Abstract
Parkinson's disease (PD) is a common neurodegenerative disorder worldwide. Currently, treatment options can only relieve symptoms but cannot prevent, slow, or halt the neurodegenerative process of PD. Much evidence has suggested that microglia-mediated neuroinflammation is involved in the pathophysiology of PD. As an anti-inflammatory agent, curcumin may exert a neuroprotective effect on PD. However, its mechanism has yet to be demonstrated clearly. Our results indicated that curcumin alleviated rotenone-induced behavioral defects, dopamine neuron loss, and microglial activation. Besides, the NF-κB signaling pathway, the NLRP3 inflammasome, and pro-inflammatory cytokines, including IL-18 and IL-1β, contributed to the microglia-mediated neuroinflammation in PD. Furthermore, Drp1-mediated mitochondrial fission causing mitochondrial dysfunction also had an etiological role in the process. This study suggests that curcumin protects against rotenone-induced PD by inhibiting microglial NLRP3 inflammasome activation and alleviating mitochondrial dysfunction in mice. Thus, curcumin may be a neuroprotective drug with promising prospects in PD.
Keywords: Curcumin, Parkinson's disease, Dopaminergic neuron, Microglia, NLRP3 inflammasome, Mitochondrial dysfunction
1. Introduction
As a common neurodegenerative disorder, Parkinson's disease (PD) has brought a great burden to society [1]. It has been reported that the incidence of PD in the population over the age of 60 is approximately 1% [2]. PD is often characterized by tremor, muscle rigidity, and motor retardation [3]. Severe patients with PD will lose the ability to move to complete daily life activities [4]. The major pathological features of PD are the reduction of dopamine neurons and deposition of alpha-synuclein in the substantia nigra (SN). However, recently, therapeutic approaches targeting neither the replacement of regenerative stem cells nor the clearance of alpha-synuclein have shown clinically long-term benefits [5]. Much evidence indicates that neuroinflammation is implicated in the progression of PD [[6], [7], [8]]. Neuroinflammation has a neurotoxic effect on dopamine neurons and contributes to the onset and progression of PD [7]. As the main innate immune cells of the brain, microglia have been reported to play a critical role in mediating neuroinflammation [9,10]. The activated microglia acquire cytotoxic phenotypes, subsequently releasing pro-inflammatory cytokines that can exacerbate neuron damage during the neurodegenerative processes of PD [11]. Thus, a therapeutic approach targeting microglia-mediated neuroinflammation has been considered a pivotal strategy for treating PD.
As microglia-mediated neuroinflammation is implicated in the progression of PD, interventions focused on anti-inflammation may exert a neuroprotective effect. Curcumin, a member of the ginger family, is derived from the spice turmeric [12]. Much evidence has suggested that curcumin has many pharmacological and biological activities, including anti-inflammatory effect [13,14], antivirulence effect [15], antimicrobial activity [16], antineoplastic activity [17,18], and antioxidant effect [19,20]. As an anti-inflammatory agent, curcumin may exert a neuroprotective effect on PD. Actually, increasing studies have shown that curcumin has pharmaceutical potential against PD [21]. However, its mechanism has yet to be demonstrated clearly. Is the effect of curcumin on PD associated with microglia-mediated neuroinflammation? This study used curcumin to treat PD, and the underlying mechanism was explored.
It is generally known that microglia can recognize danger signals through NOD-like receptor protein 3 (NLRP3) [22]. NLRP3 is the sensor protein of the NLRP3 inflammasome complex. The NLRP3 inflammasome consists of an NLRP3 receptor, an apoptosis-associated speck-like protein containing a CARD (ASC), and a caspase-1 [23]. The activation of nuclear factor kappa B (NF-κB) promotes the transcription of NLRP3 [24]. The NLRP3 inflammasome then activates caspase-1, which can cleave pro-interleukin-18 (IL-18) and pro-interleukin-1beta (IL-1β) to IL-18 and IL-1β, respectively. Briefly, the NLRP3 inflammasome promotes the maturation and release of IL-18 and IL-1β [25], contributing to the progression of neurodegeneration in PD. Furthermore, mitochondrial dysfunction has been implicated as a key mediator in the activation of microglial NLRP3 inflammasome [26]. Research with rotenone-induced rodent models of PD demonstrated that microglial mitochondrial dysfunction could enhance the NLRP3 inflammasome signaling, exacerbating the dopaminergic neurodegeneration [26]. Similarly, another study revealed that dynamin-related protein 1 (Drp1)-dependent mitochondrial fission causing mitochondrial damage induces NF-κB nuclear translocation and NLRP3 inflammasome activation, contributing to rotenone-induced olfactory bulb disturbances in rats [27]. Drp1, a dynamics-related protein, mediates mitochondrial fission, fusion, and mitophagy. Drp1 is located predominantly in the cytosol. Once activated, it forms oligomers and is recruited to punctate spots on the mitochondrial surface. Mitochondrial adaptor binding by Drp1 leads to the assembly of future fission sites and the severing of the mitochondrial membrane under GTP hydrolysis conditions [28]. It is important to note that not only is Drp1-mediated fission required for mitophagy that exerts a neuroprotective effect, but abnormal mitochondrial fission and mitophagy can cause neuronal survival or death, suggesting that Drp1 may play a critical role in PD pathogenesis [29]. Taken together, we speculate that the NF-κB signaling pathway, the NLRP3 inflammasome, pro-inflammatory cytokines, and mitochondrial fission in microglia are involved in the effect of curcumin in PD.
In this study, we aim to explore curcumin's effects on behavioral defects, dopamine neuron loss, microglial activation, NLRP3 inflammasome activation, overexpression of pro-inflammatory cytokines, activation of the NF-κB signaling pathway, and excessive mitochondrial fission in SN induced by rotenone in mice. To achieve these goals, rotenone, a mitochondrial complex I inhibitor, was utilized to construct a classical PD mouse model [[30], [31], [32]]. Curcumin was used to treat PD [[32], [33], [34]], and the underlying mechanism was studied. To study the effect of curcumin treatment on the behavioral alterations induced by rotenone in mice, we performed the open field test and the rotarod test. Tyrosine hydroxylase (TH) is a dopaminergic neuron marker. To evaluate the effect of curcumin treatment on the survival of dopamine neurons, we characterized TH expression by immunohistochemistry. Then, the protein expression levels of Iba-1, NLRP3, ASC, caspase-1, cleaved caspase-1, IL-18, and IL-1β were assessed by immunohistochemistry, Western blot, or ELISA to assess the activation of the NLRP3 inflammasome. Finally, Drp1-dependent mitochondrial fission was tested to evaluate the mitochondrial function by Western blot by assessing the mitochondrial and cytoplasmic Drp1 protein levels.
2. Materials and methods
2.1. Ethics statement
This study was carried out in accordance with the National Institute of Health Guide for the Care and Use of Laboratory Animals. The protocol was approved by the Animal Ethics Committee of the Affiliated Hospital of Qingdao University (No.20200829C576J701118002).
2.2. Animals
This study used male C57BL/6J mice (8 weeks). These mice took food and drink freely and were housed on a 12-h-light/dark cycle. The room temperature is 23 °C.
2.3. Rotenone administration
Rotenone can induce dopaminergic neuron loss, dopamine level reduction, and alpha-synuclein accumulation, resulting in behavioral defects [35]. This study obtained rotenone from Shanghai Jingdu Biotechnology Co., Ltd, China. The mice were administered with rotenone (3 mg/kg/day [36], subcutaneously) for 35 days, while the control mice were administered with sunflower oil.
2.4. Curcumin treatment
Curcumin was obtained from Sigma-Aldrich. Mice were administered with curcumin (50 mg/kg/day) or 0.1% DMSO intraperitoneally for 21 days.
2.5. Rotarod performance test
The training was performed three times on the first day. Moreover, training and test were both performed once on the second day. The latency to fall from the rotarod (the performance time) was measured for 5 min and recorded in seconds [35]. The test was performed with rotarod instrumentation and analyzed by software (Rota Rod Rotamex 5, Columbus Instruments).
2.6. Open field test
Mice were placed in the box for 5 min. In the assessment, each mouse was placed in the middle of the box and allowed to explore freely. To eliminate any possible bias caused by scents left by previous mice, the floor and walls were rinsed with 70% ethyl alcohol after each test [35]. The total distance traveled was recorded. The moving trail was recorded and analyzed by software (EthovisionXT, Noldus).
2.7. Immunohistochemistry
After the behavioral tests, brains were collected, fixed in 4% PFA overnight, and first dehydrated with 20% sucrose and then 30% sucrose. They were then cut into slices using a cryostat (Leica) and rinsed three times in PBS. For quenching endogenous peroxidase, we incubated the sections for 30 min in an aqueous solution of 20% methanol and 0.5% H2O2. Sections were rinsed three times again in PBS. Sections were incubated with blocking solution, primary antibodies, secondary antibodies, avidin-biotin-HRP complex, and diaminobenzidine tetrahydrochloride in turn. The sections were analyzed using a microscope (Leica), and the number of positive cells was quantified. The primary antibodies were: tyrosine hydroxylase (TH, Abcam, ab137869) and Iba-1 (Abcam, ab178847).
2.8. Western blot
SN proteins were extracted by RIPA lysis buffer. The samples were centrifuged at 12,000 rpm for 20 min in the subsequent step. The Bradford method was used to measure the protein concentration of each sample [37]. Samples were separated using 10% SDS- PAGE. The membranes were blocked with 5% nonfat milk for 1 h at room temperature and then incubated with primary antibodies. Membranes were then incubated with secondary antibodies for 2 h at 4 °C and finally visualized with the ECL. The relative protein expression was normalized to β-Actin. The primary antibodies were as follows: Iba-1 (Abcam, ab178847), NLRP3 (CST, # 15101S), ASC (CST, # 67824S), Caspase-1 (CST, # 24232S), Cleaved Caspase-1 (CST, # 89332S), p- NF-κB (CST, # 3039S), NF-κB (CST, # 8242S) and β-Actin (CST, # 8457S).
2.9. ELISA
The SN tissues were homogenized with the cell lysis buffer 2 (R&D Systems). IL-18 and IL-1β levels in SN tissue homogenates were quantified by ELISA (R&D Systems). OD value was measured and analyzed. The IL-1β and IL-18 concentrations were expressed as pg/mg SN protein.
2.10. Isolation of mitochondria
Tissue Mitochondria Isolation Kit (Beyotime, C3606) was used to isolate mitochondria according to the manufacturer's instructions. Precipitates are isolated mitochondria, which can be used to determine mitochondrial protein content. The supernatant can be used for cytoplasmic protein determination. Then, the Western blot was conducted. The primary antibodies were as follows: Drp1 (Abcam, ab184247), COX IV (CST, # 11967S), and β-Actin (CST, # 8457S). The COX IV antibody has proven to be effective for mitochondrial loading control.
2.11. Experimental procedure
To determine the efficacy of curcumin on behavioral defects and microglial activation in SN induced by rotenone in mice, a total of 40 mice were divided into four groups: the vehicle + vehicle group, the vehicle + curcumin group, the rotenone + vehicle group, and the rotenone + curcumin group. N = 10/group. The mice of the rotenone group were injected with rotenone for 35 days. The mice of the curcumin group were injected with curcumin for 21 days. Then, the behavioral tests, including the rotarod test and the open field test, were conducted. After the behavioral tests were finished, the brains were collected to evaluate the efficacy of curcumin on microglial activation in SN of PD mice and to study the underlying mechanism.
2.12. Statistical analysis
This study adopts a completely randomized grouping method. The mice are randomly assigned to each group, and then compare the differences between the mean values of each group for statistical significance to infer the effect of treatment factors. The observations are independent of each other. The observed values can be divided into multiple groups (4 groups). Therefore, the one-way analysis of variance (ANOVA) is selected. The data were analyzed with one-way ANOVA using Graphpad Prism 6.01. Then, Turkey is used for the Post-Hoc test. The data are shown as Mean ± Standard Error (S.E.M.).
3. Results
3.1. Curcumin treatment reversed rotenone-induced behavioral defects in mice
To study the effect of curcumin treatment on the behavioral alterations induced by rotenone in mice, we performed the open field test and the rotarod test. The experimental procedure is shown in Fig. 1A. Rotenone induced significant behavioral defects in PD mice, as shown by decreased total distance in the open field test (Fig. 1B and C) and reduced performance time in the rotarod test (Fig. 1D). As expected, curcumin treatment remarkably normalized rotenone-induced behavioral defects in mice as shown by increased total distance in the open field test (Fig. 1B and C) and performance time in the rotarod test (Fig. 1D). These results indicate that curcumin treatment can reverse rotenone-induced behavioral defects in mice.
Fig. 1.
Curcumin treatment reversed rotenone-induced behavioral defects in mice. A, Experimental procedure. B, Behavior trace of mice in the open field test. C, The total distance of mice in the open field test. D, The rotarod performance time of mice in the rotarod test. N = 10/group. All data are shown as mean ± S.E.M. *P < 0.05, **P < 0.01, ***P < 0.001.
3.2. Curcumin treatment alleviated rotenone-induced reduction of dopamine neurons in SN in mice
To evaluate the effect of curcumin treatment on the survival of dopamine neurons, we characterized TH expression by immunohistochemistry. Rotenone induced obvious loss of dopamine neurons in SN in PD mice (Fig. 2A and B). Curcumin treatment significantly rescued rotenone-induced reduction of dopamine neurons in mice (Fig. 2A and B). This result indicates that curcumin treatment can alleviate rotenone-induced pathological features in SN in mice.
Fig. 2.
Curcumin treatment alleviated the rotenone-induced reduction of dopamine neurons in SN in mice. A, Representative immunohistochemistry staining for TH in the SN. Scale bar is 50 μm. B, Quantitative analysis of the relative number of TH-positive cells in the SN. N = 5/group. All data are shown as mean ± S.E.M. **P < 0.01, ***P < 0.001.
3.3. Curcumin treatment inhibited microglial activation in SN induced by rotenone in mice
As microglia-mediated neuroinflammation is implicated in the progression of PD, we observed the effect of curcumin treatment on microglial activation in PD mice. Immunohistochemistry (Fig. 3A and B) and Western blot (Fig. 3C and D) both revealed that curcumin treatment inhibited rotenone-induced microglial activation in SN in mice. These results indicate that curcumin treatment can dampen microglial activation induced by rotenone in mice.
Fig. 3.
Curcumin treatment inhibited microglial activation in SN induced by rotenone in mice. A, Representative immunohistochemistry staining for Iba-1 in the SN. Scale bar is 50 μm. B, Quantitative analysis of the relative number of Iba-1-positive cells in the SN. C, Representative western blots for Iba-1 in the SN. D, Quantification of the relative intensity of Iba-1 in the SN. N = 5/group. All data are shown as mean ± S.E.M. *P < 0.05, ***P < 0.001.
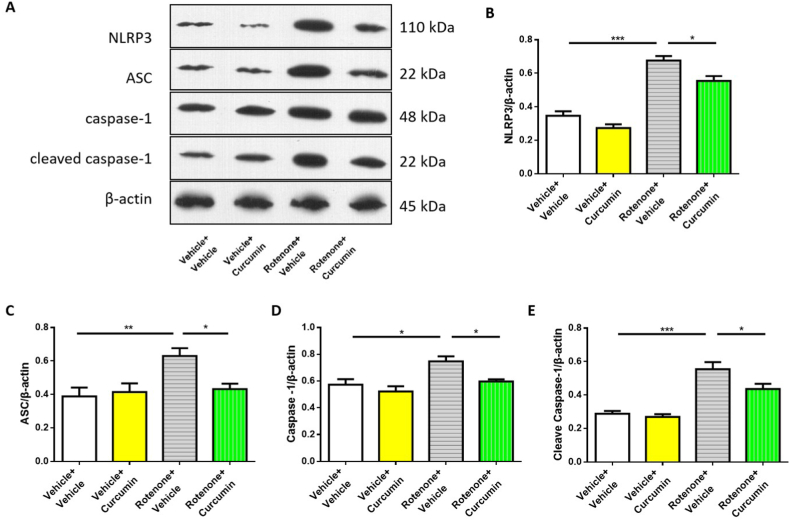
3.4. Curcumin treatment inhibited the activation of NLRP3 inflammasome in SN induced by rotenone in mice
The NLRP3 inflammasome plays an important role in the function of microglia. To study the contribution of the change of the NLRP3 inflammasome to the efficacy of curcumin treatment on PD mice, we observed the protein expression levels of NLRP3, ASC, caspase-1, and cleaved caspase-1 in SN in PD mice (Fig. 4A). In our study, NLRP3 (Fig. 4B), ASC (Fig. 4C), caspase-1 (Fig. 4D), and cleaved caspase-1 (Fig. 4E) displayed higher protein expression levels in PD mice, which were all significantly reversed by curcumin treatment. The results indicate that the change of the NLRP3 inflammasome contributes to the efficacy of curcumin on PD mice.
Fig. 4.
Curcumin treatment inhibited the activation of NLRP3 inflammasome in SN induced by rotenone in mice. A, Representative western blots for NLRP3, ASC, caspase-1, and cleaved caspase-1. B, Quantification of the relative intensities of NLRP3. C, Quantification of the relative intensities of ASC. D, Quantification of the relative intensities of caspase-1. E, Quantification of the relative intensities of cleaved caspase-1. N = 5/group. All data are shown as mean ± S.E.M. *P < 0.05, **P < 0.01, ***P < 0.001.
3.5. Curcumin treatment inhibited the overexpression of cytokines and the activation of the NF-κB signaling pathway in SN induced by rotenone in mice
The NLRP3 inflammasome promotes the maturation and release of IL-18 and IL-1β. To assess the effects of curcumin treatment on the contents of IL-18 and IL-1β in SN in PD mice, we tested them by ELISA. IL-18 (Fig. 5A) and IL-1β (Fig. 5B) both displayed higher protein expression levels in PD mice, which were significantly reversed by curcumin treatment. The activation of the NF-κB signaling pathway promotes the transcription of NLRP3. To further study the signaling pathway in SN in mice, we conducted western blots to determine p–NF–κB and NF-κB levels. As shown in Fig. 5C, the protein expression of p–NF–κB was significantly up-regulated in SN in PD mice, and curcumin treatment remarkably ameliorated this activation (Fig. 5D). These results indicate that the NF-κB signaling pathway may be associated with the effect of curcumin treatment on PD mice.
Fig. 5.
Curcumin treatment inhibited the overexpression of cytokines and the activation of the NF-κB signaling pathway in SN induced by rotenone in mice. A, IL-18 content. B, IL-1β content. C, Representative western blots for p–NF–κB and NF-κB. D, Quantification of the relative intensities of p–NF–κB/NF-κB. N = 5/group. All data are shown as mean ± S.E.M. *P < 0.05, **P < 0.01, ***P < 0.001.
3.6. Curcumin treatment inhibited the excessive mitochondrial fission in SN induced by rotenone in mice
As shown in Fig. 6, Drp1-dependent mitochondrial fission was tested by assessing the mitochondrial and cytoplasmic Drp1 protein levels. We found that the mitochondrial Drp1 protein level was significantly increased (Fig. 6A and B). In contrast, cytosolic Drp1 was remarkably decreased (Fig. 6C and D) in SN of PD mice. The alterations were significantly reversed by curcumin treatment. These results suggest that curcumin treatment can inhibit the excessive mitochondrial fission induced by rotenone in mice. To sum up, the microglial NF-κB -NLRP3 inflammasome signaling pathway and mitochondrial fission may be associated with the effect of curcumin treatment on PD mice.
Fig. 6.
Curcumin treatment inhibited the excessive mitochondrial fission in SN induced by rotenone in mice. A, Representative western blots for mitochondrial Drp1. B, Quantification of the relative intensities of mitochondrial Drp1. C, Representative western blots for cytosolic Drp1. D, Quantification of the relative intensities of cytosolic Drp1. N = 5/group. All data are shown as mean ± S.E.M. *P < 0.05, **P < 0.01, ***P < 0.001.
4. Discussion
PD is a common neurodegenerative disorder all over the world. It is often characterized by tremor, muscle rigidity, and motor retardation. The major pathological features of it are the reduction of dopamine neurons and deposition of alpha-synuclein in SN. Currently, treatment options can only relieve symptoms but cannot prevent, slow, or halt the neurodegenerative process of PD [[38], [39], [40]]. Because of the multifactorial nature of PD, the single-target therapeutic strategy cannot provide significant benefits, so multitarget approaches should be proposed. Thus, new mechanisms and novel targets of disease progression need to be studied. Much evidence has suggested that neuroinflammation is involved in the pathophysiology of PD [41]. The glial activation and overexpression of pro-inflammatory cytokines are common characteristics of the PD brain [42]. Our study used rotenone to construct the classical PD mouse model. This model mimics most clinical features of PD [43,44] and is suitable for exploring neuroprotective drugs because of the slow onset of degeneration [45]. Our results indicated that rotenone induced behavioral defects in mice, as shown by decreased total distance in the open field test and reduced performance time in the rotarod test. In addition, rotenone induced reduction of dopamine neurons in SN in mice. These results demonstrated that rotenone successfully induced behavioral defects and pathological features in PD mice, which aligned with previous studies [[46], [47], [48]]. As the main innate immune cells of the brain, microglia have been reported to play a critical role in mediating neuroinflammation. Many studies have shown that microglial activation participates in the pathophysiology of PD [[49], [50], [51]]. However, its mechanism needs to be better interpreted. The NLRP3 inflammasome plays an important role in the function of microglia. Besides, the activation of NF-κB promotes the transcription of NLRP3. The NLRP3 inflammasome promotes the maturation and release of pro-inflammatory cytokines, including IL-18 and IL-1β. It has also been suggested that mitochondrial dysfunction triggers the NLRP3 inflammasome activation. Specifically, Drp1-dependent mitochondrial fission induces NF-κB nuclear translocation and NLRP3 inflammasome activation in PD rats. Our results indicated that rotenone induced microglial activation in SN in mice. The mitochondrial fission, the NF-κB signaling pathway, the NLRP3 inflammasome, and pro-inflammatory cytokines were all abnormal, contributing to the microglia-mediated neuroinflammation in PD.
As an anti-inflammatory drug, curcumin can cross the blood-brain barrier and has a neuroprotective effect in PD [52]. In our study, curcumin treatment significantly normalized rotenone-induced behavioral defects in mice, as shown by increased total distance in the open field test and performance time in the rotarod test. Besides, curcumin treatment significantly rescued rotenone-induced reduction of dopamine neurons in mice. These results confirmed that curcumin has neuroprotective effects on PD. Evidence has shown curcumin's neuroprotective effects in PD, including inhibiting inflammation and oxidative stress, reducing monoamine oxidase B, and inhibiting the aggregation of alpha-synuclein. However, whether NLRP3 inflammasome and mitochondrial fission play a role in the effect of curcumin in PD is not known. In this study, curcumin treatment inhibited rotenone-induced microglial activation in SN in mice. Furthermore, curcumin treatment inhibited the activation of the NLRP3 inflammasome, the overexpression of cytokines, the activation of the NF-κB signaling pathway, and the excessive mitochondrial fission in SN induced by rotenone in mice. These results indicated that microglial NLRP3 inflammasome and mitochondrial fission mediate the effect of curcumin in PD. Our study adds to the evidence that curcumin has neuroprotective effects in PD. The novelty of this study is that we found that curcumin rescued rotenone-induced dopaminergic neuron loss by inhibiting microglial NLRP3 inflammasome activation and alleviating mitochondrial dysfunction.
It has been documented that NLRP3 inflammasome-mediated neuroinflammation participates in the progression of neurodegeneration in PD, and mitochondrial impairment augments the process [53]. Drp1-dependent mitochondrial fission causing mitochondrial damage induces NF-κB nuclear translocation and NLRP3 inflammasome activation. As an anti-inflammatory agent, curcumin may exert a neuroprotective effect on PD. However, its mechanism has yet to be demonstrated clearly. Hence, this study aims to clarify whether NLRP3 inflammasome-mediated neuroinflammation and mitochondrial fission are involved in the effect of curcumin in PD. Our results indicated that curcumin alleviated rotenone-induced behavioral defects, dopamine neuron loss, and microglial activation. Besides, the NF-κB signaling pathway, the NLRP3 inflammasome, and pro-inflammatory cytokines, including IL-18 and IL-1β, contributed to the microglia-mediated neuroinflammation in PD. Furthermore, Drp1-mediated mitochondrial fission causing mitochondrial dysfunction also had an etiological role in the process. This study suggests that curcumin protects against rotenone-induced PD by inhibiting microglial NLRP3 inflammasome activation and alleviating mitochondrial dysfunction in mice. Thus, curcumin may be a neuroprotective drug with promising prospects in PD. The limitation of this study is that curcumin cannot penetrate the blood-brain barrier (BBB) effectively, thus limiting its therapeutic effects [54]. The approaches to facilitate curcumin permeation across the BBB may resolve drug delivery-related problems. Further studies need to develop central nervous system penetrant curcumin to treat PD.
Author contribution statement
Long Xu: Conceived and designed the experiments; Performed the experiments.
Li-Ping Hao: Performed the experiments.
Jing Yu: Performed the experiments; Analyzed and interpreted the data.
Shao-Yuan Cheng: Analyzed and interpreted the data.
Fan Li; Shou-Mei Ding: Contributed reagents, materials, analysis tools or data.
Rui Zhang: Conceived and designed the experiments; Wrote the paper.
Data availability statement
Data will be made available on request.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Footnotes
Supplementary data related to this article can be found at https://doi.org/10.1016/j.heliyon.2023.e16195.
Appendix A. Supplementary data
The following is the supplementary data related to this article:
References
- 1.Eusebi P., et al. Incidence and prevalence of Parkinson's disease in the Italian region of Umbria: a population-based study using healthcare administrative databases. Neurol. Sci. 2019;40(8):1709–1712. doi: 10.1007/s10072-019-03872-w. [DOI] [PubMed] [Google Scholar]
- 2.Hirsch L., et al. The incidence of Parkinson's disease: a systematic review and meta-analysis. Neuroepidemiology. 2016;46(4):292–300. doi: 10.1159/000445751. [DOI] [PubMed] [Google Scholar]
- 3.Reich S.G., Savitt J.M. Parkinson's disease. Med. Clin. 2019;103(2):337–350. doi: 10.1016/j.mcna.2018.10.014. [DOI] [PubMed] [Google Scholar]
- 4.Bouça‐Machado R., et al. Measurement instruments to assess functional mobility in Parkinson's disease: a systematic review. Movement disorders clinical practice. 2020;7(2):129–139. doi: 10.1002/mdc3.12874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Schwab A.D., et al. Immunotherapy for Parkinson's disease. Neurobiol. Dis. 2020;137 doi: 10.1016/j.nbd.2020.104760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kaur K., et al. Neuroinflammation-A major cause for striatal dopaminergic degeneration in Parkinson's disease. J. Neurol. Sci. 2017;381:308–314. doi: 10.1016/j.jns.2017.08.3251. [DOI] [PubMed] [Google Scholar]
- 7.Troncoso-Escudero P., et al. Outside in: unraveling the role of neuroinflammation in the progression of Parkinson's disease. Front. Neurol. 2018;9:860. doi: 10.3389/fneur.2018.00860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lee Y., et al. Significant roles of neuroinflammation in Parkinson's disease: therapeutic targets for PD prevention. Arch Pharm. Res. (Seoul) 2019;42(5):416–425. doi: 10.1007/s12272-019-01133-0. [DOI] [PubMed] [Google Scholar]
- 9.Lazdon E., Stolero N., Frenkel D. Microglia and Parkinson's disease: footprints to pathology. J. Neural. Transm. 2020;127(2):149–158. doi: 10.1007/s00702-020-02154-6. [DOI] [PubMed] [Google Scholar]
- 10.Machado V., et al. Microglia-mediated neuroinflammation and neurotrophic factor-induced protection in the MPTP mouse model of Parkinson's disease-lessons from transgenic mice. Int. J. Mol. Sci. 2016;17(2):151. doi: 10.3390/ijms17020151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kelly R., et al. Microglial phenotypes and their relationship to the cannabinoid system: therapeutic implications for Parkinson's disease. Molecules. 2020;25(3):453. doi: 10.3390/molecules25030453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Seo H.J., et al. Curcumin as a putative antidepressant. Expert Rev. Neurother. 2015;15(3):1–12. doi: 10.1586/14737175.2015.1008457. [DOI] [PubMed] [Google Scholar]
- 13.Sandur S.K., et al. Curcumin, demethoxycurcumin, bisdemethoxycurcumin, tetrahydrocurcumin and turmerones differentially regulate anti-inflammatory and anti-proliferative responses through a ROS-independent mechanism. Carcinogenesis. 2007;28(8):1765–1773. doi: 10.1093/carcin/bgm123. [DOI] [PubMed] [Google Scholar]
- 14.Jurenka J.S. Anti-inflammatory properties of curcumin, a major constituent of Curcuma longa: a review of preclinical and clinical research. Alternative Medicine Review A Journal of Clinical Therapeutic. 2009;14(2):141. [PubMed] [Google Scholar]
- 15.Darmani H., Smadi E.A., Bataineh S.M. Blue light emitting diodes enhance the antivirulence effects of Curcumin against Helicobacter pylori. J. Med. Microbiol. 2020;69(4):617–624. doi: 10.1099/jmm.0.001168. [DOI] [PubMed] [Google Scholar]
- 16.Esmaeili E., et al. The biomedical potential of cellulose acetate/polyurethane nanofibrous mats containing reduced graphene oxide/silver nanocomposites and curcumin: antimicrobial performance and cutaneous wound healing. Int. J. Biol. Macromol. 2020;152:418–427. doi: 10.1016/j.ijbiomac.2020.02.295. [DOI] [PubMed] [Google Scholar]
- 17.Ma C., et al. Curcumin has anti-proliferative and pro-apoptotic effects on tongue cancer in vitro: a study with bioinformatics analysis and in vitro experiments. Drug Des. Dev. Ther. 2020;14:509. doi: 10.2147/DDDT.S237830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Weng W., Goel A. Seminars in Cancer Biology. Elsevier; 2020. Curcumin and colorectal cancer: an update and current perspective on this natural medicine. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Dogaru G., et al. Effect of liposomal curcumin on acetaminophen hepatotoxicity by down-regulation of oxidative stress and matrix metalloproteinases. In vivo. 2020;34(2):569–582. doi: 10.21873/invivo.11809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Celebioglu A., Uyar T. Fast-dissolving antioxidant curcumin/cyclodextrin inclusion complex electrospun nanofibrous webs. Food Chem. 2020;317 doi: 10.1016/j.foodchem.2020.126397. [DOI] [PubMed] [Google Scholar]
- 21.Ji H.-F., Shen L. The multiple pharmaceutical potential of curcumin in Parkinson's disease. CNS Neurol. Disord. - Drug Targets. 2014;13(2):369–373. doi: 10.2174/18715273113129990077. [DOI] [PubMed] [Google Scholar]
- 22.Ye Y., et al. Meisoindigo protects against focal cerebral ischemia-reperfusion injury by inhibiting NLRP3 inflammasome activation and regulating microglia/macrophage polarization via TLR4/NF-κB signaling pathway. Front. Cell. Neurosci. 2019;13:553. doi: 10.3389/fncel.2019.00553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.He Y., Hara H., Núñez G. Mechanism and regulation of NLRP3 inflammasome activation. Trends Biochem. Sci. 2016;41(12):1012–1021. doi: 10.1016/j.tibs.2016.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Choi A.J., Ryter S.W. Inflammasomes: molecular regulation and implications for metabolic and cognitive diseases. Mol. Cell. 2014;37(6):441. doi: 10.14348/molcells.2014.0104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hughes M.M., O'Neill L.A. Metabolic regulation of nlrp 3. Immunol. Rev. 2018;281(1):88–98. doi: 10.1111/imr.12608. [DOI] [PubMed] [Google Scholar]
- 26.Sarkar S., et al. Mitochondrial impairment in microglia amplifies NLRP3 inflammasome proinflammatory signaling in cell culture and animal models of Parkinson's disease. npj Parkinson's Disease. 2017;3(1):1–15. doi: 10.1038/s41531-017-0032-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zhang X., et al. Drp1, a potential therapeutic target for Parkinson's disease, is involved in olfactory bulb pathological alteration in the Rotenone-induced rat model. Toxicol. Lett. 2020;325:1–13. doi: 10.1016/j.toxlet.2020.02.009. [DOI] [PubMed] [Google Scholar]
- 28.Hu D., Qi X. Dynamin Superfamily GTPases. Springer; 2020. Measuring Drp1 activity in mitochondrial fission in vivo; pp. 189–195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Feng S.-T., et al. Dynamin-related protein 1: a protein critical for mitochondrial fission, mitophagy, and neuronal death in Parkinson's disease. Pharmacol. Res. 2020;151 doi: 10.1016/j.phrs.2019.104553. [DOI] [PubMed] [Google Scholar]
- 30.Parkhe A., et al. Protective effect of alpha mangostin on rotenone induced toxicity in rat model of Parkinson's disease. Neurosci. Lett. 2020;716 doi: 10.1016/j.neulet.2019.134652. [DOI] [PubMed] [Google Scholar]
- 31.Wang T., et al. Neuroprotective effects of Danshensu on rotenone-induced Parkinson's disease models in vitro and in vivo. BMC complementary medicine and therapies. 2020;20(1):1–10. doi: 10.1186/s12906-019-2738-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Khatri D.K., Juvekar A.R. Neuroprotective effect of curcumin as evinced by abrogation of rotenone-induced motor deficits, oxidative and mitochondrial dysfunctions in mouse model of Parkinson's disease. Pharmacol. Biochem. Behav. 2016;150:39–47. doi: 10.1016/j.pbb.2016.09.002. [DOI] [PubMed] [Google Scholar]
- 33.Chiu S., et al. Liposomal-formulated curcumin [Lipocurc™] targeting HDAC (histone deacetylase) prevents apoptosis and improves motor deficits in park 7 (DJ-1)-knockout rat model of Parkinson's disease: implications for epigenetics-based nanotechnology-driven drug platform. J. Compl. Integr. Med. 2013;10(1):75–88. doi: 10.1515/jcim-2013-0020. [DOI] [PubMed] [Google Scholar]
- 34.Sharma N., Nehru B. Curcumin affords neuroprotection and inhibits α-synuclein aggregation in lipopolysaccharide-induced Parkinson's disease model. Inflammopharmacology. 2018;26(2):349–360. doi: 10.1007/s10787-017-0402-8. [DOI] [PubMed] [Google Scholar]
- 35.Sayed A.S., et al. Xanthotoxin modulates oxidative stress, inflammation, and MAPK signaling in a rotenone-induced Parkinson's disease model. Life Sci. 2022;310 doi: 10.1016/j.lfs.2022.121129. [DOI] [PubMed] [Google Scholar]
- 36.Sherer T.B., et al. Subcutaneous rotenone exposure causes highly selective dopaminergic degeneration and alpha-synuclein aggregation. Exp. Neurol. 2003;179(1):9–16. doi: 10.1006/exnr.2002.8072. [DOI] [PubMed] [Google Scholar]
- 37.Assaf N., et al. Neuroprotective effect of PPAR alpha and gamma agonists in a mouse model of amyloidogenesis through modulation of the Wnt/beta catenin pathway via targeting alpha- and beta-secretases. Prog. Neuro-Psychopharmacol. Biol. Psychiatry. 2020;97 doi: 10.1016/j.pnpbp.2019.109793. [DOI] [PubMed] [Google Scholar]
- 38.Cheong S.L., et al. The current status of pharmacotherapy for the treatment of Parkinson's disease: transition from single-target to multitarget therapy. Drug Discov. Today. 2019;24(9):1769–1783. doi: 10.1016/j.drudis.2019.05.003. [DOI] [PubMed] [Google Scholar]
- 39.Mao Q., et al. Recent advances in dopaminergic strategies for the treatment of Parkinson's disease. Acta Pharmacol. Sin. 2020;41(4):471–482. doi: 10.1038/s41401-020-0365-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Zahoor I., Shafi A., Haq E. Exon Publications; 2018. Pharmacological Treatment of Parkinson's Disease; pp. 129–144. [PubMed] [Google Scholar]
- 41.Bassani T.B., Vital M.A., Rauh L.K. Neuroinflammation in the pathophysiology of Parkinson's disease and therapeutic evidence of anti-inflammatory drugs. Arquivos de neuro-psiquiatria. 2015;73:616–623. doi: 10.1590/0004-282X20150057. [DOI] [PubMed] [Google Scholar]
- 42.Wang Q., Liu Y., Zhou J. Neuroinflammation in Parkinson's disease and its potential as therapeutic target. Transl. Neurodegener. 2015;4(1):1–9. doi: 10.1186/s40035-015-0042-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Xiong N., et al. Mitochondrial complex I inhibitor rotenone-induced toxicity and its potential mechanisms in Parkinson's disease models. Crit. Rev. Toxicol. 2012;42(7):613–632. doi: 10.3109/10408444.2012.680431. [DOI] [PubMed] [Google Scholar]
- 44.Johnson M.E., Bobrovskaya L. An update on the rotenone models of Parkinson's disease: their ability to reproduce the features of clinical disease and model gene–environment interactions. Neurotoxicology. 2015;46:101–116. doi: 10.1016/j.neuro.2014.12.002. [DOI] [PubMed] [Google Scholar]
- 45.Schmidt W., Alam M. Parkinson’s Disease and Related Disorders; 2006. Controversies on new animal models of Parkinson's disease pro and con: the rotenone model of Parkinson's disease (PD) pp. 272–276. [PubMed] [Google Scholar]
- 46.Tao H., Liu Y., Hou Y. miRNA-384-5p regulates the progression of Parkinson's disease by targeting SIRT1 in mice and SH-SY5Y cell. Int. J. Mol. Med. 2020;45(2):441–450. doi: 10.3892/ijmm.2019.4426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Yang X., et al. Longitudinal analysis of fecal microbiome and pathologic processes in a rotenone induced mice model of Parkinson's disease. Front. Aging Neurosci. 2018;9:441. doi: 10.3389/fnagi.2017.00441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Zhang Y., et al. Involvement of Akt/mTOR in the neurotoxicity of rotenone-induced Parkinson's disease models. Int. J. Environ. Res. Publ. Health. 2019;16(20):3811. doi: 10.3390/ijerph16203811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Machado M.M.F., et al. PPAR-γ agonist pioglitazone reduces microglial proliferation and NF-κB activation in the substantia nigra in the 6-hydroxydopamine model of Parkinson's disease. Pharmacol. Rep. 2019;71(4):556–564. doi: 10.1016/j.pharep.2018.11.005. [DOI] [PubMed] [Google Scholar]
- 50.Kang Y., et al. Noninvasive PK11195‐PET image analysis techniques can detect abnormal cerebral microglial activation in Parkinson's disease. J. Neuroimaging. 2018;28(5):496–505. doi: 10.1111/jon.12519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Kim B., et al. 2-(5-(4-Chlorophenyl)-1-(2, 4-dichlorophenyl)-4-methyl-1H-pyrazol-3-yl)-N-(2-hydroxyethyl)-2-oxoacetamide (CDMPO) has anti-inflammatory properties in microglial cells and prevents neuronal and behavioral deficits in MPTP mouse model of Parkinson's disease. Neuropharmacology. 2020;166 doi: 10.1016/j.neuropharm.2019.107928. [DOI] [PubMed] [Google Scholar]
- 52.B Mythri R., Srinivas Bharath M.M. Curcumin: a potential neuroprotective agent in Parkinson's disease. Curr. Pharmaceut. Des. 2012;18(1):91–99. doi: 10.2174/138161212798918995. [DOI] [PubMed] [Google Scholar]
- 53.Han Q.Q., Le W. NLRP3 inflammasome-mediated neuroinflammation and related mitochondrial impairment in Parkinson's disease. Neurosci. Bull. 2023 doi: 10.1007/s12264-023-01023-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Askarizadeh A., et al. Neuroprotection by curcumin: a review on brain delivery strategies. Int. J. Pharm. 2020;585 doi: 10.1016/j.ijpharm.2020.119476. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data will be made available on request.