Highlights
-
•
Defensive aggression in wētā is influenced broadly by sex designation and habitat.
-
•
Protected females display high rates of aggression.
-
•
Wētā in non-protected areas are likely to be more wary than protected counterparts.
-
•
Mammalian predator presence within a system influences tree wētā behaviour.
Keywords: Anti-predator behaviour, Behavioural adaptation, Behavioural repeatability, Consistent individual differences, Ecosanctuary, Introduced predator, Predation, Predator-free fencing
Abstract
Insects have evolved a wide range of behavioural traits to avoid predation, with anti-predator behaviours emerging as important adaptive responses to the specific strategies employed by predators. These responses may become ineffective, however, when a species is introduced to a novel predator type. When individuals cannot recognise an introduced predator for instance, they may respond in ways that mean they fail to avoid, escape, or neutralize a predator encounter. New Zealand's endemic insect fauna evolved in the absence of terrestrial mammalian predators for millions of years, resulting in the evolution of unique fauna like the large, flightless Orthopteran, the wētā. Here we investigate how experience with introduced mammalian predators might influence anti-predator behaviours by comparing behaviours in a group of Wellington tree wētā (Hemideina crassidens) living in an ecosanctuary, Zealandia, protected from non-native mammalian predators, and a group living in adjacent sites without mammalian predator control. We used behavioural phenotyping assays with both groups to examine rates of activity and defensive aggression shortly after capture, and again after a period of acclimation. We found that wētā living in protected areas were more active shortly after capture than wētā in non-protected habitats where mammalian predators were present. Male wētā living in non-protected areas tended to be less aggressive than any other group. These results suggest that lifetime experience with differing predator arrays may influence the expression of antipredator behaviour in tree wētā. Disentangling innate and experiential drivers of these behavioural responses further will have important implications for insect populations in rapidly changing environments.
1. Introduction
Prey animals that have evolved alongside predators may display hard-wired defensive traits that help them escape or avoid predation. However, some anti-predator traits may require experience with predators in order to be fully expressed and may not persist with a lack of experience (Blumstein, 2006; Curio, 1993). Similarly, animals that lack shared evolutionary history with predators will display a pattern of naïveté towards those predators when they are present in their environment (Sih et al., 2010). That is, prey may be exposed to ‘novel’ predators, such as invasive species, which do not conform to existing predator archetypes. Together, this means that prey animals that encounter potential predators with which they are unfamiliar may have an inadequate capability to recognise and respond, resulting in delayed anti-predator behavioural responses (Banks and Dickman, 2007; Carthey and Banks, 2014; Cox and Lima, 2006).
Patterns of naïveté in the presence of unfamiliar predators have been observed in a range of species. For example, Iberian anuran tadpoles confronted with chemical cues from predators with whom they evolved reduce their swimming activity while these cues are present in the surrounding water. The same tadpoles fail to react to cues originating from recently introduced terrapins with which they have no prior experience (Polo-Cavia et al., 2010). Similar phenomena has been observed in insect species. When exposed to chemical cues from two species of predatory ladybugs, one co-evolved native and one introduced non-native, a European population of pea aphids (Acyrthosiphon pisum) is able to recognize and avoid native cues; however, they do not respond to cues stemming from non-native ladybugs, and are unable to avoid subsequent predation events (Ünlü et al., 2020). An evolutionarily divergent population of the same species sourced from North America was found to generalize its avoidance strategy, responding to cues from all ladybug species presented. Similarly, larvae of a Canadian damselfly (Enallagma boreale) collected from waterways where a predatory pike is not present fail to recognise environmental cues of pike presence, only decreasing foraging activity when sensing chemical cues from injured conspecifics, whereas larvae gathered from waterways with noted pike populations are able to recognize both sets of cues and decrease their foraging activity in response (Wisenden et al., 1997). These studies demonstrate how a lack of ability to recognise and respond to novel predators can put a species at risk.
In many island systems, including New Zealand, geographical isolation has rendered many native species vulnerable to predation by recently arrived invasive mammals (Blumstein, 2002; Blumstein and Daniel, 2005; Coss, 1999; Duncan and Blackburn, 2004). The activities of humans and accompanying species over the last 800 years in New Zealand have also led to a dramatic reduction in habitat availability, and population size, for many endemic invertebrates (Gibbs, 2009; St Clair, 2011). In these island systems, as well as in other environments with high numbers of endemic species at risk of extinction, fenced ecosanctuaries or aggressive trapping programmes have reduced or eradicated invasive predators for the benefit of native species. These approaches thus provide an environment with a predator array closer to that before introduced species arrival (Christensen et al., 2021; Ringma et al., 2020).
In New Zealand, fifteen fenced and registered ecosanctuaries have been established since the 1980s, with habitats that are protected from invasive mammalian predators. In addition, over one hundred offshore islands have been cleared of rats, stoats, possums and other invasive species (Towns et al., 2013). Although these ecosanctuaries and mammal-free offshore islands play a valuable role in the protection of native animals (Binny et al., 2021; Innes et al., 2019), they create a conundrum. Animals living in these ecosanctuaries, that have neither co-evolved nor have had experience of invasive predators may lose anti-predator traits over time, resulting in behavioural differences between protected groups and those sharing habitats with invasive predators (Muralidhar et al., 2019). For example, Muralidhar et al. showed that endemic South Island robins (Petroica australis) experience rapid loss of antipredator behaviour after relocation from habitat sites with invasive predators (most notably the Norwegian rat, Rattus norvegicus) and the stoat (Mustela erminea) to fenced ecosanctuaries. Likewise, habitat use by a population of tree wētā (Hemideina crassidens) on Nukuwaiata Island in Cook Strait changed markedly after kiore (the Polynesian rat Rattus exulans) populations on the island were eradicated in 1995 (Rufaut and Gibbs, 2003). During the five-year period following kiore eradication, wētā inhabited refuges (cavities or galleries in tree trunks or branches) that had larger entrance holes and were closer to the ground, and spent more time foraging away from their refuges. Adult wētā spent less time guarding refuge entrances. Together, these behaviour changes suggest a more ‘relaxed’ state overall in wētā with no experience of rat predation (Rufaut and Gibbs, 2003). These examples of behavioural changes in animals in ecosanctuaries and on predator-free islands suggest that an end to invasive predator-free status (for example, through damaged fencing in ecosanctuaries, or predator re-invasion of islands) could place protected species at future predation risk.
Here, we examine the behaviour of two groups of tree wētā (H. crassidens), one collected within the predator-free ecosanctuary Zealandia in Wellington (protected wētā), and one collected from adjacent areas in Wellington that did not control invasive mammalian predators (non-protected wētā). We asked if these two groups differed in their rate of activity, and their levels of aggression in response to a simulated predator attack. Specifically, we predicted that both male and female wētā living outside of protected areas, where they are likely to encounter introduced mammalian predators, would express higher levels of defensive aggression and lower levels of activity compared to wētā living in protected areas where introduced predators are absent.
2. Methods
2.1. Study species
Hemideina crassidens (Orthoptera, Anostostomatidae) is a large (3–6 g weight) (Wehi et al., 2013a) omnivorous, nocturnal tree wētā that occupies tree cavities and other refuges in lowland podocarp and broadleaf forests of the lower North Island and West Coast of the South Island of New Zealand. They are also commonly found in urban environments that occupy their original range. H. crassidens juveniles reach maturity between thirteen and eighteen months, and adults have a lifespan of up to 2 years (Field, 2001; Gibbs, 1998). H. crassidens is polygynandrous, and males use their enlarged mandibles to fight for female mates. This species is a common prey item of native predators such as ruru (Ninox novaeseelandiae), tuatara (Sphenodon punctatus), spotted skink (Oligosoma lineoocellatum), at least one large species of spider (Migas sp.), South Island robin (Petroica australis), kākā (Nestor meridionalis), saddleback (Philesturnus sp.), weka (Gallirallus australis), and long-tailed cuckoo (Urodynamis taitensis) (Field, 2001). They are also heavily preyed upon by invasive mammals, including rodents, mustelids, cats (Felis silvestris catus), and hedgehogs (Erinaceus europaeus) (Field, 2001).
Tree wētā behaviour includes striking antipredator displays consisting of the raising and stridulation of spiked hind legs to deter attacks from both predators and conspecifics, and defensive biting (Field, 2001). Although tree wētā remain common across their range, they are closely related to other species of wētā with more vulnerable classifications.
2.2. Collection sites
In November 2019, we collected a group of protected H. crassidens (15 males and 15 females) from artificially constructed refuges (wētā motels, pūriri moth hotels, and gecko foam) inside Zealandia, a 225 ha urban ecosanctuary in Wellington City (Fig. 1). Zealandia was established in 1999 with the express goal of conserving the native flora and fauna of the Wellington region. The ecosanctuary is surrounded by a purpose-designed pest-exclusion fence and regular bait setting and trapping occurs to ensure mammals are unable to establish within its bounds. Despite this, low numbers of mice (Mus musculus) remain within the ecosanctuary, and native predators (birds and reptiles) are, of course, present.
Fig. 1.
Collection sites for Wellington tree wētā from protected and unprotected areas in Wellington city. Unprotected sites are marked in white, and Zealandia sites are marked in green.
We collected a second group of H. crassidens (10 males and 17 females) from artificial refuges (wētā motels) on adjacent residential properties (Fig. 1), including four private properties that were known to have little to no pest control practices in place. Three individuals in the ‘unprotected’ group were collected from a city-owned native bush park, Otari-Wilton's Bush, where pest control practices take place, but which nevertheless does not enjoy the same predator-free status as Zealandia. All collections occurred in the mornings, and then wētā were transferred to facilities for phenotyping, either within Zealandia or at a residential workspace.
Wētā were housed in labelled cardboard holding containers for 2 - 6 h before phenotyping. After phenotyping, wētā collected from Zealandia were released back into the ecosanctuary in accordance with Zealandia's internal research policy. Wētā collected at non-protected sites were kept for further experimentation at the Zoology Department, University of Otago.
2.3. Phenotyping
After a minimum two-hour acclimation period in a darkened workspace, wētā were phenotyped using a series of trials – a filmed open arena test, and a non-filmed trial that tested aggression. We also filmed a refuge-seeking assay; however, only four wētā, one from the non-protected group and three from the protected group, in total entered the refuge so these data are not included here (Kelly, 2021). Four 26×26×26 cm clear plastic tanks with the outer walls blacked out with opaque plastic served as the arenas for the trials, as in a previous study (Parli et al., 2020). We set up a red lamp on either side of the arena setup, so we could conduct trials in the dark. In order to film the trials, we arranged a tripod with a video camera (Sony Handycam DCR-SR47E) above the arenas. Each arena floor was lined with a clean paper towel before each trial to minimise residual pheromones and create a contrast between the wētā and the floor. Each individual wētā was tested independently within a trial arena, with four arenas filmed simultaneously. Test subjects could not see each other during trials. Wētā were phenotyped a second time using the same methods, twenty-four hours after the conclusion of the first set of trials, to estimate repeatability of behaviours.
2.4. Activity
In the open field assay, each wētā was placed in the centre of the arena and covered with an opaque black cylindrical container (7 cm x 10 cm) for 5 min so the wētā could acclimate. After the acclimation period, the cover was removed and each wētā was filmed for 10 min. Once filming was completed, the wētā was re-covered. We positioned the video camera directly above the arenas, ensuring all four spaces were equally in frame and there were no blind spots where the wētā and their movements could not be seen during filming. We analysed video footage using EthoVision XT behavioural quantification software (version 11.5, Noldus Information Technology). We quantified activity as the total distance moved (cm) during the ten-minute trial.
2.5. Defensive aggression
Directly after the filmed phenotyping, we performed a ‘poke test’ to measure defensive aggression responses. Upon the conclusion of the filmed tests wētā were covered again with a black opaque container then moved to the centre of the arena and left to acclimate for 5 min. The cover was removed to initiate the ‘poke-tests’. Wētā remained in the centre of the testing arena during the aggression trial. Each wētā was gently prodded in the centre of the facial plate using a glass rod, until either a response was elicited, or for a maximum of 10 times with two counted seconds between each poke. We recorded the number of ‘pokes’ required to elicit a response, as well as the nature of the behavioural response (e.g. a typical ‘leg raise’ defensive position, mandible gaping, recoil from the stimulus). Behaviours were identified from a compiled ethogram drawn from Parli (2019) and Field (Field, 2001). If no response was elicited after ten ‘pokes’, the trial concluded.
2.6. Data analysis
All statistical analyses for this project were conducted in R version 4.0.2 (R Core Development Team 2020). For the activity and aggression models, we included three fixed effects: group (protected or unprotected), trial type (first or second trial), and sex (male or female), as well all 2 -way interactions and the 3-way interaction. Wētā I.D. was included in all models as a random effect. We analysed activity using a linear mixed effects model (LME), in the nlme package (Pinheiro et al., 2020). Aggression was analysed using a generalised linear mixed model (GLMM) with a Poisson error structure, in the lme4 package (Bates et al., 2015). Model selection was used in all analyses to determine the best-fit model for each dataset. Small sample-size-corrected Akaike Information Criterion (AICc; (Burnham and Anderson, 2002)) determined whether or not interaction terms were included, using the ‘dredge’ function in the ‘MuMIn’ package (Barton, 2018). Parameters were model-averaged within 4 AICc units (when ∆AICc≤4; Symonds and Moussalli 2011) using the ‘model.avg’ function (Burnham and Anderson, 2002). The repeatability of each behaviour was analysed using the ‘rptR’ package (Stofel et al., 2017), with permutations and bootstrap set to 1000.
3. Results
3.1. Activity
Wētā moved on average 77.45 cm over the course of the ten-minute testing period; however, these data include 32% of trials (34 of 106 trials) with no movement from the wētā. During activity trials, one non-protected male moved significantly more than all other individuals across both groups, moving 966 cm, i.e. 275 cm more than any other wētā. This male was subsequently removed from analyses.
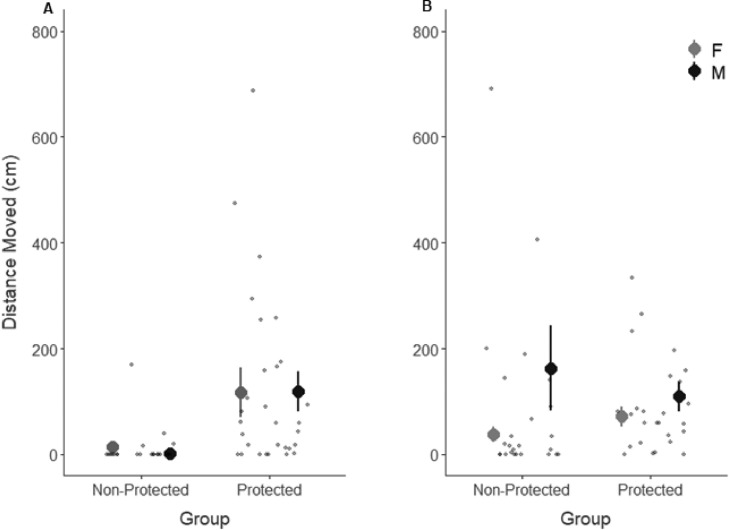
Overall, protected wētā were more active than non-protected wētā (p = <0.01, z = 4.814; Table 1; Fig. 2), though we also found a significant trial effect (p = <0.01, z = 3.007; Table 1) and a significant interaction between location and trial (p = 0.031, Table 1). The difference in activity between groups was evident in the first phenotyping session, two hours after collection (Fig. 2A), during which most non-protected wētā moved very little, whereas after 24 h, both protected and non-protected wētā were active (Fig. 2B). Notably, females in the non-protected group remained inactive, but male non-protected wētā became more active (Fig 2). However, we did not detect any sex effects, nor interactions between location and sex (removed during model selection) or trial and sex (Table 1). The differences between trials was also supported by the repeatability analysis, which showed that distance moved was not repeatable (R = 0.086; CI = [0, 0.365]).
Table 1.
Activity (distanced moved) and aggression (# pokes required to elicit a response) model outputs of Hemideina crassidens. Trial (initial after two hours of acclimation and repeat after twenty-four hours), location (protected habitat within Zealandia ecosanctuary and unprotected sites in the Wellington urban area), and sex were modelled as fixed effects. Data are shown with model estimates, variance (standard errors of fixed effects and variance of random effects), test statistic (z) and significance levels (p) from LMEs using model selection. Significant effects are indicated in bold.
| Model Parameter | Estimate | Variance | z | p |
|---|---|---|---|---|
| Activity | ||||
| Intercept | 0.128 | 0.463 | 0.273 | 0.785 |
| Location | 3.102 | 0.633 | 4.814 | <0.01 |
| Trial | 2.061 | 0.672 | 3.007 | <0.01 |
| Sex | 0.151 | 0.507 | 0.292 | 0.770 |
| Location x Trial | −1.879 | 0.8263 | 2.148 | 0.031 |
| Sex x Trial | 0.754 | 0.840 | 0.847 | 0.380 |
| Aggression | ||||
| Intercept | 1.213 | 0.199 | 6.07 | <0.01 |
| Location | −0.186 | 0.291 | −0.64 | 0.521 |
| Trial | 0.141 | 0.165 | 0.85 | 0.394 |
| Sex | 0.521 | 0.303 | 1.71 | 0.085 |
| Location x Sex | −0.389 | 0.424 | −0.91 | 0.359 |
| Location x Trial | −0.519 | 0.286 | −1.81 | 0.070 |
| Sex x Trial | −0.256 | 0.244 | −1.05 | 0.293 |
| Location x Sex x Trial | 0.962 | 0.384 | 2.50 | 0.012 |
Fig. 2.
Mean ± SE distance moved (cm) of non-protected (n = 9 males, 16 females) and protected (n = 14 males, 14 females) Hemideina crassidens during activity tests. A) The initial ten-minute trial, and B) the ten-minute trial following twenty-four hours of acclimation.
3.2. Defensive aggression
Unlike activity, defensive aggression was significantly repeatable between trials (R = 0.403; CI = [0.046, 0.628]), and trial non-significant in our models (Table 1). We did, however, detect a significant three-way interaction between location, sex, and trial (p = 0.0123, z = 2.503). Protected females were the most aggressive, requiring just three pokes, on average, to elicit an aggressive response, whereas non-protected males were the least aggressive, requiring six pokes, on average, to respond, though this pattern is only evident in the initial trial (Fig. 3).
Fig. 3.
Mean ± SE number of facial pokes required to elicit an aggressive response from non-protected (n = 9 males, 16 females) and protected (n = 14 males, 14 females) Hemideina crassidens. Aggression was measured twice over a twenty-four-hour period. A) Shows the combined results of both trials, B) shows the results of the initial set of trials, while C) shows the results of the second set of trials. Fewer pokes needed to react indicates a higher level of aggression.
4. Discussion
We compared the behaviour of two groups of Wellington tree wētā, Hemideina crassidens, to determine if exposure to mammalian predators during an individual's lifetime influenced behaviour. Protected wētā living inside Zealandia ecosanctuary showed higher rates of activity following initial capture than non-protected wētā, and protected females showed more defensive aggression than their non-protected counterparts. Together, these findings suggest that predator experience may influence the behaviour of H. crassidens.
Protected wētā were more active overall, suggesting that non-protected wētā might be more wary. Wariness is known to increase in species facing strong predation pressure (Lima and Bednekoff, 1999). For example, crickets from populations with predatory Hogna spiders exhibit greater immobility than those from populations where Hogna spiders are not present (Storm and Lima, 2010). Tonic immobility is a widespread anti-predation strategy across taxa (Humphreys and Ruxton, 2018), and may be related to the ‘freeze’ response to common visual predators exhibited by tree wētā (Field, 2001; Moller, 1985). Non-protected wētā may encounter predator cues from a wide range of mammalian predators, such as rats, hedgehogs, cats, and mustelids, as well as from native avian and reptilian predators (Gibbs, 1998). Protected wētā, however, are very unlikely to encounter these invasive mammalian predator assemblages. It is therefore possible that a decrease in costly activity, as well as increased wariness, is advantageous for non-protected wētā. There is an exception, as Zealandia is host to a small population of mice; however densities in the months leading up to our collections were very low (on average 1.481 caught over a 100-day trapping period) and this likely had little to no influence on our results (personal communication).
The behaviour observed in protected weta is consistent with the activity that Rufaut and Gibbs (Rufaut and Gibbs, 2003) observed after the eradication of predators on Nukuwaiata island, where wētā tended to use refuges with larger entrance holes close to the ground and spent more time foraging away from their refuges. In other Orthopterans, wary behaviours are correlated with the intensity of predation pressure acting on a given population. In the field cricket Gryllus integer, males whose mothers were sourced from a population facing pressure from a dense and highly diverse predator array exhibited wary hiding behaviour for significantly longer following a disturbance than males hailing from a comparatively low-predation area (Hedrick and Kortet, 2006). A similar study found that males of another field cricket species, Teleogryllus oceanicus, collected from Hawaiian islands with higher levels of parasitoid fly predation rates, were more wary in their mate-attracting calling than males from populations with lower parasitoid predation rates (Lewkiewicz & Zuz 2004). Both of these studies show that anti-predator behaviour, here in the form of avoidance and reduced conspicuity, is influenced by predator array and density and varies between populations experiencing differing predation pressure.
Introduced mammalian predators to New Zealand, such as rats, frequently hunt using smell, in contrast to the visual hunting of native predators such as owls and lizards (Gibbs, 2010). The initial ‘wary’ behaviour in non-protected males may therefore also potentially result from the ineffective nature of classical aggressive responses against these new mammalian predators. In his study of the defensive behaviours of H. crassidens (Field, 2001), Field found that the defence behaviours of wētā were partially successful against their native predators, but less so against invasive predators. ‘Freeze’ behaviours work well against detection by movement-hunting reptilians, and ‘Leg Raise’ behaviours initiated following capture by a reptilian predator often resulted in the successful escape of the captured wētā as the predator is wounded by the wētā’s leg spines, typically in the ocular region. Behaviours we classified as aggressive displays deterred some avian predators, while cryptic body colouration and a nocturnal lifestyle limit detection by these visual hunters. However, typical displays of aggression showed severely reduced effectiveness when encountering mammalian predators, proving to be virtually useless against stoats and only occasionally successful against rats. Because many behaviours and adaptations that are successful in deterring native predator encounters are rendered ineffective against mammalian predation, the expression of these behaviours in the non-protected population may have been downregulated over time, leaving the population with a reduced tendency toward aggression during a predator-prey interaction. In contrast, the protected group inhabit an environment in which mammalian predators are largely absent, and native species make up the vast majority of predators, and so continue to express behaviours proven to be effective in deterring their extant predator array.
In this study, non-protected males tended to increase their activity when re-tested after twenty-four hours in captivity, whereas females in the same group remained inactive. This result suggests sustained wariness in females over the habituation period. Changes in wētā activity between the first trial and the second trial twenty-four hours later could have been influenced by environmental and temporal factors, including habituation of captive wētā to the testing arenas or to captivity itself. Male Hemideina crassidens range further than females when searching for a mate (C. Kelly, 2006), and this study did take place at the beginning of the breeding season, which could account for the increased movement in non-protected males. Male H. crassidens are more likely to move between refuges (C. Kelly, 2006), whereas female H. crassidens are thought to be largely sedentary within harems, moving only occasionally, in contrast to female activity patterns seen in the related species Hemideina thoracica (PM Wehi et al., 2013b). However, protected males did not alter their movement rates over time, and were instead equally as active as their protected female counterparts. Conversely, the inactivity in the initial trial and subsequent increase in activity shown by non-protected males may reflect an exaggerated response to a perceived major disturbance (i.e. the collection process and travel to testing sites). H. crassidens living in rat-infested areas can develop a heightened threat awareness that results in a reduced rate of movement when threatened (Rufaut, 1995), explaining the disparate behaviours displayed between trials.
There were several aggressive female wētā in the protected group, especially during the second trial. Female tree wētā may become aggressive in instances of gallery overcrowding (Field, 2001), and it is possible that the population density of H. crassidens was higher within Zealandia, in the absence of mammalian predation, than outside the ecosanctuary (Fitzgerald et al., 2021; Ruscoe et al., 2012; Watts et al., 2011). If population numbers are indeed higher within Zealandia, as with other protected areas (Watts et al., 2011), long-term overcrowding of refuges could influence female behaviour, and result in females that are reactive to disturbance. Further work to establish how mobility levels change with habitat as well as a range of perceived threats may therefore be useful. Despite male Hemideina crassidens generally being more aggressive than female wētā (C. Kelly, 2006), we found that male wētā from the non-protected sites tended to be less aggressive than females. The low rates of defensive aggression in non-protected males may be a response to reduced population sizes outside the ecosanctuary environment, where conspecific interactions could be limited by both predation and habitat fragmentation of urban habitat. These factors may lead to reduced male-male competition; research on the closely related H. thoracica suggests that male cohabitation, and male-male interaction, is reduced significantly in areas of low population density compared to areas of high population density wherein large mixed-sex groups are seen to occupy single roosts (Griffin et al., 2020). Furthermore, if non-protected wētā are indeed influenced by a state of hypervigilance as is suggested by Rufaut (Rufaut, 1995), the increase of defensive aggression seen in non-protected males between the first and second aggression trials could be attributed to the apparent reduction in wary behaviour seen over the course of the activity trials.
In this study we did not measure associated habitat-related variables when we collected wētā, although all locations had trees or other cover present. Other studies suggest that the amount of cover, for example, can influence behaviour and assessment of risk (Stankowich and Blumstein, 2005), and further investigation into how the life histories of protected and unprotected wētā might vary in relation to habitat is warranted. Differences in behaviour and phenotypic expression between the two groups could potentially result from the combined influences of habitat quality and predation risk, as well as variable population density and resource availability. However, we did have one suburban non-protected site with few trees (Adelaide), and the wētā from that site behaved more similar to the wētā from the protected site (see Supplementary Material) . The study was also limited by sample size issues, largely due to the short duration of the field season. While increasing sample size in future projects may help eliminate the issues with data variation we experienced in this research, the sample sizes used here were nevertheless within a similar range to some other behavioural studies (C. Kelly, 2006); Bulgarella et al. 2015; Muralidhar et al., 2019; Parli et al., 2020).
Future studies of predator behaviour across a range of protected sites could be useful, although also challenging to interpret in New Zealand, given both the distances between fenced protected ecosanctuaries (for example, Zealandia and Bushy Park are >200 km apart), and differing environmental factors between locations. Nonetheless, similar studies using single-sanctuary areas have produced conclusions which highlight an emerging trend in the loss of boldness in highly-predated, non-protected groups across taxa (Herczeg et al., 2009; Jolly et al., 2021; Moynes et al., 2019; Muralidhar et al., 2019; Roy and Bhat, 2018). For example, an examination of anti-predator behaviour between protected and non-protected populations in Bluegill fish (Lepomis macrochirus) within Lake Opinicon in Ontario, Canada, found that individuals gathered from two protected regions of the waterway (i.e., an Aquatic Protected Area where fisheries are disallowed) were bolder and exhibited shorter flight initiation distances than those gathered from areas of the lake where APA protections did not extend and where predation pressure from human activity is high (Moynes et al., 2019). This, along with the findings of Muralidhar in 2019, show that a loss of anti-predator behaviour is likely in populations isolated from heavy predation pressure, even those which may be close by or occasionally overlapping with non-protected areas.
Overall, the results of this study imply that the behaviour of tree wētā changes with predator array in the wider environment. The results demonstrate that variation in experiences with predators may influence H. crassidens behaviour. Wētā living in non-protected areas were less active and females tended to be more aggressive when compared to protected wētā. If wētā are indeed capable of modifying anti-predator behaviours to contend with the novel predation strategies used by non-native predators, understanding more about the mechanisms underlying these changes could have benefits for an array of threatened species, including translocated invertebrates such as giant wētā.
CRediT authorship contribution statement
Meg Kelly: Conceptualization, Methodology, Investigation, Visualization, Writing – original draft, Writing – review & editing, Formal analysis. Priscilla M Wehi: Conceptualization, Methodology, Writing – review & editing, Formal analysis, Visualization, Supervision, Resources, Funding acquisition. Sheri L Johnson: Conceptualization, Methodology, Writing – review & editing, Supervision.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgements
We thank Peter Ritchie, Alison MacDiarmid, Melinda Divers and Otari-Wilton's Bush for allowing us to collect from the wētā motels on their properties, as well as the Zealandia ecosanctuary who allowed us use of the park as both a collection and research site. Thanks also to Simon Lamb and Eva Kakrada who assisted with wētā collection. The Behavioural Ecology & Evolution Group at Otago, especially Jenny Jandt, Jo Monks, and Stephanie Godfrey, made helpful comments on the manuscript. Chris Garden created the site map. MK was funded by the University of Otago Department of Zoology Master's research fund, and the University of Otago postgraduate publishing bursary. PMW was funded by a Rutherford Discovery Fellowship from the Royal Society of New Zealand (14-LCR-001).
Footnotes
Supplementary material associated with this article can be found, in the online version, at doi:10.1016/j.cris.2023.100058.
Appendix. Supplementary materials
Data Availability
Data will be made available on request.
References
- Banks P., Dickman C. Alien predation and the effects of multiple levels of prey naiveté. Trends Ecol. Evol. (Amst.) 2007;22:229–230. doi: 10.1016/j.tree.2007.02.006. [DOI] [PubMed] [Google Scholar]
- Barton K. (2018) MuMIN: multi-modal Inference. Available at: https://cran.r-project.org/web/packages/MuMIn/MuMIn.pdf
- Binny R., Innes J., Fitzgerald N., Pech R., James A., Price R., Gillies C., Byrom A. Long-term biodiversity trajectories for pest-managed ecological restorations: eradication vs. suppression. Ecol Monogr. 2021;91(2):e01439. [Google Scholar]
- Blumstein D. Moving to suburbia: ontogenetic and evolutionary consequences of life on predator-free islands. J. Biogeogr. 2002;29:685–692. [Google Scholar]
- Blumstein D., Daniel J. The loss of anti-predator behaviour following isolation on islands. Proc. R. Soc. B. 2005;272:1663–1668. doi: 10.1098/rspb.2005.3147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blumstein D. The multipredator hypothesis and the evolutionary persistence of antipredator behaviour. Ethology. 2006;112:209–217. [Google Scholar]
- Burnham K.P., Anderson D.R. 2nd Ed. Springer; New York: 2002. Model Selection and Multimodel inference: a Practical Information-Theoretic Approach. [Google Scholar]
- Carthey A., Banks P. Naïveté in novel ecological interactions: lessons from theory and experimental evidence. Biol. Rev. 2014;89:932–949. doi: 10.1111/brv.12087. [DOI] [PubMed] [Google Scholar]
- Christensen D.L., Harmon K.C., Wehr N.H., Price M.R. Mammal-exclusion fencing improves the nesting success of an endangered native Hawaiian waterbird. PeerJ. 2021;9:e10722. doi: 10.7717/peerj.10722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coss R. In: Geographic Variation in Behavior: Perspectives on Evolutionary Mechanisms. 1st ed. Foster S, Endler J, editors. Oxford University Press; New York: 1999. Effects of relaxed natural selection on the evolution; pp. 180–208. ed. by. [Google Scholar]
- Cox J., Lima S. Naiveté and an aquatic–terrestrial dichotomy in the effects of introduced predators. Trends Ecol. Evol. (Amst.) 2006;21:674–680. doi: 10.1016/j.tree.2006.07.011. [DOI] [PubMed] [Google Scholar]
- Curio E. Advances in the Study of Behaviour. 22 ed. Academic Press, Inc. p; San Diego. California: 1993. Proximate and developmental aspects of antipredator behaviour; pp. 135–238. Slater P, Rosenblatt J, Snowdon C, Milinski M, ed. by. [Google Scholar]
- Bates D., Maechler M., Bolker B., Walker S. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 2015;67(1):1–48. [Google Scholar]
- Duncan R., Blackburn T. Extinction and endemism in the New Zealand avifauna. Global Ecol. Biogeogr. 2004;13:509–517. [Google Scholar]
- Field L. CABI Pub; Wallingford, Oxon., UK: 2001. The Biology of wētās, King Crickets and Their Allies. [Google Scholar]
- Fitzgerald N., Innes J., Watts C., Thornburrow D., Bartlam S., Collins K., Byers D., Burns B. Increasing urban abundance of tūī (Prosthemadera novaeseelandiae) by pest mammal control in surrounding forests. Notornis. 2021;68:93–107. [Google Scholar]
- Gibbs G. Why are some wētā (Orthoptera: stenopelmatidae) vulnerable yet others are common? J. Insect Conserv. 1998;2:161–166. [Google Scholar]
- Gibbs G. The end of an 80-million year experiment: a review of evidence describing the impact of introduced rodents on New Zealand's ‘mammal-free’ invertebrate fauna. Biol. Invasions. 2009;11:1587–1593. [Google Scholar]
- Gibbs G. Do New Zealand invertebrates reflect the dominance of birds in their evolutionary history? N Z J. Ecol. 2010;34(1):152–157. [Google Scholar]
- Griffin M., Holwell G., Symonds M. From quiet-night-in to party animal: sex ratio and density affect male/female aggregations in a ‘harem’ polygynous insect. Anim. Behav. 2020;170:111–118. [Google Scholar]
- Hedrick A.V., Kortet R. Hiding behaviour in two cricket populations that differ in predation pressure. Anim Behav. 2006;72(5):1111–1118. [Google Scholar]
- Herczeg G., Gonda A., Merila J. Predation mediated population divergence in complex behaviour of nine-spined stickleback (Pungitius pungitius) J. Evol. Biol. 2009;22(3):544–552. doi: 10.1111/j.1420-9101.2008.01674.x. [DOI] [PubMed] [Google Scholar]
- Humphreys R., Ruxton G. A review of thanatosis (death feigning) as an anti-predator behaviour. Behav. Ecol. Sociobiol. 2018;72 doi: 10.1007/s00265-017-2436-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Innes J., Fitzgerald N., Binny R., Byrom A., Pech R., Watts C., Gillies C., Maitland M., Campbell-Hunt C., Burns B. New Zealand ecosanctuaries: types, attributes and outcomes. J. R. Soc. N. Z. 2019;49:370–393. [Google Scholar]
- Jolly C.J., Smart A.S., Moreen J., Webb J.K., Gillespie G.R., Phillips B.L. Trophic cascade driven by behavioural fine-tuning as naïve prey rapidly adjust to a novel predator. Ecology. 2021;102(7) doi: 10.1002/ecy.3363. [DOI] [PubMed] [Google Scholar]
- Kelly C. Fighting for harems: assessment strategies during male–male contests in the sexually dimorphic Wellington tree wētā. Anim Behav. 2006;72:727–736. [Google Scholar]
- Kelly C. Movement patterns and gallery use by the sexually dimorphic Wellington tree wētā. N Z J Ecol. 2006;30:273–278. [Google Scholar]
- Kelly M. University of Otago; 2021. Examining Behavioural Differences in Predator Aware and Predator Naïve Wellington tree wētā, Hemideina crassidens. [MSc] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lima S., Bednekoff P. Temporal variation in danger drives antipredator behavior: the predation risk allocation hypothesis. Am. Nat. 1999;153:649–659. doi: 10.1086/303202. [DOI] [PubMed] [Google Scholar]
- Moller H. Tree wētās (Hemideina crassicruris) (Orthoptera: stenopelmatidae) of Stephens Island, Cook Strait. N.Z. J. Zool. 1985;12:55–69. [Google Scholar]
- Moynes Emily N., Lawrence Michael J., Zolderdo Aaron J., Prystay Tanya S., Abrams Alice E.I., Holder Peter, Cooke Steven J. Comparative behavior of wild bluegill captured inside and outside of a long-standing aquatic protected area. Trans. Am. Fish. Soc. 2019;149:174–183. [Google Scholar]
- Muralidhar A., Moore F., Easton L., Jamieson I., Seddon P., van Heezik Y. Know your enemy? Conservation management causes loss of antipredator behaviour to novel predators in New Zealand robins. Anim Behav. 2019;149:135–142. [Google Scholar]
- Parli A., Besson A., Wehi P., Johnson S. Sub-lethal exposure to a mammalian pesticide bait alters behaviour in an orthopteran. J. Insect Conserv. 2020;24:535–546. [Google Scholar]
- Pinheiro J., Bates D., DebRoy S., Sarkar D., R. Core Team (2020). _nlme: linear and Nonlinear Mixed Effects Models_. R package version 3.1-148, URL: https://CRAN.R-project.org/package=nlme.
- Polo-Cavia N., Gonzalo A., López P., Martín J. Predator recognition of native but not invasive turtle predators by naïve anuran tadpoles. Anim Behav. 2010;80:461–466. [Google Scholar]
- Ringma J., Barnes M.D., Bode M. Australian birds could benefit from predator exclusion fencing. Conservation Science and Practice. 2020;2(4) [Google Scholar]
- Roy T., Bhat A. Repeatability in boldness and aggression among wild zebrafish (Danio rerio) from two differing predation and flow regimes. J Comp Psychol. 2018;132(4):349–360. doi: 10.1037/com0000150. [DOI] [PubMed] [Google Scholar]
- Rufaut C. Victoria University of Wellington; 1995. A Comparative Study of the Wellington tree wētā, Hemideina crassidens (Blanchard 1851), in the Presence and Absence of Rodents. [MSc] [Google Scholar]
- Rufaut C., Gibbs G. Response of a Tree Wētā Population (Hemideina crassidens) After Eradication of the Polynesian Rat from a New Zealand Island. Restoration Ecology. 2003;11:13–19. [Google Scholar]
- Ruscoe W., Sweetapple P., Perry M., Duncan R. Effects of spatially extensive control of invasive rats on abundance of native invertebrates in mainland New Zealand forests. Conservation Biology. 2012;27:74–82. doi: 10.1111/j.1523-1739.2012.01932.x. [DOI] [PubMed] [Google Scholar]
- Sih A., Bolnick D., Luttbeg B., Orrock J., Peacor S., Pintor L., Preisser E., Rehage J., Vonesh J. Predator-prey naïveté, antipredator behaviour, and the ecology of predator invasions. Oikos. 2010;119:610–621. [Google Scholar]
- St Clair J. The impacts of invasive rodents on island invertebrates. Biol. Conserv. 2011;144:68–81. [Google Scholar]
- Stankowich T., Blumstein D. Fear in animals: a meta-analysis and review of risk assessment. Proc. R. Soc. B. 2005;272:2627–2634. doi: 10.1098/rspb.2005.3251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stofel M.A., Nakagawa S., Schielzeth H. rptR: repeatability estimation and variance decomposition by generalized linear mixedefects models. Methods Ecol. Evol. 2017;8(11):1639–1644. [Google Scholar]
- Storm J., Lima S. Mothers Forewarn Offspring about Predators: a Transgenerational Maternal Effect on Behaviour. Am. Nat. 2010;175:382–390. doi: 10.1086/650443. [DOI] [PubMed] [Google Scholar]
- Towns D., West C., Broome K. Purposes, outcomes and challenges of eradicating invasive mammals from New Zealand islands: an historical perspective. Wildlife Res. 2013;40(2):94–107. [Google Scholar]
- Ünlü A.G., Obrycki J.J., Bucher R. Comparison of native and non-native predator consumption rates and prey avoidance behaviour in North America and Europe. Ecol. Evol. 2020;10(23):13334–13344. doi: 10.1002/ece3.6932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Watts C., Armstrong D., Innes J., Thornburrow D. Dramatic increases in wētā (Orthoptera) following mammal eradication on Maungatautari – evidence from pitfalls and tracking tunnels. N Z J. Ecol. 2011;35:261–272. [Google Scholar]
- Wehi P., Jorgensen M., Morgan-Richards M. Sex- and season-dependent behaviour in a flightless insect, the Auckland tree wētā (Hemideina thoracica) N Z J. Ecol. 2013;37:75–83. [Google Scholar]
- Wehi P.M., Raubenheimer D., Morgan-Richards M. Tolerance for nutrient imbalance in an intermittently feeding herbivorous cricket, the wellington tree weta. PLoS ONE. 2013;8(12) doi: 10.1371/journal.pone.0084641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wisenden B.D., Chivers D.P., Smith R.J.F. Learned recognition of predation risk by enallagma damselfly larvae (Odonata, Zygoptera) on the basis of chemical cues. J. Chem. Ecol. 1997;23(1):137–151. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data will be made available on request.