Abstract
Background:
During the 2010 Deepwater Horizon (DWH) disaster, oil spill response and cleanup (OSRC) workers were exposed to toxic volatile components of crude oil. Few studies have examined exposure to individual volatile hydrocarbon chemicals below occupational exposure limits in relation to neurologic function among OSRC workers.
Objectives:
To investigate the association of several spill-related chemicals (benzene, toluene, ethylbenzene, xylene, n-hexane, i.e., BTEX-H) and total petroleum hydrocarbons (THC) with neurologic function among DWH spill workers enrolled in the Gulf Long-term Follow-up Study.
Methods:
Cumulative exposure to THC and BTEX-H across the oil spill cleanup period were estimated using a job-exposure matrix that linked air measurement data to detailed self-reported DWH OSRC work histories. We ascertained quantitative neurologic function data via a comprehensive test battery at a clinical examination that occurred 4–6 years after the DWH disaster. We used multivariable linear regression and modified Poisson regression to evaluate relationships of exposures (quartiles (Q)) with 4 neurologic function measures. We examined modification of the associations by age at enrollment (<50 vs. ≥50 years).
Results:
We did not find evidence of adverse neurologic effects from crude oil exposures among the overall study population. However, among workers ≥50 years of age, several individual chemical exposures were associated with poorer vibrotactile acuity of the great toe, with statistically significant effects observed in Q3 or Q4 of exposures (range of log mean difference in Q4 across exposures: 0.13–0.26 micron). We also observed suggestive adverse associations among those ≥ age 50 years for tests of postural stability and single-leg stance, although most effect estimates did not reach thresholds of statistical significance (p<0.05).
Conclusions:
Higher exposures to volatile components of crude oil were associated with modest deficits in neurologic function among OSRC workers who were age 50 years or older at study enrollment.
Keywords: crude oil, hydrocarbons, volatile organic compounds, neurologic function, peripheral nervous system, oil spill
Introduction
The Deepwater Horizon (DWH) disaster is the largest marine oil spill in U.S. history (National Commission on the BP Deepwater Horizon Oil Spill and Offshore Drilling 2011). The explosion of the drilling rig on April 20, 2010 resulted in the release of an estimated 4.9 million barrels of crude oil into the Gulf of Mexico (National Commission on the BP Deepwater Horizon Oil Spill and Offshore Drilling 2011). To stop the spill and remove the oil from the environment, tens of thousands of workers and volunteers participated in an oil spill response and cleanup (OSRC) operation, which was largely completed by June 2011 (U.S. Coast Guard 2011). While working on response and cleanup, workers were exposed to a range of chemical hazards, including crude oil hydrocarbons and particulate matter from combustion of oil/gas and diesel-powered vessel engines (Kwok et al. 2017).
Several volatile crude oil chemicals, including benzene, toluene, ethylbenzene, xylene, and n-hexane (BTEX-H), are known neurotoxicants (ATSDR 2004). In addition to being components of the crude oil, they are also present in gasolines and used as solvents and industrial raw materials (ATSDR 1999, 2000, 2007a, b, 2010). In animal models and human studies, acute inhalation of high levels of these chemicals has been associated with impaired neurologic function, as demonstrated by overt symptoms of neurotoxicity, changes in electrophysiological measures of peripheral nerve performance, and reduced neurobehavioral test performance (ATSDR 1999, 2000, 2007a, b, 2010). Results of these and other studies informed the development of health-based occupational standards (e.g., permissible exposure limits (OSHA 2022)) and guidelines (threshold limit values) (ACGIH 2012) for short-term exposure to BTEX-H.
Neurological effects of longer-term, lower-level exposure to volatile hydrocarbons have been investigated in occupational studies. Most studies assessed exposure by comparing workers chronically exposed to organic solvents or petroleum fuels with a group with lower or no exposure (Gong et al. 2003; Jovanović et al. 2004; Mutti et al. 1982b; Shih et al. 2011; Stĕtkárová et al. 1993). In these studies, exposure to volatile hydrocarbon mixtures predominated by one or more BTEX-H chemicals at levels below the occupational limit were associated with reduced cutaneous sensitivity to vibration and reduced motor/sensory conduction velocities (Sanagi et al. 1980; Shih et al. 2011), postural instability (Kuo et al. 1996; Sanagi et al. 1980), deficits in visual contrast sensitivity (Gong et al. 2003), and color vision impairment (Cavalleri et al. 2000), with two studies showing effects that persisted many months after exposure (Cianchetti et al. 1976; Mutti et al. 1982b). It is unclear, however, which agent among these mixture components contribute to the neurologic effects. Quantitative exposure estimates to individual BTEX-H chemicals ascertained from air samples or from urinary biomarkers were evaluated in relation to neurologic function in a few studies, with inconsistent results (Bates et al. 2019; Bleecker et al. 1991; Gong et al. 2003; Jovanović et al. 2004; Lee et al. 2007; Mutti et al. 1982b; Neghab et al. 2012; Smith et al. 1997). In addition, most of these studies were small (n<100) or had extensive exclusion criteria, which limited the generalizability of the findings.
The neurologic effects of OSRC work have been explored in previous studies of oil spill workers. Higher prevalence of acute (Ha et al. 2012) and persistent neurological symptoms (Na et al. 2012) was associated with longer durations of exposure among workers responding to the Hebei Spirit oil spill. Following the DWH disaster, self-reported exposures to crude oil were associated with acute neurologic symptoms (Krishnamurthy et al. 2019) and longer-term risk of neurological outcomes among Coast Guard responders who participated in the cleanup (Denic-Roberts et al. 2023). Modestly worse performance in some neurobehavioral tests was observed among DWH OSRC workers with higher single daily maximum exposure to total petroleum hydrocarbons (THC), a composite measure of the volatile components of the crude oil, several years after the spill (Quist et al. 2019). These studies provide some evidence that medium-term or chronic exposure to lower levels of volatile crude oil chemicals may induce adverse, possibly persistent, neurologic effects. The objective of the present study was to evaluate associations of quantitative estimates of crude oil-related volatile hydrocarbon exposures (THC and BTEX-H) with tests of neurologic function among DWH oil spill workers 4–6 years after the spill.
Methods
Study population
The Gulf Long-Term Follow-up Study (GuLF Study) is a prospective cohort study of the potential health effects of the DWH disaster. Study participants included individuals ≥21 years of age who either had participated in OSRC work for at least one day (workers) or had completed safety training but were not hired (non-workers) (Kwok et al. 2017). Enrollment occurred from March 2011 to May 2013 via computer-assisted telephone interviews that collected information on socio-demographics, lifestyle, health, and a detailed work history of DWH OSRC activities (Kwok et al. 2017). A total of 32,608 participants were enrolled in the study. Between August 2014 and July 2016, cohort members who resided within 60 miles of study clinics in New Orleans, LA or Mobile, AL were invited to a follow-up clinical examination during which their neurologic function was evaluated, as described below (Kwok et al. 2017).
Among the 3,401 participants who completed the clinical examination, we restricted our analysis to the 2,873 workers. Oil spill chemical exposures were not assessed for non-workers, as they did not have an opportunity to be occupationally exposed to the crude oil chemicals. We included in our sample the workers who completed at least one test of neurologic function (N=2,868). We excluded workers who reported a peripheral neuropathy (N=32) or diabetes diagnosis (n=142) before the DWH disaster. Finally, we excluded 84 workers with missing covariates needed for analysis and reached a final analytical sample of 2,610 participants. All participants provided written informed consent. The study was approved by the Institutional Review Board of the National Institute of Environmental Health Sciences.
Exposure assessment
Cumulative exposure to THC and five volatile hydrocarbon chemicals (BTEX-H) across each participant’s work period were estimated via a job-exposure matrix that linked air measurement data to OSRC work histories. The primary source of measurement data came from approximately 28,000 full-shift personal air samples collected on a subset of DWH OSRC workers, including some who enrolled in the GuLF Study, during their work shifts from April 2010 to June 2011 using organic vapor passive dosimeters (Stewart et al. 2022). Laboratory assays of these samples resulted in >143,000 measurements of THC and BTEX-H (Stenzel et al. 2022a; Stewart et al. 2022). In addition to these personal measurements, direct-reading volatile organic compound (VOC) area measurements were collected on 38 large vessels. Study industrial hygienists converted these VOC measurements to equivalent full-shift THC and BTEX-H estimates to supplement measurements on days where there were few or no personal measurements on these vessels (Groth et al. 2022a; Ramachandran et al. 2022).
To estimate exposures for each study participant, the industrial hygienists created over 3,000 exposure groups (EGs) based on three exposure determinants: job/activity (e.g., driller, burning oil, skimming), location of work (hot zone, source, offshore, nearshore, and land, separately by Gulf state), and time period (periods of time during the cleanup reflecting changes in oil weathering and OSRC events) (Stenzel et al. 2022b). Each EG was a unique combination of these three determinants and represented workers who were expected to have similar distributions of exposures. The average of the air measurements corresponding to each EG was estimated for the EG (Groth et al. 2017; Groth et al. 2022a; Groth et al. 2022b; Huynh et al. 2022a, b; Huynh et al. 2022c; Ramachandran et al. 2022). Because some EGs contained a large number of measurements below the analytical method’s limit of detection, a left-censored Bayesian framework was used to estimate THC/BTEX-H exposure averages and other exposure statistics for the EGs (Groth et al. 2017; Groth et al. 2022b). More details on the statistical methods used in the exposure assessment have been described in the papers cited above.
Based on their reported DWH OSRC work history, study participants were matched to the appropriate EGs for assigning exposure estimates. Because many participants reported multiple work activities across the cleanup, some of which occurred on the same day, daily average exposure estimates were developed for each day worked by taking the average of the exposure estimates across all jobs/activities performed on a day. To examine the total burden of exposure experienced by each participant during the cleanup, cumulative exposure across all workdays was calculated by summing up the daily average exposure estimates across all of each participant’s workdays within a time period and then across all time periods.
Neurologic Function Testing
The neurologic function testing battery included tests of vibrotactile threshold, visual acuity, visual contrast sensitivity, postural sway, and ability to maintain single leg stance. These tests were administered at the clinical examination by trained clinical examiners who were blinded to each participant’s exposure levels.
The Vibratron II electromechanical vibrometer (Physitemp, Inc.) was used to measure bilateral great toe sensory acuity at a frequency of 120Hz using a standard protocol (Gerr et al. 1990). Examiners manually controlled the delivered vibration amplitude. A total of five vibration threshold values (three descending and two ascending values) were obtained for each great toe. After discarding the first value, the final vibrotactile threshold for each toe was the median of the remaining four values. We report vibrotactile threshold in log microns of peak-to-peak amplitude and examined the single bilateral mean (i.e., average of the two toes) in analyses. Higher values of vibrotactile threshold indicate worse performance.
Visual acuity was assessed binocularly using the Optec 1000 (Optec, Inc.) to determine eligibility for the statistical analysis of visual contrast sensitivity (Campagna et al. 1995). Visual contrast sensitivity was evaluated with the Optec 1000 Functional Assessment of Contrast Sensitivity test. In the test, circular stimuli consisting of alternating light and dark bars were presented to the participants. Nine stimuli of decreasing contrast were presented at each of 5 spatial frequencies (1.5, 3, 6, 12, and 18 cycles per degree). At each spatial frequency, the weakest contrast score correctly identified was selected and examined in analyses. Higher scores of contrast sensitivity indicate better performance.
Postural stability was evaluated with an Advanced Mechanical Technology, Inc. force platform (AMTI) (Letz et al. 1994). Participants were instructed to stand upright on the platform, and four trials were administered, two with eyes open and two with eyes closed. The mean sway speed (millimeters per second) was calculated from force signals recorded by the platform during the trials. We examined the average sway speeds across the eyes-open trials and, separately, the average value across the eyes-closed trials in analyses. Higher postural sway speed indicates less stability.
Trained staff evaluated participants’ ability to stand on one leg and maintain upright balance for 30 seconds (s) (Tyson and Connell 2009). If the participant was unable to maintain balance for the entire 30-s interval, then the procedure was repeated up to two additional times. We modeled the outcome as inability to maintain single-leg stance in any attempt (yes; no).
Statistical modeling
We used multivariable linear regression to estimate the mean differences and 95% confidence intervals (95% CI) in vibrotactile threshold, visual contrast sensitivity, and postural sway associated with increasing quartiles (Q) of exposures among workers. We checked assumptions of linear regression via diagnostic plots of the residuals. Assumptions were met for all continuous outcomes except for postural sway, for which we removed six outliers and ln-transformed mean sway speeds (both eyes open and eyes closed) to improve normality and achieve homoscedasticity of the residuals. For test of maintaining single leg stance, we used multivariable modified Poisson regression to estimate prevalence ratios (PR) and 95% CI with each quartile exposure of interest (Zou 2004). In addition to assessing quartile exposures, we also examined ln-transformed continuous exposure levels in all tests.
We selected covariates to be adjusted for based on a directed acyclic graph (DAG) and included the minimally sufficient adjustment set and predictors of the outcome that are not descendants of the exposure in the model (Greenland et al. 1999) (Figure S1). In the main analysis, we adjusted for continuous age, sex (male; female), self-reported race (White; Black; other/multiracial), cigarette smoking (current; former; never), highest educational attainment (less than high school; high school diploma or general equivalency diploma; some college or 2-year degree; 4-year college graduate or more), current binge drinking (consumption of ≥5 drinks on one occasion ≥12 times in the year before the clinical examination), previous oil industry experience (yes; no), and residential proximity to the spill (living in a coastal county directly affected by the spill; living in a Gulf or non-Gulf state further from the spill) in all models. For analyses, we aggregated participants who self-identified as races other than White or Black and those who identified as multiple races into the “other/multiracial” group because of the small numbers of workers in these groups (see Supplemental Text S1 for all race selection options). We used self-reported race as a proxy for the downstream effects of socioeconomic disparities as well as differential life experiences due to structural racism (e.g., health care access and utilization, neighborhood ambient air pollution exposure) that might have influenced workers’ health and neurologic function (Lane et al. 2022; Saadi et al. 2017; Xu et al. 2016). All covariates, with the exception of binge drinking, which was self-reported during the clinical examination, were ascertained in the enrollment interview. For analysis of vibrotactile threshold, we additionally adjusted for height measured in the clinical examination. For visual contrast sensitivity, we restricted analyses to participants with better than 20/50 visual acuity and additionally adjusted for use of vision correction (i.e., wearing glasses or corrective lenses). For tests of postural stability and ability to maintain single leg stance, we conducted analyses with and without additional adjustment for ever performing OSRC work on water vessels to try to account for baseline vestibular function.
In a sub-analysis, we investigated potential effect measure modification (EMM) of the associations between crude oil exposures and neurologic function by age at enrollment (<50 vs. ≥50 years). We evaluated EMM of the associations by assessing stratified models to determine effects in the subgroups and by performing likelihood ratio tests across strata and reporting the p-values. In another sub-analysis, we examined associations restricted to men; there were too few women in the study population to conduct analysis in this subgroup. In a separate sensitivity analysis, we additionally adjusted in the model for body mass index (BMI) (underweight or normal; overweight; obese) calculated from height and weight measured at the clinical examination, as obesity has been associated with neurologic function with mechanisms beyond insulin dysregulation (Wang et al. 2016). We did not account for BMI in the main analysis because crude oil chemical exposures have been associated with diabetes in this cohort (Jardel et al. 2022) and with obesity in other studies (Lee et al. 2022); thus, BMI ascertained after exposure might act as a mediator in the association of crude oil exposure and neurological outcomes. To address potential confounding from exposure to higher levels of fine particulate matter (PM2.5) from controlled burning activities (Chen et al. 2022; Pratt et al. 2022), which has been modestly associated with neurological test performance in the same cohort (Norris et al., in press), we repeated analyses excluding 210 workers with higher PM2.5 exposures from burning-related activities. Because age-related declines in neurologic function have been well-documented (Kalisch et al. 2008; Seidler et al. 2010), with studies showing a loss of mechanoreceptors on the skin, declines in nerve conduction velocity (Kalisch et al. 2008; Seidler et al. 2010), and changes in the structure and biochemistry of the brain (Vassar and Rose 2014) with increasing age, we also assessed whether our estimates were sensitive to how age was specified in the model by investigating restrictive cubic splines as an alternative functional form of age. In addition to examining cumulative exposures, we also examined average exposures, calculated as the ratios of cumulative exposures and exposure durations, as measures of exposure intensity.
All analyses were performed using SAS, version 9.4 (SAS Institute Inc., Cary, NC, USA). An alpha level of 0.05 was considered statistically significant for all analyses.
Results
Around 63% of the analytical sample were younger than 50 years at enrollment, and about three quarters were male (Table 1). Half of all participants self-reported race as White, while 41% of them self-reported as Black. The remaining participants (8%) self-identified as other races or belonging to multiple race categories. At enrollment, less than half of participants had at least some college education, and around one third were current smokers. About 11% of the participants were categorized as binge drinkers. Approximately 16% of the participants had prior experience working in the oil industry, and the vast majority of participants (93%) resided in a Gulf state coastal county/parish directly impacted by the spill at time of enrollment. The highest daily exposure to each BTEX-H chemical was well below the threshold limit values established by the American Conference of Governmental Industrial Hygienists (benzene: 500 ppb; toluene: 20,000 ppb; ethylbenzene: 20,000 ppb; xylene: 20,000 ppb; n-hexane: 50,000 ppb) or the permissible exposure limits established by the Occupational Safety and Health Administration (benzene: 1000 ppb; toluene: 200,000 ppb; ethylbenzene: 100,000 ppb; xylene: 100,000 ppb; n-hexane: 500,000 ppb). Three quarters of workers were exposed for ≤8 months (Table S1).
Table 1.
Characteristics of DWH disaster oil spill workers who completed at least one neurologic function test (N=2,610)
| Characteristic | n (%) |
|---|---|
| Age (y) | |
| <30 | 427 (16.4) |
| 30–39 | 542 (20.8) |
| 40–49 | 677 (25.9) |
| 50–59 | 650 (24.9) |
| ≥60 | 314 (12.0) |
| Sex | |
| Male | 2013 (77.1) |
| Female | 597 (22.9) |
| Race | |
| White | 1336 (51.2) |
| Black | 1061 (40.7) |
| Other/multiracial | 213 (8.2) |
| Education | |
| Less than high school | 555 (21.3) |
| High school diploma/GED | 913 (35.0) |
| Some college/2-year degree | 786 (30.1) |
| 4-year college graduate or more | 356 (13.6) |
| Smoking status | |
| Current smoker | 896 (34.3) |
| Former smoker | 548 (21.0) |
| Never smoker | 1166 (44.7) |
| Current binge drinkera | |
| No | 2332 (89.4) |
| Yes | 278 (10.7) |
| Previous oil industry experience | |
| No | 2181 (83.6) |
| Yes | 429 (16.4) |
| Residential county proximity to Gulf of Mexicob | |
| Direct contact | 2432 (93.2) |
| Indirect contact or other residence | 178 (6.8) |
| Vision correctionc | |
| Missing | 78 |
| No | 1602 (63.3) |
| Yes | 930 (36.7) |
| Poor visiond | |
| Missing | 77 |
| No | 2066 (81.6) |
| Yes | 467 (18.4) |
| Weight classification | |
| Missing | 1 |
| Underweight or normal (BMI <25) | 572 (21.9) |
| Overweight (25 ≤ BMI < 30) | 828 (31.7) |
| Obese (BMI ≥ 35) | 1209 (46.3) |
| Median (min, max) height (in) | 68.6 (55.4–78.3) |
Abbreviations: DWH, Deepwater Horizon; GED, General Equivalency Diploma
Binge drinking is defined as self-reporting ≥5 drinks on one occasion ≥12 times during the year before the clinical exam
Direct proximity is defined as living in a county directly adjacent to the Gulf of Mexico; indirect proximity or other residence is defined as living in a Gulf or non-Gulf state further from the spill.
Vision correction includes wearing corrective glasses and contact lenses.
Poor vision is defined as binocular visual acuity score of 20/50 or worse.
In tests of vibrotactile threshold, we observed modestly poorer vibrotactile acuity among workers in upper quartiles of exposure to benzene, toluene, ethylbenzene, and n-hexane compared to the respective referent groups (Figure 1, Table S2). However, the increases were not statistically significant, nor was there evidence of a monotonic exposure-response trend. No apparent associations were observed in analyses of continuous exposures.
Figure 1.
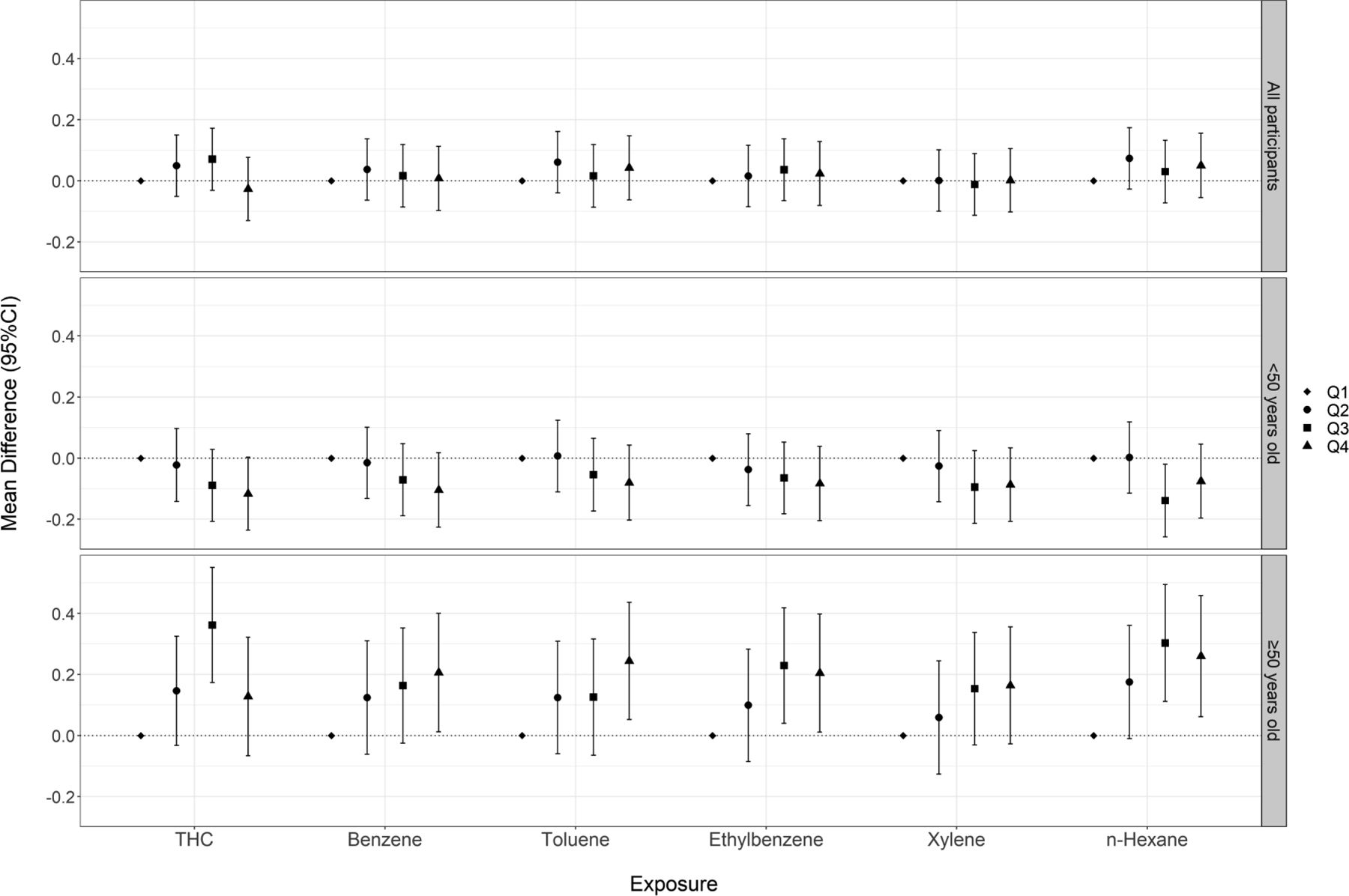
Cumulative exposure (quartiles) to crude oil chemicals and vibrotactile threshold (micron) among all participants and stratified by age at enrollment
We did not observe a positive association between the crude oil exposures and visual contrast sensitivity score (Figure 2, Table S3). At a spatial frequency of 1.5 cycles/degree, we observed unexpectedly better performance (i.e., higher contrast sensitivity scores) in contrast sensitivity across all upper quartiles of exposure to THC and BTEX-H compared to the first quartile, with some chemicals showing a monotonic trend. Effect estimates at spatial frequencies of 3, 6, and 12 cycles/degree were modestly above the null, but with no evidence of exposure-response trends. We observed non-monotonic and non-significant decreases in contrast sensitivity score at 18 cycles/degree.
Figure 2.
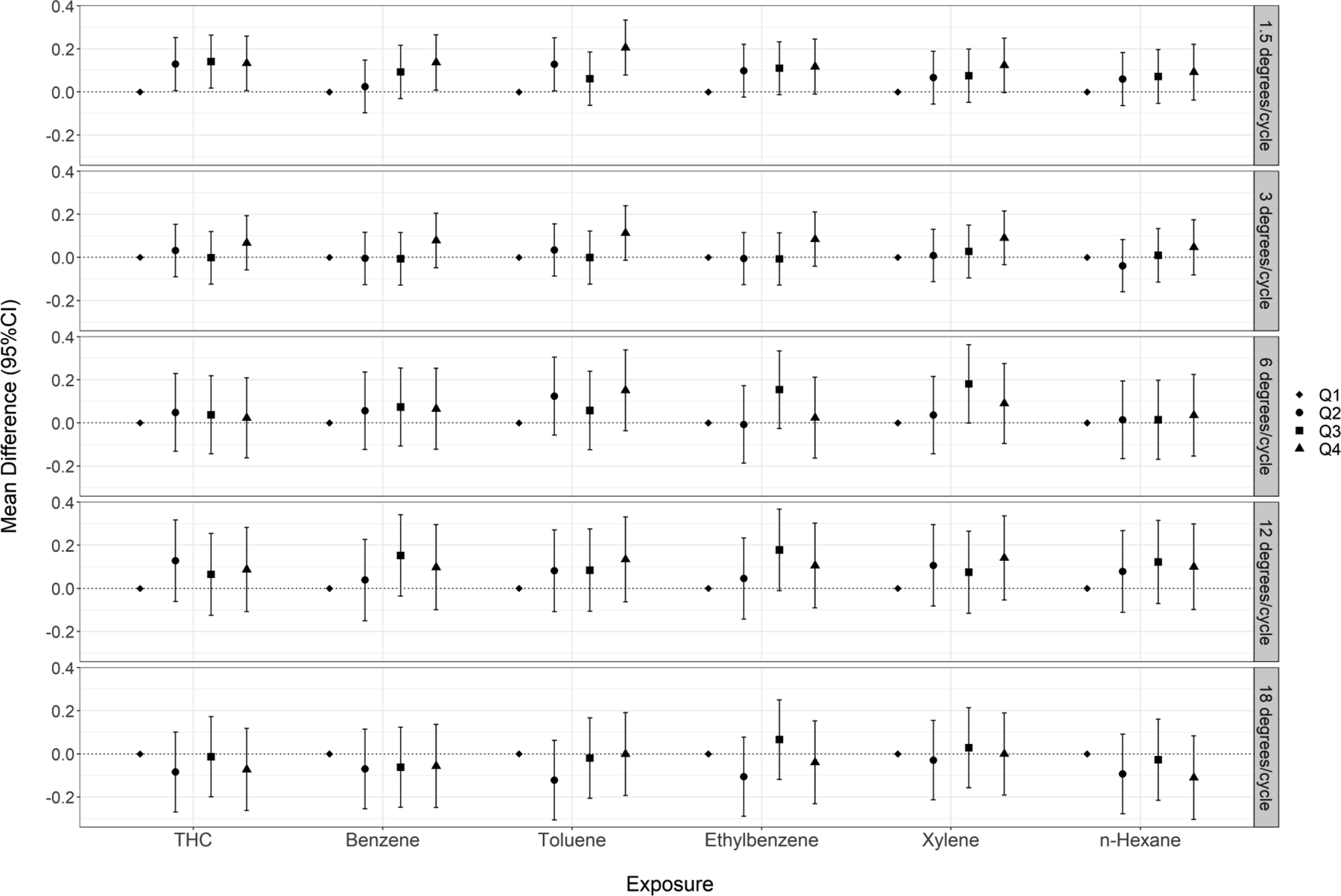
Cumulative exposure (quartiles) to crude oil chemicals and visual contrast sensitivity performance score among DWH disaster oil spill workers
There was no consistent relationship between crude oil chemical exposures and ln-transformed postural sway speed (Figure 3, Table S4). In analyses of continuous exposures, we observed a statistically significant positive association for xylene when participants performed the test eyes open. Patterns of association were similar when participants performed the test with their eyes closed (Figure 4, Table S5).
Figure 3.
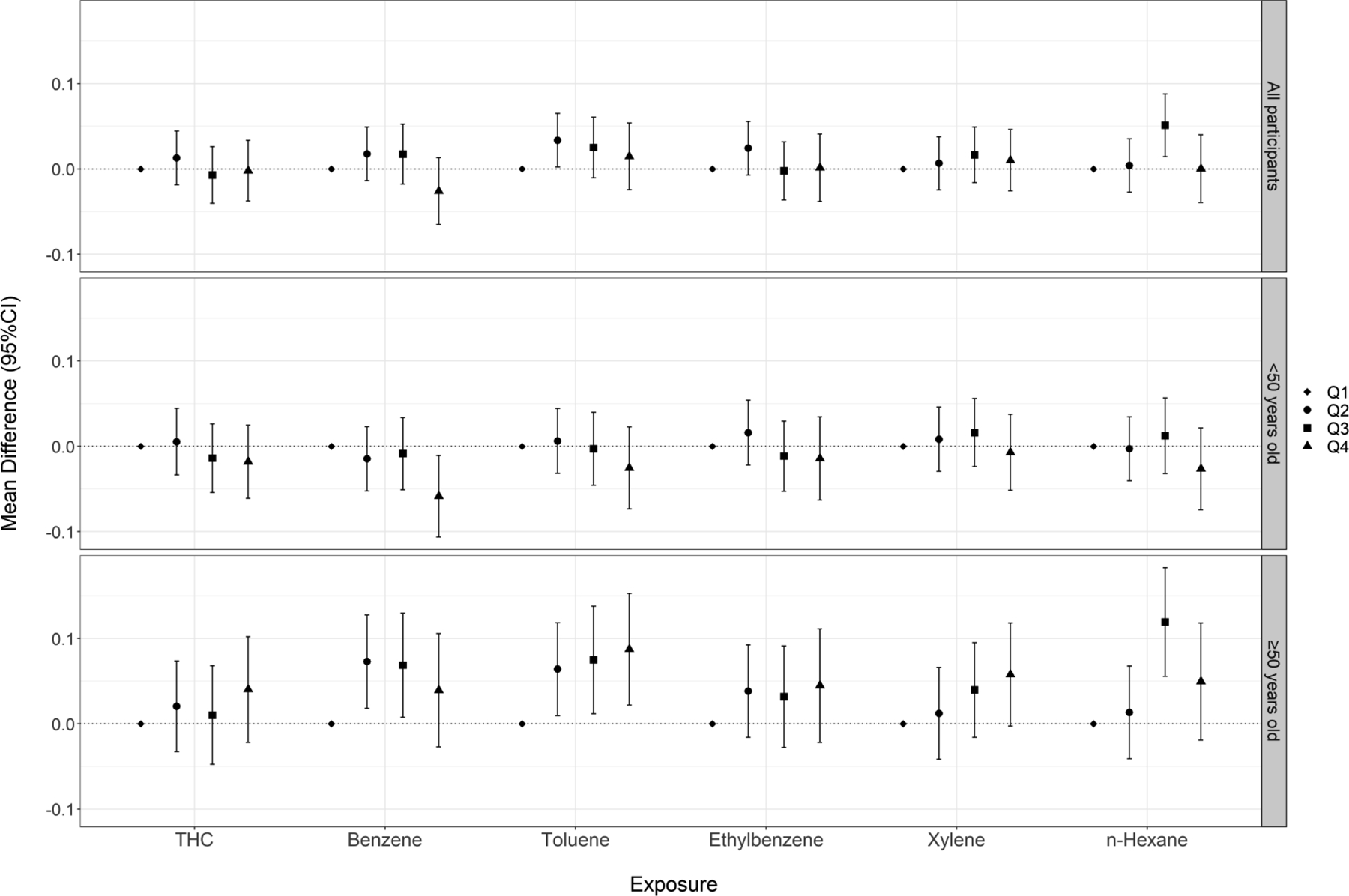
Cumulative exposure (quartiles) to crude oil chemicals and ln-transformed postural sway speed (mm/s) (eyes open) among all participants and stratified by age at enrollment
Figure 4.
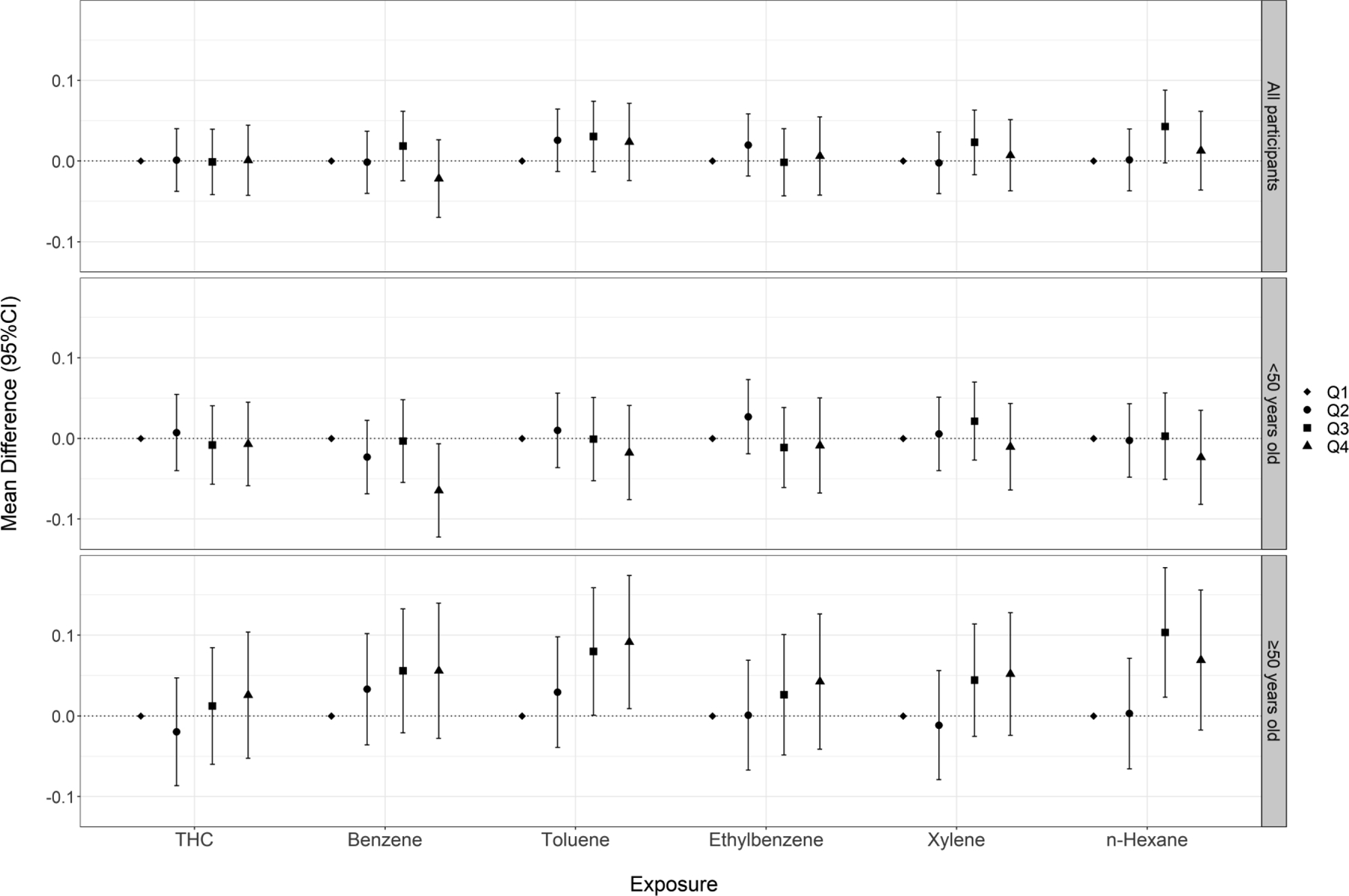
Cumulative exposure (quartiles) to crude oil chemicals and ln-transformed postural sway speed (mm/s) (eyes closed) among all participants and stratified by age at enrollment
We observed no associations between THC or benzene exposure and inability to maintain a single leg stance (Figure 5, Table S6). Modest but non-significant increases in PR were observed in Q4 for toluene, ethylbenzene, and xylene compared to the respective referent groups (range of PR: 1.13–1.18); however, there was no evidence of an exposure-response relationship across quartile exposures or in analyses of continuous exposures.
Figure 5.
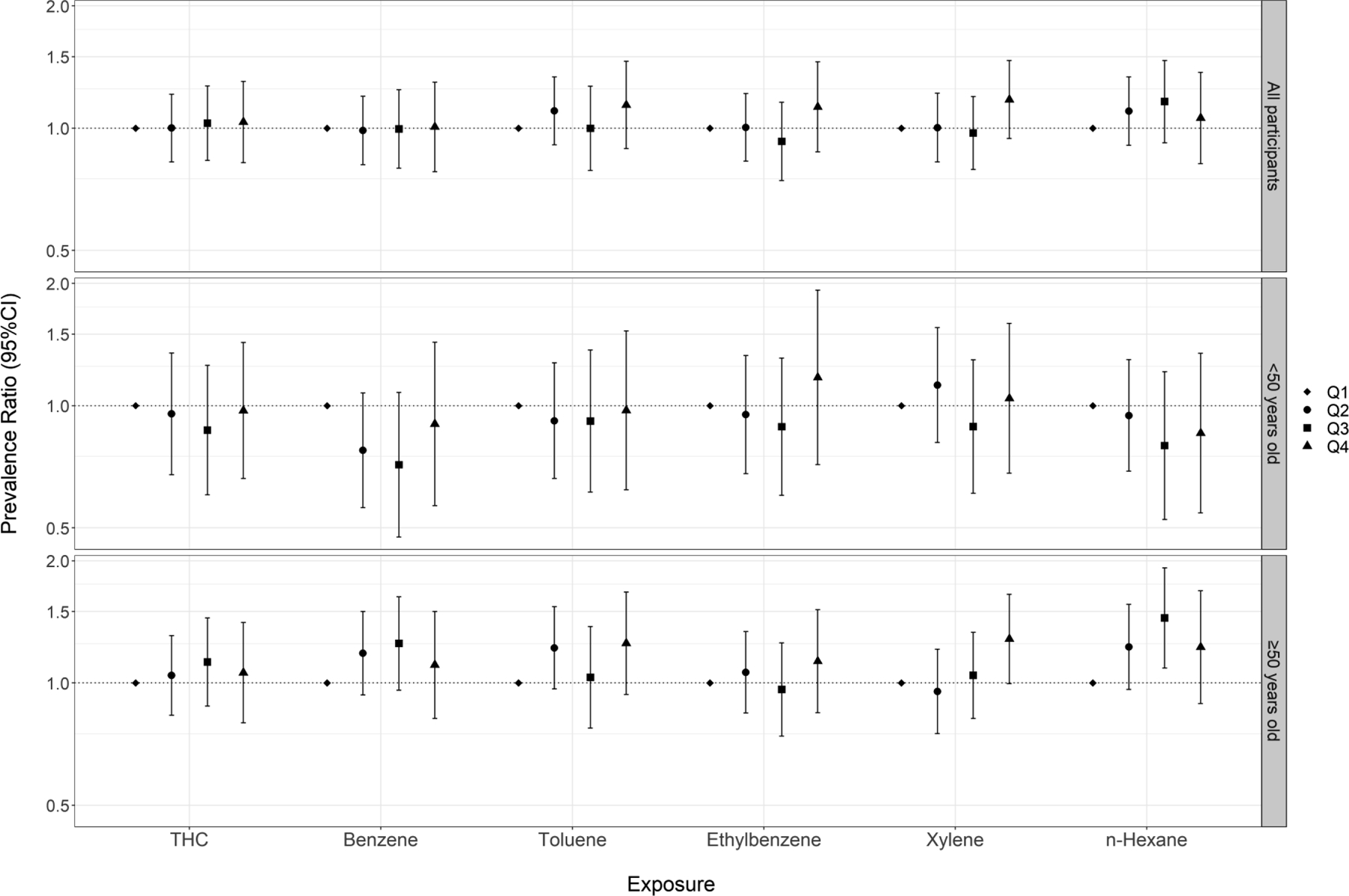
Cumulative exposure (quartiles) to crude oil chemicals and inability to maintain single leg stance among all participants and stratified by age at enrollment (note that y-axis is on the log scale)
When analyses were stratified by age at enrollment (<50 vs. ≥50 years), we observed heterogeneous associations for vibrotactile threshold (Figure 1, Table S2) and postural sway tests (Figures 3, Tables S4–5), with stronger associations observed among participants ≥50 years of age. When examining vibrotactile threshold within the older subgroup, we observed monotonic trends across quartile exposures to benzene, toluene, and n-hexane and statistically significant effect estimates in the top quartile of these chemicals. Within the younger subgroup, we observed non-significant associations in the inverse direction. When examining the eyes-open postural sway test, we saw elevated effect estimates across higher quartiles of exposure to THC and BTEX-H within the older subgroup, including monotonic trends for toluene and xylene. Within the younger subgroup, effect estimates were close to or below the null. Associations were also stronger within the older subgroup, particularly for benzene, toluene, and n-hexane when participants performed the test with their eyes closed. In the single leg stance test, we observed stronger associations for several chemicals (benzene, toluene, xylene, n-hexane) among the older subgroup compared to the younger group, although likelihood ratio tests did not reach statistical significance (Figure 4, Table S6). There was no evidence of heterogeneity in associations between crude oil exposures and contrast sensitivity by age (Figure S2, Table S7). We also examined associations among workers age 60 years or older; however, because of the relatively small number of these subjects, effect estimates were unstable and consequently difficult to interpret. Therefore, we have not presented these results.
There were no substantive differences in results when we restricted the analyses to men (Figures S3–7, Tables S8–13). We observed similar results in sensitivity analyses in which we additionally adjusted for BMI or modeled age as splines (results not shown) or restricted to workers with lower levels of burning-related PM2.5 exposure (Figures S3–7, Tables S8–13). Associations were somewhat reduced in analyses examining average exposures and in analyses not adjusted for ever working on water vessels (results not shown).
Discussion
In this study, we examined the relationship of exposure to THC and BTEX-H with several tests of neurological performance among oil spill workers 4–6 years after the DWH disaster. In the subgroup older than 50 years at enrollment, workers with higher exposures showed worse performance in the vibrotactile threshold test and modestly worse performance in postural stability and single-leg stance tests compared to those with lower exposures. The most consistent associations in this age group were observed for vibrotactile threshold, where effect estimates were above the null across all quartiles of exposure compared to the referent group, and several chemicals showed monotonic exposure-response trends. We did not find evidence of adverse neurological effects in the younger subgroup. Among the overall study population, we observed unexpectedly better performance in visual contrast sensitivity among workers with higher exposure, with several monotonic trends observed at a spatial frequency of 1.5 cycles/degree.
Numerous epidemiologic studies have shown adverse neurologic effects among populations exposed to high levels of volatile hydrocarbons (Grisold and Carozzi 2021; Valentine 2020). n-Hexane has been established as a cause of peripheral neuropathy among factory workers and persons with glue-sniffing addiction (Takeuchi 1993). Overt or subclinical changes in peripheral nerves, assessed via electromyography or vibrotactile threshold test, have also been detected among workers exposed to organic solvents containing one or more components of BTEX at concentrations above the occupational guidelines (Baslo and Aksoy 1982; Costa et al. 2012; Demers et al. 1991; Indhushree et al. 2016; Shih et al. 2011).
Only a few studies have examined quantitative measures of peripheral nerve function among populations exposed to lower levels of volatile hydrocarbons. In Bleecker et al. (1991), lifetime-weighted average occupational exposure to total hydrocarbons was associated with higher vibrotactile thresholds among paint manufacturing workers. Significant changes in nerve conduction measures were linked to longer duration of work and higher levels of urinary hippuric acid (a metabolite of toluene), but not with urinary methylhippuric acid (a metabolite of xylene), in a study of paint and lacquer factory workers exposed to sub-occupational limit levels of toluene and xylene (Jovanović et al. 2004). In a small study of shoe factory workers, urinary 2,5-hexanedione (a metabolite of n-hexane) concentration was associated with significant changes in sensory nerve action potential; however, no association was found for time-weighted average n-hexane exposure estimated from breathing zone samples (Mutti et al. 1982b). In our study, we found evidence of adverse associations between THC/BTEX-H exposures and vibrotactile threshold only among workers older than 50 years of age. In contrast to the GuLF Study participants who were exposed to crude oil chemicals for only a few months (median: 5 months, Q1-Q3: 3–8 months), workers in these earlier occupational studies were typically exposed for many years, which might account for the stronger associations observed in these studies. In addition, while the majority of occupational studies assessed neurologic function while workers were still exposed, our study examined neurologic function several years after the cessation of the spill-related occupational exposures. This might have allowed some workers to recover from the neurotoxic insult and may explain the weaker associations observed in our study.
Exposure to toluene, styrene, and mixed solvents at levels above the occupational limit have been associated with color vision loss and/or lower contrast sensitivity among workers (Boeckelmann and Pfister 2003; Costa et al. 2012; Gobba and Cavalleri 2003; Indhushree et al. 2016). Visual dysfunction among workers has also been observed at lower concentrations of volatile hydrocarbons, with several studies reporting impaired color vision among exposed workers (Campagna et al. 2001; Cavalleri et al. 2000; Lee et al. 2007) and one study linking reduced contrast sensitivity to urinary metabolite of xylene among workers (Gong et al. 2003). In our study, we did not observe positive associations between THC/BTEX-H exposure and contrast sensitivity. Unexpectedly, we observed modestly better performance in this test among workers with higher exposure, particularly at spatial frequency of 1.5 cycles/degree, although few associations were statistically significant. This could result from residual confounding by workers’ baseline visual function (“healthier worker effect”) (Chowdhury et al. 2017). Indirect evidence for this comes from a cross-tabulation of job group and use of vision correction, which showed that the proportion of vision correction use was lowest among response (33.3%) and operations workers (31.9%), who experienced the highest crude oil exposures, and highest among support workers (50.9%), who had the lowest exposures. It is plausible that workers with better visual function were selected into jobs that, coincidentally, also exposed them to higher levels of crude oil, and differences in baseline visual function could not be completely accounted for by restricting this analysis to workers with better than 20/50 visual acuity and adjusting for use of vision correction.
Maintenance of posture and balance requires coordination among the peripheral nervous system, the central nervous system, and the musculoskeletal system (Vassar and Rose 2014). The whole process involves the receipt of sensory signals by the visual, vestibular, and somatosensory systems, neural transmission of sensory information to the brain for processing, and coordinated motor responses transmitted from the brain via motor neurons to muscles for postural adjustment (Vassar and Rose 2014). In our study, we evaluated workers’ ability to maintain balance by measuring postural sway speed when participants stood on a platform and, separately, by asking participants to maintain a 30-second single-leg stance. We observed only suggestive associations, and only among workers older than 50 years at enrollment. Although symptoms of dizziness/vertigo, unsteady gait, and muscle weakness have been reported in some studies of workers exposed to organic solvents (Cianchetti et al. 1976; Herbert et al. 1995; Jovanović et al. 2004; Mutti et al. 1982a; Sanagi et al. 1980), few studies have evaluated balance using quantitative tests. In a small study of aircraft maintenance personnel exposed to jet fuel vapor concentrations above the occupational guidelines, postural stability was significantly associated with cumulative exposure to benzene, toluene, and xylene (Smith et al. 1997). The relationship between acute exposure to low concentrations of m-xylene and postural sway was assessed in two studies of volunteers, with positive associations found in one study (Savolainen et al. 1984; Savolainen et al. 1985). Kuo et al. (1996) explored postural sway in a small group of sewer workers chronically exposed to low levels of volatile hydrocarbons and found positive associations with a composite exposure metric measured as “total benzene equivalents”.
Several studies have investigated the neurologic effects of OSRC work. In analyses of Hebei Spirit oil spill workers, higher prevalence of neurological symptoms was reported a few weeks after the spill among workers with inhalation exposure to crude oil (Sim et al. 2010) and longer duration of OSRC work but not with urinary hippuric acid or mandelic acid (Ha et al. 2012). In addition, a persistent but non-significant association was observed between cleanup duration and vestibular symptoms one year after that spill (Na et al. 2012). In the DWH disaster, increasing frequency of inhalation exposure to crude oil was associated with higher prevalence of neurologic symptoms during the OSRC cleanup among a group of Coast Guard responders who participated in the DWH spill cleanup (Krishnamurthy et al. 2019). In the same cohort, ever inhalation exposure to crude oil was associated with higher hazards of International Classification of Diseases-coded neurological diagnoses ascertained from military health encounter records up to 5.5 years post-DWH (Denic-Roberts et al. 2023). A previous analysis in the GuLF Study examined participants’ neurobehavioral function 4–6 years after the spill using 16 quantitative neurocognitive tests and found modest decreases in some tests among workers with higher single daily maximum exposure to THC compared to those with lower maximum exposures (Quist et al. 2019). Blood concentrations of benzene and toluene were associated with self-reported neurologic symptoms among participants of the GuLF Study, although the blood samples were collected 2–3 years after the disaster and reflected recent ambient exposures rather than cleanup-related exposures (Werder et al. 2019).
In our study, we observed heterogeneity in associations between crude oil exposures and performance on vibrotactile threshold, postural stability, and single-leg stance tests by age, with effects seen primarily in the workers over 50 years of age. Although few studies have examined EMM of the relationship between volatile hydrocarbons exposure and neurologic function by age, studies of chemotherapies (du Bois et al. 1999) and other neurotoxicants (Albers et al. 1988) have shown higher risk or severity of toxic neuropathy among older participants. Research has shown that the peripheral nerves have the capacity to regenerate, and certain regions in the brain have the ability to compensate for neurotoxic insult (Tilson and Mitchell 1983); however, this capacity decreases with age (Verdú et al. 2000). The length of time in our study between exposure and the clinical examination (median: 4.6 years, range: 4.2–6.1 years) may have allowed the younger participants to recover from any neurotoxic deficits induced by crude oil chemicals, while older workers were less able to recover. It is also possible that older workers are more susceptible to neurotoxicants because they are less able to detoxify and excrete these chemicals. Studies have shown that the metabolic clearance of many drugs decreases with age, likely due to reduced liver and renal blood flow and functions (Mangoni and Jackson 2004; Soejima et al. 2022). Another possibility is that older individuals have less physiological reserves for responding to toxic insults; age-related declines in neurologic function have been well-documented in studies (Kalisch et al. 2008; Seidler et al. 2010). Lastly, the lipophilic nature of BTEX-H chemicals may have allowed them to accumulate more easily among older workers, who likely have a higher body fat percentage from the aging process compared to younger workers (proportion of overweight/obese in our study by age: 75.2% among <40 years; 78.6% among 40–49 years; 80.3% among 50–59 years; 81.2% among ≥60 years) (Palmer and Kirkland 2016).
The mechanisms by which BTEX-H and other volatile hydrocarbons impair neurologic function have been investigated in animal and cellular studies (Tormoehlen et al. 2014). BTEX-H chemicals have been hypothesized to lead to axonopathy by changing the integrity of the cellular membrane or interacting with the membrane proteins to impair cellular functions (ATSDR 1999, 2000, 2007a, b, 2010). Other proposed mechanisms involve generation of reactive oxygen species, induction of neurofilament accumulation that interrupts axonal transport (ATSDR 1999, 2007a; Valentine 2020), and demyelination of axons that leads to impaired signal conduction (Valentine 2020). These processes occur not only in the peripheral nervous system, but also affect the central nervous system (including the optic nerve), as the high lipophilicity and relatively low molecular weight of BTEX-H and their metabolites allow them to readily cross the blood-brain barrier (Cruz et al. 2014; Luo et al. 2021; National Research Council Committee on Acute Exposure Guideline 2010; World Health Organization 2010). Studies have demonstrated the presence of these chemicals in brains of exposed animal models and humans (Ameno et al. 1992; Avis and Hutton 1993; Ito et al. 2002; Kishi et al. 1988). The brain white matter, which contains a high concentration of lipid-rich myelin, may be more susceptible to the toxicity (Filley 2013). Consistent with this mechanism, studies have observed accumulations of neurofilaments in the axoplasm, myelin degeneration, and/or structural and biochemical changes in the brain in animals and humans exposed to these chemicals (Andersson et al. 1981; Bjornaes and Naalsund 1988; Hsieh et al. 1988; Mutti et al. 1988; Puri et al. 2007; Rosenberg et al. 1988; Schaumburg and Spencer 1976).
One strength of our study is the rigorous assessment of participants’ cumulative exposures using a job-exposure matrix based on extensive personal air samples collected on a subset of DWH oil spill workers, including some who enrolled in the GuLF Study, during the spill and detailed DWH spill work histories self-reported by all study participants, which allowed us to investigate individual etiologic agents potentially responsible for any observed neurologic effects among these workers. This study also contributes to the larger occupational literature on volatile hydrocarbons by investigating neurological effects of several chemicals at exposures below occupational limits. Previous studies of these chemicals have examined more highly exposed worker populations, with most relying on crude proxies of exposure (e.g., exposed vs. control group, duration of work). The few studies that examined exposure to individual chemicals below the occupational limits were of modest size (Bleecker et al. 1991; Gong et al. 2003; Jovanović et al. 2004). With a larger cohort, we were better powered to conduct subgroup analyses and found adverse neurologic effects among older workers that persisted several years after exposure. Persistent neurological effects have been observed in a few studies of workers exposed to organic solvents for years (Cianchetti et al. 1976; Mutti et al. 1982b), but we are the first to report such effects after several months of exposure. Another strength of the study was the use of validated quantitative measures to assess neurologic function, including subtle, subclinical effects that may occur earlier in disease progression (Gerr et al. 2000). These tests have been shown to have good test-retest reliability (Gerr and Letz 1988; Hohberger et al. 2007; Letz et al. 1994) and have been used in many other studies (Bates et al. 2019; Indhushree et al. 2016; Starks et al. 2012; Yokoyama et al. 1997). Lastly, because the GuLF Study obtained data on a wide range of factors, we were able to account for numerous potential confounders of the associations to obtain more accurate effect estimates.
Our study has a few limitations. First, study findings need to be interpreted cautiously in the context of the cross-sectional study design. There could also be bias if participants were assigned to specific jobs/activities based on health-related factors at the time of spill (e.g., baseline neurological function) that were predictive of their neurologic function at the clinical examination. We accounted for several indicators of baseline health by excluding participants with pre-spill diabetes or peripheral neuropathy and by adjusting for BMI and, for the visual contrast sensitivity test, vision correction. For tests of postural stability and single-leg stance, we found slightly stronger associations when we additionally adjusted for ever performing OSRC work on a water vessel, a proxy for baseline vestibular function. Still, there could be residual confounding from unmeasured confounders (e.g., other medical conditions that affected neurologic function) or imperfect measurement of existing covariates (e.g., inaccuracy in self-reported medical diagnoses and smoking/drinking) in the models, which may in part explain the apparent better performance observed for visual contrast sensitivity. We lacked the data to adjust for certain other potential exposures experienced during the oil spill cleanup (Middlebrook et al. 2012), although we accounted for PM2.5 from the controlled burning, which has been modestly associated with some neurologic function test performance in this cohort (Norris et al., in press) and found similar results. We did not collect data on participants’ occupational exposures after the end of their spill cleanup. It is possible that some workers who had higher spill-related exposures continued to have higher occupational exposures after the spill cleanup that could have affected their neurologic function at the time of the clinical exam. We adjusted in the models for pre-spill oil industry work as a proxy for pre-spill crude oil exposures; this variable may also serve as a reasonable indicator of participants’ propensity to work in the oil industry after the spill. Additionally, participants who reported working in the oil industry accounted for <18% of participants in the highest quartile of THC and BTEX-H exposures. Thus, continued occupational exposures from oil industry work is insufficient to explain the associations observed in our study.
Second, the exposure estimates assigned to workers contain some degree of uncertainty, (Stewart et al. 2022). Exposure group average exposures were estimated using a left-censored Bayesian method (Groth et al. 2017; Groth et al. 2022b; Huynh et al. 2016) shown to have low bias and imprecision for most exposure groups, but more uncertainty for those with extreme censoring or few measurements (Stenzel et al. 2022a). However, this is unlikely to have substantively biased our risk estimates in analyses using exposure quartiles because the majority of workers in these low-exposure groups fell into the lowest exposure category.
Third, there could be a selection bias if workers who participated in the clinical examination differed in important ways from workers eligible for the clinical examination (based on residential proximity to a study clinic) (Kwok et al. 2017) who did not participate. It is possible that workers who had greater neurological symptoms were more motivated to participate in the clinical examination; alternatively, any adverse neurological or other health effects from OSRC-related exposures might have reduced their ability to travel to the clinic. To explore the potential for selection bias, we compared characteristics, including neurologic symptoms, ascertained at enrollment between eligible workers who participated in the clinical examination and those who did not (Table S14). We found minimal differences between the two groups, mitigating concerns about this potential bias.
Our study showed modest associations between cumulative exposures to several volatile components of crude oil and several tests of neurologic function 4–6 years after the oil spill among spill workers who were 50 years of age or older. We observed no apparent associations among the younger workers, perhaps because younger workers were less susceptible to the neurotoxic exposures or because they experienced greater recovery of neurologic function after cessation of the exposure. The positive associations were consistent with findings from previous studies of individuals exposed to oil spills and workers exposed to organic solvents and provide evidence that exposure to these agents at levels below current occupational guidelines may induce adverse neurologic effects in a susceptible population. Additional follow-up time for these workers would help determine whether the observed associations persist beyond 4–6 years after the spill. Research is also needed in other populations and settings to confirm these adverse neurologic effects observed for crude oil chemical exposures below occupational limits.
Supplementary Material
Highlights.
The Deepwater Horizon oil spill workers were exposed to crude oil chemicals.
Exposures to BTEX and n-hexane were elevated, but below occupational limits.
Neurologic function was assessed via a battery of four tests at a clinical exam.
The clinical exam occurred 4–6 years after the spill.
Exposures were linked to worse neurologic function among workers ≥50 years of age.
Acknowledgments:
The authors would like to thank Christina Norris for statistical support on this project. This research was supported by the Intramural Research Program of the NIH, National Institute of Environmental Health Sciences (ZO1 ES 102945), and NIH/NIEHS R01ES031127.
Abbreviations:
- BMI
body mass index
- BTEX-H
benzene, toluene, ethylbenzene, xylene, and n-hexane
- CI
confidence interval
- DWH
Deepwater Horizon
- EMM
effect measure modification
- OSRC
oil spill response and cleanup
- PR
prevalence ratio
- THC
total petroleum hydrocarbons
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
The authors declare they have nothing to disclose.
CRediT Author Statement
Dazhe Chen: Conceptualization, Methodology, Formal analysis, Investigation, Writing - Original Draft
Emily J. Werder: Writing - Review & Editing
Patricia A. Stewart: Writing - Review & Editing
Mark R. Stenzel: Writing - Review & Editing
Fredric E. Gerr: Writing - Review & Editing
Kaitlyn G. Lawrence: Writing - Review & Editing
Caroline P. Groth: Writing - Review & Editing
Tran B. Huynh: Writing - Review & Editing
Gurumurthy Ramachandran: Writing - Review & Editing
Sudipto Banerjee: Writing - Review & Editing
W. Braxton Jackson II: Writing - Review & Editing, Data Curation
Kate Christenbury: Validation, Writing - Review & Editing, Data Curation
Richard K. Kwok: Writing - Review & Editing
Dale P. Sandler: Conceptualization, Writing - Review & Editing, Supervision, Funding acquisition
Lawrence S. Engel: Conceptualization, Methodology, Writing - Review & Editing, Supervision, Funding acquisition
Declaration of interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- ACGIH. 2012. Appendix b. Acgih threshold limit values (tlvs) and biological exposure indices (beis). In: 2012 tlvs and beis, Part 7:American Conference of Governmental Industrial Hygienists. [Google Scholar]
- Albers JW, Kallenbach LR, Fine LJ, Langolf GD, Wolfe RA, Donofrio PD, et al. 1988. Neurological abnormalities associated with remote occupational elemental mercury exposure. Ann Neurol 24:651–659. [DOI] [PubMed] [Google Scholar]
- Ameno K, Kiriu T, Fuke C, Ameno S, Shinohara T, Ijiri I. 1992. Regional brain distribution of toluene in rats and in a human autopsy. Arch Toxicol 66:153–156. [DOI] [PubMed] [Google Scholar]
- Andersson K, Fuxe K, Nilsen OG, Toftgård R, Eneroth P, Gustafsson JA. 1981. Production of discrete changes in dopamine and noradrenaline levels and turnover in various parts of the rat brain following exposure to xylene, ortho-, meta-, and para-xylene, and ethylbenzene. Toxicol Appl Pharmacol 60:535–548. [DOI] [PubMed] [Google Scholar]
- ATSDR. 1999. Toxicological profile for n-hexane. Atlanta, GA: U.S. Department of Health and Human Services, Public Health Service, Agency for Toxic Substances and Disease Registry. [Google Scholar]
- ATSDR. 2000. Toxicological profile for toluene.
- ATSDR. 2004. Interaction profile for: Benzene, toluene, ethylbenzene, and xylenes (btex). Atlanta, GA: U.S. Department of Health and Human Services, Public Health Service, Agency for Toxic Substances and Disease Registry. [PubMed] [Google Scholar]
- ATSDR. 2007a. Toxicological profile for benzene. [PubMed]
- ATSDR. 2007b. Toxicological profile for xylene. [PubMed]
- ATSDR. 2010. Toxicological profile for ethylbenzene. [PubMed]
- Avis SP, Hutton CJ. 1993. Acute benzene poisoning: A report of three fatalities. J Forensic Sci 38:599–602. [PubMed] [Google Scholar]
- Baslo A, Aksoy M. 1982. Neurological abnormalities in chronic benzene poisoning. A study of six patients with aplastic anemia and two with preleukemia. Environ Res 27:457–465. [DOI] [PubMed] [Google Scholar]
- Bates MN, Pope K, So YT, Liu S, Eisen EA, Hammond SK. 2019. Hexane exposure and persistent peripheral neuropathy in automotive technicians. Neurotoxicology 75:24–29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bjornaes S, Naalsund LU. 1988. Biochemical changes in different brain areas after toluene inhalation. Toxicology 49:367–374. [DOI] [PubMed] [Google Scholar]
- Bleecker ML, Bolla KI, Agnew J, Schwartz BS, Ford DP. 1991. Dose-related subclinical neurobehavioral effects of chronic exposure to low levels of organic solvents. Am J Ind Med 19:715–728. [DOI] [PubMed] [Google Scholar]
- Boeckelmann I, Pfister EA. 2003. Influence of occupational exposure to organic solvent mixtures on contrast sensitivity in printers. J Occup Environ Med 45:25–33. [DOI] [PubMed] [Google Scholar]
- Campagna D, Mergler D, Huel G, Bélanger S, Truchon G, Ostiguy C, et al. 1995. Visual dysfunction among styrene-exposed workers. Scand J Work Environ Health 21:382–390. [DOI] [PubMed] [Google Scholar]
- Campagna D, Stengel B, Mergler D, Limasset JC, Diebold F, Michard D, et al. 2001. Color vision and occupational toluene exposure. Neurotoxicol Teratol 23:473–480. [DOI] [PubMed] [Google Scholar]
- Cavalleri A, Gobba F, Nicali E, Fiocchi V. 2000. Dose-related color vision impairment in toluene-exposed workers. Arch Environ Health 55:399–404. [DOI] [PubMed] [Google Scholar]
- Chen D, Sandler DP, Keil AP, Heiss G, Whitsel EA, Pratt GC, et al. 2022. Fine particulate matter and incident coronary heart disease events up to 10 years of follow-up among deepwater horizon oil spill workers. Environ Res 217:114841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chowdhury R, Shah D, Payal AR. 2017. Healthy worker effect phenomenon: Revisited with emphasis on statistical methods - a review. Indian J Occup Environ Med 21:2–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cianchetti C, Abbritti G, Perticoni G, Siracusa A, Curradi F. 1976. Toxic polyneuropathy of shoe-industry workers. A study of 122 cases. J Neurol Neurosurg Psychiatry 39:1151–1161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Costa TL, Barboni MT, Moura AL, Bonci DM, Gualtieri M, de Lima Silveira LC, et al. 2012. Long-term occupational exposure to organic solvents affects color vision, contrast sensitivity and visual fields. PLoS One 7:e42961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cruz SL, Rivera-García MT, Woodward JJ. 2014. Review of toluene action: Clinical evidence, animal studies and molecular targets. J Drug Alcohol Res 3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Demers RY, Markell BL, Wabeke R. 1991. Peripheral vibratory sense deficits in solvent-exposed painters. J Occup Med 33:1051–1054. [PubMed] [Google Scholar]
- Denic-Roberts H, Engel LS, Buchanich JM, Miller RG, Talbott EO, Thomas DL, et al. 2023. Risk of longer-term neurological conditions in the deepwater horizon oil spill coast guard cohort study - five years of follow-up. Environ Health 22:12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- du Bois A, Schlaich M, Lück HJ, Mollenkopf A, Wechsel U, Rauchholz M, et al. 1999. Evaluation of neurotoxicity induced by paclitaxel second-line chemotherapy. Support Care Cancer 7:354–361. [DOI] [PubMed] [Google Scholar]
- Filley CM. 2013. Toluene abuse and white matter: A model of toxic leukoencephalopathy. Psychiatr Clin North Am 36:293–302. [DOI] [PubMed] [Google Scholar]
- Gerr F, Hershman D, Letz R. 1990. Vibrotactile threshold measurement for detecting neurotoxicity: Reliability and determination of age- and height-standardized normative values. Arch Environ Health 45:148–154. [DOI] [PubMed] [Google Scholar]
- Gerr F, Letz R, Green RC. 2000. Relationships between quantitative measures and neurologist’s clinical rating of tremor and standing steadiness in two epidemiological studies. Neurotoxicology 21:753–760. [PubMed] [Google Scholar]
- Gerr FE, Letz R. 1988. Reliability of a widely used test of peripheral cutaneous vibration sensitivity and a comparison of two testing protocols. Br J Ind Med 45:635–639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gobba F, Cavalleri A. 2003. Color vision impairment in workers exposed to neurotoxic chemicals. Neurotoxicology 24:693–702. [DOI] [PubMed] [Google Scholar]
- Gong Y, Kishi R, Kasai S, Katakura Y, Fujiwara K, Umemura T, et al. 2003. Visual dysfunction in workers exposed to a mixture of organic solvents. Neurotoxicology 24:703–710. [DOI] [PubMed] [Google Scholar]
- Greenland S, Pearl J, Robins JM. 1999. Causal diagrams for epidemiologic research. Epidemiology 10:37–48. [PubMed] [Google Scholar]
- Grisold W, Carozzi VA. 2021. Toxicity in peripheral nerves: An overview. Toxics 9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Groth C, Banerjee S, Ramachandran G, Stenzel MR, Sandler DP, Blair A, et al. 2017. Bivariate left-censored bayesian model for predicting exposure: Preliminary analysis of worker exposure during the deepwater horizon oil spill. Ann Work Expo Health 61:76–86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Groth CP, Banerjee S, Ramachandran G, Stewart PA, Sandler DP, Blair A, et al. 2022a. Methods for the analysis of 26 million voc area measurements during the deepwater horizon oil spill clean-up. Ann Work Expo Health 66:i140–i155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Groth CP, Huynh TB, Banerjee S, Ramachandran G, Stewart PA, Quick H, et al. 2022b. Linear relationships between total hydrocarbons and benzene, toluene, ethylbenzene, xylene, and n-hexane during the deepwater horizon response and clean-up. Ann Work Expo Health 66:i71–i88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ha M, Kwon H, Cheong HK, Lim S, Yoo SJ, Kim EJ, et al. 2012. Urinary metabolites before and after cleanup and subjective symptoms in volunteer participants in cleanup of the hebei spirit oil spill. Sci Total Environ 429:167–173. [DOI] [PubMed] [Google Scholar]
- Herbert R, Gerr F, Luo J, Harris-Abbott D, Landrigan PJ. 1995. Peripheral neurologic abnormalities among roofing workers: Sentinel case and clinical screening. Arch Environ Health 50:349–354. [DOI] [PubMed] [Google Scholar]
- Hohberger B, Laemmer R, Adler W, Juenemann AG, Horn FK. 2007. Measuring contrast sensitivity in normal subjects with optec 6500: Influence of age and glare. Graefes Arch Clin Exp Ophthalmol 245:1805–1814. [DOI] [PubMed] [Google Scholar]
- Hsieh GC, Parker RD, Sharma RP. 1988. Subclinical effects of groundwater contaminants. Ii. Alteration of regional brain monoamine neurotransmitters by benzene in cd-1 mice. Arch Environ Contam Toxicol 17:799–805. [DOI] [PubMed] [Google Scholar]
- Huynh T, Quick H, Ramachandran G, Banerjee S, Stenzel M, Sandler DP, et al. 2016. A comparison of the β-substitution method and a bayesian method for analyzing left-censored data. Ann Occup Hyg 60:56–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huynh TB, Groth CP, Ramachandran G, Banerjee S, Stenzel M, Blair A, et al. 2022a. Estimates of inhalation exposures among land workers during the deepwater horizon oil spill clean-up operations. Ann Work Expo Health 66:i124–i139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huynh TB, Groth CP, Ramachandran G, Banerjee S, Stenzel M, Blair A, et al. 2022b. Estimates of inhalation exposures to oil-related components on the supporting vessels during the deepwater horizon oil spill. Ann Work Expo Health 66:i111–i123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huynh TB, Groth CP, Ramachandran G, Banerjee S, Stenzel M, Quick H, et al. 2022c. Estimates of occupational inhalation exposures to six oil-related compounds on the four rig vessels responding to the deepwater horizon oil spill. Ann Work Expo Health 66:i89–i110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Indhushree R, Monica R, Coral K, Angayarkanni N, Punitham R, Subburathinam BM, et al. 2016. Visual functions of workers exposed to organic solvents in petrochemical industries. Indian J Occup Environ Med 20:133–137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ito T, Yoshitome K, Horike T, Kira S. 2002. Distribution of inhaled m‐xylene in rat brain and its effect on gab aa receptor binding. Journal of occupational health 44:69–75. [Google Scholar]
- Jardel HV, Engel LS, Lawrence KG, Stewart PA, Stenzel MR, Curry MD, et al. 2022. The association between oil spill cleanup-related total hydrocarbon exposure and diabetes. Environ Res 212:113591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jovanović JM, Jovanović MM, Spasić MJ, Lukić SR. 2004. Peripheral nerve conduction study in workers exposed to a mixture of organic solvents in paint and lacquer industry. Croat Med J 45:769–774. [PubMed] [Google Scholar]
- Kalisch T, Tegenthoff M, Dinse HR. 2008. Improvement of sensorimotor functions in old age by passive sensory stimulation. Clin Interv Aging 3:673–690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kishi R, Harabuchi I, Ikeda T, Yokota H, Miyake H. 1988. Neurobehavioural effects and pharmacokinetics of toluene in rats and their relevance to man. Br J Ind Med 45:396–408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krishnamurthy J, Engel LS, Wang L, Schwartz EG, Christenbury K, Kondrup B, et al. 2019. Neurological symptoms associated with oil spill response exposures: Results from the deepwater horizon oil spill coast guard cohort study. Environ Int 131:104963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuo W, Bhattacharya A, Succop P, Linz D. 1996. Postural stability assessment in sewer workers. J Occup Environ Med 38:27–34. [DOI] [PubMed] [Google Scholar]
- Kwok RK, Engel LS, Miller AK, Blair A, Curry MD, Jackson WB, et al. 2017. The gulf study: A prospective study of persons involved in the deepwater horizon oil spill response and clean-up. Environ Health Perspect 125:570–578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lane HM, Morello-Frosch R, Marshall JD, Apte JS. 2022. Historical redlining is associated with present-day air pollution disparities in u.S. Cities. Environ Sci Technol Lett 9:345–350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee EH, Eum KD, Cho SI, Cheong HK, Paek do M. 2007. Acquired dyschromatopsia among petrochemical industry workers exposed to benzene. Neurotoxicology 28:356–363. [DOI] [PubMed] [Google Scholar]
- Lee I, Park H, Kim MJ, Kim S, Choi S, Park J, et al. 2022. Exposure to polycyclic aromatic hydrocarbons and volatile organic compounds is associated with a risk of obesity and diabetes mellitus among korean adults: Korean national environmental health survey (konehs) 2015–2017. Int J Hyg Environ Health 240:113886. [DOI] [PubMed] [Google Scholar]
- Letz R, Gerr F, Harris-Abbott D. 1994. Heterogeneity of effects of ethanol ingestion on postural stability as measured by two devices. Neurotoxicology 15:603–607. [PubMed] [Google Scholar]
- Luo M, Shi X, Guo Q, Li S, Zhang Q, Sun X, et al. 2021. 2,5-hexanedione induced apoptosis in rat spinal cord neurons and vsc4.1 cells via the prongf/p75ntr and jnk pathways. Biosci Rep 41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mangoni AA, Jackson SH. 2004. Age-related changes in pharmacokinetics and pharmacodynamics: Basic principles and practical applications. Br J Clin Pharmacol 57:6–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Middlebrook AM, Murphy DM, Ahmadov R, Atlas EL, Bahreini R, Blake DR, et al. 2012. Air quality implications of the deepwater horizon oil spill. Proc Natl Acad Sci U S A 109:20280–20285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mutti A, Cavatorta A, Lucertini S, Arfini G, Falzoi M, Franchini I. 1982a. Neurophysiological changes in workers exposed to organic solvents in a shoe factory. Scand J Work Environ Health 8 Suppl 1:136–141. [PubMed] [Google Scholar]
- Mutti A, Ferri F, Lommi G, Lotta S, Lucertini S, Franchini I. 1982b. N-hexane-induced changes in nerve conduction velocities and somatosensory evoked potentials. Int Arch Occup Environ Health 51:45–54. [DOI] [PubMed] [Google Scholar]
- Mutti A, Falzoi M, Romanelli A, Bocchi MC, Ferroni C, Franchini I. 1988. Brain dopamine as a target for solvent toxicity: Effects of some monocyclic aromatic hydrocarbons. Toxicology 49:77–82. [DOI] [PubMed] [Google Scholar]
- Na JU, Sim MS, Jo IJ, Song HG. 2012. The duration of acute health problems in people involved with the cleanup operation of the hebei spirit oil spill. Mar Pollut Bull 64:1246–1251. [DOI] [PubMed] [Google Scholar]
- National Commission on the BP Deepwater Horizon Oil Spill and Offshore Drilling. 2011. Deep water: The gulf oil disaster and the future of offshore drilling, report to the president. 0160873711;9780160873713.National Commission on the BP Deepwater Horizon Oil Spill and Offshore Drilling. [Google Scholar]
- National Research Council Committee on Acute Exposure Guideline L. 2010. In: Acute exposure guideline levels for selected airborne chemicals: Volume 9. Washington (DC):National Academies Press (US) Copyright 2010 by the National Academy of Sciences. All rights reserved. [PubMed] [Google Scholar]
- Neghab M, Soleimani E, Khamoushian K. 2012. Electrophysiological studies of shoemakers exposed to sub-tlv levels of n-hexane. J Occup Health 54:376–382. [DOI] [PubMed] [Google Scholar]
- OSHA. 2022. Occupational chemical database. Occupational Safety and Health Administration, US Department of Labor. [Google Scholar]
- Palmer AK, Kirkland JL. 2016. Aging and adipose tissue: Potential interventions for diabetes and regenerative medicine. Exp Gerontol 86:97–105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pratt GC, Stenzel MR, Kwok RK, Groth CP, Banerjee S, Arnold SF, et al. 2022. Modeled air pollution from in situ burning and flaring of oil and gas released following the deepwater horizon disaster. Ann Work Expo Health 66:i172–i187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Puri V, Chaudhry N, Tatke M. 2007. N-hexane neuropathy in screen printers. Electromyogr Clin Neurophysiol 47:145–152. [PubMed] [Google Scholar]
- Quist AJL, Rohlman DS, Kwok RK, Stewart PA, Stenzel MR, Blair A, et al. 2019. Deepwater horizon oil spill exposures and neurobehavioral function in gulf study participants. Environ Res 179:108834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramachandran G, Groth CP, Huynh TB, Banerjee S, Stewart PA, Engel LS, et al. 2022. Using real-time area voc measurements to estimate total hydrocarbons exposures to workers involved in the deepwater horizon oil spill. Ann Work Expo Health 66:i156–i171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosenberg NL, Spitz MC, Filley CM, Davis KA, Schaumburg HH. 1988. Central nervous system effects of chronic toluene abuse--clinical, brainstem evoked response and magnetic resonance imaging studies. Neurotoxicol Teratol 10:489–495. [DOI] [PubMed] [Google Scholar]
- Saadi A, Himmelstein DU, Woolhandler S, Mejia NI. 2017. Racial disparities in neurologic health care access and utilization in the united states. Neurology 88:2268–2275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanagi S, Seki Y, Sugimoto K, Hirata M. 1980. Peripheral nervous system functions of workers exposed to n-hexane at a low level. Int Arch Occup Environ Health 47:69–79. [DOI] [PubMed] [Google Scholar]
- Savolainen K, Kekoni J, Riihimäki V, Laine A. 1984. Immediate effects of m-xylene on the human central nervous system. Arch Toxicol Suppl 7:412–417. [DOI] [PubMed] [Google Scholar]
- Savolainen K, Riihimäki V, Muona O, Kekoni J, Luukkonen R, Laine A. 1985. Conversely exposure-related effects between atmospheric m-xylene concentrations and human body sense of balance. Acta Pharmacol Toxicol (Copenh) 57:67–71. [DOI] [PubMed] [Google Scholar]
- Schaumburg HH, Spencer PS. 1976. Central and peripheral nervous system degeneration produced by pure n-hexane. Trans Am Neurol Assoc 101:153–156. [PubMed] [Google Scholar]
- Seidler RD, Bernard JA, Burutolu TB, Fling BW, Gordon MT, Gwin JT, et al. 2010. Motor control and aging: Links to age-related brain structural, functional, and biochemical effects. Neurosci Biobehav Rev 34:721–733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shih HT, Yu CL, Wu MT, Liu CS, Tsai CH, Hung DZ, et al. 2011. Subclinical abnormalities in workers with continuous low-level toluene exposure. Toxicol Ind Health 27:691–699. [DOI] [PubMed] [Google Scholar]
- Sim MS, Jo IJ, Song HG. 2010. Acute health problems related to the operation mounted to clean the hebei spirit oil spill in taean, korea. Mar Pollut Bull 60:51–57. [DOI] [PubMed] [Google Scholar]
- Smith LB, Bhattacharya A, Lemasters G, Succop P, Puhala E 2nd, Medvedovic M, et al. 1997. Effect of chronic low-level exposure to jet fuel on postural balance of us air force personnel. J Occup Environ Med 39:623–632. [DOI] [PubMed] [Google Scholar]
- Soejima K, Sato H, Hisaka A. 2022. Age-related change in hepatic clearance inferred from multiple population pharmacokinetic studies: Comparison with renal clearance and their associations with organ weight and blood flow. Clin Pharmacokinet 61:295–305. [DOI] [PubMed] [Google Scholar]
- Starks SE, Hoppin JA, Kamel F, Lynch CF, Jones MP, Alavanja MC, et al. 2012. Peripheral nervous system function and organophosphate pesticide use among licensed pesticide applicators in the agricultural health study. Environ Health Perspect 120:515–520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stenzel MR, Groth CP, Banerjee S, Ramachandran G, Kwok RK, Engel LS, et al. 2022a. Exposure assessment techniques applied to the highly censored deepwater horizon gulf oil spill personal measurements. Ann Work Expo Health 66:i56–i70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stenzel MR, Groth CP, Huynh TB, Ramachandran G, Banerjee S, Kwok RK, et al. 2022b. Exposure group development in support of the niehs gulf study. Ann Work Expo Health 66:i23–i55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stĕtkárová I, Urban P, Procházka B, Lukás E. 1993. Somatosensory evoked potentials in workers exposed to toluene and styrene. Br J Ind Med 50:520–527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stewart P, Groth CP, Huynh TB, Gorman Ng M, Pratt GC, Arnold SF, et al. 2022. Assessing exposures from the deepwater horizon oil spill response and clean-up. Ann Work Expo Health 66:i3–i22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takeuchi Y 1993. N-hexane polyneuropathy in japan: A review of n-hexane poisoning and its preventive measures. Environ Res 62:76–80. [DOI] [PubMed] [Google Scholar]
- Tilson HA, Mitchell CL. 1983. Neurotoxicants and adaptive responses of the nervous system. Introductory remarks. Fed Proc 42:3189–3190. [PubMed] [Google Scholar]
- Tormoehlen LM, Tekulve KJ, Nañagas KA. 2014. Hydrocarbon toxicity: A review. Clin Toxicol (Phila) 52:479–489. [DOI] [PubMed] [Google Scholar]
- Tyson SF, Connell LA. 2009. How to measure balance in clinical practice. A systematic review of the psychometrics and clinical utility of measures of balance activity for neurological conditions. Clin Rehabil 23:824–840. [DOI] [PubMed] [Google Scholar]
- U.S. Coast Guard 2011. On scene coordinator report: Deepwater horizon oil spill.U.S. Dept. of Homeland Security, U.S. Coast Guard. [Google Scholar]
- Valentine WM. 2020. Toxic peripheral neuropathies: Agents and mechanisms. Toxicol Pathol 48:152–173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vassar RL, Rose J. 2014. Motor systems and postural instability. Handb Clin Neurol 125:237–251. [DOI] [PubMed] [Google Scholar]
- Verdú E, Ceballos D, Vilches JJ, Navarro X. 2000. Influence of aging on peripheral nerve function and regeneration. J Peripher Nerv Syst 5:191–208. [DOI] [PubMed] [Google Scholar]
- Wang C, Chan JS, Ren L, Yan JH. 2016. Obesity reduces cognitive and motor functions across the lifespan. Neural Plast 2016:2473081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Werder EJ, Engel LS, Blair A, Kwok RK, McGrath JA, Sandler DP. 2019. Blood btex levels and neurologic symptoms in gulf states residents. Environ Res 175:100–107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- World Health Organization. 2010. Who guidelines approved by the guidelines review committee. In: Who guidelines for indoor air quality: Selected pollutants. Geneva:World Health Organization; Copyright © 2010, World Health Organization. [PubMed] [Google Scholar]
- Xu X, Ha SU, Basnet R. 2016. A review of epidemiological research on adverse neurological effects of exposure to ambient air pollution. Front Public Health 4:157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yokoyama K, Araki S, Murata K, Morita Y, Katsuno N, Tanigawa T, et al. 1997. Subclinical vestibulo-cerebellar, anterior cerebellar lobe and spinocerebellar effects in lead workers in relation to concurrent and past exposure. Neurotoxicology 18:371–380. [PubMed] [Google Scholar]
- Zou G 2004. A modified poisson regression approach to prospective studies with binary data. Am J Epidemiol 159:702–706. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


