Abstract
Human coronaviruses (HCoV) cause common colds but can also infect neural cell cultures. To provide definitive experimental evidence for the neurotropism and neuroinvasion of HCoV and its possible association with multiple sclerosis (MS), we have performed an extensive search and characterization of HCoV RNA in a large panel of human brain autopsy samples. Very stringent reverse transcription-PCR with two primer pairs for both viral strains (229E and OC43), combined with Southern hybridization, was performed on samples from 90 coded donors with various neurological diseases (39 with MS and 26 with other neurological diseases) or normal controls (25 patients). We report that 44% (40 of 90) of donors were positive for 229E and that 23% (21 of 90) were positive for OC43. A statistically significant higher prevalence of OC43 in MS patients (35.9%; 14 of 39) than in controls (13.7%; 7 of 51) was observed. Sequencing of nucleocapsid protein (N) gene amplicons revealed point mutations in OC43, some consistently found in three MS patient brains and one normal control but never observed in laboratory viruses. In situ hybridization confirmed the presence of viral RNA in brain parenchyma, outside blood vessels. The presence of HCoV in human brains is consistent with neuroinvasion by these respiratory pathogens. Further studies are needed to distinguish between opportunistic and disease-associated viral presence in human brains.
Multiple sclerosis (MS) is a central nervous system (CNS) disease characterized by patches of demyelination and infiltration of inflammatory cells (21). The etiology of this disabling disease has not yet been determined, but both genetic factors, such as genes encoding human leukocyte antigens, T-cell receptors, and myelin basic protein (MBP) (18, 19, 26), and environmental factors such as viruses have been implicated (31). At least four human demyelinating diseases have a known viral etiology: subacute sclerosing panencephalitis as a late complication of measles virus infection of childhood (35), progressive multifocal leukoencephalopathy caused by the JC papovavirus (57), encephalopathy and myelopathy (neuro-AIDS) caused by human immunodeficiency virus (43), and human T-lymphotropic virus type 1-associated myelopathy/tropical spastic paraparesis (28). Over the last decades, several viruses have been associated with MS, based on detection of virions, viral nucleic acids, or viral proteins in CNS or the presence of antiviral antibodies in serum and/or cerebrospinal fluid. A confirmed association with MS is awaited but may involve more than one virus.
Several studies have associated human coronaviruses (HCoV) with MS. Coronavirus-like particles were detected in autopsied brain tissue from an MS patient (56). Two coronaviruses that are molecularly related to murine neurotropic coronaviruses were isolated from brain material obtained at autopsy from two MS patients (9). Intrathecal anti-HCoV antibody synthesis indicative of a CNS infection was reported in MS patients (45). HCoV RNA was detected in MS patient brains (37, 51) and in cerebrospinal fluid of MS and other neurological disease (OND) patients (15). Coronavirus antigens were also detected in MS patient brains (37). Moreover, we have shown that HCoV can infect human astrocytes and microglia in primary cultures (8) and can acutely and persistently infect immortalized human glial cells (4, 5). Thus, accumulating evidence suggests that these viruses, first isolated as pathogens of the respiratory tract and now associated with up to one-third of human common colds (39), might be neurotropic, neuroinvasive, and neurovirulent in humans, as is the case for their murine counterpart, the coronavirus mouse hepatitis virus (MHV). Interestingly, upper respiratory infections of viral origin were shown to be an important trigger of MS attacks (2, 40, 48). Moreover, coronavirus seasonal patterns fit the observed occurrence of MS exacerbations (48).
MHV-induced demyelinating disease involving coronaviruses is used as an animal model for elucidating the complex pathogenesis of MS. As for MS, MHV pathogenesis is multifactorial (27). Its outcome is influenced by genetics of the host and virus, dose and route of inoculation, and host age and immunological status at the time of infection (58). Neurotropic MHV strains could invade the CNS following an intranasal inoculation in mice (34) and could also gain access to the CNS via the hematogenous and/or lymphatic systems in mice (7) and in primates (11). Given that HCoV are respiratory viruses, they might also invade the CNS following a primary infection of the upper respiratory tract. Moreover, both known strains of HCoV, OC43 and 229E, can infect macrophages (13, 41) and 229E can infect human brain endothelial cells (10), which are possible alternative routes for CNS invasion.
Given these observations for animal models and humans, demonstration and characterization of the neurotropism, neuroinvasion, and neurovirulence of HCoV are necessary to elucidate the possible link between these ubiquitous viruses and CNS pathologies such as MS and possibly others. In order to characterize the possible in vivo neurotropism and neuroinvasion of HCoV in humans, we analyzed human brain samples for the presence of both known strains, OC43 and 229E. We looked for the presence of HCoV RNA within the CNS of a large panel of donors comprising those with MS or OND and healthy patients. Very stringent reverse transcription-PCR (RT-PCR) coupled to Southern hybridization was performed. We report the detection of HCoV RNA in a large proportion of donors. A preferential association of the presence of viral strain OC43 with MS was observed. Moreover, we detected viral RNA by Northern hybridization and in situ hybridization. Our results provide a strong indication for the neurotropism and neuroinvasion of these respiratory pathogens.
MATERIALS AND METHODS
Brain samples.
Frozen coded brain samples from MS patients and control donors were received from the following brain banks: The Multiple Sclerosis Society Tissue Bank, Institute of Neurology, University of London, London, United Kingdom (Jia Newcombe); Laboratoire de Neuropathologie Raymond Escourolle, Groupe Hospitalier Pitié-Salpétrière, Paris, France (Jean-Jacques Hauw and Danielle Seilhean); Rocky Mountain Multiple Sclerosis Center Tissue Bank, Englewood, Colo. (Catalin Butunoi and Ronald S. Murray); Multiple Sclerosis Human Neurospecimen Bank, West Los Angeles VA Medical Center, Los Angeles, Calif.) (Virginia Sanders and Wallace W. Tourtellotte); Douglas Hospital Research Center Brain Bank, McGill University, Verdun, Québec, Canada (Danielle Cécyre, Yvan Dumont, and Rémi Quirion). Samples were stored at −70°C.
RNA, RT-PCR, and Southern hybridization.
Total cellular RNA from human brain tissues was extracted as already described (4). Briefly, tissues were lysed with guanidinium isothiocyanate buffer and then layered onto a cesium chloride cushion for a 12- to 20-h centrifugation at 150,000 × g. The RNA pellet was resuspended in H2O. The pairs of primers used for amplification of the RNA of HCoV-OC43, HCoV-229E, and MBP (internal gene control) are described in Table 1. Forty picomoles of the inversely complementary primer was incubated with total cellular RNA at 65°C for 5 min to denature RNA; this was followed by a slow cool down to 37°C for annealing. Reverse transcription with Moloney murine leukemia virus reverse transcriptase (38 U; Pharmacia, Baie d'Urfé, Québec, Canada) was performed at 42°C for 90 min in the presence of a mixture of 48 to 75 U of RNA Guard (Pharmacia), 0.4 mM (each) deoxynucleoside triphosphate (dNTP) (Pharmacia), and 1× reverse transcriptase buffer (50 mM Tris-HCl [pH 8.0], 62.5 mM KCl, 3 mM MgCl2, 10 mM dithiothreitol [DTT]). Optimal PCR conditions were determined with cDNA transcribed from positive-control RNAs. For PCR, one-fifth of the synthesized cDNA was incubated in the presence of either 20 (O1 and O3, O7 and O9, and M1 and M3 primer pairs) or 50 pmol (E7 and E9 and E11 and E13 primer pairs) of the sense and antisense primers, with either 2.0 (E7 and E9 and O7 and O9 primer pairs), 2.5 (O1 and O3 and M1 and M3 primer pairs), or 4.0 mM (E11 and E13 primer pair) MgCl2 (Roche Diagnostics, Laval, Québec, Canada), 1× PCR buffer (10 mM Tris-HCl [pH 8.3], 50 mM KCl; Roche Diagnostics), dNTP (0.2 mM dATP, dCTP, and dGTP and 0.6 mM dUTP; Roche Diagnostics), and 2 U of uracil DNA glycosylase (UNG; Roche Diagnostics) at 20°C for 10 min (UNG elimination of any potential contamination from a previous PCR) and then at 94°C for 10 min (inactivation of UNG and denaturation of DNA), and finally at 60°C for another 5 min (annealing). After the addition of Taq DNA polymerase (2.5 U; Roche Diagnostics) 30 cycles of 2 min at 72°C, 1 min at 94°C, and 2 min at 60°C were performed, with a final elongation step of 10 min at 72°C. DNA amplicons were separated by electrophoresis in a 1.5% (wt/vol) agarose gel. Southern hybridization was performed on amplicons according to standard techniques using a 32P-labeled internal oligonucleotide (O2, O8, E8, E12, or M2; Table 1). The suitability of RNA for RT-PCR amplification was verified by an RT-PCR specific for an internal control, MBP RNA. We worked in very stringent conditions in order to avoid any contamination from laboratory viruses or previous PCR amplicons. Positive-displacement pipettes and the UNG carryover prevention system were used. Moreover, a strict physical separation of pre- and post-PCR areas was enforced. RNA extracted from infected cells (HRT-18 cells infected by HCoV-OC43 and L-132 cells infected by HCoV-229E) were used as positive controls in our RT-PCR. A negative control consisting of master mixture solution without added RNA or cDNA was included in each reaction of RT and PCR, respectively, and put also on each agarose gel that was eventually transferred onto a nylon membrane and hybridized with a radiolabeled oligonucleotide. The sensitivity of the combination of the RT-PCR and Southern testing was estimated by preparing in vitro-transcribed RNA from the N gene cloned in pGEM-T vector as described below (“Preparation of riboprobes”). The RNA was purified from an agarose gel, quantified by spectrometry measurements, and then diluted and used to perform the RT-PCR and Southern hybridization under the same conditions used for brain RNA. The sensitivity of our RT-PCR/Southern hybridization test was estimated at 102 copies of the viral N gene (data not shown). We do not consider it possible that the viral sequences detected by RT-PCR could have been DNA since the MBP-specific RT-PCR encompassed different exons. A DNA contamination would have been detected by a larger PCR product than expected without introns, which we have never experienced with the numerous samples we have handled.
TABLE 1.
Primers used to amplify HCoV-229E and HCoV-OC43 N and MBP genes
| RNA amplified (references) | Primer
|
|
|---|---|---|
| Sequence (5′–3′) | Designation: corresponding region on published sequence | |
| HCoV-OC43 N (4, 29) | CCCAAGCAAACTGCTACCTCTCAG | O1 sense: 215–238 |
| GTAGACTCCGTCAATATCGGTGCC | O3 antisense: 497–520 | |
| GATGGCAACCAGCGTCAACTGCTG | O2 probe: 419–442 | |
| CATCAGGAGGGAATGTTGTACC | O1.1 nested sense: 243–264 | |
| TACTGGTCTTTAGCATGCGGTC | O3.1 nested antisense: 474–495 | |
| GGATGCCACTAAACCTCAGCAAG | O7 sense: 920–942 (738–760)a | |
| GGTGCGAGTTCTGCAAAGATGG | O9 antisense: 1114–1135 (954–975)a | |
| GGAGCCCCAATAAACAATGC | O8 probe: 1005–1024 (821–840)a | |
| HCoV-229E N (5, 47) | TCTGCCAAGAGTCTTGCTCG | E7 sense: 819–838 |
| AGCATAGCAGCTGTTGACGG | E9 antisense: 1035–1054 | |
| GGAAGTGCAGGTGTTGTGGC | E8 probe: 969–988 | |
| CAAAAGAACAAAAGCATGAAATCG | E7.1 nested sense: 856–879 | |
| GCTCAGCAAATTGTGGATAGC | E9.1 nested antisense: 1009–1029 | |
| TCAGAACTAGAAAGGGCAAACGG | E11 sense: 337–359 | |
| GGGAGCTTTTGATTGAAGTGTGG | E13 antisense: 525–547 | |
| CGTGTTGAAGGTGTCGTCTGG | E12 probe: 432–452 | |
| MBP (52) | AGAACTGCTCACTACGGCTCCCTG | M1 sense: 274–297b |
| TCCAGAGCGACTATCTCTTCCTCC | M3 antisense: 550–573b | |
| CTGTCCCTGAGCAGATTTAGCTGG | M2 probe: 406–429b | |
| HCoV-229E N riboprobe (47) | TACAGTCAAATGGGCTGATG | ISH229E-H: 152–171 |
| GCGTTCACCTAGGTTCAGTAAC | ISH229E-I: 1543–1562 | |
| HCoV-OC43 N riboprobe (29) | CTGGTAATCGCATCCTCAAG | ISHOC43-H: 138–157 |
| AGAAGGCTGATGTCCTCTGC | ISHOC43-I: 1465–1484 | |
Error in the original publication for nucleotide numbers (the corrected numbers are in parentheses).
Position on the coding region only, without introns.
Nested PCR, cloning, and sequencing of amplicons.
Since PCR products were most of the time not detectable on an agarose gel, a nested PCR was performed on amplicons that were positive for the first round as detected by Southern hybridization, in order to obtain enough material for cloning and sequencing. PCR products yielding a positive signal in Southern blotting were used for nested PCR to allow sequencing of specific amplicons. Ten microliters of a 1/100 dilution of the first PCR product was incubated with 50 pmol (O1.1 and O3.1 and E7.1 and E9.1 primer pairs) of sense and antisense primers–0.4 mM (each) dNTP–1× PCR buffer–2.5 mM MgCl2 for 5 min at 94°C and then for 5 min at 60°C. Taq DNA polymerase was then added, and tubes were incubated for 30 cycles as described above. PCR products were cloned into the pCR2.1 vector (Invitrogen, Carlsbad, Calif.) and XL-1 blue bacteria and sequenced with an automated sequencer (ALF; Pharmacia) in both directions; at least three clones were used for each cloning reaction. Nucleotide sequences and predicted amino sequences were analyzed with Geneworks software, version 2.5.1 for Macintosh (Oxford Molecular Ltd., Oxford, United Kingdom).
Preparation of riboprobes.
RNA from infected cells (HRT-18 cells infected with HCoV-OC43 and L-132 cells infected with HCoV-229E; both laboratory HCoV strains were originally obtained from the American Type Culture Collection) was used to amplify a fragment of the N-coding RNA of each viral strain with primers described in Table 1. RT was performed as described above. PCR was done as for nested PCR described above; 1/10 of the cDNA was used with 20 pmol of sense and antisense primers. PCR products were cloned into the pGEM-T vector (Promega; distributed by Fisher Scientific Ltd., Nepean, Ontario, Canada) and then partially sequenced with an automated sequencer (ALF; Pharmacia) in both directions to verify the integrity of each sequence as well as its orientation. Plasmids were then linearized, and 1 to 10 μg of DNA was transcribed into RNA for 2 h at 37°C in the presence of a mixture consisting of SP6 or T7 RNA polymerase (20 U; Roche Diagnostics), 40 mM Tris-HCl (pH 7.4), 6 mM MgCl2, 20 mM DTT, 4 mM spermidine, RNase inhibitor (40 U; Roche Diagnostics), and 0.5 mM ATP, CTP, and GTP and 170 μCi of [32P]UTP (800 Ci/mmol, 20 mCi/ml; Amersham) for the riboprobes to be used for Northern hybridization or 0.5 mM ATP and GTP and 250 μCi of 35S-UTP and 35S-CTP (10 mCi/ml, >1,000 Ci/mmol; Amersham) for the riboprobes to be used for in situ hybridization. Plasmid DNA was digested with RNase-free DNase (10 U; Roche Diagnostics) in the presence of a mixture of 25 μg of yeast tRNA, 40 mM Tris-HCl (pH 7.4), 6 mM MgCl2, 10 mM DTT, 4 mM spermidine, and RNase inhibitor (40 U; Roche Diagnostics) for 15 min at 37°C. A phenol-chloroform extraction was performed, and the upper phase was transferred onto a G-50 Sephadex column to eliminate nonincorporated radiolabeled nucleotides. For in situ hybridization, probes were hydrolyzed in the presence of 40 mM NaHCO3–60 mM Na2CO3 for 15 min at 60°C, followed by a neutralization and precipitation step with 91 mM sodium acetate–0.5% acetic acid–15 μg of yeast tRNA–608 mM ammonium acetate to which was added 2.5 volumes of cold ethanol. The RNA pellet was resuspended in H2O. Probes labeled with 35S were maintained under reducing conditions by adding a final concentration of 10 mM DTT.
For Northern hybridization, 32P-labeled riboprobes were diluted in the hybridization buffer (5% [wt/vol] sodium dodecyl sulfate [SDS], 400 mM NaPO4, 1 mM EDTA, 1 mg/ml bovine serum albumin, 50% (vol/vol) formamide). For in situ hybridization, 35S-labeled riboprobes were diluted in the in situ hybridization buffer (75% [vol/vol] formamide, 3× SSC [1× SSC is 0.15 M NaCl plus 0.017 M sodium citrate], 0.1× Denhardt's solution, 200 μg of yeast tRNA/ml, 50 mM NaPO4 [pH 7.4], 0.1 g of dextran sulfate/ml) at a concentration of 3.3 × 104 cpm/μl.
Northern hybridization.
Five micrograms of denatured total RNA was separated by electrophoresis on a 1.5% (wt/vol) agarose–20 mM HEPES–1 mM EDTA–6% (vol/vol) formaldehyde gel. Bands were transferred overnight to a positively charged nylon membrane (Nytran; Xymotech, Mont-Royal, Québec, Canada) by capillary action in 10× SSC buffer (1.5 M NaCl, 0.17 M sodium citrate, pH 7.0). The membrane was dried at 65°C for 1 h, and then RNA was fixed with UV light. The membrane was stained with methylene blue to verify adequate transfer. Prehybridization (prehybridization buffer: 5% [wt/vol] SDS, 400 mM NaPO4, 1 mM EDTA, 1 mg of bovine serum albumin/ml, 50% [vol/vol] formamide) for at least 2 h at 60°C preceded overnight hybridization at 60°C with 32P-labeled riboprobes. Blots were washed for 20 min and then twice for 1 h in 0.1× SSC–0.1% (wt/vol) SDS–1 mM EDTA at 70°C and exposed to Kodak X-OMAT autoradiography film (17).
In situ hybridization.
In situ hybridization was performed with a slight modification of a published protocol (46). Tissue sections 5 to 10 μm thick were fixed in 4% (vol/vol) phosphate-buffered paraformaldehyde solution for 1 h at room temperature and then washed twice in phosphate-buffered saline, pH 7.4. A proteinase K treatment of 10 min at 37°C was performed (0.1 μg/ml) followed by the following washes: 5 min in H2O, 5 min in 0.1 M triethanolamine, 10 min in 0.1 M triethanolamine containing 0.25% (vol/vol) acetic anhydride, and then 5 min in 2× SSC. The washes were followed by dehydration in graded ethanol baths (50 to 95% [vol/vol]). A 35S-labeled (106-cpm) riboprobe was added onto each tissue section, and an overnight incubation at 55°C in a humid chamber with 75% (vol/vol) formamide was performed. Sections were washed with 2× SSC to remove excess hybridization buffer and then treated for 45 min at 37°C with RNase A (40 μg/ml), and finally the following washes were performed: three 10-min washes at room temperature in 2×, 1×, and 0.5× SSC and then one wash for 45 min at 60°C in 0.1× SSC. Sections were dehydrated in graded ethanol (50 to 100% [vol/vol]) prior to coating with K-5 nuclear emulsion (Illford), exposition for 1 to 2 months at 4°C, and then development with D19 (Kodak) for 2 min and fixation for 4 min with sodium thiosulfate (30% [wt/vol]). Tissues were dehydrated in graded ethanol baths and counterstained with cresyl violet. The counterstain performed after the hybridization procedure does not result in a typical Nissls staining. Neuron nuclei were weakly stained and are large and pale, while glial cell nuclei were stained strongly purple-blue and are smaller in size (17). Slides were observed with an Eclipse 800 Nikon microscope and a Darklite illuminator (Micro Video Instruments Inc., Avon, Mass.). A positive signal was characterized by a significant number of silver grains developed over cells compared with background.
RESULTS
Detection of HCoV RNA in human brains.
HCoV or murine-like coronavirus RNA has been previously detected in human brain samples, with an apparent preferential association with MS (37, 51). However, the establishment of the statistical significance of these observations justified the requirement for a larger number of patients. Therefore, we verified the presence of HCoV RNA within a large panel of human brain autopsy samples. We looked for viral RNA and not viral protein or infectious virions since it has been established that during persistent MHV infections of murine CNS, viral proteins and virions could not be detected for periods as long as could viral RNA (22). Coded brain samples were treated blindly, and only when experiments were completed were the codes broken to ensure complete objectivity in the interpretation of results. We amplified by RT-PCR segments of the nucleocapsid protein (N) gene of HCoV since this RNA is expressed in larger amounts during a viral infection and also because its sequence is present on all subgenomic and genomic viral RNAs (32). For each viral strain, two distinct regions of the N gene were amplified. In order to increase both the sensitivity and specificity of our assay, a Southern hybridization was performed on PCR amplicons. An experiment was considered successful and was included in our final results when both negative controls (RT and PCR) did not show any signal and also when the positive control showed a very strong hybridization signal. A brain donor was considered positive for one viral strain only when both RT-PCR primer pairs for that specific strain showed a positive signal on the Southern blot or RNA from two distinct extractions amplified by the same primer pair yielded positive Southern hybridization signals.
A description of patients is presented in Table 2. The time postdeath prior to brain removal varied between samples but did not influence the suitability of RNA for amplification by RT-PCR since we were able to amplify MBP RNA for at least one block from every patient from which a sample was obtained. A description of the histological localization of positive signals for viral detection by donors is presented in Table 3, when this information was available from brain banks. A positive signal for HCoV detection was often observed in more than one brain tissue block obtained from the same patient. Signals were observed in plaque samples but also in grey and white matter from both controls and MS patients. There was no apparent correlation between parts of the brain used for HCoV detection and results obtained.
TABLE 2.
Description of autopsy brain samples used and summary of HCoV RT-PCR results
| Diagnosis | Sex | Mean age ± range | HCoV detectiona
|
||
|---|---|---|---|---|---|
| 229E | OC43 | Both | |||
| MS | Male | 53 ± 12 | 11/20 | 7/20 | 5/20 |
| Female | 45 ± 13 | 9/19 | 7/19 | 6/19 | |
| Normal control | Male | 64 ± 19 | 9/19 | 4/19 | 4/19 |
| Female | 67 ± 17 | 2/5 | 1/5 | 0/5 | |
| ONNeurologicalD | |||||
| Alzheimer's | Male | 80 ± 2 | 0/2 | 0/2 | 0/2 |
| Female | 80 ± 6 | 2/8 | 1/8 | 0/8 | |
| Parkinson's | Male | 75 ± 3 | 3/4 | 0/4 | 0/4 |
| Female | 66 ± 18 | 1/2 | 0/2 | 0/2 | |
| Schizophrenia | Male | 65 ± 7 | 1/3 | 1/3 | 1/3 |
| Female | 67 | 0/1 | 0/1 | 0/1 | |
| Depression | Male | 81 | 1/1 | 0/1 | 0/1 |
| Female | 79 ± 3 | 0/2 | 0/2 | 0/2 | |
| Amyotrophic lateral sclerosis | Male | 80 | 0/1 | 0/1 | 0/1 |
| Bacterial meningitis | Male | 70 | 0/1 | 0/1 | 0/1 |
| Meningoencephalitis | Male | 68 | 1/1 | 1/1 | 1/1 |
Number of samples positive for viral strain(s)/number of samples tested.
TABLE 3.
Information concerning histological localization of positive signals in patients' brains
| Diagnosis | Brain localization | Detection of HCoV strain:
|
|
|---|---|---|---|
| 229E | OC43 | ||
| MS | Plaque | − | + |
| Normal grey matter | − | − | |
| Normal white matter | − | + | |
| MS | Normal tissue encircling plaque | + | − |
| Normal grey matter | − | − | |
| Active MS plaque | + | − | |
| MS | Normal tissue | + | − |
| Normal grey matter, cerebellum | + | − | |
| Normal tissue encircling plaque | − | + | |
| Active plaque | + | − | |
| Plaque | − | + | |
| Plaque | − | + | |
| Chronic plaque, Pons | + | − | |
| MS | Plaque | − | + |
| MS | Normal white matter | + | − |
| Plaque | − | + | |
| MS | Normal white matter | + | − |
| Plaque | + | − | |
| MS | White matter | − | + |
| White matter | + | − | |
| Grey matter | + | − | |
| Inactive plaque | + | − | |
| MS | Inactive plaque | + | − |
| MS | Subcortical grey matter | + | − |
| Active and inactive plaque | + | − | |
| MS | Active plaque | + | − |
| MS | Inactive plaque | + | − |
| MS | White matter | + | − |
| MS | Inactive plaque | + | − |
| Grey matter | − | + | |
| MS | White matter | − | + |
| Normal | Grey matter | + | − |
| White matter | + | − | |
| Normal | Grey matter | − | + |
| Grey matter | + | − | |
| Grey matter | + | − | |
| White matter | − | + | |
| Normal | White matter | + | − |
| Normal | White matter | + | − |
| Schizophrenia | Grey matter | + | − |
| White matter | + | − | |
Overall, 48% of all donors were positive for either one or both HCoV (Table 4). Considering that we have used on average a 200-mg tissue sample per block and that a human brain weighs between 750 g and 1 kg, we used less than 1/1,000 of the total brain. Given these considerations, we conclude that the proportion of human brain samples which were positive for one or both HCoV (48% of all) was relatively large. Statistical analysis of our results showed that the 229E strain was present in all groups and did not show a preferential association for women versus men or for patients with MS versus those with OND or normal controls. However, the OC43 strain was preferentially present in MS patients compared to those with OND (P = 0.0169) or compared to all controls (P = 0.0137) as calculated by chi-square and Fisher's exact tests, respectively. Normal controls did not show a statistically different proportion of HCoV-OC43-positive brains compared to MS patients.
TABLE 4.
Statistical analysisa of the association of the detection of HCoV RNA in human brain samples with MS diagnosis
| Diagnosis | No. of samples positive for indicated strain(s)/ no. tested (%)
|
||
|---|---|---|---|
| 229E | OC43 | 229E and OC43 | |
| MS | 20/39 (51.3) | 14/39 (35.9) | 11/39 (28.2) |
| OND | 9/26 (34.6) | 2/26 (7.7) | 2/26 (7.7) |
| Normal | 11/25 (44.0) | 5/25 (20.0) | 4/25 (16.0) |
| Total | 40/90 (44.4) | 21/90 (23.3) | 17/90 (18.9) |
For strains 222E plus OC43 and strain 229E alone, P was >0.05 for MS versus OND, normal controls, and all controls. For strain OC43 alone, P was 0.0169 (Fisher's exact P value) for MS versus OND, >0.05 for MS versus normal controls, and 0.0137 (chi-square P value) for MS versus all controls.
Molecular adaptation of viral strains in human brains.
To ascertain the coronaviral origin and also to verify the possible molecular adaptation of these viruses in human brain samples compared to laboratory viruses, we sequenced HCoV amplicons from human brains. It has been shown for the murine counterpart of HCoV, MHV, that point mutations and deletions arise during a persistent in vivo infection of the murine CNS (1, 44). Moreover, certain point mutations were mainly observed in animals experiencing chronic demyelinating disease and rarely in asymptomatic animals (42). The viral protein N was previously shown to have mutated in the CNS of the viral host (1). We compared sequences obtained to already-published sequences for each viral strain. We did not observe point mutations or deletions among the human brain amplicons for the 229E strain compared to viral amplicons obtained from our positive control (RNA from infected cells). Two point mutations for the OC43 strain, one leading to an amino acid change (position 398; AGA→GGA; R→G) and the other silent (position 442; CTG→CTA) were observed in four different brains: one normal control and three brains of MS patients. Moreover, an additional point mutation (position 261; GTA→GCA; V→A) was observed in one of the MS patient brains. These mutations were never observed in any positive controls (viral RNA from acutely infected patient cell lines). We conclude that these point mutations were probably representative of those in viral RNA present in human brains. Indeed, these changes suggest that HCoV could have adapted to the CNS, unlike the laboratory viruses, which we obtained from the American Type Culture Collection and which were initially isolated from the upper respiratory tract. Moreover, relative consistency of the viral sequence for the N gene strongly suggests that the amplified RNA from human brains was really from HCoV and not from an unknown source.
Detection of viral RNA in human brains by Northern hybridization.
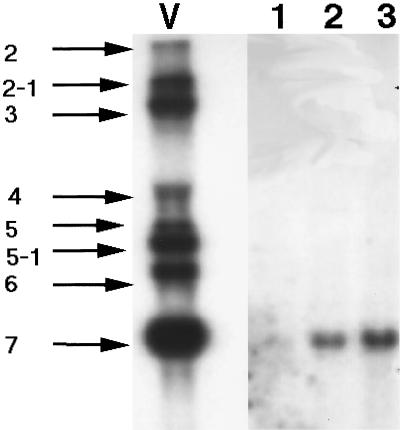
In order to evaluate the amount of viral RNA and whether a sufficient amount was detectable prior to attempting in situ hybridization experiments, we performed Northern hybridization on RNA taken from some human brains (four samples were tested, three of which were positive by RT-PCR and two of which yielded signals in Northern hybridization). Representative results obtained are shown in Fig. 1. Expected viral subgenomic RNAs, all of which retain the sequence of the N gene, were detected in the positive control (RNA from virus-infected cells). As expected (32), the subgenomic viral RNA encoding the N gene was expressed in the largest amounts in infected cells. Lanes 2, 3, and 4 represent hybridization to three different brain RNAs. We were able to detect RNA corresponding to the N gene of the OC43 strain on two of three RNA preparations. For the 229E strain, even though the positive control showed a strong signal for all subgenomic viral RNAs, we did not detect any signal in the human brain RNA tested (data not shown). Thus, viral RNA could be detected in human brains even without amplification by RT-PCR. Since this technique is less sensitive than RT-PCR coupled to Southern hybridization but has a sensitivity comparable to that of Northern blotting, this suggested that viral RNA may be present in sufficient amounts to be detected by in situ hybridization, which has even been reported to be more sensitive than Northern blotting when RNA is localized in a small portion of the tissue (3).
FIG. 1.
Northern hybridization for HCoV-OC43 on RNA extracted from human brains (lanes 1 to 3) or from positive-control HCoV-OC43-infected HRT-18 cells (lane V). HCoV-OC43 subgenomic RNAs are as follows, from top to bottom (estimated sizes and encoded proteins are indicated): RNA 2 (11.6 kb; ns2 protein); RNA 2-1 (8.8 kb; HE protein); RNA 3 (7.1 kb; S protein); RNA 4 (4.0 kb; ns4 protein); RNA 5 (3.3 kb; ns5 protein); RNA 5-1 (3.0 kb; E protein); RNA 6 (2.5 kb; M protein); RNA 7 (1.8 kb; N).
Localization of viral RNA by in situ hybridization.
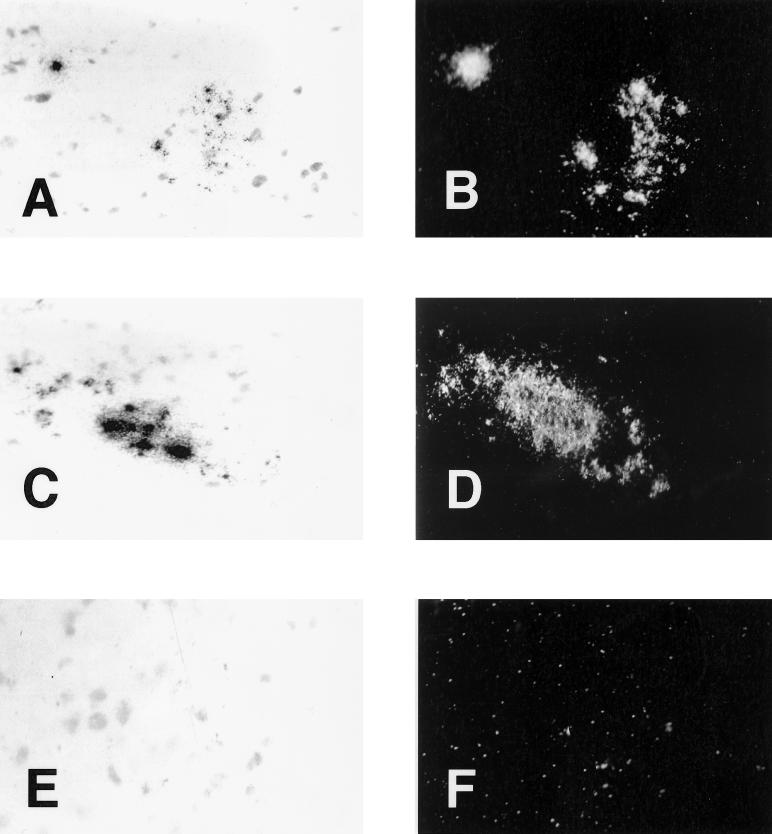
In order to verify the localization of viral RNA in the CNS, an essential confirmation of the suspected neuroinvasion, we performed in situ hybridization on human brain sections, using the same probe as for the Northern blot showing specific hybridization to viral RNA. In situ hybridization performed on immortalized human neuronal cells revealed radiolabeled riboprobe signals only on infected cells and not on noninfected cells (M. Viau, N. Arbour, and P. J. Talbot, unpublished observations). These experiments were also performed blindly, in order to provide absolute objectivity in the interpretation of results. At least two slides of the same tissue block had to be positive in order to consider the block positive. Samples from seven donors were selected on the basis of RT-PCR results. We did not observe any signal for the 229E strain, most likely because the amount of viral RNA was not sufficient for detection by this technique (data not shown). Indeed, we did not detect HCoV-229E RNA in human brains by Northern blotting, corroborating these in situ hybridization results (data not shown). On the other hand, we detected positive signals for the OC43 strain on two blocks from one MS patient and on one block from a healthy control. Samples from both of these donors were positive for HCoV-OC43 by RT-PCR. We did not detect any signal when slides adjacent to the positive ones were pretreated with RNase A prior to hybridization (data not shown). We also did not observe any positive signal with the sense probe, suggesting that the negative-strand viral RNA was not present in detectable amounts (data not shown). Hybridization of slides just adjacent to the positive ones with the 229E strain riboprobe did not yield any signal (data not shown). Examples of hybridization signals observed are shown in Fig. 2. Dark-field pictures clearly show that positive signals were really distinct from normal background. Three or four positive regions were randomly distributed on each positive slide. We often observed that more than one cell was positive in the same region. Signals obtained were never seen in blood vessels or grey matter but only in the white matter parenchyma.
FIG. 2.
Photomicrographs showing in situ hybridization for HCoV-OC43 RNA in human brain sections, using a radiolabeled riboprobe for the viral N gene. Results shown in panels A and B and in panels C and D were taken from two different blocks from the same MS patient. (E and F) Negative control; slides adjacent to those used for panels A and B; samples were treated by RNase A prior to in situ hybridization. Photographs shown in panels A, C, and E were taken under bright-field exposure, whereas those shown in panels B, D, and F were taken at the same site under dark-field exposure. Magnification = ×100.
DISCUSSION
Some murine coronaviruses are neurotropic, neuroinvasive, and neurovirulent in rodents (27, 58) and in nonhuman primates (11, 38), inducing in some cases an MS-like demyelinating disease. Our goal is to investigate the possible neurotropism, neuroinvasion, and neurovirulence of the related HCoV, which have so far only been definitively associated with respiratory infections, with occasional reports of associations with diarrhea and neurologic diseases, mainly MS (39). In the work presented herein, we searched for the presence of HCoV RNA in a large number of autopsied brain samples from patients diagnosed with MS or OND and normal controls. Viral RNA was present in a surprisingly large number of samples; 48% (44 of 90) of all donors were positive for one or both viral strains. There was no preferential association of the presence of HCoV-229E with a specific diagnosis. However, the OC43 strain was preferentially detected in MS patient brain samples (14 positive samples out of 39) compared to OND patient samples (2 positive samples out of 26) or the overall control group (OND patient samples plus normal samples; 7 positive samples out of 51). Given that HCoV-OC43 RNA was detected in normal white and grey matter as well as in plaques of MS patient brains, there was no direct association between the presence of viral RNA and the occurrence of plaques. The analysis of a larger number of brains positive for HCoV-OC43 would allow the evaluation of the preferential CNS localization of HCoV and would provide hints as to the potential effects of this neuroinfection on the CNS. Also, since the proportion of normal controls that harbored RNA from this viral strain (5 positive samples out of 25) was not statistically different from that for MS patients (14 positive samples out of 38), other studies will be necessary to clearly establish any possible association with MS. However, one has to keep in mind that some of the normal controls, who died without any known neurological disease, may have had neurological abnormalities within their CNS without visible clinical signs and could have represented patients with undiagnosed neurological disease (24). Our current results are consistent with those reported by another group showing that coronavirus RNA was also present in some donors who were not MS patients: 1 out of 5 patients with OND and 1 out of 16 normal controls (37). In the present study, we detected HCoV-229E RNA in several OND patients and normal controls. HCoV-OC43 RNA was detected in a smaller number of normal controls and OND patients than the other viral strain.
Given the fact that most human beings have been in contact with HCoV as respiratory pathogens by the age of five (39), we suggest that the presence of HCoV RNA in brain samples correlates with a persistent infection within the CNS. Indeed, it would be rather surprising that over 40% of the donors were experiencing an acute infection by HCoV leading to its detection within the CNS just prior to their death. Importantly, we have recently shown that HCoV can persist in some immortalized human neural cell lines (4, 5). Given our in vitro observations, we suggest that HCoV could also persist in vivo. However, since the blood-brain barrier is damaged in MS patients (21), we cannot rule out the possibility that HCoV-OC43 could establish a persistent infection in the CNS following onset of disease, given the easier access to the CNS in MS patients. However, our detection of HCoV RNA in normal brains strongly suggests that these viruses have access to the CNS prior to clinical disease.
Sequencing PCR amplicons confirmed that positive detection signals had a coronaviral origin and also showed that sequences of amplified fragments of the nucleocapsid protein were relatively stable. We did not detect any mutations in the 229E strain compared to the virus used in the laboratory. However, point mutations never observed in laboratory viruses were detected in amplicons of the OC43 strain; one point mutation was present in only one MS patient, and two other point mutations were present in three MS patients and one normal donor. Given that point mutations within the HCoV-OC43 amplicons were detected mainly in MS patients, we speculate that HCoV-OC43 may adapt to the human CNS and in certain cases cause neurological abnormalities, either directly or indirectly. Although we demonstrated, by sequencing coronaviral amplicons from human brains, that the N gene is relatively stable, certain positions might be important in the molecular adaptation of these viruses to the CNS. It will be important to sequence the RNA encoding the HCoV S protein since it has been shown to bear important determinants of neurovirulence in the animal model (16). Moreover, we have shown that point mutations and deletions arise during in vitro persistent HCoV infection of immortalized neural cells (4, 5). In these studies, we observed that the OC43 strain could persistently infect a larger number of cell lines and also that a greater number of mutations arose during such an infection, suggesting the emergence of viral quasispecies (4). Given our observations in human brains and in immortalized human neural cell lines, we suggest that HCoV-OC43 has the capacity to persistently infect cells in the human CNS and that such infections could lead in some cases to specific molecular adaptation of this virus to the CNS environment.
We have also been able to detect HCoV-OC43 RNA by Northern hybridization, suggesting a relatively important abundance of this viral RNA within the CNS of some patients. In situ hybridization experiments were also successfully completed and yielded convincing positive viral RNA signals, which were located outside any blood vessels but most probably within the parenchyma of white matter and plaque samples. Others have not been able to detect this virus in tissues from four patients with MS and one patient with a probable diagnosis of MS (49), although they probably did not study a sufficiently large number of patients. Murray and colleagues have previously reported the detection of viral RNA by in situ hybridization in human brain samples in 5 out of 12 MS patients for the HCoV-OC43 strain and in 12 out of 12 for the murine strain (37). However, their analysis by RT-PCR and in situ hybridization detected mouse-like coronavirus. These authors concluded that the 3′ ends of coronavirus RNAs they detected in brain were more MHV-like. They did not detect the 229E strain in any brain tested and did not detect either the OC43 strain or the murine one in controls (37). Our study was designed to specifically detect the HCoV, and it would have been surprising to have detected mouse-like sequences, since amplicons obtained by RT-PCR were either identical or very closely related to HCoV-OC43. Two tissue blocks from one MS patient representing two plaque areas were positive for HCoV-OC43 by in situ hybridization. Results using different corroborating techniques presented herein strongly suggest that HCoV-OC43 is neuroinvasive in humans.
In animal models of coronavirus infections using MHV, the presence of virus or the viral genome or proteins is not always associated with pathological alterations detectable by clinical symptoms (14). Indeed, the gene products of the JHM strain of MHV can persist for prolonged periods without any apparent neurological disease (50). On the other hand, a persistent coronavirus infection could provoke a chronic demyelination in genetically predisposed mice. The demyelination observed in MHV-JHM-infected mice was reported to be mediated by immune mechanisms (27). Demyelination in MHV-infected animals was observed even when the viral antigen and RNA were detectable only in a small proportion of glial cells and not in dying oligodendrocytes (6). By analogy, it is also possible that the outcome of a coronavirus infection in human CNS could depend on immune responses involved in demyelination or clearance of virus from the CNS. We suggest that genetic factors of the host as well as genetics of viruses (58) could determine the consequence of a persistent presence of HCoV RNA in human CNS and explain the development of disease in only some individuals. We are currently also investigating the possibility that viral RNA could be localized in different regions or in different cell types or in a different persistent state, latent or chronic, in MS patients versus other patients and that this histological localization could be related to disease development. Nevertheless, our experimental data strongly suggest that HCoV RNA frequently persists in human brains. Even though the possible pathological effects of such presence in the CNS are still unknown, investigations should be pursued given observations in animal models of neurological disease and the relevance of coronaviruses in inducing MS-like disease in laboratory animals. Moreover, the results of our study also suggest the possibility that HCoV may be associated with other neurological diseases.
It has been previously shown that the presence of viruses in the CNS does not necessarily correlate with disease. It seems that factors other than the presence of such viruses are necessary for the development of pathology. For example, measles virus (30) and JC virus (59) were detected in autopsied brain tissues from patients who had no apparent clinical symptoms. Similar observations could be envisaged for HCoV in the human CNS. Human herpesvirus 6 (HHV-6) is another virus candidate in MS etiology. It seems to be a commensal virus in human CNS since over 70% of MS patients and controls were positive by PCR (12). However, HHV-6 antigens were detected in oligodendrocytes of MS patients and not in controls (12). HCoV and HHV-6 may be part of a virus group implicated in MS etiology. Indeed, various viral pathogens have been associated with MS over the years, including, for example, measles virus and retroviruses (54). Given the large variety of clinical symptoms of this disease, the involvement of several viruses would not be impossible (54), although a common pathogenic mechanism can be envisaged. Alternatively, it is also possible that a generalized alteration of the immune system in MS patients would allow the establishment of viral persistence or the reactivation of viruses not detectable in healthy controls.
There are several possible mechanisms by which a virus could induce a demyelinating disease such as MS. It could be due to direct consequences of viral infection such as lytic infection of oligodendrocytes, as is observed for JC virus (57). It could also involve bystander effects of the viral infection, such as expression of cytotoxic molecules by glial cells. It has been shown that during an MHV-induced chronic demyelination in the CNS of mice, several inflammatory molecules, such as interleukin 1β (IL-1β), tumor necrosis factor, IL-6, type 2 nitric oxide synthase (53), cytokine response gene 2, RANTES, and macrophage-inflammatory protein 1β, or their mRNAs are detected (33). Upregulation of MHC class I mRNA and antigen expression has been observed in MHV-infected animals (25, 36). The fact that HCoV-OC43 and MHV belong to the same antigenic group (32) is consistent with the possibility that this human virus could act similarly to its murine counterpart in the CNS of its host. We are currently testing the hypothesis that HCoV could induce the secretion of proinflammatory cytokines and molecules during infection of neural cells (19a). It is also possible that a viral infection primes an immune response to cross-react with myelin antigens that are targeted in MS (20, 23). This hypothesis is being evaluated in our laboratory, and we have already shown the presence in MS patients of peripheral cross-reactive T-cell clones recognizing both HCoV and a myelin antigen (55; A. Boucher, G. Mercier, P. Duquette, and P. J. Talbot, J. Neuroimmunol. 90:33, 1998). We hypothesize that the presence of HCoV RNA could lead in certain circumstances to a low level of viral protein production that could be involved in the stimulation of immune responses within the CNS, thereby exacerbating the effect of a coronaviral infection in MS patients.
Given the observations made in animal models, we suggest that coronavirus neurovirulence may also be possible in genetically predisposed human beings, especially for the OC43 strain. Several host factors in humans could influence the outcome of the HCoV CNS infection. Animal models may provide clues for experiments to be performed with human systems. Thus, HCoV can be added to the growing list of viruses that persist in the CNS, a viral flora of the brain that could have pathological consequences in some individuals but remain subclinical and perhaps even beneficial in others.
ACKNOWLEDGMENTS
This work was supported by grant MT-9203 from the Medical Research Council of Canada to P.J.T., who also gratefully acknowledges a senior scholarship award from the Fonds de la Recherche en Santé du Québec. N.A. is grateful to the Institut Armand-Frappier as well as to the Multiple Sclerosis Society of Canada for studentship support.
We thank Weijia Dong and Xue Wen Yuan for excellent technical assistance. We thank Julie Edwards and François Denis for critically reviewing the manuscript. We are grateful to the following brain banks which have provided us with precious material for our study: Laboratoire de Neuropathologie Raymond Escourolle, Groupe Hospitalier Pitié-Salpétrière (Jean-Jacques Hauw and Danielle Seilhean); Rocky Mountain Multiple Sclerosis Center Tissue Bank (Catalin Butunoi and Ronald S. Murray); Multiple Sclerosis Human Neurospecimen Bank, West Los Angeles VA Medical Center (Virginia Sanders and Wallace W. Tourtellotte); and Douglas Hospital Research Centre Brain Bank, McGill University (Danielle Cécyre, Yvan Dumont, and Rémi Quirion).
REFERENCES
- 1.Adami C, Pooley J, Glomb J, Stecker E, Fazal F, Fleming J O, Baker S C. Evolution of mouse hepatitis virus (MHV) during chronic infection: quasispecies nature of the persisting MHV RNA. Virology. 1995;209:337–346. doi: 10.1006/viro.1995.1265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Andersen O, Lygner P E, Bergstrom T, Andersson M, Vahlne A. Viral infections trigger multiple sclerosis relapses: a prospective seroepidemiological study. J Neurol. 1993;240:417–422. doi: 10.1007/BF00867354. [DOI] [PubMed] [Google Scholar]
- 3.Angerer L M, Angerer R C. In situ hybridization to cellular RNA with radiolabelled RNA probes. In: Wilkinson D G, editor. In situ hybridization, a practical approach. New York, N.Y: IRL Press; 1992. pp. 15–32. [Google Scholar]
- 4.Arbour N, Côté G, Lachance C, Tardieu M, Cashman N R, Talbot P J. Acute and persistent infection of human neural cell lines by human coronavirus OC43. J Virol. 1999;73:3338–3350. doi: 10.1128/jvi.73.4.3338-3350.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Arbour N, Ékandé S, Côté G, Lachance C, Chagnon F, Tardieu M, Cashman N R, Talbot P J. Persistent infection of human oligodendrocytic and neuroglial cell lines by human coronavirus 229E. J Virol. 1999;73:3326–3337. doi: 10.1128/jvi.73.4.3326-3337.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Barac-Latas V, Suchanek G, Breitschopf H, Stuehler A, Wege H, Lassmann H. Patterns of oligodendrocyte pathology in coronavirus-induced subacute demyelinating encephalomyelitis in the Lewis rat. Glia. 1997;19:1–12. doi: 10.1002/(sici)1098-1136(199701)19:1<1::aid-glia1>3.0.co;2-5. [DOI] [PubMed] [Google Scholar]
- 7.Barthold S W, Smith A L. Viremic dissemination of mouse hepatitis virus-JHM following intranasal inoculation of mice. Arch Virol. 1992;122:35–44. doi: 10.1007/BF01321116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bonavia A, Arbour N, Yong V W, Talbot P J. Infection of primary cultures of human neural cells by human coronaviruses 229E and OC43. J Virol. 1997;71:800–806. doi: 10.1128/jvi.71.1.800-806.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Burks J S, DeVald B L, Jankovsky L D, Gerdes J C. Two coronaviruses isolated from central nervous system tissue of two multiple sclerosis patients. Science. 1980;209:933–934. doi: 10.1126/science.7403860. [DOI] [PubMed] [Google Scholar]
- 10.Cabirac G F, Murray R S, McLaughlin L B, Skolnick D M, Hogue B, Dorovini-Zis K, Didier P J. In vitro interaction of coronaviruses with primate and human brain microvascular endothelial cells. Adv Exp Med Biol. 1995;380:79–88. doi: 10.1007/978-1-4615-1899-0_11. [DOI] [PubMed] [Google Scholar]
- 11.Cabirac G F, Soike K F, Zhang J-Y, Hoel K, Butunoi C, Cai G Y, Johnson S, Murray R S. Entry of coronavirus into primate CNS following peripheral infection. Microb Pathog. 1994;16:349–357. doi: 10.1006/mpat.1994.1035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Challoner P B, Smith K T, Parker J D, MacLeod D L, Coulter S N, Rose T M, Schultz E R, Bennett J L, Garber R L, Chang M, Schad P A, Stewart P M, Nowinski R C, Brown J P, Burmer G C. Plaque-associated expression of human herpesvirus 6 in multiple sclerosis. Proc Natl Acad Sci USA. 1995;92:7440–7444. doi: 10.1073/pnas.92.16.7440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Collins A R. Human macrophages are susceptible to coronavirus OC43. Adv Exp Med Biol. 1998;440:635–639. doi: 10.1007/978-1-4615-5331-1_82. [DOI] [PubMed] [Google Scholar]
- 14.Compton S R, Barthold S W, Smith A L. The cellular and molecular pathogenesis of coronaviruses. Lab Anim Sci. 1993;43:15–28. [PubMed] [Google Scholar]
- 15.Cristallo A, Gambaro F, Biamonti G, Ferrante P, Battaglia M, Cereda P M. Human coronavirus polyadenylated RNA sequences in cerebrospinal fluid from multiple sclerosis patients. Microbiologica. 1997;2:105–114. [PubMed] [Google Scholar]
- 16.Dalziel R G, Lampert P W, Talbot P J, Buchmeier M J. Site-specific alteration of murine hepatitis virus type 4 peplomer glycoprotein E2 results in reduced neurovirulence. J Virol. 1986;59:463–471. doi: 10.1128/jvi.59.2.463-471.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Dong W, Marcinkiewicz M, Vieau D, Chrétien M, Seidah N G, Day R. Distinct mRNA expression of the highly homologous convertases PC5 and PACE4 in the rat brain and pituitary. J Neurosci. 1995;15:1778–1796. doi: 10.1523/JNEUROSCI.15-03-01778.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ebers G C, Kukay K, Bulman D E, Sadovnick A D, Rice G, Anderson C, Armstrong H, Cousin K, Bell R B, Hader W, Paty D W, Hashimoto S, Oger J, Duquette P, Warren S, Gray T, O'Connor P, Nath A, Auty A, Metz L, Francis G, Paulseth J E, Murray T J, Pryse-Phillips W, Nelson R, Freedman M, Brunet D, Bouchard J P, Hinds D, Risch N. A full genome search in multiple sclerosis. Nat Genet. 1996;13:472–476. doi: 10.1038/ng0896-472. [DOI] [PubMed] [Google Scholar]
- 19.Ebers G C, Sadovnick A D. The role of genetic factors in multiple sclerosis susceptibility. J Neuroimmunol. 1994;54:1–17. doi: 10.1016/0165-5728(94)90225-9. [DOI] [PubMed] [Google Scholar]
- 19a.Edwards J A, Denis F, Talbot P J. Activation of glial cells by human coronavirus OC43 infection. J Neuroimmunol. 2000;108:73–81. doi: 10.1016/S0165-5728(00)00266-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Fazakerley J F, Buchmeier M J. Pathogenesis of virus-induced demyelination. Adv Virus Res. 1993;42:249–324. doi: 10.1016/S0065-3527(08)60087-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ffrench-Constant C. Pathogenesis of multiple sclerosis. Lancet. 1994;343:271–275. doi: 10.1016/s0140-6736(94)91118-5. [DOI] [PubMed] [Google Scholar]
- 22.Fleming J O, Houtman J J, Alaca H, Hinze H C, McKenzie D, Aiken J, Bleasdale T, Baker S. Persistence of viral RNA in the central nervous system of mice inoculated with MHV-4. Adv Exp Med Biol. 1993;342:327–332. doi: 10.1007/978-1-4615-2996-5_50. [DOI] [PubMed] [Google Scholar]
- 23.Fujinami R S, Oldstone M B A. Amino acid homology and immune responses between the encephalitogenic site of myelin basic protein and virus: a mechanism for autoimmunity. Science. 1985;230:1043–1045. doi: 10.1126/science.2414848. [DOI] [PubMed] [Google Scholar]
- 24.Gilbert I J, Sadler M. Unsuspected multiple sclerosis. Arch Neurol. 1983;40:533. doi: 10.1001/archneur.1983.04050080033003. [DOI] [PubMed] [Google Scholar]
- 25.Gombold J L, Weiss S R. Mouse hepatitis virus A59 increases steady-state levels of MHC mRNAs in primary glial cell cultures and in the murine central nervous system. Microb Pathog. 1992;13:493–505. doi: 10.1016/0882-4010(92)90015-G. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hockertz M K, Paty D W, Beall S S. Susceptibility to relapsing-progressive multiple sclerosis is associated with inheritance of genes linked to the variable region of the TcR beta locus: use of affected family-based controls. Am J Hum Genet. 1998;62:373–385. doi: 10.1086/301700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Houtman J J, Fleming J O. Pathogenesis of mouse hepatitis virus-induced demyelination. J Neurovirol. 1996;2:361–376. doi: 10.3109/13550289609146902. [DOI] [PubMed] [Google Scholar]
- 28.Izumo S, Umehara F, Kashio N, Kubota R, Sato E, Osame M. Neuropathology of HTLV-1-associated myelopathy (HAM/TSP) Leukemia. 1997;11:82–84. [PubMed] [Google Scholar]
- 29.Kamahora T, Soe L H, Lai M M. Sequence analysis of nucleocapsid gene and leader RNA of human coronavirus OC43. Virus Res. 1989;12:1–9. doi: 10.1016/0168-1702(89)90048-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Katayama Y, Hotta H, Nishimura A, Tatsuno Y, Homma M. Detection of measles virus nucleoprotein mRNA in autopsied brain tissues. J Gen Virol. 1995;76:3201–3204. doi: 10.1099/0022-1317-76-12-3201. [DOI] [PubMed] [Google Scholar]
- 31.Kurtzke J F. Epidemiologic evidence for multiple sclerosis as an infection. Clin Microbiol Rev. 1993;6:382–427. doi: 10.1128/cmr.6.4.382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lai M M, Cavanagh D. The molecular biology of coronaviruses. Adv Virus Res. 1997;48:1–100. doi: 10.1016/S0065-3527(08)60286-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Lane T E, Asensio V C, Yu N, Paoletti A D, Campbell I L, Buchmeier M J. Dynamic regulation of α- and β-chemokine expression in the central nervous system during mouse hepatitis virus-induced demyelinating disease. J Immunol. 1998;160:970–978. [PubMed] [Google Scholar]
- 34.Lavi E, Fishman P S, Highkin M K, Weiss S R. Limbic encephalitis after inhalation of a murine coronavirus. Lab Investig. 1988;58:31–36. [PubMed] [Google Scholar]
- 35.Liebert U G. Measles virus infections of the central nervous system. Intervirology. 1997;40:176–184. doi: 10.1159/000150544. [DOI] [PubMed] [Google Scholar]
- 36.Massa P T, Dorries R, ter Meulen V. Viral particles induce Ia antigen expression on astrocytes. Nature. 1986;320:543–546. doi: 10.1038/320543a0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Murray R S, Brown B, Brian D, Cabirac G F. Detection of coronavirus RNA and antigen in multiple sclerosis brain. Ann Neurol. 1992;31:525–533. doi: 10.1002/ana.410310511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Murray R S, Cai G Y, Hoel K, Zhang J-Y, Soike K F, Cabirac G F. Coronavirus infects and causes demyelination in primate central nervous system. Virology. 1992;188:274–284. doi: 10.1016/0042-6822(92)90757-G. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Myint S H. Human coronaviruses: a brief review. Rev Med Virol. 1994;4:35–46. [Google Scholar]
- 40.Panitch H S. Influence of infection on exacerbations of multiple sclerosis. Ann Neurol. 1994;36:S25–S28. doi: 10.1002/ana.410360709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Patterson S, Macnaughton M R. Replication of human respiratory coronavirus strain 229E in human macrophages. J Gen Virol. 1982;60:307–314. doi: 10.1099/0022-1317-60-2-307. [DOI] [PubMed] [Google Scholar]
- 42.Pewe L, Wu G F, Barnett E M, Castro R F, Perlman S. Cytotoxic T cell-resistant variants are selected in a virus-induced demyelinating disease. Immunity. 1996;5:253–262. doi: 10.1016/s1074-7613(00)80320-9. [DOI] [PubMed] [Google Scholar]
- 43.Power C, McArthur J C, Nath A, Wehrly K, Mayne M, Nishio J, Langelier T, Johnson R T, Chesebro B. Neuronal death induced by brain-derived human immunodeficiency virus type 1 envelope genes differs between demented and nondemented AIDS patients. J Virol. 1998;72:9045–9053. doi: 10.1128/jvi.72.11.9045-9053.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Rowe C L, Baker S C, Nathan M J, Fleming J O. Evolution of mouse hepatitis virus: detection and characterization of spike deletion variants during persistent infection. J Virol. 1997;71:2959–2969. doi: 10.1128/jvi.71.4.2959-2969.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Salmi A, Ziola B, Hovi T, Reunanen M. Antibodies to coronaviruses OC43 and 229E in multiple sclerosis patients. Neurology. 1982;32:292–295. doi: 10.1212/wnl.32.3.292. [DOI] [PubMed] [Google Scholar]
- 46.Schäfer M K-H, Day R. In situ hybridization techniques to study processing enzyme expression at the cellular level. Methods Neurosci. 1994;23:16–44. [Google Scholar]
- 47.Schreiber S S, Kamahora T, Lai M M C. Sequence analysis of the nucleocapsid protein gene of human coronavirus 229E. Virology. 1989;169:142–151. doi: 10.1016/0042-6822(89)90050-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Sibley W A, Bamford C R, Clark K. Clinical viral infections and multiple sclerosis. Lancet. 1985;i:1313–1315. doi: 10.1016/S0140-6736(85)92801-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Sorensen O, Collins A, Flintoff W, Ebers G, Dales S. Probing for the human coronavirus OC43 in multiple sclerosis. Neurology. 1986;36:1604–1606. doi: 10.1212/wnl.36.12.1604. [DOI] [PubMed] [Google Scholar]
- 50.Sorensen O, Coulter-Mackie M B, Puchalski S, Dales S. In vivo and in vitro models of demyelinating disease. IX. Progression of JHM virus infection in the central nervous system of the rat during overt and asymptomatic phases. Virology. 1984;137:347–357. doi: 10.1016/0042-6822(84)90227-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Stewart J N, Mounir S, Talbot P J. Human coronavirus gene expression in the brains of multiple sclerosis patients. Virology. 1992;191:502–505. doi: 10.1016/0042-6822(92)90220-J. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Streicher R, Stoffel W. The organization of the human myelin basic protein gene. Comparison with the mouse gene. Biol Chem Hoppe-Seyler. 1989;370:503–510. doi: 10.1515/bchm3.1989.370.1.503. [DOI] [PubMed] [Google Scholar]
- 53.Sun N, Grzybicki D, Castro F, Murphy S, Perlman S. Activation of astrocytes in the spinal cord of mice chronically infected with a neurotropic coronavirus. Virology. 1995;213:482–493. doi: 10.1006/viro.1995.0021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Talbot P J. Virus-induced autoimmunity in multiple sclerosis: the coronavirus paradigm. Adv Clin Neurosci. 1997;7:215–233. [Google Scholar]
- 55.Talbot P J, Paquette J S, Ciurli C, Antel J P, Ouellet F. Myelin basic protein and human coronavirus 229E cross-reactive T cells in multiple sclerosis. Ann Neurol. 1996;39:233–240. doi: 10.1002/ana.410390213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Tanaka R, Iwasaki Y, Koprowski H. Intracisternal virus-like particles in brain of a multiple sclerosis patient. J Neurol Sci. 1976;28:121–126. doi: 10.1016/0022-510X(76)90053-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Weber T, Major E O. Progressive multifocal leukoencephalopathy: molecular biology, pathogenesis and clinical impact. Intervirology. 1997;40:98–111. doi: 10.1159/000150537. [DOI] [PubMed] [Google Scholar]
- 58.Wege H. Immunopathological aspects of coronavirus infections. Springer Semin Immunopathol. 1995;17:133–148. doi: 10.1007/BF00196162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.White F A, III, Ishaq M, Stoner G L, Frisque R J. JC virus DNA is present in many human brain samples from patients without progressive multifocal leukoencephalopathy. J Virol. 1992;66:5726–5734. doi: 10.1128/jvi.66.10.5726-5734.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]