Abstract
Investigations of hippocampal functions have revealed a dizzying array of findings, from lesion-based behavioral deficits, to a diverse range of characterized neural activations, to computational models of putative functionality. Across these findings, there remains an ongoing debate about the core function of the hippocampus and the generality of its representation. Researchers have debated whether the hippocampus’s primary role relates to the representation of space, the neural basis of (episodic) memory, or some more-general computation that generalizes across various cognitive domains. Within these different perspectives, there is much debate about the nature of feature encodings. Here, we suggest that in order to evaluate hippocampal responses – investigating, for example, whether neuronal representations are narrowly targeted to particular tasks or if they subserve domain-general purposes – a promising research strategy may be the use of multi-task experiments, or more generally switching between multiple task contexts while recording from the same neurons in a given session. We argue that this strategy – when combined with explicitly defined theoretical motivations that guide experiment design – could be a fruitful approach to better understand how hippocampal representations support different behaviors. In doing so, we briefly review key open questions in the field, as exemplified by articles in this special issue, as well as previous work using multi-task experiments, and extrapolate to consider how this strategy could be further applied to probe fundamental questions about hippocampal function.
Keywords: multi-task experiments, hippocampus, neural representation, cognitive map, spatial navigation, place cells, concept cells
Introduction
A multitude of investigations have explored the functional role(s) of the hippocampus, with results from across domains and modalities finding that responses in the hippocampus and surrounding brain structures can represent many different aspects of behavior that seemingly extend beyond one specific cognitive domain, as demonstrated by many of the works within this special issue. Even within cognitive domains – with episodic memory and spatial navigation being the most typical associated functions – multiple studies with comparable task designs aimed at detecting similar hippocampal responses often find somewhat divergent results. These myriad responses – that vary across task domains and often have seemingly discrepant results even within similar contexts – challenge our attempts at characterizing the overall function(s) of this clearly important brain area. These seeming inconsistencies imply that the hippocampus likely employs various encoding mechanism(s), with flexible, task-dependent activity, requiring us to take variability across tasks as a serious consideration to be understood and included in our explanations. The alternative is to develop theories and hypotheses that are predicated on a sub-sampling of hippocampal functions, which are likely doomed to fall apart as soon as the task changes.
In this piece, we argue that this apparent variability across domains and tasks need not only be a troublesome caveat lamented in discussion sections and review papers, but can instead be leveraged as an empirical strategy to explicitly investigate the nature of hippocampal neural representations and whether they distinguish or generalize across different contexts. Specifically, we explore the use of ‘multi-task’ experiments – meaning single experimental sessions in which multiple tasks are administered such that the responses of the same neural circuits can be examined and compared across different behavioral tasks (Figure 1). This design allows for directly asking questions about if and how neural representations in the hippocampus keep or change their tuning under different cognitive demands. While this approach does exist in the literature, we contend that it is an under-utilized strategy, in particular for the hippocampus for which there are debates around describing adequate theoretical framework(s) that address how this structure is involved in the broad range of tasks it responds to, and what function(s) it is fulfilling.
Figure 1 -. Schematic of possible designs for multi-task experiments.
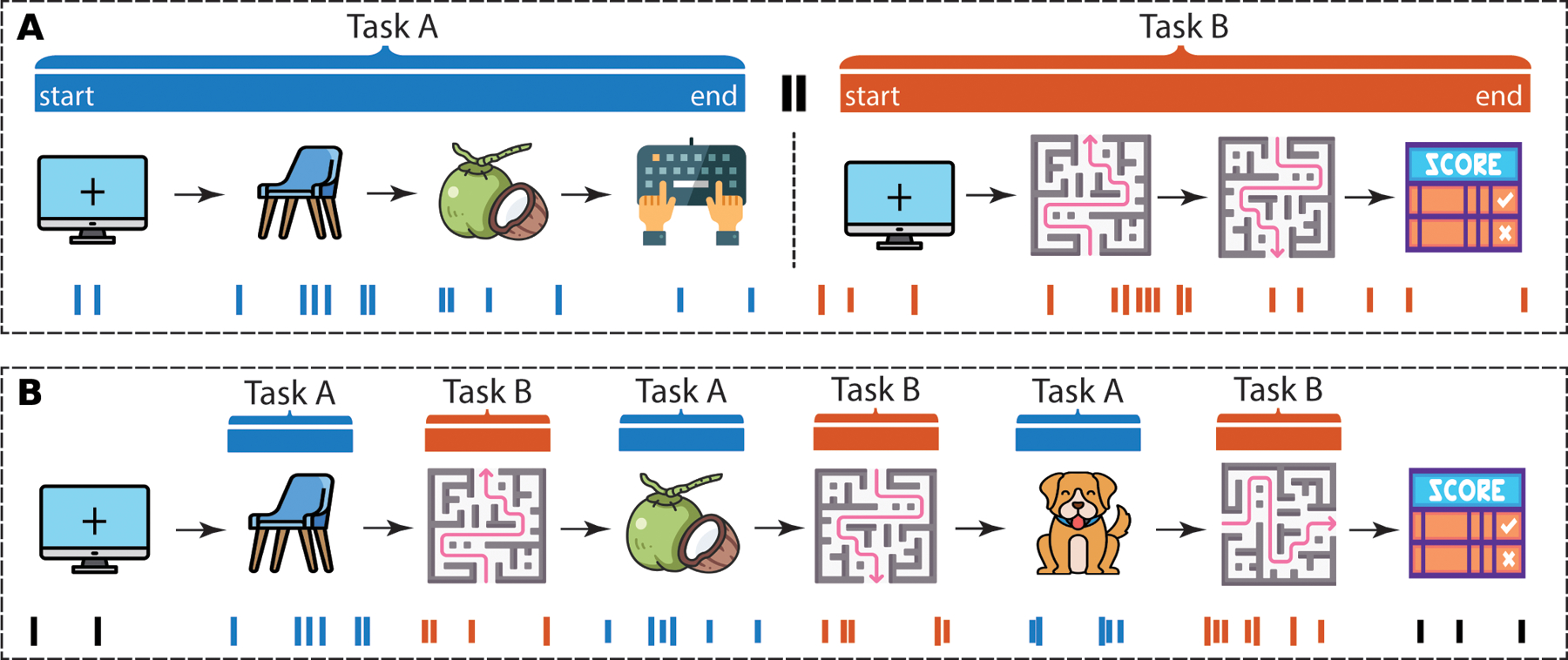
Examples show a multi-task experiment represented by a memory task (task A) and a spatial navigation task (task B) that are either administered back-to-back (A) or interleaved (B). A) Example layout of a back-to-back multi-task experiment in which subjects first complete a memory-related task, for example, viewing and responding to images of stimuli (Task A; blue). After completion of this task, subjects then immediately switch to another task, for example a space-related task, for example, navigating in a virtual/real-world environment, which could include navigating to specific locations, encountering stimuli, responding to task prompts, and receiving a reward / score based on performance accuracy (Task B; orange). The bottom raster plot indicates spikes (simulated) representing the activity on an example neuron recorded across the experiment throughout both tasks in the example session, color-coded to reflect the task to which each spike can be associated. B) Example layout of a multi-task design using the same example tasks, but in an interleaved fashion such that task A and B alternate on a block- or trial-level, with example neural activity color coded by task, with black representing non-task specific activity at the start and end of the multi-task experiment session. Note that the example tasks shown here are placeholders for any combination of task designs that can be combined. Icons from https://www.flaticon.com.
Here, we will first briefly describe an overview of what we see as major theoretical frameworks that are currently employed and discussed in relation to hippocampal function. This discussion outlines the different themes of current work demonstrating how hippocampal function varies across tasks, which we use both to discuss current research themes, and to structure topics that we believe can be productively examined using multi-task experiment designs. After this overview, we will explore examples of multi-task experiments, noting previous examples of using this strategy, as well as exploring open questions that we believe could be productively investigated through the use of multi-task designs, drawing in particular from examples from across the current special issue on view-related responses in the hippocampus. For simplicity and convenience, this paper will focus on studies analyzing single-neuron responses, though many if not all of the key points raised likely generalize to other recording modalities such as optical imaging, intracranial EEG, and/or noninvasive neuroimaging. Finally, we outline a set of recommendations that may be useful to consider when pursuing multi-task experiments.
Theoretical Frameworks of Hippocampal Function
Historically, studies on the functional role of the hippocampus have often been oriented around an underlying theoretical framework in which the hippocampus and surrounding areas reflect a dedicated system for a specific cognitive domain, albeit not with a consistent agreement across studies on what this cognitive domain is. Broadly, these frameworks can be differentiated based on whether they posit that the hippocampus has its primary role in implementing (episodic) memory, and/or in representing space. More recently, theoretical work has also posited that the hippocampus may be best described as performing a more-general relational computation that generalizes across cognitive domains. Here, we present a brief overview of each of these frameworks that suggest different primary roles for the hippocampus, introducing the key positions of the approach.
Based on the foundational work in neuropsychology of Dr. Brenda Milner and colleagues (Milner et al., 1998; Scoville & Milner, 1957), one line of work has traditionally associated the hippocampus and surrounding areas with episodic memory. Behavioral evidence for this association comes from patients who after having surgical removal of their hippocampi, were impaired in forming new episodic memories. Subsequent neuroscientific work has examined the hippocampus as a core part of a neural system dedicated to forming neural representations of episodic events that can be formed, stored, and recalled (Frankland et al., 2019; Josselyn & Tonegawa, 2020). Based on work largely done in rodents, the current view is that the hippocampus and other nearby structures contain available neurons that can be flexibly allocated to represent memory substrates and their associations, with flexible memory allocation based on task context (Josselyn & Frankland, 2018). Relatedly, in humans, single neurons in the hippocampus and surrounding areas have been found that are visually selective to faces (Heit et al., 1988; Kreiman et al., 2000), with subsequent work showing highly selective and invariant ‘concept cells’ which respond to individual identities and objects (Ison et al., 2015; Quian Quiroga, 2012; Quian Quiroga et al., 2005). Collectively, this work establishes that hippocampal circuits represent conceptual entities and associations that are thought to contribute to forming episodic memories (Quian Quiroga, 2012).
Separately, often concurrent work in behavioral neuroscience has focused on the hippocampus as being critically engaged in spatial navigation. Foundational work in this area stems from the observation of place cells, which activate whenever the navigating animal is at a particular location in space (Moser et al., 2017; O’Keefe & Dostrovsky, 1971). Subsequent research has demonstrated a veritable cornucopia of spatially-tuned responses across the medial temporal lobe (Alexander et al., 2023; Bicanski & Burgess, 2020; Moser et al., 2017; Rolls & Wirth, 2018; Zhu et al., 2023). Within the spatial domain, several lines of work have illustrated ways in which hippocampal responses are task-dependent, including remapping across different environments (Kubie et al., 2020; Kubie & Muller, 1991), attention-like modulation of place cell activity based on the availability of spatial cues (Fenton et al., 2010), task demands that can impact spatial encoding (Duvelle et al., 2023; Kobayashi et al., 1997), and experience-dependent changes in neural responses (Moore et al., 2021). Collectively, this work has established that there are ubiquitous spatial representations across the hippocampus and related areas, though with a wide variety of which spatial features they represent, and how this varies across tasks.
Despite many attempts to reconcile the role of the hippocampus in both episodic memory and spatial navigation, investigations into the mechanistic contributions of the hippocampus to episodic memory and spatial navigation remain as largely distinct lines of research. Some current theoretical formulations have suggested that the hippocampus is primarily involved in one key task, and this can be used to explain responses to other features. Some researchers support the perspective that the hippocampus is primarily an episodic-memory system, with representations such as place-cells being explained as memory responses (Benna & Fusi, 2021; Eichenbaum, 2017a). Alternatively, following the tradition of considering the hippocampus as foundationally about space, other researchers emphasize the primacy of spatially-modulated neurons in the hippocampus, and suggest that all other responses can be considered conjunctive annotations of objects or experiences that occur in space (O’Keefe & Krupic, 2021; O’Keefe & Nadel, 1978), or the similar idea that spatial navigation is the fundamental representation upon which neural representations related to episodic memory are scaffolded (Buzsáki & Moser, 2013). Alternatively, other perspectives state (or imply) that these different memory and navigation functions co-exist in the same regions in at least somewhat distinct circuits, suggesting the hippocampus may reflect a combination of multiple overlapping and interacting systems (Sugar & Moser, 2019).
As research continued in these two streams – on hippocampal-dependent memory and navigation – alternate theoretical formulations have arisen. For example, the ‘generalized cognitive map’ perspective considers that the hippocampus has a more general task of representing multidimensional feature spaces – be that the physical space of navigation or various kinds of cognitive entities (such as objects or faces) in feature space, which also gets used to encode the spatio-temporal context of episodic memories (Behrens et al., 2018; Bellmund et al., 2018; Eichenbaum & Cohen, 2014; Schiller et al., 2015). An example consistent with this perspective is the finding of hippocampal neurons that represent specific frequencies in a sound modulation task (Aronov et al., 2017). In this perspective, neither space nor episodic memory are the primary functions per se, with the proposal that both kinds of representations can be redescribed in terms of a relational memory system (Whittington et al., 2020). This framework therefore proposes an overall more general / flexible description of the functionality of the hippocampus and surrounding areas that is potentially able to encompass the variable responses of the hippocampus across different task contexts and cognitive domains.
Altogether, these different theoretical perspectives leave us with several categories, roughly construed, of theoretical priors the literature offers, with several differentiating features. One could consider the hippocampus as being a specialized system for a particular domain (either for episodic memory or spatial navigation), a collection of multiple co-embedded but distinct systems, or a more generalized but unitary system that gets applied across multiple domains. For clarity, while we do believe that adjudicating between these different frameworks is an important question, we emphasize that our goal here is not to rigorously define and contrast the relative merits of each approach or to adjudicate between them (with much other literature dedicated to these points – see (Buzsáki & Moser, 2013; Eichenbaum, 2017b; Eichenbaum & Cohen, 2014; Ekstrom & Ranganath, 2018; Epstein et al., 2017; Hasselmo, 2012; Lisman et al., 2017; Schiller et al., 2015). Rather, the goal here is to introduce a useful abstraction to discuss different theoretical backgrounds on hippocampal function that, while not always explicitly introduced, often color and constrain the tasks, analyses, and interpretations that are chosen. Here, we suggest that a productive strategy for making headway on longstanding questions about hippocampal function may be to first be explicit about these theoretical perspectives – detailing what predictions they make – and then testing these predictions using an empirical strategy that explicitly compares responses across different task contexts in multi-task experiments.
Examples of Multi-Task Research Designs
We now consider how using multi-task experiment designs can help to investigate questions of interest within and between these different theoretical frameworks. In the spatial domain, in rats, using multiple tasks in the same spatial area has been used to investigate the activity of place cells across different tasks, showing that while place cells maintain their representation across different navigation tasks (Trullier et al., 1999), changes in memory and/or attentional demands can induce remapping (Hallock & Griffin, 2013; Levy & Hasselmo, 2023; Markus et al., 1995). Similarly, by using two tasks in the same location, it has been shown that adding an associative memory task changes the representation of space in the monkey hippocampus (Gulli et al., 2020). Another study, in bats, has shown how alternating between solo-flights and cross-over flights with a conspecific induces changes whereby individual neurons can switch from representing position to representing distance-by-position conjunctions (Sarel et al., 2022). Collectively, these studies demonstrate that different behavioral contexts across tasks can induce changes to how the hippocampus encodes space and memory related representations. In the next examples we explore how other multi-task designs could help address other outstanding questions about the hippocampus, drawing from topics discussed across this special issue including local vs. remote encoding of space, allo- vs ego-centric representations, properties of memory-related representations, and theories that connect across the space and memory related functions of the hippocampus.
Example 1: Representations of Current / Self vs Remote / Other Position
Within the spatial domain, there are ongoing discussions regarding if and to what extent hippocampal neurons maintain the same kinds of spatial representations across contexts, and/or to what extent such representations can flexibly shift based on behavioral demands. Specifically, an open question is to explain when and why hippocampal responses respond to current / self or to remote / other locations. Early findings in rats showed ‘place cells’, which activate whenever the animal is in a particular location in space relative to the external environment (Moser et al., 2017; O’Keefe & Dostrovsky, 1971). However, in primate studies, hippocampal neurons can be modulated by the spatial views of the monkeys – where monkeys are looking ‘out there’ in space, regardless of their physical location (Rolls, 1999; Rolls & Wirth, 2018), as well as grid-like representations of visual space (Killian et al., 2012). Based on these differences across different animal models, it has been suggested that there may be species differences between the hippocampal representations of space (Rolls, 2023; Rolls & Wirth, 2018).
An additional, non-exclusive, explanation is that the spatial representation enacted by the hippocampus may vary depending on the behavioral task. Hippocampal neurons in monkeys do manifest place-cell-like representations in some spatial navigation and foraging tasks (Gulli et al., 2020; Ono et al., 1993), suggesting that both place and spatial-view responses can occur in the primate hippocampus. Similar discrepancies of place-related hippocampal activity have also been found in humans. Several studies have found neural responses akin to the firing of place cells in rodents using similar tasks where subjects were instructed to navigate in a virtual arena and in which subjects have to learn to use their own location in conjunction with knowledge of a partially occluded arena in order to succeed at the task (Ekstrom et al., 2003; Miller et al., 2013). However, in other experiments in which subjects have to navigate to remote locations that are always in view, spatial target cells – cells that are significantly modulated by the target locations that subjects are navigating towards – were found instead of place cells, which were observed to a much lesser extent (Donoghue et al., 2023; Tsitsiklis et al., 2020). Collectively, a potential explanation of these findings is that the difference in encoding is related to the task demands, suggesting that hippocampal neurons are spatially-modulated, but the specific nature of this spatial coding (i.e., being directed at one’s current location or on a remote / target location) can shift in a task-dependent manner, being driven by task demands that require or emphasize either self or remote locations in order to complete the task.
In order to test the potential task-dependent nature of neural responses in the hippocampus, a multi-task paradigm could be useful to investigate how and to what extent hippocampal neurons quickly adapt to encode current or remote locations. For example, a multi-task experimental design could be used that combines tasks where either one’s current or future locations are most relevant to completing the tasks, while also recording eye-tracking data (in order to disassociate between the location and view responses). Examining across these multiple navigation tasks could help elucidate i) if hippocampal neurons encode current position (place cells) in the task that focuses on the subject’s current location and encode remote location representations (spatial target cells) in the task that requires subjects to focus more strongly on remote locations, ii) if some task variants induce both representations to co-occur and iii) if individual neurons can switch from representing one’s current position to representing remote locations, or whether distinct neural populations encode the different features.
Another related distinction in hippocampal encoding of spatial variables relates to the encoding of self- vs other-locations. Experiments in both rodents and bats have revealed the existence of ‘social place cells,’ which can reflect the spatial location of both the self or another conspecific in experiments in which animals were required to observe or mimic the other rodent/bat’s trajectory in a shared environment (Danjo et al., 2018; Omer et al., 2018). An interesting prediction is therefore of ‘social target cells’, which could be investigated using a variant of a spatial navigation task tailored for target cells, where subjects observe the experimenter or another subject performing the task prior to their own completion or vice versa, with task switches between other- and self-performance (Stangl et al., 2020). Advances in virtual reality systems that are becoming more and more available in research settings also open up exciting possibilities to perform multi-task experiments by allowing rapid switches between different task and environment settings. This enables researchers to more fully characterize hippocampal coding of different spatial variables, often in combination with more complex and naturalistic environments, as further discussed elsewhere in this special issue (S. Lee et al., 2023).
Example 2: Comparing Allocentric and Egocentric Reference Frames
Parallel to the distinction of whether the hippocampus mainly represents one’s current location or remote locations, another key open question is whether the hippocampus and surrounding medial temporal lobe regions represent spatial information in allocentric or egocentric coordinates, or both (Alexander et al., 2020, 2023; Kunz et al., 2021; LaChance et al., 2019; LaChance & Taube, 2023; C. Wang et al., 2018, 2023). Allocentric neural codes represent spatial locations and directions relative to the external environment, whereas egocentric representations encode them relative to the navigating subject (Bicanski & Burgess, 2018, 2020; Burgess, 2006). Place cells are a hallmark of allocentric encoding of space because they activate whenever the animal is in a particular location in space relative to the external environment, independent of the orientation when moving through the place field (Barry et al., 2006; Muller & Kubie, 1989). Recently, however, research has shown that neurons in the hippocampus can also represent space in egocentric coordinates. For instance, hippocampal neurons in the hippocampus of mice, rats, and bats have been shown to represent egocentric directions and distances to spatial goals or reference points (Jercog et al., 2019; Ormond & O’Keefe, 2022; Sarel et al., 2017). Overall, it remains an open and important question to understand how space is represented in different coordinate frames, as well as understanding the transformations that may give rise to the allocentric nature of place cells (LaChance & Taube, 2022; C. Wang et al., 2018, 2020).
In this special issue, several studies have continued the quest of understanding allocentric versus egocentric representation in the hippocampus and surrounding areas. In a study of monkeys performing a context-object association task in a virtual environment, a considerable number of view cells were found in the hippocampus that activated whenever the monkey viewed a left or right side corridor at either end of a central corridor, thus potentially representing particular views in an egocentric rather than an allocentric coordinate framework (Corrigan et al., 2023). In another study, egocentric tuning was found in deep layers of the lateral entorhinal cortex in rats (C. Wang et al., 2023), extending previous work demonstrating egocentric tuning in the superficial layers of the lateral entorhinal cortex (C. Wang et al., 2018) and in the retrosplenial cortex (Alexander et al., 2020). These findings suggest that egocentric tuning is prevalent throughout several parahippocampal areas, suggesting these regions may perform important computations for converting allocentric hippocampal representations into an egocentric form (Alexander et al., 2023). Such egocentric representations seem to be, amongst others, useful for goal-directed navigation, for example by representing egocentric bearings and distances toward the geometric center and ‘anchor points’ of an environment (LaChance & Taube, 2023).
In order to further explore how the hippocampal formation uses allocentric and/or egocentric representations, we suggest that multi-task experiments might also constitute a powerful approach to better understand under which conditions neurons in the hippocampus and surrounding medial temporal lobe regions represent space mainly in allocentric or egocentric coordinates. For example, consecutive task sessions or alternating trials could be used in a hybrid task that requires subjects to alternately remember spatial information from either an allocentric or from an egocentric perspective. Various tasks have been developed that test allocentric and egocentric representations separately – for example, the Morris Water Maze task and adapted variants for allocentric representations (Vorhees & Williams, 2006), and the path integration tasks for egocentric representations (McNaughton et al., 2006).
Combining these pre-existing tasks into multi-task experiments could reveal whether individual neurons in the hippocampal formation show (i) the same responses in both tasks and thus exhibit a particular type of task-invariant spatial tuning; (ii) allocentric spatial tuning in allocentric tasks but egocentric spatial tuning in egocentric tasks, thus generally representing space but adapting their firing in response to the spatial information that is most important for the task at hand; and/or (iii) allocentric spatial tuning in the allocentric task but no tuning in the egocentric task and vice versa. We suggest that performing these different tasks back-to-back could resolve fundamental questions regarding the spatial tuning of neurons in the hippocampus and surrounding medial temporal regions. More specifically, they could help us understand whether spatial coding in the medial temporal lobes is invariant or flexible, whether allocentric and egocentric spatial tuning is encoded in different or overlapping neuronal populations, and whether behavioral requirements alter the prevalence of allocentric versus egocentric tuning.
Example 3: Comparing Visual Pattern Separation vs Invariant Concept Encoding
For the next example, we consider work primarily in the episodic/semantic-memory framework of hippocampal function, and in particular, how individual neurons respond to images of faces and objects. In this area of research, there is an ongoing debate over whether hippocampal neurons rely on visual pattern separation or concept association to recognize objects and form new declarative memories (Quian Quiroga, 2020), as is also discussed in the current special issue (Quian Quiroga, 2023). The concept model is supported by human single-neuron studies showing that hippocampal neurons respond highly selectively to specific persons or objects, regardless of visual appearances or stimulus modalities (Quian Quiroga, 2012; Quian Quiroga et al., 2005, 2008). It has also been shown that concept neurons encode multiple identities based on conceptual associations, personal experience, and memory (De Falco et al., 2016; Rey et al., 2018, 2020), and that these neurons can expand their selectivity through learned associations of new concepts (Ison et al., 2015). Based on these findings, it has been argued that there is no pattern separation in the human hippocampus; instead, individual concepts are represented by specific and invariant engrams and the association between related concepts is encoded via partially overlapping assemblies (Quian Quiroga, 2020).
On the contrary, a large literature on animal neurophysiology and human neuroimaging suggests visual pattern separation, which proposes that a key principle of hippocampal function is to transform similar representations or memories into dissimilar and non-overlapping representations (Leal & Yassa, 2018). Human studies using high-resolution functional neuroimaging support this model by showing that neural activity in subregions of the hippocampus is subject to adaptation effects only upon repeated display of identical objects, rather than visually changed objects, supporting pattern separation in the human hippocampus and dentate gyrus (Bakker et al., 2008). Related work in this special issue explores computational constraints on associative memory built on associated, sparse concepts (Ryom et al., 2023). Collectively, this work is consistent with the hippocampus having pattern separation.
Most studies on pattern separation focus on comparing neural response to changes in stimulus features and representations. Therefore, understanding the coding principles of the hippocampus can provide valuable insights into pattern separation and reconcile the discrepancies among human single-neuron, human fMRI, and animal physiology findings. Specifically, a relevant distinction is whether the hippocampus uses feature-invariant concept coding which has been argued to not require pattern separation (Quian Quiroga, 2020), and/or feature-based coding of stimuli, whereby visually similar stimuli would be expected to be encoded similarly such that pattern separation mechanisms may be required. For example, as well as the aforementioned work on concept codes in humans (Quian Quiroga, 2012), evidence for feature-based encoding includes that primate hippocampal neurons are more responsive to face images than names and voices, and identity coding of the same individuals does not generalize across modalities (Sliwa et al., 2016), as well as findings of neurons that encode object colors and viewpoints (Gulli et al., 2020). As such, there is currently evidence consistent with both kinds of encoding in the primate hippocampus.
Recent studies in humans have also shown hippocampal neurons that encode faces based on visual similarity but not on concept associations, showing region-based feature coding (Cao et al., 2022; J. Wang et al., 2022). These studies may help to reconcile the discrepancy in pattern separation reported by previous studies (Leal & Yassa, 2018; Quian Quiroga, 2020, 2023). Specifically, the finding of region-based feature coding suggests that hippocampal neurons exhibit a ‘receptive field’ in high-dimensional feature space (analogous to cognitive maps and place cells), such that objects or identities encoded within the receptive field may show a unitized response and no pattern separation, while neurons can discriminate between objects or identities within and outside the receptive fields, showing pattern separation. These receptive fields are thought to exist in both visual feature space, whereby neurons that respond to specific features such as color, texture, and shape, and in concept space, in which neurons respond to abstract concepts or ideas, such as familiar faces or places. Computationally, the combination of receptive fields from different neurons can lead to a global pattern separation, potentially reconciling the evidence from human single-neuron studies (Quian Quiroga, 2020) and neuroimaging studies (Bakker et al., 2008). More broadly, discrepancies between the two models may be attributed to various experimental factors, including species differences, differences in stimulus selection and familiarity (using pre-screening for responsive stimuli or not), as well as task demands and behavioral contexts (passive viewing vs. memory tasks vs. visual navigation), all of which make it more challenging to distinguish between the proposed models.
In order to address these discrepancies, multi-task experiments with human subjects that incorporate and address the above factors may be crucial to adjudicate between these different theories. Through multi-task experiments, the engagement of feature-based vs concept encodings can be directly compared in different contexts and tasks while examining the same set of neurons. For example, a memory task and passive viewing can be applied consecutively in recording sessions using the same set of stimuli, including prescreened and novel stimuli, to examine how task context, and stimulus selection impacts neural representations. Such multi-task experiments with combined neural, behavioral, and eye-tracking could be used to examine whether both feature-based and invariant concept neurons co-occur in the human hippocampus, and if their activity is modulated by task context. For example, by combining a one-back task requiring same / different recognition, and an object/face recognition task, analyses for feature and concept neurons can be applied across tasks, comparing across task demands, novel vs familiar stimuli, as well as comparing across stimuli sets with differences in their pre-selection and/or with different feature patterns. Analyzing the data across these dimensions could help to examine (i) if invariant concept-cells can be identified across tasks and stimulus sets; (ii) if feature-coding cells can be identified across tasks and stimulus sets and if they are modulated by task context and/or by task-relevant features (for example, features that are useful for distinguishing novel or familiar items); and/or (iii) if individual neurons can shift between these different representations based on behavioral context or if these representations are enacted by distinct circuits.
Example 4: Comparing Different Theoretical Frameworks of Hippocampal Activity
The examples thus far have largely examined work within a single cognitive domain, notably spatial navigation or (episodic) memory, and as such largely reflect questions that are most salient when working within one of the aforementioned theoretical frameworks that considers the hippocampus as best described as engaged in a particular domain. Next, we turn our attention to example experiments that could be used to test these frameworks themselves, comparing them to alternatives such as a generalized cognitive map. To do so, we first revisit a key feature of the majority of neuroscientific experiments, which is that neural representations are typically only tested for a limited time across a limited behavioral repertoire. In so far as there are variations in task and behavior, they are typically measured within the same domain, for example, examining remapping across different spatial environments in a spatial context (Kubie et al., 2020; Kubie & Muller, 1991), and/or testing the invariance of concept neurons across different views or evaluations of the same entities (Ison et al., 2015; Quian Quiroga, 2012). What is much rarer to examine is the activity of the same neurons across different cognitive domains – for example comparing between spatial and non-spatial memory tasks – and yet this is precisely the kind of test that can be used to start to adjudicate between the different theoretical frameworks for hippocampal function. A spatial view of the hippocampus may posit, for example, that a place cell is always a place cell – it may be active in some environments and inactive in others, but there is often an implicit assumption that this cell should continue to carry out place-cell-like coding irrespective of the animal’s behavior – with an analogous argument to be made for the memory framework and concept cells.
This assumption – that cells maintain the same kind of representation across time and/or tasks – is rarely explicitly tested. It also need not be the case, as beyond the frameworks interpreting hippocampus as reflecting either space or episodic memory, there are also alternative frameworks that posit that hippocampus is best described by a general-purpose function that gets applied to different domains, functioning, for example, as a generalized cognitive map (Behrens et al., 2018; Whittington et al., 2020). Though there are differences across variations of approaches within this framework, individual theories within this framework make specific, testable predictions regarding how individual neurons should act across completely different behavioral paradigms. For example, a study that found that hippocampal neurons could represent ‘locations’ in frequency space also found that some individual neurons that act as frequency-place cells in the sound modulation task also act as physical-place cells in a separate foraging task (Aronov et al., 2017). This study raises the intriguing possibility that ‘cell identities’ such as ‘place cell’ may be contingent on the behavioral and environmental context of the recording session, and in another situation the same cell could appear to encode an entirely different type of behavioral information.
Testing such questions – for example, examining the consistency of neural representations across tasks and examining the capacity for individual neurons to switch representation – is particularly amenable to investigations using multi-task experiments. By specifying if and how the different frameworks make different predictions on how individual neurons should change or maintain representations across contexts, this provides a useful strategy for empirically testing key tenets of the different frameworks, in order to assist with adjudicating between them. If, for example, a place cell is expected to only respond to space, after identification, this can be tested across tasks, comparing responses to various hypotheses regarding how a neuron might change activity if it is acting inline with a generalized cognitive map. In our recent work, we have started to address this question by combining a spatial navigation task with a working-memory task, finding neurons that had task-specific and task-independent responses to relevant stimuli, as well as neurons that changed their representation across the two tasks – finding, for example, neurons that switch from representing stimuli in a working memory task, to representing serial position in a spatial navigation task – demonstrating a flexibility in the representations of hippocampal neurons (Donoghue et al., 2023).
Combining multi-task experiments with explicit testing across different frameworks allows for testing specific predictions for how representations change (or not) across tasks. For example, in a generalized cognitive map framework, whereby the hippocampus maps entities in dimensional space, a place cell in a spatial task may be predicted to represent an object in feature space in a different task context. For example, as discussed in the previous section, some cells that respond to faces and individual identities can be explained by representing locations in a ‘face-feature’ space (Cao et al., 2022). Using a multi-task experiment combining facial stimuli with a feature-based analysis with a spatial task with place- or target-cell analyses could be used to evaluate whether there are neurons acting as generalized ‘features-in-dimensional-space’ neurons. Alternatively, a prediction of the hippocampus operating as a generalized relational memory system is that such a system can abstract across structurally identical exemplars (Whittington et al., 2020), consistent with findings in primates that hippocampus neurons can abstract across spatial concepts, forming schema-like representations (Baraduc et al., 2019). This perspective offers testable hypotheses for multi-task experiments, for example, there could be overlap in the neurons that represent mentally traversing a family or phylogenetic tree as compared to a task navigating a physical maze with a series of branches.
Recommendations
Thus far, we have explored some of the key debates regarding the function of the hippocampus, and proposed that experiment designs that examine neural activity across different tasks are a useful strategy to help address them. In comparing how individual neurons respond across settings, it is important to emphasize that using multiple tasks or manipulating task contexts is not a panacea, but another tool in the toolkit. As with any other empirical strategy, multi-task experiments can be used to lesser or greater effect depending on the design. Here, we explore some themes and recommendations for such experiment designs, based in part on our own experience working with a multi-task experiment.
The first theme, as we have been exploring throughout, is to explicitly consider the theoretical underpinnings of the task(s) being employed (Levenstein et al., 2020). As discussed in the examples, the theoretical assumptions and ways in which a particular task constrains the behavior (and therefore, the neural representations) is often under-specified. This creates particular jeopardy for multi-task experiments, as combining disparate tasks without explicitly considering what assumptions these tasks may embody and what predictions they make about how neural representations may relate to each other across tasks has the risk of leading to combining tasks in ways whereby the results of the study are difficult to interpret. Overall, our recommendation is that the use of multi-task experiments is an available tool that has thus far been under-utilized, and could be productively employed to investigate and attempt to address some key outstanding questions regarding the function of the hippocampus – if it is combined with clearly defined predictions drawing from the theoretical frameworks that are to be tested.
As we briefly discussed in some of the examples, a key consideration for using multi-task experiments is to consider whether the apparent discrepancies seen across experiments relate to task-dependent or context-related responses, and/or to what extent some differences may reflect inter-species differences. Coupling, for example, allocentric and egocentric spatial navigation tasks or tasks emphasizing self vs remote locations is a good design for observing shifting neural representation – if these representations can and do shift within individuals. However, multi-task paradigms are less applicable to the extent that (at least some) of these differences relate to inter-species that may relate to evolutionary differences or potential innate differences related to species-typical behavior (Payne et al., 2021; Quian Quiroga, 2020; Rolls, 2023; Rolls & Wirth, 2018). For example, one possibility is that flexibly shifting hippocampal representation and/or the specificity of particular representations is rather specific to humans. Alternatively, the different types of perceptual signals that rodents, primates, and humans rely on during navigation could be transformed from or into self-referenced representations or allocentric reference frames in species specific ways (Alexander et al., 2023; C. Yang et al., 2023; Zhu et al., 2023). Future work on explicit cross-species comparisons should therefore be a complement to multi-task experiments to further investigate hippocampal function.
Another key element of using multi-task designs is to consider the practical implications. In animal models, there are additional training requirements that may make multi-task designs more arduous (though note there are successfully multi-task examples in mice (J. J. Lee et al., 2022), rats (Aronov et al., 2017; Hallock & Griffin, 2013; Trullier et al., 1999), bats (Sarel et al., 2022), and primates (Asaad et al., 2000; Gulli et al., 2020; Rolls & Xiang, 2005). Multi-task experiments may be particularly applicable to human experiments (Donoghue et al., 2023; Minxha et al., 2020), as it is easier to ask subjects to rapidly learn and switch between different tasks / contexts, suggesting human studies may have an important role to play in investigating the flexibility of hippocampal activity. However, limitations in human studies include that recording time is limited, requiring designs that allow for sufficient data acquisition across the tasks, conditions, and/or trial types of interest. Due to this, careful consideration needs to be taken that experiments are designed to be well powered, and appropriately counter-balanced.
We also believe that multi-task and multi-context experiments may provide a stepping stone not only to bridge between different fields of inquiry related to the hippocampus, but also to other approaches taken in neuroscience. For example, multi-context task designs including using multiple distinct mazes and different memory conditions has been used with non-invasive neuroimaging (Brown et al., 2010; Brown & Stern, 2014), which offers data that could be usefully compared to other modalities, tasks, and species. Relatedly, there has been an ongoing conversation on the use of more naturalistic and ecologically valid experiments in neuroscience (Finn et al., 2022; Geva-Sagiv et al., 2015; Matusz et al., 2019; Nastase et al., 2020; Sonkusare et al., 2019). While fully naturalistic experiments require whole new frameworks of analysis, in at least some cases, using multi-task experiments may provide an intermediate approach that allows for expanding the range of behavior and evaluating if responses generalize (or shift) across individual, constrained tasks, while maintaining the control of using (multiple) laboratory tasks. Multi-task experiments can also be used to test the robustness of responses across different contexts, such as comparing real world navigation to navigating in virtual space (Aghajan et al., 2015), in order to examine how choices in task design affect neural responses. In addition, multi-task experiments offer the potential for comparing between constrained and naturalistic tasks if such tasks can be combined together, to explicitly compare neural activity between more constrained and naturalistic task settings.
Additionally, drawing from the field of machine learning, recent work has started to explore how artificial neural networks can be trained to do multi-task learning, and exploring what kinds of representations they acquire when doing so and what this can tell us about the brain (Driscoll et al., 2022; G. R. Yang, Joglekar, et al., 2019). Related work has further specified how modeling work can be used to investigate neural mechanisms of multiple tasks (G. R. Yang, Cole, et al., 2019). This kind of theoretical and modeling work, combined with examinations of how artificial neural networks can be trained and tested across different tasks, offers another avenue that could potentially be productively linked with multi-task empirical work. Future work could investigate how the different kinds of representations that emerge in artificial neural networks trained on different task combinations relates to representations seen in the brain, and what this implies about the kinds of features and tasks these circuits appear to be most tuned to.
Conclusions
The neural representations of the hippocampus and surrounding structures have been extensively studied, across species, tasks, and theoretical frameworks, which typically focus on cognitive domains such as spatial encoding and memory-related processes. Here, we emphasize that the typical procedure of examining neural activity across a single task is limited by not allowing for examining responses across different behavioral contexts – testing whether responses change across different task contexts and whether individual neurons change their representation. This may be important to address important questions regarding hippocampal function(s). To address this limitation, in this commentary, we discussed the feasibility and advantages of using multi-task experiment designs to further disentangle the flexible and highly task-dependent responses primarily in the hippocampus – when combined with explicit specifications of theoretical assumptions and hypotheses – for investigating the nature of how the hippocampal works.
Funding Sources
This work was supported by the AFOSR (FA9550-21-1-0088), NSF (BCS-1945230, IIS-2114644), and NIH (R01MH129426, R01-MH104606).
Footnotes
Disclosures
Conflicts of Interest
The authors declare no competing interests.
Data Availability Statement
Data sharing is not applicable to this article as no new data were created or analyzed in this study.
References
- Aghajan ZM, Acharya L, Moore JJ, Cushman JD, Vuong C, & Mehta MR (2015). Impaired spatial selectivity and intact phase precession in two-dimensional virtual reality. Nature Neuroscience, 18(1), 121–128. 10.1038/nn.3884 [DOI] [PubMed] [Google Scholar]
- Alexander AS, Carstensen LC, Hinman JR, Raudies F, Chapman GW, & Hasselmo ME (2020). Egocentric boundary vector tuning of the retrosplenial cortex. Science Advances, 6(8), eaaz2322. 10.1126/sciadv.aaz2322 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alexander AS, Robinson JC, Stern CE, & Hasselmo ME (2023). Gated transformations from egocentric to allocentric reference frames involving retrosplenial cortex, entorhinal cortex, and hippocampus. Hippocampus, 33. 10.1002/hipo.23513 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aronov D, Nevers R, & Tank DW (2017). Mapping of a non-spatial dimension by the hippocampal–entorhinal circuit. Nature, 543(7647), 719–722. 10.1038/nature21692 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Asaad WF, Rainer G, & Miller EK (2000). Task-Specific Neural Activity in the Primate Prefrontal Cortex. Journal of Neurophysiology, 84(1), 451–459. 10.1152/jn.2000.84.1.451 [DOI] [PubMed] [Google Scholar]
- Bakker A, Kirwan CB, Miller M, & Stark CEL (2008). Pattern Separation in the Human Hippocampal CA3 and Dentate Gyrus. Science, 319(5870), 1640–1642. 10.1126/science.1152882 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baraduc P, Duhamel J-R, & Wirth S (2019). Schema cells in the macaque hippocampus. Science, 363(6427), 635–639. 10.1126/science.aav5404 [DOI] [PubMed] [Google Scholar]
- Barry C, Lever C, Hayman R, Hartley T, Burton S, O’Keefe J, Jeffery K, & Burgess Ν (2006). The Boundary Vector Cell Model of Place Cell Firing and Spatial Memory. Reviews in the Neurosciences, 17(1–2). 10.1515/REVNEURO.2006.17.1-2.71 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Behrens TEJ, Muller TH, Whittington JCR, Mark S, Baram AB, Stachenfeld KL, & Kurth-Nelson Z (2018). What Is a Cognitive Map? Organizing Knowledge for Flexible Behavior. Neuron, 100(2), 490–509. 10.1016/j.neuron.2018.10.002 [DOI] [PubMed] [Google Scholar]
- Bellmund JLS, Gärdenfors P, Moser EI, & Doeller CF (2018). Navigating cognition: Spatial codes for human thinking. Science, 362(6415), eaat6766. 10.1126/science.aat6766 [DOI] [PubMed] [Google Scholar]
- Benna MK, & Fusi S (2021). Place cells may simply be memory cells: Memory compression leads to spatial tuning and history dependence. Proceedings of the National Academy of Sciences, 118(51), e2018422118. 10.1073/pnas.2018422118 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bicanski A, & Burgess N (2018). A neural-level model of spatial memory and imagery. ELife, 7, e33752. 10.7554/eLife.33752 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bicanski A, & Burgess N (2020). Neuronal vector coding in spatial cognition. Nature Reviews Neuroscience, 21(9), 453–470. 10.1038/s41583-020-0336-9 [DOI] [PubMed] [Google Scholar]
- Brown TI, Ross RS, Keller JB, Hasselmo ME, & Stern CE (2010). Which Way Was I Going? Contextual Retrieval Supports the Disambiguation of Well Learned Overlapping Navigational Routes. The Journal of Neuroscience, 30(21), 7414–7422. 10.1523/JNEUROSCI.6021-09.2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown TI, & Stern CE (2014). Contributions of Medial Temporal Lobe and Striatal Memory Systems to Learning and Retrieving Overlapping Spatial Memories. Cerebral Cortex, 24(7), 1906–1922. 10.1093/cercor/bht041 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burgess N (2006). Spatial memory: How egocentric and allocentric combine. Trends in Cognitive Sciences, 10(12), 551–557. 10.1016/j.tics.2006.10.005 [DOI] [PubMed] [Google Scholar]
- Buzsáki G, & Moser EI (2013). Memory, navigation and theta rhythm in the hippocampal-entorhinal system. Nature Neuroscience, 16(2), 130–138. 10.1038/nn.3304 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cao R, Wang J, Lin C, De Falco E, Peter A, Rey HG, DiCarlo J, Todorov A, Rutishauser U, Li X, Brandmeir NJ, & Wang S (2022). Feature-based encoding of face identity by single neurons in the human amygdala and hippocampus [Preprint]. Neuroscience 10.1101/2020.09.01.278283 [DOI]
- Corrigan BM, Gulli RA, Doucet G, Mahmoudian B, Abbass M, Roussy M, Luna R, Sachs AJ, & Martinez-Trujillo JC (2023). View cells in the Hippocampus and Prefrontal cortex of macaques during virtual navigation. Hippocampus, 33. 10.1002/hipo.23534 [DOI] [PubMed] [Google Scholar]
- Danjo T, Toyoizumi T, & Fujisawa S (2018). Spatial representations of self and other in the hippocampus. Science, 359(6372), 213–218. 10.1126/science.aao3898 [DOI] [PubMed] [Google Scholar]
- De Falco E, Ison MJ, Fried I, & Quian Quiroga R (2016). Long-term coding of personal and universal associations underlying the memory web in the human brain. Nature Communications, 7(1), 13408. 10.1038/ncomms13408 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Donoghue T, Cao R, Han CZ, Holman CM, Brandmeir NJ, Wang S, & Jacobs J (2023). Single neurons in the human medial temporal lobe flexibly shift representations across spatial and memory tasks. Hippocampus, 33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Driscoll L, Shenoy K, & Sussillo D (2022). Flexible multitask computation in recurrent networks utilizes shared dynamical motifs [Preprint]. Neuroscience 10.1101/2022.08.15.503870 [DOI] [PMC free article] [PubMed]
- Duvelle É, Grieves RM, & van der Meer MA (2023). Temporal context and latent state inference in the hippocampal splitter signal. ELife, 12, e82357. 10.7554/eLife.82357 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eichenbaum H (2017a). The role of the hippocampus in navigation is memory. Journal of Neurophysiology, 117(4), 1785–1796. 10.1152/jn.00005.2017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eichenbaum H (2017b). On the Integration of Space, Time, and Memory. Neuron, 95(5), 1007–1018. 10.1016/j.neuron.2017.06.036 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eichenbaum H, & Cohen NJ (2014). Can We Reconcile the Declarative Memory and Spatial Navigation Views on Hippocampal Function? Neuron, 83(4), 764–770. 10.1016/j.neuron.2014.07.032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ekstrom AD, Kahana MJ, Caplan JB, Fields TA, Isham EA, Newman EL, & Fried I (2003). Cellular networks underlying human spatial navigation. Nature, 425(6954), 184–188. 10.1038/nature01964 [DOI] [PubMed] [Google Scholar]
- Ekstrom AD, & Ranganath C (2018). Space, time, and episodic memory: The hippocampus is all over the cognitive map. Hippocampus, 28(9), 680–687. 10.1002/hipo.22750 [DOI] [PubMed] [Google Scholar]
- Epstein R, Patai EZ, Julian JB, & Spiers HJ (2017). The cognitive map in humans: Spatial navigation and beyond. Nature Neuroscience, 20(11), 1504–1513. 10.1038/nn.4656 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fenton AA, Lytton WW, Barry JM, Lenck-Santini PP, Zinyuk LE, Kubik S, Bures J, Poucet B, Muller RU, & Olypher AV (2010). Attention-Like Modulation of Hippocampus Place Cell Discharge. Journal of Neuroscience, 30(13), 4613–4625. 10.1523/JNEUROSCI.5576-09.2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Finn ES, Glerean E, Hasson U, & Vanderwal T (2022). Naturalistic imaging: The use of ecologically valid conditions to study brain function. NeuroImage, 247, 118776. 10.1016/j.neuroimage.2021.118776 [DOI] [PubMed] [Google Scholar]
- Frankland PW, Josselyn SA, & Köhler S (2019). The neurobiological foundation of memory retrieval. Nature Neuroscience, 22(10), 1576–1585. 10.1038/s41593-019-0493-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geva-Sagiv M, Las L, Yovel Y, & Ulanovsky N (2015). Spatial cognition in bats and rats: From sensory acquisition to multiscale maps and navigation. Nature Reviews Neuroscience, 16(2), 94–108. 10.1038/nrn3888 [DOI] [PubMed] [Google Scholar]
- Gulli RA, Duong LR, Corrigan BW, Doucet G, Williams S, Fusi S, & Martinez-Trujillo JC (2020). Context-dependent representations of objects and space in the primate hippocampus during virtual navigation. Nature Neuroscience, 23(1), 103–112. 10.1038/s41593-019-0548-3 [DOI] [PubMed] [Google Scholar]
- Hallock HL, & Griffin AL (2013). Dynamic coding of dorsal hippocampal neurons between tasks that differ in structure and memory demand. Hippocampus, 23(2), 169–186. 10.1002/hipo.22079 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hasselmo ME (2012). How we remember: Brain mechanisms of episodic memory MIT Press. [Google Scholar]
- Heit G, Smith ME, & Halgren E (1988). Neural encoding of individual words and faces by the human hippocampus and amygdala. Nature, 333(6175), 773–775. 10.1038/333773a0 [DOI] [PubMed] [Google Scholar]
- Ison MJ, Quian Quiroga R, & Fried I. (2015). Rapid Encoding of New Memories by Individual Neurons in the Human Brain. Neuron, 87(1), 220–230. 10.1016/j.neuron.2015.06.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jercog PE, Ahmadian Y, Woodruff C, Deb-Sen R, Abbott LF, & Kandel ER (2019). Heading direction with respect to a reference point modulates place-cell activity. Nature Communications, 10(1), 2333. 10.1038/s41467-019-10139-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Josselyn SA, & Frankland PW (2018). Memory Allocation: Mechanisms and Function. Annual Review of Neuroscience, 41(1), 389–413. 10.1146/annurev-neuro-080317-061956 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Josselyn SA, & Tonegawa S (2020). Memory engrams: Recalling the past and imagining the future. Science, 367(6473), eaaw4325. 10.1126/science.aaw4325 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Killian NJ, Jutras MJ, & Buffalo EA (2012). A map of visual space in the primate entorhinal cortex. Nature, 491(7426), 761–764. 10.1038/nature11587 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kobayashi T, Nishijo H, Fukuda M, Bures J, & Ono T (1997). Task-Dependent Representations in Rat Hippocampal Place Neurons. Journal of Neurophysiology, 78(2), 597–613. 10.1152/jn.1997.78.2.597 [DOI] [PubMed] [Google Scholar]
- Kreiman G, Koch C, & Fried I (2000). Category-specific visual responses of single neurons in the human medial temporal lobe. Nature Neuroscience, 3(9), 946–953. 10.1038/78868 [DOI] [PubMed] [Google Scholar]
- Kubie JL, Levy ERJ, & Fenton AA (2020). Is hippocampal remapping the physiological basis for context? Hippocampus, 30(8), 851–864. 10.1002/hipo.23160 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kubie JL, & Muller RU (1991). Multiple representations in the hippocampus. Hippocampus, 1(3), 240–242. 10.1002/hipo.450010305 [DOI] [PubMed] [Google Scholar]
- Kunz L, Brandt A, Reinacher PC, Staresina BP, Reifenstein ET, Weidemann CT, Herweg NA, Patel A, Tsitsiklis M, Kempter R, Kahana MJ, Schulze-Bonhage A, & Jacobs J (2021). A neural code for egocentric spatial maps in the human medial temporal lobe. Neuron, S0896627321004608. 10.1016/j.neuron.2021.06.019 [DOI] [PMC free article] [PubMed]
- LaChance PA, & Taube JS (2022). Spatial context and the functional role of the postrhinal cortex. Neurobiology of Learning and Memory, 189, 107596. 10.1016/j.nlm.2022.107596 [DOI] [PMC free article] [PubMed] [Google Scholar]
- LaChance PA, & Taube JS (2023). A model for transforming egocentric views into goal‐directed behavior. Hippocampus, 33. 10.1002/hipo.23510 [DOI] [PMC free article] [PubMed] [Google Scholar]
- LaChance PA, Todd TP, & Taube JS (2019). A sense of space in postrhinal cortex. Science, 365(6449), eaax4192. 10.1126/science.aax4192 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leal SL, & Yassa MA (2018). Integrating new findings and examining clinical applications of pattern separation. Nature Neuroscience, 21(2), 163–173. 10.1038/s41593-017-0065-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee JJ, Krumin M, Harris KD, & Carandini M (2022). Task specificity in mouse parietal cortex. Neuron, 110(18), 2961–2969.e5. 10.1016/j.neuron.2022.07.017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee S, Shin J, & Lee I (2023). Significance of visual scene‐based learning in the hippocampal systems across mammalian species. Hippocampus, 33. 10.1002/hipo.23483 [DOI] [PubMed] [Google Scholar]
- Levenstein D, Alvarez VA, Amarasingham A, Azab H, Gerkin RC, Hasenstaub A, Iyer R, Jolivet RB, Marzen S, Monaco JD, Prinz AA, Quraishi S, Santamaria F, Shivkumar S, Singh MF, Stockton B, Traub R, Rotstein HG, Nadim F, & Redish AD (2020). On the role of theory and modeling in neuroscience 24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levy SJ, & Hasselmo ME (2023). Hippocampal remapping induced by new behavior is mediated by spatial context [Preprint]. Neuroscience 10.1101/2023.02.20.529330 [DOI]
- Lisman J, Buzsáki G, Eichenbaum H, Nadel L, Ranganath C, & Redish AD (2017). Viewpoints: How the hippocampus contributes to memory, navigation and cognition. Nature Neuroscience, 20(11), 1434–1447. 10.1038/nn.4661 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Markus E, Qin Y, Leonard B, Skaggs W, McNaughton B, & Barnes C (1995). Interactions between location and task affect the spatial and directional firing of hippocampal neurons. The Journal of Neuroscience, 15(11), 7079–7094. 10.1523/JNEUROSCI.15-11-07079.1995 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matusz PJ, Dikker S, Huth AG, & Perrodin C (2019). Are We Ready for Real-world Neuroscience? Journal of Cognitive Neuroscience, 31(3), 327–338. 10.1162/jocn_e_01276 [DOI] [PMC free article] [PubMed] [Google Scholar]
- McNaughton BL, Battaglia FP, Jensen O, Moser EI, & Moser M-B (2006). Path integration and the neural basis of the “cognitive map.” Nature Reviews Neuroscience, 7(8), 663–678. 10.1038/nrn1932 [DOI] [PubMed] [Google Scholar]
- Miller JF, Neufang M, Solway A, Brandt A, Trippel M, Mader I, Hefft S, Merkow M, Polyn SM, Jacobs J, Kahana MJ, & Schulze-Bonhage A (2013). Neural Activity in Human Hippocampal Formation Reveals the Spatial Context of Retrieved Memories. Science, 342(6162), 1111–1114. 10.1126/science.1244056 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Milner B, Squire LR, & Kandel ER (1998). Cognitive Neuroscience and the Study of Memory. Neuron, 20(3), 445–468. 10.1016/S0896-6273(00)80987-3 [DOI] [PubMed] [Google Scholar]
- Minxha J, Adolphs R, Fusi S, Mamelak AN, & Rutishauser U (2020). Flexible recruitment of memory-based choice representations by the human medial frontal cortex. Science, 368(6498), eaba3313. 10.1126/science.aba3313 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moore JJ, Cushman JD, Acharya L, Popeney B, & Mehta MR (2021). Linking hippocampal multiplexed tuning, Hebbian plasticity and navigation. Nature 10.1038/s41586-021-03989-z [DOI] [PubMed]
- Moser EI, Moser M-B, & McNaughton BL (2017). Spatial representation in the hippocampal formation: A history. Nature Neuroscience, 20(11), 1448–1464. 10.1038/nn.4653 [DOI] [PubMed] [Google Scholar]
- Muller R, & Kubie J (1989). The firing of hippocampal place cells predicts the future position of freely moving rats. The Journal of Neuroscience, 9(12), 4101–4110. 10.1523/JNEUROSCI.09-12-04101.1989 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nastase SA, Goldstein A, & Hasson U (2020). Keep it real: Rethinking the primacy of experimental control in cognitive neuroscience. NeuroImage, 222, 117254. 10.1016/j.neuroimage.2020.117254 [DOI] [PMC free article] [PubMed] [Google Scholar]
- O’Keefe J, & Dostrovsky J (1971). The hippocampus as a spatial map. Preliminary evidence from unit activity in the freely-moving rat. Brain Research, 34(1), 171–175. 10.1016/0006-8993(71)90358-1 [DOI] [PubMed] [Google Scholar]
- O’Keefe J, & Krupic J (2021). Do hippocampal pyramidal cells respond to nonspatial stimuli? Physiological Reviews, 101(3), 1427–1456. 10.1152/physrev.00014.2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- O’Keefe J, & Nadel L (1978). The hippocampus as a cognitive map Clarendon Press ; Oxford University Press. [Google Scholar]
- Omer DB, Maimon SR, Las L, & Ulanovsky N (2018). Social place-cells in the bat hippocampus. Science, 359(6372), 218–224. 10.1126/science.aao3474 [DOI] [PubMed] [Google Scholar]
- Ono T, Nakamura K, Nishijo H, & Eifuku S (1993). Monkey hippocampal neurons related to spatial and nonspatial functions. Journal of Neurophysiology, 70(4), 1516–1529. 10.1152/jn.1993.70.4.1516 [DOI] [PubMed] [Google Scholar]
- Ormond J, & O’Keefe J (2022). Hippocampal place cells have goal-oriented vector fields during navigation. Nature, 607, 741–746. 10.1038/s41586-022-04913-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Payne HL, Lynch GF, & Aronov D (2021). Neural representations of space in the hippocampus of a food-caching bird. Science, 373(6552), 343–348. 10.1126/science.abg2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quian Quiroga R (2012). Concept cells: The building blocks of declarative memory functions. Nature Reviews Neuroscience, 13(8), 587–597. 10.1038/nrn3251 [DOI] [PubMed] [Google Scholar]
- Quian Quiroga R (2020). No Pattern Separation in the Human Hippocampus. Trends in Cognitive Sciences, 24(12), 994–1007. 10.1016/j.tics.2020.09.012 [DOI] [PubMed] [Google Scholar]
- Quian Quiroga R (2023). An integrative view of human hippocampal function: Differences with other species and capacity considerations. Hippocampus, 33. 10.1002/hipo.23527 [DOI] [PubMed] [Google Scholar]
- Quian Quiroga R, Kreiman G, Koch C, & Fried I (2008). Sparse but not ‘Grandmother-cell’ coding in the medial temporal lobe. Trends in Cognitive Sciences, 12(3), 87–91. 10.1016/j.tics.2007.12.003 [DOI] [PubMed] [Google Scholar]
- Quian Quiroga R, Reddy L, Kreiman G, Koch C, & Fried I (2005). Invariant visual representation by single neurons in the human brain. Nature, 435(7045), 1102–1107. 10.1038/nature03687 [DOI] [PubMed] [Google Scholar]
- Rey HG, De Falco E, Ison MJ, Valentin A, Alarcon G, Selway R, Richardson MP, & Quian Quiroga R (2018). Encoding of long-term associations through neural unitization in the human medial temporal lobe. Nature Communications, 9(1), 4372. 10.1038/s41467-018-06870-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rey HG, Gori B, Chaure FJ, Collavini S, Blenkmann AO, Seoane P, Seoane E, Kochen S, & Quian Quiroga R (2020). Single Neuron Coding of Identity in the Human Hippocampal Formation. Current Biology, 30(6), 1152–1159.e3. 10.1016/j.cub.2020.01.035 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rolls ET (1999). Spatial view cells and the representation of place in the primate hippocampus. Hippocampus, 9(4), 467–480. [DOI] [PubMed] [Google Scholar]
- Rolls ET (2023). Hippocampal spatial view cells for memory and navigation, and their underlying connectivity in humans. Hippocampus, 33. 10.1002/hipo.23467 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rolls ET, & Wirth S (2018). Spatial representations in the primate hippocampus, and their functions in memory and navigation. Progress in Neurobiology, 171, 90–113. 10.1016/j.pneurobio.2018.09.004 [DOI] [PubMed] [Google Scholar]
- Rolls ET, & Xiang J-Z (2005). Reward-Spatial View Representations and Learning in the Primate Hippocampus. The Journal of Neuroscience, 25(26), 6167–6174. 10.1523/JNEUROSCI.1481-05.2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ryom KI, Stendardi D, Ciaramelli E, & Treves A (2023). Computational constraints on the associative recall of spatial scenes. Hippocampus, 33. 10.1002/hipo.23511 [DOI] [PubMed] [Google Scholar]
- Sarel A, Finkelstein A, Las L, & Ulanovsky N (2017). Vectorial representation of spatial goals in the hippocampus of bats. Science, 355(6321), 176–180. 10.1126/science.aak9589 [DOI] [PubMed] [Google Scholar]
- Sarel A, Palgi S, Blum D, Aljadeff J, Las L, & Ulanovsky N (2022). Natural switches in behaviour rapidly modulate hippocampal coding. Nature. 10.1038/s41586-022-05112-2 [DOI] [PMC free article] [PubMed]
- Schiller D, Eichenbaum H, Buffalo EA, Davachi L, Foster DJ, Leutgeb S, & Ranganath C (2015). Memory and Space: Towards an Understanding of the Cognitive Map. Journal of Neuroscience, 35(41), 13904–13911. 10.1523/JNEUROSCI.2618-15.2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scoville WB, & Milner B (1957). Loss of recent memory after bilateral hippocampal lesions. Journal of Neurology, Neurosurgery, and Psychiatry, 20(1), 11–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sliwa J, Planté A, Duhamel J-R, & Wirth S (2016). Independent Neuronal Representation of Facial and Vocal Identity in the Monkey Hippocampus and Inferotemporal Cortex. Cerebral Cortex, 26(3), 950–966. 10.1093/cercor/bhu257 [DOI] [PubMed] [Google Scholar]
- Sonkusare S, Breakspear M, & Guo C (2019). Naturalistic Stimuli in Neuroscience: Critically Acclaimed. Trends in Cognitive Sciences, 23(8), 699–714. 10.1016/j.tics.2019.05.004 [DOI] [PubMed] [Google Scholar]
- Stangl M, Topalovic U, Inman CS, Hiller S, Villaroman D, Aghajan ZM, Christov-Moore L, Hasulak NR, Rao VR, Halpern CH, Eliashiv D, Fried I, & Suthana N (2020). Boundary-anchored neural mechanisms of location-encoding for self and others. Nature 10.1038/s41586-020-03073-y [DOI] [PMC free article] [PubMed]
- Sugar J, & Moser M (2019). Episodic memory: Neuronal codes for what, where, and when. Hippocampus, 29(12), 1190–1205. 10.1002/hipo.23132 [DOI] [PubMed] [Google Scholar]
- Trullier O, Shibata R, Mulder AB, & Wiener SI (1999). Hippocampal neuronal position selectivity remains fixed to room cues only in rats alternating between place navigation and beacon approach tasks: Dissociation of behaviour and place cell activity. European Journal of Neuroscience, 11(12), 4381–4388. 10.1046/j.1460-9568.1999.00839.x [DOI] [PubMed] [Google Scholar]
- Tsitsiklis M, Miller JF, Qasim SE, Inman CS, Gross RE, Willie JT, Smith EH, Sheth SA, Schevon CA, Sperling MR, Sharan A, Stein JM, & Jacobs J (2020). Single-Neuron Representations of Spatial Targets in Humans. Current Biology, 30(2), 245–253.e4. 10.1016/j.cub.2019.11.048 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vorhees CV, & Williams MT (2006). Morris water maze: Procedures for assessing spatial and related forms of learning and memory. Nature Protocols, 1(2), 848–858. 10.1038/nprot.2006.116 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang C, Chen X, & Knierim JJ (2020). Egocentric and allocentric representations of space in the rodent brain. Current Opinion in Neurobiology, 60, 12–20. 10.1016/j.conb.2019.11.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang C, Chen X, Lee H, Deshmukh SS, Yoganarasimha D, Savelli F, & Knierim JJ (2018). Egocentric coding of external items in the lateral entorhinal cortex. Science, 362(6417), 945–949. 10.1126/science.aau4940 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang C, Lee H, Rao G, Doreswamy Y, Savelli F, & Knierim JJ (2023). Superficial-layer vs. Deep-layer lateral entorhinal cortex: Coding of allocentric space, egocentric space, speed, boundaries, and corners. Hippocampus, 33. 10.1002/hipo.23528 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang J, Cao R, Brandmeir NJ, Li X, & Wang S (2022). Face identity coding in the deep neural network and primate brain. Communications Biology, 5(1), 611. 10.1038/s42003-022-03557-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Whittington JCR, Muller TH, Mark S, Chen G, Barry C, Burgess N, & Behrens TEJ (2020). The Tolman-Eichenbaum Machine: Unifying Space and Relational Memory through Generalization in the Hippocampal Formation. Cell, S009286742031388X. 10.1016/j.cell.2020.10.024 [DOI] [PMC free article] [PubMed]
- Yang C, Chen H, & Naya Y (2023). Allocentric information represented by self‐referenced spatial coding in the primate medial temporal lobe. Hippocampus, 33. 10.1002/hipo.23501 [DOI] [PubMed] [Google Scholar]
- Yang GR, Cole MW, & Rajan K (2019). How to study the neural mechanisms of multiple tasks. Current Opinion in Behavioral Sciences, 29, 134–143. 10.1016/j.cobeha.2019.07.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang GR, Joglekar MR, Song HF, Newsome WT, & Wang X-J (2019). Task representations in neural networks trained to perform many cognitive tasks. Nature Neuroscience 10.1038/s41593-018-0310-2 [DOI] [PMC free article] [PubMed]
- Zhu SL, Lakshminarasimhan KJ, & Angelaki DE (2023). Computational Cross-Species Views of the Hippocampal Formation. Hippocampus, 33. 10.1002/hipo.23535 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data sharing is not applicable to this article as no new data were created or analyzed in this study.


