Abstract
Objective:
Osteoarthritis (OA) is associated with functional limitations that can impair mobility and reduce quality of life in affected individuals. Excess body weight in OA can exacerbate impaired physical function, highlighting the importance of weight management in this population. The aim of this systematic review was to compare the effects of different dietary interventions for weight loss on physical function in overweight and obese individuals with OA.
Design:
A comprehensive search of five databases was conducted to identify relevant articles for inclusion. Studies were included that examined the effect of dietary weight loss interventions, with or without exercise, on physical function in adults with OA who were overweight or obese. Quality and risk of bias were assessed using the Quality Criteria Checklist for primary research. Primary and secondary outcomes were extracted, including change in weight and physical function which included performance-based and self-report measures.
Results:
Nineteen relevant studies were included, which incorporated lifestyle interventions (n 8), diet in combination with meal replacements (DMR; n 5) and very low-energy diets (VLED; n 6) using meal replacements only. Pooled data for eight RCT indicated a mean difference in Western Ontario and McMaster Universities Arthritis Index (WOMAC) physical function of 12·4 and 12·5 % following DMR or VLED interventions, respectively; however, no statistically significant change was detected for lifestyle interventions.
Conclusions:
Our findings suggest that partial use of meal replacements is as effective as their sole use in the more restrictive VLED. Both dietary interventions are more effective than lifestyle programmes to induce significant weight loss and improvements in physical function.
Keywords: Osteoarthritis, Overweight, Obesity, Weight loss, Physical function
Osteoarthritis (OA) is ranked as the 11th highest contributor of disability in the world, with a prevalence of 3·8 and 0·85 % of knee and hip OA, respectively(1). Disability may result from ongoing decline in physical function limiting independence in activities of daily living, particularly in older adults(2). Being overweight or obese is a major risk factor for development and progression of OA(3), including worsening of functional limitations(2). Thus begins a cyclic relationship where reduced physical function contributes to reduced activity levels, resulting in further weight gain, which can negatively affect functional status. Therefore, addressing functional impairments and weight management in this population is a significant public health issue.
Best practice guidelines for the management of overweight and obesity in the general population recommend lifestyle modifications primarily to promote moderate weight loss, including a hypoenergetic diet and increased levels of exercise(4). A very low-energy diet (VLED) is utilised in some cases, where diet and exercise strategies have not been successful for weight loss(4). VLED are defined as lowering energy intake to approximately 800 kcal or less per day, with total meal replacements commonly utilised(5). In the long term, lifestyle modifications are considered more sustainable compared with VLED because of associated costs, compliance issues and potential negative effects to body composition(4). Clinical practice guidelines in OA consistently recommend exercise therapy, including aerobic and strength training, and weight management incorporating diet and exercise, for improvements in physical function(6–8). Clinical trials in OA have shown significant improvements in physical function with dietary weight loss alone. The IDEA study by Messier et al. demonstrated significant weight loss and improvements in Western Ontario and McMaster Universities Arthritis Index (WOMAC) physical function that were of a similar magnitude in participants undergoing a dietary weight loss intervention, with or without exercise(9).
Overall, the body of evidence regarding the effect of weight loss on symptoms of OA is growing rapidly. Clinical trials in OA have investigated the effects of both lifestyle interventions (diet and exercise) and VLED. Previous systematic reviews have demonstrated beneficial effects of weight loss on symptoms of OA, including physical function(10–12). However, preliminary searches determined that these reviews did not include all relevant dietary weight loss studies, in particular VLED interventions. As such, it is unclear as to which of these dietary strategies is the most effective to induce significant weight loss and improvement in symptoms, namely physical function, in this population. Therefore, the aim of this systematic review is to compare the effects of different dietary interventions for weight loss, with or without exercise, on physical function in overweight and obese adults with OA.
Methods
The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines(13) were utilised, and the systematic review protocol was registered in Prospero: International prospective register of systematic reviews and can be accessed here: http://bit.ly/2yVZCg1.
Inclusion criteria
Included studies were randomised controlled trials (RCT) and uncontrolled clinical trials investigating the effect of dietary weight loss interventions, with or without exercise, on physical function in adults classified as overweight and obese (aged 18 years and over) with OA. To assess the effects of the intervention, primary outcomes included body weight and physical function as either self-reported or performance-based measures. Studies were restricted to the English language, with no date restrictions.
Search strategy
A preliminary search of the Medline database was undertaken to identify keywords and index terms, by analysis of words in the title, abstract, description of articles and Medical Subject Headings. Using the keywords identified in the first search, a second search was completed in Medline, Cochrane, Embase, AMED and CINAHL databases (September 2017 and updated May 2019) to identify potential articles for inclusion. The search strategy included sets of search terms to describe: (i) the population group, for example, overweight, obesity and OA; (ii) intervention components, for example, hypoenergetic diet and weight loss and (iii) outcomes, for example, weight and BMI and has been reproduced (see online supplementary material, Supplementary Table 1). This allowed for identification of dietary weight loss studies and subsequently those that measured different physical function outcomes. A hand search was conducted in which relevant journals and reference lists of retrieved articles were examined to check for additional studies not identified in the database search.
Study selection
Studies identified using the search strategy were screened independently by two reviewers (E.J.W. and S.K.B.) by examining the title, abstract and keywords. Full texts of articles that met the inclusion criteria, or any that were unclear, were retrieved for further reviewing. Full texts were then screened, and articles were classified as included or excluded, and the reasons for exclusion were recorded (i.e. participants, intervention, outcome and study design). Any discrepancies throughout the selection process were resolved by discussion with a third author (P.G.O.).
Quality appraisal
All included articles were assessed for quality and risk of bias using the Quality Criteria Checklist for primary research(14). The tool comprises a checklist of questions within ten domains that address methodological rigour. Articles were given a quality rating of positive (strong quality), negative (weak quality) or neutral (neither exceptionally strong nor exceptionally weak), based on these criteria. Two authors (E.J.W. and S.K.B.) assessed the quality of articles independently, and any discrepancies were resolved by consultation with a third author (P.G.O.).
Data extraction and synthesis
Data were extracted against a standardised data extraction template by one author (E.J.W.) and checked by another (S.K.B.) with reference to the full text of the article. Any discrepancies were resolved by discussion with a third author (P.G.O.). The following information was collected from each study: author, year of study and setting; participant details including age, sample size and retention rate; details of the intervention including treatment and control group, length of intervention and follow-up period; study design; and study results including before and after measures of outcomes of interest. Studies were categorised into three groups based on diet intervention type: (i) lifestyle – instruction to consume a hypoenergetic diet using conventional foods, (ii) conventional diet with partial use of meal replacements (DMR) and (iii) VLED – approximately ≤810 kcal/d, with sole use of meal replacements, as per global food standards(5).
Study data from RCT that were comparable in terms of outcome measures and follow-up periods were pooled to complete a meta-analysis. The mean difference was calculated for studies using the same instrument for measurement (i.e. WOMAC), and results reported as mean differences with 95 % CI and displayed as a forest plot. The effect of heterogeneity was quantified by calculating I 2 (range: 0–100 %). If I 2 was >30 %, a random effects model was used to account for heterogeneity between studies(15). Where multiple intervention groups were used, the diet or diet and exercise groups were selected for comparison against controls. Where data were not presented as mean and standard deviation, conversions were performed to obtain these measures(15). All statistical analyses were conducted using the Cochrane Collaboration, Review Manager software (RevMan version 5.3, Copenhagen).
Results
Study selection
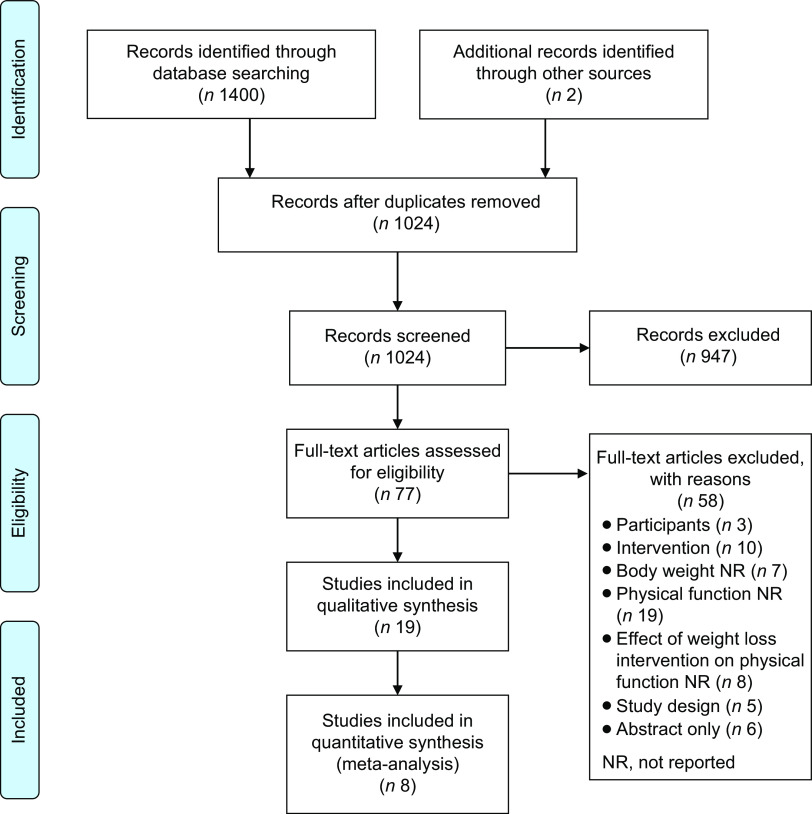
The PRISMA flow diagram (Fig. 1) provides an overview of the screening and selection process for articles to be included in the review. The initial database search identified 1024 articles after the removal of duplicates, and two articles were located via hand search. After the first screening, seventy-seven articles were retrieved for full-text review. From this, fifty-eight articles were excluded. Reasons for exclusion are provided in the PRISMA flow diagram (Fig. 1). Nineteen articles were included in the review, with eight included in the meta-analyses. Articles were categorised according to intervention type: lifestyle (n 8), conventional diet and meal replacements (DMR; n 5) and very low-energy diets (VLED; n 6).
Fig. 1.
PRISMA flow diagram. Overview of the screening and selection process for articles to be included in the review
Study quality
In terms of quality rating (Table 1), nine of the nineteen included studies received a neutral rating due to non-comparable study groups at baseline (n 1)(16) or having no comparison group (n 8)(17–24). The remaining ten studies received a positive quality rating(9,25–33), of which five were lifestyle studies(27–29,32,33), two were DMR studies(9,30) and three were VLED studies(25,26,31). These studies all specified clear inclusion criteria, had comparable study group characteristics at baseline and provided detailed information related to intervention components and outcome measures. Patient compliance was measured in eleven studies, via food diaries, exercise logs, pedometers and attendance at nutrition and exercise programmes(9,20,22–24,27–30,32,33). Due to the nature of the interventions and study design, blinding of participants was not always possible; however, outcome assessment was blinded in all studies. All studies accounted for all participants from baseline to follow-up. Seven studies did not clearly outline the study limitations or potential biases in the discussion(21,25–29,31).
Table 1.
Quality appraisal summary: a summary of the ten domains addressing scientific soundness of primary research from ADA quality criteria checklist
| Validity questions | 1. Research question | 2. Subject selection | 3. Study groups | 4. Handling withdrawals | 5. Blinding | 6. Intervention description | 7. Outcome measures | 8. Statistical analysis | 9. Conclusion | 10. Funding/sponsorship | Quality |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Study | |||||||||||
| Randomised controlled trials | |||||||||||
| Bliddal et al. (25) | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Unclear | Yes | Positive |
| Christensen et al. (26) | Yes | Yes | Yes | Yes | Unclear | Yes | Yes | Yes | Unclear | Yes | Positive |
| Hughes et al. (33) | Unclear | Yes | Yes | Yes | Unclear | Yes | Yes | Yes | Yes | Yes | Positive |
| Magrans-Courtney et al. (27) | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Unclear | Yes | Positive |
| Messier et al.(28) | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Unclear | Yes | Positive |
| Messier et al.(9) | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Positive |
| Miller et al.(30) | Yes | Yes | Yes | Yes | Unclear | Yes | Yes | Yes | Yes | Yes | Positive |
| O’Brien et al.(32) | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Positive |
| Somers et al.(16) | Yes | Yes | No | Yes | Yes | Yes | Yes | Yes | Yes | Unclear | Neutral |
| Uncontrolled clinical trials | |||||||||||
| Aaboe et al.(17) | Yes | Yes | No | Yes | N/A | Yes | Yes | Yes | Yes | Yes | Neutral |
| Atukorala et al.(18) | Yes | Yes | No | Yes | N/A | Yes | Yes | Yes | Yes | Unclear | Neutral |
| Bartels et al.(19) | Yes | Yes | No | Yes | N/A | Yes | Yes | Yes | Yes | Yes | Neutral |
| de Luis et al.(20) | Yes | Yes | No | Yes | N/A | Yes | Yes | Yes | Yes | Unclear | Neutral |
| Henriksen et al.(21) | Yes | Yes | No | Yes | N/A | Yes | Yes | Yes | Unclear | Unclear | Neutral |
| López Gómez et al.(24) | Yes | Yes | No | Yes | N/A | Yes | Yes | Unclear | Yes | Unclear | Neutral |
| Martin et al.(22) | Yes | Yes | No | Yes | N/A | Yes | Yes | Yes | Yes | Yes | Neutral |
| Messier et al.(29) | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Unclear | Yes | Positive |
| Paans et al.(23) | Yes | Yes | No | Yes | N/A | Yes | Yes | Yes | Yes | Yes | Neutral |
| Riecke et al.(31) | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Unclear | No | Positive |
Study characteristics
Of the nineteen studies included in the review, eight were lifestyle studies of which five were RCT(16,27,28,32,33), five were DMR studies of which two were RCT(9,30), and six were VLED studies of which two were RCT(25,26). The remaining ten studies were uncontrolled clinical trials(17–24,29,31). A description of study characteristics including study sample and intervention details is provided in Table 2. Overall, participant numbers ranged from 24 to 1383, with an age range of 20–90 years. For the sixteen studies that included both male and female participants, the proportion of female participants ranged from 57 to 89 %. Two lifestyle studies and one VLED study included only female participants(22,24,27). Seven studies included only obese participants (BMI ≥ 30 kg/m2)(17,19–21,24,30,31), and the average BMI of all study samples ranged from 31·7 to 40·8 kg/m2. Seventeen studies included participants who had knee OA(9,16–19,21,22,24–36); however, one VLED study included patients with hip or knee OA(20), and another lifestyle study included patients with hip OA only(23). The lifestyle interventions (n 8) provided healthy eating nutrition education, counselling, behavioural therapy and all included instruction for exercise, such as aerobic and resistance training, which were 8 weeks to 18 months in duration(16,22,23,27–29,32,33). Seven lifestyle studies provided face-to-face interventions, while O’Brien et al. used a telephone coaching programme(32). Two lifestyle studies prescribed energetic intake that ranged from 1200 to 1600 kcal/d for the intervention groups(16,27) while moderating energy intake at different stages of the intervention(27) or based on participant gender(16). Three other interventions aimed for a general reduction in energetic intake(22,28,29). The DMR studies all provided up to two meal replacements per day with meal plans for the third meal using conventional foods, and/or nutrition education and behavioural therapy, that ranged from 12 weeks to 18 months in duration(9,18,20,24,30). Two DMR studies prescribed energy intake of 1109 and 1035 kcal/d(20,24). Two other DMR studies defined a reduction in energy intake of 800–1000 kcal/d and also prescribed exercise interventions (aerobic and strength training)(9,30). The VLED studies (n 6) were the most restrictive using only meal replacements to provide a diet of <810 kcal/d. None of these studies included exercise interventions(17,19–21,24–26,31). Four VLED studies were 16 weeks(17,19,21,31), while one was 8 weeks in length(26); however, one study alternated between a VLED (810 kcal) and hypoenergetic diet (1200 kcal), over 52 weeks(25). Average prescribed energy intake was 711 kcal/d (range: 415–810 kcal/d) in the VLED treatment groups.
Table 2.
Study characteristics: description of study details, including participant and intervention information†
| Study | Participants | Intervention | Study design | BMI (kg/m2) | |
|---|---|---|---|---|---|
| Author; setting | Age (years, age range); sample size (n); retention (r) | Treatment groups (Tx); control (C); other (O) | Length of intervention (L); follow-up (F) | Baseline | Follow-up |
| Lifestyle (diet and exercise) – randomised controlled trials (RCT) | |||||
| Hughes et al.(33); USA | Age = 68 ± 6; n 413; r = 383 (93 %) | T1: Fit and Strong Plus: diet – nutrition education and behavioural therapy 30 min 3 d/week; exercise – stretching, aerobics, strength training 60 min 3 d/week T3: Fit and Strong: exercise only – stretching, aerobics, strength training 60 min 3 d/week; OA health education |
L = 2 month; F = 0, 2 and 6 months |
T1: 34·7 ± 0·4; T2: 35·0 ± 0·4 |
T1: 34·0 ± 0·4, Δ = −0·7 ± 0·1*** T2: 34·9 ± 0·4, Δ = −0·2 ± 0·1** T1 v. T2** |
| Magrans-Courtney et al.(27)
USA |
Age = 54 ± 9; n 32; r = 30 (94 %) | T1: isoenergetic low-fat higher carbohydrate (HC) diet: phase I (1 week) 1200 kcal/d; phase II (9 weeks) 1600 kcal/d; exercise: circuit resistance-training 30 min 3 d/week (n 16) T2: isoenergetic low-fat higher protein (HP) diet: phase I (1 week) 1200 kcal/d; phase II (9 weeks) 1600 kcal/d; exercise: circuit resistance-training, 30 min 3 d/week (n 14) O: GCM supplement or placebo |
L = 14 week; F = 0, 10 and 14 weeks |
T1 + T2: 33·3 ± 5·0 |
NR |
| Messier et al.(28)
USA |
Age = T1: 68 ± 1, T2: 69 ± 1, T3: 69 ± 1, C: 69 ± 0; n 316; r = 252 (80 %) | T1: diet – nutrition education and behavioural therapy, individual and group sessions, three phases: intensive (months 1–4) – weekly sessions; transition (months 5–6) – biweekly sessions; and maintenance (months 7–18) – monthly sessions and phone contact (n 82) T2: diet + exercise (n 76) T3: exercise – specialised knee exercise programme: aerobic + resistance/strength training 1 h 3 d/week (n 80) C: healthy lifestyle: usual care, health education (n 78) |
L = 18 months F = 0, 6 and 18 months |
T1: 34·5 ± 0·6 T2: 34·0 ± 0·7 T3: 34·2 ± 0·6 C: 34·2 ± 0·6 |
NR |
| O’Brien et al.(32)
Australia |
Age = 61·6 ± 12·6; n 120; r = 106 (88 %) | T: telephone coaching service (up to 10 calls over 6 months) for weight management, diet and exercise, based on national diet and physical activity guidelines (n 60) C: usual care (n 60) |
L = 6 months F = 0, 6 and 26 weeks |
T: 33·4 ± 3·4 C: 32·1 ± 3·1 |
T: 33·0 ± 4·8 C: 31·9 ± 3·6 |
| Somers et al.(16)
USA |
Age = 58 ± 10 (20–85); n 232; r = 163 (70 %) | T1: nutrition, lifestyle and behavioural therapy: 1200 kcal/d (women) and 1500 kcal/d (men); phase 1 (weeks 1–12) – group sessions 1 h/week, exercise – flexibility, strength, aerobic training 90 min 3 h/week; phase 2 (weeks 13–24) – group sessions 1 h/fortnight (n 59) T2: T1 + T3 (n 62) T3: pain coping skills training: phase 1 (weeks 1–12) – group sessions 1 h/week; phase 2 (weeks 13–24) – group sessions 1 h/fortnight (n 60) C: standard care (n 51) |
L = 6 months F = 0, 6, 12 and 24 months |
T1: 33·5 T2: 34·1 T3: 34·4 C: 34·1 |
T1: 33·9 T2: 32·7 T3: 34·3 C: 34·5 T2 v. C*** |
| Lifestyle (diet and exercise) – uncontrolled clinical trials | |||||
| Martin et al.(22); USA | Age = 61 ± 6 (49–73); n 48; r = 30 (63 %) | T: nutrition education, modifying eating behaviour 1 h/week; prescribed diet 75 % of baseline energy intake; walking programme 45 min 3 d/week (n 30) | L = 6 months F = 0 and 6 months |
T: 31·7 ± 4·9 | Δ BMI: T: −1·1 ± 2·3*** |
| Messier et al.(29); USA | Age = T1: 69 ± 5, T2: 67 ± 4; n 24; r = 21 (88 %) | T1: diet and exercise: nutrition education 1 h/week – 3 group sessions and 1 individual session/month + exercise (n 13) T2: exercise-only: aerobic and strength training 1 h 3 d/week (n 11) |
L = 6 months F = 0, 3 and 6 months |
T1: 35·0 ± 5·0 T2: 38·0 ± 6·0 |
NR |
| Paans et al.(23); The Netherlands | Age = 57 ± 12; n 35; r = 25 (71 %) | T: lifestyle programme: weight loss, nutrition education and behavioural therapy – intensive (3 months), transition (2 months) and maintenance (3 months) phases; exercise – aerobic, mobility + strength training, individual (3 months) and group (5 months) sessions 1 h/week + home-based exercise – aerobic 30 min/d; (n 35) | L = 8 months F = 0, 3 and 8 months |
T: 32 ± 3·9 | NR |
| Diet and meal replacements –RCT | |||||
| Messier et al.(9); USA | Age = 66 ± 6; n 454; r = 399 (88 %) | T1: diet: energy deficit of 800–1000 kcal/d, up to 2 meal replacements/d (Lean Shake), meal plan for third meal, nutrition education and behavioural therapy sessions – group and individual sessions weekly (months 1–6) then biweekly (months 7–18) (n 152) T2: diet + exercise (n 150) C: exercise: aerobic + strength 1 h 3 d/week (n 150) |
L = 18 months F = 0, 6 and 18 months |
T1: 33·7 ± 3·8 T2: 33·6 ± 3·7 C: 33·5 ± 3·7 |
NR |
| Miller et al.(30); USA | Age = 70; n 87; r = 79 (91 %) | T: diet: energy deficit of 1000 kcal/d, up to 2 meal replacements/d (SlimFast Foods), weekly menu plan for third meal, nutrition education and behaviour therapy 1 h/week – group and individual sessions; exercise: aerobic + strength training 1 h 3 d/week (n 44) C: bimonthly healthy lifestyle information sessions, bimonthly newsletters (n 43) |
L = 6 months F = 0 and 6 months |
T: 34·9 ± 0·7 C: 34·3 ± 0·6 |
T: 32·1 ± 0·8 Δ = −8·1 ± 0·7 C: 34·5 ± 0·7 Δ = 0·3 ± 0·9 |
| Diet and meal replacements – uncontrolled clinical trial | |||||
| Atukorala et al.(18); Australia | Age = 64 ± 9; n 1383; r = NR | T: web-based intervention: access to recipes, healthy eating and lifestyle education, and telephone support throughout; phase 1 (weeks 1–6): 2 meal replacements/d (KicStart), 1 controlled portion meal/d; phase 2 (weeks 7–12): 1 meal replacement/d, 2 portion-controlled meals/d (lunch + dinner); phase 3 (week 13 – onwards): 3 portion-controlled meals/d (n 1383) | L = 18 weeks F = 0, 6 and 18 weeks |
T: 34·4 ± 5·2 | NR |
| de Luis et al.(20); Spain | Age = 60 ± 13; n 55; r = 55 (100 %) | T: oral diet + hypoenergetic formula (Optisource) 2/d (lunch and dinner) 1109·3 kcal/d (n 55) | L = 3 months F = 0 and 3 months |
T: 38·6 ± 5·7 | T: 35·3 ± 5·7 Δ = −3·1 ± 1·5* |
| López Gómez et al.(24); Spain | Age = 62·2 ± 8·5; n 81; r = 75 (92·6 %) | T: low-fat hyperproteic diet – meal-replacement oral nutritional supplement (VEGEStart) 2/d (lunch and dinner) 1035 kcal/d, nutrition education (n 81) | L = 3 months F = 0 and 3 months |
T: 40·8 ± 4·4 | T: 37·0 ± 5·0** |
| VLED –RCT | |||||
| Bliddal et al.(25); Denmark | Age = 63 ± 11 (36–90); n 89; r = 56 (63 %) | T: alternating VLED 810 kcal/d (1–8 and 32–36 weeks) via nutrition powder (Speasy) dissolved in water taken 6/d, and hypoenergetic diet 1200 kcal/d (8–32 and 36–52 weeks), dietitian led sessions 1·5 h/week (n 44) C: 2 h education session (0, 8, 32, 36 and 52 weeks), conventional hypoenergetic, high protein diet 1200 kcal/d (n 45) |
L = 52 weeks F = 0, 8, 32, 36 and 52 weeks |
T: 36·0 ± 5·5 C: 35·2 ± 4·5 |
NR |
| Christensen et al.(26); Denmark | Age = 63 ± 11 (35–90); n 89; r = 80 (90 %) | T: VLED 3·4 MJ/d (810 kcal) via nutritional powder (Speasy) dissolved in water taken 6/d, nutrition education and behavioural therapy 1·5 h/week (n 44) C: 2 h nutrition education, diet plan booklets – conventional hypoenergetic, high protein diet ~5 MJ/d (1190 kcal) (n 45) |
L = 8 weeks F = 0 and 8 weeks |
T: 36·3 ± 5·6 C: 35·5 ± 4·6 |
NR |
| VLED – uncontrolled clinical trial | |||||
| Riecke et al.(31); Denmark | Age = 63 ± 6; n 192; r = 175 (91 %) | 1–8 weeks: 3 meal replacements/d (The Cambridge Diet), nutrition education and behavioural therapy 1·5 h/week T1: 415 kcal/d (n 96), meal replacement dissolved in water T2: 810 kcal/d (n 96), meal replacement dissolved in skimmed milk 9–16 weeks: hypoenergetic diet 1200 kcal/d, usual foods and 2 meal replacements/d, nutrition education and behavioural therapy 1·5 h/week (n 192) |
L = 16 weeks F = 0, 8 and 16 weeks |
T1: 37·5 ± 5·4 T2: 37·1 ± 4·1 |
T1: −4·8 ± 0·2*** T2: −4·0 ± 0·2*** T1 v. T2 (NS) |
| Sub-studies of Riecke et al. (2010) | |||||
| Aaboe et al.(17); Denmark | Age = 63 ± 6 (62–64); n 177; r = 157 (89 %) | T: 1–8 weeks: 3 meal replacements/d (The Cambridge Diet) 415–810 kcal/d, nutrition education and behavioural therapy 1·5 h/week 9–16 weeks: hypoenergetic diet 1200 kcal/d, usual foods + 2 meal replacements/d, nutrition education and behavioural therapy 1·5 h/week (n 157) |
L = 16 weeks F = 0 and 16 weeks |
T: 36·9 ± 4·3 | Δ BMI: T: −5·1 ± 0·1 (13·8 %)*** |
| Bartels et al.(19); Denmark | Age = 63 ± 6 (50–77); n 192; r = 175 (91 %) | T: 1–8 weeks: 3 meal replacements/d (The Cambridge Diet) 415–810 kcal/d, nutrition education and behavioural therapy 1·5 h/week 9–16 week: hypoenergetic diet 1200 kcal/d, usual foods + 2 meal replacements/d, nutrition education and behavioural therapy 1·5 h/week (n 175) |
L = 16 weeks F = 0 and 16 weeks |
T: 37·1 ± 4·4 | NR |
| Henriksen et al.(21); Denmark | Age = 63 ± 6 (50–80); n 159; r = NA | T: 1–8 weeks: 3 meal replacements/d (The Cambridge Diet) 415–810 kcal/d, nutrition education and behavioural therapy 1·5 h/week 9–16 weeks: hypoenergetic diet 1200 kcal/d, usual foods + 2 meal replacements/d, nutrition education and behavioural therapy 1·5 h/week (n 159) |
L = 16 weeks F = 0 and 16 weeks |
T: 36·1 ± 4·0 | NR |
Δ, change; GCM, glucosamine/chondroitin/methlysulphonylmethane; NR, not reported; VLED, very low-energy diet.
Results reported as mean ± sd or mean ± sem.
*P < 0·05, **P < 0·01, ***P < 0·001.
Body weight and BMI
Lifestyle interventions
Of the eight lifestyle studies, the average baseline weight was 93·1 kg (range: 86·2–108·4 kg), with average weight loss ranging between 0·1 and 8·5 kg for treatment groups (Table 3). Average baseline BMI was 34·0 kg/m2 (range: 31·7–38·0 kg/m2), with only four studies reporting on change in BMI at follow-up (Table 2)(16,22,32,33). Of the five lifestyle RCT(16,27,28,32,33), these were of varying duration between 8 weeks and 18 months(28,33). Messier et al.(28) conducted a long-term (18-month) RCT that compared three intervention groups – dietary weight loss, exercise only and diet plus exercise – with a healthy control group. The diet-only and diet plus exercise groups lost an average of 4·6 and 5·2 kg body weight, respectively, which was statistically significantly lower than the control group (−1·1 kg; P < 0·05)(28). In contrast, two six-month RCT by Somers et al. and O’Brien et al. reported no significant changes in weight or BMI(16,33). A 10-week lifestyle RCT by Magrans-Courtney et al. compared the effects of a high-carbohydrate (55 % energy intake) or high-protein (55–63 % energy intake) weight loss diet in combination with circuit resistance training, reporting small but non-significant weight loss of 1·6 and 2·4 kg, respectively(27). After the 2-month intervention, Hughes et al. reported a statistically significant reduction in weight (−2·0 kg) and BMI (−0·7 kg/m2) in the diet and exercise treatment group, compared with the exercise-only control group (P < 0·01)(33).
Table 3.
Weight and physical function outcomes for included studies: reporting of baseline and follow-up results of weight and physical function measures including performance-based and self-reported measures†
| Study | Weight (kg) | Performance-based physical function | Self-report physical function | |||
|---|---|---|---|---|---|---|
| Author | Baseline | Follow-up | Baseline | Follow-up | Baseline | Follow-up |
| Diet and exercise – randomised controlled trials (RCT) | ||||||
| Hughes et al.(33) | T1: 93·2 ± 1·1 T2: 93·7 ± 1·1 |
T1: 91·2 ± 1·1 Δ = −2·0 ± 0·2*** T2: 93·2 ± 1·1 Δ = −0·5 ± 0·2** T1 v. T2*** |
6-min walk (m): T1: 360·2 ± 6·2 T2: 357·7 ± 6·2 |
6-min walk (m): T1: 411·1 ± 6·8 Δ = 50·9 ± 4·4*** T2: 380·8 ± 6·8 Δ = 23·0 ± 4·4*** T1 v. T2*** |
WOMAC (0–68): T1: 17·7 ± 0·9 T2: 17·8 ± 0·9 |
WOMAC (0–68): T1: 12·1 ± 0·9 Δ = −5·6 ± 0·8*** T2: 15·9 ± 0·9 Δ = −1·9 ± 0·7* T1 v T2*** |
| Magrans-Courtney et al.(27) | T1: 87·4 ± 13·0 T2: 90·0 ± 14·0 |
T1: 85·8 ± 14·0** T2: 87·6 ± 14·0** T1 v T2 (NS) |
NR | NR | WOMAC (0–1700): T1 + T2: 879·0 ± 428·0 |
WOMAC (0–1700): T1 + T2: 517·0 ± 390·0** |
| Messier et al.(28) | T1: 95·0 ± 0·2 T2: 92·0 ± 0·2 T3: 92·0 ± 0·2 C: 96·0 ± 0·2 |
Δ (kg): T1: −4·6, T1 v C* T2: −5·2, T2 v C* T3: −3·5 C: −1·1 |
6-min walk (m): T1: 425·9 ± 10·9 T2: 416·2 ± 11·3 T3: 424·2 ± 11·4 C: 434·6 ± 11·0 Stair-climb time (s) T1: 9·7 ± 0·7 T2: 10·9 ± 0·7 T3: 10·5 ± 0·7 C: 9·6 ± 0·6 |
6-min walk (m): T1: 435·6 ± 12·9 Δ = 9·7 T2: 477·8 ± 13·1 Δ = 61·6 T2 v C* T3: 472·7 ± 13·1 Δ = 48·6 T3 v C* C: 429·9 ± 12·8 Δ = −4·7 Stair-climb time (s) T1: 8·4 ± 0·8 Δ = −1·3 T2: 8·5 ± 0·8 Δ = −2·5 T2 v C* T3: 8·9 ± 0·8 Δ = −1·6 C: 9·4 ± 0·8 Δ = −0·2 |
WOMAC (0–68): T1: 23·3 ± 1·3 T2: 23·6 ± 1·4 T3: 24·0 ± 1·3 C: 26·0 ± 1·3 |
Δ WOMAC: T1: −4·2 (18·0 %)* T2: −5·7 (24·0 %)* T3: −3·1 (12·0 %) C: −3·4 (13·0 %) |
| O’Brien et al.(32) | T: 93·3 ± 12·9 C: 89·9 ± 13·5 |
T: 92·9 ± 14·0 C: 89·7 ± 14·9 T v C (NS) |
NR | NR | WOMAC (0–68): T: 34·9 ± 12·6 C: 34·5 ± 12·2 T v C (NS) |
WOMAC (0–68): T: 36·5 ± 13·2 C: 32·8 ± 15·1 |
| Somers et al.(16) | T1: 92·3 T2: 92·6 T3: 96·6 C: 92·3 |
T1: 92·2 T2: 89·4, T2 v C*** T3: 94·3 C: 94·1 |
Gait velocity – normal (m/s): T1: 1·1 T2: 1·1 T3: 1·1 C: 1·1 Gait velocity – fast (m/s): T1: 1·6 T2: 1·5 T3: 1·5 C: 1·5 |
Gait velocity – normal (m/s): T1: 1·2 T2: 1·2 T3: 1·1 C: 1·2 Gait velocity – fast (m/s): T1: 1·5 T2: 1·6 T3: 1·5 C: 1·5 |
WOMAC (0–100): T1: 44·3 T2: 47·7 T3: 46·2 C: 46·1 |
WOMAC (0–100): T1: 36·0 T2: 25·1, T2 v C*** T3: 35·2 C: 37·5 |
| Diet and exercise – uncontrolled clinical trials | ||||||
| Martin et al.(22) | T: 86·2 ± 12·6 | Δ (kg): T: −5·6 ± 4·0*** |
6-min walk (m): T: 424·9 ± 53·0 |
6-min walk (m): T: 466·0 ± 54·9 Δ ( %) T: 9·7 ± 10·4*** |
WOMAC (0–68): T: 19·0 ± 15·0 |
WOMAC (0–68): T: 13·0 ± 12·0 Δ ( %) T: 21·0 ± 37·0* |
| Messier et al.(29) | T1: 91·2 ± 12·7 T2: 108·4 ± 16·8 |
Δ (kg): T1: -8·5 T2: -1·8 T1 v T2** |
6-min walk (m): T1 + T2: 429·2 Stair-climb time (s) T1 + T2: 9·8 |
6-min walk (m): T1: 523·6 ± 12·5*** T2: 555·0 ± 11·0*** Stair-climb time (s) T1: 7·4 ± 0·3*** T2: 8·7 ± 0·4** T1 v T2* |
NR | NR |
| Paans et al.(23) | NR | Δ (kg): T: −5·6*** |
6-minute walk (m): T: 433·3 ± 13·5 Gait speed- 20-m walk (s) T: 15·3 ± 0·3 |
6-minute walk (m): T: 481·4 ± 10·9 Δ = 48·1* Gait speed- 20-m walk (s) T: 14·1 ± 0·3 Δ = −1·2* |
WOMAC (0–100): T: 53·0 ± 2·9 |
WOMAC (0–100): T: 70·3 ± 2·3 Δ = −17·3* |
| Diet and meal replacements (DMR) – RCT | ||||||
| Messier et al.(9) | T1: 93·4 T2: 93·0 C: 92·3 |
T1: 84·5 Δ = -8·9 T1 v C*** T2: 82·4 Δ = -10·6 T2 v C*** C: 90·5 Δ = -1·8 |
6-min walk (m): T1: 475·0 T2: 467·0 C: 480·0 Walk speed (m/s): T1: 1·2 T2: 1·2 C: 1·2 |
6-min walk (m): T1: 502·0 Δ = 26 (6 %) T2: 537·0 Δ = 70 (15 %) T2 v. T1 & C*** C: 525·0 Δ = 45 (9 %) C v T1*** Walk speed (m/s): T1: 1·3 Δ = 0·1 (8 %) T2: 1·3 Δ = 0·1 (10 %) T2 v. T1 & C** C: 1·3 Δ = 0·1 (6 %) |
WOMAC (0–68): T1: 24·8 T2: 24·6 C: 23·1 |
WOMAC (0–68): T1: 17·7 Δ = −7·1 (29·0 %) T2: 14·2 Δ = −10·3 (42·0 %) T2 v. T1 & C*** C: 17·6 Δ = −5·5 (24·0 %) |
| Miller et al.(30) | T: 98·1 ± 2·6 C: 97·5 ± 2·4 |
T: 89·8 ± 2·7 Δ = −8·3 ± 0·8 C: 98·9 ± 2·9 Δ = −0·1 ± 0·7 T v C** |
6-min walk distance (m): T: 436·5 ± 13·0 C: 447·8 ± 14·9 Stair-climb time (s) T: 9·2 ± 0·5 C: 10·7 ± 0·8 |
6-min walk distance (m): T: 510·0 ± 15·0 Δ = 72·8 ± 10·3 C: 459·0 ± 17·4 Δ = 10·5 ± 6·3 T v. C** Stair-climb time (s) T: 7·7 ± 0·4) Δ = −1·5 ± 0·5 C: 11·2 ± 1·2 Δ = 0·8 ± 0·5 T v. C** |
WOMAC (0–68): T: 24·0 ± 1·5 C: 26·7 ± 1·9 |
WOMAC (0–68): T: 15·2 ± 1·5 Δ = −8·4 ± 1·8 C: 23·8 ± 2·0 Δ = −1·6 ± 1·6 T v. C* |
| DMR – uncontrolled clinical trial | ||||||
| Atukorala et al.(18) | T: 95·1 ± 17·2 | Δ (kg): T: −7·9 ± 4·2 (8·3 %) |
NR | NR | KOOS-FDL (0–100): T: 59·5 ± 18·3 KOOS-FSR (0–100) T: 27·6 ± 24·2 |
Δ KOOS-FDL: T: +13·3 ± 16·1 Δ KOOS-FSR T: +12·6 ± 24·8 |
| de Luis et al.(20) | T: 97·8 ± 18·3 | T: 89·4 ± 10·2 Δ = −7·3 ± 3·8* |
NR | NR | WOMAC (0–68): T: 29·6 ± 14·5 |
WOMAC (0–68): T: 24·6 ± 16·5 Δ = −5·0 ± 10·2* |
| López Gómez et al.(24) | T: 99·3 ± 14·6 | T: 91·0 ± 13·0** | NR | NR | WOMAC (0–100): T: 49·2 3 ± 27·0 |
WOMAC (0–100): T: 40·2 ± 22·1–54·4** |
| Very low-energy diet (VLED) – RCT | ||||||
| Bliddal et al.(25) | T: 95·7 ± 15·1 C: 95·5 ± 13·7 |
Δ (kg) T: −10·9 ± 0·8 C: −3·6 ± 0·8 T v C*** Δ ( %) T: –11·3 ± 0·8 C: –3·7 ± 0·8 T v C*** |
NR | NR | WOMAC (0–100) T: 40·2 ± 19·1 C: 38·3 ± 23·6 |
Δ WOMAC T: −7·5 ± 2·0 C: −3·9 ± 2·0 T v C (NS) |
| Christensen et al.(26) | T: 96·4 ± 15·5 C: 97·1 ± 13·4 |
Δ (kg) T: −11·0 ± 0·6 C: −4·4 ± 0·6 T v C*** Δ ( %) T: −11·1 ± 0·6 C: −4·3 ± 0·6 T v C*** |
NR | NR | WOMAC (0–1700) T: 678·9 ± 338·0 C: 592·1 ± 399·8 |
Δ WOMAC T: −252·5 ± 49·6 C −85·6 ± 51·9 T v C** |
| VLED – uncontrolled clinical trials | ||||||
| Riecke et al.(31) | T1: 104·1 ± 15·6 T2: 102·3 ± 14·4 |
Δ (kg) T1: −13·3 ± 0·7 T2: −12·2 ± 0·6 T1 v T2 (NS) Δ ( %) T1: −12·9 ± 0·6 T2: −12·0 ± 0·6 T1 v T2 (NS) |
NR | NR | KOOS-FDL (0–100) T1: 59·0 ± 16·9 T2: 60·8 ± 17·9 KOOS-FSR (0–100) T1: 19·6 ± 18·1 T2: 25·6 ± 21·1 |
Δ KOOS-FDL T1: 11·0 ± 0·5 T2: 11·1 ± 1·3 T1 v T2 (NS) Δ KOOS-FSR T1: 8·8 ± 1·8 T2: 8·4 ± 2·2 T1 v T2 (NS) |
| Sub-studies of Riecke et al. (2010) | ||||||
| Aaboe et al.(17) | T: 101·5 ± 1·1 | Δ (kg) T: −13·6 ± 0·2 (13·2 %)*** |
Walking speed (m/s) T: 1·1 ± 0·0 |
Δ Walking speed (m/s) T: 0·1 ± 0·1 (4 %)* |
NR | NR |
| Bartels et al.(19) | T: 102·4 ± 14·5 | Δ (kg) T: −13·4*** |
NR | NR | KOOS-FDL (0–100) T: 60·4 ± 16·8 |
Δ KOOS-FDL T: 12·5*** |
| Henriksen et al.(21) | T: 99·5 ± 12·7 | Δ (kg) T: −12·9*** |
NR | NR | WOMAC (0–100) T: 38·8 ± 16·4 |
Δ WOMAC T: −11·7*** |
Δ, change; WOMAC, Western Ontario and McMaster Universities Arthritis Index (function subscale); NR, not reported; KOOS-FDL: knee osteoarthritis outcome score – function in daily living; KOOS-FSR: knee osteoarthritis outcome score – function in sport and recreation.
Results reported as mean ± sd or mean ± sem.
*P < 0·05, **P < 0·01, ***P < 0·001.
The three uncontrolled lifestyle studies(22,23,29) utilised a combination of diet and exercise via nutrition education and behavioural therapy over 6–8 months(22,23,29), reporting a mean weight loss of 6·6 kg (range: −5·6 to −8·5 kg). Martin et al. also reported a significant reduction in BMI of 1·1 kg/m2 (P < 0·01)(22), while Messier et al.(29) and Paans et al.(23) did not report on change in BMI.
Diet and meal replacement interventions
Five studies utilised partial meal replacements (i.e. up to two per d) with conventional foods. These studies reported an average baseline weight of 95·8 kg (range: 92·3–99·3 kg), with weight loss ranging from 7·3 to 10·6 kg in the treatment groups. Average baseline BMI was 35·5 kg/m2 (range: 33·5–40·8 kg/m2), and change in BMI was reported in three DMR studies(20,24,30). Messier et al.(9) conducted a second 18-month RCT, comparing a diet-only and diet plus exercise intervention, to an exercise-only control group. The diet-only and diet plus exercise interventions resulted in statistically significant weight loss (−8·9 and −10·6 kg, respectively), compared with controls (−1·8 kg; P < 0·01)(9). Miller et al. conducted a 6-month intervention, incorporating up to two meal replacements per day, behavioural therapy and exercise (aerobic and strength training) on 3 d/week, compared with a control group provided with general health information(30). The intervention resulted in a statistically significant weight loss of 8·3 kg (P < 0·01), and a reduction in BMI of 8·1 kg/m2 (significance not reported), compared with the control group(30).
The three uncontrolled DMR studies were diet-only interventions (i.e. did not include exercise interventions), conducted over 12–18 weeks(18,20,24). All three studies reported statistically significant weight loss at an average of 7·8 kg (range: −7·3 to −8·3 kg)(18,20,24), while change in BMI was only reported in two studies (de Luis et al. and López Gómez et al.) with an average reduction of 3·5 kg/m2(20,24).
Very low-energy diet interventions
Of the six VLED studies, the average baseline weight was 99·4 kg (range: 95·7–104·1 kg), with average weight loss ranging between 10·9 and 13·6 kg. The average baseline BMI was 36·4 kg/m2 (range: 35·2–37·5 kg/m2), with only two studies reporting on change in BMI at follow-up(17,31). Of the four uncontrolled studies(17,19,21,31), these ranged between 12 and 16 weeks in duration. The CAROT study, as reported by Riecke et al., compared a VLED (415 kcal/d) with a low-energy diet (LED; 810 kcal/d) for 8 weeks, after which participants were transitioned to a diet of 1200 kcal/d using usual foods, for the final 8 weeks of the 16-week study(31). Both the VLED and LED dietary regimens demonstrated similar reductions in weight (−13·3 and −12·2 kg, respectively) and BMI (−4·8 and −4·0 kg/m2, respectively), although there was no significant difference between the two groups(31). Due to the small difference of weight loss between dietary regimens in the CAROT study, the groups were collapsed for secondary data analysis, and average weight loss was reported as 13 kg (P < 0·01)(17,19,21), and a change in BMI of −5·1 kg/m2 (P < 0·01)(17).
The only two VLED RCT reported similar weight loss results(25,26). Over the intervention period of 52 weeks, Bliddal et al. provided an alternating treatment of 810 kcal VLED (weeks 1–8 and 32–36) provided as a nutritional supplement dissolved in water (taken six times/d), and nutrition education to achieve a hypoenergetic diet of 1200 kcal/d (weeks 8–32 and 36–52)(25). The control group attended five nutrition education sessions (baseline and weeks 8, 32, 36 and 52), with instruction to consume a hypoenergetic diet using usual foods (1200 kcal/d)(25). The short-term (8-week) RCT by Christensen et al. compared a continuous VLED intervention (810 kcal/d) using a nutritional powder dissolved in water, with a healthy control group, who received nutrition education at baseline, and instruction to consume a hypoenergetic diet (1200 kcal/d)(26). Both Bliddal et al. and Christensen et al. reported a statistically significant weight loss of 11 kg (P < 0·01) in the treatment groups, compared with 4 kg in the control groups, despite the varying durations of 8 and 52 weeks of the intervention period(25,26). Change in BMI was not reported in either of these studies(25,26).
Performance-based physical function measures
Table 3 provides a summary of physical function outcomes including before and after treatment outcomes for performance-based and self-report physical function measures. A wide range of tests were used to measure performance-based physical function in the included studies; however, the most commonly used measures were 6-min walk distance (n 6), walk speed (n 3) and stair-climb time (n 3).
Of the lifestyle studies, six assessed one or more of the performance-based measures of interest(16,22,23,28,29,33). Six-minute walk distance was measured in five lifestyle studies (two RCT(28,33) and three uncontrolled studies(22,23,29)), of which the average increase in walk distance was 56·2 m (range: 9·7–125·8 m) in the treatment groups. Two RCT reported a statistically significant increase in 6-min walk distance in the diet and exercise groups, compared with controls(28,33). Two lifestyle studies measured stair-climb time reporting an average reduction in time by 1·6 s (range: −1·1 to −2·5 s) in the treatment groups(28,29). Walk speed was measured in two lifestyle studies(16,23). Paans et al. measured walk speed via the 20 m walk test, with the treatment group showing a significant increase in speed of 1·2 s (P < 0·05)(23), while Somers et al. measured gait speed at normal and fast paces, however reported only small non-significant changes in the weight loss group(16).
Miller et al.(30) and Messier et al.(9) were the only two DMR studies to measure performance-based physical function. Both RCT measured 6-min walk distance, reporting an average increase of 56·3 m (range: 26·0–72·8 m) which was statistically significantly greater than the control groups(9,30). Miller et al. also reported a significant reduction in stair-climb time of 1·5 s in the treatment group, compared with the control group (P < 0·01)(30). Messier et al. reported statistically significant increase in walk speed of 0·1 m/s in both the diet-only and diet plus exercise group, compared with controls(9). Aaboe et al., the only VLED study to measure performance-based physical function, also reported a statistically significant increase in walk speed of 0·1 m/s (P = 0·02) following the 16-week intervention(17).
Self-reported functional status
The majority of studies measured physical function using self-report measures of interest, with the exception of one lifestyle study(29) and one VLED study(17). The two most common tools used were the Knee Injury and Osteoarthritis Outcome Score (KOOS) and Western Ontario and McMaster Universities Arthritis Index (WOMAC) as summarised in Table 3.
Knee injury and osteoarthritis outcome score (KOOS)
The KOOS includes two physical function subscales, one that measures function in daily living (KOOS-FDL) and another that measures function in sport and recreation (KOOS-FSR). Each subscale is scored between 0 and 100, with higher scores indicating better function. Three uncontrolled studies measured change in physical function with KOOS, including one DMR study by Atukorala et al.(18) and two VLED studies by Bartels et al. and Riecke et al.(19,31) Following the 18-week diet-only intervention, Atukorala et al. reported a statistically significant improvement in KOOS function in daily living (KOOS-FDL) and KOOS function in sport and recreation (KOOS-FSR) score by 13·3 and 12·6, respectively (P < 0·01)(18). The VLED studies reported an average increase in KOOS-FDL by 11·5 (range: 11·0–12·5)(19,31), while only Riecke et al. reported KOOS-FSR, with an average increase of 8·6 for the VLED and LED treatment groups(31).
Western Ontario and McMaster universities arthritis index
The most commonly used physical function measure was the WOMAC function subscale which was reported in seven lifestyle studies(16,22,23,27,28,32,33), four DMR studies(9,20,24,30) and three VLED studies(21,25,26). Scoring of the WOMAC was reported differently between studies, with two reporting on a scale of 0–1700(26,27), obtained using a visual analogue scale method, five reporting a scaled score of 0–100(16,21,23–25), and six reporting on a scale of 0–68(9,20,22,28,30,32), obtained using a Likert scale method. A lower score represents better function in all studies but one, by Paans et al.(23) For the uncontrolled lifestyle and DMR studies, an average improvement by 26·8(22,23) and 17·6 %, respectively, in WOMAC function score was reported(20,24). The only uncontrolled VLED study that measured WOMAC function reports a significant improvement of 30·2 % (P < 0·01)(21).
Eight RCT were included in the meta-analysis that measured change in WOMAC physical function (thirteen independent comparisons)(9,16,25,26,28,30,32,33), which were categorised based on the intervention type. In the lifestyle studies (n 4)(16,28,32,33), the pooled effects analysis did not support an improvement in WOMAC physical function (effect size (ES) −5·3, 95 % CI −15·4, 4·7; P = 0·30) (Fig. 2). This result was following a statistically significant group mean difference in weight loss of 1·6 % (95 % CI −2·2, −1·0; P < 0·01). In contrast, the DMR RCT(9,30) showed a statistically significant mean difference between the treatment and control groups in WOMAC physical function of 12·4 % (95 % CI −21·5, −3·3; P < 0·01), based on 563 randomised participants (Fig. 3). This change followed a significant mean difference in weight loss of 8·5 % (95 % CI −10·2, −6·8; P < 0·01) between treatment and control groups. Likewise, the VLED RCT(25,26) demonstrated a significant mean difference between the control and intervention groups in WOMAC physical function of 12·5 % (95 % CI −24·5, −0·6; P = 0·04) favouring the VLED intervention group (Fig. 4). This result is based on 136 randomised participants in the VLED studies, following a statistically significant group mean difference in weight loss of 7·1 % (95 % CI −8·5, −5·8; P < 0·01).
Fig. 2.
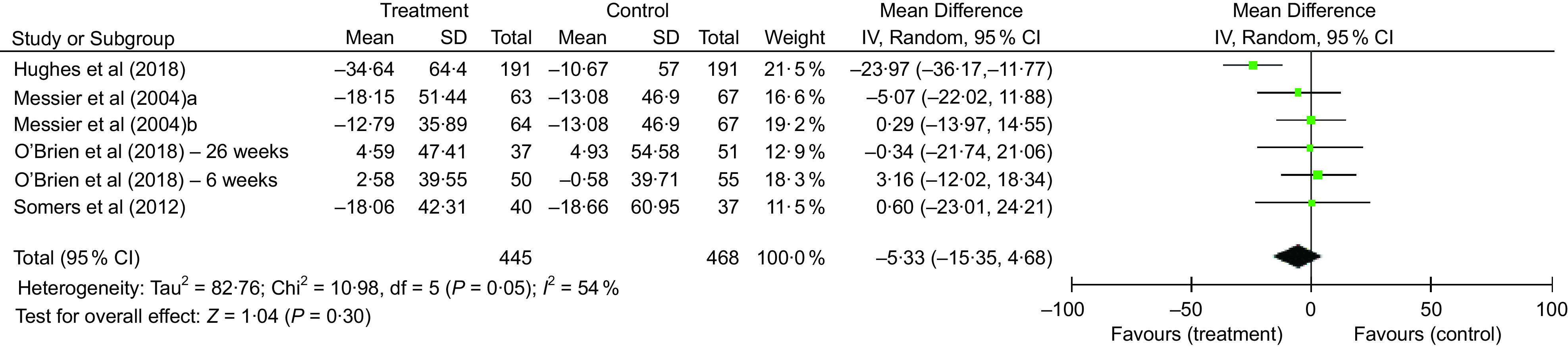
Comparison of lifestyle intervention to control for % change in Western Ontario and McMaster Universities Arthritis Index (WOMAC) physical function
Fig. 3.
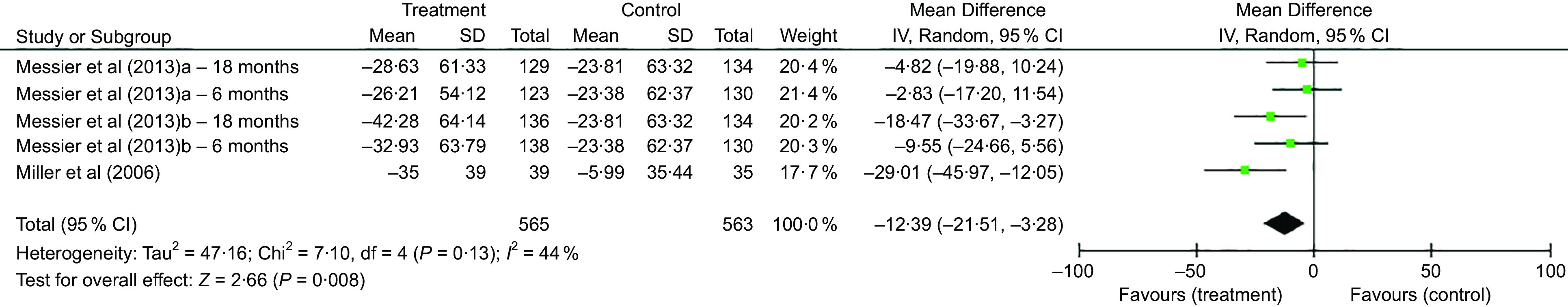
Comparison of diet and meal replacement (DMR) intervention to control for % change in Western Ontario and McMaster Universities Arthritis Index (WOMAC) physical function
Fig. 4.
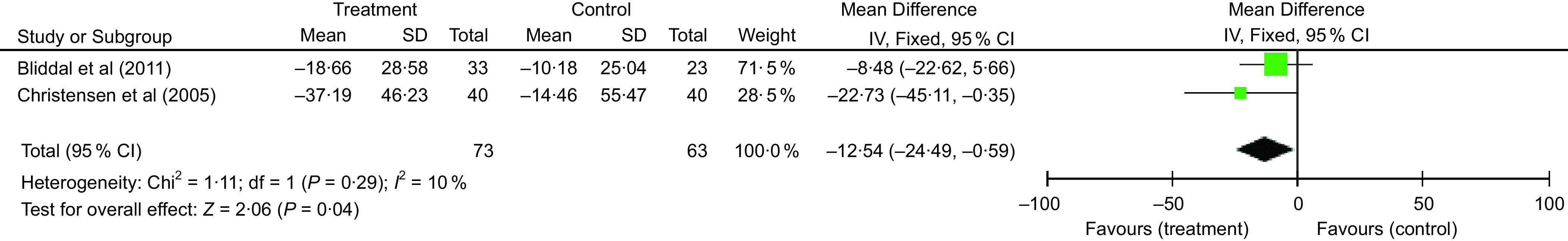
Comparison of very low energy diet (VLED) to control for % change Western Ontario and McMaster Universities Arthritis Index (WOMAC) physical function
Discussion
The present systematic review provides a comprehensive summary of studies that have assessed the effect of weight loss, comparing different dietary interventions of varying dietary restriction (lifestyle, DMR and VLED), on physical function in adults with OA. Several measures of physical function were defined including participant self-report via questionnaires (e.g. WOMAC) or objective performance-based measures (e.g. 6-min walk test). Of the nineteen included studies most reported statistically significant weight loss and improvements in physical function (n 15). Overall, there were 4178 participants (1832 from RCT and 2346 from uncontrolled studies), the majority of whom had knee OA and only forty-six had hip OA, from two uncontrolled studies(20,23). At baseline, the average BMI was 34·0 kg/m2 (range: 31·7–38·0) in the lifestyle studies, 35·5 kg/m2 (range: 33·5–40·8) in the DMR studies and 36·4 kg/m2 (range: 35·2–37·5) in the VLED studies, placing the majority of participants in the obese class I and class II categories. Of the included studies, nine (47 %) were RCT, the majority of which were lifestyle studies (n 5), and the remaining ten (53 %) studies were uncontrolled trials.
The average weight loss was variable between the lifestyle, DMR and VLED studies. The lifestyle studies incorporated nutrition education, behavioural therapy and exercise, over a period of 8 weeks to 18 months, demonstrating an average weight loss of 4 % (range: 0–9 %) in the treatment groups(16,22,23,27–29,32,33). In comparison, the DMR studies which incorporated partial use of meal replacements and nutrition education, with or without exercise, over 12 weeks to 18 months, reported an average weight loss of 9 % (range: 8–11 %)(9,18,20,24,30). As expected, the VLED intervention studies demonstrated the largest average weight loss of 12 % (range: 11–13 %) for the treatment groups(17,19,21,25,26,31). The greater energy restriction of the VLED intervention (energy intake range: 415–810 kcal/d) explains the greater weight loss compared with the lifestyle and DMR studies. Previous reports of weight loss strategies are in agreement with that hypoenergetic diets incorporating partial meal replacements provides greater long-term weight loss (up to 12 months) than a conventional diet control(37,38). While the use of VLED may provide greater weight loss, clinical guidelines report that these are ineffective in the long-term, are nutritionally inadequate and should only be used for up to 12 weeks(39,40).
The WOMAC function subscale was the most commonly reported physical function measure (n 14). In the lifestyle studies, average change in WOMAC function was 23 % (range: −5–41 %) in the treatment groups. While the DMR and VLED studies reported an average change in WOMAC function of 28 % (range: 17–42 %) and 29 % (range: 19–37 %) in the treatment groups, respectively. Meta-analysis did not demonstrate a statistically significant change in WOMAC function in the lifestyle studies (n 4). While a significantly greater improvement in WOMAC physical function by 12·4 and 12·5 % was detected in the DMR and VLED treatment groups compared with controls, with similar differences in weight changes (DMR: 8·5 % v. VLED: 7·1 %). It should be noted that only two studies were included in the VLED and DMR groups, respectively, which may limit the reproducibility of results. Previous studies have demonstrated that weight loss of ≥10 % (and up to 20 %) is conducive to better improvements in OA symptoms(18,41). While the authors reported no safety concerns, weight loss of this magnitude should be carefully monitored, particularly in older adults at risk of loss of lean and bone mass, which can lead to risk of falls, fractures and disability(42).
The findings of our review are in agreement with earlier studies including systematic reviews that have reported improvements in physical function following weight loss(10–12,43). Two of the more recent reviews looked at the effect of dietary weight loss, with or without exercise treatment, on physical function in adults with knee OA classified as overweight and obese(11,12). Pooled effects demonstrated statistically significant improvements in self-report physical function and disability following dietary weight loss(11,12). However, Hall et al. reported that these effects were not sustained in the long term (i.e. longer than 12 months)(12). Our review demonstrates a more comprehensive summary of a larger sample of both controlled and uncontrolled dietary weight loss studies, which have not been included in previous reviews(16–24,27,31). Further, we investigated the dietary modalities by which weight loss is induced, providing a comparison of three dietary weight loss interventions (lifestyle, DMR and VLED). Our findings show the change in physical function is similar between DMR and VLED weight loss interventions, and both DMR and VLED interventions are more effective than lifestyle programmes to induce significant change in physical function.
Quality of the evidence
The quality assessment highlighted limitations in study design of several of the included studies. A validated quality appraisal tool(14) was used to assess quality and risk of bias of included studies. Of the included studies, half (n 9) were of high-quality rating and low risk of bias, indicating a moderately positive quality of the review overall. The remaining nine studies received a neutral quality rating, as they were without a comparative study group (n 8)(17–24) or had non-comparable groups at baseline (n 1)(16) potentially resulting in selection bias. The three CAROT sub-studies(17,19,21) may be prone to a greater degree of bias, as research questions were not decided a priori. None of the included studies received a poor-quality rating. To be considered a high-quality study, participant retention rate is defined at 80 % or more(14) as this influences the interpretation of results. Four of the included studies did not reach this target, with retention rates of 63(22,25), 71(23) and 70 %(16), of which two were uncontrolled studies(22,23). While RCT provide the highest level of evidence, poor retention rates need to be considered when interpreting results. As with many dietary intervention studies, there was considerable variability between studies in terms of methodology, including intervention components, length of follow-up and outcome measures, which may have affected the comparability of findings of included studies and interpretation of the results.
Clinical implications
This review is the first to provide a comprehensive summary and comparison of the different dietary interventions commonly used for weight loss and change in physical function in adults with OA classified as overweight and obese. Overall, the findings of the included studies indicate that significant weight loss is generally associated with an improvement in physical function, demonstrated by objective and subjective measures. While the VLED interventions demonstrated the highest mean weight loss of 12 %, followed by the DMR (9 % WL) and lifestyle interventions (4 % WL), both DMR and VLED groups showed similar improvements in physical function. This suggests that the less restrictive dietary intervention with partial use of meal replacements results in weight loss of clinical significance between 5 and 10 %(4) and the improvements in physical function are comparable to VLED interventions that are wholly reliant on the use of meal replacements. Therefore, DMR interventions may be the most optimal treatment for improved physical function and long-term weight management in adults with OA.
Acknowledgements
Acknowledgements: The authors thank Debbie Booth, Faculty Librarian, Health and Medicine, for her assistance with the database search strategy. Financial support: This work was supported by the Australian Government Research Training Program Scholarship, of which first author E.J.W. is a recipient. Conflict of interest: None. Authorship: E.J.W. and S.K.B. completed screening, data extraction and synthesis, and assessment of study quality. All authors were responsible for the study design and completed final reviewing and editing of the manuscript. Ethics of human subject participation: Not applicable.
Supplementary material
For supplementary material accompanying this paper visit https://doi.org/10.1017/S1368980020002529.
click here to view supplementary material
References
- 1. Cross M, Smith E, Hoy D et al. (2014) The global burden of hip and knee osteoarthritis: estimates from the global burden of disease 2010 study. Ann Rheum Dis 73, 1323–1330. [DOI] [PubMed] [Google Scholar]
- 2. Manini T (2011) Development of physical disability in older adults. Curr Aging Sci 4, 184–191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Silverwood V, Blagojevic-Bucknall M, Jinks C et al. (2015) Current evidence on risk factors for knee osteoarthritis in older adults: a systematic review and meta-analysis. Osteoarthr Cartilage 23, 507–515. [DOI] [PubMed] [Google Scholar]
- 4. National Health and Medical Research Council (2013) Clinical Practice Guidelines for the Management of Overweight and Obesity in Adults, Adolescents and Children in Australia. Melbourne, Australia: National Health and Medical Research Council. [Google Scholar]
- 5. Codex Alimentarius Commission (1995) Report of the Nineteenth Session of the Codex Committee on Nutrition and Foods for Special Dietary Uses. Rome, Italy: Joint FAO/WHO Food Standards Programme. [Google Scholar]
- 6. Brosseau L, Wells GA, Tugwell P et al. (2011) Ottawa panel evidence-based clinical practice guidelines for the management of osteoarthritis in adults who are obese or overweight. Phys Ther 91, 843–861. [DOI] [PubMed] [Google Scholar]
- 7. NSW Agency for Clinical Innovation (2012) Musculoskeletal Network: Osteoarthritis Chronic Care Program Model of Care. Chatswood: NSW. [Google Scholar]
- 8. The Royal Australian College of General Practitioners (2018) Guideline for the Management of Knee and Hip Osteoarthritis. East Melbourne, VIC. [Google Scholar]
- 9. Messier SP, Mihalko SL, Legault C et al. (2013) Effects of intensive diet and exercise on knee joint loads, inflammation, and clinical outcomes among overweight and obese adults with knee osteoarthritis: the IDEA randomized clinical trial. JAMA 310, 1263–1273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Alrushud AS, Rushton AB, Kanavaki AM et al. (2017) Effect of physical activity and dietary restriction interventions on weight loss and the musculoskeletal function of overweight and obese older adults with knee osteoarthritis: a systematic review and mixed method data synthesis. BMJ Open 7, e014537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Chu IJH, Lim AYT & Ng CLW (2018) Effects of meaningful weight loss beyond symptomatic relief in adults with knee osteoarthritis and obesity: a systematic review and meta-analysis. Obes Rev 19, 1597–1607. [DOI] [PubMed] [Google Scholar]
- 12. Hall M, Castelein B, Wittoek R et al. (2018) Diet-induced weight loss alone or combined with exercise in overweight or obese people with knee osteoarthritis: a systematic review and meta-analysis. Semin Arthritis Rheum 48, 765–777. [DOI] [PubMed] [Google Scholar]
- 13. Moher D, Liberati A, Tetzlaff J et al. (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 6, e1000097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Academy of Nutrition and Dietetics (2012) Evidence Analysis Manual. Chicago, IL: Academy of Nutrition and Dietetics. [DOI] [PubMed] [Google Scholar]
- 15. The Cochrane Collaboration (2011) Cochrane Handbook for Systematic Reviews of Interventions. https://training.cochrane.org/hand (accessed October 2017).
- 16. Somers TJ, Blumenthal JA, Guilak F et al. (2012) Pain coping skills training and lifestyle behavioral weight management in patients with knee osteoarthritis: a randomized controlled study. Pain 153, 1199–1209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Aaboe J, Bliddal H, Messier SP et al. (2011) Effects of an intensive weight loss program on knee joint loading in obese adults with knee osteoarthritis. Osteoarthr Cartilage 19, 822–828. [DOI] [PubMed] [Google Scholar]
- 18. Atukorala I, Makovey J, Lawler L et al. (2016) Is there a dose-response relationship between weight loss and symptom improvement in persons with knee osteoarthritis? Arthritis Care Res 68, 1106–1114. [DOI] [PubMed] [Google Scholar]
- 19. Bartels EM, Christensen R, Christensen P et al. (2014) Effect of a 16 weeks weight loss program on osteoarthritis biomarkers in obese patients with knee osteoarthritis: a prospective cohort study. Osteoarthr Cartilage 22, 1817–1825. [DOI] [PubMed] [Google Scholar]
- 20. de Luis DA, Izaola O, Garcia Alonso M et al. (2012) Effect of a hypocaloric diet with a commercial formula in weight loss and quality of life in obese patients with chronic osteoarthritis. Nutr Hosp 27, 1648–1654. [DOI] [PubMed] [Google Scholar]
- 21. Henriksen M, Christensen R, Danneskiold-Samsoe B et al. (2012) Changes in lower extremity muscle mass and muscle strength after weight loss in obese patients with knee osteoarthritis: a prospective cohort study. Arthritis Rheum 64, 438–442. [DOI] [PubMed] [Google Scholar]
- 22. Martin K, Fontaine KR, Nicklas BJ et al. (2001) Weight loss and exercise walking reduce pain and improve physical functioning in overweight postmenopausal women with knee osteoarthritis. J Clin Rheumatol 7, 219–223. [DOI] [PubMed] [Google Scholar]
- 23. Paans N, van den Akker-Scheek I, Dilling RG et al. (2013) Effect of exercise and weight loss in people who have hip osteoarthritis and are overweight or obese: a prospective cohort study. Phys Ther 93, 137–146. [DOI] [PubMed] [Google Scholar]
- 24. Lopez-Gomez JJ, Izaola-Jauregui O, Torres-Torres B et al. (2018) Influence of a meal-replacement diet on quality of life in women with obesity and knee osteoarthritis before orthopedic surgery. Nutr Hosp 35, 71–77. [DOI] [PubMed] [Google Scholar]
- 25. Bliddal H, Leeds AR, Stigsgaard L et al. (2011) Weight loss as treatment for knee osteoarthritis symptoms in obese patients: 1-year results from a randomised controlled trial. Ann Rheum Dis 70, 1798–1803. [DOI] [PubMed] [Google Scholar]
- 26. Christensen R, Astrup A & Bliddal H (2005) Weight loss: the treatment of choice for knee osteoarthritis? a randomized trial. Osteoarthr Cartilage 13, 20–27. [DOI] [PubMed] [Google Scholar]
- 27. Magrans-Courtney T, Wilborn C, Rasmussen C et al. (2011) Effects of diet type and supplementation of glucosamine, chondroitin, and MSM on body composition, functional status, and markers of health in women with knee osteoarthritis initiating a resistance-based exercise and weight loss program. J Int Soc Sports Nutr 8, 8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Messier SP, Loeser RF, Miller GD et al. (2004) Exercise and dietary weight loss in overweight and obese older adults with knee osteoarthritis: the Arthritis, Diet, and Activity Promotion Trial. Arthr Rheum 50, 1501–1510. [DOI] [PubMed] [Google Scholar]
- 29. Messier SP, Loeser RF, Mitchell MN et al. (2000) Exercise and weight loss in obese older adults with knee osteoarthritis: a preliminary study. J Am Geriatr Soc 48, 1062–1072. [DOI] [PubMed] [Google Scholar]
- 30. Miller GD, Nicklas BJ, Davis C et al. Intensive weight loss program improves physical function in older obese adults with knee osteoarthritis. Obesity (Silver Spring). doi: 10.1038/oby.2006.139. [DOI] [PubMed] [Google Scholar]
- 31. Riecke BF, Christensen R, Christensen P et al. (2010) Comparing two low-energy diets for the treatment of knee osteoarthritis symptoms in obese patients: a pragmatic randomized clinical trial. Osteoarthr Cartilage 18, 746–754. [DOI] [PubMed] [Google Scholar]
- 32. O’Brien KM, Wiggers J, Williams A et al. (2018) Telephone-based weight loss support for patients with knee osteoarthritis: a pragmatic randomised controlled trial. Osteoarthr Cartilage 26, 485–494. [DOI] [PubMed] [Google Scholar]
- 33. Hughes SL, Tussing-Humphreys L, Schiffer L et al. (2018) Fit & strong! plus trial outcomes for obese older adults with osteoarthritis. Gerontologist 60, 558–570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Christensen P, Henriksen M, Bartels EM et al. (2017) Long-term weight-loss maintenance in obese patients with knee osteoarthritis: a randomized trial. Am J Clin Nutr 106, 755–763. [DOI] [PubMed] [Google Scholar]
- 35. Christensen R, Henriksen M, Leeds AR et al. (2015) Effect of weight maintenance on symptoms of knee osteoarthritis in obese patients: a twelve-month randomized controlled trial. Arthr Care Res 67, 640–650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Wang X, Miller GD, Messier SP et al. (2007) Knee strength maintained despite loss of lean body mass during weight loss in older obese adults with knee osteoarthritis. J Gerontol A: Biol Sci Med Sci 62, 866–871. [DOI] [PubMed] [Google Scholar]
- 37. Astbury NM, Piernas C, Hartmann-Boyce J et al. (2019) A systematic review and meta-analysis of the effectiveness of meal replacements for weight loss. Obes Rev 20, 569–587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Dietitians Association of Australia (2012) DAA Best Practice Guidelines for the Treatment of Overweight and Obesity in Adults. Canberra, Australia: Dietitians Association of Australia. [Google Scholar]
- 39. National Clinical Guideline Centre (2010) Obesity: Identification, Assessment and Management of Overweight and Obesity in Children, Young People and Adults. London, UK: National Clinical Guideline Centre. [Google Scholar]
- 40. Raynor HA & Champagne CM (2016) Position of the academy of nutrition and dietetics: interventions for the treatment of overweight and obesity in adults. J Acad Nutr Diet 116, 129–147. [DOI] [PubMed] [Google Scholar]
- 41. Messier SP, Resnik AE, Beavers DP et al. (2018) Intentional weight loss in overweight and obese patients with knee osteoarthritis: is more better? Arthr Care Res (Hoboken) 70, 1569–1575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Padilla Colon CJ, Molina-Vicenty IL, Frontera-Rodriguez M et al. (2018) Muscle and bone mass loss in the elderly population: advances in diagnosis and treatment. J Biomed (Syd) 3, 40–49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Christensen R, Bartels EM, Astrup A et al. (2007) Effect of weight reduction in obese patients diagnosed with knee osteoarthritis: a systematic review and meta-analysis. Ann Rheum Dis 66, 433–439. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
For supplementary material accompanying this paper visit https://doi.org/10.1017/S1368980020002529.
click here to view supplementary material