Abstract
The manifestation of leprosy in humans is largely determined by host immunity to Mycobacterium leprae and is a model for immunoregulation in a human disease. However, animal models available for exploration of the leprosy spectrum are inadequate. This study explored M. leprae infection in mice deficient in inducible nitric oxide synthase, and this report describes elements resembling borderline tuberculoid leprosy in humans.
A major component of the antimicrobial repertoire of effector mechanisms of activated macrophages (MΦ) is the production of reactive nitrogen intermediates (RNI), chiefly nitric oxide, generated from l-arginine by a high-output, cytokine-inducible isoform of the enzyme, inducible nitric oxide synthase (iNOS). In murine models, RNI have been shown to be necessary for the MΦ-mediated inhibition of tumor cells and a variety of extracellular and intracellular pathogens (6, 17). Evidence that RNI play a key antimicrobial role in human MΦ is less direct but is accumulating (21).
The use of the competitive inhibitor of l-arginine, NG-monomethyl-l-arginine (L-NMA), as well as other inhibitors of iNOS, including aminoguanidine (AG), allowed in vitro demonstration that MΦ RNI are important in the gamma interferon (IFN-γ)-activated MΦ killing of Mycobacterium leprae (2) and M. tuberculosis (1, 8, 11). The inclusion of L-NMA or AG in drinking water permitted in vivo demonstration of the role of RNI in host resistance to M. bovis BCG (15) and M. tuberculosis (7, 13), respectively. Recently, gene knockout (KO) mice with a targeted disruption in the calmodulin binding site of the iNOS gene (iNOS KO mice) were developed which lack a functional iNOS (16). These iNOS KO mice yielded IFN-γ-treated MΦ incapable of coping with M. tuberculosis (1). In vivo infection with virulent M. tuberculosis was greatly exacerbated in this and another strain of iNOS-deficient mice (1, 18).
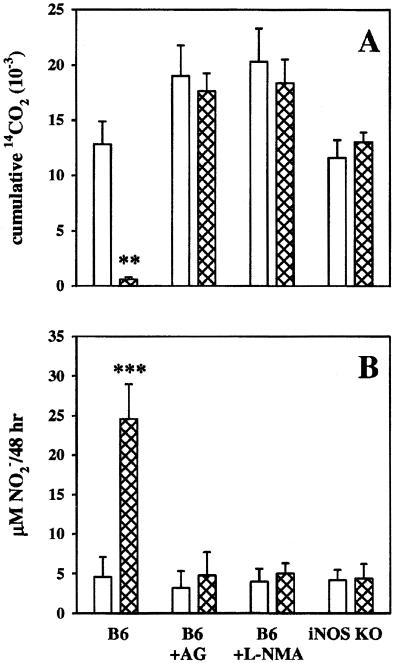
The present studies employed iNOS KO mice to explore further the role of RNI in host resistance to viable M. leprae freshly harvested from infected nu/nu mice (3). Figure 1 shows in vitro results obtained with B6 and iNOS KO peritoneal MΦ where additional controls consisted of B6 MΦ treated with the RNI inhibitors L-NMA and AG. Clearly, activated iNOS KO MΦ or B6 MΦ treated with L-NMA or AG failed to produce RNI and were unable to kill or inhibit M. leprae. Similar results were obtained with bone marrow-derived MΦ (not shown).
FIG. 1.
Effects of activated peritoneal MΦ from B6 and iNOS KO mice on M. leprae. (A) Viability of M. leprae recovered from control (open bars) or IFN-plus-lipopolysaccharide-activated (hatched bars) B6 MΦ incubated in the presence or absence of 500 μM AG or 500 μM L-NMA and from iNOS KO MΦ. Viability of bacilli was determined 48 h postinfection by radiorespirometric measurement of the oxidation of [14C]palmitic acid (19). (B) Nitrite levels in the culture supernatants were determined using the Griess reagent. Control versus activated MΦ: ∗∗, P < 0.01; ∗∗∗, P < 0.001.
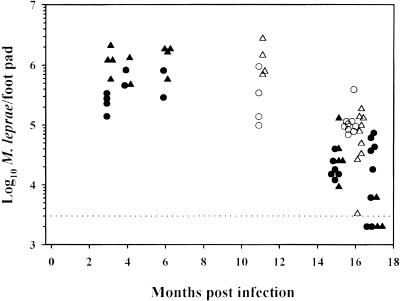
Dietary inhibition of RNI production was not a feasible approach by which to study the role of this antimicrobial mechanism of resistance to leprosy in vivo because the protracted nature of the mouse footpad model for infection with M. leprae requires several months to demonstrate growth. Therefore, M. leprae infection was carried out with iNOS KO mice to determine if, as in the M. tuberculosis model, MΦ inability to cope with M. leprae infection in vitro is also manifested in vivo by exacerbated infection. We employed the standard Shepard mouse footpad assay (20), in which the number of acid-fast bacilli (AFB) peaks at 4 to 6 months, plateaus for several months, and then gradually falls off to the counting threshold. As shown in Fig. 2, M. leprae growth followed this typical pattern in control B6 mice over 18 months. In iNOS KO mice, counts of AFB per footpad were generally higher at 3 and 6 months in one experiment and 11 months in a second experiment although statistical significance (Welch's t test) was reached only at the 3-month time point (P < 0.05). More noteworthy, considering the marked differences in granuloma formation and histopathological responses (see below), were the findings that the number of AFB per footpad fell off at the same rate and to the same level in KO mice as in controls.
FIG. 2.
Growth of M. leprae in mouse in footpads. B6 (circles) and iNOS KO (triangles) mice were infected in both hind footpads with 104 viable, nu/nu mouse-derived M. leprae organisms. At the indicated times postinfection, the number of AFB per footpad was determined. Open and closed symbols represent two independent experiments. Each symbol represents one mouse, except at the 4- and 6-month time points, where each symbol represents a pool of two mice. The threshold of counting ability (dotted line) is 3 × 103 AFB per footpad.
Clearly, growth of M. leprae was not greatly exacerbated in iNOS KO mice. These results appear to contrast with findings obtained with experimental M. tuberculosis infections, in which the absence of RNI was linked with the inability of activated MΦ to kill in vitro coupled with exacerbated growth of the tubercle bacillus and increased mouse mortality (1, 7, 8, 13, 18). Our findings also contrast with reports in which iNOS-deficient mice not only controlled M. avium growth in vivo but also did so in vitro in activated MΦ (4, 5, 12, 14). Thus, although the present studies appear to underscore the important role of RNI in MΦ-mediated resistance to M. leprae, these findings also demonstrate that control of M. leprae infection in vivo can be maintained in the absence of this potent antimicrobial mechanism. An important alternative antimicrobial pathway to be considered is the production of reactive oxygen intermediates (ROI) by MΦ. We have shown, however, that MΦ from iNOS KO mice are not deficient in the production of ROI (1) but, in the absence of RNI, ROI alone is not sufficient to kill either M. tuberculosis or M. leprae.
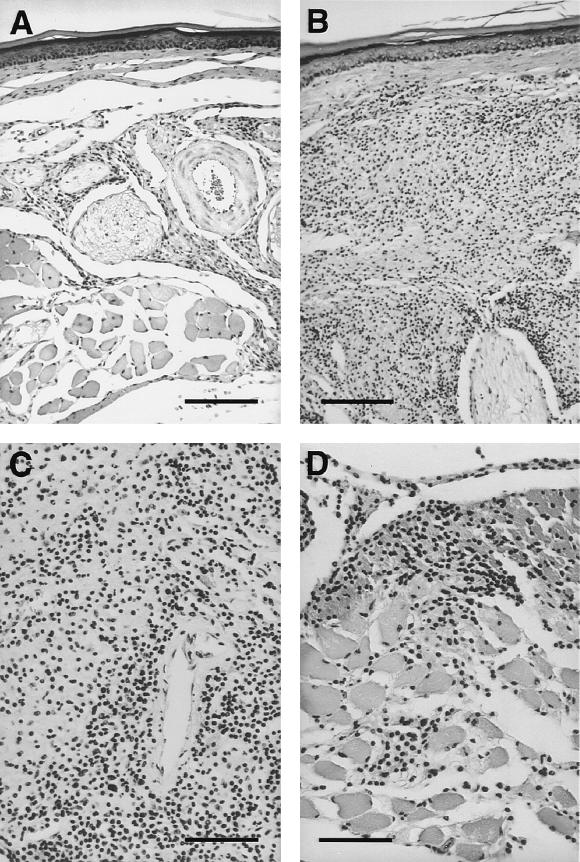
Control of M. leprae growth in iNOS-deficient mice may involve a more complicated compensatory consequence of the microenvironmental conditions associated with granuloma formation. At 15 months in B6 mice, the granuloma which formed around an injected bolus of M. leprae was confined to a small area in the footpad immediately underneath the epidermis (Fig. 3A) and consisted of small focal collections of MΦ, a few epithelioid cells, and lymphocytes which were collected around neurovascular bundles. The inflammation did not invade nerves or muscles. There were minimal superficial extensions along the tissue spaces between muscle bundles. Acid-fast staining (not shown) revealed a few clumps of bacilli inside MΦ, especially in those found immediately beneath the epidermis. In marked contrast, the granulomatous inflammation of the iNOS KO footpad was over 10 times the size of the granuloma found in the control mice (Fig. 3B). The granuloma was distributed over an area underneath the epidermis, around neurovascular bundles, and was spread along the tissue spaces between muscle bundles, even reaching the bones. The granuloma was confluent and organized and was composed of numerous epithelioid cells, a few MΦ, and dense collections of lymphocytes (Fig. 3C). In areas, the granuloma infiltrated the muscle bundles, partly destroying them (Fig. 3D), and also infiltrated the perineurium. Acid-fast staining (not shown) revealed clumps of bacilli inside MΦ mainly in the middle of the granuloma. Thus, granuloma formation in iNOS KO mice was ultimately associated with destruction of M. leprae and persisting organisms continued to evoke a granulomatous response. Two recent studies (12, 14) have shown enhanced granuloma formation in M. avium-infected mice when production of RNI was inhibited by either gene disruption or chemical inhibition, and this response was attributed to modulation of cytokine production. Ongoing immunohistopathological studies and evaluation of local cytokine profiles in our M. leprae-infected mouse footpad model may shed further light on these compensatory mechanisms of resistance.
FIG. 3.
Granulomatous response in mouse footpads in response to infection with 104 M. leprae organisms. (A) Small granuloma around a neurovascular bundle in a B6 mouse at 15 months postinfection. It is composed primarily of spindle-shaped MΦ and a few lymphocytes (hematoxylin-and-eosin staining; bar, 100 μm). (B) The granuloma in an iNOS KO mouse at 15 months postinfection was over 10 times the size of that seen in B6 mice and was composed of epithelioid cells and numerous lymphocytes (hematoxylin-and-eosin staining; bar, 100 μm). (C) Higher-power view of the granuloma in iNOS KO mice to show the epithelioid cells with abundant cytoplasm bordered by a dense collection of lymphocytes (hematoxylin-and-eosin staining; bar, 50 μm). (D) The granuloma in iNOS KO mice infiltrated the striated muscles and partly destroyed them (hematoxylin-and-eosin staining; bar, 50 μm).
Finally, it is noteworthy that our findings appear to share attributes with an important form of human leprosy. Overall, the histological picture seen in iNOS KO mice was one resembling human borderline tuberculoid leprosy. The broad clinical-immunopathological spectrum of leprosy represents a fascinating example of immunoregulation in a chronic, nonfatal human infectious disease. But current murine models of experimental leprosy generally represent only the polar forms of the disease: tuberculoid leprosy with limited growth in the footpads of conventional, immunocompetent mice and lepromatous leprosy in which growth is largely unrestricted in the footpads of athymic nu/nu mice (9, 10). The borderline area of the clinical-immunopathological manifestations of leprosy is by far the most intriguing to immunologists. Unlike the tuberculoid and lepromatous forms, the borderline form is unstable and may downgrade and evolve into a more lepromatous disease or upgrade into a more tuberculoid disease, often with the appearance of a “reversal reaction” resulting from the onset of enhanced cell-mediated immunity with damaging consequences for surrounding nerves.
In humans, understanding the mechanisms and prevention of reversal reactions is one of the main goals of leprosy research. The development of strains of mice with targeted gene disruptions has opened up the possibility of exploring models more representative of the human leprosy spectrum. As the M. leprae-infected footpad lesions of iNOS KO mice appear to resemble borderline tuberculoid lesions, the present findings may be exploitable as a model for understanding the instability of borderline disease. Experiments are under way to determine the host response to larger numbers of M. leprae organisms and explore the possibility of further manipulation of the cell-mediated immune system in infected iNOS KO mice.
Acknowledgments
We thank Nashone Soileau, J. P. Pasqua, Julie Loesch, Angelina Deming, and Greg McCormick for technical assistance and Penne Cason for clerical help.
REFERENCES
- 1.Adams L B, Dinauer M C, Morgenstern D E, Krahenbuhl J L. Comparison of the roles of reactive oxygen and nitrogen intermediates in the host response to Mycobacterium tuberculosis using transgenic mice. Tubercle Lung Dis. 1997;78:237–246. doi: 10.1016/s0962-8479(97)90004-6. [DOI] [PubMed] [Google Scholar]
- 2.Adams L B, Franzblau S G, Vavrin Z, Hibbs J B, Jr, Krahenbuhl J L. l-Arginine-dependent macrophage effector functions inhibit metabolic activity of Mycobacterium leprae. J Immunol. 1991;147:1642–1646. [PubMed] [Google Scholar]
- 3.Adams L B, Soileau N A, Battista J R, Krahenbuhl J L. Inhibition of metabolism and growth of Mycobacterium leprae by gamma-irradiation. Int J Lepr. 2000;68:1–10. [PubMed] [Google Scholar]
- 4.Appelberg R, Orme I M. Effector mechanisms involved in cytokine-mediated bacteriostasis of Mycobacterium avium infection in murine macrophages. Immunology. 1993;80:352–359. [PMC free article] [PubMed] [Google Scholar]
- 5.Bermudez L E. Differential mechanisms of intracellular killing of Mycobacterium avium and Listeria monocytogenes by activated human and murine macrophages. The role of nitric oxide. Clin Exp Immunol. 1993;91:277–281. doi: 10.1111/j.1365-2249.1993.tb05895.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bogdan C, Rollinghoff M, Diefenbach A. Reactive oxygen and reactive nitrogen intermediates in innate and specific immunity. Curr Opin Immunol. 2000;12:64–76. doi: 10.1016/s0952-7915(99)00052-7. [DOI] [PubMed] [Google Scholar]
- 7.Chan J, Tanaka K, Carroll D, Flynn J, Bloom B R. Effects of nitric oxide synthase inhibitors on murine infection with Mycobacterium tuberculosis. Infect Immun. 1995;63:736–740. doi: 10.1128/iai.63.2.736-740.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Chan J, Xing Y, Magliozzo R S, Bloom B R. Killing of virulent Mycobacterium tuberculosis by reactive nitrogen intermediates produced by activated murine macrophages. J Exp Med. 1992;175:1111–1122. doi: 10.1084/jem.175.4.1111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Chehl S, Ruby J, Job C K, Hastings R C. The growth of Mycobacterium leprae in nude mice. Lepr Rev. 1983;54:283–304. doi: 10.5935/0305-7518.19830035. [DOI] [PubMed] [Google Scholar]
- 10.Colston M J, Hilson G R. Growth of Mycobacterium leprae and M. marinum in congenitally athymic (nude) mice. Nature. 1976;262:736–741. doi: 10.1038/262399a0. [DOI] [PubMed] [Google Scholar]
- 11.Denis M. Interferon-gamma-treated murine macrophages inhibit growth of tubercle bacilli via the generation of reactive nitrogen intermediates. Cell Immunol. 1991;132:150–157. doi: 10.1016/0008-8749(91)90014-3. [DOI] [PubMed] [Google Scholar]
- 12.Ehlers S, Kutsch S, Benini J, Cooper A, Hahn C, Gerdes J, Orme I, Martin C, Rietschel E T. NOS2-derived nitric oxide regulates the size, quantity and quality of granuloma formation in Mycobacterium avium-infected mice without affecting bacterial loads. Immunology. 1999;98:313–323. doi: 10.1046/j.1365-2567.1999.00875.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Flynn J L, Scanga C A, Tanaka K E, Chan J. Effects of aminoguanidine on latent murine tuberculosis. J Immunol. 1998;160:1796–1803. [PubMed] [Google Scholar]
- 14.Gomes M S, Florido M, Pais T F, Appelberg R. Improved clearance of Mycobacterium avium upon disruption of the inducible nitric oxide synthase gene. J Immunol. 1999;162:6734–6739. [PubMed] [Google Scholar]
- 15.Granger D L, Hibbs J B, Jr, Broadnax L M. Urinary nitrate excretion in relation to murine macrophage activation. Influence of dietary l-arginine and oral NG-monomethyl-l-arginine. J Immunol. 1991;146:1294–1302. [PubMed] [Google Scholar]
- 16.Laubach V E, Shesely E G, Smithies O, Sherman P A. Mice lacking inducible nitric oxide synthase are not resistant to lipopolysaccharide-induced death. Proc Natl Acad Sci USA. 1995;92:10688–10692. doi: 10.1073/pnas.92.23.10688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.MacMicking J, Xie Q, Nathan C. Nitric oxide and macrophage function. Annu Rev Immunol. 1997;15:323–350. doi: 10.1146/annurev.immunol.15.1.323. [DOI] [PubMed] [Google Scholar]
- 18.MacMicking J D, North R J, LaCourse R, Mudgett J S, Shah S K, Nathan C F. Identification of nitric oxide synthase as a protective locus against tuberculosis. Proc Natl Acad Sci USA. 1997;94:5243–5248. doi: 10.1073/pnas.94.10.5243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ramasesh N, Adams L B, Franzblau S G, Krahenbuhl J L. Effects of activated macrophages on Mycobacterium leprae. Infect Immun. 1991;59:2864–2869. doi: 10.1128/iai.59.9.2864-2869.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Shepard C C, McRae D H. A method for counting acid-fast bacteria. Int J Lepr. 1968;36:78–82. [PubMed] [Google Scholar]
- 21.Weinberg J B. Nitric oxide production and nitric oxide synthase type 2 expression by human mononuclear phagocytes: a review. Mol Med. 1998;4:557–591. doi: 10.1007/BF03401758. [DOI] [PMC free article] [PubMed] [Google Scholar]