Abstract
Purpose To evaluate the magnitude of the association of the polymorphisms of the genes PGR, CYP17A1 and CYP19A1 in the development of endometriosis.
Methods This is a retrospective case-control study involving 161 women with endometriosis (cases) and 179 controls. The polymorphisms were genotyped by real-time polymerase chain reaction using the TaqMan system. The association of the polymorphisms with endometriosis was evaluated using the multivariate logistic regression.
Results The endometriosis patients were significantly younger than the controls (36.0 ± 7.3 versus 38.0 ± 8.5 respectively, p = 0.023), and they had a lower body mass index (26.3 ± 4.8 versus 27.9 ± 5.7 respectively, p = 0.006), higher average duration of the menstrual flow (7.4 ± 4.9 versus 6.1 ± 4.4 days respectively, p = 0.03), and lower average time intervals between menstrual periods (25.2 ± 9.6 versus 27.5 ± 11.1 days respectively, p = 0.05). A higher prevalence of symptoms of dysmenorrhea, dyspareunia, chronic pelvic pain, infertility and intestinal or urinary changes was observed in the case group when compared with the control group. The interval between the onset of symptoms and the definitive diagnosis of endometriosis was 5.2 ± 6.9 years. When comparing both groups, significant differences were not observed in the allelic and genotypic frequencies of the polymorphisms PGR +331C > T, CYP17A1 -34A > G and CYP19A1 1531G > A, even when considering the symptoms, classification and stage of the endometriosis. The combined genotype PGR +331TT/CYP17A1 -34AA/CYP19A11531AA is positively associated with endometriosis (odds ratio [OR] = 1.72; 95% confidence interval [95%CI] = 1.09–2.72).
Conclusions The combined analysis of the polymorphisms PGR-CYP17A1-CYP19A1 suggests a gene-gene interaction in the susceptibility to endometriosis. These results may contribute to the identification of biomarkers for the diagnosis and/or prognosis of the disease and of possible molecular targets for individualized treatments.
Keywords: polymorphisms, estrogens, endometriosis, biomarkers
Abstract
Resumo
Objetivo Avaliar a magnitude de associação de polimorfismos nos genes PGR, CYP17A1 e CYP19A1 no desenvolvimento da endometriose.
Métodos Este é um estudo retrospectivo do tipo caso-controle, envolvendo 161 mulheres com endometriose (casos) e 179 controles. Os polimorfismos foram genotipados pela reação em cadeia da polimerase em tempo real utilizando o sistema TaqMan. A associação dos polimorfismos estudados com a endometriose foi avaliada pela regressão logística multivariada.
Resultados As pacientes com endometriose eram significativamente mais jovens do que os controles (36,0 ± 7,3 versus 38,0 ± 8,5, respectivamente, p = 0,023), apresentaram um índice de massa corporal menor (26,3 ± 4,8 versus 27,9 ± 5,7, respectivamente, p = 0,006), maior tempo médio de duração do fluxo menstrual (7,4 ± 4,9 versus 6,1 ± 4,4 dias, respectivamente, p = 0,03) e menor tempo médio do intervalo entre as menstruações (25,2 ± 9,6 versus 27,5 ± 11,1 dias, respectivamente, p = 0,05). Uma maior prevalência dos sintomas de dismenorreia, dispareunia, dor pélvica crônica, infertilidade, alterações intestinais e urinárias foi observada no grupo casos comparado ao grupo controle. O tempo médio entre o início dos sintomas e o diagnóstico definitivo de endometriose foi de 5,2 ± 6,9 anos. Comparando os dois grupos, não foram observadas diferenças significativas nas frequências alélicas e genotípicas dos polimorfismos PGR +331C > T, CYP17A1 -34A > G e CYP19A1 1531G > A, e nem considerando os sintomas, a classificação e o estadiamento da endometriose. O genótipo combinado PGR +331TT/CYP17A1 -34AA/CYP19A11531AA está associado positivamente com a endometriose (razão de possibilidades [RP] = 1,72; intervalo de confiança de 95% [IC95%] = 1,09–2,72).
Conclusões A análise combinada dos polimorfismos PGR-CYP17A1-CYP19A1 sugere uma interação gene-gene na susceptibilidade à endometriose. Estes resultados podem contribuir para a identificação de biomarcadores para o diagnóstico e/ou prognóstico da doença, assim como de possíveis alvos moleculares para um tratamento individualizado.
Palavras-chave: polimorfismos, estrógenos, endometriose, biomarcadores
Introduction
Endometriosis is a benign gynecological estrogen-dependent disease characterized by the presence of endometrial tissue out of the uterine cavity, affecting nearly 10% of women of reproductive age. Symptoms may include dysmenorrhea, dyspareunia, chronic pelvic pain and infertility.1 The pathogenesis and the molecular mechanisms that are involved in the development of endometriosis are not yet clear, and hereditary susceptibility is an area of growing investigation for the identification of genetic polymorphisms that may lead to an increased risk of developing the disease.2 3
Estrogen performs a fundamental role in endometriosis, which predominantly occurs in women of reproductive age who have high estrogen production.4 5 An increase in enzyme expression is responsible for the estrogen synthesis and reduction of progesterone receptor (PGR) expression observed in samples of endometrial injuries, but these phenomena are not found in controls.6 7 As endometriosis is an estrogen-dependent disease, genetic polymorphisms involved in the biosynthesis and regulation of estrogens could be considered possible biomarkers for its diagnosis and/or prognosis. Cytochrome P450 17A1 (CYP17A1) is involved in the initial stages of estrogen synthesis, converting pregnenolone into 17α-hydroxypregnenolone and, subsequently, into dehydroepiandrosterone. Furthermore, cytochrome P450 19A1 (CYP19A1) acts in the final stage by converting androstenedione into estrone, and testosterone into estradiol.4 8 The enzyme CYP17A1, also known as 17 α-hydroxylase, is encoded by the gene with the same name, located in chromosome 10q24.3.8 The single nucleotide polymorphism (SNP) CYP17A1 -34A > G is located in the 5′ untranslated region (UTR) of the CYP17A1 gene, and it causes a significant increase in the expression of 17 α-hydroxylase.8 A different gene product, the aromatase enzyme, is encoded by the gene CYP19A1, which is located in chromosome 15q21. The SNP CYP19A1 1531G > A is found in the 3′UTR of this gene, and it causes a significant change in the levels of circulating estradiol.8 Progesterone is also involved in the pathogenesis of endometriosis, as it is a strong antagonist of estrogen, and thus plays an essential role in the regulation of endometrial cell proliferation.6 The PGR gene, located in chromosome 11q22–q23, is responsible for encoding both progesterone receptor isoforms (PR-A and PR-B) by transcribing from alternative promoters.9 The SNP PGR +331C/T, located in the gene promoter region, creates an additional TATA box, and causes higher transcription of PR-B.9
As the SNPs PGR +331C > T, CYP17A1 -34A > G and CYP19A1 1531G > A are located near potential elements that regulate their respective genes, interfering with the levels of expression of the corresponding proteins, it becomes relevant to evaluate the influence of these SNPs in the development of endometriosis. To date, 12 studies have evaluated the association of the SNPs PGR +331C > T, CYP17A1 -34A > G and CYP19A1 1531G > A in the development of endometriosis in different populations. However, the results of the analyses are controversial.10 11 12 13 14 15 16 17 18 19 20 21 In this context, the objective of this study was to evaluate the magnitude of the association of the SNPs PGR +331C > T, CYP17A1 -34A > G and CYP19A1 1531G > A with the development of endometriosis in women treated in two public reference hospitals in Brazil.
Methods
Study Design
This was a retrospective, case-control study approved by the Human Research Ethics Committees of two of our institutions (under protocols number 414/11 and 1.244.29 respectively), both located in the city of Rio de Janeiro, Brazil. All participating patients (n = 340) provided written informed consent, and visited one of the two institutions between March 2011 and October 2015. The research was conducted in accordance with the Declaration of Helsinki, which was revised in 2008. Patients with a surgical diagnosis (after laparoscopy or laparotomy) of endometriosis with histological confirmation of the disease, as well as those diagnosed by magnetic resonance imaging (MRI), were considered the case group (n = 161). The control group (n = 179) consisted of women with a negative diagnosis of endometriosis, after laparoscopy or laparotomy for tubal ligation (n = 48) or for the treatment of benign diseases, such as myoma (n = 52), ovarian cysts (n = 30), hydrosalpinx (n = 5), or for other reasons (n = 44). Women with any history or diagnosis of cancer or adenomyosis were excluded.2
The stage of the endometriosis was determined according to the revised American Fertility Society classification, which divides the disease into four stages: I (minimum), II (mild), III (moderate) and IV (severe).22 Regarding the classification of the endometriosis, we considered the proposal of Nisolle and Donnez:23 superficial endometriosis (SUP), ovarian endometrioma (OMA), and deep infiltrative endometriosis (DIE). Superficial endometriosis and ovarian endometrioma may be found in association with deep endometriosis,24 and the cases in which this association was observed were considered DIE.
The body mass index (BMI) was calculated as the weight (kg) divided by the height squared (m2). Only severe and incapacitating symptoms of pain were included. Women who failed to conceive after one year of regular, contraceptive-free intercourse were considered infertile. Cyclical intestinal or urinary symptoms were defined as bowel and/or urinary pain and/or bleeding coinciding with menstrual periods.
Genotyping
Genomic DNA was extracted from the peripheral blood sample using a genomic DNA extraction kit (Genomic DNA Extraction, Real Biotech Corporation, Banqiao City, Taiwan), according to the manufacturer's instructions.
Genotyping of PGR +331C > T (rs10895068), CYP17A1 -34A > G (rs743572) and CYP19A1 1531G > A (rs10046) SNPs was performed by real-time polymerase chain reaction (PCR) using the TaqMan system. Oligonucleotides and probes specific to each SNP were obtained from Applied Biosystems: rs10895068 (C_27858738_10), rs743572 (C_2852784_30) and rs10046 (C_8234731_30). For all SNPs, PCRs were performed with 30 ng of template DNA, 1× TaqMan Universal Master Mix (Applied Biosystems, Foster City, CA, US), and with each primer and probe assay at 1× dilution, and H2O to 8μL. The PCR conditions were: 95°C for 10 minutes, followed by 40 cycles of denaturation at 92°C for 15 seconds, and annealing at 60°C for 1 minute. Allele-detection was performed on a 7500 Real-Time System (Applied Biosystems, Foster City, CA, US), and the genotypes were then determined directly.
Statistical Analyses
The continuous variables were expressed as the mean ± standard deviation (SD), and the differences between means were evaluated using the Student's t-test. The categorical data were expressed as percentages, and evaluated by the Chi-square (χ2) test or Fisher's exact test, when applicable. For each SNP, the Hardy-Weinberg equilibrium (HWE) was calculated, and the allelic and genotypic distributions were compared between cases and controls by the χ2 test or Fisher's exact test. To evaluate the association between the SNPs and the development of endometriosis, as well as the presence of symptoms, the classification and staging of the disease were used to estimate the odds ratios (ORs) and their respective 95% confidence intervals (95%CIs), with adjustment for possible confounding factors, using a multivariate logistic regression. Values of p < 0.05 were considered statistically significant. All analyses were performed using the Statistical Package for the Social Sciences (SPSS, IBM Corp., Armonk, NY, US) software, version 20.0.
Results
The endometriosis cases were diagnosed through laparoscopy (n = 90, 55.9%), laparotomy (n = 27, 16.8%) or MRI (n = 44, 27.3%), and 87 (54%) were classified as DIE. Of the 117 surgery cases, 37 (31.6%) presented stages I-II diseases, and 80 (68.4%) presented stages III-IV. The most common locations of the endometriotic lesions were in the ovary (31%), followed by the intestine (19%), and the uterosacral ligaments (15%). The average age of the endometriosis patients at the time of diagnosis was 31.5 ± 7.5, and the average time for disease diagnosis, which was the time since the beginning of the symptoms until the definitive diagnosis, was 5.2 ± 6.9 years.
In Table 1, the demographic and clinical data from the study population are described. The patients with endometriosis were significantly younger than the controls (36.0 ± 7.3 versus 38.0 ± 8.5 respectively, p = 0.023), and presented a higher education level (41.6% versus 14.5%), as well as a lower BMI (26.3 ± 4.8 versus 27.9 ± 5.7 respectively, p = 0.006). The number of infertile women (primary or secondary) was significantly higher in the case group (42.9%) than in the control group (12.3%), and nearly 63% of the women from the control group had 2 or more children. The patients with endometriosis presented a higher prevalence (p < 0.001) of symptoms, including dysmenorrhea, dyspareunia, chronic pelvic pain, and urinary and intestinal changes. Considering the characteristics of the menstrual cycle, a significant difference was detected between cases and controls in relation to the average duration of the menstrual flow (7.4 ± 4.9 versus 6.1 ± 4.4 days respectively, p = 0.03) and the average interval between menstrual periods (25.2 ± 9.6 versus 27.5 ± 11.1 days respectively, p = 0.05).
Table 1. Demographic and clinical characteristics of the study population (N = 340).
| Variable | Controls (N = 179) |
Cases (N = 161) |
p* |
|---|---|---|---|
| Age (years) | n (%) | ||
| 18–29 | 30 (16.8) | 29 (18.0) | 0.011 |
| 30–39 | 60 (33.5) | 79 (49.1) | |
| ≥ 40 | 86 (48.0) | 48 (29.8) | |
| No information | 3 (1.7) | 5 (3.1) | |
| Marital status | |||
| Married/partner | 96 (53.6) | 111 (68.9) | 0.046 |
| Single | 53 (29.6) | 38 (23.7) | |
| Divorced/Widow | 5 (2.8) | 1 (0.6) | |
| No information | 25 (14.0) | 11 (6.8) | |
| Level of Schooling | |||
| Elementary education | 38 (21.3) | 24 (14.9) | < 0.001 |
| High school | 89 (49.7) | 60 (37.3) | |
| Higher education | 26 (14.5) | 67 (41.6) | |
| No information | 26 (14.5) | 10 (6.2) | |
| BMI | |||
| < 18.5 | 3 (1.7) | 7 (4.4) | 0.013 |
| 18.5–24.9 | 48 (26.8) | 48 (29.8) | |
| 25–29.9 | 50 (27.9) | 63 (39.1) | |
| 30–40 | 65 (36.3) | 35 (21.7) | |
| > 40 | 13 (7.3) | 8 (5.0) | |
| Infertility | |||
| Primary | 19 (10.6) | 53 (32.9) | < 0.001 |
| Secondary | 3 (1.7) | 16 (9.9) | |
| None** | 133 (74.3) | 58 (36.1) | |
| No attempt | 20 (11.2) | 32 (19.9) | |
| No information | 4 (2.2) | 2 (1.2) | |
| Parity | |||
| 0 | 19 (10.6) | 53 (32.8) | < 0.001 |
| 1 | 23 (12.8) | 37 (23.0) | |
| 2 | 58 (32.5) | 27 (16.8) | |
| 3 or more | 55 (30.7) | 10 (6.2) | |
| No attempt | 20 (11.2) | 32 (20.0) | |
| No information | 4 (2.2) | 2 (1.2) | |
| Symptoms*** | |||
| Dysmenorrhea | 29 (16.1) | 77 (47.5) | < 0.001 |
| Chronic pelvic pain | 70 (38.9) | 124 (76.5) | |
| Dyspareunia | 50 (27.8) | 102 (63.0) | |
| Cyclical urinary complaints**** | 9 (6.4) | 41 (27.5) | |
| Cyclical intestinal complaints**** | 8 (6.0) | 74 (49.7) | |
Abbreviation: BMI, body mass index.
Notes:*p-value obtained by Pearson's Chi-squared (χ2) test. **Number of fertile women. ***The same woman can have more than one symptom. ****Pain or bleeding during the menstrual period.
When comparing the allelic and genotypic frequencies of the SNPs PGR +331C > T, CYP17A1 -34A > G and CYP19A1 G > A between the cases and controls (Fig. 1), no significant differences were detected, even when considering the staging and endometriosis classification (data not shown). The allelic distribution of the SNPs PGR +331C > T, CYP17A1 -34A > G and CYP19A1 G > A in relation to the absence or presence of symptoms (dysmenorrhea, pelvic pain, dyspareunia, infertility, and urinary and intestinal problems, for example) in the endometriosis patients is summarized in Fig. 2. Considering the symptoms of the disease, no significant differences were found.
Fig. 1.
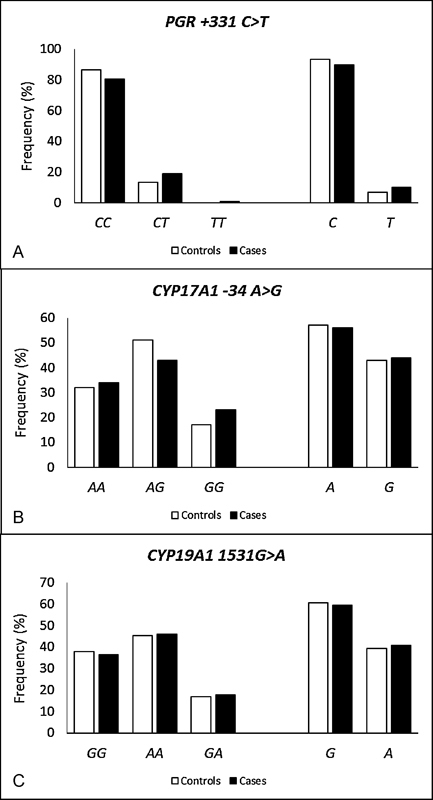
Allelic and genotypic frequencies of the polymorphisms PGR +331C > T, CYP17A1 -34A > G and CYP19A1 G > A in the study population.
Fig. 2.
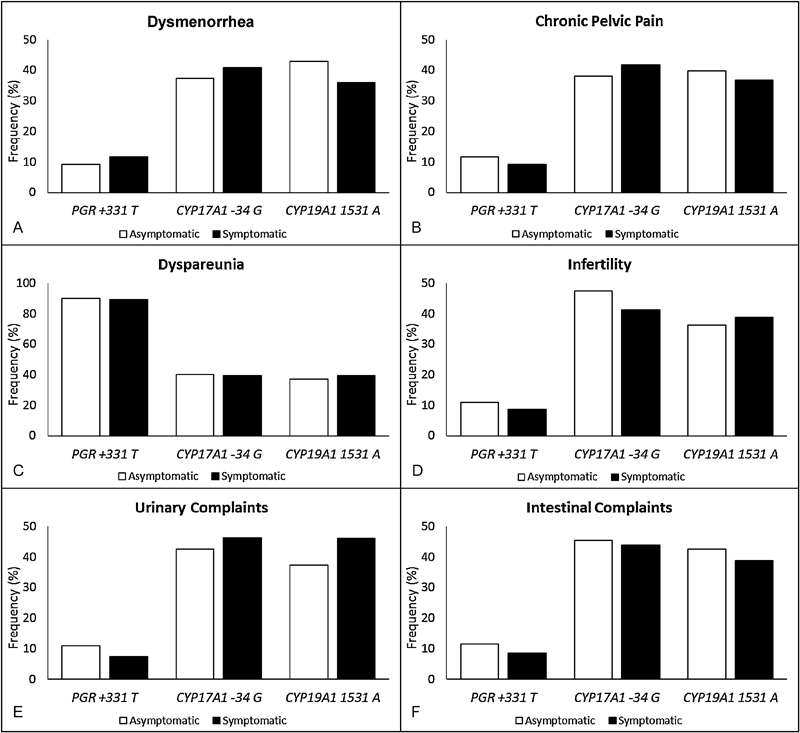
Minor allelic frequencies of the polymorphisms studied among symptomatic and asymptomatic endometriosis patients.
A combined analysis of the three studied SNPs (PGR +331C > T, CYP17A1 -34A > G and CYP19A1 G > A), compared between the endometriosis cases and controls, was performed to investigate whether the presence of more than 1 SNP would increase the risk of developing the disease (Table 2). It has been observed that relative to the combined wild-type genotype (PGR +331CC/CYP17A1 -34AA/CYP19A11531GG), the combined genotype PGR +331TT/CYP17A1 -34AA/CYP19A11531AA is associated with an increased risk of developing endometriosis. A combined analysis of the PGR +331C > T, CYP17A1 -34A > G and CYP19A1 G > A genotypes was also performed in relation to the absence or presence of symptoms (dysmenorrhea, pelvic pain, dyspareunia, infertility, and urinary and intestinal problems, for example) in the endometriosis patients. However, no significant differences were found (data not shown).
Table 2. Combined genotype frequencies of the polymorphisms PGR +331 C > T, CYP17 -34 A > G and CYP19 1531G > A between controls and cases, and their association with the risk of endometriosis.
| Genotypes | Controls N (%) |
Cases N (%) |
p * | OR (95%CI)** |
|---|---|---|---|---|
| PGR +331C > T, CYP17 -34A > G and CYP19 1531G > A | ||||
| WT / WT / WT | 23 (13.5) | 11 (7.3) | 1*** | |
| WT / WT / VAR | 29 (17.1) | 28 (18.6) | 0.20 | 1.82 (0.73–4.57) |
| WT / VAR / VAR | 70 (41.2) | 60 (40.0) | 0.17 | 1.35 (0.88–2.05) |
| VAR / WT / WT | 2 (1.2) | 4 (2.7) | 0.18 | 1.57 (0.82–3.00) |
| VAR / VAR / WT | 7 (4.1) | 10 (6.7) | 0.20 | 1.23 (0.90–1.67) |
| VAR / WT / VAR | 1 (0.6) | 7 (4.7) | 0.02 | 1.72 (1.09–2.72) |
| WT / VAR / WT | 25 (14.7) | 22 (14.7) | 0.15 | 1.13 (0.96–1.34) |
| VAR / VAR / VAR | 13 (7.6) | 8 (5.3) | 0.87 | 1.02 (0.85–1.21) |
Abbreviations: 95%CI, 95% confidence interval; OR, odds ratio.
Notes: WT/WT/WT, CC/AA/GG; WT/WT/VAR, CC/AA/GA or CC/AA/AA; WT/VAR/VAR, CC/GG/AA or CC/AG/GA or CC/AG/AA or CC/GG/GA; VAR/WT/WT, CT/AA/GG or TT/AA/GG; VAR/VAR/WT, CT/AG/GG or CT/GG/GG or TT/AG/GG or TT/GG/GG; VAR/WT/VAR, CT/AA/GA or CT/AA/AA or TT/AA/GA or TT/AA/AA; WT/VAR/WT, CC/GG/GG or CC/AG/GG; VAR/VAR/VAR, TT/GG/AA; *p-value obtained through the Chi-squared test (Pearson P-value) or Fisher's exact test. **Adjusted by age and BMI. ***Reference group.
In Table 3, we describe the variant allele frequencies of the SNPs PGR +331 T, CYP17A1 -34 G and CYP19A1A in different populations. The allele frequency PGR +331 T varied between 2% and 10% and 5% and 10% in the cases and controls respectively, based on 3 studies that have evaluated this SNP, including the present one. The SNP rs743572 has been evaluated in 7 studies, in addition to the present study, and the frequency of the CYP17A1 -34 G allele varied between 35% and 58% in the cases and between 31% and 63% in the controls. In addition to our study, 4 other studies also evaluated the SNP rs10046, and the allele frequency of CYP19A1 1531A varied between 35% and 58% and between 41% and 60% in the cases and controls respectively.
Table 3. Frequency of the studied polymorphisms among different populations (endometriosis patients and controls).
| Polymorphism | Population | N* | Frequency among the controls (%) | Frequency among the cases (%) | Association with endometriosis | Reference |
|---|---|---|---|---|---|---|
| PGR +331 C > T (rs10895068) | PGR +331 T | |||||
| Estonia | 349 | 8.8 | 6.7 | No association | Lamp et al18 | |
| United States | 823 | 4.8 | 5.1 | No association | Trabert et al19 | |
| Netherlands | 165 | 9.7 | 2.3 | Protective (T allele) | van Kaam et al15 | |
| Brazil | 179 | 6.6 | 10.1 | No association | Present study | |
| CYP17A1 -34 A > G (rs743572) | CYP17A1 -34 G | |||||
| Turkey | 93 | 30.8 | 57.6 | No association | Bozdag et al17 | |
| China | 247 | 60.2 | 50.8 | Risk (A allele) | Hsieh et al11 | |
| China | 227 | 62.9 | 50.8 | Risk (A allele) | Hsieh et al12 | |
| China | 510 | 54.3 | 58.2 | No association | Juo et al13 | |
| Japan | 317 | 39.8 | 42.9 | No association | Kado et al10 | |
| United States | 823 | 39.3 | 34.8 | No association | Trabert et al19 | |
| Italy | 190 | 37.2 | 42.3 | No association | Vietri et al16 | |
| Brazil | 180 | 42.7 | 44.4 | No association | Present study | |
| CYP19A1 1531 G > A (rs10046) | CYP19A1 1531 A | |||||
| Korea | 412 | 55.9 | 55.8 | No association | Hur et al14 | |
| Estonia | 349 | 59.8 | 55.0 | No association | Lamp et al18 | |
| China | 371 | 56.4 | 57.5 | No association | Wang et al20 | |
| China | 202 | 41.0 | 35.3 | No association | Yang et al21 | |
| Brazil | 180 | 39.4 | 40.6 | No association | Present study | |
Abbreviations: CYP17A1, cytochrome P450 17A1; CYP19A1, cytochrome P450 19A1; PGR, progesterone receptor.
Note: *N total number of individuals included in the study (cases + controls).
Discussion
Endometriosis is a complex multifactorial gynecological disease caused by the combination of hormonal, genetic and environmental factors, as well as immunological processes. Estrogen and progesterone are essential in the regulation of endometrial tissue growth, and, as such, they may play a central role in the pathogenesis of endometriosis.5 6 In this study, a positive association between the combined genotype PGR +331TT/CYP17A1 -34AA/CYP19A1 1531AA and the development of endometriosis was observed.
Supporting our results, neither Lamp et al,18 in Estonia, nor Trabert et al,19 in the United States, could find an association with only the PGR +331T allele. However, van Kaam et al,15 in the Netherlands, observed a protective effect (OR = 0.22; 95%CI = 0.06–0.77) for the development of endometriosis. In agreement with our findings, 5 studies from China,13 Japan,10 Turkey,17 Italy16 and the United States19 failed to observe an association between the SNP CYP17A1 -34G and endometriosis. However, 2 studies in the Chinese population11 12 found a positive association between endometriosis and the CYP17A1 -34A allele (p = 0.046 and p = 0.009). With relation to the SNP CYP19A1 1531G > A, none of the studies found an association with endometriosis, which is similar to our results.14 18 20 21 To date, no study has investigated the combined effect of the SNPs PGR +331TT/CYP17A1 -34AA/CYP19A1 1531AA on the development of endometriosis.
The strength of the present study is that it is the first study performed in the Brazilian population that evaluated the SNPs PGR +331C > T, CYP17A1 -34A > G and CYP19A1 1531G > A in terms of endometriosis development, while considering the symptoms of the disease. All control patients were surgically evaluated to confirm a negative diagnosis of endometriosis; 27% of them had previously undergone sterilization, and 63% had already had 2 or more children. A limitation of this study was that the controls also included women with other gynecological diseases, providing lower risk estimations. Furthermore, considering the endometriosis patients, 27% were diagnosed by MRI. However, this has been a specific and accurate method for the detection of deep endometriosis.25 26 27 28
In conclusion, the combined analysis of the polymorphisms PGR-CYP17A1-CYP19A1 suggests a gene-gene interaction in the susceptibility to endometriosis. The results may contribute to the identification of genetic biomarkers that are able to help in disease diagnosis and/or prognosis, as well as in the identification of possible molecular targets for individualized treatments.
Acknowledgment
The authors would like to thank Lucas Rafael Lopes and Caroline Passos from Centro Universitário Estadual da Zona Oeste, Rio de Janeiro, Brazil, for their technical assistance. This study was supported by the Brazilian agencies Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ) and Fundação Ary Frauzino - Oncobiologia.
Footnotes
Conflicts of Interest The authors have no conflicts of interest to disclose.
References
- 1.Acién P, Velasco I. Endometriosis: a disease that remains enigmatic. ISRN Obstet Gynecol. 2013;2013:242149. doi: 10.1155/2013/242149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Perini J A, Cardoso J V, Berardo P T et al. Role of vascular endothelial growth factor polymorphisms (-2578C > A, -460 T > C, -1154G > A, +405G > C and +936C > T) in endometriosis: a case-control study with Brazilians. BMC Womens Health. 2014;14:117. doi: 10.1186/1472-6874-14-117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Zondervan K T, Rahmioglu N, Morris A P et al. Beyond endometriosis genome-wide association study: from genomics to phenomics to the patient. Semin Reprod Med. 2016;34(04):242–254. doi: 10.1055/s-0036-1585408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bell D W, Brannigan B W, Matsuo K et al. Increased prevalence of EGFR-mutant lung cancer in women and in East Asian populations: analysis of estrogen-related polymorphisms. Clin Cancer Res. 2008;14(13):4079–4084. doi: 10.1158/1078-0432.CCR-07-5030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Barcelos I D, Donabella F C, Ribas C P et al. Down-regulation of the CYP19A1 gene in cumulus cells of infertile women with endometriosis. Reprod Biomed Online. 2015;30(05):532–541. doi: 10.1016/j.rbmo.2015.01.012. [DOI] [PubMed] [Google Scholar]
- 6.Bedaiwy M A, Dahoud W, Skomorovska-Prokvolit Y et al. Abundance and Localization of Progesterone Receptor Isoforms in Endometrium in Women With and Without Endometriosis and in Peritoneal and Ovarian Endometriotic Implants. Reprod Sci. 2015;22(09):1153–1161. doi: 10.1177/1933719115585145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lafay Pillet M C, Schneider A, Borghese B et al. Deep infiltrating endometriosis is associated with markedly lower body mass index: a 476 case-control study. Hum Reprod. 2012;27(01):265–272. doi: 10.1093/humrep/der346. [DOI] [PubMed] [Google Scholar]
- 8.Ghisari M, Eiberg H, Long M, Bonefeld-Jørgensen E C. Polymorphisms in phase I and phase II genes and breast cancer risk and relations to persistent organic pollutant exposure: a case-control study in Inuit women. Environ Health. 2014;13(01):19. doi: 10.1186/1476-069X-13-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.De Vivo I, Huggins G S, Hankinson S E et al. A functional polymorphism in the promoter of the progesterone receptor gene associated with endometrial cancer risk. Proc Natl Acad Sci U S A. 2002;99(19):12263–12268. doi: 10.1073/pnas.192172299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kado N, Kitawaki J, Obayashi H et al. Association of the CYP17 gene and CYP19 gene polymorphisms with risk of endometriosis in Japanese women. Hum Reprod. 2002;17(04):897–902. doi: 10.1093/humrep/17.4.897. [DOI] [PubMed] [Google Scholar]
- 11.Hsieh Y Y, Chang C C, Tsai F J, Lin C C, Tsai C H. Cytochrome P450c17alpha 5′-untranslated region *T/C polymorphism in endometriosis. J Genet. 2004;83(02):189–192. doi: 10.1007/BF02729896. [DOI] [PubMed] [Google Scholar]
- 12.Hsieh Y Y, Chang C C, Tsai F J, Lin C C, Tsai C H. Estrogen receptor alpha dinucleotide repeat and cytochrome P450c17alpha gene polymorphisms are associated with susceptibility to endometriosis. Fertil Steril. 2005;83(03):567–572. doi: 10.1016/j.fertnstert.2004.07.977. [DOI] [PubMed] [Google Scholar]
- 13.Juo S H, Wang T N, Lee J N, Wu M T, Long C Y, Tsai E M. CYP17, CYP1A1 and COMT polymorphisms and the risk of adenomyosis and endometriosis in Taiwanese women. Hum Reprod. 2006;21(06):1498–1502. doi: 10.1093/humrep/del033. [DOI] [PubMed] [Google Scholar]
- 14.Hur S E, Lee S, Lee J Y, Moon H S, Kim H L, Chung H W. Polymorphisms and haplotypes of the gene encoding the estrogen-metabolizing CYP19 gene in Korean women: no association with advanced-stage endometriosis. J Hum Genet. 2007;52(09):703–711. doi: 10.1007/s10038-007-0174-x. [DOI] [PubMed] [Google Scholar]
- 15.van Kaam K J, Romano A, Schouten J P, Dunselman G A, Groothuis P G. Progesterone receptor polymorphism +331G/A is associated with a decreased risk of deep infiltrating endometriosis. Hum Reprod. 2007;22(01):129–135. doi: 10.1093/humrep/del325. [DOI] [PubMed] [Google Scholar]
- 16.Vietri M T, Cioffi M, Sessa M et al. CYP17 and CYP19 gene polymorphisms in women affected with endometriosis. Fertil Steril. 2009;92(05):1532–1535. doi: 10.1016/j.fertnstert.2008.07.1786. [DOI] [PubMed] [Google Scholar]
- 17.Bozdag G, Alp A, Saribas Z, Tuncer S, Aksu T, Gurgan T. CYP17 and CYP2C19 gene polymorphisms in patients with endometriosis. Reprod Biomed Online. 2010;20(02):286–290. doi: 10.1016/j.rbmo.2009.11.007. [DOI] [PubMed] [Google Scholar]
- 18.Lamp M, Peters M, Reinmaa E et al. Polymorphisms in ESR1, ESR2 and HSD17B1 genes are associated with fertility status in endometriosis. Gynecol Endocrinol. 2011;27(06):425–433. doi: 10.3109/09513590.2010.495434. [DOI] [PubMed] [Google Scholar]
- 19.Trabert B, Schwartz S M, Peters U et al. Genetic variation in the sex hormone metabolic pathway and endometriosis risk: an evaluation of candidate genes. Fertil Steril. 2011;96(06):1401–1.406E6. doi: 10.1016/j.fertnstert.2011.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wang L, Lu X, Wang D et al. CYP19 gene variant confers susceptibility to endometriosis-associated infertility in Chinese women. Exp Mol Med. 2014;46:e103. doi: 10.1038/emm.2014.31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Yang X, Chen S Q, Liu M. [Association of the CYP19 gene polymorphism with genetic susceptibility to endometriosis] Zhonghua Yi Xue Yi Chuan Xue Za Zhi. 2010;27(06):692–696. doi: 10.3760/cma.j.issn.1003-9406.2010.06.021. [DOI] [PubMed] [Google Scholar]
- 22.Revised American Society for Reproductive Medicine classification of endometriosis: 1996. Fertil Steril. 1997;67(05):817–821. doi: 10.1016/s0015-0282(97)81391-x. [DOI] [PubMed] [Google Scholar]
- 23.Nisolle M, Donnez J. Peritoneal endometriosis, ovarian endometriosis, and adenomyotic nodules of the rectovaginal septum are three different entities. Fertil Steril. 1997;68(04):585–596. doi: 10.1016/s0015-0282(97)00191-x. [DOI] [PubMed] [Google Scholar]
- 24.Chapron C, Santulli P, de Ziegler D et al. Ovarian endometrioma: severe pelvic pain is associated with deeply infiltrating endometriosis. Hum Reprod. 2012;27(03):702–711. doi: 10.1093/humrep/der462. [DOI] [PubMed] [Google Scholar]
- 25.Bianek-Bodzak A, Szurowska E, Sawicki S, Liro M. The importance and perspective of magnetic resonance imaging in the evaluation of endometriosis. BioMed Res Int. 2013;2013:436589. doi: 10.1155/2013/436589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ma K, Majumder K, Clayton R, Rajashanker B, Ed-Osagie E.Correlation between magnetic resonance imaging results and findings at surgery for cases of severe endometriosis J Minim Invasive Gynecol 201522(6S):S55. [DOI] [PubMed] [Google Scholar]
- 27.Barcellos M B, Lasmar B, Lasmar R. Agreement between the preoperative findings and the operative diagnosis in patients with deep endometriosis. Arch Gynecol Obstet. 2016;293(04):845–850. doi: 10.1007/s00404-015-3892-x. [DOI] [PubMed] [Google Scholar]
- 28.Ito T E, Abi Khalil E D, Taffel M, Moawad G N. Magnetic resonance imaging correlation to intraoperative findings of deeply infiltrative endometriosis. Fertil Steril. 2017;107(02):e11–e12. doi: 10.1016/j.fertnstert.2016.10.024. [DOI] [PubMed] [Google Scholar]


