Abstract
Aims
Pulsed field ablation (PFA) has emerged as a promising alternative to thermal ablation for treatment of atrial fibrillation (AF). We report performance and safety using the CENTAURI™ System (Galvanize Therapeutics) with three commercial, focal ablation catheters.
Methods and results
ECLIPSE AF (NCT04523545) was a prospective, single-arm, multi-centre study evaluating safety and acute and chronic pulmonary vein isolation (PVI) durability using the CENTAURI System in conjunction with the TactiCath SE, StablePoint, and ThermoCool ST ablation catheters. Patients with paroxysmal or persistent AF were treated at two centres. Patients were analysed in five cohorts based upon ablation settings, catheter, and mapping system. Pulsed field ablation was performed in 82 patients (74% male, 42 paroxysmal AF). Pulmonary vein isolation was achieved in 100% of pulmonary veins (322/322) with first-pass isolation in 92.2% (297/322). There were four serious adverse events of interest (three vascular access complications and one lacunar stroke). Eighty patients (98%) underwent invasive remapping. Pulsed field ablation development Cohorts 1 and 2 showed a per-patient isolation rate of 38% and 26% and a per-PV isolation rate of 47% and 53%, respectively. Optimized PFA Cohorts 3–5 showed a per-patient isolation rate of 60%, 73%, and 81% and a per-PV isolation rate of 84%, 90%, and 92%, respectively.
Conclusion
ECLIPSE AF demonstrated that optimized PFA using the CENTAURI System with three commercial, contact force-sensing, solid-tip focal ablation catheters resulted in transmural lesion formation and high proportion of durable PVI with a favourable safety profile, thus providing a viable treatment option for AF that integrates with contemporary focal ablation workflows.
Keywords: Pulsed field ablation, Atrial fibrillation, Pulmonary vein isolation, Remapping, Pulsed electric fields, Focal catheter
Graphical Abstract
Graphical abstract.
What’s new?
ECLIPSE AF was the first study to evaluate the safety and chronic performance of true focal pulsed field ablation (PFA) for paroxysmal and persistent atrial fibrillation (AF) using standard, contact force-sensing, solid-tip ablation catheters for pulmonary vein isolation (PVI).
The study demonstrated 100% acute PVI using TactiCath SE, StablePoint, and ThermoCool ST catheters while meeting its primary safety endpoint.
Ninety-day invasive high-density remapping confirmed durability of focal PFA lesions and was essential for optimization of energy parameters and procedure workflows, resulting in an overall per-PV isolation rate of 89% for the optimized PFA cohorts.
ECLIPSE AF demonstrated the critical role of rigorous remapping in order to confirm and optimize treatment parameters, safety, and durability of any new ablation modality.
Introduction
Atrial fibrillation (AF) is the most common cardiac arrhythmia and is associated with an increased risk of stroke and heart failure.1 Initial treatment of AF is directed towards restoration and maintenance of sinus rhythm, usually achieved with electrical cardioversion and anti-arrhythmic drugs;2–4 however, these approaches have limited success due to complications and poor efficacy.5,6 Thermal catheter ablation has become the predominant treatment strategy for AF whereby tissue around the pulmonary veins (PVs) is targeted in order to isolate ectopic electrical activity contributing to the arrhythmia, a method known as pulmonary vein isolation (PVI).7,8 While effective at treating AF, thermal ablation is associated with complications resulting from ablative effects on non-targeted tissue.1
Pulsed field ablation (PFA) using pulsed electric fields (PEFs) has emerged as a promising non-thermal ablation alternative. Cardiac cell death is mediated by the electric field disrupting cellular homoeostasis, resulting in cell death and replacement fibrosis without affecting extracellular structures.9,10 Pre-clinical evaluation has shown PFA to target myocardial tissue while sparing critical extracardiac structures.11–16 Clinical reports suggest that PFA may be safer than thermal ablation without compromising long-term efficacy, permitting electrophysiologists to treat cardiac tissue without having to trade-off between safety and durability.17,18 However, a critical observation from previous clinical studies is the importance of PV remapping to optimize PFA dose.19–22
Although the vast majority of cardiac ablations worldwide are currently performed point by point, most emerging PFA platforms are ‘single shot’ designs.23 There have been no studies systematically exploring PFA delivered through solid-tip focal catheters in patients with AF, an approach that would provide the benefits of PFA without a learning curve or compromise to established PVI workflows. The purpose of ECLIPSE AF was to evaluate acute and chronic safety and performance of focal PFA using the CENTAURI™ System (Galvanize Therapeutics) with three commercial contact force-sensing, sold-tip ablation catheters for PVI in patients with paroxysmal and persistent AF. Chronic durability was assessed at 90 days via invasive PV remapping to optimize and validate acute PFA parameters. For the first time, we report on acute and chronic safety and durability results for a true focal PFA platform.
Methods
Trial design
The ECLIPSE AF study (NCT04523545) was a prospective, multi-centre, single-arm study performed at two centres in Europe—KBC Split (Split, Croatia) and Jessa Ziekenhuis (Hasselt, Belgium)—with approval from each centre’s Ethics Committee, corresponding national regulatory agencies, and under the principles of the Declaration of Helsinki. Patients were enrolled from September 2020 to February 2022. An independent Medical Safety Monitor reviewed all adverse events (AEs) for assessment of the primary safety endpoint.
Study population
This study enrolled patients with symptomatic paroxysmal or persistent AF24 who were resistant to Class I–IV anti-arrhythmic medication, had a left ventricular ejection fraction (LVEF) > 35%, and a left atrial (LA) anteroposterior (AP) diameter ≤ 50 mm. Patients with implanted electronic cardiovascular devices and New York Heart Association (NYHA) class III or IV heart failure were excluded from the study.
CENTAURI™ System
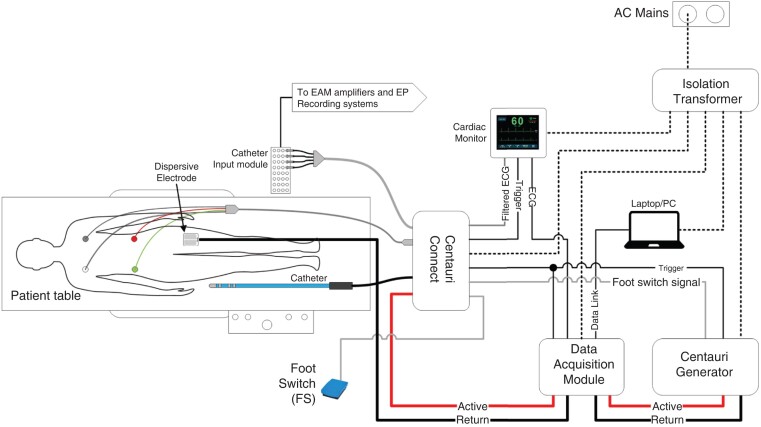
The CENTAURI System has three components: the CENTAURI Generator that delivers biphasic, monopolar PEF energy at three selectable energy settings (19, 22, and 25 A) through the tip electrode of the ablation catheter; the CENTAURI Connect device that permits connectivity of compatible focal ablation catheters and their mapping systems; and a Cardiac Monitor (Ivy Biomedical Systems) that synchronizes PFA delivery to the R-wave. The CENTAURI System was designed to integrate with standard electrophysiology (EP) lab equipment, permitting electrophysiologists to perform cardiac ablation using established workflows (Figure 1). For this study, the CENTAURI System was used sequentially with three commercial ablation catheters and their associated mapping systems: TactiCath™ SE and EnSite™ Precision (‘EnSite’); INTELLANAV STABLEPOINT™ (‘StablePoint’) and RHYTHMIA HDx™ (‘RHYTHMIA’); THERMOCOOL SMARTTOUCH™ (‘ThermoCool ST’) and CARTO®3 (‘CARTO’).
Figure 1.
Configuration of the CENTAURI System in a standard EP laboratory. ECG, electrocardiogram; EP, electrophysiology.
Study endpoints
The primary safety endpoint was the incidence of predefined system-related and procedure-related serious adverse events (SAEs) of interest (see Supplementary material online, Table S1) within 30-day post-ablation using the CENTAURI System.
The primary clinical performance endpoints were (i) acute success, defined as PVI achieved with the CENTAURI System during the index procedure assessed by entrance and exit block after a 20-min waiting period and (ii) chronic success, defined as the per-patient and per-PV isolation rate at 90 (±15) days, assessed by high-density remapping and confirmation of entrance and exit block. Thus, the primary purpose of ECLIPSE AF was to evaluate the CENTAURI System’s ability to safely and effectively create focal PVI lesion sets that remained durable at 90 days.
Cranial MRI sub-study
Patients who participated in the cranial MRI sub-study for the assessment of silent cerebral events/lesions (SCEs/SCLs) underwent a cranial MRI (DWI + FLAIR) within 7 days prior to the index procedure and again within 72-h post-procedure. Scan sequences and incidental findings were managed according to the participating centre’s standard procedures and patients with new abnormal findings returned for a 30-day follow-up cranial MRI.
Study procedure
Informed consent was obtained for each patient prior to study-related assessments. Patients underwent baseline cardiac computed tomography (CT) for the assessment of PV anatomy. The presence of intracardiac thrombus was assessed by cardiac CT or transoesophageal echocardiogram (TEE) within 24 h prior to the procedure or by intracardiac echocardiography (ICE) at the beginning of the procedure. Standard PVI, with uninterrupted oral anti-coagulation, was performed under intravenous deep sedation (fentanyl, midazolam, and propofol) or general anaesthesia (GA). Standard tools were used for access and diagnostics, and intravenous heparin was administered prior to LA access.
Similar to other PFA studies,20,22 the progressive nature of technology development resulted in sequential evaluation of the CENTAURI System paired with either the TactiCath SE/EnSite/Advisor™ HD-Grid, StablePoint/RHYTHMIA/ORION™, or ThermoCool ST/CARTO/PENTARAY™ commercial systems. Baseline electroanatomical maps were obtained prior to PVI. The ablation catheter’s irrigation rate was set to 4 mL/min. Standard wide antral circumferential ablation (WACA) was performed with a target contact force of ≥5 g while also utilizing principles of the CLOSE protocol ensuring sufficient overlap of each lesion to achieve adequate depth at, and between, each PEF application.25–28 Acute success was assessed via high-density mapping of the PVs confirming entrance and exit block following a 20-min waiting period.
Procedural safety assessments included evaluation of microbubbles on ICE and ST-segment changes on electrocardiogram (ECG) during PFA. Post-procedure safety assessments included phrenic nerve function (assessed by observing diaphragmatic motion under fluoroscopy, phrenic nerve capture, or a phrenic nerve capture threshold test pre- and post-procedure), National Institute of Health Stroke Score (NIHSS), oesophageal endoscopy, and cranial MRI (if participating in the sub-study).
Study follow-up
Study follow-up was scheduled at 7-day, 30-day, 90-day, 6-month, and 12-month post-index procedure. At 90 days, a cardiac CT was performed to reassess PV anatomy, along with invasive high-density remapping of treated PVs to reconfirm entrance and exit block. If electrically reconnected, the focal points of PV reconnection were retreated with the CENTAURI System.
Statistical analyses
This study was sized to provide >90% probability of detecting one or more serious device-related AEs, assuming such events have ∼5% risk of occurrence. Study safety and performance endpoints were analysed on the per-treatment evaluable population, which consisted of all patients who received complete PVI treatment with the CENTAURI System, who had no major protocol deviations, and for whom follow-up data was available. Descriptive statistics and graphical summaries were used to present study results. For categorial variables, counts, percentages, and where appropriate, exact 95% confidence intervals for the mean, assuming a normal distribution, were calculated.
Results
Enrolment
A total of 84 patients enrolled in the study, and 82 patients were included in the per-treatment evaluable population. One patient withdrew consent prior to the index procedure, and one patient was not treated with the CENTAURI System at the index procedure.
Baseline patient characteristics and medical history
Baseline patient characteristics are shown in Table 1. The study population was 61.0 ± 9.05 years old, mostly male (74.4%, 61/82), and included paroxysmal (51.2%, 42/82) and persistent (48.8%, 40/82) AF patients. NYHA classification ranged from Class I (57.3%, 47/82) to Class II (42.7%, 35/82) with a preserved LVEF of 60.4 ± 9.30% and LA AP diameter of 43.6 ± 4.65 mm. Most patients were at a low risk for stroke based on the baseline CHADS (median 1.0), CHA2DS2-VASc (median 2.0), and baseline NIHSS (0.1 ± 0.45).
Table 1.
Baseline demographics and clinical characteristics
| AF classification | |
|---|---|
| Paroxysmal | 42 (51.2%) |
| Persistent | 40 (48.8%) |
| Age (years) | |
| Mean (SD) | 61.0 (9.05) |
| Sex | |
| Male | 61 (74.4%) |
| Female | 21 (25.6%) |
| Body mass index (kg/m2) | |
| Mean (SD) | 29.12 (4.24) |
| NYHA classification | |
| Class I | 47 (57.0%) |
| Class II | 35 (42.7%) |
| LVEF (%) | |
| Mean (SD) | 60.4 (9.30) |
| CHA2DS2-VASC score | |
| Mean (SD) | 1.8 (1.4) |
| LA (AP) diameter (mm) | |
| Mean (SD) | 43.6 (4.3) |
Data are N (%) unless otherwise specified.
AF, atrial fibrillation; AP, anteroposterior; LA, left atrial; LVEF, left ventricular ejection fraction; SD, standard deviation.
Baseline medical history demonstrated a standard AF population undergoing first time PVI with a percentage of patients exhibiting hypertension (59.8%, 49/82), hypercholesterolaemia (19.5%, 16/82), dyslipidaemia (24.4%, 20/82), Type 1 or 2 diabetes (13.4%, 11/82), and sleep apnoea (3.7%, 3/82) (Table 2). Seven patients (8.5%, 7/82) had a history of stroke or transient ischaemic attack (TIA).
Table 2.
Baseline medical history
| Cardiovascular | |
|---|---|
| Vascular disease (including peripheral vascular disease) | 3 (3.7%) |
| Ischaemic cardiomyopathy | 0 (0.0%) |
| Non-ischaemic cardiomyopathy | 3 (3.7%) |
| Dilated cardiomyopathy | 3 (100.0%) |
| Valvular disease | 5 (6.1%) |
| Congestive heart failure | 6 (7.3%) |
| Myocardial infarction | 3 (3.7%) |
| Coronary artery disease | 5 (6.1%) |
| Percutaneous coronary intervention | 4 (4.9%) |
| Coronary bypass surgery | 1 (1.2%) |
| Stroke or Transient ischaemic attack | 7 (8.5%) |
| Thromboembolic event | 2 (2.4%) |
| Hypertension | 49 (59.8%) |
| Pulmonary hypertension | 1 (1.2%) |
| Respiratory | |
| Smoking history (n) | 21 |
| Never smoked | 6 (28.6%) |
| Current smoker | 2 (9.5%) |
| Former smoker | 13 (61.9%) |
| Sleep apnoea | 3 (3.7%) |
| Gastrointestinal | |
| (controlled/uncontrolled GERD, ulcers, strictures, oesophagitis, or structural abnormalities) | 11 (13.4%) |
| Endocrine | |
| Diabetes (Types 1 and 2) | 11 (13.4%) |
| Dyslipidaemia | 20 (24.4%) |
| Hypercholesterolaemia | 16 (19.5%) |
| Thyroid disease | 7 (8.5%) |
Procedure characteristics
The majority of patients were treated under deep sedation (58.5%, 48/82), while the remaining patients were treated under GA (41.5%, 34/82). One investigational centre used oesophageal temperature probes (CIRCA S-Cath) in only the first two patients (2.4%, 2/82) during their index procedures. Intracardiac echo was used for 74.4% (61/82) of index procedures.
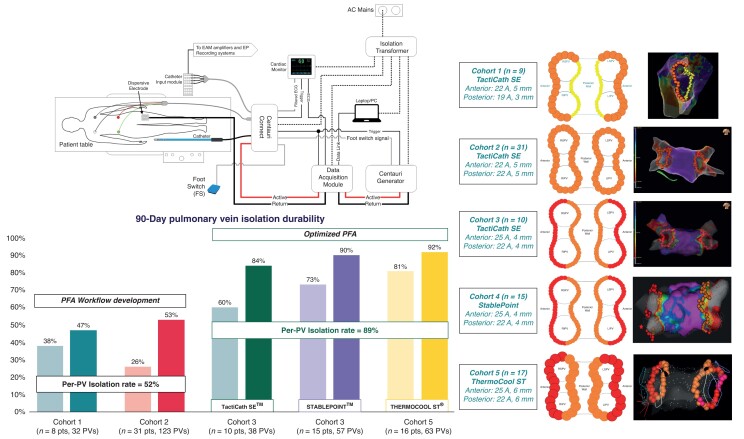
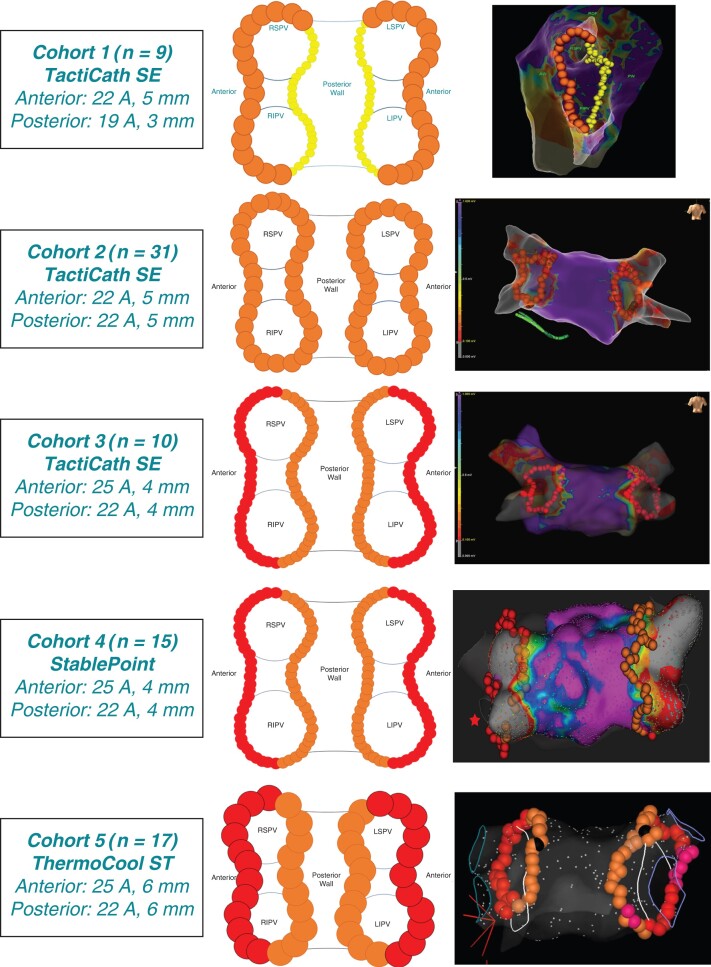
The clinical study was designed to integrate the CENTAURI System beyond the initial ablation catheter and mapping system (TactiCath SE/EnSite) once PFA dosing and workflows were optimized. Therefore, treated patients were separated into five cohorts based on ablation settings, ablation catheter, and mapping system used at the index procedure (Table 3 and Figure 2). Energy settings were selected based upon anticipated lesion characteristics determined from pre-clinical models.29 Patients in Cohort 1 (n = 9) were treated with TactiCath SE/EnSite using 19 A (delivered over 4 cardiac cycles) along the posterior region of the PVs with 3 mm diameter lesion tags, and 22 A (delivered over 7 cardiac cycles) along the anterior region of the PVs using 5 mm diameter tags. Cohort 2 (n = 31) was treated with TactiCath SE/EnSite using 22 A along the entire WACA with 5 mm diameter tags. After patients in Cohorts 1 and 2 returned for 90-day remapping, PFA was optimized to utilize 25 A along the anterior region of the PVs for remaining procedures. Therefore, Cohorts 1 and 2 were defined as the PFA workflow development cohorts and subsequently treated patients in Cohorts 3–5 were considered the optimized PFA cohorts.
Table 3.
Patient cohorts in the ECLIPSE AF study
| Cohort | Ablation settings (energy setting, energy delivery duration, and lesion tag diameter) | Cardiac ablation catheter | Mapping system | |
|---|---|---|---|---|
| PFA workflow development | 1 | Anterior: 22 A, 2.4 ms, 5 mm Posterior: 19 A, 1.4 ms, 3 mm |
TactiCath SE | EnSite |
| 2 | Anterior: 22 A, 2.4 ms, 5 mm Posterior: 22 A, 2.4 ms, 5 mm |
TactiCath SE | EnSite | |
| Optimized PFA | 3 | Anterior: 25 A, 3.4 ms, 4 mm Posterior: 22 A, 2.4 ms, 4 mm |
TactiCath SE | EnSite |
| 4 | Anterior: 25 A, 3.4 ms, 4 mm Posterior: 22 A, 2.4 ms, 4 mm |
StablePoint | RHYTHMIA | |
| 5 | Anterior: 25 A, 3.4 ms, 6 mm Posterior: 22 A, 2.4 ms, 6 mm |
ThermoCool ST | CARTO | |
Figure 2.
Cohorts were established based upon ablation settings, ablation catheter, and mapping system used during the index ablation procedure.
Cohort 3 (n = 10) was treated with TactiCath SE/EnSite using 25 A (delivered over 10 cardiac cycles) anteriorly and 22 A posteriorly, all with 4 mm diameter tags. Patients in Cohort 4 were treated with StablePoint/RHYTHMIA using 25 A anteriorly and 22 A posteriorly, all with 4 mm diameter tags. Patients in Cohort 5 were treated with ThermoCool ST/CARTO using 25 A anteriorly and 22 A posteriorly, all with 6 mm diameter tags.
Primary safety endpoint
There were four AEs in four patients (4.9%, 4/82) that met the primary endpoint definition of an SAE of interest (Table 4). All were considered to be related to the PVI procedure. Three were haemorrhagic events due to vascular access complications requiring hospitalization. One patient experienced a non-embolic cerebrovascular accident (CVA) due to exacerbated cardiac tamponade secondary to ablation catheter perforation. There were no incidences of AE fistula, diaphragmatic paralysis, myocardial infarction, pericarditis, thromboembolism, PV stenosis, TIA, or death.
Table 4.
Study complications
| Serious adverse events of interest | |
|---|---|
| Atrio-oesophageal fistula | 0 (0.0%) |
| CVA/stroke | 1 (1.2%) |
| Death (including cardiovascular death) | 0 (0.0%) |
| Diaphragmatic paralysis | 0 (0.0%) |
| Haemorrhagic event | 3 (3.7%) |
| Myocardial infarction | 0 (0.0%) |
| Pericarditis | 0 (0.0%) |
| Peripheral or organ thromboembolism | 0 (0.0%) |
| Pulmonary vein stenosis | 0 (0.0%) |
| TIA | 0 (0.0%) |
| Other procedural complications | |
| Catheter perforation, cardiac effusion, or tamponade | 2 (2.4%) |
| Oesophageal injury | 2 (2.7%) |
| ST-elevation during PVI | 0 (0%) |
CVA, cerebrovascular accident; PVI, pulmonary vein isolation; TIA, transient ischaemic attack.
Additional safety assessments
Catheter perforation and cardiac effusion/tamponade
There were two instances of catheter perforation in two patients. In one patient, a combination of map shift and PFA-induced cough possibly led to perforation along a region of thin tissue near an accessory vein of the right PV roof. Intracardiac echo identified a moderate pericardial effusion that was successfully treated via pericardiocentesis. The patient was monitored overnight and discharged after TTE confirmed resolution of the effusion. In the second patient, high ablation catheter contact force likely led to perforation, but identification of the ensuing pericardial effusion and cardiac tamponade was masked due to simultaneous haemodynamic instability caused by frequent runs of AF/AFL and a wide-complex tachycardia. The cardiac effusion was identified on TEE, and a perforation was identified along the base of the LA appendage during subsequent cardiac surgery. The perforation and effusion were resolved with complete haemodynamic recovery; however, due to the duration of tamponade and prolonged hypotension secondary to tamponade, the event resulted in a non-embolic CVA.
Microbubbles and ST elevation
During the index procedures in which ICE was used, investigators visualized the ablation catheter tip to capture the presence/absence of microbubbles during PFA due to the potential of gaseous emboli.30 Investigators also monitored ST-segment changes before, during, and after each PEF application. Microbubbles were not observed at any time during the 61 ICE-monitored procedures, and there were no incidences of ST elevation during any PEF applications for PVI.
Cranial MRI sub-study
Thirty-six patients (5 in Cohort 2, 7 in Cohort 3, 13 in Cohort 4, and 11 in Cohort 5) participated in the cranial MRI sub-study for detection of SCEs/SCLs. At baseline, all participating patients had DWI and FLAIR sequences performed (100%, 36/36), and no patient had an abnormal finding. Post-procedure cranial MRIs were performed for all but one patient due to claustrophobia. Results revealed SCEs and SCLs in 11.4% and 0% of patients, respectively, as four patients had a positive finding detected by DWI only (11.4%, 4/35) and not by FLAIR (0%, 0/35). All four patients were a part of Cohort 4, which utilized StablePoint/RHYTHMIA/ORION. There was no incidence of neurological dysfunction, increase in NIHSS, or detected microbubbles in these four patients. The 30-day follow-up cranial MRI confirmed resolution of all abnormal findings.
Oesophageal monitoring
Patients were assessed for oesophageal injury via oesophageal endoscopy within 72-h post-procedure. Out of the 75 patients who underwent this assessment, two patients had a finding that met the definition of a Category 1 (mild) oesophageal lesion.31 These were the only two patients who had an oesophageal temperature probe placed during the index procedure. Both probes had been reprocessed and reused an unknown number of times despite being labelled for single use. There was no detected rise in temperature during, or immediately after, any PEF application, and there was no loss of function of the probes or temperature monitor throughout the procedure. Upon examination, insulation defects were discovered surrounding 11 of 24 thermocouples across the two probes (5/12 and 6/12; 46%), resulting in exposed metal, rough insulation surfaces, and sharp edges along the length of each probe. One of the patients was diagnosed with chronic uncontrolled GERD, which may have contributed to oesophageal finding. Both events were resolved without sequelae upon follow-up endoscopy.
Phrenic nerve monitoring
The integrity of the phrenic nerve was evaluated by observing normal diaphragmatic excursion on the right hemi-diaphragm under fluoroscopy or by verifying functionality via pace capture. Phrenic nerve monitoring was performed on 80 patients at the index procedure using fluoroscopy (58.8%, 47/80), phrenic nerve pacing (23.8%, 19/80), or phrenic nerve capture threshold assessment (17.5%, 14/80). In all cases, there was no phrenic nerve injury. Notably, the phrenic nerve capture threshold assessment showed that the pre-ablation threshold (3.49 ± 0.93 mA) was unchanged following PFA (2.60 ± 1.09 mA), further suggesting that PFA did not affect the phrenic nerve.
Pulmonary vein imaging
Cardiac CT was performed on 80 patients at baseline and the 90-day visit to assess PV anatomy after PFA. The per cent narrowing of PVs was calculated by comparing the measured PV diameters from baseline to 90 days. The mean per cent narrowing was 2.29% for the left superior PV (LSPV), 0.91% for left inferior PV (LIPV), 3.55% for the right superior PV (RSPV), 3.01% for the right inferior PV (RIPV), and 0.48% for all other PV types indicating no chronic effect to PV diameters after PFA.
Clinical performance endpoints
Acute pulmonary vein isolation
Acute procedural success, confirmed by both entrance and exit block after a 20-min waiting period, was achieved in all 82 (100%) treated patients and 322 (100%) treated PVs, with first-pass isolation in 92.2% (297/322) of PVs.
Chronic pulmonary vein isolation at 90-day remap
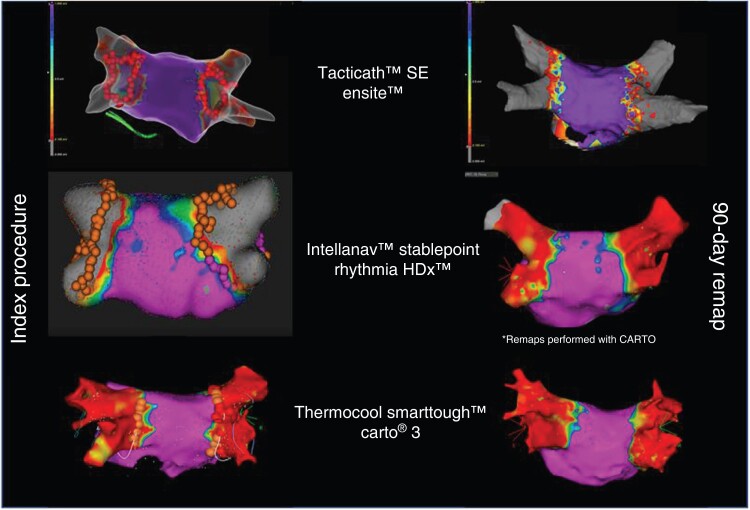
Chronic PVI durability was assessed at 90 days by calculating the proportion of patients and PVs with documented PVI evaluated by high-density mapping to verify entrance and exit block (Figure 3).
Figure 3.
Acute PVI and 90-day chronic PVI durability were assessed using high-density remapping to verify entrance and/or exit block. Voltage maps correlated with placement of PEF applications, and 90-day remaps confirmed no degradation of the line of block created at the index procedure. PEF, pulsed electric field; PVI, pulmonary vein isolation.
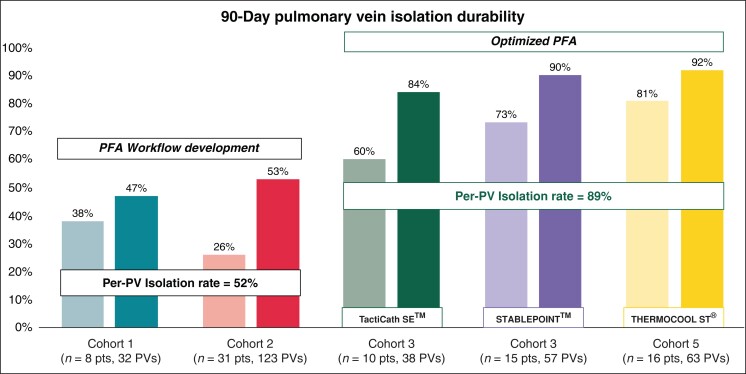
Eighty of the 82 treated patients (98%) returned for 90-day remapping at 97.6 ± 25.0 days. Each treated PV was circumnavigated with a high-density mapping catheter (Advisor HD-Grid or PENTARAY) to confirm lesion durability along the entire circumference. Patients in the PFA workflow development Cohorts 1 and 2 showed a per-patient isolation rate of 38% and 26%, and a per-PV isolation rate of 47% and 53%, respectively. Patients in the optimized PFA Cohorts 3–5 showed a per-patient isolation rate of 60%, 73%, and 81% and a per-PV isolation rate of 84%, 90%, and 92%, respectively (Figure 4). Reconnected PVs were retreated at the identified point of reconnection using the CENTAURI System connected to a compatible focal ablation catheter (Figure 5).
Figure 4.
Chronic PFA lesion durability, measured by per-patient and per-PV isolation rate at 90 days, for each cohort. Patients in the PFA workflow development Cohorts 1 and 2 showed a per-patient isolation rate of 38% and 26%, and a per-pulmonary vein isolation rate of 47% and 53%, respectively. Patients in the optimized PFA Cohorts 3–5 showed a per-patient isolation rate of 60%, 73%, and 81% and a per-PV isolation rate of 84%, 90%, and 92%, respectively. There were no AEs associated with the optimization in energy settings and clinical workflows. PFA, pulsed field ablation; PV, pulmonary vein.
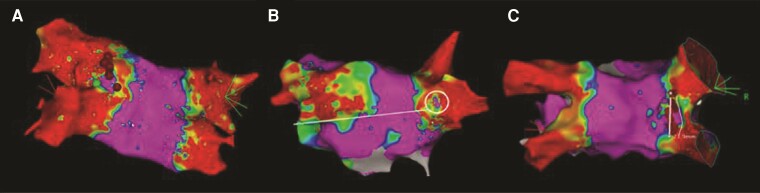
Figure 5.
Points of reconnection along the PVs were addressed by delivering focal touch-up PFA using the CENTAURI System. As depicted in these examples, PV reconnections typically required minimal focal touch-up applications (a) posterior left PV carina indicated by red ablation tags, (b) epicardial sleeve posterior right carina, and (c) posterior right carina indicated by dotted ablation tags to re-isolate the vein, emphasizing the overall chronic durability of the WACAs created at the index procedure. PV, pulmonary vein; WACA, wide antral circumferential ablation.
Additional study objectives
Procedure times
The overall procedure, LA dwell, PEF PVI, and fluoroscopy times for all patients were 140.0 ± 42.44, 117.0 ± 33.14, 61.8 ± 23.03, and 10.3 ± 4.31 min, respectively, and included the 20-min waiting period to verify acute PVI. For the optimized workflow in Cohort 5, the overall procedure, LA dwell, and PEF PVI times were reduced to 137.1 ± 40.42, 112.4 ± 21.55, and 58.5 ± 16.70 min, respectively.
Pulmonary vein isolation applications
It took 101.8 ± 31.14 focal PFA lesions to achieve PVI for all patients. This was dependent on the LA size, PEF energy setting, and the lesion tag diameter utilized for each cohort. With the optimized workflow in Cohort 5, the number of PEF applications to achieve PVI was reduced to 83.5 ± 19.32.
Discussion
Clinical performance of focal pulsed field ablation
The ECLIPSE AF study aimed to evaluate the safety and performance of focal PFA using the CENTAURI System with three commercial contact force-sensing, solid-tip ablation catheters for standard PVI in patients with paroxysmal or persistent AF. In 82 treated patients, acute success was achieved in 100% of targeted PVs with 92.2% first-pass isolation. Acute success with PFA is common but has been shown to be a poor indicator of chronic efficacy.19,20 A critical element of ECLIPSE AF was the iterative use of 90-day remapping to optimize PFA dosing and workflows and to confirm that the intended treatment plan at the index procedure was accomplished and durable. The study demonstrated an overall 89% per-PV chronic durability rate within the Optimized PFA Cohorts, with comparable results across all three tested ablation catheters and mapping systems.
The cornerstone of an effective ablation is confidence that the intended ablation plan at index results in chronic durability. This study, similar to other PFA assessments, highlighted the necessity for remapping to understand the nature of lesion durability for any new PFA system.20–22 Initial results from Cohorts 1 and 2 were inconsistent, and learnings from invasive remapping validated optimization of PFA workflows for the subsequent cohorts. This stepwise approach ensured safe application prior to increasing energy settings, resulting in substantial improvements to per-PV durability within the optimized PFA cohorts (89%) compared with the PFA workflow development cohorts (52%) without any effect on safety. Performing a per-PV analysis provided the largest sample size to adequately assess the effectiveness of each focal application. Analysis of the limited points of PV reconnection in the optimized cohorts yielded no consistent finding with respect to acute procedural factors; however, the spatial pattern of PV gaps was analysed and demonstrated most reconnections along regions of relatively thicker tissues (e.g. superior anterior ridge of the left PVs and carina region of left and right PVs) (see Supplementary material online, Figure S2), which are common areas for PV reconnection.32,33
The CENTAURI System was able to integrate with established EP lab tools, mapping systems, and ablation workflows. While there were workflow learnings, such as operators anticipating temporary deactivation of catheter visualization during PFA, they were likely minimal compared with alternative ablation platforms that require integration of new catheters, sheaths, and mapping tools.34,35 Although GA provided the most stable procedural environment, there was no difference in acute performance, safety, or chronic durability between procedures performed under GA and deep sedation. Study procedure times did include additional time to evaluate PFA workflows, system troubleshooting, and a few cases with additional lesion sets (e.g. CTI ablation and posterior wall isolation). Since the CENTAURI System is used with standard RF ablation catheters, an appropriate comparator of procedure times is standard or high-power short-duration RF ablation, which have reported procedure times of 111.15 ± 27.9 and 89.5 ± 23.9 min, respectively.36 Procedure and ablation times were also driven by the number of focal PEF applications required to achieve PVI, which initially was dependent on LA size, energy setting, and associated lesion tag diameter size. Ultimately, the final optimized workflow consolidated lesion tag diameters to 6 mm for all energy settings making the number of lesions to achieve PVI dependent on LA anatomy, which tends to be larger in persistent AF patients (∼50% of this study population).
Clinical safety of focal PFA
Given the novelty of PFA, it is important to understand the basis behind any safety event that may be attributed to the use of PEF energy. The SAEs of interest (4.9%) were a combination of haemorrhagic events due to vascular access complications and a non-embolic CVA that was a result of an exacerbated cardiac tamponade caused by catheter perforation; all considered related to the standard PVI procedure only. Additionally, the lack of AEs associated with the inclusion of additional ablation catheters and the optimization of PEF energy settings highlights the safety of PFA with the CENTAURI System.
In this study, there were two incidences of detected superficial oesophageal ulcers in the only two patients that utilized repeatedly reprocessed oesophageal temperature probes (CIRCA S-Cath). Initial speculation is that PFA caused a thermal response in the oesophagus; however, there are counter arguments to that theory: (i) pre-clinical assessments have demonstrated that the limited effect PEF energy has on oesophageal tissue,14,37,38 (ii) during the procedures, there was absolutely no evidence of temperature rise detected by the probes to indicate any type of thermal effect in the oesophagus, (iii) there was no loss of function or aberrant behaviour of the thermal probe/monitoring system during PFA, and (iv) post-procedure examination of the specific temperature probes utilized in these two patients demonstrated thermocouples with degraded insulation, sharp edges of insulating material, and exposed metal electrodes. As with RF ablation, the possibility of PFA energy interacting with exposed metal electrodes on the reprocessed temperature probes cannot be ruled out.39 Importantly, all remaining patients who received post-procedure esophagogastroduodenoscopy (n = 73) showed no signs of oesophageal injury after PFA.
There were two incidences of catheter perforation leading to pericardial effusion that required pericardiocentesis. Perforation is an anticipated risk with focal catheter ablation.40,41 The temporary deactivation of catheter visualization during PEF energy delivery and the incidence of PFA-induced cough (commonly seen in patients under deep sedation) are important to anticipate when considering catheter and mapping stability for PFA procedures.17,20,42 Improved catheter visualization during ablation will require further integration with mapping systems. While the CENTAURI PFA waveform has been optimized to mitigate muscle stimulation, the nature of PFA-induced cough is still under evaluation. Early hypotheses suggest that this phenomenon may be attributed to stimulation of vagal afferent receptors within pulmonary or bronchiole tissue.43,44 General anaesthesia or deeper levels of intravenous sedation currently are the best methods to limit this cough phenomenon.
The risk of TIA or stroke after AF catheter ablation is ∼0.5–1%.45 MRI studies after ablation have reported a 7–40% prevalence of silent cerebral infarction, and these studies continue to grow a body of evidence suggesting an association between AF and dementia.45–47 A European registry of the FARAPULSE™ Pulsed Field Ablation System (Boston Scientific), a commercially available PFA system in Europe, was associated with a 0.39% risk of stroke and 0.11% risk of TIA.48 One of the proposed mechanisms linking AF to cognitive decline includes silent cerebral ischaemia due to subclinical microemboli, and silent cerebral microemboli caused by microbubbles observed from ablation catheters.20,49,50 In order to understand the embolic fingerprint of ablation using the CENTAURI System, the incidence of PFA-induced microbubbles and ST-elevation was evaluated in this study. Assessing more than 6000 PEF applications delivered in this study, at no time were microbubbles observed on ICE or ST-elevation observed on ECG. To investigate the incidence of acute SCE/SCL post-ablation, cranial MRI scans were performed for a subset of patients. Four patients (11.1%) had detected SCEs post-ablation by DWI only (11.1%, 4/35), but there were no detected SCLs by FLAIR (0.0%, 0/35). There were no microbubbles observed and no neurological changes measured by the post-procedure NIHSS in these four patients. All four patients were in Cohort 4, which utilized RHYTHMIA with the ORION™ high-density mapping catheter for baseline and post-ablation mapping, a catheter that is associated with a higher rate of embolic events, as compared with other standard high-density mapping catheters, due to its design and method of insertion into the introducer sheath.51 No other patients that participated in this sub-study experienced any positive cranial MRI finding. Although a limited sample size, these cranial MRI results are favourable to published results using established alternative ablation methods52,53 and comparable with data observed in IMPLULSE, PEFCAT, and PEFCAT II where brain MRIs performed on 18 patients showed 11.1% (2/18) DW-positivity indicative of an acute events post-procedure,17 as well as in MANIFEST-PF where 114 patients underwent brain MRIs with 17.5% (20/114) showing asymptomatic abnormalities on MRI post-procedure.48 When using the Sphere-9 lattice-tip catheter (Medtronic), post-procedure brain MRIs performed in 51 patients revealed 9.8% (5/51) SCEs (DWI-positive/FLAIR-negative) and 5.9% (3/51) SCLs (DWI-positive/FLAIR-positive).54
There were no reported incidents of phrenic injury as assessed by diaphragmatic excursion on fluoroscopy, phrenic nerve pace capture, or phrenic nerve capture threshold compared at pre- and post-procedure. Additionally, there was no evidence of PV stenosis assessed by PV diameter measurements on cardiac CT at baseline and at 90 days. These results further support the favourable safety profile of PFA and present this focal PFA system as another notable step forward for the treatment of AF.
Study limitations
The ECLIPSE AF study was a non-randomized study without a direct comparison with a thermal ablation control arm, thus limiting the ability to draw concrete conclusions about relative safety and efficacy. Clinical performance of CENTAURI was evaluated sequentially due to serial development of compatibility with the three ablation systems; therefore, we cannot rule out impact of a learning curve on results across optimized cohorts. Due to similarity of catheter specifications, sample size was limited within the optimized cohorts. Additionally, operator experience with each mapping system should be taken into consideration. Early recurrence of atrial arrhythmia within the 90-day blanking period was not captured but will be an important analysis for future research. The focus of this study and publication was on follow-up through 90 days, including invasive 90-day remapping of the PVs as chronic PVI durability data are the true measure of acute procedural success and are a better predictor of long-term ablation success. However, 1-year freedom from atrial arrhythmia continues to be the benchmark for which AF ablation strategies are judged and will be the subject of a future publication. It must be emphasized that comparison across PFA platforms is not straightforward for various reasons including (i) PFA waveforms employed by each manufacturer are proprietary, (ii) electrode configuration has an established effect on the resulting PFA profile,9 and (iii) most PFA platforms have not assessed lesion durability or optimized ablation parameters using clinical remapping studies.
Conclusion
In the ECLIPSE AF study, focal PFA using the CENTAURI System with three commercial contact force-sensing, solid-tip ablation catheters demonstrated 100% acute PVI, with four SAEs on interest attributed to the standard PVI procedure only, and an overall per-PV 90-day chronic durability rate of 89% for the optimized patient cohorts. The overall goal of this study was to validate safe and effective treatment using focal PFA, with emphasis placed on thorough assessment of safety, as well as chronic durability of PFA lesions. The cornerstone of effective AF ablation is confidence that the intended ablation plan has been faithfully executed and will be chronically durable. Based upon the results of the optimized cohorts, we have established high confidence that the treatment plan executed in this study results in safe and durable PVI through 90 days. It is our hope that by validating an optimized ablation workflow, we have provided a solid scientific foundation for future studies to prospectively test new ablation strategies with greater scrutiny on safety and durability of the intended treatment plan.
Supplementary Material
Acknowledgements
We would like to thank Dr Quim Castellvi, Dr Robert Neal, Tim Gundert, and Armaan Vachani for their guidance in developing and optimizing PFA with the CENTAURI System. We would like to thank Dr John C. Evans, Dr Prof. Tim R. Betts, and Dr William S. Krimsky for their contributions to medical safety monitoring and interpretation of safety data. We would like to thank Charlotte Baecke, Julie Bollen, Dr Melda Fabrio, Paul Schelfhout, Dr Lori Rusch, Sophy Yohannan, Wendy Winters, Russell Dawson, and Serina Loi for their commitment to the success of the clinical study. We would like to thank Ante Borovina, Igor Visković, Sara Milanović, Mirna Žaja, Nela Lemo, Stijn Christiaens, Kim Vandeweyer, Krista Hagfors, and David Gareau for their clinical and technical expertise during support of study procedures.
Funding
This study was sponsored and funded by Galvanize Therapeutics (previously Galaxy Medical).
Data availability
The data that support the findings of this study are available from the corresponding author, A.A., upon reasonable request.
Contributor Information
Ante Anić, Heart and vascular center, Klinički Bolnički Centar (KBC) Split, Spinčićeva ul. 1, Split 21000, Croatia.
Thomas Phlips, Jessa Ziekenhuis, Hasselt, Belgium.
Toni Brešković, Heart and vascular center, Klinički Bolnički Centar (KBC) Split, Spinčićeva ul. 1, Split 21000, Croatia.
Pieter Koopman, Jessa Ziekenhuis, Hasselt, Belgium.
Steven Girouard, Galvanize Therapeutics, Inc. (previously Galaxy Medical), Redwood City, CA, USA.
Vikramaditya Mediratta, Galvanize Therapeutics, Inc. (previously Galaxy Medical), Redwood City, CA, USA.
Zrinka Jurišić, Heart and vascular center, Klinički Bolnički Centar (KBC) Split, Spinčićeva ul. 1, Split 21000, Croatia.
Ivan Sikirić, Heart and vascular center, Klinički Bolnički Centar (KBC) Split, Spinčićeva ul. 1, Split 21000, Croatia.
Lucija Lisica, Heart and vascular center, Klinički Bolnički Centar (KBC) Split, Spinčićeva ul. 1, Split 21000, Croatia.
Johan Vijgen, Jessa Ziekenhuis, Hasselt, Belgium.
Supplementary material
Supplementary material is available at Europace online.
References
- 1. Calkins H, Hindricks G, Cappato R, Kim Y-H, Saad EB, Aguinaga Let al. 2017 HRS/EHRA/ECAS/APHRS/SOLAECE expert consensus statement on catheter and surgical ablation of atrial fibrillation. Europace 2018;20(1):e1–e160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Waldo AL. Management of atrial fibrillation: the need for AFFIRMative action. Atrial fibrillation follow-up investigation of rhythm management. Am J Cardiol 1999;84:698–700. [DOI] [PubMed] [Google Scholar]
- 3. Jones DG, Haldar SK, Hussain W, Sharma R, Francis DP, Rahman-Haley SLet al. A randomized trial to assess catheter ablation versus rate control in the management of persistent atrial fibrillation in heart failure. J Am Coll Cardiol 2013;61:1894–903. [DOI] [PubMed] [Google Scholar]
- 4. Falk RH. Atrial fibrillation. N Engl J Med 2001;344:1067–78. [DOI] [PubMed] [Google Scholar]
- 5. Calkins H, Reynolds MR, Spector P, Sondhi M, Xu Y, Martin Aet al. Treatment of atrial fibrillation with antiarrhythmic drugs or radiofrequency ablation: two systematic literature reviews and meta-analyses. Circ Arrhythm Electrophysiol 2009;2:349–61. [DOI] [PubMed] [Google Scholar]
- 6. Morillo CA, Verma A, Connolly SJ, Kuck KH, Nair GM, Champagne Jet al. Radiofrequency ablation vs antiarrhythmic drugs as first-line treatment of paroxysmal atrial fibrillation (RAAFT-2): A randomized trial. JAMA 2014;311:692–700. [DOI] [PubMed] [Google Scholar]
- 7. Haïssaguerre M, Jaïs P, Shah DC, Takahashi A, Hocini M, Quiniou Get al. Spontaneous initiation of atrial fibrillation by ectopic beats originating in the pulmonary veins. N Engl J Med 1998;339:659–66. [DOI] [PubMed] [Google Scholar]
- 8. Haïssaguerre M, Jaïs P, Shah DC, Garrigue S, Takahashi A, Lavergne Tet al. Electrophysiological end point for catheter ablation of atrial fibrillation initiated from multiple pulmonary venous foci. Circulation 2000;101:1409–17. [DOI] [PubMed] [Google Scholar]
- 9. Verma A, Asivatham SJ, Deneke T, Castellvi Q, Neal RE. Primer on pulsed electrical field ablation. Circ Arrhythm Electrophysiol 2021;14:e010086. [DOI] [PubMed] [Google Scholar]
- 10. Sugrue A, Maor E, Del-Carpio Munoz F, Killu AM, Asirvatham SJ. Cardiac ablation with pulsed electric fields: principles and biophysics. Europace 2022;24:1213–22. [DOI] [PubMed] [Google Scholar]
- 11. Davalos RV, Mir LM, Rubinsky B. Tissue ablation with irreversible electroporation. Ann Biomed Eng 2005;33:223–31. [DOI] [PubMed] [Google Scholar]
- 12. Wittkampf FH, Van Driel VJ, Van Wessel H, Vink A, Hof IE, Gründeman PFet al. Feasibility of electroporation for the creation of pulmonary vein ostial lesions. J Cardiovasc Electrophysiol 2011;22:302–9. [DOI] [PubMed] [Google Scholar]
- 13. van Driel VJHM, Neven K, van Wessel H, Vink A, Doevendans PAFM, Wittkampf FHM. Low vulnerability of the right phrenic nerve to electroporation ablation. Heart Rhythm 2015;12:1838–44. [DOI] [PubMed] [Google Scholar]
- 14. Neven K, van Es R, van Driel V, van Wessel H, Fidder H, Vink Aet al. Acute and long-term effects of full-power electroporation ablation directly on the porcine esophagus. Circ Arrhythm Electrophysiol 2017;10. [DOI] [PubMed] [Google Scholar]
- 15. Koruth J, Kuroki K, Iwasawa J, Enomoto Y, Viswanathan R, Brose Ret al. Preclinical evaluation of pulsed field ablation: electrophysiological and histological assessment of thoracic vein isolation. Circ Arrhythmia and Electrophysiology 2019;12:12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Moshkovits Y, Grynberg D, Heller E, Maizels L, Maor E. Differential effect of high-frequency electroporation on myocardium vs. non-myocardial tissues. Europace 2023;25:748–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Reddy VY, Dukkipati SR, Neuzil P, Anic A, Petru J, Funasako Met al. Pulsed field ablation of paroxysmal atrial fibrillation. JACC Clin Electrophysiol 2021;7:614–27. [DOI] [PubMed] [Google Scholar]
- 18. Verma A, Haines DE, Boersma LV, Sood N, Natale A, Marchlinski FEet al. Pulsed field ablation for the treatment of atrial fibrillation: PULSED AF pivotal trial. Circulation 2023;147:1422–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Ramirez FD, Reddy VY, Viswanathan R, Hocini M, Jaïs P. Emerging technologies for pulmonary vein isolation. Circ Res 2020;127:170–83. [DOI] [PubMed] [Google Scholar]
- 20. Reddy VY, Neuzil P, Koruth JS, Petru J, Funosako M, Cochet Het al. Pulsed field ablation for pulmonary vein isolation in atrial fibrillation. J Am Coll Cardiol 2019;74:315–26. [DOI] [PubMed] [Google Scholar]
- 21. Turagam MK, Neuzil P, Petru J, Funasako M, Koruth JS, Reinders Det al. PV isolation using a spherical array PFA catheter. JACC: Clinical Electrophysiology 2023;9:638–48. [DOI] [PubMed] [Google Scholar]
- 22. Reddy VY, Peichl P, Anter E, Rackauskas G, Petru J, Funasako Met al. A focal ablation catheter toggling between radiofrequency and pulsed field energy to treat atrial fibrillation. JACC Clin Electrophysiol 2023:S2405-500X(23)00219-0. [DOI] [PubMed] [Google Scholar]
- 23. Boersma L. New energy sources and technologies for atrial fibrillation catheter ablation. Europace 2022;24(Supplement_2):ii44–51. [DOI] [PubMed] [Google Scholar]
- 24. Hindricks G, Potpara T, Dagres N, Arbelo E, Bax JJ, Blomström-Lundqvist Cet al. 2020 ESC guidelines for the diagnosis and management of atrial fibrillation developed in collaboration with the European Association of Cardio-Thoracic Surgery (EACTS). Eur Heart J 2020;42:373–498. [DOI] [PubMed] [Google Scholar]
- 25. Nakagawa H, Castellvi Q, Neal R, Girouard S, Ikeda A, Kuroda Set al. B-PO03-131 effects of contact force on lesion size during pulsed field ablation. Heart Rhythm 2021;18:S242–3. [Google Scholar]
- 26. Taghji P, El Haddad M, Phlips T, Wolf M, Knecht S, Vandekerckhove Yet al. (CLOSE protocol) evaluation of a strategy aiming to enclose the pulmonary veins with contiguous and optimized radiofrequency lesions in paroxysmal atrial fibrillation. JACC: Clinical Electrophysiology 2018;4:99–108. [DOI] [PubMed] [Google Scholar]
- 27. Nakagawa H, Castellvi Q, Neal RE, Girouard S, An Y, Hussein AAet al. PO-710-06 assessment of lesion durability produced by pulsed electric field ablation: evaluation in a swine atrial linear lesion model. Heart Rhythm 2022;19:S475. [Google Scholar]
- 28. Nakagawa H, Castellvi Q, Neal RE, Girouard S, An Y, Hussein AAet al. PO-710-03 temporal and spatial evolution of reversible and irreversible ventricular lesion boundaries produced by pulsed electric field ablation. Heart Rhythm 2022;19:S473–4. [Google Scholar]
- 29. Verma A, Neal R, Evans J, Castellvi Q, Vachani A, Deneke Tet al. Characteristics of pulsed electric field cardiac ablation porcine treatment zones with a focal catheter. Cardiovasc Electrophysiol 2023;34:99–107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Es R, Groen MHA, Stehouwer M, Doevendans PA, Wittkampf FHM, Neven K. In vitro analysis of the origin and characteristics of gaseous microemboli during catheter electroporation ablation. J Cardiovasc Electrophysiol 2019;30:2071–9. [DOI] [PubMed] [Google Scholar]
- 31. Halbfass P, Berkovitz A, Pavlov B, Sonne K, Nentwich K, Ene Eet al. Incidence of acute thermal esophageal injury after atrial fibrillation ablation guided by prespecified ablation index. J Cardiovasc Electrophysiol 2019;30:2256–61. [DOI] [PubMed] [Google Scholar]
- 32. Tohoku S, Chun KRJ, Bordignon S, Chen S, Schaack D, Urbanek Let al. Findings from repeat ablation using high-density mapping after pulmonary vein isolation with pulsed field ablation. Europace 2023;25:433–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Bohnen M, Weber R, Minners J, Jadidi A, Eichenlaub M, Neumann FJet al. Characterization of circumferential antral pulmonary vein isolation areas resulting from pulsed-field catheter ablation. Europace 2023;25:65–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Schmidt B, Bordignon S, Tohoku S, Chen S, Bologna F, Urbanek Let al. 5S Study: safe and simple single shot pulmonary vein isolation with pulsed field ablation using sedation. Circ Arrhythm Electrophysiol 2022;15:.e010817. [DOI] [PubMed] [Google Scholar]
- 35. Koruth J, Kawamura I, Dukkipati SR, Neuzil P, Reddy VY. Preclinical assessment of the feasibility, safety and lesion durability of a novel ‘single-shot’ pulsed field ablation catheter for pulmonary vein isolation. Europace 2023;25:1369–78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Kottmaier M, Popa M, Bourier F, Reents T, Cifuentes J, Semmler Vet al. Safety and outcome of very high-power short-duration ablation using 70 W for pulmonary vein isolation in patients with paroxysmal atrial fibrillation. Europace 2020;22:388–93. [DOI] [PubMed] [Google Scholar]
- 37. Koruth JS, Kuroki K, Kawamura I, Brose R, Viswanathan R, Buck EDet al. Pulsed field ablation vs radiofrequency ablation: esophageal injury in a novel Porcine model. Circ Arrhythm Electrophysiol 2020;13:e008303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Evans JC, Evans Neal R, Vachani A, Girouard S, Castellvi Q. B-PO01-053 the susceptibility and recovery of esophageal tissue layers to pulsed electric fields. Heart Rhythm 2021;18:S72. [Google Scholar]
- 39. Müller P, Dietrich JW, Halbfass P, Abouarab A, Fochler F, Szöllösi Aet al. Higher incidence of esophageal lesions after ablation of atrial fibrillation related to the use of esophageal temperature probes. Heart Rhythm 2015;12:1464–9. [DOI] [PubMed] [Google Scholar]
- 40. Cappato R, Calkins H, Chen SA, Davies W, Iesaka Y, Kalman Jet al. Prevalence and causes of fatal outcome in catheter ablation of atrial fibrillation. J Am Coll Cardiol 2009;53:1798–803. [DOI] [PubMed] [Google Scholar]
- 41. Mujović N, Marinković M, Marković N, Marković N, Kocijančić A, Kovačević Vet al. Management and outcome of periprocedural cardiac perforation and tamponade with radiofrequency catheter ablation of cardiac arrhythmias: a single medium-volume center experience. Adv Ther 2016;33:1782–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Reddy VY, Anic A, Koruth J, Petru J, Funasako M, Minami Ket al. Pulsed field ablation in patients with persistent atrial fibrillation. J Am Coll Cardiol 2020;76:1068–80. [DOI] [PubMed] [Google Scholar]
- 43. Mazzone SB. An overview of the sensory receptors regulating cough. Cough 2005;1:2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Canning BJ, Mori N, Mazzone SB. Vagal afferent nerves regulating the cough reflex. Resp Physiol Neurobiol 2006;152:223–42. [DOI] [PubMed] [Google Scholar]
- 45. Medi C, Evered L, Silbert B, Teh A, Halloran K, Morton Jet al. Subtle post-procedural cognitive dysfunction after atrial fibrillation ablation. J Am Coll Cardiol 2013;62:531–9. [DOI] [PubMed] [Google Scholar]
- 46. Deneke T, Jais P, Scaglione M, Schmitt R, Di Biase L, Christopoulos Get al. Silent cerebral events/lesions related to atrial fibrillation ablation: a clinical review. J Cardiovasc Electrophysiol 2015;26:455–63. [DOI] [PubMed] [Google Scholar]
- 47. Gaita F, Leclercq JF, Schumacher B, Scaglione M, Toso E, Halimi Fet al. Incidence of silent cerebral thromboembolic lesions after atrial fibrillation ablation may change according to technology used: comparison of irrigated radiofrequency, multipolar nonirrigated catheter and cryoballoon. J Cardiovasc Electrophysiol 2011;22:961–8. [DOI] [PubMed] [Google Scholar]
- 48. Ekanem E, Reddy VY, Schmidt B, Reichlin T, Neven K, Metzner Aet al. Multi-national survey on the methods, efficacy, and safety on the post-approval clinical use of pulsed field ablation (MANIFEST-PF). Europace 2022;24:1256–66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Gerstenfeld EP. Catheter ablation of atrial fibrillation using electroporation. JACC Clin Electrophysiol 2018;4:996–8. [DOI] [PubMed] [Google Scholar]
- 50. Rivard L, Khairy P. Mechanisms, clinical significance, and prevention of cognitive impairment in patients with atrial fibrillation. Can J Cardiol 2017;33:1556–64. [DOI] [PubMed] [Google Scholar]
- 51. Nakamura K, Sasaki T, Take Y, Minami K, Inoue M, Kishi Set al. Impact of the type of electroanatomic mapping system on the incidence of cerebral embolism after radiofrequency catheter ablation of left atrial tachycardias. Heart Rhythm 2020;17:250–7. [DOI] [PubMed] [Google Scholar]
- 52. Deneke T, Nentwich K, Krug J, Müller P, Grewe PH, Mügge A. Silent cerebral events after atrial fibrillation ablation—overview and current data. J Atr Fibrillation 2014;6:6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Miyazaki S, Kajiyama T, Yamao K, Hada M, Yamaguchi M, Nakamura Het al. Silent cerebral events/lesions after second-generation cryoballoon ablation: how can we reduce the risk of silent strokes? Heart Rhythm 2019;16:41–8. [DOI] [PubMed] [Google Scholar]
- 54. Anter E, Neužil P, Rackauskas G, Peichl P, Aidietis A, Kautzner Jet al. A lattice-tip temperature-controlled radiofrequency ablation catheter for wide thermal lesions. JACC Clin Electrophysiol 2020;6:507–19. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data that support the findings of this study are available from the corresponding author, A.A., upon reasonable request.