Abstract
Neurogenesis, the formation of new neurons in the brain, occurs throughout the lifespan in the subgranular zone of the dentate gyrus and subventricular zone (SVZ) lining the lateral ventricles of the mammal brain. In this process, gamma-aminobutyric acid (GABA) and its ionotropic receptor, the GABAA receptor (GABAAR), play a critical role in the proliferation, differentiation, and migration process of neural stem/progenitor cells (NPC). Taurine, a non-essential amino acid widely distributed throughout the central nervous system, increases the proliferation of SVZ progenitor cells by a mechanism that may involve GABAAR activation. Therefore, we characterized the effects of taurine on the differentiation process of NPC expressing GABAAR. Preincubation of NPC-SVZ with taurine increased microtubule-stabilizing proteins assessed with the doublecortin assay. Taurine, like GABA, stimulated a neuronal-like morphology of NPC-SVZ and increased the number and length of primary, secondary, and tertiary neurites compared with control NPC of the SVZ. Furthermore, neurite outgrowth was prevented when simultaneously incubating cells with taurine or GABA and the GABAAR blocker, picrotoxin. Patch-clamp recordings revealed a series of modifications in the NPCs’ passive and active electrophysiological properties exposed to taurine, including regenerative spikes with kinetic properties similar to the action potentials of functional neurons.
Supplementary Information
The online version contains supplementary material available at 10.1007/s11064-023-03883-2.
Keywords: Taurine, GABA Receptors, Subventricular Cells, Stem/progenitor Cell, Neurogenesis, Differentiation, Morphogenesis
Introduction
Neurogenesis, the process of generating functional neurons from precursor cells, was traditionally associated with mammals’ embryonic and perinatal developmental stages [1]. However, the milestone discovery by Altman and Das (1965) [2] demonstrating the presence of newborn neurons in the adult brain showed for the first time that active neurogenesis continues throughout the lifespan of the mammalian brain [2]. Today, active adult neurogenesis is acknowledged as a neurodevelopmental process that includes cell proliferation, migration, differentiation, cell fate decision, and functional integration into pre-existing neuronal networks [3]. Adult neurogenesis is a dynamic and accurate process regulated by multiple physiological, pharmacological, and pathological stimuli [4]. Previous studies demonstrated that pharmacological modulation of gamma-aminobutyric acid (GABA) transmission regulates neurogenesis in the adult brain [5, 6], suggesting that GABA-activated responses trigger a series of molecular and biochemical mechanisms relevant to the neurogenesis process.
Under physiological conditions, neurogenesis is restricted to two neurogenic regions: the subgranular zone of the dentate gyrus, an area that generates new granule cells [2]; and the subventricular zone (SVZ) of the lateral ventricles, where new neurons are generated and migrate through the rostral migratory stream to the olfactory bulb where they mature into interneurons [7].
It is well documented that GABA has a depolarizing activity in neural precursor cells during brain development, controlling cell division and contributing to neuronal migration and maturation. Moreover, neural stem/progenitor cells (NPC) express at least GABAA and metabotropic GABA receptors (GABABR) in the subventricular and subgranular zones [8]; in these cells, GABA causes depolarization and affects proliferation, migration, and neuronal maturation [9]. Electrophysiological studies have documented that the tonic release of GABA activates GABAA receptors (GABAAR) expressed in neuroblasts and stem cells [10–13]. In addition, GABA, or the selective GABAAR agonist, muscimol, activates a chloride current in neonatal NPC-SVZ [10]. In line with these findings, acute treatment of type B and C SVZ cells with muscimol decreases cell proliferation, assessed with a bromodeoxyuridine incorporation assay [14]. Interestingly, taurine, a neuroactive molecule and structurally related analog of GABA, mimics the GABA action by activating GABAAR [15, 16].
Taurine (2-aminoethane sulfonic acid) is an abundant free amino acid found in the mammalian brain [17] and is also present in the heart, retina, and muscle tissues [18]. Humans obtain taurine directly from meat consumption, as a secondary source, or via a reduced biochemical synthesis process [19]. Taurine is synthesized from cysteine, in which cysteine dioxygenase and cysteine sulfinic acid decarboxylase form hypotaurine. Hypotaurine is converted by the hypotaurine dehydrogenase enzyme to taurine [20]. Experimental evidence demonstrates that taurine acts as a trophic factor, stimulates proliferation, and promotes neurogenesis of NPC-SVZ cultures [21]. These observations suggest that taurine plays a central role in the process of proliferation, differentiation, and migration of progenitor cells during the embryonic development of the central nervous system. However, the mechanism by which taurine participates in NPC differentiation of the SVZ is unclear. Because taurine is one of the most abundant free neuroactive molecules from an early CNS developmental stage and GABAAR is present on SVZ progenitor cells, the present study aimed to evaluate the effect of taurine on the differentiation process of NPC-SVZ through interaction with GABAAR.
Materials and Methods
Cell Culture
All the animals were handled following the guidelines of the National Institutes of Health Guide for Care and Use of Laboratory Animals and with the approval of the Institutional Animal Care and Use Committee of the National University of Mexico (protocol # 068-2019/018-CIC-2019). Only 8-day-old CD1 mice were used to obtain NPC-SVZ, in total 100 animals were used for 4 experimental groups (25 animals per group). Animals were collected from a laboratory animal facility and decapitated as soon as they arrived at the laboratory, the brain was removed and the SVZ was isolated. No randomization was performed to allocate subjects in the study and no blinding was performed. The tissue obtained was mechanically dissociated, and placed in DMEM/F12 (Dulbecco’s Modified Eagle Medium/Nutrient Mixture F-12. Gibco; Thermo Fisher Scientific, Inc., Waltham, MA, USA) medium, and centrifuged for 5 min at 1000 rpm. The supernatant was removed, and the button was resuspended and cultured in a growth medium (DMEM/F12 supplemented with B27, plus epidermal growth factor and fibroblast growth factor 2) to promote progenitor cells proliferation and SVZ-neurosphere formation. The culture was kept at 37 °C and 5% CO2. After 7 days, primary neurospheres were disaggregated with trypsin for 5 min, and a trypsin inhibitor (1:1) was added to stop the reaction. Once the cells were separated, they were recultured in a fresh growth medium for 4–5 days at 37 °C and 5% CO2. For the differentiation process of NPC-SVZ, disaggregated cells of the secondary SVZ-neurospheres were cultured in 12 wells pretreated with poly-D-lysine (Sigma-Aldrich, St. Louis, MO, USA), and differentiation medium (DMEM/F12 supplemented with 1% fetal bovine serum [FBS]) and complemented with taurine (10 mM) or GABA (100 µM). A dose-response was performed to determine the effective concentration for taurine, cell cultures were exposed to taurine at several concentrations (from 2.5 to 60 mM). To evaluate the role of GABAAR in the differentiation process, cells were preincubated for 1 h with picrotoxin (5 µM, 50 µM, and 100 µM); after this time, the medium was stirred to withdraw the stimulus and a differentiation medium or treatment with taurine (10 mM) or GABA (100 µM) was applied. GABA was used as a positive control to compare the mechanism of taurine on NPC differentiation processes.
Immunofluorescence Assay
Cells were fixed with cold 4% paraformaldehyde, washed with phosphate-buffered saline (PBS) + 0.1% bovine serum albumin (BSA; 3 × 5 min each), permeabilized/blocked with PBS + 0.1% BSA + 10% FBS + 0.3% Triton X-100 for 1 h at room temperature. The cells were subsequently incubated overnight with the primary antibodies nestin (Cell Signaling Technology Cat# 4760, RRID:AB_2235913) [1:300]; glial fibrillary acidic protein (GFAP; Cell Signaling Technology Cat# 12,389, RRID:AB_2631098) [1:100]; and doublecortin (DCX; Santa Cruz Biotechnology Cat# sc-271,390, RRID:AB_10610966) [1:100]. The primary antibodies were removed the following day, followed by 1-h incubation with the secondary antibodies Alexa Fluor-488 anti-rabbit 9 for GFAP and SOX-2) and Alexa Fluor-647 anti-mouse (for nestin and DCX). Next, the excess secondary antibodies were washed and removed from the preparation assembly. The nuclei were stained with Hoechst (20 µg/mL) included in the mounting solution (100/500 µL).
Quantitative Analysis for cell Markers
Microphotographs were obtained with the cell imaging multi-mode plate reader Cytation™ 5 (BioTek Instruments Inc., Winooski, VT, USA) using a green fluorescent protein (GFP) filter cube (excitation 469/35 nm; emission 525/39 nm; dichroic mirror 497 nm) and 465 nm LED; Texas Red filter cube (excitation 586/15 nm; emission 647/67 nm; dichroic mirror 605 nm) and 590 nm LED; and 4′,6-diamidino-2-phenylindole (DAPI) filter cube (excitation 377/50 nm; emission 447/60 nm; dichroic mirror 409) and 365 nm LED. Microphotographs of five different fields were randomly selected from each coverslip, and the number of totals (Hoescht positive) cells and cells positive for the different markers in the selected fields were quantified. Cell images were processed using Gen5™ software (BioTek Instruments Inc.). The results were expressed as the percentage of positive cells of the total cell number.
Morphometric Analysis of DCX Positive Cells
Microphotographs of DCX positive (DCX+) cells were randomly obtained with a 20x magnification lens. DCX + cells were analyzed for each experimental condition (control, taurine, and GABA). The number of primary, secondary, and tertiary neurites and the length of the primary neurites were determined with Gen 5 3.04. The difference between primary, secondary, and tertiary neurites was analyzed in the different experimental conditions. Primary neurites were defined as those that originated directly from the soma; secondary and tertiary neurites were defined as those that originated from primary and secondary neurites, respectively [22].
Whole-cell Patch-clamp Recordings
Dissociated NPC-SVZ from SVZ-neurospheres were taken from a petri dish containing cell culture, mounted on a coverslip, and transferred to a submersion recording chamber. They were visualized with infrared differential interference contrast optics coupled to a Nikon FN1 microscope (Nikon Corporation, Minato, Tokyo, Japan). The NPC-SVZ were continuously perfused with Ringer’s solution at the rate of 3–4 mL.min− 1 with the help of a peristaltic pump (120 S, Watson-Marlow, Wilmington, MA, USA). The composition of Ringer’s solution was the following (mM): 125 NaCl, 2.5 KCl, 1.25 Na2HPO4, 25 NaHCO3, 2 MgCl2, 2 CaCl2, and 10 D-glucose; pH 7.30–7.40; osmolarity = 280–290 mOsm. The recording chamber temperature was maintained at 32 ± 1 °C with a single channel temperature controller (TC-324 C, Warner Instruments, Hamden, CT, USA). For the whole-cell patch-clamp recordings of hippocampal interneurons included in this study, the parameters were published elsewhere [23]. The patch pipettes were pulled from borosilicate glasses using a micropipette puller (P57, Sutter Instruments, Novato, CA, USA). The puller was programmed to obtain pipettes with a tip resistance of 5–7 MΩ when filled with an intracellular solution containing (mM): 135 K+-gluconate, 10 KCl, 5 NaCl, 1 EGTA, 10 HEPES, 2 Mg2+-ATP, 0.4 Na+-GTP, 10 phosphocreatine, and pH 7.20–7.30. Whole-cell recordings were performed with an Axopatch 200B amplifier (Molecular Devices, San José, CA, USA). Acquired data were digitized at a sampling frequency of 10 kHz and filtered at 5 kHz with a Digidata 1322 A (Axon Instruments, Palo Alto, CA, USA). Signals were acquired and analyzed offline with pCLAMP 10.6 software (Molecular Devices).
Determination of Intrinsic Properties
The resting membrane potential (RMP) was determined after the initial break-in from giga-seal to whole-cell configuration. Once the cell was switched to current-clamp mode, a series of negative and positive current steps from − 300 to rheobase (1 s, 30 pA increments) were injected to determine input resistance (RN) and membrane time constant (τmemb). RN was calculated as the slope value of a first-order polynomial function f(x) = mx + b fitted to the current-voltage relationship near RMP. τmemb was determined by fitting a mono-exponential decay function to a voltage response elicited by injecting a negative current step (1 s, –30 pA). Membrane capacitance (Cm) was calculated as the ratio of τmemb to RN, as previously reported [24].
Determination of Firing Output Properties
We analyzed the firing properties of recorded NPC-SVZ by constructing current-frequency plots. For these measurements, the firing frequency was plotted against the intensity of the injected current. Experimental data were fitted to a three-parameter logistic function.
Spike and Action Potential Analyses
The spike threshold was calculated using the third derivative’s first peak intersecting with the spike onset. The spike kinetic properties were analyzed by constructing phase plots. In this analysis, the first derivative of the action potential (AP) / (mV.ms−1) was plotted against the membrane potential, as previously reported [25].
Statistical Analysis
No statistical method was used to determine the sample size. The sample size was arbitrarily set to 100 (4 groups with 25 animals each). Statistical analysis was performed with GraphPad Prism 6. The graphs represent the mean ± standard error of the mean. Data normality was previously determined with a Kolmogorov-Smirnov test. Most of the data did not meet the normality criteria, so non-parametric tests were applied: Mann-Whitney U for comparing two groups and Kruskal-Wallis for comparing more than two groups. A one-way analysis of variance (ANOVA) test was applied, followed by Tukey’s post hoc test in cases where normal distribution was observed. In cases where the comparison was between two groups, the Student’s t-test was applied. In all cases, the accepted level of significance was P < 0.05.
Results
Characterization of NPC Markers in SVZ-derived Neurospheres
Cultured SVZ-neurospheres are widely used to mimic NPC’s proliferative capacity, self-renewal, and differentiation into mature cell lineages; therefore, we used SVZ neurospheres to characterize the effects of taurine on the differentiation processes of NPC-SVZ. The neurospheres were obtained from 8-day-old CD1 mice and cultured in a growth medium with two passages (see Materials and Methods for details). Primary, secondary, and tertiary SVZ-neurospheres were formed at 7 ± 1, 12 ± 1, and 17 ± 1 days, respectively (Suppl Fig. 1A–C). For this study, all the experimental manipulations were restricted to secondary SVZ-neurospheres (Suppl Fig. 1B) to minimize biased interpretations given the cellular changes that occur during self-renewal (unorganized structure) or senescence (necrotic center) of SVZ-neurospheres [26].
We first determined the presence of progenitor and neuronal markers in the secondary SVZ-neurospheres. Immunofluorescence image analysis revealed nestin and SOX-2 immunopositivity (Suppl Fig. 2A, B); contrary to this observation, we did not detect DCX immunofluorescence (Suppl Fig. 2C). The immunopositivity to SOX-2 and nestin confirms the progenitor origin of the NPC-SVZ, whereas the lack of DCX immunopositivity indicates a lack of differentiation to immature neurons.
Expression of Neuronal Markers in NPC-SVZ Exposed to Taurine or GABA
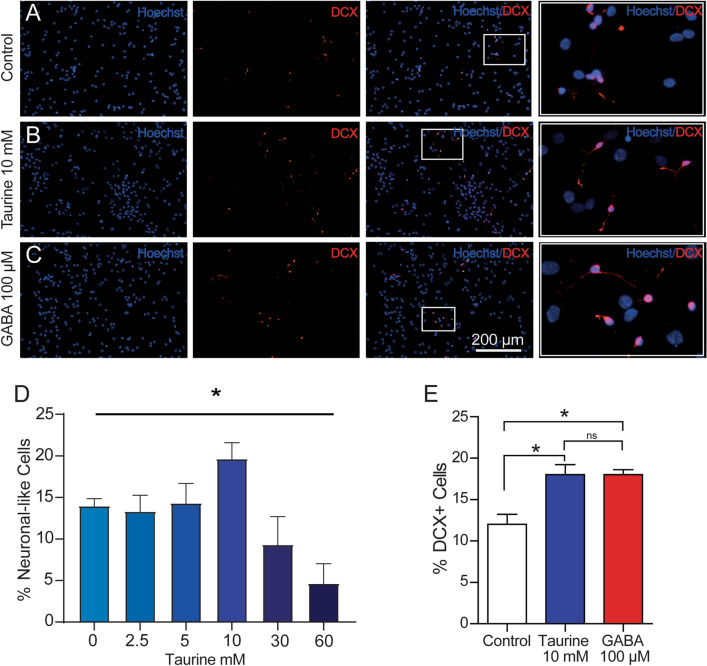
Next, the expression of the neuronal markers during the differentiation process was analyzed in disaggregated cells. Because previous studies have documented the expression of neuronal markers in the presence of GABA [27], we used this pharmacological condition as a positive control. Therefore, cells were exposed to taurine (10 mM) or GABA (100 µM) for 7 days, and the immunocytochemical analyses were performed against DCX, a standard marker to identify the neuronal identity of differentiated cells [28]. Compared with control cells (Fig. 1A), the cultures exposed to taurine or GABA (Fig. 1B, C, respectively) exhibited increased immunopositivity to DCX (one-way ANOVA; P < 0.05), with no statistical difference in DCX expression between the treated groups (Fig. 1E). Moreover, the cells exposed to taurine exhibited a clear morphological difference compared with control cells: small and elongated or multipolar somata with cytoplasmic outgrowths or neurites with a neuronal-like morphology (Fig. 1B; right panel). Although cells exposed to GABA also exhibited neurites, these structures were less conspicuous than those observed with taurine (Fig. 1C; right panel). The dose-response relationship showed a significant increase in the rate of DCX + cells exposed to taurine at 10 mM (Fig. 1D). These results suggest that taurine stimulates the expression of neuronal markers and has stronger induction properties than GABA to generate neurites in disaggregated NPC-SVZ.
Fig. 1.
Immunolabeling for neuronal markers in disaggregated NPC-SVZ Immunopositivity of disaggregated cells to DCX was observed consistently in A control cells, B cells treated with 10 mM taurine, and C cells treated with 100 µM GABA (positive control). The nuclei were stained with Hoescht (blue labeling). Right panels are magnifications obtained from the white boxes in the preceding panels showing the neurite outgrowths in the three experimental conditions. D Dose-response relationship with taurine, observe that 10 mM taurine was the effective concentration to generate neuron-like cells. E Boxplot graph showing a normalized count of DCX + cells in each experimental condition. Treatment with taurine or GABA significantly increases the number of DCX + cells. *P < 0.05
Taurine Induces neuronal-like Characteristics in NPC-SVZ
Because the NPC-SVZ can differentiate to both glial and neuronal cells [29], we next determined if taurine or GABA influences the differentiation of the disaggregated NPC-SVZ to glia. Compared with control cells (Fig. 2B), treatment with taurine (10 mM) or GABA (100 µM; Fig. 2B, C) promoted somatic elongation and neurite outgrowth that conferred a neuronal-like morphology of the treated cells. However, this morphology was restricted to DCX + cells (middle panels in Fig. 2B, C); more importantly, it was absent in cells immunopositive to GFAP (right panels in Fig. 2A–C; see also Suppl. Figure 3). The DCX + cells with neurite outgrowths exhibited rounded somata, had a length of 52.07 ± 4.8 μm, and a nuclei size of 12.18 ± 1.05 μm. By contrast, the GFAP + cells exhibited several somatic shapes, including elongated, stellate, or round morphology with abundant cytoplasm (133.3 ± 9,21 μm) and elongated nuclei (14.05 ± 0.8 μm) (right panels in Fig. 2A and Suppl. Fig. 3).
Fig. 2.
Neurite outgrowth is restricted to DCX + and absent in GFAP + cells. A–C Left panels, disaggregated NPC-SVZ showing immunopositivity to DCX. Middle panels: corresponding magnified microphotographs from the left panels. Treatment with taurine or GABA stimulated the formation of neurites. Right panels: cells positive to GFAP lacked neurite outgrowths. D Bar graph showing the average number of neurite outgrowths measured from somata. E Bar graph showing the average length of primary neurites in the three experimental conditions
A morphometric analysis of DCX + cells revealed differences in the differentiation process in each experimental condition. For instance, cells treated with taurine or GABA exhibited numerous secondary and tertiary neurites compared with control cells. The number of primary, secondary, and tertiary neurites in each experimental condition is summarized in the bar graph in Fig. 2D (one-way ANOVA; P > 0.05). Regarding the length of primary neurites, we did not find a statistical difference in the three experimental conditions (Fig. 2E).
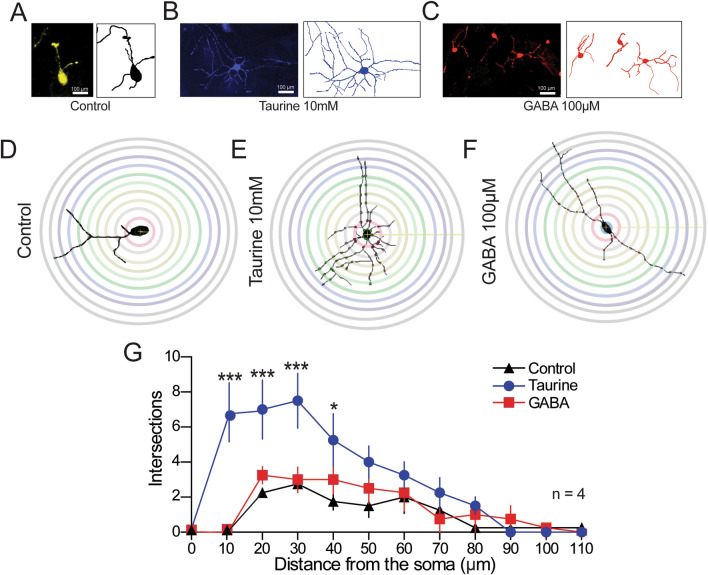
We performed a Sholl analysis to achieve more accurate morphometry of the differentiated cells. Representative microphotographs of cells from the three experimental conditions and their corresponding digital reconstructions are depicted in Fig. 3A–C. Concentric rings superimposed on the digital reconstructions for the Sholl analysis are depicted in Fig. 3D–F. Control cells exhibited unipolar morphology with fewer neurites (Fig. 3A, D). In sharp contrast, cells treated with taurine exhibited multipolar morphology and a robust number of neurites and arborizations (Fig. 3B, E). Furthermore, cells treated with GABA exhibited a bipolar-like morphology, with a reduced number of neurites and no arborizations compared with those observed in taurine-treated cells. (Fig. 3 C, F). The number of neurite intersections is plotted against neurite length from the soma, thus summarizing neuronal complexity, in Fig. 3G. As expected, the number of neurite intersections was larger in cells treated with taurine than those expressed in control cells or from cells treated with GABA. These morphometric analyses strongly suggest that taurine acts as a strong promoter of neurite complexity and contributes to the neuronal maturation of differentiated NPC.
Fig. 3.
Representation of the Sholl analysis. A–C Microphotographs of representative NPC-SVZ in the three experimental conditions showing the typical neurite outgrowth and their corresponding digital reconstructions. D–F Schematic representation of the morphometric Sholl analysis performed on control, taurine- and GABA-treated cells. G The number of intersections between neurite outgrows and concentric rings (10 μm increments) from the center of the soma were used to determine primary, secondary, and tertiary ramifications (n = 5). *P < 0.05; ***P < 0.001
Blockade of GABAAR Suppress the Morphogenic Process Induced by Taurine
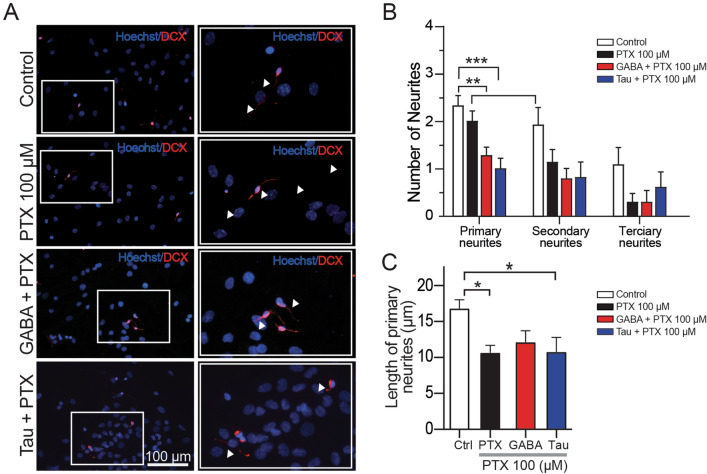
Previous studies demonstrated that GABAAR participates in the morphogenic process of NPC-SVZ [5, 30]. To investigate if GABAAR are involved in the morphogenic process mediated by taurine, disaggregated NPC-SVZ were differentiated in increasing concentrations of the GABAAR antagonist, PTX (5, 50, and 100 µM, respectively). We first corroborated that preincubation with the increasing doses of PTX did not induce cell damage. For this, a cell counting kit-8 assay determined that none of the PTX concentrations caused significant cell damage (data not shown). These observations also showed that 100 µM PTX caused a considerable reduction of neurites in cells exposed to taurine or GABA. Therefore, the number and length of neurites were analyzed in DCX + cells exposed to 100 µM PTX. Differentiated cells were preincubated with 100 µM PTX for 1 h. Under these conditions, there was a significant decrease in the number of primary neurites in DCX + cells treated with either GABA plus PTX (one-way ANOVA; P < 0.01; middle panel, Fig. 4A and red bars in B) or taurine plus PTX (one-way ANOVA; P < 0.001; bottom panel, Fig. 4A and blue bars in B). We also determined that blockade of GABAAR with 100 µM PTX reduced the number of secondary and tertiary neurites (one-way ANOVA; P > 0.05; Fig. 6B).
Fig. 4.
Effect of PTX on the differentiation and maturation process. A Left panels, disaggregated NPC-SVZ immunopositive to DCX; the cells were incubated with 100 µM PTX. Right panels, corresponding magnifications showing the reduced number of neurites in the presence of PTX. Arrowheads indicate neurite outgrowth. B Bar graph showing the average number of primary, secondary, and tertiary neurite outgrowths measured from the somata. C Bar graph contrasting the average length of primary neurites in control and the three experimental conditions. *P < 0.05; **P < 0.01; ***P < 0.001
Fig. 6.
Kinetic properties of NCP-SVZ spikes. A Superimposed spikes from control, GABA-, and taurine-treated cells. The green trace corresponds to an actual AP from a hippocampal interneuron recorded from an acute hippocampal slice. Note the control spike’s slow onset, reduced overshoot, and lack of AHP. In contrast, GABA and taurine treatment increased the spike’s onset, overshoot, and repolarization. B1 Representative examples of the first and third derivatives (grey and black trace, respectively) of an actual AP or spike. The Vth for spike initiation was determined by the third derivative changes from negative to positive. B2 Box plots and individual experiments showing Vth for each experimental group, including the values obtained from an actual AP (green box and symbols). GABA and taurine treatment shifted Vth to a more hyperpolarized membrane potential. C) Example of an actual AP (from a hippocampal interneuron) and the resulting phase plot indicating the kinetic parameters measured. MDS = maximum depolarization slope; MRS = maximum repolarization slope; h-w = spike half-width. D Superimposed phase plots from control, GABA-, and taurine-treated cells and an actual AP. Box plots and individual experiments (symbols) summarizing the kinetic parameters obtained from spikes in each experimental condition. GABA and taurine treatment: E increased spike amplitude; F decreased spike h-w, G increased spike MDS, and H decreased MRS. *P < 0.05; **P < 0.01; ***P < 0.01; n = 7 disaggregated cells for each experimental condition
Likewise, the length of primary neurites exhibited a significant decrease in the presence of PTX alone or combined with taurine or GABA (one-way ANOVA; P < 0.05 for all comparisons; Fig. 6C). These experiments demonstrate for the first time that activation of GABAAR is a necessary step in the taurine-mediated morphogenic process and neurite outgrowth of NPC-SVZ.
Taurine or GABA Treatment Induces Electrophysiological Responses in Disaggregated NPC
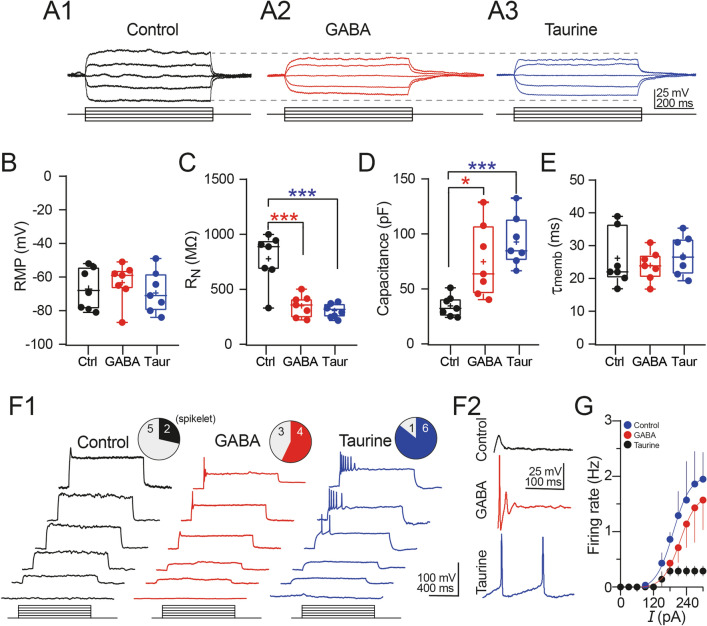
Because of our previous finding that cells differentiated with taurine or GABA express neuronal markers and exhibit a neuronal-like morphology, we next determined the passive and active electrophysiological properties of the NPC-SVZ with whole-cell patch-clamp recordings. We measured membrane properties from 21 cells divided into control, taurine- and GABA-treated cells (n = 7 for each group). Figure 5A1–3 show representative voltage responses to current injections obtained under the three experimental conditions. Compared with control cells, the decreased voltage deflections observed in GABA- or taurine-treated cells (see the continuous horizontal dash lines) indicate increased functionality of ion channels active near the RMP, possibly voltage-dependent and inward-rectifying potassium channels. From the current-voltage relationships, we determined that neither GABA nor taurine altered the RMP compared with control cells (RMP in: control cells = -67.1 ± 4.7 mV; GABA-treated cells = –63.3 ± 4.4 mV; taurine treated cells = –69.4 ± 4.6 mV; one-way ANOVA; F(2,18) = 0.456; P = 0.641; Fig. 5B). Consistent with the decreased voltage deflections as a result of increased ion channel activity near the RMP, we observed a marked decrease in the somatic RN of NPC-SVZ treated with GABA or taurine compared with control cells (RN in: control cells = 777.7 ± 87.66 MΩ; GABA-treated cells = 350.1 ± 38.2 MΩ [Tukey’s test; P < 0.001]; taurine-treated cells = 307.3 ± 22.55 MΩ [Tukey’s test; P < 0.001]; one-way ANOVA; F(2,18) = 21.040; P < 0.001; Fig. 5C). The changes in RN were accompanied with an increase in the cell’s Cm (Cm in: control cells = 34.6 ± 6.7 pF; GABA-treated cells = 75.9 ± 12.6 pF [Tukey’s test; P = 0.012]; taurine-treated cells = 92.7 ± 8.7 pF [Tukey’s test; P < 0.001]; one-way ANOVA; F(2,18) = 10.860; P < 0.001; Fig. 5D). Furthermore, the Cmwas higher in cells treated with taurine than those treated with GABA, consistent with the cell’s volume regulation being mediated by taurine [31, 32]. Lastly, we did not find differences in tmemb between the three experimental conditions (τmemb in: control cells = 25.7 ± 3.21 ms; GABA-treated cells = 23.9 ± 1.74 ms; taurine-treated cells = 27.8 ± 2.25 ms; one-way ANOVA; F(2,18) = 0.616; P = 0.550; Fig. 5E).
Fig. 5.
Electrophysiological properties of disaggregated NPC-SVZ treated with taurine or GABA. A1–3 Representative current-voltage responses elicited at − 60 mV from control, GABA, and taurine-treated cells. Note the decreased voltage response of cells treated with GABA or taurine (continuous dashed lines). Box plots and individual experiments (circles) showing: B RMP, C somatic RN, D somatic Cm, and E tmemb of control, GABA-, and taurine-treated cells. The solid line in the boxplots represents the median and the cross represents the mean. F1 Representative voltage responses elicited at -60 mV. Increasing depolarization triggered a ‘spikelet’ in control cells (left panels); in GABA- (middle panel), and taurine-treated cells (right panel), a brief burst of spikes were observed. The upper pie charts show the proportion of cells capable of eliciting a spike in response to depolarizing current injection. F2 Magnification of representative spikes obtained in the three experimental conditions. GABA- and taurine-treated cells exhibited a well-defined AHP conductance following individual AP. G Plot of the averaged maximum spike firing frequency response to depolarizing current steps for each experimental condition. *P < 0.05; ***P < 0.01 or higher statistical significance; n = 7 disaggregated cells for each experimental condition
Next, we explored the active membrane properties of disaggregated NPC-SVZ. Increasing current steps were injected to test if a sustained membrane depolarization triggers AP. Control cells did not elicit an AP, as illustrated in Fig. 6F1 (left panels), and only two control cells exhibited a short, voltage-sensitive ‘spikelet’ (upper panel, Fig. 6F1) upon sustained depolarization. On the other hand, cells treated with GABA exhibited an increased proportion of cells that elicited at least one fast spike that resembled an actual AP (middle panel, Fig. 6F1). Contrary to these observations, repetitive AP discharge was observed in the taurine-treated cells (right panel, Fig. 6F1). The number of cells that exhibited AP-like waveforms (solid fill) versus those with no response (grey fill) in the three experimental conditions is summarized in the upper pie charts of Fig. 6F1. The proportion of: control cells eliciting a spikelet = 28.57%; GABA-treated cells eliciting at least one spike = 57.14%; taurine-treated cells eliciting at least one spike = 85.71%).Interestingly, the AP observed in the taurine-treated cells was followed by an afterhyperpolarization (AHP) that resembled the Ca2+-dependent slow AHP that follows the AP of central neurons [33]. The firing pattern observed in the three experimental conditions and the resulting AHP is depicted in Fig. 6F2. We also expressed the repetitive firing in terms of AP frequency. Figure 6G summarizes the AP frequency as a function of the injected current of control (black symbols), GABA- (red symbols), and taurine-treated cells (blue symbols). Together, these results support the hypothesis that taurine is a potent inductor of phenotypic differentiation of NPC maintained in vitro and show that taurine is more effective than GABA in transforming NPC-SVZ to a neuron-like phenotype with electrophysiological properties resembling those of central neurons.
Action Potential Properties of the Differentiated NPC-SVZ
The voltage traces in Fig. 6F2 show that incubation with GABA or taurine reshapes the spike waveform. This effect is more evident in the superimposed, magnified spikes in Fig. 6A. As previously stated, the control ’spikelet’ was smaller and slower, with an amplitude that barely exceeded the AP overshoot, lacked an AHP, and required strong depolarization for initiation. On the other hand, GABA- and taurine-treated cells exhibited fast and regenerative spikes with prominent overshoots followed by an AHP conductance (Fig. 6A). Given these findings, we next analyzed the kinetic properties of the spikes and compared the resulting parameters with averaged values obtained from actual AP spikes from hippocampal interneurons. First, we determined the voltage membrane threshold (Vth) for spike initiation by computing the third derivative of the spike, a mathematical method that identifies an actual AP threshold [34] (Fig. 6B1). Compared with control cells, we found that both GABA and taurine treatment shifts the spike Vth to a more hyperpolarized membrane potential, closer to the Vth of an actual AP (spike Vth in: control cells = −21.1 ± 2.00 mV; GABA-treated cells = −32.5 ± 2.50 mV [Tukey’s test; P = 0.022]; taurine-treated cells = −35.2 ± 1.44 mV [Tukey’s test; P = 0.005]; one-way ANOVA; F(2,9) = 9.352; P = 0.006; actual AP Vth = −42.33 ± 1.54 mV [Tukey’s test; P < 0.001]; one-way ANOVA; F(3,14) = 15.840; P < 0.001; Fig. 6B2).
Next, we analyzed the kinetic parameters of the spikes utilizing a phase plot analysis. The left panel in Fig. 6C shows an actual AP spike from a hippocampal interneuron, and in the right panel, its resulting geometrical representation or phase plot; the superimposed phase plots from the three experimental conditions plus an actual AP (green trace) are superimposed in Fig. 6D.
Compared with control cells, GABA and taurine treatment increased the spike amplitude (spike amplitude in: control cells = 27.1 ± 2.55 mV; GABA-treated cells = 66.9 ± 6.83 mV [Tukey’s test; P = 0.021]; taurine-treated cells = 81.9 ± 3.58 mV [Tukey’s test; P = 0.001]; one-way ANOVA; F(2,9) = 14.330; P = 0.002; Fig. 6E). NPC-SVZ treated with taurine exhibited an amplitude closer to the actual AP amplitude (actual AP amplitude = 90.97 ± 2.12 mV [Tukey’s test; P < 0.001]; one-way ANOVA; F(3,14) = 20.940; P < 0.001; Fig. 6E). Consistent with this finding, the spike half-width (h-w) was shorter in the GABA- and taurine-treated cells and in the latter group was closer to the actual AP h-w (spike h-w in: control cells = 3.4 ± 0.70 ms; GABA-treated cells = 2.9 ± 0.20 ms [Tukey’s test; P = 0.379]; taurine-treated cells = 2.0 ± 0.09 ms [Tukey’s test; P = 0.009]); one-way ANOVA; F(2,9) = 9.198; P = 0.007; actual AP h-w = 1.7 ± 0.14 [Tukey’s test; P < 0.001]; one-way ANOVA; F(3, 14) = 12.910; P < 0.001; Fig. 6F). Likewise, the maximum depolarization slope (MDS) of the spike, a parameter that reflects activity of the AP’s voltage-dependent Na+ conductance, was increased in GABA- and taurine-treated cells (MDS in: control cells = 18.7 ± 1.80 mV/ms; GABA-treated cells = 32.8 ± 4.42 mV/ms [Tukey’s test; P = 0.049]; taurine-treated cells = 58.1 ± 2.57 mV/ms [Tukey’s test; P < 0.001]; one-way ANOVA; F(2,9) = 32.260; P < 0.001; actual AP MDS = 88.7 ± 2.88 mV/ms [Tukey’s test; P < 0.001]; one-way ANOVA; F(3, 14) = 79.130; P < 0.001; Fig. 6G). Lastly, we also computed the maximum repolarization slope (MRS) as an indirect measure of voltage-dependent K+ conductances underlying the AP repolarization phase. Compared with control cells, GABA and taurine treatment increased the speed of the MRS (MRS in: control cells = −22.7 ± 2.50 mV/ms; GABA-treated cells = −47.7 ± 4.32 mV/ms [Tukey’s test; P = 0.011]; taurine-treated cells = −79.5 ± 3.08 mV/ms [Tukey’s test; P < 0.001]; one-way ANOVA; F(2,9) = 48.870; P < 0.001; actual AP MRS = −84.7 ± 3.56 mV/ms [Tukey’s test; P < 0.001]; one-way ANOVA; F(3, 14) = 42.630; P < 0.001; Fig. 6H). Together, these data demonstrate that taurine, like GABA, induces phenotypic plasticity and modulates electrophysiological properties of disaggregated NPC-SVZ. Moreover, our data strongly suggest that taurine has a stronger effect compared with GABA in the differentiation process of NPCs.
Discussion
In this work, we have provided experimental evidence regarding the multipotentiality of NPC-SVZ to generate cells that express neuronal markers and develop electrophysiological properties following treatment with the non-essential amino acid taurine. In line with this finding, we demonstrated the critical role of the ionotropic GABAAR in the differentiation process mediated by taurine. The interaction between the GABAAR and taurine promotes a neuron-like phenotype of NPC-SVZ, increases their dendritic complexity, and favors an electrophysiological behavior that resembles actual neuron activity, including the onset of repetitive firing.
We first confirmed that NPC-SVZ matured in neurospheres expressing nestin and SOX-2 markers, as reported by Sanchez-Mendoza (2016) [35]. Moreover, disaggregation of neurospheres to monolayer cultures treated with taurine increased the percentage of cells expressing the neuronal marker DCX, an observation previously reported for GABA [27] and confirmed in this study.
Ramos-Mandujano (2014) [36] proposed that taurine promotes mitochondrial efficiency and increases ATP availability, an immediate effect that increases neuronal proliferation. In this work we offered data demonstrating both the critical role that the GABAAR has in the differentiation process of NPC-SVZ, and that blockade of this receptor suppresses differentiation. In agreement with our findings, the participation of GABAAR in the neurogenic process has been previously observed in cortical precursor cells, in which activation of the GABAAR triggers a sustained depolarization in postnatal and adult precursor cells, promoting axon elongation, neurite growth, and synaptogenesis [37–39]. Therefore, one possible way to explain the role of GABAAR in the taurine-mediated NPC-SVZ differentiation process is that upon their activation, a GABAAR-mediated sustained depolarization favors cytosolic calcium accumulation and activates Ca2+-dependent mechanisms associated with neurite formation [40]. However, additional experiments are required to confirm this. As previously stated, the critical role of GABAAR in the neuronal differentiation of NPC-SVZ was demonstrated when disaggregated NPC-SVZ were simultaneously exposed to taurine and the GABAAR blocker PTX. Blockade of GABAAR decreased the number of cells immunopositive to DCX; furthermore, these cells exhibited fewer secondary and tertiary neurons. These observations strongly indicate that taurine modulates the differentiation processes of NPC-SVZ to neuronal cells via activation of GABAAR. In this sense, it is important to emphasize that modulation of GABAAR by taurine may promote other neurogenic regulatory mechanisms, including the decreased activity of glial cells [40]; activation of the sonic hedgehog signaling pathway [41]; increased mitochondrial activity [35, 42], or activation of GABABR.
Our Sholl analyses confirmed that taurine has a stronger differentiation effect than GABA. This observation was confirmed with the morphometric analyses that verified the more complex neurite branching and increased formation of secondary and tertiary neurites in taurine- versus GABA-treated cells. Thus, it can be assumed that taurine participates in the maturation and incorporation of NPC-SVZ to perform neuronal circuits by promoting axonal differentiation, dendrite arborization, and the formation of synaptic connections [37–39].
Another remarkable observation of this study is the emergence of defined electrophysiological properties of disaggregated NPC-SVZ exposed to taurine or GABA. Compared with control cells that barely exhibited neurite outgrowth, cells exposed to GABA, and more importantly taurine, exhibited changes in their passive and active membrane properties. Taurine caused a dramatic drop in the RN and increased the activity of membrane conductances active near the RMP. Moreover, the proportion of cells generating repetitive APs was increased. In line with this observation, taurine-treated cells exhibited a well-defined AHP phase that resembles the Ca2+-activated K+ conductances that underlie and shape the AP output of mature neurons. Taurine caused NPC-SVZ cells to behave as an actual AP of a mature neuron by altering several kinetic parameters, including AP threshold, spike amplitude, and h-w, as well as AP depolarization and repolarization rates. More importantly, these effects were more marked in cells treated with taurine than GABA, supporting the hypothesis that taurine represents a stronger morphogen than GABA.
Conclusion
We have provided in vitro evidence regarding the role of taurine as a key neurogenic factor in the differentiation process of NPC-SVZ. We have demonstrated that taurine induces functional neurons by a mechanism that requires the activation of GABAAR. The morphological and electrophysiological changes mediated by taurine support the hypothesis that this non-essential sulfur-containing amino acid has functional implications in the neurogenesis process, including gene transcription, neural protein expression, and transport system activity. Given the non-active biochemical nature of taurine, our observations may provide a novel pharmacological tool to renew, increase, or maintain the pools of stem cells that participate in the neurogenesis process during or after a pathological condition, or for the treatment of the age-related reduction in adult neurogenesis.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file1 (TIF 6966 KB)—Neurospheres obtained from SVZ from a mouse brain. Representative bright-field microphotographs of A) primary, B) secondary, and C) tertiary neurospheres. Microphotographs in the left panels were taken with a 10x magnification lens; right panel, 20x. Neurospheres were formed at 7, 12, and 17 days, respectively
Supplementary file2 (TIF 7172 KB)—Expression of NPC markers in NPC- neurospheres.Representative immunofluorescence microphotographs of secondary neurospheres before differentiation. The nuclei were stained with Hoescht (blue signal). Neurospheres were immunopositive to A) nestin and B) SOX-2. C) Neurospheres did not exhibit immunopositivity for DCX. Microphotographs were taken with a 20x magnification lens. The scale bar applies to all panels
Supplementary file3 (TIF 34018 KB)—Effect of taurine and GABA on the GFAP expression of disaggregated NPC-SVZ. A–C) Representative immunofluorescence microphotographs of disaggregated NPC-SVZ after the differentiation process in the three experimental conditions showing GFAP+ cells, including their characteristic morphology: abundant cytoplasm with elongated, stellate, or round shapes. The nuclei were stained with Hoescht (blue signal). The scale bar applies to all the microphotographs. B) Bar graph showing the percentage of GFAP+ cells in each experimental condition
Acknowledgements
We thank Dr. Claudia Verónica Rivera Cerecedo head of the laboratory animal facility at the Institute of Cell Physiology, UNAM.
Author Contributions
Gutierrez-Castañeda: Investigation, Formal analysis, Writing – original draft preparation. González-Corona: Investigation. Griego: Investigation, Formal analysis, writing. Galván: Resources, writing, review, and editing. Ochoa-de la Paz: Conceptualization, Resources, Writing, Visualization, Project administration.
Funding
This work was supported by grants PAPIIT-UNAM IN221820 and Presupuesto Interno Facultad de Medicina (Id 49), Departamento de Investigación, Asociación para Evitar la Ceguera en México IAP Hospital “Dr. Luis Sánchez Bulnes”, Consejo Nacional de Ciencia y Tecnología, México grants CB-2016–281617 to EJG, fellowships 783563 (NEGC), 727269 (EG).
Data Availability
The datasets generated during and/or analyzed during the current study are available under request.
Declarations
Conflict of interest
The authors declare no competing financial interests. All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest or non-financial interest in the subject matter or materials discussed in this manuscript.
Ethical Approval
All the animals were handled following the guidelines of the National Institutes of Health Guide for Care and Use of Laboratory Animals and with the approval of the Institutional Animal Care and Use Committee of the National University of Mexico (protocol # 068–2019/018-CIC-2019). The study was not pre-registered.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Ming GL, Song H. Adult neurogenesis in the mammalian central nervous system. Annu Rev Neurosci. 2005;28:223–250. doi: 10.1146/annurev.neuro.28.051804.101459. [DOI] [PubMed] [Google Scholar]
- 2.Altman J, Das GD. Autoradiographic and histological evidence of postnatal hippocampal neurogenesis in rats. J Comp Neurol. 1965;124(3):319–335. doi: 10.1002/cne.901240303. [DOI] [PubMed] [Google Scholar]
- 3.Obernier K, et al. Adult neurogenesis is sustained by symmetric self-renewal and Differentiation. Cell Stem Cell. 2018;22(2):221–234 e228. doi: 10.1016/j.stem.2018.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Alvarez-Buylla A, Lim DA. For the long run: maintaining germinal niches in the adult brain. Neuron. 2004;41(5):683–686. doi: 10.1016/s0896-6273(04)00111-4. [DOI] [PubMed] [Google Scholar]
- 5.Duveau V, et al. Spatiotemporal specificity of GABAA receptor-mediated regulation of adult hippocampal neurogenesis. Eur J Neurosci. 2011;34(3):362–373. doi: 10.1111/j.1460-9568.2011.07782.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Felice D, et al. Blockade of the GABA(B) receptor increases neurogenesis in the ventral but not dorsal adult hippocampus: relevance to antidepressant action. Neuropharmacology. 2012;63(8):1380–1388. doi: 10.1016/j.neuropharm.2012.06.066. [DOI] [PubMed] [Google Scholar]
- 7.Gage FH. Mammalian neural stem cells. Science. 2000;287(5457):1433–1438. doi: 10.1126/science.287.5457.1433. [DOI] [PubMed] [Google Scholar]
- 8.Khozhai LI. Immunohistochemical detection of GABA and α1-Subunit of GABAA-Receptor in cells of the Subventricular Zone of the rat brain in the neonatal period. Cell Tiss Biol. 2022;16:38–44. doi: 10.1134/s1990519x22010060. [DOI] [Google Scholar]
- 9.Salazar P, et al. GABA effects during neuronal differentiation of stem cells. Neurochem Res. 2008;33(8):1546–1557. doi: 10.1007/s11064-008-9642-8. [DOI] [PubMed] [Google Scholar]
- 10.Stewart RR, et al. Neural progenitor cells of the neonatal rat anterior subventricular zone express functional GABA(A) receptors. J Neurobiol. 2002;50(4):305–322. doi: 10.1002/neu.10038. [DOI] [PubMed] [Google Scholar]
- 11.Wang DD, et al. GABA depolarizes neuronal progenitors of the postnatal subventricular zone via GABAA receptor activation. J Physiol. 2003;550(Pt 3):785–800. doi: 10.1113/jphysiol.2003.042572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bolteus AJ, Bordey A. GABA release and uptake regulate neuronal precursor migration in the postnatal subventricular zone. J Neurosci. 2004;24(35):7623–7631. doi: 10.1523/JNEUROSCI.1999-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Liu X, et al. Nonsynaptic GABA signaling in postnatal subventricular zone controls proliferation of GFAP-expressing progenitors. Nat Neurosci. 2005;8(9):1179–1187. doi: 10.1038/nn1522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Fernando RN, et al. Cell cycle restriction by histone H2AX limits proliferation of adult neural stem cells. Proc Natl Acad Sci U S A. 2011;108(14):5837–5842. doi: 10.1073/pnas.1014993108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.El Idrissi A, Trenkner E. Taurine as a modulator of excitatory and inhibitory neurotransmission. Neurochem Res. 2004;29(1):189–197. doi: 10.1023/b:nere.0000010448.17740.6e. [DOI] [PubMed] [Google Scholar]
- 16.Jia F, et al. Taurine is a potent activator of extrasynaptic GABA(A) receptors in the thalamus. J Neurosci. 2008;28(1):106–115. doi: 10.1523/JNEUROSCI.3996-07.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Huxtable RJ. Taurine in the central nervous system and the mammalian actions of taurine. Prog Neurobiol. 1989;32(6):471–533. doi: 10.1016/0301-0082(89)90019-1. [DOI] [PubMed] [Google Scholar]
- 18.Ripps H, Shen W. "Review: taurine: a “very essential” amino acid. Mol Vis. 2012;18:2673–2686. [PMC free article] [PubMed] [Google Scholar]
- 19.Jacobsen JG, Smith LH. Biochemistry and physiology of taurine and taurine derivatives. Physiol Rev. 1968;48(2):424–511. doi: 10.1152/physrev.1968.48.2.424. [DOI] [PubMed] [Google Scholar]
- 20.Vitvitsky V, et al. Taurine biosynthesis by neurons and astrocytes. J Biol Chem. 2011;286(37):32002–32010. doi: 10.1074/jbc.M111.253344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hernandez-Benitez R, et al. Taurine enhances the growth of neural precursors derived from fetal human brain and promotes neuronal specification. Dev Neurosci. 2013;35(1):40–49. doi: 10.1159/000346900. [DOI] [PubMed] [Google Scholar]
- 22.O’Neill KM, et al. Assessing effects on dendritic arborization using novel Sholl analyses. Front Cell Neurosci. 2015;9:285. doi: 10.3389/fncel.2015.00285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Griego E, Galvan EJ. Metabotropic glutamate receptors at the aged mossy fiber - CA3 Synapse of the Hippocampus. Neuroscience. 2021;456:95–105. doi: 10.1016/j.neuroscience.2019.12.016. [DOI] [PubMed] [Google Scholar]
- 24.Griego E, et al. Functional expression of TrkB receptors on interneurones and pyramidal cells of area CA3 of the rat Hippocampus. Neuropharmacology. 2021;182:108379. doi: 10.1016/j.neuropharm.2020.108379. [DOI] [PubMed] [Google Scholar]
- 25.Herrera-Lopez G, Galvan EJ. Modulation of hippocampal excitability via the hydroxycarboxylic acid receptor 1. Hippocampus. 2018;28(8):557–567. doi: 10.1002/hipo.22958. [DOI] [PubMed] [Google Scholar]
- 26.da Silva Siqueira L, et al. Neurospheres: a potential in vitro model for the study of central nervous system disorders. Mol Biol Rep. 2021;48(4):3649–3663. doi: 10.1007/s11033-021-06301-4. [DOI] [PubMed] [Google Scholar]
- 27.Brooke CJ, Shane TH. Expression of GABAergic and glutamatergic phenotypic markers in hypothalamic proopiomelanocortin neurons. J Comp Neurol. 2012;520(17):3863–3876. doi: 10.1002/cne.23127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Walker TL, et al. "The doublecortin-expressing population in the developing and adult brain contains multipotential precursors in addition to neuronal-lineage cells. J Neurosci. 2007;27(14):3734–3742. doi: 10.1523/JNEUROSCI.5060-06.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Gil-Perotin S, et al. Adult neural stem cells from the subventricular zone: a review of the neurosphere assay. Anat Rec (Hoboken) 2013;296(9):1435–1452. doi: 10.1002/ar.22746. [DOI] [PubMed] [Google Scholar]
- 30.Pallotto M, Deprez F. Regulation of adult neurogenesis by GABAergic transmission: signaling beyond GABAA-receptors. Front Cell Neurosci. 2014;8:166. doi: 10.3389/fncel.2014.00166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Morán J, Maar T, Pasantes-Morales H. Cell volume regulation in taurine deficient cultured astrocytes. Adv Exp Med Biol. 1994;359:361–7. doi: 10.1007/978-1-4899-1471-2_37. [DOI] [PubMed] [Google Scholar]
- 32.Netti V, et al. Release of taurine and glutamate contributes to cell volume regulation in human retinal Müller cells: differences in modulation by calcium. J Neurophysiol. 2018;120(3):973–984. doi: 10.1152/jn.00725.2017. [DOI] [PubMed] [Google Scholar]
- 33.Garduño J, Galván E, Fernández de Sevilla D, Buño W. 1-Ethyl-2-benzimidazolinone (EBIO) suppresses epileptiform activity in in vitro hippocampus. Neuropharmacology. 2005;49(3):376–388. doi: 10.1016/j.neuropharm.2005.03.021. [DOI] [PubMed] [Google Scholar]
- 34.Henze DA, et al. The multifarious hippocampal mossy fiber pathway: a review. Neuroscience. 2000;98(3):407–427. doi: 10.1016/s0306-4522(00)00146-9. [DOI] [PubMed] [Google Scholar]
- 35.Sanchez-Mendoza EH, et al. Characterization of seeding conditions for studies on differentiation patterns of subventricular zone derived neurospheres. Front Cell Neurosci. 2016;10:55. doi: 10.3389/fncel.2016.00055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ramos-Mandujano G, et al. Multiple mechanisms mediate the taurine-induced proliferation of neural stem/progenitor cells from the subventricular zone of the adult mouse. Stem Cell Res. 2014;12(3):690–702. doi: 10.1016/j.scr.2014.02.009. [DOI] [PubMed] [Google Scholar]
- 37.Sernagor E, et al. GABAergic control of neurite outgrowth and remodeling during development and adult neurogenesis: general rules and differences in diverse systems. Front Cell Neurosci. 2010;4:11. doi: 10.3389/fncel.2010.00011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Maric D, et al. "GABA expression dominates neuronal lineage progression in the embryonic rat neocortex and facilitates neurite outgrowth via GABA(A) autoreceptor/Cl- channels. ” J Neurosci. 2001;21(7):2343–2360. doi: 10.1523/JNEUROSCI.21-07-02343.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kilb W, et al. Role of tonic GABAergic currents during pre- and early postnatal rodent development. Front Neural Circuits. 2013;7:139. doi: 10.3389/fncir.2013.00139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ageta H, et al. Layer-specific sulfatide localization in rat hippocampus middle molecular layer is revealed by nanoparticle-assisted laser desorption/ionization imaging mass spectrometry. Med Mol Morphol. 2009;42(1):16–23. doi: 10.1007/s00795-008-0427-6. [DOI] [PubMed] [Google Scholar]
- 41.Huang X, et al. Taurine enhances mouse cochlear neural stem cell transplantation via the cochlear lateral wall for replacement of degenerated spiral ganglion neurons via sonic hedgehog signaling pathway. Cell Tissue Res. 2019;378(1):49–57. doi: 10.1007/s00441-019-03018-6. [DOI] [PubMed] [Google Scholar]
- 42.Li XW, et al. The role of taurine in improving neural stem cells proliferation and differentiation. Nutr Neurosci. 2017;20(7):409–415. doi: 10.1080/1028415X.2016.1152004. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary file1 (TIF 6966 KB)—Neurospheres obtained from SVZ from a mouse brain. Representative bright-field microphotographs of A) primary, B) secondary, and C) tertiary neurospheres. Microphotographs in the left panels were taken with a 10x magnification lens; right panel, 20x. Neurospheres were formed at 7, 12, and 17 days, respectively
Supplementary file2 (TIF 7172 KB)—Expression of NPC markers in NPC- neurospheres.Representative immunofluorescence microphotographs of secondary neurospheres before differentiation. The nuclei were stained with Hoescht (blue signal). Neurospheres were immunopositive to A) nestin and B) SOX-2. C) Neurospheres did not exhibit immunopositivity for DCX. Microphotographs were taken with a 20x magnification lens. The scale bar applies to all panels
Supplementary file3 (TIF 34018 KB)—Effect of taurine and GABA on the GFAP expression of disaggregated NPC-SVZ. A–C) Representative immunofluorescence microphotographs of disaggregated NPC-SVZ after the differentiation process in the three experimental conditions showing GFAP+ cells, including their characteristic morphology: abundant cytoplasm with elongated, stellate, or round shapes. The nuclei were stained with Hoescht (blue signal). The scale bar applies to all the microphotographs. B) Bar graph showing the percentage of GFAP+ cells in each experimental condition
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available under request.