Abstract
Fixed-ratio combination injection therapy (FRC) is a fixed-ratio mixture containing basal insulin and glucagon-like peptide-1 receptor agonist (GLP-1 RA) in a single injection for the treatment of patients with type 2 diabetes. The two types of FRC products contain different concentrations and mixing ratios of basal insulin and GLP-1 RA. Both products demonstrated satisfactory blood glucose control throughout the day, with less hypoglycemia and weight gain. However, few studies have examined the differences in the actions of the two formulations. Herein, we present a case of a 71-year-old man with pancreatic diabetes and significantly impaired intrinsic insulin secretion capacity, who demonstrated a marked difference in glycemic control following treatment with two different FRC formulations. Treatment with IDegLira, an FRC product, demonstrated suboptimal glucose control in the patient. However, after a change in therapy to another FRC product, IGlarLixi, his glucose control markedly improved, even with a decrease in the injection dose. This difference could have been due to lixisenatide, a short-acting GLP-1RA contained in IGlarLixi, which exerts a postprandial hypoglycemic effect irrespective of intrinsic insulin secretion capacity. In conclusion, IGlarLixi has the potential to achieve good fasting and postprandial glucose control with a once-daily injection, even in patients with type 2 diabetes who have a reduced intrinsic insulin secretion capacity.
Supplementary Information
The online version contains supplementary material available at 10.1007/s13340-023-00621-5.
Keywords: Fixed ratio of combination injection therapy, Short-acting GLP-1 RA, CPR index, Pancreatic diabetes
Introduction
Fixed-ratio combination therapy (FRC) is a therapeutic option for patients with type 2 diabetes in which a fixed-ratio mixture of basal insulin and glucagon-like peptide-1 receptor agonist (GLP-1 RA) is subcutaneously injected once daily [1]. FRC provides better glycemic control before and after meals while reducing the risk of hypoglycemia, weight gain, and gastrointestinal symptoms [2, 3].
Currently, two products, IDegLira (1 dose of IDegLira contains 1 unit of insulin degludec and 0.036 mg liraglutide) and IGlarLixi (1 dose of IGlarLixi contains 1 unit of insulin glargine and 1 μg of lixisenatide), are available in Japan [4]. And they may be suitable for a diverse number of patients due to the differences in the mixing ratio of basal insulin and GLP-1 receptor agonists, as well as the characteristics of the GLP-1 receptor agonists in the formulations. Herein, we report a case of pancreatic diabetes mellitus with significantly impaired intrinsic insulin secretion capacity, and the differences in the properties of the two products resulted in marked variations in hypoglycemic effects.
Case presentation
The patient was a 71-year-old man who had diabetes for the past 12 years. Upon the first diagnosis, he was treated with oral hypoglycemic agents (sulfonylurea, metformin, and dipeptidyl peptidase-4 (DPP-4) inhibitors); however, his glycemic control gradually deteriorated. Therefore, basal supported oral therapy with insulin degludec (IDeg) was introduced when the patient was 70 years old. Despite this, his blood glucose levels did not improve. Hence, the DPP-4 inhibitor was discontinued and IDeg was replaced with IDegLira, an FRC formulation. The blood glucose level of the patient did not improve after intake of 500 mg/day of metformin and eight doses of IDegLira and thus was referred to our hospital.
Clinical history taking revealed that the patient drank three cups of sake daily, and had a medical history of stroke at the age of 68, with no residual disability. He also had a family history (father) of type 2 diabetes mellitus.
Despite a history of obesity in his 40 s, the BMI of the patient was 21.5 kg/m2 on arrival to our institution. His vital signs were normal and physical examination indicated just the loss of bilateral Achilles tendon reflexes.
The patient’s laboratory data on the day of referral are shown in Table S1. His HbA1c and fasting plasma glucose levels were 9.4% and 170 mg/dL, respectively. His C-peptide level was 0.5 ng/mL, and his CPR index (C-peptide level divided by plasma glucose level multiplied by 100) was 0.29. Abdominal computed tomography revealed multiple pancreatic stones with no evidence of liver cirrhosis (Fig. S1). We diagnosed the patient with pancreatic diabetes based on his history of alcohol consumption, imaging findings, and the low insulin secretion capacity of the patient. The use of metformin in patients who consume alcohol poses as a risk factor for lactic acidosis, thus, the patient was discontinued on metformin.
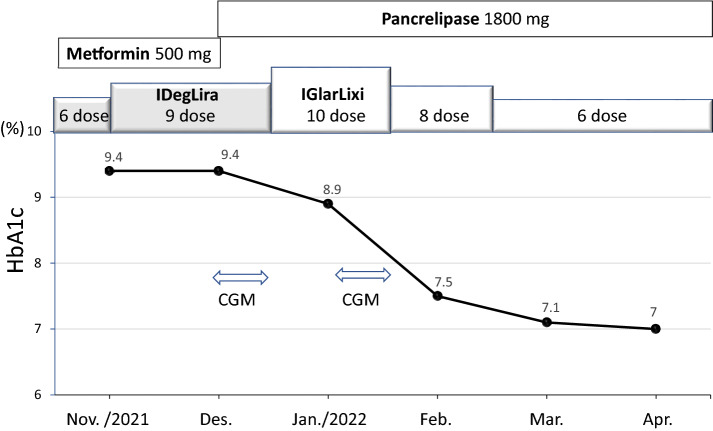
We initiated pancreatic enzyme preparations for pancreatic diabetes. Basal-bolus insulin injection therapy was recommended; however, the patient refused. Therefore, we performed continuous glucose monitoring (CGM, FreeStyle Libre Pro®) to help the patient track changes in his blood glucose levels. The CGM results demonstrated that he had marked hyperglycemia after breakfast and lunch (Fig. 1a). An increase in the dose of IDegLira was not possible due to the relatively low fasting glucose level of the patient. We again recommended basal-bolus insulin therapy, but the patient refused and requested a change in the FRC formulation. GLP-1 RA should be used with caution in patients with chronic pancreatitis, but since FRC containing GLP-1 has been used in the past, we decided to change to the other FRC formulation. Thus, his treatment regimen of 9 doses of IDegLira were replaced with 10 doses of IGlarLixi, and CGM was conducted again (Fig. 1b). This revealed a decrease in post-breakfast blood glucose levels, an improvement in hyperglycemia after lunch, and pre-breakfast hypoglycemia in the patient. Therefore, the dosage of IGlarLixi was gradually decreased to 6 doses at 3 months after initiation of IGlarLixi therapy, and the patient’s HbA1c level decreased to 7.0% (Fig. 2). His body weight did not change remarkably after the initiation of IGlarLixi.
Fig. 1.
CGM data using different FRC formulations. a IDegLira (9 doses). Although fasting blood glucose levels were acceptable, postprandial hyperglycemia was remarkable. b IGlarLixi (10 doses). Postprandial blood glucose levels improved, although there was slight hypoglycemia during fasting. CGM continuous glucose monitoring, FRC fixed-ratio combination, TIR time in range, TAR time above range, TBR time below range
Fig. 2.
Therapeutic response. Transition from IDegLira to IGlarLixi therapy resulted in a decrease in HbA1c despite the reduced injection dose
Discussion
In this study, we report a case with pancreatic diabetes and reduced insulin secretory capacity, who demonstrated a marked difference in glycemic improvement following treatment with two different FRC products. The FRC consists of basal insulin and a GLP-1 RA in a single injection [1]. The basal insulin in FRC controls fasting blood glucose levels whereas GLP-1 RA regulates postprandial blood glucose levels, contributing to better glycemic control throughout the day. Further, FRC is less likely to cause hypoglycemia and weight gain compared to treatment with basal insulin alone and also causes fewer gastrointestinal symptoms such as nausea compared to therapy with GLP-1RA injection alone [2, 3]. FRC has been reported to cause less weight gain than basal plus rapid-acting insulin therapy [5] and less weight gain and hypoglycemia than premixed insulin therapy [6]. Furthermore, patient satisfaction and adherence to therapy were also reported to be high [7, 8]. The two FRC formulations, IGlarLixi and IDegLira, differ in the basal insulin used, GLP-1RA formulation, and their mix ratio. However, there have been no direct head-to-head comparisons between the actions and effects of iGlarLixi and IDegLira.
In this patient with pancreatic diabetes and reduced insulin secretion capacity, basal-bolus injection therapy was the preferred choice of therapy, but the patient refused. Therefore, the dose of IDegLira was increased to achieve a fasting blood glucose level of 130 mg/dl; however, the HbA1c levels did not improve. Postprandial blood glucose levels were noticeably elevated, as observed by the CGM. Liraglutide, a GLP-1 RA present in IDegLira, is a long-acting GLP-1 RA whose hypoglycemic effect is known to be dependent on endogenous insulin secretion capacity [9]. Fortunately, after switching to IGlarLixi from IDegLira, postprandial blood glucose levels decreased. Lixisenatide is a short-acting GLP-1 RA, with the ability to lower postprandial blood glucose levels by delaying gastric emptying [10], and this effect is independent of the endogenous insulin secretory capacity [11, 12, 13]. To switch from IDegLira to IGlarLixi, the dose of IGlarLixi was increased by 1 unit, according to our results of a randomized controlled trial examining the effects of IDeg and IGlar on fasting blood glucose levels in patients with type 1 diabetes [14]. However, the effect of IGlarLixi on the resolution of glucotoxicity [15] which could result in fasting hypoglycemia was also considered, which necessitated a reduction in the injection dose.
Pancreatic enzyme supplementation and MDI in treating pancreatic diabetes often result in weight gain. Although we could not examine the effect on endogenous insulin secretion after initiation of IGlarLixi, no change in weight, in this case, might mean that the postprandial glycemic amelioration was achieved without an increase in endogenous insulin secretion. Interestingly, since pancreatic enzyme supplementation was reported to slow gastric emptying [16], improving postprandial blood glucose might not only be due to the effect of lixisenatide.
In conclusion, IGlarLixi may provide effective glycemic control with fewer injections in patients with impaired intrinsic insulin secretion as a replacement for the basal-bolus method.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file 1—Fig. 1 Computed tomography of the abdomen. The arrow indicates pancreatic stones.
Acknowledgements
We are grateful to the patient and staff of the General Internal Medicine and Diabetology and Endocrinology Department of Kobe University Hospital for their support.
Data Availability
The data that support the findings of this study are available on request from the corresponding author, SA. The data are not publicly available due to their containing information that could compromise the privacy of research participants.
Declarations
Conflict of interest
KS received lecture fees from Sanofi, Novo Nordisk Pharma, and Sumitomo Pharma. The remaining authors have no conflict of interest to declare.
Informed consent
Written informed consent was obtained from the patient.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Gomez-Peralta F, Al-Ozairi E, Jude EB, Li X, Rosenstock J. Titratable fixed-ratio combination of basal insulin plus a glucagon-like peptide-1 receptor agonist: a novel, simplified alternative to premix insulin for type 2 diabetes. Diabetes Obes Metab. 2021;23:1445–1452. doi: 10.1111/dom.14365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Watada H, Takami A, Spranger R, Amano A, Hashimoto Y, Niemoeller E. Efficacy and safety of 1:1 fixed-ratio combination of insulin glargine and lixisenatide versus lixisenatide in Japanese patients with Type 2 diabetes inadequately controlled on oral antidiabetic drugs: The LixiLan JP-O1 randomized clinical trial. Diabetes Care. 2020;43:1249–1257. doi: 10.2337/dc19-2452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Pei Y, Agner BR, Luo B, Dong X, Li D, Liu J, et al. Dual II China: Superior HbA1c reductions and weight loss with insulin degludec/liraglutide (IDegLira) versus insulin degludec in a randomized trial of Chinese people with type 2 diabetes inadequately controlled on basal insulin. Diabetes Obes Metab. 2021;23:2687–2696. doi: 10.1111/dom.14522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kaneto H, Koshida R, Baxter M. Fixed-ratio combination of basal insulin and glucagon-like peptide-1 receptor agonists in the treatment of Japanese people with type 2 diabetes: an innovative solution to a complex therapeutic challenge. Diabetes Obes Metab. 2020;22(Suppl 4):24–34. doi: 10.1111/dom.14095. [DOI] [PubMed] [Google Scholar]
- 5.McCrimmon RJ, Cheng AYY, Galstyan G, Djaballah K, Li X, Coudert M, et al. iGlarLixi versus basal plus rapid-acting insulin in adults with type 2 diabetes advancing from basal insulin therapy: the SoliSimplify Real-World study. Diabetes Obes Metab. 2022 doi: 10.1111/dom.14844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Polonsky WH, Giorgino F, Rosenstock J, Whitmire K, Lew E, Coudert M, et al. Improved patient-reported outcomes with iGlarLixi versus premix BIAsp 30 in people with type 2 diabetes in the SoliMix trial. Diabetes Obes Metab. 2022;24:2364–2372. doi: 10.1111/dom.14822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Inoue M, Lorenz M, Muto H, Wesch R, Hashimoto Y. Effect of a single dose of insulin glargine/lixisenatide fixed ratio combination (iGlarLixi) on postprandial glucodynamic response in Japanese patients with type 2 diabetes mellitus: a phase I randomized trial. Diabetes Obes Metab. 2019;21:2001–2005. doi: 10.1111/dom.13757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Watada H, Kaneko S, Komatsu M, Agner BR, Nishida T, Ranthe M, et al. Superior HbA1c control with the fixed-ratio combination of insulin degludec and liraglutide (IDegLira) compared with a maximum dose of 50 units of insulin degludec in Japanese individuals with type 2 diabetes in a phase 3, double-blind, randomized trial. Diabetes Obes Metab. 2019;21:2694–2703. doi: 10.1111/dom.13859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Takabe M, Matsuda T, Hirota Y, Hashimoto N, Nakamura T, Sakaguchi K, et al. C-peptide response to glucagon challenge is correlated with improvement of early insulin secretion by liraglutide treatment. Diabetes Res Clin Pract. 2012;98:e32–e35. doi: 10.1016/j.diabres.2012.09.036. [DOI] [PubMed] [Google Scholar]
- 10.Kuwata H, Yabe D, Murotani K, Fujiwara Y, Haraguchi T, Kubota S, et al. Effects of glucagon-like peptide-1 receptor agonists on secretions of insulin and glucagon and gastric emptying in Japanese individuals with type 2 diabetes: a prospective, observational study. J Diabetes Investig. 2021;12:2162–2171. doi: 10.1111/jdi.13598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Yabe D, Ambos A, Cariou B, Duvnjak L, Evans M, González-Gálvez G, et al. Efficacy of lixisenatide in patients with type 2 diabetes: a post hoc analysis of patients with diverse β-cell function in the GetGoal-M and GetGoal-S trials. J Diabetes Complicat. 2016;30:1385–1392. doi: 10.1016/j.jdiacomp.2016.05.018. [DOI] [PubMed] [Google Scholar]
- 12.Bonadonna RC, Blonde L, Antsiferov M, Berria R, Gourdy P, Hatunic M, et al. Lixisenatide as add-on treatment among patients with different β-cell function levels as assessed by HOMA-β index. Diabetes Metab Res Rev. 2017;33:e2897. doi: 10.1002/dmrr.2897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Juel CTB, Lund A, Andersen MM, Hansen CP, Storkholm JH, Rehfeld JF, et al. The GLP-1 receptor agonist lixisenatide reduces postprandial glucose in patients with diabetes secondary to total pancreatectomy: a randomised, placebo-controlled, double-blinded crossover trial. Diabetologia. 2020;63:1285–1298. doi: 10.1007/s00125-020-05158-9. [DOI] [PubMed] [Google Scholar]
- 14.Nakamura T, Sakaguchi K, So A, Nakajima S, Takabe M, Komada H, et al. Effects of insulin degludec and insulin glargine on day-to-day fasting plasma glucose variability in individuals with type 1 diabetes: a multicentre, randomised, crossover study. Diabetologia. 2015;58:2013–2019. doi: 10.1007/s00125-015-3648-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Miura H, Muramae N, Mori K, Otsui K, Sakaguchi K. Successful resolution of glucose toxicity with the use of fixed-ratio combination injection of basal insulin and short-acting glucagon-like peptide 1 (GLP-1) receptor agonist. Cureus. 2022;14:e25889. doi: 10.7759/cureus.25889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Perano SJ, Couper JJ, Horowitz M, Martin AJ, Kritas S, Sullivan T, Rayner CK. Pancreatic enzyme supplementation improves the incretin hormone response and attenuates postprandial glycemia in adolescents with cystic fibrosis: a randomized crossover trial. J Clin Endocrinol Metab. 2014;99:2486–2493. doi: 10.1210/jc.2013-4417. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary file 1—Fig. 1 Computed tomography of the abdomen. The arrow indicates pancreatic stones.
Data Availability Statement
The data that support the findings of this study are available on request from the corresponding author, SA. The data are not publicly available due to their containing information that could compromise the privacy of research participants.