Abstract
Cardiovascular disease (CVD) is the number one cause of death worldwide, with hypertension as the leading risk factor for both sexes. As sex may affect responsiveness to antihypertensive compounds, guidelines for CVD prevention might necessitate divergence between females and males. To this end, we studied the effectiveness of calcium channel blockers (CCB) on blood pressure (BP), heart rate (HR) and cardiac function between sexes. We performed a systematic review and meta-analysis on studies on CCB from inception to May 2020. Studies had to present both baseline and follow-up measurements of the outcome variables of interest and present data in a sex-stratified manner. Mean differences were calculated using a random-effects model. In total, 38 studies with 8202 participants were used for this review. In females as compared to males, systolic BP decreased by −27.6 mmHg (95%CI −36.4; −18.8) (−17.1% (95%CI −22.5;−11.6)) versus −14.4 mmHg (95%CI −19.0; −9.9) (−9.8% (95%CI −12.9;−6.7)) (between-sex difference p < 0.01), diastolic BP decreased by −14.1 (95%CI −18.8; −9.3) (−15.2%(95%CI −20.3;−10.1)) versus −10.6 mmHg (95%CI −14.0; −7.3) (−11.2% (95%CI −14.8;−7.7)) (between-sex difference p = 0.24). HR decreased by −1.8 bpm (95%CI −2.5; −1.2) (−2.5% (95%CI −3.4; −1.6)) in females compared to no change in males (0.3 bpm (95% CI −1.2; 1.8)) (between-sex difference p = 0.01). In conclusion, CCB lowers BP in both sexes, but the observed effect is larger in females as compared to males.
Keywords: hypertension, cardiovascular disease, calcium-channel blockers, sex differences, systematic review, meta-analysis
1. Introduction
Raised blood pressure primarily antedates cardiovascular disease (CVD), the number one cause of death worldwide [1,2]. As such, effective treatment of hypertension represents a key strategy for reducing cardiovascular diseases [2]. Pharmacological treatment and modulation of behavioral risk factors, amongst smoking cessation, weight reduction, physical activity, reduction in alcohol use and a healthy diet, lower the development of hypertension and CVD [1].
While in the past more males died from CVD, nowadays females have surpassed the other sex [3]. Females are considered more protected against cardiovascular events during the fertile period, but this protection diminishes after menopause. Consequently, CVD is a major cause of death in females above 65 years of age. However, in the younger population, gender-related factors, such as psychological stress and low socioeconomic status, are expected to have a significant influence on vascular ageing [4]. Timely treatment reduces the risk of CVD and its mortality for both sexes [5]. Weighing sex differences in risk factors such as smoking, system-biology, clinical manifestations, treatment effects and outcomes of CVD in guidelines may contribute to improved outcomes in prevention of CVD [6,7,8,9,10,11].
The predominantly used antihypertensive compounds are calcium-channel blockers (CCB), angiotensin-converting enzyme inhibitors (ACEI), angiotensin-receptor blockers (ARB), beta blockers (BB) and diuretics (DIU). CCB are one of the most widely used classes of antihypertensive agents. All approved CCB types exert their effects as pharmacologic agents inhibiting transmembrane calcium inflow through calcium channels, reducing actin–myosin interaction and the subsequent contraction of myocytes and with it, its vascular smooth muscle tone, thereby attributing to reduction in blood pressure. A remarkable number of clinical trials has investigated and proven the sufficient haemodynamic effect of CCB on cardiovascular and haemodynamic variables [12,13,14]. However, almost none of these trials have investigated the effects in females and males separately [15]. This raises the question whether CCB are equally effective in both sexes and whether treatment strategies should differentiate between both sexes.
To this end, we studied in a systematic review and meta-analysis the acute (0–14 days), sub-acute (15–30 days) and chronic (>30 days) intervention effects of CCB’s treatment on cardiovascular and haemodynamic variables in female versus male adults diagnosed with hypertension. The variables of primary interest are systolic blood pressure (SBP) and diastolic blood pressure (DBP), mean arterial blood pressure (MAP) and heart rate (HR). Variables of secondary interest are cardiac output (CO), left ventricular ejection fraction (LVEF) and left ventricular mass (LVM).
2. Materials and Methods
2.1. Literature Search
An extensive systematic literature search was conducted on articles evaluating the effects of antihypertensive medication on cardiovascular and haemodynamic variables using PubMed (NCBI) and Embase (Ovid) databases. PubMed and Embase provided publications published from inception to May 2020. The search terms are presented in Table S1. The search strategy aimed at studying the effect of the five antihypertensive drugs (CCB, ACEI, ARB, BB and DIU) on blood pressure, cardiac function and geometry. For this review, the articles reporting on CCB were used. The search limits used were ‘humans’ and ‘journal article’. The search served to study the following objectives:
To study differences and similarities between males and females in the effect of antihypertensive medication on cardiac function and structure.
To determine the representation of females in studies on the effect of antihypertensive drugs on CVD for the past century.
2.2. Guidelines
PRISMA 2020 reporting guidelines were taken into account. Our review was registered in the PROSPERO database with registration number CRD42021273583.
2.3. Eligibility Criteria
Studies had to focus on acute, sub-acute and/or chronic therapy with at least one type of CCB in female and/or male adults (≥18 years) diagnosed with hypertension. Moreover, studies had to include the mean with standard deviation (SD), standard error (SE), or 95% confidence interval (95% CI) of the baseline and follow-up measurements of one of the predefined variables (SBP, DBP, MAP, HR, CO, LVEF, and/or LVM). Studies also had to report the mean dose or dose range and treatment duration. Finally, the antihypertensive treatment had to be compared to a reference group (control, placebo or antihypertensive medication group other than CCB under study). Mean values with SD were requested from the authors by email if articles presented their data differently (for example, median with interquartile range).
2.4. Study Selection
After the initial search, studies were screened based on title and abstract. During this selection, other systematic reviews and meta-analyses, literature reviews, case reports, animal studies, and in vitro studies were excluded. Studies with subjects younger than 18 years and articles in another language than English or Dutch were excluded as well. The remaining studies were screened for suitability based on full-text using the eligibility criteria. Studies were excluded if they did not separate outcomes by antihypertensive medication (if participants received more than one antihypertensive medication as intervention) or did not report the treatment duration or a mean dose or dose range for the antihypertensive medication. Studies with individuals undergoing invasive operations, participants who were exercising during measurements, undergoing dialysis or chemotherapy were excluded as well. In case the articles did not stratify the data for sex, but all other eligibility criteria were met, authors from articles published after 1980 were contacted to request sex-specific data. If no contact details were found or if authors did not respond within three weeks after sending a reminder, the article was excluded. The reason for exclusion was registered for the full-text selection. Both selection steps were performed in pairs in a blinded standardized manner (title-abstract pairs: MA-EV, CD-SL, EL-DM, ZM-JW, MV-NW; full-text pairs: CD-NW, EL-MV, DM-SL, EV-JW). Discrepancies were resolved by mutual agreement.
2.5. Data Extraction
Study characteristics (sample size, control group, study design), anthropometric data (age, ethnicity), intervention characteristics (dose, duration, method of measurement) and effect measures (mean and SD at baseline and after CCB intervention of the predefined variables) were collected in a predesigned format. The study results were separately extracted for females and males. In this systematic review, only blood pressure data measured using non-invasive methods were extracted. For the other variables, multiple methods were allowed. Data extraction was performed by two investigators (RA, LK). This step of the process was not performed in duplicate.
2.6. Quality Assessment
The included studies were assessed for quality and risk of bias using the Cochrane recommended Risk of Bias 2 (RoB2) tool [16]. Studies were scored with “Low risk of bias”, “Some concerns” or “High risk of bias” on five domains including randomization process, deviations from intended interventions, missing data, outcome measurement and data reporting. To receive an overall risk-of-bias judgement of “Low risk of bias”, all domains had to receive this judgement. To receive an overall judgment of “High risk of bias”, at least one of the domains was scored as such. All other domain score combinations would rate a study with an overall judgement of “Some concerns”. The quality assessment was performed by two reviewers (RA, LK) and differences were solved by a third independent reviewer (DM, SL).
2.7. Statistical Analysis
If an SE or 95% CI was reported in the article, the SD was calculated according to the Cochrane Handbook for Systematic Review of Interventions [17]. Changes in the cardiovascular and haemodynamic variables from baseline were separately analyzed for females and males using a random-effects model as described by DerSimonian and Laird [18]. Because the included studies had some variation in study population and design, the random-effects model was chosen to account for this interstudy variation [18]. Egger’s regression test for funnel plot asymmetry was conducted to test for publication bias for each cardiovascular variable [19]. The primary outcome was the mean difference and 95% CI between baseline and follow-up of the intervention, visualized in forest plots. The relative change from baseline in percentage including 95% CI was also calculated and reported in parentheses behind the mean difference in the text. The I2 statistic, the ratio between heterogeneity and variability, was calculated as a measure of consistency and expressed as percentage in the forest plots. I2 is able to distinguish heterogeneity in data from solely sampling variance [17]. Interpretation of I2 was based on the guidelines in the Cochrane Handbook for Systematic Review of Interventions [17]. Sources of clinical heterogeneity (CCB type, treatment duration, and dosage) and methodological heterogeneity (quality of study) were investigated by meta-regression analyses using a mixed-effects model [17]. For the meta-analyses and meta-regression analyses, the meta package in the statistical program R version 4.0.3. was used [20,21].
3. Results
3.1. Study Selection
The literature search in PubMed and Embase provided a total of 73,867 unique records after removing duplicates (Figure 1). During the first screening, 58,737 articles were excluded resulting in 15,130 articles that were assessed based on the full text. Of those articles, 14,916 met at least one exclusion criterion and were excluded. For 766 articles (5%), it was not possible to find or access the full text at the university library or online. In total, 1141 articles (8%) had an unsuitable study design. This criterion was met when, for example, only measurements were taken during exercise, or SBP and DBP were measured using an arterial catheter. In total, 1058 articles (7%) did not report original research data; these articles were reviews, for example. In 1886 articles (13%), no antihypertensives were given to the patients participating. In 2141 articles (14%), antihypertensives were given, but treatment results were not stratified by those. In total, 1949 articles (13%) were excluded because treatment results were not stratified by sex. In total, 153 articles (1%) did not have reference measurements. In total, 3864 articles (26%) did not contain any measurements of interest. In 536 articles (4%), data were not suitably reported. In 984 articles (6%), no information was provided regarding either dose, duration, or both. Finally, there were 438 articles (3%) excluded because of other complications. At the end of the selection procedure, a total of 214 articles were classified suitable for inclusion (Figure 1). Eventually, in 38 of those articles, CCB were the provided treatment.
Figure 1.
Flowchart of systematic selection process.
3.2. Study Characteristics
Study characteristics and anthropometric data are visualized in Table 1. Data of 8202 subjects using CCB were included in this meta-analysis, of whom 3264 (39.8%) were female. The mean age of the subjects from the included studies was 66.2 ± 8.9 (SD) years.
Thirteen studies analyzed the effects of nifedipine [22,23,24,25,26,27,28,29,30,31,32,33,34], seven of diltiazem [29,35,36,37,38,39,40], five of amlodipine [41,42,43,44,45] and nicardipine [46,47,48,49,50], three of verapamil [51,52,53] and two of nitrendipine [54,55] and felodipine [56,57]. Lercanidipine [43], mibefradil [42], isradipine [58], lacidipine [26] and gallopamil [59] all had one study reporting on them. One study reported on both amlodipine and lercanidipine [43]. Another studied amlodipine together with mibefradil [42]. Additionally, diltiazem and nifedipine were studied together [29].
SBP was studied in 17 studies [26,28,29,33,35,36,37,39,40,43,44,45,51,52,54,55,57], DBP in 15 studies [26,29,33,35,37,39,40,43,44,45,51,52,54,55,57], MAP in 4 studies [34,41,45,57], HR in 28 studies [22,23,24,25,26,27,28,29,31,32,34,35,36,39,40,41,42,45,46,47,49,50,51,52,53,54,56,57], CO in 5 studies [39,42,56,57,58], LVEF in 12 studies [26,27,28,30,32,33,38,48,51,52,58,59] and LVM in 2 studies [40,54].
Sixteen studies measured acute [22,23,24,25,27,29,31,32,34,39,41,47,49,51,56,57] effect. In total, 4 included studies evaluated the acute and sub-acute effects of CCB [35,36,48,50], 5 evaluated only sub-acute effects [28,43,44,53,59] and 13 studies measured the chronic effects of CCB treatment [23,26,30,33,37,38,40,42,45,46,52,54,55,58].
Study designs consisted of 18 randomized controlled trials (RCT) [23,26,28,29,30,31,34,37,40,41,42,43,44,45,52,54,58,59] of which three were also a crossover study (34, 41, 52). Of the other studies, 1 was a retrospective cohort study [33] and 19 were prospective cohort studies [22,24,25,27,32,35,36,38,39,46,47,48,49,50,51,53,55,56,57]. Of the included studies containing CCB interventions, 27 studies included only male subjects [22,24,25,26,27,28,29,30,31,33,34,36,37,39,40,41,46,47,48,50,52,53,54,56,57,58,59], none included only female subjects, and the remaining 11 studies contained subjects of both sexes [23,32,35,38,42,43,44,45,49,51,55]. Publication bias assessed via Eggers’s regression showed significant bias for HR in males, but no significant bias for all other variables included (Table 2).
Table 2.
Publication bias using Eggers’s regression for all variables.
| Male | Female | cMD Male | cMD Female | |
|---|---|---|---|---|
| SBP | 0.3503 | 0.1627 | - | - |
| DBP | 0.7937 | 0.3185 | - | - |
| MAP | 0.2550 | - | - | - |
| HR | 0.0283 | 0.6273 | −2.09 (−3.60; −0.58) | - |
| CO | 0.4162 | - | - | - |
| LVEF | 0.1651 | - | - | - |
| LVM | - | - | - | - |
Table 1.
Characteristics of studies.
| Study | Patient | Ethnicity | CCB Treatment(Administration) | Mean Dose (mg/Day) | % Max Dose * | Subjects CCB (n) | Control Group ** | Controls (n) | Age (Years + SD) | Intervention Duration(Days) | Study Design | Extracted Variables | Mentioned Method(s) of Measurement | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Total | M | F | Total | M | F | ||||||||||||
| Mehlum (2020) [45] | HTN, DM, HF, MI, LVH | W, B, A | Amlodipine (oral) |
5 | 0.5 | 7477 | 4305 | 3172 | Valsartan | 7519 | 4332 | 3187 | 67.2 (8.1) | 180 | RCT | SBP, DBP, HR, MAP | Sphygmomanometry |
| Baysal (2017) [44] | HTN | - | Amlodipine (oral) |
10 | 1 | 38 | 22 | 16 | Telmisartan | 39 | 22 | 17 | 48.0 (10) |
30 | RCT | SBP, DBP | Sphygmomanometry, ECG, echo |
| Thuc Sinh (2015) [43] | HTN, DM | - | Amlodipine (oral) |
10 | 1 | 52 | 34 | 18 | - | - | - | - | 65.3 (10.7) | 28 | RCT | SBP, DBP, | Sphygmomanometry |
| Lercanidipine (oral) |
20 | 1 | 52 | 32 | 20 | ||||||||||||
| Lindqvist (2007) [42] | HTN | - | Amlodipine (oral) |
7.5 | 0.75 | 14 | 11 | 3 | - | - | - | - | 59 (***) | 42 | RCT | HR, CO | Catheterization, ECG, echo |
| Mibefradil (oral) |
75 | 0.75 | 14 | 11 | 3 | ||||||||||||
| Petrella (2000) [53] | HF | - | Verapamil (oral) |
240 | 0.33 | 10 | 10 | 0 | - | - | - | - | 73.0 (4.0) | 26 | Prospective cohort | HR | Echo |
| Burggraaf (1998) [34] | Healthy | - | Nifedipine (oral) |
20 | 0.33 | 9 | 9 | 0 | Captopril | 9 | 9 | - | 18–35 (***) | 0.125 | RCT, crossover | HR, MAP | Echo |
| Gottdiener (1998) [40] | HTN | W, B | Diltiazem (oral) |
240 | 0.67 | 185 | 185 | 0 | Atenolol, captopril, clonidine, hydrochlorothiazide or prazosin | 920 | 920 | 0 | 58.8 (10) | 730 | RCT | SBP, DBP, LVM, HR | Sphygmomanometry |
| Goldsmith (1997) [41] |
HF | - | Amlodipine (oral) |
7.5 | 0.75 | 7 | 7 | 0 | Placebo | 7 | 7 | 0 | 56 (***) | 10 | RCT, crossover | MAP, HR | Sphygmo-manometry, echo |
| Tomiyama (1997) [33] |
HT | - | Nifedipine (oral) |
30 | 0.5 | 13 | 13 | 0 | Acebutolol | 9 | 9 | 0 | 46 (7) |
1095 | Retrospective cohort | SBP, DBP, LVEF | Sphygmo-manometry, echo |
| Seki (1996) [32] | MI | - | Nifedipine (sublingual) |
10 | 0.17 | 8 | 7 | 1 | - | - | - | - | 63 (10) | 0.021 | Prospective cohort | HR, LVEF | Catheterization |
| Naritomi (1995) [55] | HTN | - | Nitrendipine (oral) |
10 | 0.25 | 10 | 7 | 3 | - | - | - | - | 60.5 (***) | 56 | Prospective cohort | SBP, DBP | Sphygmo-manometry |
| Risoe (1993) [31] | HF, MI | - | Nifedipine (sublingual) |
20 | 0.33 | 8 | 4 | 0 | Untreated | 4 | 4 | 0 | **** | 0.03 | RCT | HR | Catheterization |
| Heywood (1991) [39] | HF, CAD | - | Diltiazem (iv) |
25 | 0.07 | 9 | 9 | 0 | - | - | - | - | 68 (9) | 0.02 | Prospective cohort | SBP, DBP, CO, HR | Sphygmo-manometry, echo, ecg, catheterization |
| Sheiban (1991) [30] |
HTN | - | Nifedipine (oral) |
52 | 0.87 | 7 | 7 | 0 | Untreated | 10 | 10 | 0 | 41 (8.1) | 180 | RCT | LVEF | Echo, ecg, sphygmo-manometry |
| Lacidipine (oral) |
5 | 0.83 | 8 | 8 | 0 | ||||||||||||
| Bekheit (1990) [29] |
MI | - | Diltiazem (oral) |
180 | 0.5 | 9 | 0 | 0 | Metoprolol | 8 | 8 | 0 | 62 (13) | 6 | RCT | SBP, DBP, HR | Ecg, sphygomomanometry |
| Nifedipine (oral) |
30 | 0.5 | 10 | 0 | 0 | ||||||||||||
| Senda (1990) [38] |
HTN, LVH | - | Diltiazem (oral) |
180 | 0.5 | 9 | 6 | 3 | - | - | - | - | 60 (***) | 180 | Prospective cohort | LVEF | Echo, ecg, sphygomomanomatry |
| Setaro (1990) [52] |
HF, HTN, CAD, MI, DM | W, B | Verapamil (orally) |
256 | 0.36 | 20 | 20 | 0 | Placebo (4 day washout interval after verapamil) | 20 | 20 | 0 | 68 (5) | 32 | RCT, crossover | SBP, DBP, HR, LVEF | Echo, ECG, sphygomomanometry |
| Binetti (1989) [57] |
HF, CAD, | - | Felodipine (iv) |
0.85 | 0.85 | 10 | 10 | 0 | - | - | - | - | 53 (***) | 0.042 | Prospective cohort | SBP, DBP, CO, MAP, HR | Catheterization, ecg, sphygomomanometry |
| Crawford (1989) [28] |
HF | - | Nifedipine (oral) |
66 | 1.1 | 10 | 10 | 0 | Digoxin, hydralazin (same patients, after CCB) |
10 | 10 | 0 | 54 (***) | 30 | RCT | SBP, LVEF, HR | Ecg, |
| La Rovere (1989) [50] |
MI | - | Nicardipine (iv and oral) |
5 (iv) |
0.01 (iv) |
10 | 10 | 0 | - | - | - | - | 54 (9) | 0.0069 (iv) | Prospective cohort | HR | Ecg, |
| 60 (oral) |
0.17 (oral) |
21 (oral) | |||||||||||||||
| McGrath (1989) [58] |
HF | - | Isradipine(oral) | 15 | 0.75 | 9 | 9 | 0 | Placebo | 9 | 9 | 0 | 54 (***) |
84 | RCT | CO, LVEF, HR | Catheterization |
| Szlachcic (1989) [37] |
HTN | - | Diltiazem (oral) |
240 | 0.67 | 13 | 13 | 0 | Placebo | 11 | 11 | 0 | 48 (10) | 112 | RCT | SBP, DBP | Echo, sphygomomanometry, ecg |
| Bostr.m (1988) [36] |
MI | - | Diltiazem (oral) |
120 | 0.33 | 12 | 12 | 0 | - | - | - | - | 61 (***) | 0.104 | Prospective cohort | SBP, HR | Ecg, sphygomomanometry |
| 180 | 0.5 | 14 | |||||||||||||||
| Fisman (1988) [59] |
MI, CAD | - | Gallopamil (oral) |
75 | 0.38 | 9 | 9 | 0 | Placebo | 6 | 6 | 0 | 60.3 (5.5) | 21 | RCT | LVEF | Echo |
| 112.5 | 0.56 | 9 | 9 | 0 | |||||||||||||
| 150 | 0.75 | 8 | 8 | 0 | |||||||||||||
| Mookherjee (1988) [27] |
HF | - | Nifedipine (sublingual) |
80 | 1.33 | 12 | 12 | 0 | - | - | - | - | 55–77 (***) |
1 | Prospective cohort | LVEF, HR | Echo, catheterization |
| Burlew (1987) [49] |
HF, CAD, HTN | - | Nicardipine (oral) |
75 | 0.21 | 10 | 7 | 3 | - | - | - | - | 54 (***) |
9 | Prospective cohort | HR | Catheterization, ecg |
| Fagard (1987) [56] |
HTN | - | Felodipine (oral) |
6 | 0.6 | 10 | 10 | 0 | - | - | - | - | 41 (9) | 0.06 | Prospective cohort | HR, CO | Echo, catheterization |
| Giles (1987) [54] |
HTN, LVH | W, B | Nitrendipine (oral) |
20 | 0.5 | 9 | 9 | 0 | Hydrochlorothiazide | 9 | 0 | 0 | 66 (3) | 56 | RCT | SBP, DBP, HR, LVM, | Echo, sphygomomanometry |
| Sheiban (1987) [26] |
HTN | - | Nifedipine (oral) |
30 | 0.5 | 8 | 8 | 0 | Captopril | 8 | 8 | 0 | 38 (10) | 180 | RCT | SBP, DBP, LVEF, HR | Echo, sphygomomanometry |
| Lahiri (1986) [48] |
HF, MI | - | Nicardipine (oral) |
10 | 0.03 | 10 | 10 | 0 | - | - | - | - | 63 (***) | 0.02 | Prospective cohort | LVEF | Sphygomomanometry |
| 90 | 0.25 | 28 | |||||||||||||||
| Ortiz (1986) [51] |
HTN | - | Verapamil (oral) |
240 | 0.33 | 18 | 5 | 13 | - | - | - | - | 53.6 (***) | 0.125 | Prospective cohort | SBP, DBP, HR, LVEF | Echo, auscultation method |
| Kubo (1985) [24] |
HF | - | Nifedipine (oral) |
10 | 0.17 | 7 | 7 | 0 | - | - | - | - | 60 (***) | 0.08 | Prospective cohort | HR | Catheterization, |
| Nakamura (1985) [25] |
HF | - | Nifedipine (sublingual) |
20 | 0.33 | 8 | 8 | 0 | - | - | - | - | 55 (***) | 0.02 | Prospective cohort | HR | Echo, ecg |
| Silke (1984) [47] |
CAD | - | Nicardipine (iv) |
1.25 | 0.003 | 10 | 10 | 0 | - | - | - | - | 47 (***) | 0.08 | Prospective cohort | HR | Ecg |
| 2.5 | 0.007 | ||||||||||||||||
| 5 | 0.01 | ||||||||||||||||
| 10 | 0.03 | ||||||||||||||||
| Suwa (1984) [35] |
LVH, HF | - | Diltiazem (iv and oral) |
10 (iv) | 0.03 | 13 | 11 | 2 | Propranol | 13 | 11 | 2 | 43 (***) | 0.02 (iv) | Prospective cohort | SBP, DBP, HR | Ecg, echo, sphygomomanometry |
| 180 (oral) |
0.5 | 14 (oral) | |||||||||||||||
| Amende (1983) [22] |
CAD | - | Nifedipine (iv) |
0.1 (iv) | 0.002 | 8 | 8 | 0 | - | - | - | - | 53.5 (***) | 0.007 | Prospective cohort | HR | Ecg |
| Fujita (1983) [46] |
HTN | - | Nicardipine (oral) |
60 | 0.17 | 10 | 10 | 0 | - | - | - | - | 52.1 (1.7) | 120 | Prospective cohort | HR | Echo, sphygomomanometry |
| Paulus (1983) [23] |
LVH, HF | - | Nifedipine (sublingual) |
10 | 0.17 | 10 | 3 | 7 | Nitroprusside | 10 | 3 | 7 | 48.3 (***) | 0.02 | RCT | HR | Echo, ecg |
Data presented as mean ± SD or percentages. DM = diabetes mellitus, HTN = hypertension, HF = heart failure, CAD = coronary artery disease, MI = myocardial infarction, LVH = left ventricular hypertrophy, W = white, B = black, A = Asian, SD = standard deviation, RCT = randomized controlled trial, MAP = mean arterial pressure, SBP = systolic blood pressure, DBP = diastolic blood pressure, HR = heart rate, LVEF = left ventricular ejection fraction, LVM = left ventricular mass, ECG = electrocardiography, echo = echocardiography. * Percentage of maximal dosage for the indication hypertension. Amlodipine 10 mg/day orally [60]; Lercanidipine 20 mg/day [61]; Mibefradil 100 mg/day [62]; Nicardipine 360 mg/24 h [63]; Verapamil 720 mg/day orally [64]; Nifedipine 60 mg/day [65]; Diltiazem 360 mg/day [66]; Nitrendipine 40 mg/day [67]; Lacidipine 6 mg/day [68]; Felodipine 1 mg/24 h (iv) 10 mg/day (orally) [69]; Isradipine 20 mg/day [70]; Gallopamil 200 mg/day [71]. ** Control group: other antihypertensive treatment, placebo or non-drug intervention. *** SD not reported. **** Not mentioned.
3.3. Quality Assessment
Table 3 summarizes the quality assessment per domain according to the RoB2 tool [16]. Six studies had a low overall risk of bias [34,40,52,54,58,59]. Twenty-eight studies were rated with a high overall risk of bias [22,23,24,25,27,28,29,30,31,32,35,36,38,39,41,43,44,45,47,48,49,50,51,53,55,56,57,59]. The remaining four studies were scored as having some concerns [26,33,37,46].
Table 3.
Quality assessment.
| Random Sequence Allocation (Selection Bias) | Allocation Concealment (Selection Bias) | Incomplete Outcome Data (Attrition Bias) | Measure-ments Outcomes (Detection Bias) | Selective Reporting (Reporting Bias) | Overall Bias | |
|---|---|---|---|---|---|---|
| Lindqvist et al. (2007) [42] | Low | High | High | Low | High | High |
| McGrath et al. (1989) [58] | Low | Low | Low | Low | Low | Low |
| Heywood et al. (1991) [39] | High | Some concerns | Low | Some concerns | Low | High |
| Binetti et al. (1989) [57] | High | Some concerns | Low | Low | Low | High |
| Fagard et al. (1987) [56] | High | Some concerns | Low | Low | Low | High |
| Mehlum et al. (2020) [45] | High | Low | Low | Low | Low | High |
| Baysal et al. (2017) [44] | Low | High | Low | High | High | High |
| Thuc Sinh et al. (2015) [43] | High | Some concerns | Low | Some concerns | Low | High |
| Gottdiener et al. (1998) [40] | Low | Low | Low | Low | Low | Low |
| Tomiyama et al. (1997) [33] | Some concerns | Low | Low | Low | Low | Some concerns |
| Naritomi et al. (1995) [55] | High | Low | Low | Low | Low | High |
| Setaro et al. (1990) [52] | Low | Low | Low | Low | Low | Low |
| Bekheit et al. (1990) [29] | High | Low | Low | Low | Low | High |
| Szlachcic et al. (1989) [37] | Low | Low | Low | Low | Some concerns | Some concerns |
| Sheiban et al. (1987) [26] | Low | Some concerns | Low | Low | Low | Some concerns |
| Giles et al. (1987) [54] | Low | Low | Low | Low | Low | Low |
| Ortiz et al. (1986) [51] | High | Low | Low | Low | Low | High |
| Suwa et al. (1984) [35] | High | Some concerns | Low | Low | Low | High |
| Seki et al. (1996) [32] | High | Low | Low | Low | Low | High |
| Sheiban et al. (1991) [30] | High | Low | Low | Low | Low | High |
| Senda et al. (1990) [38] | High | Low | Low | Low | Low | High |
| Crawford et al. (1989) [28] | High | Some concerns | Low | Low | High | High |
| Fisman et al. (1988) [59] | Low | Low | Low | Low | Low | Low |
| Mookherjee et al. (1988) [27] | High | Low | Low | Low | Low | High |
| Lahiri et al. (1986) [48] | High | Low | Low | Low | Low | High |
| Petrella et al. (2000) [53] | High | Low | Low | Low | Low | High |
| Burggraaf et al. (1998) [34] | Low | Low | Low | Low | Low | Low |
| Risoe et al. (1993) [31] | High | Low | Low | Low | Low | High |
| La Rovere et al. (1989) [50] | High | Some concerns | Low | Low | Low | High |
| Bostrom et al. (1988) [36] | High | Some concerns | Low | Low | Low | High |
| Burlew et al. (1987) [49] | High | Low | Low | Low | Some concerns | High |
| Kubo et al. (1985) [24] | High | Some concerns | Low | Low | Some concerns | High |
| Silke et al. (1984) [47] | High | Low | Low | Low | Low | High |
| Fujita et al. (1983) [46] | Some concerns | Some concerns | Low | Low | Low | Some concerns |
| Amende et al. (1983) [22] | High | Some concerns | Low | Low | Low | High |
| Paulus et al. (1983) [23] | High | Some concerns | Low | Low | Low | High |
| Goldsmith et al. (1997) [41] | High | Low | Low | Low | Low | High |
| Nakamura et al. (1985) [25] | High | Some concerns | Low | Low | Low | High |
3.4. Systolic Blood Pressure
The mean difference and relative change from baseline in percentage for systolic blood pressure (SBP) are reported in Table 4 and Figure 2. The mean SBP in the female population was 162.6 mmHg (95% CI 156.9; 168.2) and the mean SBP in the male population was 146.0 mmHg (95% CI 140.1; 151.8) (p < 0.0001). In females as compared to males, SBP decreased by −27.6 mmHg (95% CI −36.4; −18.8) (%change −17.1 (95% CI −22.5; −11.6)) versus a decrease of 14.4 mmHg (95% CI −19.0; −9.9) (%change, −9.8 (95% CI −12.9; −6.7)). This change was statistically significant between sexes (p-value = 0.009). Heterogeneity was moderate to high in female (I2 = 77%) and high in male (I2 = 83%) data. Heterogeneity in SBP response was significantly affected by the CCB diltiazem, delineated as a clinical source of heterogeneity (Table 5).
Table 4.
Pooled changes in cardiovascular and haemodynamic parameters for females and males.
| Parameter | Females | Males | |
|---|---|---|---|
| SBP (mmHg) | MD % |
−27.6 (−36.4; −18.8) −17.1 (−22.5; −11.6) |
−14.4 (−19.0; −9.9) −9.8 (−12.9; −6.7) |
| DBP (mmHg) | MD % |
−14.1 (−18.8; −9.3) −15.2 (−20.3; −10.1) |
−10.6 (−14.0; −7.3) −11.2 (−14.8; −7.7) |
| MAP (mmHg) | MD % |
−13.9 (−14.5; −13.2) −12.5 (−13.1; −12.0) |
−8.7 (−14.1; −3.3) −8.9 (−14.5; −3.4) |
| HR (bpm) | MD % |
−1.8 (−2.5; −1.2) −2.5 (−3.4; −1.6) |
0.3 (−1.2; 1.8) 0.4 (−1.7; 2.4) |
| CO (L/min) | MD % |
−0.2 (−1.8; 1.4) −4.0 (−35.8; 27.7) |
0.8 (0.1; 1.6) 18.2 (2.1; 34.2) |
| LVEF (%) | MD % |
−7.8 (−12.4; −3.2) −11.4 (−18.0; −4.7) |
2.9 (−0.4; 6.1) 5.3 (−0.7; 11.3) |
| LVM (g) | MD % |
- - |
−15.9 (−48.2; 16.4) −4.9 (−15.0; 5.1) |
Values are reported as mean difference (MD) and relative change (%) compared to baseline with 95% CI. SBP = systolic blood pressure, DBP = diastolic blood pressure, MAP = mean arterial pressure, HR = heart rate, CO = cardiac output, LVEF = left ventricular ejection fraction, LVM = left ventricular mass.
Figure 2.
Forest plot of systolic blood pressure (SBP) change in mmHg after CCB use compared to baseline for females and males [26,28,29,33,35,36,37,39,40,43,44,45,51,52,54,55,57]. MD = mean difference.
Table 5.
p-values of meta-regression analysis.
| Sources of Heterogeneity | SBP | DBP | MAP | HR | CO | LVEF |
|---|---|---|---|---|---|---|
| Diltiazem | 0.0033 | 0.0169 | - | 0.3344 | 0.6689 | - |
| Felodipine | 0.4463 | 0.8744 | 0.8968 | 0.0013 | 0.0179 | - |
| Isradipine | - | - | - | 0.1107 | 0.4874 | 0.9644 |
| Gallopamil | - | - | - | - | - | 0.7769 |
| Mibefradil | - | - | - | <0.0001 | 0.0469 | - |
| Lacidipine | - | - | - | - | - | 0.6467 |
| Nicardipine | - | - | - | 0.1601 | - | 0.4624 |
| Lercanidipine | 0.7665 | 0.2490 | - | - | - | - |
| Nifedipine | 0.3323 | 0.8709 | 0.0059 | 0.0005 | - | 0.7478 |
| Nitrendipine | 0.8774 | 0.8796 | - | 0.9458 | - | - |
| Verapamil | 0.4007 | 0.4838 | - | 0.8234 | - | 0.2200 |
| Low quality | 0.0765 | 0.0954 | 0.8467 | 0.8485 | - | 0.8701 |
| Moderate quality | 0.2036 | 0.8437 | 0.8034 | 0.3195 | 0.0015 | 0.8598 |
| Treatment duration | 0.5907 | 0.6152 | 0.3061 | 0.5232 | 0.0075 | 0.6331 |
| % max dose | 0.1842 | 0.7350 | 0.8570 | 0.2724 | 0.0605 | 0.2966 |
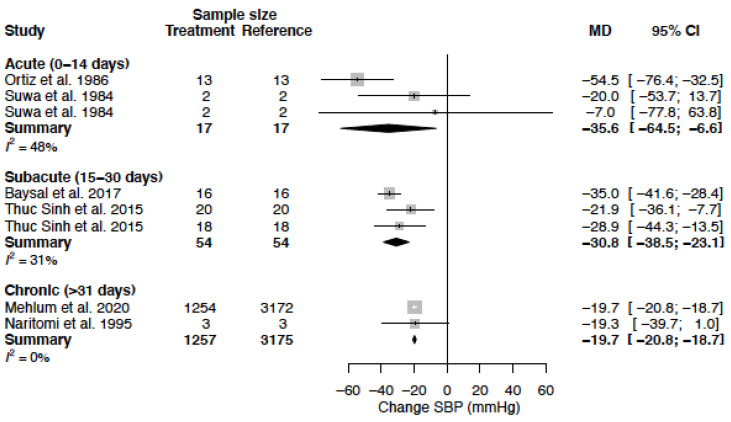
The mean difference for SBP by treatment duration is reported in Table 6. In females, the observed decrease in SBP is greatest in the acute and sub-acute treatment phase, while in males the observed decrease is largest during sub-acute and chronic treatment (Figure 3, Figure 4 and Figure 5).
Table 6.
Pooled changes in cardiovascular and haemodynamic parameters by treatment duration for females and males.
| Parameter | Females | Males | |
|---|---|---|---|
| SBP (mmHg) | MD acute | −35.6 (−64.5; −6.6) | −9.4 (−15.2; −3.7) |
| MD sub-acute | −30.8 (−38.5; −23.1) | −21.8 (−28.8; −14.7) | |
| MD chronic | −19.7 (−20.8; −18.7) | −15.1 (−23.8; −6.4) | |
| DBP (mmHg) | MD acute | −20.4 (−32.2; −8.6) | −6.9 (−9.7; −4.1) |
| MD sub-acute | −14.6 (−23.7; −5.5) | −15.1 (−20.0; −10.2) | |
| MD chronic | −10.9 (−11.5; −10.3) | −10.9 (−16.1; −5.8) |
Values are reported as mean difference (MD) compared to baseline with 95% CI. Acute = 0–14 days, sub-acute = 15–30 days, chronic = >31 days, SBP = systolic blood pressure, DBP = diastolic blood pressure.
Figure 3.
Forest plot of systolic blood pressure (SBP) change in mmHg after acute, sub-acute and chronic CCB use compared to baseline for females [35,43,44,45,51,55]. MD = mean difference.
Figure 4.
Forest plot of systolic blood pressure (SBP) change in mmHg after acute, sub-acute and chronic CCB use compared to baseline for males [26,28,29,33,35,36,37,39,40,43,44,45,51,52,54,55,57]. MD = mean difference.
Figure 5.
Meta-regression curve of systolic blood pressure (SBP) by treatment duration (days). Every circle represents one article and the size represents the number of participants included in the study, shown as a small or larger circle.
3.5. Diastolic Blood Pressure
The mean DBP in the female population was 93.1 mmHg (95% CI 88.0; 98.2) and the mean DBP in the male population was 93.2 mmHg (95% CI 89.1; 97.3) (p-value = 0.973). DBP decreased by −14.1 (95% CI −18.8; −9.3) (%change, −15.2 (95% CI −20.3; −10.1)) in females as compared to −10.6 mmHg (95% CI −14.0; −7.3) (%change −11.2 (95% CI −14.8; −7.7)) in males (Table 4, Figure 6). This effect did not reach statistical significance between sexes (p-value = 0.244). Heterogeneity was moderate to high in female (I2 = 61%) and high in male (I2 = 94 %) data. Only one clinical source of heterogeneity (diltiazem) significantly affected the change in DBP (Table 5). The mean difference for DBP by treatment duration is reported in Table 6. The observed decrease in DBP is, in females, the largest after acute and sub-acute treatment. For males, the effect is the greatest in the sub-acute phase (Figure 7, Figure 8 and Figure 9).
Figure 6.
Forest plot of diastolic blood pressure (DBP) change in mmHg after CCB use compared to baseline for females and males [26,29,33,35,37,39,40,43,44,45,51,52,54,55,57]. MD = mean difference.
Figure 7.
Forest plot of diastolic blood pressure (DBP) change in mmHg after acute, sub-acute and chronic CCB use compared to baseline for females [35,43,44,45,51,55]. MD = mean difference.
Figure 8.
Forest plot of diastolic blood pressure (DBP) change in mmHg after acute, sub-acute and chronic CCB use compared to baseline for males [26,29,33,35,37,39,40,43,44,45,51,52,54,55,57]. MD = mean difference.
Figure 9.
Meta-regression curve of diastolic blood pressure (DBP) by treatment duration (days). Every circle represents one article and the size represents the number of participants included in the study, shown as a small or larger circle.
3.6. Mean Arterial Pressure
The mean MAP in the female population was 110.6 mmHg (95% CI 110.2; 111.0) and the mean MAP in the male population was 95.6 mmHg (95% CI 82.7; 108.5) (p-value = 0.023). In females, MAP changed by −13.9 mmHg (95% CI −14.5; −13.2) (%change −12.5 (95% CI −13.1; −12.0)) as compared to −8.7 mmHg (95% CI −14.1; −3.3) (%change, −8.9 (95% CI −14.5; −3.4)) in males (Table 4, Figure 10) but the difference between sexes (p-value = 0.061) did not reach statistical significance. Heterogeneity could not be calculated in females as only one study was included. Heterogeneity was high (I2 = 85%) in males. The clinical source of heterogeneity detected by meta-regression analysis was nifedipine. Methodological sources of heterogeneity did not significantly contribute to the observed change in MAP (Table 5).
Figure 10.
Forest plot of mean arterial pressure (MAP) change in mmHg after CCB use compared to baseline for females and males [34,41,45,57]. MD = mean difference.
3.7. Heart Rate
The mean HR in the female population was 74.8 bpm (95% CI 68.0; 81.7) and the mean HR in the male population was 74.4 bpm (95% CI 72.9; 76.0) (p-value = 0.919). Heart rate (beats per minute (bpm)) decreases in females (−1.8 bpm (95% CI −2.5; −1.2) (%change −2.5 (95% CI −3.4; −1.6)). In males, HR did not change appreciably (0.3 bpm (95% CI −1.2; 1.8) (%change 0.4 (95% CI −1.7; 2.4)) (Table 4, Figure 11). This sex-difference is significant (p-value = 0.011). The heterogeneity is low in females I2= 0% and moderate in males I2 = 72%. Three clinical sources of heterogeneity (felodipine, mibefradil and nifedipine) significantly affected the change in HR (Table 5).
Figure 11.
Forest plot of heart rate (HR) change in bmp after CCB use compared to baseline for females and males [22,23,24,25,26,27,28,29,31,32,34,35,36,39,40,41,42,45,46,47,49,50,51,52,53,54,56,57]. MD = mean difference.
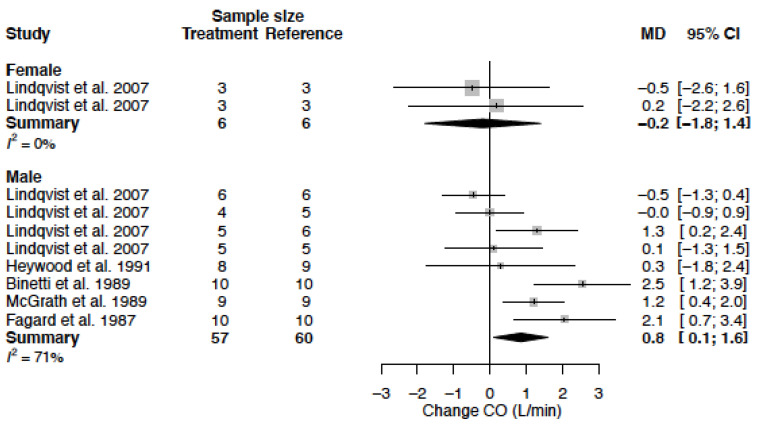
3.8. Cardiac Output
The mean CO in females was 5.0 L/min (95% CI 3.6; 6.5) and in males 4.7 L/min (95% CI 4.2; 5.1) (p-value = 0.635). A comparable effect between females and males was observed in CO after CCB use, which was also not statistically significant between sexes (p-value = 0.244). CO remained unaltered in females (−0.2 L/min (95% −1.8; 1.4) (%change −4.0 (95% CI −35.8; 27.7)) but significantly increased in males by 0.8 L/min (95% CI 0.1; 1.6) (%change 18.2 (95% CI 2.1; 34.2)) (Table 4, Figure 12). Heterogeneity is low in female data (I2 = 0%), as one study could be included, and moderate to high in male data (I2 = 71%). The clinical source of heterogeneity detected by meta-regression analysis was felodipine and mibefradil. The moderate study quality and treatment duration, both methodological sources of heterogeneity, did also significantly contribute to the observed change in CO (Table 5).
Figure 12.
Forest plot of cardiac output (CO) change in L/min after CCB use compared to baseline for males and females [39,42,56,57,58]. MD = mean difference.
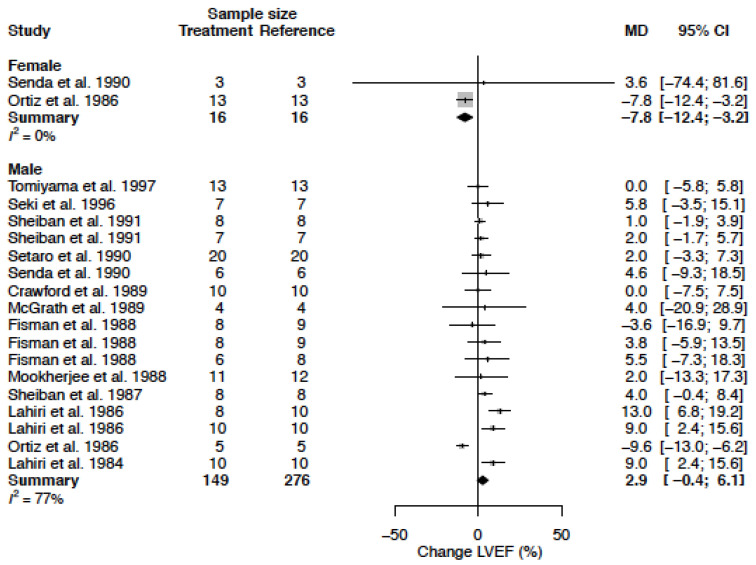
3.9. Left Ventricular Ejection Fraction
The mean LVEF in the female population was 68.7 % (95% CI 65.9; 71.5) and the mean LVEF in the male population was 51.2% (95% CI 42.0; 60.4) (p-value = 0.0004). LVEF change was opposite in females as compared to males (Table 4, Figure 13). In females, a decrease in LVEF by −7.8 % (95% CI −12.4; −3.2) (%change −11.4 (95% CI −18.0; −4.7) was observed, whereas in males, LVEF remained unaltered (2.9% (95% CI −0.4; 6.1) (%change 5.3 (95% CI −0.7; 11.3)) (Table 3). The sex-difference was statistically significant (p-value < 0.001). Heterogeneity is low in female data (I2 = 0%) and moderate to high in male data (I2 = 77%). The change in LVEF was not significantly affected by clinical or methodological sources of heterogeneity (Table 5).
Figure 13.
Forest plot of left ventricular ejection fraction (LVEF) change in % after CCB use compared to baseline for females and males [26,27,28,30,32,33,38,48,51,52,58,59]. MD = mean difference.
3.10. Left Ventricular Mass
LVM could only be extracted from two studies, and only from males. The mean LVM in the population was 322.5 g (95% CI 304.2; 340.8). In these studies, LVM remained unaltered by CCB (−15.9 g (95% CI −48.2; 16.4) (%change −4.9 (95% CI −15.0; 5.1))) (Table 4, Figure 14). Heterogeneity was low in these studies (I2 = 0%).
Figure 14.
Forest plot of left ventricular mass (LVM) change in grams after CCB use compared to baseline for males [40,54]. MD = mean difference.
4. Discussion
In this systematic review and meta-analysis, we observed that CCB treatment significantly lowered SBP, DBP and MAP in both sexes but that the decrease is greater in females as compared to males. HR and LVEF decreased in females, while remaining unaltered in males. In females, prolonged treatment duration slightly attenuated the blood pressure-lowering effect, whereas in males the effect on blood pressure was the highest after 14 days of treatment.
System-biological cardiovascular and regulatory differences between sexes are likely underlying possible differences in responsiveness to CCB in the treatment of hypertension [13,72,73]. These differences may be expected in the most important blood pressure regulatory systems amongst sympathetic nervous system (SNS), renin-angiotensin system [29], endothelin-1 (ET-1), vasomotion and sex hormones [73].
The SNS regulates the vascular tone by mediating vasoconstriction and vasodilation by transmitting norepinephrine targeting alpha- and beta adrenoceptors [74,75]. Studies have shown that sensitivity to adrenergic stimulation is sex-specific, where males have greater sensitivity to norepinephrine compared to females, leading to more vasoconstriction in males [76,77,78,79]. These findings may be affected by the presence of estrogen receptors on endothelial and vascular smooth muscle cells. Stimulation by estrogen results in weaker vascular reactivity to adrenergic stimulation [79].
The RAS consists of a classic and non-classical pathway. The classic pathway is currently defined as the ACE-Ang II AT1R axis promoting vasoconstriction, sodium, water retention as well as inflammation, oxidative stress, cellular growth, and fibrosis. On the contrary, the non-classical RAS pathway is composed primarily of the angiotensin-(1-7)-ACE2-MasR/AT2R pathways which opposes the actions of the classical stimulated Ang II-AT1R axis by causing an increase in nitric oxide and prostaglandins, mediating vasodilation, natriuresis, diuresis, and lowering oxidative stress. Female estrogen mediates downregulation of angiotensin II and upregulation of angiotensin-(1-7)-ACE2-MasR/AT2R pathways causing vasodilatation, whereas male testosterone stimulates the classic pathway leading to vasoconstriction [80,81,82,83,84,85,86]. Another important vasoconstrictor is ET-1, which is secreted by endothelial cells. Sex hormones influence the release of ET-1 in opposite ways, whereby testosterone causes an increase in ET-1 release and estrogen and progesterone cause inhibition of ET-1 release [73,87].
The prevalence of hypertension increases with age in both sexes. Remarkable is the higher incidence of hypertension in younger males compared to younger females. However, as age increases, more females suffer from hypertension compared to age-matched men. This may depend on menopause causing a drop in estrogen [88,89]. Clinical studies suggest that estrogen protects against hypertension by stimulation of the vasodilator pathway mediated by nitric oxide and prostacyclin and inhibition of the vasoconstrictor pathway mediated by the sympathetic nervous system and angiotensin [90,91].
Pharmacologically, CCB act by binding to the L-type long-acting voltage gated-calcium channels in the heart, vascular smooth muscles (coronary and peripheral arterials) and pancreas. Blocking the calcium channels leads to inhibition of Ca2+ influx into excitable cells preventing Ca2+ to serve as an intracellular messenger. As intracellular Ca2+ leads to vasoconstriction, CCB leads to vasodilatation.
Two types of CCBs are known. First are the non-dihydropyridines, amongst verapamil, primarily influencing the sinoatrial and atrioventricular node resulting in a reduction in cardiac conduction and contractility. Therefore, this group is mostly used for treating hypertension, reduction in oxygen demand and controlling heart rate in arrhythmias. Verapamil is known to be one of the first well characterized P-glycoprotein (P-gp) substrates, as well as a substrate of CYP3A4 resulting in extensive first-pass hepatic and intestinal metabolism [13]. The second group is dihydropyridines, for example, amlodipine. Dihydropyridines are known for their vasodilating effect on peripheral arterials, useful in hypertension treatment, migraines and cranial vasospasms [12,14]. Amlodipine is not generally considered to be a P-gp substrate and has low rates of first-pass metabolism and a high bioavailability [13].
Splitting the analysis into a non-dihydropyridines and a dihydropyridines group results in comparable findings according for BP. However, HR seems to decrease more in males compared to females using non-dihydropyridines. LVEF, CO and LVM are hard to compare as only few included studies investigated those parameters (Table S2).
Clinically, previous studies comparing the effects of CCB between females and males show sex differences for some types of CCB, which might relate to differences in metabolization and metabolic rate. In a study on the influence of sex on the pharmacokinetics of verapamil and norverapamil, Dadashzadeh et al., showed the blood concentration of norverapamil compared to verapamil to be significantly higher in females [92]. The substrate CYP3A4 metabolizes verapamil to norverapamil. As the mean residence time of norverapamil was significantly shorter in females compared to males, the study concluded that the production of norverapamil is more extensive in females and therefore a sex-dependent process, most likely because of a higher activity of CYP3A4 or lower activity of P-gp leading to faster clearance. Higher expression of CYP3A4 in females has been found in previous studies [13,93,94,95]. However, some studies showed the contrary; Cummins at al. suggest that sex-specific differences depend on P-gp activity [96]. If a drug is not only metabolized by CYP3A4 but is also a substrate of P-gp, intracellular hepatic levels will rise, resulting in more opportunities to encounter its metabolizing enzymes and higher clearance rates, even when enzyme protein levels are equal in sexes. Cummins concludes that intracellular drugs concentration will not differ between sexes if a drug is a substrate of CYP3A4 but not a P-gp substrate.
Several studies have assessed the efficacy and safety of amlodipine which has been safely used in patients with New York Heart Association classes II and III heart failure. Kloner et al. conducted a prospective, multicentric trial including 1084 patients (mean age 55.5 years; 35% females and 65% males) with mild to moderate hypertension and showed greater DBP changes from baseline in females (91.4%) compared to males (83.0%) (p-value = 0.001) using amlodipine [97]. These results remained significant after adjusting for baseline differences in, e.g., weight, age and weight normalized dose [97,98]. The authors suggest that this difference in response can be explained by vascular reactivity, distribution, metabolism of the drug and differences in the etiology of hypertension between females and males [13]. Kang et al. conducted a study on the effect of age on the oral clearance of amlodipine [99]. Approximately 210 elderly subjects (mean age females 79 years, males 72 years) were included and showed significant effects for sex. In accordance with previous research on other substrates of CYP3A4, faster clearance in females compared to males was observed. Contributing to the hypothesis that CYP3A4 expression in female liver tissue is greater than in males are many in vivo studies showing a greater decrease in CYP3A4 in aging males compared to aging females [100]. This should be taken into consideration as older patients are most likely to receive antihypertensive medication.
Strengths and Limitations
One of the strengths of our systematic review and meta-analysis is the large number of studies we examined for inclusion. This extensive search resulted in a population of 8202 patients using CCB and having sex-stratified data available. Another strength is that two independent reviewers screened all articles. Furthermore, we used the RoB2-tool, as recommended by Cochrane, to detect forms of bias.
On the other hand, some limitations should be mentioned. First, our meta-analysis included remarkably more male patients compared to female patients (60.2% vs. 39.8%), causing female underrepresentation. However, this is rather a clinical reflection than a lack of our study selection method. Second, the ROB2 tool classified more than half of the included articles as ‘high risk of bias’. This could be explained as the ROB2 tool classifies non-RCTs lower than RCTs. Half of the included articles were prospective non-RCTs and therefore qualified as lower quality, contributing to the high risk of bias. This means that we most likely overestimated the risk of bias for some studies. Third, clinical sources of heterogeneity (use of different CCB) significantly affected the change for some parameters. Treatment duration, a methodological source of heterogeneity, significantly contributed to the observed change in CO. Furthermore, in a lot of the included studies patients received co-medication during the study protocol. On one hand, this could have biased the observed effect. On the other hand, since co-medication was used before and after CCB initiation, this bias could be considered constant. Maybe more importantly, this could be a reliable representation of reality, contributing to the external validity of our study.
5. Conclusions and Recommendations
In individuals with hypertension, CCB lowers BP in both sexes. However, the decrease, especially in SBP, is significantly greater in females as compared to males. The lowering effect on HR and LVEF is only significant in females. When normalizing blood pressure, differences in these sex-dependent effects may be taken into account.
Acknowledgments
The authors greatly thank the biomedical sciences students (Lieke Knapen, Rosamel Abeka, Thom Knoben, Samantha Schwengle, Ryan van den Akker) and medical students of the Maastricht University honours program (Cédric Dikovec, Jan Wiesenberg, Mohamad Almutairi) involved in data extraction and risk of bias assessment.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/biomedicines11061622/s1, Table S1: Literature search: strategy for PubMed (NCBI) and Embase (Ovid) databases. Table S2: Chemical-pharmacological distinction dihydropyridines (D) and non-dihydropyridines (ND). Table S2.1: Pooled changes in cardiovascular and haemodynamic parameters for females and males using dihydropyridines. Table S2.2: Pooled changes in cardiovascular and haemodynamic parameters for females and males using non-dihydropyridines. Table S2.3: P-values males vs. females distinction dihydropyridines (D) and non-dihydropyridines (ND).
Author Contributions
E.M.v.L., E.W.P.V., M.A.M.V., N.W., D.A.M.M., S.A.J.S.L., Z.M.-A., S.d.H., M.E.A.S. and C.G.-D. performed the search, study selection and data extraction. E.M.v.L.: analyzed the data. E.M.v.L. wrote the initial draft of the paper, revised the paper and finalized the manuscript. E.M.v.L. wrote the paper. M.E.A.S., C.G.-D. initiated the project, developed the idea and coordinated the writing process. E.M.v.L., N.W., M.E.A.S., C.G.-D. wrote the paper and critically reviewed the content. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No individual patient data are included in this study. Search strategy and results of included papers are presented within the manuscript and are available at the corresponding author upon request.
Conflicts of Interest
All other authors declare no interest. Furthermore, there are no known conflicts of interest associated with this publication, and there has been no significant financial support for this work that could have influenced its outcome. We confirm that the manuscript has been read and approved by all named authors and that there are no other persons who satisfied the criteria for authorship but are not listed. We further confirm that all have approved the order of authors listed in the manuscript.
Funding Statement
This research received no external funding.
Footnotes
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
References
- 1.World Health Organization Cardiovascular Diseases (CVDs) 2021. [(accessed on 8 July 2022)]. Available online: https://www.who.int/en/news-room/fact-sheets/detail/cardiovascular-diseases-(cvds)
- 2.World Health Organization . Hypertension. WHO; Geneva, Switzerland: 2021. [Google Scholar]
- 3.Di Giosia P., Passacquale G., Petrarca M., Giorgini P., Marra A.M., Ferro A. Gender differences in cardiovascular prophylaxis: Focus on antiplatelet treatment. Pharmacol. Res. 2017;119:36–47. doi: 10.1016/j.phrs.2017.01.025. [DOI] [PubMed] [Google Scholar]
- 4.Bruno R.M., Varbiro S., Pucci G., Nemcsik J., Lonnebakken M.T., Kublickiene K., Schluchter H., Park C., Mozos I., Guala A., et al. Vascular function in hypertension: Does gender dimension matter? J. Hum. Hypertens. 2023 doi: 10.1038/s41371-023-00826-w. [DOI] [PubMed] [Google Scholar]
- 5.Carey R.M., Muntner P., Bosworth H.B., Whelton P.K. Prevention and Control of Hypertension: JACC Health Promotion Series. J. Am. Coll. Cardiol. 2018;72:1278–1293. doi: 10.1016/j.jacc.2018.07.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Jin X., Chandramouli C., Allocco B., Gong E., Lam C.S.P., Yan L.L. Women’s Participation in Cardiovascular Clinical Trials From 2010 to 2017. Circulation. 2020;141:540–548. doi: 10.1161/CIRCULATIONAHA.119.043594. [DOI] [PubMed] [Google Scholar]
- 7.Melloni C., Berger J.S., Wang T.Y., Gunes F., Stebbins A., Pieper K.S., Dolor R.J., Douglas P.S., Mark D.B., Newby L.K. Representation of women in randomized clinical trials of cardiovascular disease prevention. Circ. Cardiovasc. Qual. Outcomes. 2010;3:135–142. doi: 10.1161/CIRCOUTCOMES.110.868307. [DOI] [PubMed] [Google Scholar]
- 8.Mosca L., Linfante A.H., Benjamin E.J., Berra K., Hayes S.N., Walsh B.W., Fabunmi R.P., Kwan J., Mills T., Simpson S.L. National study of physician awareness and adherence to cardiovascular disease prevention guidelines. Circulation. 2005;111:499–510. doi: 10.1161/01.CIR.0000154568.43333.82. [DOI] [PubMed] [Google Scholar]
- 9.Regitz-Zagrosek V., Seeland U. Handbook of Experimental Pharmacology. Springer; New York, NY, USA: 2012. Sex and gender differences in clinical medicine. [DOI] [PubMed] [Google Scholar]
- 10.Tamargo J., Rosano G., Walther T., Duarte J., Niessner A., Kaski J.C., Ceconi C., Drexel H., Kjeldsen K., Savarese G., et al. Gender differences in the effects of cardiovascular drugs. Eur. Heart J. Cardiovasc. Pharmacother. 2017;3:163–182. doi: 10.1093/ehjcvp/pvw042. [DOI] [PubMed] [Google Scholar]
- 11.Mozos I., Maidana J.P., Stoian D., Stehlik M. Gender Differences of Arterial Stiffness and Arterial Age in Smokers. Int. J. Environ. Res. Public Health. 2017;14:565. doi: 10.3390/ijerph14060565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.McKeever R.G., Hamilton R.J. Calcium Channel Blockers. StatPearls Publishing; St. Petersburg, FL, USA: 2021. [PubMed] [Google Scholar]
- 13.Ueno K., Sato H. Sex-related differences in pharmacokinetics and pharmacodynamics of anti-hypertensive drugs. Hypertens. Res. 2012;35:245–250. doi: 10.1038/hr.2011.189. [DOI] [PubMed] [Google Scholar]
- 14.Katz A.M. Pharmacology and mechanisms of action of calcium-channel blockers. J. Clin. Hypertens. 1986;2:28S–37S. [PubMed] [Google Scholar]
- 15.Tocci G., Battistoni A., Passerini J., Musumeci M.B., Francia P., Ferrucci A., Volpe M. Calcium channel blockers and hypertension. J. Cardiovasc. Pharmacol. Ther. 2015;20:121–130. doi: 10.1177/1074248414555403. [DOI] [PubMed] [Google Scholar]
- 16.Sterne J.A.C., Savovic J., Page M.J., Elbers R.G., Blencowe N.S., Boutron I., Cates C.J., Cheng H.Y., Corbett M.S., Eldridge S.M., et al. RoB 2: A revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366:l4898. doi: 10.1136/bmj.l4898. [DOI] [PubMed] [Google Scholar]
- 17.JPT Higgins J.T. Cochrane Handbook for Systematic Reviews of Interventions. 2nd ed. Wiley-Blackwell; Hoboken, NJ, USA: 2020. [Google Scholar]
- 18.DerSimonian R., Laird N. Meta-analysis in clinical trials. Control Clin. Trials. 1986;7:177–188. doi: 10.1016/0197-2456(86)90046-2. [DOI] [PubMed] [Google Scholar]
- 19.Egger M., Davey Smith G., Schneider M., Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315:629–634. doi: 10.1136/bmj.315.7109.629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Schwarzer G. Meta: An R package for meta-analysis. R News. 2007;7:40–45. [Google Scholar]
- 21.R Core Team . R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing; Vienna, Austria: 2016. [Google Scholar]
- 22.Amende I., Simon R., Hood W.P., Jr., Hetzer R., Lichtlen P.R. Intracoronary nifedipine in human beings: Magnitude and time course of changes in left ventricular contraction/relaxation and coronary sinus blood flow. J. Am. Coll. Cardiol. 1983;2:1141–1145. doi: 10.1016/S0735-1097(83)80341-6. [DOI] [PubMed] [Google Scholar]
- 23.Paulus W.J., Lorell B.H., Craig W.E., Wynne J., Murgo J.P., Grossman W. Comparison of the effects of nitroprusside and nifedipine on diastolic properties in patients with hypertrophic cardiomyopathy: Altered left ventricular loading or improved muscle inactivation? J. Am. Coll. Cardiol. 1983;2:879–886. doi: 10.1016/S0735-1097(83)80235-6. [DOI] [PubMed] [Google Scholar]
- 24.Kubo S.H., Fox S.C., Prida X.E., Cody R.J. Combined hemodynamic effects of nifedipine and nitroglycerin in congestive heart failure. Am. Heart J. 1985;110:1032–1034. doi: 10.1016/0002-8703(85)90205-4. [DOI] [PubMed] [Google Scholar]
- 25.Nakamura Y., Ikeda T., Takata S., Yamamoto M., Kitamura T., Hattori N. Effects of nifedipine on forearm vascular resistance and venous capacitance in normal subjects and in patients with congestive heart failure. Int. J. Cardiol. 1985;9:27–36. doi: 10.1016/0167-5273(85)90400-0. [DOI] [PubMed] [Google Scholar]
- 26.Sheiban I., Arcaro G., Covi G., Accardi R., Zenorini C., Lechi A. Regression of cardiac hypertrophy after antihypertensive therapy with nifedipine and captopril. J. Cardiovasc. Pharmacol. 1987;10((Suppl. 10)):S187–S191. doi: 10.1097/00005344-198710100-00062. [DOI] [PubMed] [Google Scholar]
- 27.Mookherjee S., Ashutosh K., Dunsky M., Hill N., Vardan S., Smulyan H., Warner R. Nifedipine in chronic cor pulmonale: Acute and relatively long-term effects. Clin. Pharmacol. Ther. 1988;44:289–296. doi: 10.1038/clpt.1988.152. [DOI] [PubMed] [Google Scholar]
- 28.Crawford M.H., Wilson R.S., O’Rourke R.A., Vittitoe J.A. Effect of digoxin and vasodilators on left ventricular function in aortic regurgitation. Int. J. Cardiol. 1989;23:385–393. doi: 10.1016/0167-5273(89)90199-X. [DOI] [PubMed] [Google Scholar]
- 29.Bekheit S., Tangella M., El-Sakr A., Rasheed Q., Craelius W., El-Sherif N. Use of heart rate spectral analysis to study the effects of calcium channel blockers on sympathetic activity after myocardial infarction. Am. Heart J. 1990;119:79–85. doi: 10.1016/S0002-8703(05)80085-7. [DOI] [PubMed] [Google Scholar]
- 30.Sheiban I., Arosio E., Montesi G., Tonni S., Montresor G., Lechi A. Remodeling of left ventricular geometry and function induced by lacidipine and nifedipine SR in mild-to-moderate hypertension. J. Cardiovasc. Pharmacol. 1991;17((Suppl. 4)):S68–S74. doi: 10.1097/00005344-199117041-00015. [DOI] [PubMed] [Google Scholar]
- 31.Risoe C., Smiseth O.A., Rootwelt K., Sire S., Simonsen S. Effect of nifedipine on splanchnic and pulmonary vascular capacitance. Clin. Physiol. 1993;13:247–255. doi: 10.1111/j.1475-097X.1993.tb00324.x. [DOI] [PubMed] [Google Scholar]
- 32.Seki K., Katayama K., Hiro T., Yano M., Miura T., Kohno M., Fujii T., Matsuzaki M. The effect of nifedipine on ventriculoarterial coupling in old myocardial infarction. Jpn. Circ. J. 1996;60:35–42. doi: 10.1253/jcj.60.35. [DOI] [PubMed] [Google Scholar]
- 33.Tomiyama H., Doba N., Kushiro T., Yamashita M., Yoshida H., Kanmatsuse K., Kajiwara N., Hinohara S. Effects of long-term antihypertensive therapy on physical fitness of men with mild hypertension. Hypertens. Res. 1997;20:105–111. doi: 10.1291/hypres.20.105. [DOI] [PubMed] [Google Scholar]
- 34.Burggraaf J., Schoemaker R.C., Kroon J.M., Cohen A.F. The influence of nifedipine and captopril on liver blood flow in healthy subjects. Br. J. Clin. Pharmacol. 1998;45:447–451. doi: 10.1046/j.1365-2125.1998.00709.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Suwa M., Hirota Y., Kawamura K. Improvement in left ventricular diastolic function during intravenous and oral diltiazem therapy in patients with hypertrophic cardiomyopathy: An echocardiographic study. Am. J. Cardiol. 1984;54:1047–1053. doi: 10.1016/S0002-9149(84)80142-3. [DOI] [PubMed] [Google Scholar]
- 36.Bostrom P.A., Lilja B., Johansson B.W., Meier K. The effect of oral diltiazem on left ventricular performance in postinfarction patients. Clin. Cardiol. 1988;11:739–742. doi: 10.1002/clc.4960111103. [DOI] [PubMed] [Google Scholar]
- 37.Szlachcic J., Tubau J.F., Vollmer C., Massie B.M. Effect of diltiazem on left ventricular mass and diastolic filling in mild to moderate hypertension. Am. J. Cardiol. 1989;63:198–201. doi: 10.1016/0002-9149(89)90285-3. [DOI] [PubMed] [Google Scholar]
- 38.Senda Y., Tohkai H., Kimura M., Shida Y., Tutumi S., Koshiyama H., Tanaka T., Shimaji Y., Nogita T. ECG-gated cardiac scan and echocardiographic assessments of left ventricular hypertrophy: Reversal by 6-month treatment with diltiazem. J. Cardiovasc. Pharmacol. 1990;16:298–304. doi: 10.1097/00005344-199008000-00017. [DOI] [PubMed] [Google Scholar]
- 39.Heywood J.T., Graham B., Marais G.E., Jutzy K.R. Effects of intravenous diltiazem on rapid atrial fibrillation accompanied by congestive heart failure. Am. J. Cardiol. 1991;67:1150–1152. doi: 10.1016/0002-9149(91)90885-O. [DOI] [PubMed] [Google Scholar]
- 40.Gottdiener J.S., Reda D.J., Williams D.W., Materson B.J., Cushman W., Anderson R.J. Effect of single-drug therapy on reduction of left atrial size in mild to moderate hypertension: Comparison of six antihypertensive agents. Circulation. 1998;98:140–148. doi: 10.1161/01.CIR.98.2.140. [DOI] [PubMed] [Google Scholar]
- 41.Goldsmith S.R. Effect of amlodipine on norepinephrine kinetics and baroreflex function in patients with congestive heart failure. Am. Heart J. 1997;134:13–19. doi: 10.1016/S0002-8703(97)70101-7. [DOI] [PubMed] [Google Scholar]
- 42.Lindqvist M., Kahan T., Melcher A., Ekholm M., Hjemdahl P. Long-term calcium antagonist treatment of human hypertension with mibefradil or amlodipine increases sympathetic nerve activity. J. Hypertens. 2007;25:169–175. doi: 10.1097/HJH.0b013e3280104dba. [DOI] [PubMed] [Google Scholar]
- 43.Cao T.S., Huynh V.M., Tran V.H. Effects of lercanidipine versus amlodipine in hypertensive patients with cerebral ischemic stroke. Curr. Med. Res. Opin. 2015;31:163–170. doi: 10.1185/03007995.2014.964855. [DOI] [PubMed] [Google Scholar]
- 44.Baysal S.S., Pirat B., Okyay K., Bal U.A., Ulucam M.Z., Oztuna D., Muderrisoglu H. Treatment-associated change in apelin concentration in patients with hypertension and its relationship with left ventricular diastolic function. Anatol. J. Cardiol. 2017;17:125–131. doi: 10.14744/AnatolJCardiol.2016.7035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Mehlum M.H., Liestol K., Kjeldsen S.E., Wyller T.B., Julius S., Rothwell P.M., Mancia G., Parati G., Weber M.A., Berge E. Blood Pressure-Lowering Profiles and Clinical Effects of Angiotensin Receptor Blockers Versus Calcium Channel Blockers. Hypertension. 2020;75:1584–1592. doi: 10.1161/HYPERTENSIONAHA.119.14443. [DOI] [PubMed] [Google Scholar]
- 46.Fujita T., Noda H. Hemodynamic changes associated with long-term antihypertensive therapy with new calcium antagonist. Jpn. Heart J. 1983;24:587–593. doi: 10.1536/ihj.24.587. [DOI] [PubMed] [Google Scholar]
- 47.Silke B., Verma S.P., Nelson G.I., Hussain M., Taylor S.H. Haemodynamic dose-response effects of i.v. nicardipine in coronary artery disease. Br. J. Clin. Pharmacol. 1984;18:717–724. doi: 10.1111/j.1365-2125.1984.tb02534.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Lahiri A., Robinson C.W., Kohli R.S., Caruana M.P., Raftery E.B. Acute and chronic effects of nicardipine on systolic and diastolic left ventricular performance in patients with heart failure: A pilot study. Clin. Cardiol. 1986;9:257–261. doi: 10.1002/clc.4960090605. [DOI] [PubMed] [Google Scholar]
- 49.Burlew B.S., Gheorghiade M., Jafri S.M., Goldberg A.D., Goldstein S. Acute and chronic hemodynamic effects of nicardipine hydrochloride in patients with heart failure. Am. Heart J. 1987;114:793–804. doi: 10.1016/0002-8703(87)90790-3. [DOI] [PubMed] [Google Scholar]
- 50.La Rovere M.T., Mortara A., Opasich C., Specchia G. Acute and chronic effects of nicardipine on rest and exercise haemodynamics in post-myocardial infarction patients with latent cardiac failure. Eur. Heart J. 1989;10:429–436. doi: 10.1093/oxfordjournals.eurheartj.a059506. [DOI] [PubMed] [Google Scholar]
- 51.Ortiz J., Matsumoto A., Monaco C.A., Barretto A.C. Left ventricular function after a single large dose of verapamil. Am. J. Cardiol. 1986;57:30D–34D. doi: 10.1016/0002-9149(86)90802-7. [DOI] [PubMed] [Google Scholar]
- 52.Setaro J.F., Zaret B.L., Schulman D.S., Black H.R., Soufer R. Usefulness of verapamil for congestive heart failure associated with abnormal left ventricular diastolic filling and normal left ventricular systolic performance. Am. J. Cardiol. 1990;66:981–986. doi: 10.1016/0002-9149(90)90937-V. [DOI] [PubMed] [Google Scholar]
- 53.Petrella R.J., Cunningham D.A., Paterson D.H. Left ventricular diastolic filling and cardiovascular functional capacity in older men. Exp. Physiol. 2000;85:547–555. doi: 10.1111/j.1469-445X.2000.02026.x. [DOI] [PubMed] [Google Scholar]
- 54.Giles T.D., Sander G.E., Roffidal L.C., Thomas M.G., Given M.B., Quiroz A.C. Comparison of nitrendipine and hydrochlorothiazide for systemic hypertension. Am. J. Cardiol. 1987;60:103–106. doi: 10.1016/0002-9149(87)90994-5. [DOI] [PubMed] [Google Scholar]
- 55.Naritomi H., Shimizu T., Miyashita K., Oe H., Sawada T. Pilot study on the effects of nitrendipine on cerebral blood flow in hypertensive patients with a history of cerebral infarction. Curr. Ther. Res. 1995;56:649–655. doi: 10.1016/0011-393X(95)85134-8. [DOI] [Google Scholar]
- 56.Fagard R., Staessen J., Amery A. The use of Doppler echocardiography to assess the acute haemodynamic response to felodipine and metoprolol in hypertensive patients. J. Hypertens. 1987;5:143–149. doi: 10.1097/00004872-198704000-00003. [DOI] [PubMed] [Google Scholar]
- 57.Binetti G., Rubino I., Varani E., Spadoni R., Ferretti R.M., Cervi V., Magnani B. Felodipine in severe chronic congestive heart failure: Acute effects on central hemodynamics and regional blood flow distribution. Cardiovasc. Drugs Ther. 1989;3:903–911. doi: 10.1007/BF01869580. [DOI] [PubMed] [Google Scholar]
- 58.McGrath B.P., Newman R., Older P. Hemodynamic study of short- and long-term isradipine treatment in patients with chronic ischemic congestive heart failure. Am. J. Med. 1989;86:75–80. doi: 10.1016/0002-9343(89)90196-4. [DOI] [PubMed] [Google Scholar]
- 59.Fisman E.Z., Pines A., Ben-Ari E., Shiner R.J., Drory Y., Friedman B.A., Kellermann J.J. Echocardiographic evaluation of the effects of gallopamil on left ventricular function. Clin. Pharmacol. Ther. 1988;44:100–106. doi: 10.1038/clpt.1988.119. [DOI] [PubMed] [Google Scholar]
- 60.Kompas F. Amlodipine (Dihydropyridinen), Dosering. [(accessed on 1 July 2022)]. Available online: https://www.farmacotherapeutischkompas.nl/bladeren/preparaatteksten/a/amlodipine#dosering.
- 61.Kompas F. Lercanidipine (Dihydropyridinen), Dosering. [(accessed on 1 July 2022)]. Available online: https://www.farmacotherapeutischkompas.nl/bladeren/preparaatteksten/l/lercanidipine#doseringen.
- 62.Welker H.A. Single- and multiple-dose mibefradil pharmacokinetics in normal and hypertensive subjects. J. Pharm. Pharmacol. 1998;50:983–987. doi: 10.1111/j.2042-7158.1998.tb06912.x. [DOI] [PubMed] [Google Scholar]
- 63.Kompas F. Nicardipine (Dihydropyridinen), Dosering. [(accessed on 1 July 2022)]. Available online: https://www.farmacotherapeutischkompas.nl/bladeren/preparaatteksten/n/nicardipine#dosering.
- 64.Kompas F. Verapamil (Calciumantagonisten, Overige), Dosering. [(accessed on 1 July 2022)]. Available online: https://www.farmacotherapeutischkompas.nl/bladeren/preparaatteksten/v/verapamil#dosering.
- 65.Kompas F. Nifedipine (Dihydropyridinen), Dosering. [(accessed on 1 July 2022)]. Available online: https://www.farmacotherapeutischkompas.nl/bladeren/preparaatteksten/n/nifedipine.
- 66.Kompas F. Diltiazem (Calciumantagonisten, Overige), Dosering. [(accessed on 1 July 2022)]. Available online: https://www.farmacotherapeutischkompas.nl/bladeren/preparaatteksten/d/diltiazem.
- 67.Mroczek W.J., Burris J.F. Nitrendipine monotherapy in severe hypertension. Angiology. 1988;39 (1 Pt 2):81–86. [PubMed] [Google Scholar]
- 68.Kompas F. Lacidipine (Dihydropyridinen), Dosering. [(accessed on 1 July 2022)]. Available online: https://www.farmacotherapeutischkompas.nl/bladeren/preparaatteksten/l/lacidipine#dosering.
- 69.Kompas F. Felodipine (Dihydropyridinen), Dosering. [(accessed on 1 July 2022)]. Available online: https://www.farmacotherapeutischkompas.nl/bladeren/preparaatteksten/f/felodipine#dosering.
- 70.Sundstedt C.D., Ruegg P.C., Keller A., Waite R. A multicenter evaluation of the safety, tolerability, and efficacy of isradipine in the treatment of essential hypertension. Am. J. Med. 1989;86:98–102. doi: 10.1016/0002-9343(89)90201-5. [DOI] [PubMed] [Google Scholar]
- 71.Rose E.L., Lahiri A., Raftery E.B. Antianginal Efficacy of Sustained Release Gallopamil. Drug Investig. 2012;5:212–221. doi: 10.1007/BF03258449. [DOI] [Google Scholar]
- 72.Rossello X., Ferreira J.P., Pocock S.J., McMurray J.J.V., Solomon S.D., Lam C.S.P., Girerd N., Pitt B., Rossignol P., Zannad F. Sex differences in mineralocorticoid receptor antagonist trials: A pooled analysis of three large clinical trials. Eur. J. Heart Fail. 2020;22:834–844. doi: 10.1002/ejhf.1740. [DOI] [PubMed] [Google Scholar]
- 73.Song J.J., Ma Z., Wang J., Chen L.X., Zhong J.C. Gender Differences in Hypertension. J. Cardiovasc. Transl. Res. 2020;13:47–54. doi: 10.1007/s12265-019-09888-z. [DOI] [PubMed] [Google Scholar]
- 74.Hirst G.D., Edwards F.R. Sympathetic neuroeffector transmission in arteries and arterioles. Physiol. Rev. 1989;69:546–604. doi: 10.1152/physrev.1989.69.2.546. [DOI] [PubMed] [Google Scholar]
- 75.Wier W.G., Morgan K.G. Alpha1-adrenergic signaling mechanisms in contraction of resistance arteries. Rev. Physiol. Biochem. Pharmacol. 2003;150:91–139. doi: 10.1007/s10254-003-0019-8. [DOI] [PubMed] [Google Scholar]
- 76.Kneale B.J., Chowienczyk P.J., Cockcroft J.R., Coltart D.J., Ritter J.M. Vasoconstrictor sensitivity to noradrenaline and NG-monomethyl-L-arginine in men and women. Clin. Sci. 1997;93:513–518. doi: 10.1042/cs0930513. [DOI] [PubMed] [Google Scholar]
- 77.Harvey R.E., Barnes J.N., Charkoudian N., Curry T.B., Eisenach J.H., Hart E.C., Joyner M.J. Forearm vasodilator responses to a beta-adrenergic receptor agonist in premenopausal and postmenopausal women. Physiol. Rep. 2014;2:e12032. doi: 10.14814/phy2.12032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Loria A.S., Brinson K.N., Fox B.M., Sullivan J.C. Sex-specific alterations in NOS regulation of vascular function in aorta and mesenteric arteries from spontaneously hypertensive rats compared to Wistar Kyoto rats. Physiol. Rep. 2014;2:e12125. doi: 10.14814/phy2.12125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Al-Gburi S., Deussen A., Zatschler B., Weber S., Kunzel S., El-Armouche A., Lorenz K., Cybularz M., Morawietz H., Kopaliani I. Sex-difference in expression and function of beta-adrenoceptors in macrovessels: Role of the endothelium. Basic Res. Cardiol. 2017;112:29. doi: 10.1007/s00395-017-0617-2. [DOI] [PubMed] [Google Scholar]
- 80.Fischer M., Baessler A., Schunkert H. Renin angiotensin system and gender differences in the cardiovascular system. Cardiovasc. Res. 2002;53:672–677. doi: 10.1016/S0008-6363(01)00479-5. [DOI] [PubMed] [Google Scholar]
- 81.Komukai K., Mochizuki S., Yoshimura M. Gender and the renin-angiotensin-aldosterone system. Fundam. Clin. Pharmacol. 2010;24:687–698. doi: 10.1111/j.1472-8206.2010.00854.x. [DOI] [PubMed] [Google Scholar]
- 82.Hilliard L.M., Nematbakhsh M., Kett M.M., Teichman E., Sampson A.K., Widdop R.E., Evans R.G., Denton K.M. Gender differences in pressure-natriuresis and renal autoregulation: Role of the Angiotensin type 2 receptor. Hypertension. 2011;57:275–282. doi: 10.1161/HYPERTENSIONAHA.110.166827. [DOI] [PubMed] [Google Scholar]
- 83.Brown R.D., Hilliard L.M., Head G.A., Jones E.S., Widdop R.E., Denton K.M. Sex differences in the pressor and tubuloglomerular feedback response to angiotensin II. Hypertension. 2012;59:129–135. doi: 10.1161/HYPERTENSIONAHA.111.178715. [DOI] [PubMed] [Google Scholar]
- 84.Zimmerman M.A., Sullivan J.C. Hypertension: What’s sex got to do with it? Physiology. 2013;28:234–244. doi: 10.1152/physiol.00013.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Sullivan J.C., Rodriguez-Miguelez P., Zimmerman M.A., Harris R.A. Differences in angiotensin (1–7) between men and women. Am. J. Physiol. Heart Circ. Physiol. 2015;308:H1171–H1176. doi: 10.1152/ajpheart.00897.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Wang L., Wang X., Qu H.Y., Jiang S., Zhang J., Fu L., Buggs J., Pang B., Wei J., Liu R. Role of Kidneys in Sex Differences in Angiotensin II-Induced Hypertension. Hypertension. 2017;70:1219–1227. doi: 10.1161/HYPERTENSIONAHA.117.10052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Wilbert-Lampen U., Seliger C., Trapp A., Straube F., Plasse A. Female sex hormones decrease constitutive endothelin-1 release via endothelial sigma-1/cocaine receptors: An action independent of the steroid hormone receptors. Endothelium. 2005;12:185–191. doi: 10.1080/10623320500227275. [DOI] [PubMed] [Google Scholar]
- 88.Barton M., Meyer M.R. Postmenopausal hypertension: Mechanisms and therapy. Hypertension. 2009;54:11–18. doi: 10.1161/HYPERTENSIONAHA.108.120022. [DOI] [PubMed] [Google Scholar]
- 89.Virani S.S., Alonso A., Aparicio H.J., Benjamin E.J., Bittencourt M.S., Callaway C.W., Carson A.P., Chamberlain A.M., Cheng S., Delling F.N., et al. Heart Disease and Stroke Statistics-2021 Update: A Report From the American Heart Association. Circulation. 2021;143:e254–e743. doi: 10.1161/CIR.0000000000000950. [DOI] [PubMed] [Google Scholar]
- 90.Orshal J.M., Khalil R.A. Gender, sex hormones, and vascular tone. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2004;286:R233–R249. doi: 10.1152/ajpregu.00338.2003. [DOI] [PubMed] [Google Scholar]
- 91.Ashraf M.S., Vongpatanasin W. Estrogen and hypertension. Curr. Hypertens. Rep. 2006;8:368–376. doi: 10.1007/s11906-006-0080-1. [DOI] [PubMed] [Google Scholar]
- 92.Dadashzadeh S., Javadian B., Sadeghian S. The effect of gender on the pharmacokinetics of verapamil and norverapamil in human. Biopharm. Drug. Dispos. 2006;27:329–334. doi: 10.1002/bdd.512. [DOI] [PubMed] [Google Scholar]
- 93.Wolbold R., Klein K., Burk O., Nussler A.K., Neuhaus P., Eichelbaum M., Schwab M., Zanger U.M. Sex is a major determinant of CYP3A4 expression in human liver. Hepatology. 2003;38:978–988. doi: 10.1002/hep.1840380424. [DOI] [PubMed] [Google Scholar]
- 94.George J., Byth K., Farrell G.C. Age but not gender selectively affects expression of individual cytochrome P450 proteins in human liver. Biochem. Pharmacol. 1995;50:727–730. doi: 10.1016/0006-2952(95)00192-3. [DOI] [PubMed] [Google Scholar]
- 95.Schmucker D.L., Woodhouse K.W., Wang R.K., Wynne H., James O.F., McManus M., Kremers P. Effects of age and gender on in vitro properties of human liver microsomal monooxygenases. Clin. Pharmacol. Ther. 1990;48:365–374. doi: 10.1038/clpt.1990.164. [DOI] [PubMed] [Google Scholar]
- 96.Cummins C.L., Wu C.Y., Benet L.Z. Sex-related differences in the clearance of cytochrome P450 3A4 substrates may be caused by P-glycoprotein. Clin. Pharmacol. Ther. 2002;72:474–489. doi: 10.1067/mcp.2002.128388. [DOI] [PubMed] [Google Scholar]
- 97.Kloner R.A., Sowers J.R., DiBona G.F., Gaffney M., Wein M. Sex- and age-related antihypertensive effects of amlodipine. The Amlodipine Cardiovascular Community Trial Study Group. Am. J. Cardiol. 1996;77:713–722. doi: 10.1016/S0002-9149(97)89205-3. [DOI] [PubMed] [Google Scholar]
- 98.Kalibala J., Pechere-Bertschi A., Desmeules J. Gender Differences in Cardiovascular Pharmacotherapy-the Example of Hypertension: A Mini Review. Front. Pharmacol. 2020;11:564. doi: 10.3389/fphar.2020.00564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Kang D., Verotta D., Schwartz J.B. Population analyses of amlodipine in patients living in the community and patients living in nursing homes. Clin. Pharmacol. Ther. 2006;79:114–124. doi: 10.1016/j.clpt.2005.09.007. [DOI] [PubMed] [Google Scholar]
- 100.Greenblatt D.J., Harmatz J.S., von Moltke L.L., Wright C.E., Shader R.I. Age and gender effects on the pharmacokinetics and pharmacodynamics of triazolam, a cytochrome P450 3A substrate. Clin. Pharmacol. Ther. 2004;76:467–479. doi: 10.1016/j.clpt.2004.07.009. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
No individual patient data are included in this study. Search strategy and results of included papers are presented within the manuscript and are available at the corresponding author upon request.