Abstract
Background
The Mediterranean diet has been associated with lower risk of breast cancer (BC) but evidence from prospective studies on the role of Mediterranean diet on BC survival remains sparse and conflicting. We aimed to investigate whether adherence to Mediterranean diet prior to diagnosis is associated with overall and BC-specific mortality.
Methods
A total of 13,270 incident breast cancer cases were identified from an initial sample of 318,686 women in 9 countries from the European Prospective Investigation into Cancer and Nutrition (EPIC) study. Adherence to Mediterranean diet was estimated through the adapted relative Mediterranean diet (arMED), a 16-point score that includes 8 key components of the Mediterranean diet and excludes alcohol. The degree of adherence to arMED was classified as low (score 0–5), medium (score 6–8), and high (score 9–16). Multivariable Cox proportional hazards models were used to analyze the association between the arMED score and overall mortality, and Fine-Gray competing risks models were applied for BC-specific mortality.
Results
After a mean follow-up of 8.6 years from diagnosis, 2340 women died, including 1475 from breast cancer. Among all BC survivors, low compared to medium adherence to arMED score was associated with a 13% higher risk of all-cause mortality (HR 1.13, 95%CI 1.01–1.26). High compared to medium adherence to arMED showed a non-statistically significant association (HR 0.94; 95% CI 0.84–1.05). With no statistically significant departures from linearity, on a continuous scale, a 3-unit increase in the arMED score was associated with an 8% reduced risk of overall mortality (HR3-unit 0.92, 95% CI: 0.87–0.97). This result sustained when restricted to postmenopausal women and was stronger among metastatic BC cases (HR3-unit 0.81, 95% CI: 0.72–0.91).
Conclusions
Consuming a Mediterranean diet before BC diagnosis may improve long-term prognosis, particularly after menopause and in cases of metastatic breast cancer. Well-designed dietary interventions are needed to confirm these findings and define specific dietary recommendations.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12916-023-02934-3.
Keywords: Mediterranean diet, Breast cancer, Cancer survivors, Dietary patterns, Prospective studies
Background
Breast cancer (BC) is the most commonly diagnosed cancer and the fifth most common cause of cancer death worldwide, accounting for approximately one in four cancer cases and one in six cancer deaths in 2020 [1, 2]. The survival rate for BC varies depending on the stage of the cancer at the time of diagnosis, age of the patient, and type of BC diagnosed. Despite differences in survival across world regions, with lower rates in transitioning countries than in transitioned countries, early detection and advances in treatment are leading to an increase in the number of BC survivors [3].
Recent reviews show persuasive evidence that body fatness and physical activity may predict important outcomes for patients with breast cancer [4, 5]. However, evidence concerning diet is rather limited or inconclusive, and BC survivors are advised to follow cancer prevention guidelines once their treatment is completed [6, 7].
Adherence to the Mediterranean diet (MD) has been associated with lower risk of breast cancer [8]. Accordingly, most recommendations of the World Cancer Research Fund (WCRF) for cancer survivors are compatible with the MD pattern [4]. However, few studies have explored the role of this dietary pattern in relation to BC survival [9–12]. These studies were conducted in the USA and used slightly different versions of the MD pattern, different dietary assessment methods and collection times, and overall the results were inconclusive.
Our objective was to assess the association between adherence to a MD pattern, by means of the adapted relative Mediterranean diet (arMED) score [8], using dietary data collected prior to diagnosis, and all-cause and BC-specific mortality in women diagnosed with BC from nine European countries.
Methods
Study population
We used data from the European Prospective Investigation into Cancer and Nutrition (EPIC) study, a prospective, multicenter European cohort with more than half a million women and men recruited between 1992 and 2000. Full details of the study design and data collection have been described elsewhere [13, 14]. Participants completed questionnaires on diet, lifestyle, and medical history at the time of recruitment and anthropometric measurements were also obtained. All participants provided written informed consent and the study was approved by the ethical review committees of the International Agency for Research on Cancer (IARC-Lyon, France) and all local centers.
Dietary assessment
The dietary assessment was conducted using a combination of methods, including detailed dietary questionnaires, and food frequency questionnaires (FFQs). At recruitment, participants completed a validated country- or center-specific dietary questionnaire that included questions on the frequency and portion sizes of foods and drinks consumed in the previous year and was designed to capture the geographical specificity of the diet [13].
Derivation of the arMED score
To measure adherence to the MD, we used the arMED score [8], a variant of the original MD scale defined by Trichopoulou et al. [15]. The arMED score was based on tertiles of energy-adjusted intake of eight foods/food groups to reflect consumption in relation to the individual's total daily energy intake. Unlike the original score [15], the arMED includes olive oil instead of monounsaturated fats, and alcohol was excluded from the list of components due to its causal association with BC carcinogenesis. For five items presumed to fit the MD, a score of 0 to 2 was assigned to tertiles of intake: fruits (including nuts and seeds), vegetables (excluding potatoes), legumes, fish, and cereals. The scoring was inverted for the components presumed to not fit MD: meat (red meat and processed meat) and dairy products. The score was slightly modified for olive oil due to the relatively high proportion of non-consumers in some countries; a score of 0 was assigned to non-consumers, 1 to participants below the median (calculated among consumers), and 2 to participants at or above the median. Thus, the arMED score ranged from 0 to 16, with higher scores indicating greater adherence to MD.
Ascertainment of breast cancer cases
The International Classification of Diseases for Oncology (ICD-O-2) codes C50.0–50.9 were used to define BC cases. Women with prevalent tumors at recruitment, no follow-up data, no information on lifestyle and diet, or implausible diets were excluded; furthermore, BC cases (N = 50) with unknown vital status, inconsistent follow-up data, or with non-epithelial morphology were also excluded. Out of 318,686 women from nine countries (Denmark, France, Germany, Italy, the Netherlands, Norway, Spain, Sweden, and the UK), a total of 13,270 incident primary malignant breast cancers (including 14 in situ) were diagnosed during the follow-up and were included in the present analysis.
Statistical analyses
The baseline characteristics of the participants were described as mean (SD) for continuous variables and frequencies for categorical variables. Cox proportional hazard models were used to prospectively analyze associations between the arMED score and overall mortality. Fine-Gray competing risks models were performed to evaluate the association with BC-specific mortality, with other causes of death considered competing events. Entry time was defined as the date of diagnosis of primary breast cancer, and exit time was defined as the date of death or end of follow-up. The arMED score was assessed as a categorical variable according to low (score 0–5), medium (score 6–8), and high (score 9–16) adherence, using the medium category as the reference, as well as per 3-unit increase in the score. Restricted cubic spline models with five knots were fitted, and non-linearity was tested using the likelihood (LR) ratio test.
All models were stratified by country, menopausal status at diagnosis (women aged ≥ 55 years at diagnosis were considered postmenopausal regardless of the baseline information) and stage of the tumor (non-metastatic, metastatic, unknown) and adjusted for: age at diagnosis (5-years categories), education level (no formal education, primary school, secondary school, technical or professional training, university, and not specified), body mass index (BMI) (kg/m2, continuous), physical activity (inactive, moderately inactive, moderately active, active, unknown), alcohol consumption (non-drinker, 0 to < 3 g/day, 3 to < 12 g/day, 12 to < 24 g/day, ≥ 24 g/day, unknown), smoking status and intensity (never smokers, current smokers 1–15, 16–25, and > 25 cigarettes/day, former quit ≤ 10, 11–20, and > 20 years before recruitment, current smoker of cigars, pipes and occasional current smokers, current smokers with unknown intensity, and not specified), ever use of hormone replacement therapy for menopause at diagnosis (yes, no, unknown), grade of tumor (well differentiated, moderately differentiated, poorly differentiated or undifferentiated, not determined), and tumor receptor status (positive, negative, unknown) for estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor 2 (HER2). BMI was modeled as a restricted cubic spline to account for its non-linear association with mortality [5, 16].
Separate models for pre- and postmenopausal cases were performed and heterogeneity was tested by comparing models with and without the cross-product terms using the Likelihood ratio (LR) test. The proportional hazards assumptions were tested by using the Schoenfeld goodness-of-fit test.
Stratified analyses were performed for overall mortality according to potential modifiers of the association with arMED: BMI, physical activity, smoking status, tumor stage, and hormone receptor status (ER, PR, HER2, and triple negative), and adherence to dietary patterns related to underlying biological mechanisms of BC previously associated with BC survival [17]: low/high adherence to the Diabetes Risk Reduction Diet [DRRD] [18] and Inflammatory Score of Diet [ISD] [19].
In sensitivity analyses, we examined whether further adjustment for the time interval between recruitment (time at which dietary information was collected) and BC diagnosis modified our main results, as well as for the period of diagnosis, to account for the potential influence of improvements in treatment and diagnosis over time. Comorbidities, including cardiovascular disease and presence of diabetes at baseline, and a combined variable with mechanistic dietary patterns (DRRD-ISD) were also used to further adjust separate models and test the robustness of the results.
Direct adjusted cumulative incidence function (CIF) curves for the three levels of adherence to the arMED score and overall mortality were derived from the multivariable Cox proportional hazards model [20].
All analyses were performed using R version 4.2.2. We used a significance level of 0.05, but also considered the confidence intervals and point estimate magnitudes. Data analysis was conducted from October 1, 2022, to January 13, 2023.
Results
Our population included 13,270 incident cases of breast cancer. During a mean follow-up of 8.6 years from the date of diagnosis, 2340 women died, including 1475 specifically from breast cancer. Baseline characteristics of the women in relation to three levels of adherence to the arMED score are summarized in Table 1. Women with low arMED were older, with lower educational level, more likely to be current smokers, more likely to have a higher BMI, and were mostly postmenopausal. Women with low adherence to the arMED were also less likely to have well-differentiated tumors, more likely to be diagnosed at an earlier stage, and with a higher proportion of ER-positive and PR-positive tumors. Mediterranean countries in the cohort had the highest arMED scores (Spain, Italy, mean 10), and lowest for Sweden and the Netherlands (mean 5) (Additional file 1: Table S1). The adjusted HRs for overall and BC-specific mortality according to levels of adherence to the arMED score are summarized in Table 2. Among all BC survivors, low compared to medium adherence to arMED was associated with a 13% higher risk of all-cause mortality (HR 1.13, 95% CI: 1.01–1.26). Compared to medium adherence, the association for high adherence was not statistically significant (HR 0.94; 95% CI 0.84–1.05). Despite this, an assessment of the association of arMED and overall and BC-specific mortality using restricted cubic splines (Additional file 1: Fig. S1) showed no statistically significant departures from linearity (p = 0.8). Therefore, on a continuous scale, a 3-unit increase in the arMED score was associated with an 8% (HR3-unit 0.92, 95% CI: 0.87–0.97) reduced risk of overall mortality. In postmenopausal BC survivors, the arMED score showed an 8% (HR3-unit 0.92, 95% CI: 0.86–0.98) lower risk of overall mortality per 3-unit increase, whereas there was no statistically significant association in premenopausal BC survivors (HR3-unit 0.94, 95% CI: 0.84–1.06). Heterogeneity between the association of arMED and overall mortality according to menopausal status did not reach statistical significance (p = 0.437). Multivariable models for BC-specific mortality showed no evidence of association with the arMED score, in all BC survivors and in neither premenopausal nor postmenopausal BC survivors. Stratified analyses by Mediterranean and non-Mediterranean countries (Additional file 1: Table S2) showed overall inverse associations by increasing the arMED score and overall mortality with stronger associations among Mediterranean (HR3-unit 0.81, 95% 0.69–0.95) countries and no longer statistically significant among non-Mediterranean countries.
Table 1.
Baseline characteristics of the 13,270 breast cancer (BC) cases in the EPIC cohort according to the adherence to Mediterranean diet as measured by the adapted relative Mediterranean diet (arMED) score
| All BC cases (N = 13,270) | Adherence measured by the arMED score | |||
|---|---|---|---|---|
| Low (N = 3427) | Medium (N = 5162) | High (N = 4681) | ||
| Age at diagnosis, mean (SD) | 61 (8.8) | 63 (8.8) | 61 (8.5) | 60 (8.9) |
| Educational level | ||||
| None/primary | 3281 (24.7) | 998 (29.1) | 1049 (20.3) | 1234 (26.4) |
| Technical/professional | 3035 (22.9) | 1148 (33.5) | 1242 (24.1) | 645 (13.8) |
| Secondary | 3200 (24.1) | 622 (18.1) | 1317 (25.5) | 1261 (26.9) |
| Longer education | 3143 (23.7) | 588 (17.2) | 1294 (25.1) | 1261 (26.9) |
| Smoking status | ||||
| Never | 5940 (44.8) | 1465 (42.7) | 2276 (44.1) | 2199 (47) |
| Former | 3139 (23.7) | 806 (23.5) | 1233 (23.9) | 1100 (23.5) |
| Current | 2476 (18.7) | 895 (26.1) | 936 (18.1) | 645 (13.8) |
| Miscellaneous | 1487 (11.2) | 236 (6.9) | 605 (11.7) | 646 (13.8) |
| Alcohol consumption | ||||
| Non drinker | 1774 (13.4) | 422 (12.3) | 598 (11.6) | 754 (16.1) |
| > 0–3 g/day | 3820 (28.8) | 990 (28.9) | 1455 (28.2) | 1375 (29.4) |
| > 3–12 g/day | 4066 (30.6) | 1035 (30.2) | 1641 (31.8) | 1390 (29.7) |
| > 12–24 g/day | 2069 (15.6) | 523 (15.3) | 801 (15.5) | 745 (15.9) |
| > 24 g/day | 1541 (11.6) | 457 (13.3) | 667 (12.9) | 417 (8.9) |
| Body mass index (BMI) | ||||
| Normal weight | 7612 (57.4) | 1834 (53.5) | 3058 (59.2) | 2720 (58.1) |
| Overweight | 3943 (29.7) | 1105 (32.2) | 1473 (28.5) | 1365 (29.2) |
| Obesity | 1509 (11.4) | 440 (12.8) | 556 (10.8) | 513 (11) |
| Underweight | 206 (1.6) | 48 (1.4) | 75 (1.5) | 83 (1.8) |
| Physical activity | ||||
| Inactive | 2673 (20.1) | 583 (17) | 891 (17.3) | 1199 (25.6) |
| Moderately inactive | 4720 (35.6) | 1200 (35) | 1820 (35.3) | 1700 (36.3) |
| Moderately active | 3606 (27.2) | 867 (25.3) | 1555 (30.1) | 1184 (25.3) |
| Active | 2076 (15.6) | 709 (20.7) | 809 (15.7) | 558 (11.9) |
| Menopausal status at diagnosis | ||||
| Premenopausal | 3070 (23.1) | 613 (17.9) | 1152 (22.3) | 1305 (27.9) |
| Postmenopausal | 10,200 (76.9) | 2814 (82.1) | 4010 (77.7) | 3376 (72.1) |
| Ever use of hormones for menopause | ||||
| No | 7487 (56.4) | 1785 (52.1) | 2805 (54.3) | 2897 (61.9) |
| Yes | 5323 (40.1) | 1434 (41.8) | 2166 (42) | 1723 (36.8) |
| Unknown | 460 (3.5) | 208 (6.1) | 191 (3.7) | 61 (1.3) |
| Grade of tumor differentiation | ||||
| Well differentiated | 1298 (9.8) | 269 (7.8) | 547 (10.6) | 482 (10.3) |
| Moderately differentiated | 2917 (22) | 603 (17.6) | 1176 (22.8) | 1138 (24.3) |
| Undifferentiated or poorly diff | 2503 (18.9) | 462 (13.5) | 982 (19) | 1059 (22.6) |
| Not determined | 6552 (49.4) | 2093 (61.1) | 2457 (47.6) | 2002 (42.8) |
| Stage of tumor | ||||
| Stage 0/I | 1954 (14.7) | 573 (16.7) | 798 (15.5) | 583 (12.5) |
| Stage II | 1593 (12) | 506 (14.8) | 624 (12.1) | 463 (9.9) |
| Stage III | 303 (2.3) | 98 (2.9) | 119 (2.3) | 86 (1.8) |
| Non-metastatic, unknown stage | 3984 (30) | 773 (22.6) | 1622 (31.4) | 1589 (33.9) |
| Stage IV (metastatic) | 1777 (13.4) | 350 (10.2) | 699 (13.5) | 728 (15.6) |
| Unknown | 3659 (27.6) | 1127 (32.9) | 1300 (25.2) | 1232 (26.3) |
| ER status | ||||
| Negative | 1678 (12.6) | 391 (11.4) | 646 (12.5) | 641 (13.7) |
| Positive | 7500 (56.5) | 1739 (50.7) | 2968 (57.5) | 2793 (59.7) |
| Unknown | 4092 (30.8) | 1297 (37.8) | 1548 (30) | 1247 (26.6) |
| PR status | ||||
| Negative | 2612 (19.7) | 571 (16.7) | 1029 (19.9) | 1012 (21.6) |
| Positive | 5072 (38.2) | 1129 (32.9) | 1983 (38.4) | 1960 (41.9) |
| Unknown | 5586 (42.1) | 1727 (50.4) | 2150 (41.7) | 1709 (36.5) |
| HER2 status | ||||
| Negative | 3587 (27) | 931 (27.2) | 1377 (26.7) | 1279 (27.3) |
| Positive | 856 (6.5) | 202 (5.9) | 312 (6) | 342 (7.3) |
| Unknown | 8827 (66.5) | 2294 (66.9) | 3473 (67.3) | 3060 (65.4) |
Except for values where the mean and SD are specified, all values are presented as the total number (N) and %
Cut-off points of arMED categories: low adherence, 0–5; medium adherence, 6–8; high adherence, 9–16
Unknown categories of educational level (N = 611), smoking habit and intensity (N = 228), and physical activity (N = 195) are not shown in this table
Table 2.
Multivariable hazard ratios (HR) and 95% confidence interval (95%CI) of overall and breast cancer-specific mortality according to the adherence to the Mediterranean diet measured by the arMED score
| Categories of adherence of the arMED scorea | arMED score, continuous | |||
|---|---|---|---|---|
| Low | Medium | High | 3-units increase | |
| Overall mortality | ||||
| All BC survivorsb | 1.13 (1.01–1.26) | 1.00 reference | 0.94 (0.84–1.05) | 0.92 (0.87–0.97) |
| N (deaths) | 3427 (736) | 5162 (871) | 4681 (733) | 13,270 (2340) |
| Premenopausal BC survivorsc,d | 1.21 (0.94–1.56) | 1.00 reference | 1.11 (0.89–1.38) | 0.94 (0.84–1.06) |
| N (deaths) | 613 (131) | 1152 (183) | 1305 (213) | 3070 (527) |
| Postmenopausal BC survivorsc,e | 1.10 (0.98–1.24) | 1.00 reference | 0.89 (0.78–1.01) | 0.92 (0.86–0.98) |
| N (deaths) | 2814 (605) | 4010 (688) | 3376 (520) | 10,200 (1813) |
| P value heterogeneity* | 0.437 | |||
| BC-specific mortality | ||||
| All BC survivorsb | 1.12 (0.98–1.29) | 1.00 reference | 0.99 (0.86–1.13) | 0.97 (0.90–1.04) |
| N (deaths) | 3427 (441) | 5162 (541) | 4681 (493) | 13,270 (1475) |
| Premenopausal BC survivorsc,d | 1.23 (0.92–1.63) | 1.00 reference | 1.16 (0.91–1.48) | 0.97 (0.85–1.11) |
| N (deaths) | 613 (103) | 1152 (145) | 1305 (180) | 3070 (428) |
| Postmenopausal BC survivorse | 1.10 (0.94–1.28) | 1.00 reference | 0.93 (0.78–1.09) | 0.97 (0.89–1.05) |
| N (deaths) | 2814 (338) | 4010 (396) | 3376 (313) | 10,200 (1047) |
| P value heterogeneity* | 0.992 | |||
Abbreviations: arMED Adapted relative Mediterranean diet, N Number of breast cancer cases
aCategories of arMED: low adherence, 0–5; medium adherence, 6–8; high adherence, 9–16
bModel stratified by country, stage (metastatic, non-metastatic, unknown) and menopausal status at diagnosis and adjusted for age at diagnosis, attained level of education, physical activity, body mass index (modeled as restricted cubic spline), alcohol consumption reported at recruitment, smoking habit and intensity at recruitment, ever use of hormones for menopause at diagnosis, grade of differentiation, and tumor receptor status (ER, PR, HER2)
cSame model as above without stratification for menopausal status al diagnosis
dModel premenopause: variable age into 2 categories, < 50 and > = 50, and not adjusted for every use of hormone therapy replacement for menopause
eModel postmenopause: variable age into 2 categories, < 65 and > = 65
*Pvalue for heterogeneity between pre- and postmenopausal subgroups for the association of arMED and mortality outcomes
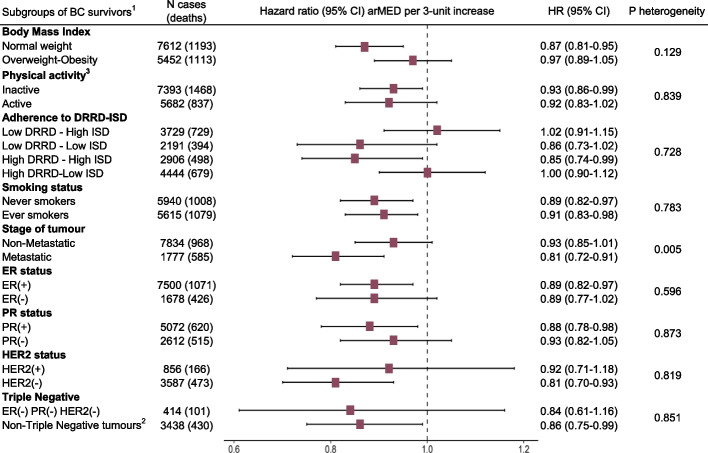
Results for arMED and overall mortality were unchanged with further adjustment for time between dietary data collection and BC diagnosis, period of diagnosis, or presence of comorbidities at baseline (Table 3). However, adjustment for adherence to DRRD-ISD dietary patterns attenuated the association. We also examined the association between arMED and non-BC-related deaths, including deaths from CVD and cancers other than BC (Additional file 1: Table S3). The arMED score was not associated with CVD mortality, whereas a statistically significant inverse association was observed for total non-BC-related mortality (p < 0.01) and cancer mortality (excluding BC) (p < 0.01). Figure 1 shows subgroup analyses for arMED per 3-unit increase in relation to overall mortality. Although there were clear associations in some subgroups but not others, there were no statistically significant interactions for the association between arMED score and overall mortality with BMI, physical activity, adherence to the DRRD-ISD dietary patterns, smoking status, or hormone receptor status, including triple negative tumors.
Table 3.
Sensitivity analysis: association between overall mortality and adherence to the arMED score by additional adjustments for time from baseline to diagnosis, period of diagnosis, presence of comorbidities, and other dietary patterns (N = 13,270, events/deaths = 2340)
| Categories of adherence of the arMED scorea | arMED score, continuous | |||
|---|---|---|---|---|
| Low (N = 3427) | Medium (N = 5162) | High (N = 4681) | 3-units increase | |
| Reference modelb | 1.13 (1.01–1.25) | 1.00 Reference | 0.94 (0.84–1.05) | 0.92 (0.87–0.97) |
| Model further adjusted for: | ||||
| Time from baseline to diagnosisc | 1.13 (1.02–1.26) | 1.00 Reference | 0.94 (0.84–1.04) | 0.92 (0.87–0.97) |
| Period of diagnosisd | 1.13 (1.01–1.25) | 1.00 Reference | 0.94 (0.84–1.05) | 0.92 (0.87–0.97) |
| Comorbiditiese | 1.13 (1.02–1.26) | 1.00 Reference | 0.93 (0.84–1.04) | 0.92 (0.86–0.97) |
| Mechanistic dietary patterns (DRRD-ISD)f | 1.10 (0.99–1.23) | 1.00 Reference | 0.98 (0.87–1.09) | 0.95 (0.89–1.01) |
Abbreviations: arMED Adapted relative Mediterranean diet, N Number of breast cancer cases
aCategories of arMED: low adherence, 0–5; medium adherence, 6–8; high adherence, 9–16
bModel stratified by country, stage (metastatic, non-metastatic, unknown) and menopausal status at diagnosis and adjusted for age at diagnosis, attained level of education, physical activity, body mass index (modeled as restricted cubic spline), alcohol consumption reported at recruitment, smoking habit and intensity at recruitment, ever use of hormones for menopause at diagnosis, grade of differentiation, and tumor receptor status (ER, PR, HER2)
cCategories of time from baseline to diagnosis are < 5 years, 5 to < 8 years, 8 to < 12 years, and equal or more than 12 years
dCategories for period of diagnosis are before 2000, between 2000 and < 2004, between 2004 and < 2008, 2008 onwards
eComorbidities include the presence of diabetes (categories yes, no, unknown) and the presence of cardiovascular problem reported at recruitment (categories yes, no, unknown)
fAdjusted for a 4-level variable that combines two dietary patterns in high-low adherence categories: Diabetes Reduced Risk Diet (DRRD) and Inflammatory Score of Diet (ISD)
Fig. 1.
Associations between arMED score and overall survival among subgroups of breast cancer survivors. Abbreviations: BC, breast cancer; BMI, body mass index; HR, hazard ratio; CI, confidence interval; ER, estrogen receptor; PR, progesterone receptor; HER2, human epidermal growth factor receptor 2; DRRD, Diabetes Risk Reduction Diet; ISD, Inflammatory Score of the Diet; N, number of breast cancer cases. 1Each model has excluded of their stratification/adjustment the specific variable that represents the subgroup in each case. Models stratified by country, stage (metastatic, non-metastatic, unknown) and menopausal status at diagnosis and adjusted for age at diagnosis, attained level of education, physical activity, body mass index (modeled as restricted cubic spline with five internal knots placed at equally spaced percentiles), alcohol consumption reported at recruitment, smoking habit and intensity as cigarettes per day at recruitment, ever use of hormones for menopause at diagnosis, grade of differentiation, and tumor receptor status: ER, PR, HER2. 2Only BC survivors with known status of the receptor (positive or negative) have been considered. 3Categories of physical activity. Inactive: includes moderately inactive and inactive. Active: includes moderately active and active
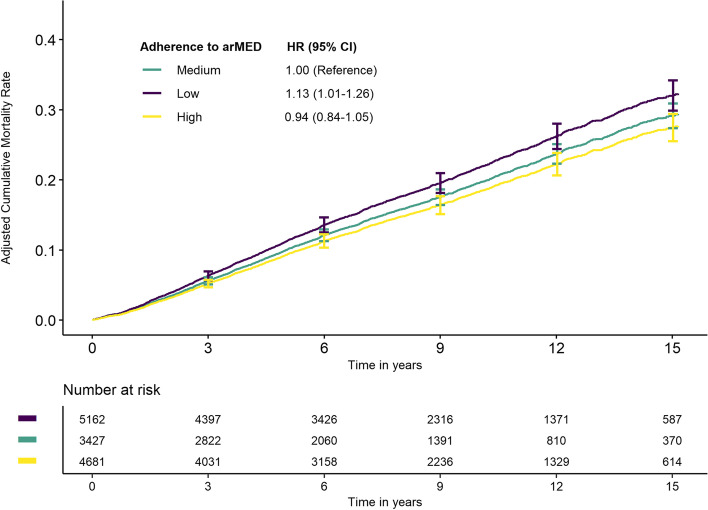
The only statistically significant interaction was observed with tumor stage at diagnosis (non-metastatic versus metastatic tumors) (p < 0.01): a protective association of arMED was observed in metastatic BC survivors for overall mortality (HR3-unit 0.81, 95% CI: 0.72–0.91): The same was observed for BC-specific mortality, with HR3-unit 0.86 (95% CI: 0.76–0.98), with p-value heterogeneity < 0.01 (Additional file 1: Table S4). The direct adjusted CIF curves for overall mortality derived from the Cox model are presented in Fig. 2. The 15-year cumulative mortality estimates were 32% (30–34%), 29% (27–31%), and 27% (26–29%) for low, medium, and high adherence to the arMED score, respectively.
Fig. 2.
Direct adjusted cumulative incidence function curves for overall mortality by categories of adherence to arMED score. HR hazard ratio, CI confidence interval, arMED adapted relative Mediterranean diet. Categories of arMED: low adherence, 0–5; medium adherence, 6–8; high adherence, 9–16. HRs and CI 95% from multivariable Cox model stratified by country, stage (metastatic, non-metastatic, unknown) and menopausal status at diagnosis and adjusted for age at diagnosis, attained level of education, physical activity, body mass index (modeled as restricted cubic spline), alcohol consumption reported at recruitment, smoking habit and intensity at recruitment, ever use of hormones for menopause at diagnosis, grade of differentiation, and tumor receptor status (ER, PR, HER2)
Discussion
In this prospective cohort study including 13,270 BC survivors, low adherence to a MD pattern before BC diagnosis was positively associated with all-cause mortality (13% higher risk compared to medium adherence). The results also showed an 8% lower overall mortality in all BC survivors for each 3-unit increase in the arMED score, which was sustained when restricted to postmenopausal women. Although no significant heterogeneity was observed by menopausal status, the smaller number of premenopausal cases may have limited our statistical power to detect significant associations. Additionally, while a statistically significant lower risk of all-cause mortality was observed with the arMED score modeled continuously, the association did not reach statistical significance when comparing high versus medium adherence. Moreover, there were no clear associations for BC-specific mortality. In addition, high adherence to MD appears to be particularly beneficial against overall mortality in Mediterranean countries, whereas in non-Mediterranean countries, the impact of increased adherence to MD seems more relevant when moving from low to medium levels of adherence.
Two recent systematic reviews have evaluated the relationship of several dietary patterns with BC survival [21, 22]. Only three studies included in these reviews evaluated the role of a MD pattern on prognostic outcomes in BC survivors [9–11]. Our study found a statistically significant inverse association between adherence to MD and all-cause mortality which conflicts with the null results in these studies. This discrepancy could be owing to a number of differences, including the study population, the score used to measure adherence to MD, the time when diet was measured with respect to BC diagnosis, and the different method of dietary assessment. For instance, the three studies [9–11], conducted in the USA, did not consider olive oil, but used a ratio of monounsaturated to saturated fats instead, and all of them included alcohol consumption. One study was restricted to postmenopausal women with stage I-III BC [9], and another [10] was a retrospective cohort with a small sample size (N = 110) of self-reported BC survivors whose dietary information was based on a single 24-h dietary recall. Finally, whereas our study assessed diet before diagnosis, other studies did it either close to the time of diagnosis [11] or after diagnosis [9].
Despite this, and in line with our results, a recent study observed statistically significant inverse associations with overall mortality for higher adherence to the Alternative Mediterranean Diet (aMED), a version of the MD adapted to the US population [12]. However, the Breast Cancer Family Registry is based on families with multiple cases of BC and the findings may not be generalizable to women without a family history of BC.
In our study, 63% of deaths were related specifically to BC, and BC used to be the most common cause of death following a BC diagnosis [23]. However, evidence now suggests that cardiovascular disease (CVD) accounts for a substantial number of non-BC-related deaths in the 10 years following diagnosis [23, 24]. Although MD is well-known to be protective against CVD [25], in our study, most deaths by causes other than BC were related to other cancers (13%) and only 150 (6%) were from CVD (Additional file 1: Table S3). Our findings showed statistically significant inverse associations between arMED score and overall non-BC mortality, including cancer mortality (excluding BC), but a non-significant association with CVD-related mortality. This warrants further investigation in future studies with a greater number of deaths related to these causes. On the other hand, Cox models additionally adjusted for the presence of comorbidities, including CVD or diabetes at the time of recruitment, showed no apparent differences with respect to the main results (Table 2), suggesting that the effect of the MD on mortality could occur beyond its protective role on these diseases.
Consistent with our results, the Nurses’ Health Study found a statistically significant reduced risk of all-cause mortality with higher adherence to MD restricted to BC survivors with low physical activity [9]. Our subgroup analyses also showed a statistically significant inverse association between adherence to MD and all-cause mortality in BC survivors who were inactive, and additionally in those with normal weight. Similarly, a recent study reported a statistically significant inverse association for adherence to MD with overall mortality in BC cases of normal BMI range [12]. A plausible interpretation for this might be that the impact of diet is more evident in BC survivors without the presence of other strong prognostic determinants such as obesity and physical activity, where perhaps the mechanisms underlying these conditions may blur the modest effect of diet. However, no heterogeneity was found, hence no firm conclusions can be drawn.
With respect to hormone receptors, BC survivors with ER/PR-positive and HER2-negative tumors showed lower overall mortality with increasing arMED score, although there was no statistically significant interaction between groups (p > 0.05). Metastatic BC survivors seemed to benefit more from adherence to the MD when assessed by the arMED score than non-metastatic BC survivors, with a statistically significant reduced risk of overall mortality and BC-specific mortality, however, we do not have a clear interpretation for these results.
The positive effect on health attributed to the MD [26] is thought to be due to several mechanisms, including weight control, antioxidant potential, improvement of glycemic profile, and anti-inflammatory properties [27, 28]. Some of these mechanisms are also involved in the prognosis of breast cancer [29]. However, to our knowledge, the impact of the MD pattern on these mechanisms in relation to BC prognosis has rarely been investigated [30]. We have recently reported that two dietary patterns related to lower insulin resistance and the inflammatory potential of the diet (the DRRD and the ISD, respectively) were associated with overall mortality among BC survivors [17]. To investigate the independent effect of the arMED score we adjusted a model with a variable combining high and low adherence to the DRRD and ISD scores. We found that the observed association of the arMED with mortality was attenuated, probably reflecting that, at least partially, the observed effect of MD may be explained by mechanisms of inflammation and insulin resistance or hyperinsulinemia.
Strengths and limitations
This study has several strengths. The prospective study design and long follow-up time allowed us to identify large numbers of deaths among a large sample of BC survivors. One specific advantage of this study is the inclusion of populations from Mediterranean and non-Mediterranean countries, which allowed us to have a broad representation of degrees of adherence to the MD pattern, with enough participants (and events) in different categories of the arMED.
One important limitation is the lack of information on treatment, a strong determinant of prognosis and survival. We used the available information on tumor stage at diagnosis, grade of tumor differentiation, and hormone receptor status as potential surrogates for treatment, since these characteristics are strong determinants of the therapeutic strategy, at least in most settings where the EPIC cohorts were recruited. Although we investigated differences by hormone receptor status for BC mortality outcomes, estimates were imprecise probably due to the missingness among these variables in our analysis. In addition, our results are based on a single dietary assessment, which may not reflect possible changes in dietary habits. To address to what extent the diet collected at baseline remains constant until diagnosis we performed sensitivity analyses adjusting for the time from diet measurement to diagnosis, and the results were unchanged. Similarly, we considered the possibility that survivors with an older diagnosis would be less likely to benefit from the early detection and advances in treatments, resulting in a worse prognosis, as compared to those with a more recent diagnosis. We performed sensitivity analyses adjusting for different periods of diagnosis and the observed results remained stable.
Conclusions
Our research suggests that adherence to a Mediterranean diet before BC diagnosis may improve long-term prognosis, especially in postmenopausal women. Furthermore, we found that the protective effect of MD appears to be stronger in women diagnosed with metastatic BC tumors. The link between postdiagnosis dietary patterns and BC outcomes remains unclear and BC survivors are still advised to follow general guidelines for cancer prevention once their treatment is completed. Further research, including large, well-designed dietary interventions will help provide more specific dietary recommendations for breast cancer survivors. In the meantime, research on the underlying biological mechanisms of various dietary patterns may provide relevant insights on the role of nutrition in breast cancer prognosis.
Supplementary Information
Additional file 1. Association of Mediterranean diet with survival after breast cancer diagnosis in women from nine European countries: results from the EPIC cohort study. Table S1. Summary of the arMED score in women from different EPIC countries. Table S2. Multivariable hazard ratiosand 95% confidence intervalof risk of overall mortality according to adherence to the Mediterranean diet across Mediterranean and non-Mediterranean countries in the EPIC study. Table S3. Multivariable hazard ratiosand 95% confidence intervalfor the adherence to the Mediterranean diet measured by the arMED score and other causes of death in all BC survivors. Table S4. Associations between the arMED score and BC-specific survival among breast cancer cases with non-metastatic and metastatic tumours. Figure S1. Assessment of linear associations between arMED score and overall and BC-specific mortality using restricted cubic spline models.
Acknowledgements
We thank CERCA Programme/Generalitat de Catalunya for institutional support. We also thank the National Institute for Public Health and the Environment (RIVM), Bilthoven, the Netherlands, for their contribution and on-going support to the EPIC Study. The authors thank all EPIC participants and staff for their outstanding contribution to the study.
IARC disclaimer
Where authors are identified as personnel of the International Agency for Research on Cancer / World Health Organization, the authors alone are responsible for the views expressed in this article and they do not necessarily represent the decisions, policy, or views of the International Agency for Research on Cancer / World Health Organization.
Abbreviations
- arMED
Adapted relative Mediterranean diet
- BC
Breast cancer
- BMI
Body mass index
- CI
Confidence interval
- DRRD
Diabetes Risk Reduction Diet
- EPIC
European Prospective Investigation into Cancer and Nutrition
- ER
Estrogen receptor
- HER2
Human epidermal growth factor receptor 2
- HR
Hazard ratio
- ISD
Inflammatory score of the diet
- MD
Mediterranean diet
- PR
Progesterone receptor
- SD
Standard deviation
Authors’ contributions
CCE and AA designed the study. AA supervised the statistical analyses. CCE performed the statistical analyses. CCE and AA drafted the manuscript. CB, MCB, NNZ, AT, LM, MHL, TT, VK, CLC, MBS, FJ, GM, SS, SP, CDG, GS, KBB, KSO, MJS, PA, MDC, MG, MS, SB, MJG, EMGG, EW, IAB, KKT, AKH, DA, and LD contributed to the acquisition and interpretation of data and critically revised the manuscript for important intellectual content. All authors read and approved the final manuscript.
Funding
Carlota Castro-Espin is funded by Instituto de Salud Carlos III through the Grant FI19/00197 (co-funded by the European Social Fund. ESF investing in your future). This study is funded by the AECC Scientific Foundation (Project PRYES211366AGUD). The coordination of EPIC is financially supported by International Agency for Research on Cancer (IARC) and by the Department of Epidemiology and Biostatistics, School of Public Health, Imperial College London, which has additional infrastructure support provided by the NIHR Imperial Biomedical Research Centre (BRC). The national cohorts are supported by: Danish Cancer Society (Denmark); Ligue Contre le Cancer, Institut Gustave Roussy, Mutuelle Générale de l’Education Nationale, Institut National de la Santé et de la Recherche Médicale (INSERM) (France); German Cancer Aid, German Cancer Research Centre (DKFZ), German Institute of Human Nutrition Potsdam-Rehbruecke (DIfE), Federal Ministry of Education and Research (BMBF) (Germany); Associazione Italiana per la Ricerca sul Cancro-AIRC-Italy, Compagnia di SanPaolo and National Research Council (Italy); Dutch Ministry of Public Health, Welfare and Sports (VWS), Netherlands Cancer Registry (NKR), LK Research Funds, Dutch Prevention Funds, Dutch ZON (Zorg Onderzoek Nederland), World Cancer Research Fund (WCRF), Statistics Netherlands (The Netherlands); Health Research Fund (FIS)—Instituto de Salud Carlos III (ISCIII), Regional Governments of Andalucía, Asturias, Basque Country, Murcia and Navarra, and the Catalan Institute of Oncology—ICO (Spain); Swedish Cancer Society, Swedish Research Council and County Councils of Skåne and Västerbotten (Sweden); Cancer Research UK (14136 to EPIC-Norfolk; C8221/A29017 to EPIC-Oxford), Medical Research Council (1000143 to EPIC-Norfolk; MR/M012190/1 to EPIC-Oxford) (United Kingdom). The funders of this study had no role in the decisions about the analysis or interpretation of the data, or preparation, review or approval of the manuscript.
Availability of data and materials
EPIC data are available for investigators who seek to answer important questions on health and disease in the context of research projects that are consistent with the legal and ethical standard practices of IARC/WHO and the EPIC centers. The primary responsibility for accessing the data belongs to IARC and the EPIC centers. For information on how to submit an application for gaining access to EPIC data and/or biospecimens, please follow the instructions at https://login.research4life.org/tacsgr0epic_iarc_fr/access/index.php.
Declarations
Ethics approval and consent to participate
This study complies with the Declaration of Helsinki. EPIC was approved by the Ethics Committee of the International Agency for Research on Cancer (IARC) (ref IEC 14–02), Lyon, France, as well as the local ethics committees of the study centers. All participants provided written informed consent for data collection and storage, as well as individual follow-up.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests to disclose.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71(3):209–249. doi: 10.3322/caac.21660. [DOI] [PubMed] [Google Scholar]
- 2.Soerjomataram I, Bray F. Planning for tomorrow: global cancer incidence and the role of prevention 2020–2070. Nat Rev Clin Oncol. 2021;18(10):663–672. doi: 10.1038/s41571-021-00514-z. [DOI] [PubMed] [Google Scholar]
- 3.Allemani C, Matsuda T, Di Carlo V, et al. Global surveillance of trends in cancer survival: analysis of individual records for 37,513,025 patients diagnosed with one of 18 cancers during 2000–2014 from 322 population-based registries in 71 countries (CONCORD-3) Lancet. 2018;391(10125):1023–1075. doi: 10.1016/S0140-6736(17)33326-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.WCRF/AICR. World Cancer Research Fund/American Institute for Cancer Research. Continuous update project expert report 2018. Survivors of breast cancer and other cancers. Published online 2018. https://www.wcrf.org/wp-content/uploads/2021/02/Cancer-Survivors.pdf. Accessed 29 Nov 2021.
- 5.Tsilidis KK, Cariolou M, Becerra-Tomás N, et al. Postdiagnosis body fatness, recreational physical activity, dietary factors and breast cancer prognosis: Global Cancer Update Programme (CUP Global) summary of evidence grading. Int J Cancer. 2023;152(4):635–644. doi: 10.1002/ijc.34320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Rock CL, Thomson CA, Sullivan KR, et al. American Cancer Society nutrition and physical activity guideline for cancer survivors. CA Cancer J Clin. 2022;72(3):230–262. doi: 10.3322/caac.21719. [DOI] [PubMed] [Google Scholar]
- 7.After a cancer diagnosis follow our recommendations, if you can | Cancer prevention. WCRF International. https://www.wcrf.org/diet-activity-and-cancer/cancer-prevention-recommendations/after-a-cancer-diagnosis-follow-our-recommendations-if-you-can/. Accessed 5 Jan 2023.
- 8.Buckland G, Travier N, Cottet V, et al. Adherence to the mediterranean diet and risk of breast cancer in the European prospective investigation into cancer and nutrition cohort study. Int J Cancer. 2013;132(12):2918–2927. doi: 10.1002/ijc.27958. [DOI] [PubMed] [Google Scholar]
- 9.Kim EHJ, Willett WC, Fung T, Rosner B, Holmes MD. Diet quality indices and postmenopausal breast cancer survival. Nutr Cancer. 2011;63(3):381–388. doi: 10.1080/01635581.2011.535963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Karavasiloglou N, Pestoni G, Faeh D, Rohrmann S. Post-diagnostic diet quality and mortality in females with self-reported history of breast or gynecological cancers: results from the Third National Health and Nutrition Examination Survey (NHANES III) Nutrients. 2019;11(11):E2558. doi: 10.3390/nu11112558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ergas IJ, Cespedes Feliciano EM, Bradshaw PT, et al. Diet quality and breast cancer recurrence and survival: the pathways study. JNCI Cancer Spectr. 2021;5(2):pkab019. doi: 10.1093/jncics/pkab019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Haslam DE, John EM, Knight JA, et al. Diet quality and all-cause mortality in women with breast cancer from the breast cancer family registry. Cancer Epidemiol Biomarkers Prev. Published online March 1, 2022:EPI-22–1198. 10.1158/1055-9965.EPI-22-1198 [DOI] [PMC free article] [PubMed]
- 13.Riboli E, Hunt KJ, Slimani N, et al. European Prospective Investigation into Cancer and Nutrition (EPIC): study populations and data collection. Public Health Nutr. 2002;5(6b):1113–1124. doi: 10.1079/PHN2002394. [DOI] [PubMed] [Google Scholar]
- 14.Riboli E, Kaaks R. The EPIC Project: rationale and study design. European prospective investigation into cancer and nutrition. Int J Epidemiol. 1997;26(Suppl 1):S6–14. doi: 10.1093/ije/26.suppl_1.s6. [DOI] [PubMed] [Google Scholar]
- 15.Trichopoulou A, Costacou T, Bamia C, Trichopoulos D. Adherence to a Mediterranean diet and survival in a Greek population. N Engl J Med. 2003;348(26):2599–2608. doi: 10.1056/NEJMoa025039. [DOI] [PubMed] [Google Scholar]
- 16.Bonet C, Crous-Bou M, Tsilidis KK, et al. The association between body fatness and mortality among breast cancer survivors: results from a prospective cohort study. Eur J Epidemiol. Published online March 29, 2023. 10.1007/s10654-023-00979-5. [DOI] [PMC free article] [PubMed]
- 17.Castro-Espin C, Bonet C, Crous-Bou M, et al. Dietary patterns related to biological mechanisms and survival after breast cancer diagnosis: results from a cohort study. Br J Cancer. Published online February 3, 2023. 10.1038/s41416-023-02169-2. [DOI] [PMC free article] [PubMed]
- 18.Kang JH, Peng C, Rhee JJ, et al. Prospective study of a diabetes risk reduction diet and the risk of breast cancer. Am J Clin Nutr. 2020;112(6):1492–1503. doi: 10.1093/ajcn/nqaa268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Castro-Espin C, Agudo A, Bonet C, et al. Inflammatory potential of the diet and risk of breast cancer in the European Investigation into Cancer and Nutrition (EPIC) study. Eur J Epidemiol. 2021;36(9):953–964. doi: 10.1007/s10654-021-00772-2. [DOI] [PubMed] [Google Scholar]
- 20.Hu ZH, Peter Gale R, Zhang MJ. Direct adjusted survival and cumulative incidence curves for observational studies. Bone Marrow Transplant. 2020;55(3):538–543. doi: 10.1038/s41409-019-0552-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Castro-Espin C, Agudo A. The role of diet in prognosis among cancer survivors: a systematic review and meta-analysis of dietary patterns and diet interventions. Nutrients. 2022;14(2):348. doi: 10.3390/nu14020348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Becerra-Tomás N, Balducci K, Abar L, et al. Postdiagnosis dietary factors, supplement use and breast cancer prognosis: Global Cancer Update Programme (CUP Global) systematic literature review and meta-analysis. Int J Cancer. 2023;152(4):616–634. doi: 10.1002/ijc.34321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Afifi AM, Saad AM, Al-Husseini MJ, Elmehrath AO, Northfelt DW, Sonbol MB. Causes of death after breast cancer diagnosis: a US population-based analysis. Cancer. 2020;126(7):1559–1567. doi: 10.1002/cncr.32648. [DOI] [PubMed] [Google Scholar]
- 24.Riihimäki M, Thomsen H, Brandt A, Sundquist J, Hemminki K. Death causes in breast cancer patients. Ann Oncol. 2012;23(3):604–610. doi: 10.1093/annonc/mdr160. [DOI] [PubMed] [Google Scholar]
- 25.Estruch R, Ros E, Salas-Salvadó J, et al. Primary prevention of cardiovascular disease with a Mediterranean diet supplemented with extra-virgin olive oil or nuts. N Engl J Med. 2018;378(25):e34. doi: 10.1056/NEJMoa1800389. [DOI] [PubMed] [Google Scholar]
- 26.Dinu M, Pagliai G, Casini A, Sofi F. Mediterranean diet and multiple health outcomes: an umbrella review of meta-analyses of observational studies and randomised trials. Eur J Clin Nutr. 2018;72(1):30–43. doi: 10.1038/ejcn.2017.58. [DOI] [PubMed] [Google Scholar]
- 27.Jannasch F, Kröger J, Schulze MB. Dietary patterns and type 2 diabetes: a systematic literature review and meta-analysis of prospective studies. J Nutr. 2017;147(6):1174–1182. doi: 10.3945/jn.116.242552. [DOI] [PubMed] [Google Scholar]
- 28.Sureda A, Bibiloni MDM, Julibert A, et al. Adherence to the Mediterranean diet and inflammatory markers. Nutrients. 2018;10(1):62. doi: 10.3390/nu10010062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Cava E, Marzullo P, Farinelli D, et al. Breast Cancer Diet “BCD”: a review of healthy dietary patterns to prevent breast cancer recurrence and reduce mortality. Nutrients. 2022;14(3):476. doi: 10.3390/nu14030476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Skouroliakou M, Grosomanidis D, Massara P, et al. Serum antioxidant capacity, biochemical profile and body composition of breast cancer survivors in a randomized Mediterranean dietary intervention study. Eur J Nutr. 2018;57(6):2133–2145. doi: 10.1007/s00394-017-1489-9. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1. Association of Mediterranean diet with survival after breast cancer diagnosis in women from nine European countries: results from the EPIC cohort study. Table S1. Summary of the arMED score in women from different EPIC countries. Table S2. Multivariable hazard ratiosand 95% confidence intervalof risk of overall mortality according to adherence to the Mediterranean diet across Mediterranean and non-Mediterranean countries in the EPIC study. Table S3. Multivariable hazard ratiosand 95% confidence intervalfor the adherence to the Mediterranean diet measured by the arMED score and other causes of death in all BC survivors. Table S4. Associations between the arMED score and BC-specific survival among breast cancer cases with non-metastatic and metastatic tumours. Figure S1. Assessment of linear associations between arMED score and overall and BC-specific mortality using restricted cubic spline models.
Data Availability Statement
EPIC data are available for investigators who seek to answer important questions on health and disease in the context of research projects that are consistent with the legal and ethical standard practices of IARC/WHO and the EPIC centers. The primary responsibility for accessing the data belongs to IARC and the EPIC centers. For information on how to submit an application for gaining access to EPIC data and/or biospecimens, please follow the instructions at https://login.research4life.org/tacsgr0epic_iarc_fr/access/index.php.