Abstract
Methamphetamine use causes spikes in blood pressure. Chronic hypertension is a major risk factor for cerebral small vessel disease (cSVD). The aim of this study is to investigate whether methamphetamine use increases the risk of cSVD. Consecutive patients with acute ischemic stroke at our medical center were screened for methamphetamine use and evidence of cSVD on MRI of the brain. Methamphetamine use was identified by self-reported history and/or positive urine drug screen. Propensity score matching was used to select non-methamphetamine controls. Sensitivity analysis was performed to assess the effect of methamphetamine use on cSVD. Among 1369 eligible patients, 61 (4.5%) were identified to have a history of methamphetamine use and/or positive urine drug screen. Compared with the non-methamphetamine group (n = 1306), the patients with methamphetamine abuse were significantly younger (54.5 ± 9.7 vs. 70.5 ± 12.4, p < 0.001), male (78.7% vs. 54.0%, p < 0.001) and White (78.7% vs. 50.4%, p < 0.001). Sensitivity analysis showed that methamphetamine use was associated with increased white matter hyperintensities, lacunes, and total burden of cSVD. The association was independent of age, sex, concomitant cocaine use, hyperlipidemia, acute hypertension, and stroke severity. Our findings suggest that methamphetamine use increases the risk of cSVD in young patients with acute ischemic stroke.
Subject terms: Neuroscience, Neurology
Introduction
Methamphetamine abuse has emerged as a risk factor for both hemorrhagic and ischemic stroke in recent years1–10. Although population-based study and forensic analysis of fatal strokes showed significant predominance of hemorrhagic stroke in methamphetamine users3,6,7, methamphetamine abuse has also been increasingly reported to be associated with acute ischemic stroke (AIS)1,2,4–6.
The mechanisms by which methamphetamine causes stroke are still unknown. Case studies and forensic analysis showed atherosclerotic stenoses, arterial dissection, and berry aneurysms in patients with methamphetamine-associated stroke2,4,6,7. Methamphetamine abuse was also shown to produce a dose-dependent elevation of blood pressure and chronic hypertension11–13.
Chronic hypertension is a major risk factor for cerebral small vessel disease (cSVD) and stroke13–16. cSVD refers to a group of pathological processes that affect the small perforating vessels and capillaries in the brain16,17. Radiographically, it is characterized by deep white matter hyperdensities (WMHs), lacunar infarcts, microbleeds, and enlarged perivascular spaces (PVS) on magnetic resonance imaging (MRI) of the brain17. cSVD is a common cause of stroke, cognitive impairment, and vascular dementia15–17.
The aim of this study is to investigate whether methamphetamine abuse increases the risk of cSVD in patients with acute ischemic stroke.
Methods
This retrospective study was approved by the University of California Irvine Institutional Review Board (IRB) and the Ethics Committee. Informed consents were waived due to retrospective study design and minimal harm to the patients. All methods in the study were performed in accordance with the relevant guidelines and regulations.
Study population
Consecutive AIS patients admitted at the University of California Irvine Medical Center from January 1, 2013 to December 30, 2018 were included. The patient list was generated from the prospectively maintained American Heart Association (AHA)'s Get-With-The-Guideline stroke data registry at our medical center. The registry uses a web-based patient management tool to collect clinical data on consecutively admitted patients, to provide decision support, and to enable real-time online reporting18. Patients with TIA, stroke mimics, subacute stroke, inpatient stroke, and primary brain hemorrhage were excluded. Stroke transfers from outside facilities were also excluded.
All patients underwent standard diagnostic evaluation and treatment per AHA guidelines19. Based on the history of methamphetamine abuse and/or urine drug screen (UDS), patients were divided into Meth and Non-Meth group. The UDS was performed using EMIT II Plus Amphetamines Assay (Beckman Coulter, Inc) with a sensitivity and specificity of 94.3% and 93.3%, respectively20.
Study parameters
MRI images of the brain were reviewed by an experienced neurologist (Zhu Z) to assess cSVD using modified Fazekas scale13–15,21. Both deep and periventricular WMHs were rated from 0 to 3 on fluid-attenuated inversion recovery (FLAIR)- and T2-weighted sequences15. Lacunes were defined as small (< 15 mm) subcortical infarcts13,21. Cerebral microbleeds (CMBs) were rated on susceptibility-weighted imaging (SWI) based on their numbers (< 5, 5–10, or > 10). Enlarged perivascular spaces (PVS) were defined as small (< 3 mm) punctate hyperintensities on FLAIR or T2-weighted images and rated from 0 to 3: 0 (absent), 1 (< 10), 2 (10–25), and 3 (> 25).
Total burdens of cSVD were rated from 0 to 413. One point was added for each of the following findings: confluent deep or periventricular WMHs (Fazekas grade 2 and 3), ≥ 1 lacune, ≥ 1 CMBs, and > 10 enlarged PVS in the basal ganglia on at least one side of the brain.
Propensity score matching
Propensity score matching with a 1:1 ratio was performed to select patients from the Non-Meth group as control. The propensity score was estimated using a logistic regression model based on age, sex, hypertension, diabetes mellitus, hyperlipidemia, obesity, and initial NIHSS score as described (1:1 match, nearest neighbor approach)22.
Statistical analysis
Continuous variables were described by mean ± standard deviation (SD) or median with interquartile range (IQR) based on the results of normality testing. Categorical variables were expressed by counts with percentages. Baseline characteristics and outcome at discharge were compared between Meth and Non-Meth groups by Mann Whitney test for continuous variables and chi-square test for categorical variables. Sensitivity analysis was performed to investigate the effect of methamphetamine abuse and other variables on the development of cSVD. All statistical analyses were performed using SPSS software (version 23.0). A 2-tailed value of p < 0.05 was considered statistically significant. All statistical analyses were reviewed and verified by a biostatistician (Qian T).
Results
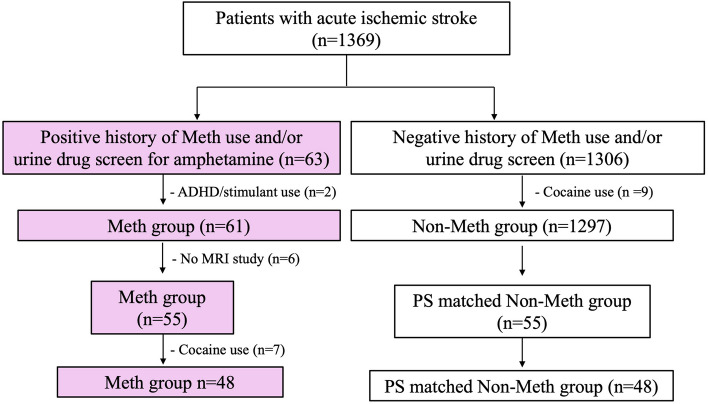
From the 1,369 patients with AIS, 63 were found to have a history of methamphetamine abuse or positive UDS. 2 patients were identified to have a positive UDS due to stimulant use for Attention Deficit Hyperactivity Disorders (ADHD) and were excluded from the study. Of the 61 patients in the Meth group, 6 had no MRI study of the brain and were excluded for the investigation of cSVD. In the remaining 55 patients, 7 had concomitant cocaine use and were excluded during the final statistical analysis. In patients with a negative history of meth use or UDS, 9 had cocaine use and was excluded. A 1:1 propensity score-matched control group was selected from the Non-Meth group for sensitivity analysis. The study flowchart is shown in Fig. 1.
Figure 1.
Study flowchart.
The characteristics of the Meth and Non-Meth groups are summarized in Table 1. Compared with Non-Meth group, patients in the Meth group were significantly younger (54.5 ± 9.7 vs. 70.5 ± 12.4, p < 0.001), more likely male (78.7% vs. 54.0%, p < 0.001) and White (78.7% vs. 50.4%, p < 0.001). There appeared to be proportionally less Asians with methamphetamine-associated stroke (3.3% vs. 24.1%, p < 0.001).
Table 1.
Demographics and clinical characteristics in the Meth group versus Non-meth group.
| Variables | Meth group | Non-meth group | p |
|---|---|---|---|
| n | 61 | 1297 | – |
| Age | 54.5 ± 9.7 | 70.5 ± 12.4 | < 0.001 |
| Male (%) | 48 (78.7%) | 709 (54.0%) | < 0.001 |
| Race | |||
| White | 48 (78.7%) | 654 (50.4%) | < 0.001 |
| Hispanic | 6 (9.8%) | 231 (17.8%) | 0.197 |
| Black | 3 (4.9%) | 30 (2.3%) | 0.213 |
| Asian | 2 (3.3%) | 312 (24.1%) | < 0.001 |
| Other | 2 (3.3%) | 70 (5.4%) | 0.471 |
| Hypertension | 35 (57.4%) | 935 (71.4%) | 0.013 |
| Diabetes | 20 (32.8%) | 447 (36.4%) | 0.788 |
| Hyperlipidemia | 16 (26.2%) | 523 (40.0%) | 0.028 |
| Obesity | 10 (16.4%) | 178 (13.6%) | 0.555 |
| Smoking | 35 (57%) | 390 (30.1%) | < 0.001 |
| Cocaine use | 7 (11.5%) | 9 (0.7%) | < 0.001 |
| Statin use | 10 (16%) | 525 (40.5%) | < 0.001 |
| Antithrombotic use | 14 (23%) | 452 (34.8%) | 0.056 |
| Initial NIHSS score | 4 (2, 10) | 4 (2, 14) | 0.154 |
| Baseline LDL cholesterol | 104 ± 38 | 99 ± 39 | 0.213 |
| Mechanism of stroke | |||
| Large artery disease | 15 (25%) | 312 (24.1%) | 0.924 |
| Cardioembolism | 21 (34%) | 592 (45.6%) | 0.085 |
| Small vessel disease | 19 (31%) | 361 (27.8%) | 0.573 |
| Other causes | 2 (3%) | 52 (4%) | 0.775 |
NIHSS National Institutes of Health Stroke Scale, LDL low density lipoprotein cholesterol.
Significant are in value [bold].
Non-Meth group had a statistically significant higher rate of hypertension, hyperlipidemia, and statin use than in the Meth group. There was no statistically significant difference between the two groups in the history of diabetes, obesity, antithrombotic use, initial NIHSS score, baseline LDL cholesterol levels, or mechanisms of stroke per TOAST classification (Table 1). However, the patients in the Meth group had a higher rate of smoking (57% vs. 30.1%, p < 0.001) or cocaine use (11.5% vs. 0.7%, p < 0.001) than those in the Non-Meth group.
Sensitivity analysis
Sensitivity analysis was performed to investigate the effect of methamphetamine use as independent variable. Compared with age- and sex-matched control group, there were proportionally more White and less Asian patients in the Meth group (Table 2). There was no significant difference in the history of hypertension, diabetes, hyperlipidemia, obesity, or initial NIHSS score between the Meth group and propensity score matched control group.
Table 2.
Comparison of cerebral SVD in Meth versus propensity score-matched control group after removing the patients with stimulant use for ADHD.
| Variables | Meth group | PS-matched control group | p |
|---|---|---|---|
| N | 55 | 55 | |
| Age | 55 ± 10 | 55 ± 10 | 0.978 |
| Male (%) | 45 (81.8) | 38 (69.1) | 0.183 |
| Race | 0.006 | ||
| White | 43 (78.2) | 25 (45.5) | |
| Hispanic | 6 (10.9) | 4 (7.3) | |
| Black | 3 (5.5) | 11 (20) | |
| Asian | 2 (3.6) | 10 (18.2) | |
| Other | 1 (1.8) | 5 (9.1) | |
| Hypertension | 36 (65.5) | 31 (56.4) | 0.329 |
| Diabetes | 19 (34.5) | 20 (36.4) | 0.329 |
| Hyperlipidemia | 15 (27.3) | 20 (36.4) | 0.306 |
| Obesity | 10 (18.2) | 13 (23.6) | 0.482 |
| NIHSS | 5 (6) | 4 (6) | 0.245 |
| Cerebral SVD | |||
| WMHs | 3 (3) | 2 (3) | < 0.001 |
| Lacunes | 2 (4) | 1 (2) | 0.032 |
| Microbleeds | 2 (4) | 0 (4) | 0.369 |
| Enlarged perivascular space | 1 (1) | 1 (1) | 0.346 |
| Total burden | 2 (1) | 2 (1) | 0.006 |
| Baseline SBP | 159 ± 31 | 170 ± 39 | 0.083 |
| Baseline DBP | 97 ± 20 | 95 ± 20 | 0.573 |
| SBP at discharge | 130 ± 17 | 133 ± 20 | 0.411 |
| DBP at discharge | 77 ± 13 | 76 ± 13 | 0.703 |
| Reduction in SBP | 27 ± 27 | 40 ± 30 | 0.038 |
| Reduction in DBP | 19 ± 21 | 20 ± 20 | 0.915 |
| Numbers of Antihypertensives | 3 (3) | 2 (3) | 0.790 |
The Meth group in this analysis excluded patients with history of stimulant use for ADHD. Propensity score-matched patients from the Non-Meth group was used as control.
Data are expressed as n (%), mean ± SD, or median (interquartile range, IQR).
ADHD attention deficit hyperactivity disorder, DBP diastolic blood pressure, NIHSS National Institutes of Health Stroke Scale, PS propensity score, SBP systolic blood pressure, WMHs white matter hyperdensities.
Significant are in value [bold].
The representative MRI images of cSVD were shown in Fig. 2. The Meth group were found to have more WMHs, lacunes, and total burdens of cSVD than propensity score-matched control group (Table 2).
Figure 2.
Representative images of cerebral small vessel disease (cSVD). One point is added for each of the following findings to estimate cSVD score: severe white matter hyperintensities (periventricular Fazekas grade 2 or 3); ≥ 1 lacunar infarcts; ≥ 1 cerebral microbleed; and > 10 perivascular spaces visible in the basal ganglia on at least one side of the brain.
There were no significant differences in systolic or diastolic blood pressure (BP) at admission or hospital discharge, DBP reduction during hospitalization, and numbers of antihypertensives required for BP control between the Meth and propensity score-matched control group. The statistically more significant reduction in SBP in the control group was likely due to higher baseline SBP than in the Meth group (Table 2). These results suggested that the increased burdens of cSVD in the Meth group are independent of age, sex, comorbidities, acute hypertension, and stroke severity as measured by initial NIHSS scores.
We performed an additional sensitivity analysis after excluding the 7 patients with concomitant cocaine use in the Meth groups. As showed in Table 3, the results were consistent with what were shown in Table 2. Methamphetamine use remains an independent predictor for cSVD.
Table 3.
Comparison of cerebral SVD in the Meth group versus PS-matched control group after removing the patients with stimulant use for ADHD and cocaine use.
| Variables | Meth | PS-matched Control | p |
|---|---|---|---|
| N | 48 | 48 | |
| Age | 54 ± 10 | 54 ± 10 | 0.984 |
| Male (%) | 34 (70.8) | 39 (81.3) | 0.232 |
| Race | 0.019 | ||
| White | 37 (77.1) | 22 (45.8) | |
| Hispanic | 3 (6.3) | 3 (6.3) | |
| Black | 5 (10.4) | 10 (20.8) | |
| Asian | 2 (4.2) | 8 (16.7) | |
| Other | 1 (2.1) | 5 (10.4) | |
| Hypertension | 31 (64.6) | 25 (52.1) | 0.214 |
| Diabetes | 18 (37.5) | 20 (41.7) | 0.676 |
| Hyperlipidemia | 14 (29.2) | 17 (35.4) | 0.513 |
| Obesity | 9 (18.8) | 11 (22.9) | 0.615 |
| NIHSS | 5 (6) | 4 (7) | 0.116 |
| Cerebral SVD | |||
| WMHs | 3 (3) | 2 (3) | < 0.001 |
| Lacunes | 2 (4) | 1 (2) | 0.016 |
| Microbleeds | 1 (4) | 0 (3) | 0.445 |
| Enlarged perivascular space | 1 (1) | 1 (1) | 0.206 |
| Total burden | 2 (2) | 2 (1) | 0.009 |
| Baseline SBP | 159 ± 31 | 172 ± 35 | 0.070 |
| Baseline DBP | 90 ± 20 | 96 ± 20 | 0.691 |
| SBP at discharge | 132 ± 17 | 133 ± 20 | 0.838 |
| DBP at discharge | 78 ± 13 | 76 ± 13 | 0.481 |
| Reduction in SBP | 27 ± 25 | 40 ± 30 | 0.029 |
| Reduction in DBP | 19 ± 20 | 20 ± 19 | 0.756 |
| Numbers of Antihypertensives | 1 (2) | 2 (3) | 0.985 |
The Meth group in this analysis excluded the patients with history of ADHD/stimulant use or cocaine use. Propensity score-matched patients from the Non-Meth group was used as control.
Data are expressed as n (%), mean ± SD, or median (interquartile range, IQR).
DBP diastolic blood pressure, NIHSS National Institutes of Health Stroke Scale, PS propensity score, SBP systolic blood pressure, WMHs white matter hyperdensities.
Significant are in value [bold].
Discussion
This single center data demonstrates that methamphetamine abuse is seen in 4.5% of the patients with acute ischemic stroke at our medical center. The patients with methamphetamine use are significantly younger (54.5 ± 9.7 vs. 70.5 ± 12.4, p < 0.001) and more likely male (78.7% vs. 54.0%, p < 0.001). In addition, sensitivity analysis with propensity score-matched controls showed that methamphetamine abuse is associated with increased burdens of cSVD, independent of age, sex, cocaine use, comorbidities, acute hypertension, and stroke severity.
Previous studies showed that elevated blood pressure levels are associated with each of the MRI markers of cSVD14,15. Effective treatment of hypertension may reduce the rates of cSVD and stroke13,15. In this retrospective study, there was no significant difference in blood pressures at admission or hospital discharge, blood pressure reduction during hospitalization, and the numbers of antihypertensives required to control hypertension between the Meth or Meth + group and propensity score-matched Non-Meth controls. It is possible that chronic hypertension from methamphetamine abuse plays an important role in the development of cSVD13.
Advanced age and male sex were reported to be major risk factors for cSVD16,23–25. In a recent study, cerebral SVD was seen in 18.9% of age group 70s as compared to 3% in age group 40s23. In a Chinese population-based study, advanced age was shown to be independently associated with the prevalence of cSVD24. In our study, we demonstrated that methamphetamine abuse increases the risk of cSVD in young stroke patients.
In a cohort study of homeless and unstably housed women (n = 30) from San Francisco community, 86% patients had a history of cocaine use and 54% patients had WMHs25. In our cohort, more patients in the Meth group had a history of cocaine abuse than the Non-Meth group (11.5% vs. 0.7%). After excluding patients with cocaine use in both Meth and Non-Meth groups, methamphetamine use remains an independent predictor for cSVD.
Although the Non-Meth group had statistically significant higher rate of hyperlipidemia (40.0% vs. 26.2%, p = 0.028) and statin use (40.5% vs. 15%, p < 0.001) than the Meth group, there was no statistically significant difference in baseline LDL cholesterol levels between the 2 groups. In the sensitivity analysis with propensity-score matched control, Meth use remains an independent predictor of cSVD after adjusting for hyperlipidemia.
There were also significantly more patients with a history of smoking in the Meth group than in the Non-Meth group (57% vs. 30%, p < 0.001). Since previous study demonstrated a dose-dependent association between pack-years of smoking and WMH progression26, smoking is likely a confounding factor in our retrospective study.
There was no statistically significant difference in stroke mechanisms between the Meth and Non-meth group. The Non-meth group had a trend of more cardioembolic stroke, likely due to significantly older ages. There was only one case of possible Reversible Cerebral Vasoconstriction Syndrome (RCVS) as the possible stroke mechanism in the Meth group.
Since cSVD increases the risk of cognitive impairment and vascular dementia, methamphetamine abuse in young adults may have more significant long-term public health concerns than stroke and other detrimental effects.
Our study has a few limitations. First, our data suggested a possible association and cannot prove causal relationship between methamphetamine use and cSVD. Additional studies are required to corroborate our findings. Second, some patients were unable to provide history of drug use due to aphasia or severe neurological deficit. Only 932 patients (71%) in the Non-Meth group had a UDS. The rate of methamphetamine abuse was likely underestimated. Third, there was no information regarding the route, frequency, and duration of methamphetamine abuse. Lastly, smoking, and other confounding factors cannot be ruled out in this retrospective study. Further studies are warranted to adjust for all confounding factors and to investigate the temporal relationship between methamphetamine abuse and cSVD.
Of note, it was challenging to get accurate information on polysubstance abuse, particularly, in patients with neurological deficit. It may be also unethical to conduct randomized controlled studies on methamphetamine abuse. Well-designed prospective registry may be a good option to further investigate the effect of chronic methamphetamine use on cSVD.
In conclusion, our preliminary results demonstrates that methamphetamine abuse is common in young adults with acute ischemic stroke and increases the risk of cSVD. Given increased prevalence of methamphetamine abuse in young adults, additional studies are warranted to investigate the effects of chronic methamphetamine use on the pathogenesis of stroke, cSVD, and vascular dementia.
Acknowledgements
We thank Xiaoqi Cheng & Dongmei Liao International Stroke Research Scholarship for generous support of this research project.
Author contributions
Z.Z. contributed to study design, data acquisition, statistical analysis, and interpretation. B.V., S.J.L., H.B. and D.S. contributed to data acquisition. T.Q. contributed to discussion and verification of statistical analysis. M.S., J.E.S., D.C., and P.C. contributed to data interpretation and draft revision. W.Y. contributed to study design, data analysis, drafting and finalizing the manuscript.
Data availability
Data of this study are available from the corresponding author on reasonable request.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Rothrock JF, Rubenstein R, Lyden PD. Ischemic stroke associated with methamphetamine inhalation. Neurology. 1988;38(4):589–592. doi: 10.1212/WNL.38.4.589. [DOI] [PubMed] [Google Scholar]
- 2.McIntosh A, Hungs M, Kostanian V, Yu W. Carotid artery dissection and middle cerebral artery stroke following methamphetamine use. Neurology. 2006;67(12):2259–2260. doi: 10.1212/01.wnl.0000249180.61312.d3. [DOI] [PubMed] [Google Scholar]
- 3.Westover AN, McBride S, Haley RW. Stroke in young adults who abuse amphetamines or cocaine: A population-based study of hospitalized patients. Arch. Gen. Psychiatry. 2007;64(4):495–502. doi: 10.1001/archpsyc.64.4.495. [DOI] [PubMed] [Google Scholar]
- 4.Ho EL, Josephson SA, Lee HS, Smith WS. Cerebrovascular complications of methamphetamine abuse. Neurocrit. Care. 2009;10(3):295–305. doi: 10.1007/s12028-008-9177-5. [DOI] [PubMed] [Google Scholar]
- 5.Huang MC, Yang SY, Lin SK, Chen KY, Chen YY, Kuo CJ, Hung YN. Risk of cardiovascular diseases and stroke events in methamphetamine users: A 10-year follow-up study. J. Clin. Psychiatry. 2016;77(10):1396–1403. doi: 10.4088/JCP.15m09872. [DOI] [PubMed] [Google Scholar]
- 6.Lappin JM, Darke S, Farrell M. Stroke and methamphetamine use in young adults: A review. J. Neurol. Neurosurg. Psychiatry. 2017;88(12):1079–1091. doi: 10.1136/jnnp-2017-316071. [DOI] [PubMed] [Google Scholar]
- 7.Darke S, Lappin J, Kaye S, Duflou J. Clinical characteristics of fatal methamphetamine-related stroke: A national study. J. Forensic. Sci. 2018;63(3):735–739. doi: 10.1111/1556-4029.13620. [DOI] [PubMed] [Google Scholar]
- 8.Nakagawa K, Vento MA, Ing MM, Seto TB. Racial disparities in methamphetamine-associated intracerebral hemorrhage. Neurology. 2015;84(10):995–1001. doi: 10.1212/WNL.0000000000001339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Swor DE, Maas MB, Walia SS, Bissig DP, Liotta EM, Naidech AM, Ng KL. Clinical characteristics and outcomes of methamphetamine-associated intracerebral hemorrhage. Neurology. 2019;93(1):e1–e7. doi: 10.1212/WNL.0000000000007666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zhu Z, Osman S, Stradling D, Shafie M, Yu W. Clinical characteristics, and outcomes of methamphetamine associated versus non methamphetamine intracerebral hemorrhage. Sci. Rep. 2020;10(1):6375. doi: 10.1038/s41598-020-63480-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hart CL, Gunderson EW, Perez A, Kirkpatrick MG, Thurmond A, Comer SD, Foltin RW. Acute physiological and behavioral effects of intranasal methamphetamine in humans. Neuropsychopharmacology. 2008;33(8):1847–1855. doi: 10.1038/sj.npp.1301578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Darke S, Kaye S, McKetin R, Duflou J. Major physical and psychological harms of methamphetamine use. Drug. Alcohol. Rev. 2008;27(3):253–262. doi: 10.1080/09595230801923702. [DOI] [PubMed] [Google Scholar]
- 13.de Leeuw FE, de Groot JC, Oudkerk M, Witteman JC, Hofman A, van Gijn J, Breteler MM. Hypertension and cerebral white matter lesions in a prospective cohort study. Brain. 2002;125(Pt 4):765–772. doi: 10.1093/brain/awf077. [DOI] [PubMed] [Google Scholar]
- 14.Klarenbeek P, van Oostenbrugge RJ, Rouhl RP, Knottnerus IL, Staals J. Ambulatory blood pressure in patients with lacunar stroke: Association with total MRI burden of cerebral small vessel disease. Stroke. 2013;44(11):2995–2999. doi: 10.1161/STROKEAHA.113.002545. [DOI] [PubMed] [Google Scholar]
- 15.Staals J, Makin SD, Doubal FN, Dennis MS, Wardlaw JM. Stroke subtype, vascular risk factors, and total MRI brain small-vessel disease burden. Neurology. 2014;83(14):1228–1234. doi: 10.1212/WNL.0000000000000837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Pantoni L. Cerebral small vessel disease: from pathogenesis and clinical characteristics to therapeutic challenges. Lancet Neurol. 2010;9(7):689–701. doi: 10.1016/S1474-4422(10)70104-6. [DOI] [PubMed] [Google Scholar]
- 17.Wardlaw JM, Smith C, Dichgans M. Mechanisms of sporadic cerebral small vessel disease: Insights from neuroimaging. Lancet Neurol. 2013;12(5):483–497. doi: 10.1016/S1474-4422(13)70060-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Schwamm LH, Fonarow GC, Reeves MJ, Pan W, Frankel MR, Smith EE, et al. Get With the Guidelines-Stroke is associated with sustained improvement in care for patients hospitalized with acute stroke or transient ischemic attack. Circulation. 2009;119(1):107–115. doi: 10.1161/circulationaha.108.783688. [DOI] [PubMed] [Google Scholar]
- 19.Kernan, W.N., Ovbiagele, B., Black, H.R., et al. Guidelines for the prevention of stroke in patients with stroke and transient ischemic attack: A guideline for healthcare professionals from the American Heart Association/American Stroke Association [published correction appears in Stroke. 2015 Feb;46(2):e54]. Stroke. 2014;45(7):2160–2236. 10.1161/STR.0000000000000024. [DOI] [PubMed]
- 20.Osman S, Zhu Z, Farag M, Groysman M, Dastur CK, Akbari Y, Stern-Nezer SRJ, Stradling D, Yu W. Intracerebral hemorrhage: Who gets tested for methamphetamines and why might it matter? BMC Neurol. 2020;27(20):392. doi: 10.1186/s12883-020-01967-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Fazekas F, Kleinert R, Offenbacher H, Schmidt R, Kleinert G, Payer F, Radner H, Lechner H. Pathologic correlates of incidental MRI white matter signal hyperintensities. Neurology. 1993;43(9):1683–1689. doi: 10.1212/WNL.43.9.1683. [DOI] [PubMed] [Google Scholar]
- 22.Deb S, Austin PC, Tu JV, Ko DT, Mazer CD, Kiss A, Fremes SE. A review of propensity-score methods and their use in cardiovascular research. Can. J. Cardiol. 2016;32(2):259–265. doi: 10.1016/j.cjca.2015.05.015. [DOI] [PubMed] [Google Scholar]
- 23.Nylander R, Fahlstrom M, Rostrup KJ, Damangir S, Ahlstrom H, et al. Quantitative and qualitative MRI evaluation of cerebral small vessel disease in an elderly population: A longitudinal study. Acta Radiol. 2018;59:612–618. doi: 10.1177/0284185117727567. [DOI] [PubMed] [Google Scholar]
- 24.Han F, Zhai FF, Wang Q, et al. Prevalence and risk factors of cerebral small vessel disease in a Chinese population-based sample. J. Stroke. 2018;20(2):239–246. doi: 10.5853/jos.2017.02110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Riley ED, Chow FC, Josephson SA, Dilworth SE, Lynch KL, Wade AN, Braun C, Hess CP. Cocaine use and white matter hyperintensities in homeless and unstably housed women. J. Stroke Cerebrovasc. Dis. 2021;30(5):105675. doi: 10.1016/j.jstrokecerebrovasdis.2021.105675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Power MC, Deal JA, Sharrett AR, Jack CR, Jr, Knopman D, Mosley TH, Gottesman RF. Smoking and white matter hyperintensity progression: The ARIC-MRI study. Neurology. 2015;84(8):841–848. doi: 10.1212/WNL.0000000000001283. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data of this study are available from the corresponding author on reasonable request.