Background.
Normothermic machine perfusion (NMP) is an alternative to hypothermic machine perfusion (HMP) for donor kidney preservation before transplantation. Contrary to HMP, NMP allows for functional assessment of donor kidneys because normothermic conditions allow for metabolic activity. The kidneys are key producers of hormones. Yet, it remains unknown whether donor kidneys during NMP display endocrine functions.
Methods.
Fifteen donor kidneys were subjected to HMP followed by 2 h of NMP before transplantation. NMP perfusate was collected at 3 time points (0, 1, 2 h) for the measurements of prorenin/renin, erythropoietin (EPO), and vitamin D, and urine samples were collected at 1 h and 2 h for urodilatin measurement. Fifteen HMP perfusate samples were collected for the same measurements.
Results.
Kidneys on NMP secreted significantly more prorenin, renin, EPO, and active vitamin D than during HMP. EPO and vitamin D secretion remained stable during 2 h of NMP, whereas the prorenin release rate increased and renin release rate decreased after 1 h. Donation after brain death kidneys secreted more vitamin D and less EPO during NMP than donation after circulatory death kidneys. Twelve donor kidneys produced urine during NMP and released detectable levels of urodilatin. Kidneys exhibited a large variation in hormone release rates. No significant differences were found in hormone release capacity between delayed graft function (DGF) and non-DGF kidneys, and no significant correlations were found between hormone release rates and the duration of DGF or 1-mo posttransplant serum creatinine levels.
Conclusions.
Human transplant kidneys display endocrine activity during NMP. To explore whether correlations exist between hormone release rates and posttransplant kidney function, large numbers of kidneys are required.
Ex vivo machine perfusion has been introduced as a technique to preserve donor kidneys and potentially improve kidney quality. Donor kidney preservation through hypothermic machine perfusion (HMP), which is usually performed at 4 °C, has been shown to improve posttransplant outcomes compared with static cold storage.1,2 Normothermic machine perfusion (NMP), which is performed at 37 °C, is emerging as a novel preservation method that allows metabolic activity of the kidney.3 NMP of suboptimal donor kidneys appears to be a safe and feasible technique in early clinical trials.4,5 There is evidence that during NMP of kidneys, cellular metabolism is improved and adenosine triphosphate levels are replenished,6-9 which may enable repair processes.10 Importantly, NMP provides the opportunity to examine kidney function before transplantation through analysis of perfusate fluid and urine. These analyses may include the measurement of injury factors, but at least equally important is the quantitative determination of kidney function.
Kidneys function as a major endocrine organ, secreting a number of essential hormones, including prorenin/renin, erythropoietin (EPO), and 1,25-dihydroxy vitamin D (1,25(OH)2D), as well as urodilatin.11 Renin, the rate-limiting enzyme of the renin–angiotensin system, is crucial for the regulation of blood pressure and maintenance of salt and volume homeostasis. Prorenin is an inactive precursor of renin. Both prorenin and renin are synthesized and secreted by juxtaglomerular cells. Prorenin is released constitutively, whereas renin is released actively from stored granules.12,13
EPO is a hypoxia-induced erythropoietic hormone, primarily produced by renal EPO-producing cells in adult kidney.14 Scholz et al15 found that isolated perfused rat kidneys are able to synthesize EPO de novo. It is unknown whether isolated human kidneys display EPO-producing capacity.
1,25(OH)2D is the physiologically active form of vitamin D and plays a key role in the maintenance of calcium homeostasis and regulation of bone formation. It is derived from inactive 25-hydroxy vitamin D [25(OH)D] produced in the liver,16 and synthesized in the proximal tubular cells. Rosenthal et al17 observed that 1,25(OH)2D could be detected in isolated perfused rat kidneys after the addition of 25(OH)D to perfusate and showed that its release rate reduced over time during perfusion. This indicates that 25(OH)D was converted to 1,25(OH)2D by the kidney.
Urodilatin is produced by distal tubular cells and secreted into the urine, unlike the other hormones mentioned earlier, which are released into the circulation. It can promote natriuresis and regulate water homeostasis.18 Kirchhoff et al19 found that urodilatin was released constitutively by the isolated perfused rat kidney. It remains unknown whether urodilatin is released by human kidneys during machine perfusion.
We propose that the endocrine function of human donor kidneys during NMP may be a relevant determinant of kidney function. In the present study, we investigated whether human kidneys during NMP display endocrine function through analysis of prorenin/renin, EPO, and 1,25(OH)2D in perfusate fluid and urodilatin in urine, and subsequently correlated hormone release capacity of kidneys during NMP with donor and transplant characteristics, as well as posttransplant kidney function.
MATERIALS AND METHODS
Kidney Perfusion
After kidney procurement from deceased donors, donor kidneys were preserved and transported on pulsatile, nonoxygenated, or oxygenated HMP. All donation after circulatory death (DCD) kidneys of donors aged ≥50 y were subjected to oxygenated HMP based on the results of the COMPARE trial,20 and the other donor kidneys underwent nonoxygenated HMP. Belzer MPS solution (Bridge to Life, USA) was used as the perfusion solution for HMP without additional substances. The materials used for this study came from an ongoing randomized controlled trial, which was registered in clinicaltrials.gov (NCT04882254) in which donor kidneys were randomly assigned to either the HMP control or NMP intervention group. For the NMP intervention group, 15 donor kidneys underwent 2 h additional, end-ischemic NMP with a plasma-free red cell–based solution before transplantation upon arrival at the Erasmus Medical Center. The perfusate composition of NMP in this study is listed in Table S1 (SDC, http://links.lww.com/TXD/A542) and the perfusate did not contain 25(OH)D.21 Perfusate samples were collected at time point 0, 1, and 2 h NMP, followed by the removal of red blood cells with a 2-μm filter (Whatman). Then the perfusate was snap-frozen and stored at –80 °C for the measurements of prorenin/renin, EPO, and vitamin D levels. Urine was collected at time point 1 h and 2 h during NMP for urodilatin measurement. For the HMP control group, 15 donor kidneys were kept on HMP until transplantation. HMP perfusate samples were only collected at the end of the perfusion period and centrifuged at 2000 rpm for 20 min at 4 °C to remove cell debris. The supernatant was aliquoted and stored at –80 °C. Urine samples were not collected during HMP for urodilatin measurement, as urine produced during HMP was recirculated into the circuit. The release of prorenin/renin, EPO, and vitamin D was expressed per hour, correcting for their levels by the duration of NMP or HMP and total perfusate volume.
The study was conducted in compliance with the Declaration of Helsinki. All patients included in this study provided written informed consent, which was approved by the Medical Ethical Committee of Erasmus Medical Center (MEC 2020-0366).
Measurement of Renin and Total Renin by Cisbio Immunoradiometric Assay or enzyme kinetic assay
Renin concentration in the kidney perfusate was measured by a commercial immunoradiometric assay (IRMA; Renin III, Cisbio, Gif-sur-Yvette, France). For total renin (including renin and prorenin) concentration, the kidney perfusate samples were incubated with 10 μmol/L aliskiren at 4 °C for 48 h, which converted prorenin to its open conformation and thus exposed its active site to allow the recognition by the active site-directed antibody of the Cisbio IRMA. Prorenin concentration was determined by subtracting the renin concentration from the total renin concentration. The limit of detection was 1 pg/mL.
Renin activity was measured by the enzyme kinetic assay (EKA), both before (= renin) and after prorenin activation (= renin + prorenin). Prorenin was converted to renin by incubating the samples with sepharose-bound trypsin at 4 °C for 72 h. After prorenin activation, the samples were centrifuged at 13 000 rpm for 10 min at 4 °C. Then, the supernatant was transferred to a new tube and stored at –80 °C until use. During the EKA, samples were incubated at 37 °C with excess sheep angiotensinogen in the presence of angiotensinase inhibitors and the generated Ang I was quantified by radioimmunoassay. The detection limit was 0.17 ng Ang I/mL/h. Prorenin was calculated by subtracting renin from total renin. Ang I–generating activity levels were converted to picograms per milliliter based on the fact that 1 ng Ang I/mL/h corresponds to 2.6 pg human renin/mL.22
Measurement of EPO
Perfusate samples were thawed at 37 °C and centrifuged at 4000×g for 5 min. Cell-free hemoglobin was measured for the determination of hemolysis. EPO was measured by the Access Erythropoietin chemiluminescence immunoassay (Beckman Coulter, CA) on the Access 2 Immunoassay System (Beckman Coulter, CA). Three EPO-positive controls were included after 10 measurements. Once a month, 3 external controls from UK National External Quality Assessment Services were measured for interlaboratory standardization. The detection range of the immunoassay is 0.5 to 750 mIU/mL.
Measurement of 1,25(OH)2D Levels
The IDS-iSYS 1,25 VitDXP kit (Immunodiagnostic Systems, IS-2000) was used to determine 1,25(OH)2D levels in kidney perfusate on the IDS-iSYS Multi-Discipline Automated System. The assay was performed strictly in accordance with the protocol of the kit. The measurement range of the assay is 18 to 360 pmol/L.
Measurement of Urodilatin
Urodilatin ELISA kit S-1231 (BMA Biomedicals, Augst, Switzerland) was used for urodilatin detection. Urodilatin levels were measured in 50 µL urine samples according to the manufacturer’s manual (Handbook, protocol III). Signals were read using the Tecan reader Infinite 200 pro (Tecan Ag, Switzerland). The urodilatin release rate was calculated based on the volume of urine produced. Given urine produced during NMP was removed every 30 min, we calculated the urodilatin release rate from 0.5 to 1 h and from 1.5 to 2 h during NMP based on urodilatin levels and volume of urine produced.
Statistical Analysis
Data analysis was performed with GraphPad Prism version 8 (GraphPad software, La Jolla, CA). Data are presented as median with interquartile range, because of nonnormal population distribution of values. The statistical significance of differences in hormone release rates between the first and second hour of NMP was determined by the Wilcoxon matched-pairs signed rank test. The Mann-Whitney U test was used for the comparisons between 2 unpaired groups. Linear regression was carried out by the “lm: fitting linear model” function in R studio (version 3.0.1) to determine the relationship between prorenin and renin secretion during 2 h of NMP. Correlations between 2 variables were assessed by a nonparametric Spearman correlation test. A 2-tailed P value of <0.05 was considered statistically significant.
RESULTS
Donor Characteristics
In this study, 15 deceased donor kidneys were included in the NMP intervention group, 12 of which were procured from DCD donors, whereas the remaining 3 were from expanded criteria donation after brain death (ECD-DBD) donors aged ≥60 y. Donor and transplant characteristics are displayed in Table 1. Eleven out of the 15 donors were male. The median age of donors was 67 (interquartile range [IQR], 49–72) y. The median total cold ischemia time (CIT) was 10.82 (IQR, 7.38–12.02) h, consisting of CIT before NMP (referred to as CIT-1) and relatively short CIT on static cold storage after disconnecting from the NMP device until transplantation (referred to as CIT-2). The median CIT-1 was 7.43 (IQR, 6.25–9.85) and median CIT-2 was 1.17 (IQR, 0.87–2.70) h. Eight of the 15 donor kidneys received oxygenated HMP and the remaining 7 underwent nonoxygenated HMP before NMP.
TABLE 1.
Donor characteristics and transplant characteristics of 15 donor kidneys included in NMP group
| Patient no. | Donor type | Donor gender | Donor age (y) | HMP | CIT-1 (h) | CIT-2 (h) | Total CIT (h) |
|---|---|---|---|---|---|---|---|
| 1 | DCD | Male | 49 | Nonoxygenated | 7.43 | 2.27 | 9.70 |
| 2 | DCD | Male | 29 | Nonoxygenated | 6.25 | 0.87 | 7.12 |
| 3 | DCD | Female | 72 | Oxygenated | 8.32 | 3.48 | 11.8 |
| 4 | DBD | Male | 74 | Nonoxygenated | 5.28 | 0.23 | 5.52 |
| 5 | DCD | Male | 65 | Oxygenated | 13.25 | 1.08 | 14.33 |
| 6 | DCD | Male | 72 | Oxygenated | 12.78 | 0.52 | 13.30 |
| 7 | DBD | Male | 69 | Nonoxygenated | 11.15 | 1.17 | 12.32 |
| 8 | DBD | Female | 70 | Nonoxygenated | 6.30 | 1.03 | 7.33 |
| 9 | DCD | Male | 36 | Nonoxygenated | 4.53 | 2.85 | 7.38 |
| 10 | DCD | Male | 71 | Oxygenated | 6.62 | 2.70 | 9.32 |
| 11 | DCD | Male | 57 | Oxygenated | 9.73 | 1.43 | 11.17 |
| 12 | DCD | Female | 49 | Nonoxygenated | 7.10 | 0.50 | 7.60 |
| 13 | DCD | Male | 67 | Oxygenated | 5.30 | 6.72 | 12.02 |
| 14 | DCD | Female | 74 | Oxygenated | 8.65 | 2.50 | 11.15 |
| 15 | DCD | Male | 63 | Oxygenated | 9.85 | 0.97 | 10.82 |
CIT, cold ischemia time; DBD, donation after brain death; DCD, donation after circulatory death; HMP, hypothermic machine perfusion; NMP, normothermic machine perfusion.
In the HMP control group, 14 of 15 donors were DCD donors and 1 was an ECD-DBD donor. HMP donor characteristics are shown in Table 2. Ten of the 15 HMP donors were male. The median age of HMP donors was 62 (IQR, 41–67) y and did not differ significantly from that of NMP donors. The median perfusion time for HMP was 4.67 (IQR, 3.53–5.50) h. Seven of the donor kidneys in the HMP group underwent oxygenated HMP and 8 received nonoxygenated HMP.
TABLE 2.
Donor characteristics and transplant characteristics of 15 donor kidneys included in HMP group
| Patient no. | Donor type | Donor gender | Donor age (y) | HMP | Perfusion time (h) |
|---|---|---|---|---|---|
| 16 | DCD | Male | 49 | Nonoxygenated | 3.75 |
| 17 | DCD | Female | 48 | Nonoxygenated | 4.25 |
| 18 | DCD | Male | 65 | Oxygenated | 3.13 |
| 19 | DBD | Female | 70 | Nonoxygenated | 4.67 |
| 20 | DCD | Female | 41 | Nonoxygenated | 3.53 |
| 21 | DCD | Male | 36 | Nonoxygenated | 3.25 |
| 22 | DCD | Male | 71 | Oxygenated | 4.98 |
| 23 | DCD | Female | 62 | Oxygenated | 2.50 |
| 24 | DCD | Male | 69 | Oxygenated | 5.05 |
| 25 | DCD | Male | 62 | Oxygenated | 8.15 |
| 26 | DCD | Male | 40 | Nonoxygenated | 5.50 |
| 27 | DCD | Female | 49 | Nonoxygenated | 4.73 |
| 28 | DCD | Male | 65 | Oxygenated | 10.25 |
| 29 | DCD | Male | 67 | Oxygenated | 3.82 |
| 30 | DCD | Male | 19 | Nonoxygenated | 9.00 |
DBD, donation after brain death; DCD, donation after circulatory death; HMP, hypothermic machine perfusion.
Kidneys Display Endocrine Function During NMP
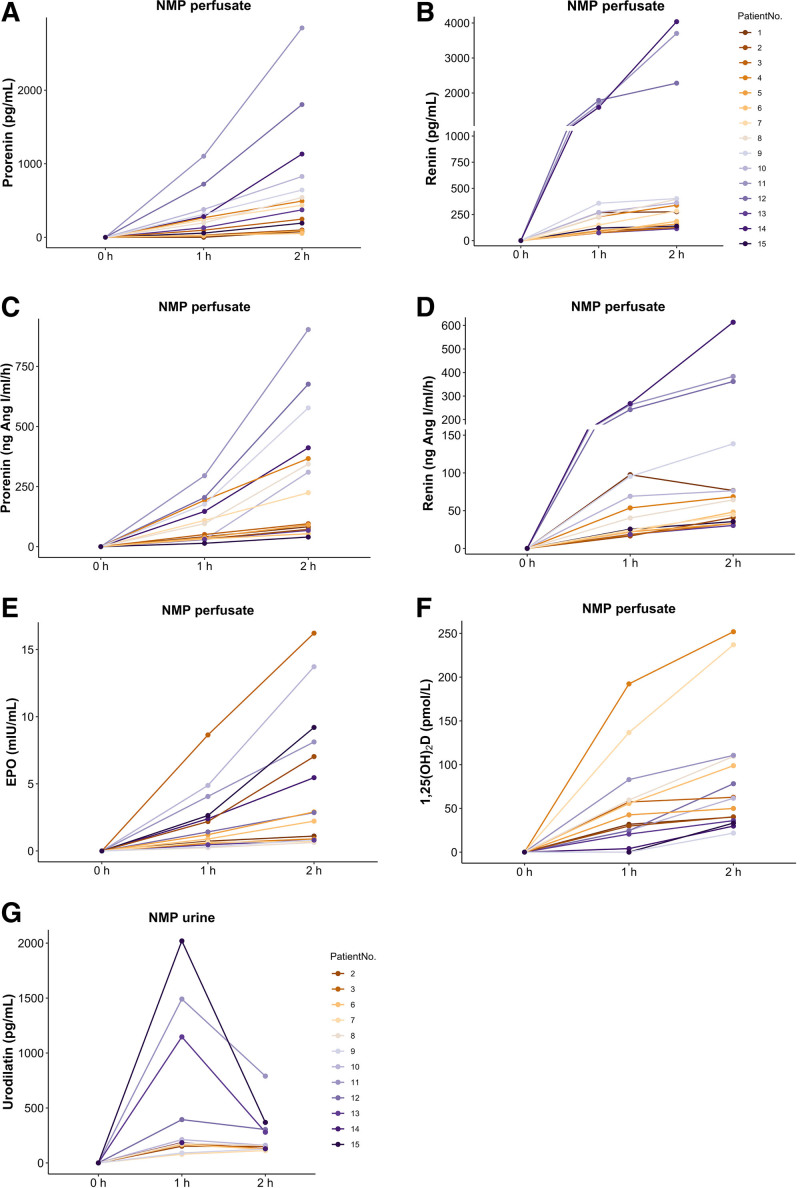
To determine whether human kidneys during NMP secrete hormones, perfusate fluid during NMP was collected for the measurements of prorenin/renin, EPO, and vitamin D, and urine samples were collected for the measurement of urodilatin. Prorenin, renin, EPO, and active vitamin D were detected in NMP perfusate (Figure 1A–F).
FIGURE 1.
Hormone release from human donor kidneys during NMP. Prorenin (A) and renin (B) levels measured by IRMA; prorenin (C) and renin (D) activities measured by EKA; EPO (E) and 1,25(OH)2D (F) levels measured in perfusate from 15 donor kidneys at t = 0, 1, and 2 h of NMP. The above data are corrected by the levels measured at t = 0 h. Urodilatin levels (G) measured in urine from 12 donor kidneys after 1 or 2 h of NMP. No urine was available for urodilatin measurements in patients no. 1, 4, and 5. EKA, enzyme-kinetic assay; EPO, erythropoietin; IRMA, immunoradiometric assay; NMP, normothermic machine perfusion; 1,25(OH)2D, 1,25-dihydroxy vitamin D.
Median release rates of prorenin in the first and second hour of NMP, measured by IRMA, were 196 (IQR, 30–311) and 227 (IQR, 77–448) ng/h, respectively (Figure 1A; Figure S1A, SDC, http://links.lww.com/TXD/A542), and renin release rates in the first and second hour of NMP were 228 (IQR, 94–357) and 92 (IQR, 40–167) ng/h separately (Figure 1B; Figure S1B, SDC, http://links.lww.com/TXD/A542). When compared with the first hour of NMP, the prorenin release rate during the second hour of NMP remarkably increased (P = 0.0015), whereas there was a 2.5-fold decrease in the renin release rate, which did not reach statistical significance. Results obtained by EKA demonstrated the same findings, that is, median release rates of prorenin and renin during the first hour of NMP were 131 (IQR, 83–460) and 105 (IQR, 56–254) ng/h, respectively, followed by a median secretion rate of 300 (IQR, 105–731) ng/h in prorenin and a release rate of 52 (IQR, 28–112) ng/h in renin during the second hour of NMP (Figure 1C and D; Figure S1C and D, SDC, http://links.lww.com/TXD/A542). Prorenin release rate increased (P = 0.013), whereas there was a 2.0-fold reduction in renin release rate (P = 0.022) in the second hour of NMP when compared with the first hour of NMP. This indicates that the 2 assays yielded the same outcome, demonstrating that the renin and prorenin levels detected by IRMA represented native human (pro)renin with functional Ang I–generating capacity. Furthermore, renin secretion was positively correlated with prorenin secretion during 2 h of NMP (R2 = 0.69, P = 0.00011; Figure S2, SDC, http://links.lww.com/TXD/A542).
EPO was secreted by kidneys with a median rate of 20 (IQR, 10–44) mIU/min during the first hour of NMP and with a median rate of 24 (IQR, 6–81) mIU/min in the second hour without significant difference (Figure 1E; Figure S1E, SDC, http://links.lww.com/TXD/A542).
The median secretion rates of 1,25(OH)2D were 32 (IQR, 21–60) and 28 (IQR, 11–50) pmol/h during the first and second hour of NMP, respectively, without significant difference (Figure 1F; Figure S1F, SDC, http://links.lww.com/TXD/A542).
Twelve donor kidneys produced urine during the first and second hour of NMP and displayed detectable urodilatin levels (Figure 1G; Figure S1G, SDC, http://links.lww.com/TXD/A542). Urodilatin could not be measured in patients no.1, 4, and 5 because there was no urine available for urodilatin measurements. Urodilatin release rates did not differ between the first and second hour of NMP (first hour: 62 [IQR, 39–125] ng/h; second hour: 73 [IQR, 36–115] ng/h).
Overall, these results indicate that human kidneys display endocrine activity during NMP by the release of prorenin, renin, EPO, active vitamin D, and urodilatin.
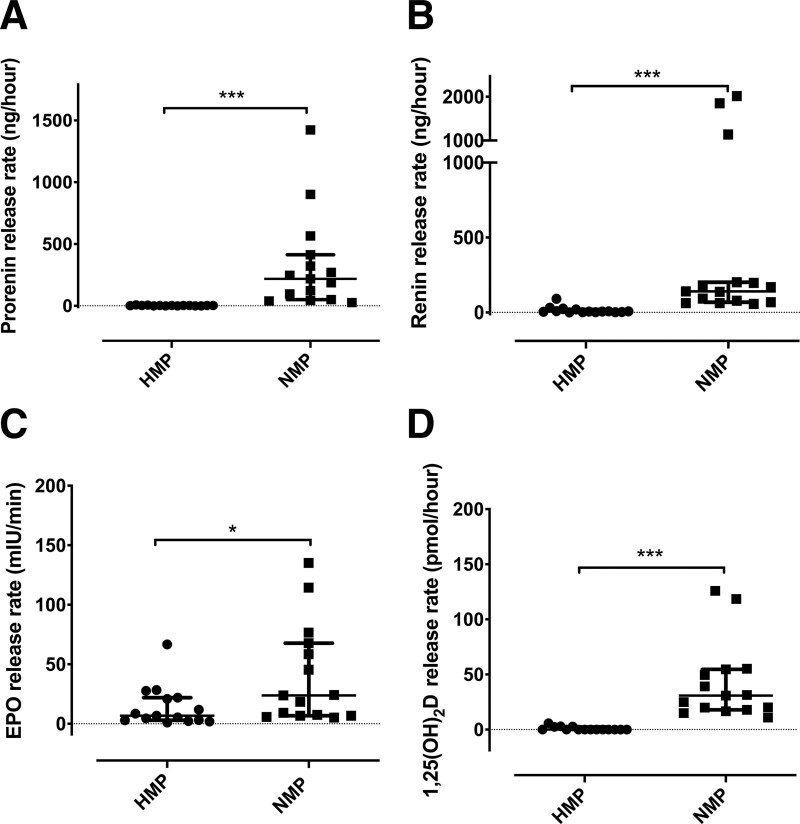
Comparison of Hormone Release Rates Between NMP and HMP
Given a marked difference in cellular metabolic activity between NMP and HMP, we next investigated whether this is reflected by a difference in the hormone release capacity of kidneys between NMP and HMP. Median release rates of prorenin during 2 h of NMP were 91-fold higher than in HMP perfusates (P < 0.0001; Figure 2A), whereas median renin release rates during 2 h of NMP were increased 22-fold in comparison with HMP (P < 0.0001; Figure 2B). The median EPO release rate during 2 h of NMP was 3.5-fold higher than in HMP perfusates (P = 0.024; Figure 2C). In addition, 1,25(OH)2D was undetectable in HMP perfusate, whereas kidneys during 2 h of NMP released significant amounts of 1,25(OH)2D (P < 0.0001; Figure 2D). The above data demonstrate that NMP increases the hormone release capacity of transplant kidneys compared with HMP.
FIGURE 2.
Comparison of hormone release rates of kidneys between HMP and NMP. Prorenin (A), renin (B), EPO (C), and 1,25(OH)2D (D) release rates in 15 kidneys during HMP and 15 kidneys during 2 h of NMP. Data are presented as median with IQR. *P < 0.05, ***P < 0.001. EPO, erythropoietin; HMP, hypothermic machine perfusion; IQR, interquartile range; NMP, normothermic machine perfusion; 1,25(OH)2D, 1,25-dihydroxy vitamin D.
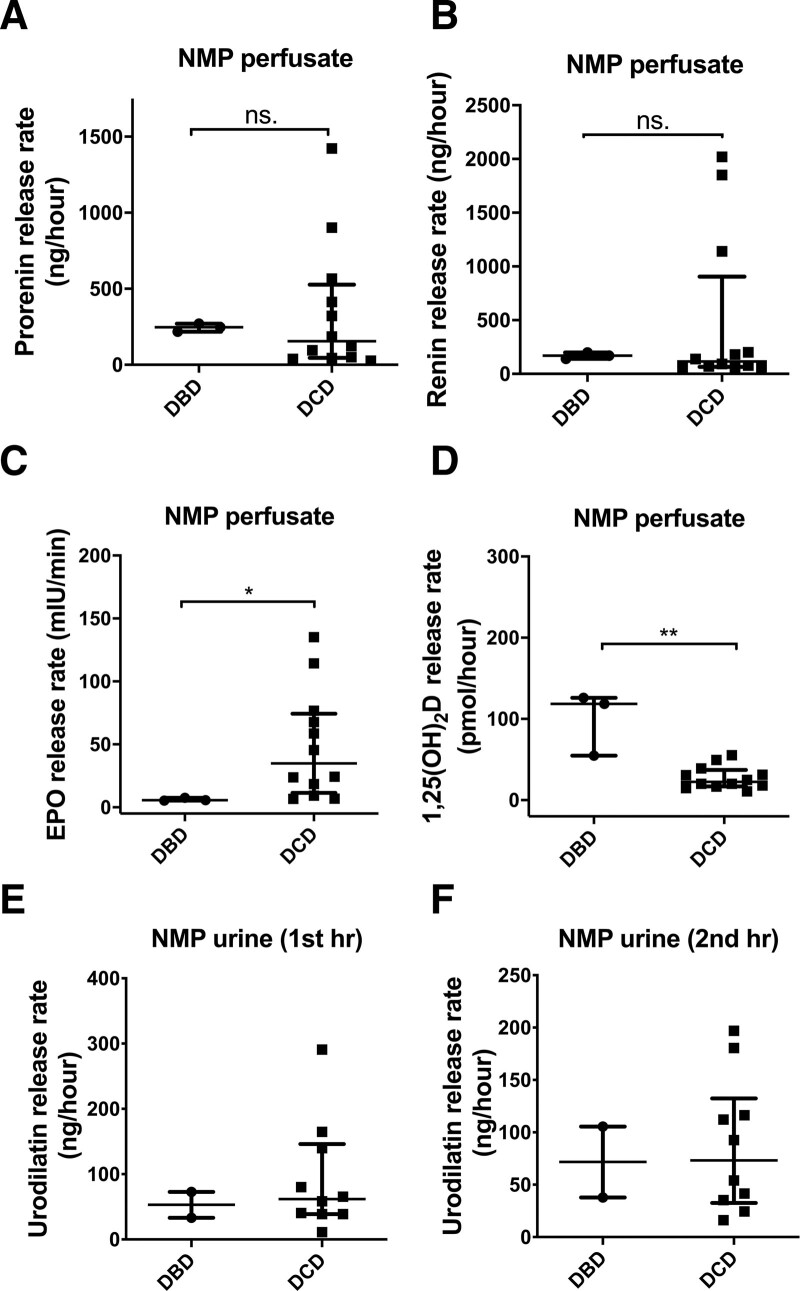
Effects of Donor Type on Hormone Release Capacity
After determining that kidneys are capable of producing hormones during NMP, we next examined whether the hormone release capacity of the grafts was affected by donor type. Neither prorenin nor renin release rates during 2 h of NMP were different between donation after brain death (DBD) and DCD kidneys (Figure 3A and B). Interestingly, DBD kidneys were found to secrete less EPO than DCD kidneys during 2 h of NMP (P = 0.018; Figure 3C), whereas there was a higher active vitamin D release rate during NMP in DBD kidneys compared with DCD kidneys (P = 0.0088; Figure 3D). Statistical analysis of the difference in urodilatin release rates during NMP between DBD and DCD kidneys could not be performed because only 2 of 12 urine samples with detectable urodilatin levels were from DBD kidneys (Figure 3E and F). In general, the data demonstrate that donor type has an impact on the hormone release capacity of donor kidneys during NMP.
FIGURE 3.
Effects of donor type on hormone release capacity of human donor kidneys. Comparison of prorenin (A), renin (B), EPO (C), and 1,25(OH)2D (D) release rates during 2 h of NMP between DBD and DCD donor kidneys (DBD: n = 3, DCD: n = 12). Urodilatin release rates during the first (E) and second (F) h of NMP were compared between DBD and DCD donor kidneys (DBD: n = 2, DCD: n = 10). Data are presented as median with IQR. ns, *P < 0.05, **P < 0.01. DBD, donation after brain death; DCD, donation after circulatory death; EPO, erythropoietin; IQR, interquartile range; NMP, normothermic machine perfusion; ns, not significant; 1,25(OH)2D, 1,25-dihydroxy vitamin D.
Effects of Transplant Characteristics on Hormone Secretion Capacity
We examined whether there were correlations between the hormone secretion ability of donor kidneys during NMP and transplant characteristics. CIT-1 did not correlate with the secretion capacity of the hormones measured during 2 h of NMP (data not shown). In addition, we examined whether the hormone release ability of kidneys during NMP was affected by oxygenation during HMP preceding NMP. The data show that kidneys undergoing nonoxygenated HMP released significantly less EPO during 2 h of NMP than those that received oxygenated HMP (P = 0.029), whereas the prorenin, renin, 1,25(OH)2D, and urodilatin release rates during NMP were similar in the oxygenated and nonoxygenated groups (Figure S3, SDC, http://links.lww.com/TXD/A542). Additionally, we correlated hormone release rates during 2 h of NMP to renal perfusate flow at 2 h of NMP, and no significant correlation was found (Table S2, SDC, http://links.lww.com/TXD/A542).
Does Hormone Release Capacity of Kidneys Predict Posttransplant Outcomes?
Results of graft outcome in the NMP intervention group are summarized in Table S3 (SDC, http://links.lww.com/TXD/A542). Six of the 15 patients had immediate kidney function, and there were 2 patients with primary nonfunction (defined as a persistent need for dialysis 3 mo after transplantation) and 7 patients with delayed graft function (DGF, defined as the need for dialysis in the first week after transplantation). No significant difference was found in hormone release capacity of kidneys between DGF and non-DGF groups (data not shown). The duration of DGF (defined as the time between the day of transplantation and the day of the last dialysis session) of these recipients is listed in Table S3 (SDC, http://links.lww.com/TXD/A542) and the median duration of DGF was 5 (IQR, 4–7) d. No significant correlations were observed between the release rates of the hormones measured and the duration of DGF (Table S4, SDC, http://links.lww.com/TXD/A542).
To determine whether hormones produced by kidneys on NMP can act as functional markers to predict posttransplant outcomes, we correlated median release rates of the hormones measured to serum creatinine levels at 1 mo posttransplant. No significant correlation was found between the secretion ability of all measured hormones and serum creatinine levels at 1 mo posttransplant (Table 3).
TABLE 3.
The Spearman correlation analyses between hormone release rates during 2 h of NMP and serum creatinine levels at 1 mo posttransplant
| Spearman correlation | Serum creatinine levels at 1 mo posttransplant |
|---|---|
| Prorenin (NMP 2 h) | Rs = –0.37, P = 0.18 |
| Renin (NMP 2 h) | Rs = –0.48, P = 0.071 |
| EPO (NMP 2 h) | Rs = –0.29, P = 0.29 |
| 1,25(OH)2D (NMP 2 h) | Rs = 0.31, P = 0.27 |
| Urodilatin (NMP first h) | Rs = –0.063, P = 0.85 |
| Urodilatin (NMP second h) | Rs = –0.10, P = 0.75 |
EPO, erythropoietin; NMP, normothermic machine perfusion; 1,25(OH)2D, 1,25-dihydroxy vitamin D; Rs, Spearman’s rank correlation coefficient.
DISCUSSION
The control of body homeostasis via hormone secretion is one of the main functions of the kidney.11 Studies reveal that there is a correlation between the expression levels of renin–angiotensin system components in human kidney biopsies and the existence and severity of underlying kidney disease.23 For instance, renin mRNA levels have been shown to be upregulated in mesangial cells of kidney tissue from patients with kidney disease.24 EPO production is reduced in damaged kidneys,25,26 and decreased circulating levels of 1,25(OH)2 are observed in patients with kidney disease.25 The above-mentioned evidence indicates that kidney injury affects hormone release. Thus, we propose that the hormone release capacity of kidneys may be used to assess transplant kidney quality and function and predict transplant outcomes. To test this hypothesis, we identified whether kidneys during machine perfusion have the capacity to secrete hormones. In this study, we determined the presence of several key hormones in perfusate fluid and urine of human kidneys during HMP and NMP and demonstrated that NMP-mediated activation of metabolism leads to increased endocrine activity of the kidney. To our knowledge, this is the first report to show that human kidneys during machine perfusion release prorenin/renin, EPO, active vitamin D, and urodilatin. The renin released in the NMP perfusate was enzymatically active, and thus in combination with its recognition in the renin assays, represents normal human renin in all aspects. Our data are in line with the fact that prorenin is secreted constitutively, whereas renin release is an active process, in which renin release started with an initial burst, and then diminished to lower values. This is most likely because of the fact that after a rapid burst, renin storage sites may have become depleted. A strong positive correlation between prorenin and renin secretion from human kidneys during NMP was found, which is in line with observations in human plasma.27,28 In addition, EPO and urodilatin release remained stable during NMP, indicating that EPO and urodilatin are released upon de novo synthesis in the kidney, which is consistent with the observations in the isolated perfused rat kidneys.15,19 1,25(OH)2D that was detected in perfusates was likely secreted by kidneys from intracellular stores rather than converted from 25(OH)D, as 25(OH)D substrate is absent in the perfusate.
We have strong evidence that the hormones detected during NMP are released as a result of physiologic activity of the kidneys, rather than as a result of cell death or cell leakage, or release from circulatory cells. In both the NMP intervention group and HMP control group, the transplant kidneys were flushed with kidney perfusion solution to remove blood remnants before HMP. During NMP, the kidneys were perfused with red blood cells, which do not produce the measured hormones. Furthermore, the hormone levels detected in kidney perfusate during the first and second hour of NMP were normalized to those measured at t = 0 h, which removed the background from the perfusion fluid. Additionally, significantly higher release rates of hormones during NMP were observed than during HMP, which further rules out the possibility that hormone secretion detected in NMP perfusate is just wash-out. Therefore, the elevated endocrine activity of kidneys during NMP is most likely because of higher cellular metabolic rates. In the setting of NMP, kidney temperature is within the physiological range, and thus optimal for enzymatic activity, allowing production and release of hormones. Unexpectedly, significantly less EPO was secreted during NMP from the kidneys subjected to nonoxygenated HMP before NMP than those subjected to oxygenated HMP. This finding is inconsistent with the fact that EPO production is normally stimulated in response to hypoxia.29-31 The explanation for this may be that EPO synthesis did not increase in response to hypoxia stress under low-temperature conditions. Preoxygenated kidneys were potentially better equipped to upregulate EPO production during NMP. Whether this is the case requires further investigation.
We demonstrated that human kidneys during NMP release hormones, which provides a potential tool to assess graft quality and function. Interestingly, kidneys showed a high degree of variation in hormone release rates. Notably, only 15 suboptimal-quality kidneys comprising 12 DCD and 3 ECD-DBD kidneys were included in the NMP group of this study, which may have been insufficient for finding correlations between endocrine activity and posttransplant kidney function. However, we found that 1,25(OH)2D release rate during NMP in DGF kidneys tends to be higher than in non-DGF kidneys (P = 0.051). In addition, renin release rate during 2 h of NMP seemed to negatively correlate with serum creatinine levels at 1 mo posttransplant (Rs = –0.48, P = 0.071). The focus of future research should be to increase the number of inclusions to increase the power of the analyses. The use of suboptimal kidneys only is another limitation of our study. The hormone release profile of standard criteria kidneys still remains unknown and it is therefore not clear whether the hormone release rates found in our study are comparable with the rates of standard criteria donor kidneys. Hence, healthier and younger kidneys should be included in the study to allow the stratification of donor kidneys based on their endocrine activity during NMP.
Nowadays, the shortage of suitable donor kidneys is a major limitation for transplantation. The increased use of ECD and DCD kidneys enlarges the kidney donor pool and tackles the organ shortage to some extent; however, the quality of these suboptimal donor kidneys varies, which is likely to affect posttransplant outcomes. The hormone release capacity of donor kidneys during NMP may become a criterion to define whether suboptimal donor kidneys can be transplantable, consequently preventing undesired transplantation of kidneys with a high risk of primary nonfunction and potentially enlarging the kidney donor pool by decreasing unnecessary discards of suboptimal donor kidneys. Consequently, more patients should be included in further studies to determine whether the secretion ability of these hormones during NMP can act as functional markers to predict posttransplant outcomes.
In conclusion, the data in this study indicate that human kidneys release functional hormones during machine perfusion. NMP conditions promote the hormone release capacity. Improved sample size may allow correlation of kidney hormone release capacity with posttransplant kidney function.
ACKNOWLEDGMENTS
The authors would like to thank all organ donors and their families.
Supplementary Material
Footnotes
H.L. and Z.D. contributed equally to this work. R.C.M. and M.J.H. are joint senior authors.
The authors declare no funding or conflicts of interest.
H.L. and Z.D. designed and performed experiments, analyzed results, and wrote the article. S.B., E.R., I.C., and A.O. performed experiments and reviewed the article. J.C. and A.H.J.D. provided expertise and reviewed the article. R.C.M. and M.J.H. designed the experiments, provided expertise and feedback, and reviewed the article.
Supplemental digital content (SDC) is available for this article. Direct URL citations appear in the printed text, and links to the digital files are provided in the HTML text of this article on the journal’s Web site (www.transplantationdirect.com).
REFERENCES
- 1.Moers C, Smits JM, Maathuis MH, et al. Machine perfusion or cold storage in deceased-donor kidney transplantation. N Engl J Med. 2009;1:7–19. [DOI] [PubMed] [Google Scholar]
- 2.Tingle SJ, Figueiredo RS, Moir JA, et al. Machine perfusion preservation versus static cold storage for deceased donor kidney transplantation. Cochrane Database Syst Rev. 2019;3:cd011671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Arykbaeva AS, De Vries DK, Doppenberg JB, et al. Metabolic needs of the kidney graft undergoing normothermic machine perfusion. Kidney Int. 2021;2:301–310. [DOI] [PubMed] [Google Scholar]
- 4.Hosgood SA, Saeb-Parsy K, Wilson C, et al. Protocol of a randomised controlled, open-label trial of ex vivo normothermic perfusion versus static cold storage in donation after circulatory death renal transplantation. BMJ Open. 2017;1:e012237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Rijkse E, De Jonge J, Kimenai H, et al. Safety and feasibility of 2 h of normothermic machine perfusion of donor kidneys in the Eurotransplant senior program. BJS Open. 2021;5:zraa024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bagul A, Hosgood SA, Kaushik M, et al. Experimental renal preservation by normothermic resuscitation perfusion with autologous blood. Br J Surg. 2008;1:111–118. [DOI] [PubMed] [Google Scholar]
- 7.Hosgood SA, Barlow AD, Yates PJ, et al. A pilot study assessing the feasibility of a short period of normothermic preservation in an experimental model of non heart beating donor kidneys. J Surg Res. 2011;1:283–290. [DOI] [PubMed] [Google Scholar]
- 8.Hosgood SA, Patel M, Nicholson ML. The conditioning effect of ex vivo normothermic perfusion in an experimental kidney model. J Surg Res. 2013;1:153–160. [DOI] [PubMed] [Google Scholar]
- 9.Ferdinand JR, Hosgood SA, Moore T, et al. Cytokine absorption during human kidney perfusion reduces delayed graft function-associated inflammatory gene signature. Am J Transplant. 2021;6:2188–2199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hosgood SA, Van Heurn E, Nicholson ML. Normothermic machine perfusion of the kidney: better conditioning and repair? Transpl Int. 2015;6:657–664. [DOI] [PubMed] [Google Scholar]
- 11.Acharya V, Olivero J. The kidney as an endocrine organ. Methodist Debakey Cardiovasc J. 2018;4:305–307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kurtz A, Schweda F. Osmolarity-induced renin secretion from kidneys: evidence for readily releasable renin pools. Am J Physiol Renal Physiol. 2006;4:f797–f805. [DOI] [PubMed] [Google Scholar]
- 13.Kurtz A. Control of renin synthesis and secretion. Am J Hypertens. 2012;8:839–847. [DOI] [PubMed] [Google Scholar]
- 14.Souma T, Suzuki N, Yamamoto M. Renal erythropoietin-producing cells in health and disease. Front Physiol. 2015;6:167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Scholz H, Schurek HJ, Eckardt KU, et al. Oxygen-dependent erythropoietin production by the isolated perfused rat kidney. Pflugers Arch. 1991;3:228–233. [DOI] [PubMed] [Google Scholar]
- 16.Bikle DD. Vitamin D metabolism, mechanism of action, and clinical applications. Chem Biol. 2014;3:319–329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Rosenthal AM, Jones G, Kooh SW, et al. 25-Hydroxyvitamin d3 metabolism by isolated perfused rat kidney. Am J Physiol. 1980;1:e12–e20. [DOI] [PubMed] [Google Scholar]
- 18.Forssmann W, Meyer M, Forssmann K. The renal urodilatin system: clinical implications. Cardiovasc Res. 2001;3:450–462. [DOI] [PubMed] [Google Scholar]
- 19.Kirchhoff K, Forssmann WG. Release of urodilatin from perfused rat kidney and from cultured neonatal rat kidney cells. Pflugers Arch. 1995;5:739–744. [DOI] [PubMed] [Google Scholar]
- 20.Jochmans I, Brat A, Davies L, et al. Oxygenated versus standard cold perfusion preservation in kidney transplantation (compare): a randomised, double-blind, paired, phase 3 trial. Lancet. 2020;10263:1653–1662. [DOI] [PubMed] [Google Scholar]
- 21.Rijkse E, Bouari S, Kimenai H, et al. Additional normothermic machine perfusion versus hypothermic machine perfusion in suboptimal donor kidney transplantation: protocol of a randomized, controlled, open-label trial. Int J Surg Protoc. 2021;1:227–237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Krop M, Van Veghel R, Garrelds IM, et al. Cardiac renin levels are not influenced by the amount of resident mast cells. Hypertension. 2009;2:315–321. [DOI] [PubMed] [Google Scholar]
- 23.Lin H, Geurts F, Hassler L, et al. Kidney angiotensin in cardiovascular disease: formation and drug targeting. Pharmacol Rev. 2022;3:462–505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lai KN, Leung JC, Lai KB, et al. Gene expression of the renin-angiotensin system in human kidney. J Hypertens. 1998;1:91–102. [DOI] [PubMed] [Google Scholar]
- 25.Banaei S, Rezagholizadeh L. The role of hormones in renal disease and ischemia-reperfusion injury. Iran J Basic Med Sci. 2019;5:469–476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Santos EJF, Dias RSC, Lima JFB, et al. Erythropoietin resistance in patients with chronic kidney disease: current perspectives. Int J Nephrol Renovasc Dis. 2020;13:231–237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Danser AH, Derkx FH, Schalekamp MA, et al. Determinants of interindividual variation of renin and prorenin concentrations: evidence for a sexual dimorphism of (pro)renin levels in humans. J Hypertens. 1998;6:853–862. [DOI] [PubMed] [Google Scholar]
- 28.Cho ME, Sweeney C, Fino N, et al. Longitudinal changes in prorenin and renin in the chronic renal insufficiency cohort. Am J Nephrol. 2021;2:141–151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Varma S, Cohen HJ. Co-transactivation of the 3’ erythropoietin hypoxia inducible enhancer by the HIF-1 protein. Blood Cells Mol Dis. 1997;2:169–176. [DOI] [PubMed] [Google Scholar]
- 30.Stolze I, Berchner-Pfannschmidt U, Freitag P, et al. Hypoxia-inducible erythropoietin gene expression in human neuroblastoma cells. Blood. 2002;7:2623–2628. [DOI] [PubMed] [Google Scholar]
- 31.Haase VH. Regulation of erythropoiesis by hypoxia-inducible factors. Blood Rev. 2013;1:41–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.