Abstract
Objective
As thoracic aortic disease (TAD) is generally asymptomatic, biomarkers are needed to provide insight into early progression. We aimed to examine the association between circulating blood biomarkers and the maximal thoracic aortic diameter (TADmax).
Methods
In this cross-sectional study, consecutive adult patients with a thoracic aortic diameter ≥40 mm and/or genetically proven hereditary TAD (HTAD) visiting our specialised outpatient clinic between 2017 and 2020 were prospectively included. Venous blood sampling and CT angiography and/or transthoracic echocardiography of the aorta were performed. Linear regression analyses were performed and estimates were presented as mean difference in TADmax in mm per doubling of standardised biomarker level.
Results
In total, 158 patients were included (median age 61 (50.3–68.8) years, 37.3% female). HTAD diagnosis was confirmed in 36 of 158 (22.7%) patients. TADmax was 43.9±5.2 mm in men vs 41.9±5.1 in women (p=0.030). In unadjusted analysis, significant associations with TADmax were found for interleukin-6 (1.15 (95% CI 0.33 to 1.96), p=0.006), growth differentiation factor-15 (1.01 (95% CI 0.18 to 1.84), p=0.018), microfibrillar-associated protein 4 (MFAP4) (−0.88 (95% CI −1.71 to 0.05), p=0.039) and triiodothyronine (T3) (−2.00 (95%CI −3.01 to 0.99), p<0.001). The association of MFAP4 with TADmax was stronger in women (p for interaction=0.020) and for homocysteine, an inverse association with TADmax was observed when compared with men (p for interaction=0.008). When adjusted for age, sex, hyperlipidaemia and HTAD, total cholesterol (1.10 (95% CI 0.27 to 1.93), p=0.010) and T3 (−1.20 (95% CI −2.14 to 0.25), p=0.014) were significantly associated with TADmax.
Conclusions
Circulating biomarkers indicative of inflammation, lipid metabolism and thyroid function might be associated with TAD severity. Possible distinct biomarker patterns for men and women warrant further investigation.
Keywords: Biomarkers, Aortic Diseases, Aortic Aneurysm
WHAT IS ALREADY KNOWN ON THIS TOPIC
An enlarged aorta poses a risk of aortic complications, which are life-threatening events. In order to identify patients at high risk of these events, biomarkers could provide more information, yet data are scarce in thoracic aortic disease (TAD).
WHAT THIS STUDY ADDS
In this cross-sectional study of 158 patients with TAD, several blood biomarkers were found to be associated with the size of the thoracic aorta. The significantly associated blood biomarkers were mostly indicators of inflammation, lipid metabolism and thyroid function, and to a smaller extent, aortic wall composition.
Our findings suggest that in two biomarkers, there may be different patterns in men and women, warranting sex-stratified analysis in future biomarker studies to elucidate potentially distinct pathophysiological processes.
HOW THIS STUDY MIGHT AFFECT RESEARCH, PRACTICE OR POLICY
Blood biomarkers might provide for a low-cost method for risk stratification in patients with TAD alongside the aortic diameter. Our findings imply pathophysiological processes in patients with TAD, yet more research is warranted before clinical implementation. We encourage prospective studies following aortic dimensions with preferably multiple biomarker measurements and male/female-specific analysis.
Introduction
Thoracic aortic disease (TAD) represents a significant health burden with an estimated incidence proportion of thoracic aortic aneurysm and dissection of 7.6 and 4.6 per 100.000 annually.1 Dilation of the thoracic aorta poses a risk for aortic dissection and rupture, both life-threatening events.2 Since aortic dilatation and growth are generally asymptomatic, early detection of the disease remains challenging. Imaging of the thoracic aorta is the standard monitoring of TAD3; however, these scan protocols are expensive and may expose patients unnecessarily to radiation. In addition, aortic dissection can occur at diameters below the threshold for preventive surgery,4 so imaging is certainly not the holy grail. Blood biomarkers might reflect pathophysiological disease processes even before progression of aortic dilatation is present or timely detection of a rise in biomarkers might prevent a clinical complication. Therefore, circulating biomarkers could provide for a low-cost screening method to identify patients with TAD at high risk of rupture or dissection.5 6
Previous literature on biomarkers in aortic disease has focused mainly on abdominal aortic pathology and thoracic aortic dissection.5 6 Some general blood tests encompassing haemostatic markers, inflammatory proteins and proteolytic enzymes have been shown elevated in patients with thoracic aortic dissection.7–9 In patients with abdominal aortic aneurysm (AAA), several biomarkers associated with lipid metabolism, inflammation, proteolysis and oxidative stress were found associated with aortic size.10 Furthermore, markers of collagen and elastin metabolism might be of interest in TAD, due to the structural wall abnormalities.11
Additionally, male–female differences have been observed in cardiovascular disease blood biomarkers.12 In the existing literature on aortic disease biomarkers, little attention is given to male–female differences,5 6 while one study only included male patients.10
The aim of this single-centre cross-sectional study was to study the association between 23 selected biomarkers and the maximal thoracic aortic diameter (TADmax) in patients with TAD, with special attention to male–female differences.
Methods
Study population
Consecutive patients of our tertiary care centre visiting the specialised TAD outpatient clinic between October 2017 and January 2020 were eligible for inclusion in this cross-sectional study. In the specialised outpatient clinic, patients visiting a cardiologist and/or specialised physician assistant in light of cardiovascular family screening or surveillance for TAD were invited to participate. The following inclusion criteria were used: adult age (≥18 years) and a thoracic aortic diameter ≥40 mm and/or a genetically confirmed hereditary TAD (HTAD).
Data collection
Patient characteristics were collected from the hospital records using a standardised case report form, and were documented using a secure web-based application (GEneric Medical Survey Tracker, Erasmus Medical Center and Equipe Zorgbedrijven, latest release 2019, V.1.8.6, open source). Body surface area (BSA) was calculated using the DuBois and DuBois formula.13 Hypertension and hyperlipidaemia were defined as described in patient history and/or receiving medical treatment.
Biomarker measurement
Venous blood (6 mL) sampling was performed at the study visit and the blood samples were collected in vacuum tubes containing EDTA. The samples were centrifuged and the plasma stored at −80°C in separate parts within 2 hours after withdrawal, until batch analysis was performed.
We sought to provide an extensive explorative analysis of biomarkers which might be associated with the thoracic aortic diameter. Therefore, a selection of 23 potentially relevant biomarkers was made in consultation with multidisciplinary experts associated with aortic disease or HTAD, cardiac disease, inflammation, lipid profile, haematology, thyroid function and nephrology. All blood biomarker measurements were performed in the clinical chemistry laboratory of the Erasmus Medical Center. Laboratory analysis of the samples was performed blinded to the patients’ characteristics and study endpoint.
In the fresh blood samples, levels of the following biomarkers were determined according to clinical practice: N-terminal pro B-type natriuretic peptide, triglycerides, total cholesterol, high-density lipoprotein (HDL), low-density lipoprotein (LDL), total cholesterol/HDL, homocysteine, haemoglobin, red cell distribution width, thyroid-stimulating hormone, free thyroxine, thyroxine, triiodothyronine (T3), creatinine, estimated glomerular filtration rate and urea. Levels of interleukin-6 (IL-6), high-sensitivity C reactive peptide (hsCRP), high-sensitivity troponin T (hsTNT) and growth differentiation factor-15 (GDF-15) were measured in our clinical chemistry laboratory on a cobas analyser (Roche Diagnostics, Rotkreuz, Switzerland) in batch analysis for the purpose of this study. Newer biomarkers included microfibrillar-associated protein 4 (MFAP4), human fibrillin-1 (FBN1) and procollagen III N-terminal propeptide (PIIINP), which were obtained using commercially available electrochemiluminescence immunoassays.
For analytical purposes, in case the lower limit of a blood biomarker was reached, it was substituted with a value half of the lower limit, (eg, a value of <0.16 was coded as 0.08 for the biomarker considered). In case the upper detection limit was reached, the value of the upper limit was assigned (eg, a value of >10 was coded as 10). Lower limits of hsTnT, IL-6, hsCRP and MFAP4 were 3 ng/L, 2 pg/mL, 0.2 mg/L and 0.16 ng/mL, respectively. For hsCRP and MFAP4, there were upper limits of detection: 20 mg/L and 10 ng/mL.
Imaging
The endpoint of this study was the TADmax, measured at the same visit of the blood sampling. The thoracic aortic dimensions were obtained using contrast-enhanced and ECG-gated CT imaging or transthoracic echocardiography (TTE), once no CT scan was performed on the date of blood sampling. A radiologist of the Erasmus Medical Center, who was blinded to the biomarker results, performed thoracic aortic measurements according to a standardised protocol. Diameters of the sinuses of Valsalva were measured from the cusp to commissure; ascending aorta and descending aorta were measured in two directions at the level of the pulmonary bifurcation using the double-oblique method in a reconstruction, perpendicular to the vessel axis. Aortic measurements on TTE were done using the parasternal long-axis view during late diastole with the leading edge to leading edge method. For the linear regression analysis, the TADmax was used. For descriptive purposes, the maximal aortic sized index (mm/m2) was calculated: TADmax (mm) divided by BSA (m2).
Statistical analysis
Patient characteristics, aortic diameters and blood biomarker levels were reported stratified by sex. Data distribution of continuous variables was checked visually with the use of density plots and formally tested with the Shapiro-Wilk test. Normally distributed continuous data were presented as the mean and SD and compared between men and women using the Student’s t-test. If continuous data were skewed, the median and 25th–75th percentile were presented, and data for men and women were compared with the Mann-Whitney U test. Categorical data were presented as percentages or frequencies, and the Χ2 test or Fisher’s exact test was used comparing men with women, as appropriate.
Linear regression analyses were performed to study the association between the biomarkers and the study endpoint. A logarithmic transformation and standardisation was performed for all blood biomarkers; therefore, all linear regression estimates show the mean difference in TADmax per doubling of the standardised biomarker value.
First, univariable analyses were performed with blood biomarkers as independent variables and the TADmax as dependent variable. Also, the interaction between each biomarker and sex associated with the study endpoint was assessed, and if significant, effect plots were constructed. Second, biomarker values were adjusted for age and sex, and third, biomarker values were adjusted for a model with clinically relevant patient characteristics, as described in the next section.
A multivariable model was constructed with all patient characteristics mentioned in table 1. The variables with a p value of <0.10 on univariable analysis and limited collinearity (Pearson correlation coefficient <0.7) were entered a backward selection process, applying a threshold p value of 0.05. Age and sex were forced in the final model. Each biomarker was then added to this model to examine its additive value.
Table 1.
Patient characteristics stratified by sex
| All patients (n=158) | Men | Women | P value | Missing % | |
| (n=99) | (n=59) | ||||
| Patient demographics | |||||
| Age, years (median (IQR)) | 61.0 (50.3–68.8) | 62.0 (52.0–69.0) | 57.0 (47.5–66.0) | 0.257 | 0 |
| Height, m (median (IQR)) | 1.79 (1.72–1.87) | 1.85 (1.79–1.90) | 1.70 (1.66–1.75) | <0.001 | 0 |
| Weight, kg (median (IQR)) | 86.0 (76.0–96.0) | 91.0 (83.3–100.0) | 73.0 (65.5–84.5) | <0.001 | 0.6 |
| BSA (mean (SD)) | 2.05 (0.22) | 2.15 (0.19) | 1.87 (0.16) | <0.001 | 0.6 |
| Systolic blood pressure (mm Hg) (median (IQR)) | 136 (127–147) | 137 (129–148) | 135 (123–146) | 0.429 | 0.6 |
| Diastolic blood pressure (mm Hg) (mean (SD)) | 86 (12) | 87 (11) | 84 (12) | 0.18 | 0.6 |
| Hypertension | 91 (57.6) | 60 (60.6) | 31 (52.5) | 0.368 | 0.6 |
| Hyperlipidaemia | 51 (32.5) | 40 (40.4) | 11 (19.0) | 0.010 | 0.6 |
| Active smoking | 15 (12.9) | 7 (9.6) | 8 (18.6) | 0.266 | 26.6 |
| Diabetes | 6 (3.8) | 5 (5.1) | 1 (1.7) | 0.412* | 0 |
| CVA | 11 (7.0) | 7 (7.1) | 4 (6.8) | 1.000* | 0 |
| COPD | 19 (12.0) | 6 (6.1) | 13 (22.0) | 0.006 | 0 |
| BAV | 6 (3.8) | 6 (6.1) | 0 (0.0) | 0.085* | 0 |
| Renal dysfunction | 5 (3.2) | 3 (3.0) | 2 (3.4) | 1.000* | 0 |
| LVEF† | 0.686* | 34.8 | |||
| Normal | 91 (88.3) | 62 (89.9) | 29 (85.3) | ||
| Slightly reduced | 10 (9.7) | 5 (7.2) | 5 (14.7) | ||
| Moderately reduced | 1 (1.0) | 1 (1.4) | 0 (0.0) | ||
| Severely reduced | 1 (1.0) | 1 (1.4) | 0 (0.0) | ||
| Medication | 0 | ||||
| Beta blocker use | 55 (34.8) | 40 (40.4) | 15 (25.4) | 0.082 | |
| ARB use | 27 (17.1) | 20 (20.2) | 7 (11.9) | 0.259 | |
| ACEi use | 34 (21.5) | 26 (26.3) | 8 (13.6) | 0.093 | |
| Diuretics | 27 (17.1) | 17 (17.2) | 10 (16.9) | 1.000 | |
| Cholesterol | 46 (29.1) | 35 (35.4) | 11 (18.6) | 0.040 | |
| Antithrombotics | 63 (39.9) | 48 (48.5) | 15 (25.4) | 0.007 | |
| Hereditary TAD diagnosis | 0.091* | 0 | |||
| Loeys-Dietz syndrome | 18 (11.4) | 7 (7.1) | 11 (18.6) | ||
| Marfan syndrome | 11 (7.0) | 9 (9.1) | 2 (3.4) | ||
| Ehlers-Danlos syndrome | 3 (1.9) | 1 (1.0) | 2 (3.4) | ||
| Other | 4 (2.5) | 3 (3.0) | 1 (1.7) | ||
| Positive family history of aortic disease | 66 (41.8) | 39 (39.8) | 27 (45.8) | 0.571 | 0.6 |
| Previous aortic surgery | 30 (19.0) | 20 (20.2) | 10 (16.9) | 0.768 | 0 |
| Previous aortic dissection | 17 (10.8) | 9 (9.1) | 8 (13.6) | 0.541 | 0 |
| Abdominal aortic aneurysm | 11 (7.0) | 9 (9.1) | 2 (3.4) | 0.212* | 0 |
| Peripheral aneurysm | 18 (11.4) | 9 (9.1) | 9 (15.3) | 0.357 | 0 |
| Aortic diameters | |||||
| Maximal diameter thoracic aorta (mm) | 43.2±5.2 | 43.9±5.2 | 41.9±5.1 | 0.030 | 3.8 |
| Indexed diameter thoracic aorta (mm/BSA) | 21.2±2.9 | 20.5±2.9 | 22.5±3.5 | <0.001 | 4.4 |
Normally distributed continuous variables are expressed as mean±SD, skewed continuous variables are expressed as median and 25th–75th percentile, and categorical values are expressed as percentages.
P values of <0.05 are depicted in bold.
*Fisher’s exact test.
†For patients who underwent transthoracic echocardiography.
ACEi, ACE inhibitor; ARB, angiotensin receptor blocker; BAV, bicuspid aortic valve; BSA, body surface area; COPD, chronic obstructive pulmonary disease; CVA, cerebrovascular accident; LVEF, left ventricular ejection fraction; TAD, thoracic aortic disease.
The missing data pattern of patient characteristics was assumed to be missing at random and missing values were imputed using single imputation and 10 iterations. Patient characteristics were imputed up to a 15% rate of missing values. Biomarker values were not imputed and a complete case analysis was performed.
For the univariable analyses, three sensitivity analyses were performed: (1) excluding patients with prior aortic dissection or aortic surgery; (2) excluding patients with HTAD diagnosis; (3) including patients with measurements on CT only.
Two-sided p values below 0.05 were considered statistically significant. The data analysis was performed with statistical and computing program R (R Foundation for Statistical Computing, Vienna, Austria, V.4.1.2) and data imputation was performed using SPSS statistics (IBM SPSS Statistics V.28.0).
Results
Patient characteristics
In total, the study population comprised of 158 patients; all patient characteristics and the TADmax are shown in table 1. The flow chart of the patient selection is shown in figure 1. The median age was 61 years (IQR: 50–69), and 59 of 158 (37%) patients were female.
Figure 1.
Flow chart of patient selection. *Reasons for exclusion were not showing up for venous blood sampling or other logistical difficulties to the clinical chemistry laboratory.
CT imaging was performed in 106 of 158 patients (67%) and TTE was used in the remaining 52 patients to obtain the TADmax. All aorta measurements performed at CT or TTE combined at different levels in the thoracic aorta are shown in online supplemental appendix I.
openhrt-2023-002317supp001.pdf (354.1KB, pdf)
Circulating biomarker measurements were shown for all patients and stratified by sex in online supplemental appendix II.
Associations with aortic diameter
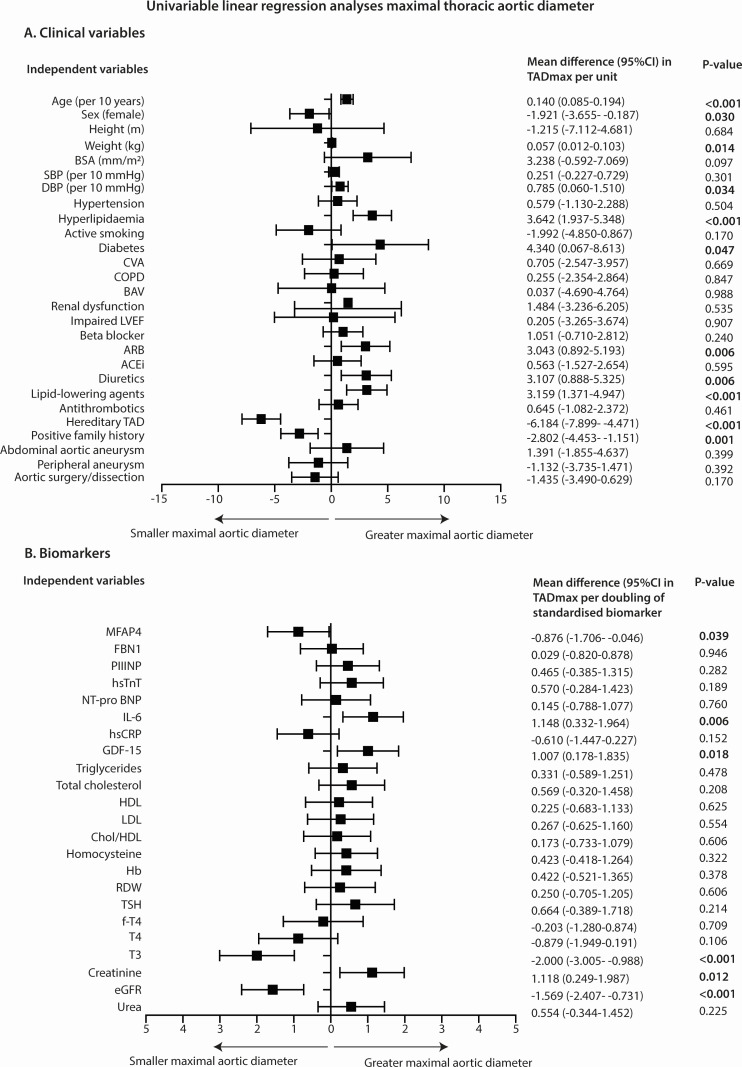
The univariable analysis of clinical variables and biomarkers with TADmax is shown in figure 2.
Figure 2.
Univariable linear regression analyses with the TADmax as dependent variable and clinical variables (A) and biomarkers (B) as independent variables. Estimates and corresponding 95% CI are derived from linear regression analysis with log2-transformed and standardised biomarkers as independent variables, that is, doubling of the standardised MFAP4 value results in a decrease of 0.876 mm in TADmax. P values of <0.05 are depicted in bold. ACEi, ACE inhibitor; ARB, angiotensin receptor blocker; BAV, bicuspid aortic valve; BSA, body surface area; COPD, chronic obstructive pulmonary disease; CVA, cerebrovascular accident; DBP, diastolic blood pressure; eGFR, estimated glomerular filtration rate; FBN1, human fibrillin-1; fT4, free thyroxine; GDF-15, growth differentiation factor-15; Hb, haemoglobin; HDL, high-density lipoprotein; hsCRP, high-sensitivity C reactive protein; hsTnT, high-sensitivity troponin T; IL-6, interleukin-6; LDL, low-density lipoprotein; LVEF, left ventricular ejection fraction; MFAP4, microfibrillar-associated protein 4; NT-proBNP, N-terminal pro B-type natriuretic peptide; PIIINP, procollagen III N-terminal propeptide; RDW, red cell distribution width; SBP, systolic blood pressure; T3, triiodothyronine; T4, thyroxine; TAD, thoracic aortic disease; TADmax, maximal thoracic aortic diameter; TSH, thyroid-stimulating hormone.
Significant interactions with sex were observed for MFAP4 (p=0.020) and homocysteine (p=0.008): the effect of MFAP4 is amplified in women and for homocysteine, a positive association with TADmax is shown in women, whereas a negative association was observed in men, as shown in the effect plots in online supplemental appendix III. The mean difference (95% CI) in TADmax per doubling of the standardised biomarker for MFAP4 was −2.373 (–3.691 to –1.054), p<0.001) for women and −0.325 (–1.358 to 0.708), p=0.534) for men. For homocysteine, the mean difference (95% CI) in TADmax per doubling of the standardised biomarker was 1.687 (0.432 to 2.942), p=0.009) for women and −0.594 (–1.685 to 0.497), p=0.283) for men.
For each biomarker, the unadjusted and adjusted estimates are shown in table 2. When adjusted for the clinical model, a positive association was observed for total cholesterol and T3 was negatively associated with the TADmax (table 2). The univariable analysis and multivariable analysis for construction of the multivariable clinical model are shown in online supplemental appendix IV.
Table 2.
Unadjusted and adjusted linear regression analyses between circulating biomarkers and TADmax (n=158)
| Biomarker | Unadjusted analysis | Adjusted analysis: age and sex | Adjusted analysis: clinical model* | |||
| Mean difference (95% CI) in TADmax per doubling of the standardised biomarker | P value | Mean difference (95% CI) in TADmax per doubling of the standardised biomarker | P value | Mean difference (95% CI) in TADmax per doubling of the standardised biomarker | P value | |
| MFAP4 | −0.876 (−1.706 to −0.046) | 0.039 | −0.698 (−1.476 to 0.081) | 0.079 | −0.421 (−1.144 to 0.301) | 0.251 |
| FBN1 | 0.029 (−0.820 to 0.878) | 0.946 | −0.327 (−1.129 to 0.475) | 0.422 | −0.208 (−0.943 to 0.528) | 0.577 |
| PIIINP | 0.465 (−0.385 to 1.315) | 0.282 | 0.475 (−0.317 to 1.267) | 0.238 | 0.284 (−0.446 to 1.013) | 0.443 |
| hsTnT | 0.570 (−0.284 to 1.423) | 0.189 | −1.099 (−2.056 to −0.143) | 0.025 | −0.500 (−1.426 to 0.428) | 0.289 |
| NT-proBNP | 0.145 (−0.788 to 1.077) | 0.760 | −0.514 (−1.422 to 0.393) | 0.264 | −0.127 (−0.981 to 0.726) | 0.768 |
| IL-6 | 1.148 (0.332 to 1.964) | 0.006 | 0.635 (−0.158 to 1.427) | 0.116 | 0.482 (−0.252 to 1.217) | 0.197 |
| hsCRP | −0.610 (−1.447 to 0.227) | 0.152 | −0.526 (−1.299 to 0.248) | 0.181 | −0.218 (−0.938 to 0.502) | 0.550 |
| GDF-15 | 1.007 (0.178 to 1.835) | 0.018 | 0.022 (−0.876 to 0.920) | 0.962 | 0.184 (−0.642 to 1.010) | 0.661 |
| Triglycerides | 0.331 (−0.589 to 1.251) | 0.478 | 0.363 (−0.496 to 1.221) | 0.405 | 0.279 (−0.511 to 1.068) | 0.486 |
| Total cholesterol | 0.569 (−0.320 to 1.458) | 0.208 | 0.802 (−0.020 to 1.624) | 0.056 | 1.098 (0.269 to 1.928) | 0.010 |
| HDL | 0.225 (−0.683 to 1.133) | 0.625 | 0.458 (−0.451 to 1.367) | 0.321 | 0.337 (−0.504 to 1.177) | 0.429 |
| LDL | 0.267 (−0.625 to 1.160) | 0.554 | 0.440 (−0.378 to 1.258) | 0.290 | 0.767 (−0.054 to 1.589) | 0.067 |
| Chol/HDL | 0.173 (−0.733 to 1.079) | 0.706 | 0.180 (−0.671 to 1.030) | 0.677 | 0.346 (−0.451 to 1.143) | 0.392 |
| Homocysteine | 0.423 (−0.418 to 1.264) | 0.322 | −0.075 (−0.869 to 0.719) | 0.853 | 0.184 (−0.546 to 0.915) | 0.619 |
| Haemoglobin | 0.422 (−0.521 to 1.365) | 0.378 | 0.568 (−0.429 to 1.565) | 0.262 | 0.478 (−0.438 to 1.394) | 0.304 |
| RDW | 0.250 (−0.705 to 1.205) | 0.606 | −0.396 (−1.304 to 0.512) | 0.390 | −0.429 (−1.261 to 0.403) | 0.310 |
| TSH | 0.664 (−0.389 to 1.718) | 0.214 | 0.619 (−0.370 to 1.607) | 0.217 | 0.544 (−0.336 to 1.423) | 0.223 |
| fT4 | −0.203 (−1.280 to 0.874) | 0.709 | −0.197 (−1.207 to 0.814) | 0.700 | −0.070 (0.968 to 0.829) | 0.878 |
| T4 | −0.879 (−1.949 to 0.191) | 0.106 | −0.496 (−1.519 to 0.526) | 0.337 | −0.015 (−0.927 to 0.897) | 0.974 |
| T3 | −2.000 (−3.005 to −0.988) | <0.001 | −1.740 (−2.751 to −0.729) | <0.001 | −1.196 (−2.143 to −0.249) | 0.014 |
| Creatinine | 1.118 (0.249 to 1.987) | 0.012 | 0.158 (−0l831 to 1.147) | 0.752 | 0.191 (−0.720 to 1.102) | 0.679 |
| eGFR | −1.569 (−2.407 to −0.731) | <0.001 | −0.322 (−1.350 to 0.706) | 0.537 | −0.472 (−1.417 to 0.473) | 0.325 |
| Urea | 0.554 (−0.344 to 1.452) | 0.225 | −0.498 (−1.400 to 0.401) | 0.276 | −0.311 (−1.152 to 0.530) | 0.466 |
Estimates and corresponding 95% CI are derived from linear regression analysis with log2-transformed and standardised biomarkers as independent variables, that is, doubling of the standardised MFAP4 value results in a decrease of 0.876 mm in TADmax.
P values of <0.05 are depicted in bold.
*Adjusted for age, sex, hyperlipidaemia and hereditary thoracic aortic disease diagnosis.
eGFR, estimated glomerular filtration rate; FBN1, human fibrillin-1; fT4, free thyroxine; GDF-15, growth differentiation factor-15; HDL, high-density lipoprotein; hsCRP, high-sensitivity C reactive protein; hsTnT, high-sensitivity troponin T; IL-6, interleukin-6; LDL, low-density lipoprotein; MFAP4, microfibrillar-associated protein 4; NT-proBNP, N-terminal pro B-type natriuretic peptide; PIIINP, procollagen III N-terminal propeptide; RDW, red cell distribution width; T3, triiodothyronine; T4, thyroxine; TADmax, maximal thoracic aortic diameter; TSH, thyroid-stimulating hormone.
Sensitivity analyses of the univariable analysis and the study endpoint are reported in online supplemental appendix V. When excluding patients with prior aortic dissection or aortic surgery (n=120), significant associations with TADmax were observed for hsCRP and T3 (mean difference in TADmax per doubling of standardised biomarker: −1.021 (95% CI −1.800 to −0.243), p=0.011 and −1.407 (95% CI −2.560 to −0.255), p=0.017); see online supplemental appendix Va). As depicted in online supplemental appendix Vb, in the study population excluding patients with HTAD (n=122), IL-6 was significantly associated with TADmax (mean difference in TADmax per doubling of standardised biomarker: 0.950 (95% CI 0.186 to 1.713), p=0.015). In the sensitivity analysis including patients with measurements on CT only (online supplemental appendix Vc), no substantial differences were observed when compared with the main analysis.
Discussion
In this cross-sectional study investigating patients with TAD, we investigated a wide set of 23 biomarkers, allowing for exploratory analysis. IL-6 and GDF-15 were significantly associated with greater TADmax, whereas MFAP4 and T3 were associated with smaller TADmax. After adjusting for a model with important clinical variables, a positive association was observed for total cholesterol levels, and T3 was negatively associated with TADmax. Interaction with sex was present for the association with biomarkers and the TADmax for MFAP4 and homocysteine, depicting an amplified negative association with MFAP4 in women and reversed associations for homocysteine.
Inflammation
Our study showed significant associations between IL-6 and GDF-15 and a greater TADmax in univariable analysis. Both IL-6 and GDF-15 are considered general inflammatory markers. IL-6 is a cytokine secreted by various cell types in the cardiovascular system, which plays a central role in the downstream inflammatory reaction enhancing atherosclerosis.14 15 Interestingly, genetic variants in the IL-6 receptor were associated with reduced risk of aortic aneurysm in a phenome-wide association study.15 Therefore, IL-6 might be a valuable marker for disease progression in TAD, and the found relationship warrants further investigation.
GDF-15 is part of the transforming growth factor β cytokine superfamily, enacting in cell growth and differentiation processes.16 In patients with AAA, GDF-15 in combination with cystatin B was the optimal diagnostic measure and GDF-15 was associated with greater diameters.10 However, the exact mechanism of GDF-15 in aneurysm formation is unclear. In pulmonary hypertension, GDF-15 is thought to play a role in vascular endothelial remodelling.17 GDF-15 could also be a reflection of concomitant disease in our cohort; around 60% of the patients were known with hypertension and 33% with hyperlipidaemia. In a general population free of cardiovascular disease, GDF-15 was associated with cardiovascular risk factors such as smoking, diabetes and antihypertensive treatment.18 Although GDF-15 is not a specific marker for aortic disease, it seems a promising marker for underlying cardiovascular disease processes, which might help identify patients at high risk of progression of aortic disease.
Thyroid function
To our knowledge, this is the first study that assesses the association with thyroid hormones and thoracic aortic diameter in TAD: lower levels of T3 were associated with greater aortic diameters on univariable analysis, and also after adjustment for important clinical factors. The connection between hypothyroidism and cardiovascular disease has been described previously. Hypothyroidism is thought to affect cardiovascular haemodynamics through several molecular mechanisms, resulting in a decreased cardiac output and arterial compliance, whereas the systemic vascular resistance and diastolic pressure are increased. In addition, hypothyroidism might affect the lipid metabolism, increasing the levels of total cholesterol and LDL.19
For aortic disease specifically, some cases have been described in the early 1970s of associations between aortic dissection and autoimmune thyroiditis20 and AAA and hypothyroidism.21 Furthermore, Wang et al showed that preoperative subclinical hypothyroidism was associated with mortality after surgery for acute type A aortic dissection.22 In women with Turner syndrome, treatment for thyroiditis was associated with thoracic aortic dilatation.23 Possibly, the combination of immune complex deposition due to the autoimmune nature of hypothyroidism and its role in atherosclerosis increases the risk of aortic complications.
Lipid metabolism
Total cholesterol levels were associated with greater TADmax when adjusting for sex, age, hyperlipidaemia and HTAD in this study. In univariable analysis, the association was not significant (p=0.208), possibly due to confounding. In population-based studies, lipid levels were associated with the risk of thoracic aortic aneurysm mortality.24 Also in AAA, associations were observed between lipid levels and aortic dimensions.10 Besides tackling hypertension in TAD,25 lipid metabolism might also play an important role in thoracic aneurysm formation, warranting accurate preventive medication. The pathophysiological mechanism for TAD is generally thought to be more genetic than inflammatory and atherosclerotic, when compared with AAA.26 However, treatment with statins might reduce thoracic aortic aneurysm growth rate and progression to dissection, rupture or death.27 28
Aortic wall composition
Unfortunately, in our cohort, we have not observed associations between aortic wall-specific markers homocysteine, FBN1, PIIINP and TADmax. We did observe an inverse association between MFAP4 and TADmax. Possibly, this reflects the pattern of HTAD with smaller aortic diameters at earlier diagnosis included in the study. In the sensitivity analysis excluding patients with HTAD, the association was diminished (online supplemental appendix IIIb) and HTAD was also associated with smaller aortic diameters. MFAP4 is a protein associated with remodelling of the extracellular matrix, which is part of the fibrinogen-related protein superfamily. It plays a role in the innate immunity, cardiovascular system and endothelial functioning.29 In proteome analysis, MFAP4 was found associated with aortic aneurysms in patients with Marfan syndrome.30 Therefore, we argue that MFAP4 might influence the pathophysiology of HTAD in a greater degree than degenerative TAD.
Male–female differences
Literature on male–female differences in biomarkers for aortic disease is scarce, probably due to the low prevalence of aortic disease among women in comparison with men.1 In a study on circulating biomarkers in cardiovascular disease, several adipokines and inflammatory markers were higher in women, while fibrotic and platelet markers were higher in men.12 In our study, significant interactions with sex and homocysteine and MFAP4 were observed: showing a stronger negative association with MFAP4 in women and reversed associations for homocysteine. The findings might be explained by distinct aetiology, as HTAD seemed more common in women than men (27% vs 20%, p=0.091), and some differences in comorbidities were observed. These results might imply different disease processes in men and women; however, the small study population (99 men and 59 women, respectively) should be kept in mind when interpreting these results.
Limitations
Some limitations to this study need to be addressed. The study population is relatively small for investigation of patterns of different biomarkers. Furthermore, the study population might not be representable as patients were included in a tertiary centre, with possibly a higher proportion of inherited TAD. However, we believe the prospective inclusion of a homogeneous patient population at the specialised TAD outpatient clinic is a major strength of this study. Due to the multiple tests performed in the analyses, type I errors might be present. As it was an exploratory analysis in a relatively small study population, we decided not to correct for multiple testing.
Conclusions
Several circulating biomarkers associated with disease severity in TAD were identified in this study. The associated markers were mostly indicators of inflammation, lipid metabolism and thyroid function, and to a lesser extent, aortic wall composition. Especially in diseases with considerable heterogeneity and comorbidities, non-tissue-specific biomarkers might provide more insights into prognosis. Further investigation of circulating biomarkers in prospective studies with multiple biomarker and aortic diameter measurements focusing on clinical endpoints will help clarify the association between biomarkers and aortic pathology. Different associations might be present for men and women; therefore, we encourage male–female stratification in future biomarker studies to unravel potentially distinct pathophysiological processes.
Acknowledgments
The analytical contribution of the medical laboratory analyst Mrs Barry Koelewijn in performing the ELISAs has been gratefully appreciated.
Footnotes
Contributors: Study design—FM, CGET, LRB, EB, JJMT and JWR-H. Data collection—FM, CGET, SD, LRB and YBdR. Data analysis—FM. Data interpretation—all authors. Manuscript draft—FM. Critical revision, editing and approval of the final manuscript—all authors, Guarantor: JWR-H.
Funding: This study was supported by ZonMw (project number: 84920001). Roche diagnostics provided hsCRP, GDF-15, hsTnT and IL-6 assays free of charge for this investigator-initiated study.
Competing interests: None declared.
Provenance and peer review: Not commissioned; externally peer reviewed.
Supplemental material: This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.
Data availability statement
Data are available upon reasonable request.
Ethics statements
Patient consent for publication
Obtained.
Ethics approval
This study involves human participants and approval was obtained from the Erasmus MC ethics committee (MEC-2017-057). The study was conducted according to local and international Good Clinical Practice guidelines. All participating patients provided written informed consent.
References
- 1.McClure RS, Brogly SB, Lajkosz K, et al. Epidemiology and management of thoracic aortic dissections and thoracic aortic aneurysms in Ontario, Canada: a population-based study. J Thorac Cardiovasc Surg 2018;155:2254–64. 10.1016/j.jtcvs.2017.11.105 [DOI] [PubMed] [Google Scholar]
- 2.Evangelista A, Isselbacher EM, Bossone E, et al. Insights from the International Registry of acute aortic dissection: a 20-year experience of collaborative clinical research. Circulation 2018;137:1846–60. 10.1161/CIRCULATIONAHA.117.031264 [DOI] [PubMed] [Google Scholar]
- 3.Erbel R, Aboyans V, Boileau C, et al. 2014 ESC guidelines on the diagnosis and treatment of aortic diseases: document covering acute and chronic aortic diseases of the thoracic and abdominal aorta of the adult. The task force for the diagnosis and treatment of aortic diseases of the European society of cardiology (ESC). Eur Heart J 2014;35:2873–926. 10.1093/eurheartj/ehu281 [DOI] [PubMed] [Google Scholar]
- 4.Pape LA, Tsai TT, Isselbacher EM, et al. Aortic diameter >or = 5.5 cm is not a good predictor of type A aortic dissection: observations from the International Registry of acute aortic dissection (IRAD). Circulation 2007;116:1120–7. 10.1161/CIRCULATIONAHA.107.702720 [DOI] [PubMed] [Google Scholar]
- 5.van Bogerijen GHW, Tolenaar JL, Grassi V, et al. Biomarkers in TAA-the Holy Grail. Prog Cardiovasc Dis 2013;56:109–15. 10.1016/j.pcad.2013.05.004 [DOI] [PubMed] [Google Scholar]
- 6.Balmforth D, Harky A, Adams B, et al. Is there a role for biomarkers in thoracic aortic aneurysm disease Gen Thorac Cardiovasc Surg 2019;67:12–9. 10.1007/s11748-017-0855-0 [DOI] [PubMed] [Google Scholar]
- 7.Suzuki T, Distante A, Zizza A, et al. Diagnosis of acute aortic dissection by D-Dimer: the International registry of acute aortic dissection substudy on biomarkers (IRAD-bio) experience. Circulation 2009;119:2702–7. 10.1161/CIRCULATIONAHA.108.833004 [DOI] [PubMed] [Google Scholar]
- 8.Kuehl H, Eggebrecht H, Boes T, et al. Detection of inflammation in patients with acute aortic syndrome: comparison of FDG-PET/CT imaging and serological markers of inflammation. Heart 2008;94:1472–7. 10.1136/hrt.2007.127282 [DOI] [PubMed] [Google Scholar]
- 9.Sangiorgi G, Trimarchi S, Mauriello A, et al. Plasma levels of metalloproteinases-9 and -2 in the acute and subacute phases of type A and type B aortic dissection. J Cardiovasc Med (Hagerstown) 2006;7:307–15. 10.2459/01.JCM.0000223251.26988.c5 [DOI] [PubMed] [Google Scholar]
- 10.Memon AA, Zarrouk M, Ågren-Witteschus S, et al. Identification of novel diagnostic and prognostic biomarkers for abdominal aortic aneurysm. Eur J Prev Cardiol 2020;27:132–42. 10.1177/2047487319873062 [DOI] [PubMed] [Google Scholar]
- 11.Iliopoulos DC, Kritharis EP, Giagini AT, et al. Ascending thoracic aortic aneurysms are associated with compositional remodeling and vessel stiffening but not weakening in age-matched subjects. J Thorac Cardiovasc Surg 2009;137:101–9. 10.1016/j.jtcvs.2008.07.023 [DOI] [PubMed] [Google Scholar]
- 12.Lau ES, Paniagua SM, Guseh JS, et al. Sex differences in circulating biomarkers of cardiovascular disease. J Am Coll Cardiol 2019;74:1543–53. 10.1016/j.jacc.2019.06.077 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Du Bois D, Du Bois EF. A formula to estimate the approximate surface area if height and weight be known. 1916. Nutrition 1989;5:303–11. [PubMed] [Google Scholar]
- 14.Hartman J, Frishman WH. Inflammation and atherosclerosis: a review of the role of Interleukin-6 in the development of atherosclerosis and the potential for targeted drug therapy. Cardiol Rev 2014;22:147–51. 10.1097/CRD.0000000000000021 [DOI] [PubMed] [Google Scholar]
- 15.Ridker PM, Rane M. Interleukin-6 signaling and anti-Interleukin-6 therapeutics in cardiovascular disease. Circ Res 2021;128:1728–46. 10.1161/CIRCRESAHA.121.319077 [DOI] [PubMed] [Google Scholar]
- 16.Paralkar VM, Vail AL, Grasser WA, et al. Cloning and characterization of a novel member of the transforming growth factor-beta/bone morphogenetic protein family. J Biol Chem 1998;273:13760–7. 10.1074/jbc.273.22.13760 [DOI] [PubMed] [Google Scholar]
- 17.Geenen LW, Baggen VJM, Kauling RM, et al. Growth differentiation factor-15 as candidate predictor for mortality in adults with pulmonary hypertension. Heart 2020;106:467–73. 10.1136/heartjnl-2019-315111 [DOI] [PubMed] [Google Scholar]
- 18.Ho JE, Mahajan A, Chen M-H, et al. Clinical and genetic correlates of growth differentiation factor 15 in the community. Clin Chem 2012;58:1582–91. 10.1373/clinchem.2012.190322 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Klein I, Danzi S. Thyroid disease and the heart. Circulation 2007;116:1725–35. 10.1161/CIRCULATIONAHA.106.678326 [DOI] [PubMed] [Google Scholar]
- 20.Hilton AM, Whittaker RS. Dissecting aneurysm and autoimmune thyroiditis. Br Med J 1972;3:827. 10.1136/bmj.3.5830.827-b [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Niarchos AP, Finn R. Letter: association between hypothyroidism and abdominal aneurysm. Br Med J 1973;4:110. 10.1136/bmj.4.5884.110-b [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Wang S-P, Xue Y, Li H-Y, et al. High-TSH subclinical hypothyroidism is associated with postoperative mortality in acute type A aortic dissection. Front Endocrinol (Lausanne) 2022;13:844787. 10.3389/fendo.2022.844787 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Donadille B, Tuffet S, Cholet C, et al. Prevalence and progression of aortic dilatation in adult patients with turner syndrome: a cohort study. Eur J Endocrinol 2020;183:463–70. 10.1530/EJE-20-0284 [DOI] [PubMed] [Google Scholar]
- 24.Sidloff D, Choke E, Stather P, et al. Mortality from thoracic aortic diseases and associations with cardiovascular risk factors. Circulation 2014;130:2287–94. 10.1161/CIRCULATIONAHA.114.010890 [DOI] [PubMed] [Google Scholar]
- 25.Baumgartner H, De Backer J, Babu-Narayan SV, et al. 2020 ESC guidelines for the management of adult congenital heart disease. Eur Heart J 2020;41:4153–4. 10.1093/eurheartj/ehaa701 [DOI] [PubMed] [Google Scholar]
- 26.Suzuki T, Bossone E, Sawaki D, et al. Biomarkers of aortic diseases. Am Heart J 2013;165:15–25. 10.1016/j.ahj.2012.10.006 [DOI] [PubMed] [Google Scholar]
- 27.Stein E, Mueller GC, Sundaram B. Thoracic aorta (Multidetector computed tomography and magnetic resonance evaluation). Radiol Clin North Am 2014;52:195–217. 10.1016/j.rcl.2013.08.002 [DOI] [PubMed] [Google Scholar]
- 28.Jovin IS, Duggal M, Ebisu K, et al. Comparison of the effect on long-term outcomes in patients with thoracic aortic aneurysms of taking versus not taking a statin drug. Am J Cardiol 2012;109:1050–4. 10.1016/j.amjcard.2011.11.038 [DOI] [PubMed] [Google Scholar]
- 29.Wulf-Johansson H, Lock Johansson S, Schlosser A, et al. Localization of microfibrillar-associated protein 4 (MFAP4) in human tissues: clinical evaluation of serum MFAP4 and its association with various cardiovascular conditions. PLoS One 2013;8:e82243. 10.1371/journal.pone.0082243 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Pilop C, Aregger F, Gorman RC, et al. Proteomic analysis in aortic media of patients with Marfan syndrome reveals increased activity of calpain 2 in aortic aneurysms. Circulation 2009;120:983–91. 10.1161/CIRCULATIONAHA.108.843516 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
openhrt-2023-002317supp001.pdf (354.1KB, pdf)
Data Availability Statement
Data are available upon reasonable request.