ABSTRACT
Leaf spot disease caused by Cercospora beticola Sacc. is the most damaging foliar disease threatening sugar beet production worldwide. The wide spread of disease incurs a reduction of yield and economic losses. The in-depth knowledge of disease epidemiology and virulence factor of pathogen is crucial and basic for preventing fungal disease. The integrated control strategies are needed for an efficient and sustainable disease management. The rotation of fungicides and crop could reduce the initial inoculum and delay the emergence of resistant pathogens. Spraying fungicides under the guide of forecasting models and molecular detecting techniques may hinder the onset of disease prevalence. The resistant varieties of sugar beet to cercospora leaf spot could be obtained by combining classical and molecular breeding methods. More effective approaches are supposed to develop for prevention and control for fungal disease of sugar beet.
KEYWORDS: Sugar beet, leaf spot disease, Cercospora beticola, epidemic, disease management
1. Introduction
Sugar beet (Beta vulgaris L.) domesticated from the sea beet (Beta maritima L.) has been worldwide cultivated in more than 40 countries.1 It is the second most cultivated sugar crop after sugarcane and provides about 20% of the global sugar production.2 Cercospora leaf spot (CLS) caused by C. beticola is one of the destructive plant diseases of sugar beet. It reduces the quality of sugar beet and results in a loss exceeding 30% of the yield. To mitigate the impact of CLS on sugar beet, much progress in understanding the pathogen and its epidemiology has been made by global researchers. The epidemic and pathogenic mechanism of C. beticola, genetical resistance research and disease management were reviewed to summarize the strategies for CLS disease prevention and control.
2. Epidemic of cercospora leaf spot disease
The genus Cercospora was first described by Saccardo in Italy.3 In 1886, the pathogen distribution map, spread and pathogenicity, as well as disease symptoms of CLS were initially documented.4 Several decades after its initial description, the global dissemination of the CLS pathogen emerged as a significant challenge for sugar beet cultivation. According to a recent survey on the distribution of CLS, more than one-third of the global sugar beet area is affected by it. The United States, Austria, Greece, Italy, Hungary, etc. have a high incidence of the disease.5
Cercospora beticola overwinters underground on infected plant residues in the form of pseudostroma. Dry environments are conducive to survival of pathogens. When temperature and humidity levels become suitable in the spring, the conidia begin to generate and invade plants through rain, wind, and insects.6 The abaxial leaf surface and petiole of sugar beet are the primary infection sites. The process of infection initiates with spore germination and then germ tubes penetrate the stomata. After appressorium formation, hyphae spread in mesophyll tissue and then produce toxins,7,8 which cause cell necrosis and leaf spots (Figure 1).
Figure 1.
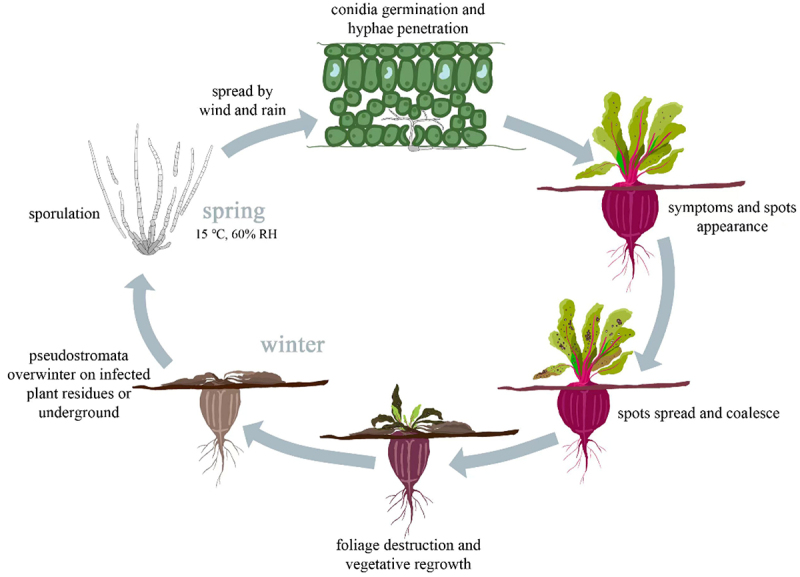
Life cycle of cercospora leaf spot disease of beets.
The spread of conidia mainly occurs in a limited range of agricultural field.9 However, long-distance asexual spread can also occur through machinery, insects, water flow, and other factors. Strains of C. beticola collected from different fields show low level of population differentiation and high genotype duplication.10 The trade of sugar beet also provides an opportunity for further spread of pathogen across different continents.11 During the quality inspection of sugar beet seeds, C. beticola is often detected to exist in the seed coat and then induce CLS along with sowing.12
3. Virulence factors of C. beticola
Cercosporin is a photoactive toxin, which was first isolated by Kuyama in 1957.13 Its toxicity in mice was related to the intensity of light.14 Cercosporin exists widely in the fungi of genus Cercospora. It could stimulate the generation of reactive oxygen species (ROS) in organisms. When ROS was excessive, cell structure collapse was caused by membrane lipid peroxidation and electrolyte leakage.15 The first cercosporin biosynthesis gene CTB1 was found in the mutant of C. nicotianae.16 CbCTB2 encodes a O-methyltransferase, of which the mutant failed to produce cercosporin and colonize sugar beet leaves.17 It proves that cercosporin is not only involved in the initial penetration and colonization but also plays a role in biotrophic phase of the pathogen. Genome sequencing indicated C. beticola held 63 secondary metabolic gene clusters. The CTB gene cluster had taken multiple duplications and horizontal transfer to other pathogenic fungi.18
Beticolin is a non-host-specific phytotoxin. This yellow compound was first named in studying the effect of toxins of C. beticola on cell membrane.19 Twenty beticolin compounds have been found from C. beticola but the biosynthetic pathways and toxicity were still unclear.20,21 In addition to cercosporin and beticolins, there are some potential virulence factors, including cellulases, pectinases and melanin.22 In 2021, Ebert et al. found that C. beticola secreted an effector protein CbNip1 that could penetrate the sugar beet leaves and lead to necrosis. Darkness is essential for full CbNip1-triggered cell necrosis. Infiltration of CbNip1 protein into sugar beet leaves showed that host cell death occurred more rapidly and extensively under dark conditions compared to light conditions.23 It complements photoactivated necrosis formation by cercosporin. The expression of plant pathogenesis-related genes is usually inhibited by darkness, which is benefit for effector proteins to produce toxicity in leaf cells.24 The study on CbNip1 is helpful to illustrate the plant–pathogen interaction process in darkness and further understand the occurrence of CLS.
4. Disease management
The symptom development of CLS on sugar beet crop is a gradual progress. In the first stage, necrotic spots emerge on the primary leaves. As CLS disease progresses, the primary foliage of the plant starts to wilt and fall off. This can lead to a vegetative regrowth to maintain photosynthetic capacity. However, this regrowth is stimulated at the expense of sugar reserves in root. It results in a significant loss of root weight and sugar content, so the control measures for CLS are very necessary for the field management.25,26
4.1. Cropping system
Crop rotation is an important method for reducing the amount of plant pathogens in field. Rotation with non-host crop is a practical strategy for disease management that could reduce initial inoculum load in the next year. At least 2-y rotation is necessary to reduce the levels of initial infection, as shorter rotations may still allow for significant survival of fungal pathogens and result in greater disease prevalence and intensity in sugar beet crops.27,28 In addition, crop rotation can also help improve soil fertility and structure, reduce soil erosion, and manage weeds or pests. Deep tillage may further relieve initial inoculum pressure in the next season. Because spores can disperse long-distance by wind, the sugar beet should be planted far away from the previously infested fields.
4.2. Assessment and monitoring
Image analysis is a technology that collects the images of sugar beet leaves with either a camera or other imaging tools. Software such as Scion Image and MATLAB can then be used to analyze the data.29 Artificial image collection holds the disadvantages of low efficiency and high error rate, while unmanned aerial vehicles (UAVs) and unmanned ground vehicles (UGVs) are high throughput vectors that overcome the defect in fieldwork. Although UAVs are limited to illumination and wind conditions, they could supply the images of high spatial resolution. UGVs can provide high-resolution images to identify the CLS symptoms by short-distance active sensors, but they require strict soil conditions in practical operation.30 ASSESS is a piece of image analysis software that identifies the necrosis region of leaves. It calculates the area by pixels which avoids overestimation of disease severity by visual assessment.31,32 In recent years, deep learning and artificial intelligence technology have offered great convenience to agricultural production. Convolutional neural network (CNN) is a feed-forward neural network constructed by imitating biological visual perception, which holds powerful capabilities in the field of image processing.33 The regression model developed by combining CNN with dataset can process images with the fewest errors.34,35
Polymerase chain reaction (PCR) is highly specific and sensitive, which could be applied for identifying the potential risks of CLS prevalence. It detects the unique DNA fragments of C. beticola from asymptomatic leaves, alternate hosts, and seeds, even spores from artificial traps. It is particularly useful in the initial stage of disease diagnosis and in the prevention of CLS spread.36,37 The enzyme-linked immunosorbent assay (ELISA) is an antigen-based detection method. By combining ELISA with PCR technology, it could not only screen the pathogens in soil quickly but also quantify the mycelial biomass sensitively.38,39
4.3. Chemical and biological treatments
Fungicide is the most effective tool employed in CLS disease prevention. At present, a series of protective and curative fungicides such as dithiocarbamates, benzimidazoles and amines are used by growers for disease control.40 Protective fungicides are locally applied to hinder the infection of conidia, while curative fungicides can be transported between different plant organs. The alternation of different fungicides is encouraged to avoiding the acceleration of resistant strains.41 When the mineral elements (e.g., CuSO4, S and H3BO3) were sprayed mixed with fungicides, the activity of catalase, peroxidase and polyphenolosxidase of sugar beet increased significantly that could ease the damage of plant cells.42
Biological control is an alternative approach that shows the potential for CLS prevention. Trichoderma strains could not only inhibit the growth and sporulation of fungi in vitro, but also cut down the overwintering conidia of C. beticola.43 After inoculating sugar beet with the strains of Bacillus, Emericella and Epicoccum, the severity of CLS was reduced and the activity of chitinase and glucanase all increased. The pathogen-resistant proteins chitinase and glucanase are recognized as two molecular markers associated with pathogen-induced systemic acquired resistance. They have a synergistic effect leading to fungal pathogen control, which is not evident when either occurs independently. The enzymes might reduce fungal disease severity by degrading the chitin and glucan components of fungal cell walls.44,45 Additionally, the isolates of Paenibacillus and Pseudomonas could be used as biocontrol agents for CLS of beet. The activities of peroxidase, polyphenoloxidase and phenylalanine ammonia lyase in beet plants increased significantly after treatment with them.46 Phenylalanine ammonia lyase initiates the phenylpropanoid pathway, which leads to the production of phytoalexins and phenolics. Polyphenoloxidase and peroxidase could induce lignin biosynthesis and oxidative cross-linking of plant cell walls, which can help prevent the spread of pathogens.47,48
4.4. Resistant varieties
Endowing sugar beet varieties with genetic resistance is effective for limiting CLS in field. A series of defensive responses initiated by the related genes expression is responsible for delaying the infection process and disease development.49 As a wild ancestor of sugar beet, sea beet is often used as a source of resistant genes in breeding programs, in which the backcrossing method is often applied.50 Resistance to CLS is inherited in a quantitative mode and 4–5 genes are estimated to be involved.51 These quantitative trait loci (QTLs) that are mostly partially dominant or additive distribute on different chromosomes. The heredity is affected by environmental factors.52 The broad and narrow sense heritabilities for resistance are 70% and 25%, respectively, and the environmental variation range from 44% to 62%.53
In order to select the genotypes of sugar beet that are highly resistant to CLS, the biochemical-molecular substances such as peroxidase, polyphenol oxidase and chitinase are often used as biomarkers.54 Meanwhile, the abundance of bacterial endophytes such as Methylobacterium and Mucilaginibacter can be correlated with increased sensitivity to CLS disease.55 These biomarkers could be employed as indicators for breeding resistant varieties of sugar beet. Compared with the analysis of differential proteins expression, metabolites profile is a more convenient method in practice. 4-Aminobutyrate, fructose and glutamine could differentiate the susceptibility of genotypes.56 In addition, fungitoxic saponins and polyphenolic compounds are associated with higher host resistance.57,58
4.5. Integrated management
Varieties of sugar beet have been developed with genetic resistance to CLS, and these varieties have been used successfully in many parts of the world where sugar beet cultivation faces challenges with this disease. However, the negative association between disease resistance and sugar yield can make it difficult to develop sugar beet varieties that combine both high levels of disease resistance and high productiveness.59 When disease pressure is low or absent, very productive but susceptible sugar beet varieties that receive a modest number of fungicide treatments may perform better than resistant varieties because they have better inherent yield potential.60 When CLS are prevalent and heavy epidemics occur frequently, economic yields typically require the combined use of resistant varieties and repeated fungicide treatments.61 However, it is important to take into account the drawbacks of fungicides, including potential negative effects on human health and the environment, increased production costs, and the development of fungicide-resistant pathogen strains. Field monitoring and epidemic forecasting models are more efficient integrated approaches that could minimize the number of fungicidal treatments. By providing early warning of disease onset and progression, the models allow growers to schedule fungicide treatments more effectively and prevent unnecessary applications. Some models based on weather data calculate the cumulative daily infection value of CLS and predict the infection process.62 Other models take into account the disease incidence, agronomic characteristics and weather data, which assist disease management comprehensively.63,64
5. Future prospects
As mentioned above, enormous progress has been made to manage the cercospora leaf spot disease, which continues to be a serious problem for sugar beet. The advanced and integrated approaches are in great need for production. Spraying fungicides should be with the mode of alternation to reduce the risk of pathogen resistance. Comprehensive models are needed for more precise prediction of disease, which could reduce the use of fungicide.
Molecular methods are useful for detecting the pathogen at the early stage of disease prevalence. It also could determine alternative and asymptomatic hosts that serve as pathogen reservoirs.49 The technologies for biological control of CLS are being deployed. In addition to the microbes that could be used as antagonist to C. beticola, some classes of enzyme isolated from them might detoxify cercosporin.65,66 The plant diseases vaccine such as oligochitosan, which is effective at eliciting plant innate immunity against plant diseases, is a fascinating research direction.67
The transgenic sugar beet has significant potential and commercial value for CLS control. The resistance can be acquired by the expression of transgenes that encode detoxifying enzymes, anti-fungal peptides and phytoalexins.68,69 Gene editing has developed rapidly for crop improvement in recent years. When it is adopted for resistance breeding, the targets are often genes that negatively regulate the anti-disease responses. It also aims to disease responses including the proteins encoding genes that are hijacked by pathogens for infection. To achieve the success that has been attained in wheat resistance breeding for powdery mildew, gene editing techniques will be applied on sugar beet soon.70
Acknowledgments
This research was funded by the National Sugar Beet Germplasm Resources Platform (NCGRC-2021-017); the Ministry of Agriculture and Rural Areas programme (19210157); the Identification, Evaluation, Cataloging and Warehousing of Sugar Beet Germplasm Resources programme (19210911); the Safe Preservation of Sugar Beet Germplasm Resources (19211031); the National Sugar Industry Technology System Project (CARS-170102); the Scientific Research Foundation of the Higher Education Institutions of Heilongjiang Province (2020-KYYWF-1049).
Funding Statement
The work was supported by the Identification, Evaluation, Cataloging and Warehousing of Sugar Beet Germplasm Resources programme [19210911]; Safe Preservation of Sugar Beet Germplasm Resources [19211031]; Ministry of Agriculture and Rural Areas programme [19210157]; Scientific Research Foundation of the Higher Education Institutions of Heilongjiang Province [2020-KYYWF-1049]; National Sugar Beet Germplasm Resources Platform [NCGRC-2021-017]; National Sugar Industry Technology System Project [CARS-170102].
Disclosure statement
No potential conflict of interest was reported by the authors.
References
- 1.Geng G, Yang J.. Sugar beet production and industry in China. Sugar Tech. 2015;17(1):13–6. doi: 10.1007/s12355-014-0353-y. [DOI] [Google Scholar]
- 2.Stevanato P, Chiodi C, Broccanello C, Concheri G, Biancardi E, Pavli O, Skaracis G. Sustainability of the sugar beet crop. Sugar Tech. 2019;21(5):703–716. doi: 10.1007/s12355-019-00734-9. [DOI] [Google Scholar]
- 3.Saccardo PAFVNVC. Fungi Veneti Novi vel Critici. Series V. Nuovo Giornale Botanico Italiano. 1876;8:162–211. [Google Scholar]
- 4.von Thümen F. Die Bekämpfung der Pilzkrankheiten unserer Culturgewächse: Versuch einer Pflanzentherapie zum praktischen Gebrauche für Land-und Forstwirthe, Gärtner, Obst-und Weinzüchter;. Wien: Verlag von Georg Paul Fasey; 1886. [Google Scholar]
- 5.Holtschulte B. Cercospora beticola-worldwide distribution and incidence. Cercospora beticola. 2000;2:5–16. [Google Scholar]
- 6.Pool VW, McKay MB. Climatic conditions as related to Cercospora beticola. J Agric Res. 1916;6:21–60. [Google Scholar]
- 7.Solel Z. Infection process of Cercospora beticola in sugarbeet in relation to susceptibility. Phytopathology. 1971;61(5):463–466. doi: 10.1094/Phyto-61-463. [DOI] [Google Scholar]
- 8.Steinkamp MP, Martin SS, Hoefert LL, Ruppel EG. Ultrastructure of lesions produced by Cercospora beticola in Leaves of Beta vulgaris. Physiol Plant Pathol. 1979;15(1):13–26. doi: 10.1016/0048-4059(79)90035-3. [DOI] [Google Scholar]
- 9.Imbusch F, Liebe S, Erven T, Varrelmann M. Dynamics of cercospora leaf spot disease determined by aerial spore dispersal in artificially inoculated sugar beet fields. null. 2021;70(4):853–861. doi: 10.1111/ppa.13337. [DOI] [Google Scholar]
- 10.Vaghefi N, Kikkert JR, Bolton MD, Hanson LE, Secor GA, Nelson SC, Pethybridge SJ, Chiang T-Y. Global genotype flow in Cercospora beticola populations confirmed through genotyping-by-sequencing. PLos One. 2017;12(10):e0186488. doi: 10.1371/journal.pone.0186488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Knight NL, Vaghefi N, Kikkert JR, Bolton MD, Secor GA, Rivera VV, Hanson LE, Nelson SC, Pethybridge SJ. Genetic diversity and structure in regional Cercospora beticola populations from Beta vulgaris subsp. vulgaris suggest two clusters of separate origin. Phytopathology®. 2019;109(7):1280–1292. doi: 10.1094/phyto-07-18-0264-r. [DOI] [PubMed] [Google Scholar]
- 12.Spanner R, Neubauer J, Heick TM, Grusak MA, Hamilton O, Rivera-Varas V, de Jonge R, Pethybridge S, Webb KM, Leubner-Metzger G, et al. Seedborne cercospora beticola can initiate cercospora leaf spot from sugar beet (Beta vulgaris) fruit tissue. Phytopathology®. 2022;112(5):1016–1028. doi: 10.1094/PHYTO-03-21-0113-R. [DOI] [PubMed] [Google Scholar]
- 13.Kuyama S, Tamura TC. Cercosporin. A pigment of Cercosporina Kikuchii Matsumoto et Tomoyasu. II. Physical and chemical properties of cercosporin and its derivatives. J Am Chem Soc. 1957;79(21):5726–5729. doi: 10.1021/ja01578a039. [DOI] [Google Scholar]
- 14.Yamazaki S, Okubo A, Akiyama Y, Fuwa K. Cercosporin, a novel photodynamic pigment isolated from Cercospora kikuchii. Agric Biol Chem. 1975;39(1):287–288. doi: 10.1271/bbb1961.39.287. [DOI] [Google Scholar]
- 15.Daub ME, Ehrenshaft M. The photoactivated cercospora toxin cercosporin: contributions to plant disease and fundamental biology. Annu Rev Phytopathol. 2000;38(1):461–490. doi: 10.1146/annurev.phyto.38.1.461. [DOI] [PubMed] [Google Scholar]
- 16.Choquer M, Dekkers KL, Chen HQ, Cao L, Ueng PP, Daub ME, Chung KR. The CTB1 gene encoding a fungal polyketide synthase is required for cercosporin biosynthesis and fungal virulence of Cercospora nicotianae. Mol Plant-Microbe Interactions. 2005;18(5):468–476. doi: 10.1094/mpmi-18-0468. [DOI] [PubMed] [Google Scholar]
- 17.Staerkel C, Boenisch MJ, Kröger C, Bormann J, Schäfer W, Stahl D. CbCTB2, an O-methyltransferase is essential for biosynthesis of the phytotoxin cercosporin and infection of sugar beet by Cercospora beticola. BMC Plant Biol. 2013;13(1):50. doi: 10.1186/1471-2229-13-50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.de Jonge R, Ebert MK, Huitt-Roehl CR, Pal P, Suttle JC, Spanner RE, Neubauer JD, Jurick WM II, Stott KA, Secor GA, et al. Gene cluster conservation provides insight into cercosporin biosynthesis and extends production to the genus Colletotrichum. Proc Natl Acad Sci. 2018;115(24):E5459–5466. doi: 10.1186/1471-2229-13-50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Milat ML, Blein JP. Cercospora beticola toxins III. Purification, Thin-layer and aigh performance liquid chromatographic analyses. J Chromatogr A. 1995;699(1–2):277–283. doi: 10.1016/0021-9673(95)00079-3. [DOI] [Google Scholar]
- 20.Goudet C, Milat ML, Sentenac H, Thibaud JBB, Nonpeptidic. Beticolins, nonpeptidic, polycyclic molecules produced by the phytopathogenic fungus cercospora beticola, as a new family of ion channel-forming toxins. Mol Plant-Microbe Interactions. 2000;13(2):203–209. doi: 10.1094/MPMI.2000.13.2.203. [DOI] [PubMed] [Google Scholar]
- 21.Rangel LI, Spanner RE, Ebert MK, Pethybridge SJ, Stukenbrock EH, de Jonge R, Secor GA, Bolton MD. Cercospora beticola: the intoxicating lifestyle of the leaf spot pathogen of sugar beet. Mol Plant Pathol. 2020;21(8):1020–1041. doi: 10.1111/mpp.12962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ebert MK, Spanner RE, de Jonge R, Smith DJ, Holthusen J, Secor GA, Thomma B, Bolton MD. Gene cluster conservation identifies melanin and perylenequinone biosynthesis pathways in multiple plant pathogenic fungi. Environ Microbiol. 2019;21(3):913–927. doi: 10.1111/1462-2920.14475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ebert MK, Rangel LI, Spanner RE, Taliadoros D, Wang X, Friesen TL, de Jonge R, Neubauer JD, Secor GA, Thomma BPHJ, et al. Identification and characterization of Cercospora beticola necrosis-inducing effector CbNip1. Mol Plant Pathol. 2021;22(3):301–316. doi: 10.1111/mpp.13026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Roberts MR, Paul, Paul ND. Seduced by the dark side: integrating molecular and ecological perspectives on the influence of light on plant defence against pests and pathogens. New Phytol. N.D;2006170(4):677–699. doi: 10.1111/j.1469-8137.2006.01707.x. [DOI] [PubMed] [Google Scholar]
- 25.Rossi V, Meriggi P, Biancardi E, Rosso F. Effect of cercospora leaf spot on sugarbeet growth, yield and quality. Cercospora Beticola Sacc Biology, Agronomic Influence And Control Measures In Sugar Beet. 2000:49–76. [Google Scholar]
- 26.Skaracis GN, Pavli OI, Biancardi E. Cercospora leaf spot disease of sugar beet. Sugar Tech. 2010;12(3–4):220–228. doi: 10.1007/s12355-010-0055-z. [DOI] [Google Scholar]
- 27.Khan J, Del Río LE, Nelson R, Khan MFR. Improving the cercospora leaf spot prediction model for sugar beet in Minnesota and North Dakota. Plant Dis. 2007;91(9):1105–1108. doi: 10.1094/PDIS-91-9-1105. [DOI] [PubMed] [Google Scholar]
- 28.Rossi V, Battilani P. CERCOPRI: a forecasting model for primary infections of cercospora leaf spot of sugarbeet1. null. 1991;21(3):527–531. doi: 10.1111/j.1365-2338.1991.tb01284.x. [DOI] [Google Scholar]
- 29.Wijekoon CP, Goodwin PH, Hsiang T. Quantifying fungal infection of plant leaves by digital image analysis using scion image software. J Microbiol Meth. 2008;74(2–3):94–101. doi: 10.1016/j.mimet.2008.03.008. [DOI] [PubMed] [Google Scholar]
- 30.Jay S, Comar A, Benicio R, Beauvois J, Dutartre D, Daubige G, Li W, Labrosse J, Thomas S, Henry N, et al. Scoring cercospora leaf spot on sugar beet: comparison of UGV and UAV phenotyping systems. null. 2020;2020:1–18. doi: 10.34133/2020/9452123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Bock C, Poole G, Parker PE, Gottwald T. Plant disease severity estimated visually, by digital photography and image analysis, and by hyperspectral imaging. Crit Rev Plant Sci. 2010;29(2):59–107. doi: 10.1080/07352681003617285. [DOI] [Google Scholar]
- 32.De Coninck BMA, Amand O, Delauré SL, Lucas S, Hias N, Weyens G, Mathys J, De Bruyne E, Cammue BPA. The use of digital image analysis and Real-time PCR fine-tunes bioassays for quantification of cercospora leaf spot disease in sugar beet breeding. null. 2012;61(1):76–84. doi: 10.1111/j.1365-3059.2011.02497.x. [DOI] [Google Scholar]
- 33.Ozguven MM, Adem K. Automatic detection and classification of leaf spot disease in sugar beet using deep learning algorithms. Physica A. 2019;535:122537. doi: 10.1016/j.physa.2019.122537. [DOI] [Google Scholar]
- 34.Afridi M, Liu X, McGrath J. An automated system for plant-level disease rating in real fields. In Proceedings of the 2014 22nd International Conference on Pattern Recognition, Stockholm, Sweden, 24–28 August 2014. [Google Scholar]
- 35.Liu Q, Xiao L, Yang J, Wei Z. CNN-enhanced graph convolutional network with pixel- and superpixel-level feature fusion for hyperspectral image classification. IEEE T Geosci Remote. 2021;59(10):8657–8671. doi: 10.1109/TGRS.2020.3037361. [DOI] [Google Scholar]
- 36.Lartey RT, Weiland JJ, Caesar-TonThat T, Bucklin-Comiskey S. A PCR protocol for rapid detection of Cercospora beticola in sugarbeet tissues. J Am Soc Sugar Beet Technol. 2003;40(1):1–10. doi: 10.5274/jsbr.40.1.1. [DOI] [Google Scholar]
- 37.Wieczorek TM, Jørgensen LN, Hansen AL, Munk L, Justesen AF. Early detection of sugar beet pathogen Ramularia beticola in leaf and air samples using qPCR. null. 2014;138(4):775–785. doi: 10.1007/s10658-013-0349-6. [DOI] [Google Scholar]
- 38.Caesar-TonThat TC, Lartey RL, Shelver WL. Enzyme-linked immunosorbent assay for Cercospora beticola in soil. J Am Soc Sugar Beet Technol. 2007;44(1):51–70. doi: 10.5274/jsbr.44.1.51. [DOI] [Google Scholar]
- 39.Lartey RT, Caesar-TonThat TC, Lenssen AW, Eckhoff J, Hanson SL, Evans RG. Direct polymerase chain reaction-based detection of cercospora beticola in field soils. Plant Dis. 2010;94(9):1100–1104. doi: 10.1094/PDIS-94-9-1100. [DOI] [PubMed] [Google Scholar]
- 40.Ioanidis PM, Karaoglanidis GS, Lartey RT. Control of cercospora leaf spot and powdery mildew of sugar beet with fungicides and tolerant cultivars. In: Cercospora leaf spot of sugar beet and related species. Lartey RT, Weiland JJ, Panella L, Crous PWWindels CE. APS Press: St. Paul; < MN. 2010. pp. 259–274. [Google Scholar]
- 41.Khan MFR, Smith LJ. Evaluating fungicides for controlling cercospora leaf spot on sugar beet. Crop Protection. 2005;24(1):79–86. doi: 10.1016/j.cropro.2004.06.010. [DOI] [Google Scholar]
- 42.Ghazy N, Shahin AA, Mustafa FA. Effect of some mineral elements on the yield, sugar contents and improving resistance to cercospora leaf spot of sugar beet. Environ Biodivers Soil Secur. 2020;4:73–83. doi: 10.21608/jenvbs.2020.28240.1090. [DOI] [Google Scholar]
- 43.Galletti S, Burzi PL, Cerato C, Marinello S, Sala E. Trichoderma as a potential biocontrol agent for cercospora leaf spot of sugar beet. BioControl. 2008;53(6):917–930. doi: 10.1007/s10526-007-9113-1. [DOI] [Google Scholar]
- 44.Esh AMH, Atia MMM, Tohamy MRA, Taghian S. Systemic resistance in sugar beet eliciated by non-pathogenic, phyllosphere-colonizing Bacillus pumilus and B. subtilus against the pathogen Cercospora beticola sacc. J Plant Prot Pathol. 2011;2(1):67–83. doi: 10.21608/jppp.2011.84657. [DOI] [Google Scholar]
- 45.Esh A, Taghian S. Role of Emericella Nidulans and Epicoccum Nigrum in Controlling sugar beet leaf spot disease. Egypt J Basic Appl Sci. 2021;1(2):55–65. doi: 10.21608/eajast.2021.90993.1001. [DOI] [Google Scholar]
- 46.Sarhan EAD. Induction of induced systemic resistance in fodder beet (Beta vulgaris L.) to cercospora leaf spot caused by (Cercospora beticola Sacc.). Egyptian Journal Of Phytopathology. 2018;46(2):39–59. doi: 10.21608/EJP.2018.91706. [DOI] [Google Scholar]
- 47.Seleim MA, Abo-Elyousr KAM, Mohamed AAA, Al-Marzoky HA. Peroxidase and polyphenoloxidase activities as biochemical markers for biocontrol efficacy in the control of tomato bacterial wilt. J Plant Physiol Pathol. 2014;2(01):2–8. doi: 10.4172/2329-955X.1000117. [DOI] [Google Scholar]
- 48.Nicholson RL, Hammerschmid TR. Phenolic compounds and their role in disease resistance. Ann Rev Phytopathol. 1992;30(1):369–389. [Google Scholar]
- 49.Weiland J, Koch G. Sugarbeet leaf spot disease (Cercospora beticola Sacc.)+. Mol Plant Pathol. 2004;5(3):157–166. doi: 10.1111/j.1364-3703.2004.00218.x. [DOI] [PubMed] [Google Scholar]
- 50.Bilgen T, Gaskill JO, Hecker RJ, Wood DR. Transferring cercospora leaf spot resistance from Beta maritima to sugarbeet by backcrossing. J Am Soc Sugar Beet Technol. 1969;15(5):444–449. doi: 10.5274/jsbr.15.5.444. [DOI] [Google Scholar]
- 51.Smith GA, Gaskill JO. Inheritance of resistance to cercospora leaf spot in sugarbeet. J Am Soc Sugar Beet Technol. 1970;16(2):172–180. doi: 10.5274/jsbr.16.2.172. [DOI] [Google Scholar]
- 52.Setiawan A, Koch G, Barnes SR, Jung C. Mapping quantitative trait loci (QTLs) for resistance to cercospora leaf spot disease (Cercospora beticola Sacc.) in sugar beet (Beta vulgaris L.). Theor Appl Genet. 2000;100(8):1176–1182. doi: 10.1007/s001220051421. [DOI] [Google Scholar]
- 53.Smith GA, Ruppel EG. Herability of resistance to cercospora leaf spot in sugarbeet1. Crop Sci. 1974;14(1):113–115. doi: 10.2135/cropsci1974.0011183x001400010034x. [DOI] [Google Scholar]
- 54.Abd El-Fatah BES, Hashem M, Abo-Elyousr KAM, Khalil Bagy HMM, Alamri SAM. Genetic and biochemical variations among sugar beet cultivars resistant to cercospora leaf spot. Physiol Mol Plant P. 2020;109:101455. doi: 10.1016/j.pmpp.2019.101455. [DOI] [Google Scholar]
- 55.Broccanello C, Ravi S, Deb S, Bolton M, Secor G, Richards C, Maretto L, Lucia MCD, Bertoldo G, Orsini E, et al. Bacterial endophytes as indicators of susceptibility to cercospora leaf spot (CLS) disease in Beta vulgaris L. null. 2022;12(1):10719. doi: 10.1038/s41598-022-14769-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Sekiyama Y, Okazaki K, Kikuchi J, Ikeda S. NMR-based metabolic profiling of field-grown leaves from sugar beet plants harbouring different levels of resistance to cercospora leaf spot disease. Metabolites. 2017;7(1):4. doi: 10.3390/metabo7010004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Schlosser E. A review of some mechanisms of resistance of sugar beet to Cercospora beticola. Journal IIRB. 1969;4:185–191. [Google Scholar]
- 58.Maag GW, Hecker RJ, Payne MG, Remmenga EE, Harrison EM. The interaction of 3-hydroxytyramine and polyphenol oxidase with weight per root and percent sucrose in sugar beets. J Am Soc Sugar Beet Technol. 1967;14(8):709–726. doi: 10.5274/jsbr.14.8.709. [DOI] [Google Scholar]
- 59.Smith GA, Campbell LG. Association between resistance to cercospora and yield in commercial sugarbeet hybrids. Plant Breeding. 1996;115(1):28–32. doi: 10.1111/j.1439-0523.1996.tb00866.x. [DOI] [Google Scholar]
- 60.Mechelke W. Züchtungs-und Sortenstrategien zur Resistenz bei Zuckerrüben gegenüber Cercospora beticola. Zuckerindustrie. 2000;125:688–692. [Google Scholar]
- 61.Skaracis GN, Ioannidis PM, Ioannidis PI. Integrated management systems against sugarbeet cercospora leaf spot disease. Proceedings of the 59th IIRB congress, Brussels. 1996, 45–54. [Google Scholar]
- 62.Pethybridge SJ, Sharma S, Hansen Z, Kikkert JR, Olmstead DL, Hanson LE. Optimizing cercospora leaf spot control in table beet using action thresholds and disease forecasting. Plant Dis. 2020;104(6):1831–1840. doi: 10.1094/pdis-02-20-0246-re. [DOI] [PubMed] [Google Scholar]
- 63.Wolf PFJ, Verreet JA. An integrated pest management system in germany for the control of fungal leaf diseases in sugar beet: the IPM sugar beet model. Plant Dis. 2002;86(4):336–344. doi: 10.1094/pdis.2002.86.4.336. [DOI] [PubMed] [Google Scholar]
- 64.Racca P, Jörg E. CERCBET 3 ? a forecaster for epidemic development of Cercospora beticola. null. 2007;37(2):344–349. doi: 10.1111/j.1365-2338.2007.01136.x. [DOI] [Google Scholar]
- 65.Bargabus RL, Zidack NK, Sherwood JE, Jacobsen BJ. Characterisation of systemic resistance in sugar beet elicited by a non-pathogenic, phyllosphere-colonizing Bacillus mycoides, biological control agent. Biological Control Agent Physiol Mol Plant P. 2002;61(5):289–298. doi: 10.1006/pmpp.2003.0443. [DOI] [Google Scholar]
- 66.Caesar-TonThat TC, Lartey RT, Solberg-Rodier LL, Caesar AJ. Effects of basidiomycete laccase on cercosporin. J Plant Pathol. 2009;91:347–355. [Google Scholar]
- 67.Yin H, Zhao X, Du Y. Oligochitosan: a plant diseases vaccine—a review. Carbohyd Polym. 2010;82(1):1–8. doi: 10.1016/j.carbpol.2010.03.066. [DOI] [Google Scholar]
- 68.Daub ME, Herrero S, Chung KR. Photoactivated perylenequinone toxins in fungal pathogenesis of plants. FEMS Microbiol Lett. 2005;252(2):197–206. doi: 10.1016/j.femsle.2005.08.033. [DOI] [PubMed] [Google Scholar]
- 69.Gao AG, Hakimi SM, Mittanck CA, Wu Y, Woerner BM, Stark DM, Shah DM, Liang J, Rommens CM. Fungal pathogen protection in potato by expression of a plant defensin peptide. Nat Biotechnol. 2000;18(12):1307–1310. doi: 10.1038/82436. [DOI] [PubMed] [Google Scholar]
- 70.Li S, Lin D, Zhang Y, Deng M, Chen Y, Lv B, Li B, Lei Y, Wang Y, Zhao L, et al. Genome-edited powdery mildew resistance in wheat without growth penalties. Nature. 2022;602(7897):455–460. doi: 10.1038/s41586-022-04395-9. [DOI] [PubMed] [Google Scholar]


