Abstract
BACKGROUND
Induced pluripotent stem cells (iPSCs) show great ability to differentiate into any tissue, making them attractive candidates for pathophysiological investigations. The rise of organ-on-a-chip technology in the past century has introduced a novel way to make in vitro cell cultures that more closely resemble their in vivo environments, both structural and functionally. The literature still lacks consensus on the best conditions to mimic the blood-brain barrier (BBB) for drug screening and other personalized therapies. The development of models based on BBB-on-a-chip using iPSCs is promising and is a potential alternative to the use of animals in research.
AIM
To analyze the literature for BBB models on-a-chip involving iPSCs, describe the microdevices, the BBB in vitro construction, and applications.
METHODS
We searched for original articles indexed in PubMed and Scopus that used iPSCs to mimic the BBB and its microenvironment in microfluidic devices. Thirty articles were identified, wherein only 14 articles were finally selected according to the inclusion and exclusion criteria. Data compiled from the selected articles were organized into four topics: (1) Microfluidic devices design and fabrication; (2) characteristics of the iPSCs used in the BBB model and their differentiation conditions; (3) BBB-on-a-chip reconstruction process; and (4) applications of BBB microfluidic three-dimensional models using iPSCs.
RESULTS
This study showed that BBB models with iPSCs in microdevices are quite novel in scientific research. Important technological advances in this area regarding the use of commercial BBB-on-a-chip were identified in the most recent articles by different research groups. Conventional polydimethylsiloxane was the most used material to fabricate in-house chips (57%), whereas few studies (14.3%) adopted polymethylmethacrylate. Half the models were constructed using a porous membrane made of diverse materials to separate the channels. iPSC sources were divergent among the studies, but the main line used was IMR90-C4 from human fetal lung fibroblast (41.2%). The cells were differentiated through diverse and complex processes either to endothelial or neural cells, wherein only one study promoted differentiation inside the chip. The construction process of the BBB-on-a-chip involved previous coating mostly with fibronectin/collagen IV (39.3%), followed by cell seeding in single cultures (36%) or co-cultures (64%) under controlled conditions, aimed at developing an in vitro BBB that mimics the human BBB for future applications.
CONCLUSION
This review evidenced technological advances in the construction of BBB models using iPSCs. Nonetheless, a definitive BBB-on-a-chip has not yet been achieved, hindering the applicability of the models.
Keywords: Induced pluripotent stem cells, Cell differentiation, Blood-brain barrier, Neurovascular unit, Organ-on-a-chip, Microfluidic device
Core Tip: This systematic review provided a current perspective on the applicability of induced pluripotent stem cells within blood-brain barrier (BBB)-on-a-chip with high technology advances in commercial chips and promotion of an efficient human neurovascular unit, able to screen for drugs, mimic brain dysfunctions, such as stroke and Huntington’s disease, and suitable for future personalized therapeutic approaches. However, the composition and construction of the BBB models lack consensus in the literature.
INTRODUCTION
Induced pluripotent stem cells (iPSCs) have been largely studied for their numerous applications in drug screening, toxicological studies, cell therapy, and disease modeling[1]. Their relevance may be justified by their resemblance to embryonic stem cells, given their ability to differentiate into any one of the three germ layers: ectoderm, mesoderm, and endoderm[1,2], making them attractive candidates for cell therapy-based regenerative medicine[3].
Different from primary cells, iPSCs are easily attainable and able to mature into almost any desired cell type. In general, they can be formed by reprogramming cells obtained from a tissue biopsy or from more accessible sources such as peripheral blood, renal epithelial cells, or dental pulp[4,5]. These characteristics, as opposed to primary cells, have promoted more reliable models for complex human structures such as the blood-brain barrier (BBB)[6].
The BBB secures the brain with a homeostatic environment, controlling the interaction, communication, and molecular and ion exchange between the central nervous system (CNS) and the peripheral blood[7]. The neurovascular unit (NVU) is generally composed of brain microvascular endothelial cells (BMECs), astrocytes (ACs), and pericytes (PCs), being directly involved in regulating CNS blood flow, and consequently, neuronal activity[8].
Recreating the BBB microenvironment in vitro allows for the investigation of barrier dysfunction in neurodegenerative diseases, and drug delivery to the CNS, in addition to the evaluation and screening of the permeability of substances across the BBB[9,10]. In this regard, classic BBB in vitro models have traditionally utilized Transwell® technology, which enables the construction of multicellular models with paracrine interactions between the co-cultures of ECs and ACs or PCs. Notwithstanding its usefulness, Transwell® technology lacks a key element for the in vivo-like functioning of ECs: Laminar flow[6,9]. Moreover, many of the existing BBB models employ primary BMECs, immortalized BMECs, or human umbilical vein ECs (HUVECs) as EC-layer cultures. Even though they can constitute fair BBB constructs, access to primary cell sources can be difficult for ethical purposes and immortalized lines often fail to mimic the actual BBB function found in vivo[6,9].
In the building of in vitro BBB mimics, iPSC-derived BMECs (iBMECs) present improved barrier properties compared to primary BMECs, allowing for the modeling of genetic diseases and personalized therapy approaches[2]. These properties become more apparent when iBMECs are cultured in a complex environment. The traditional in vitro culture model, despite being simple and reproducible, fails to mimic cell-cell and cell-extracellular matrix (ECM) interactions[11]. Animal models, which present ideal barrier permeability, transport mechanisms, and morphological characteristics, present low reliability given that 80% of clinical trials fail in drug-delivery tests[12]. These limitations may be overcome with the development of organs-on-a-chip that simulate complex morphology in a three-dimensional (3D) culture[13].
The construction and applications of BBB-on-a-chip using microfluidic devices provide a lower-cost alternative with an impact on ethical issues, allowing for the reduced use of animals, reagents, and sample volume, and also permitting real-time microscopic analysis, with shortened reaction and analysis times, high throughput, automation, and portability[14,15]. Furthermore, from a morphological point of view, the microdevices assure a compatible reproduction of the BBB model due to the presence of laminar flow and shear stress, which convey better maintenance of barrier functions, homeostasis, and transport. This model has already been applied to several studies of neurological disorders such as Alzheimer’s disease, stroke, infectious diseases, and brain cancer[15]. Despite being the best alternative for simulation and in-depth study of the BBB, studies involving microfluidic devices lack clear protocols for large-scale manufacturing and adequate liquid perfusion, making the process imprecise and hard to operate, in addition to the difficulty in collecting channel material for detailed assessments[16].
Regarding the construction of BBB in vitro models on-a-chip using iPSCs, the literature still lacks a consensus on the best conditions to mimic BBB dysfunction upon CNS disorders and to screen for drugs and other therapeutic approaches. In view of these gaps, this systematic review searched the current literature for examples that meet the abovementioned criteria, that is, iPSCs, BBB models, and microfluidic devices.
MATERIALS AND METHODS
Search strategy
The articles used in this review were identified through searches performed in the PubMed and Scopus databases following the PRISMA guidelines[17]. The following selected criteria of interest, keyword strings ((Induced Pluripotent Stem Cell) AND (Microfluidic Device) AND (Blood-Brain Barrier)) and Boolean operators (DecS/MeSH) were used:
PubMed: (((((((“organs-on-chips”[Title/Abstract]) OR (“organs-on-a-chip”[Title/Abstract])) OR (“microfluidic device”[Title/Abstract])) OR (“lab-on-chips”[Title/Abstract])) OR (microfluidics[Title/Abstract])) AND (((“blood-brain barrier”[Title/Abstract]) OR (“Brain Blood Barrier”[Title/Abstract])) OR (“Blood Brain Barrier”[Title/Abstract]))) AND (((“Induced Pluripotent Stem Cell”[Title/Abstract]) OR (“Induced pluripotent stem cells”[Title/Abstract])) OR (iPSC[Title/Abstract]))) NOT (review[Publication Type]);
Scopus: ((TITLE-ABS-KEY (ipsc) OR TITLE-ABS-KEY (“induced pluripotent stem cell”) OR TITLE-ABS-KEY (“induced pluripotent stem cells”))) AND (((TITLE-ABS-KEY (“organs-on-chips”) OR TITLE-ABS-KEY (“organs-on-a-chip”) OR TITLE-ABS-KEY (“microfluidic device”) OR TITLE-ABS-KEY (“lab-on-chips”) OR TITLE-ABS-KEY (microfluidics) OR TITLE-ABS-KEY (“lab-on-chip”))) AND ((TITLE-ABS-KEY (“blood brain barrier”) OR TITLE-ABS-KEY (“brain blood barrier”) OR TITLE-ABS-KEY (“blood-brain barrier”)))) AND (LIMIT-TO (LANGUAGE, “English”)) AND (LIMIT-TO (DOCTYPE, “ar”)).
Inclusion and exclusion criteria
Only original full-text articles written in English have been included, without publication year limit, containing: (1) iPSCs; (2) microfluidic devices; and (3) simulation of the BBB by specialized cells. From the perspective of the Problem, Intervention, Comparison, and Outcome criterion, the addressed Problem was unclear literature on the best conditions to mimic the BBB using microfluidic device technology (BBB-on-a-chip) and IPSCs; the Intervention was to analyze the conditions for BBB-on-a-chip manufacture regarding the design and material for an adequate construction of the NVU with a functional environment for testing; the Comparison was related to conventional analyses; and the Outcome was a microdevice that mimics the BBB applied in neurological conditions or not. The exclusion of articles followed the following criteria: (1) Book chapters; (2) reviews; (3) duplicate articles in the databases; and (4) a study that did not report the use of iPSCs in the BBB-on-a-chip.
Data extraction
The selected articles under evaluation were analyzed under four topics, which were represented in three tables and one figure that addressed the following characteristics: (1) Microfluidic devices design and fabrication; (2) characteristics of the iPSCs used in the BBB models and their cultivation conditions; (3) iPSC differentiation process into iBMECs for reconstruction of the BBB-on-a-chip model; and (4) applications of BBB microfluidic 3D models using iPSCs.
Data compilation and review
In this systematic review, the preselection of titles was performed by authors Alves ADH and Gamarra LF from the defined search strategies. The ten authors (Alves ADH, Nucci MP, Ennes do Valle NM, Missina JM, Rego GNA, Mamani JB, Dias OFM, Garrigós MM, de Oliveira FA, Gamarra LF), in pairs, independently, and randomly reviewed and analyzed the eligibility of the articles according to the selection criteria mentioned above. In case of discrepancy in study selection between two authors, the criteria were discussed with a third reviewer and resolved.
Ennes do Valle NM and Mamani JB searched for the fabrication and characteristics of microfluidic devices; Alves ADH, Nucci MP, Ennes do Valle NM, Missina JM, and Rego GNA searched for differentiation and characterization protocols for iPSCs; Alves ADH, Nucci MP, Ennes do Valle NM, Missina JM, and Gamarra LF searched for the insertion and manipulation of cells in the microfluidic device; de Oliveira FA, Mamani JB, Dias OFM, and Garrigós MM searched for the objectives and applications of the selected studies. Analyses of data extracted from tables and flowcharts were performed by full peer consensus, respecting the above distribution. In this review, all authors wrote the entire text.
Risk of bias assessment
The selection of articles was performed in pairs and a third independent author decided if the articles should be included. The data selected in the tables were divided by the authors into the groups already described above, and data verification was carried out by the following group. The final inclusion of studies into the systematic review was by agreement of all reviewers.
Data analysis
The variables grouped in the tables and charts were distributed in percentages or range of distribution and used to characterize and illustrate the most frequently used results in this review.
RESULTS
Overview of the reviewed literature
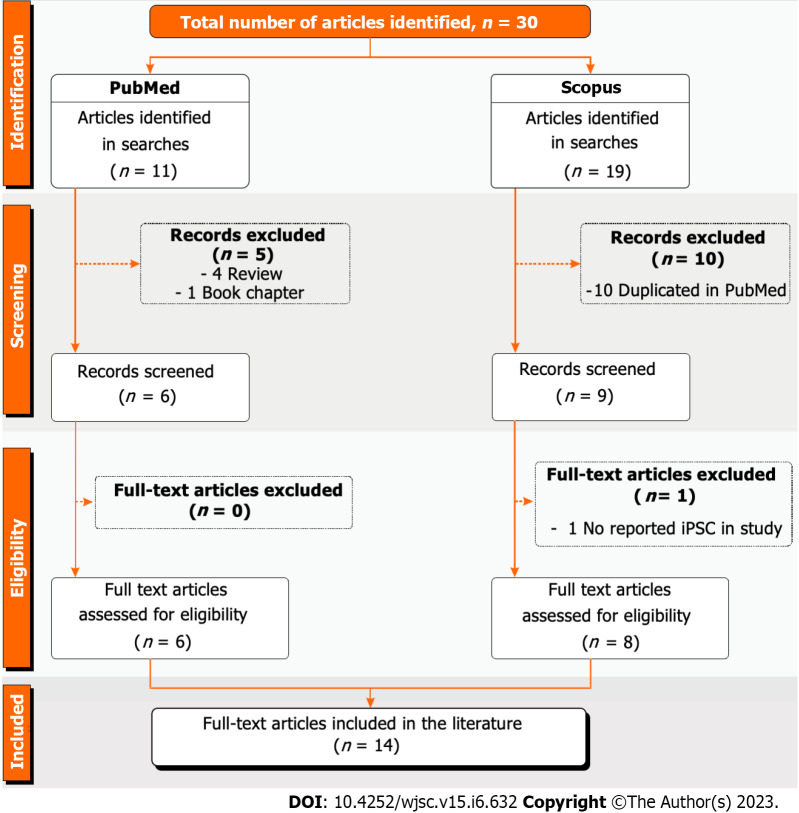
A search was performed based on the abovementioned selection strategy and keywords, resulting in 30 original articles in English: 11 from PubMed and 19 from Scopus. Following the exclusion criteria, 5 of the 11 articles from PubMed were excluded: 4 under the “review article” criterion and 1 under the “book chapter” criterion. Regarding the studies found in Scopus, 10 were manuscripts duplicated by the other database. After the eligibility analysis, only two articles were withdrawn: One for not having reported a study using iPSCs, and the other for not involving BBB functionality. Finally, this systematic review included 14 articles[18-31] that met all selection and inclusion criteria established by the authors (Figure 1).
Figure 1.
PRISMA flow chart of the study selection process applied in this systematic review. iPSC: Induced pluripotent stem cell.
Microfluidic device design and fabrication
The main theme of this review has been studied in current years (2017-2022), as shown in Table 1. The latest studies were performed in commercial chips[18-21], whereas older studies were performed using chips manufactured in-house[22-31]. In general, the studies with commercial chips did not detail the type of polymer[18-20] or the technology or mold used[18-21]. Only one of them specified the use of polydimethylsiloxane (PDMS), a well-known polymer for manufacturing microfluidic chips[21]. The majority of studies that manufactured the chip in-house used PDMS (57%)[22,25-29,31], but two studies adopted the less than usual materials polymethylmethacrylate (PMMA)[23] and Objet VeroClear® photopolymer[30]. Most of the chips fabricated in-house were molded by soft lithography[24-29,31], except the study by Choi et al[23], which employed computer numerical control; and the study by Wang et al[30], which used a 3D object printer. Details about mold material and fabrication were seldom explored in soft lithography applications; however, the mainly reported material was SU-8 made by photolithography[25-27].
Table 1.
Microfluidic device design and fabrication
|
Ref.
|
Manufacturing
|
Characteristics of microdevices
|
||||
|
Fabrication
|
Main material of device
|
Technology used
|
Mold details
|
Membranes
|
Dimensions as width × height
|
|
| Kurosawa et al[18], 2022 | MIMETAS® (OrganoPlate® 3-lane plate) | Unspecified polymer | NR | NR | Membrane-free | Top and bottom channels: 320 μm × 220 μm. Gel channels: 360 μm × 220 μm. PhaseGuides®: 100 μm × 55 μm |
| Fengler et al[19], 2022 | MIMETAS® (OrganoPlate® 3-lane plate) | Unspecified polymer | NR | NR | Membrane-free | Top and bottom channels: 320 μm × 220 μm. Gel channels: 360 μm × 220 μm. PhaseGuides®: 100 μm × 55 μm (width × height) |
| Wevers et al[20], 2021 | MIMETAS® (OrganoPlate® 3-lane plate) | Unspecified polymer | NR | NR | Membrane-free | Top and bottom channels: 320 μm × 220 μm. Gel channels: 360 μm × 220 μm. PhaseGuides®: 100 μm × 55 μm |
| Noorani et al[21], 2021 | Emulate® (brain on-a-chip) | PDMS | NR | NR | PDMS membrane | Brain channel: 1 mm × 1 mm. Blood channel: 1 mm × 0.2 mm |
| Middelkamp et al[22], 2021 | In house | PDMS | Soft lithography | Material: PMMA. Fabrication: Micrommilling | PETE membrane (5 μm thick) | Straight bottom channel: 500 μm × 500 μm. Open-top compartment: 500 μm × 1500 μm |
| Choi et al[23], 2021 | In house | PMMA | CNC | NA | PETE membrane | HUVEC microchannels: 800 μm × 200 μm. iBMECs microchannels: 800 μm × 500 μm |
| Motallebnejad et al[24], 2020 | In house | NR | Soft lithography | NR | Membrane-free | 800 μm × 100 μm |
| Lee et al[25], 2020 | In house | PDMS | Soft lithography | Material: SU-8. Fabrication: Photolithography | Membrane-free | Fluidic channel: 1340 μm × 150 μm. Main channel: 2200 μm × 150 μm |
| Jagadeesan et al[26], 2020 | In house | PDMS | Soft lithography | Material: SU-8. Fabrication: Photolithography | PDMS membrane (50 μm thick) | Top microchannel: 1 mm × 1 mm. Bottom microchannel: 1 mm × 0.2 mm |
| Vatine et al[27], 2019 | In house | PDMS | Soft lithography | Material: SU-8. Fabrication: Photolithography | PDMS membrane (50 μm thick) | Top microchannel: 1 mm × 1 mm. Bottom microchannel: 1 mm × 0.2 mm |
| Park et al[28], 2019 | In house | PDMS | Soft lithography | Material: Prototherm. Fabrication: 3D printed (Proto labs) | PE membrane (20 μm thick) | Hollow microchannels: 1 mm × 1 mm. Top channel: 1 mm × 1 mm. Bottom channel: 1 mm × 0.2 mm |
| Campisi et al[29], 2018 | In house | PDMS | Soft lithography | Material: Silicon Wafer. Fabrication: NR | Membrane-free | Fluidic channel: 1000 μm × 150 μm. Main channel: 1300 μm × 150 μm. Distance between posts: 200 μm |
| Wang et al[30], 2017 | In house | Objet VeroClear photopolymer | 3D object printer (Objet 30Pro, Stratasys Ltd., Rehovot, Israel) | NA | PC membrane (0.4 μm pore size) | Main channel: 300 μm × 160 μm |
| DeStefano et al[31], 2017 | In house | PDMS | Soft lithography | Material: Aluminum mold. Fabrication: NR | Membrane-free | 390 μm, 450 μm, 550 μm, and 770 μm (different width) |
3D: Three-dimensional; CNC: Computer numerical control; hBMECs: Human brain microvascular endothelial cells; HUVEC: Human umbilical vein endothelial cells; NA: Not applied; NR: Not reported; PDMS: Polydimethylsiloxane; PC: Porous polycarbonate; PE: Polyethylene terephthalate; PETE: Polyester track-etched; PMMA: Poly(methyl methacrylate); SU-8: Negative photoresist.
Half of the articles evaluated displayed membrane-free microfluidic chips[18-20,24,25,29,31], as shown in Table 1. The remaining studies reported the presence of a membrane dividing the channels within the chip: Three (21%) made of PDMS[21,26,27], one made of polyethylene terephthalate (PE)[28], two made of polyester (PETE)[22,23], and one of porous polycarbonate (PC)[30]. Almost all microfluidic chips with membranes were designed to comprise a top and a bottom channel[21-23,26-28]. An exception was found in the study by Wang et al[30], whose chip design was more complex, containing four parts that were stacked to compose the BBB model. Five studies (36%) presented three-channel microfluidic chips: Two side channels that were generally used to perfuse medium, and one middle channel where the cells were generally cultured[18-20,25,29]. The remaining studies employed simpler chip designs that comprised a single[24] or multiple channels[31] parallel to each other. Channel dimensions were largely variable (from 160 μm to 2200 μm) among the selected articles, defined according to the desired use of the chip.
Characteristics of the iPSCs used in the BBB models and their cultivation and differentiation conditions
To build the BBB, as shown in Table 2 and Figure 2A, IMR90-C4 from human fetal lung fibroblasts was the main iPSC line used (41.1%)[18,19,21,23,24,28,30], commonly cultivated in Matrigel-coated flasks. Few studies (17.6%) have reported the use of different iPSC lines[20,26,27], one of which used nine different types of iPSCs[27] that were extracted from skin fibroblasts or the peripheral blood of healthy (5) or unhealthy donors (2). Two of these skin fibroblast lines from unhealthy donors were modified by clustered regularly interspaced short palindromic repeat (CRISPR)[27]. These diverse cell lines were useful in modeling BBB dysfunctions and their corrections[27].
Table 2.
Characteristics of the induced pluripotent stem cells used in the blood-brain barrier model, their cultivation and differentiation conditions
| Ref. | Cell origin |
Cell differentiation
|
BBB components model
|
||||
|
iPSCs line
|
Flask coating
|
Medium
|
Supplement
|
Differentiated cell/medium
|
Co-culture/medium
|
||
| Kurosawa et al[18] | Human fetal lung fibroblast | IMR90-C4 | Matrigel | Day 3-2: mTeSR1-cGMP | Day 3: With Y27632 | iBMECs (107) in ESFM | NA |
| Day 2: Without Y27632 | |||||||
| Day 0-5: UM | KOSR (20%) + glutamax (0.5%) + NEAA (1%) + β-mercaptoethanol (0.0007%) | ||||||
| Day 6-8: EC+/+ (HESFM) | hPDS (1%) + RA (10 μM) + hFGF2 (20 ng/mL) | ||||||
| Fengler et al[19] | Human fetal lung fibroblast | IMR90-C4 | Matrigel | Day 3-1: mTeSR1 | Y27632 (10 μM) | iBMECs purified1 (106) in HBVP conditioned | NA |
| Day 0-5: UM (DMEM/F12-HEPES) | Glutamax + KOSR + NEAA + β-Mercaptoethanol | ||||||
| Day 6: EC+/+ (HESFM) | bPPP (1%) + RA (10 μM) + bFGF (20 ng/mL) | ||||||
| Wevers et al[20] | Human astrocytes | iPSCs | Geltrex | DMEM | FBS (10%) + N2 (1 ×) + P/S (1%) | iBMECs (104) in NR medium | Astrocyte-neuron cells (1:4) (1.5 × 104 cells/μL) in N2B27 |
| Human neural stem cells | Ax0018 | Matrigel-GFR | Day 0-21: N2B27 | BDNF (20 ng/mL) + GDNF (10 ng/mL) + AAc (100 μM) + db-cAMP1 (10 μM) | |||
| Noorani et al[21] | Human fetal lung fibroblast | IMR90-C4 | NR | Day 1: E8 | Y27632 (10 μm) | iBMECs (1.5 × 107 cells/mL) in NR medium | Primary ACs (106 cells/mL) and PCs (3.5 × 105 cells/mL) (3:1) in NR medium |
| Day 0-6: UM | KOSR (20%) + NEAA (1%) + Glutamax (0.5%) + β-Mercaptoethanol (0.1 mM) | ||||||
| Day 7-8: EC+/+ (HESFM) | bPPP (1%) + bFGF (20 ng/mL) + RA (10 μm) | ||||||
| Day 9: EC-/- (HESFM) | Without bFGF and RA | ||||||
| Middelkamp et al[22] | Adult skin fibroblasts | GM25256 | Laminin | Day 1: DMEM/F12 | Primocin (0.1 mg/mL) + DX (4 μg/mL) + N2 (1 ×) + MEM-NEAA (1 ×) + NT3-RHP (10 ng/mL) + BDNF-RHP (10 ng/mL) | iNeurons (104) in E8 | Rat ACs (1:1) and HUVECs (4 × 104) in ECGM-2 |
| Day 1 (after two hours): E8 | RevitaCell (1 ×) + DX (4 μg/mL) | ||||||
| Day 3-38: Neurobasal | Day 3: Primocin (0.1 mg/mL) + B-27 (1 ×) serum free + glutamax (1 ×) + DX (4 μg/mL) + NT-3 (10 ng/mL) + BDNF (10 ng/mL) + arabinoside hydrochloride (2 μm) | ||||||
| Day 5: Refresh medium without arabinoside hydrochloride (2 μm) | |||||||
| Day 9-38: Refresh medium with 2.5% fetal calf serum | |||||||
| Choi et al[23] | Human fetal lung fibroblast | IMR90-C4 | Matrigel | Day 3: mTeSR1™-E8™ | NR | iBMECs purified1 (1.2 × 107 cells/mL) in HESFM | AC (106 cells/mL) in EC-/- |
| Day 0-5: UM (DMEM/F12) | KOSR (20%) + NEAA (100x) + glutamax (0.5%) + β-Mercaptoethanol (0.007%) (5% O2) | ||||||
| Day 6-8: EC+/+ (HESFM) | Human serum (1%) + bFGF (20 ng/mL) + RA (10 μm) (5% O2) | ||||||
| Day 9: EC-/- (HESFM) | Without bFGF and RA | ||||||
| Motallebnejad et al[24] | Human fetal lung fibroblast | IMR90-C4 | Matrigel | Days 3-1: mTeSR1-cGMP | Day 3: Y27632 (10 μm) | iBMECs purified1 in NR medium | NA |
| Day 2: Without Y27632 | |||||||
| Days 0-5: UM | NR | ||||||
| Days 6-8: EC+/+ (HESFM) | Days 6-7: hPDS (1%) + RA (10 μM) + FGF2 (20 ng/mL) | ||||||
| Day 8: bPPP (1%) or FBS (2%) + FGF (20 ng/mL) + RA (10 μM) | |||||||
| Day 9: EC-/- (HESFM) | Without bFGF and RA | ||||||
| Healthy human African American male from the bone marrow CD34+ cells | ACS-1024 | LN 511-E8 or LN 411-E8 or collagen IV + fibronectin | HESFM | Day 8: bPPP (1%) or FBS (2%) + FGF (20 ng/mL) + RA (10 μM) | |||
| Day 9: Without bFGF and RA | |||||||
| Lee et al[25] | Endothelial cells | hiPSC-ECs | Human fibronectin | VascuLife VEGF | iCell media supplement | hiPSC-ECs (6 × 106 cells/mL) in VascuLife VEGF with thrombin | PCs and ACs (106 cells/mL) in VascuLife VEGF with thrombin |
| Jagadeesan et al[26] | Neural progenitors | EZ-Spheres | NA | Day 1: EZ-sphere medium (DMEM/F12) | bFGF (100 ng/mL) + EGF (100 ng/mL) + heparin (5 μg/mL) + B27 (2%) | iNPCs (106 cells/mL) in NDM | ACs (9 × 105 cells/mL) and PCs (3 × 105 cells/mL) in in DMEM |
| B27 (2%) + vitamin A + N2 (1%) + hBDNF, (20 ng/mL) | |||||||
| NR | hiPSCs | Basement membrane matrix-coated | Day 1: mTeSR1 | NR | iBMECs [(14-20) × 106 cells/mL] in EC-/- (HESFM) | ||
| Day 2-8: UM (DMEM/F12) | KOSR (10%) + NEAA (1%) + glutamine (0.5%) + β-Mercaptoethanol (100 μm) | ||||||
| Day 9-10: EC+/+ (HESFM) | bPPP (1%) + bFGF (20 ng/mL) + RA (10 μM) | ||||||
| Day 11: EC-/- (HESFM) | Without bFGF and RA | ||||||
| Vatine et al[27] | Adult skin fibroblasts | CS03iCTR | Matrigel | Day 3: mTeSR1 | NR | iNPCs (106 cells/mL) in NDM and iBMECs (1.4 × 104 cells/mL) | ACs (9 × 105 cells/mL) and PCs (3 × 105 cells/mL) in DMEM |
| CS83iCTR | |||||||
| CS03iCTRmut, 2 | |||||||
| CS01iMCT8 | |||||||
| CS01iMCT8cor, 2 | |||||||
| Peripheral blood | CS0172iCTR | ||||||
| CS0188iCTR | Day 0: UM | Without bFGF | |||||
| CS0617iCTR | |||||||
| Huntington’s disease | CS81iHD | ||||||
| Park et al[28] | Human fetal lung fibroblast | IMR90-C4 | Matrigel | Day 3: mTeSR1 | NR (5% O2) | iBMECs purified1 (2.3 × 107 cells/mL) in EC | ACs (7 × 105 cells/mL) and PCs (3 × 105 cells/mL) in ACM |
| Day 0-6: UM (DMEM/F12) | KOSR (100 mL) + NEAA (5 mL) + glutamax (2.5 mL) + β-Mercaptoethanol (3.5 μL) (5% O2) | ||||||
| Day 7-9: EC+/+ | RA (5% O2) | ||||||
| Campisi et al[29] | Blood from 30-39-year-old healthy females | hiPSC-ECs | Human fibronectin | VascuLife VEGF | iCell media supplement + VEGF (50 ng/mL) | hiPSC-ECs (2 × 106 cells/mL) in EBM-2 | Monoculture hiPSC-ECs (6 × 106 cells/mL) in EBM-2 |
| PCs (2 × 106 cells/mL) in EGM-2 MV | |||||||
| PCs and ACs (2 × 106 cells/mL) in EGM-2 MV | |||||||
| Wang et al[30] | Human fetal lung fibroblast | IMR90-C4 | Matrigel | Day 3: mTeSR1 | NR | iBMECs in HESFM | Rat primary ACs in AGM |
| Day 0-5: UM (DMEM/F12) | HEPES + KOSR (20%) + MEM-NEAA (1 ×) + L-glutamine (1 mM) + β-Mercaptoethanol (0.1 mM) | ||||||
| Day 6-8: EC+/+ (HESFM) | hPDS (1%) + bFGF (20 ng/mL) + RA (10 μM) | ||||||
| DeStefano et al[31] | CD34 positive bone marrow | BC1-hiPSCs | Matrigel | Day 4-3: mTeSR1-E8 | NR | iBMECs (105) in EC | NA |
| Day 3: Y27632 (10 μm) | |||||||
| Day 0-5: UM (DMEM/F12) | KOSR (20%) + NEAA (1%) + L-Glutamine (0.5%) + β-Mercaptoethanol (0.84 μm) | ||||||
| Day 6-7: EC+/+ (HESFM) | hPDS (1%) + bFGF (20 ng/mL) + RA (10 μm) | ||||||
| Day 8: EC (HESFM) | DB-cAMP (400 μm) or Y27632 (10 μm) | ||||||
Induced pluripotent stem cell-derived brain microvascular endothelial cells were selectively expanded before to seed in the chip.
Modified by clustered regularly interspaced short palindromic repeats. For human induced pluripotent stem cells-brain microvascular endothelial cells purification, day 8: ECM [1 μg/cm2 LN 511-E8 or LN 411-E8 (iMatrix, iWAi), 1 μg/cm2 full length laminin 511 (Biolamina), 100 μg/mL fibronectin (Millipore Sigma), or a mixture of 400 μg/mL collagen IV (Millipore Sigma) and 100 μg/mL of fibronectin] protein-coated ThinCert cell culture inserts (Greiner Bio-One), well plates or ibidi μ-slides. AT: Adipose tissue; BDNF-RHP: Brain-derived neurotrophic factor recombinant human protein; EGF: Epidermal growth factor; BM: Bone marrow; bPPP: Platelet-poor plasma derived bovine serum; CXCR4: C-X-C chemokine receptor type 4; db-cAMP1: 2’-O-Dibutyryladenosine-3’,5’-cyclic monophosphate; DMEM: Dulbecco’s modified eagle medium; E8 medium: Essential 8 medium; EGF: Epidermic growth factor; FDF2: Fibroblast growth factor 2; FL: Fetal lung; GFP: Green fluorescent protein; HESFM: Human endothelial serum free medium; hFGF2: Human fibroblast growth factor 2; hiPSC: Human induced pluripotent stem cell; HOXB4: Homeobox B4; hPDS: Human serum from platelet-poor human plasma; IB: Intrabone; IP: Intraperitoneal; IV: Intravenous; KOSR: Knockout serum replacement; Matrigel-GFR: Growth factor reduced matrigel; MEC: Brain microvascular endothelial cell; MEM: Minimum essential medium; NA: Not applied; NEAA: Non-essential amino acids; Nos2-/-: Deficient in type 2 nitric oxide; NR: Not reported; NT3-RHP: Neurotrophin-3 recombinant human protein; P/S: Penicillin/streptomycin; PDGFB: Platelet-derived growth factor subunit B; PPDS: Platelet poor derived serum; AAc: Ascorbic acid; SDF-1: Stromal cell-derived factor 1; solG-CSFR: Soluble granulocyte colony-stimulating factor decoy receptor; TPO: Thrombopoietin; UCB: Umbilical cord blood; MMP3: Matrix metallopeptidase 3.
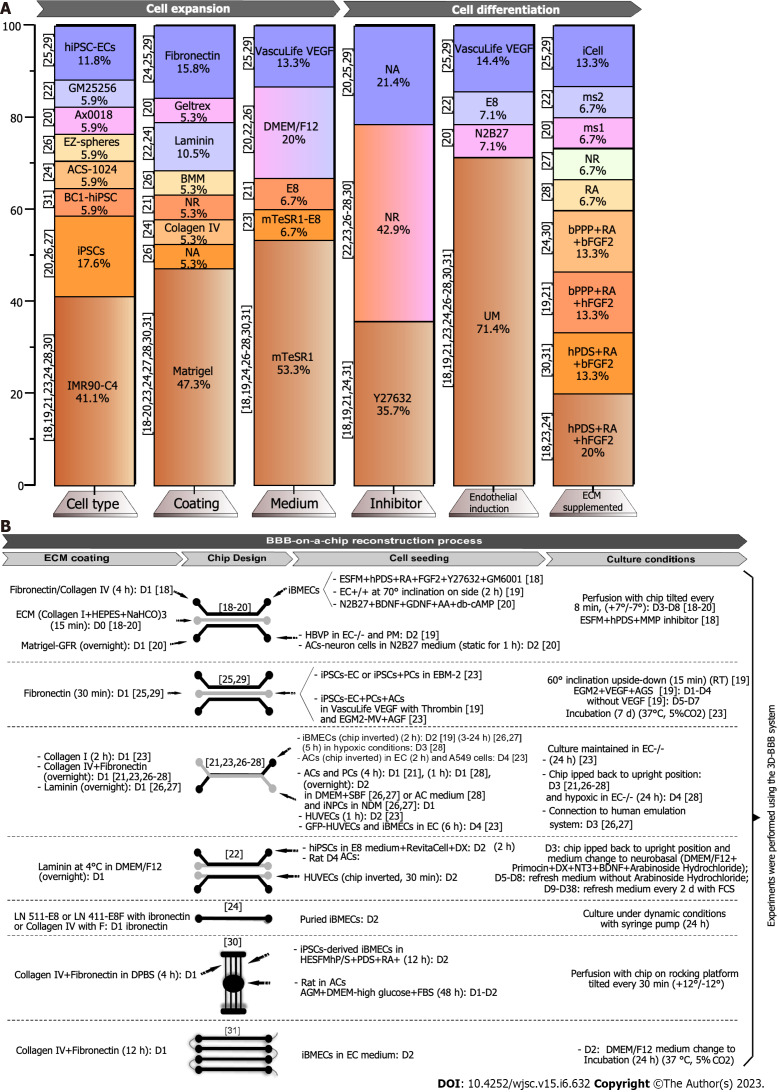
Figure 2.
Induced pluripotent stem cell differentiation process into induced pluripotent stem cell-derived brain microvascular endothelial-like cells for reconstruction of the blood-brain barrier-on-a-chip model. A: Percent analysis of cells and culture media used in induced pluripotent stem cell expansion and differentiation before blood-brain barrier (BBB)-on-a-chip reconstruction; B: Schematic summary of the main findings in this systematic review on the cells and culture conditions applied in the BBB-on-a-chip reconstruction process. Studies were grouped by similar device designs. AAc: Ascorbic acid; AC: Astrocyte; ACS-1024: Bone-induced pluripotent stem cell line; AGF: Astrocyte growth factor; AGM: Astrocyte growth medium; BC1: Lymphoma cell line; BDNF: Brain-derived neurotrophic factor; bFGF: Bovine fibroblast growth factor; BMM: Basement membrane matrix; bPPP: Basic platelet-poor plasma; D1: Day 1; D2: Day 2; D3: Day 3; D38: Day 38; D4: Day 4; D5: Day 5; D6: Day 6; D8: Day 8; D9: Day 9; db-cAMP1: 2’-O-Dibutyryladenosine-3’,5’-cyclic monophosphate; DMEM/F12: Dulbecco’s modified Eagle medium with F12; DPBS: Dulbecco’s phosphate-buffered saline; DX: Doxycycline; E8: Essential 8 medium; EC-/-: Human endothelial serum-free medium without retinoic acid + basic fibroblast growth factor; EC: Endothelial cell; EC+/+: Human endothelial serum-free medium with retinoic acid + basic fibroblast growth factor; ECM: Extracellular matrix; EGM-2MV: Microvascular endothelial cell growth medium-2; ESFM: Endothelial serum-free medium; FBS: Fetal bovine serum; FCS: Fetal calf serum; FGF2: Fibroblast growth factor 2; GDNF: Glial cell line-derived neurotrophic factor; GFP: Green fluorescent protein; GFR: Growth factor reduced; GM25256: Cell line of induced pluripotent stem cell derived from adult skin fibroblasts; GM6001: Broad spectrum MMP inhibitor; HBVP: Human brain vascular pericytes; HEPES: 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid; HESFM: Human endothelial serum-free medium; hFGF: Human fibroblast growth factor; hiPSC-EC: Human-induced pluripotent stem cell-derived endothelial cell; hPDS: Human serum from platelet-poor human plasma; hPDS: Platelet-poor plasma-derived human serum; HUVEC: Human umbilical vein endothelial cell; iBMECs: Induced pluripotent stem cell-derived brain microvascular endothelial cells; IMR90-C4: Induced pluripotent stem cell line; iPSCs: Induced pluripotent stem cells; LN: Laminin; ms1: Brain-derived neurotrophic factor + glial cell line-derived neurotrophic factor + ascorbic acid + 2’-O-Dibutyryladenosine-3’,5’-cyclic monophosphate; ms2: Primocin + glutamax + doxycycline + neurotrophin-3 + brain-derived neurotrophic factor + fetal calf serum; mTeSR1: Basal medium type for induced pluripotent stem cells; N2B27: Culture medium; NA: Not applied; NR: Not reported; NT3: Neurotrophin-3; P/S: Penicillin/streptomycin; PCs: Pluripotent cells; PM: Pericyte medium; RA: Retinoic acid; RA: Retinoic acid; RT: Room temperature; SFB: Serum-free medium; UM: Unconditioned medium; VEGF: Vascular endothelial growth factor; Y27632: Dihydrochloride inhibitor.
Concerning the coating applied for culturing the iPSC lines, fibronectin was selected for cultivation of human iPSC (hiPSC)-ECs (11.8%)[25,29]. Other particular iPSC lines, such as Ax0018 (neural stem cells)[20] and BC1-hiPSC (bone marrow)[31], were cultured on Matrigel, whereas GM25256 (skin fibroblast)[22], and ACS-1024 (bone marrow)[24] were cultured on laminin. The ECM on which EZ-Spheres (neural progenitors) were grown was not defined[26]. Interestingly, the study by Motallebnejad et al[24] compared cultivation of the ACS-1024 line on different laminin isoforms (LN511 and LN411), or on collagen IV associated with fibronectin[24]. Overall, Matrigel was the coating of choice in the majority of the selected studies (47.4%)[18-20,23,24,27,28,30,31]. The least common coatings, such as basement membrane matrix coating[26] and geltrex[20], appeared in 5.3% of the analyzed articles (Figure 2A and Table 2).
The iPSCs generally go through a complex differentiation process, involving multiple medium replacements with the addition of supplements and composition changes. Most iPSC cultivation protocols were undertaken in mTeSR1 medium (53.3%) associated[23] or not[18,19,24,26-28,30,31] with essential medium 8 (E8, 6.7%) for about 3 d, mainly for culturing the IMR90-C4 cell line. Part of these studies (35.7%)[18,19,21,24,31] added the Rho-associated, coiled-coil containing protein kinase inhibitor (Y27632) to the cultures overnight to inhibit cell proliferation before differentiation[32,33]. Unconditioned medium (UM), normally composed of Dulbecco’s modified Eagle medium (DMEM)/F12 supplemented with knockout serum replacement + non-essential amino acids (NEAA) or minimum essential medium (MEM) with NEAA (MEM-NEAA) + glutamax or L-glutamine + β-mercaptoethanol, was used in 71.4% of the studies[18,19,21,23,24,26-28,30,31] to promote differentiation to ECs (for 6 d on average), followed by a change to human endothelial serum-free medium containing diverse substances, such as retinoic acid (RA), platelet-poor-derived bovine serum (bPPP) or human serum from platelet-poor human plasma (hPDS), and basic fibroblast growth factor, to induce BMEC formation (Figure 2A and Table 2).
Few studies have described different processes to generate iBMECs. Lee et al[25] used solely vasculife vascular endothelial growth factor medium with iCell media supplement[29]. The study by Wevers et al[20] used DMEM/F12 and N2B27 supplemented with brain-derived neurotrophic factor (BDNF) + glial cell line-derived neurotrophic factor + ascorbic acid + 2’-O-Dibutyryladenosine-3’,5’-cyclic monophosphate. Finally, the study by Middelkamp et al[22] utilized DMEM/F12 supplemented with primocin + doxycycline (DX) + N2 + MEM-NEAA + neurotrophin-3 (NT3) recombinant human protein + BDNF recombinant human protein, followed by a change to E8 supplemented with revitacell + DX, and a later change to neurobasal medium supplemented first with primocin + B27SF + glutamax + DX + NT3 + BDNF, in addition to arabinoside hydrochloride for 24 h, followed by the addition of fetal calf serum. DX induced the overexpression of neurogenin 2 in GM25256 iPSCs, being one of the key factors for the differentiation of GM25256 into iNeurons (Table 2).
All of the abovementioned processes were described for iPSC differentiation on a plate, with the exception of the study by Campisi et al[29], whose cell differentiation step occurred inside the microfluidic chips. Furthermore, two studies used hypoxic conditions during the cell differentiation process[23,28].
BBB-on-a-chip reconstruction process
To build the BBB-on-a-chip model, the microdevices must first receive a coating layer to sustain 3D cell growth, as depicted in Figure 2B. From this perspective, diverse coating substances and different cell-seeding strategies have been described, depending on the chip design. As seen in Figure 2B, previously to cell culture on-a-chip, fibronectin was the principal ECM component coating the microfluidic channels (39.3%)[18,21,23-26,28-31], among which 28.6% corresponded to a mixture of fibronectin + collagen type IV. More recent studies have applied an ECM composed of collagen type I + 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid + sodium bicarbonate (10.7%)[18-20]. Laminin coating was used in 14.3% of the analyzed studies[22,26,27], wherein a mixture of different types of laminin + fibronectin was employed in the study by Motallebnejad et al[24]. Additionally, one study utilized only Matrigel[20], and another utilized collagen type I[23] as channel coating. The mentioned coatings were carefully selected for each channel, depending on the desired environment and cultured cell type.
Regarding cell seeding, five studies have described an iBMEC single culture[18,19,24,29,31], in which two studies applied a selective cell expansion step before seeding[19,24]. A co-culture of iBMEC + ACs was reported in three studies[20,23,30], one of which used PCs in the co-culture[29]. Yet, most studies have built their BBB models based on an approach involving more than two cell types in co-culture, normally associating ACs and PCs with other ECs such as HUVECs[22], iBMECs[21,28], and iPSC-ECs[25], with the exception of Jagadeesan et al[26] and Vatine et al[27], who associated neural progenitor cells with ACs and PCs in the brain-side, and iBMECs in the blood-side of their BBB model (Figure 2B).
The particular aspects of cell culture inside the chips are displayed in Figure 2B. Among the observed aspects, we highlight the inversion or inclination of the chip to seed the cells[21-23,25-28], maintenance of the chip under hypoxic conditions[28], and tilting of the chips for gravity-dependent bidirectional medium flow[18-20,30].
Applications of BBB microfluidic 3D models using iPSCs
Regarding the general objective of the studies (Table 3), the great majority of the articles (roughly 80%)[18-21,23,24,26-30] were aimed at developing a microfluidic BBB model, as well as assessing the convenience of the manufactured model by quantifying common molecular markers that are normally expressed in cells that make up the BBB in vivo. Five of these studies (35.7%) analyzed drug transport for BBB characterization purposes[18,19,27,28,30]. Only two studies described actual applications for their models: Wevers et al[20] used their developed chip to model BBB disruption upon ischemic stroke, and Lee et al[25] tested the permeability of their model to commercial and synthesized polymer nanoparticles (NPs). Middelkamp et al[22] compared a 2D Transwell BBB model to one built on-a-chip, whereas DeStefano et al[31] evaluated the effect of shear stress on their BBB microfluidic chips.
Table 3.
Applications of blood-brain barrier microfluidic three-dimensional models using induced pluripotent stem cells
|
Ref.
|
Application
|
Characterization
|
Evaluation technique
|
Outcomes
|
| Kurosawa et al[18] | Build and evaluate a BBB 3D in vitro model | Capillary structure formation and tight junction proteins expression | Immunocytochemistry | Formation of the capillary structure, functional tight proteins; lower expression of ABC transporters than levels found in vivo, except for BCRP; expression of functional SLC transporters |
| Transport proteins and receptors expression | Immunocytochemistry | |||
| qPCR | ||||
| Tight junction functionality | Fluorescence (lucifer yellow and antipyrine) | |||
| HPLC-MS/MS (test-drug transport) | ||||
| Transport proteins function | HPLC-MS/MS (test-drug transport) | |||
| Fengler et al[19] | Build and evaluate a BBB 3D in vitro model | Tight junction proteins expression | Immunocytochemistry | Capillary diameter CA. 40 times larger than in vivo brain vessels; physiologically relevant TEER values; physiologically similar localization of BCRP and GLUT-1 proteins. Promising BBB model for future drug screening tests |
| Microvessel integrity | Fluorescence (DEX-A647 and sodium fluorescein) | |||
| Microvessel permeability | Diazepam, Emricasan, Ac-YVAD-CMK, Z-DEVD-FMK, ZVAD (OH)-FMK, Staurosporine, and IL-1β | |||
| Tight junction functionality | ELISA (Diazepam) | |||
| TEER measurements | ||||
| Wevers et al[20] | Build and evaluate a BBB 3D in vitro model | Tight junction proteins expression | Immunocytochemistry | Barrier functionality similar to that found in vivo; microfluidic model suitable for evaluating disruption of the BBB; successful ischemic stroke modeling. Potential use for modeling the BBB under sub-optimal conditions (disease) and for evaluating potential therapies |
| Tight junction functionality | TEER measurements | |||
| Microvessel permeability | Fluorescence (sodium fluorescein) | |||
| Transport proteins expression | Fluorescence: P-gp inhibition | |||
| qPCR | ||||
| Neuronal functionality | Calcium fluorescence imaging | |||
| Ischemic stroke modeling | Microvessel permeability | Fluorescence (FITC-dextran) | ||
| Mitochondrial membrane potential | Luminescence (CellTiter-GLO) | |||
| ATP quantification | ||||
| Noorani et al[21] | Build and evaluate a BBB 3D in vitro model | Tight junction proteins expression | Immunocytochemistry | BBB functionality remains intact for up to 7 d and is similar to that found in vivo; a more physiologically relevant BBB model; shear stress contributes positively to BBB tightness |
| Microvessel permeability | UPLC-MS/MS: [13C12] sucrose and [13C6] mannitol | |||
| Transport proteins expression | Immunocytochemistry | |||
| Fluorescence: P-gp inhibition | ||||
| Middelkamp et al[22] | Compare 2D cultures to microfluidic chip cultures | Neuronal differentiation and characterization of HUVECs | Immunocytochemistry | Culture in microfluidic chips promotes gene expression that more closely resembles that found in vivo |
| RNA sequencing | ||||
| Transcriptomic analysis | ||||
| Choi et al[23] | Build and evaluate a BBB 3D in vitro model | Tight junction proteins expression | Immunocytochemistry | cECMTE membrane with 10 m pores in microfluidic device were successful in mimicking the in vivo BBB, also allowing for cancer cell tissue migration. Promising BBB model for studying cancer metastasis, cell communication, and migration |
| qPCR | ||||
| Tight junction functionality | Fluorescence (lucifer yellow) | |||
| Transendothelial migration of cancer cells (CellMask) | ||||
| Immunocytochemistry | ||||
| Transport proteins expression | Immunocytochemistry | |||
| Motallebnejad et al[24] | Build and evaluate a BBB 3D in vitro model | Tight junction proteins expression | Immunocytochemistry | LM511-E8 ECM contributes to long-lasting endothelial cell and BBB function, in addition to promoting better shear stress responses. Authors recommend the use of LM511-E8 ECM for future studies involving BBB function |
| Fluorescence (F-actin staining) | ||||
| qPCR | ||||
| Tight junction functionality | TEER measurements | |||
| Fluorescence (rhodamine B-labeled neutral dextran) | ||||
| Lee et al[25] | BBB permeability to polymer nanoparticles | Tight junction and transport proteins expression | qPCR | Fast analysis of polymer nanoparticles permeability; physiologically reliable BBB model |
| Permeability to polymer nanoparticles | Fluorescence (polymer nanoparticles and FITC-dextran) | |||
| 3D fluorescence intensity maps | ||||
| Jagadeesan et al[26] | Build and evaluate a BBB 3D in vitro model | Tight junction proteins expression | Immunocytochemistry | Successful fabrication of BBB model personalized for different human individuals; BBB models were able to mimic physiological differences between healthy and ill individuals |
| Tight junction functionality and microvessel permeability | Fluorescence: FITC-dextran | |||
| Transport proteins expression | Immunocytochemistry | |||
| Neuronal differentiation | ||||
| Vatine et al[27] | Build and evaluate a BBB 3D in vitro model | Tight junction proteins expression | Immunocytochemistry | Successful fabrication of BBB model personalized for different human individuals; BBB models were able to mimic physiological differences between healthy and ill individuals |
| Transcriptional analysis | ||||
| Microvessel permeability and tight junction functionality | Fluorescence (FITC-dextran and 2NDBG) | |||
| ELISA (human albumin, IgG and transferrin) | ||||
| LC-MS/MS (T3, colchicine, levetiracetam and retigabine) | ||||
| Transmission light microscopy | ||||
| TEER measurements | ||||
| Immunocytochemistry | ||||
| Transport proteins expression | Immunocytochemistry | |||
| Transcriptional analysis | ||||
| Transport protein function | Fluorescence (rhodamine-123) | |||
| Whole-blood neuronal toxicity | Colorimetric assay (quantification of lactic dehydrogenase) | |||
| Neuronal functionality | Immunocytochemistry | |||
| Calcium fluorescence imaging | ||||
| Park et al[28] | Build and evaluate a BBB 3D in vitro model | Tight junction proteins expression | Immunocytochemistry | BBB functionality remains intact for up to 7 d. Promising BBB model for future drug and antibody transport studies |
| Multiplex qPCR | ||||
| MS (proteomics) | ||||
| Tight junction functionality and microvessel permeability | Electron transmission microscopy | |||
| TEER measurements | ||||
| Fluorescence (dextrans, cetuximab, angiopep-2, MEM75, 13E4) | ||||
| ELISA (dextrans, cetuximab) | ||||
| Transport proteins expression | Immunocytochemistry | |||
| MS | ||||
| Transport proteins function | Fluorescence (rhodamine-123 and doxorubicin) | |||
| Campisi et al[29] | Build and evaluate a BBB 3D in vitro model | Tight junction proteins expression | Immunocytochemistry | Tri-culture of human iPSC-derived endothelial cells, astrocytes and pericytes spontaneously arranged into a BBB-like model. Promising BBB model for future preclinical experiments |
| qPCR | ||||
| Tight junction functionality and microvessel permeability | Fluorescence (FITC-dextran) | |||
| Characterization of astrocytes and pericytes | Immunocytochemistry | |||
| Wang et al[30] | Build and evaluate a BBB 3D in vitro model | Tight junction proteins expression | Immunocytochemistry | Pumpless media perfusion system that resembles the blood residence time within brain tissues; physiologically relevant TEER values maintained for up to 10 d. Promising BBB model for future drug permeability studies |
| Tight junction functionality and microvessel permeability | TEER measurements | |||
| Fluorescence: FITC-dextran and doxorubicin | ||||
| LC-MS/MS (caffeine and cimetidine) | ||||
| DeStefano et al[31] | Evaluate BBB upon shear stress | Characterization of iPSC-derived endothelial cells morphology and function | Microscopy (time-lapse imaging analysis using ImageJ) | BBB endothelial cells display unique features that differ from endothelial cells from other tissues; shear stress plays a key role in BBB-like function in microfluidic models |
| Tight junction proteins expression | Immunocytochemistry | |||
| Western blot | ||||
| qPCR | ||||
| Transport proteins expression | qPCR |
2NDBG: 2-deoxy-2-[(7-nitro-2,1,3-benzoxadiazol-4-yl)-amino]-D-glucose; 3D: Three-dimensional; Ac-YVAD-CMK: Caspase-1 inhibitor; ATP: Adenosine triphosphate; UPLC-MS/MS: Ultra-performance liquid chromatography-mass spectrometry (tandem); BBB: Blood-brain barrier; BCRP: Breast cancer resistance protein; cECMTE: Condensed extracellular matrix track-etched; CellTiter-GLO: Luminescent cell viability assay; DEX-A647: Dextran conjugated to Alexa 647; ECM: Extracellular matrix; ELISA: Enzyme-linked immunoassay; FITC-dextran: Fluorescein isothiocyanate dextran; GLUT-1: Glucose transporter 1; hBMEC: Human brain microvascular endothelial cells; HPLC-MS/MS: High-performance liquid chromatography-mass spectrometry (tandem); HUVEC: Human umbilical vein endothelial cell; iBMEC: Induced pluripotent stem cell-derived brain microvascular endothelial-like cell; IgG: Immunoglobulin G; IL-1β: Interleukin 1 beta; iPSC: Induced pluripotent stem cell; LC-MS/MS: Liquid chromatography-mass spectrometry (tandem); LM511-E8: Fragment E8 of laminin 511; MS: Mass spectrometry; P-gp: Permeability glycoprotein; qPCR: Quantitative polymerase chain reaction; RNA: Ribonucleic acid; SLC: Solute carrier protein; TEER: Trans-epithelial electrical resistance; Z-DEVD-FMK: Caspase-3 inhibitor; ZVAD (OH)-FMK: Pan-caspase inhibitor.
The characterization of the BBB model was performed by means of structural and functional analyses. Concerning structural analyses, the expression of tight junction proteins was evaluated in 85.7% of the studies using immunocytochemistry (85.7%)[18-21,23-31]. Part of these studies also performed quantitative polymerase chain reaction (qPCR) (42.9%)[23-25,29,31]. Less frequently used techniques were fluorescence (F-actin staining)[24], transcriptional analysis[27], mass spectrometry (MS) (proteomics)[28], and western blot analysis[31]. Transport protein and receptor expression was evaluated in 57.1% of the cases[18,20,21,23,26-28,31] by immunocytochemistry (42.9%)[18,21,23,26-28], qPCR (21.4%)[18,20,31], fluorescence (permeability glycoprotein inhibition, 14.3%)[20,21], transcriptional analysis (7.1%)[27], and MS (7.1%)[28]. Two studies (14.3%) characterized neuronal differentiation by immunocytochemistry[22,26], one of which also applied RNA sequencing and transcriptomic analysis to further characterize the HUVECs[22]. Structural characterization of ACs and PCs was performed by immunocytochemistry by Campisi et al[29]. Lastly, DeStefano et al[31] used microscopic techniques to structurally and functionally characterize ECs seeded on-a-chip[31] (Table 3).
Functional characterization of the BBB model was more varied, with the use of diverse techniques. Tight junction functionality (71.4%) was mostly analyzed by fluorescence (50%)[18-20,22,24,26,30] using diverse fluorescent markers[18,23,24,26,28-30] such as zonula occludens-1 (ZO-1), claudin-5[21,22,26,28], occludin (OCLN)[21,22], glucose transporter type 1[21,24,26], platelet/EC adhesion molecule-1 (PECAM-1)[22,24,26,27], and vascular endothelial (VE)-cadherin[22]. Other techniques used to evaluate tight junction functionality were transepithelial electrical resistance (TEER) measurements (42.9%)[19,20,24,27,28,30], enzyme-linked immunosorbent assay (21.4%)[19,27,28], (ultra or high performance) liquid chromatography with tandem MS (LC-MS/MS) (21.4%)[18,27,30], immunocytochemistry[23,27] (14.3%), transendothelial migration of cancer cells[23], electron transmission microscopy (7.1%)[28], and transmitted light microscopy (7.1%)[27]. Microvessel permeability (57.1%)[19-21,25,27-30] and microvessel integrity (7.1%)[19] were assessed by fluorescence (57.1%)[19-21,25,27-30]. Transport protein function was evaluated by 21.4% of the studies[18,27,28] by fluorescence (14.3%)[27,28] and high-performance LC-MS (7.1%)[18]. Neuronal functionality was evaluated by calcium fluorescence imaging (14.3%)[20,27] and immunocytochemistry (7.1%)[27]. Mitochondrial membrane potential was analyzed by luminescence and ATP quantification (7.1%)[20]. In a diverse approach, the study by Vatine et al[27], among other evaluations, perfused whole human blood through the chip channels to perform a whole-blood neuronal toxicity assay using a colorimetric technique, focusing on the quantification of lactic dehydrogenase[27] (Table 3).
Overall, the analyzed studies were successful in building structural and functional BBB models on-a-chip using iPSCs. Most of them highlighted the future use of their models for drug screening tests, in addition to mimicking physiological conditions found in healthy and unhealthy individuals, wherein one of the studies reported the applicability of their device for modeling cancer cell invasion and migration through the BBB. The studies also focused on the potential for the development of personalized therapies, mainly in conditions of disease. Some interesting outcomes have reported long-lasting BBB function for the developed chips, with an average BBB function duration of 7 d[21,28] to 10 d[30] (Table 3).
DISCUSSION
This systematic review showed that this theme, BBB models with iPSCs in microdevices, is quite novel in the scientific literature; without setting a time limit, only articles from 2017 to 2022 were found. Furthermore, the articles published in the last 2 years have already shown great technological advances in BBB-on-a-chip in commercial devices[18-21]. Curiously, the same chip by MIMETAS® was used in three studies by different labs around the world (Japan[18], the Netherlands[19], and Germany[20]), and another commercial BBB-on-a-chip was also used by an American lab[21]. These advances in technological on-chip manufacturing allow the scientific community to direct further efforts to apply the developed BBB-on-a-chip to model pathological conditions, such as stroke[21], and design personalized therapeutic approaches.
Among the developed BBB models on a microdevice, some showed distinctive features, mainly for the chips fabricated in house. For example, the study by Choi et al[23] described a BBB microchip whose functionality was based on a track-etched 10 μm pore polyester membrane covered with a collagen type I ECM. This study reported that the ECM-covered membrane with a larger pore size allowed for better modeling of the communications between neural cells and ECs, in addition to enabling the evaluation of cancer cell metastasis through the BBB[23]. Other articles also based their 3D BBB models on microfluidic channels separated by porous membranes of various materials - PDMS[21,26,27], PC[30], PE[28], PETE[22,23] - and various pore sizes that ranged from 0.4 mm to 7 mm. Nevertheless, none of these studies focused on the importance of the membrane material or pore size to the envisaged BBB function.
However, half of the selected articles, and curiously, the most recent ones, showed similar geometries with straight membrane-free channels. The advantage of the presence of a membrane is controversial in the literature regarding its role in the BBB model. Due to the relatively greater thickness of the artificial membrane compared to the basal lamina in vivo, cell-cell interactions are limited[34,35]. More recent articles use the diffusible factors produced by the cells in culture themselves for indirect communication between ACs and the BBB endothelium, without the physical restriction imposed by the artificial membrane[35].
Organs-on-chips are a currently sought alternative due to the possibility of physiologically relevant 3D in vitro single- or pluricellular culture and, more importantly, due to the possibility of laminar medium flow, mimicking the environment and shear stress found in capillary vessels. Also, the microchip culture of neural cells has shown more in vivo-like results, with more mature neurons and ACs[22]. However, the establishment of laminar flow might be cumbersome, given the need for apparatuses such as syringes, capillary tubes, and peristaltic pumps adapted for the sterile environment required for cell culture. Some studies have attempted to overcome this problem with creative alternatives, such as rocking platforms that allow the bidirectional flow of media within the microchips[18-20,30]. Regardless, the cerebral tissue is rather complex and involves a multitude of interacting cell types and ECM proteins. In vitro modeling at such a level is already challenging per se, whether or not the laminar flow is involved.
Regarding the material used for chip fabrication, conventional PDMS was used in most cases (57%)[22,25-29,31]. One of the studies adopted PMMA[23], which consists of the least hydrophobic material utilized in microfabrication. In addition, this material is ideal for mass production, and it is stiffer than PDMS. However, rapid prototyping is somewhat complicated by the fact that PMMA bonding is more difficult than the straightforward bonding of PDMS[36,37]. Conversely, PDMS may nonselectively adsorb proteins and hydrophobic molecules, which could possibly interfere with the characterization process of the BBB-on-a-chip and future drug screening applications[38]. Hence, some studies have presented alternatives to tackle this issue, such as covering the microchip surface with bovine serum albumin[39], grafting with anti-fouling molecules[40], or silanization[41].
For construction of the in vitro NVU, the cell cultures have been classically established in Transwell plates, mainly using primary brain ECs isolated from animal and human sources, which retain very tight barriers in vitro and may be useful tools for studying paracellular permeability[42,43]. Immortalized EC lines, such as HUVECs, have also been used in these BBB models, although they do not provide the permeability profile or the protein expression typically present in the human BBB[44]. Moreover, there is great difficulty in obtaining primary human brain ECs from healthy individuals[45].
The aforementioned drawbacks have been recently addressed with the use of iPSCs. After going through a complex differentiation protocol, these stem cells have shown in vivo-like barrier function[27]. Their BBB phenotype can be further enhanced by co-cultivation with other cells that exert barrier function in vivo, such as ACs and PCs[24,27]. In view of this aspect, we found in this systematic review a great diversity of iPSC commercially available cell lines from human sources, such as the IPS cell line IMR90-C4 from human fetal lung fibroblasts[18,19,21,23,24,28,30]. We further verified that one study used an iPSC line from unhealthy donors, with the advanced purpose of verifying BBB dysfunction in a pathological model and an attempt to correct this dysfunction through gene editing (CRISPR)[27]. In addition, building BBB models from human-derived iPSCs may be a valuable tool for personalized medicine, wherein a patient’s CNS disease could be precisely modeled in vitro, enabling the evaluation of personalized treatment.
On the other hand, iPSC-based BBB models have shown short-lived BBB function, which is generally maintained for up to 2 d[21]. Current efforts have been aimed at extending the BBB profile of these cultures[21,28,30]. 3D BBB on-a-chip devices form more physical and physiologically accurate morphologies. The cell-cell and cell-matrix interactions produced in microfluidic models generally provide a robust BBB function, with permeability to different substances (drugs, antibodies, proteins) that resemble that observed in vivo[18,19,27,28].
Most of the studies featured in this review (86%) differentiated their iPSCs into BMECs. This is expected since BMECs are the specialized cells in the CNS vasculature that show barrier properties. On the other hand, four studies differentiated the iPSCs into neural cells (neurons and ACs)[20,22,26,27]. The NVU, which makes up the BBB, is mainly composed of strict interactions among BMECs, the ECM, basal lamina, PCs, ACs, and adjacent neurons[11,46]. Even though neurons are indeed considered to be part of the BBB, they are physiologically positioned farther from the microvessels that make up the NVU (10 μm to 20 μm) than ACs or PCs[47]. The studies that reported co-culture of ECs with neurons mentioned in the current review did not clearly state the reasons for using neurons in their BBB models, but some studies stated that co-culture of iBMECs with neurons induces the upregulation of membrane transporters typical of the BBB, promoting a more robust function[29,48], as reported in a study by Wevers et al[20]. Microfluidic chips co-cultured both with iBMECs and neural cells showed a more robust phenotype regarding BMECs and strong indicators of epithelial cell barrier integrity and permeability (TEER from 1000 to 4000 Ω × cm2)[18,19,21,27,30].
Although studies have pointed out drawbacks using primary BMECs or iBMECs in the EC monolayer in BBB-on-a-chip, they still constitute a more reliable model. One of the biggest challenges in using iBMECs in BBB research is the difficulty in producing an in vivo-like BMEC phenotype in vitro following the currently available protocols, which generally result in more epithelial-like phenotypes with the suboptimal expression of membrane transporters[6,20,26,27,49]. A recent meta-analysis study showed that iPSCs could only be reliably differentiated into BMECs through exposition to endothelial ETS transcription factors ETS variant transcription factor 2, Friend leukemia integration 1 transcription factor, and Ets-related gene[49]. Nevertheless, the differentiation of iPSCs into iBMECs can further benefit from co-culture with neural cells, such as ACs and neurons[27,50]. While a more robust differentiation protocol is not established, some studies make use of alternative ways to make an EC layer, such as the use of HUVECs[22], primary BMECs[20], or even immortalized lines[10]. On the other hand, the isolation of human primary BMECs is a challenge, since they comprise less than 0.1% of the cells in the CNS and tend to de-differentiate in culture[27,51]. Studies with HUVECs generally point out some limitations, such as the formation of an incomplete EC monolayer[22]. Immortalized cells, despite being easily commercialized, generally constitute BBB models with poor barrier properties[27,52].
Fully iPSC-based chips were useful in modeling BBB disruptions from diseased patients, such as the alterations found in Huntington’s disease and in monocarboxylate transporter 8 deficiency[26,27]. Wevers et al[20] and Middelkamp et al[22] also differentiated the iPSCs into neurons, albeit not providing further justifications for doing so. With the exception of Vatine et al[27], who observed improved barrier functions with a fully iPSC-derived BBB chip, the other studies considered in this review have not versed on whether their obtained outcomes were positive or negatively dependent on the use of iPSCs.
Normally the microfluidic chips receive a coating made of collagen type IV/fibronectin before cell seeding[21,23,24,26-28,30,31]. These coating agents improve cell adhesion to the chip substrate through micropatterning, offering a fibrous base to guide cell differentiation and cell-to-cell contacts[16]. Motallebnejad et al[24] considered that the in vivo brain endothelial basement membrane is made up of laminins. Keeping this in mind, they compared the characteristics of the iPSC-derived ECs grown on an ECM based on E8 fragments of laminin 411 or laminin 511 (laminin511-E8) to ECs grown on the common collagen IV/fibronectin ECM. The authors found that the ECM made of laminin511-E8 presented more significant results, with the iPSC-ECs grown on this ECM exhibiting a longer-lasting EC phenotype and improved barrier stability compared to cells grown on the collagen IV/fibronectin ECM. iPSC-ECs grown on laminin511-E8 presented higher expression levels of junctional proteins such as claudin-5, VE-cadherin, and PECAM-1. Curiously, three other articles used a laminin ECM coating[22,26,27], but on the “brain side” channel of their microfluidic devices. One of the aforementioned studies also used the laminin coating on the “blood side” channel for culturing HUVECs, but without mentioning the importance of the laminin ECM[22].
Another important aspect of microfluidic device analyses in many studies is perfusion, whether by culture media or other biologically-relevant liquids, due to the shear stress and their important advantage in in vitro studies[10,16,53]. For BBB models, the role of shear stress on BBB function remains inconclusive. Among the studies selected for this review, eight (57%) considered shear stress[19-22,24,27,30,31], whereas two[22,30] designed their BBB mimics to minimize shear stress. Vatine et al[27] reported an increase in the expression of tight junction genes, such as tight junction protein 1/ZO-1, OCLN, PECAM-1, and cadherin 5/VE-cadherin, even under shear stress as low as 0.01 or 0.5 dyne/cm2. A study by Noorani et al[21] found ambiguous results for cultures under shear stress; perfusion did not induce changes in tight junction gene expression but decreased the permeability levels of sucrose and mannitol. In turn, the work by DeStefano et al[31] focused solely on the effect of increasing shear stress (0, 4, or 12 dyne/cm2) on the genotype and phenotype of iPSC-derived ECs. The authors found no correlation between the presence of shear stress and the expression of a more robust EC phenotype, since tight junctions are already formed under static conditions. Some authors considered that perfusion makes a bigger difference in BBB models made of non-iPSC-derived EC cultures such as HUVECs or primary ECs[31,54], whereas iPSC-derived ECs do not experience drastic changes in BBB markers upon shear stress[21,31]. Motallebnejad et al[24] gathered evidence that culturing iPSC-derived ECs on laminin511-E8 ECM improves the response of ECs to shear stress, wherein the cells exhibit changes in morphology, surface area, and the upregulation of tight junction proteins.
Regarding BBB applications, the specialized literature has not yet been able to establish a consistent microfluidic BBB model that can cover a broad range of applications, from modeling CNS disorders, including cancer metastasis, to screening for drugs able to permeate the BBB to treat such maladies. This is reflected in the goals of most studies selected to be part of this review. The great majority of the articles (roughly 80%) were aimed at developing a microfluidic BBB model, as well as assessing the convenience of the manufactured model by quantifying common molecular markers that are normally expressed in cells that make up the BBB in vivo[18-21,23,24,26-30]. Only one of the studies focused not on the BBB model itself but on the permeation potential of either commercial or synthesized polymer NPs through the in vitro 3D BBB mimic[25]. The BBB is a significant obstacle to the effective transport of large molecules for the treatment of brain disorders, and a study by Lee et al[25] showed that iPSC-ECs are appropriate for generating the 3D in vitro BBB model[25]. Surface modification of NPs with ligands for specific binding to BBB cells enhances NP accumulation in the brain through receptor-mediated endocytosis, providing good conditions to explore personalized therapies[25].
Another different application for the 3D BBB model was reported in a study by Wevers et al[20], which employed hypoxia together with hypoglycemia and lack of perfusion to successfully mimic ischemic stroke on-a-chip[20]. This hypoxic condition was also used to improve the iPSC differentiation to BMECs inside the microfluidic device by Park et al[28]. Based on the hypoxic environment found during the embryological development of the BBB in vivo, the whole differentiation step of iPSCs into ECs, both on a plate and in the microfluidic chip, was performed under low oxygen (5% O2). For the cells cultured under hypoxia, the authors observed the upregulation of diverse tight junction proteins and membrane transporters, contributing to a more robust BBB function. Moreover, they found that the differentiated ECs produce hypoxia-inducible factor 1α, a molecular marker whose presence was found to improve the function and durability of the BBB model[28].
Concerning the characterization techniques, the structural evaluation of the BBB model was mainly targeted by immunocytochemistry, whereas functional aspects have been mainly assessed using fluorescence techniques. Immunocytochemistry assays are the golden standard for identifying proteins related to the BBB, as well as for identifying different stages of BBB maturation (early, intermediate, and late phases of BBB development)[55]. Fluorescence assays are easily accessible and generally offer a vast array of markers that can be used for various purposes, from evaluating BBB permeability and integrity to neuronal functionality. Many studies mentioned TEER measurements as an important method to characterize BBB function[20,21,24,27,28,30,56]. However, they pose a number of disadvantages. The TEER values across BBB culture models are frequently influenced by disregarded physical and technical factors such as temperature, viscosity, and current density, generated by various electrode types, surface size, circumference, and porosity of the insert membrane, leading to severalfold differences within the same biological model[57]. Additionally, these TEER values can vary according to cell type (primary, immortalized, iPSC-derived), cell source (animal or human), and culture type (2D, 3D culture, co-culture)[58]. Therefore, the comparison of TEER values among different studies is highly discouraged in the relevant literature.
This systematic review highlights the main aspects of the current literature on BBB models on-a-chip, from iPSC differentiation to the NVU formation and characterization inside the microfluidic devices, also considering the functional duration of the proposed models by diverse approaches, focusing on future applications. However, the lack of uniform nomenclature and protocols made it a challenge to deeply analyze and comprehend all the steps in the processes involved.
The construction of BBB-on-a-chip still faces numerous challenges in producing feasible, reproducible, and reliable models. This becomes apparent in that, among the 14 articles studied in this review, 12 focused on the development of a robust in vitro model[18,19,21-24,26-31], and only 2 focused on the application of a model to study BBB disruption following ischemic stroke[20] and study the BBB permeability to polymer NPs[25]. Even though the use of iPSCs in microfluidic systems poses an advancement in these BBB constructs, there remain obstacles in the use of this type of cells, especially related to differentiation protocols, which need to be further developed in order to result in more dependable cell phenotypes. Moreover, on-chip technology remains a costly in vitro alternative, and the in-house manufacture of microfluidic devices is hampered by the need for a clean room and specialized equipment and personnel.
The integration of sensors to monitor barrier permeability and neuronal response to drug exposure is an important aspect scarcely addressed. The addition of such sensors could enable more precise and real-time measurements of drug transport across the BBB and its effects in brain cells. In terms of potential impacts, this technology can accelerate the development of new drugs by allowing for faster and more accurate screening of compounds that cross the BBB for therapeutic effects on the brain. One important step towards this scenario was already taken in the study by Vatine et al[27], who were successful in perfusing whole human blood through their microfluidic device, creating promising conditions to evaluate disease[27]. Moreover, more reliable BBB-on-a-chip constructs may help to better understand the in vivo functioning of the BBB across different individuals, representing a significant impact on personalized medicine.
CONCLUSION
Despite the well-known literature on iPSCs, microfluidic devices, and BBB in vitro models in isolation, the combination of the three subjects is a currently relevant theme, already presenting with advanced technology in the use of commercial microdevices for modeling the BBB. The BBB-on-a-chip models, in spite of their particularities in each study, have shown to be efficient for reconstructing the NVU in vitro, being able to reproduce the characteristics found in vivo in terms of structure and function. They have displayed promising qualities for investigating barrier disorders, drug delivery, mimicking disease, and personalized medicine.
ARTICLE HIGHLIGHTS
Research background
Induced pluripotent stem cells (iPSCs) offer a potential alternative to building blood-brain barrier (BBB)-on-a-chip models that more closely resemble the structure and functions found in vivo.
Research motivation
iPSC-derived BBB models on-a-chip are a promising field that still lacks improvements and uniformity within the specialized literature.
Research objectives
To search the literature and analyze the selected data on the cultivation of iPSCs within microfluidic environments to mimic the human BBB.
Research methods
A literature search using the PRISMA approach using the following terms: “iPSC,” “BBB,” and “microfluidic device,” wherein 14 studies were selected based on the inclusion and exclusion criteria, and data were organized into three tables and one flow chart.
Research results
Studies have been found from 2017 to 2022, wherein the microdevices were either commercially available or manufactured in-house using soft-lithography. iPSCs were differentiated into endothelial or neural cells and seeded in the chips individually or in co-culture onto an extracellular-matrix layer mainly made of collagen IV/fibronectin. The selected studies focused principally on the structural and functional design of the human BBB model on-a-chip, displaying future application potential for drug screening and disease modeling.
Research conclusions
Despite the lack of consensus in protocols, the studies analyzed herein were able to efficiently reproduce a human microfluidic BBB in vitro making use of iPSCs.
Research perspectives
The developed BBB models on-a-chip have shown great potential to study physiopathological conditions related to the central nervous system, to apply advanced technology approaches for novel therapies (drug delivery through nanoparticles), and to develop genetic modification therapies for BBB dysfunctions through clustered regularly interspaced short palindromic repeats methodologies.
Footnotes
Conflict-of-interest statement: The authors have no conflicts of interest to declare.
PRISMA 2009 Checklist statement: The authors have read the PRISMA guidelines, and the manuscript was prepared and revised according to the PRISMA 2009 Checklist.
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Corresponding Author’s Membership in Professional Societies: Hospital Israelita Albert Einstein.
Peer-review started: February 15, 2023
First decision: April 10, 2023
Article in press: May 8, 2023
Specialty type: Cell and tissue engineering
Country/Territory of origin: Brazil
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C, C
Grade D (Fair): D
Grade E (Poor): 0
P-Reviewer: Chen YJ, China; Lu Q, China; Thummer RP, India S-Editor: Wang JJ L-Editor: Filipodia P-Editor: Zhang XD
Contributor Information
Arielly da Hora Alves, Hospital Israelita Albert Einstein, São Paulo 05529-060, São Paulo, Brazil.
Mariana Penteado Nucci, Hospital Israelita Albert Einstein, São Paulo 05529-060, São Paulo, Brazil; Hospital das Clínicas HCFMUSP, Faculdade de Medicina, Universidade de São Paulo, São Paulo 05403-010, São Paulo, Brazil.
Nicole Mastandrea Ennes do Valle, Hospital Israelita Albert Einstein, São Paulo 05529-060, São Paulo, Brazil.
Juliana Morais Missina, Hospital Israelita Albert Einstein, São Paulo 05529-060, São Paulo, Brazil.
Javier Bustamante Mamani, Hospital Israelita Albert Einstein, São Paulo 05529-060, São Paulo, Brazil.
Gabriel Nery Albuquerque Rego, Hospital Israelita Albert Einstein, São Paulo 05529-060, São Paulo, Brazil.
Olivia Furiama Metropolo Dias, Hospital Israelita Albert Einstein, São Paulo 05529-060, São Paulo, Brazil.
Murilo Montenegro Garrigós, Hospital Israelita Albert Einstein, São Paulo 05529-060, São Paulo, Brazil.
Fernando Anselmo de Oliveira, Hospital Israelita Albert Einstein, São Paulo 05529-060, São Paulo, Brazil.
Lionel Fernel Gamarra, Hospital Israelita Albert Einstein, São Paulo 05529-060, São Paulo, Brazil. lgamarra@einstein.br.
References
- 1.Shi Y, Inoue H, Wu JC, Yamanaka S. Induced pluripotent stem cell technology: a decade of progress. Nat Rev Drug Discov. 2017;16:115–130. doi: 10.1038/nrd.2016.245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lippmann ES, Al-Ahmad A, Azarin SM, Palecek SP, Shusta EV. A retinoic acid-enhanced, multicellular human blood-brain barrier model derived from stem cell sources. Sci Rep. 2014;4:4160. doi: 10.1038/srep04160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Wiegand C, Banerjee I. Recent advances in the applications of iPSC technology. Curr Opin Biotechnol. 2019;60:250–258. doi: 10.1016/j.copbio.2019.05.011. [DOI] [PubMed] [Google Scholar]
- 4.Ray A, Joshi JM, Sundaravadivelu PK, Raina K, Lenka N, Kaveeshwar V, Thummer RP. An Overview on Promising Somatic Cell Sources Utilized for the Efficient Generation of Induced Pluripotent Stem Cells. Stem Cell Rev Rep. 2021;17:1954–1974. doi: 10.1007/s12015-021-10200-3. [DOI] [PubMed] [Google Scholar]
- 5.Sundaravadivelu PK, Raina K, Thool M, Ray A, Joshi JM, Kaveeshwar V, Sudhagar S, Lenka N, Thummer RP. Tissue-Restricted Stem Cells as Starting Cell Source for Efficient Generation of Pluripotent Stem Cells: An Overview. Adv Exp Med Biol. 2022;1376:151–180. doi: 10.1007/5584_2021_660. [DOI] [PubMed] [Google Scholar]
- 6.Workman MJ, Svendsen CN. Recent advances in human iPSC-derived models of the blood-brain barrier. Fluids Barriers CNS. 2020;17:30. doi: 10.1186/s12987-020-00191-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Sweeney MD, Zhao Z, Montagne A, Nelson AR, Zlokovic BV. Blood-Brain Barrier: From Physiology to Disease and Back. Physiol Rev. 2019;99:21–78. doi: 10.1152/physrev.00050.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kugler EC, Greenwood J, MacDonald RB. The "Neuro-Glial-Vascular" Unit: The Role of Glia in Neurovascular Unit Formation and Dysfunction. Front Cell Dev Biol. 2021;9:732820. doi: 10.3389/fcell.2021.732820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Williams-Medina A, Deblock M, Janigro D. In vitro Models of the Blood-Brain Barrier: Tools in Translational Medicine. Front Med Technol. 2020;2:623950. doi: 10.3389/fmedt.2020.623950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wevers NR, Kasi DG, Gray T, Wilschut KJ, Smith B, van Vught R, Shimizu F, Sano Y, Kanda T, Marsh G, Trietsch SJ, Vulto P, Lanz HL, Obermeier B. A perfused human blood-brain barrier on-a-chip for high-throughput assessment of barrier function and antibody transport. Fluids Barriers CNS. 2018;15:23. doi: 10.1186/s12987-018-0108-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Abbott NJ, Patabendige AA, Dolman DE, Yusof SR, Begley DJ. Structure and function of the blood-brain barrier. Neurobiol Dis. 2010;37:13–25. doi: 10.1016/j.nbd.2009.07.030. [DOI] [PubMed] [Google Scholar]
- 12.Van Norman GA. Limitations of Animal Studies for Predicting Toxicity in Clinical Trials: Is it Time to Rethink Our Current Approach? JACC Basic Transl Sci. 2019;4:845–854. doi: 10.1016/j.jacbts.2019.10.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Clevers H. Modeling Development and Disease with Organoids. Cell. 2016;165:1586–1597. doi: 10.1016/j.cell.2016.05.082. [DOI] [PubMed] [Google Scholar]
- 14.Halldorsson S, Lucumi E, Gómez-Sjöberg R, Fleming RMT. Advantages and challenges of microfluidic cell culture in polydimethylsiloxane devices. Biosens Bioelectron. 2015;63:218–231. doi: 10.1016/j.bios.2014.07.029. [DOI] [PubMed] [Google Scholar]
- 15.Cui B, Cho SW. Blood-brain barrier-on-a-chip for brain disease modeling and drug testing. BMB Rep. 2022;55:213–219. doi: 10.5483/BMBRep.2022.55.5.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Liang Y, Yoon JY. In situ sensors for blood-brain barrier (BBB) on a chip. Sens Actuators Rep. 2021;3:100031. [Google Scholar]
- 17.Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gøtzsche PC, Ioannidis JP, Clarke M, Devereaux PJ, Kleijnen J, Moher D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. PLoS Med. 2009;6:e1000100. doi: 10.1371/journal.pmed.1000100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kurosawa T, Sako D, Tega Y, Debori Y, Tomihara Y, Aoyama K, Kubo Y, Amano N, Deguchi Y. Construction and Functional Evaluation of a Three-Dimensional Blood-Brain Barrier Model Equipped With Human Induced Pluripotent Stem Cell-Derived Brain Microvascular Endothelial Cells. Pharm Res. 2022;39:1535–1547. doi: 10.1007/s11095-022-03249-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Fengler S, Kurkowsky B, Kaushalya SK, Roth W, Fava E, Denner P. Human iPSC-derived brain endothelial microvessels in a multi-well format enable permeability screens of anti-inflammatory drugs. Biomaterials. 2022;286:121525. doi: 10.1016/j.biomaterials.2022.121525. [DOI] [PubMed] [Google Scholar]
- 20.Wevers NR, Nair AL, Fowke TM, Pontier M, Kasi DG, Spijkers XM, Hallard C, Rabussier G, van Vught R, Vulto P, de Vries HE, Lanz HL. Modeling ischemic stroke in a triculture neurovascular unit on-a-chip. Fluids Barriers CNS. 2021;18:59. doi: 10.1186/s12987-021-00294-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Noorani B, Bhalerao A, Raut S, Nozohouri E, Bickel U, Cucullo L. A Quasi-Physiological Microfluidic Blood-Brain Barrier Model for Brain Permeability Studies. Pharmaceutics. 2021;13 doi: 10.3390/pharmaceutics13091474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Middelkamp HHT, Verboven AHA, De Sá Vivas AG, Schoenmaker C, Klein Gunnewiek TM, Passier R, Albers CA, 't Hoen PAC, Nadif Kasri N, van der Meer AD. Cell type-specific changes in transcriptomic profiles of endothelial cells, iPSC-derived neurons and astrocytes cultured on microfluidic chips. Sci Rep. 2021;11:2281. doi: 10.1038/s41598-021-81933-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Choi B, Choi JW, Jin H, Sim HR, Park JH, Park TE, Kang JH. Condensed ECM-based nanofilms on highly permeable PET membranes for robust cell-to-cell communications with improved optical clarity. Biofabrication. 2021;13 doi: 10.1088/1758-5090/ac23ad. [DOI] [PubMed] [Google Scholar]
- 24.Motallebnejad P, Azarin SM. Chemically defined human vascular laminins for biologically relevant culture of hiPSC-derived brain microvascular endothelial cells. Fluids Barriers CNS. 2020;17:54. doi: 10.1186/s12987-020-00215-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lee SWL, Campisi M, Osaki T, Possenti L, Mattu C, Adriani G, Kamm RD, Chiono V. Modeling Nanocarrier Transport across a 3D In Vitro Human Blood-Brain-Barrier Microvasculature. Adv Healthc Mater. 2020;9:e1901486. doi: 10.1002/adhm.201901486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Jagadeesan S, Workman MJ, Herland A, Svendsen CN, Vatine GD. Generation of a Human iPSC-Based Blood-Brain Barrier Chip. J Vis Exp. 2020 doi: 10.3791/60925. [DOI] [PubMed] [Google Scholar]
- 27.Vatine GD, Barrile R, Workman MJ, Sances S, Barriga BK, Rahnama M, Barthakur S, Kasendra M, Lucchesi C, Kerns J, Wen N, Spivia WR, Chen Z, Van Eyk J, Svendsen CN. Human iPSC-Derived Blood-Brain Barrier Chips Enable Disease Modeling and Personalized Medicine Applications. Cell Stem Cell. 2019;24:995–1005.e6. doi: 10.1016/j.stem.2019.05.011. [DOI] [PubMed] [Google Scholar]
- 28.Park TE, Mustafaoglu N, Herland A, Hasselkus R, Mannix R, FitzGerald EA, Prantil-Baun R, Watters A, Henry O, Benz M, Sanchez H, McCrea HJ, Goumnerova LC, Song HW, Palecek SP, Shusta E, Ingber DE. Hypoxia-enhanced Blood-Brain Barrier Chip recapitulates human barrier function and shuttling of drugs and antibodies. Nat Commun. 2019;10:2621. doi: 10.1038/s41467-019-10588-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Campisi M, Shin Y, Osaki T, Hajal C, Chiono V, Kamm RD. 3D self-organized microvascular model of the human blood-brain barrier with endothelial cells, pericytes and astrocytes. Biomaterials. 2018;180:117–129. doi: 10.1016/j.biomaterials.2018.07.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wang YI, Abaci HE, Shuler ML. Microfluidic blood-brain barrier model provides in vivo-like barrier properties for drug permeability screening. Biotechnol Bioeng. 2017;114:184–194. doi: 10.1002/bit.26045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.DeStefano JG, Xu ZS, Williams AJ, Yimam N, Searson PC. Effect of shear stress on iPSC-derived human brain microvascular endothelial cells (dhBMECs) Fluids Barriers CNS. 2017;14:20. doi: 10.1186/s12987-017-0068-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Watanabe K, Ueno M, Kamiya D, Nishiyama A, Matsumura M, Wataya T, Takahashi JB, Nishikawa S, Muguruma K, Sasai Y. A ROCK inhibitor permits survival of dissociated human embryonic stem cells. Nat Biotechnol. 2007;25:681–686. doi: 10.1038/nbt1310. [DOI] [PubMed] [Google Scholar]
- 33.Vernardis SI, Terzoudis K, Panoskaltsis N, Mantalaris A. Human embryonic and induced pluripotent stem cells maintain phenotype but alter their metabolism after exposure to ROCK inhibitor. Sci Rep. 2017;7:42138. doi: 10.1038/srep42138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Jiang L, Li S, Zheng J, Li Y, Huang H. Recent Progress in Microfluidic Models of the Blood-Brain Barrier. Micromachines (Basel) 2019;10 doi: 10.3390/mi10060375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kaisar MA, Sajja RK, Prasad S, Abhyankar VV, Liles T, Cucullo L. New experimental models of the blood-brain barrier for CNS drug discovery. Expert Opin Drug Discov. 2017;12:89–103. doi: 10.1080/17460441.2017.1253676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Young EW, Berthier E, Guckenberger DJ, Sackmann E, Lamers C, Meyvantsson I, Huttenlocher A, Beebe DJ. Rapid prototyping of arrayed microfluidic systems in polystyrene for cell-based assays. Anal Chem. 2011;83:1408–1417. doi: 10.1021/ac102897h. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Chen Y, Zhang L, Chen G. Fabrication, modification, and application of poly(methyl methacrylate) microfluidic chips. Electrophoresis. 2008;29:1801–1814. doi: 10.1002/elps.200700552. [DOI] [PubMed] [Google Scholar]
- 38.Toepke MW, Beebe DJ. PDMS absorption of small molecules and consequences in microfluidic applications. Lab Chip. 2006;6:1484–1486. doi: 10.1039/b612140c. [DOI] [PubMed] [Google Scholar]
- 39.Ostuni E, Chen CS, Ingber DE, Whitesides GM. Selective deposition of proteins and cells in arrays of microwells. Langmuir. 2001;17:2828–2834. [Google Scholar]
- 40.Gokaltun A, Yarmush ML, Asatekin A, Usta OB. Recent advances in nonbiofouling PDMS surface modification strategies applicable to microfluidic technology. Technology (Singap World Sci) 2017;5:1–12. doi: 10.1142/S2339547817300013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Jon S, Seong J, Khademhosseini A, Tran TNT, Laibinis PE, Langer R. Construction of nonbiofouling surfaces by polymeric self-assembled monolayers. Langmuir. 2003;19:9989–9993. [Google Scholar]
- 42.Lippmann ES, Azarin SM, Kay JE, Nessler RA, Wilson HK, Al-Ahmad A, Palecek SP, Shusta EV. Derivation of blood-brain barrier endothelial cells from human pluripotent stem cells. Nat Biotechnol. 2012;30:783–791. doi: 10.1038/nbt.2247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Helms HC, Abbott NJ, Burek M, Cecchelli R, Couraud PO, Deli MA, Förster C, Galla HJ, Romero IA, Shusta EV, Stebbins MJ, Vandenhaute E, Weksler B, Brodin B. In vitro models of the blood-brain barrier: An overview of commonly used brain endothelial cell culture models and guidelines for their use. J Cereb Blood Flow Metab. 2016;36:862–890. doi: 10.1177/0271678X16630991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Man S, Ubogu EE, Williams KA, Tucky B, Callahan MK, Ransohoff RM. Human brain microvascular endothelial cells and umbilical vein endothelial cells differentially facilitate leukocyte recruitment and utilize chemokines for T cell migration. Clin Dev Immunol. 2008;2008:384982. doi: 10.1155/2008/384982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Delsing L, Herland A, Falk A, Hicks R, Synnergren J, Zetterberg H. Models of the blood-brain barrier using iPSC-derived cells. Mol Cell Neurosci. 2020;107:103533. doi: 10.1016/j.mcn.2020.103533. [DOI] [PubMed] [Google Scholar]
- 46.Banerjee S, Bhat MA. Neuron-glial interactions in blood-brain barrier formation. Annu Rev Neurosci. 2007;30:235–258. doi: 10.1146/annurev.neuro.30.051606.094345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Wong AD, Ye M, Levy AF, Rothstein JD, Bergles DE, Searson PC. The blood-brain barrier: an engineering perspective. Front Neuroeng. 2013;6:7. doi: 10.3389/fneng.2013.00007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Appelt-Menzel A, Cubukova A, Günther K, Edenhofer F, Piontek J, Krause G, Stüber T, Walles H, Neuhaus W, Metzger M. Establishment of a Human Blood-Brain Barrier Co-culture Model Mimicking the Neurovascular Unit Using Induced Pluri- and Multipotent Stem Cells. Stem Cell Reports. 2017;8:894–906. doi: 10.1016/j.stemcr.2017.02.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Lu TM, Houghton S, Magdeldin T, Durán JGB, Minotti AP, Snead A, Sproul A, Nguyen DT, Xiang J, Fine HA, Rosenwaks Z, Studer L, Rafii S, Agalliu D, Redmond D, Lis R. Pluripotent stem cell-derived epithelium misidentified as brain microvascular endothelium requires ETS factors to acquire vascular fate. Proc Natl Acad Sci U S A. 2021;118 doi: 10.1073/pnas.2016950118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Canfield SG, Stebbins MJ, Morales BS, Asai SW, Vatine GD, Svendsen CN, Palecek SP, Shusta EV. An isogenic blood-brain barrier model comprising brain endothelial cells, astrocytes, and neurons derived from human induced pluripotent stem cells. J Neurochem. 2017;140:874–888. doi: 10.1111/jnc.13923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Cecchelli R, Berezowski V, Lundquist S, Culot M, Renftel M, Dehouck MP, Fenart L. Modelling of the blood-brain barrier in drug discovery and development. Nat Rev Drug Discov. 2007;6:650–661. doi: 10.1038/nrd2368. [DOI] [PubMed] [Google Scholar]
- 52.Kamiichi A, Furihata T, Kishida S, Ohta Y, Saito K, Kawamatsu S, Chiba K. Establishment of a new conditionally immortalized cell line from human brain microvascular endothelial cells: a promising tool for human blood-brain barrier studies. Brain Res. 2012;1488:113–122. doi: 10.1016/j.brainres.2012.09.042. [DOI] [PubMed] [Google Scholar]
- 53.Seebach J, Dieterich P, Luo F, Schillers H, Vestweber D, Oberleithner H, Galla HJ, Schnittler HJ. Endothelial barrier function under laminar fluid shear stress. Lab Invest. 2000;80:1819–1831. doi: 10.1038/labinvest.3780193. [DOI] [PubMed] [Google Scholar]
- 54.DeStefano JG, Williams A, Wnorowski A, Yimam N, Searson PC, Wong AD. Real-time quantification of endothelial response to shear stress and vascular modulators. Integr Biol (Camb) 2017;9:362–374. doi: 10.1039/c7ib00023e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Dermietzel R, Krause D. Molecular anatomy of the blood-brain barrier as defined by immunocytochemistry. Int Rev Cytol. 1991;127:57–109. doi: 10.1016/s0074-7696(08)60692-0. [DOI] [PubMed] [Google Scholar]
- 56.Srinivasan B, Kolli AR, Esch MB, Abaci HE, Shuler ML, Hickman JJ. TEER measurement techniques for in vitro barrier model systems. J Lab Autom. 2015;20:107–126. doi: 10.1177/2211068214561025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Vigh JP, Kincses A, Ozgür B, Walter FR, Santa-Maria AR, Valkai S, Vastag M, Neuhaus W, Brodin B, Dér A, Deli MA. Transendothelial Electrical Resistance Measurement across the Blood-Brain Barrier: A Critical Review of Methods. Micromachines (Basel) 2021;12 doi: 10.3390/mi12060685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Shah B, Dong X. Current Status of In vitro Models of the Blood-brain Barrier. Curr Drug Deliv. 2022;19:1034–1046. doi: 10.2174/1567201819666220303102614. [DOI] [PMC free article] [PubMed] [Google Scholar]