Abstract
Chronic lymphocytic leukemia (CLL) is the most common leukemia in adults. Despite its indolent clinical course, therapy refractoriness and disease progression still represent an unmet clinical need. Before the advent of pathway inhibitors, chemoimmunotherapy (CIT) was the commonest option for CLL treatment and is still widely used in areas with limited access to pathway inhibitors. Several biomarkers of refractoriness to CIT have been highlighted, including the unmutated status of immunoglobulin heavy chain variable genes and genetic lesions of TP53, BIRC3 and NOTCH1. In order to overcome resistance to CIT, targeted pathway inhibitors have become the standard of care for the treatment of CLL, with practice-changing results obtained through the inhibitors of Bruton tyrosine kinase (BTK) and BCL2. However, several acquired genetic lesions causing resistance to covalent and noncovalent BTK inhibitors have been reported, including point mutations of both BTK (e.g., C481S and L528W) and PLCG2 (e.g., R665W). Multiple mechanisms are involved in resistance to the BCL2 inhibitor venetoclax, including point mutations that impair drug binding, the upregulation of BCL2-related anti-apoptotic family members, and microenvironmental alterations. Recently, immune checkpoint inhibitors and CAR-T cells have been tested for CLL treatment, obtaining conflicting results. Potential refractoriness biomarkers to immunotherapy were identified, including abnormal levels of circulating IL-10 and IL-6 and the reduced presence of CD27+CD45RO− CD8+ T cells.
Keywords: chronic lymphocytic leukemia, predictive biomarkers, chemoimmunotherapy, pathway inhibitors, immunotherapy
1. Introduction
Chronic lymphocytic leukemia (CLL) is a hematologic neoplasm characterized by the clonal proliferation of mature B cells [1,2]. Although leukemia was first described in 1845 by R. Virchow and J.H. Bennett, it was only in 1924 that CLL was fully characterized by G.B. Minot and R. Isaacs as a distinct clinical entity [3]. However, CLL has been extensively studied in more recent times, with the identification of key genetic abnormalities implied in its pathogenesis, such as deletion 11q, deletion 13q, and trisomy 12 [4]. For a clinical diagnosis, a lymphocyte blood count of ≥5 × 109/L is required, as well as a specific immunophenotypic profile that necessarily includes the clonal expression of CD19, CD5, CD20, CD23 and dim surface immunoglobulins with κ or λ light-chain restriction [1,2,5]. CLL is the most common leukemia in adults, with an incidence of 4.6/100,000 per year in the US, and is characterized by a relatively low mortality, displaying a 5-year relative survival rate of nearly 90% [6]. The median age at diagnosis is ~70 years, implying that CLL is a disease mainly of older adults [6].
Despite its indolent clinical course, CLL is still not curable, and patients refractory to available therapies are prone to disease progression or transformation to aggressive lymphoma and eventually death, highlighting the need to overcome treatment refractoriness in this disease [7,8]. The staging of CLL is based on the historically used scores of the Binet and Rai systems that are based on clinical variables, namely anemia, thrombocytopenia, hepatosplenomegaly and lymphadenopathy [9,10]. In recent times and thanks to the availability of novel biomarkers, several prognostic scoring systems have been developed, such as the CLL International Prognostic Index (CLL-IPI) and the International Prognostic Score for Asymptomatic Early-Stage Disease (IPS-E), allowing a more precise prognostic assessment of patients on the basis of clinical and molecular features [11,12].
In asymptomatic patients with early-stage disease (Rai 0/Binet A), which account for the majority of CLL cases, a watch-and-wait approach should be adopted, while treatment requirement at diagnosis or during follow-up is based on specific indications according to the International Workshop on Chronic Lymphocytic Leukemia (iwCLL) guidelines [13,14]. Remarkably, 2–10% of CLL patients develop an aggressive lymphoma, defined as Richter syndrome (RS), which is particularly refractory to chemoimmunotherapy (CIT) and still represents a life-threatening condition and a major unmet clinical need [15].
Until the advent of pathway inhibitors, the front-line treatment for CLL was represented by CIT, in particular the FCR (fludarabine, cyclophosphamide and the anti-CD20 monoclonal antibody [mAb] rituximab), BR (bendamustine and rituximab) and Chl-O (chlorambucil and the anti-CD20 mAb obinutuzumab) regimens [13]. CIT has now been largely abandoned due to the lower efficacy and higher incidence of adverse events compared to pathway inhibitors [16,17]. CIT can still be evaluated as a treatment option in chemo-sensitive patients, especially in less-resourced countries, where pathway inhibitors are hardly available or affordable [18,19,20,21].
Current front-line treatment for CLL is based on pathway inhibitors with or without anti-CD20 mAbs. The European Society for Medical Oncology (ESMO) guidelines for front-line treatment of CLL include Bruton tyrosine kinase inhibitors (BTKi), namely ibrutinib and acalabrutinib, the B-cell lymphoma 2 inhibitor (BCL2i) venetoclax with or without obinutuzumab, and the phosphoinositide 3 kinase inhibitor (PI3Ki) idelalisib with rituximab, although the latter regimen is seldom used because of infectious toxicity [18]. Consistently, the latest National Comprehensive Cancer Network (NCCN) guidelines recommend the use of a BTKi in monotherapy (ibrutinib or zanubrutinib) or a combination of acalabrutinib or venetoclax plus obinutuzumab for the front-line treatment of CLL [21]. For relapsed/refractory (R/R) CLL, the guidelines recommend the use of one of the above-mentioned pathway inhibitors based on response to front-line therapy; several innovative drugs are also under development [18,21]. Although allogeneic hematopoietic stem cell transplantation (allo-HSCT) is the only curative option for CLL, with a 5-year progression-free survival (PFS) rate of ~40%, it is rarely used since the procedure is affected by a significant mortality rate (approximately from 10% to 20%), primarily due to graft-versus-host disease (GVHD) and its complications [22,23]. As different treatment options were progressively utilized for the treatment of CLL, several biomarkers of refractoriness have been identified (Table 1). Here, we aim to provide a comprehensive overview of the biomarkers of CLL refractoriness to available therapies, including CIT as well as pathway inhibitors and immunotherapy.
Table 1.
Summary of identified biomarkers of treatment refractoriness to CLL.
| Biomarker | Prevalence before Treatment | Prevalence at Progression | Mechanism of Resistance | Predictive Value | References |
|---|---|---|---|---|---|
| Unmutated IGHV gene | ~40% | 70–80% | Increased BCR signaling capacity | Poor response to CIT | [24,25,26] |
| Del(17p) | 5% | ~30% | Genomic instability, survival advantage, and reduced DNA damage response | Poor response to CIT, BTKi, and BCL2i | [24,25,27,28,29,30] |
| TP53 mutations | 7% | 30–40% | Genomic instability, survival advantage, and reduced DNA damage response | Poor response to CIT, BTKi, and BCL2i | [24,25,27,28,29,30] |
| BIRC3 mutations | 2–6% | ~8% | Upregulation of non-canonical NF-κB signaling pathway | Poor response to CIT | [24,31,32,33,34,35,36] |
| NOTCH1 mutations | 8–10% | 30% | Transcriptional activation of cell survival and proliferation and reduced expression of CD20 | Poor response to CIT and anti-CD20 mAbs | [24,25,27,37,38,39] |
| BTK point mutations of C481: C481S/R/Y/G | N/A | ~50% | Reduced affinity for covalent BTKi | Poor response to covalent BTKi | [40] |
| BTK point mutations of the tyrosine kinase domain: L528W, V416L, T474I, M437R, A428D | N/A | ~16% | Binding impairment of non-covalent BTKi | Poor response to covalent and non-covalent BTKi | [41] |
| PLCG2 mutations: R665W, L845G, C849R, D993H | N/A | 13% | Constitutively active PLCγ2 | Poor response to BTKi | [42,43] |
| BCL2 mutations: G101V, D103Y, F104I | N/A | ~15% | Binding impairment of BCL2is | Poor response to BCL2i | [44] |
| Upregulation of MCL-1 and/or BCL-xL | N/A | N/A | Enhanced apoptosis evasion | Poor response to BCL2i | [45,46] |
| High serum [IL-10] | N/A | N/A | Reduced T cell response through IL-10R stimulation | Poor response to PD-1/PD-L1 immune checkpoint inhibitors | [47] |
| Low serum [IL-6] | N/A | N/A | CAR-T cell exhaustion due to defective IL-6R stimulation | Poor response to CAR-T cells | [48] |
| Low levels of CD27+CD45RO− CD8+ T cells | N/A | N/A | Reduced population of active CAR-T cells | Poor response to CAR-T cells | [48] |
Abbreviations: IGHV, immunoglobulin heavy variable; del(17p), deletion of the short arm of chromosome 17; BCR, B cell receptor; CIT, chemoimmunotherapy; BTK, Bruton tyrosine kinase; BTKi, BTK inhibitors; BCL2, B cell lymphoma 2; BCL2i, BCL2 inhibitors; PLCG2, phospholipase-C-gamma-2; PLCγ2, phospholipase-C-γ-2; IL-10, Interleukin-10; IL-10R, IL-10 receptor; IL-6, Interleukin-6; IL-6R, IL-6 receptor; CAR, chimeric antigen receptor; mAbs, monoclonal antibodies.
2. Biomarkers of Refractoriness to Chemoimmunotherapy
Historically, chemotherapy had been the most widely used option for the treatment of CLL, which was subsequently replaced by CIT based on the results of practice-changing clinical studies [49]. The most used chemotherapy regimens were initially based on monotherapy with alkylating agents, such as chlorambucil and bendamustine, or purine analogues, namely fludarabine and cladribine [4]. As for combination therapies, the most adopted was the FC regimen (fludarabine and cyclophosphamide), which granted favorable results compared to monotherapies [4]. CIT for CLL consists of the combination of chemotherapy and anti-CD20 mAbs in order to obtain a synergistic effect against tumor cells [13]. The CLL8 phase III randomized trial compared the chemotherapy regimen FC versus FCR in fit CLL patients, demonstrating a significant superiority of the chemoimmunotherapeutic approach [27,50]. Specifically, FCR outperformed FC in both median PFS (56.8 vs. 32.9 months, respectively) and overall survival (OS, not reached vs. 86.0 months, respectively) with a comparable toxicity profile [27].
Different CIT regimens for CLL patients with comorbidities were evaluated in the CLL11 phase III randomized trial, where the Chl-O regimen showed better median PFS and OS compared to chlorambucil plus rituximab or chlorambucil monotherapy [28,51]. The adoption of CIT regimens for CLL has led to the identification of several biomarkers of refractoriness to this therapeutic approach, including the unmutated status of immunoglobulin heavy-chain variable (IGHV) genes and genetic lesions of TP53, BIRC3 and NOTCH1 [24]. In order to overcome resistance to CIT, pathway inhibitors were adopted for the treatment of CLL, with practice-changing results obtained by BTKi and BCL2i [49].
2.1. IGHV Mutational Status
Mature B cells express the B cell receptor (BCR) on their surface, a key component for antigen recognition and B cell activation, composed of an immunoglobulin (Ig) and a signaling subunit [52]. In order to expose an Ig on the external cell membrane, B cells must perform a genetic recombination of the variable Ig genes through a process termed V(D)J rearrangement, which ensures a very high degree of heterogeneity in the BCR repertoire [53]. After antigen recognition, naïve B cells move to lymph node germinal centers (GCs), where somatic hypermutation (SHM) of IGHV genes takes place, potentially increasing the BCR affinity for the recognized antigen [53,54].
Based on the mutational status of IGHV genes, CLL can be divided into two molecular subgroups: (i) IGHV unmutated CLL (U-CLL, ~40% of all CLL), which reflect mature B cells that have not experienced the GC reaction and have undergone maturation in a T-cell-independent manner; and (ii) IGHV mutated CLL (M-CLL, ~60% of all CLL), which reflect mature B cells that have experienced the GC reaction and have undergone the SHM process [25,55,56]. In particular, to be considered as M-CLL, the threshold used in the clinical practice is a deviation in ≥2% of the patient’s IGHV sequence from the germline nucleotide sequence [57]. Unmutated IGHV genes associate more commonly with progressive or R/R CLL, while mutated IGHV genes are more frequently detected in asymptomatic or treatment-naïve disease [58,59]. Importantly, unmutated IGHV genes occur in up to 60% of CLL, requiring treatment according to guidelines.
Beyond its prognostic value, IGHV mutational status is also a predictive biomarker, as shown by the lower response of U-CLL to all the available CIT regimens when compared to M-CLL [27,60,61,62]. Clinical trials evaluating continuous treatment with ibrutinib, acalabrutinib and zanubrutinib have displayed favorable efficacy outcomes in U-CLL, superimposable to those reached with M-CLL, overcoming treatment refractoriness due to IGHV mutational status [63,64,65].
2.2. TP53 Disruption
TP53, located on the short arm of chromosome 17 (17p), is an onco-suppressor gene encoding the p53 protein, also called “the guardian of the genome”, which exerts a proapoptotic function in response to DNA damage [66]. Consistently, the disruption of TP53 results in increased resistance to apoptosis induced by DNA-damaging agents, including chemotherapy and, by extension, CIT [67]. Somatic mutations are the most common genetic lesions of TP53 in CLL, followed by del(17p) [68,69].
The disruption of TP53 has been found in 4% to 8% of newly diagnosed CLL, while, as the disease progresses, the frequency of TP53 abnormalities rises, reaching a prevalence of 10–12% at the time of first treatment requirement, ~40% in patients refractory to fludarabine, and 50–60% in those who develop RS [69]. Consequently, genetic lesions of TP53 can be defined as both prognostic and predictive biomarkers. Among patients treated with FCR, the CLL8 trial reported a median PFS of 15.4 months and a median OS of 49 months in TP53-mutated patients, while the median PFS and OS in TP53 wild-type patients were 59 months and not reached, respectively [27]. Similar unfavorable outcomes were reached with the BR and Chl-O regimens in TP53-disrupted CLL, while BTKi-based therapies have obtained remarkable results, which are comparable with those of TP53 wild-type patients [27,28,61,63,64,65].
Due to the significant clinical impact exerted by TP53 disruption, the iwCLL guidelines recommend testing del(17p) via fluorescence in situ hybridization (FISH) and TP53 mutation status via DNA sequencing before every line of treatment [14]. In addition to these recommendations, the European Research Initiative on CLL (ERIC) endorses the possible use of next-generation sequencing (NGS) for TP53 mutation testing since this methodology is characterized by a higher sensitivity compared to traditional Sanger sequencing [70].
2.3. BIRC3 Disruption
The BIRC3 gene has been found to be mutated or deleted in 2–6% of CLL cases [31,32,33]. BIRC3 encodes for the protein c-IAP2, which negatively regulates the MAP3K14 kinase (or NIK–NF-κB-inducing kinase), the key activator of the noncanonical NF-κB signaling pathway, leading to the transcription of genes linked to cell proliferation and survival [71]. Additionally, the aberrant activation of NF-κB signaling in c-IAP2 knockdown models has been shown to increase p53 degradation via the E3 ubiquitin ligase MDM2 [72]. Therefore, the mutational inactivation or deletion of BIRC3 in CLL results in constitutive NF-κB pathway activation, providing pro-survival signals to the leukemic clone, e.g., through the up-regulation of several anti-apoptotic genes, as demonstrated in ex vivo models [34,73].
A retrospective evaluation of the outcome of FCR-treated CLL patients showed a similar median PFS rate between BIRC3- and TP53-disrupted CLL patients (2.2 and 2.6 years, respectively), significantly inferior to the PFS of BIRC3 wild-type patients [34]. Moreover, the CLL14 phase III randomized trial, evaluating front-line treatment with venetoclax plus obinutuzumab versus Chl-O in CLL, has shown poor outcomes in BIRC3 mutated patients treated with Chl-O, with a median PFS of 16.8 months [35,74]. However, ibrutinib- and/or venetoclax-based therapies appear to overcome the resistance conferred by BIRC3 disruption [34,74,75,76,77].
2.4. NOTCH1 Mutations
NOTCH1 codes for the transmembrane protein NOTCH1, which acts as a surface receptor for ligands of the SERRATE/JAGGED or DELTA families [78,79]. After being cleaved by γ-secretase, the active subunit of the receptor migrates into the nucleus and acts as a transcription factor for genes involved in cell survival and proliferation, including MYC and components of the NF-κB pathway [37,78]. In CLL, NOTCH1 mutations disrupt the PEST domain, responsible for the promotion of proteasomal degradation of the NOTCH1 protein, resulting in the aberrant activation of the receptor [80,81].
At diagnosis, ~8% of CLL patients harbor a NOTCH1 mutation, but the prevalence of this genetic lesion rises in fludarabine-refractory CLL and RS patients (20.8% and 31.1%, respectively) [38]. Furthermore, genetic lesions of NOTCH1 are thought to be involved in resistance to CLL immunotherapy [79]. The predictive value of NOTCH1 mutations for the treatment with an immunotherapeutic agent has been investigated in the above-mentioned CLL8 trial [27]. In particular, NOTCH1-mutated CLL showed no improvement due to the addition of rituximab since the 5-year PFS rate of NOTCH1-mutated patients was 25.8% for the FC cohort and 26.7% for the FCR cohort (p-value of 0.974) [27]. Similar results were obtained by the COMPLEMENT 1 study, a phase III randomized trial that compared chlorambucil alone versus chlorambucil plus the anti-CD20 mAb ofatumumab, highlighting the role of NOTCH1 mutations in predicting refractoriness to anti-CD20 mAb-based immunotherapy [39,82].
The proposed mechanism for NOTCH1-mediated resistance appears to be linked to the HDAC-mediated repression of the surface exposure of CD20 in NOTCH1-mutated CLL cells, as shown by in vitro models [83]. Remarkably, the CLL11 trial demonstrated a clear superiority of Chl-O over chlorambucil alone, showing better PFS and OS in the Chl-O arm, independent of the presence of NOTCH1 genetic lesions [28]. Hence, these data suggest that the higher clinical efficacy of obinutuzumab may overcome the effect of NOTCH1 mutations. Pathway inhibitors represent a viable option to overcome NOTCH1-mediated refractoriness to CIT and immunotherapy, as shown in the RESONATE phase III randomized trial, where no difference in PFS was detected between NOTCH1 mutated and wild-type CLL treated with ibrutinib [63].
3. Biomarkers of Refractoriness to BTK Inhibitors
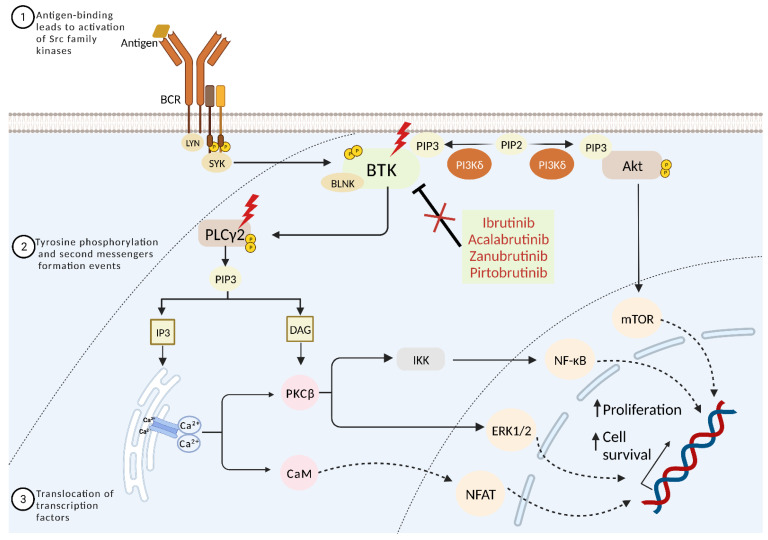
The BCR signaling pathway plays an essential role in the development of B cells and the pathogenesis of CLL [84]. BCR signaling is activated by antigen binding to surface immunoglobulins (sIg), resulting in coupling and autophosphorylation of the immunoreceptor tyrosine-based activation motifs (ITAMs) on the cytoplasmic tails of CD79A (Igα)/CD79B (Igβ) by the Src family protein kinase LYN. The phosphorylation of ITAMs creates docking sites for spleen tyrosine kinase SYK, which activates the B cell linker scaffold protein (BLNK). In the presence of BLNK, BTK is activated through phosphorylation at its Y551 aminoacidic residue by either LYN or SYK [85]. Once activated, BTK triggers downstream pathways, namely the PI3K-Akt and phospholipase-γ-2 (PLCγ2) pathways, finally leading to the induction of different transcription factors, including mTOR, NF-κB, ERK1/2, and NFAT, and it is involved in the survival, differentiation, and proliferation of B cells [85,86]. Additionally, BTK can be triggered by other receptors, including growth factors, cytokine receptors, and G-protein coupled receptors (GPCRs), such as chemokine receptors and integrins [87]. In CLL, the BCR is constitutively active through ligand-dependent and independent mechanisms, causing constitutive BTK signaling activation, which confers a survival and proliferation advantage to the neoplastic cells (Figure 1) [88].
Figure 1.
BCR signaling pathway and BTKis resistance in CLL. Upon antigen binding, the B cell receptor initiates the formation of a signaling complex through the phosphorylation of immunoreceptor-based activation motif (ITAM) residues on the cytoplasmic tails of CD79A(Igα) and CD79B(Igβ) proteins. This event activates SYK, which then triggers the activation of BTK, PLCγ2, and PI3K. The downstream signaling response includes PKC activation and Ca2+ mobilization and Akt activation, leading to the promotion of transcript factors NF-κB, ERK1/2, NFAT, and mTOR. This signaling cascade can be effectively inhibited by BTK inhibitors. However, BTK and PLCγ2 point mutations can result in BTKi resistance.
3.1. Refractoriness to First-Generation Covalent BTK Inhibitors
Ibrutinib, the first-in-class and orally bioavailable BTKi, was approved by the FDA in 2014 for the treatment of R/R CLL, changing the landscape for the treatment of this leukemia. Ibrutinib can be administrated both as a first-line therapy and in R/R CLL patients [86]. Its safety and efficacy as front-line treatment were established by the randomized phase III RESONATE-2 trial, which documented the superiority of ibrutinib versus chlorambucil [89]. In R/R CLL, ibrutinib was proven to be superior to the anti-CD20 monoclonal antibody ofatumumab by the randomized phase III RESONATE trial [90].
Ibrutinib irreversibly blocks BTK by covalently binding to the C481 aminoacidic residue in the ATP-binding domain of the protein [91]. The occupancy of the ATP-binding site by ibrutinib leads to a lack of phosphorylation of different downstream targets such as Akt and PLCγ2, resulting in BTK signaling inhibition that, in turn, reduces BCR signaling both in vitro and in vivo [92]. Besides this on-target effect, ibrutinib deactivates several off-targets, e.g., EGFR, ErbB2, ITK, and TEC, which might contribute to the antitumor effect but, at the same time, result in adverse events, such as atrial fibrillation (AF) and bleeding [93,94].
3.1.1. Primary Resistance to First-Generation Covalent BTK Inhibitors
Biologic resistance can be divided into primary (or intrinsic) and secondary (or acquired) resistance [95]. Primary resistance to ibrutinib can develop in 10–16% of the cases, but the molecular mechanisms are still unclear [96,97]. Resistance to ibrutinib in CLL patients has been associated with high-risk genomic features and heavy pretreatment. Genetic alterations unrelated to the BCR pathway, involving the ATM, BIRC3, NOTCH1, SF3B1, and TP53 genes, are found in a fraction of treatment-naïve CLL patients [96,97].
Importantly, baseline features, such as del(17p)/TP53 and complex karyotype (≥3 chromosomal abnormalities), increase the risk of disease progression in patients treated with ibrutinib [29,98,99]. TP53 disruption is the only independent molecular factor that predicts inferior OS and PFS in patients receiving ibrutinib treatment [100]. Therefore, despite major improvements compared to the CIT era, the prognosis of patients with del(17p)/TP53 remains suboptimal, at least to a certain extent, even in the era of targeted therapy. Additionally, del(18p), which occurs in a small percentage of untreated cases, is found at a higher frequency in ibrutinib-relapsed patients and is associated with BTK mutation development, suggesting a potential role of this chromosomal abnormality in refractoriness to ibrutinib [29].
3.1.2. Secondary Resistance to First-Generation Covalent BTK Inhibitors
Secondary resistance eventually develops in about 60% of CLL patients treated with BTK covalent inhibitors [42]. Shortly after ibrutinib was approved in 2014, the first BTKi resistance mutations were reported. These mutations can be divided into two categories: (i) point mutations that prevent ibrutinib from binding covalently to BTK by changing the targeted cysteine residue (C481) in the kinase domain; and (ii) mutations that constitutively activate downstream signaling through PLCγ2 (Figure 1) [101].
In an early analysis, Woyach and colleagues performed whole-exome sequencing in six patients with acquired resistance to ibrutinib and documented that four of them had a cysteine-to-serine mutation in BTK at position 481 (C481S), corresponding to the ibrutinib binding site [43]. This pivotal investigation also revealed that ibrutinib refractoriness exploited at least another molecular mechanism. In fact, one patient with a low-frequency C481S mutation in BTK harbored three different PLCG2 mutations: R665W, L845F, and S707Y. The sixth patient had an arginine-to-tryptophan mutation in PLCG2 at position 665 (R665W). In all patients, the mutations identified at the time of refractoriness were absent before the start of treatment [43].
The C481S mutation was later confirmed to be the most common ibrutinib resistance-mediating mutation in CLL patients [102]. The functional characterization of the C481S mutation has shown reduced affinity of BTK for ibrutinib, allowing only reversible inhibition rather than irreversible blockade [43,103]. In addition to the substitution of cysteine in 481 by serine, the substitution by other amino acids such as tyrosine, arginine, phenylalanine, tryptophan, and glycine has also been reported [42,99]. Besides mutations of BTK, also the PLCG2 gene, which acts downstream of BTK in the BCR signaling cascade, is involved in resistance to ibrutinib. In particular, the R665W mutation in the SH2 domain of PLCγ2 is a gain-of-function mutation that causes activation of PLCγ2 independent of BTK signaling stimulation [104].
Deep sensitivity genetic analysis of sequential samples of progressive CLL patients on ibrutinib allowed the detection of BTK- and/or PLCG2-mutated clones at a median of eight and nine months before progression, respectively [105,106]. More recently, an analysis of a French registry cohort showed an incidence of BTK mutations of 57% in patients who were still responding to ibrutinib, suggesting that a substantial proportion of CLL patients receiving ibrutinib monotherapy already have resistance-mediating mutations though still responding clinically to treatment [42]. In this respect, BTK and/or PLCG2 mutations might represent potential biomarkers to detect preclinical resistance development to BTKi, although current guidelines do not recommend testing for BTK and PLCG2 mutations before the development of clinical refractoriness. Remarkably, only approximately 70–80% of patients with acquired ibrutinib resistance carry mutations in BTK and/or PLCG2 [40]. Several chromosomal aberrations have also been associated with secondary resistance to ibrutinib, including MYC amplification, del(18p), and del(8p), which causes haploinsufficiency of TRAIL-R, generating resistance of the neoplastic cells to TRAIL-induced apoptosis [29,34,99,107].
3.2. Refractoriness to Second-Generation Covalent BTK Inhibitors
Acalabrutinib and zanubrutinib are irreversible, potent and covalent BTK inhibitors with higher selectivity than ibrutinib for the C481 residue of the binding site [108,109]. Therefore, these drugs have less off-target inhibition of other kinases of the TEC family, including EGFR and ITK, and consequently, fewer adverse events [108,109]. The phase III randomized ELEVATE-RR trial showed the non-inferiority of acalabrutinib compared to ibrutinib in terms of PFS in R/R CLL [108]. Recently, a phase III randomized controlled trial comparing ibrutinib and zanubrutinib in R/R CLL patients showed that zanubrutinib had a higher ORR, superior PFS, and a lower rate of atrial fibrillation/flutter compared with ibrutinib [109]. However, secondary resistance to second-generation BTKi has been reported as well. In patients treated with acalabrutinib, the C481S mutation was found to be the most common acquired mutation in disease progression, similar to ibrutinib [110]. Moreover, the development of PLCG2 mutations was also detected in the same cohort [110]. Conversely, in CLL patients with progressive disease treated with zanubrutinib, the BTK L528W mutation is detected at a certain rate and appears to be responsible for progression (Figure 1) [111].
3.3. Strategies to Overcome Resistance to Covalent BTK Inhibitors
Point mutations of C481 in the ATP-binding pocket of BTK are responsible for a sizable fraction of CLL resistance to ibrutinib [40]. To overcome this resistance, reversible and non-covalent BTK inhibitors have been developed, including vecabrutinib, fenebrutinib, nemtabrutinib (ARQ 531), and pirtobrutinib (LOXO-305) [112,113,114]; these compounds have demonstrated to be effective on both C481-mutated and unmutated BTK in preclinical studies. Remarkably, the BRUIN phase I/II trial has shown that pirtobrutinib achieves an ORR of 62% in CLL R/R to multiple lines of treatment, the majority of which had been previously treated with a covalent BTKi [115]. On these grounds, non-covalent BTKi offer a new frontier for CLL patients that are refractory to other pathway inhibitors, including covalent BTKi. Despite these clinically important advances, several mutations causing acquired resistance to non-covalent BTKi and some covalent BTKi have been recently reported, including point mutations in the tyrosine kinase domain of BTK such as V416L, A428D, M437R, T474I, and L528W [41]. Functional analysis of these mutations indicates that these amino acid changes impair BTK binding to both non-covalent and covalent BTKi [41]. Consistently, Blombery et al. recently described an enrichment of the BTK-L528W mutation in patients receiving the covalent BTKi zanubrutinib compared to patients treated with ibrutinib, raising the possibility of cross-resistance between zanubrutinib and the novel reversible and non-covalent BTKi (Figure 1) [116].
Different strategies to overcome the above-cited resistance to BTKi are currently under investigation. In particular, concerning ibrutinib-resistant CLL associated with PLCG2 mutations, in vitro studies have shown that the inhibition of SYK and LYN, both necessary for the activation of PLCγ2 independently of BTK, can overcome sustained survival signaling [104]. In a recent phase II study, the SYK inhibitor entospletinib resulted in a response in patients previously treated with inhibitors of the B cell receptor pathway, even in patients who had BTK and PLCG2 mutations. However, the ORR was low (33%), and PFS was 5.56 months [117].
Furthermore, proteolysis-targeting chimeras (PROTACs) may be a new approach to overcome BTKi resistance. PROTACs were shown to be effective against in vitro mutant BTK-C481 cells by inducing BTK degradation through ubiquitin-mediated protein degradation [118]. NX-2127 is the first-in-class targeted protein degrader of BTK, which in a preclinical study, was shown to induce the degradation of both wild-type and mutant BTK [119]. The clinical results of a first-in-human phase I trial on NX-2127 were reported recently. A total of 23 R/R CLL patients with a median of six prior therapies (2–11) were enrolled in the study. All patients had been previously treated with a covalent BTKi and/or venetoclax. This patient group, for whom no other therapeutic options were available, NX-2127, resulted in an ORR of 33% in 12 evaluable patients at a median follow-up of 5.6 months. These data support the use of BTK degraders in double- or triple-refractory patients, regardless of BTK or BCL2 mutation status [119].
4. Biomarkers of Refractoriness to BCL2 Inhibitors
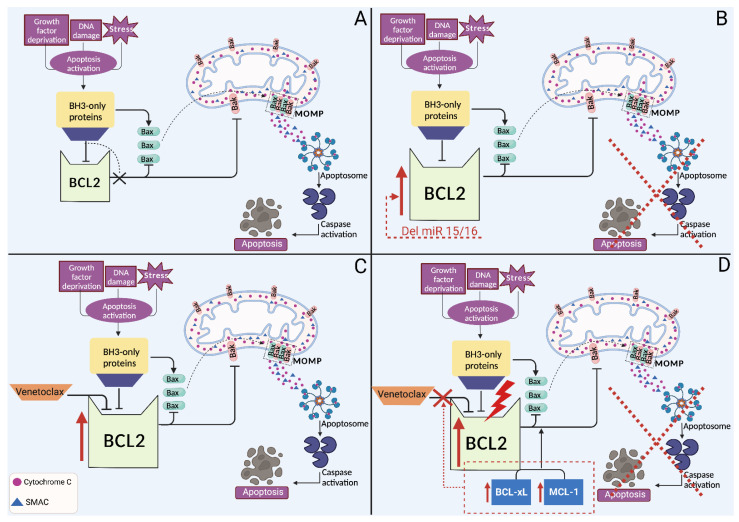
Mitochondrial apoptosis is controlled by the BCL2 family, which includes pro-apoptotic proteins (Bak and Bax), anti-apoptotic proteins (namely BCL2, BCL-xL, and MCL1) and BH3-only proteins (namely BIM, BID, BAD, PUMA, BIK, and NOXA) [120]. In normal cells, anti-apoptotic proteins play their role by binding and sequestering the mitochondria pore-forming proteins Bak and Bax. Upon cellular stress, BH3-only proteins bind to anti-apoptotic proteins via their BH3 domain, releasing pore-forming proteins. These pore-forming proteins bind to the outer mitochondrial membrane to trigger membrane permeability and the release of cytochrome C from mitochondria, allowing the assembly and activation of the apoptosome, which leads to the activation of a caspase cascade that initiates apoptosis (Figure 2) [120].
Figure 2.
Intrinsic apoptosis pathway and venetoclax resistance in CLL. (A) Upon activation of the intrinsic apoptosis pathway by cellular stress, BH3-only proteins inhibit the anti-apoptotic proteins BCL2, BCL-xL, and MCL1. This inhibition leads to the activation and oligomerization of Bak and Bax, resulting in mitochondrial outer membrane permeabilization (MOMP). MOMP-mediated release of cytochrome c and SMAC leads to the formation of apoptosome, which results in the activation of caspase proteins leading to cell death. The anti-apoptotic BCL2 proteins inhibit this process by sequestering the pro-apoptotic proteins by binding to their BH3 motifs. (B) In CLL, BCL2 is overexpressed in 20–40% of the cases. The loss of miR-15 and miR-16 miRNAs leads to increased levels of BCL2, providing a survival advantage for the tumor. (C) Venetoclax induces apoptosis by binding to BCL2 protein, which is commonly found to be overexpressed in CLL. (D) Among venetoclax resistance mechanisms (i) BCL2 point mutations, which reduce the affinity for venetoclax (ii) upregulation of MCL-1 and BCL-xL abrogates venetoclax antitumor effect.
Different aberrant mechanisms lead to the overexpression of BCL2 in the primary phase of CLL tumorigenesis, among which the loss of miR-15 and miR-16 at 13q14, which is detectable in 40–60% of CLL patients and hypomethylation of the BCL2 gene [121,122]. The miR-15 and miR-16 miRNAs physiologically inhibit the translation of the BCL2 protein by binding to a specific sequence on the corresponding mRNA. In CLL, the loss of these two miRNAs leads to increased levels of BCL2, providing a survival advantage for the tumor [121,122].
Venetoclax (formerly known as ABT-199) is a first-in-class, orally administrable, BH-3 mimetic drug designed to have high affinity and selectivity for BCL2 and a low affinity for MCL1 and BCL-xL, which are crucial for platelet survival [123]. Venetoclax was initially approved in 2016 as a monotherapy for relapsed CLL patients with del(17p) or TP53 mutation or who are not suitable for BCR inhibitors, as well as for patients without del(17p) or TP53 mutation and refractory to CIT and BCR pathway inhibitors. Despite the major clinical achievements obtained with the combination of venetoclax and an anti-CD20 mAb, a fraction of patients fail therapy and progress [35,124]. In that regard, long-term venetoclax treatment can eventually result in the expansion of resistant clones and progression, driven by clonal evolution [125]. Multiple mechanisms have been proposed to explain venetoclax resistance, including reduced drug binding due to gene mutations, upregulation of BCL2-related anti-apoptotic family members, and microenvironmental alterations [120].
4.1. Primary Resistance to BCL2 Inhibitors
Primary therapeutic resistance to venetoclax has been associated with intra-tumoral heterogeneity and clonal evolution. Pre-existent mutations in CLL sub-clones likely contribute to resistance by conferring a certain growth advantage or access to supportive microenvironment niches [126,127]. Primary venetoclax resistance may also be due to epigenetic mechanisms, including DNA methylation, post-translational histone modifications, and chromatin remodeling [128]. These alterations regulate the growth rate and response to environmental pressures, which ultimately influence tumor heterogeneity and clonal evolution [128]. On top of the mechanisms mentioned above, various microenvironmental signals (IL-10, CD40L, etc.) also contribute to intrinsic resistance by stimulating TLR9, that in turn activates NF-κB signaling [45]. Importantly, the activation of the transcription factor NF-κB leads to increased expression of the anti-apoptotic proteins BCL-xL and MCL1 [45].
4.2. Secondary Resistance to BCL2 Inhibitors
In a similar way to ibrutinib resistance, point mutations causing venetoclax resistance have also been reported. The first identified and most common mutation is G101V in the BH3-binding groove of BCL2, leading to refractoriness in about 15% of CLL patients treated with venetoclax [44]. This mutation reduces by 180-fold the affinity of BCL2 for venetoclax, preventing the drug from displacing proapoptotic BH-3-only proteins (e.g., BIM) from BCL2 [129]. In addition, the D103Y and F104I mutations were also found to cause drug resistance (Figure 2) [130]. Interestingly, both G101V and D103Y were detectable in several patients before the occurrence of clinical relapse, which raises the hypothesis that, in the future, these genetic changes might be considered as predictive biomarkers of treatment failure and lead to early intervention, e.g., by the addition of other therapeutic agents, such as BTKi [131].
The fact that BCL2 mutations were found only in a subset of patients suggested the involvement of other mechanisms leading to venetoclax resistance [129,130]. It is well established that the overexpression of the anti-apoptotic BCL-xL and MCL1 is associated with a higher risk of drug resistance [46]. Consistently, Ghia and colleagues found that higher expression of ROR1 before and after one year of venetoclax treatment is associated with accelerated disease progression and shorter OS regardless of IGHV mutation status. Notably, the increased expression of ROR1 is accompanied by the upregulation of WNT5a-ROR1 signaling, leading to the higher expression of ERK1/2 and NF-κB-target genes, including the BCL-xL protein, which may enhance venetoclax resistance since it inhibits apoptosis and is not significantly targeted by venetoclax [132]. Furthermore, the amplification of 1q23 encompassing MCL1 and PRKAB2 (a component of the AMPK pathway) and the overexpression of these two genes have been demonstrated in patients with venetoclax resistance [46].
Curiously, whole-exome sequencing and methylation profiling in a cohort of CLL patients before venetoclax treatment and at the time of venetoclax resistance revealed no genetic alterations in BCL2 [30]. However, most patients developed mutations in other cancer-related genes, including BRAF, NOTCH1, RB1, and TP53 or had a homozygous deletion of CDKN2A/B, suggesting a potential mechanism of resistance that involves the deregulation of these genes [30]. Lastly, the overexpression of NOTCH2 has also been potentially implicated as a novel mechanism of venetoclax resistance. In fact, Fiorcari et al. have recently reported that CLL patients harboring trisomy 12 have high levels of NOTCH2, leading to the upregulation of MCl-1, which in turn promotes cell survival by evading the proapoptotic effect of venetoclax [133].
4.3. Strategies to Overcome Resistance to BCL2 Inhibitors
Because BTKi and BCL2i inhibit different biological pathways, an obvious clinical strategy is to treat venetoclax-resistant patients with a BTKi. In two retrospective series with similar results, BTKi achieved an ORR of 84% to 91% and a median PFS of 32 to 34 months for patients with progressive disease (PD) after venetoclax, including those harboring BCL2 mutations [134,135]. Patients with prior BTKi intolerance who had developed venetoclax resistance could achieve durable remissions with BTKi rechallenging, preferentially with an alternative agent compared to the BTKi used previously in their clinical course [135]. In contrast, PI3Ki are associated with poor outcomes after venetoclax (median PFS, 5 months) [136].
Recent studies have shown that targeting epigenetic mechanisms with HDAC inhibitors, DNA methyltransferase inhibitors, or bromodomain reader protein inhibitors can downregulate the gene expression signature responsible for venetoclax resistance in different hematological malignancies [137]. In this respect, an in vitro analysis has demonstrated that the bromodomain and extra terminal proteins (BET) inhibitor JQ1 has an antitumor effect in CLL and, more importantly, that the combination of JQ1 and venetoclax enhances the apoptotic effect of the BCL2i [138]. These data point to the potential efficacy of BET inhibitors as a second-line treatment in case of venetoclax resistance. Additionally, because upregulation of MCL1 has been demonstrated to be involved in venetoclax resistance, the direct MCL1 antagonist AMG-176 has been shown to selectively kill CLL cells and act synergistically with venetoclax in vitro [139]. However, a phase I trial (NCT03797261) with the drug had to be discontinued due to safety concerns, leaving the door open for future MCL1 inhibitors development.
5. Biomarkers of Refractoriness to Immunotherapy
Cancer immunotherapy is a type of treatment that modulates the immune system in order to obtain an immune-mediated response against the tumor [140]. Immune checkpoint inhibitors and cellular immunotherapy for the treatment of CLL are currently under investigation, with conflicting results [141]. Due to its altered immune microenvironment and the promotion of immune dysfunction, CLL seemed to be an optimal candidate for immunotherapy, but for the same reasons, several difficulties have been encountered with immunotherapeutic approaches [141,142]. Several novel agents are under investigation for the immunotherapy of CLL, namely immune checkpoint inhibitors, chimeric antigen receptor (CAR)-T cells and bispecific/trispecific T/NK cells engagers [141]. Potential biomarkers of treatment refractoriness have been highlighted in recent years, including abnormal levels of circulating IL-10 and IL-6 and the reduced presence of specific memory T cell populations [24,47,48].
5.1. Abnormal IL-10 Levels
Higher circulating levels of IL-10 have been reported in advanced-stage CLL compared to early-stage and healthy patients [143]. IL-10 is an anti-inflammatory cytokine secreted by CLL neoplastic cells, and it is responsible, at least in part, for the suppression of the antitumor immune response observed in CLL through the intracellular signaling of its receptor IL-10R, expressed by various cell types, including T cells [144,145,146]. Recently, IL-10 has been proposed to be involved in refractoriness to immunotherapy with PD-1/PD-L1 immune checkpoint inhibitors [47].
The PD-1/PD-L1 axis is one of the most codified immune checkpoints, with an important role in the prevention of abnormal T-cell response [147]. PD-1 is a molecule expressed on the cellular membrane of normal T cells, which binds to its ligand PD-L1, physiologically found on the surface of antigen-presenting cells [148]. This interaction triggers the transduction of signaling pathways by the intracellular domain of PD-1, leading to the inhibition of PI3K/Akt and MAPK activation and finally resulting in T cell exhaustion and impaired function [147]. Since PD-1 and/or PD-L1 overexpression occurs in various tumors, the block of this axis by anti-PD1/PD-L1 mAbs has proven to be effective in several cancer types, including hematologic malignancies [149]. CLL patients overexpress PD-1 on the surface of T cells and PD-L1 on the external membrane of neoplastic B cells, thus providing an immunotolerant milieu that allows tumor escape from apoptosis [150].
Despite the apparently solid rationale for the treatment with immune checkpoint inhibitors, the results of a phase II study have shown the absence of efficacy of the anti-PD-1 mAb pembrolizumab, with an overall response rate (ORR) of 0% in relapsed CLL patients [151]. Because IL-10 serum levels are involved in refractoriness to immune checkpoint inhibitors, a combined approach of immune checkpoint inhibitors together with IL-10 inhibition has been attempted. Recently, initial promising results have been achieved with simultaneous treatment with IL-10-suppressing agents and pembrolizumab in CLL murine xenograft models [47]. The efficacy of pembrolizumab in reducing the tumor burden was amplified by 4.5-fold thanks to IL-10 suppression, paving the way for further investigations on the potential role of IL-10 as a predictive biomarker of resistance as well as a druggable target [47].
5.2. Potential Biomarkers of Refractoriness to CAR-T Cells
CAR-T cells are T lymphocytes extracted from the patient and engineered to express synthetic receptors, capable of specifically targeting and killing tumor cells through cytotoxic mechanisms [152]. After T cell extraction and ex vivo genetic engineering, CAR-T cells undergo the expansion process and are infused in the patient, who has been previously treated with a lymphodepleting conditioning regimen [153]. CARs consist of the fusion of an antigen-binding extracellular domain, a signaling CD3ζ subunit and one or more co-stimulatory intracellular domains [152,154]. Surprisingly, anti-CD19 CAR-T cell treatment has not reached the expected outcomes in CLL, with an average complete remission rate of ~30%, significantly lower compared to that obtained in acute lymphoblastic leukemia and diffuse large B cell lymphoma [155,156,157,158]. Several mechanisms of resistance to the treatment with anti-CD19 CAR-T cells in CLL have been proposed, principally due to T cell exhaustion promoted by the neoplastic clone [155]. The proposed mechanisms of resistance include impaired immune synapse formation, reduced expression of CD19 on the surface of CLL cells, production of tumor-derived extracellular vesicles, and higher expression of transmembrane inhibitory receptors on T cells, such as CTLA-4, LAG3, and PD-1 [159,160,161,162,163].
Recently, a genomic, phenotypic, and functional evaluation of CLL patients treated with anti-CD19 CAR-T cells has identified potential predictive biomarkers of response to treatment [48]. Higher blood levels of IL-6 correlated positively with response to treatment, making low serum IL-6 a biomarker of refractoriness to CAR-T cell-based immunotherapy [48]. Another candidate predictive biomarker was the presence of a significant population of CD27+CD45RO− CD8+ T cells in responders before CAR-T cell generation, but further investigation is needed to determine the clinical value of these findings [48]. Since refractoriness to immunotherapy with CAR-T cells is primarily due to T cell exhaustion, immunotherapeutic approaches with CAR-NK cells are under evaluation for the treatment of CLL [155,164]. CAR-NK cells are engineered NK cells of healthy donors, extracted from umbilical cord blood or peripheral blood, designed to express on their surface a CAR receptor in order to take advantage of NK cytotoxic response against target tumor cells [165]. Although early promising results have been obtained in ongoing phase I/II trials (NCT03056339, NCT04796675, NCT04245722) with CAR-NK cells in CLL treatment, additional testing is required to assess the safety and efficacy profiles of this novel therapeutic approach [155].
6. Conclusions and Perspectives
During the last few years, several pathway inhibitors have been introduced for the treatment of CLL, leading, on the one hand, to chemo-free therapeutic strategies and higher rates of clinical response, and on the other hand, to the emergence of new mechanisms of refractoriness. Remarkably, treatment resistance to pathway inhibitors, namely BTKi and BCL2i, has been progressively clarified in patients treated with these innovative agents. The need to overcome these novel challenges has paved the way to understanding the detailed molecular mechanisms of refractoriness and developing new pathway inhibitors. The search for predictive biomarkers that might facilitate the identification of preclinical resistance in CLL treated with pathway inhibitors has also represented a matter of investigation, although the current guidelines do not recommend sequential testing of resistance mutations in patients who are responding to pathway inhibitors.
The imposition of continuous selection pressure through the administration of uninterrupted monotherapies with pathway inhibitors, in particular BTKi, is likely to facilitate the emergence of resistance mutations in a substantial proportion of patients. Consequently, the use of time-limited treatment approaches to avoid continued drug exposure and the selection of BTKi- and venetoclax-resistant clones represents a potential strategy to circumvent resistance development [166]. An encouraging strategy to overcome resistance once it has emerged is the development of next-generation BTKi that do not covalently bind to the target and are, therefore, still active in CLL cells, with the most common resistance mutations at the binding domain. In that regard, pirtobrutinib has shown to be effective in R/R CLL patients carrying BTKi mutations associated with refractoriness to covalent BTKi. At the same time, novel acquired mutations have been described to cause resistance to non-covalent BTKi, such as pirtobrutinib.
Finally, knowledge of biomarkers of refractoriness to CLL immunotherapy is still scant. Predictive biomarkers of response to treatment with CAR-T cells include levels of serum IL-6 and CD27+CD45RO− CD8+ T cells, while no sufficient data are available on refractoriness to bispecific and trispecific T/NK cells engagers. Therefore, further investigations are needed to corroborate the initial findings of preclinical studies and early stage clinical trials.
Author Contributions
Conceptualization, all authors; investigation, all authors; writing—original draft preparation, N.M. and S.M.; writing—review and editing, all authors; funding acquisition, G.G. All authors have read and agreed to the published version of the manuscript.
Conflicts of Interest
N.M., S.M., B.F.M. and A.F.A. declare no conflict of interest for this specific work. G.G. declares advisory board and speaker’s bureau honoraria from AbbVie, AstraZeneca, BeiGene, Incyte, Janssen, and Roche.
Funding Statement
Work by the authors described in this review has been supported by Molecular bases of disease dissemination in lymphoid malignancies to optimize curative therapeutic strategies, (5 × 1000 No. 21198), Associazione Italiana per la Ricerca sul Cancro Foundation Milan, Italy; Progetti di Rilevante Interesse Nazionale (PRIN; 2015ZMRFEA), Rome, Italy; the AGING Project—Department of Excellence—DIMET, Università del Piemonte Orientale, Novara, Italy; Ricerca Finalizzata 2018 (project RF-2018-12365790), MoH, Rome, Italy; and AIL Novara Onlus, Novara, Italy.
Footnotes
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
References
- 1.Swerdlow S.H., Campo E., Harris N.L., Jaffe E.S., Pileri S.A., Stein H., Thiele J. Who Classification of Tumours of Haematopoietic and Lymphoid Tissues, Revised. 4th ed. Volume 2 International Agency for Research on Cancer Lyon; Lyon, France: 2017. [Google Scholar]
- 2.Alaggio R., Amador C., Anagnostopoulos I., Attygalle A.D., Araujo I.B.O., Berti E., Bhagat G., Borges A.M., Boyer D., Calaminici M., et al. The 5th edition of the World Health Organization Classification of Haematolymphoid Tumours: Lymphoid Neoplasms. Leukemia. 2022;36:1720–1748. doi: 10.1038/s41375-022-01620-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Rai K.R. 1 Progress in chronic lymphocytic leukaemia: A historical perspective. Baillière’s Clin. Haematol. 1993;6:757–765. doi: 10.1016/S0950-3536(05)80174-X. [DOI] [PubMed] [Google Scholar]
- 4.Hallek M., Al-Sawaf O. Chronic lymphocytic leukemia: 2022 update on diagnostic and therapeutic procedures. Am. J. Hematol. 2021;96:1679–1705. doi: 10.1002/ajh.26367. [DOI] [PubMed] [Google Scholar]
- 5.Rawstron A.C., Kreuzer K.-A., Soosapilla A., Spacek M., Stehlikova O., Gambell P., McIver-Brown N., Villamor N., Psarra K., Arroz M., et al. Reproducible diagnosis of chronic lymphocytic leukemia by flow cytometry: An European Research Initiative on CLL (ERIC) & European Society for Clinical Cell Analysis (ESCCA) Harmonisation project. Cytom. Part B Clin. Cytom. 2018;94:121–128. doi: 10.1002/cyto.b.21595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chronic Lymphocytic Leukemia-Cancer Stat Facts. Surveillance, Epidemiology, and End Results Program. [(accessed on 11 June 2023)];2023 Available online: https://seer.cancer.gov/statfacts/html/clyl.html.
- 7.Woyach J.A. Management of relapsed/refractory Chronic Lymphocytic Leukemia. Am. J. Hematol. 2022;97((Suppl. S2)):S11–S18. doi: 10.1002/ajh.26683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Smolej L. Refractory Chronic Lymphocytic Leukemia: A Therapeutic Challenge. Curr. Cancer Drug Targets. 2016;16:701–709. doi: 10.2174/1568009616666160408150032. [DOI] [PubMed] [Google Scholar]
- 9.Binet J.L., Auquier A., Dighiero G., Chastang C., Piguet H., Goasguen J., Vaugier G., Potron G., Colona P., Oberling F., et al. A new prognostic classification of chronic lymphocytic leukemia derived from a multivariate survival analysis. Cancer. 1981;48:198–206. doi: 10.1002/1097-0142(19810701)48:1<198::AID-CNCR2820480131>3.0.CO;2-V. [DOI] [PubMed] [Google Scholar]
- 10.Rai K.R., Sawitsky A., Cronkite E.P., Chanana A.D., Levy R.N., Pasternack B.S. Clinical staging of chronic lymphocytic leukemia. Blood. 1975;46:219–234. doi: 10.1182/blood.V46.2.219.219. [DOI] [PubMed] [Google Scholar]
- 11.The International CLL-IPI Working Group An international prognostic index for patients with chronic lymphocytic leukaemia (CLL-IPI): A meta-analysis of individual patient data. Lancet Oncol. 2016;17:779–790. doi: 10.1016/S1470-2045(16)30029-8. [DOI] [PubMed] [Google Scholar]
- 12.Condoluci A., Terzi di Bergamo L., Langerbeins P., Hoechstetter M.A., Herling C.D., De Paoli L., Delgado J., Rabe K.G., Gentile M., Doubek M., et al. International prognostic score for asymptomatic early-stage chronic lymphocytic leukemia. Blood. 2020;135:1859–1869. doi: 10.1182/blood.2019003453. [DOI] [PubMed] [Google Scholar]
- 13.Shadman M. Diagnosis and Treatment of Chronic Lymphocytic Leukemia: A Review. JAMA. 2023;329:918–932. doi: 10.1001/jama.2023.1946. [DOI] [PubMed] [Google Scholar]
- 14.Hallek M., Cheson B.D., Catovsky D., Caligaris-Cappio F., Dighiero G., Döhner H., Hillmen P., Keating M., Montserrat E., Chiorazzi N., et al. iwCLL guidelines for diagnosis, indications for treatment, response assessment, and supportive management of CLL. Blood. 2018;131:2745–2760. doi: 10.1182/blood-2017-09-806398. [DOI] [PubMed] [Google Scholar]
- 15.Mouhssine S., Gaidano G. Richter Syndrome: From Molecular Pathogenesis to Druggable Targets. Cancers. 2022;14:4644. doi: 10.3390/cancers14194644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Shanafelt T.D., Wang X.V., Hanson C.A., Paietta E.M., O’Brien S., Barrientos J., Jelinek D.F., Braggio E., Leis J.F., Zhang C.C., et al. Long-term outcomes for ibrutinib-rituximab and chemoimmunotherapy in CLL: Updated results of the E1912 trial. Blood. 2022;140:112–120. doi: 10.1182/blood.2021014960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Shanafelt T.D., Wang X.V., Kay N.E., Hanson C.A., O’Brien S., Barrientos J., Jelinek D.F., Braggio E., Leis J.F., Zhang C.C., et al. Ibrutinib–Rituximab or Chemoimmunotherapy for Chronic Lymphocytic Leukemia. N. Engl. J. Med. 2019;381:432–443. doi: 10.1056/NEJMoa1817073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Eichhorst B., Robak T., Montserrat E., Ghia P., Niemann C.U., Kater A.P., Gregor M., Cymbalista F., Buske C., Hillmen P., et al. Chronic lymphocytic leukaemia: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2021;32:23–33. doi: 10.1016/j.annonc.2020.09.019. [DOI] [PubMed] [Google Scholar]
- 19.Moia R., Dondolin R., De Propris M.S., Talotta D., Mouhssine S., Perutelli F., Reda G., Mattiello V., Rigolin G.M., Motta M., et al. Long-term benefit of IGHV mutated patients in a real-life multicenter cohort of FCR-treated chronic lymphocytic leukemia. Hematol. Oncol. 2022 doi: 10.1002/hon.3080. [DOI] [PubMed] [Google Scholar]
- 20.Ravinetto R., Guenzi P.D., Massat P., Gaidano G. Globalisation of clinical trials and ethics of benefit sharing. Lancet Haematol. 2014;1:e54–e56. doi: 10.1016/S2352-3026(14)00004-0. [DOI] [PubMed] [Google Scholar]
- 21.National Comprehensive Cancer Network Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma (Version 2.2023) [(accessed on 4 May 2023)]. Available online: https://www.nccn.org/professionals/physician_gls/pdf/cll.pdf.
- 22.Roeker L.E., Dreger P., Brown J.R., Lahoud O.B., Eyre T.A., Brander D.M., Skarbnik A., Coombs C.C., Kim H.T., Davids M., et al. Allogeneic stem cell transplantation for chronic lymphocytic leukemia in the era of novel agents. Blood Adv. 2020;4:3977–3989. doi: 10.1182/bloodadvances.2020001956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Shadman M., Maloney D.G. Immune Therapy for Chronic Lymphocytic Leukemia: Allogeneic Transplant, Chimeric Antigen Receptor T-cell Therapy, and Beyond. Hematol. Oncol. Clin. N. Am. 2021;35:847–862. doi: 10.1016/j.hoc.2021.03.011. [DOI] [PubMed] [Google Scholar]
- 24.Condoluci A., Rossi D. Genetic mutations in chronic lymphocytic leukemia: Impact on clinical treatment. Expert Rev. Hematol. 2019;12:89–98. doi: 10.1080/17474086.2019.1575130. [DOI] [PubMed] [Google Scholar]
- 25.Mansouri L., Thorvaldsdottir B., Sutton L.A., Karakatsoulis G., Meggendorfer M., Parker H., Nadeu F., Brieghel C., Laidou S., Moia R., et al. Different prognostic impact of recurrent gene mutations in chronic lymphocytic leukemia depending on IGHV gene somatic hypermutation status: A study by ERIC in HARMONY. Leukemia. 2023;37:339–347. doi: 10.1038/s41375-022-01802-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Vardi A., Agathangelidis A., Sutton L.-A., Ghia P., Rosenquist R., Stamatopoulos K. Immunogenetic Studies of Chronic Lymphocytic Leukemia: Revelations and Speculations about Ontogeny and Clinical Evolution. Cancer Res. 2014;74:4211–4216. doi: 10.1158/0008-5472.CAN-14-0630. [DOI] [PubMed] [Google Scholar]
- 27.Fischer K., Bahlo J., Fink A.M., Goede V., Herling C.D., Cramer P., Langerbeins P., von Tresckow J., Engelke A., Maurer C., et al. Long-term remissions after FCR chemoimmunotherapy in previously untreated patients with CLL: Updated results of the CLL8 trial. Blood. 2016;127:208–215. doi: 10.1182/blood-2015-06-651125. [DOI] [PubMed] [Google Scholar]
- 28.Estenfelder S., Tausch E., Robrecht S., Bahlo J., Goede V., Ritgen M., van Dongen J.J., Langerak A.W., Fingerle-Rowson G., Kneba M., et al. Gene Mutations and Treatment Outcome in the Context of Chlorambucil (Clb) without or with the Addition of Rituximab (R) or Obinutuzumab (GA-101, G)—Results of an Extensive Analysis of the Phase III Study CLL11 of the German CLL Study Group. Blood. 2016;128:3227. doi: 10.1182/blood.V128.22.3227.3227. [DOI] [Google Scholar]
- 29.Kadri S., Lee J., Fitzpatrick C., Galanina N., Sukhanova M., Venkataraman G., Sharma S., Long B., Petras K., Theissen M., et al. Clonal evolution underlying leukemia progression and Richter transformation in patients with ibrutinib-relapsed CLL. Blood Adv. 2017;1:715–727. doi: 10.1182/bloodadvances.2016003632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Herling C.D., Abedpour N., Weiss J., Schmitt A., Jachimowicz R.D., Merkel O., Cartolano M., Oberbeck S., Mayer P., Berg V., et al. Clonal dynamics towards the development of venetoclax resistance in chronic lymphocytic leukemia. Nat. Commun. 2018;9:727. doi: 10.1038/s41467-018-03170-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Nadeu F., Delgado J., Royo C., Baumann T., Stankovic T., Pinyol M., Jares P., Navarro A., Martín-García D., Beà S., et al. Clinical impact of clonal and subclonal TP53, SF3B1, BIRC3, NOTCH1, and ATM mutations in chronic lymphocytic leukemia. Blood. 2016;127:2122–2130. doi: 10.1182/blood-2015-07-659144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Rossi D., Rasi S., Spina V., Bruscaggin A., Monti S., Ciardullo C., Deambrogi C., Khiabanian H., Serra R., Bertoni F., et al. Integrated mutational and cytogenetic analysis identifies new prognostic subgroups in chronic lymphocytic leukemia. Blood. 2013;121:1403–1412. doi: 10.1182/blood-2012-09-458265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Baliakas P., Hadzidimitriou A., Sutton L.A., Rossi D., Minga E., Villamor N., Larrayoz M., Kminkova J., Agathangelidis A., Davis Z., et al. Recurrent mutations refine prognosis in chronic lymphocytic leukemia. Leukemia. 2015;29:329–336. doi: 10.1038/leu.2014.196. [DOI] [PubMed] [Google Scholar]
- 34.Diop F., Moia R., Favini C., Spaccarotella E., De Paoli L., Bruscaggin A., Spina V., Terzi-di-Bergamo L., Arruga F., Tarantelli C., et al. Biological and clinical implications of BIRC3 mutations in chronic lymphocytic leukemia. Haematologica. 2020;105:448–456. doi: 10.3324/haematol.2019.219550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Al-Sawaf O., Zhang C., Tandon M., Sinha A., Fink A.M., Robrecht S., Samoylova O., Liberati A.M., Pinilla-Ibarz J., Opat S., et al. Venetoclax plus obinutuzumab versus chlorambucil plus obinutuzumab for previously untreated chronic lymphocytic leukaemia (CLL14): Follow-up results from a multicentre, open-label, randomised, phase 3 trial. Lancet Oncol. 2020;21:1188–1200. doi: 10.1016/S1470-2045(20)30443-5. [DOI] [PubMed] [Google Scholar]
- 36.Ljungström V., Cortese D., Young E., Pandzic T., Mansouri L., Plevova K., Ntoufa S., Baliakas P., Clifford R., Sutton L.A., et al. Whole-exome sequencing in relapsing chronic lymphocytic leukemia: Clinical impact of recurrent RPS15 mutations. Blood. 2016;127:1007–1016. doi: 10.1182/blood-2015-10-674572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Benedetti D., Tissino E., Pozzo F., Bittolo T., Caldana C., Perini C., Martorelli D., Bravin V., D’Agaro T., Rossi F.M., et al. NOTCH1 mutations are associated with high CD49d expression in chronic lymphocytic leukemia: Link between the NOTCH1 and the NF-κB pathways. Leukemia. 2018;32:654–662. doi: 10.1038/leu.2017.296. [DOI] [PubMed] [Google Scholar]
- 38.Fabbri G., Rasi S., Rossi D., Trifonov V., Khiabanian H., Ma J., Grunn A., Fangazio M., Capello D., Monti S., et al. Analysis of the chronic lymphocytic leukemia coding genome: Role of NOTCH1 mutational activation. J. Exp. Med. 2011;208:1389–1401. doi: 10.1084/jem.20110921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Offner F., Robak T., Janssens A., Govind Babu K., Kloczko J., Grosicki S., Mayer J., Panagiotidis P., Schuh A., Pettitt A., et al. A five-year follow-up of untreated patients with chronic lymphocytic leukaemia treated with ofatumumab and chlorambucil: Final analysis of the Complement 1 phase 3 trial. Br. J. Haematol. 2020;190:736–740. doi: 10.1111/bjh.16625. [DOI] [PubMed] [Google Scholar]
- 40.Bonfiglio S., Sutton L.-A., Ljungström V., Capasso A., Pandzic T., Weström S., Foroughi-Asl H., Skaftason A., Gellerbring A., Lyander A., et al. BTK and PLCG2 remain unmutated in one third of patients with CLL relapsing on ibrutinib. Blood Adv. 2023 doi: 10.1182/bloodadvances.2022008821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Wang E., Mi X., Thompson M.C., Montoya S., Notti R.Q., Afaghani J., Durham B.H., Penson A., Witkowski M.T., Lu S.X., et al. Mechanisms of Resistance to Noncovalent Bruton’s Tyrosine Kinase Inhibitors. N. Engl. J. Med. 2022;386:735–743. doi: 10.1056/NEJMoa2114110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Quinquenel A., Fornecker L.M., Letestu R., Ysebaert L., Fleury C., Lazarian G., Dilhuydy M.S., Nollet D., Guieze R., Feugier P., et al. Prevalence of BTK and PLCG2 mutations in a real-life CLL cohort still on ibrutinib after 3 years: A FILO group study. Blood. 2019;134:641–644. doi: 10.1182/blood.2019000854. [DOI] [PubMed] [Google Scholar]
- 43.Woyach J.A., Furman R.R., Liu T.M., Ozer H.G., Zapatka M., Ruppert A.S., Xue L., Li D.H., Steggerda S.M., Versele M., et al. Resistance mechanisms for the Bruton’s tyrosine kinase inhibitor ibrutinib. N. Engl. J. Med. 2014;370:2286–2294. doi: 10.1056/NEJMoa1400029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Blombery P., Thompson E.R., Nguyen T., Birkinshaw R.W., Gong J.N., Chen X., McBean M., Thijssen R., Conway T., Anderson M.A., et al. Multiple BCL2 mutations cooccurring with Gly101Val emerge in chronic lymphocytic leukemia progression on venetoclax. Blood. 2020;135:773–777. doi: 10.1182/blood.2019004205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Jayappa K.D., Portell C.A., Gordon V.L., Capaldo B.J., Bekiranov S., Axelrod M.J., Brett L.K., Wulfkuhle J.D., Gallagher R.I., Petricoin E.F., et al. Microenvironmental agonists generate de novo phenotypic resistance to combined ibrutinib plus venetoclax in CLL and MCL. Blood Adv. 2017;1:933–946. doi: 10.1182/bloodadvances.2016004176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Guièze R., Liu V.M., Rosebrock D., Jourdain A.A., Hernández-Sánchez M., Martinez Zurita A., Sun J., Ten Hacken E., Baranowski K., Thompson P.A., et al. Mitochondrial Reprogramming Underlies Resistance to BCL-2 Inhibition in Lymphoid Malignancies. Cancer Cell. 2019;36:369–384.e313. doi: 10.1016/j.ccell.2019.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Rivas J.R., Liu Y., Alhakeem S.S., Eckenrode J.M., Marti F., Collard J.P., Zhang Y., Shaaban K.A., Muthusamy N., Hildebrandt G.C., et al. Interleukin-10 suppression enhances T-cell antitumor immunity and responses to checkpoint blockade in chronic lymphocytic leukemia. Leukemia. 2021;35:3188–3200. doi: 10.1038/s41375-021-01217-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Fraietta J.A., Lacey S.F., Orlando E.J., Pruteanu-Malinici I., Gohil M., Lundh S., Boesteanu A.C., Wang Y., O’Connor R.S., Hwang W.T., et al. Determinants of response and resistance to CD19 chimeric antigen receptor (CAR) T cell therapy of chronic lymphocytic leukemia. Nat. Med. 2018;24:563–571. doi: 10.1038/s41591-018-0010-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Small S., Ma S. Frontline Treatment for Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma (CLL/SLL): Targeted Therapy vs. Chemoimmunotherapy. Curr. Hematol. Malig. Rep. 2021;16:325–335. doi: 10.1007/s11899-021-00637-1. [DOI] [PubMed] [Google Scholar]
- 50.Hallek M., Fischer K., Fingerle-Rowson G., Fink A.M., Busch R., Mayer J., Hensel M., Hopfinger G., Hess G., von Grünhagen U., et al. Addition of rituximab to fludarabine and cyclophosphamide in patients with chronic lymphocytic leukaemia: A randomised, open-label, phase 3 trial. Lancet. 2010;376:1164–1174. doi: 10.1016/S0140-6736(10)61381-5. [DOI] [PubMed] [Google Scholar]
- 51.Goede V., Fischer K., Busch R., Engelke A., Eichhorst B., Wendtner C.M., Chagorova T., de la Serna J., Dilhuydy M.S., Illmer T., et al. Obinutuzumab plus chlorambucil in patients with CLL and coexisting conditions. N. Engl. J. Med. 2014;370:1101–1110. doi: 10.1056/NEJMoa1313984. [DOI] [PubMed] [Google Scholar]
- 52.Dal Porto J.M., Gauld S.B., Merrell K.T., Mills D., Pugh-Bernard A.E., Cambier J. B cell antigen receptor signaling 101. Mol. Immunol. 2004;41:599–613. doi: 10.1016/j.molimm.2004.04.008. [DOI] [PubMed] [Google Scholar]
- 53.Maizels N. Immunoglobulin Gene Diversification. Annu. Rev. Genet. 2005;39:23–46. doi: 10.1146/annurev.genet.39.073003.110544. [DOI] [PubMed] [Google Scholar]
- 54.Di Noia J.M., Neuberger M.S. Molecular mechanisms of antibody somatic hypermutation. Annu. Rev. Biochem. 2007;76:1–22. doi: 10.1146/annurev.biochem.76.061705.090740. [DOI] [PubMed] [Google Scholar]
- 55.Bosch F., Dalla-Favera R. Chronic lymphocytic leukaemia: From genetics to treatment. Nat. Rev. Clin. Oncol. 2019;16:684–701. doi: 10.1038/s41571-019-0239-8. [DOI] [PubMed] [Google Scholar]
- 56.Klein U., Tu Y., Stolovitzky G.A., Mattioli M., Cattoretti G., Husson H., Freedman A., Inghirami G., Cro L., Baldini L., et al. Gene expression profiling of B cell chronic lymphocytic leukemia reveals a homogeneous phenotype related to memory B cells. J. Exp. Med. 2001;194:1625–1638. doi: 10.1084/jem.194.11.1625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Visentin A., Facco M., Gurrieri C., Pagnin E., Martini V., Imbergamo S., Frezzato F., Trimarco V., Severin F., Raggi F., et al. Prognostic and Predictive Effect of IGHV Mutational Status and Load in Chronic Lymphocytic Leukemia: Focus on FCR and BR Treatments. Clin. Lymphoma Myeloma Leuk. 2019;19:678–685.e674. doi: 10.1016/j.clml.2019.03.002. [DOI] [PubMed] [Google Scholar]
- 58.Damle R.N., Wasil T., Fais F., Ghiotto F., Valetto A., Allen S.L., Buchbinder A., Budman D., Dittmar K., Kolitz J., et al. Ig V Gene Mutation Status and CD38 Expression As Novel Prognostic Indicators in Chronic Lymphocytic Leukemia: Presented in part at the 40th Annual Meeting of The American Society of Hematology, held in Miami Beach, FL, December 4–8, 1998. Blood. 1999;94:1840–1847. doi: 10.1182/blood.V94.6.1840. [DOI] [PubMed] [Google Scholar]
- 59.Hamblin T.J., Davis Z., Gardiner A., Oscier D.G., Stevenson F.K. Unmutated Ig VH Genes Are Associated With a More Aggressive Form of Chronic Lymphocytic Leukemia. Blood. 1999;94:1848–1854. doi: 10.1182/blood.V94.6.1848. [DOI] [PubMed] [Google Scholar]
- 60.Rossi D., Terzi-di-Bergamo L., De Paoli L., Cerri M., Ghilardi G., Chiarenza A., Bulian P., Visco C., Mauro F.R., Morabito F., et al. Molecular prediction of durable remission after first-line fludarabine-cyclophosphamide-rituximab in chronic lymphocytic leukemia. Blood. 2015;126:1921–1924. doi: 10.1182/blood-2015-05-647925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Fischer K., Cramer P., Busch R., Böttcher S., Bahlo J., Schubert J., Pflüger K.H., Schott S., Goede V., Isfort S., et al. Bendamustine in combination with rituximab for previously untreated patients with chronic lymphocytic leukemia: A multicenter phase II trial of the German Chronic Lymphocytic Leukemia Study Group. J. Clin. Oncol. 2012;30:3209–3216. doi: 10.1200/JCO.2011.39.2688. [DOI] [PubMed] [Google Scholar]
- 62.Gentile M., Zirlik K., Ciolli S., Mauro F.R., Di Renzo N., Mastrullo L., Angrilli F., Molica S., Tripepi G., Giordano A., et al. Combination of bendamustine and rituximab as front-line therapy for patients with chronic lymphocytic leukaemia: Multicenter, retrospective clinical practice experience with 279 cases outside of controlled clinical trials. Eur. J. Cancer. 2016;60:154–165. doi: 10.1016/j.ejca.2016.03.069. [DOI] [PubMed] [Google Scholar]
- 63.Byrd J.C., Hillmen P., O’Brien S., Barrientos J.C., Reddy N.M., Coutre S., Tam C.S., Mulligan S.P., Jaeger U., Barr P.M., et al. Long-term follow-up of the RESONATE phase 3 trial of ibrutinib vs ofatumumab. Blood. 2019;133:2031–2042. doi: 10.1182/blood-2018-08-870238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Cull G., Burger J.A., Opat S., Gottlieb D., Verner E., Trotman J., Marlton P., Munoz J., Johnston P., Simpson D., et al. Zanubrutinib for treatment-naïve and relapsed/refractory chronic lymphocytic leukaemia: Long-term follow-up of the phase I/II AU-003 study. Br. J. Haematol. 2022;196:1209–1218. doi: 10.1111/bjh.17994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Sharman J.P., Egyed M., Jurczak W., Skarbnik A., Pagel J.M., Flinn I.W., Kamdar M., Munir T., Walewska R., Corbett G., et al. Efficacy and safety in a 4-year follow-up of the ELEVATE-TN study comparing acalabrutinib with or without obinutuzumab versus obinutuzumab plus chlorambucil in treatment-naïve chronic lymphocytic leukemia. Leukemia. 2022;36:1171–1175. doi: 10.1038/s41375-021-01485-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Duffy M.J., Synnott N.C., Crown J. Mutant p53 as a target for cancer treatment. Eur. J. Cancer. 2017;83:258–265. doi: 10.1016/j.ejca.2017.06.023. [DOI] [PubMed] [Google Scholar]
- 67.Norbury C.J., Zhivotovsky B. DNA damage-induced apoptosis. Oncogene. 2004;23:2797–2808. doi: 10.1038/sj.onc.1207532. [DOI] [PubMed] [Google Scholar]
- 68.Buccheri V., Barreto W.G., Fogliatto L.M., Capra M., Marchiani M., Rocha V. Prognostic and therapeutic stratification in CLL: Focus on 17p deletion and p53 mutation. Ann. Hematol. 2018;97:2269–2278. doi: 10.1007/s00277-018-3503-6. [DOI] [PubMed] [Google Scholar]
- 69.Rossi D., Gaidano G. The clinical implications of gene mutations in chronic lymphocytic leukaemia. Br. J. Cancer. 2016;114:849–854. doi: 10.1038/bjc.2016.78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Malcikova J., Tausch E., Rossi D., Sutton L.A., Soussi T., Zenz T., Kater A.P., Niemann C.U., Gonzalez D., Davi F., et al. ERIC recommendations for TP53 mutation analysis in chronic lymphocytic leukemia—Update on methodological approaches and results interpretation. Leukemia. 2018;32:1070–1080. doi: 10.1038/s41375-017-0007-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Zarnegar B.J., Wang Y., Mahoney D.J., Dempsey P.W., Cheung H.H., He J., Shiba T., Yang X., Yeh W.C., Mak T.W., et al. Noncanonical NF-kappaB activation requires coordinated assembly of a regulatory complex of the adaptors cIAP1, cIAP2, TRAF2 and TRAF3 and the kinase NIK. Nat. Immunol. 2008;9:1371–1378. doi: 10.1038/ni.1676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Lau R., Niu M.Y., Pratt M.A. cIAP2 represses IKKα/β-mediated activation of MDM2 to prevent p53 degradation. Cell Cycle. 2012;11:4009–4019. doi: 10.4161/cc.22223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Asslaber D., Wacht N., Leisch M., Qi Y., Maeding N., Hufnagl C., Jansko B., Zaborsky N., Villunger A., Hartmann T.N., et al. BIRC3 Expression Predicts CLL Progression and Defines Treatment Sensitivity via Enhanced NF-κB Nuclear Translocation. Clin. Cancer Res. 2019;25:1901–1912. doi: 10.1158/1078-0432.CCR-18-1548. [DOI] [PubMed] [Google Scholar]
- 74.Tausch E., Schneider C., Robrecht S., Zhang C., Dolnik A., Bloehdorn J., Bahlo J., Al-Sawaf O., Ritgen M., Fink A.-M., et al. Prognostic and predictive impact of genetic markers in patients with CLL treated with obinutuzumab and venetoclax. Blood. 2020;135:2402–2412. doi: 10.1182/blood.2019004492. [DOI] [PubMed] [Google Scholar]
- 75.Burger J.A., Robak T., Demirkan F., Bairey O., Moreno C., Simpson D., Munir T., Stevens D.A., Dai S., Cheung L.W.K., et al. Outcomes of First-Line Ibrutinib in Patients with Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma (CLL/SLL) and High-Risk Genomic Features with up to 6.5 Years Follow-up: Integrated Analysis of Two Phase 3 Studies (RESONATE-2 and iLLUMINATE) Blood. 2020;136:25–26. doi: 10.1182/blood-2020-134437. [DOI] [Google Scholar]
- 76.Moia R., Gaidano G., Rossi D. Reply to Aron P. Kater et al. Haematologica. 2020;105:e384. doi: 10.3324/haematol.2020.255596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Woyach J.A., Barr P.M., Kipps T.J., Barrientos J.C., Ahn I.E., Ghia P., Girardi V., Hsu E., Jermain M., Burger J.A. Characteristics and Clinical Outcomes of Patients with Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma Receiving Ibrutinib for ≥5 Years in the RESONATE-2 Study. Cancers. 2023;15:507. doi: 10.3390/cancers15020507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Lobry C., Oh P., Aifantis I. Oncogenic and tumor suppressor functions of Notch in cancer: It’s NOTCH what you think. J. Exp. Med. 2011;208:1931–1935. doi: 10.1084/jem.20111855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Rosati E., Baldoni S., De Falco F., Del Papa B., Dorillo E., Rompietti C., Albi E., Falzetti F., Di Ianni M., Sportoletti P. NOTCH1 Aberrations in Chronic Lymphocytic Leukemia. Front. Oncol. 2018;8:229. doi: 10.3389/fonc.2018.00229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Di Ianni M., Baldoni S., Rosati E., Ciurnelli R., Cavalli L., Martelli M.F., Marconi P., Screpanti I., Falzetti F. A new genetic lesion in B-CLL: A NOTCH1 PEST domain mutation. Br. J. Haematol. 2009;146:689–691. doi: 10.1111/j.1365-2141.2009.07816.x. [DOI] [PubMed] [Google Scholar]
- 81.Rosati E., Sabatini R., Rampino G., Tabilio A., Di Ianni M., Fettucciari K., Bartoli A., Coaccioli S., Screpanti I., Marconi P. Constitutively activated Notch signaling is involved in survival and apoptosis resistance of B-CLL cells. Blood. 2009;113:856–865. doi: 10.1182/blood-2008-02-139725. [DOI] [PubMed] [Google Scholar]
- 82.Hillmen P., Robak T., Janssens A., Babu K.G., Kloczko J., Grosicki S., Doubek M., Panagiotidis P., Kimby E., Schuh A., et al. Chlorambucil plus ofatumumab versus chlorambucil alone in previously untreated patients with chronic lymphocytic leukaemia (COMPLEMENT 1): A randomised, multicentre, open-label phase 3 trial. Lancet. 2015;385:1873–1883. doi: 10.1016/S0140-6736(15)60027-7. [DOI] [PubMed] [Google Scholar]
- 83.Pozzo F., Bittolo T., Arruga F., Bulian P., Macor P., Tissino E., Gizdic B., Rossi F.M., Bomben R., Zucchetto A., et al. NOTCH1 mutations associate with low CD20 level in chronic lymphocytic leukemia: Evidence for a NOTCH1 mutation-driven epigenetic dysregulation. Leukemia. 2016;30:182–189. doi: 10.1038/leu.2015.182. [DOI] [PubMed] [Google Scholar]
- 84.Ahn I.E., Brown J.R. Targeting Bruton’s Tyrosine Kinase in CLL. Front. Immunol. 2021;12:687458. doi: 10.3389/fimmu.2021.687458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Pal Singh S., Dammeijer F., Hendriks R.W. Role of Bruton’s tyrosine kinase in B cells and malignancies. Mol. Cancer. 2018;17:57. doi: 10.1186/s12943-018-0779-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Burger J.A. Bruton Tyrosine Kinase Inhibitors: Present and Future. Cancer J. 2019;25:386–393. doi: 10.1097/PPO.0000000000000412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Burger J.A., Wiestner A. Targeting B cell receptor signalling in cancer: Preclinical and clinical advances. Nat. Rev. Cancer. 2018;18:148–167. doi: 10.1038/nrc.2017.121. [DOI] [PubMed] [Google Scholar]
- 88.Burger J.A., Chiorazzi N. B cell receptor signaling in chronic lymphocytic leukemia. Trends Immunol. 2013;34:592–601. doi: 10.1016/j.it.2013.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Burger J.A., Tedeschi A., Barr P.M., Robak T., Owen C., Ghia P., Bairey O., Hillmen P., Bartlett N.L., Li J., et al. Ibrutinib as Initial Therapy for Patients with Chronic Lymphocytic Leukemia. N. Engl. J. Med. 2015;373:2425–2437. doi: 10.1056/NEJMoa1509388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Byrd J.C., Brown J.R., O’Brien S., Barrientos J.C., Kay N.E., Reddy N.M., Coutre S., Tam C.S., Mulligan S.P., Jaeger U., et al. Ibrutinib versus ofatumumab in previously treated chronic lymphoid leukemia. N. Engl. J. Med. 2014;371:213–223. doi: 10.1056/NEJMoa1400376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Pan Z., Scheerens H., Li S.J., Schultz B.E., Sprengeler P.A., Burrill L.C., Mendonca R.V., Sweeney M.D., Scott K.C., Grothaus P.G., et al. Discovery of selective irreversible inhibitors for Bruton’s tyrosine kinase. Chem. Med. Chem. 2007;2:58–61. doi: 10.1002/cmdc.200600221. [DOI] [PubMed] [Google Scholar]
- 92.Guarini A., Chiaretti S., Tavolaro S., Maggio R., Peragine N., Citarella F., Ricciardi M.R., Santangelo S., Marinelli M., De Propris M.S., et al. BCR ligation induced by IgM stimulation results in gene expression and functional changes only in IgV H unmutated chronic lymphocytic leukemia (CLL) cells. Blood. 2008;112:782–792. doi: 10.1182/blood-2007-12-127688. [DOI] [PubMed] [Google Scholar]
- 93.Ganatra S., Sharma A., Shah S., Chaudhry G.M., Martin D.T., Neilan T.G., Mahmood S.S., Barac A., Groarke J.D., Hayek S.S., et al. Ibrutinib-Associated Atrial Fibrillation. JACC Clin. Electrophysiol. 2018;4:1491–1500. doi: 10.1016/j.jacep.2018.06.004. [DOI] [PubMed] [Google Scholar]
- 94.Kaur V., Swami A. Ibrutinib in CLL: A focus on adverse events, resistance, and novel approaches beyond ibrutinib. Ann. Hematol. 2017;96:1175–1184. doi: 10.1007/s00277-017-2973-2. [DOI] [PubMed] [Google Scholar]
- 95.Holohan C., Van Schaeybroeck S., Longley D.B., Johnston P.G. Cancer drug resistance: An evolving paradigm. Nat. Rev. Cancer. 2013;13:714–726. doi: 10.1038/nrc3599. [DOI] [PubMed] [Google Scholar]
- 96.Lee J., Wang Y.L. Prognostic and Predictive Molecular Biomarkers in Chronic Lymphocytic Leukemia. J. Mol. Diagn. 2020;22:1114–1125. doi: 10.1016/j.jmoldx.2020.06.004. [DOI] [PubMed] [Google Scholar]
- 97.Smith C.I.E., Burger J.A. Resistance Mutations to BTK Inhibitors Originate From the NF-κB but Not From the PI3K-RAS-MAPK Arm of the B Cell Receptor Signaling Pathway. Front. Immunol. 2021;12:689472. doi: 10.3389/fimmu.2021.689472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Campo E., Cymbalista F., Ghia P., Jäger U., Pospisilova S., Rosenquist R., Schuh A., Stilgenbauer S. TP53 aberrations in chronic lymphocytic leukemia: An overview of the clinical implications of improved diagnostics. Haematologica. 2018;103:1956–1968. doi: 10.3324/haematol.2018.187583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Maddocks K.J., Ruppert A.S., Lozanski G., Heerema N.A., Zhao W., Abruzzo L., Lozanski A., Davis M., Gordon A., Smith L.L., et al. Etiology of Ibrutinib Therapy Discontinuation and Outcomes in Patients With Chronic Lymphocytic Leukemia. JAMA Oncol. 2015;1:80–87. doi: 10.1001/jamaoncol.2014.218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Ahn I.E., Tian X., Ipe D., Cheng M., Albitar M., Tsao L.C., Zhang L., Ma W., Herman S.E.M., Gaglione E.M., et al. Prediction of Outcome in Patients With Chronic Lymphocytic Leukemia Treated With Ibrutinib: Development and Validation of a Four-Factor Prognostic Model. J. Clin. Oncol. 2021;39:576–585. doi: 10.1200/JCO.20.00979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Lama T.G., Kyung D., O’Brien S. Mechanisms of ibrutinib resistance in chronic lymphocytic leukemia and alternative treatment strategies. Expert Rev. Hematol. 2020;13:871–883. doi: 10.1080/17474086.2020.1797482. [DOI] [PubMed] [Google Scholar]
- 102.Nakhoda S., Vistarop A., Wang Y.L. Resistance to Bruton tyrosine kinase inhibition in chronic lymphocytic leukaemia and non-Hodgkin lymphoma. Br. J. Haematol. 2023;200:137–149. doi: 10.1111/bjh.18418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Cheng S., Guo A., Lu P., Ma J., Coleman M., Wang Y.L. Functional characterization of BTK(C481S) mutation that confers ibrutinib resistance: Exploration of alternative kinase inhibitors. Leukemia. 2015;29:895–900. doi: 10.1038/leu.2014.263. [DOI] [PubMed] [Google Scholar]
- 104.Liu T.M., Woyach J.A., Zhong Y., Lozanski A., Lozanski G., Dong S., Strattan E., Lehman A., Zhang X., Jones J.A., et al. Hypermorphic mutation of phospholipase C, γ2 acquired in ibrutinib-resistant CLL confers BTK independency upon B-cell receptor activation. Blood. 2015;126:61–68. doi: 10.1182/blood-2015-02-626846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Ahn I.E., Underbayev C., Albitar A., Herman S.E., Tian X., Maric I., Arthur D.C., Wake L., Pittaluga S., Yuan C.M., et al. Clonal evolution leading to ibrutinib resistance in chronic lymphocytic leukemia. Blood. 2017;129:1469–1479. doi: 10.1182/blood-2016-06-719294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Woyach J.A., Ruppert A.S., Guinn D., Lehman A., Blachly J.S., Lozanski A., Heerema N.A., Zhao W., Coleman J., Jones D., et al. BTK(C481S)-Mediated Resistance to Ibrutinib in Chronic Lymphocytic Leukemia. J. Clin. Oncol. 2017;35:1437–1443. doi: 10.1200/JCO.2016.70.2282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Burger J.A., Landau D.A., Taylor-Weiner A., Bozic I., Zhang H., Sarosiek K., Wang L., Stewart C., Fan J., Hoellenriegel J., et al. Clonal evolution in patients with chronic lymphocytic leukaemia developing resistance to BTK inhibition. Nat. Commun. 2016;7:11589. doi: 10.1038/ncomms11589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Byrd J.C., Hillmen P., Ghia P., Kater A.P., Chanan-Khan A., Furman R.R., O’Brien S., Yenerel M.N., Illés A., Kay N., et al. Acalabrutinib Versus Ibrutinib in Previously Treated Chronic Lymphocytic Leukemia: Results of the First Randomized Phase III Trial. J. Clin. Oncol. 2021;39:3441–3452. doi: 10.1200/JCO.21.01210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Brown J.R., Eichhorst B., Hillmen P., Jurczak W., Kaźmierczak M., Lamanna N., O’Brien S.M., Tam C.S., Qiu L., Zhou K., et al. Zanubrutinib or Ibrutinib in Relapsed or Refractory Chronic Lymphocytic Leukemia. N. Engl. J. Med. 2023;388:319–332. doi: 10.1056/NEJMoa2211582. [DOI] [PubMed] [Google Scholar]
- 110.Woyach J., Huang Y., Rogers K., Bhat S.A., Grever M.R., Lozanski A., Doong T.-J., Blachly J.S., Lozanski G., Jones D., et al. Resistance to Acalabrutinib in CLL Is Mediated Primarily By BTK Mutations. Blood. 2019;134:504. doi: 10.1182/blood-2019-127674. [DOI] [Google Scholar]
- 111.Handunnetti S.M., Tang C.P.S., Nguyen T., Zhou X., Thompson E., Sun H., Xing H., Zhang B., Guo Y., Sutton L.A., et al. BTK Leu528Trp—A Potential Secondary Resistance Mechanism Specific for Patients with Chronic Lymphocytic Leukemia Treated with the Next Generation BTK Inhibitor Zanubrutinib. Blood. 2019;134:170. doi: 10.1182/blood-2019-125488. [DOI] [Google Scholar]
- 112.Bond D.A., Woyach J.A. Targeting BTK in CLL: Beyond Ibrutinib. Curr. Hematol. Malig. Rep. 2019;14:197–205. doi: 10.1007/s11899-019-00512-0. [DOI] [PubMed] [Google Scholar]
- 113.Puła B., Gołos A., Górniak P., Jamroziak K. Overcoming Ibrutinib Resistance in Chronic Lymphocytic Leukemia. Cancers. 2019;11:1834. doi: 10.3390/cancers11121834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Frustaci A.M., Deodato M., Zamprogna G., Cairoli R., Montillo M., Tedeschi A. Next Generation BTK Inhibitors in CLL: Evolving Challenges and New Opportunities. Cancers. 2023;15:1504. doi: 10.3390/cancers15051504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Mato A.R., Shah N.N., Jurczak W., Cheah C.Y., Pagel J.M., Woyach J.A., Fakhri B., Eyre T.A., Lamanna N., Patel M.R., et al. Pirtobrutinib in relapsed or refractory B-cell malignancies (BRUIN): A phase 1/2 study. Lancet. 2021;397:892–901. doi: 10.1016/S0140-6736(21)00224-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Blombery P., Thompson E.R., Lew T.E., Tiong I.S., Bennett R., Cheah C.Y., Lewis K.L., Handunnetti S.M., Tang C.P.S., Roberts A., et al. Enrichment of BTK Leu528Trp mutations in patients with CLL on zanubrutinib: Potential for pirtobrutinib cross-resistance. Blood Adv. 2022;6:5589–5592. doi: 10.1182/bloodadvances.2022008325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Awan F.T., Thirman M.J., Patel-Donnelly D., Assouline S., Rao A.V., Ye W., Hill B., Sharman J.P. Entospletinib monotherapy in patients with relapsed or refractory chronic lymphocytic leukemia previously treated with B-cell receptor inhibitors: Results of a phase 2 study. Leuk Lymphoma. 2019;60:1972–1977. doi: 10.1080/10428194.2018.1562180. [DOI] [PubMed] [Google Scholar]
- 118.Buhimschi A.D., Armstrong H.A., Toure M., Jaime-Figueroa S., Chen T.L., Lehman A.M., Woyach J.A., Johnson A.J., Byrd J.C., Crews C.M. Targeting the C481S Ibrutinib-Resistance Mutation in Bruton’s Tyrosine Kinase Using PROTAC-Mediated Degradation. Biochemistry. 2018;57:3564–3575. doi: 10.1021/acs.biochem.8b00391. [DOI] [PubMed] [Google Scholar]
- 119.Mato A.R., Wierda W.G., Ai W.Z., Flinn I.W., Tees M., Patel M.R., Patel K., O’Brien S., Bond D.A., Roeker L.E., et al. NX-2127-001, a First-in-Human Trial of NX-2127, a Bruton’s Tyrosine Kinase-Targeted Protein Degrader, in Patients with Relapsed or Refractory Chronic Lymphocytic Leukemia and B-Cell Malignancies. Blood. 2022;140:2329–2332. doi: 10.1182/blood-2022-164772. [DOI] [Google Scholar]
- 120.Kapoor I., Bodo J., Hill B.T., Hsi E.D., Almasan A. Targeting BCL-2 in B-cell malignancies and overcoming therapeutic resistance. Cell Death Dis. 2020;11:941. doi: 10.1038/s41419-020-03144-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Ruefli-Brasse A., Reed J.C. Therapeutics targeting Bcl-2 in hematological malignancies. Biochem. J. 2017;474:3643–3657. doi: 10.1042/BCJ20170080. [DOI] [PubMed] [Google Scholar]
- 122.Cimmino A., Calin G.A., Fabbri M., Iorio M.V., Ferracin M., Shimizu M., Wojcik S.E., Aqeilan R.I., Zupo S., Dono M., et al. miR-15 and miR-16 induce apoptosis by targeting BCL2. Proc. Natl. Acad. Sci. USA. 2005;102:13944–13949. doi: 10.1073/pnas.0506654102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Lasica M., Anderson M.A. Review of Venetoclax in CLL, AML and Multiple Myeloma. J. Pers. Med. 2021;11:463. doi: 10.3390/jpm11060463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Seymour J.F., Kipps T.J., Eichhorst B., Hillmen P., D’Rozario J., Assouline S., Owen C., Gerecitano J., Robak T., De la Serna J., et al. Venetoclax-Rituximab in Relapsed or Refractory Chronic Lymphocytic Leukemia. N. Engl. J. Med. 2018;378:1107–1120. doi: 10.1056/NEJMoa1713976. [DOI] [PubMed] [Google Scholar]
- 125.Izzo F., Landau D.A. A BAX door to venetoclax resistance. Blood. 2022;139:1124–1126. doi: 10.1182/blood.2021013788. [DOI] [PubMed] [Google Scholar]
- 126.Landau D.A., Carter S.L., Stojanov P., McKenna A., Stevenson K., Lawrence M.S., Sougnez C., Stewart C., Sivachenko A., Wang L., et al. Evolution and impact of subclonal mutations in chronic lymphocytic leukemia. Cell. 2013;152:714–726. doi: 10.1016/j.cell.2013.01.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Gutierrez C., Wu C.J. Clonal dynamics in chronic lymphocytic leukemia. Blood Adv. 2019;3:3759–3769. doi: 10.1182/bloodadvances.2019000367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Guièze R., Wu C.J. Genomic and epigenomic heterogeneity in chronic lymphocytic leukemia. Blood. 2015;126:445–453. doi: 10.1182/blood-2015-02-585042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Blombery P., Anderson M.A., Gong J.N., Thijssen R., Birkinshaw R.W., Thompson E.R., Teh C.E., Nguyen T., Xu Z., Flensburg C., et al. Acquisition of the Recurrent Gly101Val Mutation in BCL2 Confers Resistance to Venetoclax in Patients with Progressive Chronic Lymphocytic Leukemia. Cancer Discov. 2019;9:342–353. doi: 10.1158/2159-8290.CD-18-1119. [DOI] [PubMed] [Google Scholar]
- 130.Tausch E., Close W., Dolnik A., Bloehdorn J., Chyla B., Bullinger L., Döhner H., Mertens D., Stilgenbauer S. Venetoclax resistance and acquired BCL2 mutations in chronic lymphocytic leukemia. Haematologica. 2019;104:e434–e437. doi: 10.3324/haematol.2019.222588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Blombery P. Mechanisms of intrinsic and acquired resistance to venetoclax in B-cell lymphoproliferative disease. Leuk Lymphoma. 2020;61:257–262. doi: 10.1080/10428194.2019.1660974. [DOI] [PubMed] [Google Scholar]
- 132.Ghia E.M., Rassenti L.Z., Choi M.Y., Quijada-Álamo M., Chu E., Widhopf G.F., 2nd, Kipps T.J. High expression level of ROR1 and ROR1-signaling associates with venetoclax resistance in chronic lymphocytic leukemia. Leukemia. 2022;36:1609–1618. doi: 10.1038/s41375-022-01543-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Fiorcari S., Maffei R., Atene C.G., Mesini N., Maccaferri M., Leonardi G., Martinelli S., Paolini A., Nasillo V., Debbia G., et al. Notch2 Increases the Resistance to Venetoclax-Induced Apoptosis in Chronic Lymphocytic Leukemia B Cells by Inducing Mcl-1. Front. Oncol. 2021;11:777587. doi: 10.3389/fonc.2021.777587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Lin V.S., Lew T.E., Handunnetti S.M., Blombery P., Nguyen T., Westerman D.A., Kuss B.J., Tam C.S., Roberts A.W., Seymour J.F., et al. BTK inhibitor therapy is effective in patients with CLL resistant to venetoclax. Blood. 2020;135:2266–2270. doi: 10.1182/blood.2020004782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Mato A.R., Roeker L.E., Jacobs R., Hill B.T., Lamanna N., Brander D., Shadman M., Ujjani C.S., Yazdy M.S., Perini G.F., et al. Assessment of the Efficacy of Therapies Following Venetoclax Discontinuation in CLL Reveals BTK Inhibition as an Effective Strategy. Clin. Cancer Res. 2020;26:3589–3596. doi: 10.1158/1078-0432.CCR-19-3815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Lew T.E., Anderson M.A., Lin V.S., Handunnetti S.M., Came N.A., Blombery P., Westerman D.A., Wall M., Tam C.S., Roberts A.W., et al. Undetectable peripheral blood MRD should be the goal of venetoclax in CLL, but attainment plateaus after 24 months. Blood Adv. 2020;4:165–173. doi: 10.1182/bloodadvances.2019000864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Prado G., Kaestner C.L., Licht J.D., Bennett R.L. Targeting epigenetic mechanisms to overcome venetoclax resistance. Biochim. Biophys. Acta Mol. Cell Res. 2021;1868:119047. doi: 10.1016/j.bbamcr.2021.119047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Carrà G., Nicoli P., Lingua M.F., Maffeo B., Cartellà A., Circosta P., Brancaccio M., Parvis G., Gaidano V., Guerrasio A., et al. Inhibition of bromodomain and extra-terminal proteins increases sensitivity to venetoclax in chronic lymphocytic leukaemia. J. Cell. Mol. Med. 2020;24:1650–1657. doi: 10.1111/jcmm.14857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Yi X., Sarkar A., Kismali G., Aslan B., Ayres M., Iles L.R., Keating M.J., Wierda W.G., Long J.P., Bertilaccio M.T.S., et al. AMG-176, an Mcl-1 Antagonist, Shows Preclinical Efficacy in Chronic Lymphocytic Leukemia. Clin. Cancer Res. 2020;26:3856–3867. doi: 10.1158/1078-0432.CCR-19-1397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Dillman R.O. Cancer immunotherapy. Cancer Biother. Radiopharm. 2011;26:1–64. doi: 10.1089/cbr.2010.0902. [DOI] [PubMed] [Google Scholar]
- 141.Perutelli F., Jones R., Griggio V., Vitale C., Coscia M. Immunotherapeutic Strategies in Chronic Lymphocytic Leukemia: Advances and Challenges. Front. Oncol. 2022;12:837531. doi: 10.3389/fonc.2022.837531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Griggio V., Perutelli F., Salvetti C., Boccellato E., Boccadoro M., Vitale C., Coscia M. Immune Dysfunctions and Immune-Based Therapeutic Interventions in Chronic Lymphocytic Leukemia. Front. Immunol. 2020;11:594556. doi: 10.3389/fimmu.2020.594556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Fayad L., Keating M.J., Reuben J.M., O’Brien S., Lee B.-N., Lerner S., Kurzrock R. Interleukin-6 and interleukin-10 levels in chronic lymphocytic leukemia: Correlation with phenotypic characteristics and outcome. Blood. 2001;97:256–263. doi: 10.1182/blood.V97.1.256. [DOI] [PubMed] [Google Scholar]
- 144.Alhakeem S.S., McKenna M.K., Oben K.Z., Noothi S.K., Rivas J.R., Hildebrandt G.C., Fleischman R.A., Rangnekar V.M., Muthusamy N., Bondada S. Chronic Lymphocytic Leukemia-Derived IL-10 Suppresses Antitumor Immunity. J. Immunol. 2018;200:4180–4189. doi: 10.4049/jimmunol.1800241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Hanna B.S., Llaó-Cid L., Iskar M., Roessner P.M., Klett L.C., Wong J.K.L., Paul Y., Ioannou N., Öztürk S., Mack N., et al. Interleukin-10 receptor signaling promotes the maintenance of a PD-1(int) TCF-1(+) CD8(+) T cell population that sustains anti-tumor immunity. Immunity. 2021;54:2825–2841.e2810. doi: 10.1016/j.immuni.2021.11.004. [DOI] [PubMed] [Google Scholar]
- 146.Domagala M., Ysebaert L., Ligat L., Lopez F., Fournié J.J., Laurent C., Poupot M. IL-10 Rescues CLL Survival through Repolarization of Inflammatory Nurse-like Cells. Cancers. 2021;14:16. doi: 10.3390/cancers14010016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Ai L., Xu A., Xu J. Roles of PD-1/PD-L1 Pathway: Signaling, Cancer, and Beyond. Adv. Exp. Med. Biol. 2020;1248:33–59. doi: 10.1007/978-981-15-3266-5_3. [DOI] [PubMed] [Google Scholar]
- 148.Flies D.B., Sandler B.J., Sznol M., Chen L. Blockade of the B7-H1/PD-1 pathway for cancer immunotherapy. Yale J. Biol. Med. 2011;84:409–421. [PMC free article] [PubMed] [Google Scholar]
- 149.Han Y., Liu D., Li L. PD-1/PD-L1 pathway: Current researches in cancer. Am. J. Cancer Res. 2020;10:727–742. [PMC free article] [PubMed] [Google Scholar]
- 150.Brusa D., Serra S., Coscia M., Rossi D., D’Arena G., Laurenti L., Jaksic O., Fedele G., Inghirami G., Gaidano G., et al. The PD-1/PD-L1 axis contributes to T-cell dysfunction in chronic lymphocytic leukemia. Haematologica. 2013;98:953–963. doi: 10.3324/haematol.2012.077537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Ding W., LaPlant B.R., Call T.G., Parikh S.A., Leis J.F., He R., Shanafelt T.D., Sinha S., Le-Rademacher J., Feldman A.L., et al. Pembrolizumab in patients with CLL and Richter transformation or with relapsed CLL. Blood. 2017;129:3419–3427. doi: 10.1182/blood-2017-02-765685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Srivastava S., Riddell S.R. Engineering CAR-T cells: Design concepts. Trends Immunol. 2015;36:494–502. doi: 10.1016/j.it.2015.06.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Miliotou A.N., Papadopoulou L.C. CAR T-cell Therapy: A New Era in Cancer Immunotherapy. Curr. Pharm. Biotechnol. 2018;19:5–18. doi: 10.2174/1389201019666180418095526. [DOI] [PubMed] [Google Scholar]
- 154.Chmielewski M., Abken H. TRUCKs: The fourth generation of CARs. Expert Opin. Biol. Ther. 2015;15:1145–1154. doi: 10.1517/14712598.2015.1046430. [DOI] [PubMed] [Google Scholar]
- 155.Todorovic Z., Todorovic D., Markovic V., Ladjevac N., Zdravkovic N., Djurdjevic P., Arsenijevic N., Milovanovic M., Arsenijevic A., Milovanovic J. CAR T Cell Therapy for Chronic Lymphocytic Leukemia: Successes and Shortcomings. Curr. Oncol. 2022;29:3647–3657. doi: 10.3390/curroncol29050293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Park J.H., Rivière I., Gonen M., Wang X., Sénéchal B., Curran K.J., Sauter C., Wang Y., Santomasso B., Mead E., et al. Long-Term Follow-up of CD19 CAR Therapy in Acute Lymphoblastic Leukemia. N. Engl. J. Med. 2018;378:449–459. doi: 10.1056/NEJMoa1709919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Locke F.L., Ghobadi A., Jacobson C.A., Miklos D.B., Lekakis L.J., Oluwole O.O., Lin Y., Braunschweig I., Hill B.T., Timmerman J.M., et al. Long-term safety and activity of axicabtagene ciloleucel in refractory large B-cell lymphoma (ZUMA-1): A single-arm, multicentre, phase 1-2 trial. Lancet Oncol. 2019;20:31–42. doi: 10.1016/S1470-2045(18)30864-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Schuster S.J., Bishop M.R., Tam C.S., Waller E.K., Borchmann P., McGuirk J.P., Jäger U., Jaglowski S., Andreadis C., Westin J.R., et al. Tisagenlecleucel in Adult Relapsed or Refractory Diffuse Large B-Cell Lymphoma. N. Engl. J. Med. 2019;380:45–56. doi: 10.1056/NEJMoa1804980. [DOI] [PubMed] [Google Scholar]
- 159.Riches J.C., Ramsay A.G., Gribben J.G. Immune dysfunction in chronic lymphocytic leukemia: The role for immunotherapy. Curr. Pharm. Des. 2012;18:3389–3398. doi: 10.2174/138161212801227023. [DOI] [PubMed] [Google Scholar]
- 160.Cox M.J., Lucien F., Sakemura R., Boysen J.C., Kim Y., Horvei P., Manriquez Roman C., Hansen M.J., Tapper E.E., Siegler E.L., et al. Leukemic extracellular vesicles induce chimeric antigen receptor T cell dysfunction in chronic lymphocytic leukemia. Mol. Ther. 2021;29:1529–1540. doi: 10.1016/j.ymthe.2020.12.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Shalabi H., Kraft I.L., Wang H.W., Yuan C.M., Yates B., Delbrook C., Zimbelman J.D., Giller R., Stetler-Stevenson M., Jaffe E.S., et al. Sequential loss of tumor surface antigens following chimeric antigen receptor T-cell therapies in diffuse large B-cell lymphoma. Haematologica. 2018;103:e215–e218. doi: 10.3324/haematol.2017.183459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Vlachonikola E., Stamatopoulos K., Chatzidimitriou A. T Cells in Chronic Lymphocytic Leukemia: A Two-Edged Sword. Front. Immunol. 2020;11:612244. doi: 10.3389/fimmu.2020.612244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Shapiro M., Herishanu Y., Katz B.Z., Dezorella N., Sun C., Kay S., Polliack A., Avivi I., Wiestner A., Perry C. Lymphocyte activation gene 3: A novel therapeutic target in chronic lymphocytic leukemia. Haematologica. 2017;102:874–882. doi: 10.3324/haematol.2016.148965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Wang Z.H., Li W., Dong H., Han F. Current state of NK cell-mediated immunotherapy in chronic lymphocytic leukemia. Front. Oncol. 2022;12:1077436. doi: 10.3389/fonc.2022.1077436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Herrera L., Santos S., Vesga M.A., Anguita J., Martin-Ruiz I., Carrascosa T., Juan M., Eguizabal C. Adult peripheral blood and umbilical cord blood NK cells are good sources for effective CAR therapy against CD19 positive leukemic cells. Sci. Rep. 2019;9:18729. doi: 10.1038/s41598-019-55239-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Fürstenau M., Eichhorst B. Novel Agents in Chronic Lymphocytic Leukemia: New Combination Therapies and Strategies to Overcome Resistance. Cancers. 2021;13:1336. doi: 10.3390/cancers13061336. [DOI] [PMC free article] [PubMed] [Google Scholar]