Abstract
An increasing number of patients are surviving critical illness, but some experience new or worsening long-lasting impairments in physical, cognitive and/or mental health, commonly known as post-intensive care syndrome (PICS). The need to better understand and improve PICS has resulted in a growing body of literature exploring its various facets. This narrative review will focus on recent studies evaluating various aspects of PICS, including co-occurrence of specific impairments, subtypes/phenotypes, risk factors/mechanisms, and interventions. In addition, we highlight new aspects of PICS, including long-term fatigue, pain, and unemployment.
Keywords: Critical illness, Intensive care, Long-term outcomes
Introduction
Intensive care units (ICUs) were established in the mid-1900s [1, 2]. With advances in life-saving interventions, survival improved over the past decades, positively impacting a large number of patients [1, 3, 4]. However, ICU survivors often report long-lasting impairments in physical, cognitive and/or mental health after hospital discharge [4]. In 2010, the Society of Critical Care Medicine (SCCM) convened an international multi-stakeholder group that created the term “Post-Intensive Care Syndrome” (PICS). PICS was created with multiple objectives, including: (1) to raise awareness among clinicians, patients/families and the general public, (2) to increase screening for specific impairments occurring after critical illness, (3) to facilitate further research into specific morbidities [4]. More specifically, PICS was defined “as new onset or worsening of impairment(s) in physical, cognitive, and/or mental health that arose after the ICU and persisted beyond hospital discharge” [4]. Furthermore, the PICS term can be applied to experiences of a family member (PICS-F) of a survivor of critical illness [4]. It is important to note that PICS is not a medical diagnosis, but a concept for improving education and awareness of post-ICU impairments [4].
Some recent publications highlighted in this narrative review evaluated data from the ARDSNet Long Term Outcomes Study (ALTOS). ALTOS is a multi-center study (including 41 hospitals in the USA) that prospectively examined physical, cognitive and mental health status at 6 and 12 months after Acute Respiratory Distress Syndrome (ARDS). This large study has recently expanded our understanding of PICS with data evaluating ICU survivors’ fatigue, pain, and delayed return to work [5]. In addition to highlighting these new data, this review is to present findings from additional recent PICS-related studies that focus on co-occurrence of specific morbidities, subtypes/phenotypes, risk factors, and interventions.
General updates
Incidence of post-ICU impairments
Determining the incidence of new or worsening impairments after critical illness is challenging due to a lack of data on pre-ICU baseline status [6]. As a result, most studies evaluate the prevalence of post-ICU impairments. However, a recent study of 2,345 ICU survivors in the Netherlands, collected baseline health status via questionnaires completed by patients or their proxies [6]. Among patients urgently admitted to the ICU, patients/proxies rated baseline health status retrospectively, while for those admitted for elective surgery, baseline questionnaires were disseminated at patients’ pre-operative visit and completed a few days before ICU admission [6]. Among those admitted to the ICU for medical (N = 649, 28%), urgent surgery (284, 12%), and elective surgery (1412, 60%), 58%, 64%, 43%, respectively, experienced new physical, cognitive and/or mental problems (Table 1) [6]. Notably, physical problems were measured using a non-validated questionnaire. The incidence of frailty, fatigue, muscle weakness, anxiety, depression, and cognitive impairment at 1 year post-ICU was more common among urgent surgical patients compared to elective surgery [6]. Patients undergoing elective surgery tended to have a shorter ICU length of stay than urgent surgery or medical patients [6]. Additionally, elective surgery patients were more likely to demonstrate improvements in physical and mental functioning at 1 year follow-up; however, baseline fatigue and anxiety were more common in elective surgery patients [6]. Overall, this landmark study provided new insights regarding the incidence of new impairments.
Table 1.
Percentage of patients with new impairments at 1 year, by reason for admission
| New Impairment | Reason for ICU admission | ||
|---|---|---|---|
| Urgent Surgery (n = 284) |
Medical (n = 649) |
Elective Surgery (n = 1,412) |
|
| Fatiguea | 45 | 36 | 24 |
| Depression symptomsb | 20 | 18 | 10 |
| Anxiety symptomsb | 20 | 14 | 9 |
| Frailtyc | 12 | 12 | 4 |
| PTSD symptomsd | 6 | 6 | 4 |
Data from a prospective cohort study across 4 hospitals in the Netherlands between 2016 and 2019 [7]
PTSD: post-traumatic stress disorder
aMeasured using 8-item subscale of the 20-Item Checklist Individual Strength (CIS-20)
bMeasured using the Hospital Anxiety and Depression Scale (HADS)
cMeasured using the Clinical Frailty Scale (CFS)
dMeasured using the Impact of Event Scale-Revised (IES-R)
An earlier smaller-sized study (N = 293) conducted in the United Kingdom (UK) found that ICU survivors experience more mobility issues, self-care issues, pain, and anxiety/depression after the ICU compared to their pre-ICU status based on the EQ-5D subscales [7]. However, this study is limited by potential for recall bias regarding baseline status and by use of only simple one-item assessments in the five subscales within the EQ-5D. Another earlier study (N = 36) conducted in the UK evaluated anxiety and depression symptoms among ICU survivors, excluding patients with pre-existing psychological symptoms; thus attempting to identify new symptoms after critical illness [8]. At 1 month after discharge, they found 16 (44%) and 17 (47%) of participants fell into the “disorder likely” category for anxiety and depression, respectively, based on scores from the Hospital Anxiety and Depression Scale (HADS) [8].
To better understand what long-term impairments are attributed to patients’ critical illness, we need further validation of methods of estimating baseline status [9, 10]. Additionally, future research should focus on evaluating the severity of impairments using continuous measures and via using validated and recommended measurement instruments [11], which would help have greater comparability in research findings and assist in understanding the magnitude of worsening of pre-existing impairments.
Subtypes of physical, cognitive and mental health outcomes
To better understand PICS, researchers have conducted analyses to identify subtypes. From the ALTOS study with 698 ARDS survivors evaluated at 6- and 12-month follow-up, four subtypes were identified via weighted network analysis and recursive partitioning [12]: (1) mildly impaired physical and mental health status (22%), (2) moderately impaired physical and mental health status (39%), (3) severely impaired physical and moderately impaired mental health status (15%), and (4) severely impaired physical and mental health status (24%) [12]. As illustrated by these subtypes, physical and mental impairment, and severity of impairment, demonstrated close associations that were distinct from the presence and severity of cognitive impairment [12]. ICU-related variables and severity of illness were not associated with these subtypes of patient outcomes [12]. Notably, when considering retrospectively-assessed baseline status, patients in all four subtypes demonstrated declines from their baseline status.
Another recent study evaluating clustering of impairments among COVID-19 survivors reported that physical and mental impairments were closely related, but did not co-occur with cognitive impairments [13]. Notably, this study included both ICU and non-ICU patients. Another COVID-19 study evaluating outcomes at 1-year follow-up of ICU survivors reported that cognitive and mental impairments always occurred together [14].
Given common co-occurrence of physical and mental health impairments, future interventions should consider jointly targeting these impairments, such as considered with a novel behavioral activation-rehabilitation (the BEHAB trial) being evaluated via a pilot randomized trial [15]. Furthermore, distinct interventions targeting cognitive impairments are needed.
Risk factors: patient/ICU specific
A multitude of risk factors for PICS-related impairments have been identified along with possible mechanisms for these impairments. A systematic review of 89 publications identified 60 risk factors, with approximately half categorized as patient-related and half as ICU-related [16]. Advanced age, female sex, a history of mental illness, severity of illness, poor ICU patient experience (including negative memories of the ICU), and delirium were significantly associated with physical, mental and/or cognitive impairments [16]. More specifically, a negative ICU patient experience and delirium have a strong impact on anxiety, Post-Traumatic Stress Disorder (PTSD), and cognitive function [16]. Although patient-related variables cannot be altered, they are helpful in identifying patients at highest risk for aspects of PICS. Interventions should target modifiable ICU-related risk factors; for instance, a negative ICU patient experience may be modified by implementing strategies to reduce delirium, increase early mobilization, optimize pain management, and reduce and/or modify the use of restraints [16]. The implementation of these strategies may facilitate alignment with patient-centeredness and improve patients’ ICU experiences; thus, addressing relevant risk factor for post-ICU impairments [17].
Potential mechanisms: inflammatory subphenotypes
Recent research, using data from the ALTOS study, has explored the relationship between ICU-based hyper- vs. hypo-inflammatory subphenotypes with physical, cognitive and mental health impairments over 12-month follow-up [18]. The hyper-inflammatory phenotype was associated with decreased survival within 90 days [18]. However, survival did not differ beyond 90 days based on inflammatory phenotype [18]. Additionally, physical, cognitive, and mental outcomes at 6- and 12-month follow-up were similar across the two inflammatory subphenotypes [18].
Recent research also has demonstrated that acute systemic inflammation and coagulation markers measured early in critical illness are not associated with cognitive function at 3 and 12-month follow-up. Moreover, only 2 markers were associated with disability in activities of daily living over follow-up [19].
Hence, based on these two studies, inflammation during critical illness may not an appropriate mechanistic target for future intervention. However, evaluating associations of prolonged inflammation after hospital discharge with PICS-related impairments merits more investigation [20].
Interventions
A recent systematic review of 36 studies with 5,165 patients, evaluated the effectiveness of non-pharmacological interventions for improving long-term outcomes after critical illness [21]. The study classified interventions into early mobilization and physical rehabilitation (56%), post-ICU follow-up (14%), psychosocial programs (8%), ICU diaries (8%), and educational activities (6%) [21]. Results from each of these 36 studies are summarized in Table 2 [8, 21–56]. Only 31% of these studies included interventions after hospital discharge. Given the prolonged impairments experienced by patients, further studies evaluating the impact of interventions post-discharge are needed [21]. Notably, existing studies have risk of bias from incomplete reporting and loss to follow-up, along with lack of standardization in instruments used to measure outcomes [21]. Hence, further improvement in study design is needed. Overall, the design and evaluation of non-pharmacological interventions targeting aspects of PICS is at an early stage and needs further investigation to improve our understanding of potential efficacy.
Table 2.
Evaluations of non-pharmacological interventions for improving long-term outcomes after critical illness
| Study (County, Year, Sample Sizea) | Summary of results at last follow-up time point (intervention group vs. control group) |
|---|---|
| Pre-Hospital Exercise | |
| Arthur et al. [32] (Canada, 2000, N = 249) |
Hospital LOS [Median (IQR)]: 6 (5–7) vs. 7 (6–8) days; MD (95% CI) 1.0 (0.0 to 1.0), p = 0.002 Mean (SD) change from baseline SF-36 Physical Component Summary Score during the pre-operative phase: 1.6 (7.5) vs. − 1.5 (7.8); MD (95% CI) 3.0 (1.0 to 5.0) |
| In-ICU Exercise/Physical Rehabilitation | |
| Wright et al. [43] (UK, 2018, N = 308) | Mean (SD) SF-36 Physical Component Summary Score at 6 months: 37 (12) vs. 37 (11). MD (95% CI) − 1.1 (− 7.1 to 5.0) |
| Morris et al. [51] (USA, 2016, N = 300) |
Mean (95% CI) SF-36 Physical Functioning scale score at 6 months: 56 (50–62) vs. 44 (38–50). MD (95% CI) 12 (3.8 to 21), p = 0.001 Mean (95% CI) Functional Performance Inventory score at 6 months: 2.2 (2.1–2.4) vs. 2.0 (1.9–2.2). MD (95% CI) 0.2 (0.04 to 0.4), p = 0.02 Mean (95% CI) Short Physical Performance Battery at 6 months: 9.0 (8.3–9.7) vs. 8.0 (7.2–8.7). MD (95% CI) 1.1 (0.04 to 2.1), p = 0.04 |
| Hodgson et al. [52] (Australia/New Zealand, 2016, N = 50) |
Mean (SD) EQ-5D score at 6 months: 0.63 (0.27) vs. 0.63 (0.33), p = 0.25 Mean (SD) IADL score at 6 months: 7 (2) vs. 7 (1), p = 0.81 Mean (SD) HADS score at 6 months: 12 (9) vs. 11 (7), p = 0.91 |
| Schaller et al. [53] (Austria/Germany/USA, 2016, N = 200) | Median (IQR) mini-modified FIM at hospital discharge: 8 (4–8) vs. 5 (2–8). MD (95% CI) 3.0 (1.0 to 4.0), p = 0.0002 |
| Kayambu et al. [54] (Australia, 2015, N = 50) |
Mean (SD) SF-36 Physical function score at 6 months: 82 (22) vs. 60 (29), p = 0.04 Mean (SD) SF-36 Physical role score at 6 months: 61 (44) vs. 17 (34), p = 0.005 |
| Moss et al. [55] (USA, 2016, N = 120) | Mean (SD) Continuous Scale Physical Functional Performance Test short form (CS-PFP-10) at 6 months: 40 (4) vs. 44 (4), p = 0.43 |
| Denehy et al. [56] (Australia, 2013, N = 150) |
Mean (SE) 6MWT (meters) at 12 months: 410 (23) vs. 405 (23). MD (95% CI) 4.7 (− 60 to 69), p = 0.88 Mean (SD) TUG (seconds) at 12 months: 10 (6.2) vs. 14 (25). MD (95% CI) − 7.3 (− 19 to 4.4), p = 0.22) Mean (SD) Assessment of Quality of Life Measure at 12 months: 0.5 (0.4) vs. 0.5 (0.4). MD (95% CI) 0.0 (− 0.1 to 0.2), p = 0.75 Mean (SD) SF-36 Physical function score at 12 months: 41 (13) vs. 44 (11). MD (95% CI) 1.6 (− 3.7 to 7), p = 0.54 Mean (SD) SF-36 Physical Component Summary score at 12 months: 45 (11) vs. 46 (9). MD (95% CI) 0.3 (− 4.3 to 4.8), p = 0.9 Mean (SD) SF-36 Mental Component Summary score at 12 months: 48 (12) vs. 45 (16). MD (95% CI) 5 (− 1.1 to 11.1), p = 0.12 |
| Chen et al. [22] (Taiwan, 2010, N = 34) | Median (IQR) Total FIM score at 1 year: 78 (62–126) vs. 31 (21–50), p < 0.001 |
| Post-ICU Exercise/Physical Rehabilitation | |
| Vitacca et al. [23] (Italy, 2016, N = 48) |
Mean (95% CI) change in maximal inspiratory pressure (cmH2O) at 6 months: 14 (5.8–22) vs. − 0.2 (− 7.8 to 7.4), p = 0.007 Mean (95% CI) change in Basic Activities of Daily Living at 6 months: 1 (0–4) vs. 1 (0–4), p = 0.63 Mean (95% CI) change in EQ-5D score at 6 months: 0.23 (− 0.29 to 0.73) vs. 0.032 (− 0.29 to 0.24), p = 0.04 |
| Brummel et al. [24] (USA, 2013, N = 87) |
*3 Groups: Cognitive + Physical Therapy vs. Physical Therapy vs. Usual Care Median (IQR) Tower test (executive functioning) at 3 months: 10 (8–11) vs. 11 (11–12) vs. 10 (9–12), p = 0.2 Median (IQR) Dysexecutive questionnaire (executive functioning) at 3 months: 9 (2–18) vs. 10 (5–17) vs. 18 (9–29), p = 0.08 Median (IQR) Mini-mental state exam (global cognition) at 3 months: 29 (28–30) vs. 29 (27–30) vs. 28 (27–29), p = 0.64 Median (IQR) TUG (functional mobility) at 3 months: 11 (9–13) vs. 10 (8–13) vs. 8 (8–14), p = 0.79 Median (IQR) Katz ADL at 3 months: 0 (0–2) vs. 0 (0–1) vs. 0 (0–0), p = 0.69 Median (IQR) EQ-5D at 3 months: 75 (54–80) vs. 80 (62–89) vs. 75 (61–86), p = 0.44 |
| Jones et al. [25] (UK, 2003, N = 126) |
SF-36 Physical function scores at 6 months. when controlling for length of ICU stay, were significantly different between group, p = 0.006 (numerical scores not reported) Number (%) of patients HAD anxiety scale score > 11 at 6 months: 19 (33%) vs. 15 (34%), p = 0.71 |
| Battle et al. [26] (UK, 2019, N = 60) |
Mean (SE) 6MWT at 12 months: 345 (63) vs. 295 (57). MD (95% CI) -50 (-224 to 124), p = 0.37 Mean (SE) HAD-A at 12 months: 4 (1) vs. 9 (1). MD (95% CI) − 4 (1 to 5), p = 0.006 Mean (SE) HAD-D at 12 months: 5 (1) vs. 7 (1). MD (95% CI) − 3 (1 to 3), p = 0.11 |
| Shelly et al. [27] (India, 2017, N = 35) |
Median (IQR) difference in SF-36 Physical component summary at 4 weeks: 10.3 (8.5–14.9) vs. 7.4 (3.7–8.5), p = 0.003 Median (IQR) difference in SF-36 Mental component summary at 4 weeks: 21.8 (15.7–24.1) vs. 14.1 (10.8–19.5), p = 0.006 |
| McDowell et al. [28] (UK, 2016, N = 60) |
Mean (SD) change in SF-36 Role physical score at 6 weeks: 12 (9.8) vs. 5.4 (12). MD (95% CI) 6.6 (0.73 to 12.5), p = 0.03 Mean (SD) change in Incremental Shuttle Walk Test (meters) at 6 weeks: 136 (120) vs. 52 (127). MD (95% CI) 83 (8.3 to 158), p = 0.03 Mean (SD) change in functional limitation profile score at 6 weeks: − 7.8 (7.4) vs. − 3.0 (6.3). MD (95% CI) − 4.8 (− 8.7 to − 0.9), p = 0.02 |
| McWilliams et al. [29] (UK, 2015, N = 63) |
Mean (95% CI) change in SF-36 Physical component summary score at 8–10 weeks: 8.6 (5.4 to 10.6) vs. 3.5 (1.6 to 6.7) Mean (95% CI) change in SF-36 mental component summary score at 8–10 weeks: 10 (6.9 to 14) vs. 4.3 (0.5 to 7.6) |
| Connolly et al. [30] (UK, 2015, N = 20) |
Median (IQR) change in Incremental Shuttle Walk Test (meters) at 3 months: 115 (− 3 to 238) vs. 170 (40 to 315) Median (IQR) change in 6MWT (meters) at 3 months: 140 (36 to 210) vs. 185 (40 to 285) Median (IQR) change in SF-36 Physical component summary at 3 months: 2 (− 7 to 16) vs. 11 (4 to 28) Median (IQR) change in SF-36 Mental component summary at 3 months: 14 (− 3 to 27) vs. 11 (− 19 to 19) Median (IQR) change in HADS at 3 months: − 6 (− 9 to 3) vs. − 5 (− 13 to − 3) |
| Batterham et al. [31] (UK, 2014, N = 59) |
Mean (SD) anaerobic threshold at week 26: 13 (18) vs. 12 (20). MD (95% CI) of 0.6 (− 1.6–2.8) Mean (SD) SF-36 Physical function score at week 26: 47 (21) vs. 47 (25). MD (95% CI) of 0.1 (− 6.0 6.2) Mean (SD) SF-36 Mental health score at week 26: 51 (21) vs. 47 (25). MD (95% CI) 4.4 (− 2.4 to 11.2) |
| Jackson et al. [33] (USA, 2012, N = 21 |
Median (IQR) Tower test at 3 months: 13 (12–14) vs. 7.5 (4.0–8.5), adjusted p < 0.01 Median (IQR) TUG at 3 months: 9 (9–12) vs. 10 (9–12), adjusted p = 0.51 Median (IQR) Functional Activities Questionnaire Score at 3 months: 1.0 (0.0–3.0) vs. 8.0 (6.0–12), adjusted p = 0.04 Median (IQR) Dysexecutive questionnaire (executive functioning) at 3 months: 8 (6–14) vs. 16 (8–19), adjusted p = 0.74 Median (IQR) Mini-mental state exam (global cognition) at 3 months: 30 (29–30) vs. 27 (25–29), adjusted p = 0.25 |
| Elliott et al. [34] (Australia, 2011, N = 195) |
Mean (95% CI) SF-36 Physical function score at 26 weeks: 15 (12–18) vs. 14 (11–16). MD (95% CI) 1 (− 3 to 5), effect size 0.08 Mean (95% CI) 6MWT at 26 weeks: 126 (99–153) vs. 116 (86–147). MD (95% CI) 9.6 (− 31 to 51), effect size 0.08 Mean (95% CI) SF-36 Physical component summary score at 26 weeks: 11 (8–14) vs. 11 (8–13). MD (95% CI) 0 (− 3 to 4), effect size 0.03 Mean (95% CI) SF-36 Mental component summary score at 26 weeks: 10 (6–13) vs. 8 (5–11). MD (95% CI) 2 (− 3 to 6), effect size 0.10 |
| Follow-up services | |
| Jonasdottir et al. [35] (Iceland, 2018, N = 168) |
Mean (SD) HADS-A total score at 12 months: 4 (3) vs. 2.5 (2.8), p = 0.005 Mean (SD) HADS-D total score at 12 months: 4 (3) vs. 4 (4), p = 0.895 Mean (SD) IES-R Score at 12 months: 20 (17) vs. 14 (15), p = 0.066 |
| Jensen et al. [36] (Denmark, 2016, N = 386) |
MD (95% CI) SF-36 Physical component summary score at 12 months: 1.4 (− 1.5 to 4.4), p = 0.35 MD (95% CI) SF-36 Mental component summary score at 12 months: 1.9 (− 1.1 to 4.9), p = 0.21 MD (95% CI) HADS-A at 12 months: − 0.21 (− 1.22 to 0.80), p = 0.68 MD (95% CI) HADS-D at 12 months: − 0.20 (− 1.12 to 0.72), p = 0.67 MD (95% CI) HTQ-IV score (PTSD severity) at 12 months: − 1.42 (− 3.94 to 1.11), p = 0.27 |
| Schmidt et al. [37] (Germany, 2016, N = 143) |
MD (SD) difference in SF-36 Physical component summary score at 12 months: 10 (12) vs. 8 (14). MD (95% CI) 1.1 (− 2.7 to 4.9), p = 0.56 MD (SD) difference in SF-36 Mental component summary score at 12 months: 4 (13) vs. 2 (13). MD (95% CI) 1.4 (− 2.4 to 5.2), p = 0.47 |
| Cuthbertso et al. [38] (UK, 2009, N = 286) |
Mean (SD) SF-36 Physical component summary score at 12 months: 42 (11) vs. 41 (12). MD (95% CI) 1.1 (− 1.9 to 4.2), p = 0.46 Mean (SD) SF-36 Mental component summary score at 12 months: 47 (13) vs. 47 (12). MD (95% CI) 0.4 (− 3.0 to 3.7), p = 0.83 Mean (SD) EQ-5D Utility score at 12 months: 0.58 (0.37) vs. 0.60 (0.30). MD (95% CI) 0 (− 0.1 to 0.1), p = 0.57 Mean (SD) HADS-A score at 12 months: 6 (5) vs. 6 (4). MD (95% CI) − 0.8 (− 1.9 to 0.4), p = 0.57 |
| Douglas et al. [39] (USA, 2007, N = 335) |
No difference in SF-8 physical scores at 2 months, p = 0.40, controlling for baseline scores and APACHE III No difference in SF-8 mental scores at 2 months, p = 0.22, controlling for baseline scores and APACHE III |
| Psychosocial Programs | |
| Cox et al. [40] (USA, 2018, N = 80) |
Mean (95% CI) change from baseline for PHQ-9 at 3 months: Telephone group (TG) − 3.9 (− 5.6 to − 2.2), Mobile group (MG) − 4.8 (− 6.6 to − 2.9), Education group (EG) − 3.0 (− 5.3 to 0.8). MD (95% CI) TG vs. EG − 0.9 (− 3.7 to 2.0), p = 0.41. MD (95% CI) MG vs. EG − 1.7 (− 4.7 to 1.2), p = 0.25 Mean (95% CI) change from baseline for GAD-7 at 3 months: TG − 1.6 (− 3.0 to − 0.1), MG − 2.1 (− 3.7 to − 0.5), EG − 0.6 (− 2.5 to 1.3). MD (95% CI) TG vs. EG − 1.0 (− 3.3 to 1.4), p = 0.43. MD (95% CI) MG vs. EG − 1.5 (− 3.9 to 1.0), p = 0.24 Mean (95% CI) change from baseline for PTSD at 3 months: TG − 2.2 (− 5.6 to 1.2), MG − 2.6 (− 6.3 to 1.2), EG − 3.5 (− 8.0 to 1.0). MD (95% CI) TG vs. EG 1.3 (− 4.4 to 7.0), p = 0.65. MD (95% CI) MG vs. EG − 0.9 (− 4.9 to 6.8), p = 0.75 Mean (95%) change from baseline for PHQ-15 at 3 months: TG − 3.7 (− 5.2 to − 2.2), MG − 5.3 (− 7.0 to − 3.7), EG − 4.8 (− 6.8 to 2.7). MD (95% CI) TG vs. EG 1.1 (− 1.5 to 3.6), p = 0.41. MD (95% CI) MG vs. EG − 0.6 (− 3.2 to 2.0), p = 0.52 |
| Cox et al. [41] (USA, 2017, N = 175) |
Mean (SE) HADS at 6 months: 16 (1) vs. 16 (1). MD (95% CI) − 0.3 (− 2.7 to 2.0), p = 0.78 Mean (SE) IES-R at 6 months: 29 (3) vs. 26 (3). MD (95% CI) 3.6 (− 2.7 to 10.0), p = 0.26 Mean (SE) EQ-5D at 6 months: 61 (3) vs. 61 (3). MD (95% CI) 0.3 (− 5.9 to 6.6), p = 0.92 |
| Agren et al. [42] (Sweden, 2014, N = 84) | No difference in SF-36 between groups at 12 months (numeric data and p-value not reported) |
| Diaries | |
| Garrouste et al. [44] (France, 2012, N = 216) | Mean (SD) of IES-R data score at 12 months pre-diary 35 (16), diary 21 (12), post diary 30 (15), p = 0.03 |
| Jones et al. [45] (6 European countries, 2010, N = 352) | Incidence of PTSD 5% vs 13%, p = 0.02 |
| Knowles et al. [8] (UK, 2009, N = 36) |
Number (%) of patients with HADS-anxiety score ≥ 8 at ~ 7 weeks: 2 (11) vs. 7 (39), p < 0.05 Number (%) of patients with HADS-depression score ≥ 8 at ~ 7 weeks: 3 (17) vs. 8 (44), p < 0.07 |
| Information & Education Programs | |
| Demircelik et al. [46] (Turkey, 2016, N = 100) |
Mean (SD) in change in HADS (anxiety) from ICU to 1 week discharge: 4.2 (0.58) vs. 0.6 (0.42), p < 0.01 Mean (SD) in change in HADS (depression) from ICU to 1 week discharge: 3.5 (0.53) vs. 0.3 (0.46), p < 0.01 |
| Fleisher et al. [47] (Germany, 2014, N = 211) |
Mean (SD) SF-12 Physical component summary score at 3 months: 41 (9) vs. 40 (10). MD (95% CI) 0.3 (− 3.1 to 3.6), p = 0.87 Mean (SD) SF-12 Mental component summary score at 3 months: 47 (11) vs. 48 (11). MD (95% CI) − 1.3 (− 5.3 to 2.6), p = 0.5 |
| ABCDE Bundle | |
| Sosnowski et al. [48] (Australia, 2018, N = 30) |
Mean (SD) SF-36 Physical component summary score at 90 days: 44 (12) vs. 38 (11) Mean (SD) SF-36 Mental component summary score at 90 days: 47 (16) vs. 40 (16) |
| Earplugs and eye mask during ICU | |
| Demoule et al. [49] (France, 2017, N = 64) |
Median (IQR) HADS-A at 90 days: 8 (4–11) vs. 6 (4–12), p = 0.69 Median (IQR) HADS-D at 90 days: 6 (3–12) vs. 6 (2–9), p = 0.63 Median IES-R at 90 days: 11 (5–18) vs. 16 (9–27), p = 0.15 |
| Structured mirrors during ICU | |
| Giraud et al. [50] (UK, 2016, N = 223) | Mean (SD) EQ-5D at 12 weeks: 73 (19) vs. 77 (15), p = 0.13 |
LOS: Length of stay; IQR: Interquartile range; MD: Mean Difference; SD: Standard deviation; EQ-5D: EuroQol-5 Dimension; IADL: Instrumental Activities of Daily Living; HADS: Hospital Anxiety and Depression Scale; FIM: Functional independence measure; 6MWT: 6 min-walk-test; TUG: Timed Up and Go; HRQOL: Health related quality of life; IES-R: Impact of Event Scale-revised; PHQ-9: Patient health questionnaire-9; GAD-7: General anxiety disorder-7; PHQ-15: Patient health questionnaire-15; PTSD: post-traumatic stress disorder; HTQ-IV: Harvard Trauma Questionnaire Part IV
Studies identified based on systematic review by Geense et al. [21]
aSample size = total patients enrolled in the study
Recent data on additional aspects of PICS
Fatigue
Survivors of acute respiratory failure commonly experience fatigue with growing empirical evaluation of this symptom. An analysis of data from the ALTOS study (n = 732) evaluated fatigue symptoms using the validated Functional Assessment of Chronic Illness Therapy-Fatigue Scale (FACIT-F) [5], with 70% and 66% reporting fatigue at 6 and 12 months respectively [5]. At 12-month versus 6-month follow-up, 28% of participants reported their symptoms were worse, 31% reported no change, and 41% reported improved symptoms. Increased fatigue was associated with female sex and unemployment prior to hospital admission [5]. At 6 and 12 months, patients with fatigue symptoms had worse physical functioning and higher psychological impairments [5]. Thus, health care providers should screen for both physical and psychological impairments among ICU survivors reporting fatigue. Importantly, in this cohort of ARDS survivors, there was no association between fatigue and ICU length of stay or severity of illness [5]. Additionally, a prospective study among a broader population of ICU survivors, rather than exclusively ARDS survivors, reported a high prevalence of fatigue at 12 month follow-up among medical, urgent surgery, and elective surgery ICU survivors as follows: 36%, 45%, and 24%, respectively [6].
Pain
In the ALTOS study, nearly 50% of ARDS survivors reported clinically significant pain during the first year after ARDS [57]. Unemployment and the use of opioids in the ICU were associated with greater pain at 6- and 12-month follow-up [57]. Among those with pain, 78% also reported anxiety and/or depressive symptoms and 78% reported cognitive and/or physical function impairment. This prevalence in the ALTOS study was similar to another study that reported 31% and 35% of medical and surgical ICU survivors having moderate to severe pain at 3 and 12 months, respectively [58]. In contrast, the prevalence of pain in the community is substantially lower, with only 20% of the US population reporting chronic pain [15]. A prior study using the brief pain inventory (BPI) measurement instrument in 295 patients from medical and surgical ICUs examined pain intensity and its effect on patients after hospital discharge [58]. Cumulative ICU opioid exposure was not associated with increased pain intensity or increased pain interference of daily life after the intensive care unit [58]. The authors suggest that patients with underlying chronic pain may report higher pain after hospital discharge due to opioid tolerance, hyperalgesia, or predisposition to developing a pain syndrome [58].
Delayed return to work and joblessness
Previously-employed survivors of critical illness experience challenges in returning to work after hospital discharge (Fig. 1) [59]. Some issues commonly encountered are delayed return to work, loss of job after return to work, and the need to change occupations [59, 60]. These problems frequently lead to a financial burden for patients and their families [59]. A meta-analysis, including 52 studies evaluating 10,015 previously-employed ICU survivors, assessed return to work [60]. Approximately 36%, 64%, 60% of patients reported return to work at 1 to 3, 6, and 12- month follow-up, respectively [60]. Furthermore, results from the ALTOS study, including 326 previously-employed ARDS survivors, found that 48% and 43% were jobless at 6- and 12-month follow-up [61]. Patients with pain or fatigue were less likely to return to work [61]. At 6 and 12 months, the imbalance between occupational workload requirements and ARDS survivors’ functional ability occurred in 90% of ALTOS participants [62]. Furthermore, having imbalance in both physical and psychosocial areas at 6 months was significantly associated with joblessness at 6 and 12 months [62]. The findings from these studies highlight the need to improve patient’s functional abilities, and to decrease work load via workplace accommodations for ICU survivors [62, 63].
Fig. 1.
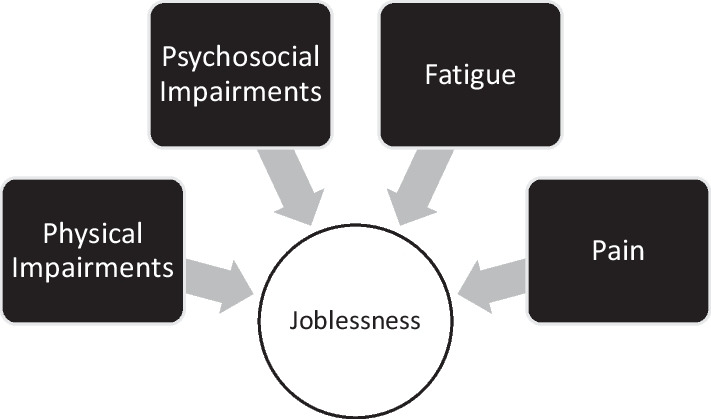
Barriers to return to work after critical illness
Conclusion
Post-intensive care syndrome is experienced by many ICU survivors who have new or worsening physical, cognitive, and/or mental health impairments. These impairments often co-occur and may include pain and fatigue. Together these impairments and symptoms create substantial challenges in returning to work for previously-employed ICU survivors. Evaluation of ARDS survivor subtypes/phenotypes demonstrate that physical and mental health impairments are closely associated, without association with cognitive outcomes. The biological mechanisms underlying many of these long-standing impairments are uncertain despite exploration into inflammatory biomarkers in the ICU setting. Increased understanding of risk factors, especially across different types of ICU patients has improved our ability to potentially identify high-risk patients for screening and intervention. However, in terms of interventions, evaluation of non-pharmacological interventions, including early mobilization and physical rehabilitation in the ICU, ICU diaries, psychological interventions, multi-disciplinary post-ICU follow-up clinics and interventions, and educational activities, are still in an early stage. Future well-designed studies are needed to better understand mechanisms and potential interventions to improve post-intensive care syndrome.
Acknowledgements
Not applicable.
Abbreviations
- ALTOS
ARDSNet Long Term Outcomes Study
- ARDS
Acute Respiratory Distress Syndrome
- BPI
Brief pain intensity
- COVID-19
Coronavirus disease 2019
- FACIT-F
Functional assessment of chronic illness therapy fatigue scale
- HADS
Hospital Anxiety and Depression Scale
- ICU
Intensive Care Unit
- PICS
Post-intensive care syndrome
- PTSD
Post-traumatic stress disorder
- SCCM
Society of Critical Care Medicine
- UK
United Kingdom
Author contributions
SLH, AF, and MA performed literature review and primary first draft. DMN performed critical revisions. All authors read and approved the final manuscript.
Funding
No specific funding supported this work.
Availability of data and materials
Not applicable.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Calvin JE, Habet K, Parrillo JE. Critical care in the United States who are we and how did we get here? Crit Care Clin. 1997;13(2):363–376. doi: 10.1016/s0749-0704(05)70315-8. [DOI] [PubMed] [Google Scholar]
- 2.Efu ME, Ojo B, Eke BA, Anefu GO, Ozoagu MA. Characterization of the intensive care unit (ICU) admission in the Benue State University teaching hospital. Med Surg Sci. 2019;6(4):126–128. doi: 10.32457/ijmss.2019.037.Efu. [DOI] [Google Scholar]
- 3.Kaukonen K, Bailey M, Suzuki S, Pilcher D, Bellomo R. Mortality related to severe sepsis and septic shock among critically ill patients in Australia and New Zealand, 2000–2012. JAMA. 2014;311(13):1308–1316. doi: 10.1001/jama.2014.2637. [DOI] [PubMed] [Google Scholar]
- 4.Needham DM, Davidson J, Cohen H, et al. Improving long-term outcomes after discharge from intensive care unit. Crit Care Med. 2012;40(2):502–509. doi: 10.1097/CCM.0b013e318232da75. [DOI] [PubMed] [Google Scholar]
- 5.Neufeld KJ, Leoutsakos JS, Yan H, et al. Fatigue symptoms during the first year following ARDS. Chest. 2020;158(3):999–1007. doi: 10.1016/j.chest.2020.03.059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Geense WW, Zegers M, Peters MAA, et al. New physical, mental, and cognitive problems 1 year after ICU admission. Am J Respir Crit Care Med. 2021;203(12):1512–1521. doi: 10.1164/rccm.202009-3381OC. [DOI] [PubMed] [Google Scholar]
- 7.Griffiths J, Hatch RA, Bishop J, et al. An exploration of social and economic outcome and associated health-related quality of life after critical illness in general intensive care unit survivors : a 12-month follow-up study. Crit Care. 2013;17(3):R100. doi: 10.1186/cc12745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Knowles RE, Tarrier N. Evaluation of the effect of prospective patient diaries on emotional well-being in intensive care unit survivors: a randomized controlled trial. Crit Care Med. 2009;37(1):184–191. doi: 10.1097/CCM.0b013e31819287f7. [DOI] [PubMed] [Google Scholar]
- 9.Azoulay E, Vincent J, Angus DC, et al. Recovery after critical illness: putting the puzzle together—a consensus of 29. Crit Care. 2017;21(1):296. doi: 10.1186/s13054-017-1887-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Spragg RG, Bernard GR, Checkley W, et al. Beyond mortality: future clinical research in acute lung injury. Am J Respir Crit Care Med. 2010;181(10):1121–1127. doi: 10.1164/rccm.201001-0024WS. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Needham DM, Sepulveda KA, Dinglas VD, et al. Core outcome measures for clinical research in acute respiratory failure survivors an international modified Delphi consensus study. Crit Care Med. 2017;196(9):1122–1130. doi: 10.1164/rccm.201702-0372OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Brown SM, Wilson EL, Presson AP, et al. Understanding patient outcomes after acute respiratory distress syndrome: identifying subtypes of physical, cognitive and mental health outcomes. Thorax. 2017;72(12):1094–1103. doi: 10.1136/thoraxjnl-2017-210337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.The PHOSP-COVID Collaborative Group Clinical characteristics with inflammation profiling of long COVID and association with 1-year recovery following hospitalisation in the UK : a prospective observational study. Lancet Respir Med. 2022;2600(22):1–15. doi: 10.1016/S2213-2600(22)00127-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Heesakkers H, van der Hoeven J, Corsten S, et al. Clinical outcomes among patients with 1-year survival following intensive care unit treatment for COVID-19. JAMA. 2022;327(6):559–565. doi: 10.1001/jama.2022.0040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Parker A. Behavioral activation-rehabilitation to improve depressive symptoms and physical function after acute respiratory failure (BEHAB). 2022:1-9. https://clinicaltrials.gov/ct2/show/study/NCT03431493.
- 16.Lee M, Kang J, Jeong Y. Risk factors for post-intensive care syndrome: a systematic review and meta-analysis. Aust Crit Care. 2020;33(3):287–294. doi: 10.1016/j.aucc.2019.10.004. [DOI] [PubMed] [Google Scholar]
- 17.Cabrini L, Landoni G, Antonelli M, et al. Critical care in the near future: patient-centered, beyond space and time boundaries. Ninerva Anestesiol. 2016;82(5):599–604. [PubMed] [Google Scholar]
- 18.Hashem M, Hopkins R, Colantuoni E, et al. Six-month and 12-month patient outcomes based on inflammatory subphenotypes in sepsis-associated ARDS: secondary analysis of SAILS-ALTOS trial. Thorax. 2022;77:22–30. doi: 10.1136/thoraxjnl-2020-216613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Brummel NE, Hughes CG, Thompson JL, et al. Inflammation and coagulation during critical illness and long-term cognitive impairment and disability. Am J Respir Crit Care Med. 2021;203(6):699–706. doi: 10.1164/rccm.201912-2449OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Parker A, Sinha P, Needham D. Biological mechanisms of cognitive and physical impairments after critical care rethinking the inflammatory model? Am J Respir Crit Care Med. 2021;203(6):665–667. doi: 10.1164/rccm.202010-3896ED. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Geense W, van den Boogaard M, van der Hoeven J, Vermuelen H, Hannink G, Zegers M. Nonpharmacologic interventions to prevent or mitigate adverse long-term outcomes among ICU survivors: a systematic review and meta-analysis. Crit Care Med. 2019;47(11):1607–1618. doi: 10.1097/CCM.0000000000003974. [DOI] [PubMed] [Google Scholar]
- 22.Chen S, Su CL, Wu YT, et al. Physical training is beneficial to functional status and survival in patients with prolonged mechanical ventilation. J Formos Med Assoc. 2011;110(9):572–579. doi: 10.1016/j.jfma.2011.07.008. [DOI] [PubMed] [Google Scholar]
- 23.Vitacca M, Barbano L, Vanoglio F, et al. Does 6-month home caregiver-supervised physiotherapy improve post-critical care outcomes? Am J Phys Med Rehabil. 2016;95(8):571–579. doi: 10.1097/PHM.0000000000000441. [DOI] [PubMed] [Google Scholar]
- 24.Brummel NE, Girard TD, Ely EW, et al. Feasibility and safety of early combined cognitive and physical therapy for critically ill medical and surgical patients: the activity and cognitive therapy in ICU (ACT-ICU) trial. Intensive Care Med. 2014;40(3):370–379. doi: 10.1007/s00134-013-3136-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Jones C, Skirrow P, Griffiths RD, et al. Rehabilitation after critical illness: a randomized, controlled trial. Crit Care Med. 2003;31(10):2456–2461. doi: 10.1097/01.CCM.0000089938.56725.33. [DOI] [PubMed] [Google Scholar]
- 26.Battle C, James K, Temblett P, Hutchings H. Supervised exercise rehabilitation in survivors of critical illness: a randomised controlled trial. J Intensive Care Soc. 2019;20(1):18–26. doi: 10.1177/1751143718767061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Shelly A, Prabhu N, Jirange P, Kamath A, Vaishali K. Quality of life improves with individualized home-based exercises in critical care survivors. Indian J Crit Care Med. 2017;21(2):89–93. doi: 10.4103/ijccm.IJCCM_433_16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.McDowell K, O’Neill B, Blackwood B, et al. Effectiveness of an exercise programme on physical function in patients discharged from hospital following critical illness: a randomised controlled trial (the REVIVE trial) Thorax. 2016;72(7):600–609. doi: 10.1136/thoraxjnl-2016-208723. [DOI] [PubMed] [Google Scholar]
- 29.McWilliams DJ, Benington S, Atkinson D. Outpatient-based physical rehabilitation for survivors of prolonged critical illness: a randomized controlled trial. Physiother Theory Pract. 2016;32(3):179–190. doi: 10.3109/09593985.2015.1137663. [DOI] [PubMed] [Google Scholar]
- 30.Connolly B, Thompson A, Douiri A, Moxham J, Hart N. Exercise-based rehabilitation after hospital discharge for survivors of critical illness with intensive care unit-acquired weakness: a pilot feasibility trial. J Crit Care. 2015;30(3):589–598. doi: 10.1016/j.jcrc.2015.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Batterham AM, Bonner S, Wright J, Howell SJ, Hugill K, Danjoux G. Effect of supervised aerobic exercise rehabilitation on physical fitness and quality-of-life in survivors of critical illness: an exploratory minimized controlled trial (PIX study) Br J Anaesth. 2014;113(1):130–137. doi: 10.1093/bja/aeu051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Arthur HM, Daniels C, McKelvie R, Hirsh J, Rush B. Effect of a preoperative intervention on preoperative and postoperative outcomes in low-risk patients awaiting elective coronary artery bypass graft surgery: a randomized, controlled trial. Ann Intern Med. 2000;133(4):253–262. doi: 10.7326/0003-4819-133-4-200008150-00007. [DOI] [PubMed] [Google Scholar]
- 33.Jackson J, Ely W, Morey M, et al. Cognitive and physical rehabilitation of ICU survivors: results of the RETURN randomized, controlled pilot investigation. Crit Care Med. 2012;40(4):1088–1097. doi: 10.1097/CCM.0b013e3182373115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Elliott D, McKinley S, Alison J, et al. Health-related quality of life and physical recovery after a critical illness: a multi-centre randomised controlled trial of a home-based physical rehabilitation program. Crit Care. 2011;15(3):R142. doi: 10.1186/cc10265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Jónasdóttir RJ, Jónsdóttir H, Gudmundsdottir B, Sigurdsson GH. Psychological recovery after intensive care: outcomes of a long-term quasi-experimental study of structured nurse-led follow-up. Intensive Crit Care Nurs. 2018;44:59–66. doi: 10.1016/j.iccn.2017.06.001. [DOI] [PubMed] [Google Scholar]
- 36.Jensen JF, Egerod I, Bestle MH, et al. A recovery program to improve quality of life, sense of coherence and psychological health in ICU survivors: a multicenter randomized controlled trial, the RAPIT study. Intensive Care Med. 2016;42(11):1733–1743. doi: 10.1007/s00134-016-4522-1. [DOI] [PubMed] [Google Scholar]
- 37.Schmidt K, Worrack S, Korff M, et al. Effect of a primary care management intervention on mental-health-related quality of life among survivors of sepsis: a randomized clinical trial. JAMA. 2016;315(24):2703–2711. doi: 10.1053/j.gastro.2016.08.014.CagY. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Cuthbertson BH, Rattray J, Campbell MK, et al. The PRaCTICaL study of nurse led, intensive care follow-up programmes for improving long term outcomes from critical illness: a pragmatic randomised controlled trial. BMJ. 2009;339(7728):1016. doi: 10.1136/bmj.b3723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Douglas S, Daly B, Kelley C, O’Toole E, Montenegro H. Chronically critically ill patients: health-related quality of life and resource use after a disease management intervention. Am J Crit Care. 2007;16(5):447–457. doi: 10.4037/ajcc2007.16.5.447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Cox CE, Hough CL, Jones DM, et al. Effects of mindfulness training programmes delivered by a self-directed mobile app and by telephone compared with an education programme for survivors of critical illness: a pilot randomised clinical trial. Thorax. 2019;74(1):33–42. doi: 10.1136/thoraxjnl-2017-211264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Cox CE, Hough CL, Carson SS, et al. Effects of a telephone- and web-based coping skills training program compared with an education program for survivors of critical illness and their family members a randomized clinical trial. Am J Respor Crit Care Med. 2018;197(1):66–78. doi: 10.1164/rccm.201704-0720OC. [DOI] [PubMed] [Google Scholar]
- 42.Ågren S, Berg S, Svedjeholm R, Strömberg A. Psychoeducational support to post cardiac surgery heart failure patients and their partners—a randomised pilot study. Intensive Crit Care Nurs. 2015;31(1):10–18. doi: 10.1016/j.iccn.2014.04.005. [DOI] [PubMed] [Google Scholar]
- 43.Wright SE, Thomas K, Watson G, et al. Intensive versus standard physical rehabilitation therapy in the critically ill (EPICC): a multicentre, parallel-group, randomised controlled trial. Thorax. 2018;73(3):213–221. doi: 10.1136/thoraxjnl-2016-209858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Garrouste-Orgeas M, Coquet I, Perier A, et al. Impact of an intensive care unit diary on psychological distress in patients and relatives. Crit Care Med. 2012;40:2033–2040. doi: 10.1097/CCM.0b013e31824e1b43. [DOI] [PubMed] [Google Scholar]
- 45.Jones C, Bäckman C, Capuzzo M, et al. Intensive care diaries reduce new onset post traumatic stress disorder following critical illness: a randomised, controlled trial. Crit Care. 2010;14(5):R168. doi: 10.1186/cc9260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Demircelik MB, Cakmak M, Nazli Y, et al. Effects of multimedia nursing education on disease-related depression and anxiety in patients staying in a coronary intensive care unit. Appl Nurs Res. 2016;29:5–8. doi: 10.1016/j.apnr.2015.03.014. [DOI] [PubMed] [Google Scholar]
- 47.Fleischer S, Berg A, Behrens J, et al. Does an additional structured information program during the intensive care unit stay reduce anxiety in ICU patients ?: a multicenter randomized controlled trial. BMC Anesthesiol. 2014;14(1):1–11. doi: 10.1186/1471-2253-14-48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Sosnowski K, Mitchell ML, White H, et al. A feasibility study of a randomised controlled trial to examine the impact of the ABCDE bundle on quality of life in ICU survivors. Pilot Feasibility Stud. 2018;4(1):1–12. doi: 10.1186/s40814-017-0224-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Demoule A, Carreira S, Lavault S, et al. Impact of earplugs and eye mask on sleep in critically ill patients : a prospective randomized study. Crit Care. 2017;21:284. doi: 10.1186/s13054-017-1865-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Giraud K, Pontin M, Sharples LD, et al. Use of a structured mirrors intervention does not reduce delirium incidence but may improve factual memory encoding in cardiac surgical ICU patients aged over 70 years : a pilot time-cluster randomized controlled trial. Front Aging Neurosci. 2016;8:228. doi: 10.3389/fnagi.2016.00228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Morris PE, Berry MJ, Files DC, et al. Standardized rehabilitation and hospital length of stay among patients with acute respiratory failure a randomized clinical trial. JAMA. 2016;315(24):2694–2702. doi: 10.1001/jama.2016.7201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Hodgson CL, Bailey M, Bellomo R, et al. A binational multicenter pilot feasibility randomized controlled trial of early goal-directed mobilization in the ICU. Crit Care Med. 2016;44(6):1145–1152. doi: 10.1097/CCM.0000000000001643. [DOI] [PubMed] [Google Scholar]
- 53.Schaller SJ, Anstey M, Blobner M, et al. Early, goal-directed mobilisation in the surgical intensive care unit: a randomised controlled trial. Lancet. 2016;388(10052):1377–1388. doi: 10.1016/S0140-6736(16)31637-3. [DOI] [PubMed] [Google Scholar]
- 54.Kayambu G, Boots R, Paratz J. Early physical rehabilitation in intensive care patients with sepsis syndromes: a pilot randomised controlled trial. Intensive Care Med. 2015;41(5):865–874. doi: 10.1007/s00134-015-3763-8. [DOI] [PubMed] [Google Scholar]
- 55.Moss M, Nordon-Craft A, Malone D, et al. A randomized trial of an intensive physical therapy program for patients with acute respiratory failure. Am J Respir Crit Care Med. 2016;193(10):1101–1110. doi: 10.1164/rccm.201505-1039OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Denehy L, Skinner EH, Edbrooke L, et al. Exercise rehabilitation for patients with critical illness: a randomized controlled trial with 12 months of follow-up. Crit Care. 2013;17(4):1–12. doi: 10.1186/cc12835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Probert JM, Lin S, Yan H, et al. Bodily pain in survivors of acute respiratory distress syndrome : a 1-year longitudinal follow-up study. J Psychosom Res. 2021;144:110418. doi: 10.1016/j.jpsychores.2021.110418. [DOI] [PubMed] [Google Scholar]
- 58.Hayhurst CJ, Jackson JC, Archer KR, Thompson JL, Chandrasekhar R, Hughes CG. Pain and its long-term interference of daily life after critical illness. Anesth Analg. 2018;127(3):690–697. doi: 10.1213/ANE.0000000000003358. [DOI] [PubMed] [Google Scholar]
- 59.Herridge MS, Cheung AM, Tansey CM, et al. One-year outcomes in survivors of the acute respiratory distress syndrome. N Engl J Med. 2003;348(8):683–693. doi: 10.1056/NEJMoa022450. [DOI] [PubMed] [Google Scholar]
- 60.Kamdar BB, Suri R, Suchyta MR, et al. Return to work after critical illness : a systematic review and meta-analysis. Thorax. 2020;75(1):17–27. doi: 10.1136/thoraxjnl-2019-213803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Su H, Thompson HJ, May S, et al. Association of job characteristics and functional impairments on return to work after ARDS. Chest. 2021;160(2):509–518. doi: 10.1016/j.chest.2021.03.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Su H, Hopkins RO, Kamdar BB, et al. Association of imbalance between job workload and functional ability with return to work in ARDS survivors. Thorax. 2022;77(2):123–128. doi: 10.1136/thoraxjnl-2020-216586. [DOI] [PubMed] [Google Scholar]
- 63.Su H, Thompson HJ, Pike K, et al. Interrelationships among workload, illness severity, and function on return to work following acute respiratory distress syndrome. Aust Crit Care. 2023;36(2):247–253. doi: 10.1016/j.aucc.2022.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.


