Abstract
A hemolytic toxin related to thermostable direct hemolysin (TDH), TDH-related hemolysin (TRH), produced by Kanagawa-phenomenon-negative Vibrio parahaemolyticus is suspected of playing an important, but yet-to-be-elucidated role in diarrhea caused by this organism. In cultured human colonic epithelial cells, TRH increases Cl− secretion, followed by elevation of intracellular calcium.
Vibrio parahaemolyticus is an important cause of gastroenteritis, with outbreaks now occurring all over the world (2, 17). However, the mechanisms underlying the diarrheic action of this pathogen are not completely understood. V. parahaemolyticus can be categorized according to the presence or absence of the Kanagawa phenomenon (KP), in which there is hemolysis due to thermostable direct hemolysin (TDH) produced by the organism on Wagatsuma agar medium (14). TDH has been considered a major virulence factor for this disease (8, 23); however, some cases of V. parahaemolyticus gastroenteritis are due to KP-negative strains (10, 15, 16). From clinically isolated KP-negative V. parahaemolyticus, we identified a new hemolysin, named TDH-related hemolysin (TRH), and demonstrated that this TRH was also an important virulence factor (10, 24). For example, TRH stimulated fluid secretion in the rabbit loop test (8, 10). Thus, there is a possibility that TRH induces diarrhea. TRH is immunologically related, but not identical, to TDH ordinary human virulence factor. Unlike that of TDH, the activity of TRH is labile to heat treatment at 60°C for 10 min (10). Although we recently demonstrated that TDH induced Cl− secretion in human colonic epithelial cells (22), there is no evidence how TRH induces fluid secretion.
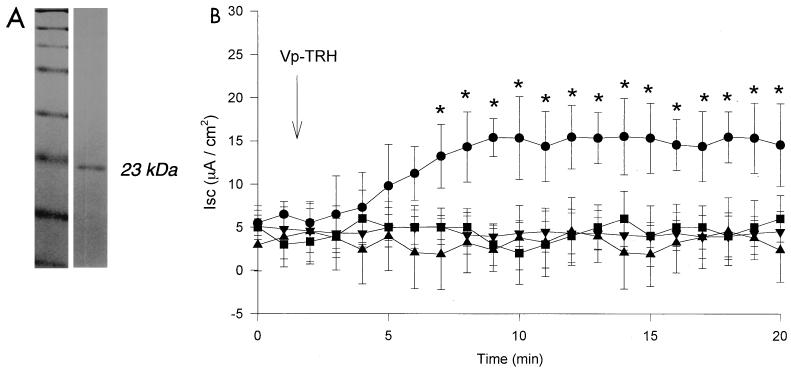
The present study was designed to investigate how TRH affects short-circuit current (Isc) in human colonic epithelial cell (Caco-2 cell) monolayers and the intracellular Ca2+ concentration ([Ca2+]in) in Caco-2 cells after exposure to TRH. The measurements of Isc and [Ca2+]in in Caco-2 cells were taken by following a previously described method (22). Briefly, stock cells were trypsinized, suspended at 20 × 104 cells/ml in medium, and seeded at confluent density onto 1.0-cm2 transwell inserts (Costar, Cambridge, Mass.). After 3 days, 2 mM sodium butyrate (Sigma) was added. Sodium butyrate is known to induce differentiation in many cells, including Caco-2 cells (1, 12, 13). It occurs naturally in normal human colon and is used as an energy source by colonocytes (18). Fecal levels of butyrate in healthy humans may be as high as 20 mM (4). Caco-2 cells treated with butyrate have higher transepithelial resistances and greater sensitivity to transepithelial Isc increases by TRH compared with those of nontreated cells (A. Takahashi and T. Honda, unpublished observations). Thus, butyrate-treated Caco-2 cells were used in this study to investigate the effects of TRH. The cells were cultured on the transwell inserts for 6 days and were mounted into a modified Ussing chamber and maintained at 37°C in modified Ringer solution (pH 7.4; gassed with 5% CO2–95% O2). Transepithelial resistance was measured by applying a 5-mV pulse at intervals of 40 to 50 s, and the resistance was calculated according to Ohm's law. [Ca2+]in was determined by microfluorometry using a fluorescent dye, 1-(2-(5′-carboxyoxazol-2′-yl)- 6-aminobenzofuran-5-oxy)-2(2′amino-5′-methylphenoxy) ethane-N,N,N′,N′-tetraacetic acid, pentaacetoxy methyl ester (Fura-2/AM; Molecular Probes, Eugene, Ore.). Using a previously described method (22, 23), TRH was purified from a clinically isolated strain of V. parahaemolyticus (RIMD 2210531), which was KP negative (TDH negative) and yielded a single band by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (Fig. 1A), suggesting that the toxins were purified to homogeneity.
FIG. 1.
(A) Sodium dodecyl sulfate-polyacrylamide gel electrophoresis gel of purified preparation of TRH. Lane 1, molecular mass markers (New England Biolabs Inc.) (175, 83, 62, 47.5, 32.5, 25, 16.5, and 6.5 kDa); lane 2, TRH. (B) Effect of gluconate ion and Ca2+ depletion on Isc in Caco-2 cell monolayers. ●, Control cells (simple exposure to TRH); ■, Cl− replaced by gluconate ion in bath solution of Ussing chamber; ▴, CaCl2 was omitted from bath solution and EGTA (1 mM) was added to bath solution of Ussing chamber. TRH was added on the apical side of the Caco-2 cells at the time indicated. ▾, negative control (no exposure to TRH). Values are expressed as means ± standard deviations (n = 5). ∗, Significant difference (P < 0.05) versus negative control.
TRH increased Isc in Caco-2 cell monolayers. Isc increased in Caco-2 cell monolayers when TRH was added to the apical side of the monolayers. When the Cl− in the bath solution was replaced with gluconate ion in both the apical and basolateral sides of the cells, Isc did not increase by addition of TRH (Fig. 1B). This indicates that the influence of TRH on Isc is dependent on extracellular Cl−. To deplete Ca2+ from the apical cell surface, CaCl2 was omitted from the bath solution and EGTA (1 mM) was added; in these conditions, after exposure to TRH, there was no change in Isc (Fig. 1B). This indicates that Isc change is also dependent on extracellular Ca2+. From these results, we hypothesized that Isc increases after exposure to TRH flow along Ca2+-activated Cl− channels.
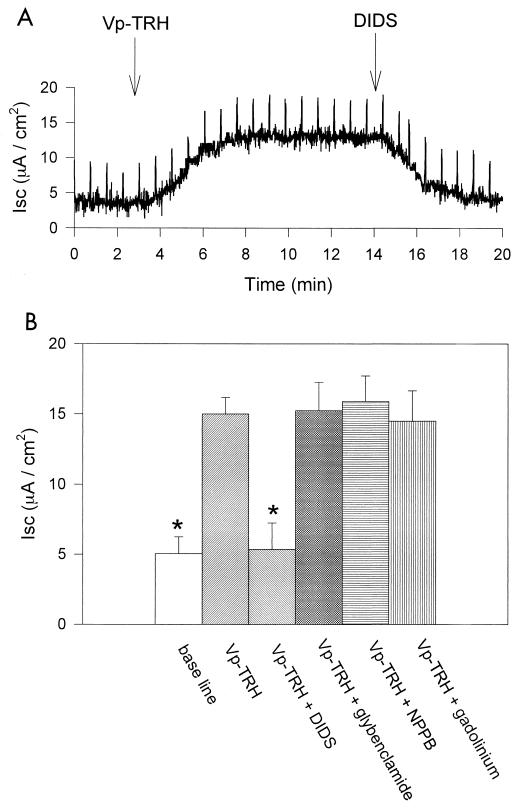
To explore this hypothesis, we used four kinds of channel inhibitors. Figure 2A shows that the influence of TRH on Isc was inhibited by 4,4′-diisothiocyanatostilbene-2,2′-disulfonic acid (DIDS) (300 μM), an inhibitor of the Ca2+-activated Cl− channel (5, 11).
FIG. 2.
Effect of inhibitors on Isc in Caco-2 cell monolayers. TRH (10 μg/ml) was added on the apical side at the time indicated. (A) DIDS (100 μM) was added on the apical side at the time indicated. (B) Effect of Cl− channel inhibitors, DIDS (100 μM), glybenclamide (300 μM), NPPB (100 μM), and gadolinium ion (Gd3+) (500 μM). Isc was measured after a 15-min exposure to TRH or 10 min after addition of Cl− channel inhibitor. Values are expressed as means ± standard deviations (n = 5). ∗, Significant difference (P < 0.05 versus TRH.
The cystic fibrosis transmembrane conductance regulator (CFTR) is one of the major Cl− channels in Caco-2 cells, and it is one of the most important Cl− secretion pathways involved in human diarrhea (6). We eliminated the possibility that CFTR is a target for TRH by testing with glybenclamide (300 μM) and 5-nitro-2-(3-phenylpropylamino)benzoic acid (NPPB) (100 μM), both of which are known to be inhibitors of CFTR (5, 19, 21); neither had any effect on the influence of TRH on Isc currents flowing through the Cl− channel (Fig. 2B).
We noted cell swelling within 15 min of adding TRH to Caco-2 cells (unpublished observation), which suggested the possibility that the stretch-activated channels may have opened. We tested with gadolinium ion (Gd3+), an inhibitor of stretch-activated channels (3, 7), and found it had no effect on Isc (Fig. 2B). This tends to rule out the association of stretch-activated channels with the Cl− secretion induced by TRH.
These observations are consistent with the conclusion that the effects of TRH on Isc are related to Ca2+-activated Cl− channels (20).
Intracellular Ca2+ concentration.
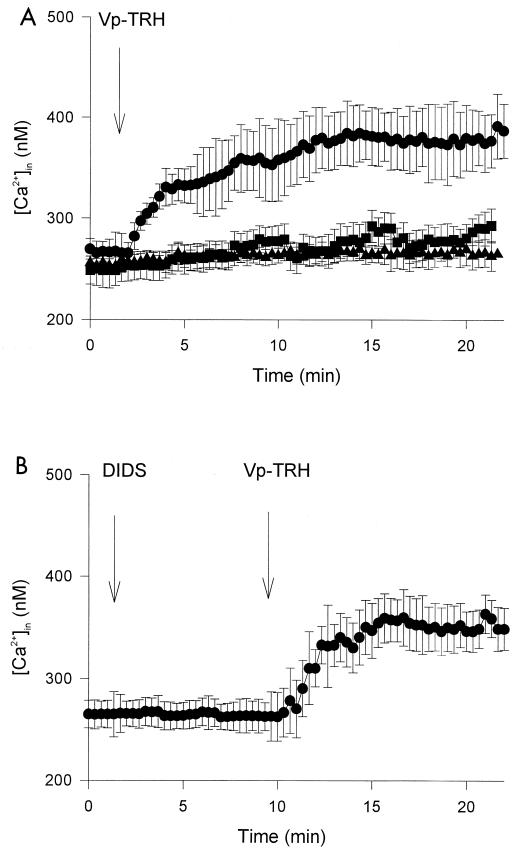
[Ca2+]in of Caco-2 cells increased in the presence of TRH (Fig. 3A). After depletion of Ca2+ from the cell culture medium by withholding CaCl2 from the bath solution and EGTA (1 mM), however, [Ca2+]in did not increase. Thus, the increase in [Ca2+]in was due to an influx of Ca2+ from the extracellular medium after exposure to TRH.
FIG. 3.
[Ca2+]in change after exposure of Caco-2 cell monolayers to TRH. (A) TRH (10 μg/ml) was added at the time indicated. ●, Positive control (simple exposure to Vp-TRH); ■, CaCl2 was depleted, and 1 mM EGTA was added to bath solution in fluorometer chamber, followed by addition of TRH and the inhibitors indicated; ▴, negative control (no exposure to Vp-TRH). (B) DIDS (300 μM) and TRH (10 μg/ml) were added at the times indicated. Values are expressed as means ± standard deviations (n = 10).
Next, we checked the effect of DIDS on [Ca2+]in. DIDS did not influence the increasing of [Ca2+]in induced by TRH (Fig. 3B).
Our data indicate that exposure to TRH resulted in greater Isc currents and that these increased currents flowed through the Cl− channels (Fig. 1B and 2).
TRH is a pore-forming toxin in erythrocytes (8, 9), suggesting the possibility that Cl− moves through pores formed by TRH. Our evidence, however, does not support this explanation: DIDS and depletion of Ca2+ from extracellular solution inhibited the effect of TRH on Cl− currents (Fig. 1A and 2). This would not have been the case if Cl− was simply passing through TRH-formed pores.
Considered overall, our findings suggest that the Cl− currents induced by TRH are mediated by the Ca2+-activated Cl− channel, because (i) there is evidence of [Ca2+]in dependency and (ii) DIDS, an inhibitor of the Ca2+-activated Cl− channel, inhibited the Isc (Fig. 1B and 2). Moreover, DIDS decreased the Cl− current without reducing the [Ca2+]in elevation (Fig. 3B), leading to the conclusion that increased Cl− currents were a secondary effect of increased [Ca2+]in.
TDH increased [Ca2+]in and activated the Ca2+-activated Cl− channel, resulting in Cl− secretion from the basolateral to the apical cell side (22). The effects of TRH to the Cl− secretion are similar to those of TDH. These confirm that TRH is also an important virulence factor. One should pay attention to TRH-producing V. parahaemolyticus, even if it is a KP-negative strain.
Acknowledgments
This work was funded by a Grant-in-Aid for the “Research for the Future” program of the Japan Society for the Promotion of Science (JSPS-RFJF 97L00704).
REFERENCES
- 1.Augeron C, Laboisse C L. Emergence of permanently differentiated cell clones in a human colonic cell line in culture after treatment with sodium butyrate. Cancer Res. 1984;44:3961–3969. [PubMed] [Google Scholar]
- 2.Centers for Disease Control and Prevention. Outbreak of Vibrio parahaemolyticus infection associated with eating raw oysters and clams harvested from Long Island Sound—Connecticut, New Jersey, and New York, 1998. Morb Mortal Wkly Rep. 1999;48:48–51. [PubMed] [Google Scholar]
- 3.Chen Y, Simasko S M, Niggel J, Sigurdson W J, Sachs F. Ca2+ uptake in GH3 cells during hypotonic swelling: the sensory role of stretch-activated ion channels. Am J Physiol. 1996;270:C1790–C1798. doi: 10.1152/ajpcell.1996.270.6.C1790. [DOI] [PubMed] [Google Scholar]
- 4.Cummings J H, Pomare E W, Branch W J, Naylor C P E, MacFarlane G T. Short chain fatty acids in human large intestine, portal, hepatic and venous blood. Gut. 1987;28:1221–1227. doi: 10.1136/gut.28.10.1221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Cunningham S A, Warrell R T, Benos D J, Frizzell R A. cAMP-stimulated ion currents in Xenopus oocytes expressing CFTR cRNA. Am J Physiol. 1992;262:C783–C788. doi: 10.1152/ajpcell.1992.262.3.C783. [DOI] [PubMed] [Google Scholar]
- 6.Fasano A. Cellular microbiology: can we learn cell physiology from microorganisms? Am J Physiol. 1999;276:C765–C776. doi: 10.1152/ajpcell.1999.276.4.C765. [DOI] [PubMed] [Google Scholar]
- 7.Garrill A, Tyerman S D, Findlay G P. Ion channels in the plasma membrane of protoplasts from the halophytic angiosperm Zostera muelleri. J Membr Biol. 1994;142:382–393. doi: 10.1007/BF00233443. [DOI] [PubMed] [Google Scholar]
- 8.Honda T, Iida T. The pathogenicity of Vibrio parahaemolyticus and the role of the thermostable direct haemolysin and related haemolysins. Rev Med Microbiol. 1993;4:106–113. [Google Scholar]
- 9.Honda T, Ni Y, Miwatani T, Kim J. The thermostable direct hemolysin of Vibrio parahaemolyticus is a pore-forming toxin. Can J Microbiol. 1992;38:1175–1180. doi: 10.1139/m92-192. [DOI] [PubMed] [Google Scholar]
- 10.Honda T, Ni Y X, Miwatani T. Purification and characterization of a hemolysin produced by a clinical isolation of Kanagawa phenomenon-negative Vibrio parahaemolyticus and related to the thermostable direct hemolysin. Infect Immun. 1988;56:961–965. doi: 10.1128/iai.56.4.961-965.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Inoue C N, Woo J S, Schwiebert E M, Morita T, Hanaoka K, Guggino S E, Guggino W B. Role of purinergic receptors in chloride secretion in Caco-2 cells. Am J Physiol. 1997;272:C1862–C1870. doi: 10.1152/ajpcell.1997.272.6.C1862. [DOI] [PubMed] [Google Scholar]
- 12.Jacewicz M S, Acheson D W, Mobassaleh M, Donohue-Rolf A, Balasubramanian K A, Keusch G T. Maturational regulation of globotriaosylceramide, the Shiga-like toxin 1 receptor, in cultured human gut epithelial cells. J Clin Investig. 1995;96:1328–1335. doi: 10.1172/JCI118168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Matsumoto H, Erikson R H, Gum J R, Yoshioka M, Gum E, Kim Y S. Biosynthesis of alkaline phosphatase during differentiation of the human colon cancer cell line Caco-2. Gastroenterology. 1990;98:1199–1207. doi: 10.1016/0016-5085(90)90334-w. [DOI] [PubMed] [Google Scholar]
- 14.Miyamoto K, Kato T, Obara Y, Akiyama S, Takizawa K, Yamai S. In vitro hemolytic characteristic of Vibrio parahaemolyticus: its close correlation with human pathogenicity. J Bacteriol. 1969;106:1147–1149. doi: 10.1128/jb.100.2.1147-1149.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Nishibuchi M, Fasano A, Russell R B, Kaper J B. Enterotoxigenicity of Vibrio parahaemolyticus with and without genes encoding thermostable direct hemolysin. Infect Immun. 1992;60:3539–3545. doi: 10.1128/iai.60.9.3539-3545.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Nishibuchi M, Taniguchi T, Misawa T, Khaeomanee-iam V, Honda T, Miwatani T. Cloning and nucleotide sequence of the gene (trh) encoding the hemolysin related to the thermostable direct hemolysin of Vibrio parahaemolyticus. Infect Immun. 1989;57:2691–2697. doi: 10.1128/iai.57.9.2691-2697.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Okuda J, Ishibashi M, Hayakawa E, Nishino T, Takeda Y, Mukhopadhyay A K, Garg S, Bhattacharya S K, Nair G B, Nishibuchi M. Emergence of a unique O3:K6 clone of Vibrio parahaemolyticus in Calcutta, India, and isolation of strains from the same clonal group from Southeast Asia travelers arriving in Japan. J Clin Microbiol. 1997;35:3150–3155. doi: 10.1128/jcm.35.12.3150-3155.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Scheppach W. Effects of short chain fatty acids on gut morphology and function. Gut. 1994;35(Suppl. 1):S35–S38. doi: 10.1136/gut.35.1_suppl.s35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Schultz B D, Singh A K, Devor D C, Bridges R J. Pharmacology of CFTR chloride channel activity. Physiol Rev. 1999;79:S109–S144. doi: 10.1152/physrev.1999.79.1.S109. [DOI] [PubMed] [Google Scholar]
- 20.Schwiebert E M, Egan M E, Hwang T H, Fulmer S B, Allen S S, Cutting G R, Guggino W B. CFTR regulates outwardly rectifying chloride channels through an autocrine mechanism involving ATP. Cell. 1995;81:1063–1073. doi: 10.1016/s0092-8674(05)80011-x. [DOI] [PubMed] [Google Scholar]
- 21.Sheppard D N, Welsh M J. Inhibition of the cystic fibrosis transmembrane conductance regulator by ATP-sensitive K+ channel regulators. Annu N Y Acad Sci. 1993;707:275–284. doi: 10.1111/j.1749-6632.1993.tb38058.x. [DOI] [PubMed] [Google Scholar]
- 22.Takahashi A, Sato Y, Shiomi Y, Cantarelli V V, Iida T, Lee M, Honda T. Mechanisms of Cl− secretion induced by thermostable direct hemolysin of Vibrio parahaemolyticus in human colonic tissue and a human intestinal epithelial cell line. J Med Microbiol. 2000;49:1–10. doi: 10.1099/0022-1317-49-9-801. [DOI] [PubMed] [Google Scholar]
- 23.Takeda Y. Thermostable direct hemolysin of Vibrio parahaemolyticus. Methods Enzymol. 1988;165:189–193. doi: 10.1016/s0076-6879(88)65029-4. [DOI] [PubMed] [Google Scholar]
- 24.Yoh M, Miwatani T, Honda T. Comparison of Vibrio parahaemolyticus hemolysin (Vp-TRH) produced by environmental and clinical isolation. FEMS Microbiol Lett. 1992;92:157–162. doi: 10.1016/0378-1097(92)90505-i. [DOI] [PubMed] [Google Scholar]