coded as 1 = good and 0 = poor, with scores ranging from 0–5 and higher scores indicating better sleep health (Dong et al., 2019). We chose the cut-off point for each dimension based on the extant literature and data from previously published studies in normative samples (Dong et al., 2019; Ohayon et al., 2017). The cut points for the five dimensions were: satisfaction (PROMIS <56.36, i.e., < 1 SD above the mean), alertness (ESS ≤ 7.5), timing (MEQ 42–58), efficiency ≥ 85%, duration 7–9 hours.
Non-sleep Risk Factors
Participants completed a sociodemographic and clinical form and other study instruments about their diabetes management via an online survey. They shared either their raw continuous glucose monitor data or a share code from Dexcom Clarity so that the research team could access and export the raw glucose data.
Statistical analysis
Prior to the analysis, we screened data for missing or out-of-range values and examined distributions of continuous variables. Data were managed using the REDCap site and exported into the Statistical Package for the Social Sciences (SPSS) version 28 for analysis. CGM data were calculated with Glyculator v. 3.0 software (Czerwoniuk et al., 2011).
A quantitative descriptive approach was used to characterize sleep health dimensions and glycemia. Glucose variability indices were calculated based on data across the days of monitoring. Descriptive statistics were used to summarize each of the variables, including the scores for multi-item scales. Self-report A1c was used to determine chronic glycemia and CGM data were used to calculate short-term day-to-day glycemia (Suh & Kim, 2015). A series of t-tests and correlations were conducted to determine differences in sleep health dimensions and glucose variables by covariates, including age, sex, T1D duration, race, and BMI.
Bivariate and multivariable linear regression models were used to examine the relationships between sleep health dimensions and glycemia. A series of linear regression models were conducted for each individual sleep health dimension and for the Sleep Health Composite score to evaluate the explanatory contributions of sleep health dimensions to glycemia. Covariates were included in multivariable models if they were significantly associated with sleep health (composite measure or individual dimension) or glycemia (glucose management indicator, J-index, coefficient of variation, or time in range) (p < .05). We also controlled for sleep apnea risk due to a priori knowledge of an independent effect of sleep apnea on glycemia (Priou et al., 2012). Statistical significance was set at p <.05.
Results
Participant characteristics
Seventy-five participants (74.7% female sex assigned at birth, 9.3% gender minority) between the ages of 18–25 years (mean 21.47, SD = 2.06) participated in the study (64.9% on an insulin pump). Participants reported a mean T1D duration of 9.4 years (SD = 5.8 years) and mean HbA1c of 6.8 % (SD = 1.04%). Mean GMI was 7.01 (SD = 0.63%) and mean time in range was 70% (SD = 17.2%) from raw CGM data. We present demographic, clinical profile, and sleep characteristics in Table 1.
Table 1.
Demographic, clinical, and sleep profile
| Demographic profile | ||
|---|---|---|
| Mean or N | SD or (%) | |
| Age in years | 21.47 | 2.056 |
| BMI (kg/m2) | 24.55 | 4.65 |
| Non-Hispanic White (%) | 65 | 86.7 |
| Female (%) | 56 | 74.7 |
| Gender minority (%) | 7 | 9.3 |
| Clinical profile | ||
| T1D duration in years | 9.37 | 5.74 |
| HbA1c (%) | 6.83 | 1.04 |
| Glucose management indicator | 7.01 | 0.63 |
| Glucose, mean | 154.64 | 26.22 |
| J Index | 44.84 | 17.84 |
| Coefficient of Variation (CV) | 34.05 | 5.30 |
| Time in range (% 70–180 mg/dL) | 69.93 | 17.17 |
| Time above range (% > 180 mg/dL) | 27.93 | 16.98 |
| Time above range (% > 250 mg/dL) | 8.13 | 10.66 |
| Time below range (% <7 0 mg/dL) | 2.14 | 2.40 |
| Time below range (% < 54mg/dL) | 0.43 | 1.01 |
| Insulin pump (n) (%) | 48 | (64.9) |
| Sleep profile | ||
| Satisfaction1 | 48.69 | 7.70 |
| Alertness2 | 7.64 | 4.26 |
| Timing3 total | 48.33 | 9.24 |
| Efficiency4 | 86.13 | 13.01 |
| Duration (PSQI)4 | 7.25 | 1.29 |
| Sleep Health Composite | 3.04 | 1.26 |
Note:
PROMIS Sleep Disturbance;
ESS Epworth Sleepiness Scale;
MEQ, Morningess Eveningness Questionnaire;
PSQI, Pittsburgh Sleep Quality Index.
Individual sleep health dimensions
In the first set of models, we examined the associations between each individual sleep health dimension and glycemia as measured by the Glucose Management Indicator (GMI). Higher satisfaction and lower sleep efficiency were significantly associated with a lower GMI (better achievement of target), accounting for 18.6% and 14.9% of the variance, respectively. Satisfaction and efficiency had a medium effect on GMI (f 2 = 0.23 and f 2 = 0.18), and timing had a small effect on GMI (f 2 = 0.04). Alertness and duration did not have an effect on GMI. However, the associations were no longer significant after considering covariates (age, T1D duration, race, insulin mode of delivery, and sleep apnea risk) (Table 2).
Table 2.
Contribution of individual sleep health dimensions to glucose management indicator (linear regression models)
| Model 1 Unadjusted | Model 2 Adjusted for Covariates | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Predictors | B | SE | β | R2 | P value | F2 | B | SE | β | R2 | P value |
| Satisfaction PROMIS |
0.55 | .018 | .431 | .186 | .004 | 0.23 | .026 | .020 | .230 | .328 | .214 |
| Alertness | .007 | .025 | .042 | .002 | .792 | 0.00 | .038 | .022 | .267 | .356 | .096 |
| Timing | −.011 | .010 | −.184 | .034 | .244 | 0.04 | .010 | .010 | .172 | .316 | .314 |
| Efficiency | -.020 | .008 | -.385 | .149 | .014 | 0.18 | −.005 | .009 | −.101 | .319 | .536 |
| Duration | .007 | .103 | .011 | .001 | .945 | 0.00 | −.049 | .100 | −.078 | .315 | .630 |
Note: Model 1 is unadjusted. Model 2 is adjusted for covariates: age, type 1 diabetes duration, race, mode of insulin delivery, and sleep apnea risk. B is the unstandardized coefficient regression coefficient. SE standard error. β is the standardized regression coefficient. R2 = coefficient of determination shown for each model.
In the second set of models, we examined the associations between each individual sleep health dimension and glycemia as measured by J-index. In these models, lower satisfaction, later timing, and lower sleep efficiency were significantly associated with lower achievement of glycemic targets as measured by J-index accounting for 14.5%, 15.3%, and 10.3% respectively; however, these relationships were no longer significant when adjusting for covariates (age, T1D duration, insulin mode of delivery, race, and sleep apnea risk) (Table 3). Satisfaction and timing had a medium effect on J-index (f 2 = 0.17 and f 2 = 0.18) and efficiency had a small effect on J-index f 2 = 0.11). In the next set of models (Table 4) we examined the associations between individual sleep health dimensions and glycemia as measured by Coefficient of Variation and none of the associations were statistically significant.
Table 3.
Contribution of individual sleep health dimensions to glucose variability J-index (linear regression models)
| Model 1 Unadjusted | Model 2 Adjusted for Covariates | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Predictors | B | SE | β | R2 | P value | F2 | B | SE | β | R2 | P value |
| Satisfaction PROMIS |
1.085 | .440 | .380 | .145 | .019 | 0.17 | .369 | .468 | .138 | .343 | .437 |
| Alertness | .179 | .737 | .041 | .002 | .809 | 0.00 | .968 | .623 | .237 | .380 | .131 |
| Timing | 11.538 | 4.517 | .392 | .153 | .015 | 0.18 | .231 | .271 | .140 | .345 | .401 |
| Efficiency | -.433 | .213 | -.320 | .103 | .050 | 0.11 | −.120 | .248 | −.077 | .345 | .633 |
| Duration | .561 | 2.917 | .032 | .001 | .849 | 0.00 | −1.312 | 2.873 | −.072 | .345 | .651 |
Note: Model 1 is unadjusted. Model 2 is adjusted for covariates: age, type 1 diabetes duration, race, mode of insulin delivery, and sleep apnea risk. B is the unstandardized coefficient regression coefficient. SE standard error. β is the standardized regression coefficient. R2 = coefficient of determination shown for each model. F2 = Cohen’s f2 effect size.
Table 4.
Contribution of individual sleep health dimensions to glucose variability – Coefficient of Variation (linear regression models)
| Model 1 Unadjusted | Model 2 Adjusted for Covariates | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Predictors | B | SE | β | R2 | P value | F2 | B | SE | β | R2 | P value |
| Satisfaction PROMIS |
.089 | .136 | .104 | .011 | .514 | 0.01 | −.010 | .138 | −.013 | .310 | .945 |
| Alertness | .183 | .210 | .136 | .019 | .389 | 0.01 | .098 | .188 | .083 | .316 | .608 |
| Timing | −.078 | .082 | −.148 | .022 | .350 | 0.02 | −.034 | .080 | −.072 | .213 | .670 |
| Efficiency | .069 | .066 | .167 | .028 | .302 | 0.03 | .008 | .074 | .018 | .308 | .912 |
| Duration | .904 | .786 | .183 | .034 | .257 | 0.04 | 5.00 | 4.44 | .178 | .308 | .270 |
Note: Model 1 is unadjusted. Model 2 is adjusted for covariates: age, type 1 diabetes duration, race, mode of insulin delivery, and sleep apnea risk. B is the unstandardized coefficient regression coefficient. SE standard error. β is the standardized regression coefficient. R2 = coefficient of determination shown for each model. F2 = Cohen’s f2 effect size.
In the next set of models, we examined the associations between each individual sleep health dimension and achievement of glycemic targets as defined by % time in glucose range (70–180 mg/dL). Higher satisfaction, earlier timing, and higher sleep efficiency were significantly associated with more time in range (better target achievement), accounting for 17.1%, 15.4% and 18.3% of the variance, respectively. Satisfaction, timing, and efficiency had a medium effect on time in range (f 2 = .021, f 2 = 0.18, and f 2 = 0.22 respectively). However, the associations were no longer significant after considering covariates (age, T1D duration, insulin mode of delivery, race, and sleep apnea risk) (Table 5).
Table 5.
Contribution of individual sleep health dimensions to time in range (linear regression models)
| Model 1 Unadjusted | Model 2 Adjusted for Covariates | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Model 1 Unadjusted | Model 2 Adjusted for Covariates | ||||||||||
| Predictors | B | SE | β | R2 | P value | F2 | B | SE | β | R2 | P value |
| Satisfaction PROMIS |
-1.137 | .417 | -.414 | .171 | .010 | 0.21 | −.547 | .414 | −.235 | .327 | .196 |
| Alertness | −.071 | .709 | −.017 | .000 | .920 | 0.00 | −.950 | .557 | −.266 | .351 | .099 |
| Timing | -11.127 | 4.343 | -.393 | .154 | .015 | 0.18 | −.160 | .245 | −.111 | .298 | .518 |
| Efficiency | .612 | .216 | .428 | .183 | .007 | 0.22 | .207 | .222 | .151 | .317 | .359 |
| Duration | .047 | 2.807 | .003 | .000 | .987 | 0.00 | 1.63 | 2.59 | .101 | .306 | .535 |
Note: Model 1 is unadjusted. Model 2 is adjusted for covariates: age, type 1 diabetes duration, race, mode of insulin delivery, and sleep apnea risk. B is the unstandardized coefficient regression coefficient. SE standard error. β is the standardized regression coefficient. R2 = coefficient of determination shown for each model. F2 = Cohen’s f2 effect size.
Sleep Health Composite Score
In the last set of models, we examined the associations between the Sleep Health Composite and all glycemia indices (GMI, J index, CV, and time in range) (Table 6). Better sleep health was significantly associated with better achievement of glycemic targets even after considering covariates (age, T1D duration, insulin mode of delivery, race, and sleep apnea risk) across all glycemic measures GMI (β = −.337, p = .031), J index (β = −.308, p = .045) and time in range (β = .434, p = .013) except for CV (p = .793) accounting for 41.3%, 42.7% and 43.4% of the variance respectively. Also, the sleep health composite had a medium effect on all glycemic metrices (GMI f 2 = 0.21, J-index, f 2 = 0.20, and f 2 = 0.24 respectively) except for CV (Table 6). We present the scatterplots of sleep satisfaction, timing, and sleep efficiency and percentage (%) time in range (70–180 mg/dL) in Figure 1.
Table 6.
Contribution of sleep health dimension composite score to glycemia (glucose management indicator, coefficient of variation, and time in range) (linear regression models)
| Model 1 Unadjusted | Model 2 Adjusted for Covariates | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Outcomes | B | SE | β | R2 | P value | F2 | B | SE | β | R2 | P value |
| Glucose Management indicator | -.247 | .088 | -.414 | .171 | .008 | 0.21 | -.175 | .077 | -.337 | .413 | .031 |
| J index | -6.634 | 2.432 | -.405 | .164 | .010 | 0.20 | -4.705 | 2.25 | -.308 | .427 | .045 |
| CV | −.405 | .747 | −.088 | .008 | .591 | 0.01 | −.188 | .712 | −.043 | .309 | .793 |
| Time in range | 6.937 | 2.304 | .439 | .193 | .005 | 0.24 | 5.194 | 1.96 | .387 | .434 | .013 |
Note: Model 1 is unadjusted. Model 2 is adjusted for covariates: age, type 1 diabetes duration, race, mode of insulin delivery, and sleep apnea risk. B is the unstandardized coefficient regression coefficient. SE standard error. β is the standardized regression coefficient. R2 = coefficient of determination shown for each model. F2 = Cohen’s f2 effect size.
Figure 1.
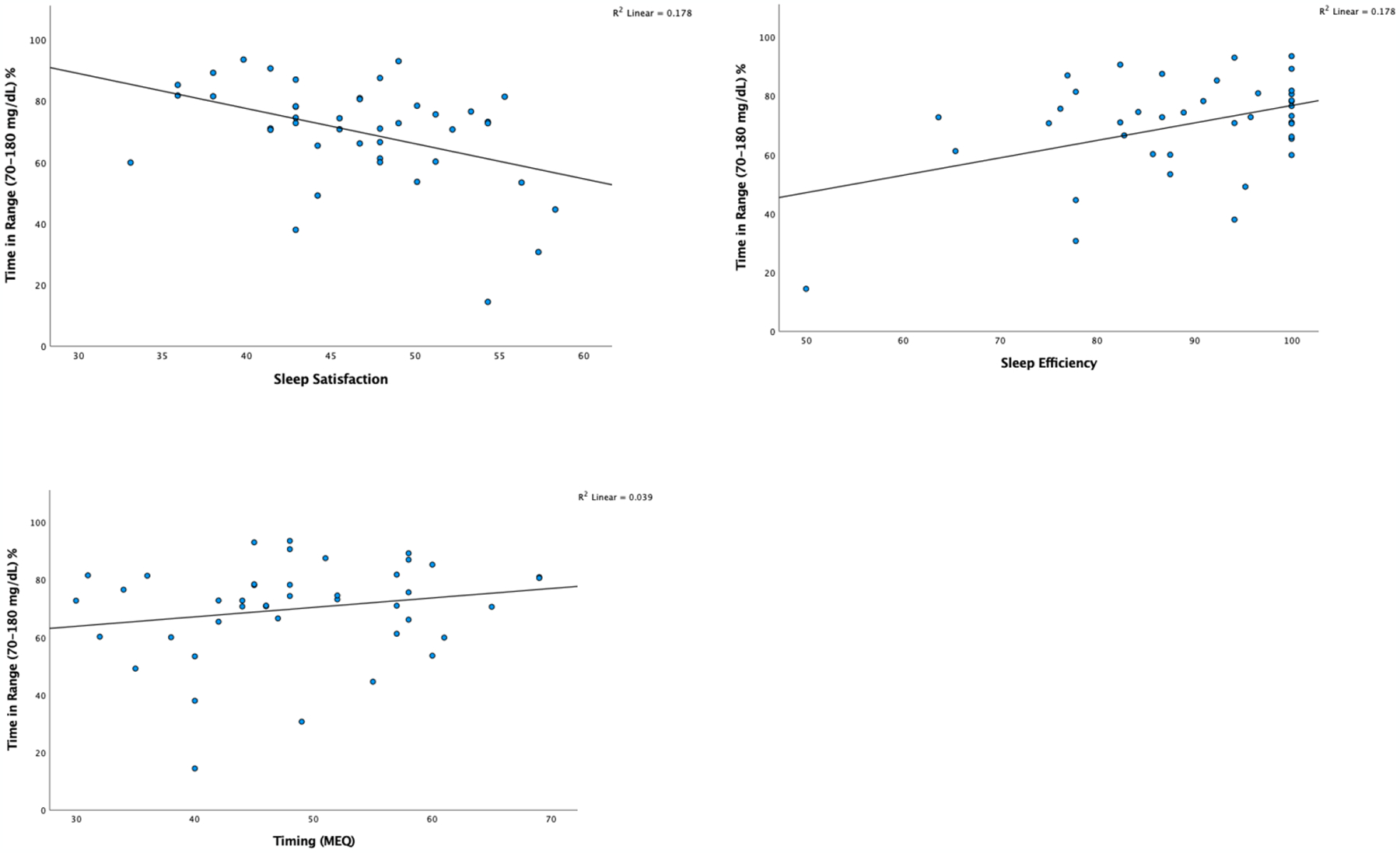
Scatterplots of Sleep Satisfaction, Timing, Sleep Efficiency (%) and Time in Range 70–180 mg/dL %
Discussion
In a sample of community-dwelling young adults with type 1 diabetes, we found that better sleep health (individual sleep health dimensions and a composite measure of sleep health dimensions) was associated with higher achievement of glycemic targets. Specifically, a better sleep health composite score was associated with higher achievement of glycemic targets as measured by continuous glucose monitoring over 7 days even after considering covariates. Only three individual sleep health dimensions, including satisfaction, timing, and efficiency, were significantly associated with achieving glycemic targets in the unadjusted models; however, these were not statistically significant after controlling for covariates. Most of the individual sleep health dimensions had a small or medium effect on each of the glucose variability indices. The composite had a medium effect size on all glucose variability indices except for coefficient of variation. To our knowledge, this is the first study where a composite measure of sleep health dimensions and glycemic targets were investigated in a sample of young adults with T1D. Although in previous studies, poorer glycemia was associated with individual dimensions of sleep health such as short duration and variability in sleep duration (Borel et al., 2009; Chontong et al., 2016; Griggs, Grey, et al., 2021; Griggs et al., 2020; Larcher et al., 2016; Patel et al., 2018), we suggest based on our findings that these sleep health dimensions have positive additive effects. Examining multidimensional sleep health profiles may advance our understanding of the relationships between sleep and glycemia in young adults with T1D and in other populations with and at risk for cardiometabolic disease.
A better sleep health composite score was associated with better glycemic target achievement across most glycemia measures supporting our hypothesis. Other studies where the association between a sleep health composite and glycemia were not available for comparison. Associations between individual sleep health dimensions and glycemia have varied across other comparison studies (Borel et al., 2009; Chontong et al., 2016; Griggs et al., 2022; Griggs, Grey, et al., 2021; Griggs, Hickman, et al., 2021; Griggs et al., 2020; Griggs, Strohl, et al., 2021; Perfect et al., 2012; Rechenberg et al., 2020; Reutrakul et al., 2016). In these studies poorer individual dimensions of sleep health (e.g., lower satisfaction, lower daytime alertness, variability in timing, lower efficiency, and or shorter duration) were associated with lower achievement of glycemic targets (Borel et al., 2009; Chontong et al., 2016; Griggs et al., 2022; Griggs, Grey, et al., 2021; Griggs, Hickman, et al., 2021; Griggs et al., 2020; Griggs, Strohl, et al., 2021; Perfect et al., 2012; Rechenberg et al., 2020; Reutrakul et al., 2016).
In the current study, poor sleep satisfaction (specifically sleep disturbance) was significantly associated with lower achievement of glycemic targets. This finding is consistent with several cross-sectional studies of small clinical samples and epidemiological studies (Matejko et al., 2015; Perfect et al., 2012; Reutrakul et al., 2016; von Schnurbein et al., 2018). In other studies of adolescents with T1D, sleep satisfaction was not associated with glycemia (Patel et al., 2018; Perfect et al., 2012). However, measures of sleep satisfaction differ among these studies. Satisfaction in the current study was measured with the PROMIS sleep disturbance scale, which measures perceptions of sleep quality, sleep depth, and restoration associated with sleep over the past 7-days. The Pittsburgh Sleep Quality Index global score or an individual sleep quality item was used in the other studies (Patel et al., 2018; Reutrakul et al., 2016).
Of the individual sleep health dimensions, sleep timing had the strongest association with glycemia. This finding is consistent with some previous studies (Griggs, Grey, et al., 2021; Reutrakul et al., 2016), but not others (Perfect et al., 2012; Siwasaranond et al., 2016). Consistent with our findings in the current study, the association between better or more stable timing and lower glycemia in individuals with T1D has been highlighted in numerous studies (Chontong et al., 2016; Griggs et al., 2022; Griggs, Grey, et al., 2021; Griggs, Hickman, et al., 2021; Griggs et al., 2020; Griggs, Strohl, et al., 2021; Patel et al., 2018; Perfect et al., 2012; Rechenberg et al., 2020; Reutrakul et al., 2016; Reutrakul & Van Cauter, 2014). Beyond cross sectional studies, in a recent clinical trial, the chronic sleep restriction with recurrent circadian disruption condition had significantly elevated postprandial plasma glucose levels whereas the condition with chronic sleep restriction and minimized circadian disruption had no adverse glycemic effects after 3 weeks of exposure among adults without chronic conditions (Yuan et al., 2021).
Neither sleep duration nor daytime alertness as individual dimensions were significantly associated with any of the glycemic target measures in the current study. This finding was not expected given the evidence of impaired glucose metabolism in adults without chronic conditions undergoing experimental sleep deprivation (Donga et al., 2010; Knutson & Van Cauter, 2008; Spiegel et al., 1999; Van Cauter et al., 2007). In previous comparison studies of adults with T1D, this association has been mixed with some researchers reporting an association between a shorter sleep duration and poorer glycemia (Borel et al., 2013; Denic-Roberts et al., 2016; Reutrakul et al., 2016; von Schnurbein et al., 2018) or between a lower daytime alertness and poorer glycemia (Griggs, Hickman, et al., 2021; Perfect et al., 2012; Zhu et al., 2021). In other studies, the associations between either sleep duration or daytime alertness and glycemia have not been significant (Griggs, Grey, et al., 2021; Griggs, Hickman, et al., 2021; Griggs et al., 2020; Patel et al., 2018; Rechenberg et al., 2020).
The current cross-sectional study and analyses have several limitations to consider when interpreting the results. First, the sample was not representative of community dwelling young adults with T1D as a majority were Non-Hispanic White (87%) and female (75%). Therefore, results cannot be generalized to the general T1D population, to other age groups (adolescents, middle or older adults), or to clinical populations with sleep disorders. However, the current sample did achieve glycemic targets at comparable rates than the T1D Exchange, a national comparison sample (mean GMI 7.0% vs. 7.3%) (Foster et al., 2019). Also, neither sex- nor race-based differences could be determined. Despite these limitations, the current study design is an important first step to gauge the utility of a sleep health composite score in young adults with T1D. Larger population-based studies are needed to evaluate the psychometric properties of a sleep health composite score in young adults with T1D as well as other clinical and non-clinical samples. Second, though we constructed items in equal weights based on clinically significant setpoints in the extant literature, different dimensions may warrant different weights especially in this unique sample of young adults with T1D. Third, while the MEQ measures self-reported timing preference, it is likely distinct from sleep variability. Although they both assess timing of sleep, the impact of a later chronotype (especially consistent late sleep timing) and sleep variability (where sleep timing varies from day to day) on achievement of glycemic targets requires further investigation. Those with a later chronotype may experience higher sleep variability consistently. In the future, researchers should incorporate objective measures of sleep health dimensions and glycemia such as concurrent actigraphy and continuous glucose monitors as well as lab drawn, or chart extracted A1c. Nevertheless, we used well validated and reliable scales to determine sleep health, and this approach is more feasible and deployable in clinical settings.
There is a paucity of studies focused on determining whether modifying sleep has an effect on glycemia in young adults with T1D. Promoting sleep duration had a positive impact on glucose targets in two pilot studies of adolescents with T1D in the short-term (3 months) (Jaser et al., 2020; Perfect et al., 2016; Perfect et al., 2018). Specifically, in a randomized controlled trial, Perfect and colleagues established that a behavioral intervention aimed at increasing sleep duration by 30 minutes per day led to an improvement in time in range after 3 months (Perfect, 2018). In a small experimental hyper insulinemic euglycemic clamp study of 7 middle-aged adults with T1D (mean age 44, SD 7 years), sleep restriction reduced the glucose disposal rate reflecting decreased peripheral sensitivity (Donga et al., 2010). It is unknown if extending sleep duration over time is sustainable, nor what the long-term impact is on clinical outcomes specifically in young adults with T1D who have unique developmental and social needs.
The current study provides support of the utility of a sleep health composite score. The results can help to establish validity in the context of chronic illness and may translate to other related populations such as those with type 2 diabetes, hypertension, or obesity. Future researchers may consider a similar approach in other populations and contexts. Linear relationships were examined in the current study, and in the future, it may be warranted to examine both linear and nonlinear associations between sleep health dimensions and glycemia. The current study was cross sectional therefore the mechanistic pathways between sleep health and glucose variability could not be determined. Future longitudinal and experimental studies can provide further insight into the findings presented here and whether poor sleep precedes greater glucose variability, or vice versa, or acts bidirectionally. In addition, age effects on multidimensional sleep health and whether relationships are consistent in broader age ranges, such as adolescents and middle to older aged adults with T1D should be examined. Promoting healthy sleep through a multidimensional framework addresses the entire diabetes population rather than only those with sleep disorders.
Acknowledgments and declaration of author contributions:
SG, PI on the grant (R00NR018886), secured the funding, designed the study, collected, analyzed, and interpreted the data and wrote the manuscript. GP interpreted the data and cowrote the manuscript. Senior author RLH contributed to the study design, interpreted the findings, and co-wrote the manuscript. All authors have seen and approved the final version of this manuscript.
Funding:
SG is funded by the National Institute of Nursing Research (NINR) R00NR018886. GP is funded by the National Center for Advancing Translational Sciences (NCATS) KL2TR002547. Its contents are solely the responsibility of the authors and do not necessarily represent the official view of NIH.
Footnotes
Conflict of interest: The authors have no conflicts of interest to disclose.
Contributor Information
Stephanie Griggs, Case Western Reserve University, Frances Payne Bolton School of Nursing, Cleveland, OH 44106.
Grant Pignatiello, Case Western Reserve University, Frances Payne Bolton School of Nursing.
Ronald L. Hickman, Jr, Case Western Reserve University, Frances Payne Bolton School of Nursing, Cleveland, OH 44106,.
References
- Beebe DW, Fallone G, Godiwala N, Flanigan M, Martin D, Schaffner L, & Amin R (2008). Feasibility and behavioral effects of an at-home multi-night sleep restriction protocol for adolescents. 49(9), 915–923. https://doi.org/ 10.1111/j.1469-7610.2008.01885.x [DOI] [PubMed] [Google Scholar]
- Beebe DW, Rausch J, Zhou A, Noe O, & Simon S (2014). Early bedtimes reduce 24-hr intake for adolescents with early chronotypes, but not those with late chronotypes. 1), A76. [Google Scholar]
- Beebe DW, Whitacre C, Catlin P, & McAlister S (2015). Feasibility of a 5-week, randomized cross-over adolescent sleep extension trial during the schoolyear 1), A123. [Google Scholar]
- Borel AL, Benhamou PY, Baguet JP, Debaty I, Levy P, Pepin JL, & Mallion JM (2009). Short sleep duration is associated with a blood pressure nondipping pattern in type 1 diabetes: the DIAPASOM study. Diabetes Care, 32(9), 1713–1715. 10.2337/dc09-0422 [doi] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borel AL, Pepin JL, Nasse L, Baguet JP, Netter S, & Benhamou PY (2013). Short sleep duration measured by wrist actimetry is associated with deteriorated glycemic control in type 1 diabetes. Diabetes Care, 36(10), 2902–2908. 10.2337/dc12-2038 [doi] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burrack AL, Martinov T, & Fife BT (2017). T Cell-Mediated Beta Cell Destruction: Autoimmunity and Alloimmunity in the Context of Type 1 Diabetes. Front Endocrinol (Lausanne), 8, 343. 10.3389/fendo.2017.00343 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buysse DJ (2014). Sleep health: can we define it? Does it matter? Sleep, 37(1), 9–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buysse DJ, Reynolds CF 3rd, Monk TH, Berman SR, & Kupfer DJ (1989). The Pittsburgh Sleep Quality Index: a new instrument for psychiatric practice and research. Psychiatry Research, 28(2), 193–213. https://doi.org/0165-1781(89)90047-4 [pii] [DOI] [PubMed] [Google Scholar]
- Chontong S, Saetung S, & Reutrakul S (2016). Higher sleep variability is associated with poorer glycaemic control in patients with type 1 diabetes. Journal of Sleep Research, 25(4), 438–444. 10.1111/jsr.12393 [DOI] [PubMed] [Google Scholar]
- Czerwoniuk D, Fendler W, Walenciak L, & Mlynarski W (2011). GlyCulator: a glycemic variability calculation tool for continuous glucose monitoring data. Journal of Diabetes Science and Technology, 5(2), 447–451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Danne T, Nimri R, Battelino T, Bergenstal RM, Close KL, DeVries JH, … Phillip M (2017). International Consensus on Use of Continuous Glucose Monitoring. Diabetes Care, 40(12), 1631–1640. 10.2337/dc17-1600 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Denic-Roberts H, Costacou T, & Orchard TJ (2016). Subjective sleep disturbances and glycemic control in adults with long-standing type 1 diabetes: The Pittsburgh’s Epidemiology of Diabetes Complications study. Diabetes Research and Clinical Practice, 119, 1–12. 10.1016/j.diabres.2016.06.013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dong L, Martinez AJ, Buysse DJ, & Harvey AG (2019). A composite measure of sleep health predicts concurrent mental and physical health outcomes in adolescents prone to eveningness. Sleep Health, 5(2), 166–174. 10.1016/j.sleh.2018.11.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Donga E, van Dijk M, van Dijk JG, Biermasz NR, Lammers GJ, van Kralingen K, … Romijn JA (2010). Partial sleep restriction decreases insulin sensitivity in type 1 diabetes. Diabetes Care, 33(7), 1573–1577. 10.2337/dc09-2317 [doi] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Farabi SS, Carley DW, & Quinn L (2017). Glucose variations and activity are strongly coupled in sleep and wake in young adults with type 1 diabetes. Biological Research for Nursing, 19(3), 249–257. 10.1177/1099800416685177 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Foster NC, Beck RW, Miller KM, Clements MA, Rickels MR, DiMeglio LA, … Smith E (2019). State of type 1 diabetes management and outcomes from the T1D Exchange in 2016–2018. Diabetes Technology & Therapeutics, 21(2), 66–72. 10.1089/dia.2018.0384 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Griggs S, Barbato E, Hernandez E, Gupta D, Margevicius S, Grey M, & Hickman RL (2022). Glucose and unstructured physical activity coupling during sleep and wake in young adults with type 1 diabetes. Sci Rep, 12(1), 5790. 10.1038/s41598-022-09728-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Griggs S, Grey M, Strohl KP, Crawford SL, Margevicius S, Kashyap SR, … Hickman RL (2021). Variations in Sleep Characteristics and Glucose Regulation in Young Adults with Type 1 Diabetes. J Clin Endocrinol Metab. 10.1210/clinem/dgab771 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Griggs S, Hickman RL, Strohl KP, Redeker NS, Crawford SL, & Grey M (2021). Sleep-wake characteristics, daytime sleepiness, and glycemia in young adults with type 1 diabetes. J Clin Sleep Med. 10.5664/jcsm.9402 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Griggs S, Redeker NS, Jeon S, & Grey M (2020). Daily variations in sleep and glucose in adolescents with type 1 diabetes. Pediatric Diabetes. 10.1111/pedi.13117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Griggs S, Strohl KP, Grey M, Barbato E, Margevicius S, & Hickman RL (2021). Circadian characteristics of the rest-activity rhythm, executive function, and glucose fluctuations in young adults with type 1 diabetes. Chronobiol Int, 1–11. 10.1080/07420528.2021.1932987 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horne JA, & Ostberg O (1976). A self-assessment questionnaire to determine morningness-eveningness in human circadian rhythms. International Journal of Chronobiology, 4(2), 97–110. [PubMed] [Google Scholar]
- Jaser SS, Hamburger ER, Bergner EM, Williams R, Slaughter JC, Simmons JH, & Malow BA (2020). Sleep coach intervention for teens with type 1 diabetes: Randomized pilot study. Pediatr Diabetes, 21(3), 473–478. 10.1111/pedi.12991 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johns MW (1992). Reliability and factor analysis of the Epworth Sleepiness Scale. Sleep, 15(4), 376–381. 10.1093/sleep/15.4.376 [DOI] [PubMed] [Google Scholar]
- Knutson KL, & Van Cauter E (2008). Associations between sleep loss and increased risk of obesity and diabetes. Annals of the New York Academy of Sciences, 1129(1), 287–304. 10.1196/annals.1417.033 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Larcher S, Gauchez AS, Lablanche S, Pepin JL, Benhamou PY, & Borel AL (2016). Impact of sleep behavior on glycemic control in type 1 diabetes: the role of social jetlag. European Journal of Endocrinology, 175(5), 411–419. 10.1530/EJE-16-0188 [doi] [DOI] [PubMed] [Google Scholar]
- Leproult R, Holmbäck U, & Van Cauter E (2014). Circadian misalignment augments markers of insulin resistance and inflammation, independently of sleep loss. Diabetes, 63(6), 1860–1869. 10.2337/db13-1546 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leproult R, & Van Cauter E (2010). Role of sleep and sleep loss in hormonal release and metabolism. Endocrine Development, 17, 11–21. 10.1159/000262524 [doi] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martyn-Nemeth P, Phillips SA, Mihailescu D, Farabi SS, Park C, Lipton R, … Quinn L (2018). Poor sleep quality is associated with nocturnal glycaemic variability and fear of hypoglycaemia in adults with type 1 diabetes. Journal of Advanced Nursing, 74(10), 2373–2380. 10.1111/jan.13765 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matejko B, Skupien J, Mrozińska S, Grzanka M, Cyganek K, Kiec-Wilk B, … Klupa T (2015). Factors associated with glycemic control in adult type 1 diabetes patients treated with insulin pump therapy. Endocrine, 48(1), 164–169. 10.1007/s12020-014-0274-2 [DOI] [PubMed] [Google Scholar]
- Morrissey EC, Casey B, Hynes L, Dinneen SF, & Byrne M (2021). Supporting self-management and clinic attendance in young adults with type 1 diabetes: development of the D1 Now intervention. Pilot and feasibility studies, 7(1), 1–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nathan DM, & DCCT EDIC Research Group. (2014). The diabetes control and complications trial/epidemiology of diabetes interventions and complications study at 30 years: overview. Diabetes Care, 37(1), 9–16. 10.2337/dc13-2112 [doi] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Netzer NC, Stoohs RA, Netzer CM, Clark K, & Strohl KP (1999). Using the Berlin Questionnaire to identify patients at risk for the sleep apnea syndrome. Annals of Internal Medicine, 131(7), 485–491. 10.7326/0003-4819-131-7-199910050-00002 [DOI] [PubMed] [Google Scholar]
- Ohayon M, Wickwire EM, Hirshkowitz M, Albert SM, Avidan A, Daly FJ, … Gozal D (2017). National Sleep Foundation’s sleep quality recommendations: first report. Sleep Health, 3(1), 6–19. [DOI] [PubMed] [Google Scholar]
- Patel NJ, Savin KL, Kahanda SN, Malow BA, Williams LA, Lochbihler G, & Jaser SS (2018). Sleep habits in adolescents with type 1 diabetes: Variability in sleep duration linked with glycemic control. Pediatric Diabetes, 19(6), 1100–1106. 10.1111/pedi.12689 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perfect MM, Beebe DW, Levine-Donnerstein D, Frye SS, Bluez GP, & Quan SF (2016). The development of a clinically relevant sleep modification protocol for youth with Type 1 diabetes. Clinical Practice in Pediatric Psychology, 4(2), 227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perfect MM, Frye S, & Bluez GP (2018). The Effects of a Sleep Extension Intervention on Glucose Control in Youth with Type 1 Diabetes. Diabetes, 67(Supplement 1). [Google Scholar]
- Perfect MM, Patel PG, Scott RE, Wheeler MD, Patel C, Griffin K, … Quan SF (2012). Sleep, glucose, and daytime functioning in youth with type 1 diabetes. Sleep, 35(1), 81–88. 10.5665/sleep.1590 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Priou P, Le Vaillant M, Meslier N, Chollet S, Masson P, Humeau MP, … Group, I. S. C. (2012). Independent association between obstructive sleep apnea severity and glycated hemoglobin in adults without diabetes. Diabetes Care, 35(9), 1902–1906. 10.2337/dc11-2538 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rechenberg K, Griggs S, Jeon S, Redeker N, Yaggi HK, & Grey M (2020). Sleep and glycemia in youth with type 1 diabetes. Journal of Pediatric Health Care. 10.1016/j.pedhc.2019.12.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reutrakul S, Thakkinstian A, Anothaisintawee T, Chontong S, Borel A-L, Perfect MM, … Knutson KL (2016). Sleep characteristics in type 1 diabetes and associations with glycemic control: systematic review and meta-analysis. Sleep Medicine, 23, 26–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reutrakul S, & Van Cauter E (2014). Interactions between sleep, circadian function, and glucose metabolism: implications for risk and severity of diabetes. Annals of the New York Academy of Sciences, 1311(1), 151–173. [DOI] [PubMed] [Google Scholar]
- Siwasaranond N, Nimitphong H, Saetung S, Chirakalwasan N, Ongphiphadhanakul B, & Reutrakul S (2016). Shorter sleep duration is associated with poorer glycemic control in type 2 diabetes patients with untreated sleep-disordered breathing. Sleep Breath, 20(2), 569–574. 10.1007/s11325-015-1243-6 [DOI] [PubMed] [Google Scholar]
- Sondrup N, Termannsen A-D, Eriksen JN, Hjorth MF, Færch K, Klingenberg L, & Quist JS (2022). Effects of sleep manipulation on markers of insulin sensitivity: A systematic review and meta-analysis of randomized controlled trials. Sleep Med Rev, 101594. [DOI] [PubMed] [Google Scholar]
- Spiegel K, Leproult R, & Van Cauter E (1999). Impact of sleep debt on metabolic and endocrine function. The Lancet, 354(9188), 1435–1439. 10.1016/S0140-6736(99)01376-8 [DOI] [PubMed] [Google Scholar]
- Spiegel K, Tasali E, Penev P, & Van Cauter E (2004). Brief communication: sleep curtailment in healthy young men is associated with decreased leptin levels, elevated ghrelin levels, and increased hunger and appetite. Annals of Internal Medicine, 141(11), 846–850. 10.7326/0003-4819-141-11-200412070-00008 [DOI] [PubMed] [Google Scholar]
- Suh S, & Kim JH (2015). Glycemic variability: how do we measure it and why is it important? Diabetes & metabolism journal, 39(4), 273–282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Cauter E, Holmback U, Knutson K, Leproult R, Miller A, Nedeltcheva A, … Spiegel K (2007). Impact of sleep and sleep loss on neuroendocrine and metabolic function. Hormone Research in Pediatrics, 67 Suppl 1, 2–9. 10.1159/000097543 [DOI] [PubMed] [Google Scholar]
- Van Dyk TR, Zhang N, Catlin PA, Cornist K, McAlister S, Whitacre C, & Beebe DW (2017). Feasibility and Emotional Impact of Experimentally Extending Sleep in Short-Sleeping Adolescents. 40(9), 01. https://doi.org/ 10.1093/sleep/zsx123 [DOI] [PubMed] [Google Scholar]
- von Schnurbein J, Boettcher C, Brandt S, Karges B, Dunstheimer D, Galler A, … Vetter C (2018). Sleep and glycemic control in adolescents with type 1 diabetes. Pediatric Diabetes, 19(1), 143–149. 10.1111/pedi.12538 [DOI] [PubMed] [Google Scholar]
- Yu L, Buysse DJ, Germain A, Moul DE, Stover A, Dodds NE, … Pilkonis PA (2012). Development of Short Forms From the PROMIS™ Sleep Disturbance and Sleep-Related Impairment Item Banks. Behavioral Sleep Medicine, 10(1), 6–24. 10.1080/15402002.2012.636266 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yuan RK, Zitting KM, Duffy JF, Vujovic N, Wang W, Quan SF, … Czeisler CA (2021). Chronic Sleep Restriction While Minimizing Circadian Disruption Does Not Adversely Affect Glucose Tolerance. Front Physiol, 12, 764737. 10.3389/fphys.2021.764737 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu B, Abu Irsheed GM, Martyn-Nemeth P, & Reutrakul S (2021). Type 1 Diabetes, Sleep, and Hypoglycemia. Curr Diab Rep, 21(12), 55. 10.1007/s11892-021-01424-1 [DOI] [PubMed] [Google Scholar]


