ABSTRACT
Sclerosing cholangitis (SC) represents a spectrum of chronic progressive cholestatic diseases of the intrahepatic and/or extrahepatic biliary system characterized by patchy inflammation, fibrosis, and stricturing. Primary and secondary SC must be distinguished given the different treatment modalities, risks of malignancy, and progression to portal hypertension, cirrhosis, and hepatic failure. This review focuses on secondary SC and the pathogenic mechanisms, risk factors, clinical presentation, and novel imaging modalities that help to distinguish between these conditions. We explore the detailed use of cholangiography and ultrasound imaging techniques.
Key words: Cholangiography, IgG4-related sclerosing cholangitis, secondary cholangitis-critically Ill patients, secondary sclerosing cholangitis, ultrasound
INTRODUCTION
The World Federation for Ultrasound in Medicine and Biology has published guidelines on the use of contrast-enhanced ultrasound (CEUS) for the evaluation of focal liver lesions[1-5] and the European Federation of Societies for Ultrasound in Medicine provided guidance for CEUS for the evaluation of nonhepatic indications.[6,7] A series of publications have been prepared to illustrate the guidelines.[8,9] Ultrasound of the biliary system is a standard method in daily routine, but there are only a few publications describing the role and typical findings of ultrasound diagnostics in the rare disease group of sclerosing cholangitis (SC). Diagnosis is usually made by cholangiographic methods. Therefore, in this article, we review the significance of ultrasound in cholangiopathies.
SC is a group of diseases with progressive narrowing and destruction of the bile ducts due to diffuse inflammation and fibrosis. Sclerosing cholangiopathies are rare and have variable etiology and prognosis. A distinction is made between primary SC (PSC) and secondary SC (SSC).
PSC is an immune-mediated disorder and considered a premalignant disease with increased risk for malignancy, especially of the bile duct and bowel. The etiology seems multifactorial including genetic predisposition, exposure to environmental antigens, and subsequent pathological immunologic response to that trigger. It has a two-peaked distribution of ages at presentation and is often (60%–80%) associated with inflammatory bowel disease,[10-12] predominantly ulcerative colitis. PSC can lead to liver cirrhosis, portal hypertension, and the need for liver transplantation.[11,13-16]
SSC has a number of well-defined causes with several different etiological factors leading to chronic inflammation and subsequent fibrosis and damage of the bile ducts. These include infections, medications, ischemia, obstruction, and autoimmune diseases[17-20] [Table 1]. For the purpose of this review, we have classified immunoglobulin G4-related SC (IgG4-SC) as an autoimmune-mediated subtype of SC. However, it has also be listed as a separate disease group by some studies.[18,21] SSCs have similar clinical features, and patients present with cholestatic symptoms, including jaundice and pruritus, and an increase in cholestatic enzymes. However, progression of the disease is variable.
Table 1.
Etiology of sclerosing cholangitis
| Ischemic |
| Critical illness patients (SSC-CIP) |
| Postliver transplantation (liver artery stenosis or thrombosis; ischemic-type biliary lesions) |
| Intraarterial chemotherapy induced |
| Hereditary hemorrhagic telangiectasia |
| Panarteriitis nodosa |
| Vascular trauma |
| Toxic or drug-induced |
| Chemotherapeutics (docetaxel), immune checkpoint inhibitors (pembrolizumab and nivolumab) |
| Chronic ketamine abuse, methimazole, moxifloxacin, and atorvastatin |
| Postoperative biliary injuries |
| Miscellaneous |
| Caroli’s disease |
| Mucoviscidosis |
| Langerhan’s cell histiocytosis |
| Infectious |
| Recurrent pyogenic infection |
| Parasitic (clonorchis sinensis and ascaris lumbricoides, echinococcus multilocularis) |
| AIDS-related opportunistic infections (CMV, cryptosporidium, microsporidia, isospora, mycobacterium avium intracellulare, and giardia) |
| Post-COVID |
| Immunological causes |
| Eosinophilic cholangiopathy |
| Immune checkpoint inhibitors |
| Behcet’s disease |
| Sarcoidosis |
| IgG4-SC |
SSC-CIP: Secondary sclerosing cholangitis in critically ill patients; AIDS: Acquired immune deficiency syndrome; IgG4-SC: Immunoglobulin G4-related sclerosing cholangitis; CMV: Cytomegalovirus; COVID: Coronavirus disease
The aim of this review is to provide a comprehensive overview of secondary SC including IgG4-SC. In addition to conventional imaging of the bile ducts by magnetic resonance cholangiopancreatography (MRCP) and ERCP, the diagnostic value of ultrasound and CEUS to discriminate SC is discussed.
ISCHEMIC CHOLANGITIS
Ischemia-induced bile duct alterations are referred to under the term ischemic cholangitis. Conventionally, an acute ischemic event can trigger formation of stenosing bile duct changes, usually intrahepatic, in the bile duct bifurcation and in the middle part of the common bile duct. This leads to the development of casts, biliomas, and not uncommonly, acalculous cholecystitis.[17,22-26] Increasingly reported is the secondary SC in critically ill patients (SSC-CIP) in association with intensive care treatment.[25-30] Other established causes of ischemic cholangiopathy include liver transplantation, intra-arterial chemotherapy in the hepatic artery, long-term ventilation, acquired immune deficiency syndrome (AIDS)-related cholangiopathy, local irradiation, hereditary hemorrhagic telangiectasia, and polyarteritis nodosa.[17,22] Possible causes of ischemic cholangiopathy are systemic diseases with microvascular involvement, for example, Schoenlein-Henoch purpura, sickle cell disease, Kawasaki disease, systemic lupus, antiphospholipid syndrome, paroxysmal nocturnal hemoglobinuria, and hypereosinophilic syndrome.[31] Most disease patterns have a multifactorial genesis, potentially also including hypercoagulopathy. Vascular injury during cholecystectomy can cause biliary strictures.
BILIARY ANATOMY AND BLOOD SUPPLY
While the hepatic parenchyma receives a dual blood supply through the hepatic artery and portal vein, the bile ducts receive only an arterial blood supply. The bile duct system is supplied by the branches of the common hepatic artery, the proper hepatic artery, the left and right hepatic artery, the cystic artery, the gastroduodenal as well as retroduodenal and retro portal arteries.[31,32] The superior and middle portions of the common bile duct are supplied axially by two main arteries that traverse the lateral margins. These vascular structures are referred to as “parabiliary arteries.” This segment is especially vulnerable to disorders of the arterial blood supply.[32,33] In contrast, the right and left hepatic ducts and the intrahepatic ducts are supplied by a rich nonaxial network of small arteries derived from the right and left hepatic arteries. A plexus of arterioles, venules, and capillaries exists in the peribiliary adventitia, with the arterial supply originating from hepatic arterial branches. Within the bile duct wall is a second plexus, which consists mainly of capillaries.[32] The peribiliary plexus (PBP) is supplied by the terminal hepatic artery branches and surrounds the bile ducts.[22]
The biliary epithelium is susceptible to injury when arterial blood flow is impaired. This impairment may occur at the level of the large, so-called hepatic arterial branches or at the microscopic level of the peribiliary capillary plexus. The middle third of the common bile duct and the biliary confluence appear to be most vulnerable to ischemic damage.
A distinction must be made between ischemic cholangiopathy and impaired bile formation under ischemic conditions. Hepatic arterial ischemia reduces the expression and function of hepatocellular transporters for biliary compounds, resulting in cholestasis.[22] This corresponds to the concept of “toxic bile.”[26,34]
LIVER TRANSPLANTATION
Nonanastomotic biliary duct strictures and necrosis of the bile ducts are both well-described complications of liver transplantation. This may be caused by the occlusion of the hepatic artery but also by nonthrombotic factors. Among 522 liver transplant patients, 4.8% developed hepatic artery thrombosis.[35] Zheng et al. saw hepatic artery stenosis in 39.0% of 172 liver transplant recipients with a median percentage of stenosis of 75.4%.[36] These are threatening events that require immediate intervention.
Ischemic cholangitis after liver transplantation usually occurs in the first 12 months after transplantation. Potential factors include prolonged cold ischemic time, delayed rearterialization of the graft, or grafts of donation after circulatory death (DCD), and ischaemia-reperfusion injury. The incidence for biliary complications and ischemic cholangitis ranges from 16% to 29% for DCD grafts versus 3% to 17% for donation after brain death (DBD grafts). The immunologic causes of ischemic cholangitis in liver grafts are damage to the bile duct epithelium and/or vascular endothelium due to chronic rejection, cytomegalovirus (CMV) infection, and ABO-incompatible transplantation.[37,38]
SECONDARY SCLEROSING CHOLANGITIS IN CRITICALLY ILL PATIENTS
This clinical picture develops after life-threatening events and intensive care therapy. These are, for example, extensive burns, trauma, or major surgery with extensive tissue damage. Whether the initial event is causative or the intensive care treatment varies. Risk factors include shock states with decreased mean arterial pressure, use of catecholamines, mechanical ventilation, ventilation strategies with high positive end-expiratory pressure (PEEP), paO2/FiO2 <150 mmHg, and disturbances in the microcirculation. Increased blood viscosity caused by colloids or myoglobin release, erythrocyte transfusion, and hypercoagulable are other factors affecting microcirculatory blood flow in the PBP.
Liver biochemistry shows cholestasis with an elevated gamma-glutamyl-transferase and alkaline phosphatase; an increase in bilirubin usually occurs later. The development of SSC takes some time. While cholestasis parameters usually increase in the 1st days after the acute event, the diagnosis of secondary sclerosing cholangitis in critically ill patients (SCC-CIP) is made within the 1st year.[26,30]
Gelbmann et al.[39] reported that all patients with cholestasis and SSC suffered from severe respiratory insufficiency with the need for mechanical ventilation (40.7 ± 32.9 days). The PaO2/FiO2 ratio until the beginning of cholestasis was 150.5 ± 43.1. Half of the patients were treated with high-frequency oscillatory ventilation and 12/17 patients by intermittent prone positioning. All patients required catecholamines for hemodynamic stabilization.[39] Jaeger et al. noted cholestasis after a median time of 6.5 days (range 4–11 days) following the initial insult.[29] Leonhardt et al. reported elevated liver enzymes of a cholestatic pattern after a mean of 7.86 ± 2.98 days (median seven days). Between the first event and the onset of cholestasis, all patients suffered an episode of severe hemodynamic instability with a reduction in mean arterial pressure to below 65 mm Hg that lasted at least 60 min, often more than 120 min. This was the most important pathogenetic factor. It was concluded that bile duct destruction in SSC-CIP is the result of a single event in which a large amount of cholangiocytes is directly and irreversibly damaged. However, the authors noted that this decrease in arterial pressure appears to contribute to critical perfusion of the PBP only when additional microcirculatory disturbances (colloids, erythrocytes, hypercoagulable state, and increased plasma viscosity) are present.[26]
Leonhardt et al. evaluated SIRS/sepsis, ischemia, high dose propofol, antimicrobials, and total parenteral nutrition as risk factors that may lead to bile duct injury (BDI) from “toxic bile” through alterations in hepatobiliary transporters. However, in the reported patient group, neither high-dose catecholamines nor total parenteral nutrition played a clear role in triggering SSC-CIP.[26] A study by Ben-Ari et al. examined patients who developed SSC-CIP after a severe burn. All these patients were mechanically ventilated and required vasopressor support, and all patients had exhibited repeated episodes of severe hypotension, defined as a systolic blood pressure <70 mmHg.[30] In a study of patients with severe acute respiratory distress syndrome (ARDS) due to influenza A (H1N1) pneumonia, those who developed SSC-CIP were the individuals who had a higher body mass, higher volume of intraperitoneal fat, and spent more time in the prone position.[40]
The importance of ketamine is controversially discussed.[41,42] Cholangiopathy is reported after chronic exposure to ketamine, both in drug users and in patients with burns. Ketamine causes drug toxic cholangiopathy, which may be an additional factor in ischemic SSC-CIP. In ketamine cholangiopathies, dilatations and strictures of the intrahepatic and extrahepatic bile ducts, biliary sepsis, and decompensated cirrhosis have been described. Liver damage is reported to be dose-dependent and progressive. Ketamine is extensively metabolized in the liver.[42] The Keta-Cov Research Group does not recommend using ketamine to sedate patients with ARDS, including those with coronavirus disease 2019 (COVID-19). If forced to prescribe, ketamine should only be used for short periods and bilirubin levels should be monitored closely.[42]
There is a high risk of complications with SSC-CIP; 75% of patients had cholangiosepsis, cholangitic liver abscesses, acalculous cholecystitis, or perforation of the gallbladder during the course.[25]
INTRAARTERIAL CHEMOTHERAPY-INDUCED ISCHEMIC CHOLANGIOPATHY
Transarterial chemotherapy through the hepatic artery has been casuistically reported as causative of SC. Significant BDI occurs in 15%–20% of patients treated with transarterial chemotherapy with floxuridine.[38,43] Secondary SC has been reported after transcatheter arterial chemoembolization with bleomycin-iodized oil for cavernous hemangiomas in the liver.[44] The most toxic combination seems to consist of chemotherapy, lipiodol, and gelatin sponge.[22,45-47]
HEREDITARY HEMORRHAGIC TELANGIECTASIA AND PANARTERIITIS NODOSA
In patients with hereditary hemorrhagic telangiectasia, arteriovenous and arterioportal shunts can cause ischemic biliary lesions when they divert blood around the PBP.[22,48] Panarteriitis nodosa is characterized by the medium and small arterial inflammation. The hepatic arteries may also be affected.[49] In autopsy studies, involvement of the bile ducts has been diagnosed in 10%–25% of patients.[22]
IMAGING IN ISCHEMIC SECONDARY SCLEROSING CHOLANGITIS
Cholangiography
The diagnosis of ischemic cholangitis is based on cholangiography and consideration of the patient’s medical history. Biliary tract imaging is performed by MRCP and in case of expected indication for endoscopic treatment by ERCP. On MRCP and ERCP, suspicion is raised by irregularities of the bile ducts with dilatation and strictures, if other causes of biliary dilatation are excluded. In the early stages, MRCP shows defects in the intrahepatic biliary tree, which manifest as bile effusions and bile leaks, occasionally forming biliomas. Extensive biliary cast formation presenting as intraductal filling defects on MRCP, and endoscopic retrograde cholangiography (ERC) might be seen. Diffuse intrahepatic bile duct strictures are observed in later stages. Remarkably, the distal common bile duct is preserved in all stages.[23,30]
ERCP
Although ERCP is considered a therapeutic method, ERCP is the gold standard for the diagnosis of SSC-CIP.[25,27,30] In the study by Jaeger et al., the first ERCP was performed a median of 69 days after injury or insult (range, 42–126 days.[29] ERCP showed multifocal stricture and destruction of intrahepatic bile ducts in all patients. In more than 50%, only the intrahepatic bile ducts were affected. In the remainder, the right and/or left hepatic ducts were also affected. In all patients, the common bile duct was normally formed or only slightly dilated. Black pigmented casts were typical.[29] Gelbmann et al.[39] described biliary casts with impaired bile flow and subsequent biliary infection, in most cases with Enterococcus faecium, as the earliest endoscopic finding in ERCP. In the further course, progressive formation of multiple strictures of the bile ducts was observed.[39]
Leonhardt et al. describe the various cholangiographic features of SSC-CIP according to approximately the following stages: (I) evidence of biliary casts corresponding to ribbon-like intraductal filling defects, which may distend the bile ducts, (II) progressive destruction of intrahepatic bile ducts beyond the second bifurcation of the intrahepatic bile ducts, and (III) picture of pruned tree [Figure 1]. The intrahepatic bile duct branches are obliterated in the course, so that only a rudimentary central biliary system can be visualized. On ERC, contrast enhancement of the bile ducts is seen only below the second branch. At the later stage, the image was that of a pruned tree. At least one side of the biliary system was sclerosed, but usually both sides were affected.[25] Biliary casts formation was a specific feature and was seen in 87% of patients.[25] In the study of Leonhardt et al., SSC-CIP is a disease mainly visibly in the intrahepatic bile ducts − the extrahepatic bile ducts were only marginally affected.[25] Regarding the distribution of biliary strictures, Voigtländer et al.[28] described multiple diffuse intrahepatic biliary strictures in 67% of patients, both intra- and extrahepatic strictures in 20% and only extrahepatic ductal changes in 6%. Biliary casts were present in 93% of patients.[28]
Figure 1.
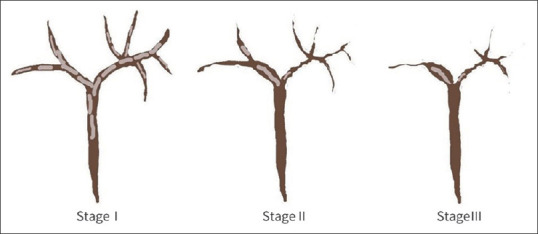
Cholangiographic stage of SCC-CIP according to Leonhardt et al. 2015.[25] Stage I: Multiple ribbon-like filling defects in the bile ducts (biliary casts). Stage II: rapidly progressive intrahepatic bile duct destruction beyond the second bifurcation of the intrahepatic bile ducts. Stage III: progressive destruction of the intrahepatic bile ducts, picture of a “pruned tree”. SCC-CIP: Secondary sclerosing cholangitis in critically ill patients
On MRCP, in analogy to ERCP, the patients of Ben-Ari et al. showed irregular intrahepatic bile ducts with multiple strictures and dilatations. In ERCP, the bile fluid of SSC-CIP patients was found to contain primarily Enterococcus faecium, Enterococcus faecalis, Pseudomonas aeruginosa, and non-albicans species of Candida.[27,28]
Ultrasound
The ultrasonographic changes of ischemic cholangitis are not specific. Ultrasound is used for differential diagnostic considerations in cases of elevated liver values such as the exclusion of biliary obstruction of other etiology or, for example, congested liver. Intrahepatic rarefication of the bile ducts is usually not seen on ultrasound until peripherally located branches are dilated. Ultrasound can visualize biliomas caused by the ischemic leaking of intrahepatic bile ducts [Figure 2].
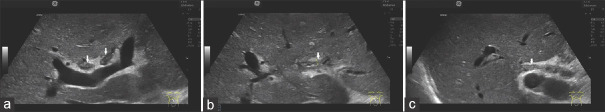
Figure 2.
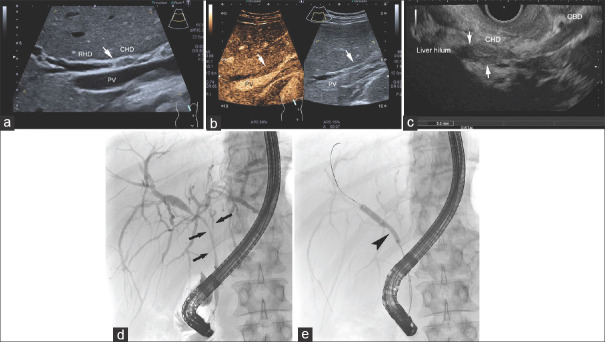
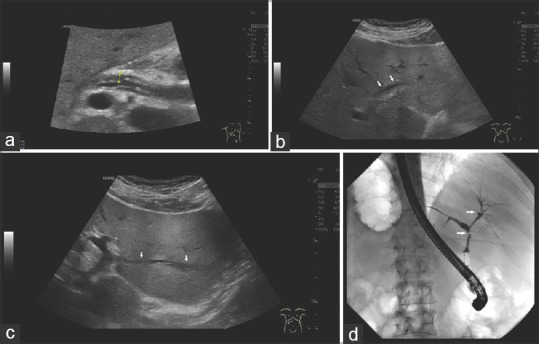
Bilioma. SSC-CIP. A 56-year-old male, intubation and ventilation for several weeks due to legionella pneumonia. Liver enzymes and cholestatic parameters are slightly elevated. Ultrasound shows a small asymmetrically shaped, smooth bordered anechoic lesion in the right liver lobe (a). The ERCP image shows destroyed and rarefied bile ducts in the left lobe of the liver. The complete right-sided biliary tree is not shown via the unblocked balloon catheter, stage III according to the classification of Leonhardt (b). SSC-CIP: Secondary sclerosing cholangitis in critically ill patients; ERCP: Endoscopic retrograde cholangiopancreaticography
Wall thickening of the bile ducts is not systematically described for SSC-CIP but can also be seen and might be helpful for differential diagnosis [Figures 3 and 4].
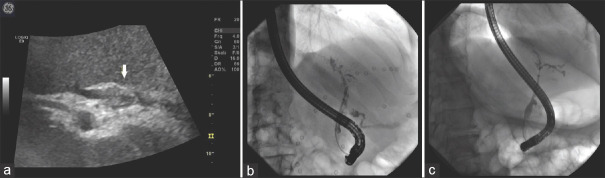
Figure 3.
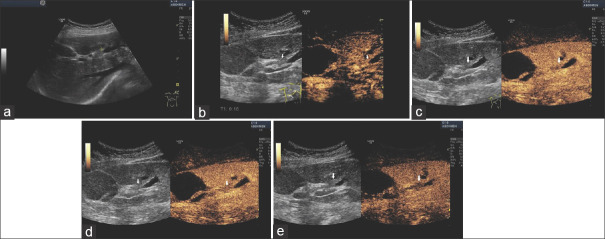
Wall thickening of the bile ducts due to SSC-CIP in a male patient after long-term intensive therapy and several weeks of intubation and ventilation. Ultrasound shows hypoechoic wall thickening and lumen narrowing of the right and left hepatic bile in the hepatic bifurcation (arrows) (a). In the left hepatic duct (b) and the right hepatic duct (c), longer segments of wall thickening are visible on ultrasound (arrows). SSC-CIP: Secondary sclerosing cholangitis in critically ill patients
Figure 4.
Irregular bile duct wall thickening due to SSC-CIP in a 62-year-old male patient after intensive therapy with long-term intubation and long-term ventilation. Recurrent removal of casts during ERCP. Ultrasound shows irregular wall thickening and lumen narrowing in the proximal common bile duct (a). The ERCP image shows contrast cavities in the hepatic sinus duct and common bile duct corresponding to casts. The right-sided bile ducts are rarefied (b). In the course, further bile duct destruction with intrahepatic rarefication occurs with an “pruned tree” appearance, stage III according to the classification of Leonhardt (c). SSC-CIP: Secondary sclerosing cholangitis in critically ill patients; Image source of ERCP: Steffen Hornoff and Matthias Mende, Sana Hospital Berlin-Lichtenberg, Endoscopy
Once the diagnosis is known, ultrasound can be helpful by detecting casts and dilated bile ducts in determining the need for repeated therapeutic ERCP.
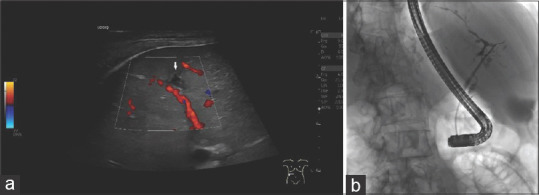
Ultrasound is very sensitive for detecting hepatic artery thrombosis in symptomatic patients in the immediate postoperative period following liver transplantation. With a frequency of 4.8%, the sensitivity of ultrasound compared with angiography was 100% for the detection of early hepatic artery thrombosis and 72.7% for late hepatic artery thrombosis. Due to arterial collateral flow, ultrasound becomes less sensitive as the interval between transplantation and diagnosis of hepatic artery thrombosis increases. Patients with irreversible hepatic artery thrombosis typically develop arterial collaterals in the interval that are visible on ultrasound.[35]
The presence of tardus parvus waveform (TPW) in the intrahepatic arteries is a screening method before arteriography in cases of suspected stenosis or thrombosis with a 97% sensitivity and 64% specificity.[50,51] TPW is defined as resistive index (RI) <0.5 and systolic acceleration time (SAT) >0.08 s.[36] Using either (1) resistive index (RI) <0.4 or SAT >0.12 s, or (2) traditional TPW (RI <0.5 and SAT >0.08 s) in the presence of abnormal liver functions as the Doppler Ultrasound criteria for hepatic artery stenosis will significantly decrease the false-positive rate compared to traditional TPW without a significant increase in the false-negative rate.[36]
Contrast enhanced ultrasound
CEUS has been reported to effectively improve flow visualization of the hepatic artery and to shorten the scanning time compared with color Doppler ultrasound (CDUS), with a 100% sensitivity and 97.8% accuracy for the detection of hepatic artery thrombosis.[52,53] When CDUS cannot detect arterial flow, CEUS should be performed immediately in the same session, even at the bedside.[51,54-56]
Leonhardt et al.[25] observed dilatation of the extrahepatic bile duct in 31% of patients with a diagnosis of SSC-CIP using ultrasound. Isolated intrahepatic cholestasis was noted in 2/16 patients. Sonographic evidence of cholecystitis was noted in 8/16 patients. Overall, 88% of patients developed sonographic signs of cirrhosis within 6 months (mean).[25] Compared with ERCP, accuracy was only 30% for ultrasound.[28]
In patients after uneventful liver transplantation without ischemic bile duct lesions, earlier and greater enhancement of the extrahepatic bile duct wall and bifurcation is found in the arterial phase with CEUS compared to the surrounding liver parenchyma. Isoenhancement or minor hypoenhancement is observed in the portal venous and late phases. In contrast, patients with reduced perfusion of the PBP showed arterial phase hypo-or nonenhancement of the bile duct wall. In addition, the initial enhancement was delayed and the time difference to portal venous contrast of the liver parenchyma was shortened.[57]
Zhang et al. used CEUS for the detection of ischemic type biliary lesions after liver transplantation. In the case of liver enzyme elevations or if CEUS showed little or no enhancement of the bile duct wall of the hilar bile duct during the arterial phase, the indication for ERCP or percutaneous transhepatic cholangiopancreatography was made early.[58]
DIFFERENTIAL DIAGNOSIS
Ischemic cholangitis has to be differentiated from PSC, Klatskin tumor especially in bismuth stages III and IV and other etiologies of SSC.[17] The most important diagnostic tool is the patient’s individual medical history with reference to the typical triggering event.
THERAPY
Therapy of ischemic cholangitis depends on the triggering pathogenetic factor, which should be specifically treated. Interdisciplinary management of this prognostically unfavorable clinical scenario is required. In case of increasing cholestasis parameters and cholangitis, ERCP with papillotomy is indicated. Casts can be removed and stenoses dilated by balloon dilatation. In individual cases, the insertion of a nasobiliary probe for flushing with sterile saline solution may be useful. Stenting with plastic endoprostheses can provoke cholangitis, especially in cases of stent occlusion.[59,60] They should be avoided as far as possible. Bile cultures should be obtained during ERCP to treat the bacterial spectrum. The indication for frequently repeated ERCP should be made with a sense of proportion, since every intervention carries the risk of cholangitis.
Ursodeoxycholic acid is often used. However, there are no studies demonstrating efficacy and a positive effect on patient outcome in SSC-CIP.[23] The indication for liver transplantation must be considered.[24]
TOXIC OR DRUG-INDUCED SECONDARY SCLEROSING CHOLANGITIS
Secondary SC can develop in the context of drug-induced liver injury (DILI). The clinical presentation of cholestatic DILI includes bland cholestasis, cholestatic hepatitis, SSC, and vanishing bile duct syndrome. Important drugs that cause DILI are amoxicillin-clavulanate, sevoflurane, amiodarone, infliximab, green tea extract, venlafaxine, atorvastatin, and celecoxib. It is not uncommon for the elevated cholestasis parameters to disappear after discontinuation of the drug. In this respect, it is advisable to check the liver parameters at the beginning of long-term therapy with drugs known for their risk to cause DILI.[17,61] The mortality associated with cholestatic DILI can be as high as 10%.[62]
Up to 10% of patients with DILI may have SC-like changes on MRCP.[63] The development of SSC has been reported after therapy with chemotherapeutics,[64] in particular Docetaxel,[65] casuistically reported for paclitaxel in combination with bevacizumab,[66] 5-FU/leucovorin combination therapy,[67] furthermore for immune checkpoint inhibitors (ICI) pembrolizumab and nivolumab,[68-73] chronic ketamine abuse, methimazole,[74,75] moxifloxacin, atorvastatin, herbal supplements.[76]
ICIs induce inflammatory reactions against neoplastic tissue, but they can also cause immune-related adverse effects. Histopathologically, ICI-related sclerosing cholangitis (ICI-SCC) is characterized by cytotoxic CD8+lymphocytes infiltrating the bile ducts and fibrosis auf the bile duct wall. Intra-and extrahepatic bile ducts can be affected.[68,77,78] Nivolumab-related cholangitis is characterized by: extrahepatic bile duct dilation without obstruction; diffuse hypertrophy of the extrahepatic bile duct wall; increase of the biliary enzymes ALP and GGT; no elevation of serum IgG4; CD8+T cell infiltration in the portal area by liver biopsy.[70,79]
Zen et al. report on hepatobiliary injury under pembrolizumab and atezolizumab. Two cases under pembrolizumab had SC. The histological changes in the common bile duct resembled those of IgG4-related SC and not PSC. The inflammatory processes were primarily within the ductal wall. The epithelium was well preserved. Diffuse wall thickening on imaging was also reminiscent of IgG4-related SC, but IgG4-positive plasma cells were <10/HPF. The patients showed only a partial response to prednisolone. Another patient on pembrolizumab who was negative antimitochondrial antibody showed intrahepatic BDI resembling primary biliary cholangitis.[73] Casuistic the typical findings are also described in the endosonography. These are, in correlation to the other imaging, diffuse hypertrophy of the extrahepatic bile duct and caliber changes of the lumen of the bile ducts.[69]
Response to glucocorticoids is only moderate or poor.[68-70] Ludwig et al. list ICI-SCC under the autoimmune genesis of SSC.[80] In toxic cholangiopathy, the imaging features commonly resemble those of PSC, with multifocal strictures of the intrahepatic and extrahepatic bile ducts.[80]
INFECTIOUS CAUSES
Recurrent pyogenic cholangitis
Recurrent pyogenic infection can lead to biliary strictures, bile duct dilatation and stone formation resulting in SSC. Complications of acute inflammation include biliomas, abscesses and portal vein thrombosis. Causes may include cholangiolithiasis, postoperative bile duct injuries, and the rare Caroli disease. Therapy is by ERCP with endoscopic interventions, in individual cases surgically. Patients with SSC due to recurrent pyogenic infection have an increased risk of cholangiocarcinoma.[81,82]
Parasitic infections
Parasitic infections such as Clonorchis sinensis and Ascaris lumbricoides are reported to cause recurrent pyogenic infections.[80,83-85] Sonographic features in the hepatobiliary system are described.[86,87] Patients with unresected alveolar echinococcosis from Echinococcus multilocularis who required benzimidazole therapy developed in 28% late biliary complications including secondary SC. The most common biliary complications during long-term chemotherapy were late-onset cholangitis, SC-like lesions, hepatolithiasis, affection of the common bile duct and secondary biliary cirrhosis.[88]
AIDS-related cholangiopathy
AIDS-related cholangiopathy is multifactorial[17] and may occur in advanced AIDS (CD4 counts typically <100/mm3).[38,89,90] Several pathogens are associated with the AIDS cholangiopathy. The main causes are opportunistic infections such as CMV-infection, cryptosporidium, microsporidia, isospora, Mycobacterium avium intracellulare, or Giardia infections. These infections induce ischemic lesions through infectious vasculitis. CMV inclusions have been observed in arterioles adjacent to bile ducts.[22,89,90]
Cholangiographic findings in AIDS cholangiopathy often show long segments of extrahepatic strictures, beading and narrowing of the intrahepatic and extrahepatic bile ducts with periductal soft tissue thickening.[82]
Post-COVID-19-cholangiopathy
In many patients, cholangiopathy developed during COVID-19-infection or after recovery from COVID-19 infection,[91] but the clinical manifestations in almost all cases occurred after recovery from COVID-19, leading to the term post-COVID-19 cholangiopathy.[92] It is a particular form of liver damage, which is a variant of SSC-ICP.[93] Among 34 critically ill patients with COVID-19, 12% developed SSC-CIP, compared with none in a similar influenza A cohort with severe ARDS.[94]
Patients with COVID-19 and ARDS who required intensive care and ventilation have the same risk factors as other patients with SSC-CIP. Nardo et al.[95] discuss that the bile ducts of patients with COVID-19 might therefore be exposed to several factors: Hypoxia due to respiratory insufficiency (possibly exacerbated by obliteration of the peribiliary arterial plexus due to vasculitic/thrombotic changes); systemic inflammatory response syndrome (SIRS) leading to a reactive cholangiocyte phenotype or a senescence-associated secretory phenotype, actively promoting inflammation as well as fibrosis; and possible viral infection of the cholangiocytes themselves.[95]
The angiotensin-converting enzyme 2 (ACE2) and the serine protease TMPRSS2 have been identified as receptors for virus entry into the host cell. ACE2 and TMPRSS2 are expressed on various cells in the human body, including cholangiocytes. A direct interaction of severe acute respiratory syndrome coronavirus type 2 (SARS-CoV-2) with the bile epithelium is suspected.[95-97] In the study by Klindt et al.,[96] analysis of gallbladder bile, gallbladder brush cytology and gallbladder and liver histology from five deceased SARS-CoV-2 patients and one patient who had undergone cholecystectomy revealed no evidence of the presence of viral RNA by polymerase chain reaction. The authors concluded that viral replication in biliary and gallbladder epithelium above the limit of detection is not generally detectable in patients with moderate and severe COVID-19 disease.[96] The viral infection with SARS-CoV-2 leads to a decreased barrier function of cholangiocytes. This in turn could cause liver injury by leakage of potentially toxic bile into the periductal space and adjacent liver parenchyma. Of note, bile acid transporter expression was downregulated by SARS-CoV-2 infection.[95] This is consistent with the “toxic bile” concept discussed earlier. Finally, the development of micro embolisms and microthrombi in the context of hypercoagulability may be another cause of local bile duct hypoxemia in SARS-CoV-2 patients.[96] SARS-CoV-2 may alter vascular homeostasis by directly infecting endothelial cells via ACE2 and induce endothelitis as proven in other organ systems.[98]
Histologically, post-COVID-19 cholangiopathy showed diffuse degenerative cholangiocyte lesions with extreme cytoplasmic vacuolization and regenerative changes. These have not been reported in SCC-CIP. Multiple causes or the combination of SSC-CIP with direct viral damage are suspected in COVID-19 SC.[92,99]
Postoperative injuries
Post-operative injuries to the bile duct and hepatic artery can result in prolonged complications and interventions. Late complications of BDI include biliary strictures, liver atrophy, cholangitis and intrahepatic lithiasis. As a result of recurrent inflammation and intraductal stone formation, multifocal strictures may form in the sense of secondary SC. Prolonged biliary obstruction with associated recurrent cholangitis is the major risk factor for onset of secondary biliary cirrhosis. BDI after cholecystectomy is defined as unintentional injury to any portion of the biliary tree, manifesting during or after operation by a bile leak or bile obstruction.[100] Its incidence ranges from 0.1% to 1.5%.[101,102] The injuries seen currently tend to be more severe, with the most severe biliary and hepatic artery or portal vein injuries often occurring after conversion from laparoscopy to open cholecystectomy. Failed attempts to repair BDIs can result in longitudinal strictures of the common bile duct. Most BDIs are recognized either during the procedure or in the immediate postoperative period, with the two most frequent scenarios being the occurrence of a bile leak or bile duct obstruction.[103,104] The management of postoperative BDIs includes endoscopic dilatation, endoscopic drainage with stenting or surgical treatment.
Congenital causes
Congenital causes leading to SSC include cystic fibrosis (CF) and Caroli’s disease.[80] CF is caused by alterations of the transmembrane conductance regulator (CTFR) protein. The CFTR protein is also expressed in the in the bile duct. The bile duct changes are also attributed to the changed mucus in CF. Bile duct abnormalities range from minor tapering to dilatation, beading to stricturing. Cholangiographic findings in CF are similar to PSC and often present as stricture of the intrahepatic and/or extrahepatic bile ducts.[80,105] Micro gallbladder and cholelithiasis may also be present and fatty replacement of the pancreas.[105] All patients (100%) with known liver disease and 50% without known liver disease in CF had intrahepatic ductal abnormalities.[106] 65% of CF patients had ductal abnormalities on MRCP in the study by King et al.[107]
In Caroli disease (discussed above), SSC is rare but may occur as a result of intrahepatic cholestasis, intraductal stone formation, recurrent cholangitis, hepaticolithiasis and stricture formation.[108]
Immunological causes
Regarding immunological causes of SSC, there are individual case reports on sarcoidosis[109-111] and Behcet’s disease,[112] reports on SCC with ICIs,[73,113] and eosinophilic cholangitis.[114]
Hepatic involvement was found in 11.5% of patients with sarcoidosis, and the clinical manifestation varies significantly. 5%–15% develop signs and symptoms of cholestasis, portal hypertension, cirrhosis and Budd-Chiari syndrome. Intrahepatic sarcoidosis can resemble PSC. Diagnosis is made by the typical cholangiographic changes and the detection of granulomas in the liver biopsy.[109]
Vasculitis is the probable cause of ischaemia and SSC in Behcet’s disease.[112]
Eosinophilic cholangiopathy is a rare condition characterized by dense transmural eosinophilic infiltration of the biliary tract and causes SC. Eosinophilic cholangiopathy is thought to be part of a spectrum of diseases caused by eosinophilic infiltration of tissues and organs with or without peripheral eosinophilia. Eosinophilic infiltration is possible not only in the biliary tract but also in other organs, including the stomach, colon, pancreas, liver, and kidney. However, peripheral eosinophilia was present in only about half of the reported cases. Bile duct wall thickening is a characteristic finding of eosinophilic cholangitis. Bile duct strictures are located diffusely from the hepatic hilum to the intrahepatic biliary tree.[114,115] Matsumoto et al. described eosinophilic cholangitis on US as an isoechoic solid lesion in the hepatic duct with irregular wall thickening of the left bile duct. CEUS using Levovist (Schering, Berlin, Germany), showed that the wall of the intrahepatic bile ducts was thickened from the hilar region to the periphery. The wall was described as well enhanced. Intraductal ultrasound examination (IDUS) correlatively described a solid, isoechoic lesion around the common hepatic duct.[115]
IGG4-SC
IgG4-SC is the biliary manifestation of IgG4 related disease.[18,116] There is debate about the nomenclature of IgG4-related hepatobiliary disease; IgG4-related cholangitis has been called IgG4-associated cholangitis, IgG4-associated SC, autoimmune cholangitis or IgG4 cholangiopathy.[18]
Tanaka et al. calculated a prevalence of IgG4-SC in 2018 of 2.18/100,000 population in a nationwide epidemiological study in Japan.[117] Bile duct strictures and concentric thickening of the bile duct wall are typical. IgG4-SC is often associated with autoimmune pancreatitis (AIP).[118-120] Type I in 75%-85% cases but can occur without pancreatic disease. Histopathological findings include lymphoplasmacytic infiltrates, obliterative phlebitis, storiform fibrosis and infiltration of >10 IgG4-positive plasma cells/high-power field. Serum IgG4 is elevated in a 70%–80% of patients. The bile duct strictures are corticosteroid responsive. However, some strictures are more advanced and fibrosed and do not experience complete resolution with steroids.
IgG4-SC can be classified according to organ-specific criteria or as part of consensus comprehensive diagnostic criteria for IgG4-related systemic disease.[121] Clinical classification can be performed according to the HISORt criteria for pancreatic and biliary disease[122] or the Clinical Diagnostic Criteria of IgG4-Related SC of 2012 and 2021 from the Japan Biliary Association.[121,123,124] The clinical classifications are based on a typical cholangiogram, laboratory findings of elevated IgG4 antibodies, presence of a systemic involvement and histological examination. Therapeutic response to corticosteroids is another diagnostic criterion.[18,19,121,123,125]
The cholangiographic classification is based on the localization of the bile duct stenoses.[126,127] According to the localization of the strictures in the biliary system, Nakazawa et al. divided four cholangiographic types of IgG4-SC [Figure 5].[126-128] The localization of the strictures is important for the differential diagnosis compared to strictures in pancreatic head carcinoma, PSC and cholangiocellular carcinoma.[126,127] In type 1 IgG4-SC, the stricture is localized only in the distal common bile duct. In type 2 of IgG4-SC, the biliary strictures are diffusely distributed throughout the intra-and extrahepatic bile ducts. Two subtypes are distinguished:
Figure 5.
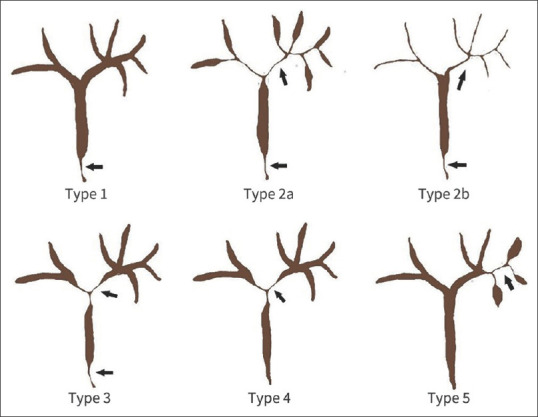
Cholangiographic classification of IgG4-SC Type 1-4 according to Nakazawa et al. 2006 and 2012[126,127] und intrahepatic IgG4-SC Type 5 according to Mo et al. 2018.[128] Type 1: stenosis only in the distal (intrapancreatic) part of the common bile duct. Type 2: stenosis in the intrahepatic and extrahepatic bile ducts; extended narrowing of intrahepatic bile ducts with pre-stenotic dilation in type 2a and extended narrowing of intrahepatic bile ducts without pre-stenotic dilation and the reduced number of bile duct branches in type 2b. Type 3: stenosis in both hilar hepatic lesions and the distal part of the common bile duct. Type 4: stenosis only in the hilar hepatic lesions without stenosis of distal common bile duct. Type 5: Strict intrahepatic bile duct stenoses proximal to the hepatic bifurcation. IgG4-SC: Immunoglobulin G4-related sclerosing cholangitis
Type 2a is characterized by biliary strictures of the intrahepatic bile ducts with prestenotic dilatation. Type 2b has reduced bile duct branching. There is no prestenotic dilatation upstream of the biliary strictures of the intrahepatic bile ducts. Type 3 IgG4-SC is characterized by biliary stricture in the hilar hepatic and intrapancreatic bile ducts. In type 4 IgG4-SC bile duct stenosis affects only the hepatic hilum region.[126,129]
Type 1 of IgG4-SC must be differentiated from pancreatic carcinoma, chronic pancreatitis and common bile duct cancer. Type 2b must be differentiated from PSC and other SCC etiologies. Type 3 and 4 should be differentiated from hilar cholangiocarcinoma.[130-132]
The presence of a distal bile duct stenosis in the intrapancreatic portion in IgG4-SC is always an indication of concurrent AIP type I.[129] Focal AIP typically presents with increased vascularisation and hyperenhancement on CEUS and contrast enhanced-EUS.[133,134] In contrast, Bauditz et al. described hilar cholangiocarcinoma in the arterial phase predominantly (87%) as hypoenhanced on CEUS.[135]
Mo et al. suggested an additional type 5: A strictly intrahepatic pattern of IgG4-SC without any affection of the hepatic hilum and common bile duct [Figure 5]. On imaging and macroscopically, the intrahepatic subtype (type 5) was divided into three subtypes: mass-forming (50%), stricturing (41.7%) and periductal infiltrating (8.3%) subtypes. The strict intrahepatic localization does not exclude the co-existence with other IgG4-related organ manifestations. Main differential diagnoses of intrahepatic localized IgG4-SC are intrahepatic cholangiocarcinoma and liver lesions.[128]
In the largest case series, a Japanese study of 872 patients with IgG4-SC by Naitoh et al.,[136] 83.7% of patients had simultaneous autoimmune pancreatitis. 69.9% of all IgG4-SC with concomitant AIP presented as cholangiographic type 1 [Figure 6]. In contrast, only 23.8% of IgG4-SCwere type 1. Most frequently, IgG4-SC without AIP corresponded to cholangiographic type 4 (30.9%).
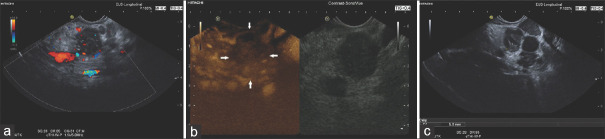
Figure 6.
IgG4-SC and IgG4-positive autoimmune pancreatitis. A 70-year-old female, history of IgG4 positive sialadenitis. Current findings: jaundice, dilated intra-and extrahepatic bile ducts and pancreas head mass. Initially a pancreatic head cancer was suspected. Serum IgG4 was 2-fold elevated. Endosonography showed a hypoechoic lesion in the pancreatic head (a) with marked enhancement in contrast-enhanced harmonic EUS (b). A sandwich-like thickening of the wall of the adjacent distal common bile duct with a smooth border of the inner and outer contour was observed (5mm, between markers; c). The diagnosis was made in the overall context of clinical history, elevated serum IgG4 and hyperenhancement of the pancreatic mass lesion. EUS-guided fine needle biopsy demonstrated no malignancy. The patient showed a rapid response to prednisolone treatment. IgG4-SC: Immunoglobulin G4-related sclerosing cholangitis
Among all IgG4-SC cases, males were predominantly affected in 74%–85%. The age of onset is in the 6th decade of life.[122,137,138] The proportion of females was significantly higher in IgG4-SC patients without AIP (28.9%) than the IgG4-SC with AIP group (20.1%).[136] 25.2% of the patients had other organ involvement besides AIP.[136] Other organ involvement can include dacryoadenitis, sialadenitis, retroperitoneal fibrosis, kidney lesion, cholecystitis, and lung disease.[18,116,136]
Cases of IgG4-SC not associated with other manifestations of IgG4-related disease are referred to as isolated IgG4-SC.[139,140]
Most frequent symptom of IgG4-SC at presentation are jaundice, followed by pruritus and abdominal pain.[19,21,136] Weight loss is commonly seen in those with co-existent AIP due to exocrine pancreatic insufficiency and deficiencies of fat-soluble vitamins.
For differentiation between IgG4-SC, PSC and cholangiocarcinoma, serum IgG4-levels, the pattern and details of biliary strictures as well as details of wall thickening may be used [Table 2]. Most relevant differential diagnoses of type 1 IgG4-SC are pancreatic head cancer and distal cholangiocarcinoma, whereas type 2 IgG4-SC has cholangiographic similarities in particular with PSC. Type 3 and 4 IgG4-SCA must be discriminated from hilar cholangiocarcinoma. Type 5 may resemble the peripheral type of PSC.[121,128,129,136]
Table 2.
Criteria for differential diagnosis of IgG4-related sclerosing cholangitis and primary sclerosing cholangitis*
| IgG4-SC | PSC | |
|---|---|---|
| Demographics and clinical presentation | ||
| Male:female | 4:1 | 7:1 |
| Predominant age at presentation (years) | >60 | <50 |
| Association with IBD | Rare (5%) | 80% ulcerative colitis in patients with large-duct PSC |
| Pancreatic involvement | 83%–92% | No |
| Other organ involvement (excluding pancreas and/or intestine) | 25%, various organs | No |
| Laboratory findings | ||
| Serum IgG4 >135 (140) mg/dL (%) | 78–88 | 12–20 |
| Serum IgG4 >270 (280) mg/dL (%) | 70 | 2 |
| Serum IgG4/IgG1-ratio >0.24 | 95% specificity for IgG4-SC | |
| Elevated serum IgE | 50% | Rare, no exact data |
| Peripheral blood eosinophilia | 40% | Rare, no exact data |
| pANCA | Rare, but no data | 80% |
| Bile duct strictures | ||
| Pattern | Short band-like strictures, beaded or pruned tree-like appearance, diverticular out pouches, continuous involvement | Segmental and long intra- and extrahepatic strictures with prestenotic dilation |
| Types | Distal (type 1), combined hilar and intrahepatic (type 2a and 2b), combined distal and hilar (type 3), hilar (type 4) and isolated intrahepatic (type 5) stricture patterns with features of type 2a and 2b overlapping with PSC | Large duct PSC (classical) and small-duct PSC |
| Wall thickening | ||
| Pattern | Circular and symmetrical with smooth inner and outer margins and homogeneous internal echo, preserved layering | Asymmetrical, inhomogeneous with irregular inner and outer contour and loss of delineation of three layers |
| Relation to strictures | Not restricted to the strictures | Restricted to the stricture |
| Histology | ||
| Infiltration with IgG4-positive cells | Lymphoplasmacytic infiltration and fibrosis in the intra- and extrahepatic bile ducts, eosinophilia | |
| Storiform fibrosis and obliterative phlebitis | Periportal sclerosis or periductal concentric “onion-skin” fibrosis | |
| >10 IgG4-positive plasma cells/high-power field | Possible | |
| Corticoid responsiveness | 90% remission rate | Rare, no exact data |
An elevated serum IgG4 level (≥135 mg/dL) was found in 78.0%-88.0% of IgG4-SC cases, and cut-off values of 135 mg/dl or 140 mg/dl were established for diagnosis.[125,136,142,143] In the large Japanese cohort, serum IgG4 levels did not differ between AIP-positive and AIP-negative IgG4-SC patients.[136] However, elevated serum IgG4-levels are non-specific for IgG4-SC and are observed in other mimickers of IgG4-SC, such as cholangiocarcinoma (13%), PSC (12%–20%) and pancreatic cancer (10%).[21,125,136,141,142] In PSC patients, IgG4 positivity is associated with a worse outcome.[141] Furthermore, elevated serum-IgG4 levels may be observed in patients with other inflammatory, infective, and autoimmune conditions such as bronchial asthma, pemphigus, and atopic dermatitis.[18,19]
If a cut-off value of 280 ng/ml was used, specificity for diagnosis of IgG4-SC was 98% in a European study. In cases with IgG4 levels between 140 and 280 ng/ml, the serum IgG4/IgG1-ration with a cut-off value of 0.24 was of discriminating value (sensitivity: 80%, specificity: 74%, positive predictive value: 55%, negative predictive value: 90%).[144] In the most recent large cohort of Japanese IgG4-SC patients the median serum IgG4 levels were 410, 348.5, 651.5 and 682 mg/dL for patients with stricture types 1-4, respectively. Given the fact, that differentiation from intrahepatic cholangiocarcinoma is most difficult for type 3 and 4 IgG4-SC, the particularly marked elevation of IgG4 in both types may be useful for differential diagnosis.[136] In another large Japanese case series, a cut-off value of 207 ng/ml had a 100% specificity to discriminate between IgG4-SC and cholangiocarcinoma.[125]
Antinuclear antibodies were found in 39% of the IgG4-SC patients. Serum IgE levels are elevated in 50% and a peripheral eosinophilia is present in 40% of IgG4-SC and is often associated with a clinical history of atopy.[145] CA 19-9 is used as a tumor marker in cholangiocellular and pancreatic carcinoma but is often also elevated in PSC and IgG4-SC and other nonmalignant causes of obstructive jaundice.[146,147]
The assignment to the different types of IgG4-SC is done by MRCP or ERCP. These methods can also show the changes in the small intrahepatic bile ducts and rarefication of the bile ducts.[125,129] Characteristic in IgG4-SC are segmental and (confluent) long strictures with pre-stenotic dilation. Band-like strictures, beaded appearance, pruned tree-like appearance and diverticular protrusions are typical of PSC and rarely found in IgG4-SC.[129,136,139,148]
Whereas thickening of the bile duct wall has been detected in the above mentioned large Japanese IgG4-SC cohort on Ultrasound (81.2%), CT (71.4%), and MRI (65.9%), high-resolution ultrasound using high frequency linear probes, EUS and IDUS are capable to visualize details of wall thickening and can therefore provide important clues for differential diagnosis. Circular and symmetrical thickening of the bile duct wall with a smooth inner and outer margin and homogeneous internal echo in the stenotic area is a characteristic feature of IgG4-SC[129,139] [Figure 7]. The representation of the smooth epithelial surface on ultrasound reflects the generalized subepithelial damage caused by the inflammatory cells.[143] Typically, wall thickening in IgG4-SC is not only observed in narrowed segments on cholangiographic modalities but extends from the distal common bile duct to the hilar region[136] [Figure 8]. However, wall thickening in a non-strictured area was significantly lower observed in AIP-negative compared to AIP-positive IgG4-SC patients (EUS, 75.3% vs. 58.8%; IDUS, 85.8% vs. 58.3%).[136] In contrast, wall thickening in PSC and cholangiocarcinoma patients is restricted to the strictured segment (s) and is more asymmetrical and heterogeneous with irregular inner margin and blurred outer contours, and disappearance of layering[129,147,149,150] [Figures 9 and 10].
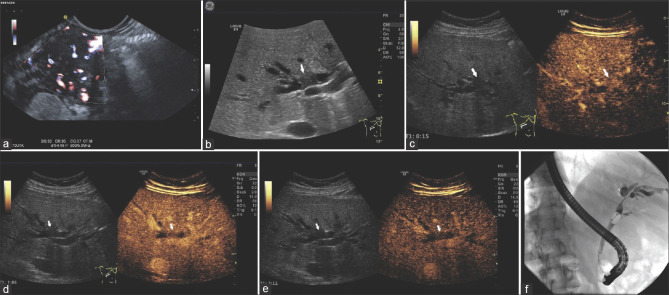
Figure 7.
IgG4-SC type 3; A 46-year-old male. Initial multifocal autoimmune pancreatitis type I with distal bile duct stenosis. EUS initially showed a hypoechoic lesion at the head of the pancreas with multiple vessels in contrast-enhanced power Doppler before starting Prednisolone therapy (a). After reduction of prednisolone therapy, recurrence of cholestasis. Ultrasound shows a smooth, symmetrical thickening of the wall of the hepatic bifurcation (b). In CEUS this is hyperenhanced in the arterial phase (c) and slightly hypoenhanced in the portal venous (d) and late phase (e). The laboratory values improved under therapy with prednisolone and then azathioprine. Balloon dilatation of stenoses in ERCP was performed several times. Via the blocked balloon catheter, ERCP reveals a short stricture involving the bifurcation of the common hepatic duct (f). IgG4-SC: Immunoglobulin G4-related sclerosing cholangitis; CEUS: Contrast-enhanced ultrasound; Image source of ERCP: Steffen Hornoff and Matthias Mende, Sana Hospital Berlin-Lichtenberg, Endoscopy
Figure 8.
IgG4-SC with AIP I (type 1) and multiple other organ manifestations of IgG4-related disease. A 48-year-old male. Longitudinal wall thickening of the bile ducts. Ultrasound shows wall thickening in the proximal common bile duct. The inner and outer contours are smooth, the inner contour is hyperechoic (a). In the left lobe of the liver, there is also a long-stretched wall thickening of the left hepatic duct (b) and of the segmental bile duct branch in the 2nd liver segment (c). The bile ducts are not dilated. ERC shows short band-like subhilar stricture (arrow) and a second in the right hepatic duct in conjunction with a small diverticular outpouching (arrow) after injection of contrast medium via the blocked balloon catheter. The left hepatic duct is prominent with no biliary strictures in the left part of the biliary tree. The intrahepatic bile ducts are rarefied (d). IgG4-SC: Immunoglobulin G4-related sclerosing cholangitis; AIP: Autoimmune pancreatitis; ERC: Endoscopic retrograde cholangiography
Figure 9.
A 55-year-old male patients presenting with mild right upper quadrant pain and elevated liver enzymes, no obstructive jaundice, history of ulcerative colitis. pANCA was positive, serum IgG4 was not elevated. Transabdominal ultrasound shows slight dilatation of the RHD, caused by hypoechoic wall thickening of the CHD and proximal RHD (arrow; a). On CEUS, in the late arterial phase, slight hypoenhancement of the thickened wall compared to liver parenchyma is observed (arrow; b). Radial EUS shows slight wall hypoechoic thickening of the extrahepatic bile duct with reduction (common bile duct, CBD) and loss of anechoic lumen and irregular outer contour (CHD, between markers; arrows are directed to the blurred outline). Compared to the IgG4-SC case in Figure 8, complete loss of layering is observed (c). ERC delineates a long stricture of the CHD extending to right and left hepatic ducts (arrows; d). Please note the difference to the short-band like strictures and diverticular out pouch in the ERC-image of the IgG4-SC case in Figure 8. Stepwise balloon dilatation is performed with initial dentation of the balloon in the area of the tightest part of the stricture (arrowhead; e). IgG4-SC: Immunoglobulin G4-related sclerosing cholangitis; CEUS: Contrast-enhanced ultrasound; ERC: Endoscopic retrograde cholangiography; RHD: Right hepatic duct; CHD: Common hepatic duct; pANCA: Perinuclear anti-neutrophil cytoplasmic antibodies
Figure 10.
Intrahepatic cholangiocarcinoma in the left liver lobe, histologically confirmed. A 77-year-old female. US was performed because of elevated liver values. This showed a segmental dilatation of the bile ducts in the left lobe of the liver. In B-mode ultrasound, the cause was a hyperechogenic thickening of the wall without definable stratification, which led to lumen stenosis (a). In CEUS, the wall-thickening, lumen-stenosing process is hyperenhanced in the arterial phase after 16 s (b). Already at the end of the arterial phase at 32 s (c), a slight washout begins. This continues in the portalvenous phase at 48 s (d) and is very clear in the late phase after more than 2 min (e). Histological confirmation was made by percutaneous US-guided biopsy and hemihepatectomy was performed. The cholangiocarcinoma presented as rather hyperechoic. CEUS showed non-specific hyperenhancement in the arterial phase with a subsequent washout, as in the IgG4-SC shown. CEUS: Contrast-enhanced ultrasound; IgG4-SC: Immunoglobulin G4-related sclerosing cholangitis; US: Ultrasound
EUS is a sufficient method to diagnose the wall thickening of the bile ducts. IDUS is more sensitive but requires the apparative and invasive effort of ERCP.[151] In the study of Naitoh et al. 2021,[136] wall thickening in a non-strictured area was found significantly more often on IDUS than on EUS (80.9% vs. 73.8%). However, EUS and IDUD proved to be as sensitive (90.4% vs. 90.5%) in visualizing a preserved triple stratification of the thickened bile duct wall.[136]
HISTOPATHOLOGY
Bile duct cytology, despite its low sensitivity, is useful for the diagnosis of biliary strictures, especially for the differential diagnosis to cholangiocarcinoma. It has not value for a positive diagnosis of IgG4-SC or PSC. Bile duct biopsy may show diffuse subepithelial lymphoplasmacytic inflammation of the wall of both intra-and extrahepatic bile ducts with fibrosis with a preserved epithelial layer.[152] However, definitive diagnosis of IgG4-SC by bile duct biopsy is often difficult because the epithelium of the bile duct may be histologically normal although the changes in the wall of the bile duct are pronounced. In the study of Naitoh et al., marked lymphoplasmacytic infiltration and fibrosis, more than 10 IgG4-positive plasma cells/HPF, storiform fibrosis and obliterative phlebitis in bile duct biopsy samples were found in 32.9%/16.9%/0.6% and 0%, respectively. In 0.3% of patients with IgG4-SC, the bile duct biopsy was classified as histopathological definitive case, and 11.8% were probable cases in 331 patients with all IgG4-SC.[136] Biopsy of the duodenal papilla with IgG4-immunostaining has been reported to be a useful supplemental method for diagnosing IgG4-SC.[129]
The clinical diagnosis of definite/probable/possible IgG4-SC is based on the constellation of several diagnostic criteria of the clinical classifications.[19,121-123]
THERAPY
Steroid therapy is the standard treatment for IgG4-SC with a response rate of 90%.[137] The recommended initial dose of prednisone is 30–40 mg daily for 2–4 weeks, followed by a reduction of 5 mg/week over 2–3 months.[18,19,153] After 2 weeks of steroid administration, the response should be evaluated.[129] Tanaka et al. described that therapy control of wall thickening in IgG4-SC type 2b was performed by ultrasound.[143] In 2019, Tanaka et al. reported that endoscopic dilatation or stenting were performed in 9% and 50% of cases, respectively. Surgical resection was performed in 9% because malignancy was suspected, mainly in those with IgG4-SC type 4.[21]
Progression to cirrhosis was noted in other reports in 5.2% and 7.5%, respectively.[122,138] The reduction and discontinuation of corticosteroid therapy will lead to a relapse of the disease in about 30% of those with IgG4-SC in the Japanese literature (3 years of steroids), and 60% in the European literature (3-6 month of steroids).[18] In case of relapse, reintroduction of corticosteroids is indicated in combination with other immunosuppressive treatments.[19] Meta-analysis and RCTs have shown significantly lower relapse rates with combination therapy from outset. Rituximab has been shown to be effective in inducing, maintaining remission, and treating relapse in patients with IgG4-related disease (RD), including IgG4-SC.[19] There has been reported an increased risk of infections with maintenance therapy, but not specifically cholangitis. Liver transplantation is rarely done. Unlike PSC or post-cholangitic strictures, patients with IgG4 SC are at a much lower risk of cholangiocellular Ca.[154] However, an increased risk of malignancy has been reported in IgG4-RD with a standardized incidence rate of 2.17, with a particular emphasis on pancreatic cancer and lymphoma.
CONCLUSION
A large number of different etiologic types of secondary SC have been reported. These must be considered alongside other cholestatic clinical pictures. Medical history is usually indicative of a triggering event. The diagnosis is mainly based on the cholangiographic images using MRCP and in some instances ERCP. Nevertheless, important diagnostic clues and complications can also be recorded in transabdominal and endoscopic ultrasound. Differential diagnosis from PSC and cholangiocarcinoma as well as correct classification of SC is a prerequisite for initiating the correct therapy because treatment and prognosis differ significantly between PSC, cholangiocarcinoma, and the various etiological types of SCC. In particular, the correct diagnosis of IgG4-SC is important, because 90% of cases respond to corticoid treatment.
Financial support and sponsorship
Nil.
Conflicts of interest
Siyu Sun is the Editor-in-Chief of the journal, Christoph F. Dietrich is a Co-Editor-in-Chief, Christian Jenssen and Michael Hocke are Editorial Board Members. This article was subject to the journal’s standard procedures, with peer review handled independently of the editors and their research groups.
REFERENCES
- 1.Dietrich CF, Averkiou M, Nielsen MB, et al. How to perform contrast-enhanced ultrasound (CEUS) Ultrasound Int Open. 2018;4:E2–15. doi: 10.1055/s-0043-123931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Claudon M, Dietrich CF, Choi BI, et al. Guidelines and good clinical practice recommendations for contrast enhanced ultrasound (CEUS) in the liver –Update 2012:A WFUMB-EFSUMB initiative in cooperation with representatives of AFSUMB, AIUM, ASUM, FLAUS and ICUS. Ultrasound Med Biol. 2013;39:187–210. doi: 10.1016/j.ultrasmedbio.2012.09.002. [DOI] [PubMed] [Google Scholar]
- 3.Claudon M, Dietrich CF, Choi BI, et al. Guidelines and good clinical practice recommendations for contrast enhanced ultrasound (CEUS) in the liver –Update 2012:A WFUMB-EFSUMB initiative in cooperation with representatives of AFSUMB, AIUM, ASUM, FLAUS and ICUS. Ultraschall Med. 2013;34:11–29. doi: 10.1055/s-0032-1325499. [DOI] [PubMed] [Google Scholar]
- 4.Dietrich CF, Nolsøe CP, Barr RG, et al. Guidelines and good clinical practice recommendations for contrast-enhanced ultrasound (CEUS) in the liver-update 2020 WFUMB in cooperation with EFSUMB, AFSUMB, AIUM, and FLAUS. Ultrasound Med Biol. 2020;46:2579–604. doi: 10.1016/j.ultrasmedbio.2020.04.030. [DOI] [PubMed] [Google Scholar]
- 5.Dietrich CF, Nolsøe CP, Barr RG, et al. Guidelines and good clinical practice recommendations for contrast enhanced ultrasound (CEUS) in the liver –Update 2020 –WFUMB in cooperation with EFSUMB, AFSUMB, AIUM, and FLAUS. Ultraschall Med. 2020;41:562–85. doi: 10.1055/a-1177-0530. [DOI] [PubMed] [Google Scholar]
- 6.Piscaglia F, Nolsøe C, Dietrich CF, et al. The EFSUMB guidelines and recommendations on the clinical practice of contrast enhanced ultrasound (CEUS):Update 2011 on non-hepatic applications. Ultraschall Med. 2012;33:33–59. doi: 10.1055/s-0031-1281676. [DOI] [PubMed] [Google Scholar]
- 7.Sidhu PS, Cantisani V, Dietrich CF, et al. The EFSUMB guidelines and recommendations for the clinical practice of contrast-enhanced ultrasound (CEUS) in non-hepatic applications:Update 2017 (short version) Ultraschall Med. 2018;39:154–80. doi: 10.1055/s-0044-101254. [DOI] [PubMed] [Google Scholar]
- 8.Möller K, Tscheu T, De Molo C, et al. Comments and illustrations of the WFUMB CEUS liver guidelines:Rare congenital vascular pathology. Med Ultrason. 2022;24:461–72. doi: 10.11152/mu-3879. [Doi:10.11152/mu-3879] [DOI] [PubMed] [Google Scholar]
- 9.Papamichalis PA, Zachou K, Papamichali RA, et al. Promyelocytic leukemia antigen expression:A histological marker for primary biliary cholangitis diagnosis? J Transl Int Med. 2021;9:43–51. doi: 10.2478/jtim-2021-0008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Tischendorf JJ, Hecker H, Krüger M, et al. Characterization, outcome, and prognosis in 273 patients with primary sclerosing cholangitis:A single center study. Am J Gastroenterol. 2007;102:107–14. doi: 10.1111/j.1572-0241.2006.00872.x. [DOI] [PubMed] [Google Scholar]
- 11.Hirche TO, Russler J, Braden B, et al. Sonographic detection of perihepatic lymphadenopathy is an indicator for primary sclerosing cholangitis in patients with inflammatory bowel disease. Int J Colorectal Dis. 2004;19:586–94. doi: 10.1007/s00384-004-0598-0. [DOI] [PubMed] [Google Scholar]
- 12.Zhang Y, Gao X, He Z, et al. Prevalence of inflammatory bowel disease in patients with primary sclerosing cholangitis:A systematic review and meta-analysis. Liver Int. 2022;42:1814–22. doi: 10.1111/liv.15339. [DOI] [PubMed] [Google Scholar]
- 13.Chapman MH, Thorburn D, Hirschfield GM, et al. British Society of Gastroenterology and UK-PSC guidelines for the diagnosis and management of primary sclerosing cholangitis. Gut. 2019;68:1356–78. doi: 10.1136/gutjnl-2018-317993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Rabiee A, Silveira MG. Primary sclerosing cholangitis. Transl Gastroenterol Hepatol. 2021;6:29. doi: 10.21037/tgh-20-266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Da Cunha T, Vaziri H, Wu GY. Primary sclerosing cholangitis and inflammatory Bowel disease:A review. J Clin Transl Hepatol. 2022;10:531–42. doi: 10.14218/JCTH.2021.00344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Tornai D, Ven PL, Lakatos PL, et al. Serological biomarkers for management of primary sclerosing cholangitis. World J Gastroenterol. 2022;28:2291–301. doi: 10.3748/wjg.v28.i21.2291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Brooling J, Leal R. Secondary sclerosing cholangitis:A review of recent literature. Curr Gastroenterol Rep. 2017;19:44. doi: 10.1007/s11894-017-0583-8. [DOI] [PubMed] [Google Scholar]
- 18.Löhr JM, Beuers U, Vujasinovic M, et al. European guideline on IGG4-related digestive disease –UEG and SGF evidence-based recommendations. United European Gastroenterol J. 2020;8:637–66. doi: 10.1177/2050640620934911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Drazilova S, Veseliny E, Lenartova PD, et al. IgG4-related sclerosing cholangitis:Rarely diagnosed, but not a rare disease. Can J Gastroenterol Hepatol. 2021;2021:1959832. doi: 10.1155/2021/1959832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Tasneem AA, Luck NH. Autoimmune hepatitis:Clinical characteristics and predictors of biochemical response to treatment. J Transl Int Med. 2020;8:106–11. doi: 10.2478/jtim-2020-0016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Tanaka A. IgG4-related sclerosing cholangitis and primary sclerosing cholangitis. Gut Liver. 2019;13:300–7. doi: 10.5009/gnl18085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Deltenre P, Valla DC. Ischemic cholangiopathy. Semin Liver Dis. 2008;28:235–46. doi: 10.1055/s-0028-1085092. [DOI] [PubMed] [Google Scholar]
- 23.Laurent L, Lemaitre C, Minello A, et al. Cholangiopathy in critically ill patients surviving beyond the intensive care period:A multicentre survey in liver units. Aliment Pharmacol Ther. 2017;46:1070–6. doi: 10.1111/apt.14367. [DOI] [PubMed] [Google Scholar]
- 24.Kirchner GI, Rümmele P. Update on sclerosing cholangitis in critically ill patients. Viszeralmedizin. 2015;31:178–84. doi: 10.1159/000431031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Leonhardt S, Veltzke-Schlieker W, Adler A, et al. Secondary sclerosing cholangitis in critically ill patients:Clinical presentation, cholangiographic features, natural history, and outcome:A series of 16 cases. Medicine (Baltimore) 2015;94:e2188. doi: 10.1097/MD.0000000000002188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Leonhardt S, Veltzke-Schlieker W, Adler A, et al. Trigger mechanisms of secondary sclerosing cholangitis in critically ill patients. Crit Care. 2015;19:131. doi: 10.1186/s13054-015-0861-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Voigtländer T, Leuchs E, Vonberg RP, et al. Microbiological analysis of bile and its impact in critically ill patients with secondary sclerosing cholangitis. J Infect. 2015;70:483–90. doi: 10.1016/j.jinf.2015.01.013. [DOI] [PubMed] [Google Scholar]
- 28.Voigtländer T, Negm AA, Schneider AS, et al. Secondary sclerosing cholangitis in critically ill patients:Model of end-stage liver disease score and renal function predict outcome. Endoscopy. 2012;44:1055–8. doi: 10.1055/s-0032-1325733. [DOI] [PubMed] [Google Scholar]
- 29.Jaeger C, Mayer G, Henrich R, et al. Secondary sclerosing cholangitis after long-term treatment in an Intensive Care Unit:Clinical presentation, endoscopic findings, treatment, and follow-up. Endoscopy. 2006;38:730–4. doi: 10.1055/s-2006-925241. [DOI] [PubMed] [Google Scholar]
- 30.Ben-Ari Z, Levingston D, Weitzman E, et al. Secondary sclerosing cholangitis following major burn. Ann Hepatol. 2015;14:695–701. [PubMed] [Google Scholar]
- 31.Rath AM, Zhang J, Bourdelat D, et al. Arterial vascularisation of the extrahepatic biliary tract. Surg Radiol Anat. 1993;15:105–11. doi: 10.1007/BF01628308. [DOI] [PubMed] [Google Scholar]
- 32.Batts KP. Ischemic cholangitis. Mayo Clin Proc. 1998;73:380–5. doi: 10.1016/S0025-6196(11)63706-3. [DOI] [PubMed] [Google Scholar]
- 33.Northover JM, Terblanche J. A new look at the arterial supply of the bile duct in man and its surgical implications. Br J Surg. 1979;66:379–84. doi: 10.1002/bjs.1800660603. [DOI] [PubMed] [Google Scholar]
- 34.Martins P, Verdelho Machado M. Secondary sclerosing cholangitis in critically ill patients:An underdiagnosed entity. GE Port J Gastroenterol. 2020;27:103–14. doi: 10.1159/000501405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Horrow MM, Blumenthal BM, Reich DJ, et al. Sonographic diagnosis and outcome of hepatic artery thrombosis after orthotopic liver transplantation in adults. AJR Am J Roentgenol. 2007;189:346–51. doi: 10.2214/AJR.07.2217. [DOI] [PubMed] [Google Scholar]
- 36.Zheng BW, Tan YY, Fu BS, et al. Tardus parvus waveforms in Doppler ultrasonography for hepatic artery stenosis after liver transplantation:Can a new cut-off value guide the next step? Abdom Radiol (NY) 2018;43:1634–41. doi: 10.1007/s00261-017-1358-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Mourad MM, Algarni A, Liossis C, et al. Aetiology and risk factors of ischaemic cholangiopathy after liver transplantation. World J Gastroenterol. 2014;20:6159–69. doi: 10.3748/wjg.v20.i20.6159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Goria O, Archambeaud I, Lemaitre C, et al. Ischemic cholangiopathy:An update. Clin Res Hepatol Gastroenterol. 2020;44:486–90. doi: 10.1016/j.clinre.2020.03.018. [DOI] [PubMed] [Google Scholar]
- 39.Gelbmann CM, Rümmele P, Wimmer M, et al. Ischemic-like cholangiopathy with secondary sclerosing cholangitis in critically ill patients. Am J Gastroenterol. 2007;102:1221–9. doi: 10.1111/j.1572-0241.2007.01118.x. [DOI] [PubMed] [Google Scholar]
- 40.Weig T, Schubert MI, Gruener N, et al. Abdominal obesity and prolonged prone positioning increase risk of developing sclerosing cholangitis in critically ill patients with influenza A-associated ARDS. Eur J Med Res. 2012;17:30. doi: 10.1186/2047-783X-17-30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Deltenre P, Moreno C, Trépo E. Progressive cholangiopathy in COVID-19 patients:Other possible diagnoses than ketamine-induced cholangiopathy should be considered. J Hepatol. 2021;75:989–90. doi: 10.1016/j.jhep.2021.02.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Keta-Cov Research Group Electronic Address:vincentmallet@aphpfr,Keta-Cov research group. Intravenous ketamine and progressive cholangiopathy in COVID-19 patients. J Hepatol. 2021;74:1243–4. doi: 10.1016/j.jhep.2021.02.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Deltenre P, Valla DC. Ischemic cholangiopathy. J Hepatol. 2006;44:806–17. doi: 10.1016/j.jhep.2006.01.009. [DOI] [PubMed] [Google Scholar]
- 44.Jin S, Shi XJ, Sun XD, et al. Sclerosing cholangitis secondary to bleomycin-iodinated embolization for liver hemangioma. World J Gastroenterol. 2014;20:17680–5. doi: 10.3748/wjg.v20.i46.17680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Richardet JP, Lons T, Sibony M, et al. Secondary sclerosing cholangitis and chemo-embolization with lipiodol. Gastroenterol Clin Biol. 1994;18:168–71. [PubMed] [Google Scholar]
- 46.Xu H, Yu X, Hu J. The risk assessment and clinical research of bile duct injury after transcatheter arterial chemoembolization for hepatocellular carcinoma. Cancer Manage Res. 2021;13:5039–52. doi: 10.2147/CMAR.S303172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Miyayama S, Yamashiro M, Okuda M, et al. Main bile duct stricture occurring after transcatheter arterial chemoembolization for hepatocellular carcinoma. Cardiovasc Intervent Radiol. 2010;33:1168–79. doi: 10.1007/s00270-009-9781-6. [DOI] [PubMed] [Google Scholar]
- 48.Blewitt RW, Brown CM, Wyatt JI. The pathology of acute hepatic disintegration in hereditary haemorrhagic telangiectasia. Histopathology. 2003;42:265–9. doi: 10.1046/j.1365-2559.2003.01579.x. [DOI] [PubMed] [Google Scholar]
- 49.Kanai R, Nakamura M, Tomisato K, et al. Cholangitis as an initial manifestation of polyarteritis nodosa. Intern Med. 2014;53:2307–12. doi: 10.2169/internalmedicine.53.2508. [DOI] [PubMed] [Google Scholar]
- 50.Dodd GD, 3rd, Memel DS, Zajko AB, et al. Hepatic artery stenosis and thrombosis in transplant recipients:Doppler diagnosis with resistive index and systolic acceleration time. Radiology. 1994;192:657–61. doi: 10.1148/radiology.192.3.8058930. [DOI] [PubMed] [Google Scholar]
- 51.Ren J, Wu T, Zheng BW, et al. Application of contrast-enhanced ultrasound after liver transplantation:Current status and perspectives. World J Gastroenterol. 2016;22:1607–16. doi: 10.3748/wjg.v22.i4.1607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Lu Q, Zhong XF, Huang ZX, et al. Role of contrast-enhanced ultrasound in decision support for diagnosis and treatment of hepatic artery thrombosis after liver transplantation. Eur J Radiol. 2012;81:e338–43. doi: 10.1016/j.ejrad.2011.11.015. [DOI] [PubMed] [Google Scholar]
- 53.Ren Y, Yuan G, Zhou Y, et al. Incidence and characteristics of benign liver space-occupying mass in 17 721 patients with chronic hepatitis B:A color Doppler ultrasound-based case-control study. Nan Fang Yi Ke Da Xue Xue Bao. 2019;39:1149–54. doi: 10.12122/j.issn.1673-4254.2019.10.03. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.García-Criado A, Gilabert R, Binachi L, et al. Impact of contrast-enhanced ultrasound in the study of hepatic artery hypoperfusion shortly after liver transplantation:Contribution to the diagnosis of artery steal syndrome. Eur Radiol. 2015;25:196–202. doi: 10.1007/s00330-014-3377-5. [DOI] [PubMed] [Google Scholar]
- 55.Saftoiu A, Napoleon B, Arcidiacono PG, et al. Do we need contrast agents for EUS? Endosc Ultrasound. 2020;9:361–8. doi: 10.4103/eus.eus_20_20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Sharma M, Hollerbach S, Fusaroli P, et al. General principles of image optimization in EUS. Endosc Ultrasound. 2021;10:168–84. doi: 10.4103/eus.eus_80_20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Ren J, Zheng BW, Wang P, et al. Revealing impaired blood supply to the bile ducts on contrast-enhanced ultrasound:A novel diagnosis method to ischemic-type biliary lesions after orthotropic liver transplantation. Ultrasound Med Biol. 2013;39:753–60. doi: 10.1016/j.ultrasmedbio.2012.12.004. [DOI] [PubMed] [Google Scholar]
- 58.Zhang YC, Qu EZ, Ren J, et al. New diagnosis and therapy model for ischemic-type biliary lesions following liver transplantation –A retrospective cohort study. PLoS One. 2014;9:e105795. doi: 10.1371/journal.pone.0105795. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 59.Mukai S, Itoi T, Sofuni A, et al. Urgent and early EUS-guided biliary drainage in patients with acute cholangitis. Endosc Ultrasound. 2021;10:191–9. doi: 10.4103/eus.eus_70_20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Yamamoto Y, Ogura T, Nishioka N, et al. Risk factors for adverse events associated with bile leak during EUS-guided hepaticogastrostomy. Endosc Ultrasound. 2020;9:110–5. doi: 10.4103/eus.eus_68_19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Grewal P, Ahmad J. Bile duct injury due to drug induced liver injury. Curr Hepatol Rep. 2019;18:269–73. doi: 10.1007/s11901-019-00474-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Sundaram V, Björnsson ES. Drug-induced cholestasis. Hepatol Commun. 2017;1:726–35. doi: 10.1002/hep4.1088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Gudnason HO, Björnsson HK, Gardarsdottir M, et al. Secondary sclerosing cholangitis in patients with drug-induced liver injury. Dig Liver Dis. 2015;47:502–7. doi: 10.1016/j.dld.2015.03.002. [DOI] [PubMed] [Google Scholar]
- 64.Sandrasegaran K, Alazmi WM, Tann M, et al. Chemotherapy-induced sclerosing cholangitis. Clin Radiol. 2006;61:670–8. doi: 10.1016/j.crad.2006.02.013. [DOI] [PubMed] [Google Scholar]
- 65.Horsley-Silva JL, Dow EN, Menias CO, et al. Docetaxel induced sclerosing cholangitis. Dig Dis Sci. 2015;60:3814–6. doi: 10.1007/s10620-015-3914-3. [DOI] [PubMed] [Google Scholar]
- 66.Kusakabe A, Ohkawa K, Fukutake N, et al. Chemotherapy-induced sclerosing cholangitis caused by systemic chemotherapy. ACG Case Rep J. 2019;6:e00136. doi: 10.14309/crj.0000000000000136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Kawabata H, Honda M, Kawamura T, et al. Acase of primary sclerosing cholangitis (PSC) complicated with acute inflammatory demyelinating polyradiculoneuropathy. Nihon Shokakibyo Gakkai Zasshi. 2002;99:1503–7. [PubMed] [Google Scholar]
- 68.Pi B, Wang J, Tong Y, et al. Immune-related cholangitis induced by immune checkpoint inhibitors:A systematic review of clinical features and management. Eur J Gastroenterol Hepatol. 2021;33:e858–67. doi: 10.1097/MEG.0000000000002280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Koya Y, Shibata M, Shinohara N, et al. Secondary sclerosing cholangitis with hemobilia induced by pembrolizumab:Casereport and reviewofpublished work. Hepatol Res. 2019;49:950–6. doi: 10.1111/hepr.13329. [DOI] [PubMed] [Google Scholar]
- 70.Kawakami H, Tanizaki J, Tanaka K, et al. Imaging and clinicopathological features of nivolumab-related cholangitis in patients with non-small cell lung cancer. Invest New Drugs. 2017;35:529–36. doi: 10.1007/s10637-017-0453-0. [DOI] [PubMed] [Google Scholar]
- 71.Kashima J, Okuma Y, Shimizuguchi R, et al. Bile duct obstruction in a patient treated with nivolumab as second-line chemotherapy for advanced non-small-cell lung cancer:A case report. Cancer Immunol Immunother. 2018;67:61–5. doi: 10.1007/s00262-017-2062-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Nakano R, Shiomi H, Fujiwara A, et al. Clinical characteristics of ICI-related pancreatitis and cholangitis including radiographic and endoscopic findings. Healthcare (Basel) 2022;10:763. doi: 10.3390/healthcare10050763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Zen Y, Chen YY, Jeng YM, et al. Immune-related adverse reactions in the hepatobiliary system:Second-generation check-point inhibitors highlight diverse histological changes. Histopathology. 2020;76:470–80. doi: 10.1111/his.14000. [DOI] [PubMed] [Google Scholar]
- 74.Schwab GP, Wetscher GJ, Vogl W, et al. Methimazole-induced cholestatic liver injury, mimicking sclerosing cholangitis. Langenbecks Arch Chir. 1996;381:225–7. doi: 10.1007/BF00571690. [DOI] [PubMed] [Google Scholar]
- 75.Turkish A, Luo JJ, Lefkowitch JH. Ketamine abuse, biliary tract disease, and secondary sclerosing cholangitis. Hepatology. 2013;58:825–7. doi: 10.1002/hep.26459. [DOI] [PubMed] [Google Scholar]
- 76.Ahmad J, Rossi S, Rodgers SK, et al. Sclerosing cholangitis-like changes on magnetic resonance cholangiography in patients with drug induced liver injury. Clin Gastroenterol Hepatol. 2019;17:789–90. doi: 10.1016/j.cgh.2018.06.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Björnsson ES, Andrade RJ. Long-term sequelae of drug-induced liver injury. J Hepatol. 2022;76:435–45. doi: 10.1016/j.jhep.2021.10.011. [DOI] [PubMed] [Google Scholar]
- 78.Patil PA, Zhang X. Pathologic manifestations of gastrointestinal and hepatobiliary injury in immune checkpoint inhibitor therapy. Arch Pathol Lab Med. 2021;145:571–82. doi: 10.5858/arpa.2020-0070-RA. [DOI] [PubMed] [Google Scholar]
- 79.Hirasawa Y, Yoshimura K, Matsui H, et al. Acase report on severe nivolumab-induced adverse events similar to primary sclerosing cholangitis refractory to immunosuppressive therapy. Medicine (Baltimore) 2021;100:e25774. doi: 10.1097/MD.0000000000025774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Ludwig DR, Anderson MA, Itani M, et al. Secondary sclerosing cholangitis:Mimics of primary sclerosing cholangitis. Abdom Radiol (NY) 2022;18:1–15. doi: 10.1007/s00261-022-03551-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Chow LT, Ahuja AT, Kwong KH, et al. Mucinous cholangiocarcinoma:An unusual complication of hepatolithiasis and recurrent pyogenic cholangitis. Histopathology. 1997;30:491–4. doi: 10.1046/j.1365-2559.1997.00542.x. [DOI] [PubMed] [Google Scholar]
- 82.Catalano OA, Sahani DV, Forcione DG, et al. Biliary infections:Spectrum of imaging findings and management. Radiographics. 2009;29:2059–80. doi: 10.1148/rg.297095051. [DOI] [PubMed] [Google Scholar]
- 83.Stunell H, Buckley O, Geoghegan T, et al. Recurrent pyogenic cholangitis due to chronic infestation with Clonorchis sinensis (2006:8b) Eur Radiol. 2006;16:2612–4. doi: 10.1007/s00330-006-0354-7. [DOI] [PubMed] [Google Scholar]
- 84.Fry LC, Mönkemüller KE, Baron TH. Sclerosing cholangitis caused by Clonorchis sinensis. Gastrointest Endosc. 2002;56:114. doi: 10.1067/mge.2002.123611. [DOI] [PubMed] [Google Scholar]
- 85.Leung JW, Yu AS. Hepatolithiasis and biliary parasites. Baillieres Clin Gastroenterol. 1997;11:681–706. doi: 10.1016/s0950-3528(97)90016-6. [DOI] [PubMed] [Google Scholar]
- 86.Dietrich CF, Sharma M, Chaubal N, et al. Ascariasis imaging:Pictorial essay. Z Gastroenterol. 2017;55:479–89. doi: 10.1055/s-0043-104781. [DOI] [PubMed] [Google Scholar]
- 87.Dietrich CF, Atkinson NS, Lee WJ, et al. Never seen before?Opisthorchiasis and clonorchiasis. Z Gastroenterol. 2018;56:1513–20. doi: 10.1055/a-0751-3078. [DOI] [PubMed] [Google Scholar]
- 88.Frei P, Misselwitz B, Prakash MK, et al. Late biliary complications in human alveolar echinococcosis are associated with high mortality. World J Gastroenterol. 2014;20:5881–8. doi: 10.3748/wjg.v20.i19.5881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Cello JP. Acquired immunodeficiency syndrome cholangiopathy:Spectrum of disease. Am J Med. 1989;86:539–46. doi: 10.1016/0002-9343(89)90381-1. [DOI] [PubMed] [Google Scholar]
- 90.Naseer M, Dailey FE, Juboori AA, et al. Epidemiology, determinants, and management of AIDS cholangiopathy:A review. World J Gastroenterol. 2018;24:767–74. doi: 10.3748/wjg.v24.i7.767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Li N, Jie Z. The application of corticosteroids in COVID-19:A two-edged sword. J Transl Int Med. 2020;8:66–70. doi: 10.2478/jtim-2020-0011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Heucke N, Keitel V. COVID-19-associated cholangiopathy:What is left after the virus has gone? Hepatology. 2022;76:1560–2. doi: 10.1002/hep.32668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Bethineedi LD, Suvvari TK. Post COVID-19 cholangiopathy –A deep dive. Dig Liver Dis. 2021;53:1235–6. doi: 10.1016/j.dld.2021.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Bütikofer S, Lenggenhager D, Wendel Garcia PD, et al. Secondary sclerosing cholangitis as cause of persistent jaundice in patients with severe COVID-19. Liver Int. 2021;41:2404–17. doi: 10.1111/liv.14971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Nardo AD, Schneeweiss-Gleixner M, Bakail M, et al. Pathophysiological mechanisms of liver injury in COVID-19. Liver Int. 2021;41:20–32. doi: 10.1111/liv.14730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Klindt C, Jensen BE, Brandenburger T, et al. Secondary sclerosing cholangitis as a complication of severe COVID-19:A case report and review of the literature. Clin Case Rep. 2021;9:e04068. doi: 10.1002/ccr3.4068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Agarwal A, Chen A, Ravindran N, et al. Gastrointestinal and liver manifestations of COVID-19. J Clin Exp Hepatol. 2020;10:263–5. doi: 10.1016/j.jceh.2020.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Perico L, Benigni A, Casiraghi F, et al. Immunity, endothelial injury and complement-induced coagulopathy in COVID-19. Nat Rev Nephrol. 2021;17:46–64. doi: 10.1038/s41581-020-00357-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Bartoli A, Cursaro C, Andreone P. Severe acute respiratory syndrome coronavirus-2-associated cholangiopathies. Curr Opin Gastroenterol. 2022;38:89–97. doi: 10.1097/MOG.0000000000000808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Barbier L, Souche R, Slim K, et al. Long-term consequences of bile duct injury after cholecystectomy. J Visc Surg. 2014;151:269–79. doi: 10.1016/j.jviscsurg.2014.05.006. [DOI] [PubMed] [Google Scholar]
- 101.Eikermann M, Siegel R, Broeders I, et al. Prevention and treatment of bile duct injuries during laparoscopic cholecystectomy:The clinical practice guidelines of the European Association for Endoscopic Surgery (EAES) Surg Endosc. 2012;26:3003–39. doi: 10.1007/s00464-012-2511-1. [DOI] [PubMed] [Google Scholar]
- 102.Törnqvist B, Strömberg C, Persson G, et al. Effect of intended intraoperative cholangiography and early detection of bile duct injury on survival after cholecystectomy:Population based cohort study. BMJ. 2012;345:e6457. doi: 10.1136/bmj.e6457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.de'Angelis N, Catena F, Memeo R, et al. 2020 WSES guidelines for the detection and management of bile duct injury during cholecystectomy. World J Emerg Surg. 2021;16:30. doi: 10.1186/s13017-021-00369-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Schmidt SC, Settmacher U, Langrehr JM, et al. Management and outcome of patients with combined bile duct and hepatic arterial injuries after laparoscopic cholecystectomy. Surgery. 2004;135:613–8. doi: 10.1016/j.surg.2003.11.018. [DOI] [PubMed] [Google Scholar]
- 105.Lavelle LP, McEvoy SH, Ni Mhurchu E, et al. Cystic fibrosis below the diaphragm:Abdominal findings in adult patients. Radiographics. 2015;35:680–95. doi: 10.1148/rg.2015140110. [DOI] [PubMed] [Google Scholar]
- 106.Durieu I, Pellet O, Simonot L, et al. Sclerosing cholangitis in adults with cystic fibrosis:A magnetic resonance cholangiographic prospective study. J Hepatol. 1999;30:1052–6. doi: 10.1016/s0168-8278(99)80259-1. [DOI] [PubMed] [Google Scholar]
- 107.King LJ, Scurr ED, Murugan N, et al. Hepatobiliary and pancreatic manifestations of cystic fibrosis:MR imaging appearances. Radiographics. 2000;20:767–77. doi: 10.1148/radiographics.20.3.g00ma08767. [DOI] [PubMed] [Google Scholar]
- 108.Yonem O, Bayraktar Y. Clinical characteristics of Caroli's disease. World J Gastroenterol. 2007;13:1930–3. doi: 10.3748/wjg.v13.i13.1930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Chen Y, Ciobanu C, Mohrmann L. Hepatic sarcoidosis resembling primary sclerosing cholangitis. BMJ Case Rep. 2021;14:e243492. doi: 10.1136/bcr-2021-243492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Alam I, Levenson SD, Ferrell LD, et al. Diffuse intrahepatic biliary strictures in sarcoidosis resembling sclerosing cholangitis. Case report and review of the literature. Dig Dis Sci. 1997;42:1295–301. doi: 10.1023/a:1018874612166. [DOI] [PubMed] [Google Scholar]
- 111.Jebran AF, Schmidt WE, Kahraman A, et al. Sarcoidosis of the intra and extrahepatic bile ducts with concomitant cholangitis in a patient with ulcerative colitis. Case Rep Gastroenterol. 2019;13:153–8. doi: 10.1159/000497470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Ben Slama Trabelsi A, Issaoui D, Ksiaa M, et al. Sclerosing cholangitis in Behçet's disease. Case Rep Med. 2013;2013:692980. doi: 10.1155/2013/692980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Agrawal R, Guzman G, Karimi S, et al. Immunoglobulin G4 associated autoimmune cholangitis and pancreatitis following the administration of nivolumab:A case report. World J Clin Cases. 2022;10:7124–9. doi: 10.12998/wjcc.v10.i20.7124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Miura F, Asano T, Amano H, et al. Resected case of eosinophilic cholangiopathy presenting with secondary sclerosing cholangitis. World J Gastroenterol. 2009;15:1394–7. doi: 10.3748/wjg.15.1394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Matsumoto N, Yokoyama K, Nakai K, et al. Acase of eosinophilic cholangitis:Imaging findings of contrast-enhanced ultrasonography, cholangioscopy, and intraductal ultrasonography. World J Gastroenterol. 2007;13:1995–7. doi: 10.3748/wjg.v13.i13.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Chen Y, Cai S, Dong L, et al. Update on classification, diagnosis, and management of immunoglobulin G4-related disease. Chin Med J (Engl) 2022;135:381–92. doi: 10.1097/CM9.0000000000001891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Tanaka A, Mori M, Kubota K, et al. Epidemiological features of immunoglobulin G4-related sclerosing cholangitis in Japan. J Hepatobiliary Pancreat Sci. 2020;27:598–603. doi: 10.1002/jhbp.793. [DOI] [PubMed] [Google Scholar]
- 118.Guo T, Xu T, Zhang S, et al. The role of EUS in diagnosing focal autoimmune pancreatitis and differentiating it from pancreatic cancer. Endosc Ultrasound. 2021;10:280–7. doi: 10.4103/EUS-D-20-00212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Shi W, Zhang S, Xu H, et al. Delayed gastric bleeding after EUS-guided fine-needle aspiration of autoimmune pancreatitis. Endosc Ultrasound. 2021;10:73–4. doi: 10.4103/eus.eus_74_20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Monachese M, Lee PJ, Harris K, et al. EUS and secretin endoscopic pancreatic function test predict evolution to overt structural changes of chronic pancreatitis in patients with nondiagnostic baseline imaging. Endosc Ultrasound. 2021;10:116–23. doi: 10.4103/EUS-D-20-00138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Nakazawa T, Kamisawa T, Okazaki K, et al. Clinical diagnostic criteria for IgG4-related sclerosing cholangitis 2020:(Revision of the clinical diagnostic criteria for IgG4-related sclerosing cholangitis 2012) J Hepatobiliary Pancreat Sci. 2021;28:235–42. doi: 10.1002/jhbp.913. [DOI] [PubMed] [Google Scholar]
- 122.Ghazale A, Chari ST, Zhang L, et al. Immunoglobulin G4-associated cholangitis:Clinical profile and response to therapy. Gastroenterology. 2008;134:706–15. doi: 10.1053/j.gastro.2007.12.009. [DOI] [PubMed] [Google Scholar]
- 123.Kawa S, Kamisawa T, Notohara K, et al. Japanese clinical diagnostic criteria for autoimmune pancreatitis, 2018:Revision of Japanese clinical diagnostic criteria for autoimmune pancreatitis, 2011. Pancreas. 2020;49:e13–4. doi: 10.1097/MPA.0000000000001443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Asaki S, Ota K, Kanazawa N, et al. Ultrasonic endoscopy. Tohoku J Exp Med. 1983;141:9–12. doi: 10.1620/tjem.141.9. [DOI] [PubMed] [Google Scholar]
- 125.Ohara H, Okazaki K, Tsubouchi H, et al. Clinical diagnostic criteria of IgG4-related sclerosing cholangitis 2012. J Hepatobiliary Pancreat Sci. 2012;19:536–42. doi: 10.1007/s00534-012-0521-y. [DOI] [PubMed] [Google Scholar]
- 126.Nakazawa T, Ohara H, Sano H, et al. Schematic classification of sclerosing cholangitis with autoimmune pancreatitis by cholangiography. Pancreas. 2006;32:229. doi: 10.1097/01.mpa.0000202941.85955.07. [DOI] [PubMed] [Google Scholar]
- 127.Nakazawa T, Naitoh I, Hayashi K, et al. Diagnostic criteria for IgG4-related sclerosing cholangitis based on cholangiographic classification. J Gastroenterol. 2012;47:79–87. doi: 10.1007/s00535-011-0465-z. [DOI] [PubMed] [Google Scholar]
- 128.Mo Z, Ramen K, Shan Y, et al. Localized intrahepatic IgG4-related sclerosing cholangitis (IgG4-SC) as an additional type of IgG4-SC:A systematic analysis of 12 cases. Scand J Gastroenterol. 2018;53:312–8. doi: 10.1080/00365521.2018.1424233. [DOI] [PubMed] [Google Scholar]
- 129.Naitoh I, Nakazawa T. Classification and diagnostic criteria for IgG4-related sclerosing cholangitis. Gut Liver. 2022;16:28–36. doi: 10.5009/gnl210116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Nakazawa T, Naitoh I, Hayashi K, et al. Diagnosis of IgG4-related sclerosing cholangitis. World J Gastroenterol. 2013;19:7661–70. doi: 10.3748/wjg.v19.i43.7661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Khoo S, Do ND, Kongkam P. Efficacy and safety of EUS biliary drainage in malignant distal and hilar biliary obstruction:A comprehensive review of literature and algorithm. Endosc Ultrasound. 2020;9:369–79. doi: 10.4103/eus.eus_59_20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Wee D, Izard S, Grimaldi G, et al. EUS assessment for intermediate risk of choledocholithiasis after a negative magnetic resonance cholangiopancreatography. Endosc Ultrasound. 2020;9:337–44. doi: 10.4103/eus.eus_57_20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Dong Y, D'Onofrio M, Hocke M, et al. Autoimmune pancreatitis:Imaging features. Endosc Ultrasound. 2018;7:196–203. doi: 10.4103/eus.eus_23_17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Hocke M, Ignee A, Dietrich CF. Contrast-enhanced endoscopic ultrasound in the diagnosis of autoimmune pancreatitis. Endoscopy. 2011;43:163–5. doi: 10.1055/s-0030-1256022. [DOI] [PubMed] [Google Scholar]
- 135.Bauditz J, Schade T, Wermke W. Sonographic diagnosis of hilar cholangiocarcinomas by the use of contrast agents. Ultraschall Med. 2007;28:161–7. doi: 10.1055/s-2007-962825. [DOI] [PubMed] [Google Scholar]
- 136.Naitoh I, Kamisawa T, Tanaka A, et al. Clinical characteristics of immunoglobulin IgG4-related sclerosing cholangitis:Comparison of cases with and without autoimmune pancreatitis in a large cohort. Dig Liver Dis. 2021;53:1308–14. doi: 10.1016/j.dld.2021.02.009. [DOI] [PubMed] [Google Scholar]
- 137.Tanaka A, Tazuma S, Okazaki K, et al. Clinical features, response to treatment, and outcomes of IgG4-related sclerosing cholangitis. Clin Gastroenterol Hepatol. 2017;15:920–6.e3. doi: 10.1016/j.cgh.2016.12.038. [DOI] [PubMed] [Google Scholar]
- 138.Huggett MT, Culver EL, Kumar M, et al. Type 1 autoimmune pancreatitis and IgG4-related sclerosing cholangitis is associated with extrapancreatic organ failure, malignancy, and mortality in a prospective UK cohort. Am J Gastroenterol. 2014;109:1675–83. doi: 10.1038/ajg.2014.223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Nakazawa T, Ikeda Y, Kawaguchi Y, et al. Isolated intrapancreatic IgG4-related sclerosing cholangitis. World J Gastroenterol. 2015;21:1334–43. doi: 10.3748/wjg.v21.i4.1334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Takagi Y, Kubota K, Takayanagi T, et al. Clinical features of isolated proximal-type immunoglobulin G4-related sclerosing cholangitis. Dig Endosc. 2019;31:422–30. doi: 10.1111/den.13320. [DOI] [PubMed] [Google Scholar]
- 141.Manganis CD, Chapman RW, Culver EL. Review of primary sclerosing cholangitis with increased IgG4 levels. World J Gastroenterol. 2020;26:3126–44. doi: 10.3748/wjg.v26.i23.3126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Oseini AM, Chaiteerakij R, Shire AM, et al. Utility of serum immunoglobulin G4 in distinguishing immunoglobulin G4-associated cholangitis from cholangiocarcinoma. Hepatology. 2011;54:940–8. doi: 10.1002/hep.24487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Tanaka Y, Kamimura K, Nakamura R, et al. Usefulness of ultrasonography to assess the response to steroidal therapy for the rare case of type 2b immunoglobulin G4-related sclerosing cholangitis without pancreatitis:A case report. World J Clin Cases. 2020;8:5821–30. doi: 10.12998/wjcc.v8.i22.5821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Boonstra K, Culver EL, de Buy Wenniger LM, et al. Serum immunoglobulin G4 and immunoglobulin G1 for distinguishing immunoglobulin G4-associated cholangitis from primary sclerosing cholangitis. Hepatology. 2014;59:1954–63. doi: 10.1002/hep.26977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Culver EL, Sadler R, Bateman AC, et al. Increases in IgE, Eosinophils, and mast cells can be used in diagnosis and to predict relapse of IgG4-related disease. Clin Gastroenterol Hepatol. 2017;15:1444–52.e6. doi: 10.1016/j.cgh.2017.02.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Marrelli D, Caruso S, Pedrazzani C, et al. CA19-9 serum levels in obstructive jaundice:Clinical value in benign and malignant conditions. Am J Surg. 2009;198:333–9. doi: 10.1016/j.amjsurg.2008.12.031. [DOI] [PubMed] [Google Scholar]
- 147.Du S, Liu G, Cheng X, et al. Differential diagnosis of immunoglobulin G4-associated cholangitis from cholangiocarcinoma. J Clin Gastroenterol. 2016;50:501–5. doi: 10.1097/MCG.0000000000000509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Hori Y, Chari ST, Tsuji Y, et al. Diagnosing biliary strictures:Distinguishing IgG4-related sclerosing cholangitis from cholangiocarcinoma and primary sclerosing cholangitis. Mayo Clin Proc Innov Qual Outcomes. 2021;5:535–41. doi: 10.1016/j.mayocpiqo.2021.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Naitoh I, Nakazawa T. Endoscopic retrograde cholangiopancreatography and intraductal ultrasonography in the diagnosis of autoimmune pancreatitis and IgG4-related sclerosing cholangitis. J Med Ultrason (2001) 2021;48:573–80. doi: 10.1007/s10396-021-01114-1. [DOI] [PubMed] [Google Scholar]
- 150.Kubota K, Kato S, Uchiyama T, et al. Discrimination between sclerosing cholangitis-associated autoimmune pancreatitis and primary sclerosing cholangitis, cancer using intraductal ultrasonography. Dig Endosc. 2011;23:10–6. doi: 10.1111/j.1443-1661.2010.01039.x. [DOI] [PubMed] [Google Scholar]
- 151.Seifert H, Fusaroli P, Arcidiacono PG, et al. Controversies in EUS:Do we need miniprobes? Endosc Ultrasound. 2021;10:246–69. doi: 10.4103/EUS-D-20-00252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Nakanuma Y, Zen Y. Pathology and immunopathology of immunoglobulin G4-related sclerosing cholangitis:The latest addition to the sclerosing cholangitis family. Hepatol Res. 2007;37(Suppl 3):S478–86. doi: 10.1111/j.1872-034X.2007.00243.x. [DOI] [PubMed] [Google Scholar]
- 153.Kamisawa T, Nakazawa T, Tazuma S, et al. Clinical practice guidelines for IgG4-related sclerosing cholangitis. J Hepatobiliary Pancreat Sci. 2019;26:9–42. doi: 10.1002/jhbp.596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Hirano K, Tada M, Sasahira N, et al. Incidence of malignancies in patients with IgG4-related disease. Intern Med. 2014;53:171–6. doi: 10.2169/internalmedicine.53.1342. [DOI] [PubMed] [Google Scholar]