Abstract
Objectives
The aim of this study was to compare the effect of steroid-impregnated spacers to that of conventional management after endoscopic sinus surgery (ESS) in patients with chronic rhinosinusitis (CRS).
Methods
Six databases were searched from inception until November 2022. Sixteen studies were found that compared the improvement of chronic sinusitis-related symptoms and postoperative outcomes between a steroid-impregnated spacer group and a control group (non-steroid-impregnated spacers). The Cochrane risk of bias tool (for randomized controlled studies) and the Newcastle-Ottawa Scale (for non-randomized controlled studies) were used to assess the quality of the works included.
Results
Regarding the endoscopic findings, the degree of mucosal edema, ethmoid inflammation, crust formation at 2–3 months postoperatively, nasal discharge, polyposis, and scarring/synechia were significantly lower in the steroid-impregnated spacer group. The steroid-impregnated spacer group also showed significantly lower Lund–Kennedy scores and perioperative sinus endoscopy scores than the control group at 2–3 weeks postoperatively. Furthermore, the steroid-impregnated spacer group had lower rates of adhesions, middle turbinate lateralization, polypoid changes, the need for oral steroid use, the need for postoperative therapeutic interventions, and lysis of adhesions than controls. However, no significant between-group differences were found in short-term (2–3 weeks postoperatively) endoscopic findings regarding nasal discharge, postoperative crusting, polyposis, or scarring/synechia.
Conclusion
Steroid-impregnated nasal packing reduced the rates of postoperative intervention and recurrent polyposis and inflammation in CRS patients undergoing ESS.
Keywords: Sinusitis, Operative Surgical Procedures, Nose, Steroids, Stents
INTRODUCTION
Chronic rhinosinusitis (CRS) is characterized by persistent inflammation of the sinus mucosa, which results in nasal congestion, postnasal drip, facial pressure, and decreased olfactory function [1,2]. Furthermore, it leads to chronic pain, depression, and social dysfunction, which impair quality of life [3]. Due to the high prevalence of CRS, the number of CRS cases that are intractable to medical treatment is also increasing. Accordingly, endoscopic sinus surgery (ESS) is becoming increasingly important [4]. Although ESS is effective in resolving persistent CRS, local sinus inflammation may sometimes persist even after surgery, and surgical site adhesions and strictures may occur during the recovery process of the surgical site. This can slow recovery, potentially leading to the need for revision surgery [5,6]. To overcome this problem, steroid-impregnated spacers have been devised as a way to reduce local inflammation around the surgical area and prevent stenosis during postoperative recovery [7–23]. In this study, we reviewed the steroid-impregnated spacers reported to date and evaluated their effects on treatment outcomes using validated scales. Additionally, the rates of postoperative adverse outcomes such as adhesions, middle turbinate lateralization, the need for postoperative therapeutic interventions, scarring/synechia, and lysis of adhesions were compared with the control group. The goal of this meta-analysis of the literature on steroid-impregnated spacers was to comprehensively assess the efficacy of steroid-impregnated spacers for improving outcomes following ESS surgery. In addition, a subgroup analysis was performed to compare the effects of absorbable nasal dressing and drug-eluting sinus stents.
MATERIALS AND METHODS
Population, Intervention, Comparison, Outcomes and Study and selection criteria
The Population, Intervention, Comparison, Outcomes and Study (PICOS) of the study were as follows: (1) population: CRS patients who underwent functional ESS (primary or revision procedure); (2) intervention: steroid-impregnated packing, spacer, and stent; (3) comparison: packing, spacer, and stent without steroids; (4) outcomes: treatment and postoperative adverse outcomes; and (5) study design: no restrictions. Review articles, case reports, and studies with missing diagnostic data were excluded. Our institution does not require institutional review board approval for a systematic review and meta-analysis based exclusively on the published literature.
Search strategy
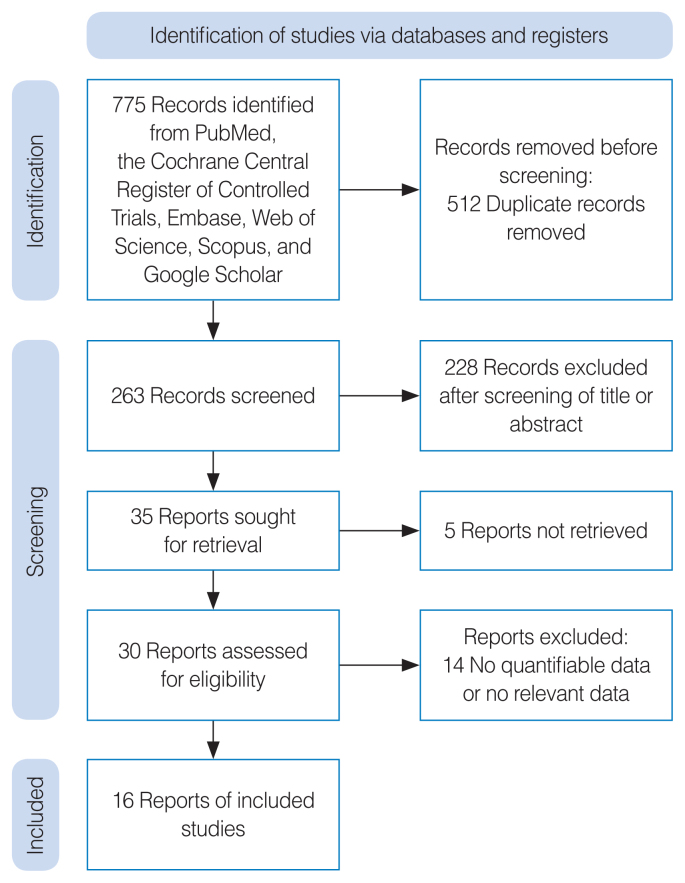
The following databases were searched from inception to November 2022: PubMed, Scopus, Embase, the Web of Science, Google Scholar, and the Cochrane database. Search terms and queries are listed in Supplementary Table 1. Two independent researchers (DHK and MAB) reviewed and screened the titles and abstracts of all potentially eligible studies and excluded those un related to our topic. If the abstract alone was not sufficient for determining whether a study was suitable for inclusion, the full text was checked. If the opinions of the two researchers differed, study eligibility was decided via discussion with a third reviewer (SWK). A flowchart of the study selection process is presented in Fig. 1. The study protocol is registered on Open Science Framework (https://osf.io/fhc42/).
Fig. 1.
Diagram of the study selection process.
Data extraction and risk of bias assessment
We extracted the following data from eligible studies: the number of patients, scale used for assessing endoscopic findings, incidence of postoperative adverse events (adhesions, middle turbinate lateralization, polypoid change, and the need for postoperative therapeutic intervention), and P-values for comparisons between treatment (steroid-impregnated spacers) and control (conventional management) groups. The studies were organized using a standardized format [24–25]. Outcome measures were post-treatment endoscopic findings (the Lund-Kennedy [LK] score and perioperative sinus endoscopy [POSE] score, postoperative crusting, nasal discharge, mucosal edema, ethmoid inflammation (evaluate scores with 100-mm visual analog scale by reviewers through endoscopic images; 0: no inflammation to 100: significant presence of severe inflammation), the need for oral steroid, polypoid change, and polyposis and the rate of postoperative adverse events for ESS. The definitions of terms were taken from a previous study [26]: “mucosal edema” refers to edema in areas where the bony contour is difficult to distinguish, “polypoid change” to outpouching filling the ethmoid cavity, and “polyposis” to polyp formation and growth beyond the middle meatus. The outcomes were compared between the treatment and control groups [7–23]. Quality assessment of the included randomized controlled studies was conducted using the Cochrane risk of bias tool. The Newcastle-Ottawa Scale was used to assess non-randomized controlled studies.
Statistical analysis
Meta-analyses were conducted using R software ver. 4.2.2 (R Foundation for Statistical Computing). Continuous variables were analyzed according to the standardized mean difference; odds ratios (ORs) were generated in all other cases. Heterogeneity was assessed by the I2 statistic (0, no heterogeneity across studies; 100, maximum heterogeneity). All results are reported with 95% confidence intervals (CIs), and all P-values were two-tailed. When significant heterogeneity among outcomes was found (I2 ≥50), the random-effects model of DerSimonian-Laird was used. This model assumes that treatment effects may differ among studies, and that the data are normally distributed. For outcomes showing no significant heterogeneity (I2<50), a fixed-effects model using the inverse variance approach was employed, in which it is assumed that all studies are based on the same population. Sensitivity analyses were also conducted to assess the effect of each study on the overall results of the meta-analysis.
RESULTS
In total, 16 of the 1,274 identified articles were included. The study characteristics are listed in Supplementary Tables 2, and the bias assessment results are presented in Supplementary Tables 3. Because the number of included studies was not sufficient (<10) to generate an adequate funnel plot or perform advanced regression analyses for all outcomes, publication bias was not evaluated.
Treatment and control group scores
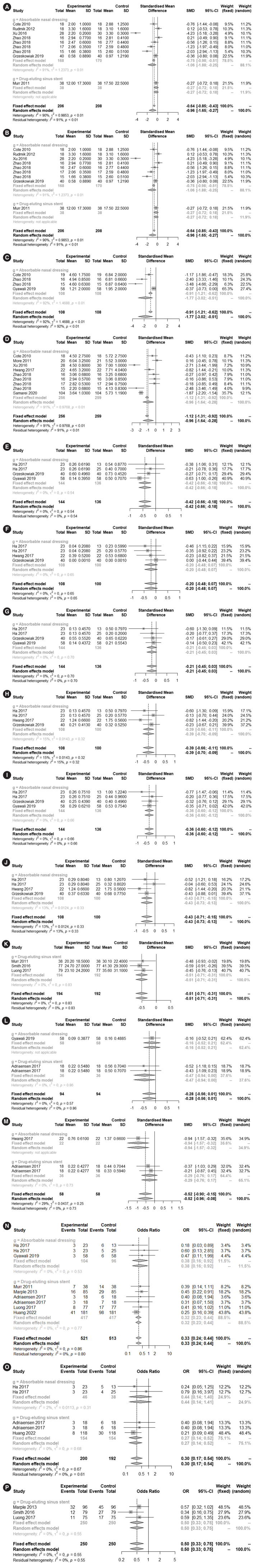
The LK and POSE scores were significantly lower in the steroid-impregnated spacer group at 2–3 weeks (−1.1495; 95% CI, −1.8207 to 0.4783; I2=87.8%; P=0.0008 and −1.7656; 95% CI, −3.0236 to 0.5075; I2=91.8%; P=0.0060, respectively) and 2–3 months postoperatively (−0.9597; 95% CI, −1.6491 to 0.2702; I2=90.2%; P=0.0064 and −0.9602; 95% CI, −1.6442 to 0.2762; I2=91.0%; P=0.0059, respectively) than in the control group (Fig. 2A–D).
Fig. 2.
Severity of postoperative adverse outcomes based on endoscopic examinations: comparison between the treatment and control groups. The Lund-Kennedy score at 2–3 weeks (A), and 2–3 months (B), perioperative sinus endoscopy score at 2–3 weeks (C) and 2–3 months (D). Crusting at 2–3 weeks (E), and 2–3 months (F), nasal discharge at 2–3 weeks (G) and 2–3 months (H), edema at 2–3 weeks (I). And 2–3 months (J), ethmoid inflammation at 1 month (K), polyposis at 2–3 weeks (L) and 2–3 months (M). Polypoid changes at 1 month (N) and 2–3 months (O), and the need for oral steroid use (P). SD, standard deviation; SMD, standardized mean difference; CI, confidence interval.
The degree of postoperative crusting at 2–3 weeks postoperatively (−0.4188; 95% CI, −0.6575 to 0.1802; I2=0.0%; P=0.0006), nasal discharge at 2–3 months postoperatively (−0.3851; 95% CI, −0.6628 to 0.1074; I2=14.5%; P=0.0066), mucosal edema at 2–3 weeks (−0.3609; 95% CI, −0.5987 to 0.1231; I2=0.0%; P=0.0029) and 2–3 months postoperatively (−0.4290; 95% CI, −0.7073 to 0.1507; I2=12.8%; P=0.0025), ethmoid inflammation at 1 month postoperatively (−0.5080; 95% CI, −0.7109 to 0.3051; I2=0.0%; P<0.0001), polypoid changes at 1 month (0.3263; 95% CI, 0.2415; 0.4408; I2=0.0%; P<0.0001) and 2–3 months (0.3021; 95% CI, 0.1697–0.5378; I2=0.0%; P< 0.0001), polyposis at 2–3 months postoperatively (−0.5226; 95% CI, −0.8958 to 0.1493; I2=28.6%; P=0.0061), and the need for oral steroid use (0.4975; 95% CI, 0.3312–0.7471; I2=0.0%; P=0.0008) were significantly lower in the steroid-impregnated spacer group than in the control group. However, there were no significant between-group differences in crusting at 2–3 months postoperatively (−0.2041; 95% CI, −0.4791 to 0.0709; I2=0.0%; P=0.1458), nasal discharge at 2–3 weeks postoperatively (−0.2106; 95% CI, −0.4471 to 0.0258; I2=0.0%; P=0.0809), or polyposis at 2–3 weeks postoperatively (−0.2750; 95% CI, −0.5629 to 0.0128; I2=0.0%; P=0.0611) (Fig. 2E–P).
Postoperative adverse outcomes: comparison between the treatment and control groups
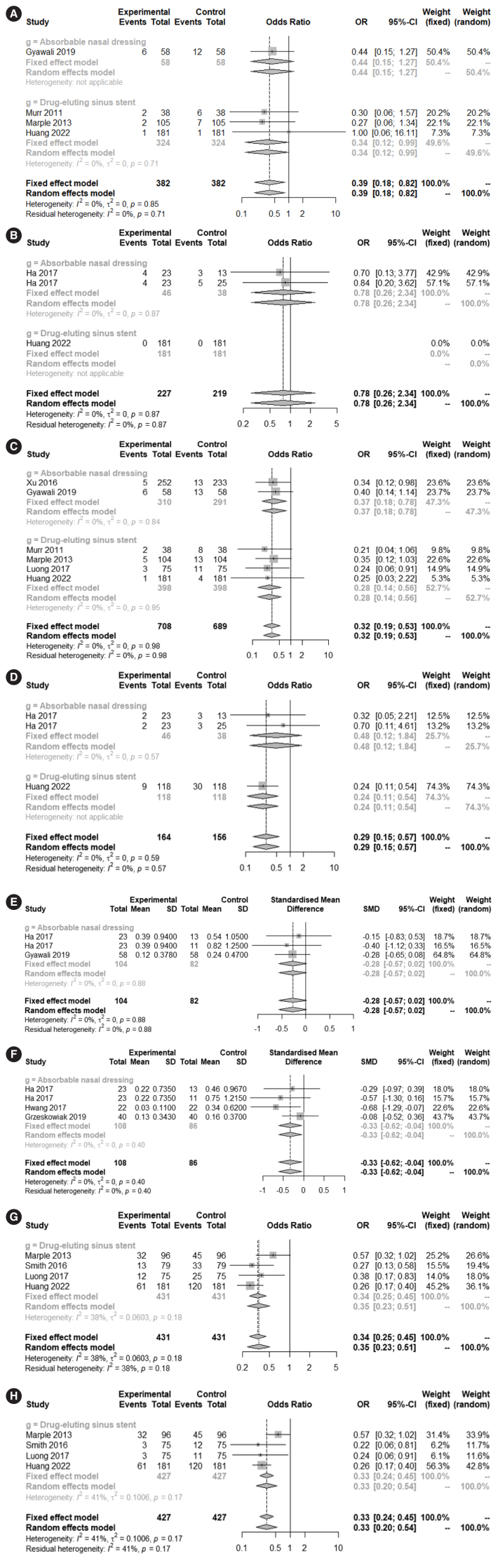
The steroid-impregnated spacer group showed significantly lower rates of middle turbinate lateralization (0.3887; 95% CI, 0.1835–0.8233; I2=0.0%; P=0.0136), non-medical or -surgical postoperative therapeutic interventions (0.3358; 95% CI, 0.2505–0.4501; I2=38.2%; P<0.0001), and lysis of adhesions (0.3259; 95% CI, 0.2350–0.4519; I2=41.0%; P=0.0001) than the control group. Although the rates of adhesion at 2 weeks (0.7788; 95% CI, 0.2589–2.3422; I2=0.0%; P=0.6563), and scarring/synechia at 2–3 weeks (−0.2752; 95% CI, −0.5697 to 0.0193; I2=0.0%; P=0.0670) did not differ between the groups postoperatively, there was a difference in adhesions at 1 month (0.3179; 95% CI, 0.1911–0.5290; I2=0.0%; P<0.0001) and both adhesions (0.2882; 95% CI, 0.1451–0.5724; I2=0.0%; P=0.0004) and scarring/synechia (−0.3326; 95% CI, −0.6225 to 0.0426; I2=0.0%; P=0.0246) at 2–3 months postoperatively (Fig. 3).
Fig. 3.
Postoperative adverse outcomes: comparison between the treatment and control groups. Middle turbinate lateralization at 1 month (A), adhesions at 2 weeks (B), 1 month (C), and 2–3 months (D). Scarring/synechia at 2–3 weeks (E) and 2–3 months (F), non-medical or non-surgical postoperative therapeutic interventions (G), and lysis of adhesions (H). OR, odds ratio; CI, confidence interval; SD, standard deviation; SMD, standardized mean difference.
Subgroup analysis comparing the incidence of postoperative outcomes between the absorbable nasal dressing and drug-eluting sinus stent groups
A comparative analysis was possible between absorbable nasal dressing and drug-eluting sinus stent groups for the adhesion and polypoid change items, which are important for the prognosis after ESS. The subgroup analysis results showed no significant difference between the two groups regarding adhesions (2–3 weeks) (P=0.5807), polypoid change (2–3 weeks) (P=0.7174), and polypoid changes (2–3 months) (P=0.4618) (Supplementary Table 4).
Sensitivity analyses
Sensitivity analyses were performed by repeating the meta-analysis several times while omitting one study on each occasion. The results were consistent with those reported above.
DISCUSSION
We performed a meta-analysis of the efficacy of steroid-impregnated spacers for CRS patients undergoing ESS. Steroid-impregnated spacers reduced the LK and POSE scores and the incidence of postoperative complications, especially after 2–3 months. The steroid-impregnated spacers in this study can be divided into absorbable nasal dressings and drug-eluting sinus stents. The steroids used included triamcinolone, budesonide, betamethasone, mometasone furoate, and fluticasone propionate. In most studies, steroid-impregnated spacers were associated with better postoperative outcomes. Important postoperative parameters such as adhesion, synechia, and polypoid changes showed no significant differences between the absorbable nasal dressing and drug-eluting sinus stent groups. In this study, the number of included studies, the number of patients included, and analysis outcome items were analyzed in more than twice as many studies as in the previously reported literature [27–29]. Therefore, it was possible to obtain improved analytical power and reliability for the analyzed items. In addition, the analysis of the effects in absorbable steroid-soaked nasal spacer and steroid-eluting sinus stent groups, which are known to show greater efficacy than the control group in individual studies, demonstrated that there was no significant difference in the effect between these two treatment groups.
Evidence is accumulating that steroid-impregnated spacers have a direct anti-inflammatory effect on inflamed sinus mucosa, and are also effective in cases of ESS-induced tissue disruption [30,31]. Furthermore, since it is difficult to precisely reach the desired site of application for topical corticosteroids, steroid-impregnated spacers could potentially be more effective than topical preparations [16]. Steroid-impregnated spacers are thought to enhance healing after surgery due to their anti-inflammatory role resulting from the steroid effects, and to prevent adhesion and synechia through the effect of the spacer [15,17]. In the sinus mucosal healing process, the first stage (1–10 days) is dominated by blood crusting without significant changes in the underlying residual mucosa, and the second stage (up to 30 days) is dominated by edematous edema of the residual mucosa [32]. Obstructive edema, which is common in this second phase, responds well to topical steroids [32]. This might be the basis for the significant results observed in follow-up observations for more than 1 month, although several items on surgical outcomes or postoperative complication may not have shown statistically significant differences compared to the control group at 2–3 weeks postoperatively.
Although absorbable nasal packing may have a shorter effect duration than drug-eluting sinus stents, the overall effect is nonetheless similar; however, additional research is needed because the number of comparative studies is small. In terms of price, steroids such as triamcinolone, dexamethasone, and betamethasone cost <$10 USD per dose, whereas Nasopore, an absorbable nasal packing used in many studies, costs $130 USD (for two 8-cm pieces). PROPEL sinus implants, which are used as drug-eluting sinus stents, cost $1,390 USD (for two implants). It is necessary to consider cost-effectiveness when selecting a steroid-impregnated spacer.
This meta-analysis had several limitations. First, variables such as patients’ baseline characteristics, the spacer types, and intervention period could have affected the results. Furthermore, preoperative medication, postoperative patient management, and patient compliance may have varied among the studies, and controlling for these variables is beyond the scope of a meta-analysis. More clinical trials using similar treatment regimens would be needed to overcome this issue. Second, various steroid drugs and spacers were used. To address this, we conducted a subgroup analysis of absorbable nasal dressings and drug-eluting sinus stents. Third, the final outcomes in most studies were evaluated within 3 months; however, a longer follow-up (e.g., 6 months) may be required in some patients. Therefore, well-designed large-scale studies with long-term follow-up are needed. Steroid-impregnated spacers will be useful for CRS patients, as they improve the surgical outcomes of ESS patients and reduce the incidence of postoperative complications.
HIGHLIGHTS.
▪ Steroid-impregnated spacers can effectively deliver a corticosteroid to the target site.
▪ Steroid-impregnated spacers improve the surgical outcomes of endoscopic sinus surgery patients, such as the degree of mucosal edema, ethmoid inflammation, crust formation, nasal discharge, polypoid changes, polyposis, Lund-Kennedy score, and the need for oral steroid usage.
▪ Steroid-impregnated spacers reduce postoperative adhesions, middle turbinate lateralization, scarring/synechia, and the need for postoperative therapeutic interventions.
ACKNOWLEDGMENTS
This work was supported by the National Research Foundation of Korea (NRF; 2022R1F1A1066232, 2019M3A9H2032424, 2019M3E5D5064110), and by the Ministry of Trade, Industry & Energy (MOTIE, Korea) (20012378). The sponsors had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Footnotes
Sung Won Kim and Do Hyun Kim are editorial board members of the journal but were not involved in the peer reviewer selection, evaluation, or decision process of this article. No other potential conflicts of interest relevant to this article were reported.
AUTHOR CONTRIBUTIONS
Conceptualization: DHK, SHH. Methodology: SWK, MAB. Formal analysis: SHH, DHK. Data curation: SWK, MAB. Visualization: SWK, MAB. Project administration: DHK, SHH. Funding acquisition: DHK, SWK. Writing–original draft: DHK, SHH. Writing–review & editing: all authors.
SUPPLEMENTARY MATERIALS
Supplementary materials can be found online at https://doi.org/10.21053/ceo.2022.01718.
Literature database search terms and queries
Results of quality assessment of randomized controlled studies using the Cochrane risk of bias tool
Results of the quality assessment of non-randomized controlled studies using the Newcastle-Ottawa scale
Results of a subgroup analysis comparing the incidence of postoperative outcomes between the absorbable nasal dressing and drug-eluting sinus stent groups
REFERENCES
- 1.Fokkens WJ, Lund VJ, Hopkins C, Hellings PW, Kern R, Reitsma S, et al. European position paper on rhinosinusitis and nasal polyps 2020. Rhinology. 2020 Feb;58(Suppl 29):1–464. doi: 10.4193/Rhin20.600. [DOI] [PubMed] [Google Scholar]
- 2.Park DY, Choi JH, Kim DK, Jung YG, Mun SJ, Min HJ, et al. Clinical practice guideline: nasal irrigation for chronic rhinosinusitis in adults. Clin Exp Otorhinolaryngol. 2022 Feb;15(1):5–23. doi: 10.21053/ceo.2021.00654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Orlandi RR, Kingdom TT, Smith TL, Bleier B, DeConde A, Luong AU, et al. International consensus statement on allergy and rhinology: rhinosinusitis 2021. Int Forum Allergy Rhinol. 2021 Mar;11(3):213–739. doi: 10.1002/alr.22741. [DOI] [PubMed] [Google Scholar]
- 4.Svider PF, Sekhsaria V, Cohen DS, Eloy JA, Setzen M, Folbe AJ. Geographic and temporal trends in frontal sinus surgery. Int Forum Allergy Rhinol. 2015 Jan;5(1):46–54. doi: 10.1002/alr.21425. [DOI] [PubMed] [Google Scholar]
- 5.DeConde AS, Mace JC, Levy JM, Rudmik L, Alt JA, Smith TL. Prevalence of polyp recurrence after endoscopic sinus surgery for chronic rhinosinusitis with nasal polyposis. Laryngoscope. 2017 Mar;127(3):550–5. doi: 10.1002/lary.26391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Stein NR, Jafari A, DeConde AS. Revision rates and time to revision following endoscopic sinus surgery: a large database analysis. Laryngoscope. 2018 Jan;128(1):31–6. doi: 10.1002/lary.26741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Cote DW, Wright ED. Triamcinolone-impregnated nasal dressing following endoscopic sinus surgery: a randomized, double-blind, placebo-controlled study. Laryngoscope. 2010 Jun;120(6):1269–73. doi: 10.1002/lary.20905. [DOI] [PubMed] [Google Scholar]
- 8.Adriaensen GF, Lim KH, Fokkens WJ. Safety and efficacy of a bioabsorbable fluticasone propionate-eluting sinus dressing in postoperative management of endoscopic sinus surgery: a randomized clinical trial. Int Forum Allergy Rhinol. 2017 Aug;7(8):813–20. doi: 10.1002/alr.21963. [DOI] [PubMed] [Google Scholar]
- 9.Grzeskowiak B, Wierzchowska M, Walorek R, Seredyka-Burduk M, Wawrzyniak K, Burduk PK. Steroid vs. antibiotic impregnated absorbable nasal packing for wound healing after endoscopic sinus surgery: a randomized, double blind, placebo-controlled study. Braz J Otorhinolaryngol. 2019 Jul–Aug;85(4):473–80. doi: 10.1016/j.bjorl.2018.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gyawali BR, Pradhan B, Thapa N. Comparison of outcomes of triamcinolone versus normal saline soaked polyvinyl alcohol pack following bilateral endoscopic sinus surgery. Rhinology. 2019 Aug;57(4):287–92. doi: 10.4193/Rhin19.004. [DOI] [PubMed] [Google Scholar]
- 11.Ha T, Valentine R, Moratti S, Hanton L, Robinson S, Wormald PJ. The efficacy of a novel budesonide chitosan gel on wound healing following endoscopic sinus surgery. Int Forum Allergy Rhinol. 2018 Mar;8(3):435–43. doi: 10.1002/alr.22057. [DOI] [PubMed] [Google Scholar]
- 12.Huang Z, Zhou B, Wang D, Zang H, Zhang H, Wang H, et al. Comparison of bioabsorbable steroid-eluting sinus stents versus nasopore after endoscopic sinus surgery: a multicenter, randomized, controlled, single-blinded clinical trial. Ear Nose Throat J. 2022 May;101(4):260–7. doi: 10.1177/0145561320947632. [DOI] [PubMed] [Google Scholar]
- 13.Hwang CS, Al Sharhan SS, Kim BR, Kim SI, Kim JW, Cho HJ, et al. Randomized controlled trial of steroid-soaked absorbable calcium alginate nasal packing following endoscopic sinus surgery. Laryngoscope. 2018 Feb;128(2):311–6. doi: 10.1002/lary.26871. [DOI] [PubMed] [Google Scholar]
- 14.Luong A, Ow RA, Singh A, Weiss RL, Han JK, Gerencer R, et al. Safety and effectiveness of a bioabsorbable steroid-releasing implant for the paranasal sinus ostia: a randomized clinical trial. JAMA Otolaryngol Head Neck Surg. 2018 Jan;144(1):28–35. doi: 10.1001/jamaoto.2017.1859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Marple BF, Smith TL, Han JK, Gould AR, Jampel HD, Stambaugh JW, et al. Advance II: a prospective, randomized study assessing safety and efficacy of bioabsorbable steroid-releasing sinus implants. Otolaryngol Head Neck Surg. 2012 Jun;146(6):1004–11. doi: 10.1177/0194599811435968. [DOI] [PubMed] [Google Scholar]
- 16.More Y, Willen S, Catalano P. Management of early nasal polyposis using a steroid-impregnated nasal dressing. Int Forum Allergy Rhinol. 2011 Sep–Oct;1(5):401–4. doi: 10.1002/alr.20067. [DOI] [PubMed] [Google Scholar]
- 17.Murr AH, Smith TL, Hwang PH, Bhattacharyya N, Lanier BJ, Stambaugh JW, et al. Safety and efficacy of a novel bioabsorbable, steroid-eluting sinus stent. Int Forum Allergy Rhinol. 2011 Jan–Feb;1(1):23–32. doi: 10.1002/alr.20020. [DOI] [PubMed] [Google Scholar]
- 18.Rudmik L, Mace J, Mechor B. Effect of a dexamethasone Sinu-Foam-TM middle meatal spacer on endoscopic sinus surgery outcomes: a randomized, double-blind, placebo-controlled trial. Int Forum Allergy Rhinol. 2012 May–Jun;2(3):248–51. doi: 10.1002/alr.21011. [DOI] [PubMed] [Google Scholar]
- 19.Sabarinath V, Harish MR, Divakaran S. Triamcinolone impregnated nasal pack in endoscopic sinus surgery: our experience. Indian J Otolaryngol Head Neck Surg. 2017 Mar;69(1):88–92. doi: 10.1007/s12070-016-1041-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Samarei R, Rasouli J, Mehdikhani F. Efficacy of triamcinolone acetonide-impregnated Gelfoam nasal pack in management of chronic sinusitis with nasal polyps following endoscopic sinus surgery: a perfectly matched, placebo-controlled trial study. Eur Arch Otorhinolaryngol. 2022 Jun;279(6):2915–24. doi: 10.1007/s00405-021-07071-2. [DOI] [PubMed] [Google Scholar]
- 21.Smith TL, Singh A, Luong A, Ow RA, Shotts SD, Sautter NB, et al. Randomized controlled trial of a bioabsorbable steroid-releasing implant in the frontal sinus opening. Laryngoscope. 2016 Dec;126(12):2659–64. doi: 10.1002/lary.26140. [DOI] [PubMed] [Google Scholar]
- 22.Xu J, Park SJ, Park HS, Han R, Rha KS, Kim YM. Effects of triamcinolone-impregnated nasal dressing on subjective and objective outcomes following endoscopic sinus surgery. Eur Arch Otorhinolaryngol. 2016 Dec;273(12):4351–7. doi: 10.1007/s00405-016-4185-0. [DOI] [PubMed] [Google Scholar]
- 23.Zhao KQ, Yu YQ, Yu HM. Effects of mometasone furoate-impregnated biodegradable nasal dressing on endoscopic appearance in healing process following endoscopic sinus surgery: a randomized, double-blind, placebo-controlled study. Int Forum Allergy Rhinol. 2018 Nov;8(11):1233–41. doi: 10.1002/alr.22213. [DOI] [PubMed] [Google Scholar]
- 24.Kim DH, Kim SW, Basurrah MA, Hwang SH. Clinical and laboratory features of various criteria of eosinophilic chronic rhinosinusitis: a systematic review and meta-analysis. Clin Exp Otorhinolaryngol. 2022 Aug;15(3):230–46. doi: 10.21053/ceo.2022.00052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hwang SH, Kim JS, Choi BY, Kim JK, Kim BG. Practical review of olfactory training and COVID-19. J Rhinol. 2022;29(3):127–33. [Google Scholar]
- 26.Wright ED, Agrawal S. Impact of perioperative systemic steroids on surgical outcomes in patients with chronic rhinosinusitis with polyposis: evaluation with the novel Perioperative Sinus Endoscopy (POSE) scoring system. Laryngoscope. 2007 Nov;117(11 Pt 2 Suppl 115):1–28. doi: 10.1097/MLG.0b013e31814842f8. [DOI] [PubMed] [Google Scholar]
- 27.Li W, Lu H, Wang H, Sun X, Wang D. Efficacy and safety of steroid-impregnated implants following sinus surgery: a meta-analysis. Laryngoscope. 2020 Dec;130(12):2754–9. doi: 10.1002/lary.28388. [DOI] [PubMed] [Google Scholar]
- 28.Stefan MM, Rabie NA, Sobhy TS, Maarouf AM. Effect of steroid-releasing sinus implants after endoscopic sinus surgery (ESS) on postoperative outcomes: a meta-analytical study. Egypt J Otolaryngol. 2019 Jul;35:250–5. [Google Scholar]
- 29.Zhao X, Grewal A, Briel M, Lee JM. A systematic review of nonabsorbable, absorbable, and steroid-impregnated spacers following endoscopic sinus surgery. Int Forum Allergy Rhinol. 2013 Nov;3(11):896–904. doi: 10.1002/alr.21201. [DOI] [PubMed] [Google Scholar]
- 30.Goshtasbi K, Abouzari M, Abiri A, Yasaka T, Sahyouni R, Bitner B, et al. Efficacy of steroid-eluting stents in management of chronic rhinosinusitis after endoscopic sinus surgery: updated meta-analysis. Int Forum Allergy Rhinol. 2019 Dec;9(12):1443–50. doi: 10.1002/alr.22443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Zhang M, Ryan PJ, Shashinder S. Efficacy of absorbable steroid-impregnated nasal packing in functional endoscopic sinus surgery for chronic rhinosinusitis: a systematic review. Laryngoscope. 2021 Aug;131(8):1704–14. doi: 10.1002/lary.29350. [DOI] [PubMed] [Google Scholar]
- 32.Hosemann W, Wigand ME, Gode U, Langer F, Dunker I. Normal wound healing of the paranasal sinuses: clinical and experimental investigations. Eur Arch Otorhinolaryngol. 1991;248(7):390–4. doi: 10.1007/BF01463560. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Literature database search terms and queries
Results of quality assessment of randomized controlled studies using the Cochrane risk of bias tool
Results of the quality assessment of non-randomized controlled studies using the Newcastle-Ottawa scale
Results of a subgroup analysis comparing the incidence of postoperative outcomes between the absorbable nasal dressing and drug-eluting sinus stent groups