Abstract
Objective
Clusterin is involved in a variety of physiological processes, including proteostasis. Several clusterin polymorphisms were associated with an increased risk of developing Alzheimer’s disease, the world-leading cause of dementia. Herein, the effect of a clusterin polymorphism, aging and dementia in the levels of clusterin in human plasma were analysed in a primary care-based cohort, and the association of this chaperone with fibrillar structures discussed.
Methods
64 individuals with dementia (CDR≥1) and 64 age- and sex-matched Controls from a Portuguese cohort were genotyped for CLU rs1136000 polymorphism, and the plasma levels of clusterin and fibrils were assessed.
Results
An increased prevalence of the CC genotype was observed for the dementia group, although no significant robustness was achieved. CLU rs11136000 SNP did not significantly change plasma clusterin levels in demented individuals. Instead, clusterin levels decreased with aging and even more in individuals with dementia. Importantly, plasma clusterin levels correlated with the presence of fibrillar structures in Control individuals, but not in those with dementia.
Conclusion
This study reveals a significant decrease in plasma clusterin in demented individuals with aging, which related to altered clusterin-fibrils dynamics. Potentially, plasma clusterin and its association with fibrillar structures can be used to monitor dementia progression along aging.
Keywords: Clusterin, Aging, Fibrils, Dementia, Alzheimer’s disease
Introduction
Population aging continues to grow being that around 10% of the world’s population is aged 65 or over, a number expected to exceed 16% by 2050 [1], [2]. As the population ages several socio-economical and health care challenges arise, including the fact that aging is the major risk factor for developing dementia [3], [4].
Dementia encompasses a set of pathological changes, among them loss of memory, language and problem-solving, all are aspects associated with cognitive decline and interfere with daily life activities. Neurodegenerative disorders, like Alzheimer’s disease (AD), Parkinson disease, Lewy bodies or frontotemporal lobar degeneration are among the main causes of dementia in the older population [5]. In fact, late-onset AD (LOAD) is the worldwide leading cause of dementia, being this disease associated with 60 to 80% of all cases [6]. LOAD, accounts for 95% of AD cases, is considered to be essentially sporadic in nature, although the contribution of risk genes such as APOE, the major risk factor identified for LOAD, can be considerable [7], [8].
Genome-wide association studies (GWAS) compared millions of single nucleotide polymorphisms (SNP) and identified several additional genetic risk determinants for LOAD [9], [10], [11]. Among these is the clusterin (CLU) gene, also known as Apolipoprotein J [12], [13]. Secreted clusterin is the most abundant form and can be found in a variety of body fluids, including plasma and cerebrospinal fluid (CSF) [13], [14], [15]. In the extracellular space, clusterin serves as a heat shock protein-like chaperone that aids in the folding of secreted proteins [16], [17] and is able to form stable complexes with misfolded proteins and peptides to prevent their aggregation or facilitate disposal [18]. Clusterin exhibits a multifunctional nature and is involved in a growing number of pathologies, placing it as a biomarker candidate and promising therapeutic target for different conditions [19]. Of note, clusterin can bind a wide variety of targets, including the amyloid-β peptide (Aβ, the main core of senile plaques in AD) [17], [20] or α-synuclein oligomers (the cytotoxic species in several α-synucleinopathies) [21], revealing an important role of this chaperone in extracellular proteostasis and neuroprotection.
Accumulation of misfolded proteins is a common hallmark in many neurodegenerative diseases and clusterin was found to co-localize with different disease-associated extracellular deposits of highly structured protein aggregates, including amyloid plaques [22]. The role of clusterin in AD etiology depends on the interaction with Aβ and tau aggregates. However, its exact contribution to the dynamics of amyloid aggregation and clearance is still ambiguous, with both beneficial and detrimental roles assigned to clusterin [23], [24], [25], [26]. Clusterin upregulation was observed in the hippocampus and cortex regions of AD brains [27], [28], and elevated clusterin levels can be detected in CSF of affected individuals [29], [30]. At a more peripheral level, some groups reported elevated clusterin levels in plasma of AD individuals [31], [32]. These levels were associated with disease severity and progression [33], but also with the transition from mild cognitive impairment (MCI) to AD, and with the risk of dementia amongst the elderly [34], [35], [36], although inconsistencies have been observed. Several CLU SNP were associated with an altered AD-risk, with the variant rs11136000 being considered the most relevant one, but exhibiting some population-specific differences [37], [38], [39]. The first studies addressing CLU rs11136000 SNP, reported that the C allele was associated with an increased risk of developing AD [40], while the T allele was considered protective [41], possibly by genetic variant rs11136000 regulation of CLU expression [42]. Individuals carrying a CC genotype have been associated with increased cognitive decline when compared to TT counterparts [43]. Additionally, genetic risk variants of rs11136000 have been associated with higher Aβ deposition [44], and with structural brain changes in cognitively normal individuals, occurring years before the appearance of AD symptoms [45], [46]. However, it was also reported that the C allele reduces the AD risk [47].
The rs11136000 SNP was also correlated with clusterin protein levels in plasma. Likewise, conflicting data was obtained with distinct authors reporting: (1) an association between lower levels of plasma clusterin and the C risk allele in cognitively healthy individuals [48]; (2) no effect of the rs11136000 SNP in plasma clusterin [33]; (3) or decreased levels in TT homozygotes of the Control group, and increased levels in MCI and AD individuals compared to Controls [31]. Other authors also reported increased plasma clusterin in type-II diabetes mellitus patients with MCI, and that CLU rs11136000 TT genotype is associated with a reduced MCI-risk [49]. Hence, this study addresses the influence of the CLU rs11136000 gene variant in the incidence of dementia in a subpopulation from a primary care-based cohort (pcb-cohort) previously established and characterized by our group [50]. The contribution of this SNP, aging and dementia to the levels of clusterin in plasma samples is likewise assessed. Finally, this study also evaluated the association between clusterin and the presence of fibrillar proteins and peptides in human plasma.
Material and methods
Characterization of the study population
Human blood and plasma samples from the primary health care-based cohort (pcb-cohort), established in the Aveiro region, Portugal [50] were used for this study. The pcb-cohort comprises a total of 568 individuals who fulfilled the inclusion criteria: ≥50 years old, no record of ongoing oncological treatments, absence of psychiatric disorders (excluding depression) or aphasia, and ability to answer the questions during the interview. These individuals were submitted to a battery of cognitive evaluations and dementia screening tests during the interview, regardless of the clinical diagnosis [51]. Herein, 64 samples from individuals screened for cognitive impairment, as determined by Clinical Dementia Rating (CDR) scores ≥ 1, and 64 age- and sex-matched Controls, randomly selected amongst cognitively normal individuals (CDR=0), were analyzed [52]. According to the CDR scale, CDR=0 indicates no dementia, CDR=0.5 suspected, questionable, or very mild dementia, while a CDR≥1 mild (CDR=1), moderate (CDR=2) or severe dementia (CDR=3) [51]. The 64 CDR≥1 individuals were designated as ‘dementia’ group and include 9 clinically diagnosed Alzheimer’s disease (AD) patients, while the 64 cognitively normal individuals were designated as ‘Control’ group. Characterization of the selected sub-group of individuals is presented in Table 1.
Table 1.
Characterization of the study group according to age, gender and clinical dementia ratio.
|
Group N |
Control 64 |
CDR ≥ 1 64 |
||
|---|---|---|---|---|
|
Gender N (%) |
Female 42 (65.6%) |
Male 22 (34.4%) |
Female 42 (65.6%) |
Male 22 (34.4%) |
| Age (years, mean ± s.d.) | 74.9 ± 7.2 | 75.7 ± 7.8 | ||
Data are presented as N (number of individuals).
Sample collection
Blood samples were collected in EDTA-tubes, according to standard procedures, and plasma was obtained by centrifugation of blood samples in EDTA gel separator tubes. Blood and plasma were promptly stored at −80 °C, as previously described [50]. Sample collection was performed in compliance with all guidelines, and approved by the Ethics Committee for Health of the Central Regional Administration of Coimbra (CES of ARS Centro, protocol N°012804-04.04.2012) and by the National Committee for Data Protection (CNPD N°369/2012). All participants gave written informed consent.
CLU genotyping
A 685 bp fragment from CLU rs1113600 polymorphic region was amplified using Phusion Blood Direct PCR Master Mix (ThermoFisher Scientific, USA), according to the manufacturer’s instructions. Briefly, 1 μL of blood was mixed with 2x Phusion Blood II DNA Polymerase Master Mix, 1 μL of each primer (Eurogentec, Belgium; Fw: 5́-CCTGGCTTAAAGAATCCACTCATC-3′, Rv: 5′-CAGGGGATTCCTTTGAGATAGAGT-3′, final concentration of 0.1 μM each) and 7 μL of ultrapure DNase distilled H2O. The PCR reaction included a denaturation step at 98 °C for 5 min, followed by 35 cycles of amplification at 94 °C for 1 min, 63 °C for 30sec and 72 °C for 46 sec, and a final extension step at 72 °C for 5 min. The mixture was centrifuged at 1000 g for 3 min and the supernatant transferred into new tubes. Amplification of PCR products was verified by agarose gel electrophoresis.
For CLU rs1113600 SNP analysis, resulting DNA product was precipitated using 1/10 vol of sodium acetate (pH 3 M, pH 5.2) and 2.5 volumes of 100% ethanol. Samples were then centrifuged at 14000 rpm for 20 min, 4 °C, and the pellet washed with 70% ethanol. Following an additional centrifugation, the resulting pellets were dried out and then resuspended in ultrapure DNase distilled H2O. 10 μL of purified PCR products were analyzed by Sanger Sequencing in an expert external laboratory (Stab Vida, Portugal), using the Fw primer 5′-CCTGGCTTAAAGAATCCACTCATC-3′ as described in [53]. Results were analyzed with the BioEdit software (Ibis Biosciences, Carlsbad USA).
Western and slot blot analysis
Clusterin levels in human plasma samples were determined by western blot analysis using an anti-human clusterin antibody (ref. 552886; BD Biosciences). For each sample, 30 µg were prepared in loading buffer and separated by SDS-PAGE. Proteins were blotted into nitrocellulose membranes and incubated with the anti-clusterin primary antibody, overnight at 4 °C, followed by an anti-mouse IgG, HRP-linked, secondary antibody (ref. 7076 s; Cell Signalling), 1 h at R.T. Clusterin was detected with ECL Select (GE Healthcare) in a Chemidoc Touch Imaging System (BioRad Laboratories, Inc.) and the resulting images were analyzed using the Image Lab software (BioRad Laboratories, Inc.). Density values were expressed as arbitrary units. A pool of plasma was used as reference sample in each blot, and across blots, to normalize and compare the obtained results.
The aggregation state of proteins and peptides present in plasma samples from Control and CDR ≥ 1 individuals, was evaluated by slot blot analysis using an anti-amyloid fibrils OC antibody (Merck KGaA), as previously described [54]. This conformation-specific and sequence-independent antibody recognizes amyloid fibrils and fibrillar structures but not prefibrillar oligomers or natively folded proteins [55]. Briefly, 10 μg of total plasma protein were resuspended in TBS and transferred to nitrocellulose membranes (GE Healthcare) using a Bio-Dot SF Microfiltration Apparatus (Bio-Rad Laboratories, Inc.). A pool of plasma was loaded in each membrane and used as a reference sample to normalize across results. Ponceau S staining (Sigma-Aldrich) was used as a loading Control. Membranes were incubated with the anti-amyloid fibrils OC antibody, overnight at 4 °C, followed by anti-rabbit IgG, HRP-conjugated, secondary antibody (7074 s, Cell Signaling). Band detection and image acquisition and analysis were performed as described above.
Statistical analysis
Statistical analysis was performed using GraphPad Prism (GraphPad Software). Data sets with a normal distribution were analyzed by two-tailed Student’s t-test and Two-way ANOVA test, and partial correlations analyzed by Pearson r coefficient. For data sets that do not follow a normal distribution the non-parametric two-tailed Mann−Whitney U test was used, and partial correlations were assessed by Spearman rank correlation. Normality of each data set was tested using the Shapiro-Wilk normality test. Chi-squared test was performed to compare non-continuous variables. Odds ratios (OR) with correspondent 95% confidence intervals (CI) were used to analyze the association of risk alleles or genotypes with dementia. Significance level was set at 0.05 and p-values≤0.05 were considered statistically significant.
Results
Genotypic and allelic characterization of the clusterin rs1113600 polymorphism in the pcb-cohort
Genotypic and allelic characterization of the CLU rs1113600 SNP was carried out in a group of individuals from the pcb-cohort [50]. The study group included 64 individuals with mild to severe dementia, according to CDR scores and collectively designated as the dementia or CDR≥1, and 64 sex- and aged-matched Controls selected amongst the cognitively normal individuals (CDR=0). Age and gender distribution of the Control and CDR≥1 groups are shown in Table 1. Overall, both groups have the same gender distribution, with an average age of 74.9 ± 7.2 and 75.7 ± 7.8 for Control and CDR≥1, respectively.
Sequencing analysis revealed that from the 64 individuals with mild to severe dementia, a total of 34 were homozygous risk-carriers (CC genotype), 19 were heterozygous risk carriers (CT genotype), and 11 were homozygous non-risk carriers (TT genotype). Comparatively, the Control group comprises 26 homozygous risk-carriers (CC genotype), 25 heterozygous risk-carriers (CT genotype) and 13 homozygous non-risk carriers (TT genotype). Despite the frequency of the CC genotype in the CDR≥1 group (34 out of 64) was higher than in Controls (26 out of 64) (Table 2), no statistical differences were observed in the distribution of CLU rs1113600 genotypes between both groups. Further, increased odds ratios were observed for the CC genotype when compared to TT genotype (OR=1.545, p=0.469), and for the recessive model CC vs TT + CT (OR=1.656, p=0.215), although none has reached statistical significance (Table 2).
Table 2.
Distribution of CLU rs11136000 genotype frequencies between the Control and dementia groups.
| Genotype | Control N |
CDR≥1 N |
Total | OR (95% CI) |
p-value |
|---|---|---|---|---|---|
| CC | 26 | 34 | 60 | 1.545 (0.601 – 4.180) |
0.469 |
| CT | 25 | 19 | 44 | 0.898 (0.313 – 2.291) |
0.999 |
| TT | 13 | 11 | 24 | 1.000 | – |
| CC | 26 | 34 | 60 | 1.656 (0.823 – 3.336) |
0.215 |
| TT + CT | 38 | 30 | 68 | 1.000 | – |
Data are presented as N (number of individuals). Genotype frequencies were compared between both groups with Fisher’s exact test. OR: odds ratio; CI: confidence interval.
Clusterin plasma levels in Control and demented individuals
Clusterin levels in the plasma of CDR≥1 individuals and the corresponding age- and sex-matched Controls were assessed by western blot. No significant differences (p=0.3515) were observed in the plasma levels of clusterin between both groups (Fig. 1a), although a slightly lower tendency could be detected in the CDR≥1 group. The impact of CLU rs11136000 SNP in clusterin plasma levels was also evaluated in the pcb-cohort, by addressing the clusterin levels in both groups according to genotype (Fig. 1b). Data suggests that the presence of the ‘C’ allele, a potential risk factor in AD, either in homozygous ‘CC’ or heterozygous ‘CT’ individuals is not associated to fluctuations in the plasma levels of this protein. The average levels of plasma clusterin are identical independently of the genotype (p=0.9059). Altogether, the presented data indicates that CLU rs11136000 SNP genotype does not affect the circulating levels of human plasma clusterin.
Fig. 1.
Levels of Clusterin in plasma of Control and CDR≥1 individuals. The levels of clusterin were evaluated in plasma of Control (○) and CDR≥1 individuals (●) as whole groups (a), and according to the CLU rs1113600 SNP genotype (b). A t-test was applied to compare the Control and CDR≥1 groups (p = 0.3515), while a Two-Way ANOVA with multiple comparisons was used to test the effect of the genotypes in the two groups (p=0.7199). All samples were normalized to the reference sample and clusterin levels are represented as fold changes. Bars represent mean ± s.e.m.
Plasma clusterin levels in aging and dementia
To address if aging can affect plasma clusterin levels, the latter was monitored and analyzed in an age-dependent manner, for individuals in both study groups. Data presented revealed a week negative correlation (Pearson’s r=−0.1802, p=0.1543) between clusterin plasma levels and aging in Control individuals (Control group, Fig. 2). A stronger decline in plasma clusterin levels was observed in samples from individuals with mild to severe dementia. Analysis of the CDR≥1 group revealed a significant negative correlation between clusterin levels and age (Pearson’s r=−0.4377, p=0.0003), which might be related to dementia progression (Fig. 2).
Fig. 2.
Plasma levels of Clusterin in Control and CDR≥1 individuals, with aging. Levels of plasma clusterin were assessed by immunoblotting and plotted according to the age of each individual. Pearson’s coefficient (Pearson r) was applied to test the correlation of both groups, (a) Controls (○) and (b) CDR≥1 (●), with aging. In Controls, (ns) stands for non-significant and p=0.1543; in CDR≥ 1, p=0.0003 (***). All samples were normalized to the reference sample and clusterin levels are represented as fold changes. Lines represent a linear regression fit with corresponding 95% confidence bands (dashed lines).
Clusterin correlation with fibrillar structures in human plasma
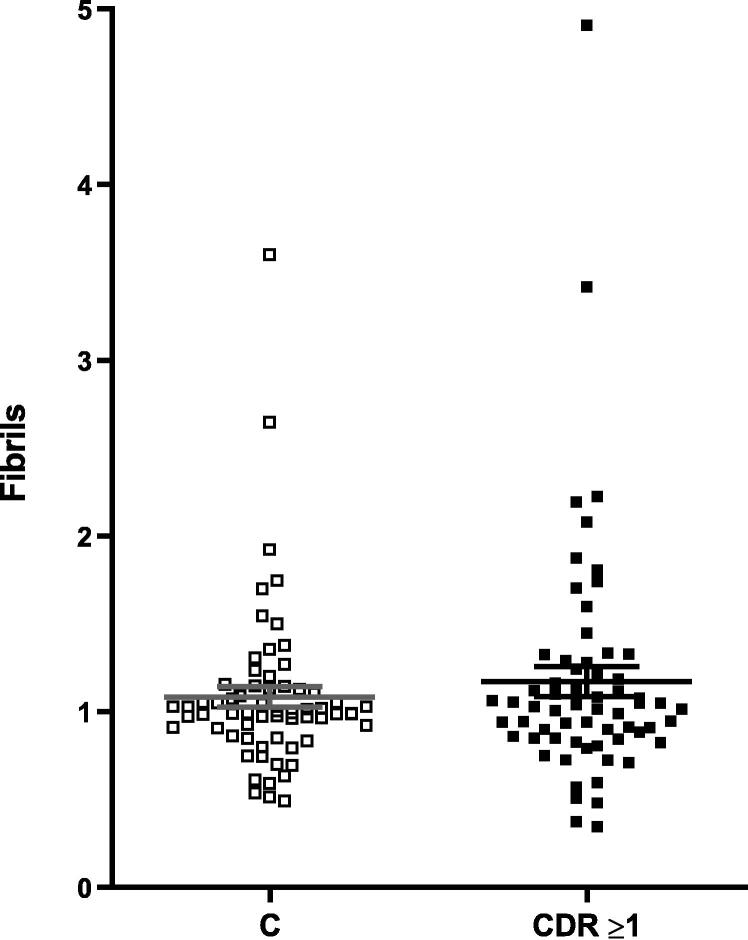
Clusterin assists in the folding of secreted proteins and functions as an extracellular chaperone to prevent the aggregation of non-native proteins, including in blood. A previous study by our group evaluated plasma protein aggregation in non-demented individuals (CDR=0) with aging [54]. A similar approach was here employed to explore how dementia or clusterin change the aggregation status of plasma proteins. Using a conformation-specific and sequence-independent antibody directed against fibrillar structures (OC), the levels of amyloid fibrils and fibrillar oligomers were evaluated in the plasma of both groups. In general, no significant differences were observed for these aggregated species after immunodetection with the OC antibody (p = 0.7190), despite the fact that even though the average level of fibrillar structures in CDR≥1 samples is slightly higher than in Controls (Fig. 3).
Fig. 3.
Levels of Fibrillar structures in Control and CDR≥1 individuals. The levels of fibrils were evaluated in plasma of Control (□) and CDR≥1 individuals (■) using a two-tailed Mann-Whitney test (p=0.7190). All samples were normalized to the reference sample and fibrils levels are represented as fold changes. Bars represent mean ± s.e.m.
Since clusterin is able to prevent protein aggregation and is involved in the clearance of extracellular misfolded proteins, correlation analyses between the plasma levels of clusterin and fibrils were carried out. In the Control group, a significant moderate negative correlation was observed (Spearman r=−0.4017, p=0.0010), indicating that a higher abundance of clusterin is associated with fewer fibrillar species (Fig. 4). Of note, this correlation was lost in the CDR≥1 study group (Spearman r=−0.1250, p=0.3252), and was no longer significant (Fig. 4).
Fig. 4.
Correlation between clusterin and fibrillar structures. The levels of clusterin and fibrils were independently evaluated in plasma of (a) Control (◊) and (b) CDR≥1 individuals (♦), and matched values plotted. Correlations were evaluated by Spearman coefficients (r), with (**) p=0.0010 for Controls and (ns) p=0.3252 for CDR≥1. All samples were normalized to the reference sample and fibrils vs clusterin levels are represented as fold changes. Lines represent a linear regression fit with corresponding 95% confidence bands (dashed lines).
Discussion
The relationship between clusterin polymorphisms and dementia is controversial, with authors either reporting an association with increased or reduced risk of dementia. To our knowledge, this is the first study addressing the association between CLU rs1113600 SNP and cognitive impairment in a Portuguese sub-population. Cognitive impairment may represent a transition stage to AD, since annualy 10–15% of MCI cases can progress to AD [56], or other dementia-associated pathologies.
Genotyping of the pcb-cohort sub-groups did not allow to determine the exact contribution of the ‘C’ allele to the development of dementia in this Portuguese population. Although it was observed a higher prevalence of CC risk homozygotes in the dementia group; and higher OR analysis for CC genotype when comparing to the TT genotype, no significant robusteness was achieved to support the association between CC genotype and dementia in this study. An increased risk for dementia was originally attributed to the C allele [40], which flagged CLU rs11136000 SNP in GWAS for risk polymorphisms associated to AD [38], [39]. Consistently, other authors also reported an association of the TT genotype with decreased risk of MCI [49], while the C risk allele may be associated with faster memory decline in the transition between MCI and AD [43]. Nevertheless, opposite data has been recently reported, suggesting that the C allele in CLU rs11136000 SNP could actually play a role in reducing the risk of AD [47]. It should be noted, however, that the association of this SNP with AD exhibits population-specific variability, being less consistent in Asian cohorts [57], than it is for European cohorts [37]. Classifying a SNP as protective or risk factor is not simple, and indeed, different observations could be associated with the presence of other SNPs at distinct loci.
No correlations were found between the rs11136000 genotypes and the plasma clusterin levels of the Control and dementia groups, in agreement with observations from other groups [33], [49]. Nonetheless, at this level, contradictory data has also been reported. While some studies showed an increase in plasma clusterin in TT homozygotes of Control individuals [48] others revealed an opposite effect [31]. None of these works detected genotype-associated differences in plasma clusterin of AD individuals.
Further, while some authors reported increased plasma clusterin levels in AD and even MCI individuals, compared to cognitively normal Controls [35], [58], others also found identical levels of this protein in the plasma of AD and Control individuals. In agreement with the later, the results reported herein reveal no differences in plasma clusterin levels between Controls and the dementia group. A recent meta-analysis analyzing 7228 individuals, including 1936 AD cases, also found no association between plasma clusterin levels and the diagnosis, risk, or severity of AD [59].
Since aging is the major risk-factor in several dementia-associated pathologies, it was also analyzed its impact in plasma clusterin. Results demonstrate a negative association between plasma clusterin and the age of individuals, as reported by others [36]. The data herein presented provides evidence for an age- and dementia-associated decrease in plasma clusterin levels, which was more pronounced in dementia group than in cognitively normal individuals. The differences observed between the Control and dementia groups support the hypothesis of clusterin as an important player in the human body’s strategy to cope with pathological processes underlying the onset and progression of dementia.
The protective role of clusterin in dementia and other proteinopathies seems to depend on its ability to bind misfolded proteins and prevent aggregation, thus maintaining extracellular proteostasis. Previous work by our group demonstrated that the plasma proteome undergoes conformational changes with aging, in cognitively normal individuals. Particularly, an age-related increase in fibrillar structures was detected [54]. Herein, no significant differences were found in the plasma levels of fibrillar proteins and peptides between Control and individuals with dementia. One cannot exclude that during disease progression the clearance rate of protein aggregates in the brain may change, but it is difficult to monitor such differences. Consistent with this observation, Chen and colleagues reported that plasma Aβ42 increases in the MCI phase and decreases just before clinical AD onset [60].
The association between clusterin and fibrillar species in human plasma was also evaluated, and a significant inverse correlation was detected in the Control group. This is consistent with the clusterin chaperone activity to preserve extracellular proteostasis [16]. Physiological concentrations of clusterin were reported to efficiently prevent the precipitation of proteins in human serum [61], supporting the association between higher levels of this protein and decreased fibrils. Remarkably, the association between clusterin and fibrils is disrupted in the dementia group, suggesting an impaired ability to properly maintain proteostasis in individuals with dementia. It cannot be excluded that the nature of the aggregates produced in demented individuals could also be slightly different (e.g., richer in fibrillar structures), and therefore more difficult to clear via clusterin. Hence, these analyses sustain the idea that pathological changes underlying dementia can disturb the proteostasis equilibrium in human plasma. A similar effect has been reported in patients with controlled hypertension [62]. Such phenomenon could be associated with changes in the plasma proteome of individuals with dementia, including differences in the aggregation state of misfolded proteins and/or impaired capacity of clusterin to prevent the formation or promote clearance of such aggregates.
This work supports that plasma clusterin levels are not affected by the different variants of the AD risk-associated CLU rs1113600 SNP but change with aging, exhibiting a more evident decline in individuals with cognitive impairment. Noticeably, clusterin levels significantly correlated with the amount of plasma fibrils in Controls, a correlation lost in dementia individuals possible reflecting a decrease in this chaperone activity. Further validation in distinct cohorts and larger populations, as well as the longitudinal assessment of plasma clusterin levels and its correlation with fibrillar structures, could be valuable for monitoring dementia. In combination with other diagnostic approaches, this can potentially contribute to evaluating the risk of developing dementia and its progression with aging.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgements
This work was supported by Centro 2020 program, Portugal 2020, EU, ADPro - CENTRO-01-0145-FEDER-181255; pAGE CENTRO-01-0145-FEDER-000003; and by Fundação para a Ciência e Tecnologia (FCT), iBiMED Grants UIDB/04501/2020 and UIDP/04501/2020. D.T. was funded through pAGE program under Grant BPD/UI98/7844/2017. M.C. was supported by FCT under Grant SFRH/BD/132995/2017 and T.S.M. under Grant SFRH/BD/145979/2019.
Thanks are due to the volunteers and their families, to all the health professionals who made this study possible.
Contributor Information
Dário Trindade, Email: dario.trindade@ua.pt.
Maria Cachide, Email: mariacachide@ua.pt.
Tânia Soares Martins, Email: martinstania@ua.pt.
Sandra Guedes, Email: sandra.silvaguedes@icr.ac.uk.
Ilka M. Rosa, Email: ilkamartins@ua.pt.
Odete A.B. da Cruz e Silva, Email: odetecs@ua.pt.
Ana Gabriela Henriques, Email: aghenriques@ua.pt.
References
- 1.W. He D. Goodkind P. Kowal An Aging World: 2015 (U.S. Census Bureau P95/16-1; International Population Reports). U.S. Government Publishing Office. 2016 https://www.census.gov/library/publications/2016/demo/P95-16-1.html.
- 2.The World Bank Population ages 65 and above (% of total population)|Data 2021 https://data.worldbank.org/indicator/SP.POP.65UP.TO.ZS.
- 3.WHO. (2022a, September 20). Dementia. https://www.who.int/news-room/fact-sheets/detail/dementia.
- 4.WHO. (2022b, October 1). Ageing and health. https://www.who.int/news-room/fact-sheets/detail/ageing-and-health.
- 5.Gale S.A., Acar D., Daffner K.R. Dementia. Am J Med. 2018;131(10):1161–1169. doi: 10.1016/j.amjmed.2018.01.022. [DOI] [PubMed] [Google Scholar]
- 6.Lopez O.L., Kuller L.H. Epidemiology of aging and associated cognitive disorders: Prevalence and incidence of Alzheimer’s disease and other dementias. Handb Clin Neurol. 2019;167:139–148. doi: 10.1016/B978-0-12-804766-8.00009-1. [DOI] [PubMed] [Google Scholar]
- 7.Bergem A.L., Engedal K., Kringlen E. The role of heredity in late-onset Alzheimer disease and vascular dementia. A twin study. Arch Gen Psychiatry. 1997;54(3):264–270. doi: 10.1001/archpsyc.1997.01830150090013. [DOI] [PubMed] [Google Scholar]
- 8.Gatz M., Reynolds C.A., Fratiglioni L., Johansson B., Mortimer J.A., Berg S., et al. Role of genes and environments for explaining Alzheimer disease. Arch Gen Psychiatry. 2006;63(2):168–174. doi: 10.1001/archpsyc.63.2.168. [DOI] [PubMed] [Google Scholar]
- 9.Deming Y., Li Z., Kapoor M., Harari O., Del-Aguila J.L., Black K., et al. Genome-wide association study identifies four novel loci associated with Alzheimer’s endophenotypes and disease modifiers. Acta Neuropathol. 2017;133(5):839–856. doi: 10.1007/s00401-017-1685-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kunkle B.W., Grenier-Boley B., Sims R., Bis J.C., Damotte V., Naj A.C., et al. Genetic meta-analysis of diagnosed Alzheimer’s disease identifies new risk loci and implicates Aβ, tau, immunity and lipid processing. Nat Genet. 2019;51(3):414–430. doi: 10.1038/s41588-019-0358-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lambert J.-C., Ibrahim-Verbaas C.A., Harold D., Naj A.C., Sims R., Bellenguez C., et al. Meta-analysis of 74,046 individuals identifies 11 new susceptibility loci for Alzheimer’s disease. Nat Genet. 2013;45(12):1452–1458. doi: 10.1038/ng.2802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.de Silva H.V., Stuart W.D., Duvic C.R., Wetterau J.R., Ray M.J., Ferguson D.G., et al. A 70-kDa apolipoprotein designated ApoJ is a marker for subclasses of human plasma high density lipoproteins. J Biol Chem. 1990;265(22):13240–13247. [PubMed] [Google Scholar]
- 13.Rizzi F., Coletta M., Bettuzzi S. vol. 104. Academic Press; 2009. Chapter 2 Clusterin (CLU): From One Gene and Two Transcripts to Many Proteins; pp. 9–23. (Advances in Cancer Research). [DOI] [PubMed] [Google Scholar]
- 14.Borghini I., Barja F., Pometta D., James R.W. Characterization of subpopulations of lipoprotein particles isolated from human cerebrospinal fluid. BBA. 1995;1255(2):192–200. doi: 10.1016/0005-2760(94)00232-n. [DOI] [PubMed] [Google Scholar]
- 15.Murphy B.F., Saunders J.R., O’Bryan M.K., Kirszbaum L., Walker I.D., d’Apice A.J. SP-40,40 is an inhibitor of C5b–6-initiated haemolysis. Int Immunol. 1989;1(5):551–554. doi: 10.1093/intimm/1.5.551. [DOI] [PubMed] [Google Scholar]
- 16.Humphreys D.T., Carver J.A., Easterbrook-Smith S.B., Wilson M.R. Clusterin has chaperone-like activity similar to that of small heat shock proteins. J Biol Chem. 1999;274(11):6875–6881. doi: 10.1074/jbc.274.11.6875. [DOI] [PubMed] [Google Scholar]
- 17.Wyatt A., Yerbury J., Poon S., Dabbs R., Wilson M. vol. 104. Academic Press; 2009. Chapter 6 The Chaperone Action of Clusterin and Its Putative Role in Quality Control of Extracellular Protein Folding; pp. 89–114. (Advances in Cancer Research). [DOI] [PubMed] [Google Scholar]
- 18.Wyatt A., Yerbury J.J., Berghofer P., Greguric I., Katsifis A., Dobson C.M., et al. Clusterin facilitates in vivo clearance of extracellular misfolded proteins. Cell Mol Life Sci. 2011;68(23):3919–3931. doi: 10.1007/s00018-011-0684-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Rodríguez-Rivera C., Garcia M.M., Molina-Álvarez M., González-Martín C., Goicoechea C. Clusterin: Always protecting. Synthesis, function and potential issues. Biomed Pharmacotherapy Biomedecine & Pharmacotherapie. 2021;134 doi: 10.1016/j.biopha.2020.111174. [DOI] [PubMed] [Google Scholar]
- 20.Ghiso J., Matsubara E., Koudinov A., Choi-Miura N.H., Tomita M., Wisniewski T., et al. The cerebrospinal-fluid soluble form of Alzheimer’s amyloid beta is complexed to SP-40,40 (apolipoprotein J), an inhibitor of the complement membrane-attack complex. Biochem J. 1993;293(Pt 1):27–30. doi: 10.1042/bj2930027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Whiten D.R., Cox D., Horrocks M.H., Taylor C.G., De S., Flagmeier P., et al. Single-Molecule Characterization of the Interactions between Extracellular Chaperones and Toxic α-Synuclein Oligomers. Cell Rep. 2018;23(12):3492–3500. doi: 10.1016/j.celrep.2018.05.074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Calero M., Rostagno A., Matsubara E., Zlokovic B., Frangione B., Ghiso J. Apolipoprotein J (clusterin) and Alzheimer’s disease. Microsc Res Tech. 2000;50(4):305–315. doi: 10.1002/1097-0029(20000815)50:4<305::AID-JEMT10>3.0.CO;2-L. [DOI] [PubMed] [Google Scholar]
- 23.Beeg M., Stravalaci M., Romeo M., Carrá A.D., Cagnotto A., Rossi A., et al. Clusterin Binds to Aβ1-42 Oligomers with High Affinity and Interferes with Peptide Aggregation by Inhibiting Primary and Secondary Nucleation. J Biol Chem. 2016;291(13):6958–6966. doi: 10.1074/jbc.M115.689539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Robbins J.P., Perfect L., Ribe E.M., Maresca M., Dangla-Valls A., Foster E.M., et al. Clusterin Is Required for β-Amyloid Toxicity in Human iPSC-Derived Neurons. Front Neurosci. 2018;12 doi: 10.3389/fnins.2018.00504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Yeh F.L., Wang Y., Tom I., Gonzalez L.C., Sheng M. TREM2 Binds to Apolipoproteins, Including APOE and CLU/APOJ, and Thereby Facilitates Uptake of Amyloid-Beta by Microglia. Neuron. 2016;91(2):328–340. doi: 10.1016/j.neuron.2016.06.015. [DOI] [PubMed] [Google Scholar]
- 26.Yuste-Checa P., Trinkaus V.A., Riera-Tur I., Imamoglu R., Schaller T.F., Wang H., et al. The extracellular chaperone Clusterin enhances Tau aggregate seeding in a cellular model. Nat Commun. 2021;12(1):4863. doi: 10.1038/s41467-021-25060-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lidström A.M., Bogdanovic N., Hesse C., Volkman I., Davidsson P., Blennow K. Clusterin (apolipoprotein J) protein levels are increased in hippocampus and in frontal cortex in Alzheimer’s disease. Exp Neurol. 1998;154(2):511–521. doi: 10.1006/exnr.1998.6892. [DOI] [PubMed] [Google Scholar]
- 28.Miners J.S., Clarke P., Love S. Clusterin levels are increased in Alzheimer’s disease and influence the regional distribution of Aβ. Brain Pathology (Zurich, Switzerland) 2017;27(3):305–313. doi: 10.1111/bpa.12392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Nilselid A.-M., Davidsson P., Nägga K., Andreasen N., Fredman P., Blennow K. Clusterin in cerebrospinal fluid: Analysis of carbohydrates and quantification of native and glycosylated forms. Neurochem Int. 2006;48(8):718–728. doi: 10.1016/j.neuint.2005.12.005. [DOI] [PubMed] [Google Scholar]
- 30.Sihlbom C., Davidsson P., Sjögren M., Wahlund L.-O., Nilsson C.L. Structural and quantitative comparison of cerebrospinal fluid glycoproteins in Alzheimer’s disease patients and healthy individuals. Neurochem Res. 2008;33(7):1332–1340. doi: 10.1007/s11064-008-9588-x. [DOI] [PubMed] [Google Scholar]
- 31.Mullan G.M., McEneny J., Fuchs M., McMaster C., Todd S., McGuinness B., et al. Plasma clusterin levels and the rs11136000 genotype in individuals with mild cognitive impairment and Alzheimer’s disease. Curr Alzheimer Res. 2013;10(9):973–978. doi: 10.2174/15672050113106660162. [DOI] [PubMed] [Google Scholar]
- 32.Schrijvers E.M.C., Koudstaal P.J., Hofman A., Breteler M.M.B. Plasma clusterin and the risk of Alzheimer disease. J Am Med Assoc. 2011;305(13):1322–1326. doi: 10.1001/jama.2011.381. [DOI] [PubMed] [Google Scholar]
- 33.Thambisetty M., Simmons A., Velayudhan L., Hye A., Campbell J., Zhang Y.i., et al. Association of plasma clusterin concentration with severity, pathology, and progression in Alzheimer disease. Arch Gen Psychiatry. 2010;67(7):739. doi: 10.1001/archgenpsychiatry.2010.78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hsu J.-L., Lee W.-J., Liao Y.-C., Wang S.-J., Fuh J.-L. The clinical significance of plasma clusterin and Aβ in the longitudinal follow-up of patients with Alzheimer’s disease. Alzheimer’s Res Therapy. 2017;9(1):91. doi: 10.1186/s13195-017-0319-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Jongbloed W., van Dijk K.D., Mulder S.D., van de Berg W.D.J., Blankenstein M.A., van der Flier W., et al. Clusterin Levels in Plasma Predict Cognitive Decline and Progression to Alzheimer’s Disease. J Alzheimer’s Disease: JAD. 2015;46(4):1103–1110. doi: 10.3233/JAD-150036. [DOI] [PubMed] [Google Scholar]
- 36.Weinstein G., Beiser A.S., Preis S.R., Courchesne P., Chouraki V., Levy D., et al. Plasma clusterin levels and risk of dementia, Alzheimer’s disease, and stroke. Alzheimer’s & Dementia (Amsterdam, Netherlands) 2016;3:103–109. doi: 10.1016/j.dadm.2016.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Foster E.M., Dangla-Valls A., Lovestone S., Ribe E.M., Buckley N.J. Clusterin in Alzheimer’s Disease: Mechanisms, Genetics, and Lessons From Other Pathologies. Front Neurosci. 2019;13:164. doi: 10.3389/fnins.2019.00164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Harold D., Abraham R., Hollingworth P., Sims R., Gerrish A., Hamshere M.L., et al. Genome-wide association study identifies variants at CLU and PICALM associated with Alzheimer’s disease. Nat Genet. 2009;41(10):1088–1093. doi: 10.1038/ng.440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Lambert J.-C., Heath S., Even G., Campion D., Sleegers K., Hiltunen M., et al. Genome-wide association study identifies variants at CLU and CR1 associated with Alzheimer’s disease. Nat Genet. 2009;41(10):1094–1099. doi: 10.1038/ng.439. [DOI] [PubMed] [Google Scholar]
- 40.Bertram L., McQueen M.B., Mullin K., Blacker D., Tanzi R.E. Systematic meta-analyses of Alzheimer disease genetic association studies: The AlzGene database. Nat Genet. 2007;39(1):Article 1. doi: 10.1038/ng1934. [DOI] [PubMed] [Google Scholar]
- 41.Lin Y.-L., Chen S.-Y., Lai L.-C., Chen J.-H., Yang S.-Y., Huang Y.-L., et al. Genetic polymorphisms of clusterin gene are associated with a decreased risk of Alzheimer’s disease. Eur J Epidemiol. 2012;27(1):73–75. doi: 10.1007/s10654-012-9650-5. [DOI] [PubMed] [Google Scholar]
- 42.Ma J., Qiu S. Genetic variant rs11136000 upregulates clusterin expression and reduces Alzheimer’s disease risk. Front Neurosci. 2022;16 doi: 10.3389/fnins.2022.926830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Thambisetty M., Beason-Held L.L., An Y., Kraut M., Nalls M., Hernandez D.G., et al. Alzheimer Risk Variant CLU and Brain Function During Aging. Biol Psychiatry. 2013;73(5):399–405. doi: 10.1016/j.biopsych.2012.05.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Tan L., Wang H.-F., Tan M.-S., Tan C.-C., Zhu X.-C., Miao D., et al. Effect of CLU genetic variants on cerebrospinal fluid and neuroimaging markers in healthy, mild cognitive impairment and Alzheimer’s disease cohorts. Sci Rep. 2016;6(1) doi: 10.1038/srep26027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.DiBattista A.M., Stevens B.W., Rebeck G.W., Green A.E. Two Alzheimer’s disease risk genes increase entorhinal cortex volume in young adults. Front Hum Neurosci. 2014;8 doi: 10.3389/fnhum.2014.00779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Haight T., Bryan R.N., Meirelles O., Tracy R., Fornage M., Richard M., et al. Associations of plasma clusterin and Alzheimer’s disease-related MRI markers in adults at mid-life: The CARDIA Brain MRI sub-study. PLoS One. 2018;13(1) doi: 10.1371/journal.pone.0190478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Balcar V.J., Zeman T., Janout V., Janoutová J., Lochman J., Šerý O. Single Nucleotide Polymorphism rs11136000 of CLU Gene (Clusterin, ApoJ) and the Risk of Late-Onset Alzheimer’s Disease in a Central European Population. Neurochem Res. 2021;46(2):411–422. doi: 10.1007/s11064-020-03176-y. [DOI] [PubMed] [Google Scholar]
- 48.Schürmann B., Wiese B., Bickel H., Weyerer S., Riedel-Heller S.G., Pentzek M., et al. Association of the Alzheimer’s Disease Clusterin Risk Allele with Plasma Clusterin Concentration. J Alzheimer’s Disease. 2011;25(3):421–424. doi: 10.3233/JAD-2011-110251. [DOI] [PubMed] [Google Scholar]
- 49.Cai R., Han J., Sun J., Huang R., Tian S., Shen Y., et al. Plasma Clusterin and the CLU Gene rs11136000 Variant Are Associated with Mild Cognitive Impairment in Type 2 Diabetic Patients. Front Aging Neurosci. 2016;8:179. doi: 10.3389/fnagi.2016.00179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Rosa I.M., Henriques A.G., Carvalho L., Oliveira J., da Cruz E., Silva O.A.B. Screening Younger Individuals in a Primary Care Setting Flags Putative Dementia Cases and Correlates Gastrointestinal Diseases with Poor Cognitive Performance. Dement Geriatr Cogn Disord. 2017;43(1–2):15–28. doi: 10.1159/000452485. [DOI] [PubMed] [Google Scholar]
- 51.Morris J.C. The Clinical Dementia Rating (CDR): Current version and scoring rules. Neurology. 1993;43(11):2412–a. doi: 10.1212/WNL.43.11.2412-a. [DOI] [PubMed] [Google Scholar]
- 52.Rosa I.M., Henriques A.G., Wiltfang J., da Cruz E., Silva O.A.B. Putative Dementia Cases Fluctuate as a Function of Mini-Mental State Examination Cut-Off Points. J Alzheimer’s Dis: JAD. 2018;61(1):157–167. doi: 10.3233/JAD-170501. [DOI] [PubMed] [Google Scholar]
- 53.Darawi M.N., Ai-Vyrn C., Ramasamy K., Hua P.P.J., Pin T.M., Kamaruzzaman S.B., et al. Allele-specific polymerase chain reaction for the detection of Alzheimer’s disease-related single nucleotide polymorphisms. BMC Med Genet. 2013;14(1):27. doi: 10.1186/1471-2350-14-27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Magalhães S., Trindade D., Martins T., Martins Rosa I., Delgadillo I., Goodfellow B.J., et al. Monitoring plasma protein aggregation during aging using conformation-specific antibodies and FTIR spectroscopy. Clinica Chimica Acta; Int J Clin Chem. 2020;502:25–33. doi: 10.1016/j.cca.2019.11.025. [DOI] [PubMed] [Google Scholar]
- 55.Kayed R., Head E., Sarsoza F., Saing T., Cotman C.W., Necula M., et al. Fibril specific, conformation dependent antibodies recognize a generic epitope common to amyloid fibrils and fibrillar oligomers that is absent in prefibrillar oligomers. Mol Neurodegener. 2007;2:18. doi: 10.1186/1750-1326-2-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Petersen R.C. Mild cognitive impairment as a diagnostic entity. J Intern Med. 2004;256(3):183–194. doi: 10.1111/j.1365-2796.2004.01388.x. [DOI] [PubMed] [Google Scholar]
- 57.Lu S.-J., Li H.-L., Sun Y.-M., Liu Z.-J., Yang P., Wu Z.-Y. Clusterin variants are not associated with southern Chinese patients with Alzheimer’s disease. Neurobiol Aging. 2014;35(11):2656.e9–2656.e11. doi: 10.1016/j.neurobiolaging.2014.05.015. [DOI] [PubMed] [Google Scholar]
- 58.Ha J., Moon M.K., Kim H., Park M., Cho S.Y., Lee J., et al. Plasma Clusterin as a Potential Link Between Diabetes and Alzheimer Disease. J Clin Endocrinol Metab. 2020;105(9):dgaa378. doi: 10.1210/clinem/dgaa378. [DOI] [PubMed] [Google Scholar]
- 59.Shi X., Xie B., Tang* Y.X. Plasma Clusterin as a Potential Biomarker for Alzheimer’s Disease-A Systematic Review and Meta-analysis. Current Alzheimer Research. 2019;16(11):1018–1027. doi: 10.2174/1567205016666191024141757. [DOI] [PubMed] [Google Scholar]
- 60.Chen T.-B., Lai Y.-H., Ke T.-L., Chen J.-P., Lee Y.-J., Lin S.-Y., et al. Changes in Plasma Amyloid and Tau in a Longitudinal Study of Normal Aging, Mild Cognitive Impairment, and Alzheimer’s Disease. Dement Geriatr Cogn Disord. 2019;48(3–4):180–195. doi: 10.1159/000505435. [DOI] [PubMed] [Google Scholar]
- 61.Poon S., Easterbrook-Smith S.B., Rybchyn M.S., Carver J.A., Wilson M.R. Clusterin is an ATP-independent chaperone with very broad substrate specificity that stabilizes stressed proteins in a folding-competent state. Biochemistry. 2000;39(51):15953–15960. doi: 10.1021/bi002189x. [DOI] [PubMed] [Google Scholar]
- 62.Teixeira M., Trindade D., Gouveia M., Eller-Borges R., Magalhães S., Duarte A., et al. Proteostasis Response to Protein Misfolding in Controlled Hypertension. Cells. 2022;11(10):1686. doi: 10.3390/cells11101686. [DOI] [PMC free article] [PubMed] [Google Scholar]