Abstract
A series of landmark experiments conducted throughout the 20th century progressively localized the regions involved in consciousness to the reticular activating system (RAS) and its ascending projections. The first description of the RAS emerged in 1949 through seminal experiments performed by Moruzzi and Magoun in feline brainstems; additional experiments in the 1950s revealed connections between the RAS and the thalamus and neocortical structures. This knowledge has allowed for the explanation of disorders of consciousness with exquisite anatomic precision. The clinical relevance of the RAS is further apparent in modern definitions of brain death/death by neurologic criteria (BD/DNC), which require demonstration of the complete and permanent loss of capacity for consciousness as one of their core criteria. BD/DNC is currently understood across jurisdictions in terms of “whole brain” and “brainstem” formulations. Although their clinical examination between formulations is indistinguishable, policies for BD/DNC declaration may differ in the rare scenario of patients with isolated infratentorial brain injuries, in which ancillary testing is advised in the whole brain formulation but not the brainstem formulation. Canadian guidelines acknowledge that the distinction between whole brain and brainstem formulations is unclear with respect to clinical implications for patients with isolated infratentorial injuries. This has led to variability in Canadian clinicians’ use of ancillary testing when the mechanism of BD/DNC is suspected to be an isolated infratentorial injury. The present narrative review highlights these concepts and explores implications for determination of BD/DNC in Canada, with specific emphasis on the RAS and its relevance to both formulations.
Keywords: brain death, infratentorial injury, neurologic determination of death, reticular activating system
Résumé
Une série d’expériences marquantes menées tout au long du 20e siècle a progressivement permis de localiser les régions impliquées dans la conscience dans le système d’activation réticulaire (SAR) et ses projections ascendantes. La première description du SAR a vu le jour en 1949 grâce à des expériences fondatrices réalisées par Moruzzi et Magoun dans des troncs cérébraux félins; d’autres expériences menées au cours des années 1950 ont révélé des liens entre le SAR et le thalamus et les structures néocorticales. Ces connaissances ont permis d’expliquer les troubles de la conscience avec une précision anatomique extraordinaire. La pertinence clinique du SAR est encore plus évidente dans les définitions modernes de la mort cérébrale / du décès déterminé par des critères neurologiques (MC/DCN), qui exigent la démonstration de la perte complète et permanente de la capacité de conscience comme l’un de ses critères de base. La mort cérébrale est actuellement comprise partout en termes de formulations de « cerveau entier » et de « tronc cérébral ». Bien que l’examen clinique ne fasse pas de distinction entre ces formulations, les politiques de déclaration de MC/DCN peuvent différer dans le rare cas de patients présentant des lésions cérébrales infratentorielles isolées, pour lesquels des examens auxiliaires sont conseillés lorsqu’on parle de cerveau entier mais pas lorsqu’on utilise la formulation de tronc cérébral. Les lignes directrices canadiennes reconnaissent que la distinction entre les termes de cerveau entier et de tronc cérébral n’est pas claire en ce qui concerne leurs implications cliniques pour les patients présentant des lésions infratentorielles isolées. Cela a entraîné une variabilité dans l’utilisation des examens auxiliaires par les cliniciens canadiens lorsqu’ils soupçonnent que le mécanisme de MC/DCN consiste en une lésion infratentorielle isolée. Ce compte rendu narratif met en lumière ces concepts et explore les implications pour la détermination de la MC/DCN au Canada, en mettant une emphase spécifique sur le SAR et sa pertinence pour les deux formulations.
What does it mean, to be conscious? For Wilder Penfield, consciousness represented “a thinking, a knowing, a focusing of attention, a planning of action, an interpretation of present experience … (an) integrated perception of the present.”1 Notwithstanding philosophical, medicolegal, and sociocultural definitions, by Penfield’s time, in the mid-20th century, it had become clear that consciousness was dependent on a specific region in the brainstem known as the reticular activating system (RAS), which together with pathways that traveled between the brainstem, thalamus, and cerebral cortex provided the neuroanatomic basis for consciousness.
Our current understanding of the brainstem and its fundamental importance to consciousness is remarkable considering that, until the late 18th century, the brainstem was still widely regarded as a simple extension of the spinal cord.2 It was not until the 19th century that anatomists began to progressively localize cranial nerves, motor pathways, and respiratory centers to different levels within the pons and medulla oblongata, lending growing support to the notion of the brainstem as a mediator of life-sustaining functions.3 By the 20th century, the core structures involved in consciousness had been mapped to regions of the brainstem and their rostral projections. In modern clinical practice, the central importance of the brainstem is manifested by its significance in neuroprognostication after brain injuries and its central place in medical and legal definitions of brain death/death by neurologic criteria (BD/DNC).
In this narrative review, we describe the evolution of our modern anatomic understanding of the RAS and explore how that knowledge has informed our understanding of disorders of consciousness and current definitions of BD/DNC. We begin by reviewing the foundational 20th-century experiments that identified the neuronal networks which mediate arousal and awareness. We then outline the connections between the RAS and link our anatomic knowledge of these structures to their central importance in consciousness and in the neurologic determination of death. We conclude by exploring important differences across jurisdictions in BD/DNC definitions and the question of inferring complete and permanent loss of consciousness—one of the core requirements to declare brain death—in patients with isolated infratentorial injuries.
Progressive anatomic mapping of the brainstem, RAS, and thalamocortical circuits
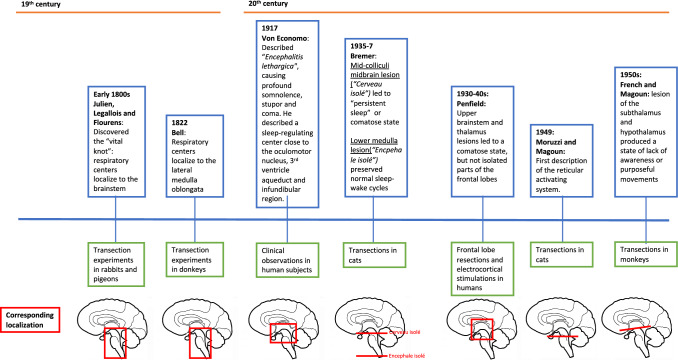
By the 19th century, neuroanatomists had shown that different parts of the brainstem mediated vital physiologic functions without which life could not be sustained. In the early part of the century, the French anatomists Cesar Julien, Jean Legallois, and Jean Pierre Flourens separately performed transection experiments in rabbits and pigeons, in which they localized respiratory activity to the brainstem and more specifically to a 1-mm region at the base of the brain, which Flourens called the noeud vital (“vital knot”).4,5 In 1822, Bell identified a similar respiratory center in donkeys, which he was able to localize to the lateral medulla oblongata.6 These transection experiments grossly localized the respiratory centers to the brainstem, but another one hundred years would pass until more sophisticated experiments allowed precise identification of the neural networks that control respiration.
The advent of new methods of neuromonitoring, specifically electroencephalography (EEG), allowed for more detailed insight into the topographical importance of the brainstem to consciousness. In 1937, the Belgian physiologist Frederic Bremer found that cutting the base of the feline brainstem at the level of the medulla (the so-called encephale isolé preparation) did not disrupt the animal’s EEG sleep-wake cycle, whereas cutting within the midbrain between the superior and inferior colliculi (cerveau isolé) resulted in a persistent EEG pattern of spindles and slow waves.7 Experiments by Giuseppe Moruzzi and Horace Magoun showed that stimulating a region between the pontine and midbrain tegmentum in Bremer’s encephale isolé animals rapidly converted a slow-wave, sleep-like EEG pattern into one resembling wakefulness. Stimulation would not modify the slow-wave EEG trace with more rostral lesions at the cerebral peduncles or tectum.8 These findings led Moruzzi and Magoun to delineate with remarkable precision the regions of the brainstem that were necessary for regulating arousal and normal sleep (of note, the specific neurons necessary for sustaining consciousness remain unknown).9 Figure 1 indicates the major findings and years over which these experiments occurred.
Fig. 1.
Historical advances in understanding the anatomy of consciousness
Around the time that Berger, Moruzzi, and Magoun were performing their animal experiments, 20th-century clinicians were beginning to link the brainstem to clinical disorders of consciousness. In 1917, the Austrian neurologist Constantin von Economo described his findings in patients afflicted by encephalitis lethargica, an atypical form of encephalitis that targeted neurons in the brainstem and caused a profound and progressive somnolence.10 Later, Canadian neurosurgeon Wilder Penfield observed that damage to structures within the brainstem produced a clinical state that was incompatible with wakefulness, whereas patients with extensive resections of their frontal lobes could still maintain arousal.11 Exploring more superior and caudal structures, Jack French and Horace Magoun further observed that destroying specific regions of the median cephalic portion of the brainstem including the midbrain tegmentum, subthalamus, hypothalamus, and medial thalamus in animal models produced “stupor waves” on the EEG similar to those of Bremner’s cerveau isolé preparation and extinguished environmental awareness or purposeful movement.12
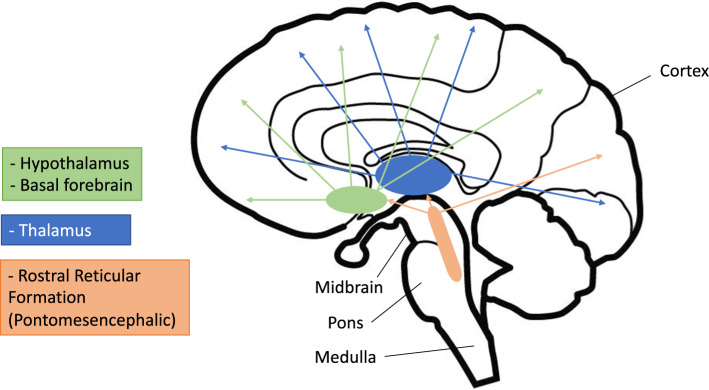
These experiments have led to our modern understanding of the reticular formation as a collection of neurons within the brainstem, extending from the level of the rostral midbrain to the rostral medulla. Named for its loose, web-like (“reticular”) appearance, this meshwork supports distinct groups of nuclei that cluster together and mediate various physiologic functions, including breathing, circulation, pain modulation, and generation of repetitive motor movements (e.g., chewing and swallowing). The ascending RAS, which is relevant to arousal and consciousness, is localized in the central and dorsal brainstem between the caudal midbrain and midpons. Projections from the RAS extend to the basal forebrain, hypothalamus, and intralaminar nuclei of the thalamus. The thalamus is a primary relay point for ascending fibers from the RAS, and thalamocortical circuits play a key role in generating oscillatory activity associated with conscious states, slow-wave sleep, and EEG spindles; disruption of these circuits has been implicated in various disorders of consciousness.13 A small number of fibers bypass the thalamus and project directly to the cortex, as depicted in Fig. 2.
Fig. 2.
Anatomy of the reticular activating system
The RAS and thalamocortical system in disorders of consciousness
The clinical impact of lesions of the RAS, thalamus, and cortex on consciousness vary depending on the location and completeness of the lesion. Lesions of the pontine tegmentum are capable of producing a state of coma, in which the sleep-wake cycle is lost, the eyes remain closed, and there is no speech or purposeful movement.14 Cortical metabolism and activity may be virtually undisturbed by such lesions and yet the clinical syndrome is one of profound unresponsiveness.15 Diffuse cortical or selective bilateral thalamic injuries may produce a persistent vegetative state or unresponsive wakefulness syndrome,16,17 in which patients maintain sleep-wake cycles but show no ability to communicate or engage purposefully with their environment. Autopsies in such patients (including Karen Ann Quinlan, whose case gained worldwide prominence)17 have confirmed severe damage to the thalamus and/or thalamocortical white matter tracts.18,19 Less profound cortical or thalamic injuries can lead to a condition known as the minimally conscious state. Patients in this condition show some level of awareness of their environment: they can often reach for objects and track movements with their eyes, although such reactions are not always consistent.20
Modern EEG and imaging studies have reinforced the association between the degree of damage to the global RAS and the severity of the resulting disorders of consciousness. Using quantitative EEG in patients with subarachnoid hemorrhage, Claassen et al. found that a greater degree of disconnection between the cortex, thalamus, and basal forebrain was associated with a lower Glasgow Coma Score.21 In studies using functional magnetic resonance imaging (fMRI), researchers observed a higher level of wakefulness in patients when both corticocortical and corticothalamic connections were preserved, whereas more profound impairments were seen when corticocortical connections were preserved but corticothalamic ones were not.22,23 Reports using MRI-based diffusion tensor imaging sequences have been used to show white matter disruption within the RAS in patients with profound disorders of consciousness (e.g., due to traumatic brain injury or cardiac arrest).24–27 Our understanding of the neuroanatomical basis for consciousness continues to evolve with new insights into the topography and connectivity among these regions. Nevertheless, the core notion that some viability of the RAS is necessary for consciousness remains unchanged.
The central importance of the brainstem and RAS in BD/DNC
Brain death/death by neurologic criteria, defined conceptually by the permanent and complete loss of the capacity for consciousness and all brainstem function, including the capacity to breathe, is distinct from disorders of consciousness.28 These foundational criteria for BD/DNC reflect the concept that vital functions for life and arousal are governed by pathways that originate from, or travel through, the brainstem. Consequently, the assessment of brainstem function plays a central role in the determination of BD/DNC, based on the premise that permanent and complete damage to the RAS (as shown by unresponsive coma and a lack of demonstrable brainstem function) precludes consciousness. This premise is further supported by evidence showing that BD/DNC is associated with damage to the brainstem on postmortem examination.29
The central importance of the brainstem and RAS for consciousness has made the permanent loss of brainstem function an enduring criteria of BD/DNC from its first description by Mollaret and Goulon in the 1950s30 to modern definitions and guidelines.31–33 Since the publication of the seminal Harvard Criteria, which provided the first set of formal criteria for establishing “irreversible coma,”31 the construct of BD/DNC has been adopted across jurisdictions with subsequent updates incorporating more specific instructions for the clinical exam and ancillary testing.28,32–34 Although there are important historic, national, and even institutional variabilities in the procedures for determining BD/DNC, all definitions consistently include the loss of brainstem function as a central feature.28
“Brainstem death,” “whole brain death,” and unanswered questions
Despite recent initiatives to reach consensus in the definition of BD/DNC, which center around the primacy of the brainstem and RAS,28 there continues to be heterogeneity in definitions across jurisdictions, including differing formulations of “whole brain death” and “brainstem death.” The UK and several other jurisdictions adopted the stance that brainstem death, as defined by the permanent and complete loss of all brainstem function and the capacity for consciousness, was sufficient for death of the whole person. This position was justified on the premise that the core processes of life and consciousness require some preserved brainstem function.35,36 By contrast, the USA adopted legislation stating that “an individual who has sustained an irreversible cessation of all functions of the entire brain, including the brainstem, is dead.”37 Despite conceptual differences and theoretical arguments that the brainstem definition entails a lower burden of proof than the whole brain definition,38 both formulations share common prerequisites and are supported by the same clinical assessment.39 This concordance stems from the fact that BD/DNC is most commonly the result of a severe supratentorial or diffuse global injury that leads to brainstem destruction by mechanical compression or complete infarction from loss of intracranial blood flow.40
Nevertheless, where the two formulations may potentially diverge is in the uncommon scenario of an isolated infratentorial injury causing BD/DNC.41,42 In jurisdictions that follow the whole brain death formulation, ancillary testing (e.g., with angiography or radionuclide imaging) is suggested when BD/DNC is suspected from an isolated brainstem lesion. Loss of intracranial blood flow with flow-based ancillary tests supports the clinical determination of BD/DNC in this scenario.28 Ancillary testing is not mandated in the brainstem formulation. Notwithstanding the limitations of ancillary testing and the fallacy of inferring intact function from intact flow, a concordant ancillary test that supports the clinical exam in patients with isolated brainstem injuries may improve clinicians’ confidence in diagnosing BD/DNC.43 One Canadian study found that clinicians ordered ancillary testing in 10/12 (85%) patients undergoing assessment for BD/DNC after infratentorial ischemic or hemorrhagic strokes compared with only 25/47 (53%) patients after supratentorial strokes.42 Interestingly, the authors documented presence of some supratentorial blood flow or perfusion in five out of six patients with infratentorial strokes, although in all cases infratentorial blood flow was absent or abnormal.42 Ancillary testing may also be perceived as providing additional certainty of BD/DNC in patients with isolated brainstem injury. This perception was supported by a Canadian survey of intensive care physicians, which found that over half believed ancillary testing should be performed in this context.44
Despite the apparent inconsistency between the two formulations of BD/DNC, there are reassuring data that the natural history of patients who appear brain dead after isolated infratentorial injuries is to progress to infarction of the supratentorial brain, thus eventually satisfying criteria of the “whole brain death” construct.28 Mechanistically, this progression may occur through progressive hydrocephalus and increased intracranial pressure leading to whole brain infarction. One study of patients with isolated brainstem injuries and clinical exams consistent with BD/DNC observed that all patients who had initial evidence of supratentorial blood flow on computed tomographic angiography (CTA) lost that flow with repeat imaging.41 A neurophysiologic study found that EEG activity persisted in 3.5% of patients who were clinically determined to be brain dead, but this activity disappeared with repeated recordings.45 Although not always temporally synchronized, there are data showing eventual convergence of ancillary testing (e.g., CTA or EEG) and a clinical diagnosis of BD/DNC.46 Nevertheless, specific situations should raise caution, as in the case of patients with posterior fossa decompressions for a primary infratentorial lesion. A case was recently reported of a patient with a cerebellar hemorrhage who underwent posterior fossa decompression and who had return of spontaneous respiration 12 hr after completing a clinical examination for BD/DNC.47 Return of respiratory activity was thought to be caused by relief of posterior brainstem compression by delayed herniation of the cerebellum through the skull defect. Ancillary testing with documentation of intracranial circulatory arrest has therefore been advised to support the determination of BD/DNC in this scenario.47
Conclusion
Clinical and neurophysiologic experiments have made clear that the RAS is a fundamental requirement for consciousness. This is further emphasized in modern medicolegal requirements of BD/DNC, in which destruction of the RAS and loss of capacity for consciousness are inferred by the complete absence of brainstem function. Nevertheless, a particular challenge for clinicians may arise in patients with isolated posterior fossa injuries, where the possibility of residual viability of the RAS or its projections makes it difficult to infer complete and permanent loss of consciousness in all cases with complete certainty. Notwithstanding that these concerns are mostly theoretical, they do provide an occasion to reflect on current clinical practices as they relate to this rare scenario. In whole brain death jurisdictions, ancillary testing has been suggested in patients with isolated infratentorial injuries before finalizing the determination of BD/DNC.28 A supportive ancillary test may also provide reassurance to clinicians when the capacity for consciousness cannot be definitively disproven—whether because of the nature or pattern of the brainstem injury or because of residual confounders. Furthermore, since clinical criteria and ancillary tests seem to achieve eventual if not immediate concordance, there is little concern that waiting will change the initial presumption of BD/DNC. It may, however, mitigate the perception of having made a premature diagnosis. These factors may collectively favor obtaining ancillary tests to support the clinical determination of death in patients with isolated infratentorial injuries.
Acknowledgments
Author contributions
Shaurya Taran and Jeff M. Singh contributed to the conception of the article. Shaurya Taran, Priti Gros, and Jeff M. Singh drafted the manuscript and prepared and/or revised figures. All authors critically revised the manuscript for intellectually important content.
Acknowledgments
The authors would like to thank the Brain Death Working Group for their comments and suggestions on a prior version of this manuscript.
Disclosures
Gordon Boyd and Jeff M. Singh report serving as regional medical leads with Ontario Health—Trillium Gift of Life Network (TGLN), a nonprofit agency of the Government of Ontario responsible for the province’s organ and tissue donation strategy, promotion, and supply. Gordon Boyd reports receiving cerebral oximeters from Edwards LifeSciences for research purposes. The remaining authors have no additional relevant associations or consultancies to declare.
Funding statement
No funding sources to declare.
Editorial responsibility
This submission was handled by Dr. James L. Bernat, Guest Editor, Canadian Journal of Anesthesia/Journal canadien d’anesthésie.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Penfield W, Roberts L. Speech and Brain Mechanisms. Princeton: Princeton University Press; 1959. [Google Scholar]
- 2.Wijdicks EF. The ascending reticular activating system. Neurocrit Care. 2019;31:419–422. doi: 10.1007/s12028-019-00687-7. [DOI] [PubMed] [Google Scholar]
- 3.Wijdicks EF. Historical awareness of the brainstem: from a subsidiary structure to a vital center. Neurology. 2020;95:484–488. doi: 10.1212/wnl.0000000000010504. [DOI] [PubMed] [Google Scholar]
- 4.Legallois J. Expériences sur le principe de la vie, notamment sure celui des mouvements du cœur, et le siège de ce principe; suivies du Rapport fait à la première classe de l’Institute sur celles relatives aux mouvements du cœur. Paris: Chez D’Hautel; 1802. [Google Scholar]
- 5.Flourens P. Note sur le point vital de la moëlle allongée. Compte Rendu des Séances de l’Académie des Sciences. 1851;33:437–439. [Google Scholar]
- 6.Bell C. Of the nerves which associate the muscles of the chest, in the actions of breathing, speaking and expression. Being a continuation of the paper on the structure and functions of the nerves. Phil Trans 1822; 112: 284–312. 10.1098/rstl.1822.0024
- 7.Bremer F. Cerveau isole et physiologie du sommeil. CR Soc Biol (Paris) 1935;118:1235–1241. [Google Scholar]
- 8.Moruzzi G, Magoun HW. Brain stem reticular formation and activation of the EEG. Electroencephalogr Clin Neurophysiol. 1949;1:455–473. doi: 10.1016/0013-4694(49)90219-9. [DOI] [PubMed] [Google Scholar]
- 9.Grady FS, Boes AD, Geerling JC. A century searching for the neurons necessary for wakefulness. Front Neurosci 2022; 16: 930514. 10.3389/fnins.2022.930514 [DOI] [PMC free article] [PubMed]
- 10.von Economo K. Encephalitis lethargica. Wien Klin Wochenschr. 1917;30:581–585. [Google Scholar]
- 11.Leblanc R. The white paper: Wilder Penfield, the stream of consciousness, and the physiology of mind. J Hist Neurosci. 2019;28:416–436. doi: 10.1080/0964704x.2019.1651135. [DOI] [PubMed] [Google Scholar]
- 12.French JD, Magoun HW. Effects of chronic lesions in central cephalic brain stem of monkeys. AMA Arch Neurol Psychiatry. 1952;68:591–604. doi: 10.1001/archneurpsyc.1952.02320230017002. [DOI] [PubMed] [Google Scholar]
- 13.Steriade M, McCormick DA, Sejnowski TJ. Thalamocortical oscillations in the sleeping and aroused brain. Science. 1993;262:679–685. doi: 10.1126/science.8235588. [DOI] [PubMed] [Google Scholar]
- 14.Parvizi J, Damasio AR. Neuroanatomical correlates of brainstem coma. Brain. 2003;126:1524–1536. doi: 10.1093/brain/awg166. [DOI] [PubMed] [Google Scholar]
- 15.Schiff ND, Ribary U, Moreno DR, et al. Residual cerebral activity and behavioural fragments can remain in the persistently vegetative brain. Brain. 2002;125:1210–1234. doi: 10.1093/brain/awf131. [DOI] [PubMed] [Google Scholar]
- 16.Kretschmer E. Das apallische Syndrom. Z Gesamte Neurol Psychiatr. 1940;169:576–579. doi: 10.1007/BF02871384. [DOI] [Google Scholar]
- 17.Jennett B, Plum F. Persistent vegetative state after brain damage. A syndrome in search of a name. Lancet 1972; 1: 734–7. 10.1016/s0140-6736(72)90242-5 [DOI] [PubMed]
- 18.Kinney HC, Korein J, Panigrahy A, Dikkes P, Goode R. Neuropathological findings in the brain of Karen Ann Quinlan. The role of the thalamus in the persistent vegetative state. N Engl J Med 1994; 330: 1469–75. 10.1056/nejm199405263302101 [DOI] [PubMed]
- 19.Adams JH, Graham DI, Jennett B. The neuropathology of the vegetative state after an acute brain insult. Brain. 2000;123:1327–1338. doi: 10.1093/brain/123.7.1327. [DOI] [PubMed] [Google Scholar]
- 20.Giacino JT, Ashwal S, Childs N, et al. The minimally conscious state: definition and diagnostic criteria. Neurology. 2002;58:349–353. doi: 10.1212/wnl.58.3.349. [DOI] [PubMed] [Google Scholar]
- 21.Claassen J, Velazquez A, Meyers E, et al. Bedside quantitative electroencephalography improves assessment of consciousness in comatose subarachnoid hemorrhage patients. Ann Neurol. 2016;80:541–553. doi: 10.1002/ana.24752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Boly M, Tshibanda L, Vanhaudenhuyse A, et al. Functional connectivity in the default network during resting state is preserved in a vegetative but not in a brain dead patient. Hum Brain Mapp. 2009;30:2393–2400. doi: 10.1002/hbm.20672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Vanhaudenhuyse A, Noirhomme Q, Tshibanda LJ, et al. Default network connectivity reflects the level of consciousness in non-communicative brain-damaged patients. Brain. 2010;133:161–171. doi: 10.1093/brain/awp313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Benson RR, Meda SA, Vasudevan S, et al. Global white matter analysis of diffusion tensor images is predictive of injury severity in traumatic brain injury. J Neurotrauma. 2007;24:446–459. doi: 10.1089/neu.2006.0153. [DOI] [PubMed] [Google Scholar]
- 25.Newcombe VF, Williams GB, Scoffings D, et al. Aetiological differences in neuroanatomy of the vegetative state: insights from diffusion tensor imaging and functional implications. J Neurol Neurosurg Psychiatry. 2010;81:552–561. doi: 10.1136/jnnp.2009.196246. [DOI] [PubMed] [Google Scholar]
- 26.Parra-Morales AM, Rudas J, Vargas JA, et al. Structural and functional connectivity of ascending reticular activating system in a patient with impaired consciousness after a cardiac arrest: a case report. Medicine (Baltimore) 2019; 98: e15620. 10.1097/md.0000000000015620 [DOI] [PMC free article] [PubMed]
- 27.Ordóñez-Rubiano EG, Johnson J, Enciso-Olivera CO, et al. Reconstruction of the ascending reticular activating system with diffusion tensor tractography in patients with a disorder of consciousness after traumatic brain injury. Cureus 2017; 9: e1723. 10.7759/cureus.1723 [DOI] [PMC free article] [PubMed]
- 28.Greer DM, Shemie SD, Lewis A, et al. Determination of brain death/death by neurologic criteria: the World Brain Death Project. JAMA. 2020;324:1078–1097. doi: 10.1001/jama.2020.11586. [DOI] [PubMed] [Google Scholar]
- 29.Mohandas A, Chou SN. Brain death. A clinical and pathological study. J Neurosurg 1971; 35: 211–8. 10.3171/jns.1971.35.2.0211 [DOI] [PubMed]
- 30.Mollaret P, Goulon M. The depassed coma (preliminary memoir) [French] Rev Neurol (Paris) 1959;101:3–15. [PubMed] [Google Scholar]
- 31.Beecher HK. A definition of irreversible coma: report of the ad hoc committee of the Harvard Medical School to examine the definition of brain death. JAMA. 1968;205:337–340. doi: 10.1001/jama.1968.03140320031009. [DOI] [PubMed] [Google Scholar]
- 32.Wijdicks EF, Varelas PN, Gronseth GS, Greer DM, American Academy of Neurology. Evidence-based guideline update: determining brain death in adults: report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology 2010; 74: 1911–8. 10.1212/wnl.0b013e3181e242a8 [DOI] [PubMed]
- 33.Shemie SD, Doig C, Dickens B, et al. Severe brain injury to neurological determination of death: Canadian forum recommendations. CMAJ. 2006;174:S1–13. doi: 10.1503/cmaj.045142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Wijdicks EF. Determining brain death in adults. Neurology. 1995;45:1003–1011. doi: 10.1212/wnl.45.5.1003. [DOI] [PubMed] [Google Scholar]
- 35.Lancet The. Diagnosis of death. Lancet. 1979;313:261–262. doi: 10.1016/S0140-6736(79)90781-5. [DOI] [PubMed] [Google Scholar]
- 36.Black D, Turnberg L, London D, et al. Criteria for the diagnosis of brain stem death. Review by a working group convened by the Royal College of Physicians and endorsed by the Conference of Medical Royal Colleges and their Faculties in the United Kingdom. J R Coll Physicians Lond 1995; 29: 381–2. [PMC free article] [PubMed]
- 37.Wijdicks EF. Brain death worldwide: accepted fact but no global consensus in diagnostic criteria. Neurology. 2002;58:20–25. doi: 10.1212/wnl.58.1.20. [DOI] [PubMed] [Google Scholar]
- 38.Bernat JL. The biophilosophical basis of whole-brain death. Soc Philos Policy. 2002;19:324–342. doi: 10.1017/S0265052502192132. [DOI] [PubMed] [Google Scholar]
- 39.Smith M. Brain death: the United kingdom perspective. Semin Neurol. 2015;35:145–151. doi: 10.1055/s-0035-1547534. [DOI] [PubMed] [Google Scholar]
- 40.Smith M, Citerio G. Death determined by neurological criteria: the next steps. Intensive Care Med. 2017;43:1383–1385. doi: 10.1007/s00134-017-4676-5. [DOI] [PubMed] [Google Scholar]
- 41.Varelas PN, Brady P, Rehman M, et al. Primary posterior fossa lesions and preserved supratentorial cerebral blood flow: implications for brain death determination. Neurocrit Care. 2017;27:407–414. doi: 10.1007/s12028-017-0442-3. [DOI] [PubMed] [Google Scholar]
- 42.Kramer AH, Kromm JA, Doig CJ, Chassé M, Couillard PL. Neurological determination of death following infratentorial stroke: a population-based cohort study. Can J Neurol Sci. 2022;49:553–559. doi: 10.1017/cjn.2021.177. [DOI] [PubMed] [Google Scholar]
- 43.Plourde G, Briard JN, Shemie SD, Shankar JJ, Chassé M. Flow is not perfusion, and perfusion is not function: ancillary testing for the diagnosis of brain death. Can J Anesth. 2021;68:953–961. doi: 10.1007/s12630-021-01988-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Chassé M, Neves Briard J, Yu M, et al. Clinical evaluation and ancillary testing for the diagnosis of death by neurologic criteria: a cross-sectional survey of Canadian intensivists. Can J Anesth. 2021;69:353–363. doi: 10.1007/s12630-021-02166-0. [DOI] [PubMed] [Google Scholar]
- 45.Fernández-Torre JL, Hernández-Hernández MA, Muñoz-Esteban C. Non confirmatory electroencephalography in patients meeting clinical criteria for brain death: scenario and impact on organ donation. Clin Neurophysiol. 2013;124:2362–2367. doi: 10.1016/j.clinph.2013.05.028. [DOI] [PubMed] [Google Scholar]
- 46.Hernández-Hernández M, Marco de Lucas E, Muñoz-Esteban C, Hernández JL, Fernández-Torre JL. The observation period after clinical brain death diagnosis according to ancillary tests: differences between supratentorial and infratentorial brain injury. J Neurol 2019; 266: 1859–68. 10.1007/s00415-019-09338-5 [DOI] [PubMed]
- 47.Walter U, Eggert M, Walther U, et al. A red flag for diagnosing brain death: decompressive craniectomy of the posterior fossa. Can J Anesth. 2022;69:900–906. doi: 10.1007/s12630-022-02265-6. [DOI] [PMC free article] [PubMed] [Google Scholar]