Abstract
Purpose
Brain metastases (BMs) are a common source of morbidity and mortality. Guidelines do not advise brain surveillance for locally advanced non-small cell lung cancer (LA-NSCLC). We describe the incidence, time to development, presentation, and management of BMs after definitive chemoradiotherapy (CRT).
Methods and Materials
We reviewed records of patients with LA-NSCLC treated with CRT within the period from 2013 to 2020. Descriptive statistics were used to characterize the population and the Kaplan-Meier method was used to estimate time to BM. Fisher exact tests and Wilcoxon rank-sum tests were used to compare outcomes between symptomatic and asymptomatic patients.
Results
A total of 219 patients were reviewed including 96 with squamous cell carcinoma, 88 with adenocarcinoma, and 35 with large cell/not otherwise specified (LC/NOS). Thirty-nine patients (17.8%) developed BMs: 35 (90%) symptomatic and 4 (10%) asymptomatic. The rate of BM was highest in LC/NOS (34.3%) and adenocarcinoma (23.9%). Ninety percent of BMs occurred within 2 years. All asymptomatic patients underwent stereotactic radiosurgery alone, compared with 40% of symptomatic patients (P = .04). Symptomatic patients were more likely to require hospitalization (65.7% vs 0%, P = .02), craniotomy (25.7% vs 0%, not significant), and steroids (91.4% vs 0%, P < .001). Cumulative BM volume was higher for symptomatic patients (4 vs 0.24 cm3, P < .001) as was median greatest axial dimension (2.18 vs 0.52 cm, P < .001).
Conclusions
We identified a high rate of BMs, particularly in LC/NOS and adenocarcinoma histology NSCLC. The majority were symptomatic. These results provide rationale for post-CRT magnetic resonance imaging brain surveillance for patients at high risk of BM.
Introduction
Brain metastases (BMs) from locally advanced non-small cell lung cancer (LA-NSCLC; stage IIB-III)1 are a common form of distant metastatic failure and a significant source of morbidity and mortality.2,3 The incidence of BM varies widely with stage, histopathology, and molecular status (EGFR, ALK, KRAS, etc.), but has been reported to be as high as 25% to 43%.4, 5, 6, 7, 8 The PACIFIC study, which established consolidative immunotherapy after definitive chemoradiotherapy (CRT) as the standard of care in LA-NSCLC, reported that at progression more than 60% of new extrathoracic recurrences involved the brain, regardless of immunotherapy receipt.9 Despite this, there is currently no standard recommendation for magnetic resonance imaging (MRI) for brain surveillance after completion of CRT. At present, the most recently updated guidelines from the National Comprehensive Cancer Network recommend repeat brain MRI and positron emission tomography/computed tomography for restaging only after recurrence.10 Given that the brain represents a common extrathoracic site of first failure, this “reactive” imaging strategy is often inadequate for early detection of BMs when they are still asymptomatic and potentially amenable to limited treatment such as stereotactic radiosurgery (SRS).9,11,12 As a result, most BMs are identified only after they become symptomatic, necessitating aggressive management such as corticosteroids, craniotomy, or whole brain radiation therapy, all of which can dramatically affect quality of life.13
In addition to the negative effect on patients’ quality of life, the delayed detection of BM until symptomatic presentation has been shown to lead to worse cancer-related outcomes and a greater financial burden on the medical system.14 Despite the established incidence and significant clinical effect of BMs in LA-NSCLC, the rate and timing of symptomatic versus asymptomatic BMs are not well reported. One of the most recent phase 3 randomized prophylactic cranial irradiation (PCI) studies, NVALT-11, specifically assessed stage III NSCLC (including adenocarcinoma and squamous cell carcinomas) and demonstrated a 27% incidence of symptomatic BM in the observation arm where most metastases occurred within 2 years of definitive therapy. Importantly, brain MRI was only ordered at the onset of clinical symptoms, and these were not ordered as surveillance examinations.11
Detection of BM after CRT for LA-NSCLC presents a unique opportunity to institute an evidence-based MRI screening strategy in LA-NSCLC patients at highest risk for BM. The development and implementation of a high-yield brain MRI schedule within the first 1 to 2 years post-CRT would be hypothesized to increase detection of asymptomatic BM. Earlier detection may mitigate the need for many invasive/morbid treatments and allow for more targeted interventions with fewer adverse effects.
Methods and Materials
Study population and treatments
In this institutional review board–approved study, we identified and reviewed consecutive patients treated at our single institution with LA-NSCLC treated with definitive CRT within the period from 2013 to 2020. We excluded patients treated with palliative radiation therapy. Clinicopathologic features including histologic subtype, T stage, N stage, and clinical prognostic stage were recorded. Treatment details were also recorded including radiation therapy dose, modality, chemotherapy agents, and receipt of adjuvant immunotherapy for consolidation.
Outcomes of interest
Development of BM was determined from the medical record and defined as any radiologically confirmed evidence of intracranial metastasis on MRI. The date of BM was determined as date of first detection of brain disease on MRI or computed tomography. The time to BM (TTBM) was defined as the time interval from completion of CRT to BM. Patients who did not develop BM were censored at last follow-up or death. Symptoms at time of BM presentation were recorded as well as methods of subsequent management. Time to local failure (TTLF) was defined as the time interval from completion of CRT to local failure (recurrent tumor with at least some portion within prior treatment volume), time to regional failure (TTRF) was defined as the time interval from completion of CRT to regional failure (new lymph nodes involved that were not included in initial treatment volume), and time to distant failure (TTDF) was defined as the time interval from completion of CRT to distant failure (metastatic disease other than BM, including subsequent lung primary). Patients without an event were censored at last follow-up or death.
Statistical analysis
Descriptive statistics were used to characterize the patient population, including incidence of BM by histology and management of symptomatic versus asymptomatic BMs. The Kaplan-Meier method was used to estimate TTBM, TTLF, TTRF, and TTDF, including the percentage of patients who were event-free at 24 months. Log-rank tests were used to compare the time-to-event outcomes by histology. Fisher exact tests were used to compare proportions between symptomatic and asymptomatic patients, and Wilcoxon rank-sum tests were used to compare continuous variables between symptomatic and asymptomatic patients and between management strategies.
Brain metastasis volume measurements
Volumetric measurements were made by delineating individual BM on the diagnostic contrast-enhanced MRI (high-resolution T1-contrasted sequence) as a cumulative structure in the treatment planning software (RayStation, RaySearch Laboratories AB, Stockholm, Sweden). The largest individual lesion was measured in the axial plane, and the single greatest dimension was recorded. The number of lesions were counted for each patient.
Results
Patient and treatment characteristics
A total of 219 patients met inclusion criteria and are summarized in Table 1. Histology included squamous cell carcinoma (SCC; n = 96), adenocarcinoma (n = 88), and large cell/not otherwise specified (LC/NOS; n = 35). Median age at the time of CRT was 67 years. Most patients (58.9%) were male; 210 (96%) were current or former smokers. Eastern Cooperative Oncology Group performance status was 0 to 1 in 76.7%. T stages were as follows: T0 (11.9%), T1 (21.9%), T2 (26.5%), T3 (18.3%), and T4 (21.5%). The distribution of N stage was as follows: N0 (5%), N1 (14.2%), N2 (60.7%), and N3 (20.1%). Clinical stage was IIB to IIIC. One patient was identified as having a single contralateral lung lesion that potentially represented a synchronous primary. This patient was treated initially with induction systemic therapy, followed by definitive chemoradiation to their locally advanced disease followed by stereotactic body radiation therapy to the contralateral nodule.
Table 1.
Patient characteristics and treatment details (N = 219)
| Characteristic | Median (IQR) or n (%) |
|---|---|
| Age (y) | 67 (60-73) |
| Sex | |
| Male | 129 (58.9) |
| Female | 90 (41.1) |
| Eastern Cooperative Oncology Group performance status | |
| 0 | 29 (13.2) |
| 1 | 139 (63.5) |
| 2 | 40 (18.3) |
| 3 | 11 (5.0) |
| Smoking | |
| Current smoker | 82 (37.4) |
| Former smoker | 128 (58.4) |
| Histologic subtype | |
| Squamous cell carcinoma | 96 (43.8) |
| Adenocarcinoma | 88 (40.2) |
| Large cell/not otherwise specified | 35 (16.0) |
| Tumor stage | |
| T0 | 26 (11.9) |
| T1 | 48 (21.9) |
| T2 | 58 (26.5) |
| T3 | 40 (18.3) |
| T4 | 47 (21.5) |
| Nodal stage | |
| N0 | 11 (5.0) |
| N1 | 31 (14.2) |
| N2 | 133 (60.7) |
| N3 | 44 (20.1) |
| Clinical disease stage | |
| IIB | 20 (9.1) |
| IIIA | 104 (47.5) |
| IIIB | 86 (39.3) |
| IIIC | 9 (4.1) |
| Patients with brain metastases | 39 (17.8) |
| Therapy received after brain metastasis diagnosis | |
| No treatment | 4 (10.3) |
| Steroids | 32 (82.1) |
| Surgery alone | 1 (2.6) |
| Stereotactic radiosurgery alone | 18 (46.2) |
| Surgery and GKRS | 8 (20.5) |
| Whole brain radiation therapy | 8 (20.5) |
Abbreviations: GKRS = gamma knife radiosurgery; IQR, interquartile range.
Incidence and timing of brain metastases
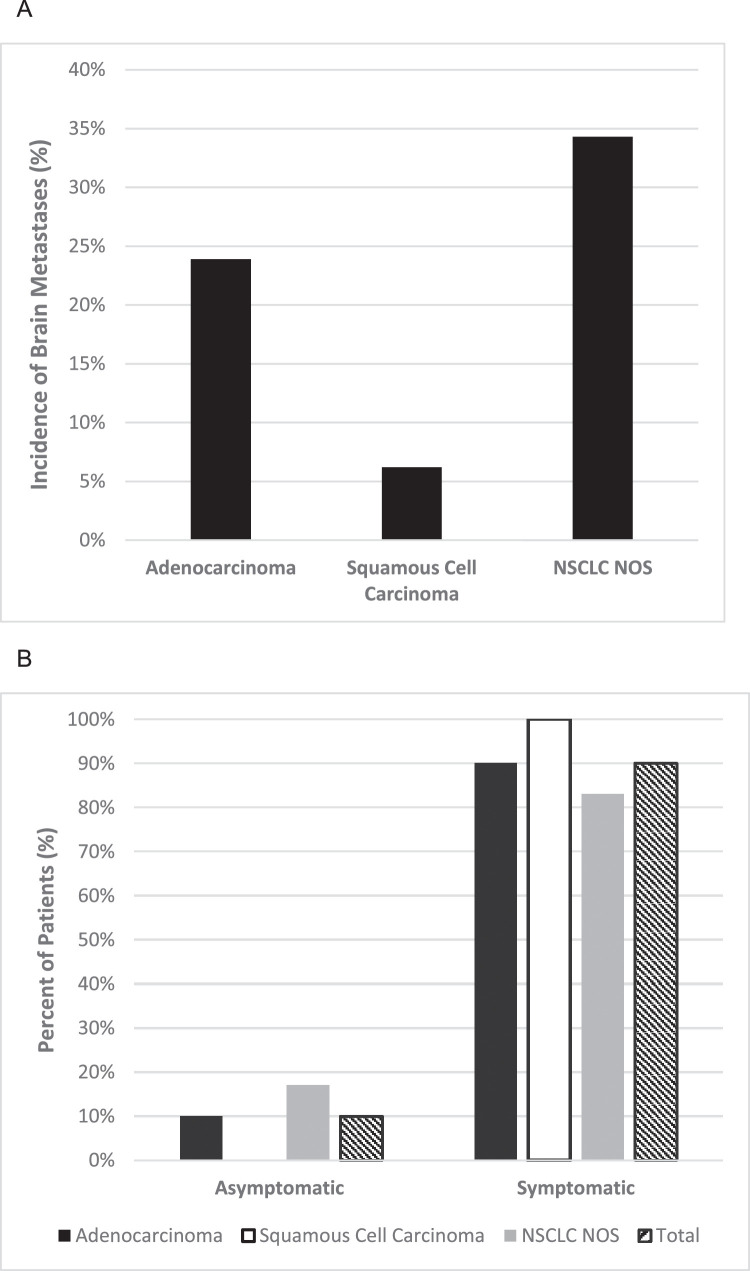
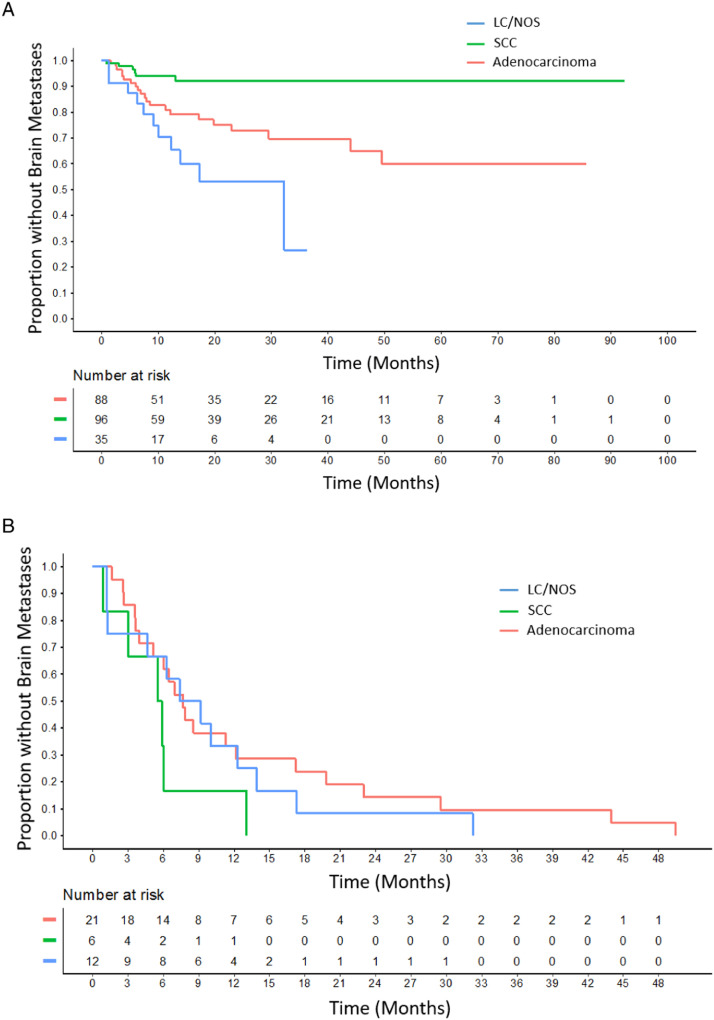
In total, 39 patients (17.8%) developed BM, 38 (97%) of whom had pretreatment staging MRI brain. One patient did not have documentation available confirming that pretreatment brain imaging had been performed. Incidence of BM was highest in the LC/NOS group (34.3%), followed by adenocarcinoma (23.9%), and SCC (6.2%) (Fig. 1A). The percentage of patients without BM at 24 months was 72.9% (95% confidence interval [CI], 62.5%-84.9%) for adenocarcinoma, 92.2% (86.3%-98.5%) for SCC, and 53.3% (35.4%-80.1%) for LC/NOS. TTBM, TTLF, TTRF, and TTDF results are shown in Table 2. Only TTBF was found to differ significantly between adenocarcinoma, SCC, and LC/NOS (P < .001), whereas TTDF did not differ significantly based on histology. Median TTBM was 7.7 months for adenocarcinoma, 5.7 months for SCC, and 8.3 months for LC/NOS. For the entire cohort, 1-year freedom from brain failure was 85% (95% CI, 79.9%-90.5%); for adenocarcinoma, 80.9% (72.3%-90.5%); for SCC, 94.0% (89.1%-99.3%); and LC/NOS, 70.4% (54.7%-90.5%). Of patients who developed BMs, overall 69.2% (95% CI, 50.7%-80.8%) occurred in the first year: 66.7% (39.0%-81.8%) for adenocarcinoma, 83.3% (0.3%-99.7%) for SCC, and 66.7% (25.8%-85.0%) for LC/NOS. TTBM among the entire cohort is shown in Fig. 2A stratified by histology. TTBM is shown for each histology only among patients who developed BM (Fig. 2B).
Figure 1.
A, Incidence of brain metastases in locally advanced NSCLC after definitive treatment by histology. B, Likelihood of symptomatic versus asymptomatic presentation at time of brain metastasis diagnosis. Abbreviations: NOS = not otherwise specified; NSCLC = non-small cell lung cancer.
Table 2.
Percentage event-free at 24 months by histology
| Histology | BM/n | TTBM, % (95% CI) | TTLF, % (95% CI) | TTRF, % (95% CI) | TTDF, % (95% CI) |
|---|---|---|---|---|---|
| Adenocarcinoma | 21/88 | 72.9 (62.5-84.9) | 79.2 (69.0-90.9) | 75.9 (65.7-87.7) | 64.8 (53.6-78.4) |
| SCC | 6/96 | 92.2 (86.3-98.5) | 64.0 (53.4-76.9) | 72.8 (62.1-85.3) | 68.2 (57.7-80.5) |
| LC/NOS | 12/35 | 53.3 (35.4-80.1) | 71.7 (54.5-94.4) | 65.9 (47.4-91.6) | 31.8 (13.6-74.4) |
| Total | 39/219 | 78.2 (71.7-85.2) | 71.0 (63.7-79.0) | 73.0 (65.8-80.9) | 62.8 (55.2-71.5) |
| P value | <.001 | .2 | .5 | .1 |
Abbreviations: BM = brain metastasis; CI = confidence interval; LC/NOS = large cell/not otherwise specified; SCC = squamous cell carcinoma; TTBM = time to brain metastasis; TTDF = time to distant failure; TTLF = time to local failure; TTRF = time to regional failure.
Figure 2.
A, Time to brain metastasis between histology groups for all patients (P < .001). B, Time to brain metastasis in patients who developed brain metastases. Time is shortest for squamous cell carcinoma (SCC) followed by large cell/not otherwise specified (LC/NOS) and adenocarcinoma.
Presentation and management of brain metastases
Thirty-five of 39 patients (89.7%) received a diagnosis of symptomatic BM based on the development of neurologic symptoms including focal neurologic deficits or signs of elevated intracranial pressure (headache, nausea, vomiting, etc) as shown in Fig. 1B. Narrative descriptions of the presentations and clinical courses for each patient who developed BM are provided in Table E1. Detection of asymptomatic BM occurred in only 4 patients, all but 1 of which were incidentally noted at time of routine restaging for extracranial progression detected during standard of care follow-up. One case involved a new adrenal metastasis, and 2 cases involved intrathoracic progression. One patient underwent follow-up brain MRI for a previously noted vascular abnormality, which resolved; however, new BMs were incidentally detected. Additional treatment related information for these patients with BM detected asymptomatic can be found in Table E4. For patients presenting with symptomatic BM, 91.4% required corticosteroids versus 0% for those without symptoms (P < .001). Patients with symptomatic BM were more likely to require hospitalization at presentation (65.7% vs 0%, P = .02). All asymptomatic patients were able to undergo SRS alone, whereas only 40% of symptomatic patients received SRS monotherapy (P = .04). There was no statistically significant difference in BM incidence based on the presence or absence of adjuvant immunotherapy for any histology (overall 17.1% vs 18.1%, P = 1.00). Eight patients received whole brain radiation therapy, and in all 8 cases, 3-dimensional plans using standard opposed lateral fields were used. All SRS cases were treated using Gamma Knife radiosurgery performed on the Leksell Gamma Knife Perfexion. Eighteen patients received SRS as upfront therapy, with 8 more patients requiring surgical resection before adjuvant SRS to the resection cavity. Median SRS dose was 20 Gy (interquartile range, 20-22) prescribed to the 50% isodose line (interquartile range, 50-80).
Brain metastasis volume and associated management
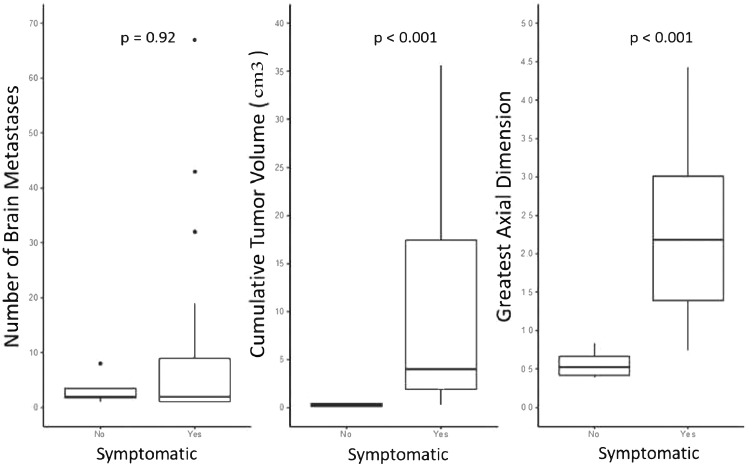
BM were found to be larger both in volume as well as single greatest axial dimension in patients presenting with neurologic symptoms compared with those discovered incidentally. Median cumulative BM volume was higher for symptomatic versus asymptomatic patients (4 vs 0.24 cm3, P < .001) as was median greatest axial dimension (2.18 vs 0.52 cm, P < .001) (Fig. 3; Table E2). Burden of brain disease compared with management (number, volume, and greatest axial dimension) is provided in Table E3 and Fig. E2. Of note, patients undergoing SRS alone as their initial treatment had fewer BMs at diagnosis (1 vs 7, P < .001), lower volume lesions (1.6 vs 16.4 cm3, P < .001), and smaller greatest axial dimensions (1.4 vs 2.4 cm, P = .01). Patients with larger cumulative tumor volume were more likely to require craniotomy as part of their initial management (22.6 vs 2.1 cm3, P = .006). Patients who required whole brain radiation therapy had significantly more metastases at diagnosis (17 vs 2, P < .001). Patients hospitalized for symptoms had larger median axial dimensions than those managed as outpatients (2.27 vs 1.22 cm, P = .04). Patients receiving steroids had greater cumulative lesion volume (4.86 vs 0.39 cm3, P = .007) and greater axial dimensions (2.28 vs 0.84 cm, P = .003). Table 3 provides further details regarding managment of symptomatic vs asymptomatic BMs.
Figure 3.
Burden of brain disease between asymptomatic and symptomatic patients.
Table 3.
Management of symptomatic versus asymptomatic brain metastases
| Treatment | Overall, n (%) | Asymptomatic, n (%) | Symptomatic, n (%) | P value |
|---|---|---|---|---|
| No treatment | 39 (18.3) | 0 (0) | 4 (11.4) | 1 |
| Steroids | 32 (82.1) | 0 (0) | 32 (91.4) | <.001 |
| Prior immunotherapy | 10 (25.6) | 0 (0) | 10 (28.6) | .56 |
| Active immunotherapy | 3 (7.7) | 0 (0) | 3 (8.6) | 1 |
| Stereotactic radiosurgery alone | 18 (46.2) | 4 (100) | 14 (40.0) | .04 |
| Surgery | 9 (35.3) | 0 (0) | 9 (25.7) | .56 |
| Hospitalization for brain met | 23 (59.0) | 0 (0) | 23 (65.7) | .02 |
| Whole brain radiation therapy | 8 (20.5) | 0 (0) | 8 (22.9) | .56 |
Discussion
Recent guidelines from the American Society of Clinical Oncology recommend against routine surveillance brain MRI for stage I to III NSCLC after treatment. This recommendation is listed as an informal consensus opinion based on the low quality of evidence and is indifferent to histology/mutation status as well as disease burden.15 With the advent of detailed molecular profiling through next-generation sequencing techniques, it is apparent that NSCLC represents an incredibly diverse group of malignancies. Adenocarcinoma and LC/NOS histologies are already known to have a higher incidence of BMs than squamous cell carcinoma.16 Furthermore, histology has been shown to be predictive of patterns of brain failure and outcomes following SRS.17
Multiple retrospective series have shown a strong association between EGFR mutation and the development of BM, with some series reporting greater than 40% incidence of BM in patients with EGFR-mutated NSCLC.18 It would stand to reason that a patient with stage IIIC EGFR-positive adenocarcinoma may benefit from closer brain follow-up than a patient with stage IIIA squamous cell carcinoma. This is especially true in the era of modern high-resolution, thin-slice, contrast-enhanced MRI techniques that are capable of detecting BM on the order of 2 to 3 mm, which would most likely still be asymptomatic.19
No randomized trials have specifically evaluated the utility of surveillance MRI in the setting of high-risk LA-NSCLC. Several randomized studies have shown that PCI in this setting can reduce the incidence of BM by over 50%. However, this approach has never translated into a clear improvement in overall survival. Given the lack of clear survival benefit, along with the potential for neurocognitive toxicity, PCI for NSCLC is not routinely recommended.15 Of note, the ongoing CC003 trial is presently evaluating the role of hippocampal avoidance in PCI to potentially further reduce the cognitive adverse effects associated with irradiation of the whole brain.20 Instead, SRS, a more focused form of radiation, has now become a mainstay for treating small BMs following detection rather than prophylactically treating the entire brain. SRS has excellent local control and minimal to mild cognitive toxicities.21 The limits on size and number of metastases are not well defined and depend on clinical scenario, but generally SRS may be considered for lesions <3 cm, and prospective studies are assessing the use of SRS in as many as 15 lesions.22 This noninvasive technique is ideal for treating most asymptomatic BM.
We demonstrated that nearly all (90%) BM detected in our series were only found as a direct result of neurologic symptoms. The included narratives for each presentation in Table E1 highlight the debilitating nature of these neurologic symptoms. If these BM had been detected while asymptomatic, long-term high-dose steroids, craniotomy, and or whole brain radiation, potentially could have been avoided with a clear effect on quality of life. Morbidities associated with surgical resection of BM are reported to occur in 2% to 25% of patients and include postoperative hemorrhage, pulmonary embolism, cerebrospinal fluid leak, and others. Death within 30 days of surgery ranges from 2% to 11%.23, 24, 25, 26 It is clear that a symptomatic presentation of BM portends a worse outcome. Lester et al14 demonstrated that patients who were treated for symptomatic BM were twice as likely to die of neurologic death from brain progression than those who presented with asymptomatic BM (42% vs 20%; P < .0001). Additionally, they reported that management of symptomatic BM required an average additional cost approximately 40% more per patient compared with those with asymptomatic disease.14
Our data highlight that there is a wide variation of disease behavior in LA-NSCLC as a group, with adenocarcinoma and LC/NOS showing an increased risk of BM over SCC. It also highlights the pattern of presentation of BM in this population. With important implications for potential future management, we found a statistically significant association with a need for steroid therapy and hospitalization in those presenting with symptomatic BM compared with those discovered when asymptomatic. We also found that patients presenting with neurologic symptoms were significantly less likely to be candidates for SRS alone to manage of their disease.
Taken in this context, our data provide promising rationale for an evidence-based brain MRI surveillance strategy for high-risk patients with LA-NSCLC (ie, nonsquamous histology) following definitive CRT. MRI surveillance at regular intervals for the first year would be expected to capture a large proportion of those lung cancer patients who will eventually fail in the brain, as we found that 90% of patients who developed BM did so within 2 years.
Our analysis is inherently limited by its retrospective nature and includes a relatively small number of patients whose BMs were detected asymptomatically. These factors limit direct comparison of the symptomatic and asymptomatic groups. Additionally, genetic markers commonly assayed in lung cancer (eg, PDL1, EGFR, ALK) would provide useful information concerning further risk stratification for the development of BMs, but these data were not routinely available in our database. However, our findings clearly indicate that when BMs occur following definitive CRT for LA-NSCLC, they are nearly always detected due to symptomatic presentation and are greater in number and volume compared with lesions detected in asymptomatic patients. Earlier detection may avoid the development of debilitating symptoms as well as the need for interventions such as steroids, emergency craniotomy, or whole brain radiation. An evidence-based MRI surveillance strategy could have a substantial clinical effect on patient outcomes and quality of life. Clinical trials are presently being designed to prospectively validate these data and to assess the role of MRI surveillance following definitive therapy for LA-NSCLC.
Conclusion
In this study, we identify high rates of BM in patients with nonsquamous histology after definitive CRT for LA-NSCLC. The vast majority of patients with BM presented with neurologic symptoms and symptomatic presentation was associated with more aggressive management. Despite the fact that current guidelines do not support surveillance brain imaging, given these findings, we believe a high-yield brain MRI surveillance strategy for the first year following CRT may increase detection of asymptomatic disease and improve patient outcomes and quality of life. Prospective study and further validation are warranted.
Footnotes
Sources of support: This work was supported by the Wake Forest Baptist Medical Center and National Center for Advancing Translational Sciences (NCATS). The National Institutes of Health funded the Wake Forest Clinical and Translational Science Institute (WF CTSI) through Grant Award Number UL1TR001. Research reported in this publication was supported by the National Center for Advancing Translational Sciences of the National Institutes of Health under Award Number KL2TR001421. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Disclosures: Dr Michael K. Farris reports financial support, administrative support, article publishing charges, and statistical analysis were provided by Wake Forest University School of Medicine. Dr Michael K. Farris reports a relationship with Wake Forest University School of Medicine that includes employment and nonfinancial support. Dr Joshua C. Farris reports a relationship with Wake Forest University School of Medicine that includes employment. Dr Hughes reports a relationship with Wake Forest University School of Medicine that includes employment. Dr Razavian reports a relationship with Wake Forest University School of Medicine that includes employment. Ms Pearce reports a relationship with Wake Forest University School of Medicine that includes employment. Dr Snavely reports a relationship with Wake Forest University School of Medicine that includes employment. Dr Chan reports a relationship with Wake Forest University School of Medicine that includes employment. Dr Steber reports a relationship with Wake Forest University School of Medicine that includes employment. Dr Leyrer reports a relationship with Wake Forest University School of Medicine that includes employment. Dr Bunch reports a relationship with Wake Forest University School of Medicine that includes employment. Dr Willey reports a relationship with Wake Forest University School of Medicine that includes employment.
Data sharing statement: Research data are stored in an institutional repository and will be shared upon request to the corresponding author.
Supplementary material associated with this article can be found in the online version at doi:10.1016/j.adro.2022.101058.
Appendix. Supplementary materials
References
- 1.Amin MB, Edge SB, Greene FL, et al. 8th ed. Springer; Chicago, IL: 2017. AJCC Cancer Staging Manual. [Google Scholar]
- 2.Barnholtz-Sloan JS, Sloan AE, Davis FG, Vigneau FD, Lai P, Sawaya RE. Incidence proportions of brain metastases in patients diagnosed (1973 to 2001) in the Metropolitan Detroit Cancer Surveillance System. J Clin Oncol. 2004;22:2865–2872. doi: 10.1200/JCO.2004.12.149. [DOI] [PubMed] [Google Scholar]
- 3.Lim JH, Um S-W. The risk factors for brain metastases in patients with non-small cell lung cancer. Ann Transl Med. 2018;6(Suppl 1):S66. doi: 10.21037/atm.2018.10.27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Carolan H, Sun AY, Bezjak A, et al. Does the incidence and outcome of brain metastases in locally advanced non-small cell lung cancer justify prophylactic cranial irradiation or early detection? Lung Cancer. 2005;49:109–115. doi: 10.1016/j.lungcan.2004.12.004. [DOI] [PubMed] [Google Scholar]
- 5.Cox JD, Scott CB, Byhardt RW, et al. Addition of chemotherapy to radiation therapy alters failure patterns by cell type within non-small cell carcinoma of lung (NSCCL): Analysis of radiation therapy oncology group (RTOG) trials. Int J Radiat Oncol Biol Phys. 1999;43:505–509. doi: 10.1016/s0360-3016(98)00429-5. [DOI] [PubMed] [Google Scholar]
- 6.Robnett TJ, Machtay M, Stevenson JP, Algazy KM, Hahn SM. Factors affecting the risk of brain metastases after definitive chemoradiation for locally advanced non-small-cell lung carcinoma. J Clin Oncol. 2001;19:1344–1349. doi: 10.1200/JCO.2001.19.5.1344. [DOI] [PubMed] [Google Scholar]
- 7.Gaspar LE, Chansky K, Albain KS, et al. Time from treatment to subsequent diagnosis of brain metastases in stage III non–small-cell lung cancer: A retrospective review by the Southwest Oncology Group. J Clin Oncol. 2005;23:2955–2961. doi: 10.1200/JCO.2005.08.026. [DOI] [PubMed] [Google Scholar]
- 8.Mitra D, Chen YH, Li R, et al. EGFR mutant locally advanced non-small cell lung cancer is at increased risk of brain metastasis. Clin Transl Radiat Oncol. 2019;18:32–38. doi: 10.1016/j.ctro.2019.06.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Raben D, Rimner A, Senan S, et al. Patterns of disease progression with durvalumab in stage III non-small cell lung cancer (PACIFIC) [abstract] Int J Rad Oncol Biol Phys. 2019;105:P683. [Google Scholar]
- 10.Ettinger DS, Wood DE, Aisner DL, Akerley W, Bauman JR, Bharat A, et al. NCCN guidelines insights: Non-small cell lung cancer, version 2.2021. J Natl Compr Canc Netw. 2021;19:254–266. doi: 10.6004/jnccn.2021.0013. [DOI] [PubMed] [Google Scholar]
- 11.De Ruysscher D, Dingemans AC, Praag J, et al. Prophylactic cranial irradiation versus observation in radically treated stage III non-small-cell lung cancer: A randomized phase III NVALT-11/DLCRG-02 study. J Clin Oncol. 2018;36:2366–2377. doi: 10.1200/JCO.2017.77.5817. [DOI] [PubMed] [Google Scholar]
- 12.Taugner J, Eze C, Käsmann L, et al. Pattern-of-failure and salvage treatment analysis after chemoradiotherapy for inoperable stage III non-small cell lung cancer. Radiat Oncol. 2020;15:148. doi: 10.1186/s13014-020-01590-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lin NU, Wefel JS, Lee EQ, et al. Challenges relating to solid tumour brain metastases in clinical trials, part 2: Neurocognitive, neurological, and quality-of-life outcomes. A report from the RANO Group. Lancet Oncol. 2013;14:e407–e416. doi: 10.1016/S1470-2045(13)70308-5. [DOI] [PubMed] [Google Scholar]
- 14.Lester SC, Taksler GB, Kuremsky JG, et al. Clinical and economic outcomes of patients with brain metastases based on symptoms: An argument for routine brain screening of those treated with upfront radiosurgery. Cancer. 2014;120:433–441. doi: 10.1002/cncr.28422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Schneider BJ, Ismaila N, Aerts J, et al. Lung cancer surveillance after definitive curative-intent therapy: ASCO guideline. J Clin Oncol. 2020;38:753–766. doi: 10.1200/JCO.19.02748. [DOI] [PubMed] [Google Scholar]
- 16.Waqar SN, Waqar SH, Trinkaus K, et al. Brain metastases at presentation in patients with non-small cell lung cancer. Am J Clin Oncol. 2018;41:36–40. doi: 10.1097/COC.0000000000000230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kuremsky JG, Urbanic JJ, Petty WJ, et al. Tumor histology predicts patterns of failure and survival in patients with brain metastases from lung cancer treated with gamma knife radiosurgery. Neurosurgery. 2013;73:641–647. doi: 10.1227/NEU.0000000000000072. discussion 647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Shin DY, Na II, Kim CH, Park S, Baek H, Yang SH. EGFR mutation and brain metastasis in pulmonary adenocarcinomas. J Thorac Oncol. 2014;9:195–199. doi: 10.1097/JTO.0000000000000069. [DOI] [PubMed] [Google Scholar]
- 19.Pope WB. Brain metastases: Neuroimaging. Handb Clin Neurol. 2018;149:89–112. doi: 10.1016/B978-0-12-811161-1.00007-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gondi V, Pugh SL, Mehta MP, et al. NRG Oncology CC003: A randomized phase II/III trial of prophylactic cranial irradiation with or without hippocampal avoidance for small cell lung cancer. J Clin Oncol. 2019;37(15 suppl):TPS8578. [Google Scholar]
- 21.Kilburn JM, Ellis TL, Lovato JF, et al. Local control and toxicity outcomes in brainstem metastases treated with single fraction radiosurgery: Is there a volume threshold for toxicity? J Neurooncol. 2014;117:167–174. doi: 10.1007/s11060-014-1373-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hughes RT, Masters AH, McTyre ER, et al. Initial SRS for patients with 5 to 15 brain metastases: Results of a multi-institutional experience. Int J Radiat Oncol Biol Phys. 2019;104:1091–1098. doi: 10.1016/j.ijrobp.2019.03.052. [DOI] [PubMed] [Google Scholar]
- 23.Haar F, Patterson RH., Jr. Surgery for metastatic intracranial neoplasm. Cancer. 1972;30:1241–1245. doi: 10.1002/1097-0142(197211)30:5<1241::aid-cncr2820300515>3.0.co;2-5. [DOI] [PubMed] [Google Scholar]
- 24.Patchell RA, Tibbs PA, Walsh JW, et al. A randomized trial of surgery in the treatment of single metastases to the brain. N Engl J Med. 1990;322:494–500. doi: 10.1056/NEJM199002223220802. [DOI] [PubMed] [Google Scholar]
- 25.Vecht CJ, Haaxma-Reiche H, Noordijk EM, et al. Treatment of single brain metastasis: Radiotherapy alone or combined with neurosurgery. Ann Neurol. 1993;33:583–590. doi: 10.1002/ana.410330605. [DOI] [PubMed] [Google Scholar]
- 26.Schödel P, Jünger ST, Wittersheim M, et al. Surgical resection of symptomatic brain metastases improves the clinical status and facilitates further treatment. Cancer Med. 2020;9:7503–7510. doi: 10.1002/cam4.3402. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.