Abstract
Catharanthus roseus L. (G.) Don is the most widely studied plant because of its high pharmacological value. In vitro culture uses various plant parts such as leaves, nodes, internodes and roots for inducing callus and subsequent plant regeneration in C. roseus. However, till now, little work has been conducted on anther tissue using plant tissue culture techniques. Therefore, the aim of this work is to establish a protocol for in vitro induction of callus by utilizing anthers as explants in MS (Murashige and Skoog) medium fortified with different concentrations and combinations of PGRs. The best callusing medium contains high α-naphthalene acetic acid (NAA) and low kinetin (Kn) concentrations showing a callusing frequency of 86.6%. SEM–EDX analysis was carried out to compare the elemental distribution on the surfaces of anther and anther-derived calli, and the two were noted to be nearly identical in their elemental composition. Gas chromatography–mass spectrometry (GC–MS) analysis of methanol extracts of anther and anther-derived calli was conducted, which revealed the presence of a wide range of phytocompounds. Some of them are ajmalicine, vindolinine, coronaridine, squalene, pleiocarpamine, stigmasterol, etc. More importantly, about 17 compounds are exclusively present in anther-derived callus (not in anther) of Catharanthus. The ploidy status of anther-derived callus was examined via flow cytometry (FCM), and it was estimated to be 0.76 pg, showing the haploid nature of callus. The present work therefore represents an efficient way to produce high-value medicinal compounds from anther callus in a lesser period of time on a larger scale.
Keywords: anther culture, flow cytometry, GC–MS, phytochemical profiling, ploidy level, secondary metabolites, SEM–EDX
1. Introduction
Catharanthus roseus (L.) G. Don, a member of the Apocynaceae family, is a popular flowering plant. It is an indigenous species to Madagascar and is widely distributed throughout the African, American, Asian and southern European regions. In India, C. roseus has been spread across all the major parts of Gujarat, Madhya Pradesh, Assam, Bihar, Uttar Pradesh, Karnataka and Tamil Nadu [1]. The plant is well known for both its ornamental and medicinal value. It produces nearly 130 alkaloids, of which vincristine and vinblastine are the two major compounds that are used in the treatment of leukemia and Hodgkin’s lymphoma [2]. For decades, this plant has been exploited for pharmaceutically active compounds from its native environments and thus is at risk of declining in the wild. Plant tissue culture proves to be an effective biotechnological tool for the rapid propagation of plants under aseptic conditions with a lesser risk of microbial infections [3]. Several in vitro studies using different explants have been successfully conducted for somatic embryogenesis [4] and organogenesis in C. roseus [5,6].
In recent times, double haploid (DH) production via anther is a promising option for developing improved plant varieties with high yields of medicinally important bioactive compounds [7]. In vitro anther culture has been attempted in various plants such as Actinidia arguta Planch [8] and Triticum aestivum L. [9]. Various factors such as stage of anther, culture conditions, plant growth regulators (PGRs) and genotypic and ploidy status determine the success of DH generation [10]. These factors necessitate ascertaining the ploidy status of anther-derived callus to generate true-to-type DH lines, which can be performed with a flow cytometric technique. The flow cytometry method (FCM) measures the genome size by examining the nuclei at a relatively faster rate and thus validates the ploidy levels of different plant tissues [11]. Recent investigations of genome size analysis using FCM have been reported for different plants [12,13]. Phytochemical profiling using gas chromatography coupled with mass spectrometry (GC–MS) has emerged as an important procedure for identifying and quantifying therapeutically significant compounds present in medicinal plants. This technique is relatively faster, accurate and needs a minimum volume of extracts to detect a wide range of bioactive compounds such as alkaloids, long-chain hydrocarbons, steroids, sugars, amino acids and nitro compounds [14]. Major bioactive compounds extracted from different plant parts of C. roseus such as stem, root and leaf include vincristine, vinblastine, reserpine, ajmalicine, vindolinine and catharine, which possess anti-cancerous, anti-diabetic, anti-fungal and anti-microbial activities [15]. GC–MS-based profiling has been recently reported for several plants including Silybum marianum L. [16] and Chukrasia velutina [17], but the information on tissue-culture-raised plants’ phytocompound profiling is relatively much less. The present work, therefore, focuses on investigating the ploidy status of anther-derived callus of C. roseus using flow cytometry. The elemental composition of both anther and anther calli was studied using a scanning electron microscopy–energy-dispersive X-ray microanalysis (SEM–EDX) technique. The identification of the bioactive compounds present in methanolic extracts of anther and anther-derived calli was conducted for the first time in C. roseus using GC–MS analysis. This report will help to understand and improve the yield of the important pharmaceutical compounds synthesized from anther-derived callus.
2. Results
2.1. Callus Induction and Proliferation
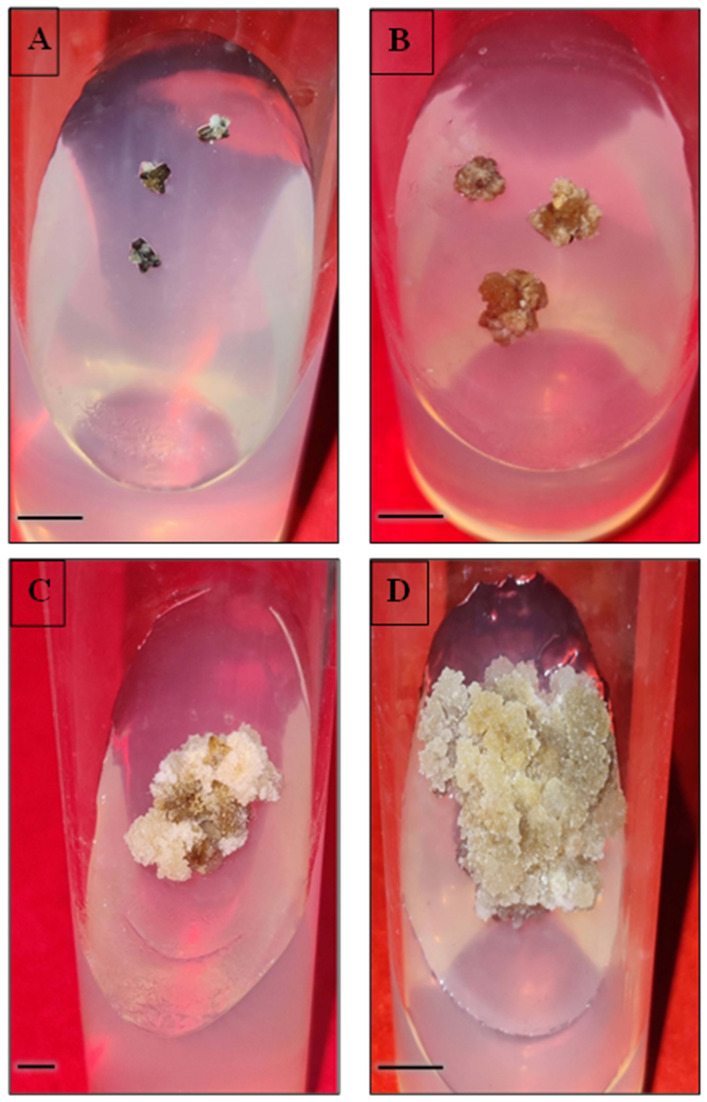
In this study, the anthers were used as explants to induce callus on MS medium augmented with different concentrations and combinations of NAA and kinetin or TDZ alone (Figure 1A). The callusing response ranged from 13.3% to 86.6% on all the tested media (Table 1). Among the PGRs utilized, a combination of NAA and kinetin produced maximum callus (86.6%) at concentrations of 1.0 mg/L and 0.1 mg/L, respectively, followed by 0.75 mg/L TDZ with a frequency of 73.3%. On the other hand, TDZ alone at 0.5 mg/L showed the least incidence of callusing efficiency (13.3%). The highest callus fresh weight was noted to be 1.7 g on MS medium containing 1.0 mg/L NAA and 0.1 mg/L kinetin. The calli obtained were white to pale yellow in color and friable in nature (Figure 1B–D). The anther callus was noted to be recalcitrant, as plant regeneration (embryogenesis and organogenesis) was not achieved on any medium added with various PGR combinations.
Figure 1.
In vitro callus induction, proliferation and scanning electron microscopic (SEM) images of anther and anther-derived callus of C. roseus. (A,B): callus initiation (bars = 0.5 cm); (C,D): callus proliferation after 6 and 9 weeks, respectively (bars (C) = 1.0 cm, (D) = 0.5 cm); (E): side view of anther (bar = 200 µm); (F): a portion of anther-derived callus (bar = 20 µm).
Table 1.
Effect of different concentrations and combinations of PGRs on callus induction and callus biomass (fresh weight) from anther explants of C. roseus.
| PGRs | Concentration (mg/L) | Callusing Frequency (%) | Mean Fresh Weight (g) |
|---|---|---|---|
| Control | 0 | 0 e | 0 c |
| NAA + Kn | 0.1 + 1.0 | 26.6 ± 12.4 cde | 0.8 ± 0.3 abc |
| 0.5 + 0.75 | 33.3 ± 14.9 cde | 0.9 ± 0.3 ab | |
| 0.75 + 0.5 | 53.3 ± 16.9 abc | 1.1 ± 0.3 ab | |
| 1.0 + 0.1 | 86.6 ± 8.1 a | 1.7 ± 1.7 a | |
| TDZ | 0.5 | 13.3 ± 8.1 de | 0.5 ± 0.3 bc |
| 0.75 | 73.3 ± 27.8 ab | 1.3 ± 0.2 ab | |
| 1 | 46.6 ± 16.9 bcd | 0.9 ± 0.2 ab |
Mean values followed by the same superscripts within a column are not significantly different according to DMRT at p ≤ 0.05 level.
2.2. Surface Morphology and Elemental Analysis
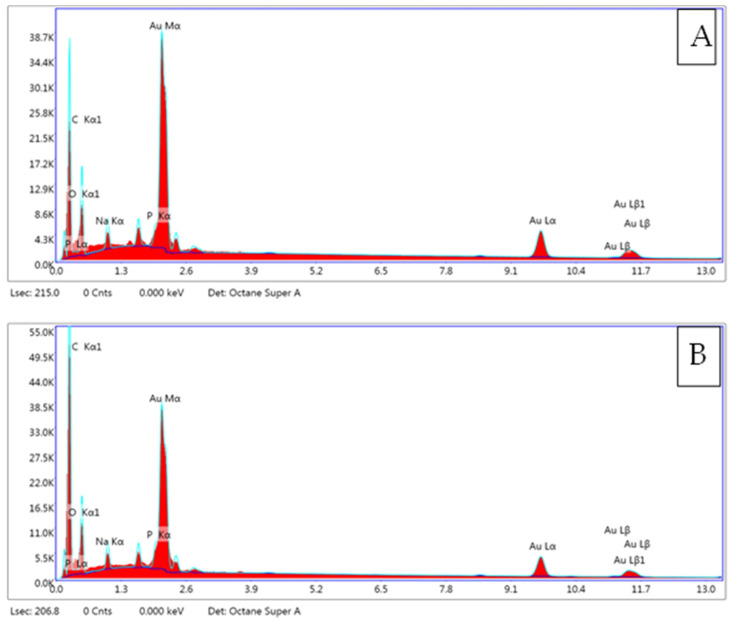
SEM–EDAX analysis was carried out to determine the elemental composition of anther as well as anther-derived callus. The SEM images and their respective spectra are shown in Figure 1E,F and Figure 2, respectively. The various peaks in both spectra reveal carbon, oxygen, sodium and phosphorous to be the major elements present on the surfaces of anther and anther-derived calli. In both the samples, the carbon and oxygen peaks are prominent and of high intensity, whereas those of sodium and phosphorous are of nearly equal intensity. The quantitative estimation of elements is presented in Table 2.
Figure 2.
SEM–EDX analysis micrographs showing elemental composition of C. roseus. (A): field grown anther; (B): anther-derived callus.
Table 2.
Elemental composition of anther and anther-derived callus of C. roseus using SEM–EDX analysis.
| S.No. | Element | Anther Explant | Anther-Derived Callus | ||
|---|---|---|---|---|---|
| Weight % | Atomic % | Weight % | Atomic % | ||
| 1 | Carbon | 33.59 | 70.67 | 47.34 | 79.42 |
| 2 | Oxygen | 12.65 | 19.97 | 11.55 | 14.55 |
| 3 | Sodium | 1.93 | 2.12 | 1.63 | 1.42 |
| 4 | Phosphorous | 0.87 | 0.71 | 1.03 | 0.67 |
2.3. GC–MS Analysis
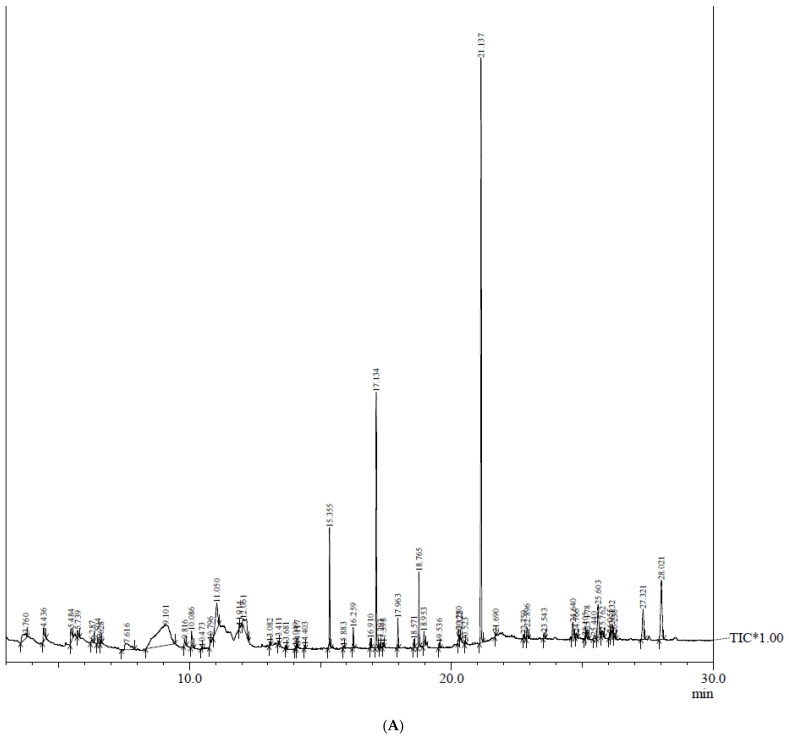
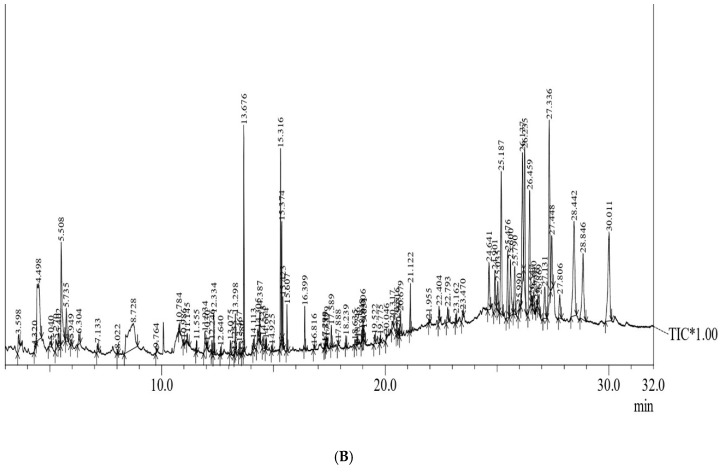
The bioactive compounds present in methanolic extracts of anthers (donor material) and anther-derived callus of C. roseus (Figure 3) were identified using the GC–MS technique. The active principles with their retention time (RT), peak area % (concentration), molecular formula and molecular weight from the NIST library are presented in Table 3 and Table 4, and the GC–MS chromatograms are presented in Figure 4A,B. The chromatograms reveal more than 50 phytocompounds in both methanolic extracts belonging to various classes such as terpenoids, phenols, lignans, steroids, alkaloids and fatty acids.
Figure 3.
Extract preparation for GC–MS analysis of C. roseus. (A): dried powder of anther-derived callus; (B): dried powder of field-grown anther; (C): methanolic extracts of the samples (A,B).
Table 3.
List of phytocompounds identified in the methanolic extract of field-grown anther of C. roseus using GC–MS analysis.
| S.No. | RT (min) | Peak Area % | Name of the Compound | Molecular Formula | Molecular Weight |
|---|---|---|---|---|---|
| 1 | 3.760 | 1.62 | Ethylcyclopentenolone | C7H10O2 | 126 |
| 2 | 4.436 | 1.12 | Pyranone | C6H8O4 | 144 |
| 3 | 5.484 | 1.17 | Coumaran | C8H8O | 120 |
| 4 | 5.739 | 0.42 | 1-monoacetin | C5H10O4 | 134 |
| 5 | 6.287 | 0.26 | 6-oxoheptanoic acid | C7H12O3 | 144 |
| 6 | 6.504 | 0.38 | Indole | C8H7N | 117 |
| 7 | 6.628 | 0.14 | 4-vinylguaiacol | C9H10O2 | 150 |
| 8 | 7.616 | 2.12 | 1,2-octanediol | C8H18O2 | 146 |
| 9 | 9.101 | 18.42 | Guanosine | C10H13N5O5 | 283 |
| 10 | 9.816 | 0.45 | 2,6-dimethoxy-4-vinylphenol | C10H12O3 | 180 |
| 11 | 10.086 | 0.78 | 1,2-benzenedicarboxylic acid, diethyl ester | C12H14O4 | 222 |
| 12 | 10.473 | 0.09 | Cedrol | C15H26O | 222 |
| 13 | 10.796 | 0.17 | Dihydromethyljasmonate | C13H22O3 | 226 |
| 14 | 11.050 | 3.78 | Quinic acid | C7H12O6 | 192 |
| 15 | 11.914 | 0.08 | 2-benzylideneoctanal | C15H20O | 216 |
| 16 | 12.061 | 2.86 | Mome inositol | C7H14O6 | 194 |
| 17 | 13.082 | 0.13 | Diisobutyl phthalate | C16H22O4 | 278 |
| 18 | 13.411 | 0.11 | Heptadecane | C17H36 | 240 |
| 19 | 13.681 | 0.09 | Methyl palmitate | C17H34O2 | 270 |
| 20 | 14.117 | 0.18 | n-hexadecanoic acid | C16H32O2 | 256 |
| 21 | 14.403 | 0.12 | Eicosane | C20H42 | 282 |
| 22 | 15.355 | 4.71 | Hexacosane | C26H54 | 366 |
| 23 | 15.883 | 0.09 | Docosanoic acid | C22H44O2 | 340 |
| 24 | 16.259 | 0.79 | Tetracosane | C24H50 | 338 |
| 25 | 16.910 | 0.35 | 9-tricosanol acetate | C25H50O2 | 382 |
| 26 | 17.134 | 9.78 | Hexatriacontane | C36H74 | 506 |
| 27 | 17.293 | 0.20 | 4,5-dihydro-2-[(8Z,11Z)-8,11-heptadecadienyl]oxazole | C20H35NO | 305 |
| 28 | 17.398 | 0.19 | 4,8-cyclododecadien-1-one | C12H18O | 178 |
| 29 | 17.963 | 1.13 | Dotriacontane | C32H66 | 450 |
| 30 | 18.571 | 0.30 | Octacosanol | C28H58O | 410 |
| 31 | 18.765 | 2.82 | n-tetracontane | C40H82 | 562 |
| 32 | 18.953 | 0.80 | alpha-monostearin | C21H42O4 | 358 |
| 33 | 19.536 | 0.22 | 1-bromotriacontane | C30H61Br | 500 |
| 34 | 20.322 | 0.11 | Linoleyl acetate | C20H36O2 | 308 |
| 35 | 20.523 | 0.12 | (-)-Coronaridine | C21H26N2O2 | 338 |
| 36 | 21.137 | 27.01 | Squalene | C30H50 | 410 |
| 37 | 22.759 | 0.19 | Arachidic acid, 3-methylbutyl ester | C25H50O2 | 382 |
| 38 | 22.896 | 0.57 | beta-tocopherol | C28H48O2 | 416 |
| 39 | 23.543 | 0.30 | Vitamin E | C29H50O2 | 430 |
| 40 | 24.640 | 1.20 | Campesterol | C28H48O | 400 |
| 41 | 24.766 | 0.30 | Ergostan-3-ol | C28H50O | 402 |
| 42 | 25.105 | 0.08 | Trans-24-ethylidenecholesterol | C29H48O | 412 |
| 43 | 25.178 | 0.52 | 3-oxocholestane | C27H46O | 386 |
| 44 | 25.440 | 0.22 | p-coumaric acid, 2-methylpropyl ether, 2-methylpropyl ester | C17H24O3 | 276 |
| 45 | 25.603 | 2.87 | gamma-sitosterol | C29H50O | 414 |
| 46 | 25.762 | 0.57 | Stigmastanol | C29H52O | 416 |
| 47 | 26.055 | 0.16 | Ergosta-4,24(28)-dien-3-one | C28H44O | 396 |
| 48 | 26.132 | 0.87 | 4-campestene-3-one | C28H46O | 398 |
| 49 | 26.230 | 0.23 | Cholestanone | C27H46O | 386 |
| 50 | 27.321 | 2.72 | Methyl commate C | C31H50O4 | 486 |
| 51 | 28.021 | 5.54 | alpha amyrin | C30H50O | 426 |
Table 4.
List of phytocompounds identified in the methanolic extract of anther-derived callus of C. roseus using GC–MS analysis.
| S.No. | RT (min) | Peak Area % | Name of the Compound | Molecular Formula | Molecular Weight |
|---|---|---|---|---|---|
| 1 | 3.598 | 0.58 | 1,3,5-triazine-2,4,6-triamine | C3H6N6 | 126 |
| 2 | 4.320 | 0.10 | Isopropylmethylnitrosamine | C4H10N2O | 102 |
| 3 | 4.498 | 5.49 | 1,2,3-propanetriol | C3H8O3 | 92 |
| 4 | 5.040 | 0.23 | 3-cis-methoxy-5-trans-methyl-1R-cyclohexanol | C8H16O2 | 144 |
| 5 | 5.270 | 0.35 | Catechol | C6H6O2 | 110 |
| 6 | 5.402 | 0.50 | 2,5,5-trimethylhepta-2,6-dien-4-ol | C10H18O | 154 |
| 7 | 5.508 | 3.89 | 5-hydroxymethylfurfural | C6H6O3 | 126 |
| 8 | 5.735 | 1.06 | 1-monoacetin | C5H10O4 | 134 |
| 9 | 5.949 | 0.15 | Decanoic acid | C10H20O2 | 172 |
| 10 | 6.304 | 0.40 | 4-oxopentyl acetate | C7H12O3 | 144 |
| 11 | 7.133 | 0.24 | Eugenol acetate | C12H14O3 | 206 |
| 12 | 8.022 | 0.07 | Indan-1,3-diol monoacetate | C11H12O3 | 192 |
| 13 | 8.728 | 6.16 | Guanosine | C10H13N5O5 | 283 |
| 14 | 9.764 | 0.08 | Dodecanoic acid | C12H24O2 | 200 |
| 15 | 10.784 | 0.15 | Dihydromethyljasmonate | C13H22O3 | 226 |
| 16 | 10.986 | 0.10 | 1-(4-isopropylphenyl)-2-methylpropyl acetate | C15H22O2 | 234 |
| 17 | 11.145 | 0.27 | Benzoic acid, 2-hydroxy-, heptyl ester | C14H20O3 | 236 |
| 18 | 11.555 | 0.19 | Methyl myristate | C15H30O2 | 242 |
| 19 | 11.934 | 0.61 | 4-((1E)-3-hydroxy-1-propenyl)-2-methoxyphenol | C10H12O3 | 180 |
| 20 | 12.030 | 0.11 | Tridecanoic acid | C13H26O2 | 214 |
| 21 | 12.246 | 0.20 | Stearic acid methyl ester | C19H38O2 | 298 |
| 22 | 12.334 | 0.72 | Octadecanoic acid, methyl ester | C19H38O2 | 298 |
| 23 | 12.640 | 0.14 | Pentadecanoic acid, methyl ester | C16H32O2 | 256 |
| 24 | 13.075 | 0.29 | Diisobutyl phthalate | C16H22O4 | 278 |
| 25 | 13.255 | 0.03 | 1-hexadecanol | C16H34O | 242 |
| 26 | 13.298 | 0.62 | Hexadecanoic acid, methyl ester | C17H34O2 | 270 |
| 27 | 13.467 | 0.19 | Methyl palmitoleate | C17H32O2 | 268 |
| 28 | 13.580 | 0.03 | 7,9-di-tert-butyl-1-oxaspiro(4,5)deca-6,9-diene-2,8-dione | C17H24O3 | 276 |
| 29 | 13.676 | 3.96 | Methyl palmitate | C17H34O2 | 270 |
| 30 | 14.113 | 0.21 | n-hexadecanoic acid | C16H32O2 | 256 |
| 31 | 14.305 | 0.49 | Decyl hexofuranoside | C16H32O6 | 320 |
| 32 | 14.387 | 0.50 | Eicosanoic acid, methyl ester | C21H42O2 | 326 |
| 33 | 14.533 | 0.36 | Cis-sinapyl alcohol | C11H14O4 | 210 |
| 34 | 14.664 | 0.15 | Heptadecanoic acid, methyl ester | C18H36O2 | 284 |
| 35 | 14.925 | 0.12 | Oxybenzone | C14H12O3 | 228 |
| 36 | 15.316 | 3.31 | Linoleic acid, methyl ester | C19H34O2 | 294 |
| 37 | 15.374 | 1.97 | Ethyl oleate | C20H38O2 | 310 |
| 38 | 15.423 | 0.72 | Oleic acid, methyl ester | C19H36O2 | 296 |
| 39 | 15.607 | 0.79 | Octadecanoic acid, methyl ester | C19H38O2 | 298 |
| 40 | 16.399 | 0.70 | cis-10-nonadecenoic acid, methyl ester | C20H38O2 | 310 |
| 41 | 16.816 | 0.08 | 4,8,13-duvatriene-1,3-diol | C20H34O2 | 306 |
| 42 | 17.290 | 0.08 | 4,5-dihydro-2-[(8Z,11Z)-8,11-heptadecadienyl]oxazole | C20H35NO | 305 |
| 43 | 17.335 | 0.03 | (Z)-2-(pentadec-8-en-1-yl)-4,5-dihydrooxazole | C18H33NO | 279 |
| 44 | 17.379 | 0.16 | Methyl arachidate | C21H42O2 | 326 |
| 45 | 17.589 | 0.45 | 6-methyladenine, TMS derivative | C9H15N5Si | 221 |
| 46 | 17.888 | 0.09 | Octadecanoic acid, 2,3-dihydroxypropyl ester | C21H42O4 | 358 |
| 47 | 18.239 | 0.15 | Henicosanal | C21H42O | 310 |
| 48 | 18.746 | 0.12 | Nonadecylpentafluoropropionate | C22H39F5O2 | 430 |
| 49 | 18.948 | 0.21 | alpha-monostearin | C21H42O4 | 358 |
| 50 | 19.006 | 0.28 | Docosanoic acid, methyl ester | C23H46O2 | 354 |
| 51 | 19.522 | 0.21 | Vindolinine | C21H24N2O2 | 336 |
| 52 | 19.775 | 0.12 | Methyl tricosanoate | C24H48O2 | 368 |
| 53 | 20.046 | 0.09 | Octocrylene | C24H27NO2 | 361 |
| 54 | 20.317 | 0.25 | n-propyl linoleate | C21H38O2 | 322 |
| 55 | 20.593 | 0.10 | Pleiocarpamine | C20H22N2O2 | 322 |
| 56 | 21.122 | 0.89 | Squalene | C30H50 | 410 |
| 57 | 22.404 | 0.36 | (+)-Pericyclivine | C20H22N2O2 | 322 |
| 58 | 22.793 | 0.47 | Ajmalicine | C21H24N2O3 | 352 |
| 59 | 23.162 | 0.20 | Cholesta-4,6-dien-3-ol | C27H44O | 384 |
| 60 | 23.470 | 0.24 | Ajmalicine oxindole | C21H24N2O4 | 368 |
| 61 | 24.641 | 1.69 | Campesterol | C28H48O | 400 |
| 62 | 24.901 | 1.23 | Stigmasta-5,20(22)-dien-3-ol | C29H48O | 412 |
| 63 | 25.035 | 0.84 | 19-epiajmalicine | C21H24N2O3 | 352 |
| 64 | 25.187 | 5.32 | 3-oxocholestane | C27H46O | 386 |
| 65 | 25.476 | 2.09 | beta-stigmasterol | C29H48O | 412 |
| 66 | 25.600 | 2.42 | gamma-sitosterol | C29H50O | 414 |
| 67 | 25.790 | 1.76 | (E)-1-(6,10-dimethylundec-5-en-2-yl)-4-methylbenzene | C20H32 | 272 |
| 68 | 25.990 | 0.30 | (22E)-ergosta-4,7,22-trien-3-one | C28H42O | 394 |
| 69 | 26.137 | 4.88 | 4-campestene-3-one | C28H46O | 398 |
| 70 | 26.235 | 4.62 | Cholestanone | C27H46O | 386 |
| 71 | 26.459 | 4.47 | Stigmasterone | C29H46O | 410 |
| 72 | 26.547 | 0.22 | 6-dehydroprogesterone | C21H28O2 | 312 |
| 73 | 26.640 | 0.56 | Cycloartenol | C30H50O | 426 |
| 74 | 26.776 | 0.20 | 3,5-cholestadien-7-one | C27H42O | 382 |
| 75 | 26.869 | 0.61 | Ergosta-4,6,22-trien-3-one | C28H42O | 394 |
| 76 | 27.336 | 7.08 | gamma-sitostenone | C29H48O | 412 |
| 77 | 27.448 | 1.97 | 24-methylenecycloartanol | C31H52O | 440 |
| 78 | 27.806 | 0.93 | Stigmasta-3,5-dien-7-one | C29H46O | 410 |
| 79 | 28.442 | 5.87 | 4,4-dimethylcholestan-3-one | C29H50O | 414 |
| 80 | 28.846 | 3.78 | (22E)-4-methylstigmast-22-en-3-one | C30H50O | 426 |
| 81 | 30.011 | 5.55 | 3-acetylcholestan-2-one | C29H48O2 | 428 |
Figure 4.
(A): GC–MS chromatogram (total ionic chromatogram) of methanolic extract of anthers of C. roseus; (B): GC–MS chromatogram (total ionic chromatogram) of methanolic extract of anther-derived callus of C. roseus.
Among the compounds identified, 1-monoacetin, guanosine, dihydromethyljasmonate, n-hexadecanoic acid, squalene, campesterol, cholestanone and gamma-sitosterol were the most prevalent present in both extracts. Only the methanolic extract of anthers contained bioactives such as cedrol (0.09%), (-)-coronaridine (0.12%), 4-vinylguaiacol (0.14%), vitamin E (0.30%), stigmastanol (0.57%), quinic acid (3.78%) and alpha amyrin (5.54%) (Table 3), and their respective mass spectra are shown in Figure S1A. The extract of anther-derived calli was found to have characteristic metabolites such as pleiocarpamine (0.10%), vindolinine (0.21%), cis-sinapyl alcohol (0.36%), (+)-pericyclivine (0.36%), ajmalicine (0.47%), cycloartenol (0.56%) and beta-stigmasterol (2.09%) (Table 4 and Table 5) having specific mass spectra (Figure S1B).
Table 5.
List of important phytocompounds identified exclusively in the methanolic extract of anther-derived callus of C. roseus using GC–MS analysis.
| S.No. | RT (min) | Name of the Compound | Molecular Formula |
|---|---|---|---|
| 1 | 7.133 | Eugenol acetate | C12H14O3 |
| 2 | 12.246 | Stearic acid methyl ester | C19H38O2 |
| 3 | 14.533 | Cis-sinapyl alcohol | C11H14O4 |
| 4 | 14.925 | Oxybenzone | C14H12O3 |
| 5 | 15.316 | Linoleic acid, methyl ester | C19H34O2 |
| 6 | 15.423 | Oleic acid, methyl ester | C19H36O2 |
| 7 | 19.522 | Vindolinine | C21H24N2O2 |
| 8 | 20.046 | Octocrylene | C24H27NO2 |
| 9 | 20.593 | Pleiocarpamine | C20H22N2O2 |
| 10 | 22.404 | (+)-Pericyclivine | C20H22N2O2 |
| 11 | 22.793 | Ajmalicine | C21H24N2O3 |
| 12 | 25.035 | 19-epiajmalicine | C21H24N2O3 |
| 13 | 25.476 | beta-stigmasterol | C29H48O |
| 14 | 26.459 | Stigmasterone | C29H46O |
| 15 | 26.547 | 6-dehydroprogesterone | C21H28O2 |
| 16 | 26.640 | Cycloartenol | C30H50O |
| 17 | 27.336 | gamma-sitostenone | C29H48O |
2.4. Flow Cytometric Analysis
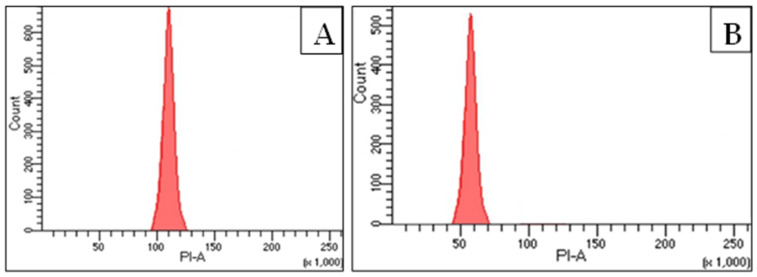
The ploidy status of callus obtained from anther was determined using a flow cytometric approach wherein good quality nuclei are a necessity. In this study, the leaves of field-grown C. roseus were utilized as an external standard reference (control). The flow cytometric histogram peak of callus reveals that its DNA content was nearly half to that of its diploid counterpart (control) (Figure 5A,B). The nuclear DNA content of anther-derived cell/callus was 0.76 pg compared to the diploid leaves’ DNA (1.51 pg) with a DNA Index (DI) of 0.51 (Table 6). This estimation confirms the haploid DNA status of callus obtained from anther.
Figure 5.
Flow cytometric histograms revealing ploidy level of (A) diploid leaves of C. roseus (standard) and (B) anther-derived callus of C. roseus.
Table 6.
Estimation of nuclear DNA content, genome size and DNA index of anther-derived callus with respect to donor plant of C. roseus using flow cytometry technique.
| Plant Sample Type | Nuclear DNA Content (pg) | Genome Size (Mbp) * | DNA Index (DI) ** |
|---|---|---|---|
| Standard (leaves) | 1.51 | 1476.7 | - |
| Anther-derived callus | 0.76 | 743.2 | 0.51 |
* 1 pg = 978 Mbp [18]. ** DNA Index = sample DNA content/standard DNA content.
3. Discussion
The present work was conducted to evaluate the callusing potentiality of anthers of C. roseus under in vitro culture conditions. The type and concentration of PGRs used in media strongly affect callusing ability and are different in different plant species. Initially, the anthers were subject to different concentrations and combinations of PGRs amended in MS medium. The results indicate that a high-to-low ratio of auxin: cytokinin concentrations was proven to be the best in inducing callus with a maximum mean fresh weight, which is very similar to Kou et al.’s [19] and Rout et al.’s [20] observations. Likewise, TDZ alone at different concentrations was found to be equally effective in producing callus and subsequent proliferation. Previous reports suggested that TDZ (a cytokinin-like PGR) alone may be used in improving callusing ability in different explants [21,22]. A comparison of the elemental distribution on the surfaces of anther and anther-derived callus was performed using SEM–EDX analysis, revealing a nearly similar elemental composition on both samples. EDX analyzes X-rays emitted from samples receiving a high-energy electron beam. This technique facilitates the qualitative and semi-quantitative detection of surface elements of samples and has been extensively used on various plant species such as sesame [23] and lemongrass [24].
Medicinal plants are an ingenious source of bioactive compounds that fight against several chronic diseases, and these phytocompounds can be identified and quantified using the GC–MS technique [25]. In the current study, phytochemical profiling with GC–MS of methanolic extracts (Figure 5) of anther and anther-derived callus of C. roseus has been conducted. The results obtained show the presence of various phytoconstituents, including carbohydrates, alkaloids, phenols, saponins, phytosterols, terpenoids, steroids, etc. A total of 14 bioactives are common in both the extracted samples. However, there are compounds that are exclusive to each sample that confer various biological properties to this plant. The presence of secondary metabolites in callus, which are otherwise not detected in anther tissue, may be due to the fact that certain bioactive compounds accumulate in specific cells or tissues or in a specific growth stage (mostly the stationary phase) of in vitro cultures [26]. Therefore, developing callus from different tissues to obtain therapeutically active compounds is of high significance.
The major compounds of medicinal value present in the methanolic extract of anthers were squalene (triterpene), alpha-amyrin (triterpene), coronaridine (alkaloid) and cedrol (essential oil), which possess anti-oxidant, gastroprotective and hepatoprotective, anti-cancerous and anti-inflammatory properties, respectively [27,28,29,30]. Similarly, in anther-derived calli exclusively, 17 compounds are present having diverse medicinal properties, and these compounds are listed in Table 5. These include stearic acid, linoleic acid, oleic acid, vindolinine, pleiocarpamine, pericyclivine, ajmalicine, 19-epiajmalicine, beta-stigmasterol, cycloartenol, etc. Ajmalicine and vindolinine are well-known alkaloids having anti-cancerous, anti-hypertensive and anti-oxidant properties [15,31]. Recently, an alkaloid named pleiocarpamine has been isolated from the stem bark of Rauvolfia caffra and is reported to possess anti-seizure activity [32]. Cycloartenol (a triterpenoid) and stigmasterol (a sterol) have also been detected in present studies and are associated with immunosuppressive, anti-hypercholestrolemic and anti-inflammatory activities, respectively [33,34]. Compounds such as cycloartenol, ajmalicine, vindolinine, pleiocarpamine and pericyclivine have been reported previously in leaf tissues of C. roseus [35,36]. Some reports of phytocompounds identified from different tissues using GC–MS were noted earlier [37,38], but till now, no information on the phytocompounds present in anther or anther-derived callus was available for C. roseus.
The ploidy status of anther-derived callus was checked using flow cytometry, and the results show that the ploidy of the calli was haploid in nature, confirming the involvement of microspores in developing callus. Similar observations have also been reported for other plant species [7,10,39]. FCM is the widely used approach for determining the ploidy of plants developed through callus, somatic embryos and other in vitro-regenerated pathways [40]. The origin of diploid plants from anthers may be due to the involvement of other somatic cells such as anther wall, filament or flower septum in developing callus. Spontaneous chromosomal doubling can also be a mechanism in the generation of polyploidy in anther-derived regenerants. In certain cases, mixoploids and aneuploids have also been noted in anther cultures of different plants [8,41], but these polyploids were not detected in this experiment. This is the first-ever report of GC–MS analysis of medically significant compounds from anther tissue of C. roseus, which enriches the phytocompound library of Catharanthus and may be utilized in the pharmaceutical and industrial sectors.
4. Materials and Methods
4.1. Anther Culture and Growth Conditions
The mature flowers of C. roseus were collected from the herbal garden, Jamia Hamdard, New Delhi, and the anthers were used as explants for experimentations. The surface sterilization of flowers was performed following the method of Bansal et al. [3] described earlier. The sterilized anthers were excised from the flowers and aseptically cultured onto agar-solidified basal Murashige and Skoog (MS) medium supplemented with various concentrations and combinations of plant growth regulators (PGRs) and sub-cultured every 3–4 weeks. The cultures were incubated at a temperature of 24 ± 2 °C with 48 μmol/m2/s2 illumination (white fluorescent light) for a 16 h photoperiod.
4.2. Callus Induction and Proliferation
The disinfected anthers were inoculated on MS augmented with different concentrations (alone or in combination) of α-naphthalene acetic acid (NAA), kinetin (Kn) and thidiazuron (TDZ) ranging from 0.1 to 1.0 mg/L for callus induction. Callus formation started within 14–16 days of culture and proliferated on the same medium with successive subculturing. The callus induction frequency and the callus fresh weight were recorded after 6 weeks of culture.
4.3. Surface Morphology and Elemental Analysis
The surface morphology and elemental profile of anther and anther-derived callus were determined using energy-dispersive X-ray microanalysis (EDX) combined with scanning electron microscopy (SEM). For this purpose, the samples were primarily fixed with Karnovsky’s fixative and washed with 0.1 M phosphate buffer at 4 °C. Afterward, a series of dehydrations with acetone (30%, 50%, 70%, 90% and 100%) were performed at 15 min intervals, and then critical-point drying was performed at 1100 p.s.i. These samples were then mounted on aluminum stubs and sputter-coated with gold having a 35 nm thick film. Finally, the coated samples were viewed at an accelerating voltage of 20 kV under a scanning electron microscope (Zeiss, Oberkochen, Germany) equipped with EDAX.
4.4. Preparation of Extracts
The methanolic extracts of both samples were prepared according to the protocol of Hussain et al. [42]. About 1.0 g of anther and anther callus were shade dried and crushed into fine powder using mortar and pestle (Figure 3A,B). Each sample was then extracted in 5.0 mL methanol in an orbital shaker for 48 h. Afterward, the extracts were filtered through Whatman filter paper no. 1 and evaporated to dryness. The obtained extracts were stored in an airtight container with proper labeling at 4 °C for further use (Figure 3C).
4.5. GC–MS Analysis
GC–MS analyses of these extracts were conducted on GC–MS QP-2010 equipment (Shimadzu, Japan) at Advanced Instrumentation Research Facility (AIRF), JNU, New Delhi. The program settings were as follows: Helium was used as a carrier gas (1 mL/min), and the initial and final temperatures were programmed at 100 °C and 260 °C, respectively, with a hold time of 18 min. Ion source temperature was 220 °C with an interface temperature of 270 °C and solvent cut time of 2.5 min. Other specifications included: detector gain mode relative to the tuning result, detector gain +0.00 kV, threshold of 1000, start time 3 min, end time 39.98 min, event time 0.3 s, scan speed of 2000, start m/z 40.00 and end m/z 600.00.
4.6. Metabolite Data Processing and Analysis
The bioactive compounds were identified using the mass spectral database of the NIST17 library. The unknown compounds’ spectra were compared with the known phytocompound spectra available in the NIST library, and the name, molecular weight and structure of the compounds were determined.
4.7. Flow Cytometric Analysis
The ploidy status of anther-derived calli was examined using the flow cytometry method as described by Galbraith [43]. A total of 3 samples of anther-derived callus were randomly chosen, along with a reference standard of diploid leaves of C. roseus with a known 2C DNA content of 1.51 pg [44]. Approximately 50 mg of callus was added to a Petri plate having 1.0 mL ice-cold Galbraith’s buffer (nuclei isolation buffer) and finely macerated with the help of a surgical blade. The homogenate was then filtered with a 100 µm nylon mesh to eliminate larger cellular remnants and was finally stained with 50 µg/mL PI RNase (propidium iodide RNase) (Sigma-Aldrich, St. Louis, MO, USA) for 8–10 min. The samples were incubated in the dark at 4 °C for about 40 min and eventually examined on a BD FACS(Calibur) flow cytometer (BD Biosciences, Franklin Lakes, NJ, USA). The relative nuclear DNA of anther-derived callus of C. roseus was estimated using the below formula [45]:
4.8. Statistical Analysis
In the tissue-culture experiment, three explants (anthers) per culture tube were inoculated with five replicates of every experimental treatment, and each experiment was repeated twice. The data are expressed as mean ± standard error, and the analysis was performed using one-way analysis of variance (ANOVA). The significance of mean difference was determined using Duncan’s multiple range test (DMRT) at p < 0.05 using SPSS Ver. 26.0 (SPSS Inc., Chicago, IL, USA) [46]. The flow cytometric study was repeated thrice with randomly chosen standard (donor plant) and callus samples.
5. Conclusions
The in vitro culture technology was successfully employed to obtain callus from anther tissue of C. roseus, an important medicinal plant. The callus was checked for its ploidy status using flow cytometry and was found to be haploid in nature. The calli obtained from anther were then subjected to GC–MS analysis for phytocompound identification. Among the bioactive compounds identified, ajmalicine, vindolinine, pleiocarpamine, pericyclivine, stigmasterol, campesterol and squalenes were detected and have a wide range of biological activities. From this study, it can then be concluded that anther-derived calli are a potent source for developing new therapeutic drugs with larger-scale applicability in pharmaceutical sectors.
Acknowledgments
The first author is thankful to the Department of Biotechnology (DBT) for the financial support given in the form of the Junior Research Fellowship and to AIRF, JNU, New Delhi, for providing the GC–MS facility. The authors are grateful for the laboratory facilities provided by the Department of Botany, Jamia Hamdard, New Delhi. The authors acknowledge Researchers Supporting Project number (RSP2023R375), King Saud University, Riyadh, Saudi Arabia.
Abbreviations
| PGRs | Plant Growth Regulators |
| MS | Murashige and Skoog |
| NAA | α-Naphthaleneacetic acid |
| Kn | Kinetin |
| TDZ | Thidiazuron |
| DH | Double Haploid |
| SEM–EDX | Scanning Electron Microscopy–Energy-Dispersive X-ray |
| FCM | Flow Cytometric Method |
| DI | DNA Index |
| GC–MS | Gas Chromatography–Mass Spectrometry |
| DMRT | Duncan’s Multiple Range Test |
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/plants12112186/s1, Figure S1A,B: Mass spectra of identified compounds from methanolic extract of anthers and anther derived callus of C. roseus.
Author Contributions
Conceptualization, A.M. and Y.B.; methodology, Y.B.; formal analysis, Y.B.; investigation, Y.B.; data curation, Y.B.; writing—original draft preparation, Y.B.; writing—review and editing, Y.B., A.M., J.M., Y.H.D. and H.Z.R.; validation, Y.H.D. and H.Z.R.; visualization, Y.H.D. and H.Z.R.; supervision, A.M.; project administration, A.M. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
All data are presented in the article.
Conflicts of Interest
The authors declare no conflict of interest.
Funding Statement
This research work is funded by the Department of Biotechnology (DBT), New Delhi, India (DBT/2020/JH/1336).
Footnotes
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
References
- 1.Das A., Sarkar S., Bhattacharyya S., Gantait S. Biotechnological advancements in Catharanthus roseus (L.) G. Don. Appl. Microbiol. Biotechnol. 2020;10:44811–44835. doi: 10.1007/s00253-020-10592-1. [DOI] [PubMed] [Google Scholar]
- 2.Dhayanithy G., Subban K., Chelliah J. Diversity and biological activities of endophytic fungi associated with Catharanthus roseus. BMC Microbiol. 2019;19:1–14. doi: 10.1186/s12866-019-1386-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bansal Y., Mujib A., Siddiqui Z.H., Mamgain J., Syeed R., Ejaz B. Ploidy status, nuclear DNA content and start codon targeted (SCoT) genetic homogeneity assessment in Digitalis purpurea L., regenerated in vitro. Genes. 2022;13:2335. doi: 10.3390/genes13122335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mujib A., Bansal Y., Malik M.Q., Syeed R., Mamgain J., Ejaz B. Internal and external regulatory elements controlling somatic embryogenesis in Catharanthus: A model medicinal plant. In: Ramirez-Mosqueda M.A., editor. Somatic Embryogenesis, Methods in Molecular Biology. Volume 2527. Humana; New York, NY, USA: 2022. pp. 11–27. [DOI] [PubMed] [Google Scholar]
- 5.Dhandapani M., Kim D.H., Hong S.B. Efficient plant regeneration via somatic embryogenesis and organogenesis from the explants of Catharanthus roseus. In Vitro Cell. Dev. Biol. Plant. 2008;44:18–25. doi: 10.1007/s11627-007-9094-x. [DOI] [Google Scholar]
- 6.Verma P., Mathur A.K. Direct shoot bud organogenesis and plant regeneration from pre-plasmolysed leaf explants in Catharanthus roseus. Plant Cell Tissue Organ Cult. 2011;106:401–408. doi: 10.1007/s11240-011-9936-4. [DOI] [Google Scholar]
- 7.Hoveida Z.S., Abdollahi M.R., Mirzaie-Asl A., Moosavi S.S., Seguí-Simarro J.M. Production of doubled haploid plants from anther cultures of borage (Borago officinalis L.) by the application of chemical and physical stress. Plant Cell Tissue Organ Cult. 2017;130:369–378. doi: 10.1007/s11240-017-1233-4. [DOI] [Google Scholar]
- 8.Wang G.F., Qin H.Y., Sun D., Fan S.T., Yang Y.M., Wang Z.X., Xu P.L., Zhao Y., Liu Y.X., Ai J. Haploid plant regeneration from hardy kiwifruit (Actinidia arguta Planch.) anther culture. Plant Cell Tissue Organ Cult. 2018;134:15–28. doi: 10.1007/s11240-018-1396-7. [DOI] [Google Scholar]
- 9.Abd El-Fatah B.E., Sayed M.A., El-Sanusy S.A. Genetic analysis of anther culture response and identification of QTLs associated with response traits in wheat (Triticum aestivum L.) Mol. Biol. Rep. 2020;47:9289–9300. doi: 10.1007/s11033-020-06007-z. [DOI] [PubMed] [Google Scholar]
- 10.Sahoo S.A., Jha Z., Verulkar S.B., Srivastava A.K., Suprasanna P. High-throughput cell analysis based protocol for ploidy determination in anther-derived rice callus. Plant Cell Tissue Organ Cult. 2019;137:187–192. doi: 10.1007/s11240-019-01561-2. [DOI] [Google Scholar]
- 11.Ejaz B., Mujib A., Mamgain J., Malik M.Q., Syeed R., Gulzar B., Bansal Y. Comprehensive in vitro regeneration study with SCoT marker assisted clonal stability assessment and flow cytometric genome size analysis of Carthamus tinctorius L.: An important medicinal plant. Plant Cell Tissue Organ Cult. 2022;148:403–418. doi: 10.1007/s11240-021-02197-x. [DOI] [Google Scholar]
- 12.Bhusare B.P., John C.K., Bhatt V.P., Nikam T.D. Induction of somatic embryogenesis in leaf and root explants of Digitalis lanata Ehrh.: Direct and indirect method. S. Afr. J. Bot. 2020;130:356–365. doi: 10.1016/j.sajb.2020.01.012. [DOI] [Google Scholar]
- 13.Mamgain J., Mujib A., Ejaz B., Gulzar B., Malik M.Q., Syeed R. Flow cytometry and start codon targeted (SCoT) genetic fidelity assessment of regenerated plantlets in Tylophora indica (Burm. f.) Merrill. Plant Cell Tissue Organ Cult. 2022;150:129–140. doi: 10.1007/s11240-022-02254-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Konappa N., Udayashankar A.C., Krishnamurthy S., Pradeep C.K., Chowdappa S., Jogaiah S. GC–MS analysis of phytoconstituents from Amomum nilgiricum and molecular docking interactions of bioactive serverogenin acetate with target proteins. Sci. Rep. 2020;10:16438. doi: 10.1038/s41598-020-73442-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Pham H.N., Vuong Q.V., Bowyer M.C., Scarlett C.J. Phytochemicals derived from Catharanthus roseus and their health benefits. Technology. 2020;8:80. doi: 10.3390/technologies8040080. [DOI] [Google Scholar]
- 16.Padma M., Ganesan S., Jayaseelan T., Azhagumadhavan S., Sasikala P., Senthilkumar S., Mani P. Phytochemical screening and GC–MS analysis of bioactive compounds present in ethanolic leaves extract of Silybum marianum (L) J. Drug. Deliv. Ther. 2019;9:85–89. doi: 10.22270/jddt.v9i1.2174. [DOI] [Google Scholar]
- 17.Jahan I., Tona M.R., Sharmin S., Sayeed M.A., Tania F.Z., Paul A., Chy M.N., Rakib A., Emran T.B., Simal-Gandara J. GC-MS phytochemical profiling, pharmacological properties, and in silico studies of Chukrasia velutina leaves: A novel source for bioactive agents. Molecules. 2020;25:3536. doi: 10.3390/molecules25153536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Dolezel J. Nuclear DNA content and genome size of trout and human. Cytom. Part A. 2003;51:127–128. doi: 10.1002/cyto.a.10013. [DOI] [PubMed] [Google Scholar]
- 19.Kou Y., Ma G., Teixeira da Silva J.A., Liu N. Callus induction and shoot organogenesis from anther cultures of Curcuma attenuata Wall. Plant Cell Tissue Organ Cult. 2013;112:1–7. doi: 10.1007/s11240-012-0205-y. [DOI] [Google Scholar]
- 20.Rout P., Naik N., Ngangkham U., Verma R.L., Katara J.L., Singh O.N., Samantaray S. Doubled Haploids generated through anther culture from an elite long duration rice hybrid, CRHR32: Method optimization and molecular characterization. Plant Biotechnol. 2016;33:177–186. doi: 10.5511/plantbiotechnology.16.0719a. [DOI] [Google Scholar]
- 21.Cappelletti R., Sabbadini S., Mezzetti B. The use of TDZ for the efficient in vitro regeneration and organogenesis of strawberry and blueberry cultivars. Sci. Hortic. 2016;207:117–124. doi: 10.1016/j.scienta.2016.05.016. [DOI] [Google Scholar]
- 22.Khan T., Abbasi B.H., Khan M.A., Shinwari Z.K. Differential effects of thidiazuron on production of anticancer phenolic compounds in callus cultures of Fagonia indica. Appl. Biochem. Biotechnol. 2016;179:46–58. doi: 10.1007/s12010-016-1978-y. [DOI] [PubMed] [Google Scholar]
- 23.Nath B., Kalita P., Das B., Basumatary S. Highly efficient renewable heterogeneous base catalyst derived from waste Sesamum indicum plant for synthesis of biodiesel. Renew. Energy. 2020;151:295–310. doi: 10.1016/j.renene.2019.11.029. [DOI] [Google Scholar]
- 24.Pandey J., Sarkar S., Verma R.K., Singh S. Sub-cellular localization and quantitative estimation of heavy metals in lemongrass plants grown in multi-metal contaminated tannery sludge. S. Afr. J. Bot. 2020;131:74–83. doi: 10.1016/j.sajb.2020.01.034. [DOI] [Google Scholar]
- 25.Mamgain J., Mujib A., Syeed R., Ejaz B., Malik M.Q., Bansal Y. Genome size and gas chromatography-mass spectrometry (GC–MS) analysis of field-grown and in vitro regenerated Pluchea lanceolata plants. J. Appl. Genet. 2022;64:1–21. doi: 10.1007/s13353-022-00727-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Leng T.C., Ping N.S., Lim B.P., Keng C.L. Detection of bioactive compounds from Spilanthes acmella (L.) plants and its various in vitro culture products. J. Med. Plant Res. 2011;5:371–378. [Google Scholar]
- 27.Diab M., Ibrahim A., Hadad G. B: Review article on chemical constituents and biological activity of Olea europaea. Rec. Pharm. Biomed. Sci. 2020;4:36–45. doi: 10.21608/rpbs.2019.19541.1048. [DOI] [Google Scholar]
- 28.Nogueira A.O., Oliveira Y.I.S., Adjafre B.L., de Moraes M.E.A., Aragão G.F. Pharmacological effects of the isomeric mixture of alpha and beta amyrin from Protium heptaphyllum: A literature review. Fundam. Clin. Pharmacol. 2019;33:4–12. doi: 10.1111/fcp.12402. [DOI] [PubMed] [Google Scholar]
- 29.Naidoo C.M., Naidoo Y., Dewir Y.H., Murthy H.N., El-Hendawy S., Al-Suhaibani N. Major bioactive alkaloids and biological activities of Tabernaemontana species (Apocynaceae) Plants. 2021;10:313. doi: 10.3390/plants10020313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Özek G., Schepetkin I.A., Yermagambetova M., Özek T., Kirpotina L.N., Almerekova S.S., Abugalieva S.I., Khlebnikov A.I., Quinn M.T. Innate immunomodulatory activity of cedrol, a component of essential oils isolated from Juniperus species. Molecules. 2021;26:7644. doi: 10.3390/molecules26247644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hemmati N., Azizi M., Spina R., Dupire F., Arouei H., Saeedi M., Laurain-Mattar D. Accumulation of ajmalicine and vinblastine in cell cultures is enhanced by endophytic fungi of Catharanthus roseus cv. Icy Pink. Ind. Crops Prod. 2020;158:112776. doi: 10.1016/j.indcrop.2020.112776. [DOI] [Google Scholar]
- 32.Chipiti T., Viljoen A.M., Cordero-Maldonado M.L., Veale C.G., Van Heerden F.R., Sandasi M., Chen W., Crawford A.D., Enslin G.M. Anti-seizure activity of African medicinal plants: The identification of bioactive alkaloids from the stem bark of Rauvolfiacaffra using an in vivo zebrafish model. J. Ethnopharmacol. 2021;279:114282. doi: 10.1016/j.jep.2021.114282. [DOI] [PubMed] [Google Scholar]
- 33.Bakrim S., Benkhaira N., Bourais I., Benali T., Lee L.-H., El Omari N., Sheikh R.A., Goh K.W., Ming L.C., Bouyahya A. Health Benefits and Pharmacological Properties of Stigmasterol. Antioxidants. 2022;11:1912. doi: 10.3390/antiox11101912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Lun W.U., Zhi-Li C.H., Yang S.U., Qiu-Hong W.A., Kuang H.X. Cycloartenol triterpenoid saponins from Cimicifuga simplex (Ranunculaceae) and their biological effects. China J. Nat. Med. 2015;13:81–89. doi: 10.1016/S1875-5364(15)60011-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Murata J., Roepke J., Gordon H., De Luca V. The leaf epidermome of Catharanthus roseus reveals its biochemical specialization. Plant Cell. 2008;20:524–542. doi: 10.1105/tpc.107.056630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Wesołowska A., Grzeszczuk M., Wilas J., Kulpa D. Gas chromatography-mass spectrometry (GC-MS) analysis of indole alkaloids isolated from Catharanthus roseus (L.) G. don cultivated conventionally and derived from in vitro cultures. Not. Bot. Horti Cluj-Napoca. 2016;44:100–106. doi: 10.15835/nbha44110127. [DOI] [Google Scholar]
- 37.Syeda A.M., Riazunnisa K. Data on GC-MS analysis, in vitro anti-oxidant and anti-microbial activity of the Catharanthus roseus and Moringa oleifera leaf extracts. Data Brief. 2020;29:105258. doi: 10.1016/j.dib.2020.105258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Rani S., Singh V., Sharma M.K., Sisodia R. GC–MS based metabolite profiling of medicinal plant-Catharanthus roseus under cadmium stress. Plant Physiol. Rep. 2021;26:491–502. doi: 10.1007/s40502-021-00595-z. [DOI] [Google Scholar]
- 39.Eshaghi Z.C., Abdollahi M.R., Moosavi S.S., Deljou A., Seguí-Simarro J.M. Induction of androgenesis and production of haploid embryos in anther cultures of borage (Borago officinalis L.) Plant Cell Tissue Organ Cult. 2015;122:321–329. doi: 10.1007/s11240-015-0768-5. [DOI] [Google Scholar]
- 40.Gulzar B., Mujib A., Mushtaq Z., Malik M.Q. Old Catharanthus roseus culture (14 years) produced somatic embryos and plants and showed normal genome size; demonstrated an increased antioxidant defense mechanism; and synthesized stress proteins as biochemical, proteomics, and flow-cytometry studies reveal. J. Appl. Genet. 2021;62:43–57. doi: 10.1007/s13353-020-00590-4. [DOI] [PubMed] [Google Scholar]
- 41.Jia Y., Zhang Q.X., Pan H.T., Wang S.Q., Liu Q.L., Sun L.X. Callus induction and haploid plant regeneration from baby primrose (Primula forbesii Franch.) anther culture. Sci. Hortic. 2014;176:273–281. doi: 10.1016/j.scienta.2014.07.018. [DOI] [Google Scholar]
- 42.Hussain S.A., Ahmad N., Anis M., Alatar A.A. Influence of meta-topolin on in vitro organogenesis in Tecoma stans L., assessment of genetic fidelity and phytochemical profiling of wild and regenerated plants. Plant Cell Tissue Organ Cult. 2019;138:339–351. doi: 10.1007/s11240-019-01631-5. [DOI] [Google Scholar]
- 43.Galbraith D.W. Simultaneous flow cytometric quantification of plant nuclear DNA contents over the full range of described angiosperm 2C values. Cytom. Part A J. Int. Soc. Adv. Cytom. 2009;75:692–708. doi: 10.1002/cyto.a.20760. [DOI] [PubMed] [Google Scholar]
- 44.Mujib A., Malik M.Q., Bansal Y., Syeed R., Ejaz B., Mamgain J. Somatic Embryogenesis in Catharanthus roseus: Proteomics of embryogenic and non-embryogenic tissues; and genome size analysis of regenerated plant. In: Kole C., editor. The Catharanthus Genome, Compendium of Plant Genomes. Springer; Cham, Switzerland: 2022. pp. 85–100. [DOI] [Google Scholar]
- 45.Doležel J., Greilhuber J., Suda J. Estimation of nuclear DNA content in plants using flow cytometry. Nat. Protoc. 2007;2:2233–2244. doi: 10.1038/nprot.2007.310. [DOI] [PubMed] [Google Scholar]
- 46.Duncan D.B. Multiple range and multiple F tests. Biome. 1955;11:1478. doi: 10.2307/3001478. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All data are presented in the article.