Abstract
Purpose
Supportive care, including exercise, nutritional and psychological support, is becoming increasingly important in cancer given their impact on ‘patients’ quality and quantity of life. The purpose of this study was to explore willingness, preferences barriers and facilitators for a multimodal intervention in patients with cancer.
Methods
An anonymous questionnaire was proposed on randomly selected days to the patients visiting the cancer outpatients’ facilities at the Oncology Unit of the University Hospital of Verona. The questionnaire investigated willingness, preferences, barriers, and facilitators associated with participation in a multimodal program designed for patients with cancer. Exercise level was estimated using two open questions, nutritional risk was identified using the Nutritional Risk Screening 2002, while distress was evaluated with the Distress Thermometer.
Results
Based on 324 participants, 65% were interested in starting a multimodal intervention. Patients declared to prefer to receive instructions from dedicated experts, with a face-to-face approach, and during the anticancer treatment. Treatment-related side effects were the major obstacles for a multimodal program, while the availability of a specialized staff as exercise kinesiologists, dietitians, and psycho-oncologists was found to be an important facilitator for increasing ‘patients’ participation.
Conclusion
Patients patients with cancer are interested in participating in a multimodal supportive care program specifically designed for them. Information from this study may help to design a tailored multimodal intervention for patients with cancer.
Supplementary Information
The online version contains supplementary material available at 10.1007/s00432-022-04232-6.
Keywords: Cancer, Exercise level, Nutritional risk, Psychological distress, Multimodal intervention
Introduction
Whereas cancer incidence increased over the years (one out of three men and one out of four women are expected to develop cancer in their lifetime), cancer mortality decreased in many countries, leading to a high prevalence of people living with cancer (Fitzmaurice et al. 2017). Nevertheless, malignancy and its treatments are often related to several side effects, potentially impairing patients’ quality of life for years even after the therapy conclusion (Devlin et al. 2017). In this sense, patients with cancer may experience a range of different symptoms and adverse events affecting their physical and psychological well-being, such as functional deconditioning, nausea, vomiting, loss of muscle mass, anxiety, and depression (Devlin et al. 2017). This scenario emphasizes the need for supportive care programs aimed to help patients in recovering from the cancer pathway. Indeed, supportive care, defined as “the prevention and management of the adverse effects of cancer and its treatment” (Berman et al. 2020), is an essential tool able to manage physical and psychological symptoms and enhance the rehabilitation and survivorship of patients. Among them, exercise, nutrition, and psychological support are important for patients with cancer. Individually, these interventions have been shown to bring a series of benefits. Exercise can increase patients' physical fitness, such as cardiorespiratory fitness, muscle mass, and strength, which are prognostic factors in patients with cancer (Campbell et al. 2019). Moreover, exercise may ameliorate adverse events of cancer and its treatments, manage cancer-related fatigue, anemia, and psychological impairments, improving peripheral neuropathy and quality of life (Campbell et al. 2019). Similarly, a nutritional screening may help to individuate, and consequently manage, malnourished patients or those at risk of malnutrition (Rock et al. 2022; Trestini et al. 2020). Nutritional intervention may facilitate maintaining an adequate nutritional intake, controlling body composition, and nutritional impact symptoms, such as nausea, vomiting, and appetite loss, to prevent loss of muscle mass and nutrient inadequacies (Caccialanza et al. 2022; Rock et al. 2022; Trestini et al. 2021a, b; Trestini et al. 2021a, b). A cancer diagnosis carries an important psychological burden for patients and their caregivers. Symptoms of anxiety, depression, fear of death, and/or recurrence frequently occur in patients with cancer. Psychological interventions, e.g., cognitive–behavioral therapy, address various psychological and social aspects that may alleviate the emotional outcomes, improving patients' quality of life (Liu et al. 2019; Travier et al. 2015).
Exercise, nutrition, and psychological aspects are strictly related, and it is reasonable to speculate that a supportive care multimodal approach could grow the benefits, as supported by previous research (Avancini et al. 2021). An 18 week exercise program including cognitive–behavioral principles of social cognitive theory found positive effects on fatigue and physical fitness levels in patients with breast cancer receiving chemotherapy (Travier et al. 2015). Similarly, a two-week prehabilitation program incorporating exercise, nutritional counseling, and psychological guidance has been shown to be effective in increasing perioperative functional capacity in patients with lung cancer undergoing thoracoscopic lobectomy (Liu et al. 2019). Nevertheless, investigations combining exercise, nutritional and psychological approaches are still few, and more research on the benefit of multimodal interventions is necessary. However, participating in a supportive care multimodal program requires a time effort from patients, making high the risk of non-compliance.
Understanding patients’ willingness, preferences, barriers, and needs may permit to develop an optimal and tailored multimodal intervention. Although the factors influencing the single lifestyle intervention in a population with cancer are investigated, studies exploring the feasibility and the willingness of patients to join a multimodal intervention are lacking. To fill this gap, this research aims to: (i) establish the willingness of patients with cancer to participate in a multimodal supportive care intervention, including exercise, nutritional and psychological approaches, (ii) analyze the patients' characteristics associated with their willingness to participate in a multimodal supportive care intervention, (iii) describe patients’ preferences about exercise, nutritional and psychological interventions, (iv) examine the perceived barriers and facilitators affecting patients adherence to the multimodal intervention.
Methods
Study design and participants
A cross-sectional survey was utilized. Between February 2020 and May 2021, an anonymous questionnaire was delivered to the patients visiting the cancer outpatients' facilities at the Oncology Unit of the University Hospital of Verona. Patients' eligibility criteria were: 18 years or older, a cancer diagnosis, and ability to understand Italian. The study staff distributed the questionnaires on randomly selected days. Patients were approached face-to-face, informed about the study, and asked whether they would be willing to complete the questionnaire. If interested, patients signed the informed consent and received a copy of the questionnaire to return directly. A duplicate check was done, looking for duplicates by date of birth, sex, education, and marital status. Approval of the Ethics Committee for Clinical Trials (Prot. No. 48647) was obtained. The study protocol adhered to Good Clinical Practice principles and the procedures were conducted following the last revision of the declaration of Helsinki as well as the declaration of Oviedo. The current report is compliant with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines (Cuschieri 2019).
Questionnaire description
A self-administered survey questionnaire was developed to collect preferences, barriers, and facilitators associated with a multimodal intervention. A pilot version of the questionnaire was created using questions derived from previous studies (Arthur et al. 2016; Avancini et al. 2020a; Weller et al. 2019), and made available to experts, including kinesiologists, oncologists, and psycho-oncologists, to make an informal peer review and develop the current version.
The questionnaire comprised 30 items and took approximately 30 min to complete. The survey included five parts: (i) General characteristics; (ii) Distress, exercise level, and risk of malnutrition; (iii) Multimodal intervention preferences; (iv) Barriers and facilitators associated with a multimodal intervention; (v) Cancer diagnosis and treatment.
General characteristics
The following demographic, and socio-economic factors were self-reported: birth date (day, month, year), sex (male/female), educational level (elementary—up to age 10–11 years/secondary—up to 14 years/secondary—up to 18–19 years/college–university/postgraduate), marital status (single/married/divorced/widowed), occupational status (retired/ homemaker/part-time employed/ full-time employed), perceived economic adequacy (inadequate/ barely adequate/adequate/ more than adequate). Weight in kilograms and height in meters were collected to obtain body mass index (BMI). BMI was calculated dividing the weight by the squared height, and categorizing as: underweight (BMI < 18.5 kg/m2), normal weight (BMI 18.5–24.9 kg/m2), overweight (BMI 25.0–29.9 kg/m2) and obese (BMI > 29.9 kg/m2) (“Physical status: the use and interpretation of anthropometry. Report of a WHO Expert Committee” 1995).
Distress, exercise level, and risk of malnutrition
Distress was assessed using the National Comprehensive Cancer Network (NCCN) Distress screening tool (Riba et al. 2019). Through a single item, representing an 11 points visual/Likert scale, from 0 (no distress) to 10 (extreme distress), the patient was asked to rate her/his level of distress experienced over the past week. Distress is classified as mild with a cutoff point < 4, whereas a score > 3 requires further screening (Riba et al. 2019). Exercise level was assessed with the two open-ended questions, adapted by Schmitz et al. (Schmitz et al. 2019): “How many days per week and times per session do you perform an aerobic activity at moderate intensity (where the heart beats faster and the breathing harder than normal, e.g., walking, cycling, running, swimming)? and “How many days per week do you perform exercise to increase muscle strength (e.g., lifting weights, bodyweight exercise, climbing)? According to the American College of Sports Medicine guidelines for patients with cancer, patients were defined as meeting the guidelines if they engaged in at least 90 min/week of aerobic exercise and performed strength activities at least two times/week (Campbell et al. 2019). The Nutritional Risk Screening 2002 (NRS-2002) was used to identify the nutritional risk. Malnutrition risk identification was not directly captured, but the questionnaire included nutrition-related questions, including severity of the disease, weight loss in the past one, two, and three months, perceived impairments in general condition, and the reduction of food intake in the preceding week, that allowed us to estimate its prevalence. NRS-2002 is evaluated through three components: nutritional status (0–3 points), the severity of disease (0–3 points), and age (0–1 points). The total NRS-2002 score ranges from 0 to 7, and patients with a score of < 3 and ≥ 3 are classified as “no nutritional risk” and “at nutritional risk”, respectively (Kondrup et al. 2003).
Multimodal intervention preferences
Preferences were explored using closed-item questions adapted from prior investigations. The first question concerned the patients’ willingness to participate in a multimodal supportive care intervention including exercise, nutritional counseling and psychological support specifically designed for patients with cancer (yes/no/maybe).
For each intervention, i.e., exercise, nutrition and psychological support, were asked patient ‘preferences concerning: who would give them instructions (oncologist/nurse/dietitian/kinesiologist/psycho-oncologist/another patient with cancer/other); how to receive instructions (face to face/over the internet/television/radio/brochure-pamphlet/other); where (at hospital/a community center outside the hospital/other); when (before treatment/during treatment/after treatment/other).
Barriers and facilitators associated with a multimodal intervention
A list of potential barriers and facilitators associated with participation in a multimodal intervention was provided. Patients may select up to three barriers and up to three facilitators.
Cancer diagnosis and treatment
Self-reported medical variables included: cancer site (breast/lung/colorectal/upper gastrointestinal/head-neck/gynecological/urogenital/melanoma/other), disease status (early/advanced/metastatic/in remission-cured/unknown), date of diagnosis (month/year), type of treatment (surgery/chemotherapy/radiotherapy/hormone therapy/other), and current treatment status (about to start/ongoing/completed/not known).
Statistical analysis
Descriptive analyses were utilized to summarize the response to survey questions. Categorical data were presented as frequencies and percentages. Logistic regression models were applied to identify patients’ characteristics (sex, age, education, body mass index, exercise level, psychological distress, risk of malnutrition, marital status, occupational status, perceived income adequacy, tumor site, disease status, cancer treatment, treatment status, and time from diagnosis) associated with their willingness to participate in a multimodal supportive care program. Additionally, logistic regression models were applied to explore patients’ characteristics associated with exercise level, risk of malnutrition and distress (Supplementary Material 1). SPSS version 28.0 software was utilized to analyze data. The significance level was set at 0.05, whereas all statistical tests were two-sided.
Results
Between February 2020 and May 2021, a total of 623 were approached, and among them, 324 agreed to participate in the survey (52% response rate).
General characteristics
The demographic and medical characteristics of the survey respondents are listed in Table 1. Overall, 57% had less than 65 years, 53% were female, 61% had a higher education (at least up to age 18–19 years), and 70% were married. Upper gastro-intestine (44%) and breast (17%) were the most frequent cancer site, and about 77% were on active anticancer treatment.
Table 1.
General characteristics of the survey’s participants1
| No | % | |
|---|---|---|
| Age (years) (n = 324) | ||
| < 65 | 184 | 57 |
| ≥ 65 | 140 | 43 |
| Sex (n = 324) | ||
| Female | 171 | 53 |
| Male | 183 | 47 |
| Education (n = 324) | ||
| Elementary (up to 10–11 years) | 27 | 8 |
| Secondary (up to 14 years) | 101 | 31 |
| Secondary (up to 18–19 years) | 120 | 37 |
| College/University | 67 | 21 |
| Postgraduate | 9 | 3 |
| Body Mass Index2 (n = 324) | ||
| Underweight | 12 | 4 |
| Normal weight | 134 | 41 |
| Overweight | 133 | 41 |
| Obese | 45 | 14 |
| Marital status (n = 324) | ||
| Single | 35 | 11 |
| Married | 228 | 70 |
| Divorced | 33 | 10 |
| Widowed | 28 | 9 |
| Occupational status (n = 324) | ||
| Retired | 154 | 48 |
| Homemaker | 47 | 16 |
| Part-time employed | 24 | 7 |
| Full-time employed | 93 | 29 |
| Other | 0 | 0 |
| Perceived income adequacy3 (n = 324) | ||
| Inadequate | 14 | 4 |
| Barely adequate | 59 | 18 |
| Adequate | 159 | 49 |
| More than adequate | 92 | 28 |
| Exercise level (n = 314) | ||
| Meeting aerobic guidelines | ||
| Yes | 94 | 30 |
| No | 220 | 70 |
| Meeting strength guidelines | ||
| Yes | 31 | 10 |
| No | 283 | 90 |
| Meeting exercise guidelines | ||
| Yes | 11 | 4 |
| No | 303 | 96 |
| Distress level (n = 306) | ||
| Mild | 129 | 42 |
| Clinically elevate | 177 | 58 |
| Risk of malnutrition (n = 320) | ||
| Yes | 97 | 30 |
| No | 223 | 70 |
| Tumor site (n = 324) | ||
| Breast | 55 | 17 |
| Lung | 18 | 6 |
| Colorectum | 20 | 6 |
| Head/neck | 19 | 6 |
| Upper gastro-intestine | 143 | 44 |
| Gynecological | 4 | 1 |
| Urogenital | 24 | 7 |
| Melanoma | 22 | 7 |
| Other | 19 | 6 |
| Disease status (n = 324) | ||
| Unknown | 59 | 18 |
| In remission/cured | 67 | 21 |
| Early | 44 | 14 |
| Advanced | 72 | 22 |
| Metastatic | 82 | 25 |
| Treatments4 (n = 324) | ||
| Surgery | 157 | 48 |
| Chemotherapy | 221 | 68 |
| Radiation therapy | 73 | 23 |
| Hormone therapy | 22 | 7 |
| Other | 56 | 17 |
| Treatment status (n = 324) | ||
| About to start | 16 | 5 |
| Ongoing | 234 | 77 |
| Completed | 43 | 14 |
| Unknown | 12 | 4 |
| Time from diagnosis (n = 324) | ||
| ≤ 30 months | 186 | 57 |
| ≥ 30 months | 138 | 43 |
1Participants of survey study conducted in Verona, Italy, from February 2020 to May 2021
2Body mass index categories are those of the World Health Organization]
3Perceived income adequacy assessed by the question: does your monthly income cover your monthly expenditure?
4Treatments, which may be completed or in course, and are not mutually exclusive
Overall, 30% and 10% of patients reported following the amount of exercise suggested for aerobic and strength activities, respectively. When the two types of exercise were merged, only 4% of survey participants resulted to follow the ACSM guidelines. Whereas the aerobic exercise levels were similar in males and females and through the age, strength training was higher in male patients with < 65 years (Supplementary material 1). The Distress Thermometer found clinically relevant levels of distress in 58% of survey participants, with percentages slightly higher in female patients. A total of 30% of patients were at risk of malnutrition. Older patients, both male and female were more frequently at high risk of malnutrition compared to < 65 years. Logistic regression models found several patients’ characteristics associated with exercise level, risk of malnutrition and distress (Supplementary Material 1).
Willingness and preferences for a multimodal supportive care intervention
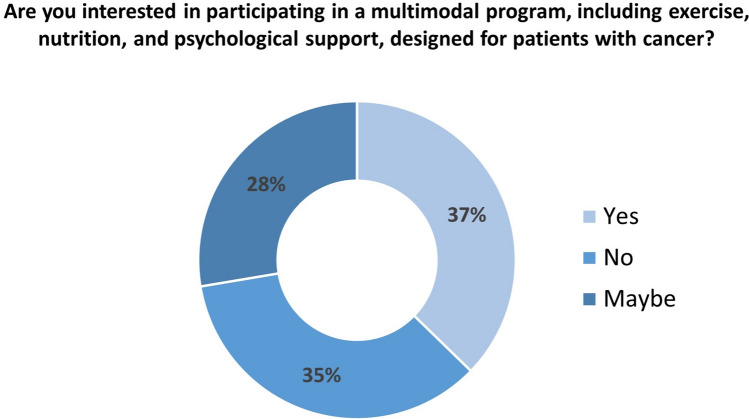
The willingness to participate to a multimodal supportive care program is shown in Fig. 1. A total of 65% of the survey participants were interested (i.e., yes, or maybe) in participating in a multimodal program, including exercise, nutrition, and psychological support, specifically designed for patients with cancer. Compared to patients having < 65 years, older subjects were less willing to participate in a multimodal intervention (OR = 0.42, 95% CI = 0.26–0.69). Patients who were single were more likely to join the multimodal intervention (OR = 2.24, 95% CI = 1.08–4.63) than those married. Patients who self-defined their cancer stage as "early were less interested in participating in an intervention, including exercise, nutrition, and psychological support (OR = 0.34, 95% CI = 0.14–0.86). Logistic regression is displayed in Supplementary material 1.
Fig. 1.
Willingness to start a multimodal intervention including exercise, nutritional counseling, and psychological support
Survey ‘respondents’ preferences are listed in Table 2. Patients preferred to receive instructions from the reference experts for each area: a kinesiologist (48%) for exercise, a dietitian (61%) for nutrition, and a psycho-oncologist (61%) for psychological support. For each intervention, the preferred way to receive information was through a face-to-face approach (66% for exercise, 69% for nutrition, and 72% for psychological support), at the hospital (78% for exercise, 87% for nutrition, and 88% for psychological support), and during anticancer treatment (44% for exercise, 47% for nutrition, and 51% for psychological support).
Table 2.
Preferences in patient with cancer for exercise, nutrition, and psychological aspect1
| Preference as expressed by answers to questions | Exercise | Nutrition | Psychology | |||
|---|---|---|---|---|---|---|
| N | % | N | % | N | % | |
| Who would you prefer to receive instructions from? | (n = 304) | (n = 312) | (n = 305) | |||
| Oncologist | 125 | 41 | 112 | 36 | 102 | 33 |
| Nurse | 2 | 1 | 1 | 0 | 2 | 1 |
| Kinesiologist | 145 | 48 | 4 | 1 | 6 | 2 |
| Nutritionist | 14 | 5 | 191 | 61 | 2 | 1 |
| Psychologist | 2 | 1 | 3 | 1 | 186 | 61 |
| Another patient cancer | 0 | 0 | 0 | 0 | 0 | 0 |
| No preference | 0 | 0 | 0 | 0 | 0 | 0 |
| Other | 16 | 5 | 1 | 0 | 7 | 2 |
| How would you prefer to receive instructions? | (n = 300) | (n = 305) | (n = 301) | |||
| Face to face | 197 | 66 | 209 | 69 | 218 | 72 |
| Television | 8 | 3 | 7 | 2 | 6 | 2 |
| Radio | 1 | 0 | 1 | 0 | 1 | 0 |
| Leaflet/pamphlet | 23 | 8 | 18 | 6 | 13 | 4 |
| Over the internet | 60 | 20 | 61 | 20 | 51 | 17 |
| No preference | 0 | 0 | 0 | 0 | 0 | 0 |
| Other | 11 | 4 | 9 | 3 | 12 | 4 |
| Where would you prefer to receive instructions? | (n = 310) | (n = 306) | (n = 306) | |||
| At the hospital | 240 | 78 | 271 | 87 | 264 | 88 |
| At a center outside the hospital | 57 | 19 | 33 | 11 | 35 | 11 |
| Other | 9 | 3 | 6 | 2 | 7 | 2 |
| When would you prefer to receive instructions? | (n = 300) | (n = 294) | (n = 292) | |||
| Before treatment | 85 | 29 | 89 | 30 | 74 | 25 |
| During treatment | 130 | 44 | 142 | 47 | 148 | 51 |
| After treatment | 34 | 12 | 26 | 9 | 27 | 9 |
| Other | 45 | 15 | 43 | 14 | 43 | 15 |
1Participants of survey study conducted in Verona, Italy, from February 2020 to May 2021
Barriers and facilitators to a multimodal intervention
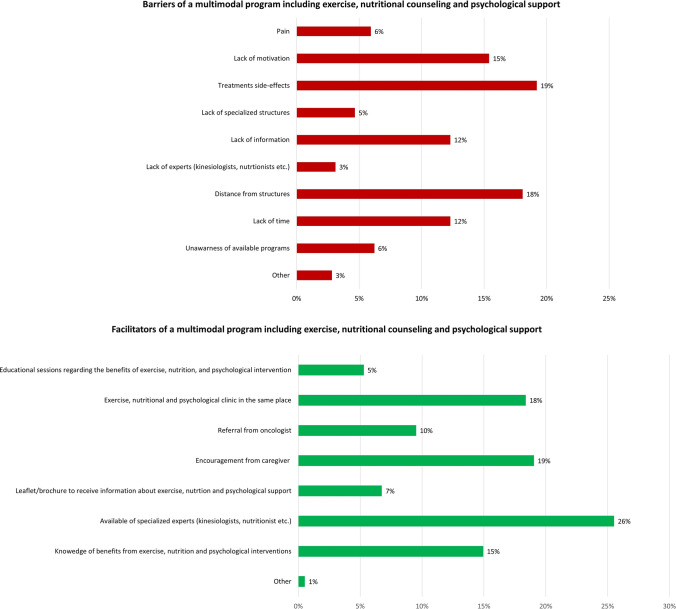
Figure 2 reported the barriers and facilitators of a multimodal program individuated by patients. The most commonly reported barriers potentially hindering participation in a multimodal supportive care intervention were: treatment-related side effects, distance from the structures, and lack of motivation. On the contrary, most commonly reported features facilitating the involvement in a program, including exercise, nutrition, and psychological support, were: availability of specialized experts, encouragement from caregivers, and having the exercise, nutritional and psychological clinics in the same place.
Fig. 2.
Barriers and facilitators associated with a multimodal intervention
Discussion
The present study is the first to explore willingness, preferences, barriers, and facilitators among patients with cancer to participate in a multimodal supportive care program including exercise, nutritional counseling, and psychological support.
We found that roughly 65% of the survey participants were willing to start a multimodal program, including exercise, nutrition, and psychological support, designed for patients with cancer. This finding is particularly crucial especially if considered that 30% of patients were at nutritional risk, 58% reported clinically relevant levels of distress, and 96% did not meet the current exercise guidelines for cancer patients, and the possible consequences in terms of quality and quantity of life of such impaired levels (Gerritsen and Vincent 2016; Hamer et al. 2009; Patel et al. 2019; Zhang et al. 2019). Prior investigations reported that patients with cancer usually express interest in health-related behaviors intervention, such as exercise (Avancini et al. 2020a), nutritional counseling (Green et al. 2014; Keaver et al. 2022), and psychological support (Arch et al. 2018). Similarly, a study exploring the preferences related to physical activity and diet interventions in pancreatic cancer survivors found that 69% of study participants were interested in participating a combined lifestyle intervention (Arthur et al. 2016). Our results support the desire of the patients with cancer to be proactive in their disease journey and their wish for a multimodal supportive care service. Moreover, this finding is crucial, especially in view of the multidisciplinary approach delivery and from a clinical point of view (Avancini et al. 2022). In this sense, different societies support the integration of supportive care alongside the cancer continuum. For instance, prehabilitation, i.e., interventions, such as exercise, smoking cessation, nutrition, and psychological support, performed between the time of cancer diagnosis and the beginning of the acute treatment, is becoming more and more relevant and a standard of care so much that different guidelines insert it in their recommendations (Low et al. 2019; Melloul et al. 2020; Nelson et al. 2019). On the other hand, the multimodal intervention has been suggested as a possible treatment for a complex syndrome like cancer-related cachexia. This multifactorial syndrome, characterized predominantly by loss of skeletal muscle mass, not fully reversed by nutritional support, and leading to a progressive functional impairment, is still orphan of effective treatments (Avancini et al. 2021). Whereas single interventions failed to gain benefits, multimodal management may be the best strategy, given its multifactorial nature (Avancini et al. 2021).
Logistic regression revealed that younger patients and those who were single were more interested in participating in a multimodal supportive care intervention. These results may be explained by the fact that aging is associated with comorbidities and growing difficulties. Prior researches on exercise found mixed results. On one side, some studies reported that more than half of older patients with cancer were interested in participating in a physical activity program (Cheung et al. 2021) (Fournier et al. 2022). On the contrary, a cross-sectional study on ovarian cancer survivors found that the interest in physical activity diminished in among participants who were aged ≥ 60 years (Stevinson et al. 2009), and Morielli and colleagues reported that age was associated with a low exercise adherence in patients with rectal cancer (Morielli et al. 2018). Regarding medical variables, patients who defined their cancer at “early” stage were less willing to participate in a multimodal supportive care program. Despite, to our knowledge, no prior studies reported a similar finding; this could intuitively explain by the fact that patients with an early stage of disease might feel less in danger, while individuals with an advanced/metastatic cancer, being aware of the severity of their disease, would try everything to feel better (Avancini et al. 2020a; Wong et al. 2018).
Regarding the preferred source for receiving instructions about each intervention, patients preferred to receive information from a dedicated expert (i.e., kinesiologist, dietitian, psycho-oncologist). These results are in line with previous studies (Gjerset et al. 2011; Nicole Culos-Reed et al. 2017), and with recommendations of international societies (Arends et al. 2017; Campbell et al. 2019). Nevertheless, a relevant percentage of patients, ranging from 33 to 41%, have indicated the oncologist as the preferred person to deliver instructions. Similarly, our previous work focusing on exercise identified the oncologist as the preferred person to deliver information (Avancini et al. 2020a). These results may be explained by the fact that the patients recognize the specificity of each approach and thus the need for specialized staff. Nevertheless, patients put their trust in their oncologists during the cancer journey, and therefore some of them may feel more reassured to receive instruction from their voice.
Most patients reported that they preferred to receive exercise and nutritional and psychological instructions face-to-face. Whereas face-to-face counseling is the most preferred counseling modality in several previous investigations (Avancini et al. 2020a; Wong et al. 2018), we found that a quarter of patients have indicated the internet as the preferred source of information. Information over the internet may have the great advantages of reaching a large number of individuals, increasing patients’ knowledge and engagement in health decision-making strategies. On the other hand, web-based informations are difficult to regulate, and often the quality control is a challenge, in which the risk of incurring misleading information becomes high, hitting more individuals in a vulnerable position, such as patients with cancer. In this sense, providing reliable websites developed by reputable institutions, such as universities or hospitals, may overcome this problem and offer evidence-based information.
The hospital was identified as the preferred place to receive information. Moreover, patients wished to receive instruction about the multimodal intervention before the beginning of therapies or during anticancer treatment. Although the time variance in the preference of lifestyle program start has been reported in the literature (Wong et al. 2018), our results are encouraging because such interventions (or the monitoring through appropriate screening tools), should be early and regularly administered (Ravasco 2019) to prevent or manage possible impairments.
Treatment side effects, distance from facilities, and lack of motivation were reported as the major barriers. These results of mixed disease specific and general obstacles to multimodal intervention mirror the current literature (Arthur et al. 2016; Avancini et al. 2020a; Clifford et al. 2018; Keaver et al. 2022). Although treatment-related adverse events, such as fatigue, and lymphoedema, are frequently reported as potential barriers in lifestyle interventions (Arthur et al. 2016), it is interesting to highlight that many of these side effects can effectively be managed through supportive care intervention. In this sense, increasing patients’ knowledge and awareness about the benefits of supportive care intervention may be a useful strategy to increase patients’ compliance. Distance from facilities and lack of motivation may be easily overcome, including tailored programs with different modality options (e.g., face-to-face approach or using telehealth), and incorporating motivational approach (e.g., goal setting, self-monitoring, action planning) to behavior change.
On the other, the availability of specialized experts, having exercise, nutrition, and psychological clinic in the same facilities, and social support were identified as facilitators for the participation in a multimodal supportive care program. Prior investigations support the experts’ supervision as facilitators to participate in a lifestyle program (Avancini et al. 2020a, b, c; Keaver et al. 2022), and may suggest that patients desire tailored intervention based on their needs. Social support may play a role in the intervention compliance, supporting patients ‘motivation and enhancing emotional well-being (Fong et al. 2017).
The present work has some limitations that should be noted. Information was self-reported and therefore open to different sources of bias. Recall bias may be a possible source of error. To minimize this issue, we have adopted the short version of the questionnaire investigating exercise, nutritional risk, and distress, asking for recent information (e.g., in the previous week). Moreover, the questionnaire did not collect information regarding participants’ diet, and that limit its ability to explore associations with other possible determinants of willingness to participate in a multimodal lifestyle program. The social desirability bias may be less likely because the questionnaire was filled and returned anonymously.
However, it cannot be excluded that patients who decided to participate in this study may be individuals more interested in the supportive care. To reduce this potential bias, the questionnaire was proposed to all patients on randomly selected days. The study participants were sampled to be representative of those attending the Verona oncology clinic, and not the full total of the patients, making our results little generalizable.
In conclusion, this study highlights that patients desire to participate in a multimodal supportive care intervention, including exercise, nutritional and psychological support, specifically designed for individuals with cancer. Patients prefer to receive instruction in the hospital, from dedicated experts, and with a face-to-face approach. Although different barriers to multimodal supportive care intervention have been identified, several facilitators may promote patient compliance.
Overall, these results support that a multimodal supportive care intervention is feasible and desired by patients with cancer, and represent the first step toward the development of a tailored program.
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgements
S.P. is supported by the Italian Association for Cancer Research AIRC-IG 20583. S.P. was supported by the International Association for Lung Cancer (IASLC).
Author contributions
All authors contributed to the study conception and design. Material preparation and data collection were performed by AA, SP, IT and DT. Analysis was performed by AA. The first draft of the manuscript was written by AA and all authors commented on previous version of the manuscript. All authors read and approved the final manuscript.
Funding
Open access funding provided by Università degli Studi di Verona within the CRUI-CARE Agreement. The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Declarations
Conflict of interest
The authors declare that this research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. M.M. reports personal fees from Pfizer, EUSA Pharma and Astra Zeneca, outside the submitted manuscript. S.P. received honoraria or speakers’ fee from Astra-Zeneca, Eli-Lilly, BMS, Boehringer Ingelheim, MSD and Roche, outside the submitted manuscript. All remaining authors declare that they have no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Alice Avancini, Email: alice.avancini@univr.it.
Ilaria Trestini, Email: ilaria.trestini@aovr.veneto.it.
Daniela Tregnago, Email: daniela.tregnago2@aovr.veneto.it.
Lorenzo Belluomini, Email: lorenzo.belluomini@univr.it.
Marco Sposito, Email: marcosposito91@gmail.com.
Jessica Insolda, Email: jessica.insolda@aovr.veneto.it.
Federico Schena, Email: federico.schena@univr.it.
Michele Milella, Email: michele.milella@univr.it.
Sara Pilotto, Email: sara.pilotto@univr.it.
References
- Arch JJ, Vanderkruik R, Kirk A, Carr AL. A closer lens: cancer survivors’ supportive intervention preferences and interventions received. Psychooncology. 2018;27(5):1434–1441. doi: 10.1002/pon.4526. [DOI] [PubMed] [Google Scholar]
- Arends J, Baracos V, Bertz H, Bozzetti F, Calder PC, Deutz NEP, Weimann A. ESPEN expert group recommendations for action against cancer-related malnutrition. Clin Nutr. 2017;36(5):1187–1196. doi: 10.1016/j.clnu.2017.06.017. [DOI] [PubMed] [Google Scholar]
- Arthur AE, Delk A, Demark-Wahnefried W, Christein JD, Contreras C, Posey JA, Rogers LQ. Pancreatic cancer survivors’ preferences, barriers, and facilitators related to physical activity and diet interventions. J Cancer Surviv. 2016;10(6):981–989. doi: 10.1007/s11764-016-0544-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Avancini A, Pala V, Trestini I, Tregnago D, Mariani L, Sieri S, Lanza M. Exercise levels and preferences in cancer patients: a cross-sectional study. Int J Environ Res Public Health. 2020 doi: 10.3390/ijerph17155351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Avancini A, Tregnago D, Rigatti L, Sartori G, Yang L, Bonaiuto C, Lanza M. Factors influencing physical activity in cancer patients during oncological treatments: a qualitative study. Integrative Cancer Treatments. 2020 doi: 10.1177/1534735420971365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Avancini A, Tregnago D, Rigatti L, Sartori G, Yang L, Trestini I, Lanza M. Factors influencing physical activity in cancer patients during oncological treatments: a qualitative study. Integr Cancer Ther. 2020;19:153473542097136. doi: 10.1177/1534735420971365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Avancini A, Trestini I, Tregnago D, Lanza M, Menis J, Belluomini L, Pilotto S. A multimodal approach to cancer-related cachexia: from theory to practice. Expert Rev Anticancer Ther. 2021;21(8):819–826. doi: 10.1080/14737140.2021.1927720. [DOI] [PubMed] [Google Scholar]
- Avancini A, Belluomini L, Borsati A, Riva ST, Trestini I, Tregnago D, Pilotto S. Integrating supportive care into the multidisciplinary management of lung cancer: we can't wait any longer. Expert Rev Anticancer Ther. 2022 doi: 10.1080/14737140.2022.2082410. [DOI] [PubMed] [Google Scholar]
- Berman R, Davies A, Cooksley T, Gralla R, Carter L, Darlington E, Higham C. Supportive care: an indispensable component of modern oncology. Clin Oncol (R Coll Radiol) 2020;32(11):781–788. doi: 10.1016/j.clon.2020.07.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Caccialanza R, Cotogni P, Cereda E, Bossi P, Aprile G, Delrio P, Pedrazzoli P. Nutritional support in cancer patients: update of the Italian Intersociety Working Group practical recommendations [Research Paper] J Cancer. 2022;13(9):2705–2716. doi: 10.7150/jca.73130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campbell KL, Winters-Stone KM, Wiskemann J, May AM, Schwartz AL, Courneya KS, Schmitz KH. Exercise guidelines for cancer survivors: consensus statement from international multidisciplinary roundtable. Med Sci Sports Exerc. 2019;51(11):2375–2390. doi: 10.1249/MSS.0000000000002116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheung DST, Takemura N, Chau PH, Ng AYM, Xu X, Lin CC. Exercise levels and preferences on exercise counselling and programming among older cancer survivors: a mixed-methods study. J Geriatr Oncol. 2021;12(8):1173–1180. doi: 10.1016/j.jgo.2021.05.002. [DOI] [PubMed] [Google Scholar]
- Clifford BK, Mizrahi D, Sandler CX, Barry BK, Simar D, Wakefield CE, Goldstein D. Barriers and facilitators of exercise experienced by cancer survivors: a mixed methods systematic review. Support Care Cancer. 2018;26(3):685–700. doi: 10.1007/s00520-017-3964-5. [DOI] [PubMed] [Google Scholar]
- Cuschieri S. The STROBE guidelines. Saudi J Anaesth. 2019;13(Suppl 1):S31–S34. doi: 10.4103/sja.SJA_543_18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Devlin EJ, Denson LA, Whitford HS. Cancer treatment side effects: a meta-analysis of the relationship between response expectancies and experience. J Pain Symptom Manage. 2017;54(2):245–258.e242. doi: 10.1016/j.jpainsymman.2017.03.017. [DOI] [PubMed] [Google Scholar]
- Fitzmaurice C, Allen C, Barber RM, Barregard L, Bhutta ZA, Brenner H, Collaboration, G.B. o. D. C. Global, regional, and National Cancer incidence, mortality, years of life lost, years lived with disability, and disability-adjusted life-years for 32 cancer groups, 1990 to 2015: a systematic analysis for the global burden of disease study. JAMA Oncol. 2017;3(4):524–548. doi: 10.1001/jamaoncol.2016.5688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fong AJ, Scarapicchia TMF, McDonough MH, Wrosch C, Sabiston CM. Changes in social support predict emotional wellbeing in breast cancer survivors. Psychooncology. 2017;26(5):664–671. doi: 10.1002/pon.4064. [DOI] [PubMed] [Google Scholar]
- Fournier B, Delrieu L, Russo C, Terret C, Fervers B, Pérol O. Interest and preferences for physical activity programming and counselling among cancer patients aged over 70 years receiving oncological treatments. Eur J Cancer Care (Engl) 2022;31(1):e13527. doi: 10.1111/ecc.13527. [DOI] [PubMed] [Google Scholar]
- Gerritsen JK, Vincent AJ. Exercise improves quality of life in patients with cancer: a systematic review and meta-analysis of randomised controlled trials. Br J Sports Med. 2016;50(13):796–803. doi: 10.1136/bjsports-2015-094787. [DOI] [PubMed] [Google Scholar]
- Gjerset GM, Fosså SD, Courneya KS, Skovlund E, Jacobsen AB, Thorsen L. Interest and preferences for exercise counselling and programming among Norwegian cancer survivors. Eur J Cancer Care (Engl) 2011;20(1):96–105. doi: 10.1111/j.1365-2354.2009.01161.x. [DOI] [PubMed] [Google Scholar]
- Green HJ, Steinnagel G, Morris C, Laakso EL. Health behaviour models and patient preferences regarding nutrition and physical activity after breast or prostate cancer diagnosis. Eur J Cancer Care (Engl) 2014;23(5):640–652. doi: 10.1111/ecc.12190. [DOI] [PubMed] [Google Scholar]
- Hamer M, Chida Y, Molloy GJ. Psychological distress and cancer mortality. J Psychosom Res. 2009;66(3):255–258. doi: 10.1016/j.jpsychores.2008.11.002. [DOI] [PubMed] [Google Scholar]
- Keaver L, O’Callaghan N, Douglas P. Nutrition support and intervention preferences of cancer survivors. J Hum Nutr Diet. 2022 doi: 10.1111/jhn.13058. [DOI] [PubMed] [Google Scholar]
- Kondrup J, Rasmussen HH, Hamberg O, Stanga Z, Group, A. H. E. W. Nutritional risk screening (NRS 2002): a new method based on an analysis of controlled clinical trials. Clin Nutr. 2003;22(3):321–336. doi: 10.1016/s0261-5614(02)00214-5. [DOI] [PubMed] [Google Scholar]
- Liu Z, Qiu T, Pei L, Zhang Y, Xu L, Cui Y, Huang Y. Two-week multimodal prehabilitation program improves perioperative functional capability in patients undergoing thoracoscopic lobectomy for lung cancer: A randomized controlled trial. Anesth Analg. 2019 doi: 10.1213/ANE.0000000000004342. [DOI] [PubMed] [Google Scholar]
- Low DE, Allum W, De Manzoni G, Ferri L, Immanuel A, Kuppusamy M, Ljungqvist O. Guidelines for perioperative care in esophagectomy: enhanced recovery after surgery (ERAS. World J Surg. 2019;43(2):299–330. doi: 10.1007/s00268-018-4786-4. [DOI] [PubMed] [Google Scholar]
- Melloul E, Lassen K, Roulin D, Grass F, Perinel J, Adham M, Demartines N. Guidelines for perioperative care for pancreatoduodenectomy: enhanced recovery after surgery (ERAS) recommendations 2019. World J Surg. 2020;44(7):2056–2084. doi: 10.1007/s00268-020-05462-w. [DOI] [PubMed] [Google Scholar]
- Morielli AR, Boulé NG, Usmani N, Joseph K, Tankel K, Severin D, Courneya KS. Predictors of adherence to aerobic exercise in rectal cancer patients during and after neoadjuvant chemoradiotherapy. Psychol Health Med. 2018;23(2):224–231. doi: 10.1080/13548506.2017.1344356. [DOI] [PubMed] [Google Scholar]
- Nelson G, Bakkum-Gamez J, Kalogera E, Glaser G, Altman A, Meyer LA, Dowdy SC. Guidelines for perioperative care in gynecologic/oncology: enhanced recovery after surgery (ERAS) society recommendations-2019 update. Int J Gynecol Cancer. 2019;29(4):651–668. doi: 10.1136/ijgc-2019-000356. [DOI] [PubMed] [Google Scholar]
- Nicole Culos-Reed S, Leach HJ, Capozzi LC, Easaw J, Eves N, Millet GY. Exercise preferences and associations between fitness parameters, physical activity, and quality of life in high-grade glioma patients. Support Care Cancer. 2017;25(4):1237–1246. doi: 10.1007/s00520-016-3516-4. [DOI] [PubMed] [Google Scholar]
- Patel AV, Friedenreich CM, Moore SC, Hayes SC, Silver JK, Campbell KL, Matthews CE. American College of Sports Medicine Roundtable Report on Physical Activity, Sedentary Behavior, and Cancer Prevention and Control. Med Sci Sports Exerc. 2019;51(11):2391–2402. doi: 10.1249/MSS.0000000000002117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Physical status: the use and interpretation of anthropometry. Report of a WHO Expert Committee (1995) World Health Organ Tech Rep Ser, 854, 1–452. [PubMed]
- Ravasco P. Nutrition in cancer patients. J Clin Med. 2019 doi: 10.3390/jcm8081211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Riba MB, Donovan KA, Andersen B, Braun I, Breitbart WS, Brewer BW, Darlow SD. Distress management, version 3.2019, NCCN clinical practice guidelines in oncology. J Natl Compr Canc Netw. 2019;17(10):1229–1249. doi: 10.6004/jnccn.2019.0048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rock CL, Thomson CA, Sullivan KR, Howe CL, Kushi LH, Caan BJ, McCullough ML. American Cancer Society nutrition and physical activity guideline for cancer survivors. CA Cancer J Clin. 2022 doi: 10.3322/caac.21719. [DOI] [PubMed] [Google Scholar]
- Schmitz KH, Campbell AM, Stuiver MM, Pinto BM, Schwartz AL, Morris GS, Matthews CE. Exercise is medicine in oncology: engaging clinicians to help patients move through cancer. CA Cancer J Clin. 2019;69(6):468–484. doi: 10.3322/caac.21579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stevinson C, Capstick V, Schepansky A, Tonkin K, Vallance JK, Ladha AB, Courneya KS. Physical activity preferences of ovarian cancer survivors. Psychooncology. 2009;18(4):422–428. doi: 10.1002/pon.1396. [DOI] [PubMed] [Google Scholar]
- Travier N, Velthuis MJ, Steins Bisschop CN, van den Buijs B, Monninkhof EM, Backx F, May AM. Effects of an 18-week exercise programme started early during breast cancer treatment: a randomised controlled trial. BMC Med. 2015;13:121. doi: 10.1186/s12916-015-0362-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trestini I, Sperduti I, Sposito M, Kadrija D, Drudi A, Avancini A, Pilotto S. Evaluation of nutritional status in non-small-cell lung cancer: screening, assessment and correlation with treatment outcome. ESMO Open. 2020 doi: 10.1136/esmoopen-2020-000689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trestini I, Caldart A, Dodi A, Avancini A, Tregnago D, Sartori G, Pilotto S. Body composition as a modulator of response to immunotherapy in lung cancer: time to deal with it. ESMO Open. 2021;6(2):100095. doi: 10.1016/j.esmoop.2021.100095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trestini I, Sperduti I, Caldart A, Bonaiuto C, Fiorio E, Parolin V, Carbognin L. Evidence-based tailored nutrition educational intervention improves adherence to dietary guidelines, anthropometric measures and serum metabolic biomarkers in early-stage breast cancer patients: a prospective interventional study. Breast. 2021;60:6–14. doi: 10.1016/j.breast.2021.08.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weller S, Oliffe JL, Campbell KL. Factors associated with exercise preferences, barriers and facilitators of prostate cancer survivors. Eur J Cancer Care (Engl) 2019;28(5):e13135. doi: 10.1111/ecc.13135. [DOI] [PubMed] [Google Scholar]
- Wong JN, McAuley E, Trinh L. Physical activity programming and counseling preferences among cancer survivors: a systematic review. Int J Behav Nutr Phys Act. 2018;15(1):48. doi: 10.1186/s12966-018-0680-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang X, Tang T, Pang L, Sharma SV, Li R, Nyitray AG, Edwards BJ. Malnutrition and overall survival in older adults with cancer: a systematic review and meta-analysis. J Geriatr Oncol. 2019;10(6):874–883. doi: 10.1016/j.jgo.2019.03.002. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.