Background:
COVID-19 forced healthcare systems to make unprecedented changes in clinical care processes. The authors hypothesized that the COVID-19 pandemic adversely impacted timely access to care, perioperative processes, and clinical outcomes for pediatric patients undergoing primary appendectomy.
Methods:
A retrospective, international, multicenter study was conducted using matched cohorts within participating centers of the international PEdiatric Anesthesia COVID-19 Collaborative (PEACOC). Patients younger than 18 yr old were matched using age, American Society of Anesthesiologists Physical Status, and sex. The primary outcome was the difference in hospital length of stay of patients undergoing primary appendectomy during a 2-month period early in the COVID-19 pandemic (April to May 2020) compared with prepandemic (April to May 2019). Secondary outcomes included time to appendectomy and the incidence of complicated appendicitis.
Results:
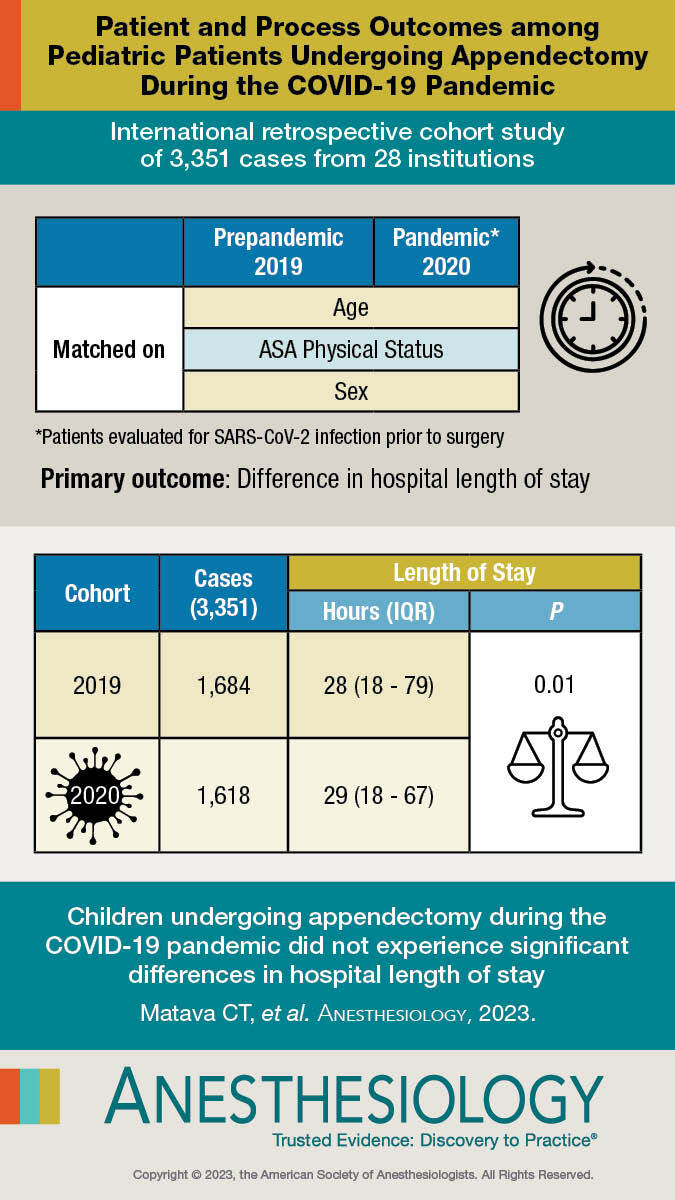
A total of 3,351 cases from 28 institutions were available with 1,684 cases in the prepandemic cohort matched to 1,618 in the pandemic cohort. Hospital length of stay was statistically significantly different between the two groups: 29 h (interquartile range: 18 to 79) in the pandemic cohort versus 28 h (interquartile range: 18 to 67) in the prepandemic cohort (adjusted coefficient, 1 [95% CI, 0.39 to 1.61]; P < 0.001), but this difference was small. Eight centers demonstrated a statistically significantly longer hospital length of stay in the pandemic period than in the prepandemic period, while 13 were shorter and 7 did not observe a statistically significant difference. During the pandemic period, there was a greater occurrence of complicated appendicitis, prepandemic 313 (18.6%) versus pandemic 389 (24.1%), an absolute difference of 5.5% (adjusted odds ratio, 1.32 [95% CI, 1.1 to 1.59]; P = 0.003). Preoperative SARS-CoV-2 testing was associated with significantly longer time-to-appendectomy, 720 min (interquartile range: 430 to 1,112) with testing versus 414 min (interquartile range: 231 to 770) without testing, adjusted coefficient, 306 min (95% CI, 241 to 371; P < 0.001), and longer hospital length of stay, 31 h (interquartile range: 20 to 83) with testing versus 24 h (interquartile range: 14 to 68) without testing, adjusted coefficient, 7.0 (95% CI, 2.7 to 11.3; P = 0.002).
Conclusions:
For children undergoing appendectomy, the COVID-19 pandemic did not significantly impact hospital length of stay.
A total of 1,618 pediatric patients across 28 institutions undergoing appendectomy from April to May 2020 were age, sex, and American Society of Anesthesiologists Physical Status matched to 1,684 similar patients treated in April to May 2019. Overall, hospital length of stay was similar, demonstrating an average of 29 h during the pandemic cohort versus 28 h prepandemic cohort.
Visual Abstract:
Editor’s Perspective.
What We Already Know about This Topic
The COVID-19 pandemic caused challenges in access to clinical care and alterations in clinical process for operative and nonoperative care
It remains unknown whether common pediatric procedures, such as appendectomy for operative management of acute appendicitis, demonstrated differences in length of stay, time to surgery, or clinical outcomes during the early stages of the pandemic
What This Article Tells Us That Is New
A total of 1,618 pediatric patients across 28 institutions undergoing appendectomy from April to May 2020 were age, sex, and American Society of Anesthesiologists Physical Status matched to 1,684 similar patients treated in April to May 2019
Overall, hospital length of stay was similar, demonstrating an average of 29 h during the pandemic cohort versus 28 h prepandemic cohort
The COVID-19 pandemic has forced healthcare systems to make unprecedented changes in clinical care pathways and processes to accommodate changes in patient volumes and to protect healthcare professionals from infection.1 Similarly, health agencies and institutions implemented policies regarding the use of personal protective equipment and preoperative testing for SARS-CoV-2 in patients.2–6 Despite the lesser burden of COVID-19 among pediatric patients, the global ramp-down of elective surgical cases and the implementation of preoperative testing were extended to many pediatric centers.7–10 Although intended to identify infected patients and protect other patients and the healthcare workforce, these processes may have led to delays in patient care, resulting in greater morbidity and mortality among patients requiring emergent surgeries, including appendectomy for appendicitis. Appendicitis is one of the most common diseases requiring emergency surgery worldwide.11 Race, insurance status, and lower rates of outpatient healthcare utilization have been shown to be associated with greater occurrence of complicated appendicitis during COVID-19 in children.7,8,12–14
We hypothesized that hospital length of stay would be longer during the COVID-19 pandemic along with longer access to care times, longer perioperative processes, and more negative clinical outcomes for pediatric patients undergoing primary appendectomy. To assess this impact, we compared perioperative care processes and patient outcomes in children undergoing appendectomy during the COVID-19 pandemic (April to May 2020) with those from a prepandemic period (April to May 2019). Centers from members of the international PEdiatric Anesthesia COVID-19 Collaborative (PEACOC) participated in the study.
Our primary aim was to determine the difference in hospital length of stay between pediatric patients undergoing primary appendectomy during a 2-month period early during the COVID-19 pandemic (April to May 2020), compared with matched cohorts during the same 2-month period the previous year (April to May 2019).
Materials and Methods
Study Design and Population
This study was approved by the Institutional Review Board at Boston Children’s Hospital (IRB-P00035907; Boston, Massachusetts). Each individual center also obtained separate institutional review board approval or waiver as per their local institutional policies. Because this was a retrospective study, the requirements for written informed consent and assent were waived by each center’s institutional review board. We designed this retrospective, observational, multicenter study using matched cohorts from participating PEdiatric Anesthesia COVID-19 Collaborative member institutions. Eligibility criteria included data from patients younger than 18 yr of age undergoing primary appendectomy for acute appendicitis. Patients who underwent appendectomy during the study periods were identified and data abstracted from each institution’s electronic medical record. Exact matching was performed manually at each study center. Exact matching criteria included patient age (< 1, 1 to 2, 3 to 5, 6 to 11, 12 to 18 yr), American Society of Anesthesiologists (ASA) Physical Status classifications (ASA Physical Status 1 to 2, 3 to 4, and 5), and sex (male, female).15,16 To minimize sampling bias, we included all pediatric patients undergoing primary appendectomy during both study periods and allowed one-to-many matching to increase the sample size and include all cases. In addition, we minimized differential measurement bias by recording data elements that are commonly used throughout the clinical course of all cases. Each center used a common data dictionary to abstract data from their electronic health record systems (Supplemental Digital Content 1, https://links.lww.com/ALN/D120, and Supplemental Digital Content 2, https://links.lww.com/ALN/D121). All data were entered into a secure, central REDCap database maintained at Boston Children’s Hospital.17 Data were reviewed for missingness or errors. Two investigators, CTM and AFS, reviewed all data entries and contacted each center to verify and validate data as needed.
Study Definitions
The exposure variables were the period of appendectomy (April to May 2019 vs. April to May 2020) and COVID-19 test result. Measured potential confounding factors that were examined included age, weight, sex, race, ethnicity, ASA Physical Status, emergent procedure, and insurance type. Our primary outcome was hospital length of stay, measured in hours. We chose hospital length of stay as a surrogate measure that encompasses the impact of timely access to care, perioperative processes, and patient-centered outcomes. Secondary outcomes were time to appendectomy (time to intervention), total operative time, time from operating room entry to intubation, total anesthesia time, postanesthesia care unit (PACU) length of stay, length of time of symptoms before presentation, occurrence of severe postoperative pain, pathology of appendicitis, surgical complexity of appendectomy, need for second procedure, occurrence of postoperative stroke, renal failure requiring renal replacement therapy, respiratory failure, need for inotropic therapy, cardiac arrest, and mortality. No variables were analyzed as effect modifiers. In addition, we compared the primary and secondary outcomes between patients from the 2020 cohort who had a positive SARS-CoV-2 test or were patients under investigation versus those with a negative SARS-CoV-2 test during the pandemic period. We used the rate of SARS-CoV2 positive tests or patients under investigation as a marker of COVID-19 burden at each center. Outcome definitions are highlighted in appendix 2 and detailed in the study protocol (Supplemental Digital Content 2, https://links.lww.com/ALN/D121) and statistical analysis plan (Supplemental Digital Content 3, https://links.lww.com/ALN/D122). The Statistical Analysis Plan was written and filed with a private entity (institutional review board) before data were accessed. We used the National Surgical Quality Improvement Program Pediatric definition to categorize primary appendectomy as simple versus complicated.18 Simple appendicitis was defined as surgical findings of appendicitis without clinical evidence of perforation or purulent fluid associated with perforation, and complicated appendicitis was defined as perforated appendicitis with clinical evidence of perforation or purulent fluid associated with perforation (appendix 2). Excluded were patients older than 18 yr, or those where appendectomy was performed as part another procedure (e.g., Ladd procedure, interval appendectomy).
Further, we aimed to assess the influence of SARS-CoV-2 polymerase chain reaction test positivity or patients under investigation on primary and secondary outcomes during the pandemic year compared with those with a negative test or no SARS-CoV-2 polymerase chain reaction test.
Statistical Analysis
No statistical power calculation was conducted before the study. All continuous data are presented as medians and interquartile ranges, and all categorical data are presented as frequencies and percentages. Comparisons of all patient characteristics between April to May 2019 versus April to May 2020 were performed using the absolute standardized mean difference as a measure of balance between the two comparison groups. Standardized mean difference values less than 0.10 were considered as reflecting good balance between the two groups on a given explanatory variable. Because good balance was determined for all baseline confounding factors between the two cohorts based on the standardized mean difference, all data were included in the analyses of outcomes. We did not perform any imputation because missing data were only observed for ethnicity, with 2% missing observation (n = 67 of 3,302). The distribution of nonmissing observations is provided in Supplemental Digital Content 4 (https://links.lww.com/ALN/D123). For both pathology of appendicitis and surgical complexity of appendectomy, logistic regression was implemented with dichotomization of complicated versus normal or simple.
For continuous outcomes, including the primary outcome of hospital length of stay, data were summarized as medians and interquartile ranges due to departure from normality as determined by the Shapiro-Wilk test. Statistical comparisons for continuous outcomes were performed using mixed-effect median regression modeling with a random effect for the matched set. Conditional logistic regression was implemented to assess binary outcomes, with a conditional specification for matched set. Median regression was performed using quantile regression on the 50th percentile (median) to compare the median of the distribution for the continuous outcomes between the time-period groups.19 Binary outcomes were expressed as frequencies and percentages, and conditional logistic regression was implemented with the matched set incorporated as a random effect in the model. Centers were incorporated into the model as a random effect. Analyses comparing pandemic versus prepandemic time periods were also performed stratified by center and geographical region. Results were summarized as adjusted coefficients or odds ratios with corresponding 95% CIs and P values, where the term “adjusted” refers to the results from conditional regression accounting for the matched set as a random effect in each model. Forest plots were constructed to visualize the results regarding key outcomes, by center and by region. Centers were compared based on outcome findings using the Wilcoxon rank sum test for continuous variables and Fisher exact test for categorical or binary data. Post hoc exploratory analysis using mixed-effects median regression was performed to assess the associations between-center volume (per 100 patients), SARS-CoV-2 testing, and outcomes. Subgroup analyses of SARS-CoV-2 testing status in 2020 according to SARS-CoV-2 positivity or patients under investigation status were performed using the same regression techniques.
All statistical analyses were performed using Stata (version 16.1, StataCorp LLC., College Station, Texas). A two-tailed P < 0.05 was considered statistically significant for the primary outcome. For all secondary outcomes using a Bonferroni correction for multiple tests (0.05 divided by 16) we determined a two-tailed P < 0.0031 as statistically significant.20 The study is reported using the Strengthening the Reporting of Observational Studies in Epidemiology statement: guidelines for reporting observational studies.21
Sensitivity Analyses
In addition to the primary analysis, we performed preplanned sensitivity analyses to look at hospital length of stay after excluding outlier centers. We defined outliers as centers experiencing an increase or decrease greater than 12 h in hospital length of stay, the primary outcome.
In response to peer review, additional post hoc sensitivity analyses were performed on all patients without including a random effect of matched set to avoid matching bias.
Results
Study Population and Patient Characteristics
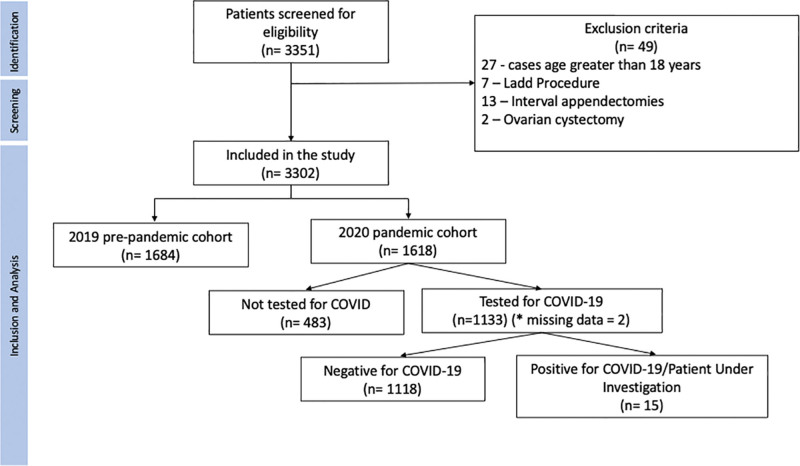
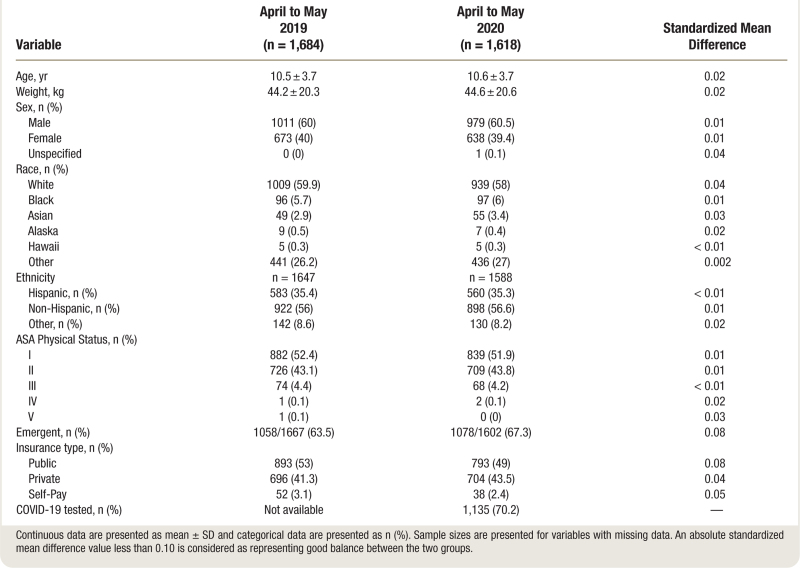
Of the 3,351 patients identified as undergoing appendectomy, 3,302 were included in the study with 1,684 patients in the prepandemic cohort and 1,618 in the pandemic cohort, and 49 patients met exclusion criteria (fig. 1). Twenty-eight institutions contributed data for the study (Supplemental Digital Content 5, https://links.lww.com/ALN/D124).22 We used all cases in the analysis, regardless of matching status. In the full dataset, we had a total of n = 3,302 primary appendectomies, and 86 cases (2.6%) did not have at least one valid match. Baseline characteristics of the participants are shown in table 1. All variables had a standardized mean difference less than 0.10 between the study groups, indicating good balance between the two study groups (table 1). The mean age was 11 yr in the prepandemic cohort versus 11 yr in the pandemic cohort. Three patients (0.27%) were confirmed SARS-CoV-2 positive and 12 were managed as patients under investigation.
Fig. 1.
Study flow chart.
Table 1.
Patient Characteristics during the Prepandemic Period vs. the Pandemic Period
Outcomes
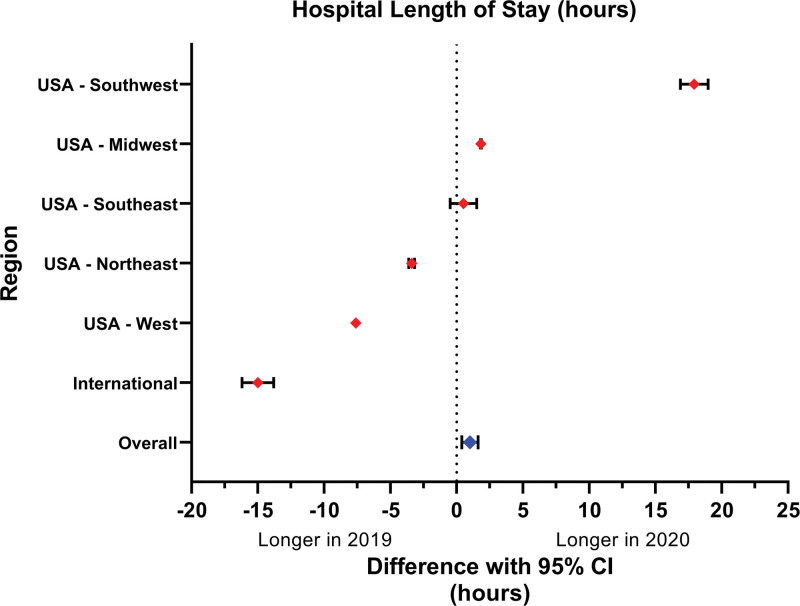
There was greater, albeit not clinically significant, hospital length of stay from a median of 28 h in the prepandemic cohort to 29 h in the pandemic group (adjusted coefficient, 1 h [95% CI, 0.39 to 1.61]; P < 0.001; table 2 and fig. 2). Individual centers had differing hospital length of stay changes from the prepandemic to the pandemic period (fig. 2). Eight centers demonstrated a statistically significantly longer hospital length of stay in the pandemic period compared with the prepandemic period, while 13 demonstrated a shorter hospital length of stay during the pandemic period (fig. 2) and 7 did not observe a statistically significant difference. Two regions demonstrated a statistically significantly longer hospital length of stay in the pandemic period than in the prepandemic period, three regions demonstrated a shorter hospital length of stay during the pandemic period, and one region did not observe a statistically significant difference (fig. 3).
Table 2.
Comparison of Outcomes during the Prepandemic Period vs. the Pandemic Period
Fig. 2.
Primary outcome of hospital length of stay difference prepandemic versus pandemic period, stratified by individual center. Pediatric patients undergoing primary appendectomy for acute appendicitis during the COVID-19 pandemic (April to May 2020) were compared with matched cohorts during the same 2-month period the previous year (April to May 2019). Overall, a statistically significant, but clinically insignificant difference was observed: 29 h (interquartile range: 18 to 79) in the pandemic cohort versus 28 h (interquartile range: 18 to 67) in the prepandemic cohort (adjusted coefficient, 1 [95% CI, 0.39 to 1.61]; P < 0.001).
Fig. 3.
Primary outcome of hospital length of stay difference prepandemic versus pandemic period, stratified by geographic region. Pediatric patients undergoing primary appendectomy for acute appendicitis during the COVID-19 pandemic (April to May 2020), were compared with matched cohorts during the same 2-month period the previous year (April to May 2019). Centers were grouped by geographical region to evaluate any geographic association with changes in hospital length of stay.
Time to appendectomy was greater in the pandemic period, 648 min (interquartile range: 357 to 1006) versus the prepandemic period, 540 min (interquartile range: 305 to 840) (adjusted coefficient, 108 min [95% CI, 106 to 110]; P < 0.001; table 2 and Supplemental Digital Content 6, https://links.lww.com/ALN/D125). Patient care processes such as total operative time, total anesthesia time, and PACU length of stay changed from the prepandemic to the pandemic period across individual institutions (table 2, Supplemental Digital Content 6, https://links.lww.com/ALN/D125) and geographical regions (Supplemental Digital Content 7, https://links.lww.com/ALN/D126). During the pandemic period, there were more patients presenting with complicated appendicitis, prepandemic 18.6% versus pandemic 24.1%, absolute difference of 5.5% (adjusted odds ratio, 1.32 [95% CI, 1.1 to 1.59]; P = 0.003).
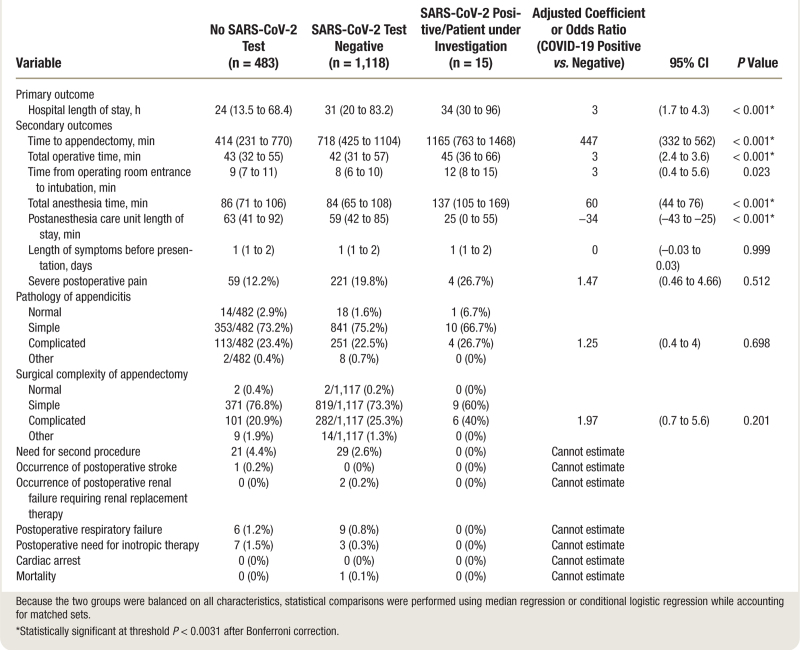
Table 3 shows the outcomes of patients presenting within the pandemic cohort. During the study period, 15 patients received a positive SARS-CoV-2 test result or were patients under investigation during the pandemic and overall had a greater and clinically significant hospital length of stay than patients who tested negative or patients who were not tested (hospital length of stay 34 h [interquartile range: 30 to 96], 31 h [interquartile range: 20 to 83], and 24 h [interquartile range: 14 to 68], respectively; adjusted coefficient, 3 h [95% CI, 1.7 to 4.3]; P < 0.001). Preoperative SARS-CoV-2 testing was associated with significantly longer hospital length of stay (31 h [interquartile range: 20 to 83] with testing versus 24 h [interquartile range: 14 to 68] without testing; adjusted coefficient, 7 [95% CI, 2.7 to 11.3]; P = 0.002). In addition, patients testing positive for SARS-CoV-2 had a greater and clinically significant median time to intervention of 1,165 min (interquartile range: 763 to 1,468) compared with 718 min (interquartile range: 425 to 1,104) for those testing negative for SARS-CoV-2 and 414 min (interquartile range: 231 to 770) for those not requiring a SARS-CoV-2 test (adjusted odds coefficient, 447 min [95% CI, 332 to 562]; P < 0.001; table 3). Preoperative SARS-CoV-2 testing was associated with significantly longer time to appendectomy (720 min [interquartile range: 430 to 1112] with testing versus 414 min [interquartile range: 231 to 770] without testing; adjusted coefficient, 306 [95% CI, 241 to 371]; P < 0.001).
Table 3.
Analysis of Outcomes by SARS-CoV-2 Status in the Pandemic Cohort
Supplemental Digital Content 8 (https://links.lww.com/ALN/D127) shows the characteristics of institutions with longer or shorter process measures in prepandemic versus pandemic time periods (post hoc analysis). Supplemental Digital Content 9 (https://links.lww.com/ALN/D128) reports the association of center volumes, proportion of SARS-CoV-2–tested patients, and SARS-CoV-2 positive results with study outcomes. For each center, the case volume of every additional 100 patients at a center was associated with a decrease of 0.41 days in hospital length of stay (adjusted coefficient, –0.41 days [95% CI, –0.42 to –0.41]). Centers with a higher volume of cases, more patients undergoing testing, and higher rate of SARS-CoV-2 testing were associated with longer time to appendectomy. For every additional 100 patients tested for SARS-CoV-2 at a given center, we estimate that this was associated with an increased median time to appendectomy of 96 min (95% CI, increase of 95.7 to 96.3). In other words, when more patients are tested for SARS-CoV-2 at a given center, this was associated with a longer time to appendectomy.
In 2020, a 1% increase in the rate of SARS-CoV-2 test positivity was associated with an increase in time to appendectomy by 17.4 h (95% CI, 15.2 to 19.5 h). A higher SARS-CoV-2 test positivity rate in a center was associated with longer time to appendectomy.
Sensitivity Analysis
In preplanned sensitivity analysis evaluating hospital length of stay after excluding outlier centers (difference in hospital length of stay from prepandemic to pandemic time period of more than 12 h), the median hospital length of stay in prepandemic was 27 h (interquartile range: 18 to 56) and the median hospital length of stay in pandemic was 26 h (interquartile range: 17 to 69; adjusted coefficient, –1 [95% CI, –3.6 to 1.6]; P = 0.456). Therefore, with outliers removed, there was no statistically or clinically significant difference in prepandemic versus pandemic hospital length of stay. Post hoc sensitivity analysis performed in response to peer review performed without including a random effect of matched set to avoid matching bias did not demonstrate any differences in the primary outcome of median hospital length of stay, with a median of 28 h (interquartile range: 18 to 67) in the prepandemic cohort to 29 h (interquartile range: 18 to 79) in the pandemic group (adjusted coefficient, 1 h [95% CI, –1.8 to 3.8]; P = 0.49).
Discussion
Among children undergoing emergency appendectomy, the COVID-19 pandemic did not impact processes and patient-centered outcomes when compared with the prepandemic period. Specifically, our study shows that the pandemic was not associated with a clinically significant greater hospital length of stay, or a clinically significant delayed access to surgical care, complicated appendicitis, and severe postoperative pain. However, children undergoing SARS-CoV-2 testing in the pandemic period, especially those patients testing positive for SARS-CoV-2, experienced greater hospital length of stay and time to appendectomy than those either not tested or with a negative SARS-CoV-2 test.
The impact of the pandemic on patient care has been reported among pediatric patients presenting for nonsurgical care10,23–29; we evaluated this impact among children presenting for a common surgical emergency across an international consortium of pediatric centers. Although the primary outcome, hospital length of stay, showed a statistically significant increase of 1 h, this increase is not clinically significant. The changes in hospital length of stay varied across centers and geographic regions. In a sensitivity analysis looking at hospital length of stay after removal of outliers and controlling for matching bias, there was no difference in hospital length of stay between the two cohorts.
Although hospital length of stay was not clinically significantly different prepandemic versus pandemic, we did observe statistically and clinically significant differences in processes of care associated with SARS-CoV-2 testing. These findings may reflect the different phases of the pandemic that influenced local hospital policies during the study period. The rolling implementation of preoperative SARS-CoV-2 testing, delays associated with the duration of SARS-CoV-2 testing, and safety practices such as operating room “settle time” and a “clearance period” before other personnel entered the room may have influenced workflows and processes, including time to intervention, operative time, anesthesia time, and turnover times.30 A higher burden of SARS-CoV-2 testing and positive tests may have played a significant role in delaying time to appendectomy in these patients. However, very few patients tested positive.
Our study was performed in what can be defined as the early phase of the pandemic, April to May 2020, when institutions were still adjusting to the pandemic. A survey conducted in June 2020 of 98 participating institutions from 31 countries reported an estimated 2-h increase in the time to diagnosis and another 2-h increase in time to intervention in patients presenting for emergency surgery.31 Changes in the perioperative workflow may have included delays in access to diagnostic imaging and delays related to processing of preoperative testing. It is possible that, as the pandemic progressed, these workflow processes impacted time to intervention among our study population. Most patients in our study underwent preoperative SARS-CoV-2 testing. Our study reports a low incidence of 0.27% positivity rate among children tested for SARS-CoV-2 before emergent appendectomy during the period April to May 2020. However, this incidence may have changed based on the “wave” of the pandemic and emergence of new variants.
Our study observed an increase in complicated appendicitis. The observed increase in time to intervention may be associated with the more complicated cases of appendicitis encountered. The overall incidence of COVID-19 was low in our study population, and so unlikely that COVID-19 itself had a direct impact on the complexity of acute appendicitis. The pandemic has also been associated with late and more severe presentations among populations with acute gastrointestinal diseases, including acute cholecystitis in adults.28 Our study did not observe an increased time to diagnosis across the prepandemic period versus pandemic period or in patients who underwent SARS-CoV-2 testing. Although reports confirm hesitancy in accessing healthcare among adults, data for this phenomenon are lacking for children.28 Further, our study demonstrates similar access-to-care times for appendectomies during the pandemic period April to May 2020 and prepandemic period April to May 2019. Our study does not report on patients that underwent nonoperative management; however, during the pandemic, a very low proportion (3.9%) of surgeons are reported to have considered this option for acute appendicitis in children.32 We observed that very few patients undergoing appendectomy were SARS-CoV-2 positive and none required preoperative oxygen or postoperative respiratory support.
The COVID-19 pandemic cohort undergoing preoperative testing and especially those testing positive or patients under investigation was associated with significantly longer hospital length of stay, time to intervention, and higher rate of complicated appendectomy while anesthesia times, operative times, and PACU length of stay were shorter. Despite a small number of patients testing SARS-CoV-2 positive or patients under investigation, these findings may indicate the influence of process changes on patient outcomes. The delays in accessing care can be explained by delayed presentation to hospitals due to pandemic-related avoidance of medical care.25 However, in our study, SARS-CoV-2 testing was associated with statistically and clinically significant increased time to intervention and hospital length of stay.
Centers with a higher volume of cases, more patients undergoing testing, and higher rate of SARS-CoV-2 testing experienced significantly longer time to appendectomy (time to intervention; Supplemental Digital Content 4, https://links.lww.com/ALN/D123). In other words, when more patients were tested for SARS-CoV-2, this was associated with longer times to appendectomy. The challenges of processing a larger number of nasal swabs with molecular testing may have played a significant role at the centers with a greater burden of COVID-19.
Our study has several limitations. We were unable to determine the prevalence of COVID-19 in the general community at each location, therefore unable to provide data on how overall prevalence may have affected the primary and secondary outcomes. Across centers, there was variation in the timing of elective surgery shutdowns and of universal testing of surgical patients. Even during the study period, the status of each location was rapidly evolving. However, the impact of the pandemic may have still affected clinical and workforce processes implemented at that time. The strengths of our study include the focus on a common global pathologic process, acute appendicitis, with standardized management and definition of outcomes within and across institutions. We also included data from a wide geographical area that allowed for both a global view of the impact of the pandemic and a more focused view of the changes at each individual institution, including a wide range of institution patient volumes. Different public health measures implemented in a variety of geographical areas included school closures, stay-at-home orders, and centralization of pediatric care to tertiary institutions; these could also have impacted volume and time from diagnosis to definitive treatment.
In summary, we studied the impact of the pandemic on pediatric perioperative care in a large, multicenter, international cohort and observed that children undergoing emergent primary appendectomy during the COVID-19 pandemic did not experience clinically significant longer hospital length of stay. There was a statistically significant increase in time to appendectomy (108 min, P < 0.001) in the pandemic cohort compared with the prepandemic cohort. In addition, children testing positive for SARS-CoV-2 or managed as patients under investigation had greater hospital length of stay, time to appendectomy, and total anesthesia time than those testing negative for SARS-CoV-2. SARS-CoV-2 testing was associated with longer processes of care, especially among those testing positive or patients under investigation despite low numbers and requires consideration during the ongoing COVID-19 pandemic and future pandemics.
Acknowledgments
The authors thank Rachel Bernier, M.P.H., and Jocelyn Booth, B.S.N., R.N., from the Department of Anesthesia, Boston Children’s Hospital (Boston, Massachusetts), for their administrative assistance in setting up and maintaining the study RedCap Database.
Research Support
All authors’ support was provided solely from institutional and/or departmental sources except for Dr. von Ungern-Sternberg who received funding from the Stan Perron Charitable Foundation (East Perth, Western Australia); Dr. Matava was supported in part by the Canadian Pediatric Perioperative Outcome National Datalake (CANNPOND; Toronto, Canada); Dr. Raman has received funding from NDS Kalstars (Columbus, Ohio), Opus Influunt (Dublin, Ohio), Merck (Rahway, New Jersey), and owns private equity in Face tomface (Columbus, Ohio) and stocks in Johnson & Johnson (New Brunswick, New Jersey).
Competing Interests
The authors declare no competing interests.
Supplemental Digital Content
Supplemental Digital Content 1. PEdiatric Anesthesia COVID-19 Collaborative data dictionary, https://links.lww.com/ALN/D120
Supplemental Digital Content 2. Study Protocol, https://links.lww.com/ALN/D121
Supplemental Digital Content 3. Statistical Analysis Protocol, https://links.lww.com/ALN/D122
Supplemental Digital Content 4. Non-missing observations, https://links.lww.com/ALN/D123
Supplemental Digital Content 5. List of Participating Institutions, https://links.lww.com/ALN/D124
Supplemental Digital Content 6. Secondary Outcomes by institution, https://links.lww.com/ALN/D125
Supplemental Digital Content 7. Secondary Outcomes according to geographical region, https://links.lww.com/ALN/D126
Supplemental Digital Content 8, Comparison of centers, https://links.lww.com/ALN/D127
Supplemental Digital Content 9. Centers and outcomes, https://links.lww.com/ALN/D128
Appendix 1. List of PEdiatric Anesthesia COVID-19 Collaborative Nonauthor Collaborators
Anita Akbar Ali, M.D.: Division of Pediatric Anesthesiology and Pain Medicine, University of Arkansas for Medical Sciences/Arkansas Children’s Hospital, Little Rock, Arkansas. Served as a scientific advisor and collected data.
Mohamad F. Al-Rabbat, M.D.: Department of Anesthesia, Sidra Medicine, Doha, Qatar. Served as a scientific advisor and collected data.
Alyssa B. Brzenski, M.D.: Department of Anesthesiology, University of California - San Diego, San Diego, California; Department of Anesthesiology, Rady Children’s Hospital, San Diego, California. Served as a scientific advisor and collected data.
William F. Casey, M.D., B.Ch., F.F.A.R.C.S.I.: Department of Anesthesia, Sidra Medicine, Doha, Qatar. Served as a scientific advisor and collected data.
Surendrasingh Chhabada, M.D.: Department of Pediatric Anesthesiology, Cleveland Clinic, Cleveland, Ohio; Department of Outcomes Research, Anesthesiology Institute, Cleveland Clinic, Cleveland Ohio. Served as a scientific advisor and collected data.
Michael Collin, M.B.B.S., F.R.A.C.S.: Department of Paediatric Surgery, Perth Children’s Hospital, Perth, Western Australia, Australia. Served as a scientific advisor and collected data.
Vipul J. Dhumak, M.D., M.P.H.: Department of Anesthesiology, West Virginia University Medicine Children’s, Morgantown, West Virginia. Served as a scientific advisor and collected data.
Ajay D’Mello, M.D.: Department of Anesthesia and Pain Medicine, Nationwide Children’s Hospital, Columbus, Ohio. Served as a scientific advisor and collected data.
Piedad C. Echeverry, M.D.: Department of Anesthesia and Pain Medicine, Nationwide Children’s Hospital, Columbus, Ohio. Served as a scientific advisor and collected data.
Pavithra R. Ellison, M.D., M.M.M.: Department of Anesthesiology, West Virginia University Medicine Children’s, Morgantown, West Virginia.
Allison M. Fernandez, M.D., M.B.A.: Department of Anesthesia, Pain and Perioperative Medicine, Johns Hopkins All Children’s Hospital, St. Petersburg, Florida.
Jake A. Fisher, M.D.: Department of Anesthesiology and Pain Medicine, University of California – Davis, Sacramento, California. Served as a scientific advisor and collected data.
Clinton L. Fuller, M.D.: Department of Anesthesiology, Perioperative and Pain Medicine, Baylor College of Medicine/Texas Children’s Hospital, Houston, Texas. Served as a scientific advisor and collected data.
Chris D. Glover, M.D.: Department of Anesthesiology, Perioperative and Pain Medicine, Baylor College of Medicine/Texas Children’s Hospital, Houston, Texas. Served as a scientific advisor and collected data.
Velu Guruswamy, M.B.B.S., D.A., F.R.C.A., F.F.A.R.C.S.I., M.B.A.: Department of Pediatric Anesthesia, Sidra Medicine, Doha, Qatar. Served as a scientific advisor and collected data.
Emily B. Hesselink, M.D.: Department of Anesthesia, University of Wisconsin-Madison, Madison, Wisconsin. Served as a scientific advisor and collected data.
Agnes I. Hunyady, M.D.: Department of Anesthesiology and Pain Medicine, Seattle Children’s Hospital, Seattle, Washington; Department of Anesthesiology and Pain Medicine, University of Washington School of Medicine, Seattle, Washington. Served as a scientific advisor and collected data.
Amanda N. Lorinc, M.D.: Department of Pediatric Anesthesiology, Lurie Children’s Hospital, Northwestern University, Feinberg School of Medicine, Chicago, Illinois. Served as a scientific advisor and collected data.
Michael King, M.D.: Department of Pediatric Anesthesiology, Lurie Children’s Hospital, Northwestern University, Feinberg School of Medicine, Chicago, Illinois.
Lavinia Mihaila, B.A.: Department of Pediatric Anesthesiology, University of California – Davis, Sacramento, California. Served as a scientific advisor and collected data.
Jonathon H. Nelson, M.D.: Division of Anesthesiology and Pain Medicine, Children’s National Hospital, Washington, D.C. Served as a scientific advisor and collected data.
Ann S. Ng, M.D.: Department of Anesthesiology, Perioperative and Pain Medicine, Baylor College of Medicine/Texas Children’s Hospital, Houston, Texas.
Joshua K. Ramjist, M.D., M.Sc.: Department of General Surgery, The Hospital for Sick Children, Toronto, Ontario, Canada. Served as a scientific advisor and collected data.
Nirop R. Ravula, M.B.B.S., F.R.C.A.: Department of Anesthesiology and Pain Medicine, University of California – Davis, Sacramento, California. Served as a scientific advisor and collected data.
Arundathi Reddy, M.D.: Division of Pediatric Anesthesiology and Pain Medicine, University of Arkansas for Medical Sciences/Arkansas Children’s Hospital, Little Rock, Arkansas. Served as a scientific advisor and collected data.
Elizabeth Rossmann Beel, M.D., M.P.H.: Department of Anesthesiology, Perioperative and Pain Medicine, Baylor College of Medicine/Texas Children’s Hospital, Houston, Texas. Served as a scientific advisor and collected data.
Rahil Rugnathx, M.D.: Department of Anesthesiology, University of Mississippi Medical Center, Jackson, Mississippi. Served as a scientific advisor and collected data.
Robert E. Shaw, M.D.: Department of Anesthesia, University of Wisconsin-Madison, Madison, Wisconsin. Served as a scientific advisor and collected data.
Michelle M. Sheth, M.D.: Department of Anesthesiology, University of Mississippi Medical Center, Jackson, Mississippi. Served as a scientific advisor and collected data.
Tripiti Sinha, M.D.: Department of Anesthesia, University of Wisconsin-Madison, Madison, Wisconsin. Served as a scientific advisor and collected data.
Aine Sommerfield, B.Sc., Ph.D., M.Sc.: Department of Anaesthesia & Pain Management, Perth Children’s Hospital, Perth, Western Australia, Australia: Perioperative Medicine Team, Telethon Kids Institute, Perth, Western Australia, Australia. Served as a scientific advisor and collected data.
Codruta Soneru, M.D.: Department of Anesthesiology & Critical Care Medicine, University of New Mexico, Albuquerque, New Mexico. Served as a scientific advisor and collected data.
Thomas W. Templeton, M.D.: Department of Anesthesiology, Wake Forest University School of Medicine, Winston-Salem, North Carolina. Served as a scientific advisor and collected data.
R.J. Williams, M.A.: Department of Anesthesia and Pain Medicine, Hospital for Sick Children, Toronto, Ontario, Canada. Served as a scientific advisor and collected data.
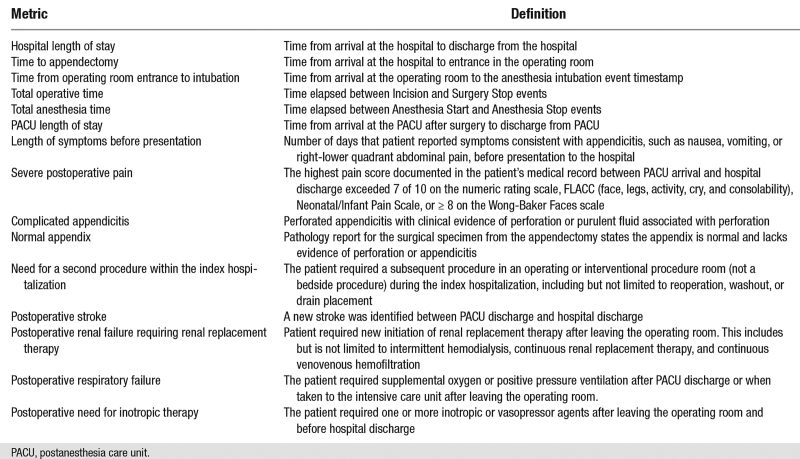
Appendix 2. Study Metrics and Outcome Definitions
Footnotes
Members of the PEdiatric Anesthesia COVID-19 Collaborative group are listed in appendix 1.
This article is featured in “This Month in Anesthesiology,” page A1.
Supplemental Digital Content is available for this article. Direct URL citations appear in the printed text and are available in both the HTML and PDF versions of this article. Links to the digital files are provided in the HTML text of this article on the Journal’s Web site (www.anesthesiology.org).
This work was presented in part at the Canadian Anesthesiologist Society Annual Conference, Halifax, Nova Scotia, June 26, 2022; the Child and Adolescent Health Service Child Health Symposium, Perth, Australia, November 3, 2021; and the Royal Australian College of Surgeons Annual Scientific Conference, Brisbane, Australia, May 4, 2022.
Published online first on April 4, 2023.
Contributor Information
Clyde T. Matava, Email: clyde.matava@sickkids.ca.
Anita Akbar Ali, Division of Pediatric Anesthesiology and Pain Medicine, University of Arkansas for Medical Sciences/Arkansas Children’s Hospital Served as a scientific advisor and collected data.
Mohamad F. Al-Rabbat, Department of Anesthesia, Sidra Medicine. Served as a scientific advisor and collected data.
Alyssa B. Brzenski, Department of Anesthesiology, University of California - San Diego Served as a scientific advisor and collected data.
William F. Casey, Department of Anesthesia, Sidra Medicine Served as a scientific advisor and collected data.
Surendrasingh Chhabada, Department of Pediatric Anesthesiology, Cleveland Clinic Served as a scientific advisor and collected data.
Michael Collin, Department of Paediatric Surgery, Perth Children’s Hospital Served as a scientific advisor and collected data.
Vipul J. Dhumak, Department of Anesthesiology, West Virginia University Medicine Children’s Served as a scientific advisor and collected data.
Ajay D’Mello, Department of Anesthesia and Pain Medicine, Nationwide Children’s Hospital Served as a scientific advisor and collected data.
Piedad C. Echeverry, Department of Anesthesia and Pain Medicine, Nationwide Children’s Hospital Served as a scientific advisor and collected data.
Pavithra R. Ellison, Department of Anesthesiology, West Virginia University Medicine Children’s
Allison M. Fernandez, Department of Anesthesia, Pain and Perioperative Medicine, Johns Hopkins All Children’s Hospital
Jake A. Fisher, Department of Anesthesiology and Pain Medicine, University of California – Davis Served as a scientific advisor and collected data.
Clinton L. Fuller, Department of Anesthesiology, Perioperative and Pain Medicine, Baylor College of Medicine/Texas Children’s Hospital Served as a scientific advisor and collected data.
Chris D. Glover, Department of Anesthesiology, Perioperative and Pain Medicine, Baylor College of Medicine/Texas Children’s Hospital Served as a scientific advisor and collected data.
Velu Guruswamy, Department of Pediatric Anesthesia, Sidra Medicine Served as a scientific advisor and collected data.
Emily B. Hesselink, Department of Anesthesia, University of Wisconsin-Madison Served as a scientific advisor and collected data.
Agnes I. Hunyady, Department of Anesthesiology and Pain Medicine, Seattle Children’s Hospital Served as a scientific advisor and collected data.
Amanda N. Lorinc, Department of Pediatric Anesthesiology, Lurie Children’s Hospital, Northwestern University, Feinberg School of Medicine Served as a scientific advisor and collected data
Michael King, Department of Pediatric Anesthesiology, Lurie Children’s Hospital, Northwestern University, Feinberg School of Medicine
Lavinia Mihaila, Department of Pediatric Anesthesiology, University of California – Davis Served as a scientific advisor and collected data
Jonathon H. Nelson, Division of Anesthesiology and Pain Medicine, Children’s National Hospital Served as a scientific advisor and collected data
Ann S. Ng, Department of Anesthesiology, Perioperative and Pain Medicine, Baylor College of Medicine/Texas Children’s Hospital
Joshua K. Ramjist, Department of General Surgery, The Hospital for Sick Children Served as a scientific advisor and collected data
Nirop R. Ravula, Department of Anesthesiology and Pain Medicine, University of California – Davis Served as a scientific advisor and collected data
Elizabeth Rossmann Beel, Department of Anesthesiology, Perioperative and Pain Medicine, Baylor College of Medicine/Texas Children’s Hospital Served as a scientific advisor and collected data
Rahil Rugnathx, Department of Anesthesiology, University of Mississippi Medical Center Served as a scientific advisor and collected data
Robert E. Shaw, Department of Anesthesia, University of Wisconsin-Madison Served as a scientific advisor and collected data
Michelle M. Sheth, Department of Anesthesiology, University of Mississippi Medical Center Served as a scientific advisor and collected data
Tripiti Sinha, Department of Anesthesia, University of Wisconsin-Madison Served as a scientific advisor and collected data
Aine Sommerfield, Department of Anaesthesia & Pain Management, Perth Children’s Hospital Served as a scientific advisor and collected data
Codruta Soneru, Department of Anesthesiology & Critical Care Medicine, University of New Mexico Served as a scientific advisor and collected data
Thomas W. Templeton, Department of Anesthesiology, Wake Forest University School of Medicine Served as a scientific advisor and collected data
R.J. Williams, Department of Anesthesia and Pain Medicine, Hospital for Sick Children Served as a scientific advisor and collected data
Collaborators: Anita Akbar Ali, Mohamad F. Al-Rabbat, Alyssa B. Brzenski, William F. Casey, Surendrasingh Chhabada, Michael Collin, Vipul J. Dhumak, Ajay D’Mello, Piedad C. Echeverry, Pavithra R. Ellison, Allison M. Fernandez, Jake A. Fisher, Clinton L. Fuller, Chris D. Glover, Velu Guruswamy, Emily B. Hesselink, Agnes I. Hunyady, Amanda N. Lorinc, Michael King, Lavinia Mihaila, Jonathon H. Nelson, Ann S. Ng, Joshua K. Ramjist, Nirop R. Ravula, Elizabeth Rossmann Beel, Rahil Rugnathx, Robert E. Shaw, Michelle M. Sheth, Tripiti Sinha, Aine Sommerfield, Codruta Soneru, Thomas W. Templeton, and R.J. Williams
References
- 1.Schuchat A; CDC COVID-19 Response Team: Public health response to the initiation and spread of pandemic COVID-19 in the United States, February 24-April 21, 2020. MMWR Morb Mortal Wkly Rep. 2020; 69:551–6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Matava CT, Kovatsis PG, Lee JK, Castro P, Denning S, Yu J, Park R, Lockman JL, Von Ungern-Sternberg B, Sabato S, Lee LK, Ayad I, Mireles S, Lardner D, Whyte S, Szolnoki J, Jagannathan N, Thompson N, Stein ML, Dalesio N, Greenberg R, McCloskey J, Peyton J, Evans F, Haydar B, Reynolds P, Chiao F, Taicher B, Templeton T, Bhalla T, Raman VT, Garcia-Marcinkiewicz A, Gálvez J, Tan J, Rehman M, Crockett C, Olomu P, Szmuk P, Glover C, Matuszczak M, Galvez I, Hunyady A, Polaner D, Gooden C, Hsu G, Gumaney H, Pérez-Pradilla C, Kiss EE, Theroux MC, Lau J, Asaf S, Ingelmo P, Engelhardt T, Hervías M, Greenwood E, Javia L, Disma N, Yaster M, Fiadjoe JE, PeDI Collaborative: Pediatric airway management in COVID-19 patients: Consensus guidelines from the Society for Pediatric Anesthesia’s Pediatric Difficult Intubation Collaborative and the Canadian Pediatric Anesthesia Society. Anesth Analg. 2020; 131: 61–73 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Peeling RW, Olliaro PL, Boeras DI, Fongwen N: Scaling up COVID-19 rapid antigen tests: Promises and challenges. Lancet Infect Dis. 2021; 21:e290–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.de la Matta M, Delgado-Sánchez JM, Gutiérrez GM, López Romero JL, Martínez Gómez MM, Domínguez Blanco A: Utility of preoperative polymerase chain reaction testing during SARS-CoV-2 pandemic: The challenge of evolving incidence. Rev Esp Anestesiol Reanim. 2021; 68:346–52 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Dibbs RP, Ferry AM, Enochs J, Ward A, Glover CD, Archer N, Taylor KD, Almaguer-Bravo J, Hollier LH, Jr.: The use of personal protective equipment during the COVID-19 pandemic in a tertiary pediatric hospital. J Healthc Risk Manag. 2021; 40:38–44 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Soneru CN, Fernandez AM, Bradford V, Staffa SJ, Raman VT, Cravero J, Zurakowski D, Meier PM; Pediatric Anesthesia COVID-19 Collaborative: A survey of the global impact of COVID-19 on the practice of pediatric anesthesia: A study from the pediatric anesthesia COVID-19 Collaborative Group. Pediatr Anesth. 2021; 31:720–9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Castagnoli R, Votto M, Licari A, Brambilla I, Bruno R, Perlini S, Rovida F, Baldanti F, Marseglia GL: Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection in children and adolescents: A systematic review. JAMA Pediatr. 2020; 174:882–9 [DOI] [PubMed] [Google Scholar]
- 8.Drake-Brockman TFE, Chambers NA, Sommerfield D, von Ungern-Sternberg BS: The impact of surgical cancellations on children, families, and the health system in an Australian paediatric tertiary referral hospital. Paediatr Anaesth. 2021; 31:578–86 [DOI] [PubMed] [Google Scholar]
- 9.Ingram M-CE, Raval MV, Newton C, Lopez ME, Berman L: Characterization of initial North American pediatric surgical response to the COVID-19 pandemic. J Pediatr Surg. 2020; 55:1431–5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Matava C, So J, Williams RJ, Kelley S, ORRACLE-Xtra Group: A Canadian weekend elective pediatric surgery program to reduce COVID-19-related backlog: Operating room ramp-up after COVID-19 lockdown ends - extra lists (ORRACLE-Xtra) implementation study. JMIR Perioper Med. 2022; 5:35584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Stewart B, Khanduri P, McCord C, Ohene-Yeboah M, Uranues S, Vega Rivera F, Mock C: Global disease burden of conditions requiring emergency surgery. Br J Surg. 2013; 101:e9–e22 [DOI] [PubMed] [Google Scholar]
- 12.Kokoska ER, Bird TM, Robbins JM, Smith SD, Corsi JM, Campbell BT: Racial disparities in the management of pediatric appenciditis. J Surg Res. 2007; 137:83–8 [DOI] [PubMed] [Google Scholar]
- 13.Rothrock SG, Pagane J: Acute appendicitis in children: emergency department diagnosis and management. Ann Emerg Med. 2000; 36:39–51 [DOI] [PubMed] [Google Scholar]
- 14.Baxter KJ, Nguyen HTMH, Wulkan ML, Raval MV: Association of health care utilization with rates of perforated appendicitis in children 18 years or younger. JAMA Surg. 2018; 153:544–50 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Rozner MA: The American Society of Anesthesiologists physical status score and risk of perioperative infection. JAMA. 1996; 275:1544. [PubMed] [Google Scholar]
- 16.Austin PC: An introduction to propensity score methods for reducing the effects of confounding in observational studies. Multivariate Behav Res. 2011; 46:399–424 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JG: Research electronic data capture (REDCap)—A metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009; 42:377–81 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Raval MV, Pawlik TM: Practical guide to surgical data sets: National Surgical Quality Improvement Program (NSQIP) and pediatric NSQIP. JAMA Surg. 2018; 153:764–5 [DOI] [PubMed] [Google Scholar]
- 19.Staffa SJ, Kohane DS, Zurakowski D: Quantile regression and its applications: A primer for anesthesiologists. Anesth Analg. 2019; 128:820–30 [DOI] [PubMed] [Google Scholar]
- 20.Armstrong RA: When to use the Bonferroni correction. Ophthalmic Physiol Opt. 2014; 34:502–8 [DOI] [PubMed] [Google Scholar]
- 21.von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP, Initiative S: The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: Guidelines for reporting observational studies. Bull World Health Organ. 2007; 85:867–72 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Matava CT, Williams RJ, Simpao AF: An open-source toolkit to assist authors and collaborators during manuscript preparation: AuthorAndCollaborator toolkit. Can J Anaesth. 2022; 69:680–1 [DOI] [PubMed] [Google Scholar]
- 23.Alvarez Garcia N, Nunez Garcia B, Perez-Gaspar M, Jimenez Gomez J, Betancourth Alvarenga J, Santiago Martinez S, Jimenez Arribas P, Esteva Miro C, Guizzo J: Immediate impact of the COVID-19 pandemic on pediatric surgery: Analysis of a tertiary healthcare facility. Cir Pediatr. 2021; 34:34–8 [PubMed] [Google Scholar]
- 24.Chidini G, Villa C, Calderini E, Marchisio P, De Luca D: SARS-CoV-2 infection in a pediatric department in Milan: A logistic rather than a clinical emergency. Pediatr Infect Dis J. 2020; 39:e79–e80 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Czeisler M, Marynak K, Clarke KEN, Salah Z, Shakya I, Thierry JM, Ali N, McMillan H, Wiley JF, Weaver MD, Czeisler CA, Rajaratnam SMW, Howard ME: Delay or avoidance of medical care because of COVID-19-related concerns - United States, June 2020. MMWR Morb Mortal Wkly Rep. 2020; 69:1250–7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Dedeilia A, Esagian SM, Ziogas IA, Giannis D, Katsaros I, Tsoulfas G: Pediatric surgery during the COVID-19 pandemic. World J Clin Pediatr. 2020; 9:7–16 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Gunadi , Idham Y, Paramita VMW, Fauzi AR, Dwihantoro A, Makhmudi A: The impact of COVID-19 pandemic on pediatric surgery practice: A cross-sectional study. Ann Med Surg (Lond). 2020; 59:96–100 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kurihara H, Marrano E, Ceolin M, Chiara O, Faccincani R, Bisagni P, Fattori L, Zago M; Lombardy Emergency Surgery Group during Covid19 outbreak: Impact of lockdown on emergency general surgery during first 2020 COVID-19 outbreak. Eur J Trauma Emerg Surg. 2021; 47:677–82 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ahluwalia R, Rocque BG, Shannon CN, Blount JP: The impact of imposed delay in elective pediatric neurosurgery: An informed hierarchy of need in the time of mass casualty crisis. Childs Nerv Syst. 2020; 36:1347–55 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Sehulster L, Chinn RY. Guidelines for environmental infection control in health-care facilities. Recommendations of CDC and the Healthcare Infection Control Practices Advisory Committee (HICPAC). MMWR Recomm Rep. 2003; 52:1–42 [PubMed] [Google Scholar]
- 31.Reichert M, Sartelli M, Weigand MA, Doppstadt C, Hecker M, Reinisch-Liese A, Bender F, Askevold I, Padberg W, Coccolini F, Catena F, Hecker A; WSES COVID-19 emergency surgery survey collaboration group: Impact of the SARS-CoV-2 pandemic on emergency surgery services–A multi-national survey among WSES members. World J Emerg Surg. 2020; 15:64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Truche P, Bowder A, Lalla AT, Crum R, Botelho F, Rice HE, Lopes BC, Greenberg S, Evans F, Meara JG, Ameh EA, Mooney DP: Perspectives on perioperative management of children’s surgical conditions during the COVID-19 pandemic in low-income and middle-income countries: a global survey. World J Pediatr Surg. 2020; 3:e000187. [DOI] [PMC free article] [PubMed] [Google Scholar]