Abstract
Many chemicals and toxicants are released into our ecosystem and environment every day, which can cause harmful effects on human populations. Agricultural compounds are used in most crop production and have been shown to cause negative health impacts, including effects on reproduction and other pathologies. Although these chemicals can be helpful for pest and weed control, the compounds indirectly impact humans. Several compounds have been banned in the European Union but continue to be used in the United States. Recent work has shown most toxicants affect transgenerational generations more than the directly exposed generations through epigenetic inheritance. While some toxicants do not impact the directly exposed generation, the later generations that are transgenerational or ancestrally exposed suffer health impacts. Due to impacts to future generations, exposure becomes an environmental justice concern. The term “environmental justice” denotes the application of fair strategies when resolving unjust environmental contamination. Fair treatment means that no group should bear a disproportionate share of negative environmental consequences resulting from industrial, municipal, and commercial operations. This article illustrates how research on directly exposed generations is often prioritized over studies on transgenerational generations. However, research on the latter generations suggests the need to take environmental justice concerns seriously moving forward, as future generations could be unduly shouldering harms, while not enjoying benefits of production.
Keywords: Environmental Justice, Biomedical Ethics, Epigenetics, Transgenerational, Inheritance, Toxicants, Vinclozolin, DDT, Glyphosate, Generational Toxicology
Introduction
Environmentally induced epigenetic transgenerational inheritance of disease may cause changes in phenotypes in both males and females, leading to much interest in fields ranging from plants to humans [1]. Differences in phenotype and inheritance of diseases can come from many environmental factors from nutrition to toxicants [2]. Dichlorodiphenyldichloroethane (DDT) was the first agricultural compound developed and heavily used in most countries in the 1950s and 1960s and is still used currently in countries in East Asia (eg, India) and Africa. DDT has affected multiple generations for the past 80 years [2]. Research has shown that DDT causes infertility issues, cancers, and more. Unfortunately, DDT is still in the soil and due to its long half-life will be in the environment for the next 500 years [2]. Vinclozolin, a fungicide used in fruit and vegetable crops [1], was one of the first toxicants shown to environmentally induce epigenetic transgenerational inheritance of disease [3]. A large number of environmental factors and toxicants have now been shown to promote epigenetic inheritance, such as hydrocarbons (jet fuel) and herbicides (glyphosate), as well as environmental stress (behavioral), and nutrition abnormalities [4], Figure 1.
Figure 1.
Environmentally induced epigenetic transgenerational inheritance. Various exposures and species investigated. Modified from [5].
The impacts of toxicants for multiple generations is termed “generational toxicology” and mediated through the germline (sperm or eggs) and involves epigenetic mechanisms [5]. The ability of an environmental exposure to impact multiple generations has been demonstrated in all organisms studied from plants to humans, Figure 1. For plants, flies, and worms, the epigenetic inheritance has been shown to impact hundreds of generations [5], while for mammals the experiments have only been done on four to six generations to date. Nearly all environmental factors and toxicants studied have been shown to promote epigenetic transgenerational inheritance, Figure 11. Although studies have demonstrated effects of nutrition and stress on epigenetic transgenerational inheritance [5], the current study will focus on generational toxicology and environmental chemicals.
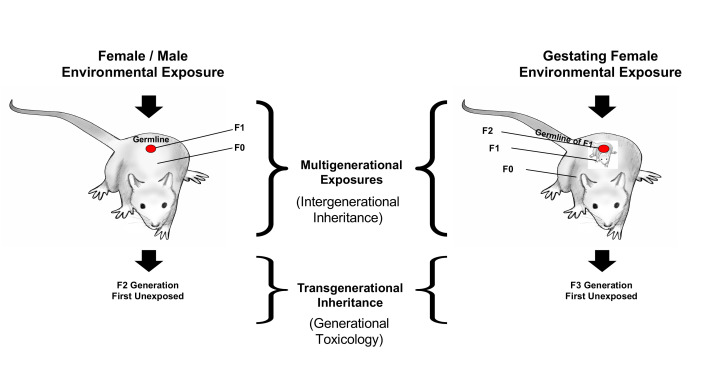
The exposure of a toxicant for an adult female or male impacts the F0 generation male or female and their germline (sperm or egg) that will generate the F1 generation offspring, so the transgenerational generation is the F2 generation grand-offspring, which does not have any direct exposure, Figure 2. Exposure of these toxicants in a gestating female effects the F0 generation female, the F1 generation fetus, and the germline that is within the F1 generation fetus that will generate the F2 generation [3]. Therefore, the first generation that is transgenerational will be the F3 generation [3], Figure 2. When a gestating female rat is exposed to vinclozolin during fetal gonadal sex determination, the reprogramming of the male or female germline epigenome develops [1]. Epigenetic transgenerational inheritance is only accomplished through the germline transmission of information that contains the altered epigenetics to change the phenotype or disease, in the absence of continued direct exposure to the toxicant [1]. The transgenerational generations are the focus as we consider the stability of induced epigenetic transgenerational inheritance and environmental justice. The current article discusses the transgenerational generations, specifically looking into the stability of transmitted epigenetic inheritance. This is a concern for environmental justice because the transgenerational generations are not directly exposed but could suffer negative health impacts. Ancestrally exposed transgenerational individuals need to be considered in environmental justice and health related conversations.
Figure 2.
Environmentally induced transgenerational epigenetic inheritance: schematic of environmental exposure and affected generations for both gestating female and adult male or female. The multigenerational direct exposures are indicated in contrast to the transgenerational generation having no direct exposure. Modified from [5].
Background: Epigenetics
Epigenetics is defined as “molecular factors and processes around DNA that regulate genome activity, independent of DNA sequence, and are mitotically stable” [5]. Although DNA sequence is inherited through genetic inheritance, heritable changes in the epigenetics can also occur through many epigenetic mechanisms [6]. The first epigenetic change identified was DNA methylation [6]. DNA methylation refers to adding methyl groups to DNA to alter DNA transcription in the methylated region (Figure 3) [7]. The importance of DNA methylation is that during gene expression the methylation can regulate the process of binding of proteins that regulate transcription [7]. When DNA methylation occurs, a methyl group is put onto a C5 position of cytosine, which is adjacent to guanine to form a 5-methylcytosine (5mC) [5,7]. Research has found that 5mC will suppress transcription while formation of 5-hydroxymethylcytocine (5hmC) by the enzyme family named ten-eleven translocation (tet) will facilitate DNA methylation erasure [5]. The ten-eleven translocation enzymes have been found to help remove the methylated region during the time of early embryo development [5]. DNA methylation is an essential component of transcription and replication, Figure 3 [5]. However, DNA methylation does not always indicate transcriptional suppression [8]. Cytosine guanine dinucleotide (CpG) islands or areas where CpG regions are dense in the genome (CGIs) can be repressed [8]. This can occur through various mechanisms [8]. One such epigenetic mechanism occurs through chromatin remodeling proteins such as polycomb structural proteins [8]. Polycomb proteins are “epigenetic regulators for cell proliferation and differentiation during development” [9]. Polycomb proteins can suppress “gene encoding master regulators” during embryonic development [8].
Figure 3.
Schematic representation of the primary epigenetic factors and processes of non-coding RNA, DNA methylation, chromatin structure, histone modifications and DNA structure presented. Modified from [5].
There are a number of other epigenetic processes that exist [9]. RNA methylation does occur on adenine sites [10]. Methylation of adenine sites (m6A) has shown to “affect various biological processes, including stem-cell pluripotency, memory formation, immune responses and tumorigenesis” [10]. Found in Drosophila, m6A can influence splicing and process biogenesis in the RNA [11]. Researchers found that in a Drosophila system there is a critical protein called Sxl [11]. Sxl is used during the determination of sex in the Drosophila fly [11]. If the Sxl involves a methyl adenine and is abnormal this may change the function and developmental biology of the organisms [11]. The study concluded that epigenetic mechanism of m6A became a critical role in the process of sex determination in Drosophila [11].
Another epigenetic process involves the modification of histones [12]. Histones are essential to the structure of DNA as the DNA is wrapped around the histone core [12,13], Figure 3. Histone modifications directly regulate gene expression [13]. In addition to regulation of gene expression, histones can alter the positioning between the nucleosomes [14]. Histone modification can also become detrimental and associate with pathologies [13,14]. The DNA and associated proteins is known as chromatin [15]. The chromatin structure is also known for being critical in gene expression and is an important epigenetic factor [15], Figure 3. An example involves changing the structure of the chromatin to be suppressive heterochromatin [13,15]. Another critical epigenetic factor involves non-coding RNA (ncRNA) [16], Figure 3. These ncRNA molecules do not need a specific DNA binding sequence for them to regulate gene expression, but gene expression is regulated from ncRNA facilitating proteins binding to DNA that alter the expression of genes [17], Figure 3. In a recent study done in rats, vinclozolin induced epigenetic transgenerational inheritance involving sperm with changes in the amounts of various ncRNAs [18]. All these epigenetic factors can alter gene expression and correlate with phenotypic change in all organisms investigated [19].
Epigenetic Inheritance
Epigenetic transgenerational inheritance refers to germline transmission of altered epigenetics between generations [5]. These epigenetic alternations are passed down through the parents’ gametes that can cause the epigenetic inheritance of pathology and disease [5]. Epigenetic transgenerational inheritance starts with the generation without continued direct exposure (eg, F3 generation) and subsequent generations [5]. The first generation exposed to these harmful toxicants will often have lower risk of disease compared to the transgenerational generations [5]. The transgenerational generations are put into a position where they will have epigenetic alterations passed down to them without their consent [5]. Ancestral exposure to toxicants that cause lifelong illnesses without the consent of the affected generations (transgenerational generations) is an environmental justice issue and will be addressed throughout this review [5].
Generational Toxicology
Due to the ever-increasing demand for goods and consumables, increasing amounts of toxicants are released into our environment and later consumed by humans and animals [2]. While toxicants can be helpful in the management of certain diseases, such as malaria, the long-lasting effects of exposure to these toxicants on later generations have not been considered. Generational toxicology reports have indicated that toxicant exposures will be seen in the subsequent generations and will continue to cause harm in the health of subsequent generations [5]. When releasing toxicants into the environment, there must be regulation set to avoid the mass destruction of fertility and an increase in other diseases in the transgenerational generations [5]. There also have been studies that have observed a profound increase in obesity, ovarian disease, and parturition abnormalities more in the transgenerational generations rather than the directly exposed generations [20]. This phenomenon is termed generational toxicology to impact future generations with no direct exposures [5]. Addressed in this review are the toxicants studied most recently and been found to have the greatest health effects on the transgenerational generations.
Dichlorodiphenyldichloroethane (DDT)
DDT was used as a pesticide and to prevent malaria in countries all over North America, including the United States [2]. While direct exposure to DDT did not affect the overall health, the consequences of transgenerational inheritance were observed in their offspring and F2 generation grand-offspring, which exhibited disease in the kidneys, ovaries, and testes [2], as well as the onset of obesity [2] (Figure 4). Most countries have banned the use of DDT due to its harmful effects on humans and animals that have been impacted transgenerationally [20]. Some countries still allow the use of DDT and large corporations spray this harmful insecticide in indoor settings [21,22]. Recent studies have shown high direct exposure to DDT in humans when spread indoors [2]. In addition to human issues, studies have shown that wildlife species suffer defects, both from direct exposure and transgenerationally [23]. While DDT was an inexpensive way to rid the world of terrible disease that still causes death in Africa today, the transgenerational effects of this toxicant should not be dismissed [2]. There are alternatives to using DDT that are less harmful that should be considered [2]. DDT has an extremely long half-life once the toxicant is released into the environment, and the toxicant will be in the environment for the next 500 years [2]. Other toxicants available (aldrin, eldrin, and dieldrin) have shorter half-lives and may not wreak havoc on subsequent generations [2].
Figure 4.
Epigenetic inheritance profile in humans. Impacts when a gestating female is exposed to environmental factors. Exposure occurs in the F0, F1, and F2 generations (see Figure 2), and F3 generation inherit transgenerational disease. Modified from [2].
A recent study researched ancestral exposure to DDT and how environmental toxicants can promote DNA methylation alterations in adipocytes [24]. The F0 generation gestating female was exposed to DDT and carried out the generations into the F3 generation [24]. The F3 generation was not exposed to DDT [24]. The results of this research found that in several metabolic pathways many genes are associated with “adipogenesis, insulin resistance, and lipolysis” [24]. Changes in these functions dealing with adipocytes can cause the promotion of obesity [24]. Other studies have also concluded that DDT, in the case of direct exposure, is associated with an increase in obesity [25]. Transgenerational impacts of DDT have been shown to cause an obese phenotype in the offspring of the F3 generation [24]. When an herbicide was used, such as atrazine, the phenotype gave a lean phenotype [24]. When species are exposed to toxicants such as DDT, epigenetic alterations occur and cause disease pathology that the directly exposed generations may not experience [24]. Environmental justice must be considered if toxicants are known to have effects on transgenerational generations [24].
Vinclozolin
Another pesticide (ie, fungicide) that is heavily used in the US and can have transgenerational effects is vinclozolin. Vinclozolin is a testosterone antagonist, blocking androgen receptors and thus suppressing androgen actions in the male [26]. When pregnant female rats were exposed orally to vinclozolin during gestation, their offspring were affected by the exposure. Impacts on male fertility and the promotion of transgenerational diseases and epimutations were observed [26]. Vinclozolin also has a transgenerational impact on male fertility [26]. DNA methylation patterns were altered in the direct exposure generations (F0-F2), as well as the first transgenerational (F3) generation [26]. One of the most common ways to study transgenerational inheritance is through DNA methylation [26]. This research suggested that the environment can induce epigenetic changes in the germline and that these can be inherited to contribute to transgenerational disease [26]. In another vinclozolin study in rats, the researchers used terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling (TUNEL) assay protocol on the transgenerational generation animals to detect cellular death (apoptosis) in the testes [18]. The F0 generation pregnant female was exposed to vinclozolin during embryonic days E8-E14 of early gestation [18]. A control lineage gestating female rat was exposed to dimethyl sulfoxide (DMSO) during the days of E8-E14 [18]. In comparison to the wild-type animals having no exposure to the toxicant, the exposed lineage had more significant counts of apoptotic cells in the testes in the transgenerational generation (F3) [18]. These results support the transgenerational phenotype of the use of the toxicant vinclozolin [18]. Exposure to vinclozolin has also been shown to transmit this phenotype from the first generation to the subsequent generations, causing widespread transgenerational pathology [26].
Glyphosate (Roundup)
Among the many agricultural chemical toxicants polluting our environment and being a driving force of transgenerational disease, (N-(phosphonomethyl) glycine), also known as glyphosate (Roundup), is known to cause the epigenetic transgenerational inheritance of disease [20]. Used as an herbicide, glyphosate is the most heavily used agricultural chemical to eliminate and kill unwanted plants. In a recent study, rats through ancestral exposure to glyphosate of an F0 generation gestating female were carried out to the F3 transgenerational generation [20]. The results showed that the directly exposed F0 and F1 generations did not have significant toxicity nor pathology [20]. However, when looking into the pathology of the F2 generation, there began to be significant changes in the rat’s ovary diseases, obesity, and parturition abnormalities [20]. The transgenerational generation (F3) of rats had significant abnormalities dealing with prostate issues, obesity, increased ovarian disease, and increased parturition abnormalities [20]. An important observation was the increased parturition abnormalities in the F2 generation, over 30% of the F2 generation female rats in the later stages of gestation died of dystocia and/or had litter mortality [20]. Glyphosate had little to no effect on the directly exposed F0 and F1 generations, but the impacts on the subsequent generations resulted in significant pathology due to epigenetic inheritance [20]. Using the rat model, investigations found that transgenerational generations now live with the result of the reluctant use of toxicants, such as glyphosate, as they will suffer from diseases that the directly exposed generations never thought of worrying about [20].
Other Generational Environmental Toxicants
According to the Federal Aviation Administration, there are about “45,000 flights” per day [27]. Jet fuel can leech into our ecosystems and fumes get into the daily air we breathe [28]. While we do not see the direct effects, the subsequent generations can be significantly affected [28]. Jet fuel contains harmful hydrocarbons and toxicants that cause immune system, respiratory and nervous system issues, such as problems with narcosis, issues with attention and memory, and coordination [4]. Dioxin (2,3,7,8-tetrachlorodibenzo[p]dioxin, TCDD) is an industrial contaminant that comes from manufactures production [29]. A study dealing with exposing dioxin to rats has shown that there was an increased problem with primordial follicle loss, polycystic ovarian disease, and higher kidney disease found in the females [29]. Tributyltin is a toxicant that is used as a biocide and is an obesogen known to cause disease in transgenerational generations [30]. Some diseases linked to the use of the toxicant tributyltin have been found to promote transgenerational hepatic steatosis [30]. When investigated with mice samples there was higher risk and increase of obesity in the transgenerational generations [30]. Plastic compound toxicants such as bisphenol A (BPA) and phthalates have been shown to promote prominent heart disorders in zebrafish [31] and male and female transgenerational pathology in mammals [32]. Ethanol influences the offspring of gestating female rats that have been exposed to ethanol vapor [33]. The results showed that the offspring had neurological alterations in the F3 generation that was similar of the syndrome found in human fetal alcohol syndrome [33]. These are just a few of the generational toxicology observations previously made in the literature [5].
Environmental Justice
Studies have been conducted on impacts of direct exposure to toxicants. However, relatively little research has been done on transgenerational impacts of environmental exposures [26]. Connections between ancestrally exposed generations and environmental justice need to be included in both research and social discussions. The term “environmental justice” came into popular parlance during the 1980s and is associated with events within the US Black Belt region [34]. Specifically, protests were carried out by communities to challenge unfair placement practices of environmental externalities, including chemical waste dumps, oil refineries, landfills, and other noxious facilities. According to Murdock, “communities of color and poor communities identified their neighborhoods as being overburdened with particular environmental ills, especially those linked with toxicity and pollution related to the fossil fuel and petrochemical industries” [34]. Grounded in the realities of these events, the term “environmental justice” denotes the application of fair strategies and processes in the resolution of inequality related to environmental contamination. Fair treatment means that no group of people should bear a disproportionate share of negative environmental consequences resulting from industrial, municipal, and commercial operations or the execution of federal, state, local, and tribal programs, and policies.
Much of the time, we have little say as to what toxicants corporations are introducing into the food we consume or into the environments which we live. However, these toxicants may cause direct negative health impacts and/or transgenerational disease [20,35]. Environmental justice brings fairness, equality, and equity to light by arguing that everyone, irrespective of location, should not be saddled with undue harms [35]. Socioeconomic standing and location could ultimately correlate with the level of toxicants you are exposed to [2,36]. For example, unfair exposure to environmental harms prompted the 1982 Warrant County, North Carolina, demonstrations against the decision to place toxic waste landfill in a primarily Black/African American community [34]. This was a pivotal event in the development of the environmental justice movement, as citizens protested the placement of a PCB (polychlorinated biphenyls) landfill, which posed an explicit environmental harm to the community. Beyond US borders, as environmental justice movements are global, diverse communities are increasingly concerned about similar exposures. For example, while the US has banned the use of DDT, India and parts of Africa are still heavily using DDT to eradicate malaria [21]. This use poses potential harms to a myriad of communities, especially those in exposure sites, which are often inhabited by lower socioeconomic groups and historically marginalized communities. Several studies, including the groundbreaking “Toxic Waste and Race in the United States at Twenty” study, found that historically marginalized groups had a higher probability of being exposed to toxicants and hazardous waste than their more affluent counterparts [37]. Another study researched the “activity of manufacturing plants found in Los Angeles, Boston, Columbus, and Houston” [38]. Results showed that the communities with poor populations received more resources than other groups, and there was an increased exposure depending on the economic status [38]. As first generations face higher levels of direct exposure to toxicants, future generations could suffer greatly and have long-lasting pathology effects [26].
Studies have also shown that environmental factors such as pesticides, stress, and behavioral issues can be inherited transgenerationally [39]. Future generations cannot avoid being ancestrally exposed to harmful toxicants that give them increased pathology in the testis, ovary, and kidney [2]. Another example rife with environmental justice concerns is when glyphosate is used as an herbicide. As discussed previously, glyphosate has been shown to cause the epigenetic transgenerational inheritance of disease [20,40]. The F0 and F1 generations had no effects on their pathology, while the F2 and the F3 generations had suffered dramatically from the ancestral exposure to this toxicant [20,40]. The rats in the F2, and the F3 generations had increased prostate disease, obesity, and ovarian abnormalities [40]. Glyphosate was used to get rid of unwanted plants, which worked well when people did not know the crucial side effects concerning humans disease susceptibility [20,40]. The knowledge we have obtained concerning glyphosate underscores the need to investigate not only directly exposed populations, but also the transgenerational generations [40]. These generations are left to suffer pathologies and diseases from ancestral environmental pollution, where initial exposure was outside their control. In the case of glyphosate and vinclozolin, the directly exposed may now be free of weeds or molds in their crops, however, the subsequent generations are now left to deal with ovarian abnormalities and low sperm count [2,20,26,40]. These subsequent generations did not give consent and are not given a choice whether they accept the impacts associated with such harmful toxicants. Concerns over fairness to future generations highlights how environmental justice should play a greater role in social discussions concerning what constitutes appropriate use of agricultural inputs. These larger justice considerations need to be included to ensure that communities are not unduly shouldering harms for the betterment of society.
Environmental Justice and Social Determinants of Health
Impacts to future generations also highlight how health impacts and environmental exposures are deeply intertwined. Communities protesting environmental injustices often argue that they are bearing a disproportionate burden of environmental health risk or facing additional burdens from social determinants of health (SDOH) [41]. SDOHs are defined as environmental conditions where people live, learn, work, and play that impact health outcomes and general quality of life. Environmental justice claims highlight the multifaceted connection between community health and environmental harms. As such, medical ethics also provides insights concerning transgenerational impacts of environmental exposures. In the late 1970s, the Belmont Report identified key ethical guidelines for biomedical research and healthcare practice. “Respect for persons [often separated into autonomy and nonmaleficence] beneficence, and justice” were cited as key criteria that need to be met to ensure that biomedical research is ethical [42]. Autonomy concerns respecting the individual choices of those involved, while nonmaleficence focuses on doing no harm to patient or research subject [42]. Justice implies that every person should be treated fairly, and benefits harms should be distributed equally across social groups [35,42]. In medical contexts, these principles guide practice to ensure that patients and research subjects are treated ethically. It should be noted that the main area of concern discussed in this paper falls outside of medical contexts. However, as environmental toxicants (which are SDOHs) impact community health [37,41], a medical ethics framework also applies to the case of transgenerational impacts. When workers spray a chemical such as vinclozolin, DDT or glyphosate on our crops, this prevents pests and weeds from thriving, improving the production of the crop [26]. However, the effects on subsequent exposed human generations are long lasting. Transgenerational generations did not consent to be exposed to a toxicant, which could impact their epigenetics and cause associated disease risk. Thus, their autonomy is violated. Nonmaleficence is also being violated as well, as transgenerational generations could be harmed by this exposure [42]. Lastly, justice considerations for future generations shouldn’t be ignored, as one could argue that future generations bear increased risk associated with exposure [40]. Multiple cases illustrate how generations exposed to toxicants may not show direct health effects, but third generations (great grand-offspring) may be greatly impacted [26]. These future generations suffer from diseases such as cancer, higher levels of dioxins in breast milk, etc. [43,44]. From this perspective, environmental exposure to vinclozolin, glyphosate, and DDT is at once an environmental justice issue and a medical ethics concern, as a social determinant of health.
Due to potential health impacts to future generations, companies should be required to research the epigenetics of at least the third generation to determine if there are impacts on human health [44]. Studies dealing with transgenerational epigenetics show that later generations accumulate new diseases, even when the first direct exposure might not [26]. To say that individuals directly exposed are the only population to potentially suffer impacts is not accurate and thus needs to be corrected. With this being said, there is also a need for more research on transgenerational impacts of environmental exposures. Research on transgenerational exposure is less common than research on direct exposures. Larger companies, who produce such toxicants, have primarily researched first-generation health impacts. In addition, governments agencies, such as the US Environmental Protection Agency (EPA), should take future generational impacts more seriously when drafting policies and regulations.
There are concerns as to the levels of exposure to environmental toxicants being higher in investigations than the lowest observed adverse effect level (LOAELs). This has been an advanced topic in toxicology. This issue removes previous concerns on high and low doses found in research today. In addition, we are not aware of any study that has not found transgenerational effects of environmental toxicants. The concerns given are irrelevant to transgenerational exposure because effects still occur. To say that direct exposure is the only viable generation to develop a disease is neither accurate nor acceptable and must be corrected to the fullest extent. Environmental toxicants that are used should be researched until at least the F3 generation due to research done on environmental toxicants having transgenerational effects [1-3,5,17,18,24,26,28,29,31,32,37]. In addition to more research being done by larger companies, there must be a change in the way policies are made in our governments agencies, such as the EPA.
Conclusion
Regardless of the initial purpose of the toxicant developed, people in policy-making positions should implement research to ensure there is no unethical impact in the later generations. Suppose there are alternatives in cases, such as the use of DDT to combat malaria. In that case, they should be explored and heavily researched to ensure what is being used is the safest, yet most effective way to address the problem. Large companies and government agencies that know the harmful effects of toxicants and continue to use them must be held responsible for the diseases and abnormalities that the transgenerational generations will experience. However, we do understand that there are concerns about the limitations of such regulations. While we do agree that it is difficult and costly to accomplish transgenerational studies, it is time industries and regulatory agencies step up and protect our future generations from the phenomenon. Independent of cost, the fact that there is an exposure impact to the transgenerational generations (F3 and onward) is more important than the direct exposure being primarily investigated today. Model systems on humans would be optimal. As generally found, the animal models provide the best assessment [45]. Using the same methods that currently and have always been used will not allow the environment to recover, and future human health is at risk as more toxicants are being released into our ecosystems. Due to the lack of action in transgenerational studies for subsequent generations there is not enough knowledge of all the epigenetic mechanisms. However, we do know that in the studies done with transgenerational generations that all epigenetic mechanisms found become permanent and show generational stability. Previously, our evolutionary model had been thought to involve primarily genetic mutations that would generate heritable phenotypes that would cause phenotypic variation, which natural selection would choose and select [46]. Studies have now shown that induced environmental epigenetic inheritance can persist in species for many generations [46]. However, the theory of epigenetic inheritance has long been pushed aside and not considered for evolutionary change. Combining the study of epigenetics and the study of genetics will allow researchers to have more information and a deeper understanding of the evolutionary model and biology as a whole. Environmental toxicant-induced epigenetic transgenerational inheritance of disease now needs to be incorporated into our consideration for future generations’ health and the evolutionary trajectory of humans. Generational toxicology needs to be equally considered as is direct exposure toxicology. Stakeholder groups concerned about environmental justice and social determinants of health need to incorporate this new science for safeguarding our future generations. Environmental justice concerns should be taken more seriously moving forward, as future generations could be unduly shouldering harms, while not enjoying benefits of production.
Acknowledgments
We thank Dr. Eric Nilsson for critically reviewing the manuscript and Ms. Heather Johnson for assistance in preparation of the manuscript. The research was supported by the John Templeton Foundation.
Glossary
- DDT
Dichlorodiphenyldichloroethane
- ncRNA
Non-coding RNA
- 5mC
5-methylcytosine
- EPA
Environmental Protection Agency
- m6A
Methylation of adenine sites
- CpG
Cytosine guanine dinucleotide
- Dioxin
2,3,7,8-tetrachlorodibenzo[p]dioxin, TCDD
- glyphosate
N-(phosphonomethyl) glycine
- SDOH
social determinants of health
Author Contributions
AAK drafted and edited the manuscript; SEN edited the manuscript; MKS obtained funding and edited the manuscript.
References
- Ben Maamar M, King SE, Nilsson E, Beck D, Skinner MK. Epigenetic transgenerational inheritance of parent-of-origin allelic transmission of outcross pathology and sperm epimutations. Dev Biol. 2020. Feb;458(1):106–19. 10.1016/j.ydbio.2019.10.030 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kabasenche WP, Skinner MK. DDT, epigenetic harm, and transgenerational environmental justice. Environ Health. 2014. Aug;13(1):62. 10.1186/1476-069X-13-62 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anway MD, Cupp AS, Uzumcu M, Skinner MK. Epigenetic transgenerational actions of endocrine disruptors and male fertility. Science. 2005;308(5727):1466-9. [pii] https://doi.org/ 10.1126/science.1108190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McDougal JN, Rogers JV. Local and systemic toxicity of JP-8 from cutaneous exposures. Toxicol Lett. 2004. Apr;149(1-3):301–8. 10.1016/j.toxlet.2003.12.041 [DOI] [PubMed] [Google Scholar]
- Nilsson EE, Ben Maamar M, Skinner MK. Role of epigenetic transgenerational inheritance in generational toxicology. Environ Epigenet. 2022;8(1):1-9. https://doi.org/ 10.1093/eep/dvac001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Handy DE, Castro R, Loscalzo J. Epigenetic modifications: basic mechanisms and role in cardiovascular disease. Circulation. 2011. May;123(19):2145–56. 10.1161/CIRCULATIONAHA.110.956839 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moore LD, Le T, Fan G. DNA methylation and its basic function. Neuropsychopharmacology. 2013. Jan;38(1):23–38. 10.1038/npp.2012.112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones PA. Functions of DNA methylation: islands, start sites, gene bodies and beyond. Nat Rev Genet. 2012. May;13(7):484–92. 10.1038/nrg3230 [DOI] [PubMed] [Google Scholar]
- Liu Q, Wang G, Li Q, Jiang W, Kim JS, Wang R, et al. Polycomb group proteins EZH2 and EED directly regulate androgen receptor in advanced prostate cancer. Int J Cancer. 2019. Jul;145(2):415–26. 10.1002/ijc.32118 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boulias K, Greer EL. Biological roles of adenine methylation in RNA. Nat Rev Genet. 2023. Mar;24(3):143–60. 10.1038/s41576-022-00534-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haussmann IU, Bodi Z, Sanchez-Moran E, Mongan NP, Archer N, Fray RG, et al. m6A potentiates Sxl alternative pre-mRNA splicing for robust Drosophila sex determination. Nature. 2016. Dec;540(7632):301–4. 10.1038/nature20577 [DOI] [PubMed] [Google Scholar]
- Turner BM. Histone acetylation as an epigenetic determinant of long-term transcriptional competence. Cell Mol Life Sci. 1998. Jan;54(1):21–31. 10.1007/s000180050122 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rothbart SB, Strahl BD. Interpreting the language of histone and DNA modifications. Biochim Biophys Acta. 2014. Aug;1839(8):627–43. 10.1016/j.bbagrm.2014.03.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bártová E, Krejcí J, Harnicarová A, Galiová G, Kozubek S. Histone modifications and nuclear architecture: a review. J Histochem Cytochem. 2008. Aug;56(8):711–21. 10.1369/jhc.2008.951251 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yaniv M. Chromatin remodeling: from transcription to cancer. Cancer Genet. 2014. Sep;207(9):352–7. 10.1016/j.cancergen.2014.03.006 [DOI] [PubMed] [Google Scholar]
- Kornfeld JW, Brüning JC. Regulation of metabolism by long, non-coding RNAs. Front Genet. 2014. Mar;5:57. 10.3389/fgene.2014.00057 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Larriba E, del Mazo J. Role of Non-Coding RNAs in the Transgenerational Epigenetic Transmission of the Effects of Reprotoxicants. Int J Mol Sci. 2016. Mar;17(4):452. 10.3390/ijms17040452 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ben Maamar M, Sadler-Riggleman I, Beck D, McBirney M, Nilsson E, Klukovich R, et al. Alterations in sperm DNA methylation, non-coding RNA expression, and histone retention mediate vinclozolin-induced epigenetic transgenerational inheritance of disease. Environment Epigenet. 2018;4(2):1-19, dvy010. https://doi.org/ 10.1093/eep/dvy010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wei JW, Huang K, Yang C, Kang CS. Non-coding RNAs as regulators in epigenetics (Review) [Review] [Review]. Oncol Rep. 2017. Jan;37(1):3–9. 10.3892/or.2016.5236 [DOI] [PubMed] [Google Scholar]
- Kubsad D, Nilsson EE, King SE, Sadler-Riggleman I, Beck D, Skinner MK. Assessment of Glyphosate Induced Epigenetic Transgenerational Inheritance of Pathologies and Sperm Epimutations: generational Toxicology. Sci Rep. 2019. Apr;9(1):6372. 10.1038/s41598-019-42860-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davies K. Strategies for eliminating and reducing persistent bioaccumulative toxic substances: common approaches, emerging trends, and level of success. J Environ Health. 2006;69(5):9-15, 36, 8. [PubMed] [Google Scholar]
- Aneck-Hahn NH, Schulenburg GW, Bornman MS, Farias P, de Jager C. Impaired semen quality associated with environmental DDT exposure in young men living in a malaria area in the Limpopo Province, South Africa. J Androl. 2007;28(3):423–34. 10.2164/jandrol.106.001701 [DOI] [PubMed] [Google Scholar]
- Hamlin HJ, Guillette LJ Jr. Birth defects in wildlife: the role of environmental contaminants as inducers of reproductive and developmental dysfunction. Syst Biol Reprod Med. 2010. Apr;56(2):113–21. 10.3109/19396360903244598 [DOI] [PubMed] [Google Scholar]
- King SE, Nilsson E, Beck D, Skinner MK. Adipocyte epigenetic alterations and potential therapeutic targets in transgenerationally inherited lean and obese phenotypes following ancestral exposures. Adipocyte. 2019. Dec;8(1):362–78. 10.1080/21623945.2019.1693747 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee DH, Porta M, Jacobs DR Jr, Vandenberg LN. Chlorinated persistent organic pollutants, obesity, and type 2 diabetes. Endocr Rev. 2014. Aug;35(4):557–601. 10.1210/er.2013-1084 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beck D, Sadler-Riggleman I, Skinner MK. Generational comparisons (F1 versus F3) of vinclozolin induced epigenetic transgenerational inheritance of sperm differential DNA methylation regions (epimutations) using MeDIP-Seq. Environmental Epigenetics. 2017;3(3):1-12, dvx016. https://doi.org/ 10.1093/eep/dvx016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hudda N, Fruin S, Durant JL. Substantial Near-Field Air Quality Improvements at a General Aviation Airport Following a Runway Shortening. Environ Sci Technol. 2022. Jun;56(11):6988–95. 10.1021/acs.est.1c06765 [DOI] [PubMed] [Google Scholar]
- Ben Maamar M, Nilsson E, Thorson JL, Beck D, Skinner MK. Epigenome-wide association study for transgenerational disease sperm epimutation biomarkers following ancestral exposure to jet fuel hydrocarbons. Reprod Toxicol. 2020. Dec;98:61–74. 10.1016/j.reprotox.2020.08.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manikkam M, Tracey R, Guerrero-Bosagna C, Skinner MK. Dioxin (TCDD) induces epigenetic transgenerational inheritance of adult onset disease and sperm epimutations. PLoS ONE. 2012;7(9):1-15, e46249. https://doi.org/ 10.1371/journal.pone.0046249. PONE-D-12-14353 [pii]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chamorro-García R, Sahu M, Abbey RJ, Laude J, Pham N, Blumberg B. Transgenerational inheritance of increased fat depot size, stem cell reprogramming, and hepatic steatosis elicited by prenatal exposure to the obesogen tributyltin in mice. Environ Health Perspect. 2013. Mar;121(3):359–66. 10.1289/ehp.1205701 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lombó M, Fernández-Díez C, González-Rojo S, Navarro C, Robles V, Herráez MP. Transgenerational inheritance of heart disorders caused by paternal bisphenol A exposure. Environ Pollut. 2015. Nov;206:667–78. 10.1016/j.envpol.2015.08.016 [DOI] [PubMed] [Google Scholar]
- Thorson JLM, Beck D, Ben Maamar M, Nilsson EE, Skinner MK. Ancestral plastics exposure induces transgenerational disease-specific sperm epigenome-wide association biomarkers. Environ Epigenet. 2021;7(1):1-13. https://doi.org/ 10.1093/eep/dvaa023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Abbott CW, Rohac DJ, Bottom RT, Patadia S, Huffman KJ. Prenatal Ethanol Exposure and Neocortical Development: A Transgenerational Model of FASD. Cereb Cortex. 2018. Aug;28(8):2908–21. 10.1093/cercor/bhx168 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murdock E. A History of Environmental Justice: Foundations, Narratives, and Perspectives. In: Coolsaet B. Environmental Justice: Key Issues. New York: Taylor and Francis; 2020. pp. 7–16. 10.4324/9780429029585-2 [DOI] [Google Scholar]
- Powell DL, Stewart V. Children. The unwitting target of environmental injustices. Pediatr Clin North Am. 2001. Oct;48(5):1291–305. 10.1016/S0031-3955(05)70375-8 [DOI] [PubMed] [Google Scholar]
- Shrader-Frechette K. Environmental Justice: Creating Equality, Reclaiming Democracy. New York: Oxford UP; 2002. 288 p. [Google Scholar]
- Bullard R, Mohai P, Saha R, Wright B. Toxic wastes and race at twenty: why race still matters after all these years. Environ Law. 2008;38(2):371–411. [Google Scholar]
- Gray WB, Shadbegian RJ, Wolverton A. Environmental Justice: Do Poor and Minority Populations Face More Hazards? In: Jefferson PN. Oxford Handbook of the Economics of Poverty. NCEE Environmental Economics Working Paper Series Oxford. Oxford University Press; 2010. pp. 1–35. [Google Scholar]
- Skinner MK. Environmental stress and epigenetic transgenerational inheritance. BMC Med. 2014. Sep;12(1):153. 10.1186/s12916-014-0153-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ben Maamar M, Beck D, Nilsson EE, Kubsad D, Skinner MK. Epigenome-wide association study for glyphosate induced transgenerational sperm DNA methylation and histone retention epigenetic biomarkers for disease. Epigenetics. 2021. Oct;16(10):1150–67. 10.1080/15592294.2020.1853319 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prochaska JD, Nolen AB, Kelley H, Sexton K, Linder SH, Sullivan J. Social Determinants of Health in Environmental Justice Communities: Examining Cumulative Risk in Terms of Environmental Exposures and Social Determinants of Health. Hum Ecol Risk Assess. 2014;20(4):980–94. 10.1080/10807039.2013.805957 [DOI] [PMC free article] [PubMed] [Google Scholar]
- OHRP. Office for Human Research Protections (OfHRP). The Belmont Report: HHS.Gov. 2021.
- Mazza A, Piscitelli P, Neglia C, Della Rosa G, Iannuzzi L. Illegal Dumping of Toxic Waste and Its Effect on Human Health in Campania, Italy. Int J Environ Res Public Health. 2015. Jun;12(6):6818–31. 10.3390/ijerph120606818 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martuzzi M, Mitis F, Biggeri A, Terracini B, Bertollini R. [Environment and health status of the population in areas with high risk of environmental crisis in Italy]. Epidemiol Prev. 2002;26(6 Suppl):suppl 1-53. [PubMed] [Google Scholar]
- Mukherjee P, Roy S, Ghosh D, Nandi SK. Role of animal models in biomedical research: a review. Lab Anim Res. 2022. Jul;38(1):18. 10.1186/s42826-022-00128-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Skinner MK, Guerrero-Bosagna C, Haque MM, Nilsson EE, Koop JAH, Knutie SA, et al. Epigenetics and the evolution of Darwin’s Finches. Genome Biology & Evolution. 2014;6(8):1972-89. https://doi.org/ 10.1093/gbe/evu158. [DOI] [PMC free article] [PubMed] [Google Scholar]