Abstract
Background
Although androgen deprivation therapy (ADT) is associated with cardiovascular risks, the extent and temporal trends of cardiovascular burden amongst patients with prostate cancer receiving ADT are unclear.
Methods
This retrospective cohort study analyzed adults with PCa receiving ADT between 1993–2021 in Hong Kong, with follow-up until 31/9/2021 for the primary outcome of major adverse cardiovascular events (MACE; composite of cardiovascular mortality, myocardial infarction, stroke, and heart failure), and the secondary outcome of mortality. Patients were stratified into four groups by the year of ADT initiation for comparisons.
Results
Altogether, 13,537 patients were included (mean age 75.5 ± 8.5 years old; mean follow-up 4.7 ± 4.3 years). More recent recipients of ADT had more cardiovascular risk factors and used more cardiovascular or antidiabetic medications. More recent recipients of ADT had higher risk of MACE (most recent (2015–2021) vs least recent (1993–2000) group: hazard ratio 1.33 [1.11, 1.59], P = 0.002; Ptrend < 0.001), but lower risk of mortality (hazard ratio 0.76 [0.70, 0.83], P < 0.001; Ptrend < 0.001). The 5-year risk of MACE and mortality for the most recent group were 22.5% [20.9%, 24.2%] and 52.9% [51.3%, 54.6%], respectively.
Conclusions
Cardiovascular risk factors were increasingly prevalent amongst patients with prostate cancer receiving ADT, with increasing risk of MACE despite decreasing mortality.
Subject terms: Outcomes research, Prostate cancer, Epidemiology
Introduction
With an estimated 1.4 million new cases and over 375,000 deaths, prostate cancer (PCa) was the third most common cancer globally in 2020 [1]. Androgen deprivation therapy (ADT), which involves the use of pharmacological agents or surgery to suppress the levels of testosterone, is one of the key components of PCa therapy [2, 3]. Though proven efficacious for PCa treatment, research in recent decades, starting with the groundbreaking work by Keating et al. [4], has demonstrated an increasingly established link between ADT and adverse cardiovascular effects, including increased risks of cardiovascular mortality, non-fatal cardiovascular diseases, myocardial infarction, and stroke [5]. Given the rising prevalence of PCa globally and cardiovascular diseases being the leading cause of non-cancer death amongst PCa patients [6–8], ADT-related adverse cardiovascular effects have become increasingly important.
Nonetheless, there is a paucity of studies characterizing the magnitude of cardiovascular burden amongst patients with PCa receiving ADT, despite the large number of studies having compared ADT to non-ADT treatments or between specific types of ADT [5]. Furthermore, it is unclear whether the cardiovascular burden of these patients has evolved over time. It would have been reasonable to hypothesize that the significant progress made in our understanding of ADT-related adverse cardiovascular effects has significantly influenced the cardiovascular outcome of these patients. As such, this study aimed to describe the cardiovascular burden amongst patients with PCa receiving ADT and explore the temporal trends in such burden over the past decades.
Methods
This retrospective cohort study was performed in accordance with the Declaration of Helsinki and the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guideline, and was approved by the Joint Chinese University of Hong Kong—New Territories East Cluster Clinical Research Ethics Committee. The requirement for patient consent has been waived due to the use of deidentified data. All underlying data is available on reasonable request to the corresponding authors.
Source of data
Data were obtained from the Clinical Data Analysis and Reporting System (CDARS), a population-based, administrative electronic medical records database of all patients attending public healthcare institutions in Hong Kong which serve an estimated 90% of the population [9]. Diagnoses were recorded by International Classification of Diseases, Ninth revision (ICD-9) codes regardless of the time of data entry, as CDARS has not implemented ICD-10 codes to date. Mortality data were obtained from the linked Hong Kong Death Registry, a governmental database containing the death record of all Hong Kong citizens, in which the cause of death is recorded using ICD-9 or ICD-10 codes. CDARS and the Hong Kong Death Registry have been used extensively in previous studies and shown to have good coding accuracy [10–15].
Patient population
Patients aged 18 years old or above with PCa who received any ADT (medical castration or bilateral orchidectomy (BO)) between January 1, 1999 and March 31, 2021 were analysed.
Patients with a prior history of myocardial infarction (MI), stroke, or heart failure (HF) were excluded from all analyses of the primary outcome. These patients were excluded for two reasons. Firstly, patients with prior occurrence of MI, stroke, or HF were likely to have further recorded attendances to follow up on these conditions, and as all diagnoses were extracted using ICD-9 codes, it would have been difficult to reliably distinguish follow-up attendances from true, recurrent occurrences of events. Second, patients with prior occurrence of MI, stroke, or HF likely had varying severity or multiple prior occurrences of these events, both of which would have caused substantial increase in the heterogeneity of their cardiovascular risks that were difficult to account for. Therefore, excluding these patients avoided this heterogeneity and allowed a better reflection of the cardiovascular risks associated with ADT.
Data collected
The following baseline variables were collected: age, type of ADT (medical castration or bilateral orchiectomy; medical castration included usage of leuprorelin, triptorelin, goserelin, or degarelix, as other gonadotropin-releasing hormone agonists and antagonists were not available in Hong Kong during the study period), duration of castration, comorbidities (hypertension, diabetes mellitus, dyslipidaemia, chronic kidney disease, chronic liver disease, stroke, MI, ischaemic heart disease, HF, anaemia, atrial fibrillation, ventricular tachyarrhythmia, chronic obstructive pulmonary disease, and known malignancy), use of medications or prior procedures (radiotherapy, chemotherapy, radical prostatectomy, angiotensin-converting enzyme inhibitor or angiotensin receptor blocker, beta-blocker, dihydropyridine calcium channel blockers, non-dihydropyridine calcium channel blockers, metformin, sulphonylurea, dipeptidyl peptidase-4 inhibitors, glucagon-like peptide-1 receptor agonist, insulin, antiplatelet, anticoagulant, corticosteroid, percutaneous coronary intervention, and coronary artery bypass graft), the number of cardiovascular medications being used at baseline, and the number of diabetic medications being used at baseline. Patients were also recorded for ever receiving radiotherapy, chemotherapy, radical prostatectomy, and any androgen receptor signalling inhibitor (ARSI; prescriptions of first-generation ARSIs (flutamide and bicalutamide) and second-generation ARSIs (abiraterone and enzalutamide) were recorded separately; other ARSIs were not available in Hong Kong during the study period). Additionally, the number of patients with available baseline records (within 3 years prior to index date which is the date of ADT initiation) of serum total cholesterol level, high-density lipoprotein cholesterol (HDL-C) level, and haemoglobin A1c (HbA1c) level were recorded, and these variables were described for patients with available records. All comorbidities were identified using ICD-9 codes, which were listed in Supplementary Table 1.
Follow-up and outcome
All patients were followed up from the date of ADT initiation until September 31, 2021. The primary outcome was major adverse cardiovascular events (MACE), which was defined as the first occurrence of cardiovascular mortality, MI, stroke, or HF. The secondary outcome was all-cause mortality. MI, stroke, and HF were identified by ICD-9 codes as listed in Supplementary Table 1. The cause of death was identified by ICD-9 or ICD-10 codes as listed in Supplementary Table 2.
Statistical analysis
Continuous variables were expressed as mean ± standard deviation. The cohort was subdivided into four groups by the year of ADT initiation, i.e. 1993–2000, 2001–2007, 2008–2014, and 2015–2021. All baseline characteristics were described for patients in each group. Continuous variables were compared between groups using one-way analysis of variance (ANOVA), while categorical variables were compared using Chi-square test. Between-group trends were tested using the Wilcoxon-type test developed by Cuzick [16]. Due to the nature of the database, missing values could only exist for laboratory test results. The number of patients with missing values were described, and only non-missing values were included in between-group comparisons without any imputation.
Kaplan–Meier incidence curves were constructed to visualize the cumulative incidence of the outcomes for each group. As the group of patients with the most recent ADT initiation (i.e. the 2015–2021 group) could be expected to have the shortest follow-up duration, several approaches were deployed to compare the outcomes between groups. First, incidence rates of the outcomes were calculated for each group with the follow-up duration of the groups restricted to the longest follow-up duration of the 2015–2021 group. The incidence rates (IR) were then compared against that of the group with the earliest ADT initiation (i.e. the 1993–2000 group) using the Mantel–Haenszel method with calculation of the corresponding incidence rate ratios (IRR), and the trends in incidence rates between groups were tested using the log-linear trend test. These analyses on IR and IRR were also repeated without any restriction on the follow-up duration. Second, survival analysis was conducted with the aforementioned restriction on follow-up duration. As no important violation of the proportional hazards assumption was found by the Schoenfield’s residuals-based test and visual inspection of the log-log plot and Kaplan–Meier plot, univariable Cox regression analysis was used to compare the cumulative incidence of the outcomes between groups with the 1993–2000 group as reference, and with hazard ratio (HR) and 95% confidence interval (CI) as summary statistics. Third, the 5-year risk of the outcomes were estimated for each group using life tables.
Two sensitivity analyses were performed. First, the restricted mean survival time (RMST) for each group were calculated. Second, as some patients died without having MACE, non-cardiovascular mortality was a competing event for MACE. Hence, univariable competing risk regression was performed for MACE using the Fine and Gray sub-distribution model with non-cardiovascular mortality as the competing event; sub-hazard ratio (SHR) and 95% CI were used as summary statistics.
Lastly, to better understand the risk factors for the outcomes, all baseline variables except laboratory test results were entered for backward stepwise Cox regression with P ≥ 0.10 as the threshold for removal and P < 0.05 as the threshold for entry. The year of ADT initiation was also entered as a categorical variable to account for any temporal difference in the outcomes. This also accounted for the temporal difference in follow-up duration; thus, no restriction was placed on the follow-up duration in this analysis.
All P values were two-sided, with P < 0.05 considered statistically significant. All statistical analyses were performed on Stata v16.1 (StataCorp LLC, College Station, Texas, USA).
Results
In total, 13,537 patients were identified and analysed (mean age 75.5 ± 8.5 years old). Baseline characteristics of included patients were summarized in Table 1. There was a trend favouring medical castration and against BO in more recent years (85.8% with medical castration and 24.4% with BO in 2015–2021 group vs 11.1% with medical castration and 91.2% with BO in 1993–2000 group; Chi-square P < 0.001 and Ptrend < 0.001 for both). Fewer of those receiving ADT in more recent years had received radiotherapy (2.5% in 2015–2021 group vs 6.1% in 1993–2000 group, Chi-square P < 0.001, Ptrend < 0.001) or radical prostatectomy (19.4% in 2015–2021 group vs 47.3% in 1993–2000 group, Chi-square P < 0.001, Ptrend < 0.001). Meanwhile, more of those who received ADT in more recent years have ever received first-generation (44.4% in 2015–2021 group vs 8.4% in 1993–2000 group, Chi-square P < 0.001, Ptrend < 0.001) or second-generation (19.8% in 2015–2021 group vs 0.3% in 1993–2000 group, Chi-square P < 0.001, Ptrend < 0.001) ARSI, as well as chemotherapy (16.1% in 2015–2021 group vs 0.8% in 1993–2000 group, Chi-square P < 0.001, Ptrend < 0.001). Amongst those who received medical castration, the duration of medical castration was shorter in the most recent group (596 ± 564 days in 2015–2021 group vs 974 ± 1409 days in 1993–2000 group, ANOVA P < 0.001, Ptrend < 0.001), although this may have been due to the shorter follow-up duration inherent to this group.
Table 1.
Characteristics for all patients and stratified by the year of androgen deprivation therapy initiation.
| All patients | 1993–2000 | 2001–2007 | 2008–2014 | 2015–2021 | P value between groups | P for trend | ||
|---|---|---|---|---|---|---|---|---|
| Number of patients, N | 13,537 | 1134 | 3017 | 4641 | 4745 | NA | NA | |
| Follow-up duration, years | 4.7 ± 4.3 | 5.7 ± 6.6 | 6.5 ± 5.6 | 5.4 ± 3.7 | 2.6 ± 1.7 | <0.001 | <0.001 | |
| Age, years | 75.5 ± 8.5 | 74.9 ± 7.8 | 75.2 ± 7.8 | 75.7 ± 8.5 | 75.8 ± 9.0 | <0.001 | <0.001 | |
| Medical castration, N (%) | 8178 (60.4) | 126 (11.1) | 1213 (40.2) | 2768 (59.6) | 4071 (85.8) | <0.001 | <0.001 | |
| Bilateral orchidectomy, N (%) | 6593 (48.7) | 1034 (91.2) | 2075 (68.8) | 2326 (50.1) | 1158 (24.4) | <0.001 | <0.001 | |
| Duration of ADT, daysa | 891 ± 905 | 842 ± 1160 | 1066 ± 1287 | 1116 ± 1026 | 687 ± 558 | <0.001 | <0.001 | |
| Hypertension, N (%) | 3624 (26.8) | 123 (10.9) | 549 (18.2) | 1293 (27.9) | 1659 (35.0) | <0.001 | <0.001 | |
| Diabetes mellitus, N (%) | 2886 (21.3) | 87 (7.7) | 484 (16.0) | 1014 (21.9) | 1301 (27.4) | <0.001 | <0.001 | |
| Dyslipidaemia, N (%) | 1270 (9.4) | 9 (0.8) | 97 (3.2) | 364 (7.8) | 800 (16.9) | <0.001 | <0.001 | |
| Chronic kidney disease, N (%) | 452 (3.3) | 18 (1.6) | 70 (2.3) | 177 (3.8) | 187 (3.9) | <0.001 | <0.001 | |
| Chronic liver disease, N (%) | 146 (1.1) | 5 (0.4) | 14 (0.5) | 56 (1.2) | 71 (1.5) | <0.001 | <0.001 | |
| Stroke, N (%) | 1216 (9.0) | 42 (3.7) | 221 (7.3) | 424 (9.1) | 529 (11.2) | <0.001 | <0.001 | |
| Myocardial infarction, N (%) | 427 (3.2) | 9 (0.8) | 73 (2.4) | 156 (3.4) | 189 (4.0) | <0.001 | <0.001 | |
| Ischaemic heart disease, N (%) | 1407 (10.4) | 76 (6.7) | 260 (8.6) | 507 (10.9) | 564 (11.9) | <0.001 | <0.001 | |
| Heart failure, N (%) | 695 (5.1) | 35 (3.1) | 116 (3.8) | 278 (6.0) | 266 (5.6) | <0.001 | <0.001 | |
| Anaemia, N (%) | 966 (7.1) | 34 (3.0) | 112 (3.7) | 418 (9.0) | 402 (8.5) | <0.001 | <0.001 | |
| Atrial fibrillation, N (%) | 610 (4.5) | 24 (2.1) | 90 (3.0) | 212 (4.6) | 284 (6.0) | <0.001 | <0.001 | |
| COPD, N (%) | 804 (5.9) | 53 (4.7) | 193 (6.4) | 316 (6.8) | 242 (5.1) | 0.001 | 0.341 | |
| Known malignancy, N (%) | 1003 (13.3) | 130 (11.5) | 403 (13.4) | 701 (15.1) | 569 (12.0) | <0.001 | 0.558 | |
| Prior PCI, N (%) | 432 (3.2) | 6 (0.5) | 47 (1.6) | 157 (3.4) | 222 (4.7) | <0.001 | <0.001 | |
| Prior CABG, N (%) | 55 (0.4) | 1 (0.1) | 10 (0.3) | 17 (0.4) | 27 (0.6) | 0.088 | 0.015 | |
| Prior radiotherapy, N (%) | 493 (3.6) | 69 (6.1) | 105 (3.5) | 199 (4.3) | 120 (2.5) | <0.001 | <0.001 | |
| Prior RP, N (%) | 3735 (27.6) | 536 (47.3) | 1071 (35.5) | 1206 (26.0) | 922 (19.4) | <0.001 | <0.001 | |
| Prior chemotherapy, N (%) | 61 (0.5) | 0 (0) | 3 (0.1) | 11 (0.2) | 47 (1.0) | <0.001 | <0.001 | |
| Ever received radiotherapy, N (%) | 3114 (23.0) | 328 (28.9) | 799 (26.5) | 1294 (27.9) | 693 (14.6) | <0.001 | <0.001 | |
| Ever received RP, N (%) | 4601 (34.0) | 603 (53.2) | 1286 (42.6) | 1456 (31.4) | 1256 (26.5) | <0.001 | <0.001 | |
| Ever received chemotherapy, N (%) | 1311 (9.7) | 9 (0.8) | 121 (4.0) | 416 (9.0) | 765 (16.1) | <0.001 | <0.001 | |
| Ever received first-generation ARSI, N (%) | 4239 (31.5) | 92 (8.4) | 663 (22.1) | 1377 (29.7) | 2107 (44.4) | <0.001 | <0.001 | |
| Ever received second-generation ARSI, N (%) | 1582 (11.7) | 3 (0.3) | 77 (2.6) | 563 (12.1) | 939 (19.8) | <0.001 | <0.001 | |
| Ever received chemotherapy or ARSI, N (%) | 5116 (37.8) | 101 (8.9) | 724 (24.0) | 1638 (35.3) | 2653 (55.9) | <0.001 | <0.001 | |
| Number of cardiovascular medications | 1.5 ± 1.7 | 0.2 ± 0.6 | 1.0 ± 1.3 | 1.7 ± 1.6 | 2.0 ± 1.8 | <0.001 | <0.001 | |
| Number of antidiabetic medications | 0.30 ± 0.73 | 0.05 ± 0.28 | 0.21 ± 0.57 | 0.32 ± 0.74 | 0.40 ± 0.85 | <0.001 | <0.001 | |
| ACEI/ARB users, N (%) | 3383 (25.0) | 23 (2.0) | 481 (15.9) | 1303 (28.1) | 1576 (33.2) | <0.001 | <0.001 | |
| Beta-blocker users, N (%) | 4130 (30.5) | 42 (3.7) | 696 (23.1) | 1638 (35.3) | 1754 (37.0) | <0.001 | <0.001 | |
| Dihydropyridine CCB users, N (%) | 5396 (39.9) | 58 (5.1) | 886 (29.4) | 2058 (44.3) | 2394 (50.5) | <0.001 | <0.001 | |
| Non-dihydropyridine CCB users, N (%) | 575 (4.3) | 11 (1.0) | 134 (4.4) | 226 (4.9) | 201 (4.3) | <0.001 | 0.002 | |
| Metformin users, N (%) | 1480 (10.9) | 14 (1.2) | 194 (6.4) | 584 (12.6) | 688 (14.5) | <0.001 | <0.001 | |
| Sulphonylurea users, N (%) | 1744 (12.9) | 29 (2.6) | 347 (11.5) | 661 (14.2) | 707 (14.9) | <0.001 | <0.001 | |
| DPP4 inhibitor users, N (%) | 150 (1.1) | 0 (0) | 0 (0) | 29 (0.6) | 121 (2.6) | <0.001 | <0.001 | |
| GLP1 receptor agonist users, N (%) | 2 (0.0) | 0 (0) | 0 (0) | 1 (0.0) | 1 (0.0) | 0.829 | 0.423 | |
| Insulin users, N (%) | 722 (5.3) | 10 (0.9) | 90 (3.0) | 222 (4.8) | 400 (8.4) | <0.001 | <0.001 | |
| Antiplatelet users, N (%) | 2962 (21.9) | 43 (3.8) | 531 (17.6) | 1112 (24.0) | 1276 (27.9) | <0.001 | <0.001 | |
| Anticoagulant users, N (%) | 458 (3.4) | 3 (0.3) | 58 (1.9) | 148 (3.2) | 249 (5.3) | <0.001 | <0.001 | |
| Corticosteroid users, N (%) | 2342 (17.3) | 29 (2.6) | 510 (16.9) | 933 (20.1) | 870 (18.3) | <0.001 | <0.001 | |
| With available total cholesterol, HDL-C, and HbA1c levels, N (%) | 3940 (29.1) | 5 (0.4) | 265 (8.8) | 1220 (26.3) | 2450 (51.6) | <0.001 | <0.001 | |
| Total cholesterol | Available, N (%) | 6727 (49.7) | 28 (2.5) | 806 (26.7) | 2458 (53.0) | 3435 (72.4) | <0.001 | <0.001 |
| Level, mmol/L | 4.41 ± 1.01 | 5.25 ± 2.20 | 4.79 ± 0.95 | 4.56 ± 1.01 | 4.21 ± 0.96 | <0.001 | <0.001 | |
| HDL-C | Available, N (%) | 6478 (47.9) | 15 (1.3) | 626 (20.8) | 2415 (52.0) | 3422 (72.1) | <0.001 | <0.001 |
| Level, mmol/L | 1.27 ± 0.37 | 1.17 ± 0.31 | 1.28 ± 0.37 | 1.27 ± 0.37 | 1.28 ± 0.37 | 0.335 | 0.299 | |
| HbA1c | Available, N (%) | 4295 (31.7) | 22 (1.9) | 403 (13.4) | 1320 (28.4) | 2550 (53.7) | <0.001 | <0.001 |
| Level, % | 6.49 ± 1.16 | 7.85 ± 1.76 | 6.94 ± 1.46 | 6.64 ± 1.18 | 6.33 ± 1.06 | <0.001 | <0.001 | |
ACEI angiotensin-converting enzyme inhibitor, ARB angiotensin receptor blocker, ARSI androgen receptor signaling inhibitor, CABG coronary artery bypass graft, CCB calcium channel blocker, COPD chronic obstructive pulmonary disease, DPP4 dipeptidyl peptidase-4, GLP1 glucagon-like peptide-1, HbA1c haemoglobin A1c, HDL-C high-density lipoprotein cholesterol, NA not applicable, RP radical prostatectomy.
aOnly including patients who received medical castration
A total of 2059 (15.2%) had a prior diagnosis of MI, stroke, or HF, and were thus excluded from all analyses of MACE, resulting in a sample size of 11,478 patients for the MACE analyses. Over a mean follow-up duration of 4.7 ± 4.3 years, 2727 patients (23.8%) had MACE, and 9124 (67.4) died. Kaplan–Meier curves showing the cumulative incidence of MACE and all-cause mortality without any restriction on follow-up durations were shown in Supplementary Figs. 1 and 2, respectively. As expected, patients with the most recent ADT initiation had the shortest follow-up duration, with the longest observed follow-up duration being 6.7 years.
Cardiovascular risk factors and medications
Overall, patients who were initiated on ADT more recently had more cardiovascular risk factors (Table 1), with higher rates of prior hypertension, diabetes mellitus, stroke, myocardial infarction, ischaemic heart, heart failure, chronic kidney disease, atrial fibrillation, and dyslipidaemia. These patients were also slightly older. Correspondingly, more of those who had ADT initiated more recently were using cardiovascular or antidiabetic medications at baseline. Also, more of those who had ADT initiated more recently had received percutaneous coronary intervention.
Additionally, more of those who had ADT initiated recently had been checked for levels of total cholesterol, HDL-C, and HbA1c prior to initiating ADT. This was accompanied by lower total cholesterol and HbA1c amongst those with available test results whose ADT was initiated recently, but without any significant differences in HDL-C levels. Overall, despite an increasing proportion of patients having all three laboratory markers profiled prior to ADT initiation, only 51.6% of the 2015–2021 group had all three markers profiled (vs 0.4% in 1993–2000 group, Chi-square P < 0.001, Ptrend < 0.001).
Major adverse cardiovascular event
With follow-up durations restricted to the longest observed follow-up duration of the 2015–2021 group (6.7 years), patients initiated on ADT more recently had higher IR of MACE (IR 5.7 [5.3, 6.2] events per 100 person-year in the 2015–2021 group vs 4.4 [95% CI 3.7, 5.1] events per 100 person-year in the 1993–2000 group; IRR 1.32 [1.10, 1.58], P = 0.003 for the 2015–2021 group compared to the 1993–2000 group; log-linear trend test P < 0.001; Table 2). Similar observations were made with IR and IRR calculated without any restriction on follow-up durations (Supplementary Table 3).
Table 2.
Incidence rates of both outcomes with the follow-up duration restricted to the longest observed follow-up duration of the 2015–2021 group (6.7 years), stratified by the year of androgen deprivation therapy initiation.
| Year of androgen deprivation therapy initiation | Major adverse cardiovascular events | All-cause mortality | ||
|---|---|---|---|---|
| Incidence ratea | Incidence rate ratio | Incidence ratea | Incidence rate ratio | |
| 1993–2000 | 4.4 [3.7, 5.1] | 1 (reference) | 20.3 [19.0, 21.8] | 1 (reference) |
| 2001–2007 | 4.7 [4.3, 5.1] | 1.07 [0.89, 1.28], P = 0.479 | 14.5 [13.9, 15.2] | 0.71 [0.66, 0.78], P < 0.001 |
| 2008–2014 | 5.4 [5.0, 5.7] | 1.23 [1.04, 1.47], P = 0.019 | 13.9 [13.4, 14.5] | 0.69 [0.63, 0.74], P < 0.001 |
| 2015–2021 | 5.7 [5.3, 6.2] | 1.32 [1.10, 1.58], P = 0.003 | 15.9 [15.2, 16.6] | 0.78 [0.72, 0.85], P < 0.001 |
aPer 100 person-year.
Incidence rate ratios displayed were referenced against the 1993–2000 group.
A similar trend was observed in the Cox regression analysis (Ptrend < 0.001), where the patients with ADT initiated in years 2015–2021 had an estimated 33% higher risk of MACE compared to those with ADT initiation in years 1993–2000 (HR 1.33 [1.11, 1.59], P = 0.002; Fig. 1a and Table 3). Correspondingly, the estimated 5-year risk of MACE was the highest in the 2015–2021 group (22.5% [20.9%, 24.2%]) and the 2008–2014 group (23.0% [21.6%, 24.5%]), followed by the 2001–2007 group (19.4% [17.7%, 21.1%], and lastly, with the lowest 5-year risk, the 1993–2000 group (17.0% [14.4%, 19.9%]).
Fig. 1. Kaplan–Meier incidence curves.
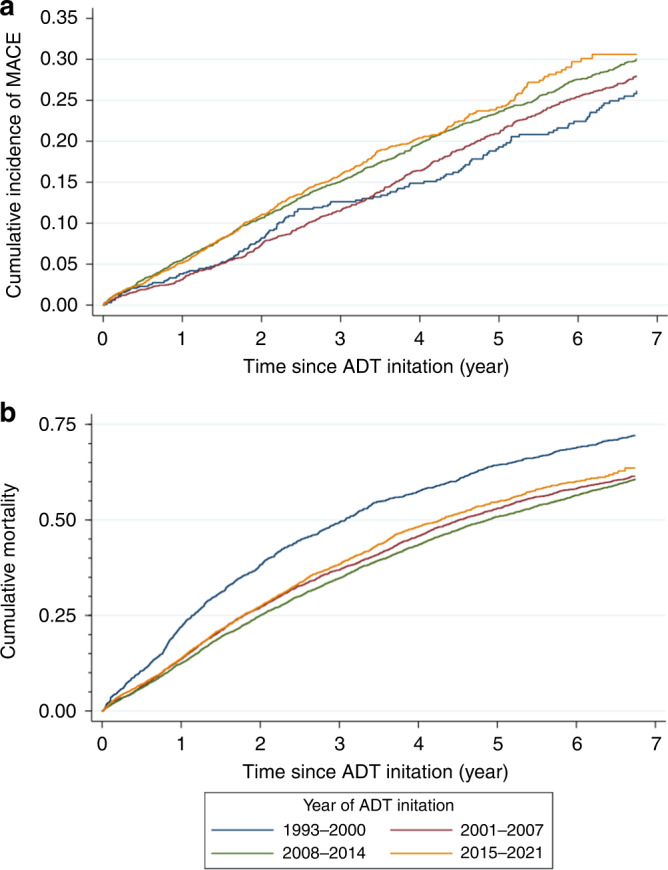
a shows the cumulative incidence of major adverse cardiovascular event (MACE), while b shows that of all-cause mortality. The follow-up durations in both curves were restircted to the longest observed follow-up duration of the 2015–2021 group (6.7 years). ADT androgen deprivation therapy.
Table 3.
Hazard ratios and accompanying 95% confidence intervals from Cox regression analysis with the follow-up duration restricted to the longest observed follow-up duration of the 2015–2021 group (6.7 years).
| Year of androgen deprivation therapy initiation | Major adverse cardiovascular event | All-cause mortality |
|---|---|---|
| 1993–2000 | 1 (reference) | 1 (reference) |
| 2001–2007 | 1.07 [0.89, 1.28], P = 0.493 | 0.72 [0.66, 0.78], P < 0.001 |
| 2008–2014 | 1.23 [1.03, 1.46], P = 0.021 | 0.69 [0.64, 0.74], P < 0.001 |
| 2015–2021 | 1.33 [1.11, 1.59], P = 0.002 | 0.76 [0.70, 0.83], P < 0.001 |
ADT androgen deprivation therapy.
Hazard ratios displayed were referenced against the 1993–2000 group.
Sensitivity analyses with RMST demonstrated similar results (RMST 5.59 [5.51, 5.68] years for the 2015–2021 group vs 5.86 [5.73, 6.00] years for the 1993–2000 group; Supplementary Table 4). Competing risk regression also found similar results, with the 2015–2021 group having an estimated 43% higher cumulative incidence of MACE than the 1993–2000 group (SHR 1.43 [1.19, 1.71], P < 0.001; Supplementary Table 5).
Backward stepwise Cox regression showed that increased age, diabetes mellitus, hypertension, anaemia, known malignancy, anticoagulant use, and using more cardiovascular medications were independently associated with higher risk of MACE, while statin use, and metformin use were independently associated with lower risk of MACE (Supplementary Table 6).
All-cause mortality
With follow-up durations restricted to the longest observed follow-up duration of the 2015–2021 group (6.7 years), patients initiated on ADT more recently had lower IR of all-cause mortality (IR 15.9 [15.2, 16.6] deaths per 100 person-year in the 2015–2021 group vs 20.3 [19.0, 21.8] deaths per 100 person-year in the 1993–2000 group; IRR 0.78 [0.72, 0.85], P < 0.001; for the 2015–2021 group compared to the 1993–2000 group; log-linear trend test P = 0.003; Table 2). However, analysis without any restriction on follow-up duration showed no significant difference between the 2015–2021 group and 1993–2000 group in the IR of all-cause mortality (IRR 0.98 [0.91, 1.06], P = 0.660; Supplementary Table 3), which was driven by a markedly lower IR in the 1993–2000 group (IR 16.2 [15.2, 17.2] deaths per 100 person-year when analyzed without any restriction on follow-up duration vs 20.3 [19.0, 21.8] deaths per 100 person-year when analyzed with restricted follow-up duration). Such difference likely resulted from the mortality rate being higher in the initial years after ADT initiation before levelling off in later years, as apparent from the Kaplan–Meier curve without any restriction on follow-up duration (Supplementary Fig. 2).
Cox regression with restricted follow-up duration showed that patients with ADT initiated more recently had lower risk of mortality (Ptrend < 0.001), with the 2015–2021 group having an estimated 24% lower risk of all-cause mortality than those in the 1993–2000 group (HR 0.76 [0.70, 0.83], P < 0.001; Fig. 1b and Table 3). The estimated 5-year risk of all-cause mortality was 64.5% [61.7%, 67.4%] for the 1993–2000 group, 53.0% [51.2%, 54.8%] for the 2001–2007 group, 50.8% [49.4%, 52.3%] for the 2008–2014 group, and 52.9% [51.3%, 54.6%] for the 2015–2021 group.
Sensitivity analysis with RMST showed similar results as Cox regression (RMST 3.55 [3.40, 3.70] years for the 1993–2000 group vs 4.14 [4.06, 4.22] years for the 2015–2021 group; Supplementary Table 4).
Backward stepwise Cox regression showed that increased age, diabetes mellitus, anaemia, atrial fibrillation, chronic liver disease, chronic obstructive pulmonary disease, known malignancy, prior chemotherapy, prior radiotherapy, insulin use, sulphonylurea use, and using more cardiovascular medications were independently associated with higher risk of all-cause mortality, while medical castration, statin use, prior radical prostatectomy, angiotensin-converting enzyme inhibitor/angiotensin receptor blocker use, and metformin use were independently associated with lower risk of all-cause mortality (Supplementary Table 7). ADT initiation during or after 2001 was identified to be independently associated with lower risk of all-cause mortality as well. There was a trend of chronic kidney disease being associated with numerically higher risk of all-cause mortality which approached statistical significance (P = 0.056).
Discussion
This population-based study demonstrated that cardiovascular risk factors were increasingly prevalent amongst Asian patients with PCa receiving ADT. This was accompanied by an increasing risk of MACE, despite reducing risk of mortality and an increasing proportion of patients with laboratory profiling relevant to cardiovascular risks.
To the best of our knowledge, this was one of the first studies that systematically quantified the temporal trends of cardiovascular burden in patients with PCa receiving ADT. We observed an increase in the risk of MACE but a decrease in the risk of mortality over time. The latter has been observed in other studies and have been postulated to be due to better treatment of PCa [7]; other factors possibly at play may include changes in lifestyle and access to healthcare over the years. Meanwhile, the former occurred despite better contemporary understanding and recognition of the adverse cardiovascular effects of ADT [5], hence more patients being tested or monitored for cardiovascular risks. While it is immediately apparent that the increasing prevalence of cardiovascular risk factors over time may have contributed significantly to this observation, the factors driving the increase in MACE incidence were likely multifactorial and intertwined. For instance, the increasing use of ARSI and chemotherapy may have contributed to such increase in the risk of MACE, as both classes of agents have been shown to carry significant cardiovascular risks [17], with the newer generation of ARSI having been shown specifically to carry significantly higher cardiovascular risks than conventional ADT [18, 19]. Nonetheless, disentangling these factors, which may further include but are not limited to the patterns of cardiometabolic screening, usage and duration of specific types of ADT, and the stage of disease when PCa was detected or when ADT was initiated, was outside the scope of the current study.
The increasing prevalence of cardiovascular risk may have been a direct result of the well-characterized, general increase in the prevalence of cardiovascular diseases [6]. Systemic factors, such as patient selection for ADT, and patients’ knowledge and perception of ADT, may have played a role also. Indeed, we showed that the preference for specific modalities of ADT changed over time. This change maybe partly related to the change in local medical reimbursement system, as well as an overwhelming preference for medical castration over BO in more recent years which was consistent with survey studies showing an estimated two-third of clinicians not considering or offering BO to eligible patients with PCa [20, 21]. It was possible that similar changes in the selection of patients for ADT have influenced the cardiovascular outcomes. This was especially relevant since the use of ADT is heavily dependent on patient preferences even within guidelines [3, 22], with studies having shown significant variations in practice [23, 24]. It may thus be reasonable to speculate that changes in patient preferences outside the scope of this study may have contributed to the differences in cardiovascular burden and outcomes, either directly or by influencing patient selection for ADT.
Previous studies have shown that cardiovascular risk factors are prevalent amongst patients with PCa. In a large, prospective Canadian cohort of 2492 patients with PCa, 22% had known cardiovascular diseases [25]. Similarly, in another smaller cohort of patients with PCa undergoing ADT, a quarter had established cardiovascular diseases [26]. We observed similar results particularly in the most contemporary subgroup of patients, with 35.0% having hypertension and 27.4% having diabetes mellitus at baseline, and over 11% having had stroke or MI. In addition, we observed that only half of the patients in the most contemporary group had had total cholesterol, HDL-C, and HbA1c profiled prior to ADT initiation, which was despite substantial improvement in such proportions over time. This was echoed by a recent study by Sun and colleagues, who found that only 68.1% of an American cohort of veterans with PCa received comprehensive cardiovascular risk factor assessment [27].
Direct clinical relevance and further directions
Given the increasing risk of MACE observed in the current study, this study serves as a timely reminder for clinicians to be vigilant in screening and managing cardiovascular burden amongst patients with PCa undergoing ADT. The increasing risk of MACE in spite of increasing metabolic screening prior to initiating ADT showed that it is insufficient to only screen these patients, and that more efforts are required to adequately manage and reduce their cardiometabolic risk, including referral of patients with multiple cardiovascular risk factors to cardiology or cardio-oncology services before initiating ADT. Such multidisciplinary approach has been recommended by the 2022 European Society of Cardiology cardio-oncology guideline [28]. This was not only relevant to urologists and oncologists, but also primary care physicians and cardiologists who may take care of patients with PCa receiving ADT. Furthermore, the observed 5-year risks of MACE and mortality reported in this study should allow clinicians to better inform patients of the risks associated with ADT, thereby facilitating shared decision-making regarding therapeutic options. It is important for clinicians to comprehensively inform patients and involve them actively in such decision-making, as greater involvements have been shown to be associated with lower risks of decision regret and higher health-related quality of life [29]. Additionally, the independent risk factors for MACE and all-cause mortality hereby identified may improve identification of patients who are possibly at higher risk of these events. Given the relatively old mean age and long duration of ADT in this study, and as the duration of ADT may be proportionally associated with the risk of adverse cardiovascular outcomes [30], the current results are the most relevant to elderly patients with PCa who will be receiving long-term ADT, especially medical castration which was the predominant modality of ADT in the most contemporary group of patients in the current study. Although the results may not be as relevant to young, otherwise healthy patients who are to be initiated on short-term ADT, the findings remain a valid reminder for clinicians to be vigilant of cardiovascular risks in patients with PCa undergoing ADT in general.
With this study demonstrating the significant and increasing cardiovascular burden amongst patients with PCa undergoing ADT, there are several gaps in evidence that are more relevant than ever. First, evidence pertinent to the optimal screening strategy for cardiovascular diseases in these patients is scarce. While previous studies have used the Framingham risk score as a surrogate of cardiovascular risk [25–27], its accuracy and validity for this specific patient group has not been adequately explored. This was echoed by the 2022 European Society of Cardiology cardio-oncology guideline [28] which pointed out the lack of cardiovascular risk scores for patients receiving ADT. More macroscopically, it is unclear, and therefore remains at clinicians’ discretion, which patients are in particular need for cardiovascular screening and monitoring. To this end, prognostic studies, possibly with exploration of novel risk scores, are sorely needed to allow better stratification of high-risk patients; our reported independent risk factors for MACE and all-cause mortality may be a starting point which such studies may reference.
Second, to the best of our knowledge, there has not been any investigation of strategies to mitigate the adverse cardiovascular effects of ADT. Whilst Bhatia and colleagues have proposed a management algorithm for ADT-related adverse cardiovascular effects, the algorithm was based on previous paradigm established for patients with breast cancer, and direct evidence supporting the algorithm or any specific management approach remains lacking [31, 32]. This was in stark contrast to many other classes of antineoplastic medications that causes adverse cardiovascular effects, for which evidence-based management algorithms have been established [33, 34]. Future studies should therefore investigate approaches and therapeutics to mitigate the adverse cardiovascular effects brought by ADT. Screening, risk stratification, and management of cardiovascular risk factors have all been specified as key areas of focus for future research by an American Heart Association scientific statement as well [35].
Last but not least, further studies are required to elucidate the factors driving the observed worsening of cardiovascular outcome in patients with PCa undergoing ADT, which are likely complex, intertwined, and multifactorial in nature. A better understanding of these factors is necessary for stifling further worsening of cardiovascular outcomes in these patients.
Generalizability, strengths, and limitations
This study included a large cohort of patients from a population-based database, meaning that the findings are representative of real-life practice locally, and likely generalizable to other developed Asian cities. The consistent findings from multiple, different statistical approaches also reinforced the validity of our findings. However, Hong Kong operates a heavily subsidized public healthcare system, from which an estimated 90% of all Hong Kong citizens receive healthcare. Given that healthcare financing structures have significant impact on access to care and the choice of therapeutics [36, 37], the findings of this study may not be directly generalizable to countries with different healthcare financing structures, such as those that are predominantly privatized. Sociodemographic and cultural differences in the population may also affect generalizability of our findings to other countries and regions.
This study had a few limitations. First, the retrospective, observational nature predisposed to residual and unmeasured confounders which may influence findings. Whilst we acknowledge that there were likely unmeasured factors that may have driven our findings, it was not within the scope of this study to disentangle these underlying drivers – we invite the readers and colleagues to further explore this topic indeed. Second, owing to the nature of the database used, cancer staging and some cardiovascular risk factors that are prognostic in cardiovascular diseases, such as blood pressure and smoking status, were not available, which may have been important confounders, and which limited the interpretation of our findings. Specifically, there have been reports showing earlier diagnosis of PCa in recent years [38], which may have contributed to differences in the incidence and risk of both all-cause mortality and MACE – this remains to be investigated in the future. Third, as the data was retrieved from a deidentified, administrative database, it could not be individually adjudicated. Nonetheless, data entry was performed by treating clinicians without the involvement of any of the authors, and none of the authors had the right or authority to alter the data. Furthermore, previous studies have demonstrated that data recorded in the database (CDARS) had good accuracy, particularly for cardiovascular outcomes [39, 40].
Conclusions
Over the past 29 years, cardiovascular risk factors have become increasingly prevalent amongst patients with PCa receiving ADT in Hong Kong. This was accompanied by an increasing incidence of MACE but a decreasing incidence of all-cause mortality. Factors underlying such observations remain to be elucidated.
Supplementary information
Author contributions
JSKC designed the study, curated the data, performed and interpreted all statistical analyses, produced all graphics, and drafted and critically revised the manuscript. DIS interpreted the statistical analyses, and drafted and critically revised the manuscript. YHAL, JMHH, and KL collected and curated data, and revised the manuscript critically. ECD and KN interpreted the statistical analyses, and revised the manuscript critically. GT and CFN supervised the study, interpreted the statistical analyses, and revised the manuscript critically. All authors approved this manuscript for submission.
Funding
No funding was obtained for this study.
Data availability
All underlying data is available on reasonable request to the corresponding authors.
Competing interests
ECD is funded in part through the Cancer Center Support Grant from the National Cancer Institute (P30 CA008748). The remaining authors declare no competing interests.
Ethics approval and consent to participate
This retrospective cohort study was performed in accordance with the Declaration of Helsinki, and has been approved by the Joint Chinese University of Hong Kong– New Territories East Cluster Clinical Research Ethics Committee. The requirement for patient consent has been waived due to the use of deidentified data.
Consent for publication
Not applicable.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Gary Tse, Email: gary.tse@kmms.ac.uk.
Chi Fai Ng, Email: ngcf@surgery.cuhk.edu.hk.
Supplementary information
The online version contains supplementary material available at 10.1038/s41416-023-02271-5.
References
- 1.Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71:209–49. doi: 10.3322/caac.21660. [DOI] [PubMed] [Google Scholar]
- 2.Mohler JL, Antonarakis ES, Armstrong AJ, D’Amico AV, Davis BJ, Dorff T, et al. Prostate cancer, version 2.2019, NCCN clinical practice guidelines in oncology. J Natl Compr Canc Netw. 2019;17:479–505. doi: 10.6004/jnccn.2019.0023. [DOI] [PubMed] [Google Scholar]
- 3.Mottet N, van den Bergh RCN, Briers E, Van den Broeck T, Cumberbatch MG, De Santis M, et al. EAU-EANM-ESTRO-ESUR-SIOG guidelines on prostate cancer-2020 update. Part 1: screening, diagnosis, and local treatment with curative intent. Eur Urol. 2021;79:243–62. doi: 10.1016/j.eururo.2020.09.042. [DOI] [PubMed] [Google Scholar]
- 4.Keating NL, O’Malley AJ, Smith MR. Diabetes and cardiovascular disease during androgen deprivation therapy for prostate cancer. J Clin Oncol. 2006;24:4448–56. doi: 10.1200/JCO.2006.06.2497. [DOI] [PubMed] [Google Scholar]
- 5.Hu JR, Duncan MS, Morgans AK, Brown JD, Meijers WC, Freiberg MS, et al. Cardiovascular effects of androgen deprivation therapy in prostate cancer. Arterioscler Thromb Vasc Biol. 2020;40:e55. doi: 10.1161/ATVBAHA.119.313046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Roth GA, Mensah GA, Johnson CO, Addolorato G, Ammirati E, Baddour LM, et al. Global burden of cardiovascular diseases and risk factors, 1990–2019: update from the GBD 2019 study. J Am Coll Cardiol. 2020;76:2982–3021. doi: 10.1016/j.jacc.2020.11.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Culp MBB, Soerjomataram I, Efstathiou JA, Bray F, Jemal A. Recent global patterns in prostate cancer incidence and mortality rates. Eur Urol. 2020;77:38–52. doi: 10.1016/j.eururo.2019.08.005. [DOI] [PubMed] [Google Scholar]
- 8.Guo Y, Dong X, Mao S, Yang F, Wang R, Ma W, et al. Causes of death after prostate cancer diagnosis: a population-based study. Oxid Med Cell Longev. 2022;2022:8145173. doi: 10.1155/2022/8145173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kong X, Yang Y, Gao J, Guan J, Liu Y, Wang R, et al. Overview of the health care system in Hong Kong and its referential significance to mainland China. J Chinese Med Assoc. 2015;78:569–73. doi: 10.1016/j.jcma.2015.02.006. [DOI] [PubMed] [Google Scholar]
- 10.Chan JSK, Zhou J, Lee S, Li A, Tan M, Leung KSK, et al. Fragmented QRS is independently predictive of long-term adverse clinical outcomes in asian patients hospitalized for heart failure: a retrospective cohort study. Front Cardiovasc Med. 2021;0:1634. doi: 10.3389/fcvm.2021.738417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Chan JSK, Lakhani I, Lee TTL, Chou OHI, Lee YHA, Cheung YM, et al. Cardiovascular outcomes and hospitalizations in Asian patients receiving immune checkpoint inhibitors: a population-based study. Curr Probl Cardiol. 2022;48:101380. doi: 10.1016/j.cpcardiol.2022.101380. [DOI] [PubMed] [Google Scholar]
- 12.Lee YHA, Zhou J, Hui JMH, Liu X, Lee TTL, Hui K, et al. Risk of new-onset prostate cancer for metformin versus sulfonylurea use in type 2 diabetes mellitus: a propensity score-matched study. J Natl Compr Canc Netw. 2022;20:674–82. doi: 10.6004/jnccn.2022.7010. [DOI] [PubMed] [Google Scholar]
- 13.Zhou J, Li H, Chang C, Wu WKK, Wang X, Liu T, et al. The association between blood pressure variability and hip or vertebral fracture risk: a population-based study. Bone. 2021;150:116015. doi: 10.1016/j.bone.2021.116015. [DOI] [PubMed] [Google Scholar]
- 14.Wai AKC, Wong CKH, Wong JYH, Xiong X, Chu OCK, Wong MS, et al. Changes in emergency department visits, diagnostic groups, and 28-day mortality associated with the COVID-19 pandemic: a territory-wide, retrospective, cohort study. Ann Emerg Med. 2022;79:148–57. doi: 10.1016/j.annemergmed.2021.09.424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wong JYH, Luk LYF, Yip TF, Lee TTL, Wai AKC, Ho JWK. Incidence of emergency department visits for sexual abuse among youth in hong kong before and during the COVID-19 pandemic. JAMA Netw open. 2022;5:e2236278. doi: 10.1001/jamanetworkopen.2022.36278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cuzick J. A Wilcoxon-type test for trend. Stat Med. 1985;4:87–90. doi: 10.1002/sim.4780040112. [DOI] [PubMed] [Google Scholar]
- 17.Wilk M, Waśko-Grabowska A, Szmit S. Cardiovascular complications of prostate cancer treatment. Front Pharmacol. 2020;11:555475. doi: 10.3389/fphar.2020.555475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Iacovelli R, Ciccarese C, Bria E, Romano M, Fantinel E, Bimbatti D, et al. The cardiovascular toxicity of abiraterone and enzalutamide in prostate cancer. Clin Genitourin Cancer. 2018;16:e645–53. doi: 10.1016/j.clgc.2017.12.007. [DOI] [PubMed] [Google Scholar]
- 19.Di Lorenzo G, Autorino R. Androgen receptor signaling inhibitors in nonmetastatic castration-resistant prostate cancer and risk of cardiovascular toxicity: all that glitters isn’t gold. Eur Urol. 2020;78:647–9. doi: 10.1016/j.eururo.2020.07.036. [DOI] [PubMed] [Google Scholar]
- 20.Schubbe ME, Gellhaus PT, Tobert CM, Mott SL, Garje R, Erickson BA. Knowledge and attitudes regarding surgical castration in men receiving androgen deprivation therapy for metastatic prostate cancer and their relationship to health-related quality of life. Urology. 2021;155:179–85. doi: 10.1016/j.urology.2021.04.027. [DOI] [PubMed] [Google Scholar]
- 21.Anderson PT, Rowe NE. Current attitudes of Canadian urologists towards surgical castration in the treatment of prostate cancer. Can Urol Assoc J. 2021;15:E281. doi: 10.5489/cuaj.6834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Schaeffer E, Srinivas S, Antonarakis ES, Armstrong AJ, Bekelman JE, Cheng H, et al. NCCN guidelines insights: prostate cancer, version 1.2021. J Natl Compr Canc Netw. 2021;19:134–43. doi: 10.6004/jnccn.2021.0008. [DOI] [PubMed] [Google Scholar]
- 23.Rot I, Wassersug RJ, Walker LM. What do urologists think patients need to know when starting on androgen deprivation therapy? The perspective from Canada versus countries with lower gross domestic product. Transl Androl Urol. 2016;5:235–47. doi: 10.21037/tau.2016.03.06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Agrawal V, Ma X, Hu JC, Barbieri CE, Nagar H. Trends in androgen deprivation use in men with intermediate-risk prostate cancer who underwent radiation therapy. Adv Radiat Oncol. 2022;7:100904. doi: 10.1016/j.adro.2022.100904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Leong DP, Fradet V, Shayegan B, Duceppe E, Siemens R, Niazi T, et al. Cardiovascular risk in men with prostate cancer: insights from the RADICAL PC study. J Urol. 2020;203:1109–16. doi: 10.1097/JU.0000000000000714. [DOI] [PubMed] [Google Scholar]
- 26.Davis MK, Rajala JL, Tyldesley S, Pickles T, Virani SA. The prevalence of cardiac risk factors in men with localized prostate cancer undergoing androgen deprivation therapy in British Columbia, Canada. J Oncol. 2015;2015:820403. doi: 10.1155/2015/820403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Sun L, Parikh RB, Hubbard RA, Cashy J, Takvorian SU, Vaughn DJ, et al. Assessment and management of cardiovascular risk factors among US veterans with prostate cancer. JAMA Netw Open. 2021;4:e210070–e210070. doi: 10.1001/jamanetworkopen.2021.0070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Lyon AR, López-Fernández T, Couch LS, Asteggiano R, Aznar MC, Bergler-Klein J, et al. 2022 ESC guidelines on cardio-oncology developed in collaboration with the European Hematology Association (EHA), the European Society for Therapeutic Radiology and Oncology (ESTRO) and the International Cardio-Oncology Society (IC-OS) Eur Heart J. 2022;43:4229–361. doi: 10.1093/eurheartj/ehac244. [DOI] [PubMed] [Google Scholar]
- 29.Wilding S, Downing A, Selby P, Cross W, Wright P, Watson EK, et al. Decision regret in men living with and beyond nonmetastatic prostate cancer in the United Kingdom: a population-based patient-reported outcome study. Psychooncology. 2020;29:886–93. doi: 10.1002/pon.5362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Chan JSK, Tang P, Hui JMH, Lee YHA, Dee EC, Ng K, et al. Association between duration of gonadotrophin-releasing hormone agonist use and cardiovascular risks: a population-based competing-risk analysis. Prostate. 2022;82:1477–80. doi: 10.1002/pros.24423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Montazeri K, Unitt C, Foody JM, Harris JR, Partridge AH, Moslehi J. ABCDE steps to prevent heart disease in breast cancer survivors. Circulation. 2014;130:e157–9. doi: 10.1161/CIRCULATIONAHA.114.008820. [DOI] [PubMed] [Google Scholar]
- 32.Bhatia N, Santos M, Jones LW, Beckman JA, Penson DF, Morgans AK, et al. Cardiovascular effects of androgen deprivation therapy for the treatment of prostate cancer: ABCDE steps to reduce cardiovascular disease in patients with prostate cancer. Circulation. 2016;133:537. doi: 10.1161/CIRCULATIONAHA.115.012519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Trapani D, Zagami P, Nicolò E, Pravettoni G, Curigliano G. Management of cardiac toxicity induced by chemotherapy. J Clin Med. 2020;9:2885. doi: 10.3390/jcm9092885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Alexandre J, Cautela J, Ederhy S, Damaj GL, Salem JE, Barlesi F, et al. Cardiovascular toxicity related to cancer treatment: a pragmatic approach to the American and European cardio-oncology guidelines. J Am Heart Assoc. 2020;9:18403. doi: 10.1161/JAHA.120.018403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Okwuosa TM, Morgans A, Rhee JW, Reding KW, Maliski S, Plana JC, et al. Impact of hormonal therapies for treatment of hormone-dependent cancers (breast and prostate) on the cardiovascular system: effects and modifications: a scientific statement from the American Heart Association. Circ Genomic Precis Med. 2021;14:E000082. doi: 10.1161/HCG.0000000000000082. [DOI] [PubMed] [Google Scholar]
- 36.Clemens J, Gottlieb JD. Do physicians’ financial incentives affect medical treatment and patient health? Am Econ Rev. 2014;104:1320–49. doi: 10.1257/aer.104.4.1320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Wendt C, Kohl J. Translating monetary inputs into health care provision: a comparative analysis of the impact of different modes of public policy. J Comp Policy Anal Res Pract. 2010;12:11–31. doi: 10.1080/13876980903076161. [DOI] [Google Scholar]
- 38.Wong HF, Yee CH, Teoh JYC, Chan SYS, Chiu PKF, Cheung HY, et al. Time trend and characteristics of prostate cancer diagnosed in Hong Kong (China) in the past two decades. Asian J Androl. 2019;21:104. doi: 10.4103/aja.aja_75_18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Wong AYS, Root A, Douglas IJ, Chui CSL, Chan EW, Ghebremichael-Weldeselassie Y, et al. Cardiovascular outcomes associated with use of clarithromycin: population based study. BMJ. 2016;352:h6926. doi: 10.1136/bmj.h6926. [DOI] [PubMed] [Google Scholar]
- 40.Lau WCY, Chan EW, Cheung CL, Sing CW, Man KKC, Lip GYH, et al. Association between dabigatran vs warfarin and risk of osteoporotic fractures among patients with nonvalvular atrial fibrillation. J Am Med Assoc. 2017;317:1151–8. doi: 10.1001/jama.2017.1363. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All underlying data is available on reasonable request to the corresponding authors.


