To the Editor,
The majority of the general population, particularly, servicemen and women deployed across the globe are exposed to various airborne hazards, including diesel exhaust particulate (DEP) and desert sand dust (DSD). 1 Notably, both DEP and DSD exhibit high reactivity and toxicity and are found to be associated with several pathological conditions, including respiratory, cardiovascular, and neurovascular dysfunction. 2 , 3 Evidence from in vitro and in vivo toxicological studies indicate that exposure to both DEP and DSD induces pathologies, in part, via the activation of multiple signaling cascades. 4 , 5 Importantly, epithelial cells from the respiratory tract are common sites for initial exposure to DEP and DSD. Exposure to these xenobiotics has been correlated to pulmonary oxidative stress via induction in reactive oxygen species (ROS) and reactive nitrogen species generation. 5 However, the fundamental mechanisms by which these hazardous pollutants act initially on the epithelium promoting both local and systemic disease remains unclear – making prevention, diagnosis, and treatment difficult.
Studies, including ours, have shown that exposure to ROS‐generating pro‐oxidative stressors ranging from environmental carcinogens to pollutants, including cigarette smoke, produces a class of potent phospholipid mediators, Platelet‐activating factor (PAF) and PAF‐like agonists via oxidative cleavage of lipid membrane glycerophosphocholines (GPCs). 6 , 7 These PAF agonists mediate their effects via binding to a widely‐expressed single G‐protein coupled, PAF‐receptor (PAFR). 6 , 7 Of importance, recent studies, including from our group, have implicated the significance of small membrane‐bound extracellular vesicles, known as microvesicle particles (MVPs), which are released in response to various stimuli from diverse cell types. 8 , 9 These MVPs contain various bioactive components, including cytokines and lipids such as PAF, and play a vital role as a mediator of cell‐to‐cell communications in diverse biological and pathological conditions. 8 , 9
Given that various pro‐oxidative stressors induce ROS generation and cell damage, which are known triggers of PAF agonist synthesis and MVP release via activation of the PAFR, 6 , 7 the current studies were designed to test the hypothesis that exposure to DEP and DSD could result in MVP release and PAF agonists generation. As lung epithelial cells are a primary target of DEP and DSD, we utilized the human nasopharyngeal cell line KB that either expressed PAFR (KBP) or was PAFR negative (KBM). 6 These cell lines were treated with various concentrations of DEP and DSD alone or in combination with a lower concentration of DEP + DSD. Cells treated with Hanks balanced salt solution with 1% BSA as a vehicle served as a negative control. While the known PAFR agonist, carbamyl‐PAF (CPAF), and PAFR‐independent agonist, phorbol myristate acetate (PMA) served as positive controls. After 4 hours of incubation, MVPs were isolated from the supernatants by differential centrifugation as previously reported. 8 , 9 The size and concentrations of MVP were analyzed by nanoparticles tracking analysis (NTA) using a NanoSight NS300 instrument as per our published protocols. 8 , 9
Our studies demonstrate that both DEP and DSD stimulated MVP release from KBP cells in a concentration‐dependent manner. Additionally, the KBP cells exhibited an increased additive effect in MVP release in response to exposure to the combined mixture of DEP + DSD at a final concentration of 25 ug/ml (12.5 ug/ml DEP + 12.5 ug/ml DSD) (Figure 1A). Notably, both CPAF and PMA induced an increased MVP release in KBP cells, indicating the possible role of the PAFR signaling in both DEP‐ and DSD‐induced effects (Figure 1A). To confirm the PAFR dependency, similar studies were performed in PAFR‐negative KBM cells, which demonstrated that neither DEP, DSD nor CPAF, but only PMA treatment resulted in MVP release (Figure 1B). To further determine if DEP and DSD‐induced MVP release in KBP cells are cell‐associated and not an artifact from the heterogeneous particles in these agents, similar studies were performed without the cells. We observed no MVP release in the DEP and DSD‐containing solutions alone (Figure 1C), confirming that MVP release was cell‐associated. Overall, these studies indicate that DEP and DSD induce MVP release in a PAFR‐dependent manner.
FIGURE 1.
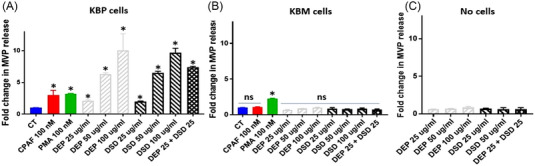
Diesel exhaust particulate (DEP) and desert sand dust (DSD) stimulate microvesicle particle (MVP) release. (A) KBP and (B) KBM cell lines or (C) media only (no cells) were treated with or without carbamyl‐platelet‐activating factor (CPAF) (100 nM), phorbol myristate acetate (PMA) (100 nM), or DEP and DSD at 25, 50, and 100 μg/ml concentrations, or a combination of DEP + DSD at 25 μg/ml final concentration (12.5 ug/ml DEP + 12.5 ug/ml DSD). MVPs released into supernatant were measured at 4 h. The data are mean ± SD from 3 separate experiments, normalized per 1 × 106 cells. *Statistically (p < 0.05) significant changes, and NS: non‐significant from control values.
Our next studies evaluated the effects of DEP and DSD exposure on PAF agonists generation using a human interleukin‐8 (IL‐8) ELISA assay. IL‐8 serves as a surrogate marker of total PAFR agonistic activity in the complex lipid mixture (Figure 2A) from KBP cells in response to diverse stimuli. 8 , 9 For these studies, we treated KBP cells with the lower concentration of DEP or DSD, the vehicle as a negative control, and CPAF as a positive control. After 4 h of incubation, IL‐8 protein released in the supernatants were measured, and presented as %CPAF response. As shown in Figure 2B, the treatments of both DEP and DSD stimulated IL‐8 release as compared to vehicle control treatment. Similar studies in KBM cells demonstrated no induction in PAFR agonistic activity by DEP, DSD, or CPAF (data not shown). These studies indicate that both DEP and DSD stimulate PAF agonist generation in a PAFR‐dependent fashion.
FIGURE 2.
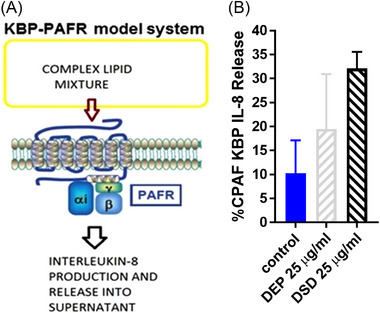
Diesel exhaust particulate (DEP) and desert sand dust (DSD) stimulate platelet‐activating factor‐receptor (PAFR) agonistic activity. (A) Schematic representation of KBP‐PAFR model system. (B) KBP cells were treated with or without carbamyl‐PAF (CPAF) (100 nM), DEP, and DSD at 25 μg/ml concentration. Interleukin‐8 (IL‐8) released into the supernatant was measured at 4 h. The data are representative of mean ± SD from three separate experiments, expressed as %CPAF KBP IL‐8 release.
Overall, these studies provide a novel mechanism by which potent airborne hazards generate MVP and PAF agonists in a PAFR‐dependent manner via induction of a pro‐oxidative stressor effect. As PAF agonists are rapidly metabolized by acetylhydrolases, these studies suggest that MVPs serve to not only protect PAF agonists from degradation but may also act as a signaling mechanism by permitting circulation of PAF agonists to exert local and systemic effects, as evidenced by our published reports. 8 , 9 Notably, DEP and DSD‐induced inflammatory signaling pathways appear to be similar to those profiles associated with the PAFR activation in the skin, vascular endothelium, and lung. 10 Thus, this novel pathway provides impetus to translate toxic airborne hazard exposures to epithelial‐lined organs into systemic effects and can be blocked via pharmacologic agents (e.g., inhibitors of MVP generation).
CONFLICT OF INTEREST STATEMENT
The authors declare no conflict of interest.
ACKNOWLEDGMENTS
This research was supported in part by grants from the National Institutes of Health grants R21 ES033806 (RPS), R01 HL062996 (J.B.T.), Elsa U. Pardee Foundation grant 671432 (RPS), and Veteran's Administration Merit Award 5I01BX000853 (J.B.T.).
DATA AVAILABILITY STATEMENT
The author has provided the required Data Availability Statement, and if applicable, included functional and accurate links to said data therein.
REFERENCES
- 1. Falvo MJ, Osinubi OY, Sotolongo AM, Helmer DA. Airborne hazards exposure and respiratory health of Iraq and Afghanistan veterans. Epidemiol Rev. 2015;37:116–30. [DOI] [PubMed] [Google Scholar]
- 2. Dursa EK, Tadesse BE, Carter CE, Culpepper WJ, Schneiderman AI, Rumm PD. Respiratory illness among Gulf War and Gulf War era veterans who use the Department of Veterans Affairs for healthcare. Am J Ind Med. 2020;63(11):980–7. [DOI] [PubMed] [Google Scholar]
- 3. Hinojosa R. Veterans' likelihood of reporting cardiovascular disease. J Am Board Fam Med. 2019;32(1):50–7. [DOI] [PubMed] [Google Scholar]
- 4. Wilfong ER, Lyles M, Rietcheck RL, et al. The acute and long‐term effects of Middle East sand particles on the rat airway following a single intratracheal instillation. J Toxicol Environ Health A. 2011;74(20):1351–65. [DOI] [PubMed] [Google Scholar]
- 5. Daniel S, Phillippi D, Schneider LJ, Nguyen KN, Mirpuri J, Lund AK. Exposure to diesel exhaust particles results in altered lung microbial profiles, associated with increased reactive oxygen species/reactive nitrogen species and inflammation, in C57Bl/6 wildtype mice on a high‐fat diet. Part Fibre Toxicol. 2021;18(1):3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Sahu RP, Petrache I, Van Demark MJ, et al. Cigarette smoke exposure inhibits contact hypersensitivity via the generation of platelet activating factor agonists. J Immunol. 2013;190(5):2447–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Sahu RP, Turner MJ, DaSilva SC, et al. The environmental stressor ultraviolet B radiation inhibits murine antitumor immunity through its ability to generate platelet‐activating factor agonists. Carcinogenesis. 2012;33(7):1360–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Liu L, Awoyemi AA, Fahy KE, et al. Keratinocyte‐derived microvesicle particles mediate ultraviolet B radiation‐induced systemic immunosuppression. J Clin Invest. 2021;131(10):e144963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Thyagarajan A, Awasthi K, Rapp CM, et al. Topical application of gemcitabine generates microvesicle particles in human and murine skin. Biofactors. 2022;48(6):1295–304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Serban KA, Rezania S, Petrusca DN, et al. Structural and functional characterization of endothelial microparticles released by cigarette smoke. Sci Rep. 2016;6:31596. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The author has provided the required Data Availability Statement, and if applicable, included functional and accurate links to said data therein.


