Abstract
Background
Immune checkpoint inhibition (ICI) has changed the melanoma treatment spectrum. Few studies have examined the characteristics and long-term outcomes of patients achieving complete response (CR) under ICI.
Materials and methods
We evaluated patients with unresectable stage IV melanoma treated with first-line ICI. The characteristics of those achieving CR were compared with those not achieving CR. Progression-free survival (PFS) and overall survival (OS) were assessed. Late-onset toxicities, response to second-line treatment, the prognostic value of clinicopathologic features, and blood markers were examined.
Results
A total of 265 patients were included; 41 (15.5%) achieved CR, while 224 (84.5%) had progressive disease, stable disease, or partial response. At the therapy start, those who had CR were more likely to be older than 65 years of age (p = 0.013), have a platelet-to-lymphocyte ratio below 213 (p = 0.036), and have lower lactate dehydrogenase levels (p = 0.008) than those not achieving a CR. For those who discontinued therapy after CR, the median follow-up time after CR was 56 months (interquartile range [IQR] 52–58) and the median time from CR to therapy end was 10 months (IQR 1–17). Five-year PFS after CR was 79% and 5-year OS was 83%. Most complete responders had a normalization of S100 at the time of CR (p < 0.001). In simple Cox regression analysis, age below 77 years at CR (p = 0.04) was associated with better prognosis after CR. Eight patients received second-line ICI; disease control was seen in 63%. Late immune-related toxicities occurred in 25% of patients, most being cutaneous immune-related toxicities.
Conclusions
Response, according to the Response Evaluation Criteria in Solid Tumors (RECIST) criteria, is, until now, the most important prognostic factor, and CR is a valid surrogate marker for long-term survival in patients treated with ICI. Our results highlight the importance of investigating the optimal therapy duration in complete responders.
Graphical Abstract
Supplementary Information
The online version contains supplementary material available at 10.1007/s40257-023-00775-7.
Key Points
| Response, according to the Response Evaluation Criteria in Solid Tumors (RECIST) v1.1, is prognostic for long-term survival, and complete response (CR) is a valid surrogate marker in patients treated with immune checkpoint inhibition. |
| Higher CR rates were seen in patients older than 65 years and in those with lower baseline LDH and platelet/lymphocyte ratio. |
| Among complete responders, 5-year progression-free survival and overall survival after CR were 79% and 83%, respectively. |
| Currently, the optimal duration of anti-PD-1-based therapy in patients achieving complete response is unknown. |
| Better tools are needed to identify patients at higher risk of progression, despite achieving a complete response |
Introduction
Anti-PD-1-based immunotherapy has improved survival in advanced melanoma patients. Therapeutic options in this setting include immune checkpoint inhibitors (ICI) and targeted therapy (TT) with BRAF and MEK inhibitors. One advantage of immunotherapy compared with TT is the sustained response off-treatment [1–4].
Prolonged treatment for up to 24 months affects the quality of life due to potential immune-related adverse events (irAEs) and psychological burden [5]. Longer therapy duration directly impacts the healthcare system through the direct therapy costs, the costs associated with managing irAEs, and the loss of working hours from the patients’ side. Therefore, there is a great need to determine the optimal duration of anti-PD-1-based therapy; however, there is currently no consensus on this matter.
Outside clinical trials, the standard total duration of ICI in advanced unresectable melanoma is 2 years if no progression or severe toxicities are documented. The standard therapy duration is based on the previous clinical trials for melanoma, where immunotherapy was given continuously for up to 2 years. Shorter therapy courses have been associated with less response durability [6]. Other studies showed that the type of response is the most important prognostic factor for survival and that patients without CR should be treated longer [7–10]. This implies that the ideal therapy duration should depend on the type of response and that patients with complete response (CR), partial response (PR), or stable disease (SD) might need different therapy durations. Moreover, this raises the question of whether additional markers, besides the type of response, could more accurately predict progression.
Around 15–25% of patients receiving ICI achieve a CR. A pooled analysis of data from stage III and IV melanoma patients treated with first-line nivolumab or nivolumab plus ipilimumab in the CheckMate 066, CheckMate 067, and CheckMate 069 studies showed that 19% of patients achieved CR under nivolumab and 23% under combined immunotherapy [11]. Another publication showed that 16% of patients with advanced melanoma treated with pembrolizumab achieved a CR and 87.6% exhibited ongoing responses 30 months after achieving CR [12].
The long-term outcomes of patients with CR treated outside clinical trials have not been systematically evaluated, although some publications have addressed this topic [8, 13–15]. Predictors of progression in complete responders have not been broadly examined. Likewise, biomarkers to identify drug-related toxicities associated with prolonged treatment in these patients are missing. Indeed, shorter therapy duration that induces the same type of response could potentially minimize these toxicities [16].
In this study, we assess the characteristics and long-term survival outcomes of a real-world cohort of patients who achieved CR under ICI. We also analyzed potential prognostic biomarkers and immune-related toxicities.
Methods
Study Design and Population
This retrospective, monocentric study includes unresectable stage IV melanoma patients who received ICI (anti-PD1 monotherapy or combination therapy with anti-PD-1 and anti-CTLA-4) as first-line systemic therapy. Patients were treated at the Dermato-Oncology Center of the University of Tuebingen between 2014 and 2018. Additional eligibility criteria were (1) patients who provided informed consent to be enrolled in the Central Malignant Melanoma Registry (CMMR); (2) age above 18 years; (3) patients achieving CR, PR, SD, or progressive disease (PD) under ICI as the best overall response (BOR); and (4) minimum follow-up (FUP) of 2 months after BOR. The exclusion criteria were (1) patients treated in an adjuvant setting; (2) patients treated with anti-CTLA-4 monotherapy as first-line therapy; (3) participation in a clinical trial; (4) patients receiving fewer than two cycles of therapy and not achieving CR or PR; and (5) diagnosis of uveal melanoma (electronic supplementary material [ESM] Fig. 1). Staging was based on the 8th edition of the American Joint Committee on Cancer (AJCC) staging system [17]. The response was assessed using the Response Evaluation Criteria in Solid Tumours (RECIST) version 1.1 [18] approximately 3 months after starting therapy and every 3 months thereafter. The severity of irAEs was documented using the National Cancer Institute’s Common Terminology Criteria for Adverse Events (CTCAE) version 4.03.
Statistical Analysis
Descriptive statistics were reported with median and interquartile range (IQR) for continuous variables, and counts (n) and percentages (%) for categorical variables. Patient characteristics of those who achieved CR were documented at the time of treatment start and compared with those who had PD, SD, or PR. The difference in CR rates between the groups was evaluated using the Chi-square test. The Mann–Whitney U test was used to compare differences in the continuous variables between the two groups.
Age, sex, body mass index (BMI), BRAF variants, histologic subtype, presence of brain or liver metastases, type of ICI received, reason for treatment discontinuation, diagnosis of irAEs, and blood markers were documented and evaluated for their prognostic value. Blood markers included lactate dehydrogenase (LDH), serum protein S100, serum neutrophil-to-lymphocyte ratio (NLR), and serum platelet-to-lymphocyte ratio (PLR). Blood biomarkers were documented at therapy start (T0), at CR (T1), and at the time of progress when applicable (T2). Their relative change was calculated to assess their prognostic significance and investigate their variation over time. Using the median, upper, and lower quartiles or the reference value of our center, we evaluated continuous variables as continuous and categorical. For PLR, the optimal cut-off was determined using the Youden index.
Late-onset irAEs were defined as irAEs occurring 6 months after treatment started. Response to second-line treatment was also documented.
Survival analysis was performed using endpoints of PD for PFS and death for OS, and patients were censored at the last FUP date. The median survival and 5-year survival rates were calculated using the Kaplan–Meier estimator, and hazard ratios (HRs) were estimated using the simple Cox proportional hazards model.
All statistical analyses and data visualization were performed using R (survival, survminer, and swimplot) version 4.1. Reported p values were two-sided and a p value < 0.05 was considered statistically significant.
Results
Patient and Disease Characteristics
Our study included 265 patients; 41 (15.5%) achieved a CR, while 224 (84.5%) had PD, SD or PR. The entire cohort had a median age of 67 years (IQR 56–77) at the therapy start, and 62% were male; 60% received anti-PD-1 monotherapy, while 40% received combination immunotherapy as first-line systemic therapy. At the therapy start, the median LDH was 217 U/L (IQR 185–275) and the median S100 was 0.1 μg/L (IQR 0.06–0.22) [ESM Table 1].
Among those achieving CR and discontinuing therapy thereafter (n = 40), most (70%) were men and the median age at CR was 74 (IQR 66–77). Of these patients, 31 (77.5%) had cutaneous melanoma, 8 (20%) had occult melanoma, and 1 (2.5%) had mucosal melanoma. Twenty-six (65%) patients received anti-PD-1 monotherapy (15% nivolumab, 50% pembrolizumab), while 14 (35%) patients received ipilimumab plus nivolumab.
The median treatment duration was 22 months (IQR 17–24), the median time from therapy start to CR was 6 months (95% confidence interval [CI] 4–8), and the median time from CR to therapy stop was 10 months (IQR 1–17). Five (12%) patients were treated for <6 months, 2 (5%) between 6 and 12 months, 5 (12%) between 12 and 18 months, 20 (50%) between 18 and 24 months, and 8 (20%) for more than 24 months. The reasons for therapy discontinuation were PD (n = 1, 2.5%), severe irAEs (n = 8, 20%), patient/physician decision (n = 17, 42.5%), and therapy discontinuation 2 years after therapy start without evidence of PD (n = 14, 35%). Of these 14 patients, 8 received therapy for more than 2 years and 6 received therapy for 2 years (ESM Table 2). In one patient, therapy is ongoing without evidence of PD.
After achieving CR, PD occurred in 8 (20%) patients within 5 years after CR. Only one patient had disease progression after CR while still receiving therapy, whereas the other seven had disease progression after therapy cessation. One more patient (patient 31) had PD 5 years after achieving CR. Three patients died during the FUP period after PD.
Survival Analysis of Complete Responders
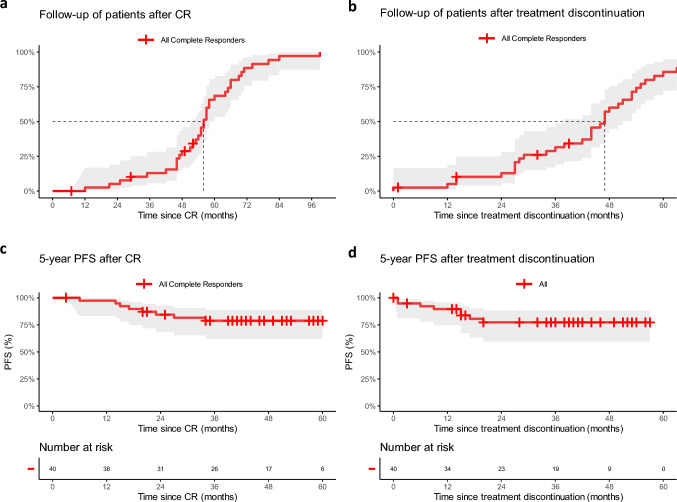
In patients achieving CR and discontinuing therapy thereafter (n = 40), the median FUP (mFUP) time after CR was 56 months (95% CI 52–58), and 47 months (95% CI 38–51) after treatment discontinuation (Fig. 1a, b). Median progression-free survival (mPFS) and median overall survival (mOS) after CR were not reached (NR) [95% CI NR–NR]. The median time to PD after CR was 17 months (95% CI 10–24). After CR, the 5-year PFS rate was 79% (95% CI 62–89%) and the 5-year OS rate was 83% (95% CI 60–93%) (Fig. 1c, d).
Fig. 1.
a Patient FUP using the reverse Kaplan–Meier method of those who achieved a CR under ICI. Time is calculated since CR. The mFUP time after CR was 56 months (95% CI 52–58). b Patient FUP using the reverse Kaplan–Meier method after treatment discontinuation. mFUP time after treatment discontinuation was 47 months (95% CI 38–51). c Five-year PFS analysis of patients (n = 40) who achieved a CR under ICI and subsequently discontinued therapy. The Kaplan–Meier curve is displayed, and time is calculated since achieving CR. The 5-year PFS was 79% (95% CI 62–89%). d Five-year PFS analysis of patients (n = 40) who achieved a CR under ICI and subsequently discontinued therapy. The Kaplan–Meier curve is displayed, and time is calculated since treatment discontinuation. ICI immune checkpoint inhibition, CR complete response, FUP follow-up, mFUP median follow-up, CI confidence interval, PFS progression-free survival
Among those achieving a CR, we saw that patients with CR within 3 months of therapy start did not develop PD, suggesting that a shorter time to CR since therapy start could potentially be associated with a more favorable prognosis (ESM Fig. 1a). However, these differences were not statistically significant, and the number of patients was small to draw any definitive conclusions.
No relation was found between total treatment duration (p = 0.754) or treatment duration after CR (p = 0.398) and survival. Furthermore, the type of immunotherapy, i.e. monotherapy versus combination therapy, was not significantly associated with response durability after CR (p = 0.551) and after therapy cessation (p = 0.663) [ESM Fig. 1b, c]. In the simple Cox regression analysis performed for the different clinicopathologic variables evaluated at the time of CR, age below 77 years (the upper quartile of the cohort) at CR was associated with a better PFS after CR (HR 0.24, 95% CI 0.06–0.95; p = 0.043) and after treatment discontinuation (HR 0.17, 95% CI 0.04–0.73; p = 0.022).
Of the 40 patients who discontinued therapy after CR, 11 had a fluorodeoxyglucose-positron emission tomography/computed tomography (FDG-PET/CT) scan to validate CR, among whom 2 (18%) had PD during FUP.
Comparison between Complete Responders and Non-Complete Responders
Patients who achieved a CR had a median age of 73 years (IQR 65–77), while those who did not achieve CR had a median age of 66 years (IQR 55–77). Complete responders were more likely to be over 65 years of age (Chi-square test, p = 0.013) and less likely to have liver metastasis (Chi-square test, p = 0.032). At treatment start, the median LDH in complete responders was lower than in non-complete responders (Mann–Whitney U test, p = 0.008), and the baseline PLR was more likely to be below 213 (Chi-square test, p = 0.036). The presence of brain metastases was not significantly different between those who had CR and those who did not (p = 0.135) (Table 1).
Table 1.
Patients and disease characteristics of patients with stage IV melanoma who achieved a complete response compared with those with progressive disease, stable disease or partial response
| Complete responders [n = 41]a | Non-complete responders [n = 224]a | CR rate (%) | p valueb | |
|---|---|---|---|---|
| Age, years | ||||
| Median (IQR) | 73 (65–77) | 66 (55–77) | 0.13 | |
| Age at therapy start, years | ||||
| ≤ 65 | 11 (27) | 107 (48) | 9.3 | 0.013 |
| > 65 | 30 (73) | 117 (52) | 20.4 | |
| Sex | ||||
| Female | 12 (29) | 88 (39) | 12 | 0.224 |
| Male | 29 (71) | 136 (61) | 17.6 | |
| First-line systemic therapy | ||||
| Anti-PD-1 monotherapy | 27 (66) | 132 (59) | 17 | 0.405 |
| Combination immunotherapy | 14 (34) | 92 (41) | 13.2 | |
| Brain metastases at therapy start | ||||
| Metastases | 6 (15) | 57 (25) | 9.5 | 0.135 |
| No metastases | 35 (85) | 167 (75) | 17.3 | |
| Liver metastases at therapy start | ||||
| Metastases | 7 (17) | 76 (34) | 8.4 | 0.032 |
| No metastases | 34 (83) | 148 (66) | 18.7 | |
| BRAF mutation | ||||
| BRAF mut | 12 (31) | 58 (30) | 17.1 | 0.898 |
| BRAF wt | 27 (69) | 137 (70) | 16.4 | |
| Missing (n) | 2 | 29 | ||
| LDH at therapy start (U/L) | ||||
| Median (IQR) | 197 (172–226) | 221 (189–279) | 0.008 | |
| Missing (n) | 1 | 20 | ||
| LDH at therapy start (U/L) | ||||
| ≤ 250 | 32 (80) | 134 (66) | 19.3 | 0.076 |
| > 250 | 8 (20) | 70 (34) | 10.3 | |
| Missing (n) | 1 | 20 | ||
| S100 at therapy start (μg/L) | ||||
| Median (IQR) | 0.1 (0.06–0.16) | 0.1 (0.06–0.24) | 0.6 | |
| Missing (n) | 3 | 21 | ||
| S100 at therapy start (μg/L) | ||||
| ≤ 0.1 | 18 (47) | 102 (50) | 15 | 0.745 |
| > 0.1 | 20 (53) | 101 (50) | 16.5 | |
| Missing (n) | 3 | 21 | ||
| BMI, kg/m2 | ||||
| Median (IQR) | 25.7 (22.6–28.5) | 26.3 (23.5–29.7) | 0.4 | |
| Missing (n) | 2 | 47 | ||
| PLR at therapy start | ||||
| Median (IQR) | 165 (132–211) | 195 (145–251) | 0.095 | |
| Missing (n) | 9 | 53 | ||
| PLR at therapy start | ||||
| ≤ 213 | 25 (78) | 100 (58) | 20 | 0.036 |
| > 213 | 7 (22) | 71 (42) | 8.9 | |
| Missing (n) | 9 | 53 | ||
| NLR at therapy start | ||||
| Median (IQR) | 3.2 (2.2–4.2) | 3.6 (2.5–5.3) | 0.2 | |
| Missing (n) | 9 | 53 | ||
BMI body mass index, CR complete response, IQR interquartile range, LDH lactate dehydrogenase, mut mutant, NLR neutrophil-to-lymphocyte ratio, PD-1 programmed death-1, PLR platelet-to-lymphocyte ratio, wt wild-type
aValues are reported as counts (n) and percentages (%) for discrete values, and as median and IQR for continuous values. For comparisons between categorical variables, Pearson’s Chi-square test was used, and for comparisons between continuous and categorical values, the Mann–Whitney U test was used
bThe Chi-square test was used and tested only for patients with information about this certain characteristic; the percentages do not include the patients for whom information was not available. Significant values are reported in bold
When comparing the OS of patients with CR and patients with PR, SD and PD, a statistically significant difference was observed between all subgroups (Fig. 2a). Of note, PFS and OS in patients with PR and CR differed significantly. Patients with PR had a 5-year PFS and OS since therapy initiation of 27% (95% CI 18–38%) and 50% (95% CI 38–61%), respectively, while those achieving CR had a 5-year PFS and 5-year OS of 78% (95% CI 61–88%) and 92% (95% CI 77–97%), respectively (Fig. 2a, b). The mPFS and mOS in patients with PR were 21 months (IQR 16–31) and 55 months (IQR 35–NR), respectively, while both were NR in those having CR.
Fig. 2.
a The 5-year OS analysis of patients according to the response. The Kaplan–Meier curve is displayed, and time is calculated since therapy initiation. b The 5-year PFS analysis of patients according to the response. The Kaplan–Meier curve is displayed, and time is calculated since treatment initiation. OS overall survival, PFS progression-free survival, RECIST Response Evaluation Criteria in Solid Tumors, CR complete response, PR partial response, SD stable disease, PD progressive disease
Blood Biomarkers
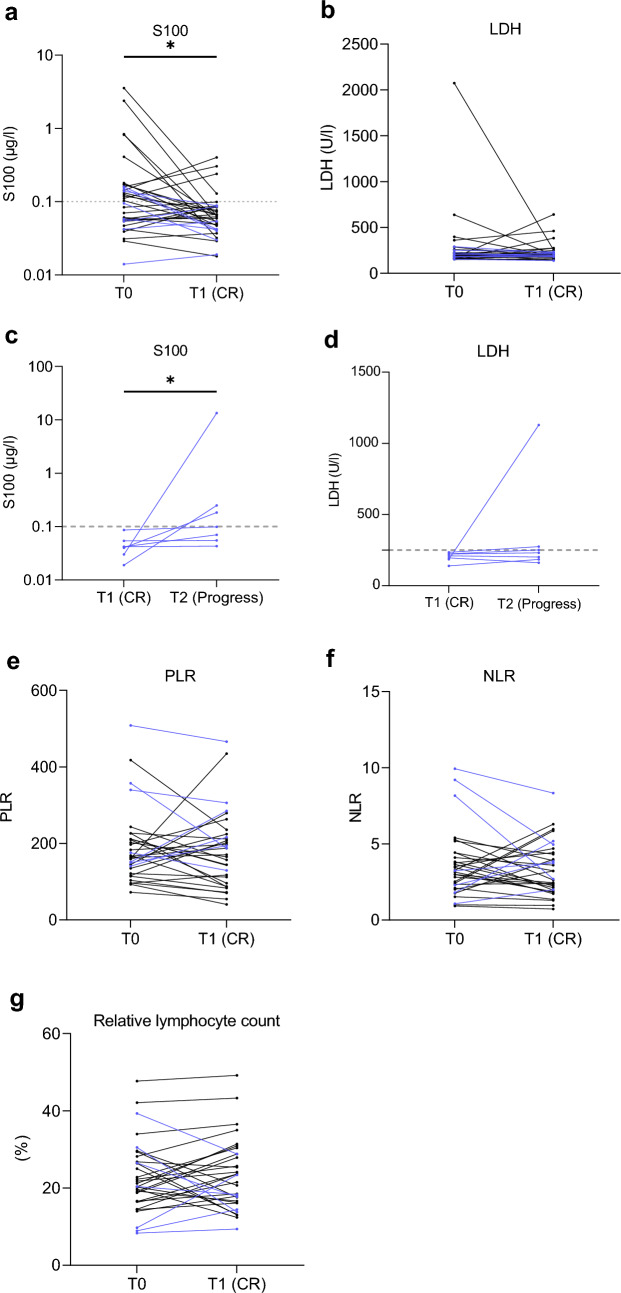
Using the Wilcoxon signed-rank test for matched pairs, we saw a statistically significant decrease in serum S100 (Wilcoxon matched-pairs signed rank test, p = 0.043) from T0 (therapy start) to T1 (time of CR) (Fig. 3a). At the time of CR, there was serum S100 normalization (Fisher test, p < 0.001). Moreover, although not statistically significant, a decrease in serum LDH (median difference = −3, mean difference T1–T0 = −44), PLR and NLR, and an increase in relative lymphocyte count from T0 (therapy start) to T1 (time of CR) was seen (Fig. 3a–g). We subsequently compared the S100 and LDH values at the time of CR (T1) and at the time of progression (T2) when these were available at both time points (n = 7). We saw a significant increase in S100 (p = 0.015) (Fig. 3c), and, although not significant, an increase in LDH was also seen (Fig. 3d).
Fig. 3.
a–g Dot plots showing the variation of blood biomarkers (LDH, S100, PLR, NLR, and relative lymphocyte count) in patients who achieved a CR from baseline value at the time of therapy start (T0) to the time of complete response (T1) and time of progression (T2). Each line represents one patient, and the plots show only patients whose values were reported at both time points. LDH lactate dehydrogenase, PLR platelet-to-lymphocyte ratio, NLR neutrophil-to-lymphocyte ratio, CR complete response
Besides this, we saw that among patients with CR who later had PD, there were patients (n = 3) who had high baseline NLR (above 7), PLR (above 340), and a low baseline relative lymphocyte count. The other patients who progressed (n = 4) and did not have a low baseline relative lymphocyte count did exhibit a decrease in the relative number of lymphocytes and an increase in NLR (Fig. 3a–g).
Rechallenge with Second-Line Immune Checkpoint Inhibitors
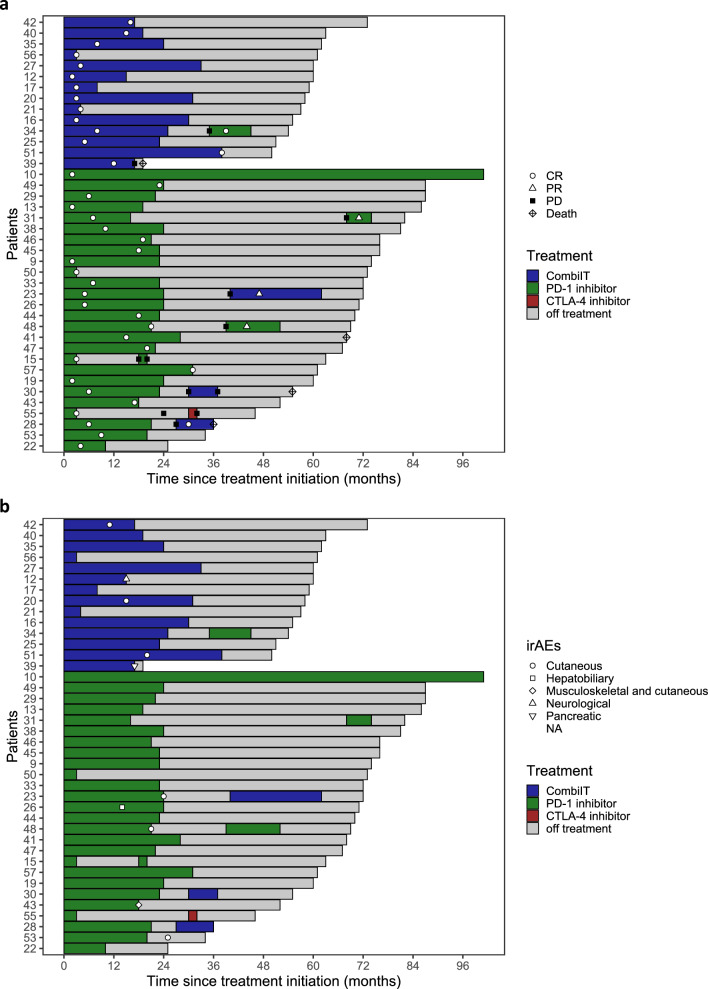
Rechallenge with ICIs was offered to the eight patients with PD after CR; seven (87.5%) patients received anti-PD-1-based immunotherapy and one patient received ipilimumab. Disease control (CR, PR, or SD) was observed in five (63%) patients, while three (37%) patients experienced PD (Fig. 4a).
Fig. 4.
a Patient swimmer plot showing the time patients achieved a CR under first-line immunotherapy. The type of ICI, therapy duration, off-treatment duration, response to rechallenge with second-line immunotherapy, disease progression, and death as events are also depicted. Type of treatment includes PD-1 inhibitor monotherapy (PD-1 inhibitor), CTLA-4 inhibitor monotherapy (CTLA-4 inhibitor), and combined CTLA-4 and PD-1 inhibitor (CombiIT). b Patient swimmer plot showing the late-onset irAEs related to first-line immunotherapy. The time of onset and late-occurring immune-related toxicities are depicted. Type of treatment includes PD-1 inhibitor monotherapy (PD-1 inhibitor), CTLA-4 inhibitor monotherapy (CTLA-4 inhibitor), and combined CTLA-4 and PD-1 inhibitor (CombiIT). CR complete response, PR partial response, SD stable disease, PD progressive disease, CombiIT combination immunotherapy, irAEs immune-related adverse events, ICI immune checkpoint inhibition, PD-1 programmed death-1, CTLA-4 cytotoxic T-lymphocyte-associated protein 4
Immune-Related Adverse Events
Overall, 28 of 40 (70%) patients had an irAE, 15 (37.5%) were cutaneous, and 15 (37.5%) were endocrinological; 8 patients had hepatitis, 7 had gastroenterological irAEs, 5 had respiratory irAEs, and 2 had neurological irAEs. Toxicities that led to therapy discontinuation were as follows: one patient was diagnosed with neuritis, one with meningitis, one with pneumonitis, two with colitis, one with hypophysitis, and one patient with pancreatitis.
Late-onset toxicities occurred in 10 (25%) patients. Nine were diagnosed during treatment, while one developed toxicity after the therapy was stopped. Drug-related adverse events included cutaneous toxicities (n = 7), hepatitis (n = 1), arthritis (n = 1), neuritis (n = 1), and pancreatitis (n = 1). The cutaneous late-onset toxicities identified were bullous pemphigoid (n = 2), vitiligo (n = 2), Grover’s disease (n = 1), rhagade formation (n = 1), and mucositis (n = 1) (Table 2 and Fig. 4b). Four late-onset cutaneous toxicities were identified at or before the time of CR.
Table 2.
Late-onset immune-related adverse events appearing after 6 months of therapy initiation
| ID | Toxicity | Type | Start after therapy (months) | Start after CR (months) | Grade |
|---|---|---|---|---|---|
| 51 | Rhagades | Cutaneous | 20 | −17 | 1 |
| 42 | Bullous pemphigoid | Cutaneous | 11 | −4 | 2 |
| 43 | Arthritis and mucositis | Arthritis and cutaneous | 18 | 0 | 1 |
| 48 | Vitiligo | Cutaneous | 21 | 0 | 1 |
| 39 | Pancreatitisa | Pancreatic | 17 | 5 | 4 |
| 12 | Neuritisa | Neurological | 15 | 12 | 4 |
| 20 | Grover’s disease | Cutaneous | 15 | 12 | 2 |
| 26 | Hepatitis | Hepatobiliary | 14 | 12 | 2 |
| 53 | Bullous pemphigoid | Cutaneous | 25 | 16 | 2 |
| 23 | Vitiligo | Cutaneous | 24 | 22 | 1 |
CR complete response
aThese immune-related adverse events led to therapy discontinuation
Discussion
In our study, with an mFUP of 56 months after CR, patients with CR, according to RECIST 1.1, displayed a survival benefit sustained at the landmark analysis of 5 years. Therefore, we can assume that CR is a surrogate marker for long-term survival in our cohort. Most patients (80%) experienced a durable response after treatment cessation, and neither the overall therapy duration nor the therapy duration after CR were related to survival. These data suggest that in most cases, it is reasonable to discontinue treatment after a CR.
In patients who achieved CR, the median time from therapy start to CR was 6 months, which coincides with the second radiological assessment. Patients with CR within 3 months of therapy initiation (n = 5) did not develop PD, suggesting that an early CR could be more favorable than CR achieved later in the course of therapy; however, the number of patients was low, therefore no definitive conclusions could be drawn.
Robert et al. showed that CR was more common in patients over 65 years of age, and other studies have demonstrated that older age might be associated with response [12, 19]. In mouse experiments, young mice had more T regulatory cells than older mice injected with the same tumors [19]. Moreover, older age has been linked to increased tumor mutation burden and a proinflammatory gut microbiome [20]. In our study, patients achieving CR were more likely to be older than 65 years of age, but patients older than 77 years of age (upper quantile) had a worse survival outcome than younger patients who also achieved CR. Our findings are in line with the meta-analysis of Wu et al., which shows that although people over 65 years of age had a more significant benefit from immunotherapy, no difference in survival was seen for those over 75 years of age [21].
Besides age, our data showed that elevated serum LDH, higher PLR, and liver metastases at the time of treatment start were associated with lower CR rates. LDH depicts the tumor burden, is an already known prognostic marker associated with worse therapy outcomes, and is incorporated into the M category of the AJCC classification of melanoma [22–24]. PLR is a marker of systemic inflammation. NLR, PLR, and C-reactive protein have been associated with a worse prognosis in cancer, including melanoma [25–28]. In our cohort, patients with liver metastases were less likely to have a CR under ICI, which aligns with other working groups showing that different metastatic sites impact therapy outcomes differently. Lee et al. reviewed that the liver microenvironment causes local immunosuppression due to its different types of antigen-presenting cells leading to an unconventional T-cell tolerance, showing that the tumor microenvironment plays a role in melanoma progression [29, 30]. Finally, Pires da Silva et al. showed that liver melanoma metastases had the lowest response rate, and melanoma patients having liver metastases had low objective response rates and worse survival outcomes [31].
In our study, the majority of patients were men, and this overrepresentation is probably due to a selection bias that cannot be overcome due to the retrospective nature of this study.
In our analysis, 20% of patients developed PD—14% received combination ICI and 6% received monotherapy. In Checkmate 066, 067, and 069, up to 30% of patients receiving nivolumab had PD after CR [12]. Therefore, it is critical to find other tools and biomarkers besides response, according to RECIST 1.1, to identify those at risk of progression [32]. It is also important to compare how well the various imaging systems (RECIST, mRECIST, irRC, and iRECIST) identify complete responders in melanoma. Combining these with PET/CT (PET Response Criteria in Solid Tumors [PERCIST] and immunotherapy-modified PERCIST [imPERCIST]), which assesses the tumors metabolically, or performing a pathological assessment of the radiographic remnants, may increase accuracy in predicting long-term survival [33–36]. Dimitriou et al. used PET/CT to evaluate patients with PR according to RECIST 1.1. In this analysis, the authors showed that patients with PR according to RECIST 1.1 and complete metabolic response in PET/CT had a better outcome [34]. Ellebaek et al. also demonstrated that in patients with CR or PR, according to RECIST 1.1, the presence of FDG-avid lesions at the time of therapy discontinuation was an independent predictor of unfavorable survival [36].
The PET STOP trial (NCT04462406) is currently evaluating the use of PET/CT and biopsy of the tumor lesions to decide on the optimal therapy duration. Specifically, patients in this study with a negative FDG-PET/CT scan or a negative biopsy from an avid FDG-PET/CT lesion will discontinue anti-PD-1 therapy (Table 3). Trials evaluating the optimal treatment duration, lower doses of ICI, and the ideal interval between doses in different solid cancers, including melanoma, are ongoing [37–39]. These are expected to shed light on the topic (Table 3).
Table 3.
Clinical trials assessing the optimal duration of immunotherapy in advanced unresectable melanoma patients. All clinical trials are currently recruiting, except the Safe-Stop IPI-NIVO, which has not yet begun to recruit
| Trial | Responsible party | Study type | No. of participants | Intervention | Primary outcome measure | Identifier | |
|---|---|---|---|---|---|---|---|
| SAFE-STOP | Netherlands | Multicenter, single-arm, prospective, interventional | 200 | Early discontinuation of therapy | 6 weeks after CR or PR | Ongoing response rate at 2 years after therapy discontinuation | NL7293 |
| Safe-Stop IPI-NIVO | Erasmus Medical Center (Netherlands) | Single-arm, prospective, interventional | 80 | Early discontinuation of therapy | Upon achieving CR or PR | Ongoing response rate at 1 year after therapy start | NCT05652673 |
| DANTE | UK | Multicenter, randomized, phase III, interventional | 1208 | Early discontinuation of therapy (Arm A) vs. SOC (Arm B) | 12 months after therapy start (arm A) | Progression-free survival at 3, 6, 9, and 12 months after therapy discontinuation | ISRCTN15837212 |
| STOP-GAP | Canadian Clinical Trials Group | Multicenter, randomized, phase III, interventional | 614 | Intermittent therapy when tumor regression (arm A) vs. SOC (Arm B) | Overall survival | NCT02821013 | |
| PET-STOP | Eastern Cooperative Oncology Group/ACRIN group | Multicenter, two-arm, non-randomized, phase II, interventional | 150 | Discontinuation of therapy in patients with disease control and negative PET/CT or biopsy (arm A) | 12 months after therapy start (arm A) | Event-free survival at 1 year after therapy discontinuation | NCT04462406 |
SOC standard of care, CR complete response, PR partial response, PET positron emission tomography, CT computed tomography
Furthermore, as we see that 20% of the patients with CR progress after therapy discontinuation, monitoring them to detect minimal residual disease and progression is essential. Our study shows that serum S100 varied greatly from baseline level to time of CR and time of PD, making S100 a good monitoring marker for serial FUP after therapy discontinuation. We showed a significant decrease in S100 at the time of CR and a significant increase when PD was documented. In the future, the use of technics such as liquid biopsy for the detection of circulating tumor DNA (ctDNA), cell-free DNA (cfDNA), circulating tumor cells (CTCs), microRNAs, extracellular vesicles, antibodies against tumor antigens, and tumor-educated platelets (TEP) could help clinicians when deciding to stop or start therapy [40–46].
Regarding safety, up to 37.5% of patients with CR presented cutaneous toxicities. Previous publications have shown that cutaneous toxicities are associated with improved survival outcomes [47]. Melanoma-associated vitiligo is associated with a good prognosis and shows increased melanoma-specific CD8+ T cells and a high interferon-γ signature, while anti-skin immunoglobulin (Ig) G has been associated with favorable outcomes [48–51]. Specifically, in a prospective observational study of patients developing vitiligo under pembrolizumab, Hua et al. showed that 18% achieved a CR and 53% achieved a PR [52]. Late-onset toxicity was documented in 25% of patients; in most, the irAEs were mild, in line with previous publications [53].
The advantages of our study include its long FUP time of up to 5 years after CR and the inclusion of patients receiving treatment outside of clinical trials, which have restricted the inclusion and exclusion criteria. We focused on the subgroup of patients achieving CR to shed light on the long-term outcomes of this patient subgroup. We included only patients receiving first-line anti-PD-1 monotherapy or combined immunotherapy, as prior treatment lines can influence response durability.
The study’s limitations include its single-center retrospective design and CR being assessed using only RECIST 1.1 criteria.
Conclusions
CR using RECIST 1.1 is a surrogate marker for long-term survival in patients receiving ICI. CR rates seem to be higher in patients older than 65 years of age and those with lower LDH and PLR before therapy started. The optimal therapy duration after achieving CR is unclear and needs to be investigated. Among complete responders, tools are still needed to identify patients at higher risk of PD.
Supplementary Information
Below is the link to the electronic supplementary material.
Declarations
Funding
Open Access funding enabled and organized by Projekt DEAL.
Conflicts of interest/competing interest
Ulrike Leiter reports research support from MSD; consulting fees and honoraria from Sun Pharma, Sanofi (personal and institutional), MSD (personal and institutional), Novartis, Roche, and Almirall Hermal; support for attending meetings from Sun Pharma; and participation on a Data Safety Monitoring Board or Advisory Board from Sun Pharma, Sanofi, MSD, Novartis, Roche, and Almirall Hermal, outside the submitted work. Ioannis Thomas reports honoraria for talks or advisory boards from BMS and Pierre Fabre; and research funding (institutional) from BMS, Pfizer, MSD, Amgen, ARGN-X, LEO, Novartis, UCB, 4SC, AstraZeneca, BioNTech, Genentech, Roche, Biotech, CureVac, HUYA, Incyte, Idera, Iovance, InflaRx, Cerpass, Kartos, Nektar, Philogen, Pierre Fabre, Regeneron, Replimune, and Sanofi. Heike Niessner reports institutional funding from Novartis and Pierre Fabre outside the submitted work. Tobias Sinnberg reports institutional funding from Novartis and Pierre Fabre outside the submitted work. Andrea Forschner served as a consultant to Roche, Novartis, MSD, BMS, and Pierre Fabre; received travel support from Roche, Novartis, BMS, and Pierre Fabre; received speaker fees from Roche, Novartis, BMS, MSD, and CeGaT; and reports institutional research grants from BMS Stiftung Immunonkologie. Lukas Flatz reports grants or contracts from Hookipa Pharma, SAKK/Immunophotonics, DFG Grant (Deutsche Forschungsgemeinschaft), Philogen, and Mundipharma; consulting fees from Philogen, Sanofi, Novartis, and BMS; participation on the Data Safety Board, University of Basel; and stocks or stock options from Hookipa Pharma, outside the submitted work. Teresa Amaral reports personal honoraria from BMS, CeCaVa, Novartis, and Pierre Fabre; institutional financial support from iFIT, NeraCare, Novartis, Sanofi, and SkylineDx, and an institutional research grant from Novartis, outside the submitted work. Eftychia Chatziioannou, Ulrike Keim, Olivia Seeber, Andreas Meiwes, Isabell Boessenecker, Stephanie Sanchez Gonzalez, and Francisco Merraz Torres have no relationships to disclose.
Ethics approval
The Ethics Commission of the Eberhard Karls University Tuebingen approved this retrospective single-center study (number 676/2016BO2).
Consent to participate
Patients provided informed consent to be enrolled in the CMMR and for the use of their clinical data.
Consent for publication
Not applicable.
Data availability
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Code availability
Not applicable.
Author contributions
Study concept: EC, TA. Data collection: EC, TS, HN, TA. Data analysis: EC, TA. Data interpretation: All authors. Writing: All authors. Final approval: All authors. All authors agree to be accountable for all aspects of this work.
References
- 1.McDermott D, Lebbé C, Hodi FS, Maio M, Weber JS, Wolchok JD, et al. Durable benefit and the potential for long-term survival with immunotherapy in advanced melanoma. Cancer Treat Rev. 2014;40(9):1056–1064. doi: 10.1016/j.ctrv.2014.06.012. [DOI] [PubMed] [Google Scholar]
- 2.Ribas A, Hodi FS, Kefford R, Hamid O, Daud A, Wolchok JD, et al. Efficacy and safety of the anti-PD-1 monoclonal antibody MK-3475 in 411 patients (pts) with melanoma (MEL) J Clin Oncol. 2014;32(15 Suppl):LBA9000-LBA. [Google Scholar]
- 3.Schadendorf D, Hodi FS, Robert C, Weber JS, Margolin K, Hamid O, et al. Pooled analysis of long-term survival data from phase II and phase III trials of ipilimumab in unresectable or metastatic melanoma. J Clin Oncol. 2015;33(17):1889–1894. doi: 10.1200/JCO.2014.56.2736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.van Zeijl MCT, van den Eertwegh AJM, Wouters M, de Wreede LC, Aarts MJB, van den Berkmortel F, et al. Discontinuation of anti-PD-1 monotherapy in advanced melanoma-outcomes of daily clinical practice. Int J Cancer. 2022;150(2):317–326. doi: 10.1002/ijc.33800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Marron TU, Ryan AE, Reddy SM, Kaczanowska S, Younis RH, Thakkar D, et al. Considerations for treatment duration in responders to immune checkpoint inhibitors. J Immunother Cancer. 2021;9(3):e001901. doi: 10.1136/jitc-2020-001901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Mäkelä S, Kohtamäki L, Laukka M, Juteau S, Hernberg M. Limited-duration anti-PD-1 therapy for patients with metastatic melanoma. Acta Oncol. 2020;59(4):438–443. doi: 10.1080/0284186X.2020.1716388. [DOI] [PubMed] [Google Scholar]
- 7.Asher N, Israeli-Weller N, Shapira-Frommer R, Ben-Betzalel G, Schachter J, Meirson T, et al. Immunotherapy discontinuation in metastatic melanoma: lessons from real-life clinical experience. Cancers (Basel). 2021;13(12):3074. doi: 10.3390/cancers13123074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Betof Warner A, Palmer JS, Shoushtari AN, Goldman DA, Panageas KS, Hayes SA, et al. Long-term outcomes and responses to retreatment in patients with melanoma treated with PD-1 blockade. J Clin Oncol. 2020;38(15):1655–1663. doi: 10.1200/JCO.19.01464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Jansen YJL, Rozeman EA, Mason R, Goldinger SM, GeukesFoppen MH, Hoejberg L, et al. Discontinuation of anti-PD-1 antibody therapy in the absence of disease progression or treatment limiting toxicity: clinical outcomes in advanced melanoma. Ann Oncol. 2019;30(7):1154–1161. doi: 10.1093/annonc/mdz110. [DOI] [PubMed] [Google Scholar]
- 10.Jansen Y, van der Veldt AAM, Awada G, Neyns B. Anti-PD-1: when to stop treatment. Curr Oncol Rep. 2022;24(7):905–915. doi: 10.1007/s11912-022-01264-6. [DOI] [PubMed] [Google Scholar]
- 11.Robert C, Long G, Larkin J, Wolchok JD, Hassel J, Schadendorf D, et al. 1082MO 5-year characterization of complete responses in patients with advanced melanoma who received nivolumab plus ipilimumab (NIVO+IPI) or NIVO alone. Ann Oncol. 2020;31:S734–S735. [Google Scholar]
- 12.Robert C, Ribas A, Hamid O, Daud A, Wolchok JD, Joshua AM, et al. Durable complete response after discontinuation of pembrolizumab in patients with metastatic melanoma. J Clin Oncol. 2018;36(17):1668–1674. doi: 10.1200/JCO.2017.75.6270. [DOI] [PubMed] [Google Scholar]
- 13.Dimitriou F, Zaremba A, Allayous C, Kähler KC, Gerard CL, Festino L, et al. Sustainable responses in metastatic melanoma patients with and without brain metastases after elective discontinuation of anti-PD1-based immunotherapy due to complete response. Eur J Cancer. 2021;149:37–48. doi: 10.1016/j.ejca.2021.02.037. [DOI] [PubMed] [Google Scholar]
- 14.Saiag P, AitMehdi R, Blom A, Longvert C, Emile J-F, Boru B, et al. Discontinuation of anti-PD-1 mAb after complete response in advanced melanoma pts. J Clin Oncol. 2018;36(15 Suppl):e21549. [Google Scholar]
- 15.De Risi I, Sciacovelli AM, Guida M. Checkpoint inhibitors immunotherapy in metastatic melanoma: when to stop treatment? Biomedicines. 2022;10(10):2424. doi: 10.3390/biomedicines10102424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Dutheil A, Belkadi D, Antigny M, Roy S, Lupu J, Vallet A, et al. Complete responders to checkpoint inhibitors in advanced melanoma: relapse risk factors, and patients' outcomes. J Clin Oncol. 2021;39(15 Suppl):9550. [Google Scholar]
- 17.Gershenwald JE, Scolyer RA, Hess KR, Sondak VK, Long GV, Ross MI, et al. Melanoma staging: Evidence-based changes in the American Joint Committee on Cancer eighth edition cancer staging manual. CA Cancer J Clin. 2017;67(6):472–492. doi: 10.3322/caac.21409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1) Eur J Cancer. 2009;45(2):228–247. doi: 10.1016/j.ejca.2008.10.026. [DOI] [PubMed] [Google Scholar]
- 19.Kugel CH, III, Douglass SM, Webster MR, Kaur A, Liu Q, Yin X, et al. Age correlates with response to anti-PD1, reflecting age-related differences in intratumoral effector and regulatory T-cell populations. Clin Cancer Res. 2018;24(21):5347–5356. doi: 10.1158/1078-0432.CCR-18-1116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Machiraju D, Schäfer S, Hassel JC. Potential reasons for unresponsiveness to anti-PD1 immunotherapy in young patients with advanced melanoma. Life (Basel). 2021;11(12):1318. doi: 10.3390/life11121318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Wu Q, Wang Q, Tang X, Xu R, Zhang L, Chen X, et al. Correlation between patients' age and cancer immunotherapy efficacy. Oncoimmunology. 2019;8(4):e1568810. doi: 10.1080/2162402X.2019.1568810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Espinosa E, Márquez-Rodas I, Soria A, Berrocal A, Manzano JL, Gonzalez-Cao M, et al. Predictive factors of response to immunotherapy-a review from the Spanish Melanoma Group (GEM) Ann Transl Med. 2017;5(19):389. doi: 10.21037/atm.2017.08.10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Weide B, Elsässer M, Büttner P, Pflugfelder A, Leiter U, Eigentler TK, et al. Serum markers lactate dehydrogenase and S100B predict independently disease outcome in melanoma patients with distant metastasis. Br J Cancer. 2012;107(3):422–428. doi: 10.1038/bjc.2012.306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Amaral T, Seeber O, Mersi E, Sanchez S, Thomas I, Meiwes A, et al. Primary resistance to PD-1-based immunotherapy: a study in 319 patients with stage IV melanoma. Cancers. 2020;12(4):1027. doi: 10.3390/cancers12041027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Qi Y, Zhang Y, Fu X, Wang A, Yang Y, Shang Y, et al. Platelet-to-lymphocyte ratio in peripheral blood: a novel independent prognostic factor in patients with melanoma. Int Immunopharmacol. 2018;56:143–147. doi: 10.1016/j.intimp.2018.01.019. [DOI] [PubMed] [Google Scholar]
- 26.Zhou X, Du Y, Huang Z, Xu J, Qiu T, Wang J, et al. Prognostic value of PLR in various cancers: a meta-analysis. PLoS ONE. 2014;9(6):e101119. doi: 10.1371/journal.pone.0101119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kim J-Y, Jung EJ, Kim J-M, Lee HS, Kwag S-J, Park J-H, et al. Dynamic changes of neutrophil-to-lymphocyte ratio and platelet-to-lymphocyte ratio predicts breast cancer prognosis. BMC Cancer. 2020;20(1):1206. doi: 10.1186/s12885-020-07700-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Guida M, Bartolomeo N, Quaresmini D, Quaglino P, Madonna G, Pigozzo J, et al. Basal and one-month differed neutrophil, lymphocyte and platelet values and their ratios strongly predict the efficacy of checkpoint inhibitors immunotherapy in patients with advanced BRAF wild-type melanoma. J Transl Med. 2022;20(1):159. doi: 10.1186/s12967-022-03359-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lee JC, Green MD, Huppert LA, Chow C, Pierce RH, Daud AI. The liver-immunity nexus and cancer immunotherapy. Clin Cancer Res. 2022;28(1):5–12. doi: 10.1158/1078-0432.CCR-21-1193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Chatziioannou E, Aydin SA, Forchhammer S, Sinnberg T, Eigentler T. Makrophagen im Melanom—von molekularen Signalen zur therapeutischen Anwendung. Die Dermatologie. 2022;73(12):915–928. doi: 10.1007/s00105-022-05077-3. [DOI] [PubMed] [Google Scholar]
- 31.Pires da Silva I, Lo S, Quek C, Gonzalez M, Carlino MS, Long GV, et al. Site-specific response patterns, pseudoprogression, and acquired resistance in patients with melanoma treated with ipilimumab combined with anti-PD-1 therapy. Cancer. 2020;126(1):86–97. doi: 10.1002/cncr.32522. [DOI] [PubMed] [Google Scholar]
- 32.Stein JE, Soni A, Danilova L, Cottrell TR, Gajewski TF, Hodi FS, et al. Major pathologic response on biopsy (MPRbx) in patients with advanced melanoma treated with anti-PD-1: evidence for an early, on-therapy biomarker of response. Ann Oncol. 2019;30(4):589–596. doi: 10.1093/annonc/mdz019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Borm FJ, Smit J, Oprea-Lager DE, Wondergem M, Haanen J, Smit EF, et al. Response prediction and evaluation using PET in patients with solid tumors treated with immunotherapy. Cancers (Basel). 2021;13(12):3083. doi: 10.3390/cancers13123083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Dimitriou F, Lo SN, Tan AC, Emmett L, Kapoor R, Carlino MS, et al. FDG-PET to predict long-term outcome from anti-PD-1 therapy in metastatic melanoma. Ann Oncol. 2022;33(1):99–106. doi: 10.1016/j.annonc.2021.10.003. [DOI] [PubMed] [Google Scholar]
- 35.Schank TE, Forschner A, Sachse MM, Dimitrakopoulou-Strauss A, Sachpekidis C, Stenzinger A, et al. Complete metabolic response in FDG-PET-CT scan before discontinuation of immune checkpoint inhibitors correlates with long progression-free survival. Cancers (Basel). 2021;13(11):2616. doi: 10.3390/cancers13112616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ellebaek E, Schina A, Andersen R, Hendel HW, Svane IM, Donia M. Clinical value of routine [18F]2-fluoro-2-deoxy-d-glucose positron emission tomography scans as a decision tool for early immunotherapy discontinuation in advanced melanoma. Int J Cancer. 2022;150(11):1870–1878. doi: 10.1002/ijc.33926. [DOI] [PubMed] [Google Scholar]
- 37.Coen O, Corrie P, Marshall H, Plummer R, Ottensmeier C, Hook J, et al. The DANTE trial protocol: a randomised phase III trial to evaluate the Duration of ANti-PD-1 monoclonal antibody Treatment in patients with metastatic mElanoma. BMC Cancer. 2021;21(1):761. doi: 10.1186/s12885-021-08509-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Mulder E, de Joode K, Litière S, Ten Tije AJ, Suijkerbuijk KPM, Boers-Sonderen MJ, et al. Early discontinuation of PD-1 blockade upon achieving a complete or partial response in patients with advanced melanoma: the multicentre prospective Safe Stop trial. BMC Cancer. 2021;21(1):323. doi: 10.1186/s12885-021-08018-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Hirsch I, Goldstein DA, Tannock IF, Butler MO, Gilbert DC. Optimizing the dose and schedule of immune checkpoint inhibitors in cancer to allow global access. Nat Med. 2022;28(11):2236–2237. doi: 10.1038/s41591-022-02029-1. [DOI] [PubMed] [Google Scholar]
- 40.Amrane K, Le Goupil D, Quere G, Delcroix O, Gouva S, Schick U, et al. Prediction of response to immune checkpoint inhibitor therapy using 18F-FDG PET/CT in patients with melanoma. Medicine (Baltimore) 2019;98(29):e16417. doi: 10.1097/MD.0000000000016417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Kitajima K, Watabe T, Nakajo M, Ishibashi M, Daisaki H, Soeda F, et al. Tumor response evaluation in patients with malignant melanoma undergoing immune checkpoint inhibitor therapy and prognosis prediction using (18)F-FDG PET/CT: multicenter study for comparison of EORTC, PERCIST, and imPERCIST. Jpn J Radiol. 2022;40(1):75–85. doi: 10.1007/s11604-021-01174-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Tan L, Sandhu S, Lee RJ, Li J, Callahan J, Ftouni S, et al. Prediction and monitoring of relapse in stage III melanoma using circulating tumor DNA. Ann Oncol. 2019;30(5):804–814. doi: 10.1093/annonc/mdz048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Vanni I, Tanda ET, Spagnolo F, Andreotti V, Bruno W, Ghiorzo P. The current state of molecular testing in the BRAF-mutated melanoma landscape. Front Mol Biosci. 2020 doi: 10.3389/fmolb.2020.00113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Boyer M, Cayrefourcq L, Dereure O, Meunier L, Becquart O, Alix-Panabières C. Clinical relevance of liquid biopsy in melanoma and merkel cell carcinoma. Cancers. 2020;12:960. doi: 10.3390/cancers12040960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Kamińska P, Buszka K, Zabel M, Nowicki M, Alix-Panabières C, Budna-Tukan J. Liquid biopsy in melanoma: significance in diagnostics, prediction and treatment monitoring. Int J Mol Sci 2021;22(18). [DOI] [PMC free article] [PubMed]
- 46.Pitcovski J, Shahar E, Aizenshtein E, Gorodetsky R. Melanoma antigens and related immunological markers. Crit Rev Oncol Hematol. 2017;115:36–49. doi: 10.1016/j.critrevonc.2017.05.001. [DOI] [PubMed] [Google Scholar]
- 47.Hasan Ali O, Diem S, Markert E, Jochum W, Kerl K, French LE, et al. Characterization of nivolumab-associated skin reactions in patients with metastatic non-small cell lung cancer. OncoImmunology. 2016;5(11):e1231292. doi: 10.1080/2162402X.2016.1231292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Failla CM, Carbone ML, Fortes C, Pagnanelli G, Datri S. Melanoma and vitiligo: in good company. Int J Mol Sci. 2019;20(22):5731. doi: 10.3390/ijms20225731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Byrne KT, Turk MJ. New perspectives on the role of vitiligo in immune responses to melanoma. Oncotarget. 2011;2(9):684–694. doi: 10.18632/oncotarget.323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Zhao J-J, Wen X-Z, Ding Y, Li D-D, Zhu B-Y, Li J-J, et al. Association between immune-related adverse events and efficacy of PD-1 inhibitors in Chinese patients with advanced melanoma. Aging. 2020;12(11):10663–10675. doi: 10.18632/aging.103285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Wu C-E, Yang C-K, Peng M-T, Huang P-W, Chang C-F, Yeh K-Y, et al. The association between immune-related adverse events and survival outcomes in Asian patients with advanced melanoma receiving anti-PD-1 antibodies. BMC Cancer. 2020;20:1018. doi: 10.1186/s12885-020-07508-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Hua C, Boussemart L, Mateus C, Routier E, Boutros C, Cazenave H, et al. Association of vitiligo with tumor response in patients with metastatic melanoma treated with pembrolizumab. JAMA Dermatol. 2016;152(1):45–51. doi: 10.1001/jamadermatol.2015.2707. [DOI] [PubMed] [Google Scholar]
- 53.Carlet C, Dalle S, Leccia M-T, Mortier L, Dalac-Rat S, Dutriaux C, et al. Late-onset adverse events of anti-PD1 therapy in melanoma patients: an observational study from MELBASE, a nationwide prospective cohort. J Am Acad Dermatol. 2022;86(2):345–352. doi: 10.1016/j.jaad.2021.06.849. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data supporting the findings of this study are available from the corresponding author upon reasonable request.