Abstract
Background
The effect of surgery on impulse control disorders (ICDs) remains unclear in Parkinson's disease (PD) patients undergoing deep brain stimulation (DBS).
Objective
To examine changes in ICD symptoms in PD patients undergoing DBS compared to a medication‐only control group.
Methods
The study was a 2‐center, 12‐month, prospective, observational investigation of PD patients undergoing DBS and a control group matched on age, sex, dopamine agonist use, and baseline presence of ICDs. Questionnaire for Impulsive‐Compulsive Disorders in Parkinson's Disease‐Rating Scale (QUIP‐RS) and total levodopa equivalent daily dose (LEDD) were collected at baseline, 3, 6, and 12 months. Linear mixed‐effects models assessed changes in mean QUIP‐RS score (sum of buying, eating, gambling, and hypersexuality items).
Results
The cohort included 54 participants (DBS = 26, controls = 28), mean (SD) age 64.3 (8.1) and PD duration 8.0 (5.2) years. Mean baseline QUIP‐RS was higher in the DBS group at baseline (8.6 (10.7) vs. 5.3 (6.9), P = 0.18). However, scores at 12 months follow‐up were nearly identical (6.6 (7.3) vs. 6.0 (6.9) P = 0.79). Predictors of change in QUIP‐RS score were baseline QUIP‐RS score (β = 0.483, P < 0.001) and time‐varying LEDD (β = 0.003, P = 0.02). Eight patients (four in each group) developed de novo ICD symptoms during follow‐up, although none met diagnostic criteria for an impulse control disorder.
Conclusions
ICD symptoms (including de novo symptoms) at 12 months follow‐up were similar between PD patients undergoing DBS and patients treated with pharmacological therapy only. Monitoring for emergence of ICD symptoms is important in both surgically‐ and medication‐only‐treated PD patients.
Keywords: Parkinson's disease, deep brain stimulation, de novo ICDs, dopaminergic medication, LEDD
Impulse control disorders (ICDs) are a major neuropsychiatric complication of Parkinson's disease (PD), occurring in up to 40%. 1 , 2 , 3 The four primary ICDs are compulsive buying, eating, gambling, and hypersexuality; other impulse control behaviors include hobbyism, punding, and dopamine dysregulation syndrome (DDS). 4 These manifestations are associated with a reduced quality of life and negatively impact family dynamics. 5
In PD, ICDs are generally considered a complication of dopaminergic medication therapy. 5 , 6 Dopamine agonists (DAs) have been identified as the most potent risk factor, though levodopa has also been associated with ICDs at higher doses, 2 , 7 , 8 and short‐acting, high‐potency dopaminergic medications with punding and DDS. 4 , 9 , 10 Other risk factors associated with ICDs include younger age, unmarried, male sex, family history of gambling, personal or family history of substance abuse, alcohol or tobacco use, history of ICDs, depression pre‐PD diagnosis, and anxiety. 1 , 2 , 11 PD itself does not appear to be a risk factor for developing ICDs. 6
Patients with advanced PD are frequently considered for deep brain stimulation surgery (DBS). 12 A growing body of literature has revealed some of the potential effects of DBS on ICDs and related disorders, 13 , 14 , 15 , 16 , 17 , 18 , 19 but with conflicting results. 20 , 21 Most studies have concluded that ICDs largely resolve following DBS, 16 , 17 , 22 usually in the setting of a decrease in dopaminergic therapy. However, recent case series and prospective observational studies have reported the possibility of worsening or re‐emergence of ICDs, and/or the development of de novo ICDs, in a small subset of patients following surgery. 13 , 14 , 17 , 23 , 24 , 25 All available studies have been limited by the absence of a non‐surgical control group which could be used to account for the natural history of ICD symptoms in medicated PD patients. The current study aimed to prospectively examine the evolution of ICD symptoms in a group of individuals with PD undergoing DBS and to compare the results over the same time period to a group without DBS.
Methods
Study Design
This was a prospective observational study that recruited patients from two centers in the United States: the Parkinson's Disease and Movement Disorders Center (PDMDC) at the University of Pennsylvania and the University of Florida Norman Fixel Institute for Neurological Diseases. The study was approved by the Institutional Review Boards (independent ethics committees) at each institution and written informed consent was obtained from each participant.
Study Sample
We recruited participants from Jan 1, 2015 to Dec 31, 2018. The DBS group was a convenience sample of individuals attending pre‐operative multidisciplinary clinics at each institution. Determination of eligibility for DBS, including neuropsychological testing to assess cognition, was performed per the standard of care at each institution. Inclusion criteria for this study were age ≥18 years, English‐speaking, and sufficient cognitive abilities to provide informed consent. Individuals who required bilateral procedures but whose second surgery was expected to occur >3 months following the first one were excluded. During the recruitment period for the DBS group, approximately 140 non‐demented PD patients underwent DBS at both institutions, but only those who had surgery scheduled and could be contacted by the study team prior to surgery were assessed for eligibility (n = 42; Fig. 1).
Figure 1.
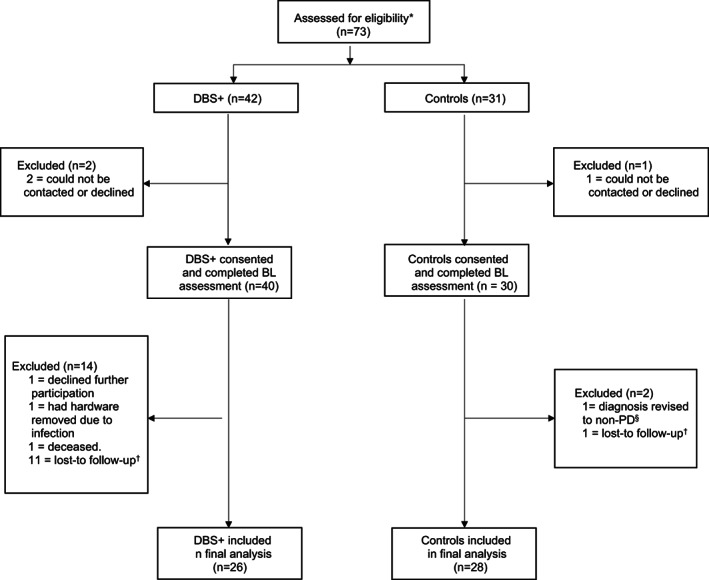
Flow diagram of the study sample: from assessment for the study to the final analytic sample. *Eligible DBS+ patients were PD patients who had surgery scheduled in one of the participating institutions and who could be contacted by the study team prior surgery. Eligible participants for the control group were selected among PD patients who were not expected to require DBS within two years of enrollment in the study. §diagnosis was revised by the clinician’s patient. †no post‐baseline assessment available.
The non‐surgical control group was a convenience sample of patients diagnosed with PD who were not expected to require DBS within 2 years of enrollment in the study. Controls were recruited from the outpatient clinic or from other observational studies occurring at either participating institution. Controls were matched consecutively, on a 1:1 basis to each individual recruited to the DBS group, based on age (±2 years), sex, dopamine agonist (DA) medication use (yes/no), and the presence or absence of an impulse control disorder (ICD) diagnosis as determined by a structured clinical interview. 26
Assessments
Collected data included demographics (age, sex, race/ethnicity, marital status, level of education), PD duration at baseline visit, and Movement Disorders Society Unified Parkinson's Disease Rating Scale part III (MDS‐UPDRS III) scores in the medication off state where available. Participants in the DBS group were interviewed at baseline (≤1 month before scheduled DBS surgery) and at 3, 6, and 12 months post‐operatively. For individuals undergoing staged implantation, the first post‐operative assessment occurred 3 months following implantation of the second lead. Participants in the control group were assessed at the same intervals, with “baseline” defined as the first interview conducted during the study period. All assessments were administered by telephone by a research coordinator experienced in assessing ICDs in PD patients (author EM).
Participants were administered the Questionnaire for Impulsive‐Compulsive Disorders in Parkinson's Disease‐Rating Scale (QUIP‐RS) at each assessment. The QUIP‐RS is a patient‐reported outcome that consists of 28 items that assess the frequency of compulsive behaviors related to buying, eating, gambling, sex, hobbyism, punding, and DDS. 26 Responses range from 0 (never) to 4 (very often), 4 and the total score ranges from 0 to 112, with higher scores implying greater symptom severity. For the primary analysis, the QUIP‐RS score was generated as the sum of the shopping, eating, gambling, and sexual behaviors items, which constitute the four primary ICDs in PD. The onset of de novo ICD symptoms was defined as a score of ≥10 in an individual whose score was <10 at baseline. 26 , 27 In secondary analyses, the total score of all items of the QUIP‐RS was examined as the outcome measure.
Data regarding medication (name and dose) were provided by participants or an informed other, and verified via electronic health record (EHR) review. Total levodopa equivalent daily dose (LEDD) and LED‐DA (LEDD from dopamine agonists only) were collected at baseline and each follow‐up assessment. 28 Study data were collected and managed using REDCap electronic data capture tools. 29
Statistical Analysis
The primary outcome was the change in QUIP‐RS score for the four primary ICDs (shopping, eating, gambling, and sexual behaviors) from baseline to month 12. Secondary outcomes were occurrence of de novo ICD symptoms and the total score of all items of the QUIP‐RS.
Unpaired t‐test or Wilcoxon rank sum test as appropriate (for continuous variables) and Chi‐squared (for categorical variables) were used to compare DBS and control groups’ baseline demographic and clinical characteristics. Changes in total QUIP‐RS scores over time and by treatment group were tested with paired t‐test. Within‐subject changes in QUIP‐RS score over time were assessed with linear mixed‐effects model (LMM) using random intercepts for each patient. Change in slope for mean QUIP‐RS scores over time in the DBS group vs. control group was first examined in an unadjusted analysis for the cohort as a whole, and in the DBS subgroup with bilateral STN DBS only. Then a multivariable linear mixed‐effect regression model was constructed including the following covariates: age, sex, disease duration, and time‐varying LEDD (or in a separate model time‐varying LED‐DA). In sensitivity analyses, dopamine agonist use (yes/no) instead of LED‐DA was adjusted for. R version 3.6.0 was used for all statistical analyses.
Results
Figure 1 shows the total number of PD patients at each stage of the study. The final analytic cohort was comprised of 54 participants; 45 were followed at Penn PDMDC (n = 22 with DBS), while the remaining nine received care at UF (n = 4 with DBS). Surgical target was as follows: bilateral STN (n = 17; one staged), bilateral GPi (n = 2, none staged), unilateral STN (n = 5), and unilateral GPi (n = 2).
Cohort Baseline Characteristics
Table 1 displays baseline demographic and clinical characteristics. At baseline, patients had a mean (SD) age of 64.3 (8.1) years and disease duration of 8.0 (5.2) years. The majority of subjects were male (81.5%) and White (94.4%). Patients in the DBS group had higher total LEDD compared to those in the control group (mean (SD) 1045.5 (516.6) vs. 793.7 (397.6), P = 0.05). LED‐DA was not significantly different between the groups (mean (SD) 63.1 (122.8) vs. 101.9 (144.3), P = 0.29). 30.8% of patients in the DBS group had QUIP‐RS scores ≥10 compared with 17.9% of patients in the control group (P = 0.27).
TABLE 1.
Demographics and Baseline Characteristics of Patients with Parkinson's Disease by Treatment Group
| DBS (n = 26) | Control (n = 28) | Total (n = 54) | P‐value | |
|---|---|---|---|---|
| Demographic | ||||
| Age, mean (SD) | 63.18 (8.5) | 65.27 (7.6) | 64.27 (8.1) | 0.35 |
| Sex, n (col %) | 0.90 | |||
| Male | 21 (80.8) | 23 (82.1) | 44 (81.5) | |
| Female | 5 (19.2) | 5 (17.9) | 10 (18.5) | |
| Race, n (col %) | 0.18 | |||
| White | 23 (88.5) | 28 (100) | 51 (94.4) | |
| Black | 2 (7.7) | 0 (0) | 2 (3.7) | |
| Asian | 0 (0) | 0 (0) | 0 (0) | |
| American Indian or Alaska Native | 0 (0) | 0 (0) | 0 (0) | |
| Native Hawaiian or other Pacific Islander | 0 (0) | 0 (0) | 0 (0) | |
| Other | 1 (3.8) | 0 (0) | 1 (1.9) | |
| Ethnicity, n (col %) | 0.17 | |||
| Hispanic or Latino | 0 (0) | 2 (7.1) | 2 (3.7) | |
| Non‐Hispanic, Non‐Latino | 26 (100) | 26 (92.9) | 52 (96.3) | |
| Marital status, n (col %) | 0.25 | |||
| Married | 22 (84.6) | 23 (82.1) | 45 (83.3) | |
| Single | 1 (3.8) | 4 (14.3) | 5 (9.3) | |
| Divorced | 3 (11.5) | 1 (3.6) | 4 (7.4) | |
| Separated | 0 (0) | 0 (0) | 0 (0) | |
| Widowed | 0 (0) | 0 (0) | 0 (0) | |
| Education, mean (SD) | 15.15 (3.2) | 15.92 (2.35) | 15.54 (2.8) | 0.32 |
| Clinical | ||||
| Disease duration, mean (SD) | 6.77 (4.4) | 9.26 (5.71) | 8.04 (5.2) | 0.08 |
| MDS‐UPDRS part III mean (SD) off* | 38.57 (11.6) | Data not available | ||
| LEDD, mean (SD) | 1045.5 (516.6) | 793.7 (397.6) | 915 (471.8) | 0.05 |
| LED‐DA, mean (SD) | 63.1 (122.8) | 101.9 (144.3) | 83.2 (134.6) | 0.29 |
| ICD symptoms | ||||
| QUIP‐RS score, mean (SD) | 8.58 (10.7) | 5.29 (6.9) | 6.87 (9.0) | 0.18 |
| QUIP‐RS ≥10, n (col %) | 0.27 | |||
| No | 18 (69.2) | 23 (82.1) | 41 (75.9) | |
| Yes | 8 (30.8) | 5 (17.9) | 13 (24.1) | |
| DDS score | 1.73 (3.18) | 0.46 (1.60) | 1.07 (2.55) | 0.76 |
| Hobbyism score | 2.42 (2.58) | 2.11 (2.95) | 2.26 (2.76) | 0.36 |
| Punding score | 2.15 (2.46) | 1.71 (2.80) | 1.93 (2.63) | 0.54 |
| ICD by DSM‐IV criteria | 2 (7.7) | 1 (3.6) | 3 (5.6) | 0.51 |
Note: MDS‐UPRS scores were not available for controls.
Abbreviations: DBS, deep brain stimulation; DDS, dopamine dysregulation syndrome; ICD, impulse control disorder; LEDD, levodopa equivalent daily dose; LED‐DA, levodopa equivalent dose from dopamine agonist; MDS‐UPDRS, movement disorder society‐sponsored revision of the unified Parkinson's disease rating scale; QUIP‐RS, questionnaire for impulse control disorders in Parkinson's disease rating scale.
MDS‐UPDRS part III was administered in the medication off state during preoperative clinical evaluation, after withholding dopaminergic medications overnight. MDS‐UPDRS III scores are missing on two patients in the DBS group.
Cohort Characteristics at Follow‐Up
Of the 54 participants, 50 (92.6%) completed the 12‐month assessment; 42 (77.8%) completed assessments at all time points. Of the four that did not complete the 12‐month assessment, three were in the DBS group, and one in the control group; they were older and had higher baseline LEDD and lower LED‐DA compared to those that had a 12‐month assessment. In the surgical group, the 12‐month assessment occurred a mean of 358.5 (SD 24.34) days following DBS surgery.
In the DBS group, compared to baseline, LEDD decreased significantly after DBS at each follow‐up assessment (mean (SD) change −349.7 (361.4), P < 0.001 at 3 months; 347.1 (431.9), P = 0.001 at 6 months; and −332.3 (482.8), P = 0.003 at 12 months). Compared to baseline, LED‐DA was significantly different in the DBS group only at the 3‐month assessment (mean (SD) −26.4 (57.6), P = 0.04). In the control group, LEDD did not change significantly during follow‐up compared to baseline (mean (SD) change 23.1 (187.7), P = 0.54 at 3 months; 21.1 (168.1), P = 0.52 at 6 months; and 68.3 (225.4), P = 0.13 at 12 months).
QUIP‐RS Scores over Time
The longitudinal change in QUIP‐RS score and in LEDD are shown in Figure 2. At baseline, mean (SD) QUIP‐RS score in the DBS group vs control group was 8.6 (10.7) vs. 5.3 (6.9), P = 0.18. At the 3‐month assessment, scores remained higher in the DBS group (mean (SD) 7.0 (8.1) vs. 4.7 (5.9), P = 0. 26). QUIP‐RS score was nearly identical in the two groups at the 6‐ and 12‐months assessments (5.3 (6.2) vs. 5.0 (6.4), P = 0.89; 6.6 (7.3) vs. 6.0 (6.9) P = 0.79, respectively).
Figure 2.
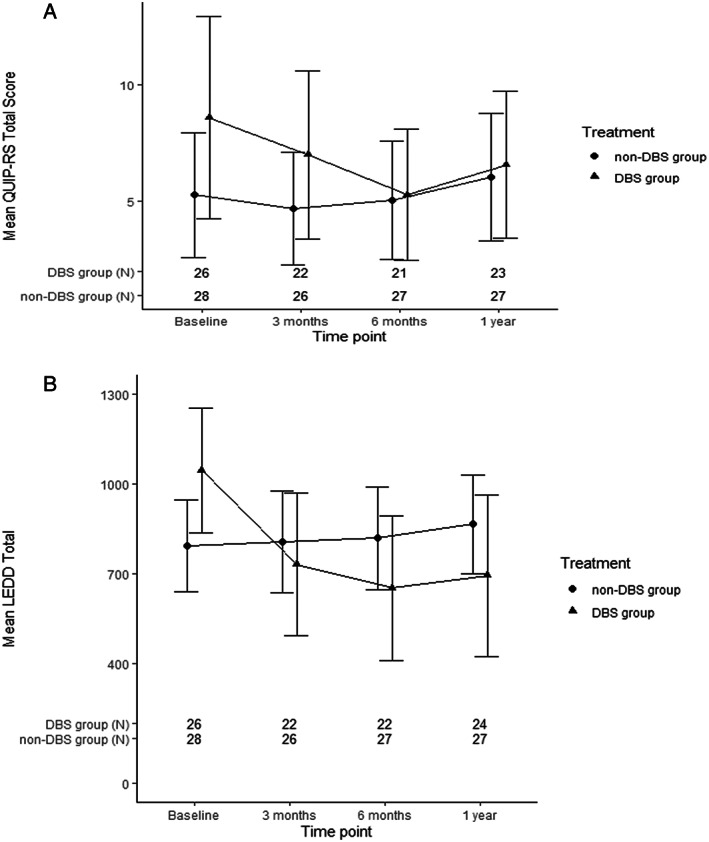
Trends in (A) QUIP‐RS scores and (B) LEDD during follow‐up.
In a univariate (unadjusted) linear‐mixed effects model, mean QUIP‐RS scores decreased significantly in the DBS group from baseline to 6 months, then slightly increased from 6 to 12 months. QUIP‐RS score did not significantly change in the control group. Significant differences in change in QUIP‐RS score over time in the DBS vs. control group were not observed; results were similar when analyses were restricted to the patients with bilateral STN DBS (Table 2).
TABLE 2.
Change in QUIP‐RS Score from Baseline in Cohort as a Whole (Model 1) and in the Subset of Surgical Group with Bilateral STN DBS only (Model 2) (Results from Unadjusted Linear Mixed‐Effects Model)
| 3 months | 6 months | 1 year | ||||
|---|---|---|---|---|---|---|
| Exposure group | Estimate (SE) | 95% CI | Estimate (SE) | 95% CI | Estimate (SE) | 95% CI |
| Model 1 | ||||||
| DBS | −2.0 (1.6) | −5.1–1.1 | −3.3 (1.6) | −6.5– −0.2 | −1.9 (1.6) | −4.9–1.2 |
| Control | −0.3 (1.5) | −3.2–2.6 | −0.3 (1.5) | −3.2–2.6 | 0.7 (1.5) | −2.2–3.6 |
| Model 2 | ||||||
| DBS, bilateral STN only | −1.3 (2.0) | −5.2–2.7 | −5.1 (2.0) | −9.1– −1.2 | −4.1 (2.0) | −8.0– −0.1 |
| Control | −0.3 (1.5) | −3.2–2.7 | −0.3 (1.5) | −3.2–2.6 | 0.7 (1.5) | −2.2–3.6 |
Abbreviations: DBS, deep brain stimulation; QUIP‐RS, questionnaire for impulse control disorders in Parkinson's disease rating scale; STN, subthalamic nucleus.
Adjusting for covariates in a linear mixed‐effect regression model, change in QUIP‐RS scores over time did not significantly differ in the DBS vs control group. Significant predictors of lower QUIP‐RS scores over time in the cohort as a whole were lower baseline QUIP‐RS (β = 0.483, <0.001) and lower time‐varying LEDD (β = 0.003, P = 0.018) (Table 3). Similar results were obtained with the linear mixed‐effect regression model incorporating time‐varying LED‐DA as a covariate instead of total LEDD (Table 4), as well as when dopamine agonist use over time was examined as a categorical variable (Table S1.). Results were similar when the total score of all items of the QUIP‐RS, including hobbyism, punding, and DDS, was examined as the outcome measure (Table S2).
TABLE 3.
Predictors of Change in QUIP‐RS Scores; Results from Linear Mixed‐Effects Model; Model Adjusting for LEDD
| Variable | β (standard error) | 95% CI | P‐value |
|---|---|---|---|
| Intercept | 9.662 (4.524) | (0.598, 18.727) | 0.037 |
| Age | −0.105 (0.067) | (−0.24, 0.029) | 0.123 |
| Female sex (Male reference) | −1.7 (1.275) | (−4.264, 0.865) | 0.189 |
| Disease duration | −0.185 (0.101) | (−0.389, 0.019) | 0.074 |
| LEDD (time‐varying) | 0.003 (0.001) | (0, 0.005) | 0.018 |
| QUIP‐RS score (Baseline) | 0.483 (0.056) | (0.37, 0.595) | <0.001 |
| Timepoint (Baseline reference) | 0.905 | ||
| 3 months | −0.077 (1.504) | (−3.05, 2.897) | 0.959 |
| 6 months | −0.407 (1.488) | (−3.349, 2.534) | 0.785 |
| 1 year | 0.686 (1.489) | (−2.257, 3.629) | 0.646 |
| DBS (control reference) | 0.356 (1.624) | (−2.851, 3.563) | 0.827 |
| Treatment (DBS) * Time | 0.796 | ||
| DBS group: 3 months | −1.139 (2.199) | (−5.483, 3.206) | 0.605 |
| DBS group: 6 months | −2.083 (2.227) | (−6.483, 2.317) | 0.351 |
| DBS group: 1 year | −1.744 (2.197) | (−6.085, 2.597) | 0.429 |
Abbreviations: DBS, deep brain stimulation; LEDD, levodopa equivalent daily dose; QUIP‐RS, questionnaire for impulse control disorders in Parkinson's disease rating scale.
TABLE 4.
Predictors of Change in QUIP‐RS Scores; Results from Linear Mixed‐Effects Model; Model Adjusting for LED‐DA
| Variable | β (standard error) | 95% CI | P‐value |
|---|---|---|---|
| Intercept | 9.021 (4.815) | (−0.633, 18.675) | 0.066 |
| Age | −0.075 (0.07) | (−0.216, 0.066) | 0.29 |
| Female sex (Male reference) | −1.899 (1.353) | (−4.62, 0.821) | 0.167 |
| Disease duration | −0.136 (0.106) | (−0.349, 0.078) | 0.207 |
| LED‐DA total | 0.001 (0.004) | (−0.006, 0.009) | 0.75 |
| QUIP‐RS score (Baseline) | 0.508 (0.058) | (0.39, 0.625) | <0.001 |
| Timepoint (Baseline reference) | 0.876 | ||
| 3 months | −0.094 (1.506) | (−3.071, 2.883) | 0.95 |
| 6 months | −0.333 (1.489) | (−3.277, 2.61) | 0.823 |
| 1 year | 0.836 (1.491) | (−2.112, 3.783) | 0.576 |
| DBS versus control | 1.149 (1.638) | (−2.085, 4.383) | 0.484 |
| Treatment (DBS) * Time | 0.456 | ||
| DBS group: 3 months | −1.895 (2.179) | (−6.202, 2.412) | 0.386 |
| DBS group: 6 months | −3.156 (2.184) | (−7.473, 1.161) | 0.151 |
| DBS group: 1 year | −2.848 (2.154) | (−7.105, 1.409) | 0.188 |
Abbreviations: DBS, deep brain stimulation; LED‐DA, levodopa equivalent dose from dopamine agonists; QUIP‐RS, questionnaire for impulse control disorders in Parkinson's disease rating scale.
De Novo ICD Symptoms
Six of 13 (46%) patients who had QUIP‐RS score ≥ 10 at baseline dropped below this cut‐off at the 12‐month assessment (four in the DBS group and two in the control group) (Fig. 3). Eight of 41 patients (19.5%) developed de novo ICD symptoms at the 12‐month assessment, four in each group (Table 5). They were male, had a mean age of 60.6 years, and disease duration ranged from 1 to 13 years. None of the de novo cases met formal criteria for an ICD based on the structured diagnostic interview. Patterns of LEDD varied in the DBS group developing incident ICD symptoms: for two patients the LEDD had decreased at 12 months compared to baseline (1000 vs. 0 and 920 vs. 750), for one patient LEDD remained the same (LEDD = 800 mg), and for another LEDD increased (1000 vs. 1250 mg). LED‐DA did not change in those developing ICD symptoms.
Figure 3.
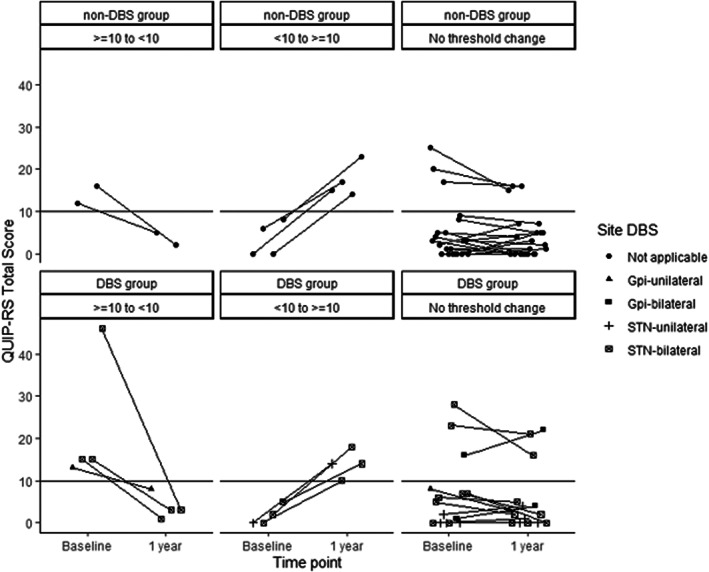
Within‐subject presence/absence of ICD based on QUIP‐RS score.
TABLE 5.
Characteristics of Patients with PD Who Experienced De Novo ICD Symptoms as defined by QUIP‐RS ≥10 at Year 1
| ID | Treatment group | Age* | Sex | Disease duration (years)** | DBS target | Unilateral or Bilateral | Baseline QUIP‐RS score | Baseline LEDD | Baseline LED‐DA | Year 1 QUIP‐RS | Year 1 LEDD | Year 1 LED‐DA |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | DBS | 61.95 | Male | 2 | STN | bilateral | 0 | 1000 | 0 | 10 | 0 | 0 |
| 2 | DBS | 55.80 | Male | 4 | STN | bilateral | 2 | 920 | 0 | 18 | 750 | 0 |
| 3 | DBS | 55.08 | Male | 4 | STN | bilateral | 5 | 800 | 0 | 14 | 800 | 0 |
| 4 | DBS | 60.45 | Male | 4 | STN | unilateral | 0 | 1000 | 0 | 14 | 1250 | 0 |
| 5 | Control | 62.82 | Male | 1 | n/a | n/a | 0 | 300 | 300 | 15 | 300 | 300 |
| 6 | Control | 61.18 | Male | 9 | n/a | n/a | 6 | 1169.5 | 0 | 17 | 1444 | 0 |
| 7 | Control | 68.64 | Male | 13 | n/a | n/a | 0 | 840 | 240 | 14 | 840 | 240 |
| 8 | Control | 58.76 | Male | 8 | n/a | n/a | 8 | 900 | 300 | 23 | 1050 | 300 |
Abbreviations: DBS, deep brain stimulation; ICD, impulse control disorder; LEDD, levodopa equivalent daily dose; LED‐DA, levodopa equivalent dose from dopamine agonists; QUIP‐RS, questionnaire for impulse control disorders in Parkinson's disease rating scale.
Age in years, at baseline visit (approximately one‐month prior to DBS surgery for patients undergoing surgery and first interview for the control group).
Disease duration (year at baseline visit—year of diagnosis of Parkinson's disease).
Discussion
This prospective observational study assessed 12‐month change in the severity of patient‐reported ICD symptoms in a group of advanced PD patients undergoing DBS when compared to a group of non‐surgical PD patients who were matched on several of the key risk factors for ICDs (age, sex, dopamine agonist use). While there were no significant differences when comparing the groups over time, QUIP‐RS scores decreased in the surgical group at 6 months follow up, approaching values observed in the control group. A similar effect was observed for medications. The relationship between higher prevalence and greater severity of ICDs and higher doses of dopaminergic medications in PD is well established. 2 , 3 , 7 , 30 It is therefore not surprising that QUIP‐RS scores decreased in the surgical group, coinciding with a reduction in LEDD. Indeed, in multivariate analyses, aside from baseline QUIP‐RS score, time‐varying LEDD was the only significant predictor of change in QUIP‐RS score over time, regardless of surgical group status. Our results are similar to another recent study without a control group which examined the short‐term effects of bilateral STN DBS in PD. 31 In that study, an overall significant change in QUIP‐RS was not observed, however baseline QUIP‐RS score and LEDD were the strongest predictors of change in post‐operative ICD symptoms. 31 In contrast to our study, it was LED‐DA rather than total LEDD that predicted the QUIP‐RS scores. This difference may be accounted for by the lower overall LED‐DA in our study, perhaps reflecting the recent reduction of dopamine agonist prescriptions in the USA. 32 , 33
There has been an extensive published literature on the change in ICD symptoms following DBS in PD, with some studies showing significant improvements in ICD symptoms post‐operatively, and others revealing no significant change. 2 What is consistent across the literature is evidence that changes in ICD symptoms post‐operatively are not uniform: some patients improve and some patients worsen. The observed variability in ICD symptoms post‐DBS may thus be in part explained by the surgical procedure itself, 2 , 5 , 34 individual susceptibility to ICD symptoms, and the interaction with medication changes and DBS. 2 When interpreting the existing literature, it is critical to acknowledge that in analyses examining the change in ICD symptoms following DBS, there is a dearth of studies providing comparison to similar PD patients without DBS. The EARLYSTIM trial 35 did perform such a comparison utilizing the Ardouin Scale of Behavior in Parkinson's Disease, with 14‐item subscale analysis showing overall significant improvement in “hyperdopaminergic behaviors” in patients with bilateral STN DBS compared to medical therapy alone. However, the improvement was mainly demonstrated in the subscale items of nocturnal hyperactivity, diurnal somnolence, creativity, and hobbyism. Differences in the rest of ICD behaviors, including punding, pathological gambling, hypersexuality, compulsive shopping, and “dopaminergic addiction,” were not observed. Although these results suggest overall improvement in hyperdopaminergic behaviors with DBS, the authors note that this study was not powered to analyze ICDs through individual Ardouin subscale items. Thus, a critical gap in knowledge persists as to the extent to which changes in ICD symptoms post‐DBS reported in the literature may have been influenced by the natural history of, and variability in, ICD symptoms irrespective of DBS therapy. This pattern of variability, that is seen both in DBS and control groups, is perhaps best exemplified in examining the cases with de novo ICD symptoms in our cohort—that is, the occurrence of ICD symptoms on follow‐up in individuals who did not have ICD symptoms at baseline.
A unique strength of the present study was the inclusion of a control, non‐surgical group, thus capturing the natural fluctuation in ICD symptoms observed in medicated PD patients over time. An important finding was that ~20% of patients in the cohort had worsening in ICD symptoms, four in the DBS group and four in the control group. Increases in LEDD could account for an increase in ICD symptoms in a subset of the patients. In addition, some may have had a baseline increased risk for ICDs such as male sex or longer PD disease duration 2 , 5 and also may have had exposure to risk factors that went unmeasured in our study. Regardless, our data suggest that both DBS‐ and medication‐ only groups should be monitored for ICDs. Additional key strengths of this study are the prospective design and recruitment from two centers in different regions of the country.
This study also had several limitations. Due to the small sample size, the study may have been underpowered to determine significant differences between DBS and control groups, and between different time points. Though the groups were matched on key demographic characteristics and medications known to be risk factors for ICDs, given the observational nature of this study, we cannot exclude the possibility of residual confounding. It is possible that our control group was different from the DBS group in unmeasured characteristics that may have confounded the relationship between DBS (or lack thereof) and the change in ICD symptoms. Our cohort was a convenience sample—only 30% of patients undergoing DBS during the study recruitment period were assessed for eligibility for the study—and the DBS group may not represent the broader PD population undergoing DBS. In addition, there may have been center‐specific differences in selection of patients for DBS candidacy or surgical approaches. Our study sample was predominantly White, male, had a high level of education and were residing in the United States, further limiting generalizability of our results. This is of particular importance when considering the variability in the prevalence of ICDs in PD, influenced by cultural and environmental factors. 2 Moreover, although patients were prospectively asked about their medication use by telephone interview, with no mechanism to ensure that patients were adherent to their medication there exists the possibility of misclassification of this important covariate; however, we would not expect this misclassification to be different between surgical and control groups. In addition, ICDs were assessed by telephone. It is possible that patients with ICDs may under‐report their symptoms, and input from informed others (caregivers) was not accounted for in assessing ICDs. Another limitation was that most of our sample had bilateral STN DBS and thus we could not examine differences in DBS targets on ICD symptoms, nor the impact of unilateral vs bilateral procedures. Finally, identifying matched non‐surgical groups to compare to patients undergoing DBS remains a challenge for the field.
Conclusions
In this observational study comparing ICD symptoms in patients undergoing DBS to a non‐surgical (medication‐only) control group, at 12 months follow‐up, both groups had similar degree of ICD symptoms. De novo ICD symptoms occurred in 8/41 patients (20%), four patients in each group, although none met formal diagnostic criteria for an actual impulse control disorder on structured interview. The findings from the study highlight the strong relationship of ICD symptoms to the overall dopaminergic medication burden and not just dopamine agonists. Observed changes in the QUIP‐RS scores inform the field on the natural variability in ICD symptoms when treating PD, regardless of DBS status. The data from this study suggest that clinicians should be monitoring both DBS and medication‐only PD groups for the emergence of ICD symptoms. Further controlled studies with larger sample sizes examining the effect of different DBS targets and with more diverse populations should be performed.
Author Roles
(1) Research project: A. Conception, B. Organization, C. Execution; (2) Statistical Analysis: A. Design, B. Execution, C. Review and Critique; (3) Manuscript Preparation: A. Writing of the first draft, B. Review and Critique.
P.H.C.: 1B, 2C, 3A, 3B
I.L.: 3A, 3B
E.M.: 1B, 1C, 3B
R.F.: 2B, 2C, 3A, 3B
A.A.: 2A, 2B, 2C, 3B
W.D.: 1B, 1C, 2C, 3B
R.B.: 1B, 1C, 3B
R.E.: 1C, 2C, 3B
M.S.: 1B, 1C, 3B
M.S.O.: 1A, 2A, 2C, 3B
D.W.: 1A, 1B, 2A, 2C, 3A, 3B
L.M.C.: 1A, 1B, 1C, 2A, 2C, 3A, 3B
Disclosures
Ethical Compliance Statement: This study was approved by the Institutional Review Boards of the University of Pennsylvania and the University of Florida. Written informed consent was obtained from each participant before study initiation, as part of pre‐operative multidisciplinary clinics research study procedures at each institution. Signed informed consents were scanned, uploaded and saved in REDCap electronic data capture tools.
All authors confirm that they have read the Journal's position on issues involved in ethical publication and affirm that this work is consistent with those guidelines.
Funding Sources and Conflicts of Interest: This project was partially funded by the University of Pittsburgh Dean's Faculty Advancement Award (Dr. Chahine, PI). The authors declare that there are no conflicts of interest relevant to this work.
Financial Disclosures for the Previous 12 Months: Pilar Hernandez‐Con, Iris Lin, Eugenia Mamikonyan, Robert Feldman, Wissam Deeb and Ryan Barmore have no disclosures to report. Andrew Althouse's institution receives clinical research funding from National Institutes of Health, Cystic Fibrosis Foundation, and Abbott. Robert S. Eisinger is supported by NINDS F30NS111841. Meredith Spindler's institution receives clinical research funding from Abbvie, Abbott, UCB, Supernus, Takeda, Sanofi, and Praxis. Dr. Spindler has received consulting fees from Medtronic and Supernus, and royalties from Wolters Kluwel for authorship. Michael S. Okun serves as Medical Advisor for the Parkinson's Foundation, and has received research grants from NIH, Parkinson's Foundation, the Michael J. Fox Foundation, the Parkinson Alliance, Smallwood Foundation, the Bachmann‐Strauss Foundation, the Tourette Syndrome Association, and the UF Foundation. Dr. Okun's research is supported by: NIH R01 NR014852, R01NS096008, UH3NS119844, U01NS119562. Dr. Okun is PI of the NIH R25NS108939 Training Grant. Dr. Okun has received royalties for publications with Demos, Manson, Amazon, Smashwords, Books4Patients, Perseus, Robert Rose, Oxford and Cambridge (movement disorders books). Dr. Okun is an associate editor for New England Journal of Medicine, Journal Watch Neurology and JAMA Neurology. Dr. Okun has participated in CME and educational activities (past 12–24 months) on movement disorders sponsored by WebMD/Medscape, RMEI Medical Education, American Academy of Neurology, Movement Disorders Society, Mediflix and by Vanderbilt University. The institution and not Dr. Okun receives grants from Medtronic, Abbvie, Boston Scientific, Abbott and Allergan and the PI has no financial interest in these grants. Dr. Okun has participated as a site PI and/or co‐I for several NIH, foundation, and industry sponsored trials over the years but has not received honoraria. Research projects at the University of Florida receive device and drug donations. Daniel Weintraub has received research funding or support from Michael J. Fox Foundation for Parkinson's Research, Alzheimer's Therapeutic Research Initiative (ATRI), Alzheimer's Disease Cooperative Study (ADCS), International Parkinson and Movement Disorder Society (IPMDS), National Institute on Health (NIH), Parkinson's Foundation; U.S. Department of Veterans Affairs and Acadia Pharmaceuticals; honoraria for consultancy from Acadia Pharmaceuticals, Alkahest, Aptinyx, Cerevel Therapeutics, CHDI Foundation, Clintrex LLC (Otsuka), EcoR1 Capital, Eisai, Ferring, Gray Matter Technologies, Great Lake Neurotechnologies, Intra‐Cellular Therapies, Janssen, Merck, Sage, Scion and Signant Health; and license fee payments from the University of Pennsylvania for the QUIP and QUIP‐RS. Lana M. Chahine receives research support from the Michael J. Fox Foundation, UPMC Competitive Medical Research Fund, National Institutes of Health, and University of Pittsburgh, is study site investigator for a study sponsored by Biogen, receives consulting fees from Gray Matter Technologies, receives royalties from Elsevier (for authorship), and receives royalties from Wolters Kluwel (for authorship).
Supporting information
Table S1. Predictors of change in QUIP‐RS score (sum of buying, eating, gambling, and hypersexuality items); results from linear mixed‐effects model; model adjusting for dopamine agonist use as a categorical variable.
Table S2. Predictors of change in QUIP‐RS total score for all items assessed (primary ICDs + hobbyism, punding, and DDS). Results from linear mixed‐effects model
Acknowledgments
This work was partially funded by the University of Pittsburgh School of Medicine Dean's Faculty Advancement Award (Dr. Chahine, PI).
We thank the research participants.
REFERENCES
- 1. Weintraub D, Koester J, Potenza MN, Siderowf AD, Stacy M, Voon V, et al. Impulse control disorders in Parkinson disease: a cross‐sectional study of 3090 patients. Arch Neurol 2010;67(5):589–595. [DOI] [PubMed] [Google Scholar]
- 2. Eisinger RS, Ramirez‐Zamora A, Carbunaru S, Ptak B, Peng‐Chen Z, Okun MS, Gunduz A. Medications, deep brain stimulation, and other factors influencing impulse control disorders in parkinson's disease. Front Neurol 2019;10:86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Ceravolo R, Frosini D, Rossi C, Bonuccelli U. Impulse control disorders in Parkinson's disease: definition, epidemiology, risk factors, neurobiology and management. Parkinsonism Relat Disord 2009;15(Suppl 4):S111–S115. [DOI] [PubMed] [Google Scholar]
- 4. Weintraub D, David AS, Evans AH, Grant JE, Stacy M. Clinical spectrum of impulse control disorders in Parkinson's disease. Mov Disord 2015;30(2):121–127. [DOI] [PubMed] [Google Scholar]
- 5. Weintraub D, Mamikonyan E. Impulse control disorders in parkinson's disease. Am J Psychiatry 2019;176(1):5–11. [DOI] [PubMed] [Google Scholar]
- 6. Weintraub D, Papay K, Siderowf A, Parkinson's Progression Markers I . Screening for impulse control symptoms in patients with de novo Parkinson disease: a case‐control study. Neurology 2013;80(2):176–180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Weintraub D. Dopamine and impulse control disorders in Parkinson's disease. Ann Neurol 2008;64(Suppl 2):S93–S100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Weintraub D, Siderowf AD, Potenza MN, Goveas J, Morales KH, Duda JE, et al. Association of dopamine agonist use with impulse control disorders in Parkinson disease. Arch Neurol 2006;63(7):969–973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Voon V, Fernagut PO, Wickens J, et al. Chronic dopaminergic stimulation in Parkinson's disease: from dyskinesias to impulse control disorders. Lancet Neurol 2009;8(12):1140–1149. [DOI] [PubMed] [Google Scholar]
- 10. Silveira‐Moriyama L, Evans AH, Katzenschlager R, Lees AJ. Punding and dyskinesias. Mov Disord 2006;21(12):2214–2217. [DOI] [PubMed] [Google Scholar]
- 11. Faouzi J, Corvol J‐C, Mariani L‐L. Impulse control disorders and related behaviors in Parkinson's disease: risk factors, clinical and genetic aspects, and management. Curr Opin Neurol 2021;34(4):547–555. [DOI] [PubMed] [Google Scholar]
- 12. Antonini A, Stoessl AJ, Kleinman LS, et al. Developing consensus among movement disorder specialists on clinical indicators for identification and management of advanced Parkinson's disease: a multi‐country Delphi‐panel approach. Curr Med Res Opin 2018;34(12):2063–2073. [DOI] [PubMed] [Google Scholar]
- 13. Shotbolt P, Moriarty J, Costello A, Jha A, David A, Ashkan K, Samuel M. Relationships between deep brain stimulation and impulse control disorders in Parkinson's disease, with a literature review. Parkinsonism Relat Disord 2012;18(1):10–16. [DOI] [PubMed] [Google Scholar]
- 14. Merola A, Romagnolo A, Rizzi L, et al. Impulse control behaviors and subthalamic deep brain stimulation in Parkinson disease. J Neurol 2017;264(1):40–48. [DOI] [PubMed] [Google Scholar]
- 15. Bandini F, Primavera A, Pizzorno M, Cocito L. Using STN DBS and medication reduction as a strategy to treat pathological gambling in Parkinson's disease. Parkinsonism Relat Disord 2007;13(6):369–371. [DOI] [PubMed] [Google Scholar]
- 16. Ardouin C, Voon V, Worbe Y, Abouazar N, Czernecki V, Hosseini H, et al. Pathological gambling in Parkinson's disease improves on chronic subthalamic nucleus stimulation. Mov Disord 2006;21(11):1941–1946. [DOI] [PubMed] [Google Scholar]
- 17. Amami P, Dekker I, Piacentini S, et al. Impulse control behaviours in patients with Parkinson's disease after subthalamic deep brain stimulation: de novo cases and 3‐year follow‐up. J Neurol Neurosurg Psychiatry 2015;86(5):562–564. [DOI] [PubMed] [Google Scholar]
- 18. Eusebio A, Witjas T, Cohen J, Fluchère F, Jouve E, Régis J, et al. Subthalamic nucleus stimulation and compulsive use of dopaminergic medication in Parkinson's disease. J Neurol Neurosurg Psychiatry 2013;84(8):868–874. [DOI] [PubMed] [Google Scholar]
- 19. Lim S‐Y, O'Sullivan SS, Kotschet K, Gallagher DA, Lacey C, Lawrence AD, et al. Dopamine dysregulation syndrome, impulse control disorders and punding after deep brain stimulation surgery for Parkinson's disease. J Clin Neurosci 2009;16(9):1148–1152. [DOI] [PubMed] [Google Scholar]
- 20. Amstutz D, Paschen S, Lachenmayer ML, Maradan‐Gachet ME, Deuschl G, Krack P, Debove I. Management of Impulse Control Disorders with subthalamic nucleus deep brain stimulation in Parkinson's disease. CNS Neurol Disord Drug Targets 2020;19(8):611–617. [DOI] [PubMed] [Google Scholar]
- 21. Mehta SH. DBS and impulse control disorders in PD: I think we got this…Wait! We don't. Parkinsonism Relat Disord 2018;56:1–2. [DOI] [PubMed] [Google Scholar]
- 22. Witjas T, Baunez C, Henry JM, Delfini M, Regis J, Cherif AA, et al. Addiction in Parkinson's disease: impact of subthalamic nucleus deep brain stimulation. Mov Disord 2005;20(8):1052–1055. [DOI] [PubMed] [Google Scholar]
- 23. Moum SJ, Price CC, Limotai N, Oyama G, Ward H, Jacobson C, et al. Effects of STN and GPi deep brain stimulation on impulse control disorders and dopamine dysregulation syndrome. Plos One 2012;7(1):e29768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Kim A, Kim YE, Kim H‐J, et al. A 7‐year observation of the effect of subthalamic deep brain stimulation on impulse control disorder in patients with Parkinson's disease. Parkinsonism Relat Disord 2018;56:3–8. [DOI] [PubMed] [Google Scholar]
- 25. Santin MN, Voulleminot P, Vrillon A, Hainque E, Béreau M, Lagha‐Boukbiza O, et al. Impact of subthalamic deep brain stimulation on impulse control disorders in parkinson's disease: a prospective study. Mov Disord 2021;36(3):750–757. [DOI] [PubMed] [Google Scholar]
- 26. Weintraub D, Mamikonyan E, Papay K, Shea JA, Xie SX, Siderowf A. Questionnaire for impulsive‐compulsive disorders in Parkinson's disease‐rating scale. Mov Disord 2012;27(2):242–247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Evans AH, Okai D, Weintraub D, Lim S‐Y, O'Sullivan SS, Voon V, et al. Scales to assess impulsive and compulsive behaviors in Parkinson's disease: critique and recommendations. Mov Disord 2019;34(6):791–798. [DOI] [PubMed] [Google Scholar]
- 28. Tomlinson CL, Stowe R, Patel S, Rick C, Gray R, Clarke CE. Systematic review of levodopa dose equivalency reporting in Parkinson's disease. Mov Disord 2010;25(15):2649–2653. [DOI] [PubMed] [Google Scholar]
- 29. Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JG. Research electronic data capture (REDCap) – a metadata‐driven methodology and workflow process for providing translational research informatics support. J Biomed Inform 2009;42(2):377–381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Balarajah S, Cavanna AE. The pathophysiology of impulse control disorders in Parkinson disease. Behav Neurol 2013;26(4):237–244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Sauerbier A, Loehrer P, Jost ST, et al. Predictors of short‐term impulsive and compulsive behaviour after subthalamic stimulation in Parkinson disease. J Neurol Neurosurg Psychiatry 2021;92(12):1313–1318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Crispo JAG, Fortin Y, Thibault DP, Emons M, Bjerre LM, Kohen DE, et al. Trends in inpatient antiparkinson drug use in the USA, 2001‐2012. Eur J Clin Pharmacol 2015;71(8):1011–1019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Orayj K, Lane E. Patterns and determinants of prescribing for parkinson's disease: a systematic literature review. Parkinson's Dis 2019;2019:9237181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Rossi PJ, Gunduz A, Okun MS. The subthalamic nucleus, limbic function, and impulse control. Neuropsychol Rev 2015;25(4):398–410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Lhommée E, Wojtecki L, Czernecki V, et al. Behavioural outcomes of subthalamic stimulation and medical therapy versus medical therapy alone for Parkinson's disease with early motor complications (EARLYSTIM trial): secondary analysis of an open‐label randomised trial. Lancet Neurol 2018;17(3):223–231. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Table S1. Predictors of change in QUIP‐RS score (sum of buying, eating, gambling, and hypersexuality items); results from linear mixed‐effects model; model adjusting for dopamine agonist use as a categorical variable.
Table S2. Predictors of change in QUIP‐RS total score for all items assessed (primary ICDs + hobbyism, punding, and DDS). Results from linear mixed‐effects model


