Abstract
Areca nut (AN) is used for traditional herbal medicine and social activities in several countries. It was used as early as about A.D. 25-220 as a remedy. Traditionally, AN was applied for several medicinal functions. However, it was also reported to have toxicological effects. In this review article, we updated recent trends of research in addition to acquire new knowledge about AN. First, the history of AN usage from ancient years was described. Then, the chemical components of AN and their biological functions was compared; arecoline is an especially important compound in AN. AN extract has different effects caused by different components. Thus, the dual effects of AN with pharmacological and toxicological effects were summarized. Finally, we described perspectives, trends and challenges of AN. It will provide the insight of removing or modifying the toxic compounds of AN extractions for enhancing their pharmacological activity to treat several diseases in future applications.
Keywords: areca nut, component, pharmacological activity, toxicological effect
1. The History and Usage of AN
Areca nut (AN) as the fruit of the palm tree Areca cattechu Linn. provides popular psychoactive substances and is used as a traditional herbal drug in countries of South and Southeast Asia, including China and India (Figure 1). AN was used for antidepression recorded in the Chinese book called YiWuZhi (The Collection of Foreign Materials) as early as the period of A.D. 25-220. AN was used as anti-parasitic medicine according to the Chinese book called YaoLu (Medicine Record) in the period of A.D. 220-265. Moreover, AN was also described to treat miasma in the Chinese book, HelinYulu, during the period of A.D. 1127-1279. In addition to its medical applications, AN also provides functions for social and religious activities [1]. AN chewers were estimated as more than 600 million people located mainly in Asian countries like India, Pakistan and Taiwan as well as migrants from South Africa and the UK, which account for at least 10% of the world population [2,3,4]. A survey from the United States showed that 17% of people self-reported that they have ever used AN products, and 31% people reported that they had friends/families of AN users, indicating the high percentage of AN usage [5]. Although AN has the potential of treating malaria, diarrhea, ascariasis, edema, stagnation of food, arthritis, and beriberi [6], it still provides a dilemma of pharmacologically useful as well as toxic effects. The biological functions of AN components and their pharmacological and toxicological effects are described in detail as below.
Figure 1.
The Areca cattechu Linn. palm tree and its fruits (AN). (A) Habitus of the Areca cattechu Linn. palm tree. (B) Fruit stands of the A. cattechu tree. (C) Areca nuts with tip removed are used for chewing. (D) AN alone or combined with betel leaf, betel stem inflorescence and slaked lime is a popular psychoactive drug in subtropical and tropical countries. (E) Dried AN boiled in water is applied in traditional Chinese medicine. (F) Fibers of ANs provide substantial risks for oral diseases.
AN provides popular compounds with several studies focusing on it. There are several articles describing the effects caused by areca nut (AN) previously [6,7,8,9]. In addition, some of the review articles focused on specific aspects, such as carcinogenic effects [10], addiction [11], AN cessation [12], genetic and epigenetic instability [13], submucous fibrosis progression [14], oral cancer [15] and liver disease [4]. We here review the results of these reports and evaluate the controversial roles of AN.
There are more than hundred articles reporting about AN and its components which indicates the importance of AN. However, these articles covered different fields. We summarized and evaluated the recently published reports in this article to update recent trends of research in addition to acquire new knowledge about AN. Moreover, this article focused on the effect of AN (Figure 1C) but not betel nut (Figure 1D). Although AN is also called betel nut, betel nut is usually combined with other materials, such as betel leaf, betel stem inflorescence and slaked lime. In order to exclude the effect caused by the materials other than AN, we did not include reports including the additions that commonly come along with betel nut consumption.
This review contains four parts. The first part, Section 1 describes the history of AN usage. Section 2 introduces the components of AN and their biological effects. Arecoline is an important compound of AN. In addition, AN components are a mixture of different components, and AN extract has different effects from each component. The dual effects of AN with pharmacological and toxicological effects were summarized. The Section 3 introduces the pharmacological effects, and the Section 4 explores the toxicological effects of AN extract, other mixture compounds and arecoline.
2. AN Components and Their Biological Effects
The components of AN include alkaloids, flavonoids, tannins, triterpenoids and steroids, fatty acids and others [6,7]. Their biological activities are described below.
2.1. Alkaloids
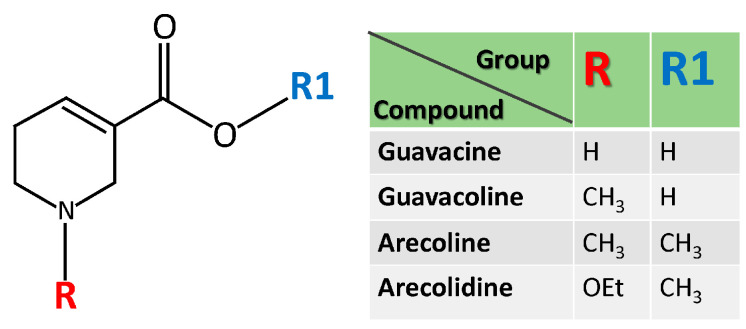
Alkaloids of AN have addictive and carcinogenic effects [16]. There are four major alkaloids, including arecoline, arecaidine, guvacoline and guvacine. Arecoline (N-methyl-1,2,5,6-tetrahydropyridine-3-carboxylic acid methyl ester) is the most abundant alkaloid and a key chemical constituent contributing to the carcinogenicity of AN [17]. The chemical structure of these components is listed in Figure 2.
Figure 2.
The chemical structure of areca alkaloids includes arecoline, arecaidine, guvacoline and guvacine. Their structures are similar but carry different functional groups, R1 and R2.
Arecaidine is the hydrolyzed product of arecoline. Arecoline and arecaidine increase collagen deposition by producing tissue inhibitors of metalloproteinases (TIMPs) in the extracellular matrix to alter the microenvironmental conditions of carcinogenic effects [18]. Guvacoline decreases exploration activity and social interaction in zebrafish [19]. Guvacine is the second abundant alkaloid followed by arecoline. The nitrosated derivate of guvacine causes DNA breaks as well as oxidative stress resulting in oral carcinogenesis [18]. Nicotine plays dual roles in anti- and pro-inflammatory effects. It exerts anti-inflammatory effects in ulcerative colitis, arthritis, sepsis, and endotoxemia but promotes diseases such as periodontitis and gingivitis due to its pro-inflammatory effects [20]. Isoguvacine, a GABA agonist, decreases blood pressure and heart rate of kidney-clipped hypertension rats by inhibiting commissural nuclei of the solitary tract [21].
2.2. Flavonoids
Flavonoids belong to polyphenolic compounds with wide biological functions for treating several disorders, such as cancer, cardiovascular complications, chronic inflammation and hypoglycemia [22]. At least 11 flavonoids were isolated from AN, including isorhamnetin, chrysoeriol, luteolin, quercetin, jacareubin and liquiritigenin [6,7]. Isorhamnetin has several pharmacological activities including cardiovascular and cerebrovascular protection, anti-inflammation, anti-tumor, antioxidation, prevention of obesity and organ protection [23]. Chrysoeriol has anticancer, antibacterial, anti-inflammatory, anti-fungal, anti-insecticide, anti-osteoporosis effects and prevents diabetes, inflammation, Parkinson’s disease, osteoporosis, and cardiovascular diseases [24]. Luteolin possesses anticancer, anti-inflammatory, antimicrobial, antioxidant and antidiabetic effects and provides health benefits for patients with Alzheimer’s disease, Parkinson’s disease, cardiac diseases and obesity [25]. Quercetin is an antioxidant, antimicrobial, antidiabetic, anticancerous and anti-inflammatory agent for preventing allergies, ulcers, microbes, cancer and Alzheimer’s disease [26]. Jacareubin is reported to have anti-inflammatory activity. The receptor-binding domain of SARS-CoV-2 Spike protein induces TNF-α synthesis and cell infiltration in the lungs of mice. Pre-treatment with jacareubin can inhibit Toll-like receptor (TLR) 4-induced lung inflammatory response caused by SARS-CoV-2 Spike protein [27]. Liquiritigenin causes antidepressant, antianxiety, antipsychostimulant, memory-enhancing and neuroprotective activities on neurodegenerative diseases, such as Parkinson’s disease, Alzheimer’s disease, stroke and brain glioma. In addition, it also has several pharmacological activities, such as radical scavenging, antibacterial and anti-inflammatory activities [28]. Another two of the new flavonoids, calquiquelignan N and calquiquelignan M, from AN provide cytotoxicity to human cancer HepG2 cell line [29].
2.3. Tannins
At least 12 tannins were isolated from AN, such as catechin and procyanidins [6,7]. Tannins are polyphenol antioxidants. They have anti-inflammatory, antimicrobial, antioxidant and anticancer effects and prevent cardiovascular, neuroprotective and general metabolic diseases [30]. Catechin shows effectiveness as anti-inflammatory, increasing memory, antidiabetic, anticancer, bactericidal, anti-arthritis, hepatoprotection and neuroprotection, through activation of NF-κB, TLR4/NF-κB, Nrf-2, COMT and MAPKs pathways [31,32]. Procyanidins have anti-inflammatory, antioxidant, antibacterial and anti-tumor effects. Therefore, they provide potential treatments for several diseases, such as Alzheimer’s disease, rheumatoid arthritis, diabetes, obesity, tumors and oral diseases. Among the procyanidins, procyanidin A1 inhibits the release of inflammatory factors IL-6, PGE2, NO and TNF-α by inactivating the IκB/NF-κB p65 pathway. Procyanidin A1 and B2 increase the level of antioxidant genes including HO-1, NQO1 and γ-GCS through activating the AMPK/Nrf2 pathway [33].
2.4. Terpenoids
At least eight terpenoids were isolated from AN, such as 3-carene, procurcumenol and ursolic acid [6]. Terpenoids have pharmacological effects including anti-pyretic, anti-analgesic, anti-inflammatory, antibacterial, antiviral and anticancer, such as in lung cancer [34]. 3-carene shows antibacterial activity to inhibit the growth of Escherichia coli in a minimum inhibitory concentration test and antioxidant activity as shown by the oxygen radical absorbance capacity activity test [35]. Procurcumenol provides anti-platelet aggregation activity in vitro promoting blood circulation [36]. In addition, triterpenoids also have biological activities including hepatoprotection, anti-inflammation, antiviral and anti-tumor [37]. Ursolic acid is a pentacyclic triterpenoid with anti-inflammatory, apoptosis induction and anticarcinogenic activities by inhibiting cancer cell proliferation and viability and preventing tumor metastatic activity and angiogenesis [38]. Ursolic acid and 3-O-acetylursolic acid arrest cell cycle and increase the apoptotic population of A375 melanoma cells by activating caspases and attenuating Bcl-2 expression [39].
2.5. Steroids
Steroids are regulators of lipid metabolism [40]. At least four steroids have been isolated from AN, such as cycloartenol and beta-sitosterol [6]. Cycloartenol increases IL-6 expression and suppresses IL-10 expression, indicating its immune potentiating effects [41]. Cycloartenol also inhibits proliferation and the colony formation by arresting Sub-G1 cell cycle and triggering apoptosis through altering the expression of Bax (Bcl-2-associated X protein) and Bcl-2 of glioma U87 cells. In addition, it inhibits the migration of glioma cells through suppressing the phosphorylation of p38 MAP kinase [42]. Cycloartenol is a putative active ingredient against type 2 diabetes mellitus through regulating the expression of AKT1, TNF-α, MAPK3 and MAPK1 pathways predicted by molecular-docking plus GO/KEGG analysis [43]. Beta-sitosterol is involved in several signaling pathways regarding cell cycle, apoptosis, proliferation, survival, invasion, angiogenesis, metastasis, anti-inflammatory, anticancer (breast, prostate, colon, lung, stomach and leukemia), hepatoprotective, antioxidant, cardioprotective and antidiabetic effects [44].
2.6. Fatty Acids
AN contains several important fatty acids, among others lauric acid, myristic acid, palmitic acid, stearic acid, arachidic acid and dodecenoic acid [6,7]. Fatty acids are key materials of anti-inflammation mediators and crucial for development, growth and preservation of human health [45]. Feeding chicks with 350 and 500 mg/kg of lauric acid monoglyceride and cinnamaldehyde improved their intestinal morphology, improved the body’s antioxidant capacity and downregulated the expression of inflammatory factors [46]. Myristic acid (200 μg/mL) can reduce skin inflammation. Myristic acid exerted anti-inflammatory activity by increasing the production of IL-10 in LPS-stimulated JA774A.1 macrophage cells. Myristic acid (12.5–100 mg/kg) also showed anti-inflammatory effects on the acute and chronic TPA-induced ear edema of mice [47]. Palmitic acid was shown to inhibit the growth and metastasis of gastric cancer. Palmitic acid (50 μM) could block the proliferation and invasion of the MGC-803 and BGC-823 gastric cells. Palmitic acid (50 mg/kg) treatment also decreased gastric cancer growth in xenografted models of nude mice. Moreover, gastric cancer cells treated with palmitic acid decreased the expression of p-STAT3, p-JAK2, N-cadherin and vimentin indicating that palmitic acid inhibited the growth of gastric cancer by blocking the STAT3 signaling pathway [48]. Stearic acid attenuates idiopathic pulmonary fibrosis by reducing profibrotic signaling. Stearic acid also reduced the expression of fibrosis markers, α-SMA and collagen type 1 and the levels of p-Smad2/3 in MRC-5 cells treated with TGF-β1. Stearic acid also reduced the levels of bleomycin-induced hydroxyproline, an amino acid present in collagen, in a mouse model indicating that stearic acid attenuates idiopathic pulmonary fibrosis through reducing the p-Smad2/3 pathway [49].
2.7. Others
The other compounds of AN also have biological effects, such as vanillin, alpha-terpineol, benzyl alcohol, capric acid, resveratrol, quinic acid, chrysophanol, physcion, p-hydroxybenzoic acid, epoxyconiferyl alcohol, protocatechuic acid, isovanillic acid, ferulic acid, vanillic acid and polyphenols. Vanillin has anti-tumor, anti-mutagenic, antioxidant, analgesic and anti-erythrocyte-sickling properties [50]. Alpha-terpineol was demonstrated to have a potential in reducing mechanical hypernociception/inflammatory response, cardiovascular and gastric lesions, anticonvulsant and antidiarrheal activities [51]. Benzyl alcohol plays important roles in bacteriostatic, anesthetic processes and relieving nerve and ganglionic pain. Benzyl alcohol also accelerates the healing process of Achilles tendon injury by increasing collagen and blood capillaries and causing fewer inflammatory cells through activating the TGF-β1/Smad2/3 pathway [52]. Capric acid has several targets including PPAR-γ, AMPA receptors, gut dysbiosis and inflammatory of oxidative stress pathways. Moreover, Capric acid exhibits an ameliorative effect of neurological diseases including affective disorders, epilepsy and Alzheimer’s disease [53]. Resveratrol shows anti-aging, anticancer and antioxidant properties [54]. Quinic acid has different biological effects, including antioxidant, aging protective, antidiabetic, anti-nociceptive, antimicrobial, anticancer, antiviral and analgesic effects. In addition, quinic acid also shows antibacterial effects by inhibiting the functions of ribosomes [55,56,57,58]. Chrysophanol has dual pharmacological and toxicity effects. Pharmacological effects include anticancer, antioxidation, neuroprotection, antibacterial, and antiviral ones and regulating blood lipids. However, the toxic effects of chrysophanol are hepatotoxic and nephrotoxic [59]. Anthraquinones are important compounds with different pharmacological effects, such as antibacterial, anti-tumor, antioxidant, laxative and other activities [60]. Physcion is a natural anthraquinone exerting anti-inflammatory, anticancer, antimicrobial and hepatoprotective properties [61]. The p-hydroxybenzoic acid is confirmed to have therapeutic advantages, such as antibacterial, anticancer, antioxidative and anti-inflammatory activities. However, various adverse effects including male infertility and female breast cancer were reported [62]. Epoxyconiferyl alcohol showed the cytotoxicity of human liver cancer cell lines HepG2 detected by the sulforhodamine B assay [63]. Protocatechuic acid has diverse pharmacological activities, such as antioxidant, neuroprotective, anti-inflammatory, antibacterial, anticancer, antiviral, antiosteoporotic and anti-aging activities and analgesia, protection from metabolic syndrome and preservation of kidney, liver and reproductive organs [64]. Isovanillic acid has inhibitory activity on tyrosinase and tyrosine acidase and enzymes for melanin production in mouse melanoma B16 cells. Isovanillic acid has a potential for whitening cosmetics [65]. Isovanillic acid can also attenuate fibrin polymer formation for blood clots and degrades clots by inhibiting fibrinoligase and procoagulant proteases and prolongs the time of coagulation. It is suggested that isovanillic acid has the potential as inhibitor of thrombin [66]. Ferulic acid is a phenolic compound with several pharmacological activities, especially in oxidative stress, inflammation, vascular endothelial injury, fibrosis, apoptosis and platelet aggregation [67]. Vanillic acid protects the liver by reducing transaminase levels, inflammatory cytokines and accumulation of collagen and preventing liver fibrosis. In addition, it decreases the adipose tissue of mice through the activation of the AMPK pathway [68]. Polyphenols in lipopolysaccharides of AN reduce the generation of reactive oxygen species by activating the Nrf2/HO-1 antioxidant pathways and inhibiting the MAPK pathway in RAW264.7 cells [69].
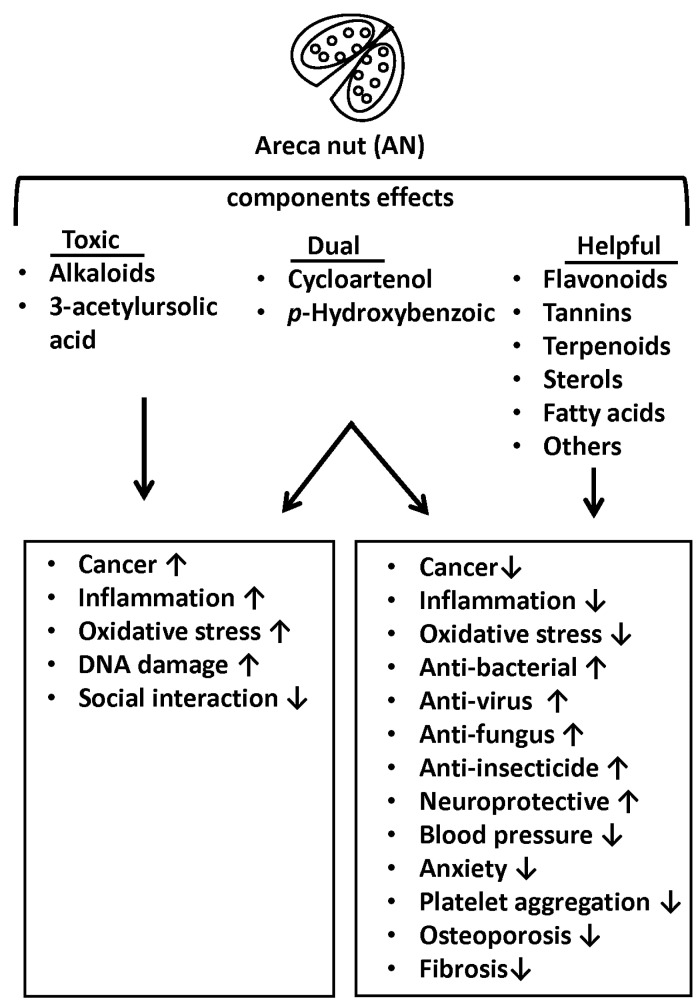
In summary, the different components of AN have their own biological or toxicological effects. Most of the components have pharmacological effects, except alkaloids and 3-acetylursolic acid have toxicological effects, and cycloartenol, chrysophanol and p-hydroxybenzoic acid have both pharmacological and toxicological effects. The biological effects of AN components are listed in Table 1. Additionally, their effects are summarized in Figure 3.
Table 1.
The biological effects of AN components.
| Effects | Components | Biological Materials | References |
|---|---|---|---|
| Cancer | ↑: Alkaloids | [16] | |
| Arecoline | Hep-2 and KB cells | [17] | |
| Arecaidine | fibroblasts | [18] | |
| Guvacine | human buccal epithelial cells | [18] | |
| ↓: Flavonoids | |||
| Isorhamnetin | >8 cells of different cancer types, mice | [23] | |
| Chrysoeriol | >3 cells of different cancer types, mice | [24] | |
| Luteolin | >9 cells of different cancer types | [25] | |
| Quercetin | >5 cells of different cancer types, mice | [26] | |
| ↓: Tannins | |||
| Catechin | HCT-116 cells | [31] | |
| Procyanidins | >4 cells of different cancer types | [33] | |
| ↓: Terpenoids | |||
| Ursolic acid | mice, human | [38] | |
| ↓: Steroids | |||
| Cycloartenol | U87 cells | [42] | |
| β-sitosterol | >16 cells of different cancer types, rat | [44] | |
| ↓: Fatty acids | |||
| Palmitic acid | MGC-803, BGC-823 cells, mice | [48] | |
| ↓: Others | |||
| Vanillin | cells, rats | [50] | |
| Resveratrol | breast cancer cells, A549, HeLa, DU-145, HepG2 | [54] | |
| Quinic acid | human | [55] | |
| Chrysophanol | HepG2, MCF-7, T47D, HCT116, A549 cells | [59] | |
| Anthraquinones | >20 cells of different cancer types, mice | [60] | |
| Physcion | in vitro, in vivo | [61] | |
| p-Hydroxybenzoic acid | rats | [62] | |
| Epoxyconiferyl alcohol | HepG2 | [63] | |
| Protocatechuic acid | >6 cells of different cancer types | [64] | |
| Inflammation | ↑: Steroids | ||
| Cycloartenol | human blood | [41] | |
| ↓: Alkaloids | |||
| Nicotine | human | [20] | |
| ↓: Flavonoids | |||
| Isorhamnetin | >6 cells, mice, rats | [23] | |
| Chrysoeriol | RAW264.7, HaCaT cells, mice, rats | [24] | |
| Luteolin | 264.7, HUVEC cells, mice, rats | [25] | |
| Quercetin | RAW264.7, HUVEC cells, rats, human | [26] | |
| Jacareubin | mice | [27] | |
| Liquiritigenin | RAW264.7 cells, rats, mice | [28] | |
| ↓: Tannins | |||
| Catechin | human stomach cancer cell lines | [31] | |
| Procyanidins | RAW264.7 cells, rats, mice | [33] | |
| ↓: Terpenoids | |||
| Ursolic acid | mice, rats | [38] | |
| ↓: Steroids | |||
| β-sitosterol | J774A.1 cells, mice, rats | [44] | |
| ↓: Fatty acids | |||
| Lauric acid | chicks | [46] | |
| Myristic acid | JA774A.1 cells, mice | [47] | |
| ↓: Others | |||
| α-Terpineol | mice | [51] | |
| Benzyl alcohol | rats | [52] | |
| Capric acid | IPEC-J2, COS-7 cells | [53] | |
| Physcion | MH7A cells | [61] | |
| p-Hydroxybenzoic acid | rats | [62] | |
| Protocatechuic acid | PC12, CGNs 2. BV2, cells, mice, porcine |
[64] | |
| Ferulic acid | mice, rat | [67] | |
| Vanillic acid | mice, | [68] | |
| Oxidative stress | ↑: Alkaloids | ||
| Guvacine | human | [18] | |
| ↓: Flavonoids | |||
| Isorhamnetin | RPE, H9C2, C2C12 cells | [23] | |
| Luteolin | HT-29, SNU-407 cells, rats | [25] | |
| Quercetin | SH-SY5Y cells, rats | [26] | |
| Liquiritigenin | [28] | ||
| Calquiquelignan N and M | HepG2 cells | [29] | |
| ↓: Tannins | |||
| Procyanidins | RAW264.7 cells | [33] | |
| ↓: Steroids | |||
| β-sitosterol | RAW 264.7, HT22 cells | [44] | |
| ↓: Others | |||
| Vanillin | B16F0 cells | [50] | |
| Capric acid | SY5Y, Neuro2a, AML12 cells | [53] | |
| Resveratrol | none | [54] | |
| Quinic acid | none | [57] | |
| Chrysophanol | BV2 cells, mice | [59] | |
| Anthraquinones | rats | [60] | |
| p-Hydroxybenzoic acid | CGN cells | [62] | |
| Protocatechuic acid | HUVEC cells, rats | [64] | |
| Ferulic acid | HEK293, SH-SY5Y, HUVEC cells, rats | [67] | |
| Polyphenols | RAW264.7 cells | [69] | |
| Antibacterial | ↑: Flavonoids | ||
| Chrysoeriol | >5 Gram positive bacteira, >5 Gram negative bacteira |
[24] | |
| Quercetin | S. aureus, S. saprophyticus, E. coli, P. aeruginosa | [26] | |
| Liquiritigenin | M. tuberculosis, M. bovis, S. aureus, S. epidermidis, S. hemolyticus | [28] | |
| ↑: Tannins | |||
| Catechin | S. aureus and L. monocytogenes | [31] | |
| Procyanidins | S. mutans, B. cereus | [33] | |
| ↑: Terpenoids | |||
| 3-carene | E. coli | [35] | |
| ↑: Others | |||
| Benzyl alcohol | rats | [52] | |
| Quinic acid | P. aeruginosa | [56] | |
| Anthraquinones | S. aureus, S. epidermidis, P. aeruginosa, E. faecalis, P. vulgaris, P. mirabilis, S. typhi, E. cloacae, E. aerogenes, K. pneumoniae, Vibrio harveyi | [60] | |
| Physcion | S. aureus, P. aeruginosa, C. albican, M. gypseum | [61] | |
| p-Hydroxybenzoic acid | rats | [62] | |
| Protocatechuic acid | E. coli, P. ceruminous, S. aureus, B. cereus, S. pneumoniae, A. barramundi, H. pylori | [64] | |
| Antivirus | ↑: Others | ||
| Quinic acid | RNA-dependent RNA polymerase (RdRp) of the SARS-CoV-2 | [58] | |
| Chrysophanol | JEV virus | [59] | |
| Protocatechuic acid | HBV, NDV, | [64] | |
| Anti-fungus | ↑: Flavonoids | ||
| Chrysoeriol | F. graminearum, P. graminicola | [24] | |
| Anti-insecticide | ↑: Flavonoids | ||
| Chrysoeriol | A. pisum, R. meliloti, S. litura | [24] | |
| Neuroprotective | ↑: Flavonoids | ||
| Liquiritigenin | D10 cells | [28] | |
| ↑: Tannins | |||
| Catechin | rats | [32] | |
| DNA damage | ↑: Alkaloids | ||
| Guvacine | human buccal epithelial cells | [18] | |
| Blood pressure | ↓: Alkaloids | ||
| Isoguvacine | rats | [21] | |
| Social interaction | ↓: Alkaloids | ||
| Guvacine | zebrafish | [19] | |
| Anxiety | ↓: Flavonoids | ||
| Liquiritigenin | mice | [28] | |
| Platelet aggregation | ↓: Terpenoids | ||
| Procurcumenol | rats | [36] | |
| Osteoporosis | ↓: Flavonoids | ||
| Chrysoeriol | MC3T3-E1 cells | [24] | |
| Fibrosis | ↓: Fatty acids | ||
| Stearic acid | MRC-5 cells, mice | [49] |
↑: Activated; ↓: Inhibited.
Figure 3.
Pharmacological and toxicological effects of AN components. Most of the AN components are helpful for organisms from a health point of view. However, some AN components have toxicological effects. ↑: Activated; ↓: Inhibited.
3. Pharmacological Effects of AN Extracts, Other Mixture Compounds and Arecoline on Several Diseases
3.1. Gastric and Intestinal Diseases
Two compounds isolated from the dried fruit of AN, 2,4-dimethoxyphenyl-β-D-glucopyranoside (26) and isorhamnetin 3-O-(6”-O-α-L-rhamnopyransoyl) β-D-glucopyranoside (34), showed weak cytotoxicity and had anticancer effects against human gastric cancer cell lines (BGC-823), indicating that AN compounds have the potential for treating gastric cancer [70]. Dried pericarp of AN extracts (Areca pericarpium extract) and arecoline can induce the contractions of porcine lower esophageal sphincter sling and clasp muscles in a dose–response manner. However, the muscarinic receptor antagonist atropine inhibited such contractions. These results showed that AN extract and arecoline induced contractions were mediated by muscarinic receptors indicating the possibility of developing as an alternative therapy for gastroesophageal reflux disease [71].
3.2. Depression and Anxiety
The metabolites of AN analyzed with the UPLC-MS/MS system showed that 93 metabolites were capable of targeting 141 depression-related genes, including L-phenylalanine, nicotinic acid, L-tyrosine, protocatechuic acid, okanin, benzocaine, benzocaine, phloretic acid, syringic acid, cynaroside and 3,4-dihydroxybenzaldehyde. These results indicated that AN is rich in bioactive antidepression compounds [72]. Arecoline increased serotonin levels and social preference and elevated brain norepinephrine but reduced turnover of serotonin for activating central monoaminergic neurotransmission of anxiolytic effects in zebrafish through upregulation of c-fos, c-jun, egr2 and ym1. These findings suggested that arecoline possesses anxiolytic-like activity [73].
3.3. Liver Diseases
With AN extract treatment, hepatocellular carcinoma cells, HepJ5 and Mahlavu cells, increased the conversion of light chain 3 (LC3)-I to LC3-II, beclin levels and anti-thymocyte globulin 5+12 for ROS-mediated autophagy and lysosome formation. They also increased apoptotic proteins, such as Bax and cleaved poly (ADP-ribose) polymerase, and decreased the level of Bcl-2 for apoptosis. Tumor sizes and tumor weights in the AN extract-treated group significantly decreased compared to vehicle-treated group of nude mice. These studies demonstrated that the AN extract can inhibit the progression of hepatocellular carcinoma cells [74].
3.4. Neurological Disorders
Arecoline evoked an enhancement of the firing rate, including an increase of spike numbers per burst, elongation in the burst duration and the burst rate of dopaminergic neurons in the ventral tegmental area of rats. These findings may be used for potentially targeting AN cessation therapy [75]. Deficiencies of neuromuscular junctions in the motor neurons resulted in the degeneration of both neurons and muscles resulting in age-related mobility decline. Arecoline is a muscarinic acetylcholine receptor agonist capable of promoting synaptic exocytosis at neuromuscular junctions. Treatment of arecoline during the early stage of aging decreased muscle tissue and increased lifespan through the GAR-2/PLCβ pathway in the motor neurons of Caenorhabditis elegans. These results suggested that arecoline has a pharmacological potential to promote lifespan when it was applied at the early stage of aging [76]. Arecoline improves cognition, memory and some behavioral disorders of patients with schizophrenia or Alzheimer’s disease through activating postsynaptic muscarinic M1 receptors. Moreover, it reduced the dopaminergic hyperactivity throughout the modulation of M1, -2 and -4 receptors to ameliorate the negative symptoms of psychosis [77].
3.5. Bacterial Infection
Ethanolic and methanolic extracts of AN fruits have antibacterial effects against S. aureus, E. aerogenes, E. coli and S. enterica detected by both methods of minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC). These results suggested that AN fruit extracts can serve as a natural preservative [78]. Orally administered AN extracts 14 days before the intraperitoneal challenge with S. aureus in rats produced increased concentrations of white blood cells but did not affect red blood cells and other cell types. In addition, no harmful effects on liver and kidney functions were observed. HPLC analysis revealed that phenolic catechin and quercetin are the major compounds of AN extracts. These findings suggested that phenolic compounds of AN extract have immunomodulatory activity against S. aureus infection [79]. Polyphenols of AN ethanol extraction, including catechin, epicatechin and epigallocatechin gallate, have anti-infective activities against bacteria belonging to M. tuberculosis, Gram-positive S. aureus and Gram-negative E. coli. These findings suggested that polyphenols of AN provide antibacterial action against multiple antibacterial resistance such as those mentioned above [80]. Several volatile components of AN fiber extracted by simultaneous hydrodistillation–extraction showed to have antimicrobial effect. Nine bacteria and yeasts were determined. The results showed that the antimicrobial effects of Gram-positive bacteria, such as Streptococci, were better than that of Gram-negative strains, such as E. coli, and the yeast Candida albicans [81].
3.6. Skin Protection
Oral administration of AN procyanidins prevented UVB-induced photoaging by detecting epidermal thickness and collagen disorientation of the dorsal skin of CD-1 mice. In addition, inhibited UVB-induced photodamage, such as expression of cyclooxygenase-2 (COX-2) and matrix metalloproteinases (MMPs), including MMP-2, MMP-9 and TIMP metallopeptidase inhibitor 1 (TIMP1), was also observed. These finding suggested that AN procyanidins could attenuate solar UVB-induced premature skin aging [82]. Overproduced melanin may cause skin disorders, such as premature aging. Tyrosinase is the critical enzyme of melanogenesis, and reduced tyrosinase activity decreased melanin production. AN extract was demonstrated to have tyrosinase inhibitory effect using 3,4-dihydroxy-L-phenylalanine (L-DOPA) as the substrate of tyrosinase [83]. Elastin is important for keeping elasticity of the skin. Elastase is a protease enzyme for the degradation of elastin. The inhibition of elastase activity can prevent skin loss of elasticity and wrinkles. AN extract was demonstrated to have elastase inhibitory effects using N-succinyl-trialanyl-paranitroanilide as the substrate of elastase [83]. Taken together, AN extract has an anti- aging skin potential.
3.7. Anthelmintic Activity
AN crude aqueous extract damaged the morphology and average number of eggs of the nematode, Ascaridia galli, found in chicken. The body weight, erythrocyte, leukocyte and hemoglobin were increased in chicken with AN extract treatment. These data indicated that AN extract has anthelmintic efficacy against A. galli in chicken [84]. AN extract was found to change the morphology and decrease the motility time of flukes Fasciola spp., from the liver of buffaloes. This demonstrates that AN extract has anthelmintic efficacy against liver fluke [85].
3.8. Antioxidant
The DPPH (2,2-diphenyl-1-picryl-hydrazyl-hydrate) assay is used to detect the scavenging ability on stable DPPH radicals of compounds with hydrogen-donating capacity. A polysaccharide PAP1b isolated from AN containing mannose, galactose, xylose and arabinose showed the ability of scavenging hydroxyl radicals using a DPPH assay in vitro. Therefore, this polysaccharide from AM had a potential antioxidant effect for the food and pharmaceutical industry [86]. The ABTS (2,2′-azino-bis (3-ethylbenzothiazoline-6-sulfonic acid)) assay is used to detect the ability of transferring hydrogen atoms for neutralizing ABTS radical cations of compounds. AN ethanol extraction with phenolic content showed high free radical scavenging activity detected by both DPPH and ABTS assays. It suggested that the phenolics of AN have high antioxidant capacities [83].
3.9. Anti-Hypoxia
AN polyphenol has antioxidant, anti-inflammatory and antibacterial effects. Recent studies showed that they have protective effects on oxidative stress caused by hypoxia via improving blood gas index of hypoxic organism through scavenging excessive free radicals. These findings indicated that AN polyphenol can be used as potential anti-hypoxia drugs [87].
3.10. Anti-Osteoarthritis
Ethanol extracts of AN show anti-inflammatory effects on lipopolysaccharide (LPS)-induced inflammation through decreasing NO and expressing materials, inducible nitric oxide synthase (iNOS) and cyclooxygenase-2 (COX-2). It also suppressed carrageenan-induced inflammation in rats by decreasing edema and prostaglandin E2 levels. Therefore, AN has a potential as an anti-osteoarthritis agent [88].
In summary, AN extracts, other mixture compounds and arecoline have several pharmacological effects to prevent certain diseases that are listed in Table 2.
Table 2.
Pharmacological effects and mechanisms of AN extract, other mixture compounds and arecoline for several diseases.
| Diseases | Pharmacological Effects | AN Components | Mechanism | Biological Materials | References |
|---|---|---|---|---|---|
| Gastric and intestine | against gastric cancer cell | 15.91 μM 2,4-Compound 26, 20.13 μM Compound 34 | BGC-823 cells | [70] | |
| esophageal sphincter muscle contractions ↑ | 300 ng/L AN pericarp extracts, 300 nM arecoline | porcine | [71] | ||
| Depression and anxiety | antidepression | 141 depression-related genes | [72] | ||
| anxiolytic-like activity | 10 mg/L arecoline | ↑ c-fos, c-jun, egr2 and ym1 | zebrafish | [73] | |
| Liver | hepatocellular carcinoma progression ↓ | 20~30 µg/mL or 20 mg/kg AN extract | ↑ autophagy and apoptosis | HepJ5 and Mahlavu cells, nude mice | [74] |
| Nerve | dopaminergic neuron firing rate ↑ | 0.2 mg/kg arecoline | ↑ dopaminergic | rats | [75] |
| synaptic exocytosis at neuromuscular junctions ↑ | 0.2 mM arecoline | ↑ GAR-2/PLCβ pathway | C. elegans. | [76] | |
| improving cognition, memory and behavioral disorders | arecoline | ↑ muscarinic M1 receptors | human | [77] | |
| Bacterial infection | antibacterial effects | 1–100 mg/mL AN extracts | S. aureus, E. aerogenes, E. coli, S. enterica | [78] | |
| against Staphylococcus aureus | 1000 mg/kg BW AN extracts | S. aureus, rats | [79] | ||
| against Mycobacterium tuberculosis, Staphylococcus aureus, Escherichia coli | 0.975 ± 0.02 μg/mL AN extracts | M. tuberculosis, S. aureus, E. coli. | [80] | ||
| antimicrobial activity of nine Gram-negative, Gram-positive bacteria and yeast | 10.3 ± 1.1–40.0 ± 3.0 AN SHDE extract | B. subtilis, E. faecalis, E. coli, P. aeruginosa, S. aureus, S. agalactiae, S. canis, S. pyogenes, Candida albicans | [81] | ||
| Skin protection | UVB-induced photoaging ↓ | AN procyanidins | ↓ COX-2, MMPs | mice | [82] |
| tyrosinase and elastase inhibition | 295.79 ± 11.97 mg/g dry weight AN extract | ↓ Tyrosinase, elastase | [83] | ||
| Anthelmintic activity | against the nematode Ascaridia galli | 250 mg/mL, 100 mg/mL AN extract | Ascaridia galli, chicken | [84] | |
| against the fluke Fasciola spp. | 10%, 20%, 40% AN extract | Fasciola spp., buffaloes | [85] | ||
| Antioxidant | antioxidant | 2 mg/mL PAP1b | ↓ hydroxyl radicals | [86] | |
| antioxidant | 20 µL AN extract | ↓ free radical | [83] | ||
| Anti-hypoxia | protecting effects caused by hypoxia | AN polyphenols | ↓ free radicals | [87] | |
| Anti-osteoarthritis | suppressed inflammation | 1 and 10 mg/kg/day AN ethanol extract | ↓ iNOS, COX-2 | rats | [88] |
↑: Activated, ↓: Inhibited.
4. Toxicological Effects of AN Extract, Other Mixture Compounds and Arecoline on Several Diseases
4.1. Oral Diseases
AN chewing is supposed to be the major etiological factor in the manifestation of oral submucous fibrosis through dysregulating of proinflammatory cytokines, such as transforming growth factor-β [89]. However, a systematic meta-analysis showed that the prevalence of oral submucous fibrosis in AN chewers was 5% suggesting a low prevalence of oral submucous fibrosis in AN chewers. These results indicate that ANs may not be the sole cause of oral submucous fibrosis [90]. However, AN chewing is significantly associated with a negative prognosis for patients with oral cancer [91]. AN extract decreased viability of human buccal epithelial KB cells. It promotes apoptosis by increasing Bax and decreasing BCL2. The AN extract also elevated cell death by decreasing cell cycle regulators, such as cyclin D1, cyclin E1, cyclin dependent kinase 4 (CDK4), retinoblastoma (Rb), proliferation cell nuclear antigen (PCNA) and tumor suppressor p53, through increasing transcription factors, including Activator Protein -1 (AP-1) subunits (c-Jun/c-Fos), p21 and p16. AP-1 is a transcription factor involved in oncogenesis. These findings suggested that AN regulated AP-1 signaling of KB cells for oral carcinogenesis [92]. Arecoline is the major compound in AN to initiate the oral submucous fibrosis process [93]. It can induce reactive oxygen species, cell cycle arrest, apoptosis and DNA damage [94]. Arecoline-induced oral pathologies are explained by the following mechanisms: a suppression of p53 for DNA repair and increased reactive oxygen species (ROS) for epithelial-mesenchymal transition (EMT) and metastasis through mitogen-activated protein kinase (MAPK), phosphatidylinositol-3-kinase (PI3K)/Akt and nuclear factor kappa-light chain enhancer of activated B-cells (NF-κB) and PKC–protein kinase C (PKC) pathways [95]. It alters the expression of several inflammatory cytokines, such as serum amyloid A1, interleukin-6, IL-36G, chemokine CCL2 and CCL20, for epithelial-mesenchymal transformation (EMT) in oral squamous cell carcinoma cells. Moreover, arecoline enhanced cervical lymph node metastasis of tongue xenografted models using nude mice. These results also suggested that arecoline induces EMT and promotes metastasis of oral cancer [96]. The phosphodiesterase (PDE)/cAMP/Epac/C-EBP-β signal cascade regulates renal fibrosis in renal tubular epithelial cells [97]. Transforming growth factor-β (TGF-β) exerts pathological effects on organ fibrosis. Arecoline enhanced phosphodiesterase 4A activity. Silence phosphodiesterase 4A reversed the effects of TGF-β-induced fibrotic changes caused by arecoline. These findings indicated that arecoline promoted TGF-β1-induced buccal mucosal fibroblast activation through enhancing phosphodiesterase 4A activity during oral submucosal fibrosis [98]. Oxidative stress-caused DNA damage also leads to nuclear anomalies such as micronuclei. The levels of glutathione antioxidant enzymes, such as reductase and superoxide dismutase, were reduced, but the micronuclei count increased in buccal exfoliated cells of AN chewers. The detection of micronuclei in AN chewers can be a useful risk biomarker for oral cancer diagnosis [99]. However, there is no direct evidence of arecoline-induced carcinogenesis in animal models, possibly because of inappropriate treatment procedure or being caused by the biological differences between human and mice or rats [100].
4.2. Liver Diseases
Twelve of fifteen studies indicated that AN is a risk factor for various liver diseases like liver cirrhosis, hepatocellular carcinoma, complicating cirrhosis, liver fibrosis and chronic liver disease [4]. Most of these studies came from Taiwan, suggesting a high prevalence and many adverse effects of AN on liver in Taiwan. Moreover, five studies showed an increased risk of hepatocellular carcinoma and liver cirrhosis associated with an increased consumption of betel quid. Arecoline increased the proliferation, increased the migration ability of the HepG2 cells and upregulated the expression of PI3K-AKT pathway factors. These findings suggested that arecoline promoted migration and proliferation of human HepG2 cells by activating the PI3K/AKT/mTOR pathway [101]. Metabolic syndrome is related to liver fibrosis, cirrhosis and hepatocellular carcinoma. Patients of both metabolic syndrome and betel quid chewing had a higher risk of liver fibrosis than those with neither metabolic syndrome nor betel quid chewing. Betel quid chewing was reared with liver fibrosis in patients with metabolic syndrome but not in those of without metabolic syndrome [102]. In addition, betel nut chewing was associated with a high risk of liver fibrosis in subjects with nonalcoholic fatty liver disease [103].
4.3. Behavior and Addiction
AN alkaloid with psychoactive activity, including arecoline, arecaidine, guvacine and guvacoline, was reported to cause behavioral alterations in zebrafish [19]. They were reported to change behaviors, such as increasing the sense of well-being, stamina and euphoria. A review article described that AN chewing claimed to produce warm sensations of the human body, palpitation and heightened alertness and tolerance to hunger. All these neurological effects may be caused by the chemicals in AN that affect the autonomic nervous system at various levels [104]. Injected dichloromethane of AN into rat brain has antidepressant properties via monoamine oxidase-A inhibition [105]. Addiction is a significant public health menace. AN cessation is needed for the chewers. A total of 16 studies showed that the determinants influence the results of AN cessation including addiction of AN, withdrawal symptoms and sociocultural factors [106]. The majority of intervention method for AN cessation is behavior control. Behavior changing interventions were proven to be more effective [104]. However, pharmacological cessation therapies are also emerging for controlling this public health problem [11]. The results of a literature search related to betel nut use provided 139 references between the years 1970 and 2019 demonstrating a lack of up-to-date statistics on betel nut use. Therefore, appropriate policies, educational and cessation programs of helping to control betel nut applications are warranted [107].
4.4. Cardiac Diseases
Intraperitoneally injected arecoline in rats induced cardiac apoptosis through the Fas/Fas ligand pathway [108]. In addition, arecoline causes of cardiotoxicity and heart damage through pathways, such as induced IL-6-activated JAK2/STAT3, MEK5/ERK5 and mitogen-activated protein kinases (MAPK) pathways [109]. Intraperitoneally injected arecoline also caused cardiac fibrosis in rats through transforming growth factor-β (TGF-β)/Smad-mediated signaling pathways [110].
4.5. Gastric and Intestinal Diseases
Feeding raw AN extract developed gastric cancer in mice. Carcinogenesis was induced here by histone H3 epigenetic modifications and the Rb/E2F1 pathway [111]. The constituents of intestinal epithelial cells, such as total hexose, sialic acid, cholesterol and enzymes, including alkaline phosphatase, Ca2+-Mg2+-ATPase and sucrase, were decreased in rat administrated with AN extract by gastric intubation. These findings suggested that prolonged chewing of AN decreased intestinal epithelial cell lining functions [112].
4.6. Genotoxicity
Arecoline and its metabolites were reviewed to have genetic toxicology, such as gene mutations, DNA damage and repair endpoints as well as cytogenetic effects by in vitro and in vivo experiments [17].
4.7. Reproduction and Development
Eating AN before pregnancy was followed by a higher preterm birth rate in China [113]. The results of a systematic database review containing 15,270 women of 28 studies showed that prenatal betel nut use is associated with a low birth weight [114]. Arecoline caused oocyte meiosis arrest by disrupting spindle assembly, actin filament dynamics and kinetochore-microtubule attachment stability in mouse oocytes. In addition, arecoline reduced ATP level and increased oxidative stress by disturbing the distribution of mitochondria to induce oocyte apoptosis [115]. Myogenic differentiation is an important process during embryonic development. The inhibitory effects of myogenesis through the signal transducer and activator of transcription 3 (Stat3) pathway caused by arecoline was reviewed [116]. Fortunately, the arecoline induced defective effects of myogenesis, including decreased nuclei in each myotube and the number of myotubes, and the level of the myogenic markers, such as myosin heavy chain and myogenin, in C2C12 myoblast cells can be prevented by N-acetyl cysteine through an extracellular signal-regulated kinase (ERK) pathway [117].
4.8. Kidney Disease
The analysis of 43,636 men from Taiwan Biobank showed that a high risk of kidney stone disease was found in subjects with betel nut chewing habits. Moreover, those who chewed more than 30 quids betel nut daily have more than 1.5-fold possibility to get a kidney stone [118]. Arecoline increased cell migration and the expression of epithelial mesenchymal transition and fibrogenesis markers, such as N-cadherin, vimentin, α-SMA and collagen but decreased another epithelial mesenchymal transition marker, E-cadherin, in human kidney (HK2) cells. The above results demonstrated that arecoline played an important role in inducing EMT and fibrogenesis of renal tubule cells for promoting the progression of chronic kidney disease [119].
4.9. Neuron Activation
Arecoline induced activation of nicotinic acetylcholine receptors (nAChR) in Xenopus oocytes expressing either human α4 and β2 nAChR subunits or α7 subunits. Arecoline analogs, isoarecolone and 1-(4-methylpiperazin-1-yl) ethanone, had the α4 nAChR selectivity without muscarinic activity. These finding indicated that α4 nAChR is at the root of nicotine addiction for betel nut addiction [120].
4.10. Abortifacient
AN was reported to have an abortifacient effect in rats and is one of the herbs considered unsafe for use during pregnancy [121].
In summary of Section 4, AN extracts, other mixture compounds and arecoline have several toxicological effects that induce or promote different diseases. Diseases associated with toxicological effects of AN are listed in Table 3.
Table 3.
The toxicological effects and mechanisms of AN extract, other mixture compounds and arecoline resulting in several diseases.
| Diseases | Toxicological Effects | AN Components | Mechanism | Biological Materials | References |
|---|---|---|---|---|---|
| Oral | dysregulating proinflammatory cytokines | AN | ↓ TGF-β | human | [89] |
| low prevalence of oral submucous fibrosis | human | [90] | |||
| deteriorate prognosis of oral cancer | AN | human | [91] | ||
| oral carcinogenesis | 0.35% AN extract | ↑ AP-1, apoptotic gnes, ↓ cell cycle regulators |
KB cells | [92] | |
| initiate oral submucous fibrosis process | Arecoline | human | [93] | ||
| reactive oxygen species ↑, cell cycle arrest, apoptosis ↓, DNA damage | 800 μg/mL Arecoline | OSCC cells | [94] | ||
| induce oral pathologies mechanisms | Arecoline | ↑ MAPK, PI3K Akt, NF-κB, PKC pathways | [95] | ||
| promote metastasis of oral cancer | 160 μg/mL Arecoline | cytokines for EMT | CAL33 and UM2 cells | [96] | |
| promoted buccal mucosal fibroblasts | 20 and 50 µg/mL Arecoline | ↑ TGF-β | buccal mucosal fibroblasts | [98] | |
| increased oxidative stress | AN | ↓ reductase, superoxide and dismutase, ↑ micronuclei in buccal exfoliated cells |
human | [99] | |
| carcinogenesis not found in animal models | Arecoline | [100] | |||
| Liver | risk factor for various liver diseases | AN | human | [4] | |
| promoting migration and proliferation of human HepG2 cells | 2.5 µM Arecoline | ↑ PI3K-AKT pathway | HepG2 cells | [101] | |
| associated with liver fibrosis | AN | human | [102] | ||
| high risk of liver fibrosis | AN | human | [103] | ||
| Behavior and Addiction | behavioral alterations | 0.001, 0.01, 0.1, or 1 ppm four alkaloid | zebrafish | [19] | |
| increase sense of well-being, stamina, and euphoria | AN | human | [104] | ||
| antidepressant | 10 mg/kg Dichloromethane fraction | ↓ monoamine oxidase-A | rat brain | [105] | |
| addiction of AN | AN | human | [106] | ||
| methods for AN cessation | AN | human | [104] | ||
| emerging pharmacological cessation therapies | AN | human | [11] | ||
| Cardiac | cardiac apoptosis | 5 and 50 mg/kg/day arecoline | ↑ Fas/Fas ligand pathway | rats | [108] |
| cardiotoxicity and heart damage | 5 and 50 mg/kg/day arecoline | ↑ JAK2/STAT3, MEK5/ERK5, MAPK pathways | rats | [109] | |
| cardiac fibrosis | 5 and 50 mg/kg/day arecoline | ↑ TGF-β)/Smad pathways | rats | [110] | |
| Gastric and intestinal | developed gastric cancer | 1 mg raw areca nutt | ↑ histone H3 epigenetic modifications and Rb/E2F1 pathway | mice | [111] |
| decreases intestinal epithelial cell lining functions | AN extract | ↓ membrane constituents | rats | [112] | |
| Genotoxicity | genetic toxicology | Arecoline | ↑ gene mutations, DNA damage and repair, cytogenetic effects | in vitro, in vivo | [17] |
| Reproduction and development | risk factor for preterm birth | AN | ↑ oxidative stress | human | [113] |
| low birth weight | AN | human | [114] | ||
| inducing oocyte apoptosis | 160, 180 and 200 μg/mL arecoline | ↑ oxidative stress | mouse oocytes | [115] | |
| Inhibiting myogenesis | 0.04 and 0.08 mM arecoline | ↓ pStat3 | C2C12 cells | [116] | |
| defective effects of myogenesis | 0.04 and 0.08 mM arecoline | ↓ myosin heavy chain, myogenin | C2C12 cells | [117] | |
| Kidney | high risk of kidney stone disease | >30 AN quids | human | [118] | |
| promoting the progression of chronic kidney disease | 10, 20, or 40 μg/mL arecoline | ↑ EMT genes, fibrogenesis markers | HK2 cells | [119] | |
| Neuron activation | nicotine addiction | 100 µM four areca alkaloid | ↑ nAChR | Xenopus oocytes | [120] |
| Abortifacient | may have abortifacient effects | AN | human | [121] |
↑: Activated, ↓: Inhibited, AN: partial or whole AN chewing.
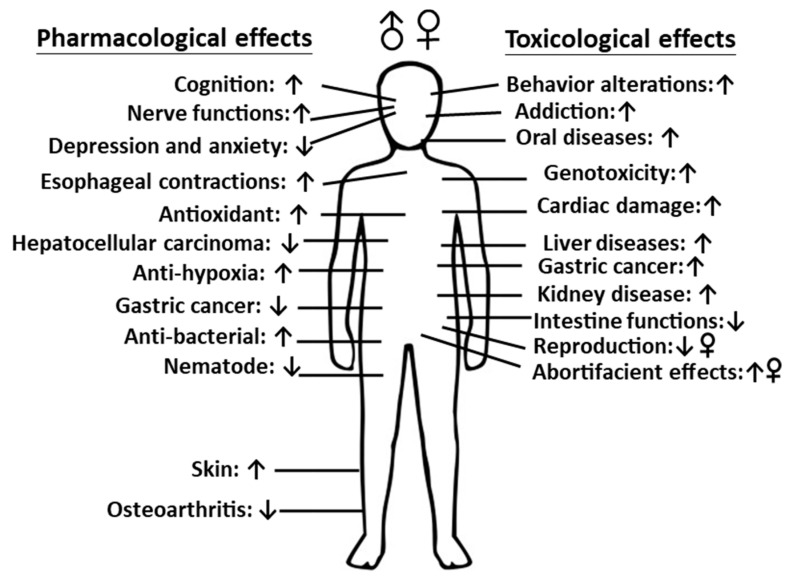
Overall, both pharmacological and toxicological effects of AN extracts, additions of combined compounds and arecoline are presented in Figure 4. Both males and a females have the same effects, except reproduction. This figure does not indicate any gender. This figure is hand-drawn using Inkscape (https://inkscape.org/zh-hant/, accessed on 8 May 2023 to 10 May 2023).
Figure 4.
The distribution of pharmacological and toxicological effects of AN extract, combined compounds and arecoline in the human body. The effects in other animals, such as mice, rats, porcine, zebrafish, C. elegans, chickens and buffaloes were included from different sources cited in this review. ↑: Activated, ↓: Inhibited.
5. Perspectives
By comparing the effects of AN components, we found that most of the components are useful in medicinal applications for a diverse array of ailments. However, there are also some toxic compounds, such as arecoline, arecaidine and guvacine, that induce toxicological effects related to carcinogenicity, increased DNA breaks and oxidative stress. In the future, removing or modifying the toxic ingredients of AN extractions may improve their pharmacological effects, which may effectively and safely treat several diseases such as cancer or speed up drug development.
One of the purposes of this review was to find out the trends and challenges of AN from recent published reports. We found several trends and perspectives. (1) One of the common functions of AN in Traditional Chinese Medicine is its anthelmintic function. However, research on anthelmintic efficacy for human was cautioned recently, due to concerns about its accompanying toxicity. However, the anthelmintic efficacy on other animals, such as chicken and buffaloes [84,85] was promising, suggesting that the toxicity of AN may also be tolerated by us. (2) Several articles studied the behavior and addiction/cessation [11,19,104,105,106], indicating one of the trends of AN research. Since arecoline activates muscarin M1 receptors, reports about the effect of behavior and addiction by AN [75,76,77] provide accelerating evidence. (3) Most articles were studying oral diseases [89,90,91,92,93,94,95,96,98,99,100]. The effects of AN on other organs, such as the liver, heart and kidney, and cells such as myoblasts [116] were recently published, indicating more diverse effects of AN than hitherto expected. (4) Several toxic materials also serve as medicine, indicating the potential of toxins as medicines, such as arecoline. Arecoline is a well-known toxin. It causes anxiolytic behavior in zebrafish [73]. To remove its toxicity by modifying its structure may be applied to animal models and hopefully to humans in the near future. The structures of four areca alkaloids are similar but have a different extent of anxiolytic-like effects in zebrafish. Therefore, the structural modification of toxic materials from AN by chemistry may develop new potential for clinical applications. (5) Volatile components of AN also provide antimicrobial effects [81]. Volatile compounds have the potential for development in fragrances for cosmetic, pharmaceutical or clinical applications. (6) Social interaction causes the popular usage of AN. It will be a great challenge to change the public opinion about the toxicological risks of AN usage. Therefore, information and education about the harmful effects of AN consumption are warranted.
6. Conclusions
AN is used for traditional herbal medicine and social activities in several countries. It was used as early as about the years A.D. 25-220 in China. The traditional usage of AN was applied for treating several diseases including malaria, diarrhea, ascariasis, edema, stagnation of food, arthritis and beriberi. However, it was also reported to have toxicological effects. Knowing the effects of single compounds as well as their synergistic interactions will be helpful for realizing the functions of AN. Effective components of AN include among others alkaloids, flavonoids, tannins, triterpenoids, fatty acids and steroids. The pharmacological effects of AN include curing gastric and intestinal diseases, depression and anxiety, liver diseases, nerve diseases, bacterial infections, skin protection, anthelmintic efficacy, antioxidant, anti-hypoxia and anti-osteoarthritis. The toxicological effects of AN include oral diseases, liver diseases, behavior and addiction, cardiac diseases, gastric and intestinal diseases, genotoxicity, reproduction and development, kidney disease, neuron and abortifacient. This article summarizes components of AN and their pharmacological and toxicological effects in several diseases. In the future, removing or modifying the toxic ingredients of AN extractions may improve their pharmacological effects, which may effectively and safely treat several diseases such as cancer cells or speed up the development of drugs. Moreover, AN extracts may be applied in veterinary medicine such as poultry as an anthelmintic agent, and the volatile components may be developed as fragrances. The effects of AN on organs and the oral cavity are emerging. Further studies will explore the effects of AN on animals. A better understanding of the mechanism of arecoline on the activation of muscarinic M1 receptors may provide mechanisms of action for behavior and addiction problems caused by AN. Finally, to modify the structure of toxic components by synthetic chemistry will be helpful for many of the above purposes.
Acknowledgments
We thank Hans-Uwe Dahms at Kaohsiung Medical University (Taiwan) for corrections of the written English.
Author Contributions
Conceptualization, P.-F.L. and Y.-F.C.; Writing—Original Draft Preparation, P.-F.L. and Y.-F.C.; Writing—Review and Editing, P.-F.L. and Y.-F.C.; Funding Acquisition, Y.-F.C. All authors have read and agreed to the published version of the manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
Funding Statement
We acknowledge the funding provided by the National Science and Technology Council (108-2314-B-037-075; 109-2320-B-037-015-MY3) and the Kaohsiung Medical University Research Foundation (KMU-M103001, KMU-M104003, KMU-TP104PR16).
Footnotes
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
References
- 1.Singh A., Dikshit R., Chaturvedi P. Betel Nut Use: The South Asian Story. Subst. Use Misuse. 2020;55:1545–1551. doi: 10.1080/10826084.2020.1753772. [DOI] [PubMed] [Google Scholar]
- 2.Ghani W.M.N., Razak I.A., Doss J.G., Yang Y.H., Rahman Z.A.A., Ismail S.M., Abraham M.T., Wan Mustafa W.M., Tay K.K., Zain R.B. Multi-ethnic variations in the practice of oral cancer risk habits in a developing country. Oral Dis. 2019;25:447–455. doi: 10.1111/odi.12995. [DOI] [PubMed] [Google Scholar]
- 3.Warnakulasuriya S., Chen T.H.H. Areca Nut and Oral Cancer: Evidence from Studies Conducted in Humans. J. Dent. Res. 2022;101:1139–1146. doi: 10.1177/00220345221092751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Khasbage S.B.D., Bhowate R.R., Khatib N. Risk of liver disease in areca nut habitual: A systematic review. J. Oral. Maxillofac. Pathol. 2022;26:128–129. doi: 10.4103/jomfp.jomfp_345_21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Tami-Maury I., Nethan S., Feng J., Miao H., Delclos G., Mehrotra R. Evidence of areca nut consumption in the United States mainland: A cross-sectional study. BMC Public Health. 2022;22:912. doi: 10.1186/s12889-022-13262-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chen X., He Y., Deng Y. Chemical Composition, Pharmacological, and Toxicological Effects of Betel Nut. Evid. Based Complement. Altern. Med. 2021;2021:1808081. doi: 10.1155/2021/1808081. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 7.Salehi B., Konovalov D.A., Fru P., Kapewangolo P., Peron G., Ksenija M.S., Cardoso S.M., Pereira O.R., Nigam M., Nicola S., et al. Areca catechu-From farm to food and biomedical applications. Phytother. Res. 2020;34:2140–2158. doi: 10.1002/ptr.6665. [DOI] [PubMed] [Google Scholar]
- 8.Garg A., Chaturvedi P., Gupta P.C. A review of the systemic adverse effects of areca nut or betel nut. Indian. J. Med. Paediatr. Oncol. 2014;35:3–9. doi: 10.4103/0971-5851.133702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Arjungi K.N. Areca nut: A review. Arzneimittelforschung. 1976;26:951–956. [PubMed] [Google Scholar]
- 10.Senevirathna K., Pradeep R., Jayasinghe Y.A., Jayawickrama S.M., Illeperuma R., Warnakulasuriya S., Jayasinghe R.D. Carcinogenic Effects of Areca Nut and Its Metabolites: A Review of the Experimental Evidence. Clin. Pract. 2023;13:326–346. doi: 10.3390/clinpract13020030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Sumithrarachchi S.R., Jayasinghe R., Warnakulasuriya S. Betel Quid Addiction: A Review of Its Addiction Mechanisms and Pharmacological Management as an Emerging Modality for Habit Cessation. Subst. Use Misuse. 2021;56:2017–2025. doi: 10.1080/10826084.2021.1963990. [DOI] [PubMed] [Google Scholar]
- 12.Dhingra K., Jhanjee S. A Review of Intervention Strategies for Areca Nut Use Cessation. Indian. J. Psychol. Med. 2023;45:117–123. doi: 10.1177/02537176221144711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Muthukumaran R.B., Bhattacharjee P., Bhowmick P., Zote L., Malsawmtluangi, Kumar N.S., Jahau L., Cooke M.S., Hu C.W., Chao M.R. Genetic and epigenetic instability induced by betel quid associated chemicals. Toxicol. Rep. 2023;10:223–234. doi: 10.1016/j.toxrep.2023.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Gayathri K., Malathi N., Gayathri V., Adtani P.N., Ranganathan K. Molecular pathways of oral submucous fibrosis and its progression to malignancy. Arch. Oral. Biol. 2023;148:105644. doi: 10.1016/j.archoralbio.2023.105644. [DOI] [PubMed] [Google Scholar]
- 15.Chuang H.C., Tsai M.H., Lin Y.T., Chou M.H., Yang K.L., Chien C.Y. Systemic and Local Effects Among Patients With Betel Quid-Related Oral Cancer. Technol. Cancer Res. Treat. 2022;21:15330338221146870. doi: 10.1177/15330338221146870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Pasupuleti R.R., Lee C.H., Osborne P.G., Wu M.T., Ponnusamy V.K. Rapid green analytical methodology for simultaneous biomonitoring of five toxic areca nut alkaloids using UHPLC-MS/MS for predicting health hazardous risks. J. Hazard. Mater. 2022;422:126923. doi: 10.1016/j.jhazmat.2021.126923. [DOI] [PubMed] [Google Scholar]
- 17.Oliveira N.G., Ramos D.L., Dinis-Oliveira R.J. Genetic toxicology and toxicokinetics of arecoline and related areca nut compounds: An updated review. Arch. Toxicol. 2021;95:375–393. doi: 10.1007/s00204-020-02926-9. [DOI] [PubMed] [Google Scholar]
- 18.Zhang P., Chua N.Q.E., Dang S., Davis A., Chong K.W., Prime S.S., Cirillo N. Molecular Mechanisms of Malignant Transformation of Oral Submucous Fibrosis by Different Betel Quid Constituents-Does Fibroblast Senescence Play a Role? Int. J. Mol. Sci. 2022;23:1637. doi: 10.3390/ijms23031637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Siregar P., Audira G., Castillo A.L., Roldan M.J.M., Suryanto M.E., Liu R.X., Lin Y.T., Lai Y.H., Hsiao C.D. Comparison of the psychoactive activity of four primary Areca nut alkaloids in zebrafish by behavioral approach and molecular docking. Biomed. Pharmacother. 2022;155:113809. doi: 10.1016/j.biopha.2022.113809. [DOI] [PubMed] [Google Scholar]
- 20.Zhang W., Lin H., Zou M., Yuan Q., Huang Z., Pan X., Zhang W. Nicotine in Inflammatory Diseases: Anti-Inflammatory and Pro-Inflammatory Effects. Front. Immunol. 2022;13:826889. doi: 10.3389/fimmu.2022.826889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Melo M.R., Gasparini S., Speretta G.F., Silva E.F., Rodrigues Pedrino G., Menani J.V., Zoccal D.B., Colombari D.S.A., Colombari E. Importance of the commissural nucleus of the solitary tract in renovascular hypertension. Hypertens. Res. 2019;42:587–597. doi: 10.1038/s41440-018-0190-6. [DOI] [PubMed] [Google Scholar]
- 22.Wen K., Fang X., Yang J., Yao Y., Nandakumar K.S., Salem M.L., Cheng K. Recent Research on Flavonoids and their Biomedical Applications. Curr. Med. Chem. 2021;28:1042–1066. doi: 10.2174/0929867327666200713184138. [DOI] [PubMed] [Google Scholar]
- 23.Gong G., Guan Y.Y., Zhang Z.L., Rahman K., Wang S.J., Zhou S., Luan X., Zhang H. Isorhamnetin: A review of pharmacological effects. Biomed. Pharmacother. 2020;128:110301. doi: 10.1016/j.biopha.2020.110301. [DOI] [PubMed] [Google Scholar]
- 24.Aboulaghras S., Sahib N., Bakrim S., Benali T., Charfi S., Guaouguaou F.E., Omari N.E., Gallo M., Montesano D., Zengin G., et al. Health Benefits and Pharmacological Aspects of Chrysoeriol. Pharmaceuticals. 2022;15:973. doi: 10.3390/ph15080973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Muruganathan N., Dhanapal A.R., Baskar V., Muthuramalingam P., Selvaraj D., Aara H., Shiek Abdullah M.Z., Sivanesan I. Recent Updates on Source, Biosynthesis, and Therapeutic Potential of Natural Flavonoid Luteolin: A Review. Metabolites. 2022;12:1145. doi: 10.3390/metabo12111145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Azeem M., Hanif M., Mahmood K., Ameer N., Chughtai F.R.S., Abid U. An insight into anticancer, antioxidant, antimicrobial, antidiabetic and anti-inflammatory effects of quercetin: A review. Polym. Bull. 2022;80:241–262. doi: 10.1007/s00289-022-04091-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Segura-Villalobos D., Roa-Velázquez D., Zavala-Vargas D.I., Filisola-Villaseñor J.G., Castillo Arellano J.I., Morales Ríos E., Reyes-Chilpa R., González-Espinosa C. Jacareubin inhibits TLR4-induced lung inflammatory response caused by the RBD domain of SARS-CoV-2 Spike protein. Pharmacol. Rep. 2022;74:1315–1325. doi: 10.1007/s43440-022-00398-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Ramalingam M., Kim H., Lee Y., Lee Y.I. Phytochemical and Pharmacological Role of Liquiritigenin and Isoliquiritigenin From Radix Glycyrrhizae in Human Health and Disease Models. Front. Aging Neurosci. 2018;10:348. doi: 10.3389/fnagi.2018.00348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Yuan M., Ao Y., Yao N., Xie J., Zhang D., Zhang J., Zhang X., Ye W. Two New Flavonoids from the Nuts of Areca catechu. Molecules. 2019;24:2862. doi: 10.3390/molecules24162862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Maugeri A., Lombardo G.E., Cirmi S., Süntar I., Barreca D., Laganà G., Navarra M. Pharmacology and toxicology of tannins. Arch. Toxicol. 2022;96:1257–1277. doi: 10.1007/s00204-022-03250-0. [DOI] [PubMed] [Google Scholar]
- 31.Musial C., Kuban-Jankowska A., Gorska-Ponikowska M. Beneficial Properties of Green Tea Catechins. Int. J. Mol. Sci. 2020;21:1744. doi: 10.3390/ijms21051744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Josiah S.S., Famusiwa C.D., Crown O.O., Lawal A.O., Olaleye M.T., Akindahunsi A.A., Akinmoladun A.C. Neuroprotective effects of catechin and quercetin in experimental Parkinsonism through modulation of dopamine metabolism and expression of IL-1β, TNF-α, NF-κB, IκKB, and p53 genes in male Wistar rats. Neurotoxicology. 2022;90:158–171. doi: 10.1016/j.neuro.2022.03.004. [DOI] [PubMed] [Google Scholar]
- 33.Chen H., Wang W., Yu S., Wang H., Tian Z., Zhu S. Procyanidins and Their Therapeutic Potential against Oral Diseases. Molecules. 2022;27:2932. doi: 10.3390/molecules27092932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Wei Z., Chen J., Zuo F., Guo J., Sun X., Liu D., Liu C. Traditional Chinese Medicine has great potential as candidate drugs for lung cancer: A review. J. Ethnopharmacol. 2023;300:115748. doi: 10.1016/j.jep.2022.115748. [DOI] [PubMed] [Google Scholar]
- 35.Muilu-Mäkelä R., Aapola U., Tienaho J., Uusitalo H., Sarjala T. Antibacterial and Oxidative Stress-Protective Effects of Five Monoterpenes from Softwood. Molecules. 2022;27:3891. doi: 10.3390/molecules27123891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Tong H., Yu M., Fei C., Ji D., Dong J., Su L., Gu W., Mao C., Li L., Bian Z., et al. Bioactive constituents and the molecular mechanism of Curcumae Rhizoma in the treatment of primary dysmenorrhea based on network pharmacology and molecular docking. Phytomedicine. 2021;86:153558. doi: 10.1016/j.phymed.2021.153558. [DOI] [PubMed] [Google Scholar]
- 37.Luo Y., Jiang Y., Chen L., Li C., Wang Y. Applications of protein engineering in the microbial synthesis of plant triterpenoids. Synth. Syst. Biotechnol. 2023;8:20–32. doi: 10.1016/j.synbio.2022.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Panda S.S., Thangaraju M., Lokeshwar B.L. Ursolic Acid Analogs as Potential Therapeutics for Cancer. Molecules. 2022;27:8981. doi: 10.3390/molecules27248981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.AlQathama A., Shao L., Bader A., Khondkar P., Gibbons S., Prieto J.M. Differential Anti-Proliferative and Anti-Migratory Activities of Ursolic Acid, 3-O-Acetylursolic Acid and Their Combination Treatments with Quercetin on Melanoma Cells. Biomolecules. 2020;10:894. doi: 10.3390/biom10060894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Dembitsky V.M. In Silico Prediction of Steroids and Triterpenoids as Potential Regulators of Lipid Metabolism. Mar. Drugs. 2021;19:650. doi: 10.3390/md19110650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Gonyela O., Peter X., Dewar J.B., van der Westhuyzen C., Steenkamp P., Fouche G. Cycloartanol and Sutherlandioside C peracetate from Sutherlandia frutescens and their immune potentiating effects. Nat. Prod. Res. 2021;35:1968–1976. doi: 10.1080/14786419.2019.1648457. [DOI] [PubMed] [Google Scholar]
- 42.Niu H., Li X., Yang A., Jin Z., Wang X., Wang Q., Yu C., Wei Z., Dou C. Cycloartenol exerts anti-proliferative effects on Glioma U87 cells via induction of cell cycle arrest and p38 MAPK-mediated apoptosis. JBUON. 2018;23:1840–1845. [PubMed] [Google Scholar]
- 43.Noor F., Rehman A., Ashfaq U.A., Saleem M.H., Okla M.K., Al-Hashimi A., AbdElgawad H., Aslam S. Integrating Network Pharmacology and Molecular Docking Approaches to Decipher the Multi-Target Pharmacological Mechanism of Abrus precatorius L. Acting on Diabetes. Pharmaceuticals. 2022;15:414. doi: 10.3390/ph15040414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Khan Z., Nath N., Rauf A., Emran T.B., Mitra S., Islam F., Chandran D., Barua J., Khandaker M.U., Idris A.M., et al. Multifunctional roles and pharmacological potential of β-sitosterol: Emerging evidence toward clinical applications. Chem. Biol. Interact. 2022;365:110117. doi: 10.1016/j.cbi.2022.110117. [DOI] [PubMed] [Google Scholar]
- 45.Custers, Emma E.M., Kiliaan, Amanda J. Dietary lipids from body to brain. Prog. Lipid Res. 2022;85:101144. doi: 10.1016/j.plipres.2021.101144. [DOI] [PubMed] [Google Scholar]
- 46.Zheng C., Xiao G., Yan X., Qiu T., Liu S., Ou J., Cen M., Gong L., Shi J., Zhang H. Complex of Lauric Acid Monoglyceride and Cinnamaldehyde Ameliorated Subclinical Necrotic Enteritis in Yellow-Feathered Broilers by Regulating Gut Morphology, Barrier, Inflammation and Serum Biochemistry. Animals. 2023;13:516. doi: 10.3390/ani13030516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Alonso-Castro A.J., Serrano-Vega R., Pérez Gutiérrez S., Isiordia-Espinoza M.A., Solorio-Alvarado C.R. Myristic acid reduces skin inflammation and nociception. J. Food Biochem. 2022;46:e14013. doi: 10.1111/jfbc.14013. [DOI] [PubMed] [Google Scholar]
- 48.Yu X., Peng W., Wang Y., Xu W., Chen W., Huang L., Xu H., He X., Wang S., Sun Q., et al. Palmitic Acid Inhibits the Growth and Metastasis of Gastric Cancer by Blocking the STAT3 Signaling Pathway. Cancers. 2023;15:388. doi: 10.3390/cancers15020388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Kim H.S., Yoo H.J., Lee K.M., Song H.E., Kim S.J., Lee J.O., Hwang J.J., Song J.W. Stearic acid attenuates profibrotic signalling in idiopathic pulmonary fibrosis. Respirology. 2021;26:255–263. doi: 10.1111/resp.13949. [DOI] [PubMed] [Google Scholar]
- 50.Rakoczy K., Szlasa W., Saczko J., Kulbacka J. Therapeutic role of vanillin receptors in cancer. Adv. Clin. Exp. Med. 2021;30:1293–1301. doi: 10.17219/acem/139398. [DOI] [PubMed] [Google Scholar]
- 51.Dos Santos Negreiros P., da Costa D.S., da Silva V.G., de Carvalho Lima I.B., Nunes D.B., de Melo Sousa F.B., de Souza Lopes Araújo T., Medeiros J.V.R., Dos Santos R.F., de Cássia Meneses Oliveira R. Antidiarrheal activity of α-terpineol in mice. Biomed. Pharmacother. 2019;110:631–640. doi: 10.1016/j.biopha.2018.11.131. [DOI] [PubMed] [Google Scholar]
- 52.You T., Yuan S., Bai L., Zhang X., Chen P., Zhang W. Benzyl alcohol accelerates recovery from Achilles tendon injury, potentially via TGF-β1/Smad2/3 pathway. Injury. 2020;51:1515–1521. doi: 10.1016/j.injury.2020.03.058. [DOI] [PubMed] [Google Scholar]
- 53.Shekhar N., Tyagi S., Rani S., Thakur A.K. Potential of Capric Acid in Neurological Disorders: An Overview. Neurochem. Res. 2023;48:697–712. doi: 10.1007/s11064-022-03809-4. [DOI] [PubMed] [Google Scholar]
- 54.Tian B., Liu J. Resveratrol: A review of plant sources, synthesis, stability, modification and food application. J. Sci. Food Agric. 2020;100:1392–1404. doi: 10.1002/jsfa.10152. [DOI] [PubMed] [Google Scholar]
- 55.Ahmad S., Sayeed S., Bano N., Sheikh K., Raza K. In-silico analysis reveals Quinic acid as a multitargeted inhibitor against Cervical Cancer. J. Biomol. Struct. Dyn. 2022;15:1–17. doi: 10.1080/07391102.2022.2146202. [DOI] [PubMed] [Google Scholar]
- 56.Lu L., Zhao Y., Yi G., Li M., Liao L., Yang C., Cho C., Zhang B., Zhu J., Zou K., et al. Quinic acid: A potential antibiofilm agent against clinical resistant Pseudomonas aeruginosa. Chin. Med. 2021;16:72. doi: 10.1186/s13020-021-00481-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Liu Y., Muema F.W., Zhang Y.L., Guo M.Q. Acyl Quinic Acid Derivatives Screened Out from Carissa spinarum by SOD-Affinity Ultrafiltration LC-MS and Their Antioxidative and Hepatoprotective Activities. Antioxidants. 2021;10:1302. doi: 10.3390/antiox10081302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Benguechoua M.I., Benarous K., Benahmed Z., Boukhalkhal S., Silva A.M.S., Yousfi M. Quinic and Digallic acids from Pistacia atlantica Desf. Leaves Extracts as Potent Dual Effect Inhibitors against main Protease and RNA-dependent RNA Polymerase of SARS-CoV-2. Curr. Comput. Aided Drug. Des. 2022;18:307–317. doi: 10.2174/1573409918666220616121449. [DOI] [PubMed] [Google Scholar]
- 59.Xie L., Tang H., Song J., Long J., Zhang L., Li X. Chrysophanol: A review of its pharmacology, toxicity and pharmacokinetics. J. Pharm. Pharmacol. 2019;71:1475–1487. doi: 10.1111/jphp.13143. [DOI] [PubMed] [Google Scholar]
- 60.Li X., Liu Y., Chu S., Yang S., Peng Y., Ren S., Wen B., Chen N. Physcion and physcion 8-O-β-glucopyranoside: A review of their pharmacology, toxicities and pharmacokinetics. Chem. Biol. Interact. 2019;310:108722. doi: 10.1016/j.cbi.2019.06.035. [DOI] [PubMed] [Google Scholar]
- 61.Adnan M., Rasul A., Hussain G., Shah M.A., Sarfraz I., Nageen B., Riaz A., Khalid R., Asrar M., Selamoglu Z., et al. Physcion and Physcion 8-O-β-D-glucopyranoside: Natural Anthraquinones with Potential Anticancer Activities. Curr. Drug. Targets. 2021;22:488–504. doi: 10.2174/1389450121999201013154542. [DOI] [PubMed] [Google Scholar]
- 62.Fan X., Song H., Xu X., Lu X., Wang Y., Duan X. Subchronic Oral Toxicity of Sodium p-Hydroxybenzoate in Sprague-Dawley Rats. Front. Pharmacol. 2022;13:843368. doi: 10.3389/fphar.2022.843368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Nguyen Thien T.V., Vo T.K.L., Dang P.H., Huynh N.V., Ngo T.T.D., Nguyen T.M.N., Hansen P.E., Ton That Q. Two new sesquiterpenes from the stems of Miliusa velutina. Nat. Prod. Res. 2022;36:553–559. doi: 10.1080/14786419.2020.1789984. [DOI] [PubMed] [Google Scholar]
- 64.Song J., He Y., Luo C., Feng B., Ran F., Xu H., Ci Z., Xu R., Han L., Zhang D. New progress in the pharmacology of protocatechuic acid: A compound ingested in daily foods and herbs frequently and heavily. Pharmacol. Res. 2020;161:105109. doi: 10.1016/j.phrs.2020.105109. [DOI] [PubMed] [Google Scholar]
- 65.Zhang R., Hu X., Zhang B., Wang Z., Hao C., Xin J., Guo Q. Whitening Activity of Constituents Isolated from the Trichosanthes Pulp. Evid. Based Complement. Altern. Med. 2020;2020:2582579. doi: 10.1155/2020/2582579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Choi J.H., Kim S. In Vitro Antithrombotic, Hematological Toxicity, and Inhibitor Studies of Protocatechuic, Isovanillic, and p-Hydroxybenzoic Acids from Maclura tricuspidata (Carr.) Bur. Molecules. 2022;27:3496. doi: 10.3390/molecules27113496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Li D., Rui Y.X., Guo S.D., Luan F., Liu R., Zeng N. Ferulic acid: A review of its pharmacology, pharmacokinetics and derivatives. Life Sci. 2021;284:119921. doi: 10.1016/j.lfs.2021.119921. [DOI] [PubMed] [Google Scholar]
- 68.Shekari S., Khonsha F., Rahmati-Yamchi M., Nejabati H.R., Mota A. Vanillic Acid and Non-Alcoholic Fatty Liver Disease: A Focus on AMPK in Adipose and Liver Tissues. Curr. Pharm. Des. 2021;27:4686–4692. doi: 10.2174/1381612827666210701145438. [DOI] [PubMed] [Google Scholar]
- 69.Yi S., Zou L., Li Z., Sakao K., Wang Y., Hou D.X. In Vitro Antioxidant Activity of Areca Nut Polyphenol Extracts on RAW264.7 Cells. Foods. 2022;11:3607. doi: 10.3390/foods11223607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Cao M., Yuan H., Daniyal M., Yu H., Xie Q., Liu Y., Li B., Jian Y., Peng C., Tan D., et al. Two new alkaloids isolated from traditional Chinese medicine Binglang the fruit of Areca catechu. Fitoterapia. 2019;138:104276. doi: 10.1016/j.fitote.2019.104276. [DOI] [PubMed] [Google Scholar]
- 71.Tey S.L., Li C.Y., Lin L.W., Chang L.C., Chen Y.L., Chang F.R., Yang S.N., Tsai C.C. Arecae pericarpium extract induces porcine lower-esophageal-sphincter contraction via muscarinic receptors. BMC Complement. Med. Ther. 2021;21:275. doi: 10.1186/s12906-021-03442-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Yang Y., Huang H., Cui Z., Chu J., Du G. UPLC-MS/MS and Network Pharmacology-Based Analysis of Bioactive Anti-Depression Compounds in Betel Nut. Drug. Des. Devel Ther. 2021;15:4827–4836. doi: 10.2147/DDDT.S335312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Serikuly N., Alpyshov E.T., Wang D., Wang J., Yang L., Hu G., Yan D., Demin K.A., Kolesnikova T.O., Galstyan D., et al. Effects of acute and chronic arecoline in adult zebrafish: Anxiolytic-like activity, elevated brain monoamines and the potential role of microglia. Prog. Neuropsychopharmacol. Biol. Psychiatry. 2021;104:109977. doi: 10.1016/j.pnpbp.2020.109977. [DOI] [PubMed] [Google Scholar]
- 74.Wei P.L., Hung C.S., Lu H.H., Batzorig U., Huang C.Y., Chang Y.J. Areca nut extract (ANE) inhibits the progression of hepatocellular carcinoma cells via activation of ROS production and activation of autophagy. Int. J. Med. Sci. 2021;18:3452–3462. doi: 10.7150/ijms.61570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Lan Q., Guan P., Huang C., Huang S., Zhou P., Zhang C. Arecoline Induces an Excitatory Response in Ventral Tegmental Area Dopaminergic Neurons in Anesthetized Rats. Front. Pharmacol. 2022;13:872212. doi: 10.3389/fphar.2022.872212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Ching T.T., Chen Y.C., Li G., Liu J., Xu X.Z.S., Hsu A.L. Short-term enhancement of motor neuron synaptic exocytosis during early aging extends lifespan in Caenorhabditis elegans. Exp. Biol. Med. 2020;245:1552–1559. doi: 10.1177/1535370220950639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Brunetti P., Lo Faro A.F., Tini A., Busardò F.P., Carlier J. Pharmacology of Herbal Sexual Enhancers: A Review of Psychiatric and Neurological Adverse Effects. Pharmaceuticals. 2020;13:309. doi: 10.3390/ph13100309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Jam N., Hajimohammadi R., Gharbani P., Mehrizad A. Evaluation of Antibacterial Activity of Aqueous, Ethanolic and Methanolic Extracts of Areca Nut Fruit on Selected Bacteria. Biomed. Res. Int. 2021;2021:6663399. doi: 10.1155/2021/6663399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Sari L.M., Hakim R.F., Mubarak Z., Andriyanto A. Analysis of phenolic compounds and immunomodulatory activity of areca nut extract from Aceh, Indonesia, against Staphylococcus aureus infection in Sprague-Dawley rats. Vet. World. 2020;13:134–140. doi: 10.14202/vetworld.2020.134-140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Raju A., De S.S., Ray M.K., Degani M.S. Antituberculosis activity of polyphenols of Areca catechu. Int. J. Mycobacteriol. 2021;10:13–18. doi: 10.4103/ijmy.ijmy_199_20. [DOI] [PubMed] [Google Scholar]
- 81.Machová M., Bajer T., Šilha D., Ventura K., Bajerová P. Volatiles Composition and Antimicrobial Activities of Areca Nut Extracts Obtained by Simultaneous Distillation-Extraction and Headspace Solid-Phase Microextraction. Molecules. 2021;26:7422. doi: 10.3390/molecules26247422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Weng C.L., Chen C.C., Tsou H.H., Liu T.Y., Wang H.T. Areca nut procyanidins prevent ultraviolet light B-induced photoaging via suppression of cyclooxygenase-2 and matrix metalloproteinases in mouse skin. Drug. Chem. Toxicol. 2022;45:353–359. doi: 10.1080/01480545.2019.1696813. [DOI] [PubMed] [Google Scholar]
- 83.Chaikhong K., Chumpolphant S., Rangsinth P., Sillapachaiyaporn C., Chuchawankul S., Tencomnao T., Prasansuklab A. Antioxidant and Anti-Skin Aging Potential of Selected Thai Plants: In Vitro Evaluation and In Silico Target Prediction. Plants. 2022;12:65. doi: 10.3390/plants12010065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Mubarokah W.W., Nurcahyo W., Prastowo J., Kurniasih K. In vitro and in vivo Areca catechu crude aqueous extract as an anthelmintic against Ascaridia galli infection in chickens. Vet. World. 2019;12:877–882. doi: 10.14202/vetworld.2019.877-882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Yamson E.C., Tubalinal G., Viloria V.V., Mingala C.N. Anthelmintic effect of betel nut (Areca catechu) and neem (Azadirachta indica) extract against liver fluke (Fasciola spp.) J. Adv. Vet. Anim. Res. 2019;6:44–49. doi: 10.5455/javar.2019.e310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Ji X., Guo J., Pan F., Kuang F., Chen H., Guo X., Liu Y. Structural Elucidation and Antioxidant Activities of a Neutral Polysaccharide From Arecanut (Areca catechu L.) Front. Nutr. 2022;9:853115. doi: 10.3389/fnut.2022.853115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Ma J., Du X., Zhao A., Wang Z., Guo Q., Qin N., Wang R. Anti-hypoxic pharmacological effects of betelnut polyphenols. J. Cent. South Univ. Med. Sci. 2022;47:512–520. doi: 10.11817/j.issn.1672-7347.2022.210582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Sartinah A., Nugrahani I., Ibrahim S., Anggadiredja K. Potential metabolites of Arecaceae family for the natural anti-osteoarthritis medicine: A review. Heliyon. 2022;8:e12039. doi: 10.1016/j.heliyon.2022.e12039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Kondaiah P., Pant I., Khan I. Molecular pathways regulated by areca nut in the etiopathogenesis of oral submucous fibrosis. Periodontol. 2000. 2019;80:213–224. doi: 10.1111/prd.12266. [DOI] [PubMed] [Google Scholar]
- 90.Yuwanati M., Ramadoss R., Kudo Y., Ramani P., Senthil Murugan M. Prevalence of oral submucous fibrosis among areca nut chewers: A systematic review and meta-analysis. Oral Dis. 2022;00:1–7. doi: 10.1111/odi.14235. [DOI] [PubMed] [Google Scholar]
- 91.Yang J., Wang Z.Y., Huang L., Yu T.L., Wan S.Q., Song J., Zhang B.L., Hu M. Do betel quid and areca nut chewing deteriorate prognosis of oral cancer? A systematic review, meta-analysis, and research agenda. Oral Dis. 2021;27:1366–1375. doi: 10.1111/odi.13456. [DOI] [PubMed] [Google Scholar]
- 92.Nagesh R., Kiran Kumar K.M., Naveen Kumar M., Patil R.H., Sharma S.C. Regulation of Jun and Fos AP-1 transcription factors by JNK MAPKs signaling cascade in areca nut extract treated KB cells. Biochem. Biophys. Rep. 2021;27:101090. doi: 10.1016/j.bbrep.2021.101090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Li J., Yao M., Zhu X., Li Q., He J., Chen L., Wang W., Zhu C., Shen T., Cao R., et al. YAP-Induced Endothelial-Mesenchymal Transition in Oral Submucous Fibrosis. J. Dent. Res. 2019;98:920–929. doi: 10.1177/0022034519851804. [DOI] [PubMed] [Google Scholar]
- 94.Tu H.F., Chen M.Y., Lai J.C., Chen Y.L., Wong Y.W., Yang C.C., Chen H.Y., Hsia S.M., Shih Y.H., Shieh T.M. Arecoline-regulated ataxia telangiectasia mutated expression level in oral cancer progression. Head. Neck. 2019;41:2525–2537. doi: 10.1002/hed.25718. [DOI] [PubMed] [Google Scholar]
- 95.Das A., Giri S. A Review on Role of Arecoline and Its Metabolites in the Molecular Pathogenesis of Oral Lesions with an Insight into Current Status of Its Metabolomics. Prague Med. Rep. 2020;121:209–235. doi: 10.14712/23362936.2020.19. [DOI] [PubMed] [Google Scholar]
- 96.Ren H., He G., Lu Z., He Q., Li S., Huang Z., Chen Z., Cao C., Wang A. Arecoline induces epithelial-mesenchymal transformation and promotes metastasis of oral cancer by SAA1 expression. Cancer Sci. 2021;112:2173–2184. doi: 10.1111/cas.14866. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 97.Ding H., Bai F., Cao H., Xu J., Fang L., Wu J., Yuan Q., Zhou Y., Sun Q., He W., et al. PDE/cAMP/Epac/C/EBP-β Signaling Cascade Regulates Mitochondria Biogenesis of Tubular Epithelial Cells in Renal Fibrosis. Antioxid. Redox Signal. 2018;29:637–652. doi: 10.1089/ars.2017.7041. [DOI] [PubMed] [Google Scholar]
- 98.Zhang B., Gao L., Shao C., Deng M., Chen L. Arecoline Enhances Phosphodiesterase 4A Activity to Promote Transforming Growth Factor-β-Induced Buccal Mucosal Fibroblast Activation via cAMP-Epac1 Signaling Pathway. Front. Pharmacol. 2021;12:722040. doi: 10.3389/fphar.2021.722040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Sanjeeta N., Banerjee S., Mukherjee S., Devi T.P., Nandini D.B., Aparnadevi P. Correlation of the activities of antioxidant enzymes superoxide dismutase and glutathione reductase with micronuclei counts among areca nut chewers of Manipuri population using exfoliative cytology: A preliminary study. J. Cancer Res. Ther. 2022;18:984–989. doi: 10.4103/jcrt.JCRT_1227_20. [DOI] [PubMed] [Google Scholar]
- 100.Shen Y.W., Shih Y.H., Fuh L.J., Shieh T.M. Oral Submucous Fibrosis: A Review on Biomarkers, Pathogenic Mechanisms, and Treatments. Int. J. Mol. Sci. 2020;21:7231. doi: 10.3390/ijms21197231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Xie H., Jing R., Liao X., Chen H., Xie X., Dai H., Pan L. Arecoline promotes proliferation and migration of human HepG2 cells through activation of the PI3K/AKT/mTOR pathway. Hereditas. 2022;159:29. doi: 10.1186/s41065-022-00241-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Chou Y.T., Sun Z.J., Shen W.C., Yang Y.C., Lu F.H., Chang C.J., Li C.Y., Wu J.S. Cumulative Betel Quid Chewing and the Risk of Significant Liver Fibrosis in Subjects With and Without Metabolic Syndrome. Front. Nutr. 2022;9:765206. doi: 10.3389/fnut.2022.765206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Chou Y.T., Li C.H., Sun Z.J., Shen W.C., Yang Y.C., Lu F.H., Chang C.J., Wu J.S. A Positive Relationship between Betel Nut Chewing and Significant Liver Fibrosis in NAFLD Subjects, but Not in Non-NAFLD Ones. Nutrients. 2021;13:914. doi: 10.3390/nu13030914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Athukorala I.A., Tilakaratne W.M., Jayasinghe R.D. Areca Nut Chewing: Initiation, Addiction, and Harmful Effects Emphasizing the Barriers and Importance of Cessation. J. Addict. 2021;2021:9967097. doi: 10.1155/2021/9967097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Dar A., Khatoon S. Behavioral and biochemical studies of dichloromethane fraction from the Areca catechu nut. Pharmacol. Biochem. Behav. 2000;65:1–6. doi: 10.1016/S0091-3057(99)00179-3. [DOI] [PubMed] [Google Scholar]
- 106.Gupta R., Nethan S.T., Sinha D.N., Gupta S., Singh S. Systematic review of determinants and interventions of areca nut cessation: Curbing a public health menace. J. Public Health. 2023;45:145–153. doi: 10.1093/pubmed/fdab411. [DOI] [PubMed] [Google Scholar]
- 107.Joo Y.J., Newcombe D., Nosa V., Bullen C. Investigating Betel Nut Use, Antecedents and Consequences: A Review of Literature. Subst. Use Misuse. 2020;55:1422–1442. doi: 10.1080/10826084.2019.1666144. [DOI] [PubMed] [Google Scholar]
- 108.Lin W.Y., Tsai B.C., Day C.H., Chiu P.L., Chen R.J., Chen M.Y., Padma V.V., Luk H.N., Lee H.C., Huang C.Y. Arecoline induces heart injure via Fas/Fas ligand apoptotic pathway in heart of Sprague-Dawley rat. Environ. Toxicol. 2021;36:1567–1575. doi: 10.1002/tox.23153. [DOI] [PubMed] [Google Scholar]
- 109.Ho T.J., Chi-Kang Tsai B., Kuo C.H., Luk H.N., Day C.H., Jine-Yuan Hsieh D., Chen R.J., Kuo W.W., Kumar V.B., Yao C.H., et al. Arecoline induces cardiotoxicity by upregulating and activating cardiac hypertrophy-related pathways in Sprague-Dawley rats. Chem. Biol. Interact. 2022;354:109810. doi: 10.1016/j.cbi.2022.109810. [DOI] [PubMed] [Google Scholar]
- 110.Ku C.W., Day C.H., Ou H.C., Ho T.J., Chen R.J., Kumar V.B., Lin W.Y., Huang C.Y. The molecular mechanisms underlying arecoline-induced cardiac fibrosis in rats. Open. Life Sci. 2021;16:1182–1192. doi: 10.1515/biol-2021-0116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Boruah N., Singh C.S., Swargiary P., Dkhar H., Chatterjee A. Securin overexpression correlates with the activated Rb/E2F1 pathway and histone H3 epigenetic modifications in raw areca nut-induced carcinogenesis in mice. Cancer Cell. Int. 2022;22:30. doi: 10.1186/s12935-022-02442-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Kumar M., Kannan A., Upreti R.K. Effect of betel/areca nut (Areca catechu) extracts on intestinal epithelial cell lining. Vet. Hum. Toxicol. 2000;42:257–260. [PubMed] [Google Scholar]
- 113.Ye C.X., Chen S.B., Wang T.T., Zhang S.M., Qin J.B., Chen L.Z. Risk factors for preterm birth: A prospective cohort study. Chin. J. Contemp. Pediatr. 2021;23:1242–1249. doi: 10.7499/j.issn.1008-8830.2108015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.De Silva M., Panisi L., Brownfoot F.C., Lindquist A., Walker S.P., Tong S., Hastie R. Systematic review of areca (betel nut) use and adverse pregnancy outcomes. Int. J. Gynaecol. Obstet. 2019;147:292–300. doi: 10.1002/ijgo.12971. [DOI] [PubMed] [Google Scholar]
- 115.Li W.D., Zang C.J., Yin S., Shen W., Sun Q.Y., Zhao M. Metformin protects against mouse oocyte apoptosis defects induced by arecoline. Cell. Prolif. 2020;53:e12809. doi: 10.1111/cpr.12809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Chang Y.F. Effects of areca nut consumption on cell differentiation of osteoblasts, myoblasts, and fibroblasts. BIOCELL. 2023;47:283–287. doi: 10.32604/biocell.2023.025743. [DOI] [Google Scholar]
- 117.Li Y.X., Hsiao C.H., Chang Y.F. N-acetyl cysteine prevents arecoline-inhibited C2C12 myoblast differentiation through ERK1/2 phosphorylation. PLoS ONE. 2022;17:e0272231. doi: 10.1371/journal.pone.0272231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Chang C.K., Lee J.I., Chang C.F., Lee Y.C., Jhan J.H., Wang H.S., Shen J.T., Tsao Y.H., Huang S.P., Geng J.H. Betel Nut Chewing Is Associated with the Risk of Kidney Stone Disease. J. Pers. Med. 2022;12:126. doi: 10.3390/jpm12020126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Hsieh Y.H., Syu R.J., Lee C.C., Lin S.H., Lee C.H., Cheng C.W., Tsai J.P. Arecoline induces epithelial mesenchymal transition in HK2 cells by upregulating the ERK-mediated signaling pathway. Environ. Toxicol. 2020;35:1007–1014. doi: 10.1002/tox.22937. [DOI] [PubMed] [Google Scholar]
- 120.Horenstein N.A., Quadri M., Stokes C., Shoaib M., Papke R.L. Cracking the Betel Nut: Cholinergic Activity of Areca Alkaloids and Related Compounds. Nicotine Tob. Res. 2019;21:805–812. doi: 10.1093/ntr/ntx187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Bernstein N., Akram M., Yaniv-Bachrach Z., Daniyal M. Is it safe to consume traditional medicinal plants during pregnancy? Phytother. Res. 2021;35:1908–1924. doi: 10.1002/ptr.6935. [DOI] [PubMed] [Google Scholar]