Abstract
Well-characterized and validated animal models are required for the development of medical countermeasures (MCMs) for acute radiation syndrome to mitigate injury due to high doses of total- or partial-body irradiation. Animal models used in MCM development must reflect a radiation dose- and time-dependent relationship, clinical presentation, and pathogenesis of organ injuries in humans. The objective of the current study was to develop the lethality curve for the Armed Forces Radiobiology Research Institute high level cobalt-60 gamma-radiation source in nonhuman primates (NHPs) after total-body irradiation. A dose-response relationship was determined using NHPs (rhesus macaques, N = 36, N = 6/radiation dose) irradiated with 6 doses in the range of 6.0 to 8.5 Gy, with 0.5 Gy increments at a dose rate of 0.6 Gy/min. Animals were provided subject-based supportive care including blood transfusions and were monitored for 60 days postirradiation. Survival was the primary endpoint of the study and the secondary endpoint included hematopoietic recovery. The lethality curve suggested LD30/60, LD50/60, and LD70/60 values as 5.71, 6.78, and 7.84 Gy, respectively. The results of this study will be valuable to provide specific doses for various lethalities of 60Co-gamma radiation to test radiation countermeasures in rhesus macaques using subject-based supportive care including blood transfusion.
INTRODUCTION
Evidence in recent years has demonstrated a dramatic increase in terrorist activity, and the dissemination of nuclear materials have increased the number of cases where military or civilian personnel may be exposed to radiation from dirty bombs or improvised nuclear devices involving a γ-radiation source, resulting in the development of acute radiation syndrome (ARS) and substantial casualties (1–3). Radiation exposures can result in different types of injuries requiring diagnostic and therapeutic intervention. The clinical progression of ARS depends on the absorbed dose and dose rate of radiation, and its distribution in the body. For humans, ARS is expressed after total-body irradiation (TBI) or partial-body irradiation (PBI) at doses above 1 Gy and delivered at relatively high dose rates (~0.05 Gy/h or above). Clinical indications of the various sub-syndromes of ARS include hematopoietic (H-ARS; 1–6 Gy), gastrointestinal (GI-ARS; 6–10 Gy), and neurovascular (NV-ARS; >10 Gy) (4). Currently, NV-ARS is considered untreatable and death generally occurs within 24–48 h. Individuals exposed to doses of radiation leading to H-ARS and GI-ARS are likely to respond to the treatment of appropriate radiation medical countermeasures (MCM). Currently, there are only four United States Food and Drug Administration (U.S. FDA)-approved MCMs that are indicated for the treatment of H-ARS (5). Neupogen® [filgrastim/granulocyte colony-stimulating factor (G-CSF)], Neulasta® (PEGfilgrastim/PEGylated G-CSF), and Leukine® [sargramostim/granulocyte-macrophage colony-stimulating factor (GM-CSF)] are approved to treat H-ARS-associated neutropenia, while Nplate® (romiplostim) is approved to treat H-ARS-associated thrombocytopenia (6–14).
The nonhuman primate (NHP) model is considered the gold standard for drug development and FDA approval due to its close resemblance to pathways and targets in relation to humans. Rhesus macaques have been used in defining radiation injury and assessing MCMs for regulatory approval (15–17). NHPs have several valuable characteristics and are considered to be the closest animal model as possible to humans (e.g., organ structure similar to humans, genetic homology – 95+% DNA sequence homology with humans, suitable for GI symptoms, metabolism similar to humans, easy sequential sampling during the course of study, and long life span) (15). Furthermore, this model closely reproduces the clinical, histopathological, and pathophysiological characteristics of radiation injury observed in humans. Such a close relationship has made NHPs attractive animal models for MCM development under the U.S. FDA Animal Rule. The most frequently used NHPs in investigation are rhesus and cynomolgus macaques (18–20).
The dose-response relationship (DRR) is an important parameter in selecting the optimal dose of radiation for MCM development, identification of biomarkers of radiation damage, and biodosimetry (21, 22). Though earlier studies from various institutions have established the DRR for H-ARS in rhesus NHPs using different radiation sources, various experimental conditions, and supportive care (18, 23, 24), a lethality curve in NHPs using the AFRRI 60Co γ-radiation source with full supportive care was needed for performing efficacy studies with MCMs of interest against specific lethal doses of γ radiation with medical management. Such study for 60Co γ radiation is important since the different energy of orthovoltage X rays, LINAC-derived photons, and γ rays may cause different biological effects (14, 25–27). With the decreasing and increasing energy of various radiation types and the use of medical management, there may be a change in hematopoietic and GI injury, requiring a dose adjustment to achieve comparable biological effects.
We accomplished a lethality curve study using male and female NHPs exposed to six different doses of total-body 60Co γ radiation, with subject-based medical management including blood products. Morbidity and mortality were assessed over a 60-day postirradiation period. Establishing a DRR for the ARS in an NHP model is pivotal in determining the efficacy of MCMs under the U.S. FDA Animal Rule (23, 26, 28).
MATERIALS AND METHODS
Experimental Design
A total of 36 animals were divided into six groups, with six animals in each group. Animals of each group were exposed to a specific dose of 60Co γ radiation (6.0, 6.5, 7.0, 7.5, 8.0 and 8.5 Gy) at a dose rate of 0.6 Gy/min (22). The animals were observed for 60 days postirradiation to score survivors.
Animals
A total of 36 naïve rhesus macaques (Macaca mulatta, Chinese substrain, 19 males and 17 females) 2.9–7.3 years of age, weighing 3.6–7.4 kg, were procured from the National Institutes of Health Animal Center (NIHAC, Poolesville, MD) and maintained in a facility accredited by the Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC)-International. Animals were quarantined for six weeks prior to initiation of the experiment. Animal quarantine, exclusion criteria, housing, health monitoring, care, and enrichment during the experimental period have been described in detail earlier (29). Animals were stratified by gender and body weight increases during the quarantine period and then assigned to different groups. All procedures involving animals were approved by the Institutional Animal Care and Use Committee (IACUC) (Armed Forces Radiobiology Research Institute Protocol #2015–12-011 and University of Maryland Baltimore Protocol #0581005) and the Department of Defense Animal Care and Use Review Office (ACURO). This study was carried out in strict accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals (30). This study used full supportive care including whole blood transfusion (14).
Irradiation
For irradiation, two NHPs were placed on the platform facing away from each other and were exposed to a specific midline dose of 60Co γ radiation at a dose rate of 0.6 Gy/min from both sides (bilateral, simultaneous exposure). This is possible since our institute’s high-level cobalt facility has two radiation sources that can be used simultaneously to irradiate animals from both sides. All irradiation procedures and dosimetry are discussed in detail earlier (29). The radiation field in the NHP location was uniform within ±1.5%. The dosimetry for photons was based on the alanine/EPR (electron paramagnetic resonance) dosimetry system (31). Dose measurements and calibrations (EMXmicro spectrometer, Bruker Corp., Billerica, MA) are based on alanine/EPR system (31, 32). This is one of the most precise dosimetry techniques at present, which is used by national standards laboratories for the most critical measurements and calibrations. The alanine dosimeters obtained from the National Institute of Standards and Technology (NIST, Gaithersburg, MD) had been calibrated in terms of absorbed dose to water using the U.S. National Standard Radiation Sources. The identical alanine dosimeters were placed midline within NHP phantoms (Plexiglas cylinders 6.9, 10, 12.5 cm in diameter and 34.5 cm length) and irradiated to approximately 100 Gy. Measurement of their EPR signals using the calibration curve constructed with alanine dosimeters from NIST-provided dose rates to water in the core bodies of NHPs. To deliver the precise dose, NHPs’ abdominal widths were measured with digital calipers. Animals were observed throughout the irradiation procedure via in-room cameras. After irradiation, animals were returned to the transport cart and to their cages in the housing area and monitored for recovery from the procedure.
Cage-Side Animal Observations
All NHPs were observed pre-exposure and for 60 days after exposure, with survival as the primary endpoint. Daily observations for signs of pain and distress were made no less than twice a day by the husbandry or research staff members. The animals were evaluated for the following parameters at least three times a week: weight, body temperature, fecal consistency, respiratory rate, heart rate, and overall health assessment (29). During the critical period (approximately 10 to 20 days postirradiation), animals were observed for signs of radiation sickness or need for further medical management and treatment. These observations were made 3 times a day no more than 10 h apart. All observations were appropriately documented in animals’ medical records.
Blood and Tissue Collection
All animals were restrained using the pole-and-collar method and placed in a chair for blood collection. The blood draw was conducted between 08:00 AM and 10:00 AM, 1–3 h after animals were fed. Blood samples were collected by venipuncture from the saphenous vein of the lower leg after the site was cleaned using a 70% isopropyl alcohol wipe and dried with sterile gauze. A 3 ml disposable luer-lock syringe with a 25-gauge needle was used to collect the desired volume of blood (29). Whole blood samples were analyzed for complete blood counts (CBC), while separated serum samples were analyzed for blood biochemistry and cytokines. For serum collection, blood samples were transferred to Capiject serum separator tubes (3T-MG; Terumo Medical Corp, Elkton, MD), allowed to clot for 30 min, and then centrifuged for 10 min at 400 × g. Serum samples were either analyzed for biochemistry or stored at −70°C for cytokine analysis. NHPs that did not reach day 60 of the study underwent necropsy along with tissue collection. Tissue samples collected were stored in 10% zinc-buffered formalin for histopathology.
CBC and Blood Biochemistry Analysis
Blood cells were counted using a Bayer Advia-120 hematology analyzer (Siemens Medical Solutions, Malvern, PA). Serum blood chemistry was analyzed using a Vitros 350 automatic biochemistry analyzer (Ortho Clinical Diagnostics, Markham, ON, Canada) (29).
Medical Management/Symptomatic Palliative Care
After specialized procedures, NHPs were monitored at least twice daily for signs of complications. The type of supportive care provided was based on CBC and blood chemistry analysis or cage side observations (33). The details of medical management are provided in Table 1. Antibiotics were initiated when the absolute neutrophil count was <500 cells/μl and continued until the count reached >500 cells/μl. In general, NHPs in the higher dose groups (7.5–8.5 Gy) had an earlier onset, longer duration, and higher incidence of neutropenia when compared to the lower dose groups (6.0–7.0 Gy). The primary antibiotic used was enrofloxacin (Baytril Bayer HealthCare LLC, Shawnee Mission, KS). The dose administered was 5 mg/kg intramuscularly (IM) or subcutaneously (SC) twice a day (BID); or 10 mg/kg administered IM or intravenous once daily (QD). If the body temperature was >39.4°C (in addition to neutrophil count <500 cells/μl), ceftiofur (Zoetis Inc.) (5 mg/kg, SC - lasts 2 days or 20 mg/kg – lasts 7 days). Alternatively, gentamicin sulfate (GentaMax, Phoenix Scientific, Inc.) (5 mg/kg, IM or iv, QD) was administered in combination with Baytril. If high fever persisted, ceftriaxone (Rocephin, Roche Laboratories Inc., Nutley, NJ) (50 mg/kg, IM, every 24 h) was administered after gentamicin sulfate was discontinued or if microbial resistance was demonstrated to enrofloxacin or gentamicin. When microbial resistance was demonstrated to enrofloxacin, gentamicin and ceftriaxone, an alternative antibiotic, at the discretion of the study’s veterinarian, was used under such situation. Additional supportive care measures included rehydration fluids, alternate antibiotics, antipyretics, antidiarrheal agents, analgesics, antiemetics, treatment for mucosal ulcers, nutritional support, and blood transfusions. The specific criteria used for administering a blood transfusion included any of the following: a platelet count of <20,000/μl and hemoglobin of <7 g/dL or hematocrit of <20% in an animal that experienced a 5-unit decrease in hematocrit in a 24 h period that resulted in a hematocrit of ≤25%, an animal that previously received a transfusion that demonstrated continued evidence of bleeding, such as bloody stools and/or any decrease in hematocrit relative to the most recent result obtained, obvious evidence of uncontrolled hemorrhage, a 7–10 unit decrease in hematocrit in a 24 h period, or a hematocrit value that was ≤25% for 2 consecutive days and platelet counts that were <3,000/μl.
TABLE 1.
Details of Medical Management/Symptomatic Palliative Care
| Drug class | Allowed medication or supportive care agents | Indication and/or criterion for administration |
|---|---|---|
|
| ||
| Parenteral fluids | Equal volume of Lactated Ringer’s Solution (LRS) and LRS with 5% Dextrose; each at 5±2.5 ml/kg body weight via slow iv push; twice daily depending on extent of dehydration; or LRS at 40±5 ml/kg body weight by iv drip over 2 h. Fluid administration rates and volume was changed at the discretion of veterinarian. | Dehydration: Mild to moderate dehydration signs and symptoms: subtle loss of skin elasticity, decreased urine output, and increased thirst. |
| Pedialyte or Gastrolyte orally 15 ± 5 ml/kg of body weight | Route: Ringer’s lactate and Ringer’s lactate with 5% dextrose via slow iv push. | |
| Bottles containing diluted fruit juice or oral rehydration solutions (Prang, Bio-Serv). | Severe dehydration signs and symptoms: rapid breathing, lethargy, severe loss of skin elasticity, sunken eyes, dry buccal mucous membranes, extremities cool to the touch, decreased body temperature. Route: Ringer’s lactate by iv drip. Pedialyte or Gastrolyte or similar commercial solutions containing sodium, potassium, and glucose was used if iv access is difficult or precluded. |
|
| Antidiarrheal | Loperamide hydrochloride (Imodium), Initial dose 0.04 mg/kg QD Diphenoxylate HCl/atropine sulfate (Lomotil), 0.1 mg/kg, (2.5 mg/tablet) was administered BID, by mouth (Po). | Fever (temperature >39.4°C) Diarrhea: At the first sign of soft-to-loose stools: Imodium was administered up to twice daily and continued for 3 days. It was discontinued earlier than 3 days if the diarrhea resolved. Severe (diarrhea or if Imodium has been administered for 3 days): Lomotil, up to BID, Po for 3 days. It was discontinued earlier than 3 days if diarrhea resolved. If the diarrhea did not resolve, the Imodium treatment was repeated. To facilitate the administration of Po medication, tablets was offered to NHP within a treat. |
| Anti-ulcer | Sucralfate (Carafate) 1 g/day, (0.5 g BID) daily from days 5–30, Po | For treatment of possible ulcers of the stomach or proximal small intestine |
| Anti-emetics | Ondansetron (1–2 mg/kg), IM, iv, or Po 25–90 min prior to irradiation and 30–45 min postirradiation to suppress emesis or Granisetron (0.25 mg/kg), Po or iv | Administered pre- and postirradiation to suppress emesis |
| Blood products | Whole blood was obtained from donor animals and blood product irradiated @2,500 cGy. Whole blood was transfused at 7–14 ml/kg of whole blood. | Whole blood was transfused if: PLT counts was <20,000/μL and hemoglobin (Hgb) <7 g/dL or hematocrit (Hct) <20% |
| Donor animals got CBC done prior to donation to confirm they do not have anemia – as per consultation with veterinarian. | • An animal experiencing a 5-unit decrease in Hct in a 24-h period that resulted in an Hct of ≤25%. • An animal that previously received a transfusion demonstrating continued evidence of bleeding, such as bloody stools and/or any decrease in Hct relative to the most recent result obtained. • If there was obvious evidence of uncontrolled hemorrhage. An animal experiencing a 7–10 unit decrease in Hct in a 24-h period • An animal’s Hct was ≤25% for 2 consecutive days and PLT counts were <3,000/L. • Packed RBCs will be administered in preference to whole blood when PLT counts are satisfactory (≥100,000/L with no clinical bleeding) but the Hgb or Hct remain low (Hgb <7 g/dL or Hct <20 g/dL). PLT counts were satisfactory (≥100,000/μL with no clinical bleeding) but the Hgb or Hct remained low (Hgb <7 g/dL or Hct <20 g/dL). |
|
| Nutritional Support | BIO-SERV Rhesus Liquidiets - To facilitate the consumption of food after irradiation, the primates were provided with an array of drinking and food products that heightened their interest. Such items included Koolaid type drinks, jello and yogurt (as examples of soft-type foods), and bananas, sweet potatoes, and celery with peanut butter (as examples of harder type foods). This brief listing is not meant to exclude any drink or food product that was in the consumption of diet designated for the animals. | Weight loss (Anorexia): Nutritional support was administered via oral gastric tube when body weight was <85% of baseline and continued if the body weight remained <85% of baseline, and the animal was not eating. |
| Analgesics | Rimadyl (Carprofen) 15 mg/tab BID, Po or 1–5 mg/kg BID, SC Buprenorphine HCL (Hospira Inc. Lake Forest IL) 0.010.02 mg/kg IM BID |
Fever (body temperature >39.4°C) or visual signs of pain as determined by the veterinarian |
| Antibiotics | Enrofloxacin (Baytril Bayer HealthCare LLC, Shawnee Mission, KS) 5 mg/kg IM or SC, twice a day (BID); or 10 mg/kg IM or SC once daily (QD). | ANC (absolute neutrophil count): Antibiotics was initiated if the absolute neutrophil count was <500/μl and was continued until the NHP ANC reached >500/μl. |
| Ceftiofur 5 mg/kg SC, lasting 2 days or 20 mg/kg SC, lasting 7 days. | Fever (body temperature 39.4°C) | |
| Gentamicin sulfate (GentaMax, Phoenix Scientific, Inc.) 5 mg/kg IM or iv, QD in combination with Enrofloxacin. | ||
| Ceftrixaone (Rocephin, Roche Laboratories Inc., Nutley, NJ) 50 mg/kg IM QD – per discretion of clinical veterinarian. | ||
| Clindamycin 12.5 mg/kg every 8 h, as per discretion of clinical veterinarian. | ||
| All iv, IM and SC procedures were performed in the same manner. The area was prepared with betadine scrub and alcohol 3× prior to administration. | ||
Euthanasia
Euthanasia was conducted in accordance with the most recently approved versions of the IACUC protocol, the Guide, and the American Veterinary Medical Association (AVMA) guidelines (30, 34). When an animal reached a state of moribundity, the animal was euthanized. Moribundity was used as a surrogate for mortality, and animals were euthanized to minimize pain and distress (29). The following parameters were used as guidelines for moribundity: significant weight loss (10%) from baseline; inappetence (complete anorexia for 2 days and deteriorating conditions); minimal or absence of response to stimuli, severe anemia (<13% hematocrit due to acute blood loss or <40 g/dL hemoglobin) and core body temperature below 96.6°F following a period of febrile neutropenia (such as >103°F and <500 neutrophils/μl); weakened/inability to obtain feed or water; severe thrombocytopenia (<10,000 platelets/μl) or other signs of severe organ dysfunction with poor prognosis as determined by the veterinarian such as dyspnea or severe cyanosis; sustained vomiting or diarrhea, GI obstruction, intussusception and peritonitis; renal failure as determined by clinical chemistry and urinalysis; sustained central nervous system depression, seizures, or paralysis of one or more extremities; non-healing wounds, repeated self-trauma, and severe skin infections; and severe organ system dysfunction with poor prognosis. Any single parameter from the above-listed guidelines did not lead to euthanasia of any animal. Moribundity status of the animal was determined by a joint effort between the institutional veterinarian, principal investigator, research staff, veterinary technicians, and husbandry staff based on the combination of criteria described above. The moribund animals were given pentobarbital sodium iv (Virbac AH Inc., Fort Worth, TX) using either saphenous or cephalic veins, needle size 20–25 gauge (100 mg/kg, 1–5 ml). Prior to pentobarbital sodium administration, animals were sedated using ketamine hydrochloride injection (Mylan Institutional LLC, Rockford, IL) (5–15 mg/kg, IM). Intra-cardiac administration was performed if unable to administer pentobarbital sodium through peripheral veins. The animals were deeply anesthetized by Isoflurane (Baxter Healthcare Corporation, Deerfield, IL) (1–5%) with oxygen at 1–4 liters per minute via mask before administering the intra-cardiac injection. The animals were euthanized only under the guidance of a staff veterinarian or a trained technician in consultation with the veterinarian. After pentobarbital sodium administration, the animals were examined by assessing the heart auscultation and pulse to confirm death. At the end of the study (60 days postirradiation), all surviving animals were sent to another facility for studying late and delayed effects of exposure to radiation.
Necropsy and Histopathology
The moribund animals were euthanized at various time points during the 60-day postirradiation period and necropsy was performed on euthanized animals. A total of eleven different tissues were collected: sternum, lung, heart, liver, kidney, spleen, duodenum, jejunum, ileum, large intestine, and urinary bladder. These tissues were fixed in 10% zinc-buffered formalin until submission to a commercial histology lab, Histoserv, Inc. (Germantown, MD), for tissue sectioning, trimming, and hematoxylin and eosin staining for microscopic evaluation. Histopathology analysis was performed on the eleven tissues from 21 decedent animals using a semi-quantitative morphologic method. Lesions, including hemorrhage, were graded semi quantitatively using a 5-point scale, where 1 = minimal, up to 5% of the area was affected; 2 = mild, up to 15% of the area affected; 3 = moderate, up to 40% of the area affected; 4 = marked, up to 75% of the area affected; and 5 = severe, greater than 75% affected, or essentially maximal.
Multiplex Analysis of Cytokines
Luminex 200 analyzer (Luminex Corp, Austin, TX) was used to detect the cytokines, chemokines, and growth factors in the serum using custom-made 48-plex multiplex kits (Bio-Rad Laboratories, Hercules, CA). A list of these 48 cytokines has been included in the supplementary file (https://doi.org/10.1667/RADE-22-001011.1.S1). Standard curves for each cytokine were prepared by serial dilution and run-in duplicates. Cytokine concentration (pg/ml) was determined by fluorescence intensity and its quantification was performed using Bio-Plex Manager software, version 6.1 (Bio-Rad Inc.) (35).
Data Analysis
A probit analysis was performed using the lethality data gathered in this study to establish a lethality curve for cobalt-60 γ radiation in NHPs with supportive care. Estimates of radiation doses were made for various death percentages based on the estimated probit model parameters. The 95% confidence intervals were estimated using the delta method based on a Taylor series expansion (36). The probit analysis was performed using STATA version 15 (StataCorp. 2017. Stata Statistical Software: Release 15. College Station, TX: StataCorp LLC.). Descriptive analyses and logistic regression were performed using SAS version 9 (SAS Institute Inc., Cary, NC). Throughout the study, confidence intervals (or standard errors) were calculated, when possible, to demonstrate the variability of the endpoints across time.
RESULTS
Effects of Different Doses of Radiation on Survival
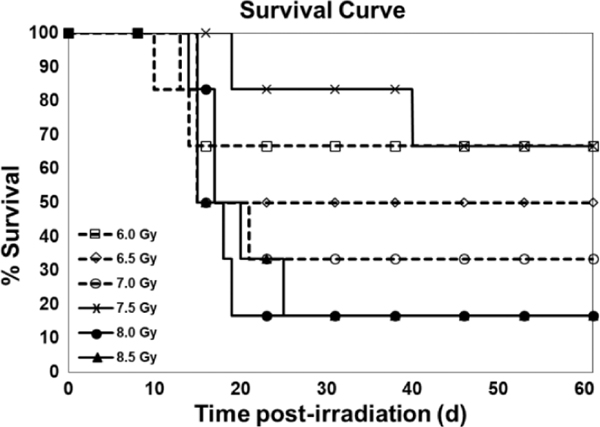
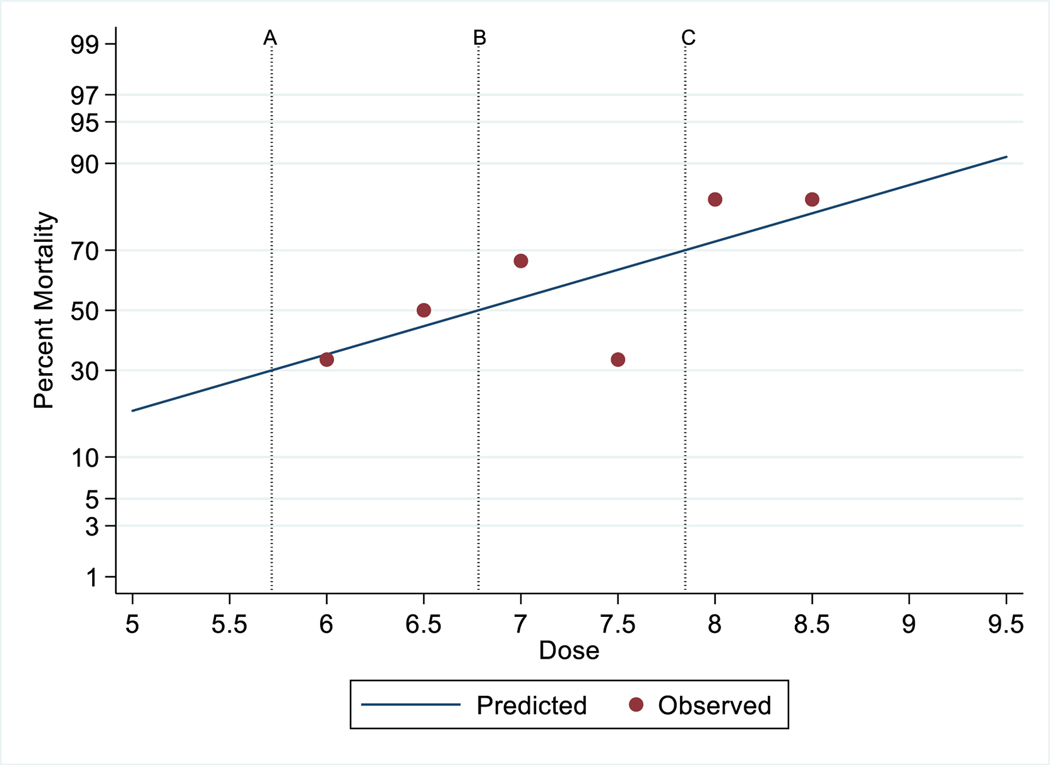
The primary endpoint was mortality since the objective of this study was to determine the lethality of 60Co γ radiation using a probit curve after exposure to various doses of radiation. All animals of any one dose were not irradiated on a single day as one cohort. This study was conducted in eight cohorts, and in each cohort three to six animals were used; animals irradiated in one day were considered as one cohort. In a single cohort, there were animals of one to four different doses. The survival of the NHPs for 60 days postirradiation is shown in Fig. 1. The need for whole blood transfusions is complicated and based on several factors. A decision to carry-out a blood transfusion requires knowledge of the most recent hematocrit and platelet counts as well as information about bleeding. The peripheral blood platelet count and number of transfusions for each animal of all groups are presented in Table 2. We have also presented pre- and post-transfusion platelet counts, transfusion incidence, and recovery in this table. Moribundity was used as a surrogate for mortality. Out of the total 36 study animals, 21 decedents showed H-ARS and associated sequelae over the dose range from 6.0 to 8.5 Gy and the results demonstrated a correlation between the dose and the lethality of NHPs as presented in Fig. 2. The observed percent survival for each radiation dose was 66.67% at 6.0 Gy, 50% at 6.5 Gy, 33.33% at 7.0 Gy, 66.67% at 7.5 Gy, and 16.67% at both 8.0 Gy and 8.5 Gy. The Kaplan-Meier survival curve demonstrated longitudinal decreased survival relative to the radiation dose over the 60 day study duration (Fig. 1). The lethality curve presented in Fig. 2 indicates that the LD30/60, LD50/60, and LD70/60 values are 5.71 (95% CI 3.87 to 7.56), 6.78 (95% CI 5.82 to 7.74), and 7.84 (95% CI 6.74 to 8.95) Gy, respectively. Mortality was less than expected in the 7.5 Gy dose group, as only 2 animals out of the 6 in this group succumbed to radiation injury (66.67% survival).
FIG. 1.
Survival curve of irradiated NHPs. Thirty-six NHPs were divided into six groups and were exposed to either 6.0, 6.5, 7.0, 7.5, 8.0, or 8.5 Gy (bilateral simultaneous, 0.6 Gy/min) 60Co γ radiation. Survival was monitored for 60 days postirradiation.
TABLE 2.
Survival Percentage for Males and Females, Pre- and Post-Transfusion Platelet Counts, and Recovery
| Dose(Gy) | Animal ID and gender | Survival for males/females |
Transfusion incidence (study day)a | Platelet count for transfusion (×103 cells/μl) |
||
|---|---|---|---|---|---|---|
| Day | Percentage on day 60 | Pre-transfusionb | Post-transfusion (study day) c | |||
|
| ||||||
| 6.0 | RA2580M | 60 | 100%/50% | 14, 16 | 0/9 | 21 (15)/12 (18) |
| RA2445M | 60 | 14, 15 | 0/9 | 20 (16) | ||
| RA0529F | 60 | None | - | - | ||
| RA2740F | 60 | 15, 19 | 0/9 | 16 (16)/63 (20) | ||
| RA0724F | 14 | 13, 14 | 0/19 | Deceased | ||
| RA0521F | 13 | None | - | Deceased | ||
| 6.5 | RA1547M | 60 | 66%/33% | 15 | 3 | 15 (16) |
| RA2576M | 60 | None | - | - | ||
| RA2705M | 15 | None | - | Deceased - | ||
| RA2118F | 60 | None | - | - | ||
| RA2267F | 15 | 15 | 0 | Deceased | ||
| RA1782F | 10 | 6, 8 | 8/10 | 8 (10), Deceased | ||
| 7.0 | RA2597M | 15 | 0%/44% | 13, 14, 15 | 0/0/1 | Deceased |
| RA2284F | 60 | 14, 15, 17 | 2/18/12 | 40 (16)/40 (18) | ||
| RA2021F | 60 | 14 | 6 | 31 (16) | ||
| RA2265F | 21 | 14, 16 | 3/4 | 7 (18), Deceased | ||
| RA2257F | 15 | None | - | Deceased | ||
| RA0456F | 15 | 14 | 0 | 20 (15), Deceased | ||
| 7.5 | RA2700M | 60 | 80%/0% | None | - | - |
| RA2709M | 60 | 14, 20 | 5/9 | 8 (16)/29 (22) | ||
| RA2592M | 60 | 16, 18 | 3/26 | 59 (20) | ||
| RA1938M | 60 | 14, 18 | 0/1 | 7 (16)/22 (20) | ||
| RA2691M | 39 | 14 | 4 | 31 (16), Deceased | ||
| RA2745F | 18 | 10, 14, 16 | 0/0/0 | 18 (12)/0 (18), Deceased | ||
| 8.0 | RA2596M | 60 | 20%/0% | 14, 24 | 20/13 | 15 (16)/34 (26) |
| RA2816M | 17 | 12, 14 | 3/17 | 11 (16), Deceased | ||
| RA2689M | 18 | 12, 14 | 2/10 | 13 (16), Deceased | ||
| RA2694M | 17 | 14, 16 | 0/0 | Deceased | ||
| RA2598M | 15 | 14 | 5 | 16 (15), Deceased | ||
| RA2733F | 19 | 14, 16, 18 | 0/3/20 | Deceased | ||
| 8.5 | RA2685M | 25 | 0%/33% | 14 | 1 | 8 (16), Deceased |
| RA2475M | 20 | 13, 14, 16 | 0/18/4 | 1 (18), Deceased | ||
| RA2318M | 15 | 12, 14 | 0/3 | 13 (15) Deceased | ||
| RA2743F | 60 | 14, 16 | 2 /13 | 23 (18) | ||
| RA2742F | 15 | 14 | 5 | Deceased | ||
| RA2116F | 14 | None | - | Deceased - | ||
Study days postirradiation on which transfusion was carried out.
Each number in this column is the number of platelets for one blood transfusion. There are one, two or three transfusions to various animals.
Numbers outside bracket indicate platelet counts and numbers inside the brackets indicate study days postirradiation.
FIG. 2.
Probit analysis of irradiated NHPs. Vertical reference lines indicate (A) LD30/60 =5.71 (95% CI 3.87 to 7.56), (B) LD50/60, = 6.78 (95% CI 5.82 to 7.74), and (C) LD70/60 = 7.84 (95% CI 6.74 to 8.95).
Generally, an earlier onset of dehydration was observed in animals exposed to the higher doses of radiation. At the first indication of dehydration (subtle loss of skin elasticity), parenteral fluids were administered. Additionally, bottles containing diluted fruit juice or oral rehydration solutions (Prang, Bio-Serv) were provided. Severe dehydration was not encountered during the study period as treatment was provided at the earliest sign of mild dehydration. Similarly, at the first sign of diarrhea, treatment was initiated [Loperamide hydrochloride (Imodium), initial dose 0.04 mg/kg QD]. After 3 days of unresolved diarrhea, Imodium was stopped and treatment with diphenoxylate HCl/atropine sulfate (Lomotil), 0.1 mg/kg, (2.5 mg/tablet) was administered BID. Treatment with Lomotil was highly effective at resolving cases of diarrhea. A higher incidence of diarrhea was observed with increased radiation doses.
This was a mixed cohort of males and females, and LD50/60 is the average between males and females. The sample size at each dose is too small to support separate estimation of LD50/60 by sexes.
Effects of Different Radiation Doses on CBC
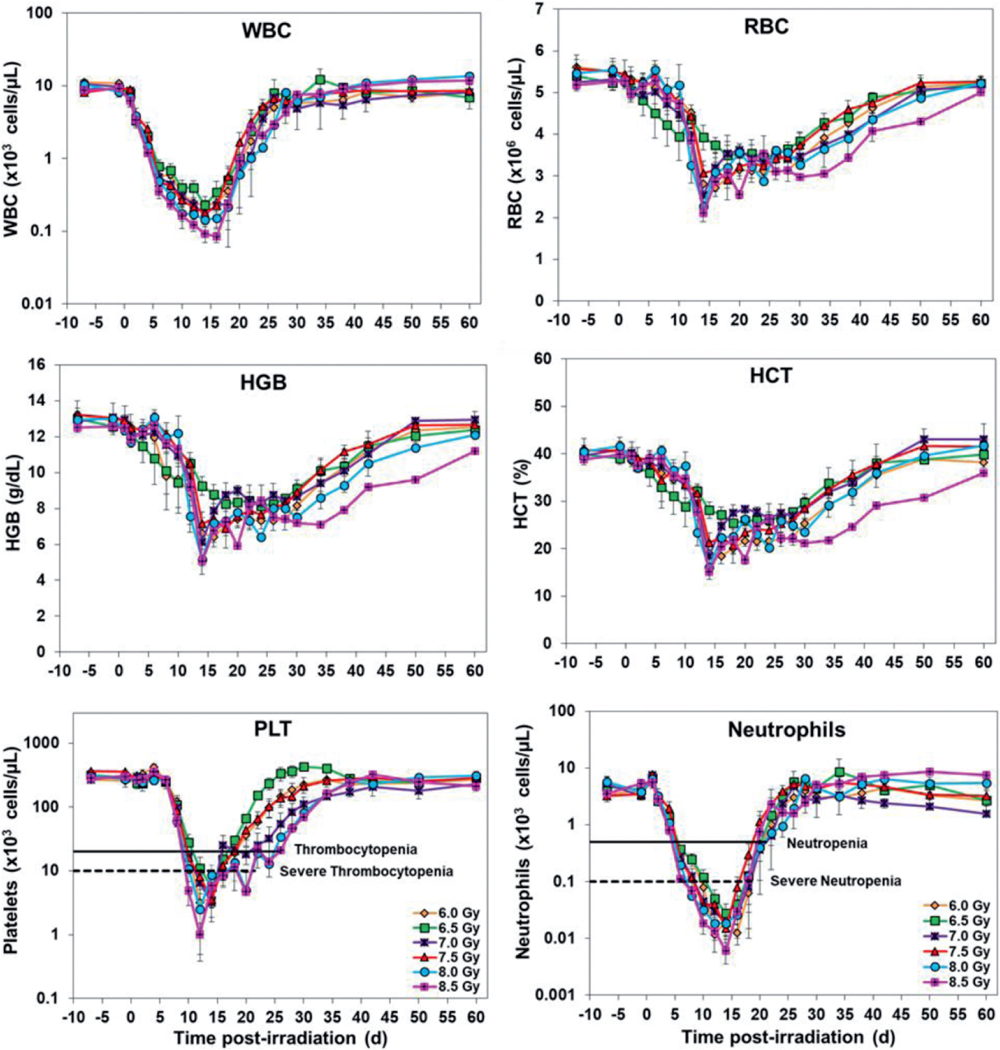
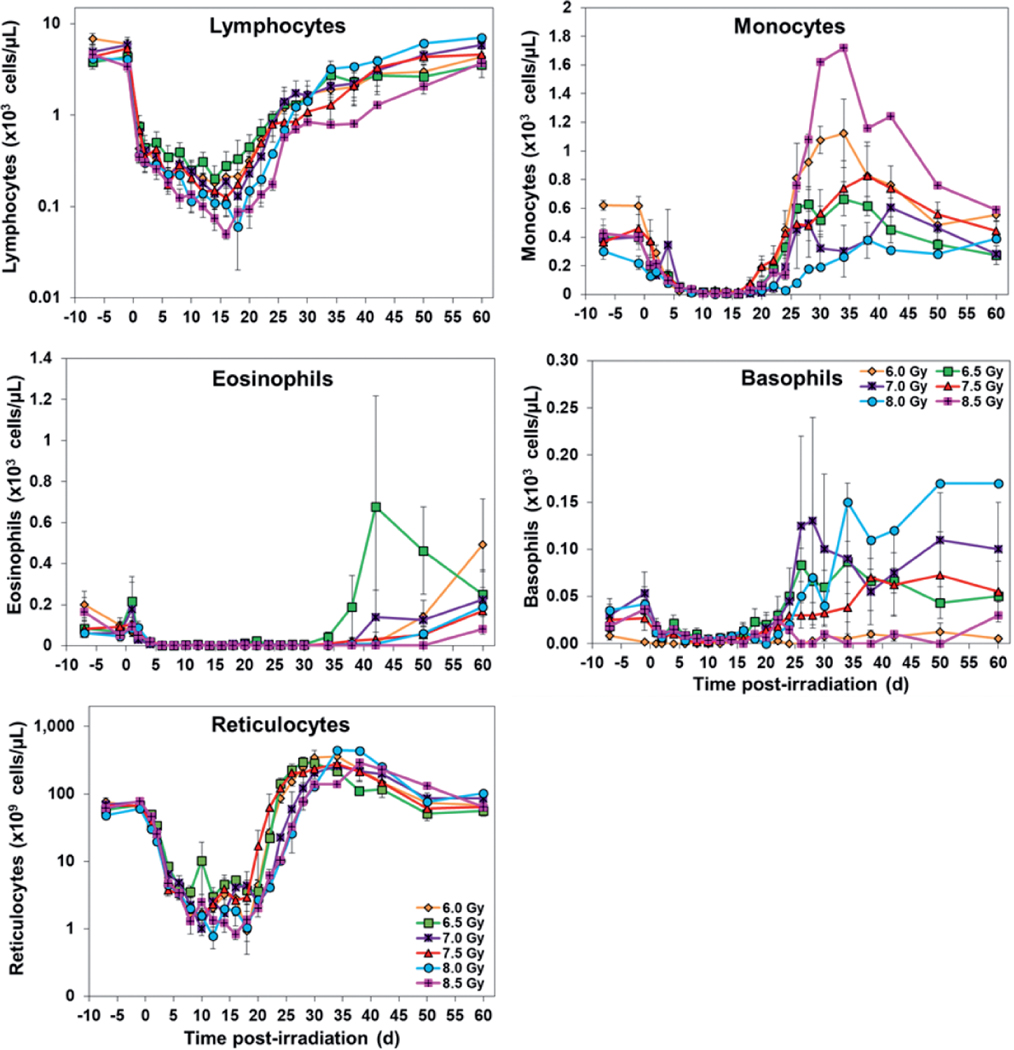
A total of eleven parameters were analyzed at various time points pre- and postirradiation, and the data presented in Figs. 3 and 4 shows postirradiation nadirs for white blood cells, platelets, neutrophils, lymphocytes, and reticulocytes in animals exposed to various doses of radiation. Overall, the results indicate that the CBC parameters in survivors returned to pre-exposure levels by the end of the study period.
FIG. 3.
Effects of various radiation doses on CBCs: white blood cells, red blood cells, hemoglobin, hematocrit, platelets, and neutrophils in irradiated NHPs. The data for each time point is presented as the mean for animals exposed to each dose of radiation.
FIG. 4.
Effects of various doses of radiation on CBCs: absolute counts of lymphocytes, monocytes, eosinophils, basophils, and reticulocytes in irradiated NHPs. The data for each time point is presented as the mean for animals exposed to each radiation dose.
White blood cells gradually decreased in values from day 1, with the lowest values noticed from days 14–16, with recovery beginning approximately 16 to 18 days postirradiation. The values eventually returned to pre-exposure levels over the course of the study. Platelets levels dropped from days 4–14 postirradiation with thrombocytopenia from days 10–26 and nadir noted from days 12–14. Severe thrombocytopenia was observed in some NHPs from days 10–20. The recovery phase started from day 14 and reached to pre-exposure levels with time. Neutrophil counts started declining after irradiation. Animals were neutropenic from days 6–20 and severely neutropenic from days 6–18 in various groups. While the nadir was observed during days 12–16, the recovery continued from days 16–18, and reached pre-exposure levels by days 24–38.
Lymphocyte values started dropping soon after irradiation and reached the lowest value from days 14–18. The recovery phase started from days 16–20 in various groups and reached pre-exposure levels gradually. Similarly, reticulocytes dropped after irradiation and the counts reached the lowest value from days 10–18 in different groups. The values started to recover from days 12–20. Red cell associated parameters (red blood cells, hematocrit and hemoglobin) showed a similar pattern where pre-exposure values decreased after irradiation, and levels returned to normal values by day 60. Other CBC parameters are also presented in Figs. 3 and 4.
The cellular values of neutropenia and thrombocytopenia, and their respective nadir, duration, and recovery for all animals in each group are presented in Tables 3 and 4. Neutrophils provide the first line of defense against opportunistic infections after irradiation. Various doses of TBI reduced the absolute neutrophil counts to <500/μl within approximately 6 day after TBI. Antibiotics were administered prophylactically when neutrophil count was <500/μl since it is expected that the neutrophil counts will decrease in values leading to grade four neutropenia (<100/μl). Various neutrophil parameters are presented in Table 3. Various doses of TBI induced severe decrease in platelet levels in all groups. The decrease, nadir, and recovery of platelet after TBI that approximate the LD30/60, LD50/60, and LD70/90 are almost similar (Fig. 3 and Table 4).
TABLE 3.
Neutrophil-Related Parameters for Rhesus Macaques after Total-Body γ Irradiation
| Dose (Gy) | Animal ID and gender | Survival day | Neutropenia (≤0.5 × 103 /μl) |
||
|---|---|---|---|---|---|
| Neutrophil nadir/(study days)a | Durationb | Recovery >0.5 × 103 cells/μLc | |||
|
| |||||
| 6.0 | RA2580M | 60 | 0.01 (16) | 6–22 (17 days) | 24 |
| RA2445M | 60 | 0.01 (12) | 10–18 (9 days) | 20 | |
| RA0529F | 60 | 0 (12) | 6–20 (15 days) | 22 | |
| RA2740F | 60 | 0.01 (12) | 6–20 (15 days) | 22 | |
| RA0724F | 14 | 0.01 (12) | * | Deceased | |
| RA0521F | 13 | 0 (12) | * | Deceased | |
| Meand | - | 0.0067 ± 0.0021 | 14 ± 1.73 | 22 ±0.82 | |
| Range | - | 0–0.01 | 9–17 | 20–24 | |
| 6.5 | RA1547M | 60 | 0.02 (16) | 6–18 (13 days) | 20 |
| RA2576M | 60 | 0.06 (14) | 6–20 (15 days) | 22 | |
| RA2705M | 15 | 0 (14) | * | Deceased | |
| RA2118F | 60 | 0.02 (14) | 8–20 (13 days) | 22 | |
| RA2267F | 15 | 0.01 (10) | * | Deceased | |
| RA1782F | 10 | 0.27 (10) | * | Deceased | |
| Meand | - | 0.063 ± 0.042 | 13.67 ± 0.67 | 21.33 ± 0.67 | |
| Range | - | 0–0.27 | 13–15 | 20–22 | |
| 7.0 | RA2597M | 15 | 0 (12) | Deceased | |
| RA2284F | 60 | 0.03 (16) | 6–22 (17 days) | 24 | |
| RA2021F | 60 | 0.02 (14) | 6–18 (13 days) | 20 | |
| RA2265F | 21 | 0.01 (12) | * | Deceased | |
| RA2257F | 15 | 0.02 (12) | * | Deceased | |
| RA0456F | 15 | 0 (12) | * | Deceased | |
| Meand | - | 0.013 ±0.0049 | 15 ± 2.0 | 22 ± 2 | |
| Range | - | 0–0.03 | 13–17 | 20–24 | |
| 7.5 | RA2700M | 60 | 0.03 (14) | 4–16 (13 days) | 18 |
| RA2709M | 60 | 0.02 (14) | 8–20 (13 days) | 22 | |
| RA2592M | 60 | 0.01 (14) | 6–18 (13 days) | 20 | |
| RA1938M | 60 | 0 (14) | 6–20 (15 days) | 22 | |
| RA2691M | 39 | 0.02 (10) | 6–16 (11 days) | 18 | |
| RA2745F | 18 | 0 (12) | * | Deceased | |
| Meand | - | 0.013 ± 0.0049 | 13 ± 0.63 | 20 ± 0.89 | |
| Range | - | 0–0.03 | 11–15 | 18–22 | |
| 8.0 | RA2596M | 60 | 0.03 (12) | 6–20 (15 days) | 22 |
| RA2816M | 17 | 0.01 (14) | * | Deceased | |
| RA2689M | 18 | 0.02 (12) | * | Deceased | |
| RA2694M | 17 | 0 (10) | * | Deceased | |
| RA2598M | 15 | 0.01 (14) | * | Deceased | |
| RA2733F | 19 | 0 (12) | * | Deceased | |
| Meandd | - | 0.012 ± 0.0048 | 15 | 22 | |
| Range | - | 0–0.03 | 22 | ||
| 8.5 | RA2685M | 25 | 0.01 (14) | * | Deceased |
| RA2475M | 20 | 0.01 (12) | * | Deceased | |
| RA2318M | 15 | 0 (10) | * | Deceased | |
| RA2743F | 60 | 0 (10) | 6–22 (17 days) | 24 | |
| RA2742F | 15 | 0.01 (12) | * | Deceased | |
| RA2116F | 14 | 0.02 (12) | * | Deceased | |
| Meand | - | 0.0083 ± 0.0031 | 17 | 24 | |
| Range | - | 0–0.02 | 24 | ||
Numbers outside bracket indicate neutrophil counts and numbers inside the brackets indicate study days postirradiation.
Numbers outside bracket indicate duration of neutropenia (first and last day) and numbers inside the brackets indicate period of neutropenia in days based on first and last neutropenic days postirradiation.
Numbers indicate day postirradiation on which animals recovered from neutropenia.
Mean data shown with the standard error of the mean (+SEM)
The values for these animals are not included for calculating the mean and ranges since these animals deceased prior to the end of the cytopenia period.
TABLE 4.
Platelet-Related Parameters for Rhesus Macaques after Total-Body γ Irradiation
| Dose (Gy) (≤20 × 103 /μl) | Animal ID and gender | Survival day | Thrombocytopenia |
||
|---|---|---|---|---|---|
| PLT nadir/(day)a | Duration days Postirradiationb | Recovery PLT >20 × 103/μl on dayc | |||
|
| |||||
| 6.0 | RA2580M | 60 | 0 (12) | 10–20 (11 days) | 22 |
| RA2445M | 60 | 0 (12) | 10–18 (9 days) | 20 | |
| RA0529F | 60 | 0 (14) | 12–16 (5 days) | 18 | |
| RA2740F | 60 | 0 (14) | 10–14 (5 days) | 16 | |
| RA0724F | 14 | 0 (12) | * | Deceased | |
| RA0521F | 13 | 0 (12) | * | Deceased | |
| Meand | - | 0 | 7.5 ±1.5 | 19 ±1.29 | |
| Range | - | 0 | 5–11 | 16–22 | |
| 6.5 | RA1547M | 60 | 3 (14) | 12–16 (5 days) | 18 |
| RA2576M | 60 | 15 (14) | 14–16 (3 days) | 18 | |
| RA2705M | 15 | 0 (14) | * | Deceased | |
| RA2118F | 60 | 10 (14) | 12–16 (5 days) | 18 | |
| RA2267F | 15 | 0 (14) | * | Deceased | |
| RA1782F | 10 | 8 (10) | * | Deceased | |
| Meand | - | 6 ± 2.46 | 4.3 ±0.67 | 18 | |
| Range | - | 0–15 | 3–5 | 18 | |
| 7.0 | RA2597M | 15 | 0 (12) | * | Deceased |
| RA2284F | 60 | 2 (14) | 10–14 (5 days) | 16 | |
| RA2021F | 60 | 6 (14) | 12–14 (3 days) | 16 | |
| RA2265F | 21 | 3 (14) | * | Deceased | |
| RA2257F | 15 | 7 (12) | * | Deceased | |
| RA0456F | 15 | 0 (14) | * | Deceased | |
| Meand | - | 3 ± 1.21 | 4 ±1.0 | 16 | |
| Range | - | 0–7 | 3–5 | 16 | |
| 7.5 | RA2700M | 60 | 12 (14) | 12–14 (3 days) | 16 |
| RA2709M | 60 | 4 (18) | 12–20 (11 days) | 22 | |
| RA2592M | 60 | 0 (12) | 10–16 (7 days) | 18 | |
| RA1938M | 60 | 0 (14) | 10–18 (9 days) | 20 | |
| RA2691M | 39 | 4 (14) | 10–14 (5 days) | 16 | |
| RA2745F | 18 | 0 (10) | * | Deceased | |
| Meand | - | 3.3 ± 1.91 | 7 ±1.41 | 18.4 ±1.17 | |
| Range | - | 0–12 | 3–11 | 16–22 | |
| 8.0 | RA2596M | 60 | 5 (20) | 10–24 (15 days) | 26 |
| RA2816M | 17 | 3 (12) | * | Deceased | |
| RA2689M | 18 | 2 (12) | * | Deceased | |
| RA2694M | 17 | 0 (12) | * | Deceased | |
| RA2598M | 15 | 3 (12) | * | Deceased | |
| RA2733F | 19 | 0 (12) | * | Deceased | |
| Meand | - | 2.2 ± 0.79 | 15 | 26 | |
| Range | - | 0–5 | 26 | ||
| 8.5 | RA2685M | 25 | 1 (12) | * | Deceased |
| RA2475M | 20 | 0 (20) | * | Deceased | |
| RA2318M | 15 | 0 (10) | * | Deceased | |
| RA2743F | 60 | 0 (10) | 10–26 (17 days) | 28 | |
| RA2742F | 15 | 2 (12) | * | Deceased | |
| RA2116F | 14 | 3 (12) | * | Deceased | |
| Meand | - | 1 ± 0.52 | 17 | 28 | |
| Range | - | 0–3 | 28 | ||
Numbers outside bracket indicate platelet counts and numbers inside the brackets indicate study days postirradiation.
Numbers outside bracket indicate duration of thrombocytopenia (first and last day) and numbers inside the brackets indicate period of thrombocytopenia in days based on first and last thrombocytopenic days postirradiation.
Numbers indicate day postirradiation on which animals recovered from thrombocytopenia.
Mean data shown with the standard error of the mean (+SEM)
The values for these animals are not included for calculating the mean and ranges since these animals deceased prior to the end of the cytopenia period.
Effects of Different Doses of Radiation on Serum Biochemistry
Blood samples were collected at selected time points pre- and postirradiation to investigate serum biochemistry. A total of 23 serum chemistry parameters were analyzed. The data presented in Supplementary Figs. S1–S3 (https://doi.org/10.1667/RADE-22-0010.1.S1) demonstrate that serum parameters including albumin, aspartate aminotransferase, alanine transferase, and cholesterol generally followed a sharp recovery in animals exposed to various doses of radiation. Overall, the results show that by the conclusion of the study, most of the serum parameters analyzed returned to pre-exposure levels.
Albumin levels started declining soon after exposure to radiation and continued to decline until day 20, a point where lowest albumin levels were observed in all doses of radiation. After day 20, the values started recovering and reached pre-exposure levels by day 60 in survivors of the study. The postirradiation concentration of aspartate aminotransferase showed a biphasic pattern, the first increase peaking on day 10 in all doses of radiation, declining thereafter till day 28, and peaking again on day 38. The concentration of aspartate aminotransferase reached the pre-exposure levels by the end of the study period on day 60. The serum alanine aminotransferase levels saw an increase soon after irradiation and continued to increase to day 10; afterwards, there was a steady decline, thereby reaching pre-irradiation levels in surviving animals by day 28. As observed in the above parameters the cholesterol levels started decreasing soon after irradiation, with the nadir on day 20. The levels started increasing thereafter, returning to pre-exposure levels by day 38 and remaining at these levels until the end of the study period. In the remaining 19 analytes assessed, no trend related to radiation exposure or dose was observed.
Effects of Different Doses of Radiation on Serum Cytokines
The cytokine profiles detected using 48 plex kits on the serum samples collected from the irradiated NHPs generally followed similar trends between radiation doses, apart from a few instances where certain radiation dose groups deviated from the other radiation dose groups. However, these were mostly isolated events, and no dose group differentiated consistently from the other groups throughout the study. Interleukin-7 (IL-7), and monocyte chemoattractant protein-1 (MCP-1 (MCAF)) concentrations displayed similar increasing trends across all six groups after irradiation. Concentrations in NHPs exposed to 8.5 Gy increased sharply on day 2, and remained elevated throughout the study, while concentrations in the 7.0 Gy group increased sharply on day 6 (Supplementary Figs. S4–S7). The 7.0 Gy group also experienced a sharp increase in the concentration of IL-7 and IL-13, with a less pronounced increase in MCP-1. Granulocyte colony-stimulating factor (G-CSF) concentrations were found to be elevated across all groups after exposure to radiation, with the most prominent raise in concentration noted on day 6. The concentration of stem cell growth factor-β (SCGF-β) and platelet derived growth factor-BB (PDGF-BB) mostly decreased across all six groups following irradiation, apart from a few exceptions. The concentrations of macrophage colony stimulating factor (M-CSF) fluctuated over the course of the study, with all NHPs experiencing a rise in this cytokine after irradiation, followed by a decline towards baseline by day 6. The concentration of IL-1β remained consistent within each group through day 2, which was followed by a substantial amount of variation on day 6. The group exposed to 7.0 Gy displayed a notable elevation in concentration on day 6. The 6.0, 6.5, and 7.5 Gy groups experienced minor increases in cytokine concentration in comparison to their baseline values, while the remaining two cohorts, 8.0 and 8.5 Gy, displayed a notable decrease in concentrations on day 6. Interleukin-2 (IL-2) concentrations had the most variation between groups. For the high-dose groups, 7.5, 8.0, and 8.5 Gy, there were no detectable concentrations noted in time points after irradiation. Concentrations for the remaining lower dose groups were consistent throughout the study. Generally, apart from the instances listed above, cytokine concentrations in NHPs exposed to one of the six radiation doses were consistent throughout the duration of the study with minimal variation between groups. All cytokine concentrations of different groups at various time points throughout the study can be viewed in Supplementary Figs. S4–S7 (https://doi.org/10.1667/RADE-22-00101.1.S1).
Effects of Different Doses of Radiation on Vital Signs
Vital signs data including heart rate, temperature, blood pressure, and weights were recorded on the days when animals were anesthetized for scheduled sample collection, as described above. Vital signs data can be viewed in Supplementary Fig. S8 (https://doi.org/10.1667/RADE-22-00101.1.S1). Heart rate, temperature, and blood pressure readings generally followed similar trends between various groups throughout the study. There were a few instances where the readings of one group were considerably higher or lower than the other dose groups. However, these were typically isolated events. Weights followed unique trends across all dose groups. The groups exposed to the lower radiation doses, 6.0 and 6.5 Gy, were the only two groups that finished the study with higher average weights than those recorded pre-irradiation.
Necropsy and Histopathological Analysis
Histopathological findings revealed radiation-induced changes in all organs examined, and results can be viewed in Table 5. These consisted of atrophy or degeneration/necrosis of rapidly dividing cell types, including bone marrow elements, splenic lymphocytes of the white pulp, epithelial necrosis of intestinal and bladder mucosa and lung bronchial and alveolar cells, as well as hepatocyte and renal tubular necrosis, and hemorrhages in various organs. Gut-associated lymphoid tissue (GALT) was present in many but not all small or large intestine sections. When present, it was consistently atrophic as lymphoid depletion. The marrow element depletion or atrophy included developing red, white and megakaryocyte precursors; thus, the hemorrhages were likely related to depleted thrombocytes and clotting dysfunction. The spleen lymphoid depletion or atrophy included splenic follicles, periarteriolar lymphoid sheaths (PALS), and marginal zone lymphocytes. Bacterial colonies were often seen, particularly in the liver or intestine.
TABLE 5.
Radiation-Induced Histopathological Changes in Decedent NHPs after Total-Body γ Irradiation
| Organs | Histological lesions | 6.0–6.5 Gy |
7.0–7.5 Gy |
8.0 Gy |
8.5 Gy |
||||
|---|---|---|---|---|---|---|---|---|---|
| MED (IQR) | IR (n = 5) | MED (IQR) | IR (n = 6) | MED (IQR) | IR (n = 5) | MED (IQR) | IR (n = 5) | ||
|
| |||||||||
| Sternum | Myeloid element atrophy | 5 (1) | 4 | 4 (2) | 6 | 4 (0) | 5 | 5 (2) | 4 |
| Hemorrhage | 3 (3) | 3 | 3 (2) | 4 | 2 (2) | 4 | 3 (1) | 4 | |
| Spleen | White pulp atrophy | 5 (0) | 5 | 5 (0) | 5 | 5 (0) | 5 | 5 (0) | 5 |
| Liver | Hepatocellular necrosis | 1 (3) | 3 | 1 (0) | 1 | 2 (1) | 4 | 4 (4) | 3 |
| Bacterial presence | 2 | 1 | 4 | 2 | |||||
| Lung | Alveolar epithelial necrosis | 2 (0) | 1 | 1 (0) | 1 | – | – | – | – |
| Bronchial epithelial necrosis | 3 (0) | 1 | – | – | 3 (2) | 3 | 4 (1) | 2 | |
| Hemorrhage | 1 (0) | 1 | – | – | 3 (4) | 2 | – | – | |
| Edema | 3 (0) | 1 | – | – | – | – | 3 (1) | 3 | |
| Interstitial mononuclear cell-infiltration | 1 (0) | 1 | – | – | 2 (1) | 3 | 2 (0) | 1 | |
| Acute alveolar inflammation | – | – | 4 (0) | 1 | – | – | – | – | |
| Heart | Bacterial presence | – | 1 | – | – | – | – | – | – |
| Hemorrhage | 1 (0) | 1 | – | – | 2 (1) | 2 | – | ||
| Myocyte necrosis | – | – | – | – | – | – | 2 (0) | 1 | |
| Kidney | Tubular necrosis | 1 (0) | 2 | – | – | 2 (1) | 2 | – | – |
| Nephropathy with casts | 5 (0) | 1 | – | – | – | – | – | – | |
| Hemorrhage | – | – | – | – | 2 (0) | 1 | 2 (0) | 1 | |
| Duodenum | Hemorrhage | 1 (0) | 2 | – | – | 3 (0) | 2 | 2 (1) | 2 |
| Mucosal erosion | 2 (0) | 1 | – | – | 3 (0) | 1 | – | ||
| Mucosal necrosis | – | – | – | – | 5 (1) | 2 | 4 (0) | 1 | |
| Mucosal ulcer | – | – | – | – | 4 (0) | 1 | – | – | |
| Jejunum | Mucosal ulcer | 4 (0) | 1 | – | – | – | – | – | – |
| Mucosal necrosis | – | – | – | – | 4 (3) | 2 | 5 (0) | 2 | |
| Mucosal erosion | 2 (0) | 1 | – | – | 4 (1) | 2 | – | – | |
| Hemorrhage | 3 (4) | 2 | – | – | 2 (2) | 3 | 3 (0) | 1 | |
| Bacterial presence | – | 1 | – | – | – | – | – | – | |
| Ileum | Hemorrhage | 2 (1) | 2 | – | – | 1 (0) | 1 | 3 (4) | 2 |
| Mucosal necrosis | – | – | – | – | 5 (1) | 2 | – | – | |
| Large intestine | Mucosal necrosis | – | – | – | – | 5 (0) | 1 | – | – |
| Hemorrhage | – | – | 3 (2) | 2 | 3 (3) | 2 | 3 90) | 1 | |
| Mucosal erosion | – | – | – | – | 1 (0) | 1 | – | – | |
| Mucosal ulcer | – | – | – | – | 5 (0) | 1 | – | – | |
| Urinary bladder | Hemorrhage | 1 (0) | 1 | 2 (1) | 2 | 1 (2) | 3 | – | – |
| Mononuclear cell infiltrate | – | – | – | – | 1 (0) | 1 | — | ||
| Epithelial necrosis | – | – | – | – | 1 (0) | 1 | |||
| GALT | Lymphocyte depletion | 5 (0) | 5 | 5 (0) | 5 | 5 (0) | 5 | 5 (0) | 5 |
Notes. Organs were collected and processed for H&E staining from NHPs that underwent unscheduled euthanasia before the completion of the study (60 days postirradiation). Sections were scored semi-quantitatively on a five-point scale [1 = minimal (≤5% of the area affected); 2 = mild (≤15%); 3 = moderate (≤40%); 4 = marked (≤75%); and 5 = severe (≥75%)]. MED (IQR) - Median score (Interquartile Range). IR - Incidence Rate, indicates number of animals with lesions out of total number (n) of irradiated NHPs. Responses from 6.0 (n = 2) and 6.5 Gy (n = 3) exposure, also from 7.0 (n=4) and 7.5 (n=2) Gy were pooled due to smaller sample size. Bacterial presence was qualitative, and no scores were given. Score of “–” indicates that the histology was within the normal limits. Gut-associated lymphoid tissue (GALT) was present in most of the small and large intestinal sections and was consistently atrophic
While both male and female NHPs were included in this study, there were no observable gender differences in the type or severity of lesions. All animals, except one, had GALT atrophy with the highest severity grade. The results concluded that increasing radiation dose resulted in a wider variety of changes mentioned above, as well as increasing the severity of such histopathological findings. Each incidence of radiation-induced histopathological changes in non-surviving NHPs can be viewed in Table 5.
DISCUSSION
Well-defined animal models are necessary for the development of effective MCMs for ARS and delayed effects of acute radiation exposure (DEARE). Specifically, the NHP model is critical for the regulatory approval of such MCMs from the US FDA for human use under the Animal Rule (28). NHPs are particularly pertinent for the investigation of the efficacy of MCMs to mitigate morbidity and mortality in animals of H-ARS, GI-ARS, and DEARE (15). In addition to NHPs, there are other animal models including ferrets, swine (minipig in particular), canines, and rats that are also used to investigate the pathophysiology of radiation injury as well as countermeasure development. Some of these species are suitable for specific endpoints (e.g., ferrets for emesis and minipig for cutaneous injury) (37, 38). However, the NHP is regarded as the model reproducing the most suitable representation of human clinical presentation with a high degree of resemblance in terms of pathways of physiological responses and targets (15). Based on these attributes, the NHP is considered the gold standard of animal models for MCM development and regulatory approval. The characterization of such models also includes a reproducible DRR. Though NHP models have long been used in studies of radiation pathophysiology, there is considerable heterogeneity in DRRs across various institutions conducting such studies involving different radiation sources, radiation qualities, level of clinical support, and other experimental environment factors (23, 25). Numerous factors contribute to this phenomenon and data describing the DRR for various radiation sources, types of radiation, and different levels of medical management are limited. The effect of animal sex, radiation type, and supportive care are important factors that may be responsible for DRR heterogeneity. It is apparent that the level of medical management and the use of blood products may shift the DRR. It is also challenging to compare such data across publications from various institutions carried out under different experimental conditions (23, 24, 39, 40).
A large data set from a male-only NHP TBI model exists. However, the dependence on only one sex may result in an incomplete understanding of sex differences in the DRR. A recent review article by Beach et al. has suggested that female NHPs are more sensitive to radiation exposure and display higher mortality than males (39). These findings may be important for the treatment of irradiated victims during a large-scale radiological incidence under a limited resource setting. Blood transfusions improve survival in males when exposed to lower doses of radiation, but not at higher doses of radiation. The beneficial effect of blood transfusions at lower doses of radiation exposure may be associated with H-ARS. However, blood transfusions may have a minimal impact on GI-ARS that presents after higher doses of radiation (39, 40). Female NHPs have been shown to have higher mortality after irradiation at various doses than male macaques. Such study emphasizes the need for using animals of both sexes in such studies. The above findings are based on TBI models, and it will be interesting to investigate whether sex differences lead to different mortality rates in PBI model. Optimally, both sexes should be included in radiation MCM studies to ensure differences between sexes are accounted for. We used both males and females in our study, but the sample size is not large enough to compare the fluctuations in various parameters between males and females. There are several key variables that affect the outcome of every radiation effect study conducted at different research sites using modified study protocols. Many variables affect the outcome of lethal irradiation, such as radiation source, exposure geometry, supportive care regimen, and well-defined study protocols. Several studies have published DRRs and LD50/30 values for rhesus macaques exposed to uniform TBI with 60Co relative to supportive care or lack thereof. The values are relatively similar within all the studies (11, 23, 33, 39, 41, 42). Additionally, though there is substantial evidence that medical management changes the DRR, a consensus about exact supportive care provided for the development of MCMs needs to be harmonized across various institutions conducting MCM development using NHP models with funding from different sources (39). This is a debatable issue from the feasibility point of view. For example, blood transfusions may not be a practicable option during a mass causality scenario as a result of a large scale radiological/nuclear event (43). Yu et al. conducted a study comparing benefits of subject-based care versus a population-based strategy in rhesus macaques (27). Estimated doses for LD30/60, LD50/60, and LD70/60 of NHPs with subject-based supportive care were 6.83 Gy, 7.44 Gy, and 8.05 Gy, respectively, compared with population-based strategy radiation doses of 5.61 Gy, 6.62 Gy, and 7.63 Gy, respectively. This study demonstrates the benefits of subject-based supportive care over population-based strategy.
Our study was performed using a cobalt-60 γ radiation source for animal exposure and we opted for bilateral simultaneous exposure. The calculated LD30/60, LD50/60, and LD70/60 values were 5.71, 6.78, and 7.84 Gy, respectively. This study was completed using a sample size of 6 animals in each radiation dose and this is a limitation in this study. Prior to conducting this study, various study options were considered to decide the number of animals in each group/dose of radiation and the number of groups. This evaluation was done for each of three models: a model consistent with AXR01 data, a model with a lower lethality curve, and one with a higher lethality curve to ensure that the results would be robust across a range of possible true curves. Four dosing regimen options were used for projection: (a) 8 doses spread uniformly from 6 to 9.5 (6.0, 6.5, 7.0, 7.5, 8.0, 8.5, 9.0, and 9.5 Gy); (b) 8 doses spread uniformly from 6.5 to 9 (6.5, 6.86, 7.21, 7.57, 7.93, 8.29, 8.64, and 9.0 Gy), (c) 6 doses spread uniformly from 6 to 9.5 (6.0, 6.7, 7.4, 8.1, 8.8, and 9.5 Gy), and (d) 6 doses spread uniformly from 6.5 to 9 (6.5, 7.0, 7.5, 8.0, 8.5, and 9.0 Gy) (36 total animals vs. 48 total animal situation).
The fourth dosing regimen provided the most precise estimates of LD30/60, LD50/60, and LD70/60, regardless of the three models. The advantage of this dosing regimen was relatively modest, but it was the clear winner. The advantage of using 48 total animals versus 36 was also relatively modest; the decision on sample size requires a judgement as it is a trade-off between increased efficiency and the total cost and number of animals used. For NHP studies, the number of animals used is important from an IACUC perspective to ensure minimal use of large animals keeping in mind the 3Rs. Thus, the fourth option was selected for the study. During execution of the study, based on mortality, it was decided to remove the 9.0 Gy and include the 6.0 Gy dose. Recently, we have also identified biomarkers of radiation exposure using multi-omics platforms (metabolomics/lipidomics and proteomics) for the six doses of radiation used in this study (22). A logistic regression model was used to develop a four-analyte panel to stratify irradiated from unirradiated NHPs with high accuracy that was agnostic for all six doses of γ radiation. This panel was comprised of acetylcarnitine, PC (16:0/22:6), Serpin Family A9, and suberylglycine, which showed 2–4-fold elevation in serum abundance upon irradiation in NHPs.
The results of our study will be invaluable to future studies conducting MCM evaluation against different lethalities of radiation using male and female animals and medical management with blood transfusions. It is important to note that several variables, such as radiation source/quality, exposure geometry, and medical management affect the outcome of lethal irradiation.
Supplementary Material
Supplementary data. Multiplex analysis of cytokines.
Supplementary Fig. S1. Effects of various radiation doses on serum biochemistry: albumin, alkaline phosphatase (ALKP), alanine transaminase (ALT), amylase, aspartate aminotransferase (AST), blood urea nitrogen (BUN), calcium, and cholesterol concentrations in irradiated NHPs. The data for each time point is presented as the mean for each radiation dose group.
Supplementary Fig. S2. Effects of various radiation doses on chloride, creatine kinase, carbon dioxide (CO2), creatinine, gamma glutamyl transferase (GGT), glucose, lactate dehydrogenase (LDH), and lipase concentrations in irradiated NHPs. The data for each time point is presented as the mean for each radiation dose group.
Supplementary Fig. S3. Effects of various radiation doses on phosphorus, potassium, sodium, total bilirubin, total protein, triglycerides, and uric acid concentrations in irradiated NHPs. The data for each time point is presented as the mean for each radiation dose group.
Supplementary Fig. S4. Effects of various radiation doses on cytokines: IL-4, IL-7, IL-13, G-CSF, MCP-1 (MCAF), and M-CSF concentrations in serum samples of irradiated NHPs. The data for each time point is presented as the mean for each radiation dose group.
Supplementary Fig. S5. Effects of various radiation doses on cytokines: IL-18, SCGF-β, PDGF-BB, and IL-1β concentrations in irradiated NHPs. The data for each time point is presented as the mean for each radiation dose group.
Supplementary Fig. S6. Effects of various radiation doses on IL-2, FGF Basic, MIP-1β, RANTES, VEGF, GROα, IL-2Rα, and LIF in irradiated NHPs. The data for each time point is presented as the mean for each radiation dose group.
Supplementary Fig. S7. Effects of various radiation doses on MIF, β-NGF, SCF, SDF-1α, HGF, and IL-1Rα in irradiated NHPs. The data for each time point is presented as the mean for each radiation dose group.
Supplementary Fig. S8. Effects of various radiation doses on heart rate, temperature, systolic blood pressure, diastolic blood pressure, and weight in irradiated NHPs. The data for each time point is presented as the mean for each radiation dose group.
ACKNOWLEDGMENTS
The authors would like to thank the staff of the Veterinary Science Department, AFRRI; the Comparative Medicine Department of the University of Maryland, Baltimore for animal care; and the staff of the Radiation Science Department, AFRRI, for dosimetry and radiation exposure to the animals. We also thank Dr. Carmen Rios and Dr. Andrea DiCarlo-Cohen for scientific discussions. The authors gratefully acknowledge the research support from the National Institute of Allergy and Infectious Diseases (AAI12044-001-07000, Work plan G) to VKS as a part of an Interagency-Agreement which was administered by The Henry M. Jackson Foundation for the Advancement of Military Medicine, Inc. All procedures were approved by the IACUC of AFRRI, The University of Maryland, Baltimore, and Department of Defense Animal Care and Use Review Office (ACURO). This study was carried out in strict accordance with the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health. The opinions or assertions contained herein are the private views of the authors and are not necessarily those of the Uniformed Services University of the Health Sciences, or the Department of Defense. The mention of specific therapeutic agents does not constitute endorsement by the U.S. Department of Defense, and trade names are used only for the purpose of clarification.
Footnotes
Editor’s note. The online version of this article (DOI: https://doi.org/10.1667/RADE-22-00101.1) contains supplementary information that is available to all authorized users.
REFERENCES
- 1.Gale RP. Medical and policy considerations for nuclear and radiation accidents, incidents and terrorism. Curr Opin Hematol 2017; 24:496–501. [DOI] [PubMed] [Google Scholar]
- 2.Gale RP, Armitage JO. Are we prepared for nuclear terrorism? N Engl J Med 2018; 378:1246–54. [DOI] [PubMed] [Google Scholar]
- 3.Andersson KG, Mikkelsen T, Astrup P, Thykier-Nielsen S, Jacobsen LH, Schou-Jensen L, et al. Estimation of health hazards resulting from a radiological terrorist attack in a city. Radiat Prot Dosimetry 2008; 131:297–307. [DOI] [PubMed] [Google Scholar]
- 4.McCann DGC. Radiation poisoning: Current concepts in the acute radiation syndrome. Am J Clin Med 2006; 3:13–21. [Google Scholar]
- 5.U.S. Food and Drug Administration. Animal Rule approvals.2021. Available at: https://www.fda.gov/drugs/nda-and-bla-approvals/animal-rule-approvals [Last accessed 2021].
- 6.Farese AM, MacVittie TJ. Filgrastim for the treatment of hematopoietic acute radiation syndrome. Drugs Today (Barc) 2015; 51:537–48. [DOI] [PubMed] [Google Scholar]
- 7.Singh VK, Seed TM. An update on sargramostim for treatment of acute radiation syndrome. Drugs Today (Barc) 2018; 54:679–93. [DOI] [PubMed] [Google Scholar]
- 8.Wong K, Bunin DI, Bujold K, Javitz HS, Bakke J, Gahagen J, et al. Romiplostim (Nplate) alone and in combination with pegfilgrastim (Neulasta) increased survival and reduces incidence, duration, and severity of thrombocytopenia post-iradiation in non-human primates. Annual Conference of Radiation Research Society. Virtual; 2020. [Google Scholar]
- 9.Singh VK, Seed TM. Radiation countermeasures for hematopoietic acute radiation syndrome: growth factors, cytokines and beyond. Int J Radiat Biol 2021; 97:1526–47. [DOI] [PubMed] [Google Scholar]
- 10.Hankey KG, Farese AM, Blaauw EC, Gibbs AM, Smith CP, Katz BP, et al. Pegfilgrastim improves survival of lethally irradiated nonhuman primates. Radiat Res 2015; 183:643–55. [DOI] [PubMed] [Google Scholar]
- 11.Clayton NP, Khan-Malek RC, Dangler CA, Zhang D, Ascah A, Gains M, et al. Sargramostim (rhu GM-CSF) improves survival of non-human primates with severe bone marrow suppression after acute, high-dose, whole-body irradiation. Radiat Res 2021; 195:191–9. [DOI] [PubMed] [Google Scholar]
- 12.Zhong Y, Pouliot M, Downey AM, Mockbee C, Roychowdhury D, Wierzbicki W, et al. Efficacy of delayed administration of sargramostim up to 120 hours post exposure in a nonhuman primate total body radiation model. Int J Radiat Biol 2021; 97:S100–S16. [DOI] [PubMed] [Google Scholar]
- 13.Singh VK, Seed TM. An update on romiplostim for treatment of acute radiation syndrome. Drugs Today (Barc) 2022; 58:133–45. [DOI] [PubMed] [Google Scholar]
- 14.Farese AM, Cohen MV, Katz BP, Smith CP, Gibbs A, Cohen DM, et al. Filgrastim improves survival in lethally irradiated nonhuman primates. Radiat Res 2013; 179:89–100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Singh VK, Olabisi AO. Nonhuman primates as models for the discovery and development of radiation countermeasures. Expert Opin Drug Discov 2017; 12:695–709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Williams JP, Brown SL, Georges GE, Hauer-Jensen M, Hill RP, Huser AK, et al. Animal models for medical countermeasures to radiation exposure. Radiat Res 2010; 173:557–78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Singh VK, Newman VL, Berg AN, MacVittie TJ. Animal models for acute radiation syndrome drug discovery. Expert Opin Drug Discov 2015; 10:497–517. [DOI] [PubMed] [Google Scholar]
- 18.Anno GH, Young RW, Bloom RM, Mercier JR. Dose response relationships for acute ionizing-radiation lethality. Health Phys 2003; 84:565–75. [DOI] [PubMed] [Google Scholar]
- 19.Graessle DH, Dorr H, Bennett A, Shapiro A, Farese AM, MacVittie TJ, et al. Comparing the hematopoetic syndrome time course in the NHP animal model to radiation accident cases from the database search. Health Phys 2015; 109:493–501. [DOI] [PubMed] [Google Scholar]
- 20.Dorr H, Lamkowski A, Graessle DH, Bennett A, Shapiro A, Farese AM, et al. Linking the human response to unplanned radiation and treatment to the nonhuman primate response to controlled radiation and treatment. Health Phys 2014; 106:129–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Singh VK, Newman VL, Romaine PL, Hauer-Jensen M, Pollard HB. Use of biomarkers for assessing radiation injury and efficacy of countermeasures. Expert Rev Mol Diagn 2016; 16:65–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Cheema AK, Li Y, Moulton J, Girgis M, Wise SY, Carpenter A, et al. Identification of novel biomarkers for acute radiation injury using multi-omics approach and nonhuman primate model. Int J Radiat Oncol Biol Phys 2022; 114:310–30. [DOI] [PubMed] [Google Scholar]
- 23.MacVittie TJ, Farese AM, Jackson W 3rd. The hematopoietic syndrome of the acute radiation syndrome in rhesus macaques: A systematic review of the lethal dose response relationship. Health Phys 2015; 109:342–66. [DOI] [PubMed] [Google Scholar]
- 24.MacVittie TJ, Farese AM, Jackson WE 3rd. A systematic review of the hematopoietic acute radiation syndrome (H-ARS) in canines and non-human primates: Acute mixed neutron/gamma vs. reference quality radiations. Health Phys 2020; 119:527–58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Bell BL, Vercellino J, Brodin NP, Velten C, Nanduri LSY, P.K.B.N, et al. Orthovoltage x-rays exhibit increased efficacy compared with γ-rays in preclinical irradiation. Cancer Res 2022; 82:2678–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Thrall KD, Love R, O’Donnell KC, Farese AM, Manning R, MacVittie TJ. An interlaboratory validation of the radiation dose response relationship (DRR) for H-ARS in the rhesus macaque. Health Phys 2015; 109:502–10. [DOI] [PubMed] [Google Scholar]
- 27.Yu JZ, Lindeblad M, Lyubimov A, Neri F, Smith B, Szilagyi E, et al. Subject-Based versus Population-Based Care after Radiation Exposure. Radiat Res 2015; 184:46–55. [DOI] [PubMed] [Google Scholar]
- 28.U.S. Food and Drug Administration. Guidance document: Product development under the Animal Rule. 2015. Available at: https://bit.ly/3CfmqFO [Last accessed 2020].
- 29.Singh VK, Kulkarni S, Fatanmi OO, Wise SY, Newman VL, Romaine PL, et al. Radioprotective efficacy of gamma-to-cotrienol in nonhuman primates. Radiat Res 2016; 185:285–98. [DOI] [PubMed] [Google Scholar]
- 30.National Research Council of the National Academy of Sciences. Guide for the care and use of laboratory animals. 8th ed. Washington, DC: National Academies Press; 2011. [Google Scholar]
- 31.Nagy V Accuracy considerations in EPR dosimetry. Appl Radiat Isot 2000; 52:1039–50. [DOI] [PubMed] [Google Scholar]
- 32.International Standardization Organization and ASTM International. Standard Practice for Use of an Alanine-EPR Dosimetry System. Geneva, Switzerland: ASTM International, ISO and West Conshohocken (US:PA):; 2013. p. 7. [Google Scholar]
- 33.Farese AM, Cohen MV, Katz BP, Smith CP, Jackson W 3rd,Cohen DM, et al. A nonhuman primate model of the hematopoietic acute radiation syndrome plus medical management. Health Phys 2012; 103:367–82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Association AVM. AVMA Guidelines for the Euthanasia of Animals: 2020 Edition. 2020. Available at: https://www.avma.org/sites/default/files/2020-01/2020-Euthanasia-Final-1-17-20.pdf [Last accessed [Google Scholar]
- 35.Kulkarni S, Singh PK, Ghosh SP, Posarac A, Singh VK. Granulocyte colony-stimulating factor antibody abrogates radio-protective efficacy of gamma-tocotrienol, a promising radiation countermeasure. Cytokine 2013; 62:278–85. [DOI] [PubMed] [Google Scholar]
- 36.Oehlert GW. A Note on the Delta Method. The American Statistician 1992; 46:27–9. [Google Scholar]
- 37.Kim JW, Lee DW, Choi WH, Jeon YR, Kim SH, Cho H, et al. Development of a porcine skin injury model and characterization of the dose-dependent response to high-dose radiation. J Radiat Res 2013; 54:823–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.King GL, Rabin BM, Weatherspoon JK. 5-HT3 receptor antagonists ameliorate emesis in the ferret evoked by neutron or proton radiation. Aviat Space Environ Med 1999; 70:485–92. [PubMed] [Google Scholar]
- 39.Beach T, Authier S, Javitz HS, Wong K, Bakke J, Gahagen J, et al. Total body irradiation models in NHPs - consideration of animal sex and provision of supportive care to advance model development. Int J Radiat Biol 2021; 97:126–30. [DOI] [PubMed] [Google Scholar]
- 40.MacVittie TJ, Farese AM, Parker GA, Jackson W, 3rd, Booth C, Tudor GL, et al. The Gastrointestinal Subsyndrome of the Acute Radiation Syndrome in Rhesus Macaques: A Systematic Review of the Lethal Dose-response Relationship With and Without Medical Management. Health Phys 2019; 116:305–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Eltringham JR. Recovery of the rhesus monkey from acute radiation exposure as evaluated by the split dose technique: preliminary results. Radiat Res 1967; 31:533. [Google Scholar]
- 42.Dalrymple GV, Lindsay IR, Ghidoni JJ. The Effect of 2-MevWhole-Body X-Irradiation on Primates. Radiat Res 1965; 25:377–400. [PubMed] [Google Scholar]
- 43.Waselenko JK, MacVittie TJ, Blakely WF, Pesik N, Wiley AL, Dickerson WE, et al. Medical management of the acute radiation syndrome: Recommendations of the Strategic National Stockpile Radiation Working Group. Ann Intern Med 2004; 140:1037–51. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary data. Multiplex analysis of cytokines.
Supplementary Fig. S1. Effects of various radiation doses on serum biochemistry: albumin, alkaline phosphatase (ALKP), alanine transaminase (ALT), amylase, aspartate aminotransferase (AST), blood urea nitrogen (BUN), calcium, and cholesterol concentrations in irradiated NHPs. The data for each time point is presented as the mean for each radiation dose group.
Supplementary Fig. S2. Effects of various radiation doses on chloride, creatine kinase, carbon dioxide (CO2), creatinine, gamma glutamyl transferase (GGT), glucose, lactate dehydrogenase (LDH), and lipase concentrations in irradiated NHPs. The data for each time point is presented as the mean for each radiation dose group.
Supplementary Fig. S3. Effects of various radiation doses on phosphorus, potassium, sodium, total bilirubin, total protein, triglycerides, and uric acid concentrations in irradiated NHPs. The data for each time point is presented as the mean for each radiation dose group.
Supplementary Fig. S4. Effects of various radiation doses on cytokines: IL-4, IL-7, IL-13, G-CSF, MCP-1 (MCAF), and M-CSF concentrations in serum samples of irradiated NHPs. The data for each time point is presented as the mean for each radiation dose group.
Supplementary Fig. S5. Effects of various radiation doses on cytokines: IL-18, SCGF-β, PDGF-BB, and IL-1β concentrations in irradiated NHPs. The data for each time point is presented as the mean for each radiation dose group.
Supplementary Fig. S6. Effects of various radiation doses on IL-2, FGF Basic, MIP-1β, RANTES, VEGF, GROα, IL-2Rα, and LIF in irradiated NHPs. The data for each time point is presented as the mean for each radiation dose group.
Supplementary Fig. S7. Effects of various radiation doses on MIF, β-NGF, SCF, SDF-1α, HGF, and IL-1Rα in irradiated NHPs. The data for each time point is presented as the mean for each radiation dose group.
Supplementary Fig. S8. Effects of various radiation doses on heart rate, temperature, systolic blood pressure, diastolic blood pressure, and weight in irradiated NHPs. The data for each time point is presented as the mean for each radiation dose group.