Abstract
This study presents the 1st method Parfait-Hounsinou, a method that makes it very easy to detect saltwater intrusion in groundwater. The method relies on the commonly sampled ion concentrations. This method involves several steps, namely: Chemical analyzes to determine the concentrations of majors ions and TDS in groundwater, production and study of the spatial distribution of chemical parameters in groundwater (TDS, Cl−…), delimitation of a probable area of saltwater intrusion in groundwater and the production and the study of spie chart where area of pie slices represents ions contents or group of ions content in the groundwater sample of the probable area of the saltwater intrusion in groundwater and radius length represents the Relative Content Index. The method is applied to groundwater data collected from the municipality of Abomey-Calavi in Benin. The method is also compared to other methods used for saltwater intrusion including the Scholler-Berkaloff and Stiff diagrams and the Revelle Index. Comparatively to Scholler-Berkaloff and Stiff diagrams, the 1st method Parfait –Hounsinou, at spie chart level, the area of pie slices make it easier to compare the major cations between them and the majors anions between them and the Relative Content Index of the chloride ion allows further confirmation of the saltwater intrusion and its extent.
-
•
There are frequent saltwater intrusion into groundwater which are important freshwater reserves for our planet.
-
•
Governments around the world should delineate the levels of ocean intrusion into groundwater using scientific methods such as this method and seek to limit this saltwater intrusion.
-
•
In this article we have proposed a simple and reliable method based on a series of irrefutable proofs of saltwater intrusion into groundwater.
Keywords: Inorganic chemistry, Hydrogeochemistry, Saltwater intrusion, Groundwater, Benin
Method name: The 1st method Parfait-Hounsinou used for the detection of saltwater intrusion in groundwater
Graphical abstract

Specifications table
| Subject area: | Environmental Science |
| More specific subject area: | Hydrogeochemistry |
| Name of your method: | The 1st method Parfait-Hounsinou method for the detection of saltwater intrusion in groundwater. |
| Name and reference of original method: | None |
| Resource availability: | Conductimeter, spectrophotometer, program arc-view, program "Diagram 2″ |
Method details
Groundwater is a fairly good quality freshwater. Unfortunately, with global warming, there is a gradual melting of the polar ice cap and the water formed flows into the oceans, the volumes of which are constantly increasing. There is then an increasing intrusion of the oceans into coastal groundwater and the quality of the latter is becoming increasingly poor. After contamination, the restoration of groundwater quality is difficult, it usually takes a long time to return to its natural state [1,2]. It is very important to monitor and control the quality of coastal groundwater in order to detect saltwater intrusions and their extent and then, if necessary, alert the public authorities who can take the necessary measures in time. The detection of a saltwater intrusion in groundwater is multidisciplinary. Recently, to detect marine intrusions in groundwater, among authors have used the Scholler-Berkaloff and Stiff diagrams [3], [4], [5], [6] and other authors have used the Revelle index [4,7].
We present here the steps of the 1st method Parfait-Hounsinou for the detection of saltwater intrusion in groundwater, the application of this method to groundwater in the municipality of Abomey-Calavi and the comparaison of the 1st method Parfait-Hounsinou method to the methods usually used to study the saltwater intrusion in groundwater.
Steps for using the 1st method Parfait-Hounsinou for the detection of saltwater intrusion in groundwater
We have proposed a new method for detecting saltwater intrusion in groundwater. This method is carried out in several stages, namely: Chemical analyzes of groundwater, realization and study of spatial distribution maps of TDS, Cl-…, delimitation of a probable zone of saltwater intrusion in groundwater and the realization and the study of spie chart.
-
-
Chemical analyzes of groundwater:
The study environment is located at the edge of a sea, an ocean or another surface saltwater. Sampling sites should be chosen within the same aquifer. Some sample sites will be close to the surface saltwater, other sample sites will be relatively far away, and other sample sites will be far from the surface saltwater. The chemical analyzes to be carried out are the measurement of the TDS and the concentrations of the major ions in the groundwater, namely Na+, K+, Mg2+, Ca2+, Cl−, SO42−, HCO3− and CO32−.
-
-
Realization and study of spatial distribution maps of TDS and Cl−:
Seawater is very excessively mineralized and in particular, seawater is excessively charged with chloride ions and sodium ions. If coastal soils are permeable enough, seawater infiltrates groundwater. The mineralization and the Cl− and Na+ ions contents of the coastal groundwater are then very high. When there is saltwater intrusion into groundwater, the TDS reflecting the overall mineralization of coastal groundwater is significantly higher than the TDS of groundwater far from the oceans or the surface saltwater. Similarly, chloride ion concentrations in coastal groundwater in which there is saltwater intrusion are significantly higher than in groundwater far from the ocean or sea or surface saltwater. When we want to detect saltwater intrusion in groundwater, it is necessary to start by making maps of the spatial variation of the TDS and the chloride ion content of the groundwater in the study area. The contrast of these values for groundwater near and far from the ocean or surface saltwater suggests saltwater intrusion.
It should be noted that the spatial distribution map of Cl− ions in groundwater is very useful for detecting saltwater intrusion in these groundwaters because the soil is devoid of Cl− ion. But it is illusory to represent a map of the spatial variation of the sodium ion concentration of these groundwaters to prove a saltwater intrusion because the soil contains various concentrations of sodium ions which can be released into the groundwater. On the other hand, it is necessary to draw the curve of variation of the concentration of sodium ions according to the concentration of chloride ions. When this curve is linear and the correlation coefficient is close to 1, we conclude that the Na+ and Cl− ions of these groundwaters have the same origin, namely the marine origin or surface saltwater origin.
Once the maps of spatial variation of TDS and Cl− are produced and the curve of variation of Na+ as a function of Cl− plotted, if it is noticed that the values of TDS and Cl− ions of coastal groundwater are clearly higher than those of groundwater far from the ocean or surface saltwater and it is observed that the high levels of Na+ and Cl− ions in coastal groundwater are both of the same origin, then there is potentially a saltwater intrusion in the groundwaters.
-
-
Delimitation of a probable zone of saltwater intrusion in groundwater:
Groundwater samples taken near the sea or near a surface saltwater and which have TDS values and Cl− contents significantly higher than those of all other samples in the study area make it possible to delimit the probable zone of saltwater intrusion in the study area.
-
-
The realization and the study of spie chart:
For each groundwater sample from the probable saltwater intrusion zone, are determined, the percentages relative to the TDS of the following concentrations: (Na+ + K+), Mg2+, Ca2+, Cl−, SO42− and (HCO3− + CO32−). Similarly, for the entire study area, the average percentage relative to the average TDS of the average concentrations of (Na+ + K+), Mg2+, Ca2+, Cl−, SO42− and (HCO3− + CO32−).
For each groundwater sample from the probable saltwater intrusion zone, for each ions or ions group (Na+ + K+), Mg2+, Ca2+, Cl−, SO42− and (HCO3− + CO32−), we determine the Index of the Relative Content by calculating the ratio of its percentage by the percentage of its average across the study area.
On the radar plot of the Relative Content Index, radius length represents the Relative Content Index corresponding to the values of the major ion contents in the groundwater of the study area (example on the Fig. 1). On this diagram, Cl− is placed at the highest peak and clockwise is placed SO42− and (HCO3− + CO32−). Always starting from Cl− in the counterclockwise direction we place the cations (Na+ + K+), Mg2+ and Ca2+.
Fig. 1.
Shape of the Radar plot of Relative Content Index for a sample of groundwater in which there is saltwater intrusion.
When there is saltwater intrusion in groundwater, the radius length of the Cl- ion is the highest in the radar plot. When the Relative Content Index of the Cl- ion of a water sample is very high (greater than 10) it means that the saltwater intrusion is concentrated around the corresponding well and that the saltwater intrusion is not too widespread in the study area.
Using the diagram of Spie chart the area of pie slices represents the contents for each sample of the likely area of saltwater intrusion and radius length represents the Relative Content Index. We have chosen to color the pie slices representing the anions with the color blue or the color green then we have chosen to color the pie slices representing the cations with the color red or the color orange or yellow (example in Fig. 2). This particular diagram of Spie chart use for detecting saltwater intrusion in groundwater is named the 1st Parfait-Hounsinou diagram.
Fig. 2.
Example of the 1st Parfait-Hounsinou diagram (Spie chart where area of pie slices represents ions contents in the probable zone of saltwater intrusion in groundwater and radius length represents the Relative Content Index).
From the highest point of the 1st Parfait-Hounsinou diagram, the surfaces of pie slices vary in a decreasing way for anions in a clockwise direction and for cations in a counterclockwise direction when there is saltwater intrusion in groundwater.
In the Spie chart 1st Parfait-Hounsinou diagram example of Fig. 2, the Ca2+ ion concentration is not visible on the diagram because the Ca2+ ion content is very low compared to the contents of the other major ions.
Application of the 1st method Parfait-Hounsinou to groundwater in the municipality of Abomey-Calavi
Measurement of physico-chemical parameters of groundwater in the municipality of Abomey-Calavi
-
-
Description of the study area:
Abomey-Calavi is located in the southern part of the Republic of Benin in the department of Atlantic, between latitudes 6°20′23.4′' and 6°42′6.6′' North and longitudes 2°14′13.8′' and 2°25′7.8′' East [8]. The municipality of Abomey-Calavi covers an area of 536 km². The municipality of Abomey-Calavi is limited to the south by the Atlantic Ocean, to the east by the municipalities of Cotonou and Sô-Ava, to the west by the municipalities of Ouidah and Tori-Bossito and to the north by the municipality of Zè (Fig. 3). The average monthly precipitation is 100 mm. The municipality of Abomey-Calavi is subdivided into 9 districts: Abomey-Calavi center, Godomey, Akassato, Zinvié, Ouèdo, Togba, Hêvié, Kpanroun and Glo-Djigbé (Fig. 4).
-
-
Sampling and chemical analyzes
Fig. 3.
Geographic location of Abomey-Calavi in Benin.
Fig. 4.
The districts of the municipality of Abomey-Calavi.
For this study, we selected half a hundred wells throughout the municipality of Abomey-Calavi (Fig. 5). The wells water samples were collected using standard sampling procedures. We analyzed in the laboratory, using the methods described in the book “Le Rodier”, the TDS and the major ions, namely Ca2+, Na+, K+, Mg2+, Cl−, SO42−, CO32− and HCO3−. To measure the TDS we used a turbidimeter and to determine the concentrations of the major ions we used a spectrophotometer.
Fig. 5.
Sites of withdrawal of waters of well.
Spatial distribution of physico-chemical parameters
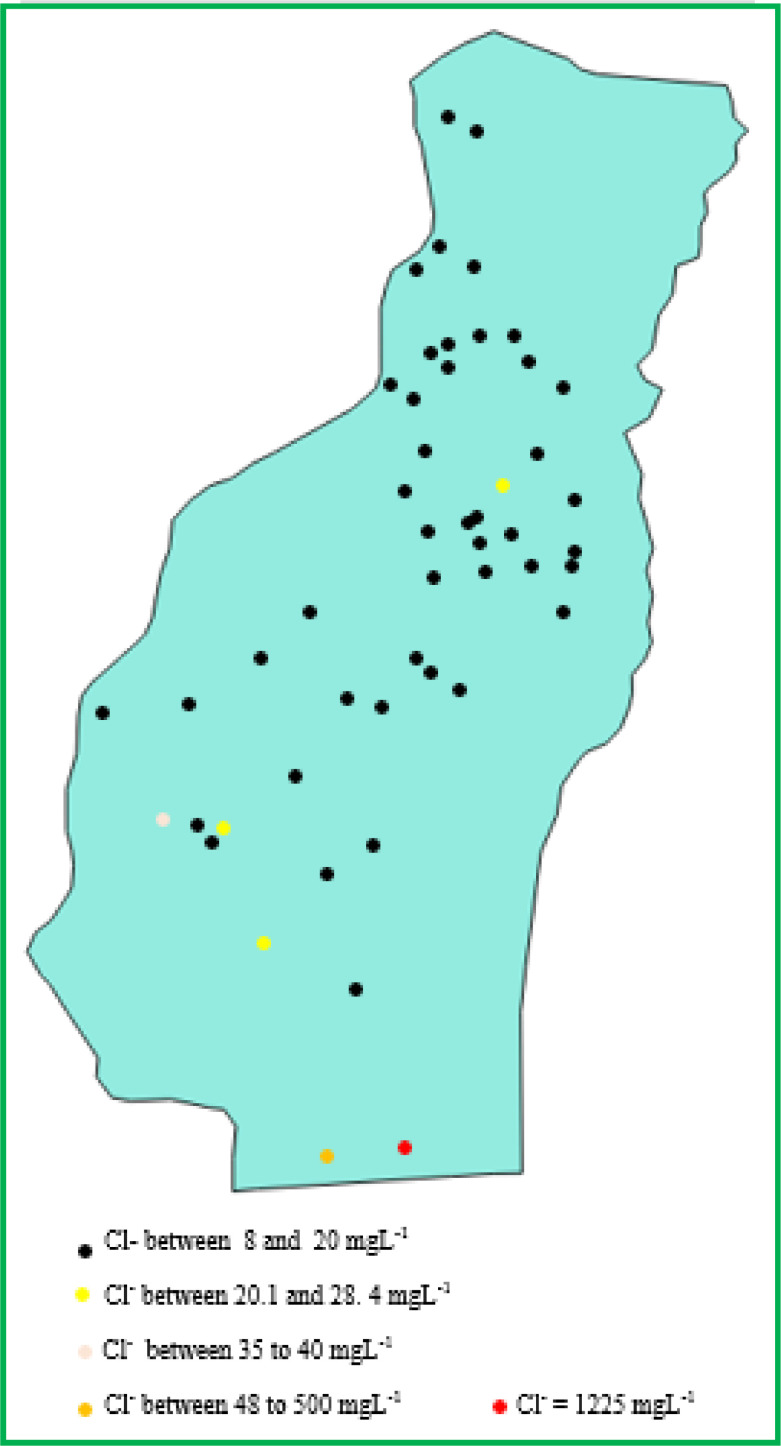
The spatial distribution of TDS and Cl− (Figs. 6 and 7) in the groundwater of the municipality of Abomey-Calavi shows that these values are significantly higher in the coastal waters of the Togbin village in the district of Godomey than in the groundwater of all the rest of the municipality. Moreover, the curve of the variation of Na+ as a function of Cl− in these groundwaters is linear with a correlation coefficient equal to 1. Na+ and Cl− in these groundwaters therefore have the same marine origin (Fig. 8).
Fig. 6.
Spatial variation of TDS values of wells water in the municipality of Abomey-Calavi.
Fig. 7.
Spatial variation in chloride content in wells water in the municipality of Abomey-Calavi.
Fig. 8.
Na+ = f (Cl−) of wells water in the district of Godomey.
There is therefore potentially a marine intrusion into the groundwater of the district of Godomey.
Delimitation of a probable zone of saltwater intrusion in groundwater in the municipality of Abomey-Calavi
The two water samples from the 2 wells of the municipality of Abomey-Calavi closest to the sea have TDS values and Cl− contents significantly higher than those of all the water samples from the other wells of the municipality of Abomey-Calavi (Figs. 6 and 7). These two water samples are located in the Godomey district precisely in the village of Togbin. The probable area of saltwater intrusion in groundwater in the municipality of Abomey-Calavi is the village of Togbin in the district of Godomey.
Production and the study of the 1st Parfait-Hounsinou diagram
We analyzed the major ions and the TDS in 68 wells of the 9 districts of the municipality of Abomey-Calavi. The district of Godomey is the district of the municipality of Abomey-Calavi located at the edge of the ocean. Therefore, we can examine more accurately the influence of seawater on the groundwater of Godomey district. We examined the content of the major ions of the water samples of the district of Godomey compared to the averages of these major ions in the municipality of Abomey-Calavi (Table 1).
-
-
Determination of the Index of the Relative Content of the water samples analyzed in the district of Godomey.
Table 1.
Determination of a new indicator: The Index of the Relative Content of majors ions in the water of the sample 1 in the district of Godomey.
| Parameters | Average of the ion content in the municipality | % of the ion content in the municipality | Content of ion in the groundwater sample 1 | % of the ion content in the groundwater sample 1 | Relative Content Index for the sample 1 |
|---|---|---|---|---|---|
| Cl− | 33.78 | 84.64% | 12.43 | 20.37% | 0.24 |
| SO42− | 3.9 | 9.77% | 2 | 3.28% | 0.33 |
| HCO3−+ CO32− | 21.9 | 54.87% | 73.239 | 120.06% | 2.18 |
| Ca2+ | 3.92 | 9.82% | 2.405 | 3.94% | 0.40 |
| Mg2+ | 1.11 | 2.78% | 36.87 | 60.44% | 21.74 |
| Na+ + K+ | 35.68 | 89.39% | 17.62 | 28.88% | 0.64 |
| TDS | 39.91 | 61 |
In Table 1 relating to sample 1, the% of the ion or group of ions content in the municipality of Abomey-Calavi is calculated by making the ratio of the average of the ion or group of ions content in the municipality by the average of the groundwater TDS of the municipality. Similarly, the% of the ion or group of ions content in the groundwater sample 1 is calculated by making the ratio of the content of ion or of group of ions in the groundwater sample 1 by the TDS of sample 1. The Relative Content Index for the sample 1 is calculated by dividing the% of an ion or group of ions content in the groundwater sample 1 by the% of that ion or group of ions content in the municipality.
Similarly, the Relative Contents Index of the other 4 groundwater samples from the Godomey district was determined (Table 2).
-
-
Realization of Radars plots of Relative Contents Index of groundwater samples analyzed in the district of Godomey
Table 2.
The values of the Relative Content Index of majors ions in the water of the samples 2, 3, 4 and 5 in the district of Godomey.
| Parameters | Relative Content Index for the sample 2 | Relative Content Index for the sample 3 | Relative Content Index for the sample 4 | Relative Content Index for the sample 5 |
|---|---|---|---|---|
| Cl− | 10.64 | 0.38 | 3.72 | 0.90 |
| SO42− | 8.08 | 0.23 | 3.22 | 7.11 |
| HCO3−+ CO32− | 1.23 | 0.38 | 0.32 | 0.47 |
| Ca2+ | 0.18 | 0.55 | 0.10 | 2.14 |
| Mg2+ | 5.35 | 1.1 9 | 2.49 | 1.38 |
| Na+ + K+ | 9.307 | 0.27 | 3.01 | 1.32 |
From the values of the Relative Contents Index of the 5 groundwater samples from the Godomey district, we constructed the Radars plots of Relative Contents Index for these samples (Fig. 9, Fig. 10, Fig. 11, Fig. 12, Fig. 13).
Fig. 9.
Radar plot of Relative Content Index for the sample 1.
Fig. 10.
Radar plot of Relative Content Index for the sample 2.
Fig. 11.
Radar plot of Relative Content Index for the sample 3.
Fig. 12.
Radar plot of Relative Content Index for the sample 4.
Fig. 13.
Radar plot of Relative Content Index for the sample 5.
The radar plot of Relative Content Index of the 5 water samples from Godomey district can be divided into 2 groups:
For each of the water samples 1, 3 and 5, the Relative Content Index of the Cl− ion is less than 1 and does not have the highest value of the Relatives Contents Index of each of these water samples. There is no saltwater intrusion into these groundwaters.
For each of water samples 2 and 4, the Relative Content Index of the Cl− ion is much greater than 1 and is greater than the Relatives Contents Index of the other ions for each of these 2 water samples.
-
-
Realization of the 1st Parfait-Hounsinou diagrams of the analyzed water samples in the district of GodomeyFig. 14, Fig. 15, Fig. 16, Fig. 17, Fig. 18
Fig. 14.
1st Parfait-Hounsinou diagram where area of pie slices represents ions contents in the groundwater for the sapmle 1 in the district of Godomey groundwater and radius length represents the Relative Content Index.
Fig. 15.
1st Parfait-Hounsinou diagram where area of pie slices represents ions contents in the groundwater for the sample 2 in the district of Godomey groundwater and radius length represents the Relative Content Index.
Fig. 16.
1st Parfait-Hounsinou diagram where area of pie slices represents ions contents in the groundwater for the sample 3 in the district of Godomey groundwater and radius length represents the Relative Content Index.
Fig. 17.
1st Parfait-Hounsinou diagram where area of pie slices represents ions contents in the groundwater for the sample 4 in the district of Godomey groundwater and radius length represents the Relative Content Index.
Fig. 18.
1st Parfait-Hounsinou diagram where area of pie slices represents ions contents in the groundwater for the sample 5 in the district of Godomey groundwater and radius length represents the Relative Content Index.
The 1st Parfait-Hounsinou diagrams of the 5 water samples from the Godomey district can be divided into 2 groups:
For each of the water samples 1, 3 and 5, the surfaces of pie slices do not respect the inequalities (Na+ + K+)› Mg2+ › Ca2+ and do not respect the inequalities Cl− › SO42− › (HCO3− + CO32−). So there is no seawater intrusion in these groundwaters.
For the 1st Parfait-Hounsinou diagrams of water samples 2 and 4, starting from the highest point of the graph, the areas of pie slices vary in a decreasing fashion for anions in the clockwise direction and for cations in the trigonometric sense. There is therefore saltwater intrusion in groundwater from wells 2 and 4. These two wells are located in the district of Godomey, more precisely in the coastal village of Togbin (Fig. 19). We recall that, for each of the samples of water 2 and 4, the radius length represents the Relative Content Index of the Cl− ion is along. This confirms the seawater intrusion in the groundwater of the village of Togbin.
Fig. 19.
Extent of seawater intrusion in groundwater in the municipality of Abomey-Calavi.
The intrusion of seawater in the village of Togbin is explained by the vulnerability of groundwater in this village. Indeed, we had conducted a previous study using the DRASTIC method which showed that the village of Togbin is the place in Abomey-Calavi where the groundwater in the aquifer of the Continental terminal is the most vulnerable [9].
Comparison of the new method to methods usually used to study saltwater intrusion in groundwater such as Schoeller-Berkaloff or Stiff diagrams and Revelle index
To verify the hypothesis of saltwater intrusion in groundwater, in the methods usually used it is important to construct the Schoeller-Berkalof diagram and/or the Stiff diagram. If the geometric shape(s) of the Schoeller-Berkaloff and Stiff diagrams of these groundwaters show that (Na+ + K+)› Mg2+ › Ca2+ and Cl− › SO42− › (HCO3− + CO32−), this confirms that there is saltwater intrusion into these groundwaters.
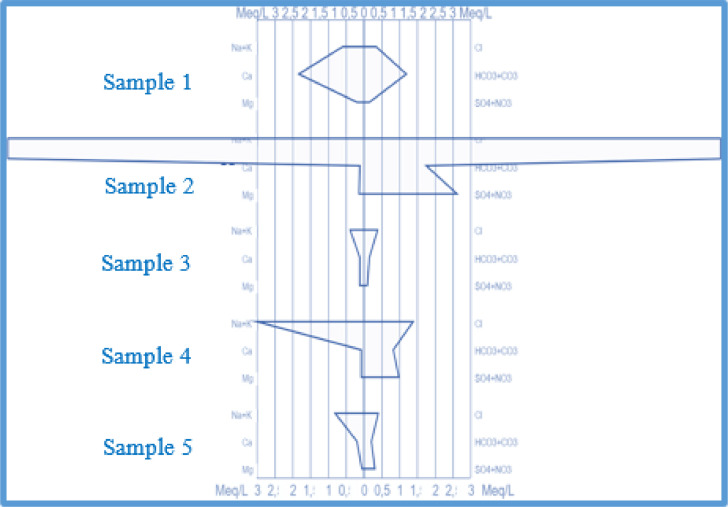
In the municipality of Abomey-Calavi, looking for a probable saltwater intrusion in groundwater, we constructed the Stiff diagrams of groundwater in the coastal district of Godomey (Fig. 20). The geometry of samples 2 and 4 located in the coastal village of Togbin shows that there is a saltwater intrusion in the groundwater of Togbin.
Fig. 20.
Stiff diagrams of chemical analysis of groundwater samples from monitoring wells in Godomey.
To verify this intrusion, it is necessary to calculate the Revelle index of the water samples analyzed.
Revelle index R = rCl− / (rHCO3– + rCO32–)
If for water samples, the Revelle index is greater than 6.6 then the saltwater intrusion is verified for these groundwaters.
To verify the saltwater intrusion in groundwater, we calculated the Revelle indices of groundwater in the municipality of Abomey-Calavi (Table 3). We noticed that only the values of the Revelle Indexes of the groundwater of the village of Togbin in the district of Godomey are higher than 6.6. Saltwater intrusion into Togbin groundwater is therefore verified.
Table 3.
Values of the Revelle index of the groundwater samples studied in the municipality of Abomey-Calavi.
| Districts names | Revelle Index (RI) |
|---|---|
| ABOMEY-CALAVI | 2.25 |
| ABOMEY-CALAVI | 2 |
| ABOMEY-CALAVI | 2.7 |
| ABOMEY-CALAVI | 3 |
| ABOMEY-CALAVI | 2.4 |
| AKASSATO | 3.5 |
| AKASSATO | 1.7 |
| AKASSATO | 0.8 |
| AKASSATO | 3.7 |
| AKASSATO | 3.5 |
| AKASSATO | 2.5 |
| AKASSATO | 1.1 |
| AKASSATO | 3 |
| AKASSATO | 0.7 |
| AKASSATO | 3.5 |
| AKASSATO | 2.5 |
| AKASSATO | 1.2 |
| AKASSATO | 2.5 |
| AKASSATO | 1.5 |
| AKASSATO | 2.2 |
| AKASSATO | 2.7 |
| AKASSATO | 2.85 |
| AKASSATO | 3 |
| AKASSATO | 1.75 |
| AKASSATO | 1.5 |
| AKASSATO | 3 |
| AKASSATO | 1.6 |
| AKASSATO | 1.7 |
| AKASSATO | 0.6 |
| AKASSATO | 1.1 |
| AKASSATO | 2.3 |
| GLO-DJIGBE | 1.7 |
| GLO-DJIGBE | 3.5 |
| GLO-DJIGBE | 1 |
| GLO-DJIGBE | 0.4 |
| GLO-DJIGBE | 1.5 |
| GLO-DJIGBE | 0.8 |
| GODOMEY | 0.3 |
| GODOMEY | 19.5 |
| GODOMEY | 2.5 |
| GODOMEY | 7 |
| GODOMEY | 2 |
| HEVIE | 5.5 |
| HEVIE | 4.3 |
| KPANROUN | 0.01 |
| OUEDO | 0 |
| OUEDO | 0.01 |
| OUEDO | 0.01 |
| OUEDO | 0 |
| OUEDO | 0.7 |
| OUEDO | 0.01 |
| OUEDO | 0 |
| TOGBA | 0.02 |
| TOGBA | 0 |
| TOGBA | 0.03 |
| TOGBA | 0 |
| ZINVIE | 0.01 |
| ZINVIE | 0 |
| ZINVIE | 0 |
| ZINVIE | 0 |
| ZINVIE | 0 |
| ZINVIE | 0 |
| ZINVIE | 0 |
| ZINVIE | 0.01 |
| ZINVIE | 0 |
| ZINVIE | 0 |
| ZINVIE | 0 |
| ZINVIE | 0.01 |
We note that the methods usually used confirm the 1st method Parfait-Hounsinou but the 1st method Parfait-Hounsinou is more efficient.
Compared to Scholler-Berkaloff and Stiff diagrams, in the 1st method Parfait-Hounsinou, at the spie chart level, the area of pie slices make it easier to compare the major cations with each other and the major anions with each other. Moreover, at the level of the spie chart of the 1st method Parfait-Hounsinou of a water sample, the comparison of the Relative Content Index of the Cl− ion with the Relative Contents Index of the other major ions of the same water sample allows further confirmation of the saltwater intrusion and to appreciate the extent of the saltwater intrusion.
Conclusion
The 1st method Parfait-Hounsinou developed during this study is easy, useful and efficacy. This method allowed us to prove in 2020 then in 2022 the saltwater intrusion in the groundwater of the municipality of Abomey-Calavi in Benin. The novelty of this method is that the parameters and analysis methods most used for water are used, but at each stage of the method, a very rigorous and mathematical interpretation is made to prove the saltwater intrusion in the groundwater.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Footnotes
Related research article:Sagnon Parfait Hounsinou, Use of graphical and multivariate statistical methods to show a marine intrusion and salinization of a coastal water table: Case study of the township of Abomey-Calavi, Benin, Heliyon, volume 8, issue 11, 2022, e11588, ISSN 205–8440. https://doi.org/10.1016/j.heliyon.2022.e11588
Data availability
The data are in the article.
References
- 1.Mazloomi S., Zarei A., Nourmoradi H., Ghodsei S., Amraei P., Haghighat G.A. Optimization of coagulation-flocculation process for turbidity removal using response surface methodology: a study in Ilam water treatment plant, Iran. Desalin. Water Treat. 2019;147:234–242. [Google Scholar]
- 2.Mirzabeygi M., Abbasnia A., Yunesian M., Nodehi R.N., Yousefi N., Hadi M., Mahvi A.H. Heavy metal contamination and health risk assessment in drinking water of Sistan and Baluchistan, Southeastern Iran. Hum. Ecol. Risk Assess.: An Int. J. 2017;23:1893–1905. [Google Scholar]
- 3.Hounsinou Sagnon Parfait. Use of graphical and multivariate statistical methods to show a marine intrusion and salinization of a coastal water table: case study of the township of Abomey-Calavi, Benin. Heliyon. 2022;8(11):e11588. doi: 10.1016/j.heliyon.2022.e11588. ISSN 205-8440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hounsinou Sagnon Parfait, Assessment of potential seawater intrusion in a coastal aquifer system at Abomey - Calavi, Benin, Heliyon 6 (2020) e03173. 10.1016/j.heliyon.2020.e03173 [DOI] [PMC free article] [PubMed]
- 5.Putra D.B.E., Hadian M.S.D., Alam B.Y.C., Yuskar Y., Yaacob W.Z.W., Datta B., Harnum W.P.D. Geochemistry of groundwater and saltwater intrusion in a coastal region of an island in Malacca Strait, Indonesia. Environ. Eng. Res. 2021;26 [Google Scholar]
- 6.Bhagat C., Puri M., Mohapatra P.K., Kumar M. Imprints of seawater intrusion on groundwater quality and evolution in the coastal districts of south Gujarat, India. Case Stud. Chem. Environ. Eng. 2021;3 [Google Scholar]
- 7.Busico G., Buffardi C., Ntona M.M., Vigliotti M., Colombani N., Mastrocicco M., Ruberti D. Actual and forecasted vulnerability assessment to seawater intrusion via GALDIT-SUSI in the Volturno river mouth (Italy) Remote Sens. 2021;13(18):3632. [Google Scholar]
- 8.Sagnon Parfait Hounsinou The Hounsinou scale: its development and use to determine the overall quality of groundwater used for drinking and bathing in the municipality of Abomey-Calavi in Benin. J.Hydrol. Regional Stud. 2021:100777. doi: 10.1016/j.ejrh.2021.100777. [DOI] [Google Scholar]
- 9.Parfait H.S., Daouda M. Using the DRASTIC method to study the vulnerability of groundwater in the aquifer of the Continental terminal of the town of Abomey-Calavi in Benin. Pyrex J. Ecol. Natural Environ. 2015;1(2) http://www.pyrexjournals.org/pjene 007-01 2 July. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data are in the article.