Summary:
Forequarter amputations in advanced local malignancy or trauma cases often leave a large defect that is challenging to reconstruct. Options for defect closure are varied. A vertical rectus abdominis myocutaneous (VRAM) flap could be an alternative to close a significantly large defect, which is relatively easier than the more technically demanding free flap. This case presents a 64-year-old man with a soft tissue sarcoma in the left shoulder that was treated by forequarter amputation and subsequent defect closure using a VRAM flap. The VRAM flap was initially used to reconstruct the chest and abdominal walls. There have been no reported uses for the shoulder defect. The repair site defect was viable even with a less aesthetic donor site, and all of the defects were closed without any signs of infection. The VRAM flap is a good option for a large defect closure at the shoulder region, particularly after forequarter amputation.
Forequarter amputation (FQA) is a type of limb ablation surgery that involves the removal of an entire upper extremity, including the shoulder girdle, which results in satisfying clinical (and oncologic) outcomes.1–3 However, it results in a large defect that necessitates a further reconstructive procedure. Various closure modalities had been proposed such as the skin graft, local flaps, pedicled flaps, and free flaps.4,5
The vertical rectus abdominis myocutaneous (VRAM) flap is a versatile myocutanous flap which is commonly used for breast, chest wall, and groin to perineal region defect reconstruction. It is well vascularized, relatively bulky, and technically easy. In this report, we present a 64-year-old man with soft tissue sarcoma treated with FQA and VRAM flap. This patient had low demand and was severely ill. Thus, we performed a nonmicrosurgical reconstruction of the FQA defect to prevent a prolonged surgery. To our knowledge, this is the first report of VRAM utilization for shoulder defect closure.
CASE PRESENTATION
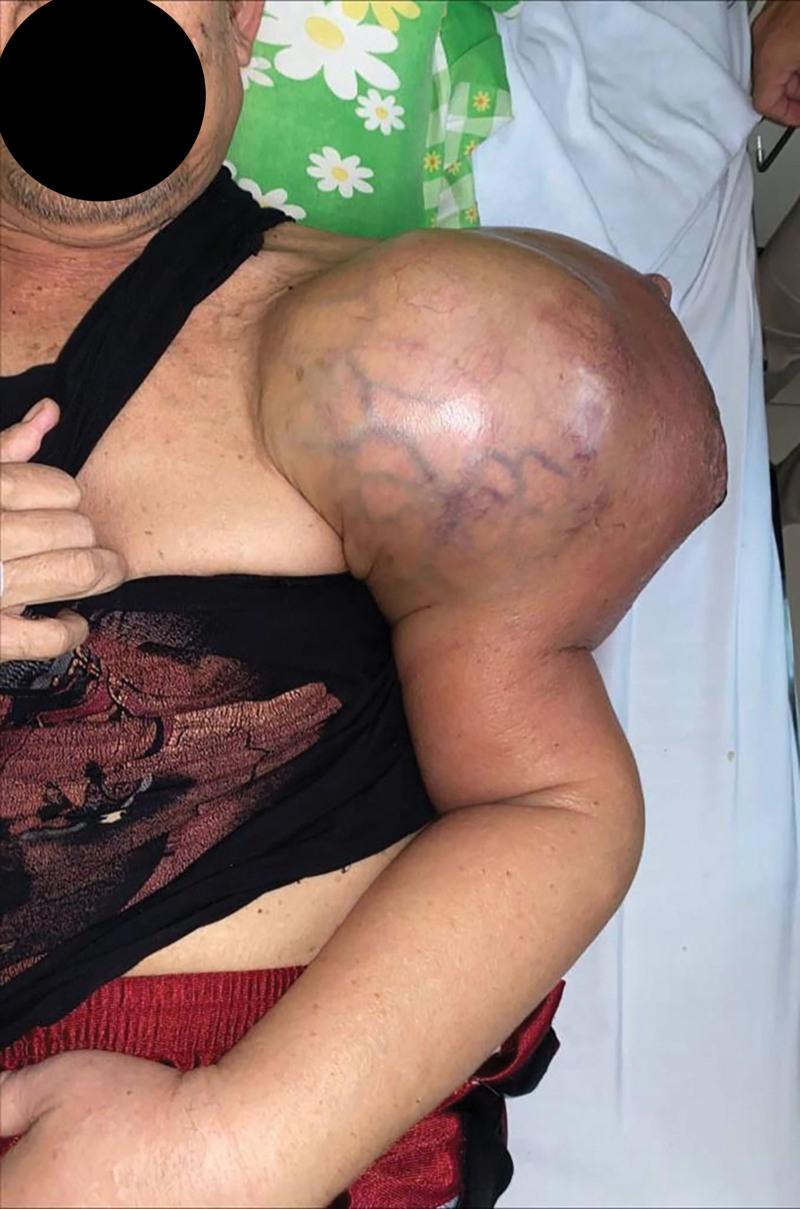
A 64-year-old man complained of a painful mass on the left shoulder that had enlarged in the past year from the size of an egg to the size of a basketball and was accompanied by dyspnea in the last 3 months. There was no previous treatment or history of other malignancies. Physical examination revealed a solid mass in the left shoulder with local tenderness visual analog score of 5, venectation, and distal edema (Fig. 1). Sensory examination revealed distal sensory hypoesthesia. A plain radiograph showed a large radiopaque mass around the proximal humerus, with a suspicion of soft tissue sarcoma. MRI showed a soft tissue mass with heterogenous hyperintensity that had destroyed the proximal humerus, scapula, and lateral end of the clavicle; caused glenohumeral joint destruction and dislocation; and involved the major brachial neurovascular bundle.
Fig. 1.
Preoperative clinical picture of the patient.
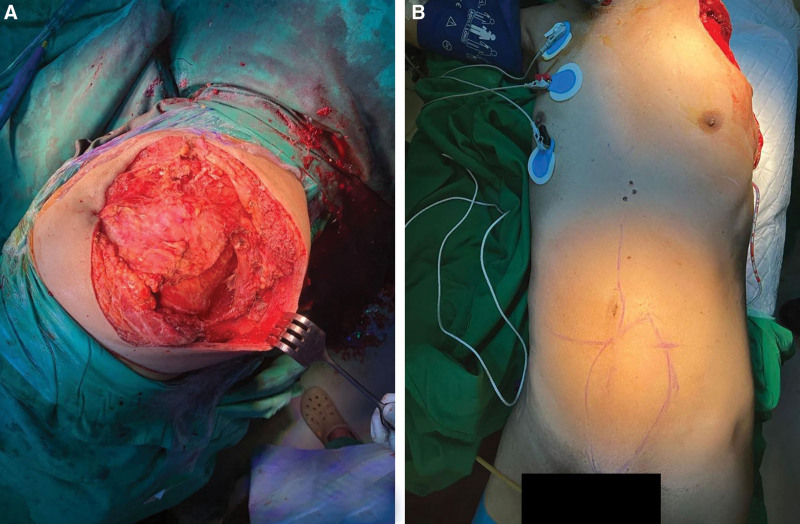
The working diagnosis was primary malignant soft tissue tumor of the left shoulder, with a suspicion of rhabdomyosarcoma. Extensive tumor excision with FQA was performed and left a 10 x 11 cm defect (Fig. 2). The axillary vessels and infraclavicular brachial plexus were involved with the tumor.
Fig. 2.
Intraoperative condition after tumor resection. A, Defect on the left shoulder. B, VRAM flap design.
Surgical Technique
A 15 x 9 cm VRAM pedicled island flap was designed based on the left deep superior epigastric vessels (Fig. 2). To ensure an adequate length of rotation, the length from the xyphoid to the edge of the defect was measured. Skin and fascia were incised, and the fascia was sutured to the skin to prevent shearing on the perforators. The rectus muscle was cut several centimeters wider than the harvested anterior rectus sheath fascia. The muscle was dissected from the posterior rectus sheath and raised from inferior to superior after ligating the inferior epigastric vessels. The donor site fascia was closed with a mesh, and the skin was primarily closed. The flap was then inset into the defect area through a subcutaneous tunnel. [See graphic, Supplemental Digital Content 1, which displays the schematic flap design. (1) The superior epigastric vasculature-based myo-cutaneous VRAM flap is raised. (2) The incision is extended superiorly, and the pedicle is freed until the level of xyphoid. (3) The flap is rotated to match the desired position before it is passed through the subcutaneous tunnel. http://links.lww.com/PRSGO/C618]
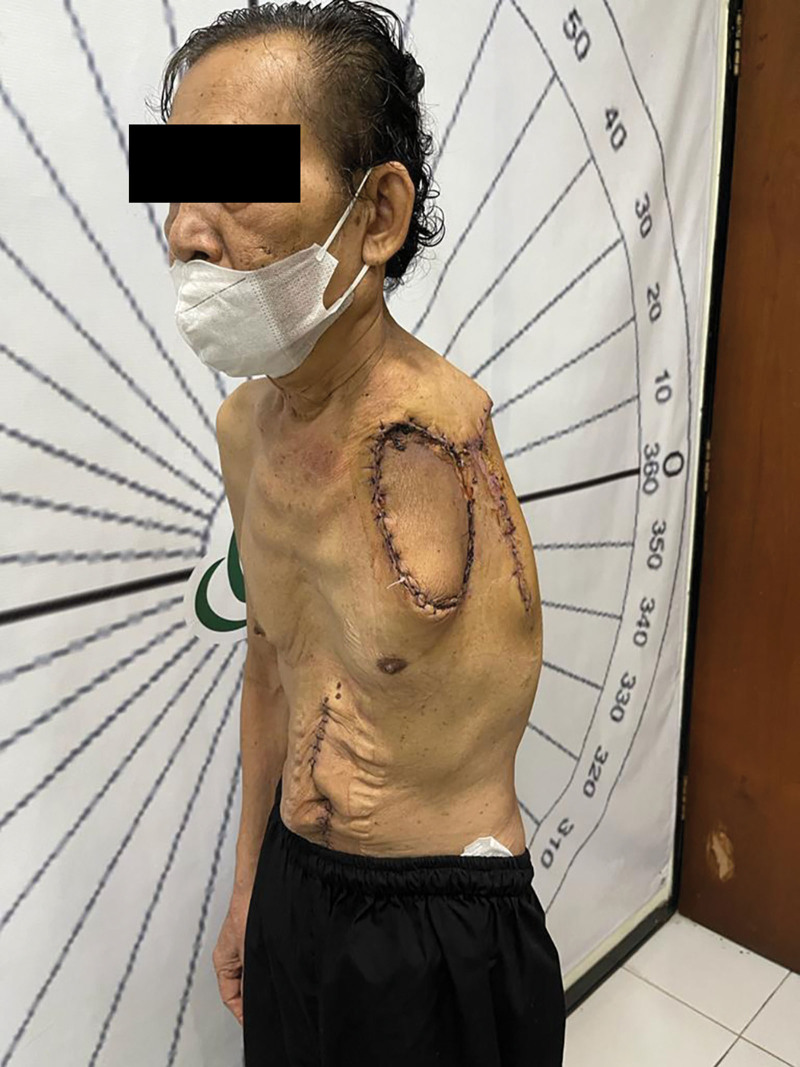
The patient reported a significant reduction in pain in the immediate postoperative period. The flap was viable without any signs of infection at the postoperative outpatient clinic visit 2 weeks after hospital discharge (Fig. 3). The pathology result confirmed the diagnosis of myxofibrosarcoma. The patient was sent for chemotherapy. Unfortunately, the patient died approximately two months after surgery, which emphasizes the severity of the condition.
Fig. 3.
Postoperative clinical examination of the patient.
DISCUSSION
In our patient, FQA was performed due to an extensive and life-threatening sarcoma that attached to the major neurovascular bundle. Compared with limb-salvage surgery, FQA may result in a higher opportunity to achieve adequate oncological margins for a curative approach. Elsner et al3 and Parsons et al6 reported an adequate oncological margin in more than 90%, with no local relapse in those who were initially treated with FQA.3,6,7 However, it leaves a large defect that necessitates a further reconstructive procedure.
The free fillet flap, which uses tissues from the amputated limb (“spare parts” concept), is one of the options without donor site morbidity.8 However, this flap primarily relies on the donor condition. Tumor extension to the amputated part is the contraindication of a free fillet flap, similar to our patient’s condition. Because of the potential difficulty in the wound healing process, lymphedema is also a relative contraindication. Moreover, any microsurgical procedures may come with a risk of anastomosis failure.9 Considering the surgical duration and patient condition, we tried to reduce the surgical duration by utilizing nonmicrosurgical reconstruction.
Options for pedicled flaps are the pectoralis major, latissimus dorsi, and rectus abdominis flaps. In our patient, the pectoralis muscle and the latissimus dorsi pedicle were involved with the tumor. Moreover, the pectoralis major flap is more suitable for defects in the sternal region, whereas the latissimus dorsi flap is more suitable in lateral defects. Other alternatives include the omental flap, which requires more invasive surgery to harvest the flap.10,11
The use of a VRAM flap in shoulder a defect has never been reported in literature. We performed VRAM flap transposition to fill the 10 cm defect in the left shoulder. It has relatively abundant tissue, which may provide bulk transfer to the recipient site.12 Compared with the transverse RAM flap, the VRAM is associated with better blood supply due to its advantage of maximum capture of perforating vessels at the defect. However, several potential drawbacks have been reported related to the VRAM flap, including donor site scarring and a smaller availability of skin paddles compared with the transverse RAM flap. The resultant changes in core strength with loss of a unilateral rectus muscle is another potential drawback, although it is usually well tolerated.13–15
Immunosuppression, obesity, smoking, and diabetes mellitus are associated with surgical complications such as flap necrosis.16 However, it can be prevented by ensuring proper tissue tension and adequate tissue perfusion17,18 In our patient, the flap was viable without skin necrosis or infection. The limitation of this study is lack of long-term follow-up on the patient. The terminally ill condition seen in many oncologic patients rarely provides sufficient follow-up for postoperative evaluation. However, we believe that this method of reconstruction is fast and effective for closing the FQA defect after malignant tumor excision.
CONCLUSIONS
The VRAM flap is a good option for managing the defect at the shoulder after FQA. It is a simple yet quick-to-harvest flap that may provide coverage in a large defect with a stable blood supply.
DISCLOSURE
The authors have no financial interest to declare in relation to the content of this article.
PATIENT CONSENT
The patient provided written consent for the use of his image.
ACKNOWLEDGMENTS
Ethical approval was waived at our institution. This article is reporting a case that consists of only one patient and is not considered human research.
Supplementary Material
Footnotes
Disclosure statements are at the end of this article, following the correspondence information.
Related Digital Media are available in the full-text version of the article on www.PRSGlobalOpen.com.
REFERENCES
- 1.Clark MA, Thomas JM. Major amputation for soft-tissue sarcoma. Br J Surg. 2003;90:102–107. [DOI] [PubMed] [Google Scholar]
- 2.Puhaindran ME, Chou J, Forsberg JA, et al. Major upper-limb amputations for malignant tumors. J Hand Surg Am. 2012;37:1235–1241. [DOI] [PubMed] [Google Scholar]
- 3.Elsner U, Henrichs M, Gosheger G, et al. Forequarter amputation: a safe rescue procedure in a curative and palliative setting in high-grade malignoma of the shoulder girdle. World J Surg Oncol. 2016;14:216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Dimas V, Kargel J, Bauer J, et al. Forequarter amputation for malignant tumours of the upper extremity: Case report, techniques and indications. Can J Plast Surg. 2007;15:83–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wamalwa AO, Khainga SO. VRAM flap for an above knee amputation stump. JPRAS Open 2020;23:11–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Parsons CM, Pimiento JM, Cheong D, et al. The role of radical amputations for extremity tumors: a single institution experience and review of the literature. J Surg Oncol. 2012;105:149–155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Qadir R, Sidhu S, Romine L, et al. Interscapulothoracic (forequarter) amputation for malignant tumors involving the upper extremity: surgical technique and case series. J Shoulder Elb Surg. 2014;23:e127–e133. [DOI] [PubMed] [Google Scholar]
- 8.Machol JA, Fang RC, Matloub HS. The free fillet flap after traumatic amputation: a review of literature and case report. HAND. 2013;8:487–490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Tran NV, Evans GRD, Kroll SS, et al. Free filet extremity flap: indications and options for reconstruction. Plast Reconstr Surg. 2000;105:99–104. [DOI] [PubMed] [Google Scholar]
- 10.Contant CME, Van Geel AN, Van Der Holt B, et al. The pedicled omentoplasty and split skin graft (POSSG) for reconstruction of large chest wall defects. A validity study of 34 patients. Eur J Surg Oncol. 1996;22:532–537. [DOI] [PubMed] [Google Scholar]
- 11.Herrera HR, Ginsburg ME. The pectoralis major myocutaneous flap and omental transposition for closure of infected median sternotomy wounds. Plast Reconstr Surg. 1982;70:465–470. [DOI] [PubMed] [Google Scholar]
- 12.Mirza AA, Srinivas KG, Amarendra S, et al. Versatility of rectus abdominis myocutaneous flap in primary reconstruction of defects in surgical oncology. Indian J Surg Oncol. 2020;11:740–745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Skoracki RJ, Chang DW. Reconstruction of the chestwall and thorax. J Surg Oncol. 2006;94:455–465. [DOI] [PubMed] [Google Scholar]
- 14.Sanna S, Brandolini J, Pardolesi A, et al. Materials and techniques in chest wall reconstruction: a review. J Vis Surg. 2017;3:95–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Beahm EK, Chang DW. Chest wall reconstruction and advanced disease. Semin Plast Surg. 2004;18:117–129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kreutz-Rodrigues L, Banuelos J, Saleem HY, et al. The use of vertical rectus abdominis myocutaneous flap for pelvic reconstruction. Plast Reconstr Surg - Glob Open. 2019;7:75. [Google Scholar]
- 17.Küntscher MV, Mansouri S, Noack N, et al. Versatility of vertical rectus abdominis musculocutaneous flaps. Microsurgery. 2006;26:363–369. [DOI] [PubMed] [Google Scholar]
- 18.Yang CE, Chung SW, Lee DW, et al. Evaluation of the relationship between flap tension and tissue perfusion in implant-based breast reconstruction using laser-assisted indocyanine green angiography. Ann Surg Oncol. 2018;25:2235–2240. [DOI] [PubMed] [Google Scholar]