Abstract
Purpose
Immune checkpoint inhibitors (ICI) are a promising treatment, but may cause hyperprogressive disease and early death. The present study investigated early mortality factors in ICI monotherapy for lung cancer.
Patients and methods
We retrospectively reviewed all patients diagnosed with advanced or metastatic non-small cell lung cancer (NSCLC) and treated with ICI monotherapy (nivolumab, pembrolizumab, and atezolizumab) between March 2016 and August 2021 at National Hospital Organization Kochi Hospital and Tokushima University. Early death was defined as patients who died within 60 days of ICI treatment.
Results
A total of 166 patients were included. The majority of patients (87%) had an Eastern cooperative oncology group (ECOG) Performance status (PS) of 0/1. There were 21 early deaths. Significant differences were observed in ECOG PS, the histological type, liver metastasis, tumor size, the white blood cell count, neutrophils (%), lymphocytes (%), the neutrophil-to-lymphocyte ratio in serum (sNLR), C-reactive protein (CRP), and albumin between the groups with or without early death. Univariate logistic regression analyses identified ECOG PS score ≥ 2, liver metastasis, tumor size ≥ 5 cm, neutrophils ≥ 69%, lymphocytes < 22%, sNLR ≥ 4, CRP ≥ 1 mg/dl, and albumin < 3.58 g/dl as significant risk factors for early death. A multivariate logistic regression analysis revealed that liver metastasis (Odds ratio [OR], 10.3; p = 0.008), ECOG PS score ≥ 2 (OR, 8.0; p = 0.007), and a smoking history (OR, 0.1; p = 0.03) were significant risk factors for early death.
Conclusion
Liver metastases, ECOG PS score ≥ 2, and a non-smoking history are early mortality factors in ICI monotherapy for advanced or metastatic NSCLC.
Keywords: Early death, Early mortality factors, Immune checkpoint inhibitors, Lung cancer, Monotherapy
Introduction
Immune checkpoint inhibitors (ICI) have caused a paradigm shift in the treatment of various cancer types. ICI monotherapy has been used as a first-line treatment for advanced or metastatic untreated non-small cell lung cancer (NSCLC) (Reck et al. 2016; Mok et al. 2019). Among patients with advanced, previously treated NSCLC, overall survival (OS) was shown to be significantly better with ICI monotherapy than with chemotherapy (Borghaei et al. 2015; Brahmer et al. 2015; Herbst et al. 2016; Rittmeyer et al. 2017). On the other hand, ICI have been implicated in the development of hyperprogressive disease (HPD) and early death (Champiat et al. 2017; Abbar et al. 2021; Ferrara et al. 2018). Although the risk factors for and mechanisms underlying HPD have been examined (Lo Russo et al. 2019; Kim et al. 2020, 2021; Castello et al. 2020; Passaro et al. 2021; Chen et al. 2021; Ku et al. 2021; Kamada et al. 2019), current knowledge is still insufficient. The selection of suitable patients for ICI monotherapy is essential. OS is the most robust indicator of cancer treatment outcomes. On the other hand, few studies have examined early mortality factors in ICI monotherapy for advanced or metastatic lung cancer in real-world settings. To the best of our knowledge, only one study investigated early death in real-world settings (Inoue et al. 2018). Therefore, the present study attempted to identify early mortality factors in ICI monotherapy for advanced or metastatic NSCLC.
Materials and methods
Patients
We retrospectively reviewed all patients diagnosed with advanced or metastatic NSCLC and treated with ICI monotherapy (nivolumab, pembrolizumab, and atezolizumab) between March 2016 and August 2021 at National Hospital Organization Kochi Hospital and Tokushima University. Early death was defined as patients who died within 60 days of ICI treatment.
Data collection
We collected data on age, sex, smoking history, the Eastern cooperative oncology group performance scale (ECOG PS), white blood cell count, neutrophil count, lymphocyte count, eosinophil count, C-reactive protein (CRP), albumin, histological type, the genotypes of mutations, type of ICI, line of ICI, date of ICI initiation, number of ICI administered, and the status of death. We also examined data obtained on the primary lesion size (maximum diameter measured on chest computed tomography), the number of metastatic sites (count of involved solid organs, not all sites), the status of specific metastasis (non-regional lymph nodes, contralateral lung, pleura, brain, liver, kidney, adrenal gland, and bone), and stage (according to the eighth edition of the tumor-node-metastasis [TNM] staging system). A computed tomography scan was performed for a radiological evaluation before ICI therapy.
Statistical analysis
Categorical and continuous variables were summarized using descriptive statistics. The independent samples t test was used to test for differences between continuous variables. Pearson’s chi-squared test and Fisher’s exact test were employed to analyze relationships between categorical variables. Univariate and multivariate logistic regression analyses were performed to identify predictive factors associated with early death with ICI. The neutrophil-to-lymphocyte ratio in serum (sNLR) was calculated as the ratio of the neutrophil count to the lymphocyte count. The cut-off value for sNLR was set at 4 based on previous studies (Kim et al. 2019b; Sacdalan et al. 2018). We conducted all statistical analyses using SPSS statistics version 27.0 (IBM, Armonk, USA). P values are presented without adjustments for multiple comparisons in an exploratory manner.
Results
Patient characteristics
A total of 166 advanced or metastatic NSCLC patients treated with ICI monotherapy were included in the present study. Twenty-one out of 166 patients (13%) died within 60 days. Twenty patients (95%) died early due to the rapid progression of lung cancer. One patient (5%) died of immune-related adverse events, including interstitial lung disease. No patient died from exacerbation of complications or other diseases. The clinical characteristics of enrolled patients are summarized in Table 1. Among 166 patients, mean age at ICI therapy was 69 years, 128 (77%) were male, and 130 (78%) were ex- or current smokers. Most patients (87%) had ECOG PS of 0–1. Twenty-one patients (13%) had postoperative recurrence, 31 (17%) had stage III, and 114 (69%) had stage IV. Ninety-eight patients (59%) exhibited adenocarcinoma histology, while 45 (27%) showed squamous cell carcinoma histology. The expression of PD-L1 was higher than 1% in 80% (132/166) of patients, but was absent in 19% (31/166). Six patients (4%) were harboring epidermal growth factor receptor (EGFR) mutations, two (1%) had the anaplastic lymphoma kinase (ALK) rearrangement, and one each (1%) harbored rearranged during transfection (RET) and ROS proto-oncogene 1 (ROS1) fusion. Forty-five patients (27%) received ICI as the first-line therapy. Sixteen patients (10%) had liver metastasis.
Table 1.
Characteristics of the study population
| Total | ≤ 60 days | > 60 days | p | ||
|---|---|---|---|---|---|
| n = 166 | n = 21 | n = 145 | |||
| Age, y | Mean age, (SD) | 69 (9) | 68 (10) | 69 (9) | 0.9* |
| Sex, n (%) | Male | 128(77) | 14 (67) | 114 (79) | 0.2*** |
| Female | 38 (23) | 7 (33) | 31 (21) | ||
| Smoking history, n (%) | Yes | 130 (78) | 13 (61) | 117 (81) | 0.1*** |
| No | 30 (18) | 6 (29) | 24 (17) | ||
| Missing | 6 (4) | 2 (10) | 4(3) | ||
| ECOG PS, n (%) | 0–1 | 145 (87) | 10 (48) | 135 (93) | 0.001*** |
| 2–4 | 21 (13) | 11 (52) | 10 (7) | ||
| Stage, n (%) | Recurrence | 21 (13) | 2 (10) | 19 (13) | 0.4*** |
| III | 31 (17) | 2 (10) | 29 (20) | ||
| IV | 114 (69) | 17 (81) | 97 (67) | ||
| Histological type, n (%) | Adeno | 98 (59) | 16 (76) | 82 (57) | 0.047*** |
| Squamous | 45 (27) | 1 (5) | 44 (30) | ||
| Others | 23 (14) | 4 (19) | 19 (13) | ||
| PD-L1, n (%) | < 1% | 31 (19) | 5 (24) | 26 (18) | 0.7*** |
| ≥ 1% | 132 (80) | 16 (76) | 116 (80) | ||
| Missing | 3 (2) | 0 (0) | 3 (3) | ||
| Driver mutation, n (%) | None | 151 (91) | 19 (90) | 132 (91) | 0.6*** |
| EGFR | 6 (4) | 2 (10) | 4 (3) | ||
| ALK | 2 (1) | 0(0) | 2(1) | ||
| RET | 1 (1) | 0 (0) | 1 (1) | ||
| ROS1 | 1(1) | 0 (0) | 1 (1) | ||
| Missing | 5 (3) | 0 (0) | 5 (3) | ||
| Treatment line, n (%) | 1 | 45 (27) | 4 (19) | 41 (28) | 0.4** |
| > 1 | 121 (73) | 17 (81) | 104 (72) | ||
| ICI drug, n (%) | Pembrolizumab | 130 (78) | 17 (81) | 113 (78) | 0.9*** |
| Nivolumab | 35 (21) | 4 (19) | 31 (21) | ||
| Atezolizumab | 1 (1) | 0 (0) | 1 (1) | ||
| Liver metastasis, n (%) | No | 150 (90) | 14 (76) | 136 (96) | < 0.001*** |
| Yes | 16 (10) | 7 (24) | 9 (4) | ||
| Tumor size, mm | Mean, (SD) | 43.7 (24.1) | 56.3 (25.2) | 41.8 (23.5) | 0.01* |
| White blood count, /μL | Mean, (SD) | 7229 (3404) | 9905 (4657) | 6842 (3011) | 0.01* |
| Neutrophils, % | Mean, (SD) | 67 (10) | 72 (8) | 66 (10) | 0.004* |
| Lymphocytes, % | Mean, (SD) | 22 (9) | 15 (5) | 23 (9) | < 0.001* |
| sNLR, ratio | Mean, (SD) | 3.9 (2.5) | 5.6 (2.5) | 3.7 (2.4) | < 0.001* |
| Eosinophils, % | Mean, (SD) | 3 (3) | 4 (5) | 3 (3) | 0.5* |
| CRP, mg/dL | Mean, (SD) | 2.6 (3.4) | 4.9 (4.9) | 2.3 (3.1) | 0.03* |
| Alb, g/dL | Mean, (SD) | 3.5 (0.7) | 2.7 (0.7) | 3.6 (0.6) | < 0.001* |
ECOG PS Eastern cooperative oncology group performance status, PD-L1 programmed death ligand 1, EGFR epidermal growth factor receptor, ALK anaplastic lymphoma kinase, RET rearrangement during transfection, ROS1 ROS proto-oncogene 1, ICI immune checkpoint inhibitor, sNLR the neutrophil-to-lymphocyte ratio in serum, CRP C-reactive protein, Alb albumin
*Independent samples t test, **Fisher’s exact test, ***Chi-squared test
Among the 21 patients who died early, mean age was 68 years, 14 (67%) were male, and thirteen (61%) were ex- or current smokers. Ten patients (48%) had an ECOG PS of 0–1. Two patients (10%) had postoperative recurrence, two (10%) had stage III, and 17 (81%) had stage IV. Sixteen patients (76%) showed adenocarcinoma histology, and one (5%) exhibited squamous cell carcinoma histology. The expression of PD-L1 was higher than 1% in 76% (16/21) of patients, but was absent in 24% (5/21). Two patients (10%) harbored EGFR mutations. Four patients (19%) received ICI as the first-line therapy, and seven (24%) had liver metastasis.
Among the 145 patients without early death, mean age was 69 years, 114 (79%) were male, and 117 (81%) were ex- or current smokers. One hundred and thirty-five patients (93%) had an ECOG PS of 0–1. Nineteen patients (13%) had postoperative recurrence, 29 (20%) had stage III, and 97 (67%) had stage IV. Eighty-two patients (57%) exhibited adenocarcinoma histology, and 44 (30%) showed squamous cell carcinoma histology. The expression of PD-L1 was higher than 1% in 80% of patients (116/145), but was absent in 18% (26/145). Four patients (3%) harbored EGFR mutations, two (1%) had the ALK rearrangement, and one each (1%) harbored RET and ROS1 fusion. Forty-one patients (28%) received ICI as the first-line therapy. Nine (4%) had liver metastasis.
Significant differences were observed in ECOG PS, the histological type, liver metastasis, tumor size, the white blood count, neutrophils (%), lymphocytes (%), sNLR, CRP, and albumin between the groups with or without early death.
Univariate analysis of factors associated with early death
As shown in Table 2, univariate logistic regression analyses identified ECOG PS score ≥ 2 (Odds ratio [OR], 14.9; 95% CI, 5.1–43.3; p < 0.001), liver metastasis (OR, 7.6; 95% CI, 2.4–23.4; p < 0.001), tumor size ≥ 5 cm (OR, 2.6; 95% CI, 1.03–6.6; p = 0.04), neutrophils ≥ 69% (OR, 3.5; 95% CI, 1.3–9.7; p = 0.01), lymphocytes < 22% (OR, 10.1; 95% CI, 2.3–45.3; p = 0.002), sNLR ≥ 4 (OR, 4.2; 95% CI, 1.6–11; p = 0.004), CRP ≥ 1 mg/dl (OR, 6.6; 95% CI, 1.9–23.4; p = 0.003), and Alb < 3.58 g/dl (OR, 3.5; 95% CI, 1.5–7.7; p = 0.002) as significant risk factors for early death during ICI monotherapy.
Table 2.
Univariate analysis of factors associated with early death
| Variable | Category | Univariate analysis Odds ratio (95% CI) |
p value |
|---|---|---|---|
| Smoking history | No | 1 | 0.1 |
| Yes | 0.4 (0.2–1.3) | ||
| ECOG PS | 0–1 | 1 | < 0.001 |
| 2–4 | 14.9 (5.1–43.3) | ||
| Histological type | Non-adeno | 1 | 0.1 |
| Adeno | 2.5 (0.9–7.1) | ||
| PD-L1 | ≥ 1% | 1 | 0.7 |
| < 1% | 1.3 (0.4–3.7) | ||
| Liver metastasis | No | 1 | < 0.001 |
| Yes | 7.6 (2.4–23.4) | ||
| Tumor size | < 5 cm | 1 | 0.04 |
| ≥ 5 cm | 2.6 (1.03–6.6) | ||
| Neutrophils | < 69% | 1 | 0.01 |
| ≥ 69% | 3.5 (1.3–9.7) | ||
| Lymphocytes | ≥ 22% | 1 | 0.002 |
| < 22% | 10.1 (2.3–45.3) | ||
| sNLR | < 4 | 1 | 0.004 |
| ≥ 4 | 4.2 (1.6–11) | ||
| Eosinophils | ≥ 1.5% | 1 | 0.08 |
| < 1.5% | 2.3 (0.9–5.8) | ||
| CRP | < 1 mg/dl | 1 | 0.003 |
| ≥ 1 mg/dl | 6.6 (1.9–23.4) | ||
| Alb | ≥ 3.58 g/dl | 1 | 0.002 |
| < 3.58 g/dl | 3.5 (1.5–7.7) |
p values <0.05 are bolded
ECOG PS Eastern cooperative oncology group performance status, adeno, adenocarcinoma, PD-L1 programmed death ligand 1, sNLR the neutrophil-to-lymphocyte ratio in serum, CRP C-reactive protein, Alb albumin
Multivariate analysis of factors associated with early death
A multivariate logistic regression analysis for the prediction of early death was performed using the following variables: a smoking history, ECOG PS, the histological type, liver metastasis, tumor size, neutrophils, lymphocytes, sNLR, CRP, and albumin (Table 3). A multivariate analysis with a multivariate logistic regression model showed that liver metastasis (OR, 10.3; 95% CI, 1.8–58; p = 0.008), ECOG PS score ≥ 2 (OR, 8.0; 95% CI, 1.8–36; p = 0.007), and a smoking history (OR, 0.1; 95% CI, 0.01–0.8; p = 0.03) were significant risk factors for early death during ICI monotherapy.
Table 3.
Multivariate analysis of factors associated with early death
| Variable | Category | Multivariate analysis Odds ratio (95% CI) |
p value |
|---|---|---|---|
| Smoking history | No | 1 | 0.03 |
| Yes | 0.1(0.01–0.8) | ||
| ECOG PS | 0–1 | 1 | 0.007 |
| 2–4 | 8.0 (1.8–36) | ||
| Histological type | Non-adeno | 1 | 0.1 |
| Adeno | 3.7 (0.7–18.8) | ||
| Liver metastasis | No | 1 | 0.008 |
| Yes | 10.3 (1.8–58) | ||
| Tumor size | < 5 cm | 1 | 0.3 |
| ≥ 5 cm | 2.5 (0.5–12.5) | ||
| Neutrophils | < 69% | 1 | 0.7 |
| ≥ 69% | 0.7 (0.1–4.5) | ||
| Lymphocytes | ≥ 22% | 1 | 0.3 |
| < 22% | 3.5 (0.3–40) | ||
| sNLR | < 4 | 1 | 0.8 |
| ≥ 4 | 1.1 (0.2–6.8) | ||
| CRP | < 1 mg/dl | 1 | 0.2 |
| ≥ 1 mg/dl | 5.0 (0.5–48.6) | ||
| Alb | ≥ 3.58 g/dl | 1 | 0.1 |
| < 3.58 g/dl | 2.9 (0.7–11.4) |
p values <0.05 are bolded
ECOG PS Eastern cooperative oncology group performance status, adeno, adenocarcinoma, sNLR the neutrophil-to-lymphocyte ratio in serum, CRP C-reactive protein, Alb albumin
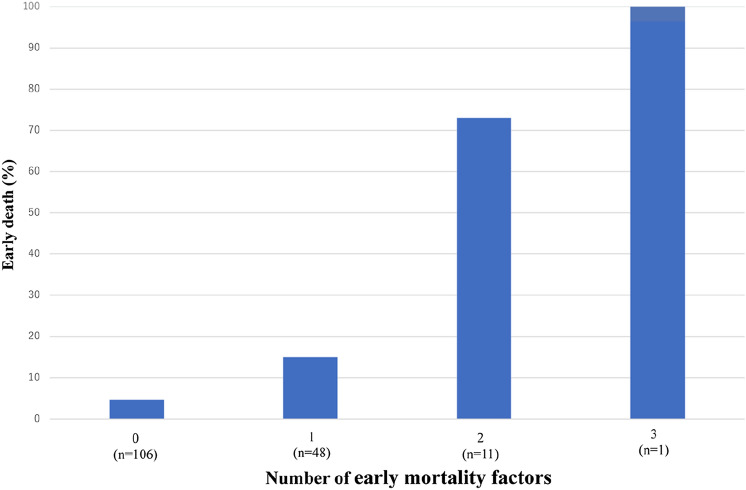
The number of early mortality factors and percentage of early deaths
Liver metastases, ECOG PS score ≥ 2, and a non-smoking history were early mortality factors in ICI monotherapy against advanced or metastatic NSCLC. The number of early mortality factors and percentage of early deaths are shown in Fig. 1. One patient had all three early mortality factors, and the early mortality rate was 100%. Eight out of 11 patients (73%) with two early mortality factors and 7 out of 48 patients (15%) with one early mortality factor died within 60 days. Furthermore, 5 out of 106 patients (4.7%) with no early mortality factors died early.
Fig. 1.
The number of early mortality factors and percentage of early deaths
Discussion
The present results identified liver metastases, ECOG PS score ≥ 2, and a non-smoking history as early mortality factors in ICI monotherapy for advanced or metastatic NSCLC. Few studies have examined early mortality factors in ICI monotherapy for advanced or metastatic lung cancer in real-world settings. To the best of our knowledge, only one study investigated early mortality in real-world settings (Inoue et al. 2018).
ICI has caused a paradigm shift in the treatment of various cancer types, but have also been implicated in the development of HPD and early patient death (Champiat et al. 2017; Abbar et al. 2021; Ferrara et al. 2018). Although the risk factors for and mechanisms underlying HPD have been examined (Lo Russo et al. 2019; Kim et al. 2020, 2021; Castello et al. 2020; Passaro et al. 2021; Chen et al. 2021; Ku et al. 2021; Kamada et al. 2019), current knowledge is still inadequate. Although PD-L1 and the tumor mutation burden (TMB) have been used as predictive biomarkers for ICI therapy, there are currently no established predictive biomarkers (Brody et al. 2017; Zou et al. 2021). The selection of suitable patients for ICI monotherapy is essential in real-world settings, and OS is the most robust indicator of cancer treatment outcomes. Therefore, we examined early mortality factors in ICI monotherapy for advanced or metastatic NSCLC in real-world settings.
ECOG PS ≥ 2, a pretreatment CRP-to-albumin ratio > 0.3, and poor responses to previous treatment have been associated with early death in ICI monotherapy (Inoue et al. 2018). In the present study, a multivariate analysis with a multivariate logistic regression model identified liver metastasis (OR, 10.3; 95% CI, 1.8–58; p = 0.008), ECOG PS score ≥ 2 (OR, 8.0; 95% CI, 1.8–36; p = 0.007), and a smoking history (OR, 0.1; 95% CI, 0.01–0.8; p = 0.03) as risk factors for early death in ICI monotherapy. ECOG PS ≥ 2 is frequently identified as an early mortality factor.
In a system review, liver metastasis, previous metastatic sites > 2, a Royal Marsden Hospital score ≥ 2, higher ECOG PS, and a lactate dehydrogenase (LDH) level greater than the normal upper limit were associated with the development of HPD (Chen et al. 2020). In another system review, liver metastasis, LDH, more than two metastatic sites, and a Royal Marsden Hospital score of 2 or higher correlated with HPD (Kim et al. 2019a). A previous study also implicated hepatic metastases, more than two metastatic sites, ECOG PS ≥ 2, and an LDH level higher than the normal upper limit in the development of HPD (Chen et al. 2021). Furthermore, liver metastasis was the only variable associated with HPD in a multivariate analysis (Abbar et al. 2021). The presence of tumors within the liver was previously shown to significantly reduce systemic tumor-specific immunity, and immunosuppression was dependent on regulatory T cells (Tregs) (Lee et al. 2020). The number of circulating Tregs was significantly higher than the baseline in patients with HPD (Kang et al. 2022). The balanced expression of PD-1 between CD8 + T cells and Tregs is essential in programmed cell death 1 (PD-1) inhibitory immunotherapy (Aksoylar and Boussiotis 2020; Kumagai et al. 2020). In a mouse model of liver metastasis due to colorectal cancer, Treg numbers were elevated, and the hepatocyte growth factor/hepatocyte growth factor receptor signaling pathway was up-regulated (Huang et al. 2020). Lactic acid has been shown to promote the expression of PD-1 in Tregs in highly glycolytic tumors, such as liver metastases (Kumagai et al. 2022).
In the present study, a non-smoking history was a significant early mortality factor. OR was 0.1 (P = 0.03) when six patients with a missing smoking history were assumed to have a smoking history and 0.2 (P = 0.03) when they were hypothesized to have a non-smoking history, indicating that non-smoking history is a risk factor regardless of the presence or absence of missing data. The reason for this result is not apparent. In adenocarcinoma, a non-smoking status was predictive of a low TMB (Sharpnack et al. 2020). The probability of EGFR mutations and ALK rearrangements was previously shown to be significantly higher in patients with a non-smoking history than in those with a smoking history (Chapman et al. 2016). Furthermore, TMB was lower in EGFR-mutant lung cancer than in EGFR wild-type lung cancer (Offin et al. 2019). EGFR mutations and ALK rearrangements are generally associated with low response rates to ICI therapy in NSCLC (Gainor et al. 2016; Lee et al. 2017). Therefore, ICI may be less effective in patients with a non-smoking history. However, in the present study, only 2 out of 21 patients who died early had EGFR mutations and no ALK rearrangement. No significant differences were observed in genetic abnormalities between the groups with and without early death (Table 1). Specific molecular alterations (MDM2/MDM4, KRAS, and serine/threonine kinase 11) have been associated with HPD (Kato et al. 2017; Kim et al. 2019b). Therefore, these genetic abnormalities in patients with a non-smoking history may be related; however, only a few have been examined to date.
The limitation of the present study is that it was a two-center retrospective analysis conducted with heterogeneous data from patient cohorts. Therefore, the results obtained are speculative and not definitive. Furthermore, since the number of cases was small, OR was significant, but CI was also large; therefore, reliability was not high. The frequency of early death within 60 days of ICI monotherapy for advanced or metastatic NSCLC was not high. Therefore, multicenter studies are needed. Although we need to consider these limitations when interpreting the present results, this study is of value because early mortality factors in ICI monotherapy against advanced or metastatic NSCLC were identified in real-world settings. Simple clinical parameters may easily predict early mortality with single-agent ICI against advanced or metastatic NSCLC and are clinically useful. Early mortality rates were 73 and 100% in patients with 2 and 3 early mortality factors, respectively. ICI-based combination therapy may be a better treatment option for patients with multiple mortality factors than ICI monotherapy for advanced or metastatic NSCLC.
Conclusion
Liver metastases, ECOG PS score ≥ 2, and a non-smoking history were identified as early mortality factors in ICI monotherapy against advanced or metastatic NSCLC.
Acknowledgements
We thank Hiroki Yoshida (Data Seed Inc.) for statistical advice.
Abbreviations
- ALK
Anaplastic lymphoma kinase
- CI
Confidence interval
- CRP
C-reactive protein
- ECOG
Eastern Cooperative oncology group
- EGFR
Epidermal growth factor receptor
- HPD
Hyperprogressive disease
- ICI
Immune checkpoint inhibitors
- LDH
Lactate dehydrogenase
- NSCLC
Non-small cell lung cancer
- PD-1
Programmed cell death 1
- PD-L1
Programmed death ligand 1
- OR
Odds ratio
- OS
Overall survival
- PS
Performance status
- RET
Rearranged during transfection
- ROS1
ROS proto-oncogene 1
- Tregs
Regulatory T cells
- sNLR
Neutrophil-to-lymphocyte ratio in serum
- TMB
Tumor mutation burden
Author contributions
Conceptualization and design: ET, HN; data collection and analysis: KK, YO, MK, YK, NK, HM, NH, TS, KN, HO; formal analysis and investigation: HM, ET; writing–original draft preparation: ET; writing–review and editing: HN, YN]; supervision: TS, HN, YN. All authors read and approved the final manuscript.
Funding
None.
Data availability
None.
Code availability
None.
Declarations
Competing interests
The authors declare no competing interests.
Conflict of interest
The authors declare no conflicts of interest.
Ethical approval
The Institutional Review Board of the National Hospital Organization Kochi Hospital (R3-3) and Tokushima University (3013–3) approved the study protocol. Informed consent was waived because of the retrospective nature of the study.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Eiji Takeuchi, Email: eijitake1964@icloud.com.
Kensuke Kondo, Email: sdt4260@gmail.com.
Yoshio Okano, Email: okano.yoshio.mu@mail.hosp.go.jp.
Michihiro Kunishige, Email: michi6910@gmail.com.
Yoshihiro Kondo, Email: bontakundog@gmail.com.
Naoki Kadota, Email: naoki_kadota.usa@kta.biglobe.ne.jp.
Hisanori Machida, Email: hsnr1967mcd@gmail.com.
Nobuo Hatakeyama, Email: nobuoh1965@gmail.com.
Keishi Naruse, Email: naruse.keishi.hj@mail.hosp.go.jp.
Hirokazu Ogino, Email: ogino@tokushima-u.ac.jp.
Hiroshi Nokihara, Email: hnokihara@hosp.ncgm.go.jp.
Tsutomu Shinohara, Email: shinohara_fau@yahoo.co.jp.
Yasuhiko Nishioka, Email: yasuhiko@tokushima-u.ac.jp.
References
- Abbar B, De Castelbajac V, Gougis P, Assoun S, Pluvy J, Tesmoingt C, Théou-Anton N, Cazes A, Namour C, Khalil A, Gounant V, Besse B, Zalcman G, Brosseau S. Definitions, outcomes, and management of hyperprogression in patients with non-small-cell lung cancer treated with immune checkpoint inhibitors. Lung Cancer. 2021;152:109–118. doi: 10.1016/j.lungcan.2020.12.026. [DOI] [PubMed] [Google Scholar]
- Aksoylar HI, Boussiotis VA. PD-1(+) T(reg) cells: a foe in cancer immunotherapy? Nat Immunol. 2020;21:1311–1312. doi: 10.1038/s41590-020-0801-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borghaei H, Paz-Ares L, Horn L, Spigel DR, Steins M, Ready NE, Chow LQ, Vokes EE, Felip E, Holgado E, Barlesi F, Kohlhaufl M, Arrieta O, Burgio MA, Fayette J, Lena H, Poddubskaya E, Gerber DE, Gettinger SN, Rudin CM, Rizvi N, Crino L, Blumenschein GR, Jr, Antonia SJ, Dorange C, Harbison CT, Graf Finckenstein F, Brahmer JR. Nivolumab versus docetaxel in advanced nonsquamous non-small-cell lung cancer. N Engl J Med. 2015;373:1627–1639. doi: 10.1056/NEJMoa1507643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brahmer J, Reckamp KL, Baas P, Crino L, Eberhardt WE, Poddubskaya E, Antonia S, Pluzanski A, Vokes EE, Holgado E, Waterhouse D, Ready N, Gainor J, Aren Frontera O, Havel L, Steins M, Garassino MC, Aerts JG, Domine M, Paz-Ares L, Reck M, Baudelet C, Harbison CT, Lestini B, Spigel DR. Nivolumab versus docetaxel in advanced squamous-cell non-small-cell lung cancer. N Engl J Med. 2015;373:123–135. doi: 10.1056/NEJMoa1504627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brody R, Zhang Y, Ballas M, Siddiqui MK, Gupta P, Barker C, Midha A, Walker J. PD-L1 expression in advanced NSCLC: insights into risk stratification and treatment selection from a systematic literature review. Lung Cancer. 2017;112:200–215. doi: 10.1016/j.lungcan.2017.08.005. [DOI] [PubMed] [Google Scholar]
- Castello A, Rossi S, Mazziotti E, Toschi L, Lopci E. Hyperprogressive disease in patients with non-small cell lung cancer treated with checkpoint inhibitors: the role of (18) F-FDG PET/CT. J Nucl Med. 2020;61:821–826. doi: 10.2967/jnumed.119.237768. [DOI] [PubMed] [Google Scholar]
- Champiat S, Dercle L, Ammari S, Massard C, Hollebecque A, Postel-Vinay S, Chaput N, Eggermont A, Marabelle A, Soria JC, Ferte C. Hyperprogressive disease is a new pattern of progression in cancer patients treated by anti-PD-1/PD-L1. Clin Cancer Res. 2017;23:1920–1928. doi: 10.1158/1078-0432.CCR-16-1741. [DOI] [PubMed] [Google Scholar]
- Chapman AM, Sun KY, Ruestow P, Cowan DM, Madl AK. Lung cancer mutation profile of EGFR, ALK, and KRAS: meta-analysis and comparison of never and ever smokers. Lung Cancer. 2016;102:122–134. doi: 10.1016/j.lungcan.2016.10.010. [DOI] [PubMed] [Google Scholar]
- Chen Y, Hu J, Bu F, Zhang H, Fei K, Zhang P. Clinical characteristics of hyperprogressive disease in NSCLC after treatment with immune checkpoint inhibitor: a systematic review and meta-analysis. BMC Cancer. 2020;20:707. doi: 10.1186/s12885-020-07206-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen S, Gou M, Yan H, Fan M, Pan Y, Fan R, Qian N, Dai G. Hyperprogressive disease caused by PD-1 inhibitors for the treatment of pan-cancer. Dis Markers. 2021;2021:6639366. doi: 10.1155/2021/6639366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferrara R, Mezquita L, Texier M, Lahmar J, Audigier-Valette C, Tessonnier L, Mazieres J, Zalcman G, Brosseau S, Le Moulec S, Leroy L, Duchemann B, Lefebvre C, Veillon R, Westeel V, Koscielny S, Champiat S, Ferté C, Planchard D, Remon J, Boucher ME, Gazzah A, Adam J, Bria E, Tortora G, Soria JC, Besse B, Caramella C. Hyperprogressive disease in patients with advanced non-small cell lung cancer treated with PD-1/PD-L1 inhibitors or with single-agent chemotherapy. JAMA Oncol. 2018;4:1543–1552. doi: 10.1001/jamaoncol.2018.3676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gainor JF, Shaw AT, Sequist LV, Fu X, Azzoli CG, Piotrowska Z, Huynh TG, Zhao L, Fulton L, Schultz KR, Howe E, Farago AF, Sullivan RJ, Stone JR, Digumarthy S, Moran T, Hata AN, Yagi Y, Yeap BY, Engelman JA, Mino-Kenudson M. EGFR mutations and ALK rearrangements are associated with low response rates to PD-1 pathway blockade in non-small cell lung cancer: a retrospective analysis. Clin Cancer Res. 2016;22:4585–4593. doi: 10.1158/1078-0432.CCR-15-3101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herbst RS, Baas P, Kim DW, Felip E, Pérez-Gracia JL, Han JY, Molina J, Kim JH, Arvis CD, Ahn MJ, Majem M, Fidler MJ, de Castro G, Jr., Garrido M, Lubiniecki GM, Shentu Y, Im E, Dolled-Filhart M, Garon EB, Pembrolizumab versus docetaxel for previously treated, PD-L1-positive, advanced non-small-cell lung cancer (KEYNOTE-010): a randomised controlled trial. Lancet. 2016;387:1540–1550. doi: 10.1016/S0140-6736(15)01281-7. [DOI] [PubMed] [Google Scholar]
- Huang X, Chen Z, Zhang N, Zhu C, Lin X, Yu J, Chen Z, Lan P, Wan Y. Increase in CD4(+)FOXP3(+) regulatory T cell number and upregulation of the HGF/c-Met signaling pathway during the liver metastasis of colorectal cancer. Oncol Lett. 2020;20:2113–2118. doi: 10.3892/ol.2020.11785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Inoue T, Tamiya M, Tamiya A, Nakahama K, Taniguchi Y, Shiroyama T, Isa SI, Nishino K, Kumagai T, Kunimasa K, Kimura M, Suzuki H, Hirashima T, Atagi S, Imamura F. Analysis of early death in japanese patients with advanced non-small-cell lung cancer treated with nivolumab. Clin Lung Cancer. 2018;19:e171–e176. doi: 10.1016/j.cllc.2017.09.002. [DOI] [PubMed] [Google Scholar]
- Kamada T, Togashi Y, Tay C, Ha D, Sasaki A, Nakamura Y, Sato E, Fukuoka S, Tada Y, Tanaka A, Morikawa H, Kawazoe A, Kinoshita T, Shitara K, Sakaguchi S, Nishikawa H. PD-1(+) regulatory T cells amplified by PD-1 blockade promote hyperprogression of cancer. Proc Natl Acad Sci U S A. 2019;116:9999–10008. doi: 10.1073/pnas.1822001116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kang DH, Chung C, Sun P, Lee DH, Lee SI, Park D, Koh JS, Kim Y, Yi HS, Lee JE. Circulating regulatory T cells predict efficacy and atypical responses in lung cancer patients treated with PD-1/PD-L1 inhibitors. Cancer Immunol Immunother. 2022;71:579–588. doi: 10.1007/s00262-021-03018-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kato S, Goodman A, Walavalkar V, Barkauskas DA, Sharabi A, Kurzrock R. Hyperprogressors after immunotherapy: analysis of genomic alterations associated with accelerated growth rate. Clin Cancer Res. 2017;23:4242–4250. doi: 10.1158/1078-0432.CCR-16-3133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim JY, Lee KH, Kang J, Borcoman E, Saada-Bouzid E, Kronbichler A, Hong SH, de Rezende LFM, Ogino S, Keum N, Song M, Luchini C, van der Vliet HJ, Shin JI, Gamerith G. Hyperprogressive disease during anti-PD-1 (PDCD1)/PD-L1 (CD274) Therapy: a systematic review and meta-analysis. Cancers (basel). 2019;11(11):1699. doi: 10.3390/cancers11111699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim Y, Kim CH, Lee HY, Lee SH, Kim HS, Lee S, Cha H, Hong S, Kim K, Seo SW, Sun JM, Ahn MJ, Ahn JS, Park K. Comprehensive clinical and genetic characterization of hyperprogression based on volumetry in advanced non-small cell lung cancer treated with immune checkpoint inhibitor. J Thorac Oncol. 2019;14:1608–1618. doi: 10.1016/j.jtho.2019.05.033. [DOI] [PubMed] [Google Scholar]
- Kim KH, Hur JY, Koh J, Cho J, Ku BM, Koh JY, Sun JM, Lee SH, Ahn JS, Park K, Ahn MJ, Shin EC. Immunological characteristics of hyperprogressive disease in patients with non-small cell lung cancer treated with anti-PD-1/PD-L1 Abs. Immune Netw. 2020;20:e48. doi: 10.4110/in.2020.20.e48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim SR, Chun SH, Kim JR, Kim SY, Seo JY, Jung CK, Gil BM, Kim JO, Ko YH, Woo IS, Shim BY, Hong SH, Kang JH. The implications of clinical risk factors, CAR index, and compositional changes of immune cells on hyperprogressive disease in non-small cell lung cancer patients receiving immunotherapy. BMC Cancer. 2021;21:19. doi: 10.1186/s12885-020-07727-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ku BM, Kim Y, Lee KY, Kim SY, Sun JM, Lee SH, Ahn JS, Park K, Ahn MJ. Tumor infiltrated immune cell types support distinct immune checkpoint inhibitor outcomes in patients with advanced non-small cell lung cancer. Eur J Immunol. 2021;51:956–964. doi: 10.1002/eji.202048966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumagai S, Togashi Y, Kamada T, Sugiyama E, Nishinakamura H, Takeuchi Y, Vitaly K, Itahashi K, Maeda Y, Matsui S, Shibahara T, Yamashita Y, Irie T, Tsuge A, Fukuoka S, Kawazoe A, Udagawa H, Kirita K, Aokage K, Ishii G, Kuwata T, Nakama K, Kawazu M, Ueno T, Yamazaki N, Goto K, Tsuboi M, Mano H, Doi T, Shitara K, Nishikawa H. The PD-1 expression balance between effector and regulatory T cells predicts the clinical efficacy of PD-1 blockade therapies. Nat Immunol. 2020;21:1346–1358. doi: 10.1038/s41590-020-0769-3. [DOI] [PubMed] [Google Scholar]
- Kumagai S, Koyama S, Itahashi K, Tanegashima T, Lin YT, Togashi Y, Kamada T, Irie T, Okumura G, Kono H, Ito D, Fujii R, Watanabe S, Sai A, Fukuoka S, Sugiyama E, Watanabe G, Owari T, Nishinakamura H, Sugiyama D, Maeda Y, Kawazoe A, Yukami H, Chida K, Ohara Y, Yoshida T, Shinno Y, Takeyasu Y, Shirasawa M, Nakama K, Aokage K, Suzuki J, Ishii G, Kuwata T, Sakamoto N, Kawazu M, Ueno T, Mori T, Yamazaki N, Tsuboi M, Yatabe Y, Kinoshita T, Doi T, Shitara K, Mano H, Nishikawa H. Lactic acid promotes PD-1 expression in regulatory T cells in highly glycolytic tumor microenvironments. Cancer Cell. 2022;40(2):201–218. doi: 10.1016/j.ccell.2022.01.001. [DOI] [PubMed] [Google Scholar]
- Lee CK, Man J, Lord S, Links M, Gebski V, Mok T, Yang JC. Checkpoint inhibitors in metastatic EGFR-mutated non-small cell lung cancer-a meta-analysis. J Thorac Oncol. 2017;12:403–407. doi: 10.1016/j.jtho.2016.10.007. [DOI] [PubMed] [Google Scholar]
- Lee JC, Mehdizadeh S, Smith J, Young A, Mufazalov IA, Mowery CT, Daud A, Bluestone JA. Regulatory T cell control of systemic immunity and immunotherapy response in liver metastasis. Sci Immunol. 2020;5(52):eaba0759. doi: 10.1126/sciimmunol.aba0759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lo Russo G, Moro M, Sommariva M, Cancila V, Boeri M, Centonze G, Ferro S, Ganzinelli M, Gasparini P, Huber V, Milione M, Porcu L, Proto C, Pruneri G, Signorelli D, Sangaletti S, Sfondrini L, Storti C, Tassi E, Bardelli A, Marsoni S, Torri V, Tripodo C, Colombo MP, Anichini A, Rivoltini L, Balsari A, Sozzi G, Garassino MC. Antibody-Fc/FcR interaction on macrophages as a mechanism for hyperprogressive disease in non-small cell lung cancer subsequent to PD-1/PD-L1 blockade. Clin Cancer Res. 2019;25:989–999. doi: 10.1158/1078-0432.CCR-18-1390. [DOI] [PubMed] [Google Scholar]
- Mok TSK, Wu YL, Kudaba I, Kowalski DM, Cho BC, Turna HZ, Castro G, Jr, Srimuninnimit V, Laktionov KK, Bondarenko I, Kubota K, Lubiniecki GM, Zhang J, Kush D, Lopes G. Pembrolizumab versus chemotherapy for previously untreated, PD-L1-expressing, locally advanced or metastatic non-small-cell lung cancer (KEYNOTE-042): a randomised, open-label, controlled, phase 3 trial. Lancet. 2019;393:1819–1830. doi: 10.1016/S0140-6736(18)32409-7. [DOI] [PubMed] [Google Scholar]
- Offin M, Rizvi H, Tenet M, Ni A, Sanchez-Vega F, Li BT, Drilon A, Kris MG, Rudin CM, Schultz N, Arcila ME, Ladanyi M, Riely GJ, Yu H, Hellmann MD. Tumor mutation burden and efficacy of EGFR-tyrosine kinase inhibitors in patients with EGFR-mutant lung cancers. Clin Cancer Res. 2019;25:1063–1069. doi: 10.1158/1078-0432.CCR-18-1102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Passaro A, Novello S, Giannarelli D, Bria E, Galetta D, Gelibter A, Reale ML, Carnio S, Vita E, Stefani A, Pizzutilo P, Stati V, Attili I, de Marinis F. Early progression in non-small cell lung cancer (NSCLC) with high PD-L1 treated with pembrolizumab in first-line setting: a prognostic scoring system based on clinical features. Cancers (basel). 2021;13(12):2935. doi: 10.3390/cancers13122935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reck M, Rodriguez-Abreu D, Robinson AG, Hui R, Csoszi T, Fulop A, Gottfried M, Peled N, Tafreshi A, Cuffe S, O'Brien M, Rao S, Hotta K, Leiby MA, Lubiniecki GM, Shentu Y, Rangwala R, Brahmer JR, Investigators K. Pembrolizumab versus chemotherapy for PD-L1-positive non-small-cell lung cancer. N Engl J Med. 2016;375:1823–1833. doi: 10.1056/NEJMoa1606774. [DOI] [PubMed] [Google Scholar]
- Rittmeyer A, Barlesi F, Waterkamp D, Park K, Ciardiello F, von Pawel J, Gadgeel SM, Hida T, Kowalski DM, Dols MC, Cortinovis DL, Leach J, Polikoff J, Barrios C, Kabbinavar F, Frontera OA, De Marinis F, Turna H, Lee JS, Ballinger M, Kowanetz M, He P, Chen DS, Sandler A, Gandara DR. Atezolizumab versus docetaxel in patients with previously treated non-small-cell lung cancer (OAK): a phase 3, open-label, multicentre randomised controlled trial. Lancet. 2017;389:255–265. doi: 10.1016/S0140-6736(16)32517-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sacdalan DB, Lucero JA, Sacdalan DL. Prognostic utility of baseline neutrophil-to-lymphocyte ratio in patients receiving immune checkpoint inhibitors: a review and meta-analysis. Onco Targets Ther. 2018;11:955–965. doi: 10.2147/OTT.S153290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharpnack MF, Cho JH, Johnson TS, Otterson GA, Shields PG, Huang K, Carbone DP, He K. Clinical and molecular correlates of tumor mutation burden in non-small cell lung cancer. Lung Cancer. 2020;146:36–41. doi: 10.1016/j.lungcan.2020.05.021. [DOI] [PubMed] [Google Scholar]
- Zou XL, Li XB, Ke H, Zhang GY, Tang Q, Yuan J, Zhou CJ, Zhang JL, Zhang R, Chen WY. Prognostic value of neoantigen load in immune checkpoint inhibitor therapy for cancer. Front Immunol. 2021;12:689076. doi: 10.3389/fimmu.2021.689076. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
None.
None.