Abstract
Mesonephric remnants (MRs) are embryonic vestiges most commonly found in female pathology specimens from the lateral wall of the cervix. The highly regulated genetic programme of mesonephric duct development has been well characterised in animals based on traditional surgical castration and knockout mouse experiments. However, the process is incompletely understood in humans. MRs are believed to give rise to mesonephric neoplasms, which are rare tumours with uncertain pathophysiology. There is a dearth of molecular studies on mesonephric neoplasms in part due to their rarity. Here, we report the results of next-generation sequencing of MR, which identified amplification of the androgen receptor gene for the first time to the best of our knowledge and discuss the potential implications in the context of the literature.
Keywords: Pathology, Obstetrics and gynaecology
Background
Remnants of the mesonephric duct (MD), also known as Wolffian duct, Malpighi-Gartner’s duct or ductus longitudinalis epoophori, were rediscovered by Hermann Gartner in 1822 and can be seen occasionally in surgical pathology specimens representing meso-ovaries, broad ligaments or lateral walls of cervix and vagina. In the absence of masculinising hormones in the female fetus, the MD regresses.1 However, vestiges of these structures may persist along the female genital tract as remnants, which may subsequently give rise to cysts and neoplasms. In most cases, mesonephric remnants/rests (MRs) are small and identified incidentally in asymptomatic women. Their prevalence varies from approximately 1% in adults to 40% in newborns and children, and their most frequent location is the lateral wall of the cervix.2 True mesonephric hyperplasia has been defined as remnants larger than 6 mm and could be clinically detected due to their association with erosions, nodularity or abnormal gynaecological cytology. The distinction from hyperplasia can be quite arbitrary, and large series on benign mesonephric neoplasms have been published.3 In contrast, malignant mesonephric tumours are very rare and have yet to be fully characterised both morphologically and molecularly.3 4
Here, we report a case of MR characterised by immunohistochemistry and next-generation sequencing (NGS), revealing amplification of the androgen receptor (AR) gene.
Case presentation
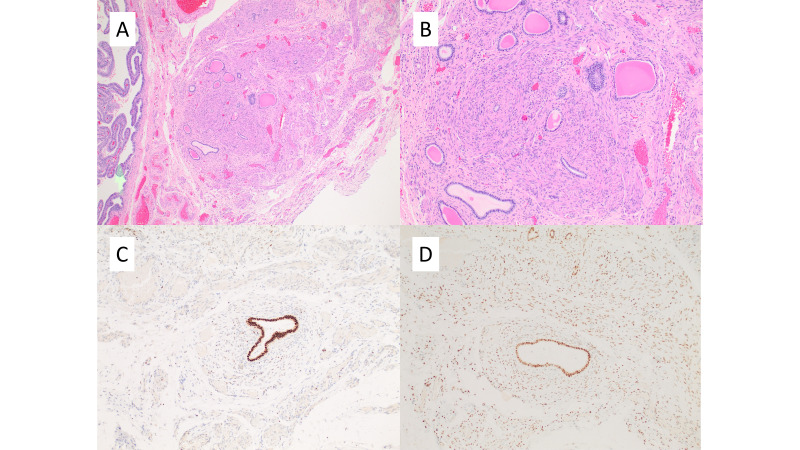
A woman in her 40s, G2P2 with a clinical history of menometrorrhagia, severe dysmenorrhoea and endometriosis underwent laparoscopic hysterectomy and bilateral salpingectomy in August 2020. Histopathology revealed benign proliferative endometrium, mild chronic cervicitis and endometriosis involving fallopian tubes. In addition, a focus of MR (figure 1) measuring up to 0.4 cm was found incidentally. Immunohistochemical staining (figure 1) showed the cuboidal epithelium of the remnants to be positive for GATA3, PAX8, CK7, P16 (focally) and AR, and the stromal cells to be positive for WT1, as expected. NGS (Oncomine Focus, ThermoFisher) of DNA revealed amplification of AR (copy number of 6.1). The patient had an uneventful postoperative recovery and has remained asymptomatic for 2 years.
Figure 1.
H&E stains of mesonephric remnants at low (40X) and medium power (100X) in A and B, respectively. Immunohistochemistry of mesonephric remnants showing positivity on epithelial cells for GATA3 (100X) and AR (100X, deeper section) in C and D, respectively.
Outcome and follow-up
One month after laparoscopic hysterectomy and bilateral salpingectomy, the patient was able to return to her regular and daily activities. Two years after the surgery, the patient is in good health.
Discussion
The MD is present both in the male and female embryo and forms the trigone of the urinary bladder. In males, it also develops into structures of the internal genitalia (epididymis, vas deferens and seminal vesicles) by a genetic programme that has been more recently characterised using knockout mice and is still incompletely understood in humans.5 In females, the absence of fetal testicular hormones (mainly testosterone and androstenedione) and anti-mullerian hormone (a product of Sertoli cells in males) determines a default process by which the MD regress,5 and female phenotypic development of the mullerian duct ensues.6
Several genes are involved in this process, including paired box genes (PAX2 and PAX8), homeobox genes (EMX2, LIM1, HOXA9, HOXA10, HOXA11 and HOXA13), anti-mullerian hormone, epidermal growth factor receptor and AR, which act in a complex interplay requiring mesenchymal/epithelial signalling. Interestingly, AR is expressed in humans only in the epithelium of the MD. On activation by binding to androgens, AR translocates from the cytoplasm to the nucleus, triggering a transcriptional cascade that prevents MD degeneration in a time-sensitive manner. Therefore, inhibition of AR (or lack of testosterone) during weeks 8 –13 of gestation, results in disrupted stabilisation of MD, as seen in normal females.7
Despite much progress, the understanding of MD development is still incomplete, compromising the characterisation of tumourigenesis in mesonephric neoplasms.8 Interestingly, MD failed to regress in some male patients with inactivating mutations of AR suffering from complete androgen insensitivity syndrome,9 underscoring the complexity of AR signalling in mesonephric development, and the fact that some AR functions are preserved despite severe AR mutations.
Mesonephric tumours have been well characterised by immunohistochemistry and are typically positive for CK7, PAX8, GATA3, CD10, calretinin, HNF1β and AR; and negative for CK20, CDX2, CEA, oestrogen receptor, progesterone receptor, TTF1, p16 (patchy), napsin A, racemase and p53.10 Recently, genetic alterations in various genes (KRAS, NRAS, BRAF, CTNNB1, PIK3CA, PTEN, ARID1A, TP53, MLL2, CDKN2A, and GATA3) and large chromosomic aberrations (gains or 1q, 10 and 12; and losses of 9) have been reported in mesonephric and mesonephric-like adenocarcinoma.2 11 12 However, molecular characterisation of benign mesonephric proliferations is lacking in PubMed and may be informative because they are believed to give rise to their malignant counterparts.
Even though AR alterations (including amplification) are well established in the pathogenesis of prostatic carcinoma and have also been reported in malignancies from other origins, including breast, lung, colon and endometrium,13 they have not been described in mesonephric tumours to the best of our knowledge. Nonetheless, we found AR amplification in MR, which is intriguing and supports the importance of AR signalling in the stabilisation of mesonephric tissue, suggesting a driver role in mesonephric proliferations.
Learning points.
Mesonephric remnants (MRs) are vestiges of the Wolffian ducts, which usually regress during normal embryonal development in women and are believed to give rise to malignant neoplasms through an incompletely understood process.
Molecular characterisation of mesonephric lesions is needed to better understand neoplastic transformation.
Here, AR gene amplification in MR is reported, which contributes to the molecular characterisation of mesonephric proliferations.
Footnotes
Contributors: GS wrote the first draft and edited additional versions. AZ contributed to the clinical and pathologic diagnosis and edited the manuscript. VEN designed the study, took micrographs and edited the manuscript.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Case reports provide a valuable learning resource for the scientific community and can indicate areas of interest for future research. They should not be used in isolation to guide treatment choices or public health policy.
Competing interests: None declared.
Provenance and peer review: Not commissioned; externally peer reviewed.
Ethics statements
Patient consent for publication
Consent obtained directly from patient(s).
References
- 1.Makiyan Z. Studies of Gonadal sex differentiation. Organogenesis 2016;12:42–51. 10.1080/15476278.2016.1145318 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Montalvo N, Redrobán L, Galarza D. Mesonephric adenocarcinoma of the cervix: a case report with a three-year follow-up, lung metastases, and next-generation sequencing analysis. Diagn Pathol 2019;14. 10.1186/s13000-019-0847-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ferry JA, Scully RE. Mesonephric remnants, hyperplasia, and neoplasia in the uterine cervix. The American Journal of Surgical Pathology 1990;14:1100–11. 10.1097/00000478-199012000-00002 [DOI] [PubMed] [Google Scholar]
- 4.Bagué S, Rodríguez IM, Prat J. Malignant mesonephric tumors of the female genital tract: a clinicopathologic study of 9 cases. Am J Surg Pathol 2004;28:601–7. [DOI] [PubMed] [Google Scholar]
- 5.Murashima A, Xu B, Hinton BT. Understanding normal and abnormal development of the Wolffian/epididymal duct by using transgenic mice. Asian J Androl 2015;17:749–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Cunha GR, Robboy SJ, Kurita T, et al. Development of the human female reproductive tract. Differentiation 2018;103:46–65.:S0301-4681(18)30100-2. 10.1016/j.diff.2018.09.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Zhao F, Yao HH-C. A tale of two tracts: history, current advances, and future directions of research on sexual differentiation of reproductive tracts†. Biol Reprod 2019;101:602–16. 10.1093/biolre/ioz079 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Hannema SE, Hughes IA. Regulation of Wolffian duct development. Horm Res Paediatr 2007;67:142–51. 10.1159/000096644 [DOI] [PubMed] [Google Scholar]
- 9.Hannema SE, Scott IS, Hodapp J, et al. Residual activity of mutant androgen receptors explains Wolffian duct development in the complete androgen insensitivity syndrome. J Clin Endocrinol Metab 2004;89:5815–22. 10.1210/jc.2004-0709 [DOI] [PubMed] [Google Scholar]
- 10.Buza N. Immunohistochemistry in gynecologic carcinomas: practical update with diagnostic and clinical considerations based on the 2020 who classification of tumors. Semin Diagn Pathol 2022;39:58–77. 10.1053/j.semdp.2021.10.006 [DOI] [PubMed] [Google Scholar]
- 11.Lin DI, Shah N, Tse JY, et al. Molecular profiling of Mesonephric and Mesonephric-like Carcinomas of Cervical, endometrial and ovarian origin. Gynecol Oncol Rep 2020;34:100652. 10.1016/j.gore.2020.100652 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.da Silva EM, Fix DJ, Sebastiao APM, et al. Mesonephric and mesonephric-like carcinomas of the female genital tract: molecular characterization including cases with mixed histology and matched metastases. Mod Pathol 2021;34:1570–87. 10.1038/s41379-021-00799-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.André F, et al. , The AACR Project GENIE Consortium, The AACR Project GENIE Consortium . The AACR project genie Consortium. AACR project genie: Powering precision medicine through an international Consortium. Cancer Discov 2017;7:818–31. [DOI] [PMC free article] [PubMed] [Google Scholar]