Abstract
Purpose:
To examine the frequency and risk factors for ocular pain after laser assisted in situ keratomileusis (LASIK) and photorefractive keratectomy (PRK).
Methods:
Prospective study of individuals undergoing refractive surgery at two different centers. Participants rated their ocular pain on a 0 – 10 Numerical Rating Scale (NRS) pre-surgery and 1 day, 3 months, and 6 months post-surgery. A clinical examination focused on ocular surface health was performed 3- and 6-months post-surgery. Persistent ocular pain was defined as an NRS score ≥ 3 at both 3- and 6-months post-surgery (cases) and this group was compared to individuals with NRS scores <3 at both time points (controls).
Results:
109 individuals underwent refractive surgery (87% LASIK; 13% PRK) and were followed for 6 months after surgery. Mean age was 34±8 years (range 23 to 57); 62% self-identified as female, 81% as White, 33% as Hispanic. Eight (7%) individuals reported ocular pain (NRS ≥ 3) prior to surgery, with the frequency of ocular pain increasing after surgery to 23% (n=25) at 3 months and 24% (n=26) at 6 months. Twelve individuals (11%) reported an NRS ≥ 3 at both time points and comprised the persistent pain group. Factors that predicted persistent pain after surgery in a multivariable analysis were: (a) ocular pain pre-surgery (b) symptom report of depression prior to surgery (Patient Health Questionnaire-9: PHQ9: odds ratio (OR) 1.3, 95% confidence interval (CI) 1.1–1.6, p=0.01); (c) use of an oral anti-allergy medication pre-surgery (OR 13.6, 95% CI 2.1–89.3, p=0.007). (d) pain intensity day 1 after surgery (OR: 1.6, 95% CI 1.2–2.2, p=0.005); Ocular surface signs of tear dysfunction were not related to pain complaints. Most individuals (>90%) were completely or somewhat satisfied with their vision at 3 and 6 months.
Conclusions:
Eleven percent of individuals reported persistent ocular pain after refractive surgery, with several pre- and peri-operative factors predicting pain after surgery.
Precis
Twelve of 109 individuals (11%) reported persistent pain after refractive surgery, defined as a Numerical Rating Scale score≥3 (range 0–10) at 3- and 6-months post-surgery. Factors that predicted persistent pain were (a) ocular pain pre-surgery; (b) depressive symptoms pre-surgery; (c) use of an oral anti-allergy medication pre-surgery; and (d) ocular pain 24 hours after surgery.
Introduction
Laser in-situ keratomileusis (LASIK) and photorefractive keratectomy (PRK) are commonly performed procedures used to correct refractive error. While patient outcomes and satisfaction are typically excellent, a potential side effect is the occurrence of unpleasant eye sensations after surgery.1 These sensations were initially labeled as ‘dry eye’ because ‘dryness’ was a commonly reported sensation. However, over time, these sensations were re-characterized as pain, given that other descriptors such as ‘burning’, ‘aching’, and ‘tenderness’ were also used2, and that patient symptoms were often discordant from dry eye (DE) signs, such as tear production and stability.1 One prospective study examined symptoms pre- and post-LASIK3 using the Ocular Surface Disease Index (OSDI), a multifaceted questionnaire that captures information on pain, vision, symptom triggers, and visual function.4 In active-duty Navy personnel, some individuals with low OSDI scores pre-surgery (OSDI≤12) had higher scores after surgery (OSDI>12: 27%, 33 of 121). On the other hand, many individuals reported an improvement in symptoms after surgery, with 59% (60 of 101) having an OSDI score >12 pre-surgery which decreased to ≤12 after surgery.3 A similar pattern was noted in a civilian population, with some individuals noting an increase, and others a decrease, in OSDI scores after refractive surgery.3 Overall, the frequency of ocular symptoms after refractive surgery has been estimated at 20–55% in studies that used different metrics to capture symptoms and had variable study designs and follow up times (Table 1).5–11 A limitation of the available studies is that ocular pain was not specifically examined, and it is thus not possible to separate pain complaints from the other eye symptoms that are captured in many standard clinical questionnaires, such as visual disturbances and tearing.
Table 1.
Summary of Some Prior Studies of Ocular Symptoms after Refractive Surgery
| Study | N | Procedure | Metric used to capture symptoms | Population, age | Study design | Outcome |
|---|---|---|---|---|---|---|
| Gong et al. 202212 | 78 | LASIK (n=24) SMILE (n=19) PRK (n=35) |
OSDI (high score=more severe symptoms) | Chinese, mean 23.92 ± 4.65 years | P | OSDI scores ↑ 1-month after surgery, returned to pre-surgery levels after 6 months. |
| Eydelman et al. 20173 | 242 | LASIK | OSDI ≤ 12 no symptoms >12 symptoms |
Navy personnel, mean 29.1 ± 6.2 years | P | 27% (33/121) without symptoms pre-surgery reported symptoms 3 months after surgery; 19.5% (23/118) reported symptoms at 6 months. 59% (60/101) with symptoms pre-surgery had no symptoms 3 months post-surgery |
| Eydelman et al. 20173 | 292 | LASIK | OSDI ≤ 12 no symptoms >12 symptoms |
US civilians, mean 31.5 ± 7.3 years | P | 27.5% (30/109) without symptoms pre-surgery reported symptoms 3 months after surgery; 59% (87/147) with symptoms pre-surgery had no symptoms 3 months post-surgery |
| Bower et al. 201513 | 143 | LASIK (n=70) PRK (n=73) |
McMonnies Dry Eye Questionnaire | US Army personnel, mean 29.9 ± 5.2 years | P | McMonnies score ↑ 3, 6, and 12-months post-surgery compared to pre-surgery |
| Shoja et al. 20075 | 190 | LASIK | DE as defined by symptoms (soreness, scratchiness, dryness, grittiness, burning) + TBUT<10 + corneal staining > 3/5 + Schirmer <10mm | Iranians, mean 31 ± 8 years | R | 20% (38/190) had DE ≥ 6 months post-LASIK |
| Tuisku et al. 20078 | 30 | LASIK (n=20) Controls (n=10) |
OSDI | Mean 35.9 ± 8.8 years | R | OSDI higher 44.2 ± 11.3 months after surgery vs. controls (18.6 ± 13.4 vs 7.5 ± 5.7) |
| Donnenfeld et al. 200310 | 52 | LASIK | Subjective rating (less dry, more dry, same as before LASIK) | Mean 40.1 years | P | 31% (16/52) reported eyes felt drier at 6 months vs. before LASIK |
| Albietz et al. 20027 | 88 | LASIK | Any symptom (sore, scratchy, dry, gritty, burning) sometimes, often, or constantly. | Mean 49 ± 9 years | R | 14% (12/88) with symptoms pre- surgery; 32% (22/88) with symptoms ≥ 6 months post-surgery |
| Hovanesian et al. 20016 | 828 | LASIK (n=587) PRK (n=241) |
Eye dryness rated as yes/no, sharp pains rated as yes/no | No demographic data | R | 41% (99/241) PRK and 45% (266/587) LASIK reported dryness, 20% (49/241) PRK and 8% (48/587) LASIK reported sharp pain ≥ 6m after surgery |
LASIK= Laser in situ keratomileusis; PRK= Photorefractive keratectomy; OSDI= Ocular surface disease index; DE= Dry eye; TBUT= tear break up time; R= retrospective; P=prospective; US=United States; vs.=versus
There are many potential causes for the occurrence of ocular pain after refractive surgery, including nociceptive and neuropathic etiologies. Tear film abnormalities (e.g., tear instability, corneal epithelial disruption) have been noted after refractive surgery, and are potential sources of nociceptive pain.5 Corneal nerve damage also occurs at the time of refractive surgery and studies have demonstrated that nerve density and sensitivity often do not return to baseline levels for years.14 Maladaptive healing of nerves may contribute to the development of neuropathic pain in some individuals, as seen in other forms of persistent post-operative pain (PPOP).1, 15
Despite recognition that ocular pain is a potential side effect of refractive surgery, no prospective studies have examined the frequency and risk factors for its development. Furthermore, retrospective studies that have examined chronic pain after refractive surgery have limitations with regards to pain definition and potential biases (e.g., recall, referral). One study identified 18 individuals of 16,000 who developed severe ocular pain (not further defined) after LASIK, with a mean time to pain onset of 9.6 months.16 Yet, another study of 50 individuals with pain after refractive surgery (recruited from a corneal neuralgia Facebook group) found that pain started within 1 month of surgery in 46% of individuals.17 To bridge knowledge gaps, we prospectively evaluated individuals pre- and post-refractive surgery with a formal protocol to examine the frequency, quality, and risk factors for developing persistent ocular pain.
Methods
Study population:
The Institutional Review Boards of the University of Miami and Oregon Health & Science University (OHSU) approved this prospective study, the methods adhered to the tenets of the Declaration of Helsinki, and all patients signed an informed consent form prior to participation. Subjects from both sites who elected to undergo refractive surgery in both eyes and who were on stable ocular and systemic medication for at least 3 months prior to surgery were eligible for inclusion in this study. Exclusion criteria included pregnancy, prior eye surgery, eye diseases that could confound ocular pain (glaucoma, herpetic eye disease), anatomic abnormalities of the eyelids, conjunctiva, or cornea, and age<18 years. Of the 127 individuals enrolled into the study, 18 did not complete their 6-month visit and were considered lost to follow up, leaving 109 individuals included for analysis.
Data collected:
At the baseline pre-surgery visit, individuals filled out questionnaires that captured demographics, co-morbidities (e.g., diabetes, hypertension, sleep apnea, migraine, thyroid problems, allergies (ocular and non-ocular), chronic pain conditions (e.g., fibromyalgia, temporomandibular joint disorder, trigeminal neuralgia, arthritis, low back pain)), medications, and eye symptoms. In addition, an anesthetized Schirmer test was performed. One day after surgery, individuals filled out questionnaires on eye symptoms since surgery. At the 3- and 6-month visits, individuals again filled out questionnaires and underwent a full ocular surface examination.
DE symptoms:
Two validated DE questionnaires, the 5 Item Dry Eye Questionnaire (DEQ5)18 and the Ocular Surface Disease Index (OSDI)4 were administered at baseline and at 3 and 6 months after surgery.
Ocular pain:
A numerical rating scale (NRS) (range 0–10) was used to assess pain intensity at all time points (pre-surgery, 1 day after surgery, 3 months, 6 months). The NRS was chosen as it is a validated pain measure that has been used across multiple studies19 and has been recommended as an outcome measure for clinical trials involving chronic pain by the International Association for the Study of Pain.20 One day after surgery, individuals were asked to rate their worst eye pain intensity since surgery. At all other time points, individuals were asked to rate the intensity of their worst eye pain over the last week. The Neuropathic Pain Symptom Inventory modified for the Eye (NPSI-Eye)21 was administrated pre-operatively and at 3- and 6-months post-surgery. The NPSI was developed to evaluate symptoms of neuropathic pain and was subsequently modified and validated for evaluation of eye pain.21 The NPSI-Eye interrogates eye pain characteristics over 5 dimensions (burning pain, paroxysmal pain, pressing pain, evoked pain, and paresthesia/dysesthesia), with individual scores (0–10) and a total score (0–100) generated.
Visual acuity:
Uncorrected and best-corrected visual acuity were obtained at each visit.
Ocular surface examination:
At the 3- and 6-month visits, ocular surface assessments included (in the order performed): (a) examination of the eyelid margin and grading of anterior blepharitis, eyelid vascularity (0=none, 1=mild, 2=moderate, 3=severe), and inferior eyelid Meibomian orifice plugging (0=none; 1=less than 1/3, 2=between 1/3 and 2/3, 3=greater than 2/3 of eyelid glands with visible plugs, graded without eyelid manipulation); (b) tear film break up time (TBUT) (average of 3 measures in each eye); (c) corneal staining assessed using the National Eye Institute (NEI) scale22; (d) pain assessment (0–10 NRS) pre- and 30 seconds post- placement of topical anesthesia; (e) anesthetized Schirmer test.
Patient Satisfaction:
At the 3- and 6-month follow up visits, subjects were asked to rate satisfaction with their vision on a five-point scale. Answer options included: completely satisfied, somewhat satisfied, neither, somewhat dissatisfied, completely dissatisfied.
Assessed co-morbidities:
At baseline, individuals filled out questionnaires regarding (a) demographics; (b) medications; (c) co-morbidities; (d) non-ocular pain via a pain history questionnaire; (e) depression via the Patient Health Questionnaire 9.23 In addition, details of the surgical procedure were recorded including treatment parameters and flap thickness.
Main outcome measure:
Individuals with persistent ocular pain after refractive surgery, defined as an NRS ≥3 (rated at its worst intensity over the past week) at both the 3 and 6 months timepoints (cases). This cut-off value was chosen based on prior work that defined an NRS of 3 as moderate pain.24
Statistical analysis:
This is an interim analysis of a dataset generated from an ongoing study whose main aim is to identify diagnostic and prognostic tear biomarkers of persistent pain after refractive surgery. In this analysis, we studied the data generated from about the first half of the target sample of ~200 individuals. All statistical analyses were performed using SPSS 26.0. Descriptive statistics were used to summarize baseline and outcome variables. Paired methodologies (t-test and McNemar, as appropriate) were used to evaluate change over time within an individual. Tests for Independent samples (t test and Chi square or Fisher exact, as appropriate) were used to examine differences between our two main groups (cases: individuals with persistent ocular pain after surgery; controls: individuals without persistent pain defined as an NRS <3 at both the 3- and 6-month time points). Multivariable logistic and linear regression analyses were performed to examine factors associated with pain after surgery after inspection of residuals. Receiver operating characteristic (ROC) curves were built based on factors that significantly predicted persistent pain on univariable analyses. Given the preliminary nature of the study, we opted to report all examined variables, with accompanying confidence intervals, and did not adjust p-values for multiple comparisons, given potential limitations with this approach.25
Results
Study population:
109 individuals (51 at the Miami site and 58 at the OHSU site) were enrolled into the study between April 2021 and March 2022 and followed for 6 months post-surgery (Figure 1). The mean age of the population was 34±8 years (range 23–57), 62% self-identified as female, 81% as White, and 33% as Hispanic. The cohort was overall healthy with 6 reporting hypertension, 5 sleep apnea, 5 arthritis (osteoarthritis n=3, rheumatoid arthritis n=2), and 4 thyroid disease (not further sub-typed). Twenty three percent (n=25) of individuals reported mild or greater depression symptoms (PHQ9 ≥5) pre-surgery, with most symptoms falling in the mild range (PHQ9 5 to 9, n=20). Most individuals wore contact lenses prior to surgery (68%, n=74), and 17% (n=18) reported a history of eye allergies. All individuals underwent bilateral eye surgery with LASIK (87%, n=95) being more common than PRK (13%, n=14).
Figure 1:
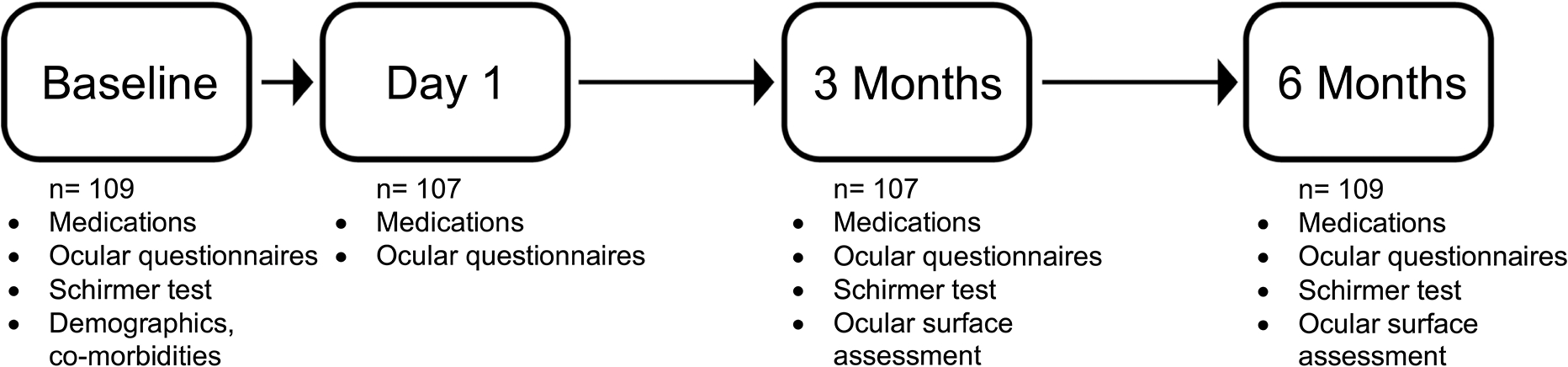
Study timepoints and assessments.
Frequency of ocular pain and DE symptoms prior to refractive surgery.
Prior to surgery, eight individuals (7%, n=8) rated their worst ocular pain as ≥3 on NRS. Two individuals (2%) had a DEQ5 score ≥12 prior to surgery, representing severe symptoms on a DE-specific questionnaire. When examined across all individuals, ocular pain scores (NRS) correlated with DE symptom scores (DEQ5: r=0.38, p<0.001; OSDI: r=0.26, p=0.006), highlighting the overlap in symptoms between pain-specific and dry-eye questionnaires.
Frequency of ocular pain after refractive surgery.
One day post-surgery, most individuals (72%, n=84, 2 individuals with missing responses) rated their worst post-operative pain as ≥3 on a 10-point NRS. There was no significant difference in acute pain at one day post-surgery when assessed by surgery type (LASIK: 5.8±3.1, PRK: 4.5±3.0, p=0.14). The frequency of ocular pain (NRS≥3) at 3 months (23%, n=25, 2 individuals with missing responses) and 6 months (24%, n=26) post-surgery was higher than pre-surgery. Overall, pain scores were greater at 3 and 6-months compared to baseline (p=0.001 for both) but were not significantly different between the 3- and 6-month time points (p=0.90) (Figure 2).
Figure 2: Ocular pain score frequencies at the pre- and post-surgery timepoints demonstrating an overall increase in pain scores at the 3- and 6-month visits compared to pre-surgery.

Numerical Rating Scale (NRS). A) Pre-surgery; B) Day 1 Post-surgery; C) Month 3 Post-surgery; D) Month 6 Post-surgery
Twelve individuals (11.0%, 11 LASIK, 1 PRK) reported an NRS ≥ 3 at both the 3- and 6-month time points and were defined as having persistent pain after refractive surgery (cases). Sixty-eight individuals (64%) had NRS<3 at both 3 and 6 months and were defined as having no persistent pain (controls). Twenty-seven individuals (25%) had an NRS ≥ 3 at one time point post-surgery but not both (n=13 at 3 months; n=14 at six months). When examining the entire study cohort, heterogeneity was noted with regards to pain intensity over time, as noted in Figure 3. Some individuals in all groups (persistent pain, no persistent pain, NRS>3 at 3 or 6 months after surgery but not both time points) had pain (NRS>3) prior to surgery and/or at one day post-surgery. In addition, some individuals in the case group had higher pain levels at 3 compared to 6 months while others had higher pain levels at 6 compared to 3 months Figure 3.
Figure 3: Individual pain intensity ratings demonstrate heterogeneity with regards to pain intensity pre-surgery and day 1 post-surgery in all three groups.

(A) 68 individuals without persistent pain (controls: NRS<3 at both 3 and 6 months); (B) 12 individuals with persistent pain (cases: NRS≥3 at 3 and 6 months); and (C) 27 individuals with pain at one, but not both, time points (3 or 6 months). Each individual line is shown in an arbitrary color to allow visual tracking of each individual over time.
Artificial tears use increased after surgery, with 13% (n=14) of individuals reporting use prior to surgery and 73% (n=79) and 65% (n=71) reporting use at 3- and 6-months post-surgery, respectively. However, it is noteworthy that use of artificial tears is often recommended by surgeons during the post-operative period.
Characteristics of ocular pain after surgery.
Characteristics of pain were extracted by examining responses to individual questions within the various pain and DE specific questionnaires. As with the pre-operative findings, NRS pain scores and symptom scores on DE questionnaires were correlated at both 3- and 6- month time points (3 months: DEQ5: r=0.68, OSDI: r=0.40; p<0.001 for both; 6 months: DEQ5: r=0.58, OSDI: r=0.62; p<0.001 for both).
At 6 months, the most common pain descriptors (via OSDI and NPSI-Eye) in the 12 individuals with persistent pain after surgery were “soreness” (n=7), “grittiness” (n=6), “burning” (n=3) and “pressure” (n=2). Three individuals reported that their eye pain increased with exposure to light, and five reported increased pain with exposure to wind. Ten individuals (n=10 of 12; 83%) reported complete resolution of pain after topical anesthesia placement, with two (n=2 of 12; 17%) reporting persistent pain in at least one eye after topical anesthesia.
Risk factors for persistent ocular pain after refractive surgery.
Several baseline risk factors were identified when comparing the 12 individuals with an NRS ≥ 3 at both the 3- and 6-month time points (cases) and the 68 individuals with NRS<3 at both time points (controls). On univariable analysis, the factors that increased the risk for persistent ocular pain after surgery were: (a) the presence of ocular pain prior to surgery, assessed with both pain and DE questionnaires and (b) rating of pain on Day 1 post-surgery (Table 2).
Table 2:
Demographics, co-morbidities, and ocular surface metrics of cases with persistent post-operative pain compared to controls without persistent pain. P values are indicated for univariable analyses of each factor.
| Persistent pain (n=12) | No persistent pain (n=68) | P value | |
|---|---|---|---|
|
| |||
| Demographics | |||
|
| |||
| Age, mean years±SD | 33.3±7.7 | 33.6±7.6 | 0.90 |
|
| |||
| Gender, % female (n) | 67% (8) | 59% (40) | 0.75 |
|
| |||
| Race, % White (n) | 58% (7) | 78% (53) | 0.07 |
| % Black (n) | 0% (0) | 6% (4) | |
| % Asian (n) | 17% (2) | 12% (8) | |
| % Other (n) | 25% (3) | 4% (3) | |
|
| |||
| Ethnicity, % Hispanic (n) | 42% (5) | 32% (22) | 0.53 |
|
| |||
| Smoker, % current (n) | 8% (1) | 15% (10) | 1.00 |
|
| |||
| Location, % Miami (n) | 58% (7) | 43% (30) | 0.34 |
|
| |||
| Co-morbidities | |||
|
| |||
| Hypertension, % (n) | 0% (0) | 6% (4) | 1.00 |
|
| |||
| Sleep apnea using CPAP, % (n) | 0% (0) | 6% (4) | 1.00 |
|
| |||
| Migraine or headache, % (n) | 17% (2) | 16% (11) | 1.00 |
|
| |||
| Non-ocular pain conditiona, % (n) | 33% (4) | 35% (24) | 1.00 |
|
| |||
| Depression, PHQ-9, mean±SD | 4.5±4.9 | 2.4±2.9 | 0.14 |
|
| |||
| Depression (PHQ-9≥5), % (n) | 33% (4) | 21% (14) | 0.45 |
|
| |||
| Self-reported non ocular allergies, % (n) | 42% (5) | 32% (22) | 0.53 |
|
| |||
| Oral medications | |||
|
| |||
| Antidepressant, % (n) | 17% (2) | 15% (10) | 1.00 |
|
| |||
| Anxiolytic, % (n) | 17% (2) | 12% (8) | 0.64 |
|
| |||
| Anti-allergy medication, % (n) | 33% (4) | 10% (7) | 0.06 |
|
| |||
| Pre-surgery considerations | |||
|
| |||
| DEQ5, mean±SD | 4.9±2.6 | 2.9±2.9 | 0.03 |
|
| |||
| OSDI, mean±SD | 8.3±6.5 | 5.0±7.4 | 0.16 |
|
| |||
| NRS worst pain over past week, mean±SD | 1.0±1.2 | 0.4±0.8 | 0.01 |
|
| |||
| NRS worst pain over past week ≥3, % (n) | 17% (2) | 3% (2) | 0.11 |
|
| |||
| NPSI-Eye, mean±SD | 1.6±2.8 | 0.8±1.8 | 0.43 |
|
| |||
| Schirmerc, mean mm at 5 minutes ±SD | 16.5±10.3 | 15.8±10.3 | 0.92 |
|
| |||
| Schirmerc <5 mm at 5 minutes, % (n) | 17% (2) | 12% (8) | 0.64 |
|
| |||
| Self-reported ocular allergies, % (n) | 33% (4) | 13% (9) | 0.10 |
|
| |||
| Contact lens wear, % (n) | 75% (9) | 75% (51) | 1.00 |
|
| |||
| Surgical considerations | |||
|
| |||
| LASIK, % (n) | 92% (11) | 90% (61) | 1.00 |
|
| |||
| Myopic ablation, % (n) | 100% (12) | 97% (66) | 1.00 |
|
| |||
| Spherical equivalent, more negative or positive value between eyes | |||
| Myopic, mean±SD | −3.5±1.3 | −4.3±1.7 | 0.11 |
| Hyperopic, mean±SD | N/A | +2.8±0.8 | NA |
|
| |||
| Flap depth, mean μm ±SD (in those who underwent LASIK) | 119±8.3 | 117±9.0 | 0.52 |
|
| |||
| NRS worst pain post-operative day 1, mean±SD | 6.3±2.4 | 4.0±3.0 | 0.02 |
|
| |||
| NRS≥3 post-operative day 1, % (n) | 92% (11) | 66% (45) | 0.10 |
|
| |||
| Ocular surface signs at 3 months | |||
|
| |||
| TBUTc, mean seconds ±SD | 6.8±1.9 | 8.0±3.3 | 0.31 |
|
| |||
| Corneal stainingb, mean ±SD | 1.6±2.2 | 1.6±2.2 | 0.95 |
|
| |||
| Schirmerc, mean mm at 5 minutes ±SD | 15.2±11.2 | 15.9±10.8 | 0.84 |
|
| |||
| Anterior blepharitisb, mean ±SD | 0.3±0.5 | 0.4±0.7 | 0.66 |
|
| |||
| Eyelid vascularityb, mean ±SD | 0.9±0.7 | 1.0±0.9 | 0.73 |
|
| |||
| Meibomian gland pluggingb, mean ±SD | 0.9±0.3 | 0.8±0.7 | 0.63 |
|
| |||
| Ocular surface signs at 6 months | |||
|
| |||
| TBUTc, mean seconds ±SD | 8.6±3.7 | 8.1±3.1 | 0.57 |
|
| |||
| Corneal stainingb, mean ±SD | 0.9±1.2 | 1.9±2.6 | 0.22 |
|
| |||
| Schirmerc, mean mm at 5 minutes ±SD | 15.7±10.2 | 14.5±9.9 | 0.71 |
|
| |||
| Anterior blepharitisb, mean ±SD | 0.6±0.7 | 0.4±0.7 | 0.38 |
|
| |||
| Eyelid vascularityb, mean ±SD | 0.8±0.6 | 0.9±0.9 | 0.71 |
|
| |||
| Meibomian gland pluggingb, mean ±SD | 1.0±0.6 | 1.0±0.6 | 0.48 |
Persistent post-operative pain defined as a Numerical Rating Scale score of worst pain intensity over a 1-week recall ≥3 at both 3- and 6-months post-surgery
SD=standard deviation; LASIK= laser assisted in situ keratomileusis; NRS=Numerical Rating Scale; DEQ5=5 item Dry Eye Questionnaires; OSDI=Ocular Surface Disease Index; NPSI-Eye=Neuropathic Pain Symptoms Inventory-Modified for the Eye; PHQ=Patient Health Questionnaire; CPAP=continuous positive airway pressure; N/A not applicable
Includes a yes response to ever having any of the following conditions for >3 months: Headache, arthritis, fibromyalgia, temporomandibular disorder, trigeminal neuralgia, low back pain, muscle pain, sciatica, shingles, post-surgical pain, tendonitis, chronic fatigue, irritable bowel syndrome, interstitial cystitis
Higher value between the two eyes
Lower value between the two eyes
A multivariable forward stepwise logistic regression model was built to examine all factors with a p value<0.1526 in Table 2, in combination. In this model, pre-operative symptoms of depression (PHQ9: OR 1.3, 95% CI 1.1–1.6, p=0.01), pain intensity the day after surgery (OR: 1.6, 95% CI 1.2–2.2, p=0.005), and use of an oral anti-allergy medication pre-surgery (OR: 13.6, 95% CI 2.1–89.3, p=0.007) were predictive of persistent pain after surgery.
Next, the significant factors on uni- or multi-variable analysis (PHQ9, DEQ5, NRS assessed pre-surgery and day 1 post-surgery, and use of an oral anti-allergy pre-surgery) were combined and examined in receiver operating curve analysis, with a resulting area under the curve (AUC) of 0.90 (standard error 0.04, 95% CI 0.83–0.97), suggesting that these factors had a reasonable ability to discriminate between those with and without persistent pain.
Risk factors for pain at 3 months and 6 months after surgery.
We next examined which factors remained predictive of pain at each separate time point (3 and 6 months) with forward stepwise linear regression analyses. In each model, we considered all captured pre-operative variables, pain intensity 1-day post-surgery, and ocular surface signs 3 or 6 months after surgery. Both analyses resulted in similar findings, pain symptoms (captured with pain or DE specific questionnaires) prior to surgery (3 months: DEQ5, standardized β = 0.29, p=0.002; 6 months: NRS, standardized β = 0.20, p=0.04) and pain 1-day post-surgery (3 months: NRS, standardized β = 0.25, p=0.008; 6 months: NRS, standardized β = 0.21, p=0.04) were predictive of post-operative pain.
Visual outcomes and patient satisfaction.
92% of individuals studied for this interim analysis achieved uncorrected vision of 20/25 or better in both eyes at 3 months. Patient satisfaction was high with 93% and 91% of the entire population indicating that they were completely or somewhat satisfied with their vision in both eyes at 3 and 6 months, respectively. The frequency of individuals satisfied or somewhat satisfied with their procedure was similar between those with, versus without, persistent pain.
Discussion
In this interim analysis, we found that 11% of individuals reported persistent ocular pain after refractive surgery, defined as pain intensity of ≥3 on a 0–10 scale at both 3 and 6 months after surgery. The pain was not related to visual acuity or ocular surface signs of disease (e.g., tear production, stability), suggesting that neuropathic mechanisms may contribute to this pain. Topical anesthesia eliminated pain in 10 of 12 individuals, further suggesting peripheral mechanisms at the 3- and 6-month times points in the majority of individuals. Risk factors for persistent pain identified on uni- or multivariable analyses included: (a) ocular pain pre- surgery, (b) symptoms of depression pre-surgery, (c) oral anti-allergy medication use pre-surgery, and (d) ocular pain 1-day post-surgery. Importantly, none of our patients had debilitating symptoms that necessitated escalation of therapy beyond that typically used in the post-operative period (e.g., artificial tears).
Our findings are similar to reports of persistent post-operative pain (PPOP) after non-ocular surgical procedures. PPOP is generally defined as pain that develops after surgery, that is at least 3–6 months in duration (after sufficient time has passed for the healing of tissues disrupted by surgery), and that is not explained by another cause of pain (e.g., infection). PPOP often has a neuropathic quality (burning, shooting, electric-like), and occurs spontaneously, in the absence of nociceptor activation.27 The 11% estimate of PPOP in our study population is within the range reported for PPOP after other surgeries including: dental implants (8.5–36%)28, inguinal hernia repair (5–30%)29, thoracotomy (5–65%)29, and breast surgery (20–50%).29 Furthermore, some risk factors identified in our study for persistent ocular pain are shared with those from prior PPOP studies, including pre-existing pain, acute pain, and depression.30–36,35,36–40
Regarding pre-existing pain, pain 1 day prior to thoracotomy (assessed via a 0–10 NRS) increased the risk of post-operative pain 12 months after surgery (OR 7.0, 95% CI 2.4–20.2, n=106).34 With respect to acute pain, pain 1 hour after hysterectomy (assessed via 0–10 NRS) increased the risk of post-operative pain 3 months after surgery (r=0.16, p<0.05, n=200).37 Regarding depression, pre-operative depressive symptoms (assessed via the Hospital Anxiety and Depression Scale) increased the risk of post-operative pain 6 months after GI surgery (NRS≥1 at rest: OR 1.1; 95% CI 1.01–1.2, p = 0.03, n=274).38 Our study adds pain and depressive symptoms pre-surgery and acute pain post-surgery as risk factors for PPOP development after refractive surgery.
Specific to the eye, we previously reported that the presence of depression and ocular pain pre-surgery were risk factors for PPOP after LASIK. In a prospective study of 43 individuals undergoing LASIK, anxiety levels pre-surgery predicted ocular pain intensity scores (average NRS over 1 week recall) 6 months after LASIK (anxiety symptom check list (SCL)-90: β= 0.61, p<0.0005). In a similar manner, depression and ocular pain scores pre-surgery predicted neuropathic eye symptoms (via NPSI-Eye) 6 months after surgery (Depression SCL-90: β = 0.50, p<0.0005; NRS: β = 0.45, p 0.0005).39 In the current study, we found that use of oral anti-allergy medication pre-surgery was associated with PPOP after surgery. Interestingly, a similar finding was noted in a prior study where oral anti-histamine use predicted PPOP using a dry eye specific questionnaire 6 months after cataract surgery (DEQ5≥6: OR=6.2, CI=2.2–17.8, p=0.0003, n=119).40 The mechanisms behind these associations are not clear and are likely multifactorial, including biological, psychological, and socioeconomic factors.41
However, some hypothesis may be postulated. For example, inflammation in multiple compartments may impact both mental health and pain42, and the propensity for a pro-inflammatory state can have genetic and epigenetic origins.43 Oral allergy medications, such as antihistamines, inhibit effects mediated by histamine. In the peripheral nervous system, this blockade can translate into activation and sensitization of nociceptive sensory nerve fibers.44, 45 Future studies are needed that examine mechanisms that may underlie our noted associations.
As with all studies, the study limitations need to be considered when interpreting our results. First, we defined persistent pain using specific questions at two time points (3 and 6 months) and thus cannot comment on pain evolution beyond the 6-month time point. Furthermore, we cannot comment on the location of pain within the eye (e.g., superficial, deep, eyelid). Second, our study recruited individuals undergoing refractive surgery in two geographical locations within the United States (US). Furthermore, due to limited space and capacity, not all individuals who met criteria for inclusion were offered enrollment into the study on a given day. As such, generalizability to geographically distinct populations and individuals not offered recruitment within our sites needs further examination. Third, we performed a comprehensive ocular surface examination at 3 and 6 months, but did not include all potentially relevant tests, such as confocal microscopy and infrared meibography. Fourth, there are measures that we cannot tease out from our study, such as the motivation for artificial tear use after surgery (e.g., used because eyes felt uncomfortable or because ordered to use by the treating physician). Finally, unaccounted confounders (e.g., diet, environmental exposures) may have influenced our outcomes measures.
Despite these limitations, this study highlights that a minority of individuals develop persistent pain after refractive surgery and identifies risk factors for its development. While these findings need to be validated in larger studies, this knowledge may inform preventive and treatment strategies that can be guided by literature on PPOP outside the eye. For example, with regards to prevention, in randomized clinical trials oral neuromodulators (e.g., gabapentin, pregabalin) have been shown to decrease PPOP incidence after many, but not all, surgical procedures.15 Dosing strategies have ranged from high dose pre-operative administration only, to a low pre-operative dose followed by an extended taper.15 While one small, randomized study using the latter approach (150 mg pregabalin twice daily, started prior to refractive surgery and continued for 14 days) did not impact ocular pain symptoms after LASIK39, future studies in larger populations, perhaps in individuals at risk for PPOP development, are still warranted. Unique to the eye, neuromodulators can also be given in topical form, and some molecules have been found to improve corneal sensitivity and nerve growth after LASIK in animal models, including topical insulin-like growth factor-1 (IGF-1)47, macrophage migration inhibitory factor, and nerve growth factor (NGF).48, 49 Similar agents have also been studied as treatments for humans with presumed neuropathic ocular pain.50, 51 These lines of investigation are worthwhile as improvement in pain can translate into improved function and quality of life, preventing the development or lessening the morbidity of this condition. The future direction of our current study is to couple the clinical measures outlined above with tear proteins to develop prognostic and diagnostic models that can be used to identify individuals at high risk for pain development and to improve treatment approaches in individuals with pain, guided by the tear proteins of interest.
Acknowledgments
We would like to thank the members of the Data Monitoring and Oversite Committee for their support of this study: Todd Margolis MD PhD, Mae Gordon PhD, Houmam Araj PhD, Jimmy Le ScD, Donald Everett MA
Funding/Support:
Supported by the National Eye Institute R61EY032468 (Drs. Aicher and Galor)
Other support:
R01EY026174 (Dr. Galor), Department of Veterans Affairs, Veterans Health Administration, Office of Research and Development, Clinical Sciences R&D (CSRD) I01 CX002015 (Dr. Galor) and Biomedical Laboratory R&D (BLRD) Service I01 BX004893 (Dr. Galor), Department of Defense Gulf War Illness Research Program (GWIRP) W81XWH-20-1-0579 (Dr. Galor) and Vision Research Program (VRP) W81XWH-20-1-0820 (Dr. Galor), NIH Center Core Grant P30EY014801 (University of Miami), and Research to Prevent Blindness Unrestricted Grant (University of Miami and Casey Eye Institute).
Footnotes
Financial Disclosures: All authors have no financial disclosures.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Levitt AE, Galor A, Weiss JS, et al. Chronic dry eye symptoms after LASIK: parallels and lessons to be learned from other persistent post-operative pain disorders. Mol Pain 2015;11:21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Kalangara JP, Galor A, Levitt RC, et al. Characteristics of Ocular Pain Complaints in Patients With Idiopathic Dry Eye Symptoms. Eye Contact Lens 2017;43(3):192–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Eydelman M, Hilmantel G, Tarver ME, et al. Symptoms and Satisfaction of Patients in the Patient-Reported Outcomes With Laser In Situ Keratomileusis (PROWL) Studies. JAMA Ophthalmol 2017;135(1):13–22. [DOI] [PubMed] [Google Scholar]
- 4.Schiffman RM, Christianson MD, Jacobsen G, et al. Reliability and validity of the Ocular Surface Disease Index. Arch Ophthalmol 2000;118(5):615–21. [DOI] [PubMed] [Google Scholar]
- 5.Shoja MR, Besharati MR. Dry eye after LASIK for myopia: Incidence and risk factors. Eur J Ophthalmol 2007;17(1):1–6. [DOI] [PubMed] [Google Scholar]
- 6.Hovanesian JA, Shah SS, Maloney RK. Symptoms of dry eye and recurrent erosion syndrome after refractive surgery. J Cataract Refract Surg 2001;27(4):577–84. [DOI] [PubMed] [Google Scholar]
- 7.Albietz JM, Lenton LM, McLennan SG. Effect of laser in situ keratomileusis for hyperopia on tear film and ocular surface. J Refract Surg 2002;18(2):113–23. [DOI] [PubMed] [Google Scholar]
- 8.Tuisku IS, Lindbohm N, Wilson SE, Tervo TM. Dry eye and corneal sensitivity after high myopic LASIK. J Refract Surg 2007;23(4):338–42. [DOI] [PubMed] [Google Scholar]
- 9.De Paiva CS, Chen Z, Koch DD, et al. The incidence and risk factors for developing dry eye after myopic LASIK. Am J Ophthalmol 2006;141(3):438–45. [DOI] [PubMed] [Google Scholar]
- 10.Donnenfeld ED, Solomon K, Perry HD, et al. The effect of hinge position on corneal sensation and dry eye after LASIK. Ophthalmology 2003;110(5):1023–9; discussion 9–30. [DOI] [PubMed] [Google Scholar]
- 11.Denoyer A, Landman E, Trinh L, et al. Dry eye disease after refractive surgery: comparative outcomes of small incision lenticule extraction versus LASIK. Ophthalmology 2015;122(4):669–76. [DOI] [PubMed] [Google Scholar]
- 12.Gong Q, Li A, Chen L, et al. Evaluation of Dry Eye After Refractive Surgery According to Preoperative Meibomian Gland Status. Front Med (Lausanne) 2022;9:833984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bower KS, Sia RK, Ryan DS, et al. Chronic dry eye in photorefractive keratectomy and laser in situ keratomileusis: Manifestations, incidence, and predictive factors. Journal of Cataract & Refractive Surgery 2015;41(12). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Chao C, Golebiowski B, Stapleton F. The role of corneal innervation in LASIK-induced neuropathic dry eye. Ocul Surf 2014;12(1):32–45. [DOI] [PubMed] [Google Scholar]
- 15.Schmidt PC, Ruchelli G, Mackey SC, Carroll IR. Perioperative gabapentinoids: choice of agent, dose, timing, and effects on chronic postsurgical pain. Anesthesiology 2013;119(5):1215–21. [DOI] [PubMed] [Google Scholar]
- 16.Moshirfar M, Bhavsar UM, Durnford KM, et al. Neuropathic Corneal Pain Following LASIK Surgery: A Retrospective Case Series. Ophthalmol Ther 2021;10(3):677–89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Baksh BS, Morkin M, Felix E, et al. Ocular Pain Symptoms in Individuals With and Without a History of Refractive Surgery: Results From a Cross-Sectional Survey. Cornea 2022;41(1):31–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Chalmers RL, Begley CG, Caffery B. Validation of the 5-Item Dry Eye Questionnaire (DEQ-5): Discrimination across self-assessed severity and aqueous tear deficient dry eye diagnoses. Cont Lens Anterior Eye 2010;33(2):55–60. [DOI] [PubMed] [Google Scholar]
- 19.Ferreira-Valente MA, Pais-Ribeiro JL, Jensen MP. Validity of four pain intensity rating scales. Pain 2011;152(10):2399–404. [DOI] [PubMed] [Google Scholar]
- 20.Dworkin RH, Turk DC, Farrar JT, et al. Core outcome measures for chronic pain clinical trials: IMMPACT recommendations. Pain 2005;113(1–2):9–19. [DOI] [PubMed] [Google Scholar]
- 21.Farhangi M, Feuer W, Galor A, et al. Modification of the Neuropathic Pain Symptom Inventory for use in eye pain (NPSI-Eye). Pain 2019;160(7):1541–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Methodologies to diagnose and monitor dry eye disease: report of the Diagnostic Methodology Subcommittee of the International Dry Eye WorkShop (2007). Ocul Surf 2007;5(2):108–52. [DOI] [PubMed] [Google Scholar]
- 23.Kroenke K, Spitzer RL, Williams JB. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med 2001;16(9):606–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Alschuler KN, Jensen MP, Ehde DM. Defining mild, moderate, and severe pain in persons with multiple sclerosis. Pain Med 2012;13(10):1358–65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Perneger TV. What’s wrong with Bonferroni adjustments. BMJ 1998;316(7139):1236–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Bursac Z, Gauss CH, Williams DK, Hosmer DW. Purposeful selection of variables in logistic regression. Source Code Biol Med 2008;3:17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.McGreevy K, Bottros MM, Raja SN. Preventing Chronic Pain following Acute Pain: Risk Factors, Preventive Strategies, and their Efficacy. Eur J Pain Suppl 2011;5(2):365–72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Al-Sabbagh M, Okeson JP, Khalaf MW, Bhavsar I. Persistent pain and neurosensory disturbance after dental implant surgery: pathophysiology, etiology, and diagnosis. Dent Clin North Am 2015;59(1):131–42. [DOI] [PubMed] [Google Scholar]
- 29.Niraj G, Rowbotham DJ. Persistent postoperative pain: where are we now? Br J Anaesth 2011;107(1):25–9. [DOI] [PubMed] [Google Scholar]
- 30.H. L, A. K, K.-å. J. Prediction of persistent pain after total knee replacement for osteoarthritis. The Journal of Bone and Joint Surgery British volume 2008;90-B(2):166–71. [DOI] [PubMed] [Google Scholar]
- 31.Fletcher D, Stamer UM, Pogatzki-Zahn E, et al. Chronic postsurgical pain in Europe: An observational study. European Journal of Anaesthesiology | EJA 2015;32(10):725–34. [DOI] [PubMed] [Google Scholar]
- 32.Gungor S, Fields K, Aiyer R, et al. Incidence and risk factors for development of persistent postsurgical pain following total knee arthroplasty: A retrospective cohort study. Medicine 2019;98(28):e16450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Khan JS, Sessler DI, Chan MTV, et al. Persistent Incisional Pain after Noncardiac Surgery: An International Prospective Cohort Study. Anesthesiology 2021;135(4):711–23. [DOI] [PubMed] [Google Scholar]
- 34.Hetmann F, Kongsgaard UE, Sandvik L, Schou-Bredal I. Prevalence and predictors of persistent post-surgical pain 12 months after thoracotomy. Acta Anaesthesiol Scand 2015;59(6):740–8. [DOI] [PubMed] [Google Scholar]
- 35.Brandsborg B, Nikolajsen L, Hansen Charlotte T, et al. Risk Factors for Chronic Pain after Hysterectomy: A Nationwide Questionnaire and Database Study. Anesthesiology 2007;106(5):1003–12. [DOI] [PubMed] [Google Scholar]
- 36.Tsirline VB, Colavita PD, Belyansky I, et al. Preoperative pain is the strongest predictor of postoperative pain and diminished quality of life after ventral hernia repair. Am Surg 2013;79(8):829–36. [DOI] [PubMed] [Google Scholar]
- 37.Benlolo S, Hanlon JG, Shirreff L, et al. Predictors of Persistent Postsurgical Pain After Hysterectomy-A Prospective Cohort Study. J Minim Invasive Gynecol 2021;28(12):2036–46.e1. [DOI] [PubMed] [Google Scholar]
- 38.Liu Q-R, Dai Y-C, Ji M-H, et al. Predictors and predictive effects of acute pain trajectories after gastrointestinal surgery. Scientific Reports 2022;12(1):6530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Galor A, Patel S, Small LR, et al. Pregabalin Failed to Prevent Dry Eye Symptoms after Laser-Assisted in Situ Keratomileusis (LASIK) in a Randomized Pilot Study. J Clin Med 2019;8(9). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Sajnani R, Raia S, Gibbons A, et al. Epidemiology of Persistent Postsurgical Pain Manifesting as Dry Eye-Like Symptoms After Cataract Surgery. Cornea 2018;37(12):1535–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Richebé P, Capdevila X, Rivat C. Persistent Postsurgical Pain: Pathophysiology and Preventative Pharmacologic Considerations. Anesthesiology 2018;129(3):590–607. [DOI] [PubMed] [Google Scholar]
- 42.Fitzcharles MA, Cohen SP, Clauw DJ, et al. Nociplastic pain: towards an understanding of prevalent pain conditions. Lancet 2021;397(10289):2098–110. [DOI] [PubMed] [Google Scholar]
- 43.Fernandez Robles CR, Degnan M, Candiotti KA. Pain and genetics. Curr Opin Anaesthesiol 2012;25(4):444–9. [DOI] [PubMed] [Google Scholar]
- 44.Obara I, Telezhkin V, Alrashdi I, Chazot PL. Histamine, histamine receptors, and neuropathic pain relief. Br J Pharmacol 2020;177(3):580–99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Arslan H, Gündoğdu EC, Sümbüllü M. The Effect of Preoperative Administration of Antihistamine, Analgesic and Placebo on Postoperative Pain in Teeth with Symptomatic Apical Periodontitis: A Randomized Controlled Trial. Eur Endod J 2016;1(1):1–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Ousler GW, Wilcox KA, Gupta G, et al. An evaluation of the ocular drying effects of 2 systemic antihistamines: loratadine and cetirizine hydrochloride. Annals of Allergy, Asthma & Immunology 2004;93(5):460–4. [DOI] [PubMed] [Google Scholar]
- 47.Wang C, Peng Y, Pan S, Li L. Effect of insulin-like growth factor-1 on corneal surface ultrastructure and nerve regeneration of rabbit eyes after laser in situ keratomileusis. Neurosci Lett 2014;558:169–74. [DOI] [PubMed] [Google Scholar]
- 48.Hyon JY, Hose S, Gongora C, et al. Effect of macrophage migration inhibitory factor on corneal sensitivity after laser in situ keratomileusis in rabbit. Korean J Ophthalmol 2014;28(2):170–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Joo MJ, Yuhan KR, Hyon JY, et al. The effect of nerve growth factor on corneal sensitivity after laser in situ keratomileusis. Arch Ophthalmol 2004;122(9):1338–41. [DOI] [PubMed] [Google Scholar]
- 50.Small LR, Galor A, Felix ER, et al. Oral Gabapentinoids and Nerve Blocks for the Treatment of Chronic Ocular Pain. Eye Contact Lens 2020;46(3):174–81. [DOI] [PubMed] [Google Scholar]
- 51.Patel S, Mittal R, Felix ER, et al. Differential Effects of Treatment Strategies in Individuals With Chronic Ocular Surface Pain With a Neuropathic Component. Front Pharmacol 2021;12:788524. [DOI] [PMC free article] [PubMed] [Google Scholar]


