Abstract
Background
The novel optic neuritis (ON) diagnostic criteria include intereye differences (IED) of optical coherence tomography (OCT) parameters. IED has proven valuable for ON diagnosis in multiple sclerosis but has not been evaluated in aquaporin-4 antibody seropositive neuromyelitis optica spectrum disorders (AQP4+NMOSD). We evaluated the diagnostic accuracy of intereye absolute (IEAD) and percentage difference (IEPD) in AQP4+NMOSD after unilateral ON >6 months before OCT as compared with healthy controls (HC).
Methods
Twenty-eight AQP4+NMOSD after unilateral ON (NMOSD-ON), 62 HC and 45 AQP4+NMOSD without ON history (NMOSD-NON) were recruited by 13 centres as part of the international Collaborative Retrospective Study on retinal OCT in Neuromyelitis Optica study. Mean thickness of peripapillary retinal nerve fibre layer (pRNFL) and macular ganglion cell and inner plexiform layer (GCIPL) were quantified by Spectralis spectral domain OCT. Threshold values of the ON diagnostic criteria (pRNFL: IEAD 5 µm, IEPD 5%; GCIPL: IEAD: 4 µm, IEPD: 4%) were evaluated using receiver operating characteristics and area under the curve (AUC) metrics.
Results
The discriminative power was high for NMOSD-ON versus HC for IEAD (pRNFL: AUC 0.95, specificity 82%, sensitivity 86%; GCIPL: AUC 0.93, specificity 98%, sensitivity 75%) and IEPD (pRNFL: AUC 0.96, specificity 87%, sensitivity 89%; GCIPL: AUC 0.94, specificity 96%, sensitivity 82%). The discriminative power was high/moderate for NMOSD-ON versus NMOSD-NON for IEAD (pRNFL: AUC 0.92, specificity 77%, sensitivity 86%; GCIP: AUC 0.87, specificity 85%, sensitivity 75%) and for IEPD (pRNFL: AUC 0.94, specificity 82%, sensitivity 89%; GCIP: AUC 0.88, specificity 82%, sensitivity 82%).
Conclusions
Results support the validation of the IED metrics as OCT parameters of the novel diagnostic ON criteria in AQP4+NMOSD.
Keywords: vision, neuroimmunology, neuroophthalmology
WHAT IS ALREADY KNOWN ON THIS TOPIC
The novel optic neuritis (ON) diagnostic criteria include intereye differences (IED) of neuroaxonal optical coherence tomography (OCT) parameters.
IED has proven valuable for ON diagnosis in multiple sclerosis but has not been evaluated in aquaporin-4 antibody seropositive neuromyelitis optica spectrum disorders (AQP4+NMOSD).
WHAT THIS STUDY ADDS
In this cohort study that included 73 AQP4+NMOSD patients and 62 healthy controls from 13 centres, the discriminative power of intereye differences of neuroaxonal OCT parameters was high for AQP4+NMOSD with a history of one unilateral ON event versus healthy controls (specificity ≥82%, sensitivity ≥75%) as well as versus AQP4+NMOSD without a history of ON (specificity ≥77%, sensitivity ≥75%).
HOW THIS STUDY MIGHT AFFECT RESEARCH, PRACTICE OR POLICY
OCT parameters of novel diagnostic ON criteria are applicable in AQP4+NMOSD.
Background
Long-awaited diagnostic criteria and a new classification schema for optic neuritis (ON) have recently been defined by an expert consortium.1 As part of these criteria, the increasing value of optical coherence tomography (OCT) as an accurate method for quantitative assessment of retinal neurodegeneration after ON in clinical diagnosis and monitoring has been recognised.
In addition to monocular atrophy of the peripapillary retinal nerve fibre layer (pRNFL) and of the combined ganglion cell and inner plexiform layer (GCIPL), retinal asymmetry of pRNFL and GCIPL can be used as a diagnostic tool for retinal neurodegeneration after ON. 2,3(p),4–6 In multiple sclerosis (MS), an intereye absolute difference (IEAD) of >3–5 µm and intereye percentage difference (IEPD) of >3%–5% are diagnostic for a history of unilateral ON and can reach a specificity up to 97% and sensitivity of up to 100%.2–5 Yet, outside of MS cohorts, ON associations with intereye differences have not been validated in other disorders.
Patients with aquaporin-4 antibody (AQP4-IgG) seropositive neuromyelitis optica spectrum disorder (NMOSD) frequently suffer from severe and recurrent ON episodes.7–9 Due to the relative rarity of these patients as compared with MS, diagnosis and appropriate treatment are often delayed.10 11 Furthermore, ON in AQP4-IgG seropositive NMOSD can present with an atypical clinical picture more commonly than in MS.11 12 In turn, inaccurate and/or delayed diagnoses can significantly negatively affect long-term clinical outcomes. Thus, IEAD and IEPD could be valuable additions to the diagnostic workup of ON in AQP4-IgG seropositive NMOSD. Yet, it remains unclear if the threshold values defined by the novel ON diagnostic criteria and established in MS are also applicable in AQP4-IgG seropositive NMOSD. Therefore, this study aims to evaluate the diagnostic accuracy of reported IEAD and IEPD values for AQP4-IgG seropositive NMOSD patients with one unilateral ON event (NMOSD-ON) as compared with healthy controls (HC).
Methods
Cohort design
We included AQP4-IgG seropositive NMOSD patients with a history of one unilateral ON (NMOSD-ON) >6 months before OCT measurements and two groups for comparison: (1) HC and (2) AQP4-IgG seropositive NMOSD patients without a history of ON (NMOSD-NON). All NMOSD and HC subjects were recruited as part of the international Collaborative Retrospective Study on retinal OCT in Neuromyelitis Optica (CROCTINO) study and their data were acquired by 13 international centres between 2000 and 2018 (online supplemental table 1).13–16 Inclusion criteria were: (1) OCT data acquired by Spectralis SD-OCT devices, (2) the absence of diseases potentially confounding OCT analyses (such as glaucoma, retinal surgery and ametropia >6 D) and (3) the absence of (further) ON attacks within 6 months before the examination date. Clinical data including time since disease onset, time since ON and treatment history were collected at the discretion of each centre. AQP4-IgG antibodies were detected in serum samples of all NMOSD patients by cell-based assays.17
jnnp-2022-330608supp001.pdf (293.1KB, pdf)
Data are reported according to the Enhancing the Quality and Transparency Of Health Research reporting guidelines.18
Optical coherence tomography
All OCT examinations were conducted at each centre using Spectralis SD-OCT devices (Heidelberg Eingineering, Heidelberg, Germany). pRNFL thickness was measured using a 12° or 3.5 mm diameter peripapillary ring scan. Macular GCIPL thickness was calculated using a 5 mm diameter annulus around the fovea excluding the central 1 mm diameter cylinder from a volume scan. All OCT data reading was performed at Charité Universitätsmedizin Berlin by five graders as previously described.14 All included OCT images fulfilled OSCAR-IB criteria19 20 and are presented adhering to the Apostel V.2.0 recommendations.21 22
Statistical analyses
Statistical analyses were conducted using R (V.4.2.1) (RStudio, Boston, Massachusetts, USA).23 Data were stratified in cohorts by diagnosis and history of ON. Continuous data were described as mean±SD, if not stated otherwise. IEAD was calculated as absolute difference between eyes in pRNFL or GCIPL, respectively. IEPD was calculated as IEAD divided by the higher pRNFL or GCIPL value, respectively (comparing both eyes of the patient). Age, IEAD and IEPD were compared between groups employing unpaired t-test. Significance was established at p<0.05. The level of diagnostic accuracy was evaluated using receiver operating characteristics (ROC) curves. Area under the curve (AUC) measurements were rated as low/no discriminative power (for AUC <0.7), moderate discriminative power (for AUC 0.7–0.9) and high discriminative power (for AUC >0.9).
Data availability
All data used in this project are available within the article and by reasonable request from the corresponding author.
Results
Cohort
Characteristics of the study cohort are summarised in table 1. Twenty-eight patients (56 eyes) with a diagnosis of AQP4-IgG seropositive NMOSD and a history of unilateral ON (NMOSD-ON) were compared with 62 healthy controls (HC, 124 eyes) and 45 patients with AQP4-IgG seropositive NMOSD without a history of ON (NMOSD-NON, 90 eyes). All control groups were age-matched with NMOSD-ON (HC: p=0.881, NMOSD-NON: p=0.942).
Table 1.
Demographic overview
| HC | NMOSD-NON | NMOSD-ON | |
| Subjects (n) | 62 | 45 | 28 |
| Eyes (n) | 124 | 90 | 56 |
| Patients with a disease duration <10 years (n) | . | 43 | 21 |
| Patients with ON as first manifestation (n) | . | . | 17 |
| Age (year, mean±SD) | 37.7±10.2 | 39.0±10.4 | 38.8±12.1 |
| Sex (male, n (%)) | 20 (32) | 2 (4) | 3 (11) |
| Time since ON (year, median (min−max)) | . | . | 2.8 (0.7–19.5) |
| Time since onset (year, mean±SD) | . | 3.8±4.0 | 6.5±5.6 |
| pRNFL thickness (µm, mean±SD) | 98.5±9.4 | 98.8±10.8 | 80.7±24.9 |
| pRNFL IEPD (%, mean±SD) | 2.7±2.3 | 3.7±4.4 | 28.6±19.9 |
| pRNFL IEAD (µm, mean±SD) | 2.7±2.2 | 3.8±4.5 | 27.0±19.8 |
| GCIPL thickness (µm, mean±SD) | 79.9±5.3 | 77.6±6.3 | 66.3±13.2 |
| GCIPL IEPD (%, mean±SD) | 1.3±1.1 | 2.7±3.5 | 19.0±14.3 |
| GCIPL IEAD (µm, mean±SD) | 1.0±0.9 | 2.1±2.9 | 14.1±10.9 |
GCIPL, combined ganglion cell and inner plexiform layer; HC, healthy control; IEAD, intereye absolute difference; IEPD, intereye percentage difference; NMOSD-NON, patients with aquaporin-4-antibody seropositive neuromyelitis optica spectrum disorders and no history of optic neuritis; NMOSD-ON, patients with aquaporin-4-antibody seropositive neuromyelitis optica spectrum disorders and a history of unilateral optic neuritis; ON, optic neuritis; pRNFL, peripapillary retinal nerve fibre layer.
Twenty-one NMOSD-ON (42 eyes, age: 36.2±11.5, sex (male, n (%)): 2 (10)) and 43 NMOSD-NON (86 eyes, age: 38.4±10.3, sex (male, n (%)): 2 (5)) had a disease duration <10 years. For 17 NMOSD-ON (34 eyes, age: 39.2±12.2, sex (male, n (%)): 2 (12)), the unilateral ON episode was their first disease manifestation.
Intereye percentage and absolute differences
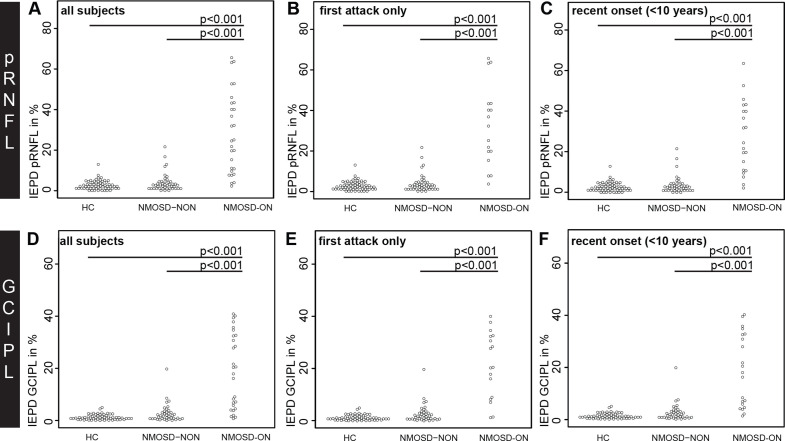
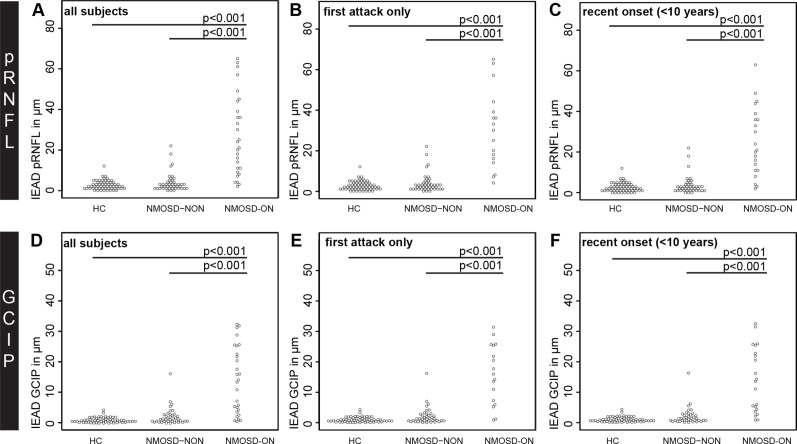
The IEAD and IEPD of pRNFL and GCIPL was higher in NMOSD-ON compared with HC and NMOSD-NON (table 2 and figures 1 and 2). This result was highly significant for both parameters (p<0.001) when comparing the whole cohort as well as subsets with (a) the unilateral ON episode as the first attack and (b) disease duration <10 years.
Table 2.
Comparison of IEAD and IEPD value for GCIPL and pRNFL
| NMOSD-ON versus HC | NMOSD-ON versus NMOSD-NON | |||
| T value | P value | T value | P value | |
| All subjects | ||||
| pRNFL IEPD (%) | −6.885 | <0.001 | −6.519 | <0.001 |
| pRNFL IEAD (µm) | −6.483 | <0.001 | −5.102 | <0.001 |
| GCIPL IEPD (%) | −6.566 | <0.001 | −6.928 | <0.001 |
| GCIPL IEAD (µm) | −6.338 | <0.001 | −5.663 | <0.001 |
| First attack only | ||||
| pRNFL IEPD (%) | −6.162 | <0.001 | −5.892 | <0.001 |
| pRNFL IEAD (µm) | −5.969 | <0.001 | −5.677 | <0.001 |
| GCIPL IEPD (%) | −6.570 | <0.001 | −6.024 | <0.001 |
| GCIPL IEAD (µm) | −6.357 | <0.001 | −5.778 | <0.001 |
| Recent onset (<10 years) | ||||
| pRNFL IEPD (%) | −6.527 | <0.001 | −6.201 | <0.001 |
| pRNFL IEAD (µm) | −6.054 | <0.001 | −5.708 | <0.001 |
| GCIPL IEPD (%) | −5.768 | <0.001 | −5.274 | <0.001 |
| GCIPL IEAD (µm) | −5.530 | <0.001 | −5.014 | <0.001 |
GCIPL, combined ganglion cell and inner plexiform layer; HC, healthy control; IEAD, intereye absolute difference; IEPD, intereye percentage difference; NMOSD-NON, patients with aquaporin-4-antibody seropositive neuromyelitis optica spectrum disorders and no history of optic neuritis; NMOSD-ON, patients with aquaporin-4-antibody seropositive neuromyelitis optica spectrum disorders and a history of unilateral optic neuritis; ON, optic neuritis; pRNFL, peripapillary retinal nerve fibre layer.
Figure 1.
IEPD comparisons by beeswarm plot for (A-C) pRNFL and (D-F) GCIPL in (A, D) all NMOSD-ON patients compared with HC and NMOSD-NON, (B, E) NMOSD-ON patients with the ON as their first attack compared with HC and NMOSD-NON and (C, F) NMOSD-ON and NMOSD-NON patients with a disease duration <10 years compared with HC. GCIPL, combined ganglion cell and inner plexiform layer; HC, healthy controls; IEPD, intereye percentage difference; NMOSD-ON, patients with aquaporin-4-antibody seropositive neuromyelitis optica spectrum disorders and a history of unilateral optic neuritis; NMOSD-NON, patients with aquaporin-4-antibody seropositive neuromyelitis optica spectrum disorders and no history of optic neuritis; pRNFL, peripapillary retina nerve fibre layer.
Figure 2.
IEAD comparisons by beeswarm plot for (A-C) pRNFL and (D-F) GCIPL in (A, D) all NMOSD-ON patients compared with HC and NMOSD-NON, (B, E) NMOSD-ON patients with the ON as their first attack compared with HC and NMOSD-NON and (C, F) NMOSD-ON and NMOSD-NON patients with a disease duration <10 years compared with HC. GCIPL, combined ganglion cell and inner plexiform layer; HC, healthy controls; IEAD, intereye absolute difference; NMOSD-ON, patients with aquaporin-4-antibody seropositive neuromyelitis optica spectrum disorders and a history of unilateral optic neuritis; NMOSD-NON, patients with aquaporin-4-antibody seropositive neuromyelitis optica spectrum disorders and no history of optic neuritis; pRNFL, peripapillary retina nerve fibre layer.
Diagnostic sensitivity and specificity of intereye differences in NMOSD
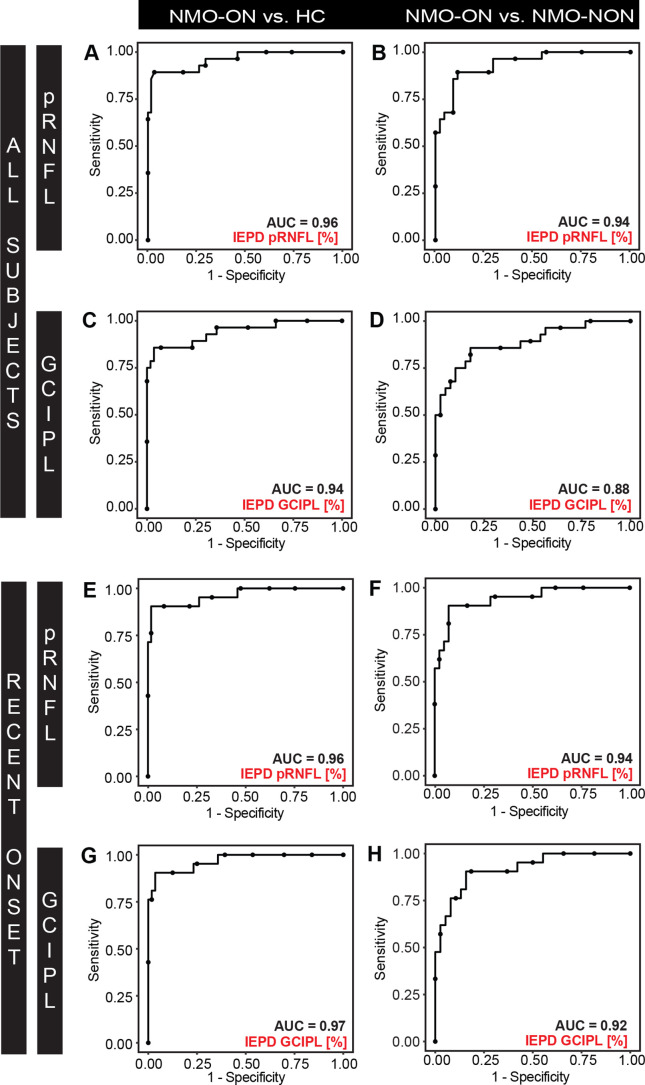
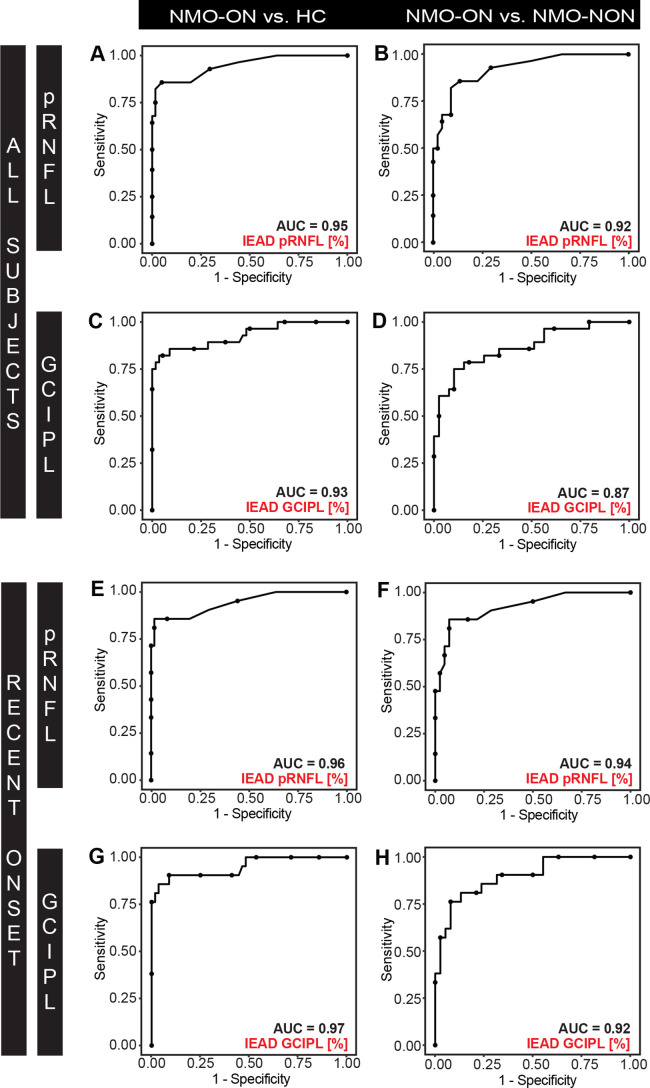
The discriminative power for IEPD and IEAD was high (AUC >0.9) for HC versus NMOSD-ON. This outcome was true for patients overall, and for patients with a disease duration <10 years (figures 3 and 4). The discriminative power for IEPD and IEAD was also high or moderate (AUC 0.8–0.9) for separating NMOSD-NON versus NMOSD-ON.
Figure 3.
ROC curves for IEPD for (A-B, E-F) pRNFL and (C-D, G-H) GCIPL discriminating between (A, C, E, G) NMOSD-ON versus HC, and (B, D, F, H) NMOSD-ON versus NMOSD-NON. ROC curves are plotted for (A-D) all subjects and (E-H) NMOSD-ON and NMOSD-NON patients with a disease duration <10 years and HC. AUC, area under the curve; GCIPL, combined ganglion cell and inner plexiform layer; HC, healthy controls; IEPD, intereye percentage difference, NMOSD-ON, patients with aquaporin-4-antibody seropositive neuromyelitis optica spectrum disorders and a history of unilateral optic neuritis; NMOSD-NON, patients with aquaporin-4-antibody seropositive neuromyelitis optica spectrum disorders and no history of optic neuritis; pRNFL, peripapillary retina nerve fibre layer; ROC, receiver operating characteristics.
Figure 4.
ROC curves for IEAD for (A-B, E-F) pRNFL and (C-D, G-H) GCIPL (A, D) discriminating between (A, C, E, G) NMOSD-ON versus HC and (B, D, F, H) NMOSD-ON versus NMOSD-NON. ROC curves are plotted for (A-D) all subjects and (E-H) NMOSD-ON and NMOSD-NON patients with a disease duration <10 years and HC. AUC, area under the curve; GCIPL, combined ganglion cell and inner plexiform layer; HC, healthy controls; IEAD, intereye absolute difference, NMOSD-ON, patients with aquaporin-4-antibody seropositive neuromyelitis optica spectrum disorders and a history of unilateral optic neuritis; NMOSD-NON, patients with aquaporin-4-antibody seropositive neuromyelitis optica spectrum disorders and no history of optic neuritis; pRNFL, peripapillary retina nerve fibre layer; ROC, receiver operating characteristics.
Evaluating previously reported threshold values (IEAD: 5 µm, IEPD: 5% for pRNFL and IEAD: 4 µm, IEPD: 4% GCIPL), NMOSD-ON could be separated with high discriminative power from HC using IEAD for pRNFL with a specificity of 82% and a sensitivity of 86% (AUC 0.95, positive predictive value (PPV) 0.68, negative predictive value (NPV) 0.93). Likewise, discriminative power was high using IEPD for pRNFL with a specificity of 87% and a sensitivity of 89% (AUC 0.96, PPV 0.76, NPV 0.95). NMOSD-ON can be equally well distinguished from HC using IEAD for GCIPL with a specificity of 98% and a sensitivity of 75% (AUC 0.93, PPV 0.94, NPV 0.90) and using IEPD for GCIPL with a specificity of 96% and a sensitivity of 82% (AUC 0.94, PPV 0.90, NPV 0.92).
NMOSD-ON can be separated with high to moderate discriminative power from NMOSD-NON using IEAD for pRNFL with a specificity of 77% and a sensitivity of 86% (AUC 0.92, PPV 0.70, NPV 0.90) and using IEPD for pRNFL with a specificity of 82% and a sensitivity of 89% (AUC 0.94, PPV 0.75, NPV 0.92). NMOSD-ON can equally well be separated from NMOSD-NON using IEAD for GCIPL with a specificity of 85% and sensitivity of 75% (AUC 0.87, PPV 0.76, NPV 0.85) and using IEPD for GCIPL with a specificity of 82% and a sensitivity of 82% (AUC 0.88, PPV 0.74, NPV 0.88). ROC results of IEAD and IEPD did not differ significantly for any discrimination (data not shown).
Discussion
The identification of prior ON episodes is a valuable step in the diagnostic workup and differential diagnosis of multiple autoimmune neuroinflammatory diseases such as AQP4-IgG seropositive NMOSD and MS.8 24 Whereas IEAD and IEPD previously proved their diagnostic value in the ON workup of patients with MS,2 3 our study demonstrates for the first time a high diagnostic accuracy of intereye difference metrics for the identification of AQP4-IgG seropositive NMOSD patients with a prior unilateral ON episode (>6 months before OCT), as compared with NMOSD patients without prior ON episodes and HC. The data also provide support for the macular GCIPL IEAD (4 µm), and IEPD (4%) thresholds as defined in the novel ON diagnostic criteria as having excellent diagnostic sensitivity and specificity for ON in AQP4-IgG seropositive NMOSD. Intereye difference metrics thereby prove to not be specific or limited to a particular demyelinating disease, but rather a reflection of the thinning of the RNFL and GCIPL that occurs after ON associated with many autoimmune neuroinflammatory diseases. The fact that these findings were consistent across an array of international centres is also encouraging with respect to uniform application of these methods and outcome interpretations with minimised centre bias globally.
The IEPD and IEAD performed equally well in our study suggesting that intereye difference metrics may not require the establishment of separate reference values for different OCT devices. Rather, it is conceivable that the IEPD threshold could be used across heterogenous devices and cohorts. Interestingly, both pRNFL and GCIPL results provided comparable assessments in this cohort, extending the application of intereye differences. Whereas pRNFL may be the faster in a standard clinical setting, GCIPL undergoes less edematous change and may therefore have special utility earlier after acute ON episodes or in clinical ON trials.25 26 The effects of recurrent or bilateral ON attacks and of chiasmic involvement on the IEAD and IEPD, as commonly seen in NMOSD, are not known. Therefore, future studies should interrogate the validity of these measurements longitudinally in NMOSD-ON.
A strength of our study rests on its size and multicentre setting since the cohort was derived from a consortium of expert NMOSD researchers in context of the international CROCTINO cohort.10 13–15 Hence, the significance of the results suggests that potential differences in use of OCT across broad geographic distances and ethnicities are overcome by the methods of acquisition and analysis used here. Yet, limitations of the current study should also be considered. First, the cohorts were not matched for gender or degree of loss of neurological function, preventing statistical evaluation. Furthermore, the prevalence of unilateral ON in our cohort is higher than expected in a real-world setting, thereby influencing PPV and NPV calculations. Also, no functional metrics such as visually evoked potentials, or MRI were included in this study, thus subclinical contralateral optic nerve involvement, which would affect IED measurements, cannot be excluded. Therefore, it remains uncertain if there is added diagnostic value of MRI and visually evoked potentials. This determination will be important for future, especially prospective, studies to further improve on diagnostic sensitivity. Nonetheless, the current findings show the astonishing value of quantitative intereye differences for ON diagnosis in AQP4-IgG seropositive NMOSD. This might be of value in making a clinical diagnosis and defining inclusion criteria for clinical trials in NMOSD.
Footnotes
Twitter: @elenahlapiscina, @NeuroVisionLab
FP and AP contributed equally.
Contributors: FCO: conceptualisation (support), data curation (support), formal analysis (lead), investigation (equal), project administration (equal), resources (equal), visualisation (lead), writing – original draft, writing – review and editing (equal); HZ: data curation (equal), formal analysis (support), project administration (equal), resources (equal), validation (lead), writing – review and editing (equal); SM: data curation (equal), project administration (equal), resources (equal), writing – review and editing (equal); CB: data curation (support), project administration (equal), resources (equal), writing – review and editing (equal), CC: data curation (equal), project administration (equal), resources (equal), writing – review and editing (equal); OA: resources (equal), writing – review and editing (equal); PA: resources (equal), writing – review and editing (equal); MR: resources (equal), writing – review and editing; AD: resources (equal), writing – review and editing (equal); LP: resources (equal), writing – review and editing (equal); EHM-L: resources (equal), writing – review and editing (equal); BS-D: resources (equal), writing – review and editing (equal); PV: resources (equal), writing – review and editing (equal); JP: resources (equal), writing – review and editing (equal); AR-F: resources (equal), writing – review and editing (equal); MIL: resources (equal), writing – review and editing (equal); SMS: resources (equal), writing – review and editing (equal); LL: resources (equal), writing – review and editing (equal); MPi: resources (equal), writing – review and editing (equal); MR: resources (equal), writing – review and editing (equal); MAL-P: resources (equal), writing – review and editing (equal); MAF: resources (equal), writing – review and editing (equal); JH: resources (equal), writing – review and editing (equal); FA: resources (equal), writing – review and editing (equal); RKa: resources (equal), writing – review and editing (equal); AD: resources (equal), writing – review and editing (equal); MPo: resources (equal), writing – review and editing (equal); RM: resources (equal), writing – review and editing (equal); AC-C: resources (equal), writing – review and editing (equal); NA: resources (equal), writing – review and editing (equal); KKS: resources (equal), writing – review and editing (equal);AJ: resources (equal), writing – review and editing (equal); SH: resources (equal), writing – review and editing (equal); YM-D: resources (equal), writing – review and editing (equal); AJG: supervision (support), writing – review and editing (equal); MRY: data curation (equal), project administration (equal), writing – review and editing (equal); TJS: data curation (equal), project administration (equal), writing – review and editing (equal); LC: data curation (equal), project administration (equal), software (lead), writing – review and editing (equal); AB: data curation (equal), funding acquisition (equal), project administration (equal), resources (equal), supervision (support), writing – review and editing (equal); FP: funding acquisition (equal), project administration (equal), resources (equal), supervision (support), writing – review and editing (equal); AP: conceptualisation (lead), formal analysis (support), methodology (lead), supervision (lead), writing – review and editing (equal).
Funding: regarding the CROCTINO project (to FP), the German Neurology Association (DGN) (to FCO) and the German Ministry for Economic Affairs and Climiate Action (to HGZ). FCO received fellowship funding from the American Academy of Neurology (AAN) and the National Multiple Sclerosis Society (NMSS).regarding the CROCTINO project (to FP), the German Neurology Association (DGN) (to FCO) and the German Ministry for Economic Affairs and Climiate Action (to HGZ). FCO received fellowship funding from the American Academy of Neurology (AAN) and the National Multiple Sclerosis Society (NMSS).
Competing interests: The authors have stated explicitly that there are no conflicts of interest in connection with this article.
Provenance and peer review: Not commissioned; externally peer reviewed.
Supplemental material: This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.
Data availability statement
Data are available on reasonable request.
Ethics statements
Patient consent for publication
Consent obtained directly from patient(s).
Ethics approval
The study was approved by local ethics committees (Charité Universitätsmedizin Berlin: EA1/097/18) and conducted in accordance with the applicable laws as well as the current version of the Declaration of Helsinki. All participants gave written informed consent.
References
- 1. Petzold A, Fraser CL, Abegg M, et al. Diagnosis and classification of optic neuritis. Lancet Neurol 2022;21:1120–34. 10.1016/S1474-4422(22)00200-9 [DOI] [PubMed] [Google Scholar]
- 2. Coric D, Balk LJ, Uitdehaag BMJ, et al. Diagnostic accuracy of optical coherence tomography inter-eye percentage difference for optic neuritis in multiple sclerosis. Eur J Neurol 2017;24:1479–84. 10.1111/ene.13443 [DOI] [PubMed] [Google Scholar]
- 3. Nolan RC, Galetta SL, Frohman TC, et al. Optimal intereye difference thresholds in retinal nerve fiber layer thickness for predicting a unilateral optic nerve lesion in multiple sclerosis. J Neuroophthalmol 2018;38:451–8. 10.1097/WNO.0000000000000629 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Bsteh G, Hegen H, Altmann P, et al. Validation of inter-eye difference thresholds in optical coherence tomography for identification of optic neuritis in multiple sclerosis. Mult Scler Relat Disord 2020;45:102403. 10.1016/j.msard.2020.102403 [DOI] [PubMed] [Google Scholar]
- 5. Petzold A, Chua SYL, Khawaja AP, et al. Retinal asymmetry in multiple sclerosis. Brain 2021;144:224–35. 10.1093/brain/awaa361 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Nolan-Kenney RC, Liu M, Akhand O, et al. Optimal intereye difference thresholds by optical coherence tomography in multiple sclerosis: an international study. Ann Neurol 2019;85:618–29. 10.1002/ana.25462 [DOI] [PubMed] [Google Scholar]
- 7. Wingerchuk DM, Banwell B, Bennett JL, et al. International consensus diagnostic criteria for neuromyelitis optica spectrum disorders. Neurology 2015;85:177–89. 10.1212/WNL.0000000000001729 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Oertel FC, Zimmermann H, Paul F, et al. Optical coherence tomography in neuromyelitis optica spectrum disorders: potential advantages for individualized monitoring of progression and therapy. EPMA J 2018;9:21–33. 10.1007/s13167-017-0123-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Jarius S, Paul F, Weinshenker BG, et al. Neuromyelitis optica. Nat Rev Dis Primers 2020;6:85. 10.1038/s41572-020-0214-9 [DOI] [PubMed] [Google Scholar]
- 10. Oertel FC, Paul F. Accelerating clinical research in neuromyelitis optica spectrum disorders. Clin Exp Neuroimmunol 2021;12:89–91. 10.1111/cen3.12637 [DOI] [Google Scholar]
- 11. Petzold A, Pittock S, Lennon V, et al. Neuromyelitis optica-IgG (aquaporin-4) autoantibodies in immune mediated optic neuritis. J Neurol Neurosurg Psychiatry 2010;81:109–11. 10.1136/jnnp.2008.146894 [DOI] [PubMed] [Google Scholar]
- 12. Zheng W, Liu X, Hou X, et al. Recurrent optic neuritis in a patient with sjogren syndrome and neuromyelitis optica spectrum disorder: a case report. Medicine (Baltimore) 2020;99:e23029. 10.1097/MD.0000000000023029 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Specovius S, Zimmermann HG, Oertel FC, et al. Cohort profile: a collaborative multicentre study of retinal optical coherence tomography in 539 patients with neuromyelitis optica spectrum disorders (CROCTINO). BMJ Open 2020;10:e035397. 10.1136/bmjopen-2019-035397 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Oertel FC, Specovius S, Zimmermann HG, et al. Retinal optical coherence tomography in neuromyelitis optica. Neurol Neuroimmunol Neuroinflamm 2021;8:e1068. 10.1212/NXI.0000000000001068 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Lu A, Zimmermann HG, Specovius S, et al. Astrocytic outer retinal layer thinning is not a feature in AQP4-igg seropositive neuromyelitis optica spectrum disorders. J Neurol Neurosurg Psychiatry 2022;93:188–95. 10.1136/jnnp-2021-327412 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Oertel FC, Sotirchos ES, Zimmermann HG, et al. Longitudinal retinal changes in MOGAD. Ann Neurol 2022;92:476–85. 10.1002/ana.26440 [DOI] [PubMed] [Google Scholar]
- 17. Waters P, Reindl M, Saiz A, et al. Multicentre comparison of a diagnostic assay: aquaporin-4 antibodies in neuromyelitis optica. J Neurol Neurosurg Psychiatry 2016;87:1005–15. 10.1136/jnnp-2015-312601 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. von Elm E, Altman DG, Egger M, et al. The strengthening the reporting of observational studies in epidemiology (STROBE) statement: guidelines for reporting observational studies. Int J Surg 2014;12:1495–9. 10.1016/j.ijsu.2014.07.013 [DOI] [PubMed] [Google Scholar]
- 19. Tewarie P, Balk L, Costello F, et al. The OSCAR-IB consensus criteria for retinal OCT quality assessment. PLoS ONE 2012;7:e34823. 10.1371/journal.pone.0034823 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Schippling S, Balk LJ, Costello F, et al. Quality control for retinal OCT in multiple sclerosis: validation of the OSCAR-IB criteria. Mult Scler 2015;21:163–70. 10.1177/1352458514538110 [DOI] [PubMed] [Google Scholar]
- 21. Cruz-Herranz A, Balk LJ, Oberwahrenbrock T, et al. The APOSTEL recommendations for reporting quantitative optical coherence tomography studies. Neurology 2016;86:2303–9. 10.1212/WNL.0000000000002774 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Aytulun A, Cruz-Herranz A, Aktas O, et al. Apostel 2.0 recommendations for reporting quantitative optical coherence tomography studies. Neurology 2021;97:68–79. 10.1212/WNL.0000000000012125 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. R Development Core Team . R: A language and environment for statistical computing. R Foundation for Statistical Computing, 2008. Available: http://www.R-project.org [Google Scholar]
- 24. Oertel FC, Zimmermann HG, Brandt AU, et al. Novel uses of retinal imaging with optical coherence tomography in multiple sclerosis. Expert Rev Neurother 2019;19:31–43. 10.1080/14737175.2019.1559051 [DOI] [PubMed] [Google Scholar]
- 25. Syc SB, Saidha S, Newsome SD, et al. Optical coherence tomography segmentation reveals ganglion cell layer pathology after optic neuritis. Brain 2012;135:521–33. 10.1093/brain/awr264 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Costello F, Pan YI, Yeh EA, et al. The temporal evolution of structural and functional measures after acute optic neuritis. J Neurol Neurosurg Psychiatry 2015;86:1369–73. 10.1136/jnnp-2014-309704 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
jnnp-2022-330608supp001.pdf (293.1KB, pdf)
Data Availability Statement
All data used in this project are available within the article and by reasonable request from the corresponding author.
Data are available on reasonable request.