Abstract
Background
Preterm birth affects lungs in several ways but few studies have follow-up until adulthood. We investigated the association of the entire spectrum of gestational ages with specialist care episodes for obstructive airway disease (asthma and chronic obstructive pulmonary disease (COPD)) at age 18–50 years.
Methods
We used nationwide registry data on 706 717 people born 1987–1998 in Finland (4.8% preterm) and 1 669 528 born 1967–1999 in Norway (5.0% preterm). Care episodes of asthma and COPD were obtained from specialised healthcare registers, available in Finland for 2005–2016 and in Norway for 2008–2017. We used logistic regression to estimate odds ratios (ORs) for having a care episode with either disease outcome.
Results
Odds of any obstructive airway disease in adulthood for those born at <28 or 28–31 completed weeks were 2–3-fold of those born full term (39–41 completed weeks), persisting after adjustments. For individuals born at 32–33, 34–36 or 37–38 weeks, the odds were 1.1- to 1.5-fold. Associations were similar in the Finnish and the Norwegian data and among people aged 18–29 and 30–50 years. For COPD at age 30–50 years, the OR was 7.44 (95% CI 3.49–15.85) for those born at <28 weeks, 3.18 (95% CI 2.23–4.54) for those born at 28–31 weeks and 2.32 (95% CI 1.72–3.12) for those born at 32–33 weeks. Bronchopulmonary dysplasia in infancy increased the odds further for those born at <28 and 28–31 weeks.
Conclusion
Preterm birth is a risk factor for asthma and COPD in adulthood. The high odds of COPD call for diagnostic vigilance when adults born very preterm present with respiratory symptoms.
Short abstract
In this population-based study of 2.4 million people from Finland and Norway, adults born preterm were at higher risk for asthma and COPD at ages 18–50 years. The risks were observed across all gestational ages before full term in a dose-response manner. https://bit.ly/3YqnUGe
Introduction
One in ten babies worldwide is born preterm, before 37 completed weeks of gestation [1]. Preterm birth disrupts normal lung development, which can have lasting effects on lung function, in particular on airflow [2–4]. This is most evident in infants born very preterm (<32 weeks) who develop bronchopulmonary dysplasia (BPD), which is characterised by alveolar septal fibrosis and inflammation or by impaired alveolar growth [3, 5, 6].
Impaired airflow is the hallmark of the obstructive airway diseases asthma and chronic obstructive pulmonary disease (COPD). In 2017, the global prevalence of asthma was estimated at 3.6% and COPD at 3.9% [7]. The prevalence of asthma is highest in childhood and, after a nadir in young adulthood, increases again with age, while the prevalence of COPD increases steadily with age from early midlife onwards. Although COPD is typically linked to smoking and indoor or outdoor air pollution, fetal and childhood factors that impair lung development and function, such as prematurity or allergy and bronchitis in childhood, are recognised risk factors for both asthma and COPD [3, 8–11].
Studies of the risk of adult obstructive airway diseases among preterm-born individuals show mixed findings. Many show an association between preterm birth and obstructive airway disease in younger ages, whereas most studies show no association in older ages [8, 12–16]. However, there is evidence of a more prominent risk for asthma in preterm-born women [17], COPD in both women and men [5, 6, 18] and increased mortality from chronic lung diseases [19] in adulthood. A separate body of research has focused on those born very preterm (<32 weeks) or at very low birth weight (<1500 g) who have lower expiratory airflow as young adults than those born at term [18]. Airflow is even lower among those who have experienced neonatal BPD [5, 6, 18]. However, several gaps in the knowledge remain. So far, few studies have had sufficiently long follow-up and adequate statistical power to assess early life origins of COPD. Moreover, many studies have assessed infants born preterm as one group, not appreciating the wide variation in degree of immaturity or related traits such as being born small for gestational age (SGA).
In this population-based register study we investigated the association between the full range of gestational ages (GA) and obstructive airway disease (asthma and/or COPD) in early and mid-adulthood, using nationwide data from two Nordic countries. Additionally, we investigated whether the association differed in subgroups of individuals born preterm who were diagnosed with BPD or were born SGA.
Material and methods
Study population
The study populations, identified from the Medical Birth Registers (MBR), comprised all live births in Finland from 1987 to 1998 (n=750 733) and in Norway from 1967 to 1999 (n=1 904 600). More information of the MBRs can be found in the supplementary material. Data from the MBRs in Finland and Norway were linked with data from other national registers, using encrypted national identification codes provided to every citizen and permanent resident of Finland or Norway [20–24].
We excluded individuals who died (n=41 494, 1.6%) or emigrated (n=8797, 0.3%) before the start of the follow-up, who had congenital anomalies (n=69 238, 2.6%), missing GA (n=123 109, 4.6%), GA <23+0 or >43+6 weeks or implausible GA and birth weight combinations (n=29 296, 1.1%), GA >33+6 completed weeks who had a diagnosis of BPD (n=33, 0.0%) and those with missing data on covariates, including triplets and quadruplets due to missing birth weight z-score (n=7121, 0.3%) (supplementary figure S1).
In Finland, this study was approved by Research Ethics Board of the Finnish Institute for Health and Welfare (THL/1960/6.02.01/2018, §810), and in Norway by the Central Norway Regional Committee for Medical Research Ethics (2018/32) and by the registers. Information on data permissions and waiver of informed consent is presented in the supplementary material.
Gestational age
GA in completed weeks was ascertained from the MBRs [23, 24]. The duration of gestation has been confirmed by ultrasonography in Finland since the late 1980s or early 1990s, and in Norway from December 1998. Prior to that, duration of gestation was estimated mainly based on data on the last menstrual period or by means of clinical examination [25, 26]. GA was categorised as extremely preterm (23–27 weeks), very preterm (28–31 weeks), moderately preterm (32–33 weeks), late preterm (34–36 weeks), early term (37–38 weeks), full term (39–41 weeks, reference) and post-term (≥42 weeks).
Obstructive airway disease outcomes
We started the follow-up at the earliest when the individuals turned 18 years: in Finland the study time frame extended from 2005 (when the oldest Finnish individuals within the study population turned 18 years) to 2016 and in Norway from 2008 (when the national identity number was introduced in the Norwegian Patient Registry (NPR), allowing data to be obtained at the individual level) to 2017 (supplementary figure S2). During these time periods, the Finnish Care Register for Health Care (CRHC) covered all specialist inpatient care and public hospital outpatient care episodes [21] and the NPR covered all publicly funded specialist inpatient and outpatient care episodes [20], with care records using the 10th revision of International Statistical Classification of Diseases and Related Health Problems (ICD) codes. For asthma, we included diagnostic codes J45–J46 [27], and for COPD diagnosis subgroups of pulmonary emphysema (J43), other COPD (J41–J42, J44.0–J44.8) and unspecified COPD (J44.9) [17, 28]. Individuals were considered as cases if they had an inpatient or outpatient hospital or specialist care episode with these codes as the main diagnosis, regardless of the number of records.
Covariates
As covariates we selected factors that are associated with preterm birth [29, 30] and asthma [31] or COPD [9, 32]. Information on sex, birth year, birth weight, single versus twin birth, parity, Caesarean section and mother's age were obtained from the MBRs. Birth weight z-scores (BWZ) were calculated using the Sankilampi growth references for singletons and twins born at GA 23–43 weeks [33]. Mother's asthma diagnosis was obtained from the CRHC in Finland and from the MBR in Norway. Information on mother's hypertensive disorders during pregnancy (chronic hypertension or gestational hypertension, and pre-eclampsia or eclampsia) was obtained from both CRHC and MBR in Finland and from the MBR in Norway. Data on the highest attained education of the mother and father were obtained from Statistics Finland and Statistics Norway. Diagnosis of BPD was obtained from the CRHC and maternal smoking during pregnancy from the MBR in Finland. The methodological appendix provides details on the covariates and the directed acyclic graph presenting the assumed relationships between the variables used in this study.
Statistical analysis
In the main analyses we used logistic regression to estimate odds ratios and associated 95% confidence intervals for having a care episode with diagnoses of obstructive airway disease in specialty care across the seven GA categories, with those born full term as the reference. We investigated the association for all obstructive airway diseases combined, for asthma and COPD separately, and for the COPD subgroups (in Norway only). In Finland, we included care episodes at 18–29 years. In Norway, we included two age categories, ages 18–29 years and 30–50 years, and the full follow-up at ages 18–50 years, and used generalised estimation equations (PROC GENMOD in SAS; SAS Institute, Cary, NC, USA) with exchangeable working correlation structure to account for intraindividual dependence of those participants who contributed to both of the two age categories [34]. We calculated the population attributable risk to estimate the risk of obstructive airway disease attributable to preterm birth and BPD (only available in the Finnish data) in the total population. Numbers of individuals included in the analyses, by age category and country, are presented in supplementary figure S1.
We constructed two models: Model 1 was adjusted for sex and birth year. Model 2 was additionally adjusted for BWZ, twin birth, parity, Caesarean section, mother's age, mother's hypertensive disorders during pregnancy, mother's asthma, and mother's and father's highest education (separate measures). In the Finnish data, we created subcategories for the extremely, very and moderately preterm groups with and without BPD diagnosis and compared them to the group born full term (interaction for BPD*GA categories p<0.001 for obstructive airway diseases combined). We also ran the models for ages 18–29 years in both countries, in subgroups born SGA (BWZ <−2 sds), appropriate for gestational age (AGA) (BWZ −2 to +2 sds) and large for gestational age (LGA) (BWZ >+2 sds), comparing categories of those born preterm to those born full term (interaction for birth weight groups*GA categories p<0.001 in both countries).
For the sensitivity analyses, in both countries, we ran the main analyses using care episodes only from 1) pulmonology speciality or 2) inpatient care, and 3) using completed weeks of GA as the exposure, with GA 40 weeks as the reference category, as well as running the analyses separately by 4) sex and 5) mother's parity, and 6) by excluding twin births. Additionally, using only data from Finland, we 7) constructed a model adjusting additionally for maternal smoking during pregnancy. Further, to account for possible survival bias, we 8) calculated survival time from the beginning of the follow-up to the first care episode of obstructive airway disease, death or emigration and estimated hazard ratios from Cox regression in Finland and Norway at age 18–29 years. Statistical analyses were conducted using SAS EG versions 7.13 and 7.15 (SAS Institute Inc.).
Results
Study population
Selection of the study population is presented in supplementary figure S1. Of the 2 655 333 individuals born alive in Finland or Norway during the baseline period, 2 376 245 (89.5%) were included in the analyses (706 717 in Finland and 1 669 528 in Norway). Detailed characteristics of the study population by country are shown in table 1 and by outcome groups in supplementary table S1. In total, 1.9% (n=45 624) of the participants had a specialised healthcare episode with diagnosis of obstructive airway disease during the follow-up (supplementary table S2). Of these individuals, 90.6% (n=41 332) had asthma only, 5.9% (n=2706) COPD only and 3.5% (n=1586) had both diseases during the follow-up.
TABLE 1.
Characteristics of the study population
| Finland, born 1987–1998 | Norway, born 1967–1999 | |
| Individuals, n | 706 717 | 1 669 528 |
| Male | 360 028 (50.9) | 852 867 (51.1) |
| GA at birth, completed weeks | ||
| Extremely preterm (23–27) | 850 (0.1) | 1359 (0.1) |
| Very preterm (28–31) | 2793 (0.4) | 6931 (0.4) |
| Moderately preterm (32–33) | 4048 (0.6) | 10 880 (0.7) |
| Late preterm (34–36) | 26 557 (3.8) | 65 057 (3.9) |
| Early term (37–38) | 125 648 (17.8) | 218 145 (13.1) |
| Full term (39–41) | 515 608 (73.0) | 1 153 153 (69.1) |
| Post-term (≥42) | 31 213 (4.4) | 214 003 (12.8) |
| Birth weight Z-score | 0.00±1.06 | 0.00±1.11 |
| Small for gestational age | 17 958 (2.5) | 77 405 (4.6) |
| Large for gestational age | 23 612 (3.3) | 47 144 (2.8) |
| First born | 281 020 (39.8) | 687 968 (41.2) |
| Singleton | 689 038 (97.5) | 1 634 803 (97.9) |
| Caesarean section | 105 846 (15.0) | 135 827 (8.1) |
| Bronchopulmonary dysplasia # | 616 (0.1) | N/A |
| Follow-up time, years | 6.0 (3.2–8.9) | 10.0 (7.5–10.0) |
| Attained age | ||
| 18–29 years | 706 717 (100.0) | 623 024 (37.3) |
| 30–50 years | – (0.0) | 1 046 504 (62.7) |
| Mother's age at giving birth, years | 29.0±5.18 | 27.4±5.24 |
| Maternal smoking during pregnancy #,¶ | 108 184 (15.6) | N/A |
| Mother's history of asthma | 8705 (1.2) | 25 616 (1.5) |
| Mother's hypertensive disorders during pregnancy | ||
| No | 668 333 (94.6) | 1 595 953 (95.6) |
| Chronic or gestational hypertension | 20 110 (2.9) | 27 339 (1.6) |
| Pre-eclampsia or eclampsia | 18 274 (2.6) | 46 236 (2.8) |
| Mother's education | ||
| Unknown | 3559 (0.5) | 10 541 (0.6) |
| Low (ISCED classes 0–2) | 71 668 (10.1) | 408 046 (24.4) |
| Intermediate (ISCED classes 3–5) | 464 997 (65.8) | 770 949 (46.2) |
| High (ISCED classes 6–8) | 166 493 (23.6) | 479 992 (28.8) |
| Father's education | ||
| Unknown | 10 676 (1.5) | 25 541 (1.5) |
| Low (ISCED classes 0–2) | 128 028 (18.1) | 372 900 (22.3) |
| Intermediate (ISCED classes 3–5) | 426 067 (60.3) | 836 690 (50.1) |
| High (ISCED classes 6–8) | 141 946 (20.1) | 434 397 (26.0) |
| Care episode at 18–29 years of age + | ||
| Obstructive airway disease§ | 9512 (1.3) | 14 763 (1.4) |
| Asthma, J45–J46 | 9412 (1.3) | 14 418 (1.4) |
| COPD, J41–J44 | 147 (0.0) | 582 (0.1) |
| Care episode at 30–50 years of age +,ƒ | ||
| Obstructive airway disease§ | N/A | 22 271 (2.1) |
| Asthma, J45–J46 | N/A | 19 971 (1.9) |
| COPD, J41–J44 | N/A | 3594 (0.3) |
| Care episodes from inpatient care | ||
| Obstructive airway disease at age 18–29 years+,§ | 520 (0.1) | 987 (0.1) |
| Obstructive airway disease at age 30–50 years+,§,ƒ | N/A | 2091 (0.2) |
| Care episodes from pulmonology speciality | ||
| Obstructive airway disease at age 18–29 years+,§ | 6398 (0.9) | 8811 (0.8) |
| Obstructive airway disease at age 30–50 years+,§,ƒ | N/A | 15 975 (1.5) |
Data are presented as n (%), mean±sd or median (interquartile range), unless otherwise stated. GA: gestational age; ISCED: International Standard Classification of Education; COPD: chronic obstructive pulmonary disease; N/A: not available. #: information not available in Norway; ¶: maternal smoking missing in Finland, n=14 028; +: number contributing to analyses at different age categories in Norway: 18–29 years: 1 065 372, 30–50 years: 1 046 611; §: obstructive airway disease comprises both asthma and/or COPD; ƒ: information not available in Finland.
Main results
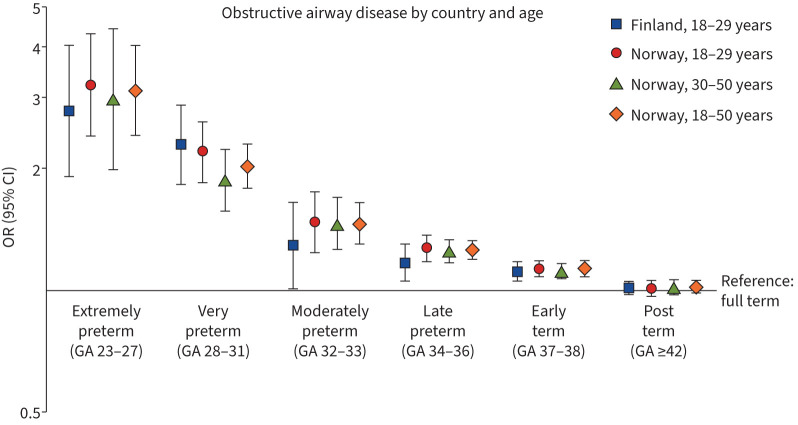
All groups born preterm had higher odds of obstructive airway disease in adulthood (figure 1). ORs for both models are presented in supplementary table S3. We observed a graded association between GA at birth and the odds of having a care episode of obstructive airway disease in adulthood (p for trend <0.001 in both countries). In the Finnish dataset, the odds of obstructive airway disease at age 18–29 years were the highest (OR 2.77, 95% CI 1.91–4.02) in the extremely preterm group compared to the group born full term. The corresponding OR in the Norwegian dataset was 3.22 (95% CI 2.40–4.31). In Norway, estimates were only slightly lower at age 30–50 years and 18–50 years (extremely preterm group: OR 2.96 (95% CI 1.98–4.43) and OR 3.12 (95% CI 2.42–4.03), respectively). Adjustment for covariates did not alter the direction or magnitude of the estimates. The odds of airway disease did not markedly differ between individuals born full term and post-term. Population attributable risk of preterm birth for obstructive airway disease was 1.2% (95% CI 0.7–1.7%) in Finland and 1.3% (95% CI 1.1–1.6) in Norway, and of BPD was 0.3% (95% CI 0.2–0.5%) in Finland.
FIGURE 1.
Associations between gestational age (GA) in weeks and any obstructive airway disease (asthma or chronic obstructive pulmonary disease), adjusted for sex and birth year.
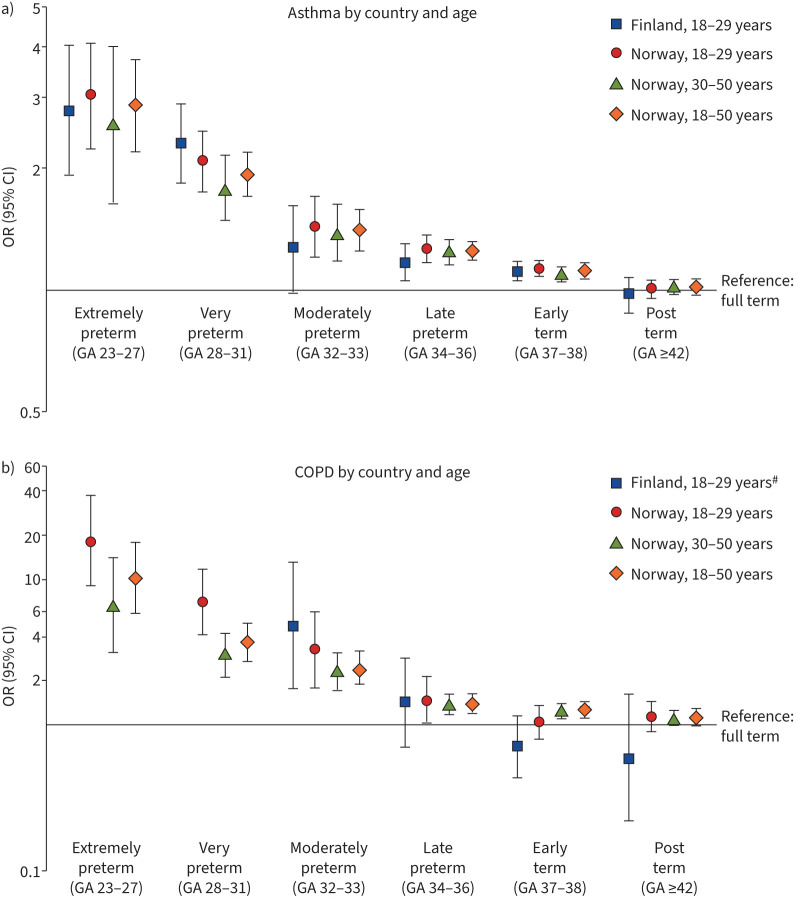
For asthma, the risk was higher in all groups born preterm, except for those born moderately preterm in Finland (figure 2a). The risk for COPD was higher in Norway in all groups born preterm, but not for those aged 18–29 years born early term (figure 2b). In Finland, there were no COPD cases in the extremely or very preterm groups. The risk of COPD was markedly increased for the moderately preterm group (figure 2b). These associations attenuated only slightly after adjustment for covariates (table 2, supplementary table S3). ORs for COPD subgroups in Norway are shown in table 2. The highest odds in those born extremely preterm were for emphysema (18–29 years: OR 31.09 (95% CI 7.54–128.14); 30–50 years: OR 16.66 (95% CI 4.13–67.12); 18–50 years: OR 21.68 (95% CI 6.47–71.63) in the fully adjusted model).
FIGURE 2.
Associations between gestational age (GA) in weeks and a) asthma and b) chronic obstructive pulmonary disease (COPD), adjusted for sex and birth year. #: missing outcome values for extremely and very preterm born in Finland.
TABLE 2.
Odds for different obstructive airway diseases
| Finland | Norway | |||
| 18–29 years | 18–29 years | 30–50 years | 18–50 years | |
| OR (95% CI) | OR (95% CI) | OR (95% CI) | OR (95% CI) | |
| Asthma, J45–J46 | ||||
| Full term# | Reference | Reference | Reference | Reference |
| Extremely preterm | 2.90 (2.00–4.21) | 3.10 (2.29–4.20) | 2.71 (1.73–4.25) | 2.96 (2.26–3.88) |
| Very preterm | 2.26 (1.80–2.85) | 2.04 (1.71–1.44) | 1.83 (1.51–2.21) | 1.94 (1.70–2.21) |
| Moderately preterm | 1.24 (0.96–1.60) | 1.42 (1.20–1.69) | 1.41 (1.20–1.66) | 1.41 (1.25–1.60) |
| Late preterm | 1.17 (1.05–1.30) | 1.28 (1.18–1.38) | 1.25 (1.17–1.34) | 1.26 (1.20–1.33) |
| Early term | 1.12 (1.06–1.18) | 1.13 (1.08–1.19) | 1.10 (1.05–1.15) | 1.11 (1.08–1.15) |
| Post-term | 0.94 (0.85–1.05) | 0.98 (0.93–1.03) | 1.00 (0.95–1.04) | 0.99 (0.96–1.02) |
| All COPD, J41–J44 | ||||
| Full term | Reference | Reference | Reference | Reference |
| Extremely preterm# | 18.92 (9.30–38.46) | 7.44 (3.49–15.85) | 9.58 (5.50–16.69) | |
| Very preterm# | 6.69 (4.00–11.18) | 3.18 (2.23–4.54) | 3.32 (2.45–4.49) | |
| Moderately preterm | 3.95 (1.38–11.32) | 3.13 (1.72–5.70) | 2.32 (1.72–3.12) | 2.28 (1.75–2.97) |
| Late preterm | 1.28 (0.60–2.71) | 1.47 (1.01–2.13) | 1.39 (1.19–1.63) | 1.41 (1.22–1.63) |
| Early term | 0.66 (0.40–1.09) | 1.03 (0.80–1.32) | 1.23 (1.12–1.35) | 1.23 (1.13–1.35) |
| Post-term | 0.61 (0.22–1.65) | 1.08 (0.85–1.38) | 1.03 (0.93–1.14) | 0.97 (0.88–1.08) |
| Emphysema, J43 | ||||
| Full term | Reference | Reference | Reference | Reference |
| Extremely preterm# | 31.09 (7.54–128.14) | 16.66 (4.13–67.23) | 21.68 (6.47–71.63) | |
| Very preterm# | 9.54 (3.09–29.45) | 2.60 (0.83–8.18) | 4.06 (1.86–8.90) | |
| Moderately preterm# | 6.28 (1.95–20.17) | 1.84 (0.68–4.98) | 2.62 (1.23–5.58) | |
| Late preterm# | 1.09 (0.34–3.44) | 1.16 (0.70–1.93) | 1.15 (0.72–1.81) | |
| Early term# | 0.62 (0.27–1.46) | 1.13 (0.84–1.52) | 1.04 (0.79–1.38) | |
| Post-term# | 1.14 (0.59–2.18) | 1.18 (0.90–1.56) | 1.17 (0.91–1.52) | |
| Other COPD, J41, J42, J44 (not J44.9) | ||||
| Full term | Reference | Reference | Reference | Reference |
| Extremely preterm# | 6.39 (0.89–45.79) | 8.33 (3.68–18.85) | 7.99 (3.77–16.97) | |
| Very preterm# | 8.27 (3.88–17.60) | 3.61 (2.48–5.24) | 4.06 (2.91–5.67) | |
| Moderately preterm# | 2.23 (0.71–6.97) | 2.61 (1.91–3.56) | 2.58 (1.91–3.48) | |
| Late preterm# | 1.01 (0.49–2.06) | 1.51 (1.28–1.79) | 1.48 (1.25–1.74) | |
| Early term# | 0.95 (0.63–1.45) | 1.23 (1.11–1.37) | 1.21 (1.09–1.34) | |
| Post-term# | 1.22 (0.84–1.77) | 1.04 (0.93–1.16) | 1.05 (0.94–1.17) | |
| Unspecified COPD, J44.9 | ||||
| Full term | Reference | Reference | Reference | Reference |
| Extremely preterm# | 32.37 (14.16–73.96) | 9.82 (4.33–22.25) | 15.13 (8.41–27.21) | |
| Very preterm# | 7.73 (3.82–15.64) | 3.59 (2.40–5.35) | 4.11 (2.85–5.94) | |
| Moderately preterm# | 3.01 (1.24–7.33) | 2.68 (1.93–3.72) | 2.71 (1.99–3.68) | |
| Late preterm# | 2.04 (1.28–3.25) | 1.56 (1.31–1.86) | 1.61 (1.36–1.90) | |
| Early term# | 1.21 (0.86–1.69) | 1.24 (1.11–1.39) | 1.23 (1.11–1.37) | |
| Post-term# | 0.97 (0.68–1.40) | 1.04 (0.93–1.17) | 1.04 (0.93–1.16) | |
Models were adjusted for sex, birth year, birth weight z-score, multiple birth, parity, Caesarean section, mother's age, mother's hypertensive disorders during pregnancy, mother's asthma, and mother's and father's education. Extremely preterm: gestational age 23–27 completed weeks; very preterm: 28–31 weeks; moderately preterm: 32–33 weeks; late preterm: 34–36 weeks; early term: 37–38 weeks; full term: 39–41 weeks; post-term: ≥42 weeks. #: missing outcome values in Finland.
Bronchopulmonary dysplasia
Data on neonatal BPD were available in Finland (figure 3). Associations with GA were stronger among those with than those without a BPD diagnosis. With term-born individuals as the reference group, the fully adjusted OR was 4.79 (95% CI 2.88–7.99) for those born extremely preterm with BPD versus 1.91 (95% CI 1.10–3.33) for those without BPD. The corresponding estimates for those born very preterm were 8.17 (95% CI 5.15–12.93) versus 1.76 (95% CI 1.34–2.31).
FIGURE 3.
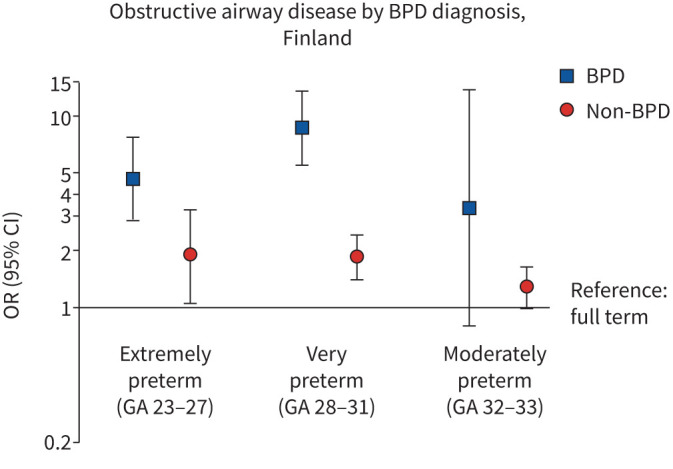
Associations between gestational age (GA) in weeks and obstructive airway disease (asthma or chronic obstructive pulmonary disease) at age 18–29 years in children born preterm with or without a diagnosis of bronchopulmonary dysplasia (BPD). Models are adjusted for sex and birth year.
Being born small for gestational age
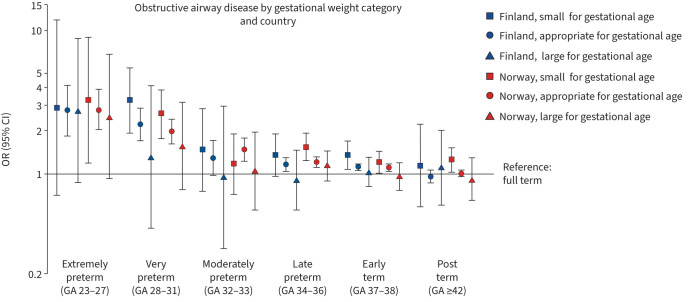
In total, 4.0% of the participants were born SGA and 3.0% LGA. In Norway, individuals born extremely, very and late preterm and early term, and in Finland those born very preterm or early term had higher odds for obstructive airway disease compared to those born full term, in both SGA and AGA categories (figure 4). However, individuals born SGA in preterm groups had somewhat higher odds than those born AGA. For example, the fully adjusted OR was 2.36 (95% CI 1.58–3.52) in the SGA very preterm group and 1.92 (95% CI 1.57–2.34) in the AGA very preterm group in Norway. In Finland, the corresponding ORs were 2.80 (95% CI 1.64–4.78) in the SGA very preterm group and 2.22 (95% CI 1.71–2.88) in the AGA very preterm group. In general, no significant associations between GA and obstructive airway disease in the LGA groups were found although the point estimates were somewhat lower than in the AGA groups.
FIGURE 4.
Associations between gestational age (GA) in weeks and obstructive airway disease (asthma or chronic obstructive pulmonary disease) at age 18–29 years in different birth weight groups. Models adjusted for sex and birth year.
Sensitivity analyses
When GA was studied week by week, the highest odds were observed among individuals born before 28 weeks, whereafter the odds decreased gradually (supplementary figure S3). Restricting the outcomes to care episodes from a pulmonology speciality (supplementary table S4) or from inpatient care (supplementary table S5) resulted in slightly higher odds compared to using all episodes. However, in Finland where the number of inpatient episodes was much smaller, only individuals born very preterm had higher odds than those born full term using model 1. There were no major differences between men and women in the associations of GA with the risk of obstructive airway diseases (supplementary figure S4) or between the first born or later born (supplementary figure S5). Excluding twins (Finland, n=17 679, 2.5%; Norway n=34 725, 2.1%) resulted in associations of similar magnitude as in the main analysis (supplementary table S6). In Finland, after adjusting model 2 additionally for maternal smoking, the odds for obstructive airway disease remained essentially similar as in the main analysis (supplementary table S7). The associations also remained similar when using Cox regression to account for survival time (supplementary table S8).
Discussion
In this two-country register study of over 2 million people, we found that preterm-born individuals had higher risks of care episodes due to asthma and/or COPD in young adulthood and middle age than those born full term. The risks at the lowest GAs were substantial, but the risks were observed across all GAs before full term in a dose-response manner. Risk remained higher over young to mid-adulthood, even after adjusting for parity, Caesarean section, twin birth, mother's age, mother's asthma and parents’ education. The risk was further increased in preterm-born individuals who were born SGA or who had a BPD diagnosis.
A key strength of our study derives from the large sets of individual-level data, with follow-up from birth to the age of 50 years. Near-complete information on births and diagnoses received in specialised healthcare, in both Finland and Norway, minimised the risk of bias from loss to follow-up. We were able to include many potential confounders by linking data from different registers. Large register data offered sufficient statistical power to study risk of obstructive airway disease across a wide spectrum of GAs.
An inherent limitation of a register study is that individual asthma and COPD diagnoses cannot be independently verified by assessing reversibility of obstructed airflow through clinical lung function measures. Nevertheless, asthma and COPD diagnoses in specialist care are typically based on thorough clinical history and paraclinical evaluations in the Nordic countries, and we evaluate any such misclassification as most probably nondifferential and thus not likely to substantially bias to the main findings. We have also shown that our results remained similar when we restricted the outcome only to care episodes from pulmonology or inpatient care. Importantly, we only included care episodes from specialist healthcare and had no data on primary care visits for the time period covered in our study. Thus, our results show the more severe end of the asthma and COPD spectrum, and not the total prevalence of the conditions. It is possible that people born extremely or very preterm could be more likely to be treated for respiratory symptoms in specialist care, e.g. because of other chronic conditions. However, we believe that in adulthood this is unlikely to cause substantial bias [35]. In addition, pregnancy dating based on last menstrual period, which was a commonly used method in Norway up to the late 1980s, may have overestimated the length of gestation for some individuals in our dataset [29]. We would expect this to produce more conservative estimates; however, the associations of obstructive airway disease at age 30–50 years in Norway (born 1967–1987) were not weaker than at age 18–29 years (born 1979–1999). Based on previous research from Finland [36] and Norway [37], we expect preterm-born young adults to be less or as likely to smoke as term-born individuals. We were able to adjust for maternal smoking during pregnancy in Finnish data with negligible changes in the results. Nevertheless, it is possible that our findings have been influenced by residual confounding from unmeasured confounders. For example, register data offer only limited information on certain prenatal or early life factors (e.g. chorioamnionitis or respiratory tract infections), health behaviours and life-style factors (e.g. personal smoking and work-related exposure to dust particles) that could contribute to the associations.
Overall, the findings in the Finnish and Norwegian datasets were consistent across the subgroups. However, the individuals in our study were born in 1967–1999, during a period of change in clinical care practices for preterm-born neonates [38], which has to be taken into account when interpreting the results. Some of the individuals, mainly from Norway, were born during the pre-surfactant era (pre-1990) [18, 38], whereas the individuals for whom we had information on BPD diagnosis were born in Finland in 1987–1998. Most of the BPD is likely to be the “new” BPD, which is considered a consequence of disrupted lung development, whereas the share of “classic” BPD caused by aggressive ventilation strategies was probably higher in the Norwegian sample for which we have no information on BPD diagnoses [6]. The results may thus not be generalisable to preterm-born individuals who received modern prenatal and neonatal care, individuals in other settings with different healthcare systems or with different measurement of duration of gestation.
Our findings on the association of GA and adult asthma are in line with previous research showing higher risk in young adulthood for those born preterm, compared to term-born individuals [8, 39]. We were also able to expand this investigation up to 50 years of age. Unlike previous research from Denmark, which reported higher risk among individuals born preterm for asthma medication purchases in childhood but not in young adulthood [13], we also found increased risk of asthma treated in specialist healthcare in middle-age, with a dose-response pattern in young adulthood similar to that observed by others [39].
Few studies have investigated COPD or obstructive airway diseases combined. In two Swedish studies, risk of obstructive airway disease was either similar [14] or higher only for women born preterm compared to full term [17]. In an Australian study, the risk of COPD at age 53 years was higher for individuals born at GA 28–33 weeks, but not for those born at 34–36 weeks [10]. We found higher risk for asthma and COPD combined, and COPD alone, in all groups born preterm, and both for men and women. The differences in the results may be due to the smaller sample size; differences in defining the outcome or the reference group; and regional, older cohorts with fewer extremely and very preterm survivors in the earlier studies. Our results support the hypothesis that preterm birth could increase the risk of COPD in adulthood, potentially via mechanisms relating to impaired lung development and function among preterm-born children, young people and adults [4, 9, 18, 40].
Earlier research has found that airway obstruction is more marked in extremely preterm-born individuals with a history of BPD [41, 42]. We can extend this finding to include very preterm-born individuals with BPD, who in our study also had a markedly higher risk for a diagnosis of obstructive pulmonary disease in young adulthood compared to very preterm-born without BPD. We were also able to investigate the combined risk of being born both preterm and SGA and found that SGA was associated with additionally higher risk for obstructive airway disease in some of the preterm categories.
Conclusion
Adults born preterm are more often treated in specialist care for obstructive airway diseases, including both asthma and COPD, compared to individuals born full term. The higher risk is extended to those born late preterm and early term and is higher for those who received a BPD diagnosis in infancy or who were born SGA. Preterm birth should be recognised as a risk factor for obstructive airway diseases in adulthood, and full medical history for people presenting with respiratory symptoms should include key perinatal data such as birth weight, gestational age and key pregnancy conditions. High-risk groups of preterm-born individuals, such as extremely or very preterm or those with a history of BPD, could benefit from more systematic follow-up with specialised teams who have knowledge on risks, possible prevention strategies, early diagnosis and treatment. In these groups, the excess risks are clinically significant and call for particular diagnostic vigilance when individuals born preterm present with respiratory symptoms.
Supplementary material
Please note: supplementary material is not edited by the Editorial Office, and is uploaded as it has been supplied by the author.
Supplementary material ERJ-01763-2022.Supplement (676.1KB, pdf)
Shareable PDF
Footnotes
This article has an editorial commentary: https://doi.org/10.1183/13993003.00662-2023
Conflict of interest: All authors have nothing to disclose.
Support statement: This project has received funding from the European Union's Horizon 2020 research and innovation programme under grant agreement number 733280 for RECAP. Funding also comes from the NORFACE Joint Research Programme on Dynamics of Inequality Across the Life-course, which is co-funded by the European Commission through Horizon 2020 under grant agreement number 724363, Academy of Finland (315690), European Union Horizon 2020 (874739, LongITools), the European Union Horizon Europe (101057739, TRIGGER), Sigrid Juselius Foundation, Foundation for Pediatric Research, Finnish Foundation for Cardiovascular Research, Diabetes Research Foundation, Novo Nordisk Foundation, Finska Läkaresällskapet, Yrjö Jahnsson Foundation, the joint research committee (FFU) between St. Olavs Hospital HF and the Faculty of Medicine NTNU (30223-2021) and the Norwegian Research Council (295989). Funders had no role in the design of the study, analysis of the data, reporting of the results or the decision to submit the manuscript for publication. Funding information for this article has been deposited with the Crossref Funder Registry.
References
- 1.Chawanpaiboon S, Vogel JP, Moller A, et al. Global, regional, and national estimates of levels of preterm birth in 2014: a systematic review and modelling analysis. Lancet Glob Health 2019; 7: e37–e46. doi: 10.1016/S2214-109X(18)30451-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.McGrath-Morrow SA, Collaco JM. Bronchopulmonary dysplasia: what are its links to COPD? Ther Adv Respir Dis 2019; 13: 1753466619892492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Carraro S, Scheltema N, Bont L, et al. Early-life origins of chronic respiratory diseases: understanding and promoting healthy ageing. Eur Respir J 2014; 44: 1682–1696. doi: 10.1183/09031936.00084114 [DOI] [PubMed] [Google Scholar]
- 4.Satrell E, Clemm H, Røksund OD, et al. Development of lung diffusion to adulthood following extremely preterm birth. Eur Respir J 2022; 59: 2004103. doi: 10.1183/13993003.04103-2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bolton CE, Bush A, Hurst JR, et al. Lung consequences in adults born prematurely. Thorax 2015; 70: 574–580. doi: 10.1136/thoraxjnl-2014-206590 [DOI] [PubMed] [Google Scholar]
- 6.Gough A, Spence D, Linden M, et al. General and respiratory health outcomes in adult survivors of bronchopulmonary dysplasia: a systematic review. Chest 2012; 141: 1554–1567. doi: 10.1378/chest.11-1306 [DOI] [PubMed] [Google Scholar]
- 7.GBD Chronic Respiratory Disease Collaborators . Prevalence and attributable health burden of chronic respiratory diseases, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet Respir Med 2020; 8: 585–596. doi: 10.1016/S2213-2600(20)30105-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Jaakkola JJK, Ahmed P, Ieromnimon A, et al. Preterm delivery and asthma: a systematic review and meta-analysis. J Allergy Clin Immunol 2006; 118: 823–830. doi: 10.1016/j.jaci.2006.06.043 [DOI] [PubMed] [Google Scholar]
- 9.Postma DS, Bush A, van den Berge M. Risk factors and early origins of chronic obstructive pulmonary disease. Lancet 2015; 385: 899–909. doi: 10.1016/S0140-6736(14)60446-3 [DOI] [PubMed] [Google Scholar]
- 10.Bui DS, Perret JL, Walters EH, et al. Association between very to moderate preterm births, lung function deficits, and COPD at age 53 years: analysis of a prospective cohort study. Lancet Respir Med 2022; 10: 478–484. doi: 10.1016/S2213-2600(21)00508-7 [DOI] [PubMed] [Google Scholar]
- 11.Bui DS, Walters HE, Burgess JA, et al. Childhood respiratory risk factor profiles and middle-age lung function: a prospective cohort study from the first to sixth decade. Ann Am Thorac Soc 2018; 15: 1057–1066. doi: 10.1513/AnnalsATS.201806-374OC [DOI] [PubMed] [Google Scholar]
- 12.Matheson MC, Olhaberriague AL D, Burgess JA, et al. Preterm birth and low birth weight continue to increase the risk of asthma from age 7 to 43. J Asthma 2017; 54: 616–623. doi: 10.1080/02770903.2016.1249284 [DOI] [PubMed] [Google Scholar]
- 13.Damgaard ALB, Gregersen R, Lange T, et al. The increased purchase of asthma medication for individuals born preterm seems to wane with age: a register-based longitudinal national cohort study. PLoS One 2018; 13: e0199884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Johansson CJ, Nilsson PM, Ignell C. Early life exposures and risk of adult respiratory disease during 50 years of follow-up. Eur J Epidemiol 2020; 35: 1157–1166. doi: 10.1007/s10654-020-00626-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Barker DJP, Osmond C, Forsén TJ, et al. Foetal and childhood growth and asthma in adult life. Acta Paediatr 2013; 102: 732–738. doi: 10.1111/apa.12257 [DOI] [PubMed] [Google Scholar]
- 16.Kotecha SJ, Dunstan FD, Kotecha S. Long term respiratory outcomes of late preterm-born infants. Semin Fetal Neonatal Med 2012; 17: 77–81. doi: 10.1016/j.siny.2012.01.004 [DOI] [PubMed] [Google Scholar]
- 17.Broström EB, Akre O, Katz-Salamon M, et al. Obstructive pulmonary disease in old age among individuals born preterm. Eur J Epidemiol 2013; 28: 79–85. doi: 10.1007/s10654-013-9761-7 [DOI] [PubMed] [Google Scholar]
- 18.Doyle LW, Andersson S, Bush A, et al. Expiratory airflow in late adolescence and early adulthood in individuals born very preterm or with very low birthweight compared with controls born at term or with normal birthweight: a meta-analysis of individual participant data. Lancet Respir Med 2019; 7: 677–686. doi: 10.1016/S2213-2600(18)30530-7 [DOI] [PubMed] [Google Scholar]
- 19.Risnes K, Bilsteen JF, Brown P, et al. Mortality among young adults born preterm and early term in 4 Nordic nations. JAMA Netw Open 2021; 4: e2032779. doi: 10.1001/jamanetworkopen.2020.32779 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Bakken IJ, Ariansen AMS, Knudsen GP, et al. The Norwegian Patient Registry and the Norwegian Registry for Primary Health Care: research potential of two nationwide health-care registries. Scand J Public Health 2020; 48: 49–55. doi: 10.1177/1403494819859737 [DOI] [PubMed] [Google Scholar]
- 21.Sund R. Quality of the Finnish Hospital Discharge Register: a systematic review. Scand J Public Health 2012; 40: 505–515. doi: 10.1177/1403494812456637 [DOI] [PubMed] [Google Scholar]
- 22.Laugesen K, Ludvigsson JF, Schmidt M, et al. Nordic health registry-based research: a review of health care systems and key registries. Clin Epidemiol 2021; 13: 533–554. doi: 10.2147/CLEP.S314959 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Gissler M, Teperi J, Hemminki E, et al. Data quality after restructuring a national medical registry. Scand J Soc Med 1995; 23: 75–80. doi: 10.1177/140349489502300113 [DOI] [PubMed] [Google Scholar]
- 24.Langhoff-Roos J, Krebs L, Klungsøyr K, et al. The Nordic medical birth registers–a potential goldmine for clinical research. Acta Obstet Gynecol Scand 2014; 93: 132–137. doi: 10.1111/aogs.12302 [DOI] [PubMed] [Google Scholar]
- 25.Sipola-Leppänen M, Vääräsmäki M, Tikanmäki M, et al. Cardiovascular risk factors in adolescents born preterm. Pediatrics 2014; 134: 1072. doi: 10.1542/peds.2013-4186 [DOI] [PubMed] [Google Scholar]
- 26.Helsedata. Variables. 2022. https://helsedata.no/en/variables/ Date last accessed: 26 April 2022. [Google Scholar]
- 27.Korhonen P, Haataja P, Ojala R, et al. Asthma and atopic dermatitis after early-, late-, and post-term birth. Pediatr Pulmonol 2018; 53: 269–277. doi: 10.1002/ppul.23942 [DOI] [PubMed] [Google Scholar]
- 28.Inghammar M, Engström G, Löfdahl CG, et al. Validation of a COPD diagnosis from the Swedish Inpatient Registry. Scand J Public Health 2012; 40: 773–776. doi: 10.1177/1403494812463172 [DOI] [PubMed] [Google Scholar]
- 29.Vogel JP, Chawanpaiboon S, Moller A, et al. The global epidemiology of preterm birth. Best Pract Res Clin Obstet Gynaecol 2018; 52: 3–12. doi: 10.1016/j.bpobgyn.2018.04.003 [DOI] [PubMed] [Google Scholar]
- 30.Goldenberg RL, Culhane JF, Iams JD, et al. Epidemiology and causes of preterm birth. Lancet 2008; 371: 75–84. doi: 10.1016/S0140-6736(08)60074-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Beasley R, Semprini A, Mitchell EA. Risk factors for asthma: is prevention possible? Lancet 2015; 386: 1075–1085. doi: 10.1016/S0140-6736(15)00156-7 [DOI] [PubMed] [Google Scholar]
- 32.Adeloye D, Song P, Zhu Y, et al. Global, regional, and national prevalence of, and risk factors for, chronic obstructive pulmonary disease (COPD) in 2019: a systematic review and modelling analysis. Lancet Respir Med 2022; 10: 447–458. doi: 10.1016/S2213-2600(21)00511-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Sankilampi U, Hannila M, Saari A, et al. New population-based references for birth weight, length, and head circumference in singletons and twins from 23 to 43 gestation weeks. Ann Med 2013; 45: 446–454. doi: 10.3109/07853890.2013.803739 [DOI] [PubMed] [Google Scholar]
- 34.Zeger SL, Liang KY. Longitudinal data analysis for discrete and continuous outcomes. Biometrics 1986; 42: 121–130. doi: 10.2307/2531248 [DOI] [PubMed] [Google Scholar]
- 35.Raju TNK, Buist AS, Blaisdell CJ, et al. Adults born preterm: a review of general health and system-specific outcomes. Acta Paediatr 2017; 106: 1409–1437. doi: 10.1111/apa.13880 [DOI] [PubMed] [Google Scholar]
- 36.Strang-Karlsson S, Räikkönen K, Pesonen A, et al. Very low birth weight and behavioral symptoms of attention deficit hyperactivity disorder in young adulthood: the Helsinki study of very-low-birth-weight adults. Am J Psychiatry 2008; 165: 1345–1353. doi: 10.1176/appi.ajp.2008.08010085 [DOI] [PubMed] [Google Scholar]
- 37.Husby IM, Stray KM, Olsen A, et al. Long-term follow-up of mental health, health-related quality of life and associations with motor skills in young adults born preterm with very low birth weight. Health Qual Life Outcomes 2016; 14: 56. doi: 10.1186/s12955-016-0458-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Lehtonen L. Treatment of premature infant is changing [Keskosen muuttuva hoito]. Duodecim 2009; 125: 1333–1339. [PubMed] [Google Scholar]
- 39.Trønnes H, Wilcox AJ, Lie RT, et al. The association of preterm birth with severe asthma and atopic dermatitis: a national cohort study. Pediatr Allergy Immunol 2013; 24: 782–787. doi: 10.1111/pai.12170 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Näsänen-Gilmore P, Sipola-Leppänen M, Tikanmäki M, et al. Lung function in adults born preterm. PloS One 2018; 13: e0205979. doi: 10.1371/journal.pone.0205979 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Vollsæter M, Røksund OD, Eide GE, et al. Lung function after preterm birth: development from mid-childhood to adulthood. Thorax 2013; 68: 767–776. doi: 10.1136/thoraxjnl-2012-202980 [DOI] [PubMed] [Google Scholar]
- 42.Gough A, Linden M, Spence D, et al. Impaired lung function and health status in adult survivors of bronchopulmonary dysplasia. Eur Respir J 2014; 43: 808–816. doi: 10.1183/09031936.00039513 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Please note: supplementary material is not edited by the Editorial Office, and is uploaded as it has been supplied by the author.
Supplementary material ERJ-01763-2022.Supplement (676.1KB, pdf)
This one-page PDF can be shared freely online.
Shareable PDF ERJ-01763-2022.Shareable (736.5KB, pdf)